A gold(III)-catalyzed Conia-ene reaction for the direct synthesis of substituted pyrroles from β-ketopropargyl amines
Sayed Mustafa
Badr
 a,
Mustafa
Durmaz
ab and
Nurettin
Menges
a,
Mustafa
Durmaz
ab and
Nurettin
Menges
 *ac
*ac
aScience and Technology Research and Application Center (BITAM), Necmettin Erbakan University, 42090, Konya, Türkiye. E-mail: nurettin.menges@erbakan.edu.tr
bDepartment of Basic Sciences, Faculty of Engineering, Necmettin Erbakan University, 42090, Konya, Türkiye
cDepartment of Biomedical Engineering, Faculty of Engineering, Necmettin Erbakan University, 42090, Konya, Türkiye
First published on 28th January 2026
Abstract
We disclose a gold-catalyzed conversion of unactivated β-ketopropargyl amines to polysubstituted pyrroles under mild, operationally simple conditions. A broad substituent survey reveals clear structure–reactivity trends. With application of the revealed protocol, leveraging the propargyl tolerance, combining AuCI3 with TBACN telescopes the sequence to a bicyclic indolizine ring.
Introduction
The Conia-ene reaction is an intramolecular cyclization of enols with alkenes or alkynes, first studied in the late 1960s by Jean-Marie Conia. This pericyclic reaction, named in honor of Conia's contributions, enables the formation of carbon–carbon bonds through the thermal cyclization of unsaturated carbonyl compounds.1 The traditional Conia-ene reaction requires high temperatures, limiting its application, but advancements in metal catalysis have enabled milder conditions, expanding its use in natural product synthesis.2 The extensive application of transition metals, particularly gold, in these transformations has been comprehensively reviewed, emphasizing the efficiency and sustainability of modern protocols.3 Following these developments, foundational studies in the field have further refined the use of metal catalysts for complex assemblies.4 Milder reaction conditions can be achieved by using activated substrates with electron-withdrawing groups or by employing external Lewis acids or bases, such as t-BuOK, to promote keto–enol interconversion. Maestri and his team developed a t-BuOK-promoted strategy for the synthesis of 1,3-dihydro-2H-pyrrol-2-one and 4-methylenepyrrolidin-2-one frameworks through a Conia-ene-like intramolecular cyclization.5 Conia-ene reactions have been widely employed with various starting materials to synthesize cyclopentane derivatives. Over time, researchers have developed novel substrates, leading to diverse transformations. For instance, Balalaie and his team reported an efficient synthesis of diketopiperazinoindolines via an indium-catalyzed intramolecular 5-exo-dig cyclization of ortho-alkynyl diketopiperazines.6 Similarly, a new indium(III)-catalyzed method was introduced for the synthesis of indolizine scaffolds through a 5-exo-dig aza Conia-ene reaction.7 Grover et al. explored a tandem C–H functionalization/Conia-ene cyclization of N-alkyne tethered indoles, enabling a dual functionalization of diazodicarbonyls to generate pyrrolo[1,2-a]-, pyrido[1,2-a]-, and azepino[1,2-a]indole derivatives.8 This strategy was also applied in cyclization reactions recently reported by Hatakeyama9 and Sun et al.,10 facilitating the synthesis of tetrahydrofurans and pyrrolidines. More recently, innovative approaches have continued to expand the scope of these transformations, offering new insights into the efficiency and selectivity of the Conia-ene process.11,12 Various approaches have been explored for Conia-ene reactions so far, and some of them have been highlighted above. However, the limited number of examples leading to heterocyclic ring formation presents an open field for further investigation.Pyrrole is a fundamental structural unit found in a wide range of natural products, pharmaceuticals, and advanced materials.13 The presence of pyrrole derivatives in bioactive compounds, including antibiotics, anti-cancer agents, and anti-inflammatory drugs, underscores their significance in medicinal chemistry.14 Considering the value of multi-substituted pyrroles, the development of efficient and large-scale synthesis approaches from simple and readily available starting materials continues to be a central focus of organic synthesis.15,16 Advances in metal-catalyzed, electrochemical, and stepwise cyclization methods continue to expand the accessibility and diversity of pyrrole derivatives, opening avenues for new applications in drug discovery and materials science.
Wang and his team reported the Conia-ene cyclization of β-keto propargylamine derivatives using AgOTf to generate pyrroline derivatives (Scheme 1). However, the study revealed some key limitations. The reaction's success was highly dependent on the nitrogen substituents. Only tosyl (Ts) and nosyl (Ns) protecting groups enabled cyclization, while other substituents such as methyl (Me), acetyl (Ac), and phenyl (Ph) failed to initiate the reaction. On the other hand, the researchers obtained pyrroline derivatives, which upon deprotection under basic conditions underwent further transformation into pyrroles. Moreover, the authors noted that the use of an Au(I)/Ag catalytic system diverted the reaction course, leading to complete hydration of the alkyne rather than the desired cyclization. Therefore, we have designed a novel strategy where β-keto propargyl amines undergo cyclization using a gold(III) catalyst in the absence of any additives for the first time. This method offers a promising alternative to overcome the limitations of the approach previously reported.17
Results and discussion
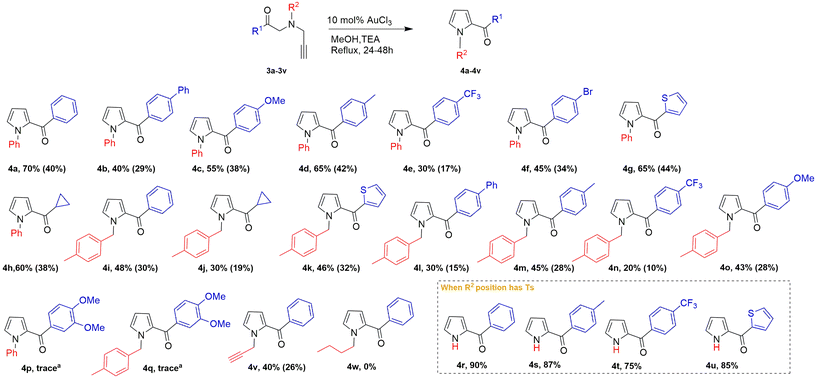
Initially, various amine derivatives were subjected to propargylation resulting in 1a–1c, followed by their reaction with acyl bromide derivatives 2a–2i to obtain the desired intermediates 3a–3q (Scheme S2). One of the starting materials 3v, having two identical propargyl units, was synthesized using α-aminoacetophenone and propargyl bromide (Scheme S4). The crude products were then purified and utilized for cyclization reactions (see the SI for experimental details).18First, we chose N-phenyl-N-(prop-2-yn-1-yl)aniline (3a) as the model substrate (Table 1). The optimization of the Conia-ene reaction was investigated using various catalysts, bases, and solvents to enhance pyrrole formation. Initially, gold, copper, and silver salts alone failed to promote the cyclization, yielding no detectable product. However, upon the addition of two equivalents of triethylamine (TEA), the reaction catalysed by AuCl3 successfully furnished the desired product, presumably due to the enolization of the starting material, which facilitated the subsequent cyclization process. Increasing the catalyst loading of gold(III) chloride from 10 mol% to 20 mol% and 30 mol% did not result in an appreciable enhancement in product yield, indicating that 10 mol% represents the optimal and most efficient catalytic amount under the established reaction conditions. On the other hand, decreasing the catalyst amount to 5 mol% reduced the yield to 50%, and 1 mol% of catalyst did not yield any product (Table 1, entries 19 and 20). Among the tested catalysts, AuCl3 demonstrated the highest activity, achieving a 70% NMR yield in methanol after 24 hours, while catalytic amounts of Ag2CO3 and AgOAc provided no product. Copper-based catalysts (CuBr, CuBr2, CuI) exhibited poor activity (1–7%), whereas ZnCl2, FeCl3, and Pd(OAc)2 were ineffective. Base additives played a crucial role, with TEA (2 eq.) improving the reaction, yielding up to 70% pyrrole, while 3 equivalents of Ag2CO3 provided moderate effects (40%). In contrast, BaCO3 was unable to facilitate the Conia-ene cyclization, indicating that the reaction cannot proceed solely in the presence of a conventional base or a simple metal source. Morpholine was also evaluated as an alternative base and afforded compound 4a in 45% NMR yield (Table 1, entry 23). This observation suggests that the steric profile of the base plays a significant role in determining the reaction efficiency, with increased steric hindrance likely being beneficial for achieving higher yields.
| Entry | Additive | Catalyst (10 mol%) | Solvent | Yield of 4a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b (%) b (%) |
|---|---|---|---|---|
| a Reaction conditions: 1 mmol of 3a, 2 mmol of triethylamine (TEA), 10 mol% catalyst and 1 mL of solvent at reflux temperature for 24 h. b Yields were obtained from 1H NMR spectra. c The reaction was run at room temperature. d The reaction was run under nitrogen. The value in parentheses refers to the isolated yield. | ||||
| 1 | TEA | AuCl3 | EtOH | 60 |
| 2 | TEA | ZnCl2 | EtOH | Trace |
| 3 | TEA | CuBr2 | EtOH | Trace |
| 4 | TEA | CuBr | EtOH | 7 |
| 5 | TEA | CuI | EtOH | Trace |
| 6 | TEA | AgOAc | EtOH | Trace |
| 7 | TEA | FeCl3 | EtOH | — |
| 8 | TEA | Pd(OAc)2 | EtOH | — |
| 9 | Ag2CO3 (3 eq.) | — | EtOH | 40 |
| 10 | BaCO3 (3 eq.) | — | EtOH | — |
| 11 | — | Ag2CO3 | EtOH | — |
| 12 | TEA | AuCl 3 | MeOH | 70 (40) |
| 13 | NaOH (2 eq.) | AuCl3 | MeOH | 50 |
| 14 | TEA | AuCl3 | DMSO | — |
| 15 | TEA | AuCl3 | Pyridine | — |
| 16 | TEA | AuCl3 | MeCN | 20 |
| 17 | TEA | AuCl3 | MeNO2 | 5 |
| 18 | TEA | AuCl3 | Toluene | 40 |
| 19 | TEA | AuCl3 (5 mol%) | MeOH | 50 |
| 20 | TEA | AuCl3 (1 mol%) | MeOH | Trace |
| 21 | TEA | — | MeOH | — |
| 22 | — | AuCl3 | MeOH | — |
| 23 | Morpholine | AuCl3 | MeOH | 45 |
| 24c | TEA | AuCl3 | MeOH | — |
| 25d | TEA | AuCl3 | MeOH | — |
The choice of solvent was found to be a crucial factor influencing the reaction outcome. Among the solvents tested, methanol provided the best performance, affording the highest yield in the presence of AuCl3. In contrast, toluene and acetonitrile resulted in moderate conversions (40% and 20%, respectively), whereas DMSO and pyridine completely suppressed the reaction. In addition, a trace formation of the desired product (5% yield) was observed in nitromethane.
These findings highlight the importance of catalyst selection, base additives, and solvent choice in optimizing the Conia-ene reaction for efficient pyrrole synthesis.
After optimization reactions, derivatization of starting materials was completed, and all derivatives were subjected to cyclization reaction protocols.
The substituent survey established substrate 4a as a reference, producing a high yield of 70% as the NMR yield. All discussed yields are given as NMR yields, and isolated yields are reported in Scheme 2. Aryl derivatives bearing electron-donating groups (4c and 4d) showed minor yield erosion (55 and 65%), likely due to intermediate stabilization during the gold-mediated cyclization. Conversely, strongly electron-withdrawing groups (e.g., CF3) significantly diminished the reactivity, with the yields dropping to 20 and 30% (4n and 4e), indicating reduced π-nucleophilicity and destabilized intermediates. Modest yields were observed for the p-bromo substrate (4f, 45%) and thiophene derivatives (4g, 65%; 4k, 46%), with the latter aligning with the softness of the π-system. Although compounds 4p and 4q could not be isolated in pure form, their characteristic product signals were observed in the 1H NMR spectra of the crude reaction mixtures. A pivotal finding was the impact of the N-substituent on both reactivity and stability. Transitioning to a tosyl (Ts) group at R2 significantly enhanced the isolated yields to 75–90% (4r–4u), even for electronically demanding substrates. Ts-protected pyrroles proved to be more robust, facilitating higher isolated yields through a desulfonylation and base-promoted aromatization pathway. The derivative set also included N-p-tolyl (R2 = p-tolyl) and benzyl versions. For these derivatives, p-tolyl generally produced modest yields (20–48%), which were lower than those for the Ts series but higher than those for the most deactivated instances. Taken together, these patterns provide a clear structure–reactivity map, confirming that EDG-rich aryls and Ts-protected scaffolds are low-risk, high-yielding options. Mechanistically, the neutral, moderately bulky benzyl group may not have provided the coordination-suppressing properties of the Ts group—such as increasing conversion and broadening substrate tolerance—but it also did not significantly inhibit cyclization, indicating that its effect is primarily steric rather than electronic.
For Ts-protected substrates, the reaction likely proceeds through a modified aromatization pathway involving desulfonylation, as the Ts group is absent in the final pyrrole products. This behavior is consistent with previously reported Ag-catalyzed base-assisted pyrrole formation by Wang et al. and may explain the enhanced yields observed for electron-deficient substrates.
Mechanistically, the reaction involves Au(III)-mediated π-activation of the alkyne, followed by a 5-endo-dig cyclization to form a cationic vinyl-gold intermediate (Scheme 3). The electron-withdrawing Ts group plays an essential role by attenuating the nucleophilicity of the nitrogen lone pair, thereby suppressing competitive N → Au(III) coordination. Coordination of unprotected N-containing starting materials to Au(III) reduces the population of catalytically active species. However, N-sulfonyl protection suppresses this inhibitory interaction and accelerates the catalytic cycle. This electronic modification prevents catalyst poisoning—a hypothesis supported by the cyclization failure of the more basic N-butyl analogue (3w) to undergo reaction. Even though the subsequent oxidation-driven aromatization appears to be the rate-determining step for derivatives 3a–3p, 3q, and 3v, a different mechanistic pathway is operative for the N-sulfonyl substrates (3r–3u). In these cases, following the formation of the pyrroline ring, the TEA abstracts a proton, leading to the departure of the tosylate (Ts) group from the pyrroline ring. Subsequently, the tosylate anion abstracts a proton adjacent to the imine nitrogen to generate the pyrrole anion, resulting in the release of TsH. This step is followed by proton abstraction from the protonated triethylamine (TEA), resulting in the regeneration of TEA to afford compounds 4r–4u (Scheme 3).
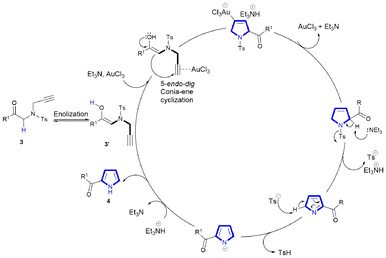 | ||
| Scheme 3 Proposed mechanistic pathway highlighting the role of N-tosyl protection in suppressing undesired N → Au(III) coordination and facilitating in situ deprotection/aromatization. | ||
To confirm the identity of pyrroline peaks, we monitored the reaction of substrate 3a at shorter time intervals. When the reaction was quenched before 24 h, the pyrroline signals in the 1H NMR spectrum were found to be significantly more intense, indicating that the Au(III)-catalyzed Conia-ene cyclization is faster than the subsequent aromatization/oxidation step. Consistent with this, HRMS analysis of 4a showed the main ion corresponding to the aromatic pyrrole ([M + Na], found: 270.0853) and trace ions at m/z 272.0906, assignable to the partially saturated pyrroline ([M + 2H + Na]). These data provide direct evidence for the unoxidized intermediate and support the proposed mechanism.
This oxidation process is particularly decisive for electron-deficient substrates, where inefficient oxidation may lead to incomplete aromatization or the formation of side products, accounting for the reduced yields observed in certain derivatives. On the other hand, Ts-protected derivatives were cyclized under a different mechanism as depicted in Scheme 3. In this reaction, there was no oxidation, but base promoted aromatization.
The protocol also demonstrated excellent chemoselectivity and scalability. The acceptable yield (40%) of 4v coming from the N-propargyl derivative 3v highlights the catalyst's ability to selectively engage one alkyne while leaving the terminal handle intact for late-stage diversification. This terminal alkyne remains accessible as a synthetically flexible handle for transformations such as CuAAC “click” reactions, Sonogashira-type couplings, or oxidative elaborations without hampering the core transformation. Importantly, the Au(III)-mediated cyclization continues despite the presence of an additional propargyl group on nitrogen, demonstrating the protocol's resistance to catalyst deactivation that more alkynes may otherwise cause.19 Furthermore, a gram-scale reaction of 3a (4.0 mmol) provided 4a in 40% yield (0.396 g). This consistency with small-scale results confirms the protocol's operational simplicity and practical utility for larger-scale heterocyclic synthesis.
Using the doubly propargylated substrate 3v, we confirmed that the AuCl3-mediated annulation tolerates the presence of two terminal alkynes. This selective reactivity underscores the chemoselective nature of the Au(III) catalyst and offers a synthetically valuable handle for further derivatization. Building on this remarkable propargyl tolerance and drawing from our previous studies,20–24 we combined gold(III) chloride with tetrabutylammonium cyanide (TBACN). This catalyst and nucleophilic reagent pairing effectively promoted an additional annulation step, enabling the formation of the bicyclic indolizine scaffold (Scheme 4). The indolizine skeleton, encompassing a fused pyridine–pyrrole system, represents a privileged heterocyclic motif in both medicinal and materials chemistry. Its extended π-conjugation imparts remarkable photophysical and electrochemical characteristics, including intense fluorescence, tuneable redox activity, and high binding propensity toward biomolecular receptors. Naturally occurring indolizines exhibit a wide spectrum of pharmacological activities, including anticancer, antimicrobial, and anti-inflammatory effects. In materials science, indolizine scaffolds serve also as versatile building blocks for organic semiconductors, nonlinear optical materials, and fluorescent probes, making them valuable platforms.25–28
Under AuCl3/TBACN in 1,4-dioxane, the intended indolizine derivative 5a was produced in 20% yield, along with N-allenyl pyrrole (6a) and 2-acetyl-1H-pyrrole (4r). This result emphasizes the protocol's chemoselectivity and robustness—its ability to run without catalyst deactivation by additional alkynes—and establishes it as a one-flask, single-operation platform appropriate for telescoped, higher-order synthesis using multifunctional propargylated substrates. With a new protocol in hand, ongoing efforts are directed toward refining the optimal reaction conditions for high yield indolizine synthesis and elucidating the scope and functional group tolerance, which will be addressed in our subsequent reports.
Conclusions
This work found that a gold(III)-catalyzed Conia-ene reaction may be used to synthesize multi-substituted pyrroles from β-ketopropargyl amines using moderate conditions. By carefully analyzing substituent effects and nitrogen-protection techniques, the study gives a clear structure–reactivity framework for explaining and predicting substrate performance. Importantly, the demonstrated tolerance for an additional propargyl moiety allows for rapid cyclization while preserving a late-stage diversification handle, thereby increasing the synthetic utility of this technique. Furthermore, the effective telescoping of this reaction into indolizine production demonstrates its potential for scaffold escalation in medicinal chemistry and materials science. These findings highlight the operational simplicity, chemoselectivity, and scalability of this gold-catalyzed protocol, paving the way for future applications in complex molecule assembly. Our current investigations are focused on broadening the derivatization scope and exploring other heteroannulation transformations to further enhance the synthetic relevance of this platform.Conflicts of interest
All authors have confirmed that they have no conflicts of interest about the study.Data availability
The datasets supporting this study are included in the supplementary information (SI). Supplementary information: full spectral files and processed summaries (NMR and HRMS) corresponding to the compounds and experiments. See DOI: https://doi.org/10.1039/d5ob01665g.Acknowledgements
The authors thank Necmettin Erbakan University Science and Technology Research and Application Center (BITAM) for their facilities. N. Menges thanks TUBİTAK and Necmettin Erbakan University Scientific Research Projects Coordination for partly funding this study (123Z183 and 2217MER03007). N. Menges also thanks to Turkish Academia of Sciences for their support (TÜBA-GEBİP).References
- J. M. Conia and P. L. Perchec, Synthesis, 2002, 1–19 Search PubMed.
- C. Peng, P. Arya, Z. Zhou and S. A. Snyder, Angew. Chem., 2020, 132, 13623–13627 Search PubMed.
- J. S. S. Neto and G. Zeni, ChemCatChem, 2020, 12, 3335–3408 CrossRef CAS.
- J. J. Kennedy-Smith, S. T. Staben and F. D. Toste, J. Am. Chem. Soc., 2004, 126, 4526–4527 CrossRef CAS PubMed.
- A. Cerveri, M. Vettori, A. Serafino and G. Maestri, Org. Biomol. Chem., 2023, 21, 7311–7315 RSC.
- H. Z. Tejeneki, A. Nikbakht, S. Balalaie and F. Rominger, J. Org. Chem., 2020, 85, 8544–8552 Search PubMed.
- M. Meazza, L. A. Leth, J. D. Erickson and K. A. Jørgensen, Chem. – Eur. J., 2017, 23, 7905–7909 Search PubMed.
- A. H. Bhat, S. Alavi and H. K. Grover, Org. Lett., 2020, 22, 224–229 CrossRef CAS PubMed.
- F. Urabe, S. Miyamoto, K. Takahashi, J. Ishihara and S. Hatakeyama, Org. Lett., 2014, 16, 1004–1007 CrossRef CAS PubMed.
- K. Liu, C. Zhu, J. Min, S. Peng, G. Xu and J. Sun, Angew. Chem., Int. Ed., 2015, 54, 12962–12967 Search PubMed.
- G. Onodera, H. Shimizu, R. Kuwahara, K. Kawasaki, N. Krause, T. Fukuda and M. Kimura, Adv. Synth. Catal., 2025, 367, e70064 Search PubMed.
- X. Ling, J. Wang, Y. Chen, P. Zhao, D. Ni, Y. Wei, S. Nie and Y. He, J. Org. Chem., 2025, 90, 7359–7371 CrossRef CAS PubMed.
- B. H. Ganesh, A. G. Raj, B. Aruchamy, P. Nanjan, C. Drago and P. Ramani, ChemMedChem, 2024, 19, e202300447 Search PubMed.
- L. Long, H. Zhang, Z. Zhou, L. Duan, D. Fan, R. Wang, S. Xu, D. Qiao and W. Zhu, Eur. J. Med. Chem., 2024, 273, 116470 CrossRef CAS PubMed.
- T. Shi, G. Yin, X. Wang, Y. Xiong, Y. Peng, S. Li, Y. Zeng and Z. Wang, Green Synth. Catal., 2023, 4, 20–34 CAS.
- H. Seyrani, S. Ramezanpour, A. Vaezghaemi and F. Kobarfard, New J. Chem., 2021, 45, 15647–15654 Search PubMed.
- S. S. K. Boominathan, W.-P. Hu, G. C. Senadi and J.-J. Wang, Adv. Synth. Catal., 2013, 355, 3570–3574 CrossRef CAS.
- N. Yi, Y. Liu, Y. Xiong, H. Gong, J.-P. Tan, Z. Fang and B. Yi, J. Org. Chem., 2023, 88, 11945–11953 CrossRef CAS PubMed.
- H.-C. Wu, C.-W. Yang, L.-C. Hwang and M.-J. Wu, Org. Biomol. Chem., 2012, 10, 6640–6648 RSC.
- S. Gul, K. S. I. Amudi, B. Kuzu and N. Menges, ACS Omega, 2025, 10, 18881–18888 Search PubMed.
- V. Taşdemir and N. Menges, Asian J. Org. Chem., 2020, 9, 2108–2111 Search PubMed.
- V. Tasdemir, H. Genç and N. Menges, Synlett, 2023, 801–806 Search PubMed.
- B. Kuzu, S. Gül, M. Tan, N. Menges and M. Balci, ChemistrySelect, 2021, 6, 2366–2372 Search PubMed.
- O. Sari, A. F. Seybek, S. Kaya, N. Menges, S. S. Erdem and M. Balci, Eur. J. Org. Chem., 2019, 5261–5274 Search PubMed.
- A. V. Botezatu, B. Furdui, A. Busuioc and R. M. Dinică, Eur. J. Med. Chem., 2025, 297, 117908 Search PubMed.
- A. A. Kalinin, G. M. Fazleeva, T. I. Burganov, L. N. Islamova, Y. B. Dudkina, A. A. Kadyrova, T. A. Vakhonina, A. R. Khamatgalimov, A. S. Mukhtarov, V. V. Bazarov, S. A. Katsyuba, Y. H. Budnikova and M. Yu. Balakina, J. Photochem. Photobiol., A, 2020, 386, 112125 Search PubMed.
- Y.-C. Yuan, T.-Z. Liu and B.-X. Zhao, J. Org. Chem., 2021, 86, 12737–12744 CrossRef CAS PubMed.
- M. Jadhav, K. Mali, V. Rajput, R. Das and A. Shard, Med. Chem. Res., 2024, 33, 1491–1510 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |