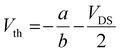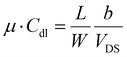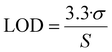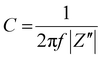Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Bio-modified hydrogels in electrolyte-gated organic field-effect transistors for sensing applications
María Jesús Ortiz-Aguayo a,
Sara Ruiz-Molinaa,
Carme Martínez-Domingo†
*a and
Marta Mas-Torrent
a,
Sara Ruiz-Molinaa,
Carme Martínez-Domingo†
*a and
Marta Mas-Torrent *ab
*ab
aInstitut de Ciència de Materials de Barcelona, ICMAB-CSIC, Campus UAB, 08193 Bellaterra, Spain. E-mail: carme.martinez@imb-cnm.csic.es; mmas@icmab.es
bCIBER-BBN, Campus UAB, 08193 Bellaterra, Spain
First published on 21st April 2026
Abstract
Electrolyte – Gated Organic Field – Effect Transistors (EGOFETs) have attracted considerable interest as electrical transducers due to their low power consumption and strong potential for label – free biosensing. Hydrogels – polymeric networks capable of absorbing and retaining large amounts of water – have emerged as effective solid electrolyte media, enabling the replacement of liquid electrolytes in EGOFETs to form hydrogel-gated OFETs (HYGOFETs). In this work, we demonstrate the use of HYGOFETs for biosensing applications by employing agarose hydrogels functionalized with avidin as the solid electrolyte. The devices were integrated into a lateral flow system, and their electrical response to biotin was systematically investigated under continuous analysis. The bio-recognition event induced a measurable change in device capacitance, allowing the achievement of a very low limit of detection in the fM range. This study highlights the relevance of bio-compatible solid electrolytes for advancing robust, integrated, and physiologically compatible organic bio-sensing platforms.
1 Introduction
Electrolyte-gated organic field-effect transistors (EGOFETs) are attracting significant interest for the development of low-power electronic devices (typically operating below 1 V).1 The EGOFET architecture relies on the replacement of the conventional dielectric layer of an organic field-effect transistor with an electrolyte medium, which is in direct contact with the organic semiconductor (OSC). The application of a gate–source voltage leads to the formation of electrical double layers at the OSC/electrolyte and electrolyte/gate interfaces, which are responsible for the OSC transport modulation.2,3 In recent years, these devices have attracted significant attention as electronic transducers for biosensing applications, particularly when operated with aqueous electrolytes.4–8 Nevertheless, alternative strategies based on the use of solid electrolytes are gaining increasing interest, as they enable low-voltage operation while providing enhanced device robustness.9–13 Hydrogels are three-dimensional, cross-linked polymer networks that can absorb and retain large amounts of water while maintaining its structural integrity.14 In addition, hydrogels show flexibility,15 tuneable mechanical strength16,17 and excellent biocompatibility.18 These materials have been widely exploited for applications such as sensors,19–22 touch panels,23,24 energy storage systems,25,26 drug delivery,27,28 heavy metal ion removal,29,30 contact lenses31,32 and tissue engineering scaffolds,33 among others.Owing to their high-water content, hydrogels exhibit ultrahigh capacitance values (on the order of μF cm−2). Consequently, hydrogels can function as gel electrolytes in EGOFETs, overcoming the limitations of liquid water-based media and enabling efficient charge accumulation and carrier density modulation at very low voltages.34–36 Hydrogel-gated-OFETs (HYGOFETs) have also been explored as pressure sensors.12 In addition, a few studies have demonstrated the use of HYGOFETs for bio-sensing applications, such as the detection of pH variations, redox reactions, or the encapsulation of bio-chemical reactions.37–40 It is well-established that hydrogels can be readily bio-functionalised,41–43 and that analyte/bio-receptor interactions may alter the material properties, for example by inducing volumetric changes within the hydrogel.44 However, to the best of our knowledge, bio-functionalised hydrogels have not yet been employed in HYGOFET architectures for bio-sensing applications. This work focuses on the production of bio-functionalized agarose hydrogel films and their integration in EGOFETs for sensing applications. As a model system, avidin-modified agarose was employed to detect biotin, exploiting the well-known high affinity and specificity avidin–biotin supramolecular complex.45 First, a protocol was developed to obtain robust and optimised hydrogel films, and the resulting HYGOFETs were electrically characterised. Subsequently, the devices were assembled by integrating a lateral flow (LF) nitrocellulose paper strip into the HYGOFET architecture (LF-HYGOFET), yielding a portable system that enables sample transport via the capillarity action of nitrocellulose and allows continuous analyte flow. Further, this approach eliminates the need to remove the hydrogel for ex situ analyte incubation, thereby simplifying the device operation and enhancing the transducer stability. The devices exhibited a linear response over a wide range of biotin concentrations, which was attributed to changes in the hydrogel capacitance, with a limit of detection in the fM range. Thus, this work demonstrates the feasibility of using bio-functionalised hydrogels for the development of reliable and efficient point-of-care diagnostics tools and sensors.
2 Results and discussion
The HYGOFET device configuration used is shown in Fig. 1a. Interdigitated gold source (S) and drain (D) electrodes as well as a coplanar gate (G) contact were fabricated on Kapton substrates. As active layer, a highly crystalline thin film of the OSC 2,8-difluoro-5,11 bis(triethylsilylethynyl)anthradithiophene (diF-TES-ADT) blended with polystyrene (PS) was prepared using the bar-assisted meniscus shearing (BAMS) technique, as previously reported (Fig. 1b).8,12,46 To fabricate the hydrogel electrolyte matrices, a commercial avidin–modified agarose suspension was employed. Before use, the hydrogel was pre-treated as described in the Experimental section. Briefly, 1 mL of commercial 6% avidin–agarose in an aqueous glycerol suspension was washed by centrifugation to remove glycerol (Fig. S1a and Experimental section). After this treatment, 0.5 mL of a solid packed gel was obtained, containing approximately 60 mg of avidin–agarose. Since avidin–agarose hydrogels alone did not yield mechanically robust and easily handled hydrogels, avidin–agarose was blended with a solution of non-modified agarose 2 wt% in water to obtain the following weight ratios avidin–agarose![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) agarose (1
agarose (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.33, 1
0.33, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 and 1
0.5 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.66) (Fig. S1b). To perform this task, the corresponding volumes of the non-modified agarose solution were directly added to the obtained 0.5 mL avidin–agarose hydrogel. In addition, as control, non-modified agarose hydrogel was also prepared (i.e., ratio 0
0.66) (Fig. S1b). To perform this task, the corresponding volumes of the non-modified agarose solution were directly added to the obtained 0.5 mL avidin–agarose hydrogel. In addition, as control, non-modified agarose hydrogel was also prepared (i.e., ratio 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Avidin–agarose
1). Avidin–agarose![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) agarose hydrogels were prepared by drop casting hot solutions (60 °C) on a square polydimethylsiloxane (PDMS) gasket of 1 cm2 placed on top of a glass slide, and letting the solution to cool down at room temperature. This process resulted in hydrogel films of approximately 3 mm thick. Afterwards, the hydrogel was removed from the substrate and placed on top of the OSC thin film and the gate area.
agarose hydrogels were prepared by drop casting hot solutions (60 °C) on a square polydimethylsiloxane (PDMS) gasket of 1 cm2 placed on top of a glass slide, and letting the solution to cool down at room temperature. This process resulted in hydrogel films of approximately 3 mm thick. Afterwards, the hydrogel was removed from the substrate and placed on top of the OSC thin film and the gate area.
The devices were then electrically characterised. Agarose HYGOFETs, as previously reported, exhibit a slightly lower product of charge carrier mobility and electrical double-layer capacitance (μ·Cdl) and transconductance (gm) values in comparison with devices measured in water (Fig. S2).12 This was attributed to a lower effective concentration of mobile ions within the hydrogel, originating from the entrapped aqueous phase, as well as to reduced ion diffusion caused by steric constraints and interactions with the polymer network, ultimately diminishing their ability to modulate the HYGOFET channel conductivity. Fig. 1c displays the transfer characteristics in linear regime of the HYGOFETs based on hydrogels prepared from the different avidin–agarose![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) agarose ratios together with the response of an HYGOFET based on non-modified agarose for comparison. The extracted μ·Cdl and threshold voltage (Vth) parameters obtained are summarised in Fig. 1d. The device performance exhibited a slight decrease in μ·Cdl in the HYGOFETs based on agarose–avidin
agarose ratios together with the response of an HYGOFET based on non-modified agarose for comparison. The extracted μ·Cdl and threshold voltage (Vth) parameters obtained are summarised in Fig. 1d. The device performance exhibited a slight decrease in μ·Cdl in the HYGOFETs based on agarose–avidin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) agarose 1
agarose 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.33 and 1
0.33 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 compared to those based on non-modified agarose, which became more evident in the 1
0.5 compared to those based on non-modified agarose, which became more evident in the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.66 mixture. Again, this is likely due to a reduced ion concentration and mobility within the matrix. Regarding the Vth, an increase was progressively observed with increasing the proportion of modified agarose. Based on these observations, the 1
0.66 mixture. Again, this is likely due to a reduced ion concentration and mobility within the matrix. Regarding the Vth, an increase was progressively observed with increasing the proportion of modified agarose. Based on these observations, the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.33 ratio was selected as the optimal hydrogel composition for sensing measurements, as it provided the best electrical performance while maximizing the concentration of avidin moieties within the hydrogel.
0.33 ratio was selected as the optimal hydrogel composition for sensing measurements, as it provided the best electrical performance while maximizing the concentration of avidin moieties within the hydrogel.
To proceed with the sensing experiments, a nitrocellulose lateral flow (LF) paper strip was integrated into the HYGOFET, resulting in LF-HYGOFET devices (Fig. S3). We have shown very recently that the combination of EGOFETs with LF assays is a very appealing approach to develop point-of-care sensors, where the nitrocellulose can serve to transport the sample to analyse.47 Additionally, the incorporation of nitrocellulose strip onto the hydrogel enables the continuous supply of the aqueous media into the HYGOFET, avoiding hydrogel dehydration and achieving a stable electrical performance over time (Fig. S4). Firstly, the nitrocellulose strip was treated with bovine serum albumin (BSA) to prevent unspecific biomolecule adsorption48 and then positioned on top of the hydrogel of the HYGOFET, together with a sample pad (SP) and an absorbent pad (AP) at each end of the paper. Prior to biotin detection, the LF-HYGOFET was electrically conditioned to achieve a stable current response (see Experimental section). Subsequently, a reference transfer characteristic curve of the device was recorded using phosphate buffered saline solution (PBS 0.1 M, pH 7.4). Following, a volume of 200 µL of biotin solutions in PBS 0.1 M (pH 7.4) with concentrations ranging from 10−17 to 10−10 M were sequentially introduced in the sample pad, starting from the lowest to the highest concentration. It should be noted that this solution volume was previously optimized to ensure complete diffusion of the sample toward the device interfaces. After flowing each sample, the device was allowed to incubate for 15 minutes followed by a rinsing step. Then, the device transfer characteristic was recorded. This procedure was repeated for each tested concentration. Fig. 2a schematically illustrates the experimental protocol followed. The operation mechanism of an HYGOFET is governed by the formation of electrical double layers at the OSC/electrolyte and gate/electrolyte interfaces, where most of the voltage drop takes place. Thus, the interfacial layer near the device interfaces with the hydrogel (typically on the order of a few angstroms to few nanometres) is expected to effectively contribute to the sensing mechanism (Fig. 2b).
Fig. 3a shows the transfer characteristics obtained upon the exposure of the LF-HYGOFET to each biotin concentration. It can be observed that source–drain current (IDS) decreases while increasing biotin concentration. Electrical variations resulting from the bio-recognition process were assessed by calculating the change in current intensity (ΔI = IDS − IDS,0) at a source–gate voltage (VGS) −0.4 V, where IDS represents the source–drain current measured at a given antigen concentration, and IDS,0 corresponds to the value prior to biotin exposure. Calibration curves, obtained by plotting the normalized ΔI against biotin concentration, are presented in Fig. 3b. A lineal response is observed in the concentration range from 10−15 to 10−11 M. In this region, the data could be fitted giving a slope of 0.05 ± 0.01 M−1 (r2 = 0.997) corresponding to the device sensitivity. Further, a low limit of detection (LOD) of 5.1 fM was extracted (see Experimental section).
In EGOFETs, the product μ·Cdl represents the effective transconductance factor, where μ denotes the OSC charge-carrier mobility and Cdl stands for the interfacial electrical double layer capacitance and quantifies the interfacial charge accumulation at device interfaces, which is responsible for the channel conductivity modulation. When EGOFETs are deployed for biosensing, a binding recognition event can induce different electrical responses depending on the dominant underlying mechanism.49 If the binding event affects the charge carrier mobility or the device capacitance, a change in the slope of the transfer curve is expected. In contrast, when the interaction primarily alters the metal work function (potentiometric effect), a lateral shift of the transfer curve is observed, manifested as a Vth shift.3,50,51 In our experiments, the sensing response leads to a reduction of the μ·Cdl, whilst Vth remains essentially unchanged (Fig. 3c). Thus, the sensing mechanism is primarily governed by a capacitive effect. As the hydrogel is exposed to a higher concentration of biotin, more avidin binding sites from the hydrogel become occupied, forming avidin–biotin complexes.45,52,53 The avidin–biotin binding event occurs throughout the hydrogel, but its electrical effect on the HYGOFET is strictly interfacial (Fig. 2b). Since the device response is governed by the electrical double layer within the Debye length, only recognition events near this region can modulate the effective capacitance, regardless of hydrogel thickness.54 The bio-recognition event might introduce conformational changes in the structure of the agarose hydrogel, as well as alter the charge distribution within the gel. In fact, avidin is positively charged at neutral pH, when it binds to biotin the net positive charge is reduced and the ionic distribution modified, affecting the properties of the electrical double layers at the hydrogel-gate and hydrogel-OSC interfaces.55
To further confirm that the sensing mechanism is capacitive, Electrochemical Impedance Spectroscopy (EIS) was employed to measure the impedance as a function of frequency of the bio-functionalized agarose hydrogels, before and after biotin exposure (Fig. 3d). For this purpose, the avidin–agarose![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) agarose (1
agarose (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.33) hydrogel was exposed to a 10−14 M biotin solution in PBS. As it can be observed, upon addition of biotin, a clear decrease in capacitance was observed, consistent with the trend observed in the transfer characteristics. Capacitance values before and after the biotin exposure were extracted and normalised by area employing the formula described in the Experimental section (Table S1).
0.33) hydrogel was exposed to a 10−14 M biotin solution in PBS. As it can be observed, upon addition of biotin, a clear decrease in capacitance was observed, consistent with the trend observed in the transfer characteristics. Capacitance values before and after the biotin exposure were extracted and normalised by area employing the formula described in the Experimental section (Table S1).
To ensure the specificity of the sensing platform and non-specific biomolecule adsorption, a control experiment was conducted by using an HYGOFET based on non-modified agarose and adding progressively increasing concentrations of the biotin solutions. The results indicate that the IDS fluctuated randomly within a 10–20% range, with no systematic drift observed (Fig. S5). These results confirm that the IDS response is due to the biotin–avidin recognition interaction.
To further examine potential cross-reactivity of the sensing mechanism, an additional experiment was carried out, in which the avidin–agarose![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) agarose (1
agarose (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.33) hydrogel-based LF-HYGOFET was exposed to increasing concentrations of ascorbic acid in PBS 0.1 M, a molecule chosen as a non-target interferent due to its chemical similarity to biotin.56–59 The resulting transfer characteristics are plotted in Fig. 4a, showing no consistent trend in the measured IDS with the exposition of the interferent (Fig. 4b). These findings provide additional evidence that the capacitive changes observed in the biotin sensing experiment are mainly attributed to the specific interaction of biotin with avidin.
0.33) hydrogel-based LF-HYGOFET was exposed to increasing concentrations of ascorbic acid in PBS 0.1 M, a molecule chosen as a non-target interferent due to its chemical similarity to biotin.56–59 The resulting transfer characteristics are plotted in Fig. 4a, showing no consistent trend in the measured IDS with the exposition of the interferent (Fig. 4b). These findings provide additional evidence that the capacitive changes observed in the biotin sensing experiment are mainly attributed to the specific interaction of biotin with avidin.
3 Conclusions
In this study, agarose-based hydrogels were employed as an alternative to conventional liquid electrolytes in EGOFETs, leading to the development of HYGOFETs. Agarose was chosen as the hydrogel matrix due to its low cost, wide availability, and simple gelation process. To demonstrate the potential of these devices for biosensing applications, a receptor biomolecule, avidin, was incorporated into the hydrogel matrix. The sensing performance was investigated by integrating the HYGOFET with a lateral flow system, enabling continuous and real-time detection. The platform was validated through the detection of avidin–biotin interactions, which induced changes in the electrical double-layer capacitance and, consequently, in the measured source–drain current, achieving a very low limit of detection of ∼5 fM.These results show that HYGOFETs retain the key advantages of conventional EGOFETs, such as low operating voltage and compatibility with aqueous environments, while offering easier handling and simplified fabrication. Furthermore, we demonstrate that, through chemical modification of the hydrogel matrix, these devices can be effectively employed for highly sensitive bio-sensing applications. Importantly, the hydrogels used in this work are biocompatible and biodegradable, making the proposed platform particularly attractive for sustainable and bio-integrated sensing technologies.
4 Experimental section
4.1 Materials
Agarose powder (Type I–B, low EEO), polystyrene (Mw = 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1), anhydrous 99.8% chlorobenzene (CB), 2,3,4,5,6-pentafluorothiophenol (PFBT), Avidin–agarose from egg white – aqueous glycerol 50% suspension, Bovine Serum Albumin (BSA, Mw = 66 kDa), biotin (≥99%, HPLC) lyophilized powder and L-ascorbic acid (99%) were purchased from Sigma-Aldrich. Polydimethylsiloxane (PDMS), Qsil216 A/B for gasket production was purchased from Farnell Components. 2,8-Difluoro-5,11-bis(triethylsilylethynyl)anthradithiophene (diF TES-ADT, purity >99%) racemic mixture was purchased from Luminisence Technology Corp (LUMTEC). Acetone and isopropanol HPLC grade were purchased from Chem-Lab. All these commercial materials were used without any further purification. Kapton® foils, used as substrate, were bought from DuPont (Kapton®HN, 125 μm thick). Lateral flow components, unbacked nitrocellulose test membrane (AE98), sample and absorbent pads (CF4) were obtained from Cytiva Lifescience.
000 g mol−1), anhydrous 99.8% chlorobenzene (CB), 2,3,4,5,6-pentafluorothiophenol (PFBT), Avidin–agarose from egg white – aqueous glycerol 50% suspension, Bovine Serum Albumin (BSA, Mw = 66 kDa), biotin (≥99%, HPLC) lyophilized powder and L-ascorbic acid (99%) were purchased from Sigma-Aldrich. Polydimethylsiloxane (PDMS), Qsil216 A/B for gasket production was purchased from Farnell Components. 2,8-Difluoro-5,11-bis(triethylsilylethynyl)anthradithiophene (diF TES-ADT, purity >99%) racemic mixture was purchased from Luminisence Technology Corp (LUMTEC). Acetone and isopropanol HPLC grade were purchased from Chem-Lab. All these commercial materials were used without any further purification. Kapton® foils, used as substrate, were bought from DuPont (Kapton®HN, 125 μm thick). Lateral flow components, unbacked nitrocellulose test membrane (AE98), sample and absorbent pads (CF4) were obtained from Cytiva Lifescience.
4.2 EGOFET manufacture
For EGOFET devices fabrication, interdigitated source (S), drain (D), and coplanar gate (G) electrodes were patterned on 125 µm thick Kapton foil substrates using positive photolithography (Micro-Writer ML2, Durham Magneto Optics Ltd). Metallization was carried out via thermal evaporation (System Auto 360, BOC Edwards), depositing a 5 nm chromium adhesion layer followed by a 40 nm gold layer. The resulting channel dimensions were 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 µm in width (W) and 50 µm in length (L), yielding a W/L ratio of 360. The area of the gate electrode was of 0.15 cm2. The lift-off process was performed by sequential cleaning in acetone and isopropanol for 5 minutes each, repeated twice.
000 µm in width (W) and 50 µm in length (L), yielding a W/L ratio of 360. The area of the gate electrode was of 0.15 cm2. The lift-off process was performed by sequential cleaning in acetone and isopropanol for 5 minutes each, repeated twice.
Prior to active layer deposition, the electrodes underwent ultrasonic cleaning in acetone and isopropanol. The organic semiconductor (OSC) deposition protocol followed previously reported procedures.46 Briefly, the substrates with patterned gold electrodes were first exposed to UV-ozone for 25 minutes, then immersed in a 15 mM solution of PFBT in isopropanol for 15 minutes and rinsed. Subsequently, a 2 wt% solution of diF-TES-ADT and polystyrene (PS) in a 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 volume ratio, dissolved in CB, was deposited using the Bar-Assisted Meniscus Shearing (BAMS) technique.46,54 The coating process was carried out at a shearing speed of 10 mm s−1 while maintaining the substrate temperature at 105 °C. Polarized optical microscopy (POM) images were acquired in reflection mode using an Olympus BX51 microscope equipped with a polarizer and analyzer set at 90°.
1 volume ratio, dissolved in CB, was deposited using the Bar-Assisted Meniscus Shearing (BAMS) technique.46,54 The coating process was carried out at a shearing speed of 10 mm s−1 while maintaining the substrate temperature at 105 °C. Polarized optical microscopy (POM) images were acquired in reflection mode using an Olympus BX51 microscope equipped with a polarizer and analyzer set at 90°.
4.3 Hydrogel preparation
Agarose films were prepared using the BAMS technique at a coating speed of 10 mm s−1 and keeping the plate temperature at 30 °C, close to the agarose jellification temperature. Before deposition, the tip of the micropipette was kept in hot agarose solution (120 °C) for some seconds to avoid the jellification inside the plastic tip. Upon cooling down to room temperature, the resulting agarose film was subsequently cut into square pieces (around 1 cm2) and immediately used as electrolyte in the transistors by sandwiching it on top on the prepared devices.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) agarose solution. Commercial agarose–avidin hydrogel is delivered initially suspended in glycerol, which acts as stabilizing agent. To remove the glycerol, 1 mL of the agarose–avidin suspension was washed with 1× phosphate-buffered saline (PBS 0.1 M; pH = 7.4) in an Eppendorf tube, followed by centrifugation at 2000 rpm. The resulting supernatant was carefully discarded to eliminate residual glycerol. This washing step was repeated three additional times using 0.5 mL of PBS per wash. From each 1 mL of the original agarose suspension, approximately 0.5 mL of solid packed hydrogel was recovered. Since these gels are hard to manipulate, the suspensions were mixed with agarose. Therefore, agarose–avidin
agarose solution. Commercial agarose–avidin hydrogel is delivered initially suspended in glycerol, which acts as stabilizing agent. To remove the glycerol, 1 mL of the agarose–avidin suspension was washed with 1× phosphate-buffered saline (PBS 0.1 M; pH = 7.4) in an Eppendorf tube, followed by centrifugation at 2000 rpm. The resulting supernatant was carefully discarded to eliminate residual glycerol. This washing step was repeated three additional times using 0.5 mL of PBS per wash. From each 1 mL of the original agarose suspension, approximately 0.5 mL of solid packed hydrogel was recovered. Since these gels are hard to manipulate, the suspensions were mixed with agarose. Therefore, agarose–avidin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) agarose mixtures were prepared using the avidin
agarose mixtures were prepared using the avidin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) agarose packed gel obtained and adding different volumes (1, 1.5 and 2 ml) of the agarose suspension 2% (w/w) to obtain the following w/w mixture ratios: 1
agarose packed gel obtained and adding different volumes (1, 1.5 and 2 ml) of the agarose suspension 2% (w/w) to obtain the following w/w mixture ratios: 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.33, 1
0.33, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5 and 1
0.5 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.66. The mixtures were heated at 60 °C, to prevent avidin denaturalization, until a homogeneous solution was obtained. A polydimethylsiloxane (PDMS) gasket was employed for the formation of a gel with a specific area. Hence, the hot solutions (60 °C) were deposited on a square PDMS pool of 1 cm2 and let to cool down at room temperature, leading to films 0.5 cm thick. To prevent dehydration of the gels, all the prepared hydrogels were stored in a refrigerator at 5 °C.
0.66. The mixtures were heated at 60 °C, to prevent avidin denaturalization, until a homogeneous solution was obtained. A polydimethylsiloxane (PDMS) gasket was employed for the formation of a gel with a specific area. Hence, the hot solutions (60 °C) were deposited on a square PDMS pool of 1 cm2 and let to cool down at room temperature, leading to films 0.5 cm thick. To prevent dehydration of the gels, all the prepared hydrogels were stored in a refrigerator at 5 °C.4.4 LF – HYGOFET assembly
The prepared hydrogel was deposited on top of the device ensuring covering the organic semiconductor channel and gate electrode. Before assembling the LF strip on the HYGOFET, 1 mL of a BSA solution in PBS (1 mg ml−1) was deposited on the nitrocellulose strip and sample pad at let it react for 20 minutes at 37 °C in order to avoid unspecific absorption along the strip test. Afterwards, the LF strip paper was positioned on top of the hydrogel. At the edges of the nitrocellulose paper, a sample pad and an absorbent pad were placed, maintaining a 2 mm overlap on each side to ensure proper capillary flow. Since the absorbent pad does not directly influence or interfere with the device response, it was not treated with BSA.4.5 LF – HYGOFET characterization
LF-HYGOFETs were electrically characterized under ambient conditions using a Keithley 2612A Source Meter controlled via a custom Phyton script. Initially, LF-HYGOFET devices were conditioned by applying a source–gate voltage (VGS) of −0.1 V and a source–drain voltage (VDS) of −0.1 V. This conditioning continued for approximately 30 minutes until the source–drain current (IDS) stabilized. Once a steady-state was achieved, the initial transfer curve and output characteristics were recorded and used as a reference to assess device performance.To monitor the electrical changes in performance induced by the sensing event, device figures of merit, including the threshold voltage (Vth) and the product of mobility (µ) and double-layer capacitance (Cdl), were derived from the slope (b) and y-intercept (a) of the linear fitting of each transfer characteristic using the following equations:
4.6 Sensing experiments
Sensing experiments were conducted using a single HYGOFET device integrated into a LF system. This setup enabled continuous operation and helped to prevent dehydration of the hydrogel. Initially, the device was conditioned by flowing 0.1 M a PBS solution (pH 7.4) through the nitrocellulose membrane to ensure proper hydration of the hydrogel. Once a steady-state drain–source current was achieved, 200 µL of biotin or interferent solutions at varying concentrations (ranging from 10−17 to 10−10 M) were applied dropwise onto the sample pad. After a 15-minute incubation period to allow complete interaction with the hydrogel, the membrane was rinsed. This rinsing step consisted of dispensing 200 µL of 0.1 M PBS onto the sample pad, allowing it to flow along the nitrocellulose strip by capillary action, in order to remove unbound or loosely adsorbed biomolecules from the nitrocellulose membrane. Following a 5-minute stabilization period, the electrical response of the LF-HYGOFET was recorded by measuring the transfer characteristics in the linear regime (VDS = −0.1 V).Sensitivity was determined by calculating the slope of the calibration curve. The limit of detection (LOD) has been calculated using the following equation:
4.7 Capacitance measurements
The electrical double layer (EDL) capacitance values were obtained by employing the Electrochemical Impedance Spectroscopy (EIS) technique. Measurements were carried out with Autolab potentiostat/galvanostat (PGSTAT128N). Setup measurements relied on the sandwiching the hydrogel in between two gold electrodes of 1 cm2, which were connected to the potentiostat and used as working and auxiliary electrodes.Impedance spectra (capacitance vs. frequency) were recorded at different DC bias potentials within the operational window of the HYGOFET, using a small-signal AC perturbation of 5 mV (VRMS). Capacitance was obtained from the imaginary part of the impedance according to:
Author contributions
M. O.-A. and S. R.-M. performed the experimental work and the data analysis. C. M.-D. and M. M.-T. supervised the project. M. M-T. conceived the idea. M. O.-A, C. M.-D. and M. M.-T. co-wrote the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data published in this paper will be provided by the authors when requested.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d6nr00527f.
Acknowledgements
This work was funded by MCIN/AEI/10.13039/501100011033/ERDF, UE, with projects SENSATION PID2022-141393OB-I00 and PDC2021-121511-I00, and through the “Severo Ochoa” Programme for Centers of Excellence in R&D (CEX2023-001263-S). The authors also acknowledge funds from the Generalitat de Catalunya (2021-SGR-00443). S. R. acknowledges her FPI fellowship. S. R. and M. O. are enrolled at the UAB Chemistry PhD program. The authors thank Dr Dean Kos for software development.References
- L. Torsi, M. Magliulo, K. Manoli and G. Palazzo, Organic field-effect transistor sensors: a tutorial review, Chem. Soc. Rev., 2013, 42, 8612 RSC.
- F. Torricelli, et al., Electrolyte-gated transistors for enhanced performance bioelectronics, Nat. Rev. Methods Primers, 2021, 1, 66 CrossRef CAS PubMed.
- L. Kergoat, et al., Tuning the threshold voltage in electrolyte-gated organic field-effect transistors, Proc. Natl. Acad. Sci. U. S. A., 2012, 109, 8394–8399 CrossRef CAS PubMed.
- G. Y. Wang, K. Lian and T.-Y. Chu, Electrolyte-Gated Field Effect Transistors in Biological Sensing: A Survey of Electrolytes, IEEE J. Electron Devices Soc., 2021, 9, 939–950 CAS.
- Y. Liang, et al., Bioinspired Electrolyte-Gated Organic Synaptic Transistors: From Fundamental Requirements to Applications, Nano-Micro Lett., 2025, 17, 198 CrossRef PubMed.
- S. K. Sailapu, et al., Standalone operation of an EGOFET for ultra-sensitive detection of HIV, Biosens. Bioelectron., 2020, 156, 112103 CrossRef CAS PubMed.
- E. Macchia, et al., Single-molecule detection with a millimetre-sized transistor, Nat. Commun., 2018, 9, 3223 CrossRef PubMed.
- S. Ricci, et al., Label-free immunodetection of α-synuclein by using a microfluidics coplanar electrolyte-gated organic field-effect transistor, Biosens. Bioelectron., 2020, 167, 112433 CrossRef CAS PubMed.
- Y. Zhong, et al., Eutectogels as a Semisolid Electrolyte for Organic Electrochemical Transistors, Chem. Mater., 2024, 36, 1841–1854 CrossRef CAS PubMed.
- A. Alyami, M. Skowrons, K. Perera, B. Lüssem and A. Jákli, Performance of Organic Electrochemical Transistors with Ionic Liquid Crystal Elastomers as Solid Electrolytes, ACS Appl. Mater. Interfaces, 2024, 16, 54282–54291 CrossRef PubMed.
- B. Singh, R. A. Mishra and D. Punetha, Optimizing performance of electrolyte-gated organic field-effect transistors for biosensing: material variations and parameter analysis, Discov. Electron., 2025, 2, 20 CrossRef.
- Q. Zhang, F. Leonardi, R. Pfattner and M. Mas-Torrent, A Solid–State Aqueous Electrolyte–Gated Field–Effect Transistor as a Low–Voltage Operation Pressure–Sensitive Platform, Adv. Mater. Interfaces, 2019, 6, 1900719 CrossRef.
- A. Miyamoto, et al., Solid Electrolyte Gas Sensor Based on a Proton-Conducting Graphene Oxide Membrane, ACS Omega, 2017, 2, 2994–3001 CrossRef CAS PubMed.
- F. Ullah, M. B. H. Othman, F. Javed, Z. Ahmad and H. M. Akil, Classification, processing and application of hydrogels: A review, Mater. Sci. Eng., C, 2015, 57, 414–433 CrossRef CAS PubMed.
- Nano Hydrogels: Physico-Chemical Properties and Recent Advances in Structural Designing, Springer Singapore, Singapore, 2021. DOI:10.1007/978-981-15-7138-1.
- A. S. Maier, et al., Cytocompatible Hydrogels with Tunable Mechanical Strength and Adjustable Swelling Properties through Photo-Cross-Linking of Poly(vinylphosphonates), ACS Appl. Mater. Interfaces, 2024, 16, 58135–58147 CrossRef CAS PubMed.
- L. Fu and H. Li, Ionic Crosslinking Improves the Stiffness and Toughness of Protein Hydrogels, Polym. Sci. Technol., 2025, 1, 342–350 CrossRef CAS PubMed.
- M. M. H. Rumon, et al., Progress in hydrogel toughening: addressing structural and crosslinking challenges for biomedical applications, Discover Mater., 2025, 5, 5 CrossRef CAS.
- D. Buenger, F. Topuz and J. Groll, Hydrogels in sensing applications, Prog. Polym. Sci., 2012, 37, 1678–1719 CrossRef CAS.
- Y. Lin, et al., Recent progress of nanomaterials-based composite hydrogel sensors for human–machine interactions, Discover Nano, 2025, 20, 60 CrossRef PubMed.
- K. Nagamine, et al., Noninvasive Sweat-Lactate Biosensor Emplsoying a Hydrogel-Based Touch Pad, Sci. Rep., 2019, 9, 10102 CrossRef PubMed.
- T. Du, Z. Zhu, M. Chen, X. Yan and Y. Li, Functional Hydrogel Strain Sensors for Smart Electronic Devices: Strategies and Recent Progress, ACS Appl. Electron. Mater., 2024, 6(8), 5402–5428 CAS.
- Y. Li, et al., An ultrastretchable and multifunctional hydrophobic/electrostatic dual-crosslinked hydrogel for self-healing flexible touch panel and sensor, npj Flexible Electron., 2025, 9, 45 CrossRef CAS.
- T. Feng, et al., Stretchable on-skin touchless screen sensor enabled by ionic hydrogel, Nano Res., 2024, 17, 4462–4470 CrossRef.
- Z. Shen, et al., Hydrogel Electrolytes-Based Rechargeable Zinc-Ion Batteries under Harsh Conditions, Nano-Micro Lett., 2025, 17, 227 CrossRef CAS PubMed.
- K. Sadaiyandy, et al., A review on redox hydrogel electrolyte for energy storage devices, Ionics, 2025, 31, 8837–8873 CrossRef CAS.
- H. Hameed, S. Faheem, A. C. Paiva-Santos, H. S. Sarwar and M. Jamshaid, A Comprehensive Review of Hydrogel-Based Drug Delivery Systems: Classification, Properties, Recent Trends, and Applications, AAPS PharmSciTech, 2024, 25, 64 CrossRef PubMed.
- M. Baghali, H. Ziyadi and A. Di Martino, Engineering smart hydrogels for intelligent drug delivery, Polym. Bull., 2025, 82, 2287–2328 CrossRef CAS.
- S. Lee, Y. Eom, J. Park, J. Lee and S. Y. Kim, Micro-hydrogel Particles Consisting of Hyperbranched Polyamidoamine for the Removal of Heavy Metal Ions from Water, Sci. Rep., 2017, 7, 10012 CrossRef PubMed.
- D. Gokhale, A. F. Hamelberg and P. S. Doyle, Multifunctional zwitterionic hydrogels for the rapid elimination of organic and inorganic micropollutants from water, Nat. Water, 2024, 2, 62–71 CrossRef CAS.
- M. Hisham, A. E. Salih and H. Butt, 3D Printing of Multimaterial Contact Lenses, ACS Biomater. Sci. Eng., 2023, 9, 4381–4391 CrossRef CAS PubMed.
- K. Ishihara, et al., Biomimetic-Engineered Silicone Hydrogel Contact Lens Materials, ACS Appl. Bio Mater., 2023, 6, 3600–3616 CrossRef CAS PubMed.
- B. S. MohanKumar, et al., Hydrogels: potential aid in tissue engineering—a review, Polym. Bull., 2022, 79, 7009–7039 CrossRef CAS.
- L. M. Dumitru, et al., A hydrogel capsule as gate dielectric in flexible organic field-effect transistors, APL Mater., 2015, 3, 014904 CrossRef.
- L. Fillaud, et al., Switchable Hydrogel-Gated Organic Field-Effect Transistors, Langmuir, 2018, 34, 3686–3693 CrossRef CAS PubMed.
- M. Azimi, A. Subramanian, J. Fan, F. Soavi and F. Cicoira, Electrical and mechanical stability of flexible, organic electrolyte-gated transistors based on iongel and hydrogels, J. Mater. Chem. C, 2023, 11, 4623–4633 RSC.
- G. Li, et al., Acrylamide Hydrogel-Modified Silicon Nanowire Field-Effect Transistors for pH Sensing, Nanomaterials, 2022, 12, 2070 CrossRef CAS PubMed.
- H. D. Chirra and J. Z. Hilt, Nanoscale Characterization of the Equilibrium and Kinetic Response of Hydrogel Structures, Langmuir, 2010, 26, 11249–11257 CrossRef CAS PubMed.
- J. L. Gall, et al., Algae-functionalized hydrogel-gated organic field-effect transistor. Application to the detection of herbicides, Electrochim. Acta, 2021, 372, 137881 CrossRef CAS.
- J. Pallu, et al., A DNA hydrogel gated organic field effect transistor, Org. Electron., 2019, 75, 105402 CrossRef CAS.
- F. Qu, Y. Zhang, A. Rasooly and M. Yang, Electrochemical Biosensing Platform Using Hydrogel Prepared from Ferrocene Modified Amino Acid as Highly Efficient Immobilization Matrix, Anal. Chem., 2014, 86, 973–976 CrossRef CAS PubMed.
- S. Hageneder, et al., Responsive Hydrogel Binding Matrix for Dual Signal Amplification in Fluorescence Affinity Biosensors and Peptide Microarrays, ACS Appl. Mater. Interfaces, 2021, 13, 27645–27655 CrossRef CAS PubMed.
- P. Shen, et al., Aptamer-functionalized smart photonic hydrogels: application for the detection of thrombin in human serum, NPG Asia Mater., 2022, 14, 94 CrossRef CAS.
- K. Cherifi and S. Matoori, Hydrogels for Analyte Sensing, ACS Meas. Sci. Au, 2025, 5(6), 771–779 CrossRef CAS PubMed.
- R. F. Delgadillo, et al., Detailed characterization of the solution kinetics and thermodynamics of biotin, biocytin and HABA binding to avidin and streptavidin, PLoS One, 2019, 14, e0204194 CrossRef CAS PubMed.
- I. Temiño, et al., A Rapid, Low–Cost, and Scalable Technique for Printing State–of–the–Art Organic Field–Effect Transistors, Adv. Mater. Technol., 2016, 1, 1600090 CrossRef.
- M. J. Ortiz-Aguayo, C. Martínez-Domingo, D. Gutiérrez, D. Kos and M. Mas-Torrent, Electrically Readable Lateral Flow Assay Using Organic Transistors for Diagnostic Applications, Adv. Mater., 2025, e13468, DOI:10.1002/adma.202513468.
- X. Jiang and P. B. Lillehoj, Lateral flow immunochromatographic assay on a single piece of paper, Analyst, 2021, 146, 1084–1090 RSC.
- K. Manoli, et al., Printable Bioelectronics To Investigate Functional Biological Interfaces, Angew. Chem., Int. Ed., 2015, 54, 12562–12576 CrossRef CAS PubMed.
- F. Leonardi, A. Tamayo, S. Casalini and M. Mas-Torrent, Modification of the gate electrode by self-assembled monolayers in flexible electrolyte-gated organic field effect transistors: work function vs. capacitance effects, RSC Adv., 2018, 8, 27509–27515 RSC.
- Q. Zhang, A. Tamayo, F. Leonardi and M. Mas-Torrent, Interplay between Electrolyte-Gated Organic Field-Effect Transistors and Surfactants: A Surface Aggregation Tool and Protecting Semiconducting Layer, ACS Appl. Mater. Interfaces, 2021, 13, 30902–30909 CrossRef CAS PubMed.
- O. Livnah, E. A. Bayert, M. Wilchekt and J. L. Sussman, Three-dimensional structures of avidin and the avidin-biotin complex, Proc. Natl. Acad. Sci. U. S. A., 1993, 90(11), 5076–5080 CrossRef CAS PubMed.
- P. Strzelczyk, D. Plażuk, J. Zakrzewski and G. Bujacz, Structural characterization of the avidin interactions with fluorescent pyrene-conjugates: 1-biotinylpyrene and 1-desthiobiotinylpyrene, Molecules, 2016, 21, 1270 CrossRef PubMed.
- G. Palazzo, et al., Detection Beyond Debye's Length with an Electrolyte–Gated Organic Field–Effect Transistor, Adv. Mater., 2015, 27, 911–916 CrossRef CAS PubMed.
- R. J. McMahon, Avidin-Biotin Interactions: Methods and Applications, Humana press, Totowa (N.J.), 2008 Search PubMed.
- S. Wang, et al., Avidin–Biotin Technology in Gold Nanoparticle-Decorated Graphene Field Effect Transistors for Detection of Biotinylated Macromolecules with Ultrahigh Sensitivity and Specificity, ACS Omega, 2020, 5, 30037–30046 CrossRef CAS PubMed.
- D. Ortiz-Aguayo, M. Bonet-San-Emeterio and M. Del Valle, Simultaneous Voltammetric Determination of Acetaminophen, Ascorbic Acid and Uric Acid by Use of Integrated Array of Screen-Printed Electrodes and Chemometric Tools, Sensors, 2019, 19, 3286 CrossRef CAS PubMed.
- H. S. Magar, A. M. Fahim and M. S. Hashem, Accurate, affordable, and easy electrochemical detection of ascorbic acid in fresh fruit juices and pharmaceutical samples using an electroactive gelatin sulfonamide, RSC Adv., 2024, 14, 39820–39832 RSC.
- S. Rantataro, L. Ferrer Pascual and T. Laurila, Ascorbic acid does not necessarily interfere with the electrochemical detection of dopamine, Sci. Rep., 2022, 12, 20225 CrossRef CAS PubMed.
Footnote |
| † Current address: Institut de Microelectrònica de Barcelona, IMB-CNM (CSIC), Campus UAB, 08193 Bellaterra, Spain. |
| This journal is © The Royal Society of Chemistry 2026 |