 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Nanowire photodetectors: path to single physical artificial neurons
Yunqiu
Chen†
,
Milad
Fathabadi†
,
Mohammad Fazel
Vafadar
and
Songrui
Zhao
 *
*
Department of Electrical and Computer Engineering, McGill University, 3480 University Street, Montreal, Quebec H3A 0E9, Canada. E-mail: songrui.zhao@mcgill.ca; Tel: (+1) 514-398-3244
First published on 31st March 2026
Abstract
Neurons are basic units for information processing in biological systems. Inspired by the information-processing efficiency of biological systems, artificial neural networks (ANNs) have been studied for decades. However, it remains challenging today to realize single physical artificial neurons so that an ANN can be constructed in a bottom-up manner. Herein, we show that by using an innovative light-detecting semiconductor nanowire structure, neuronal features much closer to single, physical artificial neurons can be emulated, including excitation and inhibition, threshold, refractory period, and temporal summation. The analogy between the presented neuron-like nanowire photodetectors and neurons in the human retina system is also discussed. This study lays the cornerstone for the bottom-up construction of a physical ANN.
Introduction
Despite the exciting progress with artificial intelligence, which has improved our everyday lives considerably, the ANNs we are dealing with today are based either on mathematical functions or large-scale circuitry that model the behavior of biological neuronal systems, rather than being constructed from single physical artificial neurons.1–10 This is because single artificial neurons have yet to be achieved. Fig. 1a illustrates the basic structure of a neuron (axodendritic kind), including the axon, which functions as the primary output pathway, and the dendrite, which receives the input from other neurons. These structures communicate at the synaptic cleft – a specialized junction that facilitates inter-neuronal communication. Signal transmission across the synaptic cleft is mediated by neurotransmitters (chemical messengers) released from the presynaptic neuron. The type of synapse between neurons (excitatory or inhibitory), along with the strength and properties of the presynaptic and the postsynaptic neurons, is highly critical for information processing in such a neural network.11To achieve single physical artificial neurons, one must demonstrate a small-scale device roughly the size of typical neurons (e.g., tens of µm) that does not rely on large-scale circuits and can emulate as many neuronal features as possible, such as temporal summation, excitation, inhibition, threshold, and refractory period. In recent years, we have witnessed an increasing number of studies using semiconductor devices to emulate neurons. However, constrained by their physical properties, existing semiconductor devices can only emulate limited neuronal features and thus do not yet closely resemble single physical neurons. For example, existing studies can only emulate excitation and temporal summation but cannot reproduce inhibition, threshold, and refractory period simultaneously.12–16
In this study, we demonstrate an innovative semiconductor nanowire photodetector based on a nanowire ensemble that can emulate features much closer to single, physical neurons (Fig. 1b). Thanks to the rich photoresponse of the presented device, rich neuronal features, including excitation, inhibition, threshold, refractory period, and temporal summation, can be emulated. The rich photoresponse demonstrated in the present semiconductor device has not been achieved previously. In the present study, the very rich photoresponse is made possible by leveraging the unique physical properties of semiconductor III-nitrides, including wide-range optical bandgap tunability that can tune the photoabsorption properties, excellent strain relaxation in nanowires that allow highly flexible material stacking with a minimum penalty from lattice mismatch, and careful engineering of device layers, such as the choice of materials and alloy composition. The operating voltage for such devices is only tens of millivolts, significantly lower than the previously reported devices (several volts to tens of volts), which can only emulate some neuronal features.15,16
Structural properties of the light-detecting nanowires
Fig. 2a illustrates the layer-by-layer structure of the individual nanowire obtained using energy dispersive X-ray spectroscopy (EDXS) imaging. Each layer is clearly visible and labeled. Such a structure is denoted as a PNNP structure. A scanning electron microscopy (SEM) image of a nanowire ensemble is shown in Fig. 2b. It is seen that nearly vertically aligned nanowires are formed. Such nanowires are formed in a self-assembled manner, without any additional processes such as substrate patterning. The nanowires also have excellent interface quality, especially at the n-GaN/n-AlGaN interface, which is critical to this study. Additional electron microscopy studies can be found in SI Note 1.The alloy compositions of the layers were examined using room-temperature photoluminescence spectroscopy (RTPL). The details can be found in SI Note 2. To summarize here, the p-InGaN layer absorbs 405 nm blue incident light (used as the control light), and 266 nm ultraviolet (UV) light (used as the external stimulus) is absorbed by both the p-InGaN and n-GaN layers.
Photocarrier dynamics
Fig. 2c illustrates the energy band diagram of the PNNP structure. Under UV light illumination, although both the GaN and p-InGaN layers absorb the light, due to the large band offset at the n-AlGaN/p-InGaN interface and the presumably better light absorption of the GaN layer compared to the InGaN layer due to the greater thickness, a net positive photocurrent is generated.17 It is important to note that, different from thin films in which the light path has to be vertical, in nanowires, the light can be scattered to the nanowire root and absorbed. Moreover, further examining the n-GaN/n-AlGaN interface, the downward band bending on the n-GaN side traps electrons. This is also illustrated in Fig. 2c. Due to the electron trap, when the light is turned off, the slow release of the trapped electrons contributes to a persistent positive photocurrent, whereas when the light is turned back on, the photocurrent continues to increase, as illustrated in Fig. 2d.The charge trap at the n-GaN/n-AlGaN interface also produces a rich photoresponse under blue light stimulation. In this case, the light is only absorbed by the p-InGaN layer, and thus a negative photocurrent is generated. However, due to the trapped electrons at the n-GaN/n-AlGaN interface (n-GaN side), a positive current spike appears immediately when the blue light is turned off with a small bias voltage (∼50 mV) applied, as illustrated in Fig. 2e. This small, applied bias is to overcome the potential barrier for the charge carrier transport. As the charge accumulation takes time, the longer that the blue light is kept on before being turned off, the larger the positive photocurrent spike will be; whereas when the blue light is chopped frequently, the positive photocurrent spike is noticeably smaller, as illustrated in the inset of Fig. 2e.
In this study, the charge trapping effect at the n-GaN/n-AlGaN interface is also examined for the comparison nanostructure (PNP), wherein the n-GaN layer between p-GaN and n-AlGaN is removed (and thus the electron trap is removed). In this case, the temporal summation of the positive photocurrent under the UV light illumination disappears. More details about the comparison between the PNNP and PNP structures can be found in SI Notes 3 and 4.
Neuron-like characteristics
Here, we will describe how to use the presented semiconductor nanowire photodetectors to emulate various neuronal features. Excitation is described first. In this case, one can simply turn off the blue control light or modulate (i.e., on and off) the control light frequently. The time-dependent photocurrent of the former is shown in Fig. 2d, whereas for the latter it is shown in Fig. 3a (light profiles) and 3b (light response). In either case, treating the time-dependent photocurrent as the time-dependent postsynaptic potential in the postsynaptic neuron, excitation can be seen, i.e., the membrane potential in the postsynaptic neuron is more positive and thus has an increased likelihood of firing. Moreover, when a pulse train is used (i.e., the UV light is repeatedly turned on and off), the consecutive pulse generates a higher photocurrent compared to the previous pulse. This mimics pulse-number-dependent plasticity (and here, it is facilitation), i.e., the consecutive pulse generates a higher membrane potential in the postsynaptic neuron compared to the previous pulse, and could be treated as short-term plasticity.18–20 Temporal summation is also seen, i.e., successive input signals add up.We next consider inhibition. In this case, as the control light leads to a negative photocurrent (when it is turned on), one can utilize this feature to introduce inhibition. For example, when the light intensity of the control light is increased continuously (Fig. 3c), the time-dependent photocurrent decreases under each consecutive pulse of the UV light (Fig. 3d). This could emulate inhibition of neurons, namely, a more negative membrane potential in the postsynaptic neuron and thus a reduced likelihood of firing. Moreover, when the input signal is a pulse strain, pulse-number-dependent plasticity is also seen (and in this case, it is depression). Furthermore, just like excitation, temporal summation is also seen for inhibition, i.e., successive input signals add up, although it is a decrease. It is further noted that: (1) simply reducing the UV light could also reduce the photocurrent under consecutive pulses, and we consider it as a trivial case; (2) the purpose of the control light is to reduce the photocurrent; as such, the choice of light intensity profile of the control light can be highly flexible. Essentially, the change of the control light may mimic the release of neurotransmitters (e.g., glutamates or gamma-aminobutyric acid [GABA], as shown in Fig. 1a). However, it needs to be made clear that, different from typical biological neurons wherein such control is coded in a protein inside the neuron, the blue control light here is external to the artificial neuron.
Lastly, we consider emulating the threshold and refractory period related to spiking neurons. For spiking neurons, an action potential can be fired when the membrane potential exceeds the threshold. In this case, at least four key features need to be shown: (1) attempt to fire, i.e., an increase of membrane potential, (2) firing action potential (a pulse signal), (3) reset, and (4) refractory period, i.e., neurons cannot fire again even if the stimulus is present.3 To emulate these features, one can use the control light profile as shown in Fig. 3e, i.e., on with brief off. The time-dependent photocurrent is shown in Fig. 3f, which can emulate attempts to fire (small photocurrent spikes), firing action potential (a large photocurrent spike), reset (the photocurrent drops back to baseline), and refractory period (gap between two large photocurrent spikes). It is also noted that pulse-number-dependent facilitation is also evident here between each fire, similar to Fig. 2d and 3b.
More analysis on pulse-number-dependent facilitation
The facilitation index is analyzed first. In this regard, as the plasticity is associated with the positive photocurrent under UV light stimulus, the blue control light was not used. The facilitation index under a pulse train is measured by the photocurrent difference between two consecutive light pulses, i.e., (In+1 − In)/In, where In is the photocurrent under the nth light stimulus (as denoted in Fig. 2d). Fig. 4a shows the facilitation index versus each stimulus under different applied bias voltages. It is seen that the facilitation index is less sensitive to bias. | ||
| Fig. 4 More analysis on pulse-number-dependent facilitation. (a and b) Facilitation index versus each stimulus under different applied bias voltages and under different excitation powers, respectively, as denoted in Fig. 2d. (c) Photocurrent profiles versus pulse numbers. (d) Time of synaptic strength decay versus pulse numbers. More details are described in the main text. For (c) and (d), the applied bias is 50 mV, and the UV light average power density is 3 mW cm−2. | ||
This is an important feature, meaning the presented neuron-like nanowire photodetectors can “learn” with very low power consumption. In contrast, for the previously demonstrated devices, much larger voltages (several volts to tens of volts) are required to operate, meaning significantly higher power consumption.16,21,22 Moreover, as the power efficiency of III-nitride semiconductor light sources can be higher than 50%, e.g., ref. 23, this means that even considering the power consumption of the additional light source for operating such devices, the overall power consumption remains significantly lower compared to those operated by large applied bias voltages.
Fig. 4b further demonstrates the facilitation index under different excitation powers, and in this case, it is seen that a higher excitation power leads to a smaller facilitation index (a lower degree of facilitation). Moreover, it is seen that whether in the voltage dependence (Fig. 4a) or the excitation power dependence (Fig. 4b), the facilitation index reduces as the pulse number increases.
The synaptic strength increase is also studied as a function of pulse numbers, i.e., 10, 20, 40, and 80, for four different pulse trains. Fig. 4c shows the corresponding photocurrent under the same experimental conditions. The grey line is a fitting curve using the Hill function:
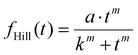 | (1) |
Finally, synaptic strength decay versus pulse number is studied. In this case, we consider the synaptic strength increase after the end of each pulse train and examine the decay time when the photocurrent is reduced to ∼37% (1/e, a natural decay is considered) of the value before the pulse train ends. For example, when the pulse number is 10, we examine the time at I1/e when I1/e/I10 ∼ 37%, as illustrated in Fig. 2d. The moment when the pulse train ends is set as 0 s. It is seen that, with a larger pulse number, the decay time increases, and close to 2 s is obtained with a pulse number of 80. This mimics the learning process in typical biological neuron systems in the sense that with more rehearsals, the plasticity lasts longer.
Analogy to artificial retina
The top panel of Fig. 5a illustrates the working principle of a human retina: briefly, photoreceptors such as cones and rods convert the optical signal into an electric signal, which forms synapses on bipolar cells. Bipolar cells can be either excitatory or inhibitory, and form synapses on ganglion cells, which fire action potentials to the optic nerve when the threshold is reached. This allows further optical information processing in the primary visual cortex or other regions of the brain for high-level processing, such as object distinction, scene segmentation, and so on.24 Neurons in the retina can also pre-process certain visual information, such as enhancing the image contrast and the signal-to-noise ratio.25–28The presented neuron-like nanowire photodetectors can mimic, to some extent, the photoreceptors – bipolar cells – ganglion cells ensemble. As shown in Fig. 3f, the presence of synaptic plasticity could emulate the plasticity of bipolar and ganglion cells, and the action potential firing in Fig. 3f could emulate the firing of ganglion cells to the optic nerves. As such, the presented neuron-like nanowire photodetectors could be readily used to build an artificial retina. Fig. 5b shows an overview of using the presented neuron-like nanowire photodetectors to construct a conceptual artificial retina. Briefly, artificial red (R), green (G), and blue (B) cells (can be more cells) detect light and pre-process light information. By further introducing optics and electronics and combining with external convolutional neural networks (CNNs) of various specific functions, complex visual information can be processed.
Discussion and outlook
As the size and the density of the nanowires can be controlled using selective area epitaxy,29–31 the presented neuron-like nanowire photodetectors can have a preset synaptic strength, allowing them to mimic the specific colour sensitivity that different species have developed through evolutionary processes. Using selective area epitaxy further allows us to design artificial sensors with multiple focus zones, mimicking multiple foveae in birds.32–34 In addition, leveraging the widely tunable bandgap of III-nitrides, which ranges from near-infrared to deep ultraviolet,35–37 the optical response of such an artificial retina can be extended beyond RGB.Methods
Nanowire epitaxial growth
The light-detecting nanowires were grown on n-type Si (111) substrates using plasma-assisted molecular beam epitaxy (MBE). The growth began with a thin n-GaN nanowire layer to mitigate the electrical mismatch between n-Si and p-GaN. This was followed by the sequential growth of p-type GaN, n-type GaN, n-type AlGaN, and p-type InGaN segments in the format of nanowires. Magnesium (Mg) and silicon (Si) were p-type and n-type dopants, respectively. For n-type doping, the Si cell temperature ranged from 1100 °C to 1200 °C, while for p-type doping, the Mg cell temperature was between 280 °C and 300 °C. These conditions correspond to doping concentrations of approximately 1018–1019 cm−3.17,38–43 The GaN, AlGaN, and InGaN segments were grown at substrate temperatures of 740 °C, 830 °C, and 590 °C, respectively. For GaN, a nitrogen flow rate of 1.5 sccm and a Ga beam equivalent pressure (BEP) of approximately 7 × 10−8 Torr were used. For AlGaN, the nitrogen flow rate was 0.9 sccm, and Ga and Al BEPs were 2.4 × 10−8 Torr and 1.7 × 10−8 Torr, respectively. The InGaN layer was grown with Ga and In BEPs of 2.4 × 10−8 Torr and 2.2 × 10−8 Torr, respectively, under a nitrogen flow rate of 0.9 sccm.Device fabrication
The neuron-like nanowire photodetectors were fabricated by standard photolithography and metallization techniques. The p-contact, which defines the device size (1 mm × 1 mm in this study), was formed by depositing a Ni/Au bilayer (15/15 nm) on the top surface of the nanowires. The n-contact was established by applying colloidal silver (Ag) paste to the backside of the highly conductive n-Si substrate. Importantly, it is noted that the p-contact size can be further reduced, and in fact, a p-contact size as small as 1 µm × 1 µm is possible.44,45 Therefore, the presented nanowire photodetectors offer a viable path towards single artificial neurons, significantly different compared with using large-scale circuits to emulate neuronal dynamics. In this study, an example photoresponse from a 300 µm × 300 µm device is shown in the SI.Device characterization
The optoelectronic properties of the neuron-like nanowire photodetectors were characterized using Keithley 2400 source meter units (SMUs). The devices were placed on an Al plate, with the colloidal Ag paste applied underneath. The ground probe from the SMU was connected to the Al plate, while the voltage biasing probe contacted the metal pad of the device. Light from the sources was focused onto the devices using focus lenses, with an area of ∼1 mm2. The 405 nm blue light (laser) is continuous-wave (CW), whereas the 266 nm UV light (laser) is pulsed. Note that the meaning of pulse here is an intrinsic property of the laser, whereas the pulse mentioned in the main text refers to periodic on/off light. A calibrated Si photodiode was used to measure the incident light intensity.Electron microscopy
The SEM images were taken using a field-emission (FE) SEM. The wafers were tilted at a 45-degree angle. To study the structural properties, TEM analysis was carried out using a Tecnai Talos F200× G2 TEM. The nanowires were mechanically transferred onto a TEM grid.Author contributions
S. Z. conceived the idea and directed the project. Y. C. and M. F. performed device characterizations. Y. C. performed experiments on neuron emulation. M. V., S. Z., and M. F. performed the MBE growth of the nanostructures. S. Z. and M. V. performed the electron microscopy experiments. M. V. fabricated the devices. S. Z. wrote the manuscript, with comments from all authors.Conflicts of interest
The authors declare no competing financial interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: additional electron microscopy studies; alloy composition of the light-detecting nanowires; comparison with the reference structure and more discussions on the photocarrier dynamics; additional analysis on Hill function fitting; photoresponse of smaller devices; microscope image of the fabricated wafer. See DOI: https://doi.org/10.1039/d6nr00513f.Acknowledgements
This work is supported by Natural Sciences and Engineering Research Council of Canada (NSERC) and Fonds de Recherche du Quebec – Nature et Technologies (FRQNT).References
- A. Krizhevsky, I. Sutskever and G. E. Hinton, Imagenet classification with deep convolutional neural networks, Adv. Neural Inf. Process. Syst., 2012, 25, 1 Search PubMed.
- Y. LeCun, Y. Bengio and G. Hinton, Deep learning, Nature, 2015, 521, 436 CrossRef CAS PubMed.
- L. D. Harmon, Artificial Neuron, Science, 1958, 129, 962 CrossRef PubMed.
- A. K. Jain, J. Mao and K. M. Mohiuddin, Artificial neural networks a tutorial, IEEE Comput., 1996, 29, 31 CrossRef.
- S.-C. Wang, Interdisciplinary computing in java programming, Springer, Boston, MA, 2003 Search PubMed.
- A. Krogh, What are artificial neural networks, Nat. Biotechnol., 2008, 26, 195 CrossRef CAS PubMed.
- D. Beniaguev, I. Segev and M. London, Single cortical neurons as deep artificial neural networks, Neuron, 2021, 109, 2727 e2723 CrossRef PubMed.
- G. R. Yang and X. J. Wang, Artificial Neural Networks for Neuroscientists: A Primer, Neuron, 2020, 107, 1048 CrossRef CAS PubMed.
- W. Zhang, et al., Neuro-inspired computing chips, Nat. Electron., 2020, 3, 382 Search PubMed.
- M. Le Gallo, et al., A 64-core mixed-signal in-memory compute chip based on phase-change memory for deep neural network inference, Nat. Electron., 2023, 6, 680 CrossRef.
- D. Purves, et al., Neurosciences, De Boeck Supérieur, 2009 Search PubMed.
- T. Ohno, et al., Short-term plasticity and long-term potentiation mimicked in single inorganic synapses, Nat. Mater., 2011, 10, 591 CrossRef CAS PubMed.
- K. He, C. Wang, Y. He, J. Su and X. Chen, Artificial Neuron Devices, Chem. Rev., 2023, 123, 13796 CrossRef CAS PubMed.
- T. Wang, et al., A chemically mediated artificial neuron, Nat. Electron., 2022, 5, 586 CrossRef CAS.
- T. Sarkar, et al., An organic artificial spiking neuron for in situ neuromorphic sensing and biointerfacing, Nat. Electron., 2022, 5, 774 CrossRef.
- G. S. Syed, Y. Zhou, J. Warner and H. Bhaskaran, Atomically thin optomemristive feedback neurons, Nat. Nanotechnol., 2023, 18, 1036 CrossRef CAS PubMed.
- Y. Chen, et al., Real-Time, Dual-Physical-Layer Encryption Directly within an Optical Sensor on a Silicon Platform, ACS Appl. Mater. Interfaces, 2025, 17, 28350 CrossRef CAS PubMed.
- T. V. Bliss and T. Lømo, Long–lasting potentiation of synaptic transmission in the dentate area of the anaesthetized rabbit following stimulation of the perforant path, J. Physiol., 1973, 232, 331 CrossRef CAS PubMed.
- D. Zipser, B. Kehoe, G. Littlewort and J. Fuster, A spiking network model of short-term active memory, J. Neurosci., 1993, 13, 3406 CrossRef CAS PubMed.
- J. C. Magee, Dendritic Ih normalizes temporal summation in hippocampal CA1 neurons, Nat. Neurosci., 1999, 2, 508 CrossRef CAS PubMed.
- H. Seung, et al., Integration of synaptic phototransistors and quantum dot light-emitting diodes for visualization and recognition of UV patterns, Sci. Adv., 2022, 8, eabq3101 CrossRef CAS PubMed.
- J. Chen, et al., Optoelectronic graded neurons for bioinspired in-sensor motion perception, Nat. Nanotechnol., 2023, 18, 882 CrossRef CAS PubMed.
- Y. Narukawa, M. Ichikawa, D. Sanga, M. Sano and T. Mukai, White light emitting diodes with super-high luminous efficacy, J. Phys. D: Appl. Phys., 2010, 43, 354002 CrossRef.
- J. Tombran-Tink and C. J. Barnstable, Visual transduction and non-visual light perception, Springer Science & Business Media, 2008 Search PubMed.
- J. M. Fuster and J. P. Jervey, Inferotemporal neurons distinguish and retain behaviorally relevant features of visual stimuli, Science, 1981, 212, 952 CrossRef CAS PubMed.
- B. E. Reese, Developmental plasticity of photoreceptors, Prog. Brain Res., 2004, 144, 3 CAS.
- Z. Deng, S. Oosterboer and W. Wei, Short-term plasticity and context-dependent circuit function: Insights from retinal circuitry, Sci. Adv., 2024, 10, eadp5229 CrossRef CAS PubMed.
- S. A. Baccus and M. Meister, Fast and slow contrast adaptation in retinal circuitry, Neuron, 2002, 36, 909 CrossRef CAS PubMed.
- M. F. Vafadar and S. Zhao, Architecture for Surface-Emitting Lasers with On-Demand Lasing Wavelength by Nanowire Optical Cavities, ACS Nano, 2024, 18, 14290 CrossRef CAS PubMed.
- M. F. Vafadar and S. Zhao, Low-Temperature Selective Area Epitaxy of GaN Nanowires: Toward a Top-Surface Morphology Controllable, Fully Epitaxial Nanophotonic Platform, ACS Appl. Nano Mater., 2022, 5, 16045 CrossRef CAS.
- M. F. Vafadar and S. Zhao, Photo–Detectable Light–Emitting Devices with Epitaxial Nanowire Photonic Crystals, Phys. Status Solidi A, 2025, 23, 2500157 CrossRef.
- D. Williams, Eagle eyed or bird brained?, Eye, 2023, 37, 2426 CrossRef PubMed.
- J. González-Martín-Moro, J. L. Hernández-Verdejo and A. Clement-Corral, The visual system of diurnal raptors: Updated review, Arch. Soc. Esp. Oftalmol., 2017, 92, 225 CrossRef PubMed.
- T. Baden and D. Osorio, The Retinal Basis of Vertebrate Color Vision, Annu. Rev. Vis. Sci., 2019, 5, 177 CrossRef CAS PubMed.
- S. Zhao, H. P. T. Nguyen, M. G. Kibria and Z. Mi, III-Nitride nanowire optoelectronics, Prog. Quantum Electron., 2015, 44, 14 CrossRef.
- S. Zhao, III-Nitride nanostructures: Selective area epitaxy, surface emitting lasers, and emerging photodetectors, Semicond. Semimetals, 2024, 116, 1 CAS.
- S. Nakamura, M. Senoh, N. Iwasa and S.-i. Nagahama, High-Brightness InGaN Blue, Green and Yellow Light-Emitting Diodes with Quantum Well Structures, Jpn. J. Appl. Phys., 1995, 34, L797 CrossRef CAS.
- M. Fathabadi, Y. Yin, S. Li and S. Zhao, Breaking the Built–In Electric Field Barrier in p–n Heterojunction for Self–Powered, Wavelength Distinguishable Photoelectrochemical Photodetectors: Toward Low Power Consumption and Secure Underwater Wireless Sensor Network, Adv. Opt. Mater., 2023, 12, 2302372 CrossRef.
- X. Hai, R. T. Rashid, S. M. Sadaf, Z. Mi and S. Zhao, Effect of low hole mobility on the efficiency droop of AlGaN nanowire deep ultraviolet light emitting diodes, Appl. Phys. Lett., 2019, 114, 101104 CrossRef.
- K. H. Li, X. Liu, Q. Wang, S. Zhao and Z. Mi, Ultralow-threshold electrically injected AlGaN nanowire ultraviolet lasers on Si operating at low temperature, Nat. Nanotechnol., 2015, 10, 140 CrossRef CAS PubMed.
- M. G. Kibria, et al., Tuning the surface Fermi level on p-type gallium nitride nanowires for efficient overall water splitting, Nat. Commun., 2014, 5, 3825 CrossRef CAS PubMed.
- S. Zhao, et al., p-Type InN Nanowires, Nano Lett., 2013, 13, 5509 CrossRef CAS PubMed.
- S. Zhao, Z. Mi, M. G. Kibria, Q. Li and G. T. Wang, Understanding the role of Si doping on surface charge and optical properties: Photoluminescence study of intrinsic and Si-doped InN nanowires, Phys. Rev. B: Condens. Matter Mater. Phys., 2012, 85, 245313 CrossRef.
- V. Veeramuthu, et al., Scalable InGaN nanowire micro-LEDs: paving the way for next-generation display technology, Natl. Sci. Rev., 2025, 12, nwae306 CrossRef CAS PubMed.
- A. Pandey, et al., Toward High Wall-Plug Efficiency in Nanowire-Based Red Micro-LEDs, ACS Photonics, 2025, 12, 4988 CrossRef CAS.
Footnote |
| † Y. C. and M. F. contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |




