 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Synthesis and characterization of platinum-decorated iron carbide nanoparticles and their potential for magnetically induced catalysis
Sheng-Hsiang
Lin
 ab,
Oscar
Suárez-Riaño†
ab,
Oscar
Suárez-Riaño†
 c,
Víctor
Varela-Izquierdo
c,
Víctor
Varela-Izquierdo
 c,
Farzan
Shabani
c,
Pier-Francesco
Fazzini
c,
Farzan
Shabani
c,
Pier-Francesco
Fazzini
 c,
Edwin A.
Baquero
c,
Edwin A.
Baquero
 d,
Simon
Tricard
d,
Simon
Tricard
 c,
Bruno
Chaudret
c,
Bruno
Chaudret
 *c,
Walter
Leitner
*c,
Walter
Leitner
 ab and
Alexis
Bordet
ab and
Alexis
Bordet
 *a
*a
aMax Planck Institute for Chemical Energy Conversion, Stiftstraße 34-36, 45470, Mülheim an der Ruhr, Germany. E-mail: alexis.bordet@cec.mpg.de
bInstitute of Technical and Macromolecular Chemistry, RWTH Aachen University, 52074, Aachen, Germany
cLaboratoire de Physique et Chimie des Nano-Objets, Université de Toulouse, LPCNO, INSA, UPS, CNRS-UMR5215, 135 Avenue de Rangueil, 31077, Toulouse, France
dEstado Sólido y Catálisis Ambiental (ESCA), Departamento de Química, Facultad de Ciencias, Universidad Nacional de Colombia, Carrera 30 No. 45-03, 111321, Bogotá, Colombia
First published on 17th February 2026
Abstract
A mild organometallic synthetic approach to platinum-decorated iron carbide nanoparticles (Pt@ICNPs) is reported. The resulting ferromagnetic Pt@ICNPs can be activated upon exposure to alternating current magnetic fields (ACMFs, magnetic induction), with a specific absorption rate of 625 W g−1 at 47 mT, 100 kHz. Pt@ICNPs are a versatile multifunctional platform with significant potential for applications in biomedicine and magnetically induced catalysis. The latter is demonstrated herein via the magnetically induced hydrogenation of various functionalities under mild conditions.
Platinum-containing nanoparticles (NPs) are important materials in electronic, energy storage, automotive, biomedicine, and chemical applications owing to their excellent electric, magnetic, and catalytic properties.1 In particular, Pt NPs catalysts are pivotal to traditional automotive exhaust gases treatment,2 petroleum refining,3 electrocatalytic water splitting,4 fuel cells,5 and hydrogenation/dehydrogenation for liquid organic hydrogen carrier (LOHC) applications.6 Pt is a rare and precious metal, prompting extensive efforts to improve the activity and durability of Pt NPs for more efficient catalytic utilization. A typical strategy is to immobilize Pt NPs on porous support materials to increase Pt dispersion and stability.7–10 While relatively inert metal oxides and carbons have been widely used as support materials for catalytic NPs,7,11 many opportunities arise once the support becomes functional,12 and even stimuli-responsive.13 In this context, core–shell NPs can combine multiple functions that do not exist in single-component compounds,14 and there is growing interest in multifunctional NPs that respond to stimuli such as heat, light, electric, and magnetic fields, with the aim to enhance catalytic performance and decrease environmental impact.15–18
In particular, magnetically-responsive NPs coated or decorated with Pt are attractive in catalysis as they combine for instance efficient Pt utilization,7 magnetic separation,19 and potential magnetic heating capabilities upon exposure to alternating current magnetic fields (ACMFs).18–20 In the past decades, various synthetic approaches have been reported for the preparation of Pt-decorated magnetic NPs (Pt@MagNPs), mainly using iron oxide NPs (IONPs) as the magnetic platform.21 However, the moderate magnetic properties of IONPs lead to limited heat generation under magnetic induction, thereby limiting the potential of Pt@IONPs for magnetically induced catalysis. In addition, Pt deposition is typically achieved using high temperatures and non-sustainable reducing agents (e.g., hydrazine and NaBH4).22–24 Thus, mild synthetic approaches to Pt@MagNPs with excellent magnetic properties are desirable, yet underdeveloped.
In this context, ferromagnetic iron carbide nanoparticles (ICNPs) possess the highest specific absorption rate (SAR) reported to date, and were showed to be excellent heating agents25,26 and active species27 in magnetically induced catalysis.18,25–31 Organometallic synthetic routes were recently reported for the decoration of ICNPs with Ru or Cu to produce multifunctional Ru@ICNPs30 and Cu@ICNPs31 catalysts with great potential for magnetically induced hydrogenation and hydrodeoxygenation reactions.
Building on this approach, we explore herein the possibility to decorate ICNPs with Pt via a mild organometallic approach. The target Pt@ICNPs material must combine ICNPs’ magnetic separation and heating capabilities with the excellent catalytic properties of Pt. This multifunctionality will be probed through the application of Pt@ICNPs to magnetically induced hydrogenation reactions of platform molecules under mild H2 pressures (Fig. 1).
Pt-decorated ICNPs were synthesized through an organometallic approach (see SI Section S2). First, ICNPs (Fe2.2C@Fe5C2 core@shell structure, ca. 15 nm) were prepared by carbidization of preformed Fe(0) NPs under syngas, following a reported procedure.25 Then, a THF solution of trans-bis(benzonitrile)dichloroplatinum(II) (trans-PtCl2(NCPh)2), was added to a colloidal dispersion of ICNPs in THF, and the resulting mixture was pressurized with hydrogen (3 bar) at 30 °C for 16 h (Fig. 2). The organometallic precursor and reaction conditions were tuned to promote the controlled decomposition of the platinum complex at the ICNPs’ surface, while suppressing side-nucleation of undesired free Pt NPs in solution (Fig. S1). Under suitable conditions (0.25 equivalent of Pt compared to Fe), homogeneous nucleation could be avoided, and Pt@ICNPs were obtained as a black magnetic powder (70% Fe, 4% Pt, and 26% ligands determined by scanning electron microscopy with energy-dispersive X-ray spectroscopy (SEM-EDX) and thermogravimetric analysis (TGA)).
Characterization of newly prepared Pt@ICNPs by transmission electron microscopy (TEM) showed small NPs (ca. 1.7 ± 0.3 nm) decorating the surface of larger core–shell structures typical of ICNPs (Fig. 2a). Scanning transmission electron microscopy in high-angle annular dark field mode (STEM-HAADF), along with elemental mapping by energy-dispersive X-ray spectroscopy (EDX), demonstrated the successful decoration of ICNPs with small Pt NPs (Fig. 2b–d). Powder X-ray diffraction (XRD) further evidenced the presence of both Fe2.2C and Pt(0) crystalline phases in the newly prepared material (Fig. 2e). X-ray photoelectron spectroscopy (XPS) analysis of the Fe 2p region of Pt@ICNPs showed the expected iron carbide peaks (2p1/2 at 720.5 eV and 2p3/2 at 707.2 eV),31,32 but also revealed the presence of FeClx species (710.5 eV and 724.0 eV) presumably located at the nanoparticle surface (Fig. 2f and Fig. S2).33 Analysis of the Pt 4f spectrum indicated platinum NPs in the metallic state (Fig. 2g),34 consistent with the XRD data. Zero-field 57Fe Mössbauer spectroscopy at 80 K showed the predominance of two iron carbide phases: Fe2.2C (68%) and Fe5C2 (25%), in the expected ratio for ICNPs,25 along with minor contributions from paramagnetic iron species (5%, including the FeClx species) and metallic iron (2%) (Fig. S3). Signals corresponding to oxidized iron species were not detected, in agreement with XPS and XRD analysis. These data indicate that the morphology, composition, and structure of ICNPs did not change substantially upon Pt decoration.
Magnetic characterization by vibrating sample magnetometry (VSM) at 300 K showed that ICNPs decoration with Pt resulted in a slight reduction in the coercive field (HC, from 94 mT to 66 mT) but no change in the saturation magnetization (MS, 161 A m2 kgFe–1) (Fig. 2h and Table S1). While the surface FeClx species may be responsible for the observed decrease in HC,35 the Pt@ICNPs are still ferromagnetic at room temperature. Furthermore, Pt@ICNPs demonstrated the ability to generate heat when exposed to ACMFs, with a SAR of 625 W g–1 measured at 47 mT and 100 kHz. This SAR value is lower than that of the starting ICNPs (>3000 W g–1),25 but remains higher than that of typical FexOy-based materials.36 This includes Pt@IONPs (no heating, see SI) and Pt@Fe/FeOx (109 W g–1) prepared here as reference materials (see Section S2 for detailed synthetic protocols and Fig. S4 and S5 for characterization). This result is consistent with our previous reports where deposition of non-magnetic elements was shown to reduce the overall heating efficiency of the starting MagNPs.30,31,37
The potential of the newly prepared Pt@ICNPs for application in magnetically induced catalysis was explored through their testing in the hydrogenation of a variety of functional groups. Reactions were carried out in Fisher–Porter bottles under a standard set of reaction conditions: ACMF (μ0Hmax = 65 mT, f = 300 Hz), 3 bar of H2, 16 h, with decane as the solvent (Table 1). The global temperature of the reaction under the conditions mentioned, recorded with an infrared camera, was 200 °C; however, recent reports demonstrate that local hot spots could reach temperatures exceeding 300 °C.27 Moreover, the magnetic nature of these Pt@ICNPs offers the possibility of easy recovery and separation from the reaction medium.
| # | Subs. | Prod. | Conv. (%) | Select. (%) |
|---|---|---|---|---|
| Reaction conditions: Pt@ICNPs (10.0 mg, 0.002 mmol of Pt), substrate (0.33 mmol), decane (0.5 mL), 3 bar H2, 65 mT, 300 kHz, 16 h.a Product distribution is provided in SI. Subs. = substrate. Prod. = product. Conv. = conversion. Select. = selectivity. | ||||
| 1 |
|

|
>99 | >99 |
| 2 |

|

|
>99 | >99 |
| 3 |
|

|
>99 | 31a |
| 4 |
|

|
>99 | >99 |
| 5 |
|

|
>99 | >99 |
| 6 |

|

|
23 | >99 |
| 7 |

|

|
0 | 0 |
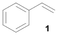
The C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond in styrene (1) and NO2 group in nitrobenzene (2) were readily hydrogenated, giving the corresponding ethylbenzene (1a) and aniline (2a) products in quantitative yields.
C bond in styrene (1) and NO2 group in nitrobenzene (2) were readily hydrogenated, giving the corresponding ethylbenzene (1a) and aniline (2a) products in quantitative yields.
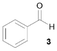
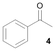
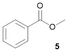
Notably, Pt@ICNPs exhibited also hydrodeoxygenation (HDO) activity, enabling the selective reduction of aromatic compounds possessing aldehyde (benzaldehyde, 3), ketone (acetophenone, 4), and ester (methylbenzoate, 5) functionalities to the corresponding methyl-substituted aromatic products. In the case of 3, the selectivity toward toluene remained modest (31%), which is due to a competitive coupling reaction yielding 1,2-diphenylethane. This result demonstrates the formation of benzyl radicals during the deoxygenation process of benzaldehyde (Scheme S1).
The stability and reusability of Pt@ICNPs were investigated through recycling experiments using the HDO of 4 as a model reaction. While the catalyst was easily separated magnetically and reused through the cycles without any change in selectivity, severe loss of activity was observed after the 3rd cycle (Table S2). This loss of catalytic activity is attributed to the aggregation/coalescence and oxidation of Pt@ICNPs, as revealed by electron microscopy (Fig. S6), powder-XRD (Fig. S7), and VSM analysis (decrease of HC (30 mT) and MS (119 A m2 kgFe–1), Table S1 and Fig. S8). While the aggregation/coalescence of M@ICNPs is consistent with previous observations using other metals (e.g. Ru@ICNPs,30 Cu@ICNPs,31etc.), NPs oxidation may have occurred due to handling during the recycling experiments/characterization.
Notably, the selective HDO of 5 to toluene is interesting as it typically requires harsh reaction conditions under conventional heterogeneous catalysis,38,39 while it is enabled under mild conditions by magnetically induced catalysis also with iron-based catalysts.27 The magnetically induced hydrogenolysis of diphenylether (6) with Pt@ICNPs also proved possible, although challenging (23% conversion, >99% selectivity toward benzene). The excellent selectivity toward benzene indicates an efficient Csp2–OH bond cleavage and no aromatic hydrogenation activity, which is remarkable compared to previously reported catalytic and magnetocatalytic systems, which yielded complex mixtures of hydrogenated and non-hydrogenated products.40–43 Notably, aromatic ring hydrogenation was suppressed in all the investigated cases, presumably due to a synergistic interaction between noble and 3d metals.44,45 The hydrogenation of 1-acetyl-3-methylpiperidine (7) proved unsuccessful with Pt@ICNPs under these standard conditions, consistent with the difficulty of amide hydrogenation with H2.46,47 Notably, we have recently shown that the magnetically induced hydrogenation of amides can be enabled under mild conditions using a different catalyst design consisting in the immobilization of ICNPs as heating agents on a supported Pt catalyst.48
Finally, reference Pt-decorated iron oxide nanoparticles (Pt@IONPs) prepared following an identical approach were found inactive for any reaction described here. The HDO of 4 could be catalysed by Pt@Fe/FeOx (>99% ethylbenzene yield) but not the hydrogenolysis of 6 (Table S2). Pt@IONPs and Pt@Fe/FeOx possess lower SAR than Pt@ICNPs, which results in insufficient thermal activation of the surface Pt for this challenging transformation. This comparison demonstrates the importance of selecting ICNPs with excellent magnetic properties to enable challenging transformation via magnetically induced catalysis.
Conclusions
In summary, we open a mild organometallic approach for the preparation of Pt-decorated iron carbide nanoparticles. Characterization of the novel Pt@ICNPs show that the morphology, composition, and structure of ICNPs do not change substantially upon Pt loading. Pt@ICNPs are ferromagnetic, and capable of heating efficiently upon exposure to ACMFs. Their potential for application in magnetically induced catalysis is demonstrated through the selective hydrogenation and hydrodeoxygenation of various functionalities under mild conditions. The importance of selecting ferromagnetic ICNPs as the magnetically-responsive core is outlined by the inactivity of reference Pt@IONPs under identical conditions. This work highlights the promise of magnetic NPs decoration as a versatile strategy for the design of novel multifunctional nanocatalysts.Author contributions
S.-H. L. participated to the formulation of the original idea, project definition and conceptual planning of the experimental workflow, performed experimental work and data interpretation and wrote the manuscript draft. O. S. R., V. V-I., and F. S. performed experimental work and edited the manuscript. P.-F. F characterized the materials by electron microscopy. E. A. B., S. T., B. C., and W. L. participated to the data interpretation and editing of the manuscript. A. B. participated to the formulation of the original idea, project definition and conceptual planning of the experimental workflow, supervised the work, contributed to data interpretation and writing of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5nr04609b.Acknowledgements
The authors acknowledge financial support by the Max Planck Society and by the Deutsche Forschungsgemeinschaft (DFG) under Germany's Excellence Strategy – Exzellenzcluster 2186 “The Fuel Science Center” ID: 390919832. A. B. acknowledges the Gordon and Betty Moore Foundation for financial support (GBMF ID #13905). Furthermore, the authors would like to thank Derya Demirbas (MPI-Kohlenforschung) for access and assistance with 57Fe Mössbauer experiments, Dr. Lukas Pielsticker, Gudrun Klihm (MPI-CEC) for assistance with XPS experiments, Annika Gurowski, Alina Jakubowski, and Justus Werkmeister (MPI-CEC) for their support with GC and GC-MS measurements. S.-H. L. acknowledges NanoX for the mobility funding support. V. V. I. and B. C. are grateful to the French State aid managed by the Agence Nationale de la Recherche under France 2030 plan, bearing the reference code ANR-22-PESP-0010: Projet ciblé “POWERCO2” within the PEPR project SPLEEN. Open access funding enabled and organized by Projekt DEAL. Open Access funding provided by the Max Planck Society.References
- A. Chen and P. Holt-Hindle, Chem. Rev., 2010, 110, 3767–3804 CrossRef CAS PubMed.
- B. K. Cho, Ind. Eng. Chem. Res., 1988, 27, 30–36 CrossRef CAS.
- M. H. Pinzón, A. Centeno and S. A. Giraldo, Appl. Catal., A, 2006, 302, 118–126 Search PubMed.
- K. L. Zhou, Z. Wang, C. B. Han, X. Ke, C. Wang, Y. Jin, Q. Zhang, J. Liu, H. Wang and H. Yan, Nat. Commun., 2021, 12, 3783 Search PubMed.
- T.-W. Song, C. Xu, Z.-T. Sheng, H.-K. Yan, L. Tong, J. Liu, W.-J. Zeng, L.-J. Zuo, P. Yin, M. Zuo, S.-Q. Chu, P. Chen and H.-W. Liang, Nat. Commun., 2022, 13, 6521 Search PubMed.
- Y. Wu, Y. Li, X. Yu, X. Ma, M. Boebinger, J. Webera and Z. Wu, Catal. Sci. Technol., 2024, 14, 1791–1801 RSC.
- D. Leybo, U. J. Etim, M. Monai, S. R. Bare, Z. Zhong and C. Vogt, Chem. Soc. Rev., 2024, 53, 10450–10490 RSC.
- L. Foppa, J. Dupont and C. W. Scheeren, RSC Adv., 2014, 4, 16583–16588 RSC.
- X. Zhang, N. Hao, X. Dong, S. Chen, Z. Zhou, Y. Zhang and K. Wang, RSC Adv., 2016, 6, 69973–69976 Search PubMed.
- L. Liu, Y. Wang, Y. Zhao, Y. Wang, Z. Zhang, T. Wu, W. Qin, S. Liu, B. Jia, H. Wu, D. Zhang, X. Qu, M. Chhowalla and M. Qin, Adv. Funct. Mater., 2022, 32, 2112207 Search PubMed.
- H. Wang, Z. Gao, B. Sun, S. Mu, F. Dang, X. Guo, D. Ma and C. Shi, Chem. Catal., 2023, 3, 100768 Search PubMed.
- P. Verma, Y. Kuwahara, K. Mori, R. Raja and H. Yamashita, Nanoscale, 2020, 12, 11333–11363 Search PubMed.
- P. Huang, R. Baldenhofer, R. P. Martinho, L. Lefferts and J. A. F. Albanese, ACS Catal., 2023, 13, 6590–6602 CrossRef CAS.
- Y. Zou, Z. Sun, Q. Wang, Y. Ju, N. Sun, Q. Yue, Y. Deng, S. Liu, S. Yang, Z. Wang, F. Li, Y. Hou, C. Deng, D. Ling and Y. Deng, Chem. Rev., 2025, 125, 972–1048 CrossRef CAS PubMed.
- A. P. Blum, J. K. Kammeyer, A. M. Rush, C. E. Callmann, M. E. Hahn and N. C. Gianneschi, J. Am. Chem. Soc., 2015, 137, 2140–2154 CrossRef CAS PubMed.
- A. Bordet and W. Leitner, Angew. Chem., Int. Ed., 2023, 62, e202301956 CrossRef CAS PubMed.
- I. Salahshoori, A. Yazdanbakhsh, M. N. Jorabchi, F. Z. Kazemabadi, H. A. Khonakdar and A. H. Mohammadi, Adv. Colloid Interface Sci., 2024, 333, 103304 CrossRef CAS PubMed.
- A. Bordet, W. Leitner and B. Chaudret, Angew. Chem., Int. Ed., 2025, 64, e202424151 CrossRef CAS PubMed.
- L. Truong-Phuoc, C. Duong-Viet, J.-M. Nhut, A. Pappa, S. Zafeiratos and C. Pham-Huu, ChemSusChem, 2025, 18, e202402335 Search PubMed.
- J.-S. Pavelić, S. Gyergyek, B. Likozar and M. Grilc, Chem. Eng. J., 2025, 505, 158928 CrossRef.
- V. Polshettiwar, R. Luque, A. Fihri, H. Zhu, M. Bouhrara and J.-M. Basset, Chem. Rev., 2011, 111, 3036–3075 CrossRef CAS PubMed.
- R. Abu-Reziq, D. Wang, M. Post and H. Alper, Adv. Synth. Catal., 2007, 349, 2145–2150 Search PubMed.
- M. Xie, F. Zhang, Y. Longa and J. Ma, RSC Adv., 2013, 3, 10329–10334 Search PubMed.
- J. Adamski, M. I. Qadir, J. P. Serna, F. Bernardi, D. L. Baptista, B. R. Salles, M. A. Novak, G. Machado and J. Dupont, J. Phys. Chem. C, 2018, 122, 4641–4650 Search PubMed.
- A. Bordet, L.-M. Lacroix, P.-F. Fazzini, J. Carrey, K. Soulantica and B. Chaudret, Angew. Chem., Int. Ed., 2016, 55, 15894 CrossRef CAS PubMed.
- H. Kreissl, J. Jin, S.-H. Lin, D. Schüette, S. Störtte, N. Levin, B. Chaudret, A. J. Vorholt, A. Bordet and W. Leitner, Angew. Chem., Int. Ed., 2021, 60, 26639 Search PubMed.
- S. Ahmedi, L.-M. Lacroix, D. Demirbas, D. J. SantaLucia, C. Weidenthaler, W. Hetaba, W. Leitner and A. Bordet, J. Am. Chem. Soc., 2025, 147, 34758–34766 CrossRef CAS PubMed.
- J. Mazarío, S. Ghosh, V. Varela-Izquierdo, L. M. Martínez-Prieto and B. Chaudret, ChemCatChem, 2025, 17, e202400683 CrossRef.
- C. Hu, Y. Dong, Q. Shi, R. Long and Y. Xiong, Chem. Soc. Rev., 2025, 54, 524–559 Search PubMed.
- J. M. Asensio, A. B. Miguel, P.-F. Fazzini, P. W. N. M. van Leeuwen and B. Chaudret, Angew. Chem., Int. Ed., 2019, 58, 11306 CrossRef CAS PubMed.
- S.-H. Lin, W. Hetaba, B. Chaudret, W. Leitner and A. Bordet, Adv. Energy Mater., 2022, 12, 2201783 Search PubMed.
- C. Yang, H. Zhao, Y. Hou and D. Ma, J. Am. Chem. Soc., 2012, 134, 15814–15821 Search PubMed.
- Y. J. Kim and C. R. Park, Inorg. Chem., 2002, 41, 6211–6216 Search PubMed.
- E. I. Vovk, A. V. Kalinkin, M. Yu. Smirnov, I. O. Klembovskii and V. I. Bukhtiyarov, J. Phys. Chem. C, 2017, 121, 17297–17304 Search PubMed.
- T. Yanai, D. Fukushima, R. Narabayashi, N. Ogushi, Y. Yamaguchi, A. Yamashita, M. Nakano and H. Fukunaga, AIP Adv., 2024, 14, 025020 CrossRef CAS.
- R. Ghazi, T. K. Ibrahim, J. A. Nasir, S. Gai, G. Ali, I. Boukhris and Z. Rehman, RSC Adv., 2025, 15, 11587–11616 RSC.
- V. Varela-Izquierdo, I. Mustieles-Marín, P.-F. Fazzini, G. Mencía, S. Guelen, R. Rachet and B. Chaudret, Angew. Chem., Int. Ed., 2024, 63, e202412421 CrossRef CAS PubMed.
- K. Sakoda, S. Yamaguchi, T. Mitsudome and T. Mizugaki, JACS Au, 2022, 2, 665–672 CrossRef CAS PubMed.
- J. Pritchard, G. A. Filonenko, R. van Putten, E. J. M. Hensen and E. A. Pidko, Chem. Soc. Rev., 2015, 44, 3808–3833 RSC.
- S. Ghosh, T. Ourlin, J. Mazarío, S. Cayez, S. Daccache, J. Carrey and B. Chaudret, Chem. Mater., 2023, 18, 7542–7553 CrossRef.
- J. Mazarío, I. M. Marin, G. Mencia, C. W. Lopes, V. Varela-Izquierdo, G. Agostini, P.-F. Fazzini, N. Ratel-Ramond and B. Chaudret, ACS Appl. Nano Mater., 2024, 8, 9412–9427 CrossRef.
- M. Guo, J. Peng, Q. Yang and C. Li, ACS Catal., 2018, 8, 11174–11183 CrossRef CAS.
- M. Wang, Y. Zhao, D. Mei, R. M. Bullock, O. Y. Gutiérrez, D. M. Camaioni and J. A. Lercher, Angew. Chem., Int. Ed., 2020, 59, 1445 Search PubMed.
- A. Bordet and W. Leitner, Acc. Chem. Res., 2021, 54, 2144–2157 CrossRef CAS PubMed.
- N. Marchenko, L.-M. Lacroix, N. Ratel-Ramond, W. Leitner, A. Bordet and S. Tricard, ACS Appl. Nano Mater., 2023, 6, 20231–20239 CrossRef CAS.
- M. C. Bryan, P. J. Dunn, D. Entwistle, F. Gallou, S. G. Koenig, J. D. Hayler, M. R. Hickey, S. Hughes, M. E. Kopach, G. Moine, P. Richardson, F. Roschangar, A. Steven and F. J. Weiberth, Green Chem., 2018, 20, 5082–5103 Search PubMed.
- J. R. Cabrero-Antonino, R. Adam, V. Papa and M. Beller, Nat. Commun., 2020, 11, 3893 CrossRef CAS PubMed.
- S.-H. Lin, S. Ahmedi, A. Kretschmer, C. Campalani, Y. Kayser, L. Kang, S. DeBeer, W. Leitner and A. Bordet, Nat. Commun., 2025, 16, 3464 CrossRef CAS PubMed.
Footnote |
| † This work is dedicated to the memory of Oscar Suarez-Riaño, a scientist and friend whose passion and positive spirit greatly influenced our group. |
| This journal is © The Royal Society of Chemistry 2026 |


