Open Access Article
Open Access ArticleThermal conductivity at solid/fluid interfaces: from adsorption to phonon scattering through the rattle effect
Nikolas Ferreira de
Souza
ab,
Cecilia
Herrero
 c,
Thomaz Rossetti
Ghizoni
a,
Luís Fernando
Mercier Franco
c,
Thomaz Rossetti
Ghizoni
a,
Luís Fernando
Mercier Franco
 *a and
Benoit
Coasne
*a and
Benoit
Coasne
 *bc
*bc
aUniversidade Estadual de Campinas (UNICAMP), Faculdade de Engenharia Química, 13083-852 Campinas, Brazil. E-mail: lmfranco@unicamp.br
bUniv. Grenoble Alpes, CNRS, LIPhy, F-38000 Grenoble, France. E-mail: benoit.coasne@univ-grenoble-alpes.fr
cInstitut Laue Langevin, F-38042 Grenoble, France
First published on 9th March 2026
Abstract
Of relevance to adsorption, separation and catalysis, fluids in nanoporous materials display intriguing phenomena arising from their underlying structural, dynamical, and thermodynamic behavior. While many effects at the solid/fluid interface in nanoporous materials are well-documented, the thermal behavior of such solids subjected to fluid adsorption remains to be deciphered. Among exotic mechanisms, the so-called rattle effect, which corresponds to the decrease in the solid thermal conductivity induced by phonon scattering at the solid/fluid interface, has received only little attention. This phenomenon, which challenges existing mixing rules and effective medium approaches, has been identified in nanoporous materials (e.g. zeolite). Considering that this nanoscale effect is necessarily restricted to a small region near the solid/fluid interface, its impact for less finely divided materials (i.e. with thicker solid/fluid domains) remains to be established. Here, we address this question by employing a molecular simulation strategy to investigate thermal transport in a prototypical nanoporous silica material filled with a simple fluid. As a result, while the conventional behavior predicting an overall thermal conductivity increase upon fluid addition is qualitatively recovered in many cases, we also observe some situations where the overall thermal conductivity κ in such hybrid systems is mostly unchanged as conductivity through fluid is counterbalanced by the rattle effect at the solid surface. Understanding such intrinsic relations and the rattle effect that govern the thermal conductivity in solid/fluid systems paves the way for the design of novel materials to harness thermal processes in practical applications.
1 Introduction
With pressing challenges in environmental engineering (e.g. agriculture, water treatment) and energy applications (e.g. storage, conversion), the development of novel separation and catalysis processes involving nanoporous materials is urgently needed.1,2 In this context, the large spectrum of available nanoporous solids is expected to be a game-changer by contributing for instance to the “seven key chemical separations to change the world”.3 In such processes, the large surface area and strong confinement within the porosity of the host nanoporous material are used to harness the broad range of phenomena occurring when the solid is set in contact with a fluid.4–7 From a more fundamental viewpoint, while the transport of fluids in nanoporous materials has been reasonably clarified in the last decades, many microscopic mechanisms that rule mass and energy transfers at the nanoscale in such systems remain to be explored. In particular, most of these transport processes are often assumed to obey effective medium theories that do not properly account for the microscopic behavior of the nanoconfined fluid and nanoporous material.8–10 Specifically, with these strategies, the properties of the nanoconfined fluid are assumed to remain identical to their bulk counterpart despite well-documented reports on interfacial effects that can affect thermal and mass transport mechanisms (phonon scattering,11 heat diffusion restriction,12 pressure-like effect,13etc.).Among the challenges associated with fluid confinement in nanoporous materials, modeling thermal transport introduces complex features such as the rattle effect – a reduction in the system's thermal conductivity caused by the presence of fluid atoms at the solid surface. This phenomenon has been observed in a wide range of materials such as skutterudites,14 carbon nanotubes,15 methane hydrates,16 metal organic frameworks (MOFs)17 and zeolites.11 Formally, the rattle effect, which can amount in some cases to a 40% decrease in the solid's thermal conductivity,11 arises from phonon scattering at the solid/fluid interface as fluid atoms collide with the atomic structure of the host nanoporous material.11,17 The effect is also observed in a experimental study with HKUST-1, which further show that liquid adsorbates can reduce thermal conductivity by up to 80%, an effect attributed to enhanced phonon scattering and not captured by effective medium approximations.18 This phenomenon is necessarily omitted from mixing rules and, in general, from effective medium theories as both the fluid and solid contributions are usually assumed to be equivalent to their bulk/fluid-free counterpart (i.e. with properties as assessed in the absence of the other phase).
The above question is even more puzzling as the available literature on the topic provides somewhat contrasting pictures regarding the rattle effect.19,20 While phonon scattering consistently appears to reduce thermal transport at the molecular level, the resulting transport properties of the fluid + solid system depend on specific features of the solid phase – such as its surface area and wall thickness – but also the solid/fluid interaction strength. This complexity makes predictions of the overall thermal conductivity through effective medium theories particularly challenging. In this context, unraveling the thermal coupling between the host solid and the confined fluid constitutes a key challenge to design novel materials and processes with tailored thermal management properties. In particular, the occurrence of rattle effects opens perspectives to modulate thermal transport at the nanoscale by considering fluids adsorbed in nanoporous materials.
Here, to tackle some of the questions reported above, we investigate thermal transport in a nanoporous system using a simple yet realistic model consisting of a silica nanopore filled with a fluid. Overall, our molecular simulations show that the rattle effect remains present for all solid/fluid interaction strengths ε due to phonon scattering at the solid/fluid interface. Depending on ε and the relative contributions of the fluid and solid phases, the overall thermal conductivity κ in such hybrid (solid + fluid) systems can be increased or unchanged with respect to that obtained for the empty solid. While the first situation (thermal conductivity increase) corresponds to the conventional result where adding a fluid into the solid porosity enhances the overall thermal conductivity, the other situation cannot be predicted using classical mixing rules. By disentangling the thermal conductivity contributions from the solid phase, the fluid phase and the solid/fluid interface, we identify the role played by the solid/fluid interaction strength ε and the adsorbed amount per unit of surface area na. We also provide a simple model which allows rationalizing the decrease in the solid's thermal conductivity by considering the average phonon lifetime resulting from intrinsic phonon scattering within the solid and from solid/fluid collisions at their interface.
2 Theoretical background and formalism
2.1 Thermal conductivity and microscopic energy flux
Through the prism of statistical mechanics, the thermal conductivity κ can be evaluated using the Green–Kubo formalism by computing the time autocorrelation function of the total heat current j(t) within molecular dynamics simulations. Due to the anisotropic nature of the slit pore geometry, the Green–Kubo formulation is applied to compute the thermal conductivity κ‖ in the parallel direction to the surface using the following formula: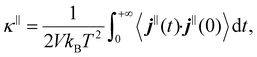 | (1) |
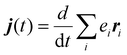 . By explicitly deriving the heat current with respect to time, we arrive at the following relation:
. By explicitly deriving the heat current with respect to time, we arrive at the following relation: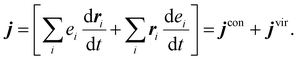 | (2) |
The first term, which corresponds to the product of the atom's energy by its velocity, is defined as the convective contribution to the total current. The second term is referred to as the virial contribution as it arises from the variation in time of the total energy of the atom (it is therefore related to the transfer of potential energy between atoms in the system).
2.2 Heat current definition
Care must be taken with multi-component systems to ensure consistency with macroscopic transport theory. Formally, the derivation of the entropy production rate leads to a coupling between the different fluxes and their underlying driving forces. The driving force for mass flux appears as the gradient of the chemical potential divided by temperature, ∇(μ/T).22 To recover the thermal conductivity as the coefficient relating the heat current j solely to the temperature gradient ∇T, it is necessary to remove the temperature dependence from the diffusive contribution. This is achieved by redefining the heat current j to exclude the energy carried by mass transport: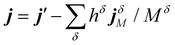 where δ corresponds to the different species within the system. In this expression, j′ is the total energy current given by eqn (2), hδ the partial molar enthalpy of species δ, jδM the mass current of component δ, and Mδ the molar mass of component δ.22,23 The latter expression for the microscopic heat current can be expressed as:
where δ corresponds to the different species within the system. In this expression, j′ is the total energy current given by eqn (2), hδ the partial molar enthalpy of species δ, jδM the mass current of component δ, and Mδ the molar mass of component δ.22,23 The latter expression for the microscopic heat current can be expressed as: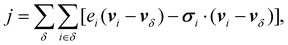 | (3) |
We recognize in eqn (3) the convective and virial contributions to the heat current (jcon and jvir) for a multi-component system. The above expression can be recast as a per-atom representation. The convective term can be given as the product of this relative velocity and the total energy of each atom while the virial term can be written as a sum of atomic stress σi distribution based on their relative positions to the geometric center 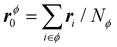 of the Nϕ interacting atoms within a given potential energy function (which may be a bond, angle, dihedral, etc.):24
of the Nϕ interacting atoms within a given potential energy function (which may be a bond, angle, dihedral, etc.):24
| jconi,δ = ei(vi − vδ), | (4a) |
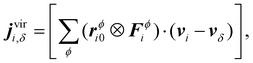 | (4b) |
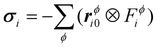 .
.
3 Computational methods
3.1 Nanoporous silica and fluid models
The slit nanopore considered in this work is built from a realistic model of amorphous silica material. The procedure employed to prepare the silica walls was described in detail elsewhere.25–27 In short, a block of crystalline silica (cristobalite) is initially melted at T = 4000 K using a molecular dynamics simulation run. The obtained system is then rapidly quenched to T = 300 K to obtain an amorphous silica block. The resulting simulation box, initially elongated along the z direction, was then cut to generate two free surfaces. The slab thickness was intentionally chosen to be Lz = 35.8 Å to match the in-plane dimensions Lx = Ly = 35.8 Å. Periodic boundary conditions were applied in the x and y directions. To ensure that the surface is realistic from a chemical viewpoint, all uncoordinated Si atoms and any O atoms not bonded to at least one Si atom are first removed and the remaining dangling bonds of the O atoms at the silica surface are saturated by adding H atoms to form silanol groups. After surface saturation, the final silica slab contained 2601 solid atoms, comprising 779 silicon, 1646 oxygen, and 176 hydrogen atoms.The number of OH bonds is adjusted to obtain a final silanol density of 6.9 OH per nm2 (this surface density is estimated by taking the geometrical surface area of the silica surface) – a value which is consistent with typical experimental conditions.28 The dimensions of the surface are Lx = 35.8 Å × Ly = 35.8 Å. The directions parallel to this surface are treated as infinite by applying periodic boundary conditions in the x and y directions. As the realistic silica surface used in this work does not exhibit a strictly identical molecular structure in the directions parallel to the surface, we looked at possible anisotropy along the x and y directions. In practice, no significant differences in thermal properties were observed when considering these two directions. Therefore, the thermal conductivity was only evaluated in the directions parallel to the silica surface. In this direction, finite-size effects associated with phonon/phonon interactions are negligible. This is supported by the fact that both the thermal conductivity and vibrational density of states for bulk amorphous silica and the silica slab from which it is extracted match (see Results section discussion).
Silica is chosen here as a prototypical surface as a large body of available materials with large specific surface area are made up of oxides. The silica slab thickness of e = 35.8 Å is chosen large enough to ensure that fluid atoms on opposite sides of the silica surface do not interact. The slit nanopore is cut so that the pore width (which is defined between the two opposite surfaces) is about H = 10 nm. The interactions between atoms of the amorphous silica were described using the force field by Carré et al., which was parameterized through a fitting scheme based on Car-Parrinello molecular dynamics simulations.29 The united atom model TraPPE was used to describe the molecular interaction between the fluid atoms (with parameters chosen to describe the whole methane molecule).30 A Lennard–Jones (LJ) potential was also used to describe the interactions between methane molecules and the Si and O atoms of the silica surface. The LJ parameters for Si and O are derived from the CLAYFF force field31 and combined using the Lorentz–Berthelot combining rules.32,33 All parameters and details are provided in the SI.
3.2 Grand canonical Monte Carlo and molecular dynamics simulations
In Grand Canonical Monte Carlo (GCMC) simulations, the system has a constant volume V and is set in equilibrium with an infinite reservoir of methane molecules imposing their chemical potential μ and temperature T.34,35 Here, the adsorption isotherm is simulated at T = 300 K by varying the chemical potential μ of the reservoir. All simulations are performed with LAMMPS which combines Monte Carlo (MC) and Molecular Dynamics (MD) steps, therefore sampling the phase space in the grand canonical ensemble.36 This hybrid scheme ensures that the system evolves through classical MD at fixed temperature, while Monte Carlo insertion and deletion moves of fluid molecules are attempted at regular intervals to enforce the matching with the prescribed chemical potential. In practice, the hybrid GCMC–MD simulations were performed with an initial equilibration of 1 × 105 steps, followed by 5 × 105 production steps, during which insertion and deletion attempts were carried out at every 10 MD steps.The absolute adsorbed amount Na(P) is estimated as the ensemble average of the number of adsorbed molecules as a function of the gas reservoir pressure P. To establish the correspondence between pressure and chemical potential, a set of reference pressures is first selected and used to define the chemical potential of an ideal gas at the target temperature. These chemical potentials are then employed as inputs for two independent GCMC simulations: the first performed within the silica nanopore system and the second carried out for a corresponding bulk methane system at the same temperature and fixed volume. After equilibration, both simulations result in an averaged number of fluid molecules. The resulting average number of particles 〈N〉 in the bulk system is then used in a NVT simulation, for which the resulting real pressures were then evaluated by assessing the average system pressure.
Starting with the configurations obtained from GCMC simulations, equilibrium molecular dynamics (EMD) simulations are conducted with LAMMPS.36 An in-house C++ code coupled to LAMMPS is used to compute the heat flux vector within the system. These molecular simulations are performed with a cutoff of 14.0 Å and using the Particle–Particle Particle–Mesh (PPPM) solver for the long-range Coulomb and van der Waals forces. All the simulations were carried with an integration time step of 0.5 fs. The molecular simulations begin with a 0.5 ns equilibration run, during which the Nosé–Hoover thermostat is applied to the silica framework (while the fluid atoms are treated in the NVE ensemble). The proper thermalization of the fluid atoms through collisions with the silica surface is verified by assessing their average kinetic energy. Once thermal equilibrium is achieved, a production run is carried out in the NVE ensemble for an additional 0.5 ns (i.e., 106 timesteps). Data for the energy flux calculations are collected at evenly spaced intervals of 1 fs during the production run. To accumulate enough statistically relevant data, 200 independent simulations are performed for each configuration considered in the present study (here, configuration refers to a pore loading with a given fluid adsorbed amount Na). These molecular simulations differ in their initialization as each simulation run starts with a unique random velocity distribution for the fluid atoms at the start of the equilibration run.
4 Results
4.1 Fluid adsorption in nanoporous silica
With insulating materials such as amorphous silica, thermal transfer occurs through atomic lattice vibrations (phonons) since the electronic contribution to heat conductivity is negligible. To assess the impact of fluid adsorption on energy transfer in nanoporous materials, we employ here a realistic model of nanoporous silica. As discussed in the Methods section, the pore surface is built from a numerical model of amorphous silica as obtained using a cook-and-quench procedure. Fig. 1(a) provides a magnified view of the final silica structure obtained using this procedure. Fig. 1(b) also shows a SiO4 tetrahedron taken from the core of the silica slab and a SiO3 (OH) silanol group located at its external surface. This silica slab is used to prepare a simulation box corresponding to a nanoporous slit having a pore size H = 10 nm. Fig. 1(c) illustrates the silica nanopore filled with methane at a pressure P = 200 atm and a temperature T = 300 K. Each methane molecule is represented as a spherical Lennard–Jones particle with parameters taken from the TraPPE force field.30 While this model adopts a simplified description of the fluid by neglecting internal degrees of freedom, previous molecular simulations have shown excellent agreement between the predicted thermal conductivity and experimental measurements (with only small deviations observed at low densities).11The number of fluid atoms confined within the silica nanopore is obtained using Monte Carlo simulations in the Grand Canonical ensemble (GCMC) as discussed in the Methods section. This approach ensures that the chemical potential and temperature of the atoms inside the nanoporosity match those of a bulk phase under specific conditions. Consequently, considering a pure fluid here, the gas pressure is also uniquely defined for a given set of temperature and chemical potential. The adsorption isotherm Na(P) for methane confined at T = 300 K within the silica nanopore is presented in Fig. 2(a). The absolute adsorbed amount Na is determined as the ensemble average of the number of adsorbed atoms as a function of the gas reservoir pressure P. This adsorption isotherm exhibits a Type I (Langmuir) behavior – with a steep initial uptake at low pressures followed by slower and slower increase until adsorption saturation. Despite the large pore size H, no capillary condensation occurs as methane is above its bulk critical temperature Tc = 190.56 K. For three pressures taken along the adsorption isotherm, we show in Fig. 2(b) the fluid density profile ρa(z) along the direction perpendicular to the pore surfaces. These density profiles exhibit a pronounced peak near the pore surface, which is indicative of the adsorbed layer formed due to the interactions between the fluid atoms and the solid atoms. Beyond this adsorbed layer, the fluid density becomes more uniform to recover the bulk fluid density in the pore center.
To explore the role of the solid/fluid affinity on adsorption and thermal transport, we varied the solid/fluid interaction strength ε. The value of the nominal interaction strength ε0 is given in the SI. In more detail, while maintaining the absorbed density Na at a constant value, the solid/fluid interaction strength was set to ε = ε0, 0.5ε0, and 2ε0 where ε0 denotes the nominal interaction strength (see Methods section). As this change affects the fluid energy landscape near the solid surface, the density profile ρa(z) is found to evolve upon changing ε. The chemical potential μ of the confined fluid evolves here as we change the solid/fluid interaction strength ε while keeping the total number of atoms constant (this explains why the fluid density in the pore center changes upon changing ε). The inset in Fig. 2(b) depicts the density profiles corresponding to each interaction strength ε as determined for the total adsorbed amount Na = 6.6 molec./nm3. The stronger solid/fluid interactions lead to a stronger adsorption peak as molecules from the pore central region migrate towards the surface. To quantify this effect, we introduce the excess density Nexa which measures the excess of fluid molecules with respect to the bulk density under the same thermodynamic conditions ρa(0) (taken as the density at the position z = 0 which corresponds to the pore center):
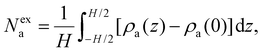 | (5) |
4.2 Vibrational spectra and solid/fluid dynamical coupling
Before discussing the overall fluid + solid thermal conductivity, we first discuss the impact of fluid adsorption on the vibrational properties of the solid and fluid.11,15,37 Throughout the remainder of this paper, α and β refer to the fluid and solid phases, respectively. As shown in Fig. 3(a), we computed the vibrational density of states (VDOS), i.e. D(ω)β, of the silica atoms within 3 Å layers taken at different positions with respect to the silica surface. We consider the following regions: surface [S], subsurface [S*], and bulk [B]. D(ω)β is obtained for each region by taking the Fourier transform of the velocity autocorrelation functions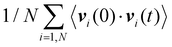 where the sum runs over all atoms i in the corresponding layer. The VDOS for each region are shown in Fig. 3(b) where we also compare the results for the empty and filled nanopores (the adsorbed fluid is taken at a pressure of 200 atm, i.e. Na = 6.6 molec./nm3). The VDOS for the silica slab Dβ(ω) exhibits some features that reflect its chemical and amorphous nature. Notably, the peak observed in the high-energy range (ħω ∼ 450 meV) can be assigned to the stretching of the hydroxyl groups at the surface. This is consistent with experimental measurements reporting silanol stretching frequencies around ∼3600 cm−1 (equivalent to 446 meV).38 Additionally, Dβ(ω) displays a broad distribution of vibrational frequencies, which arise from the disordered atomic structure of amorphous silica (this contrasts with crystalline materials where phonon modes appear as sharp peaks in the VDOS due to the strong positional and orientation ordering). As expected, the VDOS for the fluid phase Dα(ω) does not display an assembly of discrete narrow peaks, but rather a continuous distribution of states in the low energy range corresponding to ħω ∼ 0–25 meV.
where the sum runs over all atoms i in the corresponding layer. The VDOS for each region are shown in Fig. 3(b) where we also compare the results for the empty and filled nanopores (the adsorbed fluid is taken at a pressure of 200 atm, i.e. Na = 6.6 molec./nm3). The VDOS for the silica slab Dβ(ω) exhibits some features that reflect its chemical and amorphous nature. Notably, the peak observed in the high-energy range (ħω ∼ 450 meV) can be assigned to the stretching of the hydroxyl groups at the surface. This is consistent with experimental measurements reporting silanol stretching frequencies around ∼3600 cm−1 (equivalent to 446 meV).38 Additionally, Dβ(ω) displays a broad distribution of vibrational frequencies, which arise from the disordered atomic structure of amorphous silica (this contrasts with crystalline materials where phonon modes appear as sharp peaks in the VDOS due to the strong positional and orientation ordering). As expected, the VDOS for the fluid phase Dα(ω) does not display an assembly of discrete narrow peaks, but rather a continuous distribution of states in the low energy range corresponding to ħω ∼ 0–25 meV.
In agreement with previous results on fluid within zeolite cages,11 the vibrational modes of the fluid atoms at the pore surface shows a weak coupling with those of the solid atoms. However, this coupling is difficult to observe here as it only affects atoms directly located at the solid/fluid interface. Indeed, considering the large pore width H = 10 nm, the fraction of fluid atoms at the solid surface remains overall very small. In other words, even if the adsorbed density Na is large, a great number of molecules are distributed in the large porous domain available to the fluid – therefore, making the contribution of adsorbed fluid atoms small in the overall VDOS Dα(ω). As a result, Dα(ω)for the confined fluid remains very close to that of the bulk phase. In contrast, the presence of fluid atoms in contact with the solid surface impacts the vibrational properties of the solid surface layers [S] and [S*]. This effect can be seen in the low energy range where the fluid density of states Dα(ω) is non-zero. The adsorption-induced changes in Dβ(ω) indicate that the solid atoms at the solid/fluid interface display a different vibrational behavior in the presence of adsorbed fluid atoms. As will be discussed in detail below, this result provides evidence for phonon scattering by the fluid at the surface – the so-called rattle effect – and the resulting decrease in the solid's contribution to the thermal conductivity. In contrast, as expected, the fluid phase does not impact the VDOS for the solid atoms in the bulk region of the silica slab. The VDOS for bulk amorphous silica, which was used to prepare the silica nanopore, closely matches the VDOS obtained for the bulk region [B] in the silica wall. This further corroborates that the rattle mechanism stems from the coupling between the fluid and the solid at their interface.
4.3 Thermal conductivity of fluid-filled nanoporous silica
The domain sizes in the system under study lead to fluid and solid phases with quite distinct contributions to the thermal conductivities compared to their bulk counterpart value. It is therefore important to assess how each phase impacts the effective thermal transport within the fluid + solid system. As discussed in Section 2, the heat current j can be computed as a sum of individual atomic terms. For an individual atom, this involves summing over its convective jcon and virial jvir terms according to eqn (4a) and (4b), respectively. On the one hand, the convective term jcon accounts for the heat current contribution corresponding to the kinetic energy of atoms. On the other hand, the virial term jvir stands for the potential energy exchange through the multiple interactions experienced by an atom as described by the atomic stress tensor σi defined in Section 2 [eqn (4b)]. For a given fluid atom i, this atomic stress tensor σi is expressed as a sum of interactions with atoms in the same phase (αα) and with the atoms of the other phase (αβ). This yields a total stress which can be written as σi = σi,(αα) + σi,(αβ) and a virial contribution which can be decomposed as jviri = jviri,(αα) + jviri,(αβ).
Fig. 4(a) illustrates the total heat current autocorrelation function 〈j∥(t)· j∥(0)〉 at T = 300 K and P = 200 atm along the direction parallel to the surface. The effective thermal conductivity κ is obtained by integrating the heat flux autocorrelation function in eqn (1) as shown in Fig. 4(b). Fig. 4(c) shows the effective thermal conductivity κ‖ in the direction parallel to the interface, with contributions from both the solid and fluid phases, as a function of the fluid density Na within the nanopore. Changing the fluid/solid interaction strength ε impacts both the rattle effect at the solid/fluid interface but also the adsorption layer density. As a result, when considering the effect ε, the different trends in Fig. 4(c) illustrate the competition between the changes in the thermal conductivity through the solid phase and the changes in the thermal conductivity through the adsorbed layer. This point will be further discussed in the next section. The thermal conductivity of the empty silica nanopore is 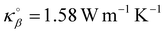 – a value slightly larger but quite consistent with the experimental thermal conductivity for amorphous silica (∼1.30 W m−1 K−1).39 Considering the empty solid for which the fluid conductivity is necessarily zero (since Na = 0), it is interesting to note that one recovers the simple mixing rule which predicts that the effective thermal conductivity is given by
– a value slightly larger but quite consistent with the experimental thermal conductivity for amorphous silica (∼1.30 W m−1 K−1).39 Considering the empty solid for which the fluid conductivity is necessarily zero (since Na = 0), it is interesting to note that one recovers the simple mixing rule which predicts that the effective thermal conductivity is given by 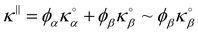 where ϕα = 0.642 and ϕβ = 0.358 are the volume fraction occupied by the fluid and solid phases while
where ϕα = 0.642 and ϕβ = 0.358 are the volume fraction occupied by the fluid and solid phases while 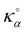 and
and 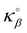 are the heat conductivity of the fluid and solid phases. In more detail, this yields κ‖ = 0.565 W m−1 K−1 which corresponds to the horizontal red dashed line in Fig. 4(c). As expected, regardless of the interaction strength ε, κ‖ tends to this value as Na → 0. In contrast, as soon as the adsorbed amount na becomes non-negligible, the interaction strength ε controls the affinity of the Na fluid atoms with the solid surface and defines the density of the adsorbed layer [Fig. 2(b)]. Although the overall thermal conductivity may remain approximately unchanged for weak solid–fluid interactions, the solid contribution κβ systematically decreases due to the rattle effect (as will be shown later in detail). However, while κβ decreases with na, we note that the intrinsic contribution κββ to κβ remains unchanged for all adsorbed amounts and fluid-interaction strengths. These data, which will be presented in detail in Fig. 5, indicate that the total conductivity κ remains unaffected by na for the lowest interaction strength as a result of the compensation between the rattle effect and the increase due to the addition of a fluid contribution. Therefore, changes in the interaction strength ε lead to different solid-fluid interfacial behavior and its contribution to the effective thermal conductivity. In the next section, this contribution will be analyzed to explain the different behaviors observed in the heat transport.
are the heat conductivity of the fluid and solid phases. In more detail, this yields κ‖ = 0.565 W m−1 K−1 which corresponds to the horizontal red dashed line in Fig. 4(c). As expected, regardless of the interaction strength ε, κ‖ tends to this value as Na → 0. In contrast, as soon as the adsorbed amount na becomes non-negligible, the interaction strength ε controls the affinity of the Na fluid atoms with the solid surface and defines the density of the adsorbed layer [Fig. 2(b)]. Although the overall thermal conductivity may remain approximately unchanged for weak solid–fluid interactions, the solid contribution κβ systematically decreases due to the rattle effect (as will be shown later in detail). However, while κβ decreases with na, we note that the intrinsic contribution κββ to κβ remains unchanged for all adsorbed amounts and fluid-interaction strengths. These data, which will be presented in detail in Fig. 5, indicate that the total conductivity κ remains unaffected by na for the lowest interaction strength as a result of the compensation between the rattle effect and the increase due to the addition of a fluid contribution. Therefore, changes in the interaction strength ε lead to different solid-fluid interfacial behavior and its contribution to the effective thermal conductivity. In the next section, this contribution will be analyzed to explain the different behaviors observed in the heat transport.
 | ||
| Fig. 4 Thermal conductivity κ of a fluid-filled silica nanopore. (a) Normalized time autocorrelation function of the heat current 〈j∥(t)·j∥(0)〉 at T = 300 K and Na ≈ 6.6 molec./nm3 along the direction parallel to the pore surface. (b) Thermal conductivity κ‖ as computed using the Green–Kubo formalism for P = 200 atm, T = 300 K and Na ≈ 6.6 molec./nm3. The variable t represents the upper time boundary in the Green–Kubo integral given in eqn (1), i.e. the cumulative integral of the heat-current autocorrelation function 〈j∥(t)·j∥(0)〉 up to time t. (c) Thermal conductivity parallel to the pore surface κ‖ as a function of the adsorbed amount Na. The data are presented for the three solid/fluid interaction strengths ε: 0.5ε0, ε0, and 2ε0 with the legend shown within the panel. The horizontal red dashed line represents the thermal conductivity of the empty nanoporous silica. | ||
Fig. S1 of the SI compares the effective thermal conductivity κ of the system as obtained in our molecular simulation and that predicted by using a classical mixing rule. While both datasets follow the same trend as a function of Na, the thermal conductivity by MD is slightly above the one predicted by the classical mixing rule. This may be explained by the thermal conductivity increase due to virial contribution on adsorbed fluid molecules, which is not take under consideration in the classical model. Moreover, we note that the breakdown of classical mixing rules can already be seen from the results shown in Fig. 4(c). Indeed, the mixing rule approach predicts a unique dependence of the effective thermal conductivity on the fluid adsorbed amount – in stark contrast with our molecular simulation results. In other words, our results reveal that changes in the solid/fluid interaction strength leads to qualitatively different dependences of κ(na) on (na) which cannot be captured by conventional mixing rules.
5 Discussion
5.1 Solid phase, fluid phase and solid/fluid interface contributions
To shed light on the impact of adsorption on thermal transport in the fluid-filled nanoporous silica, the different contributions to the overall thermal conductivity were calculated. In what follows, we decompose the total thermal conductivity κ into distinct contributions to the heat current autocorrelation function 〈j∥(t)· j∥(0)〉. As discussed in Section 2, the instantaneous heat current j(t) can be expressed as the sum of contributions from different groups of atoms. Accordingly, j(t) = jα + jβ + jαβ where jα, jβ, and jαβ are the fluid, solid, and solid/fluid interface contributions, respectively. In more detail, the fluid contribution jα to the heat current includes (i) the convective term jconα from all fluid atoms and the virial term jvir(αα) from fluid/fluid interactions. Similarly, the solid phase contribution jβ includes (i) the convective term jconβ from all solid atoms and (ii) the virial term jvir(ββ) from solid/solid interactions. Finally, the solid/fluid interfacial contribution jαβ to the heat current is equivalent to the virial term jvir(αβ) arising from solid/fluid interactions.Using the heat current decomposition into three contributions, the overall thermal conductivity 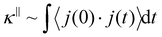 can be decomposed into 9 = 3 × 3 terms. Therefore, we define the following component contributions by the solid, fluid, and solid/fluid interface:
can be decomposed into 9 = 3 × 3 terms. Therefore, we define the following component contributions by the solid, fluid, and solid/fluid interface:
| κ‖β= κβ,α + κβ,αβ + κβ,β, | (6a) |
| κ‖α= κα,α + κα,αβ + κα,β, | (6b) |
| κ‖αβ= καβ,α + καβ,αβ + καβ,β. | (6c) |
These 9 terms narrow down to 6 terms owing to the symmetrical nature of the heat flux correlations 〈jδ(0)· jδ′(t)〉 (with δ, δ′ = α, β, αβ): 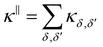 . All six independent thermal conductivity terms contributing to eqn (6) are reported in the SI. With this convention, the total in-plane thermal conductivity can be written as
. All six independent thermal conductivity terms contributing to eqn (6) are reported in the SI. With this convention, the total in-plane thermal conductivity can be written as
| κ‖ = κ‖α+ κ‖β+ κ‖αβ. | (7) |
Fig. 5 shows the three contributions κ‖α, κ‖β and κ‖αβ as a function of the adsorbed amount per unit of surface area na. We consider na rather than the total adsorbed amount Na as the latter does not capture how the fluid atoms distribute between the adsorbed layer at the pore surface and the bulk-like density in the pore center. The adsorbed amount is defined as 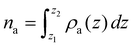 , where z1 ∼ 0 and z2 define the boundaries of the adsorbed peak in the vicinity of the silica surface. This definition of the adsorbed amount at given thermodynamic conditions allows rationalizing the variations in the overall thermal conductivity κ when changing the solid/fluid interaction strength ε0. All thermal conductivity contributions are evaluated for the different solid/fluid interaction strengths (see the caption of Fig. 5 for the labeling of each dataset). The schematic views in Fig. 5 illustrate the different contributions to the thermal conductivity as described in what follows. The top molecular configuration illustrates the phonons within the solid, which are responsible for thermal transport across amorphous silica. The middle molecular configuration illustrates the energy transfer between interacting fluid atoms within the confined phase. Finally, the bottom molecular configuration illustrates the energy transfer across the interface through the rattle effect between solid and fluid atoms.
, where z1 ∼ 0 and z2 define the boundaries of the adsorbed peak in the vicinity of the silica surface. This definition of the adsorbed amount at given thermodynamic conditions allows rationalizing the variations in the overall thermal conductivity κ when changing the solid/fluid interaction strength ε0. All thermal conductivity contributions are evaluated for the different solid/fluid interaction strengths (see the caption of Fig. 5 for the labeling of each dataset). The schematic views in Fig. 5 illustrate the different contributions to the thermal conductivity as described in what follows. The top molecular configuration illustrates the phonons within the solid, which are responsible for thermal transport across amorphous silica. The middle molecular configuration illustrates the energy transfer between interacting fluid atoms within the confined phase. Finally, the bottom molecular configuration illustrates the energy transfer across the interface through the rattle effect between solid and fluid atoms.
It is believed that the influence of the bulk-like region on κ‖α is minimal, given that the effective parallel thermal conductivity arises from surface and bulk-like contributions acting in parallel. Indeed, because κα is observed to be a unique function of the surface adsorbate density na (see Fig. 5(b)), the effective property appears to be primarily governed by the surface region. This suggests that fluid molecules outside the surface layer contribute weakly to heat transport. Moreover, the higher local density within the surface region, compared to the bulk-like region, enhances its thermal conductivity and further supports this interpretation. However, we note that, for the reasons provided above, while na is the controlling parameter discussed in this work, it is not possible to rule out that it also governs other confinement geometries.
5.2 Phonon lifetime and the rattle effect
In insulating materials such as amorphous silica, the thermal conductivity κ arises from phonon transport. We use here a simple statistical physics approach based on the microscopic kinetic theory of phonons to quantify phonon scattering in heterogeneous (i.e., solid + fluid) systems. Specifically, we model thermal transport in the solid structure by considering its vibrational modes while accounting for the so-called rattle effect. We begin with the microscopic expression for the thermal conductivity, which includes all phonon modes and their associated heat capacities within the solid phase: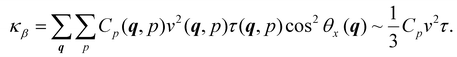 | (8) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θx(q) accounts for the velocity projection along a given Cartesian direction. The second equality is simply obtained by assuming that the material is isotropic and that both the group velocity and phonon lifetime are constant –v(q, p) ∼ v and τ(q, p) ∼ τ.
θx(q) accounts for the velocity projection along a given Cartesian direction. The second equality is simply obtained by assuming that the material is isotropic and that both the group velocity and phonon lifetime are constant –v(q, p) ∼ v and τ(q, p) ∼ τ.
To incorporate the effect of the adsorbed fluid na on phonon scattering via the rattle mechanism, we propose a stochastic model based on Matthiessen's rule.40 In this model, we implicitly assume that scattering occurs through two mechanisms independent of each other: (1) intrinsic phonon scattering within the solid (which is characterized by a relaxation rate τ0−1 that is independent of na and ε) and (2) phonon scattering at the solid/fluid interface due to collisions between fluid and solid atoms (which is characterized by a relaxation rate τa−1 that depends on na and ε). With this simple model, the number of phonons N(t + dt) not scattered at time t + dt is written as the number of phonons N(t) not scattered at time t minus the number of phonons scattered over the time interval dt: N(t + dt) = N(t)[1 − dt/τ0 − dt/τa(na, ε)]. Solving this differential equation leads to an exponential decay for the number of phonons:11,17
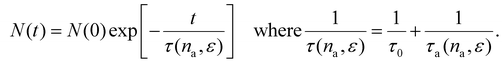 | (9) |
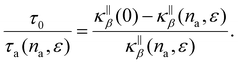 | (10) |
The relaxation time appearing in the left-hand side of eqn (10) corresponds to the ratio of the intrinsic phonon relaxation time τ0 to the relaxation time associated exclusively with fluid-induced scattering, τa(na,ε). The right-hand-side term arises from separating the total and fluid-induced scattering mechanisms through Matthiessen's rule.
We note that the phonon relaxation time τ introduced in eqn (8) (and further decomposed in τ0 and τa, which ratio is given by eqn (10)) characterizes heat transport through the solid phase – and not through the overall hybrid (i.e. solid + fluid) system. As a result, changes in τ due to the presence of adsorbed fluid at the solid/fluid interface directly impacts the solid thermal conductivity κβ. Fig. 5(d) shows the normalized phonon scattering rate,  , as a function of the adsorbed amount na for the three different solid/fluid interaction strengths: 0.5ε0, ε0, and 2ε0. The data shows that stronger solid-fluid interactions lead to a decrease in the phonon scattering rate. This result, which appears counterintuitive at first, can be rationalized as follows. Strong fluid/solid interactions lead to fluid atoms that are tightly physisorbed to the surface and, therefore, dynamically coupled to the solid molecular structure. As a result, such significant binding leads to frequent collisions between fluid and solid atoms which, in turn, leads to a decrease in the phonon lifetime and hence a decrease in the solid's heat conductivity. In fact, in agreement with prior observations, phonon scattering at the fluid/solid interface mostly depends on collisions between solid and fluid atoms and is therefore governed by momentum exchange at the solid surface.11
, as a function of the adsorbed amount na for the three different solid/fluid interaction strengths: 0.5ε0, ε0, and 2ε0. The data shows that stronger solid-fluid interactions lead to a decrease in the phonon scattering rate. This result, which appears counterintuitive at first, can be rationalized as follows. Strong fluid/solid interactions lead to fluid atoms that are tightly physisorbed to the surface and, therefore, dynamically coupled to the solid molecular structure. As a result, such significant binding leads to frequent collisions between fluid and solid atoms which, in turn, leads to a decrease in the phonon lifetime and hence a decrease in the solid's heat conductivity. In fact, in agreement with prior observations, phonon scattering at the fluid/solid interface mostly depends on collisions between solid and fluid atoms and is therefore governed by momentum exchange at the solid surface.11
These data reveal a strong dependence of  on ε with a crossover in the scattering behavior upon increasing na. Here, the dilute regime refers to the limit in which adsorbed molecules contribute independently to phonon scattering – such that each fluid atom acts as an independent phonon scatterer, leading to a linear scaling of
on ε with a crossover in the scattering behavior upon increasing na. Here, the dilute regime refers to the limit in which adsorbed molecules contribute independently to phonon scattering – such that each fluid atom acts as an independent phonon scatterer, leading to a linear scaling of 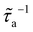 with na (even if intermolecular interactions within the adsorbed phase are non-negligible). This linear trend is confirmed by the dash-dotted line in Fig. 5(d). To quantify deviations from this ideal behavior corresponding to infinite dilution (Henry regime), we model the scattering rate as τa(na,ε)−1 ∼ f(na,ε)τ0a(ε)−1, where τ0a(ε) ∼ τa(na → 0, ε) is the reference scattering time in the dilute limit. For consistency, we define
with na (even if intermolecular interactions within the adsorbed phase are non-negligible). This linear trend is confirmed by the dash-dotted line in Fig. 5(d). To quantify deviations from this ideal behavior corresponding to infinite dilution (Henry regime), we model the scattering rate as τa(na,ε)−1 ∼ f(na,ε)τ0a(ε)−1, where τ0a(ε) ∼ τa(na → 0, ε) is the reference scattering time in the dilute limit. For consistency, we define 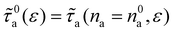 , with n0a= 10 molec./nm2 (however, we note that our results remain qualitatively independent of the exact choice as long as n0a lies within the linear regime). The inset of Fig. 5(d) shows the resulting function
, with n0a= 10 molec./nm2 (however, we note that our results remain qualitatively independent of the exact choice as long as n0a lies within the linear regime). The inset of Fig. 5(d) shows the resulting function 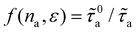 . We expect f(na,ε) to grow linearly at low na as each fluid atom scatters phonon independently of each other. In fact, while the data do not firmly show this linear regime in Fig. 5(d) with na in the low adsorbed amount range, it is theoretically expected. Indeed, in the limit of nearly non-interacting fluid molecules (Henry regime of the adsorption isotherm), we expect each fluid molecule to contribute independently of each other to the rattle effect – therefore, leading to a linear regime f(na) ∼ na. In contrast, as can be observed in the inset of Fig. 5(d) upon increasing na, f(na) becomes non linear and tends to a plateau as saturation of the rattle effect occurs when the nanoconfined fluid becomes very crowded. For the interaction strength ε = 0.5ε0, the function
. We expect f(na,ε) to grow linearly at low na as each fluid atom scatters phonon independently of each other. In fact, while the data do not firmly show this linear regime in Fig. 5(d) with na in the low adsorbed amount range, it is theoretically expected. Indeed, in the limit of nearly non-interacting fluid molecules (Henry regime of the adsorption isotherm), we expect each fluid molecule to contribute independently of each other to the rattle effect – therefore, leading to a linear regime f(na) ∼ na. In contrast, as can be observed in the inset of Fig. 5(d) upon increasing na, f(na) becomes non linear and tends to a plateau as saturation of the rattle effect occurs when the nanoconfined fluid becomes very crowded. For the interaction strength ε = 0.5ε0, the function 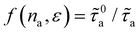 is close to unity over the evaluated range of na. This indicates that, for such weak interactions, the fluid-induced scattering rate is constant and close to that in the dilute regime (no cooperative effect between adsorbed molecules in terms of phonon scattering). Moreover, the corresponding rates
is close to unity over the evaluated range of na. This indicates that, for such weak interactions, the fluid-induced scattering rate is constant and close to that in the dilute regime (no cooperative effect between adsorbed molecules in terms of phonon scattering). Moreover, the corresponding rates 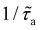 exhibit larger relative statistical fluctuations and do not display the monotonic trends observed for the other interactions strengths ε0 and 2ε0.
exhibit larger relative statistical fluctuations and do not display the monotonic trends observed for the other interactions strengths ε0 and 2ε0.
Formally, f(na) describes the dependence on the adsorbed amount na of the scattering rate 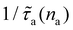 due to fluid molecules at the surface (the latter is normalized by the scattering rate in the limit of a vanishingly small number of fluid molecules
due to fluid molecules at the surface (the latter is normalized by the scattering rate in the limit of a vanishingly small number of fluid molecules 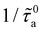 ). Within the silica pore, f(na,ε) grows linearly at low na as each fluid atom scatters phonons independently of each other. On the other hand, as na increases, fluid/fluid interactions give rise to cooperative scattering which results in a increase in f(na,ε) followed by saturation as the interface becomes crowded/saturated at very large na. Interestingly, this transition from independent (linear) to cooperative (non linear) behavior of the so-called “rattle effect” is not observed for methane confined within zeolites,11 where f(na) remains linear until saturation is reached. This qualitative difference likely stems from pore morphology/topology effects: severe confinement and very large surface areas in zeolites do not lead to collective fluid effects within the adsorbed phase as fluid/fluid interactions are negligible over fluid/solid interactions. In contrast, the open silica surface studied here leads to large fluid/fluid interactions at the surface, which result in a different dependence of the scattering rate on the adsorbed amount f(na).
). Within the silica pore, f(na,ε) grows linearly at low na as each fluid atom scatters phonons independently of each other. On the other hand, as na increases, fluid/fluid interactions give rise to cooperative scattering which results in a increase in f(na,ε) followed by saturation as the interface becomes crowded/saturated at very large na. Interestingly, this transition from independent (linear) to cooperative (non linear) behavior of the so-called “rattle effect” is not observed for methane confined within zeolites,11 where f(na) remains linear until saturation is reached. This qualitative difference likely stems from pore morphology/topology effects: severe confinement and very large surface areas in zeolites do not lead to collective fluid effects within the adsorbed phase as fluid/fluid interactions are negligible over fluid/solid interactions. In contrast, the open silica surface studied here leads to large fluid/fluid interactions at the surface, which result in a different dependence of the scattering rate on the adsorbed amount f(na).
Although differences in the phonon spectra for crystalline zeolite and amorphous silica are expected, we note that f(na,ε) relates directly to the scattering of phonons by fluid molecules at the solid/liquid interface regardless of the intrinsic features of each solid phase. In other words, as can be seen from our formal treatment using Matthiessen's rules, fluid-driven scattering of the phonon modes adds to the intrinsic phonon scattering independently of the type of phonon spectrum (crystalline versus amorphous material).
6 Conclusions
We conducted a quantitative assessment of the thermal conductivity in a prototypical system consisting of a nanoporous material filled with a confined fluid. For this, we simulated a system composed of a simple fluid confined in a realistic model of nanoporous silica made up of amorphous silica walls. By considering different solid/fluid interaction strengths ε and adsorbed amounts Na, we investigate the parameters that give rise to the rattle effect. Despite the use of thick silica walls, we find that the rattle effect remains a non-negligible phenomenon that affects the overall thermal conductivity in such solid/fluid systems. We identify that the thermal conductivity and its fluid phase, solid phase, and solid/fluid interface contributions can be rationalized by taking into account the adsorbed density na. While the thermal conductivity of the solid phase κ‖β is decreased by the rattle effect, the thermal conductivity of the fluid phase κ‖α increases as the fluid density increases within the pore. The thermal conductivity contribution from both fluid and solid phases (κ‖α and κ‖β, respectively) follows a unique and general relation with the adsorbed amount – regardless of the interaction strength between the solid and fluid atoms. We also evaluate the role of the solid/fluid interface contribution κ‖αβ to the thermal conductivity. This term is a function of the adsorbed density na but also slightly dependent of the adsorbent/adsorbate interaction strength ε.The connection between the thermal conductivity in solid/fluid systems and the fluid interfacial/adsorbed density suggests that the rattle mechanism is an important contribution. This microscopic phenomenon, which arises from phonon scattering induced by collisions of the fluid atoms with the solid surface, leads to a decrease in the solid's contribution to thermal conductivity. As for the fluid contribution to the overall thermal conductivity, we find that increasing na leads to a much larger number of collisions – which increases the virial (i.e. potential energy) contribution to the thermal conductivity. While our results suggest that the rattle effect is a general mechanism that should be taken into account in any solid/fluid systems, further evaluation is needed to assess its impact for more complex systems such as when polyatomic molecules or mixtures are considered. In practice, our findings highlight the inherent limitations of conventional mixing rules for porous materials systems. Such macroscopic approaches, which are routinely used in engineering applications, neglect the coupling between the solid and fluid phases and its consequences. As a result, describing thermal transport in such complex solid/fluid phases requires an atomistic-level approach to correctly capture the underlying mechanisms. Finally, the findings reported in the present result suggest that the solid/fluid coupling can be harnessed to engineer specific nanodevices with controlled effective thermal conductivity. Future studies may explore the impact of varying fluid types and nanopores geometries on the rattle effect as well as the effect of temperature and pressure on phonon scattering dynamics. These investigations could ultimately lead to the development of materials tailored for specific thermal management applications. This opens perspectives for the design of materials with tailored and optimized thermal properties.
Author contributions
B. C. and L. F. M. F conceived the research. N. F. S. carried out the molecular simulations. N. F. S., T. R. G., C. H. and B. C. analyzed the data and developed the model. All authors contributed to the analysis of the results. N. F. S and B. C. wrote the manuscript with inputs from all authors.Conflicts of interest
There are no conflicts to declare.Data availability
Data for this article, including the molecular configurations generated and/or analyzed in the framework of this study are available at Science Data Bank at https://doi.org/10.57760/sciencedb.30816.Supplementary information (SI): additional data, corresponding to the force field parameters, comparison of simulation data with the classical mixing rules and details about the thermal conductivity contributions defined in the text. See DOI: https://doi.org/10.1039/d5nr04591f.
Acknowledgements
This work was partially supported by São Paulo Research Foundation (FAPESP) grant no. 2018/02713-8 and 2021/11105-4, CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico), and CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior). This work was also carried within the framework of a joint project HTD-POM (ANR-23-CE08-0038-01) funded by the French Agence Nationale pour la Recherche (ANR) and the Brazilian agency FAPESP (grant no. 2023/03170-6). All (or most of) the computations presented in this paper were performed using the GRICAD infrastructure (https://gricad.univ-grenoble-alpes.fr), which is supported by Grenoble research communities agency. N. F. S. and B. C. are grateful to J. L. Barrat (Univ. Grenoble Alpes) for stimulating discussions.References
- G. Sneddon, A. Greenaway and H. H. Yiu, Adv. Energy Mater., 2014, 4, 1301873 CrossRef.
- E. Kianfar and H. Sayadi, Carbon Lett., 2022, 32, 1645–1669 CrossRef.
- D. S. Sholl and R. P. Lively, Nature, 2016, 532, 435–437 CrossRef PubMed.
- L. Bocquet, Nat. Mater., 2020, 19, 254–256 CrossRef CAS PubMed.
- N. Kavokine, R. R. Netz and L. Bocquet, Annu. Rev. Fluid Mech., 2021, 53, 377–410 CrossRef.
- A. Schlaich, J.-L. Barrat and B. Coasne, Chem. Rev., 2025, 125, 2561–2624 CrossRef CAS PubMed.
- G. Gonella, E. H. Backus, Y. Nagata, D. J. Bonthuis, P. Loche, A. Schlaich, R. R. Netz, A. Kühnle, I. T. McCrum and M. T. Koper, et al. , Nat. Rev. Chem., 2021, 5, 466–485 CrossRef CAS PubMed.
- Y. F. Han, S. Li, H.-D. Liu and W. Cui, Therm. Sci., 2020, 24, 3749–3756 CrossRef.
- D. Raabe, Modell. Simul. Mater. Sci. Eng., 2004, 12, R13 CrossRef.
- Y.-L. He and W.-Q. Tao, J. Heat Transfer, 2015, 137, 090801 CrossRef.
- N. F. Souza, C. Picard, L. F. M. Franco and B. Coasne, J. Phys. Chem. B, 2024, 128, 2516–2527 CrossRef PubMed.
- R. Alipour, M. Ghoranneviss, M. Mirzaee and A. Jafari, Heat Mass Transfer, 2014, 50, 1727–1735 CrossRef CAS.
- X. Liang, Chin. Sci. Bull., 2007, 52, 2457–2472 CrossRef CAS.
- G. Nolas, G. Slack, D. Morelli, T. Tritt and A. Ehrlich, J. Appl. Phys., 1996, 79, 4002–4008 CrossRef CAS.
- J. A. Thomas, R. M. Iutzi and A. J. McGaughey, Phys. Rev. B: Condens. Matter Mater. Phys., 2010, 81, 045413 CrossRef.
- E. J. Rosenbaum, N. J. English, J. K. Johnson, D. W. Shaw and R. P. Warzinski, J. Phys. Chem. B, 2007, 111, 13194–13205 CrossRef CAS PubMed.
- H. Babaei and C. E. Wilmer, Phys. Rev. Lett., 2016, 116, 025902 CrossRef PubMed.
- H. Babaei, M. E. DeCoster, M. Jeong, Z. M. Hassan, T. Islamoglu, H. Baumgart, A. J. H. McGaughey, E. Redel, O. K. Farha, P. E. Hopkins, J. A. Malen and C. E. Wilmer, Nat. Commun., 2020, 11, 4010 CrossRef CAS PubMed.
- V. V. Murashov, J. Condens. Matter Phys., 1999, 11, 1261 CrossRef CAS.
- C.-Y. Chen and D. I. Kopelevich, Mol. Simul., 2008, 34, 155–167 CrossRef CAS.
- H. Mizuno, S. Mossa and J.-L. Barrat, Sci. Rep., 2015, 5, 14116 CrossRef CAS PubMed.
- S. R. De Groot and P. Mazur, in Non-equilibrium thermodynamics, Courier Corporation, 2013 Search PubMed.
- G. Paolini and G. Ciccotti, Phys. Rev. A, 1987, 35, 5156 CrossRef CAS PubMed.
- D. Surblys, H. Matsubara, G. Kikugawa and T. Ohara, Phys. Rev. E, 2019, 99, 051301 CrossRef CAS PubMed.
- B. Coasne, L. Viau and A. Vioux, J. Phys. Chem. Lett., 2011, 2, 1150–1154 CrossRef CAS PubMed.
- L. N. Ho, S. Clauzier, Y. Schuurman, D. Farrusseng and B. Coasne, J. Phys. Chem. Lett., 2013, 4, 2274–2278 CrossRef CAS.
- S. Hocine, R. Hartkamp, B. Siboulet, M. Duvail, B. Coasne, P. Turq and J.-F. Dufrêche, J. Phys. Chem. C, 2016, 120, 963–973 CrossRef CAS.
- L. Zhuravlev, Langmuir, 1987, 3, 316–318 CrossRef CAS.
- A. Carré, J. Horbach, S. Ispas and W. Kob, Europhys. Lett., 2008, 82, 17001 CrossRef.
- B. L. Eggimann, A. J. Sunnarborg, H. D. Stern, A. P. Bliss and J. I. Siepmann, Mol. Simul., 2014, 40, 101–105 CrossRef CAS.
- R. T. Cygan, J.-J. Liang and A. G. Kalinichev, J. Phys. Chem. B, 2004, 108, 1255–1266 CrossRef CAS.
- H. A. Lorentz, Ann. Phys., 1881, 248, 127–136 CrossRef.
- D. Berthelot, Compt. Rendus, 1898, 126, 15 Search PubMed.
- L. N. Ho, Y. Schuurman, D. Farrusseng and B. Coasne, J. Phys. Chem. C, 2015, 119, 21547–21554 CrossRef CAS.
- B. Coasne and P. Ugliengo, Langmuir, 2012, 28, 11131–11141 CrossRef CAS PubMed.
- S. Plimpton, J. Comput. Phys., 1995, 117, 1–19 CrossRef CAS.
- N. J. English and S. T. John, Phys. Rev. Lett., 2009, 103, 015901 CrossRef PubMed.
- L. Dalstein, E. Potapova and E. Tyrode, Phys. Chem. Chem. Phys., 2017, 19, 10343–10349 RSC.
- S. Andersson and L. Dzhavadov, J. Condens. Matter Phys., 1992, 4, 6209 CrossRef CAS.
- N. W. Ashcroft and N. Mermin, Solid state, Holt Rinehart and Winston, 1976, vol. 1 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |