Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Advancing natural product R&D&I: guide for compliance with responsible research and innovation, ethics, Nagoya and Cartagena Protocols, and intellectual property protection
Susana P. Gaudêncio
 *ab,
Belma Kalamujić Stroil
*ab,
Belma Kalamujić Stroil
 c,
Lucie Novoveska
c,
Lucie Novoveska
 d,
Céline Rebours
d,
Céline Rebours
 e and
Xenia T. Schneider
e and
Xenia T. Schneider
 f
f
aAssociate Laboratory i4HB, Institute for Health and Bioeconomy, NOVA School of Science and Technology, NOVA University of Lisbon, 2819-516 Caparica, Portugal. E-mail: s.gaudencio@fct.unl.pt
bUCIBIO—Applied Molecular Biosciences Unit, NOVA School of Science and Technology, NOVA University of Lisbon, 2819-516 Caparica, Portugal
cUniversity of Sarajevo-Institute for Genetic Engineering and Biotechnology, 71000 Sarajevo, Bosnia and Herzegovina
dUK Centre for Ecology and Hydrology, Bush Estate, Penicuik, EH26 0QB, UK
eMøreforsking AS, 6021 Ålesund, Norway
fXPRO Consulting Limited, Strovolos 2021, Cyprus
First published on 1st May 2026
Abstract
Covering: January 2005 to July 2025
The interplay between Responsible Research and Innovation, the Nagoya and Cartagena Protocols and other legislative frameworks, ethical practices, and intellectual property protection plays a pivotal role in shaping the field of natural products R&D&I, and the bioeconomy. Together, these elements establish a holistic framework for the sustainable and equitable utilization of natural resources, while promoting innovation and supporting economic growth.
1. Introduction
Responsible Research and Innovation (RRI) has become a key framework for aligning science and technological development with societal values, especially in ethically sensitive fields such as the bioeconomy. Ethics is about ensuring that research and innovation activities uphold ethical principles, including respect for human rights, societal values, and environmental sustainability.The bioeconomy, particularly research, development and innovation (R&D&I) involving natural products, illustrates both the challenges and opportunities for applying RRI to real-world scientific practice. In this field, key ethical considerations include biodiversity conservation, protection of genetic resources (GR) and associated traditional knowledge (TK), ensuring equitable benefit-sharing. International legal frameworks like the Convention on Biological Diversity (CBD),1 the Nagoya Protocol,2 and the World Intellectual Property Organization (WIPO) on Genetic Resources and Associated Traditional Knowledge (GRATK) Treaty,3 provide regulatory support, enforce benefit-sharing obligations, and promote accountability. Intellectual property rights (IPR) serve as essential drivers enabling researchers and companies to profit from their discoveries while incentivizing continued research investment. However, in bioprospecting context, IPR systems should be adapted to safeguard Indigenous Peoples and Local Communities (IPLCs) knowledge through sui generis† or hybrid approaches that balance innovation with equitable recognition and benefit-sharing.
Embedding RRI and ethical governance into natural product R&D&I is essential for building trust, promoting justice, and supporting sustainable development within the growing sector of bioeconomy.
This review examines how RRI, ethical practices, legal frameworks, and intellectual property (IP) influence the development of natural products R&D&I and the broader bioeconomy. It begins by outlining the concept of RRI, its application in Europe and internationally, and its links to European Union (EU) strategies, emerging biotechnology legislation, artificial intelligence (AI), new genomic technologies (NGTs), sustainability, and the protection of GR and TK. The review then highlights ethics as a foundational element of sustainable natural product R&D&I, emphasizing the need to respect for the rights of IPLCs when accessing their GR and associated TK. It reviews European and national ethical guidelines, including a detailed example from Norway, and examines ethical challenges such as biopiracy and the misuse of GR and TK. A section is dedicated to the Nagoya and Cartagena Protocols and related legal frameworks governing access to GR and TK benefit-sharing. It discusses regulatory gaps, compliance challenges, the access and benefit sharing (ABS) workflow, tensions surrounding digital sequence information (DSI), the role of biorepositories, and emerging governance frameworks such as marine genetic resources (MGR) under the High Seas Treaty. The review then analyses IPR related to GR and TK, including how global health emergencies influence governance, the tools available for IPR protection, the complexities of patenting genetic material, and differences between global and local IPR systems.
Finally, the review presents real-world case studies of ethical and legal governance in natural product R&D&I and concludes by offering guidance on navigating the interconnected challenges of RRI, ethics, legal obligations, and IP in the bioeconomy.
These topics are particularly relevant when considering the increasing emphasis on sustainability, biodiversity conservation, equitable benefit-sharing practices, and hold special significance for the EU Bioeconomy Strategy as well as the forthcoming EU Biotech Act.4–6
2. Responsible research and innovation: framework and principles
RRI is a multifaceted concept,7–9 often difficult to define due to its inherently broad, interdisciplinary, and context-dependent nature. As a result, it has been interpreted differently across disciplines, projects, and national contexts. Rather than representing a single, fixed concept, RRI functions as a boundary subject, flexible enough to be adapted by diverse stakeholders, yet structured enough to support shared objectives, reflecting its pluralistic and evolving nature.10RRI operates as dynamic, continuously negotiated framework that aligns science and innovation with societal needs and values. It places the end-user at the centre of the R&D&I process and engages stakeholders throughout the research and innovation cycle to ensure that outcomes are ethically sound, socially desirable, and sustainable.7–9 By fostering broad participation, RRI strengthens public trust, accelerates innovation uptake, and improves science-society communication.11
2.1. Responsible research and innovation in Europe
The origins of RRI can be traced back to European policy developments in the late 1980s, when the social dimensions of research first gained attention under Framework Programme 2 (FP2) and later expended during FP5 and FP6 amid growing public distrust and scientific controversies. Events such as the Bovine Spongiform Encephalopathy (BSE) crisis, debates over genetically modified organisms (GMOs), and concerns about nanotechnology and surveillance exposed growing gaps between technological advances and societal acceptance.9 Moreover, the 2001 European Commission (EC) White Paper on Governance further emphasized the need to bring European citizens closer to decision-making and address the democratic deficit, calling for a more participatory approach to science policy, laying the groundwork for the Science and Society programme.12,13 Together with a broader EU shift toward transparency and accountability, these developments created the conditions for RRI to emerge as a framework for more democratic and inclusive science governance.In 2011, RRI formally entered the European policy discourse. Von Schomberg's widely cited definition framed it as “a transparent, interactive process by which societal actors and innovators become mutually responsive to each other with a view to the (ethical) acceptability, sustainability and societal desirability of the innovation process and its marketable products”,14 aiming to integrate science and technology properly into society. The Declaration of Rome on RRI reinforces this view, describing it as the ongoing alignment of research and innovation with societal values, needs, and expectations.15 However, operationalizing this ambitious vision into practice has proven challenging, as it requires systemic changes to governance of science and innovation.
Conceptual ambiguity has led to multiple frameworks attempting to capture RRI's essence. Stilgoe et al. (2013) proposed four dimensions: anticipation, inclusion, reflexivity, and responsiveness that support a proactive, ethically informed, and dialogical approach to emerging technologies, grounded in democratic deliberation.9,16 Simultaneously, EC institutionalized RRI through five “RRI keys”: public engagement, gender equality, science education, ethics, and open access (OA). While these keys are more concrete and measurable, their procedural application has often reduced RRI to a set of compliance tasks rather than transformative practices.17
The disconnection between normative visions and practical instruments have sparked debate. Some scholars argued that RRI has become overly technocratic, drifting away from its initial emphasis on values, responsibility, and systemic innovation governance.18,19 Institutionalizing RRI within research cultures dominated by disciplinary silos, short-term goals, and productivity metrics, has been a difficult endeavor.20
The RRI “keys”, originally action lines of the “Science in Society” programme and rebranded under the RRI banner during Horizon 2020, offered a pragmatic structure for implementation and evaluation.9,17 At a policy level, the EC institutionalized RRI to foster a model of innovation governance aimed not only to minimize risks and avoid controversy but also at shaping research agendas that are ethically acceptable, sustainable, and socially desirable.14
After 2015, the rise of the Open Science (OS) agenda, which seeks to make the entire research process more transparent, accessible, and collaborative, introduced a complementary emphasis on transparency, data sharing, and citizen science.21,22 OS includes OA to publications (see Section 4.4.1), as well as open research data, open methods and code of conduct, open peer review, research integrity, citizen science, and new incentive and evaluation systems, collectively promoting collaboration, societal engagement, and transparency across the entire research lifecycle.21 While OS shares values with RRI, such as openness and inclusivity, it focuses on efficiency and digital collaboration.22
RRI gained further momentum under Horizon 2020,10 moving beyond earlier frameworks like Ethical, Legal, and Social Issues (ELSI) and Ethical, Legal, and Social Aspects (ELSA).16 Unlike these earlier approaches, RRI advocates a more proactive, anticipatory, inclusive, and value-driven approach to research governance.18
Formally integrated in Horizon 2020, the European Strategy for RRI reinforces the expectation that research aligns with societal needs, values, and ethical standards.23 It places greater emphasis on six core dimensions: public engagement, gender equality, science education, OA, ethics, and responsible governance.23 Researchers are encouraged to involve stakeholders throughout the research cycle and to demonstrate the societal desirability and sustainability of their work, moving beyond excellence alone toward excellence with responsibility.23
In Horizon Europe (2021–2027), RRI remains a policy objective but with less prominence than in Horizon 2020. A key ethical requirement is the “Do No Significant Harm” (DNSH) principle, tied to the EU Taxonomy Regulation, which mandates that research avoids adverse effects on environmental goals, including climate change mitigation, biodiversity, and pollution prevention. To support RRI operationalization and implementation in R&D&I, there are several initiatives like the RRI Roadmap©™,24 a proprietary framework with tools integrating design thinking and change management, based on broad stakeholder consultation across sectors including the Blue Economy, providing practical resources for inserting RRI principles in research and innovation projects.25 Complementing these approaches, the One Health concept offers a collaborative framework recognizing the interdependence of human, animal, and environmental health, aiming to address shared risks and promote sustainable outcomes globally.26
To be transformative, RRI must move beyond basic compliance, by embedding reflexivity, anticipation, and stakeholder engagement into R&D&I governance, while remaining adaptive, context-sensitive, and responsive to societal needs.17,22,27
RRI, OS, and related frameworks are largely complementary with shared values (i.e., transparency, inclusivity, and societal engagement). Additionally, OS can reinforce RRI by increasing openness in data, methods, and participation, thereby enhancing accountability and supporting stakeholder involvement. However, tensions may emerge: OS often prioritizes efficiency, rapid dissemination, and standardization, which can conflict with RRI's emphasis on deliberation, reflexivity, and context-sensitive transformative governance.7–9,21
2.2. Responsible research and innovation beyond Europe
Beyond Europe, RRI has not developed into a unified or institutionalized policy framework comparable to the European model.The Global North's approach to RRI is capital-oriented, grounded in strong democratic principles and robust social and ethical values.28 While in the Global South, it is more community-oriented, closely linked to development priorities, equity, and social justice, bottom-up mannered, emphasizing grassroots knowledge co-production and inclusive participation rather than a predominantly techno-economic focus.28
In the United States of America (USA), RRI is emerging, particularly in biotechnology, with opportunities for cross-sector collaboration and public engagement. However, progress is limited by institutional barriers, diverse stakeholder beliefs, and a lack of formal frameworks.29 RRI approaches are typically included in sector-specific regulation, have a stronger focus on industry, professional ethics, integrity, and risk-based oversight, organizational practices, and innovation management, rather than formalized through funding requirements or procedural frameworks.30 Advancing RRI in the USA will require context-sensitive practices that balance innovation, institutional constraints, and societal inclusivity to build trust and legitimacy.29
In Asia, RRI is diverse, evolving, and shaped by ongoing adaptation and learning with European frameworks.31 Countries like India and China adapt RRI to local institutional, cultural, and ethical contexts, integrating elements such as inclusivity, equity, and environmental considerations into technology and innovation governance. However, progress is constrained by limited public awareness, low stakeholder participation, and the absence of formalised institutional structures, making consistent implementation complex.29,31 Japan and South Korea face similar obstacles in applying RRI.32
Despite persistent challenges in implementation, RRI has had exerted a lasting influence by promoting an innovation model grounded in care, and societal responsibility.7,20
To achieve genuine global relevance, RRI must extend beyond formal, technology-focused strategies to embrace informal, community-driven approaches. This shift requires supporting mutual learning, collaboration, and alignment with the UN Sustainable Development Goals (SDGs) and the EU Biotech Act, while recognizing and integrating diverse practices including those originating outside Europe and Global North.28
2.3. The role of RRI in the EU bioeconomy strategy and the forthcoming EU biotech act
Within the European bioeconomy, RRI complements the objectives of the EU Bioeconomy Strategy, by advancing the transition toward a sustainable, circular, and climate-neutral economy. Embedding inclusivity, transparency, and long-term insight into technologies development,4 and through anticipatory governance, ethical compliance, and OS, it guarantees that bio-based innovations reflect the values and needs of diverse stakeholders, and can enable faster uptake of results.16This alignment is further reinforced by the forthcoming European Biotech Act, announced in the EU Bioeconomy Strategy Progress Report (2022),33 which is expected to establish a binding legislative framework and underscore the relevance of RRI principles. By introducing measurable sustainability criteria, enhanced stakeholder participation, and system-wide monitoring, the Act operationalizes RRI's emphasis on early engagement and anticipatory evaluation, enabling bio-based innovations that are not only technically sound but also socially accepted and economically viable. Additionally, core components of RRI, such as stakeholder engagement, ethical and legal compliance, OA to data, and long-term impact assessment, directly support the Bioeconomy Strategy's pillars of food and energy security, ecosystem restoration, and industrial transformation.4 Early co-creation among researchers, civil society organizations, policymakers, and consumers strengthens science–policy interfaces, builds societal trust, and facilitates the uptake of innovation.
These benefits are further amplified through integration with Industry 4.0 and circular economy approaches. Digital technologies, IoT, big-data analytics, automation, enhance traceability and resource efficiency across bio-based value chains, while circular economy principles reinforce ethical sourcing, waste minimization, and equitable benefit-sharing.34 Together, RRI and these frameworks reduce policy gaps, build capacities, and reduce economic uncertainty, enabling more inclusive participation, particularly from under-represented communities, and fostering responsible bioprospecting practices aligned with the EU's long-term bioeconomy objectives.
2.4. Artificial intelligence (AI) and generative AI
Advances in computational omics, machine learning, deep learning, artificial intelligence (AI) generative AI (GAI), and large language models (LLMs), are increasingly being adopted to support natural products R&D&I. AI-driven technologies accelerate data mining, interpretation, classification, and pattern recognition, enabling in silico screening as well as the prediction or simulation of biological activity.35–39 These technologies support the synthesis and integration of complex information across heterogeneous data types, including chemical structures, spectra, DNA sequences, biosynthetic gene clusters, and bioactivity profiles, accelerating the identification of valuable biological resources and the characterization of bioactive compounds. These tools optimize molecular discovery and design workflows, advance de novo and natural-product-inspired drug design, generate data-driven hypotheses, and forecast experimental or biological outcomes.35–39 Collectively, these methods enable the exploration of under-sampled chemical and biological space and have the potential to deepen mechanistic understanding of natural products modes of action.40–44 By supporting resource prioritization and reducing the need for repeated physical access to biological materials, AI approaches also contribute to biodiversity conservation goals.40–44In parallel, AI-generated datasets that simulate experimental or sequence data are increasingly used to train models or augment existing datasets.45 Such synthetic data may further enhance bioprospecting and drug development by enabling researchers to model biological scenarios, expanding datasets without compromising sensitive information, and testing hypotheses with minimal ethical or environmental impact.45,46
Despite these advances, the impact of AI in natural products R&D&I is constrained primarily by data-related limitations rather than by a lack of algorithmic innovation.35 Available natural product datasets are often small, fragmented, heterogeneous, proprietary, and insufficiently standardized compared to those available for synthetic compounds, increasing the risk of overfitting and frequently favouring simpler, less parameterized models over complex architectures.35–37,40 Furthermore, when generating synthetic data for AI applications, there is a risk that feedback loops may arise, leading the AI to produce unreliable or self-reinforcing outputs. Addressing these challenges requires sustained investment in data standardization, curation, datasets validation, and interoperability across specialized databases, as well as long-term database maintenance.35–37,40
In alignment with RRI principles, AI-generated predictions must be rigorously validated through experimental and empirical methods to ensure reproducibility, robustness, and scientific reliability, and to avoid misdirecting research efforts or enabling the misappropriation or misuse of biological knowledge.35–44
The responsible integration of AI and GAI into natural products R&D&I depends on robust governance with adherence to ethical and legal frameworks, cybersecurity measures and interdisciplinary collaboration.47–49 Without rigorous governance, AI systems risk entrenching dominant scientific paradigms, marginalizing alternative perspectives and knowledge systems of IPLCs, and generate biased or misleading outputs that undermine evidence-based decision-making.35–39,47–49 Effective governance, therefore, requires adherence to ethical and legal frameworks with the implementation of comprehensive ethical safeguards, explainability and transparency in model development, robust validation and reproducibility of AI outputs, and mechanisms to detect and mitigate bias or misinterpretation.47–49
From a broader societal and ethical perspective, the implementation and governance of AI should consider ABS50 obligations under the Nagoya Protocol,2 particularly in relation to DSI (Section 4.4 and 4.4.1). AI can support ABS compliance by improving traceability and documentation of biological resources. However, unresolved legal ambiguities risk enabling the circumvention of fair and equitable benefit sharing if governance frameworks are not updated, reinforcing the need for strategic policy decisions on this subject.47–49
2.5. Applying RRI to new genomic techniques
NGTs are increasingly applied in natural products R&D&I, transforming the discovery, optimization, and production of bioactive secondary metabolites and enzymes from plants, animals, or microorganisms.51,52NGTs such as gene editing tools, i.e., Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR)/CRISPR-associated protein 9 (Cas9), Transcription Activator-Like Effector Nucleases (TALENs), base editing), cisgenesis, intragenesis, RNA-directed DNA methylation, and targeted mutagenesis allow targeted changes, often without introducing foreign DNA, raising novel regulatory, ethical, and societal questions.53,54
By precisely targeting biosynthetic genes, NGTs can enhance natural product yields, reduce by-products, activate silent metabolic pathways, or introduce novel functionalities. This allows the production of high-value compounds at industrial scale while reducing environmental pressures due to wild harvesting.55,56 Furthermore, NGTs have the potential to enhance crop yield, disease resistance, climate resilience, and animal breeding, while also posing scientific, environmental, and ethical challenges.53,57,58
NGTs intersect with the Cartagena Protocol on Biosafety (Sections 4 and 4.1),59 depending on whether they contain novel genetic material or traits not achievable through conventional breeding. Divergent national interpretations exist, with some countries excluding NGTs that do not introduce foreign DNA from Living Modified Organisms (LMOs) regulation, while others regulate them as LMOs, creating variability in compliance and oversight.60
The regulation and application of NGTs in the EU serve as a critical test case for implementing RRI principles, strengthening interactions between social and natural sciences is essential to support more democratic and inclusive governance of NGTs.61
Assessing NGTs solely on whether they are “safe enough” is insufficient to achieve public trust. Trustworthy governance requires transparency, inclusiveness, attention to ethical, socio-economic, and environmental dimensions, and recognition of both cognitive and emotional aspects of trust.61 NGTs governance must anticipate long-term impacts, integrate reflexivity, and ensure meaningful public engagement to comply with RRI principles.62
The EU is currently advancing a new regulatory framework for plants obtained through NGTs.63 The proposed legislation introduces a two-tier regulatory system: NGT-1 plants, whose genetic modifications could also occur naturally or through conventional breeding, would undergo a simplified verification process and be largely exempt from the stringent requirements of existing GMOs legislation, while NGT-2 plants would remain subject to full GMOs risk assessment, authorisation, traceability and labelling rules. The framework aims to maintain high safety standards while reducing administrative burdens and enabling innovation in plant breeding, particularly for traits supporting climate resilience and sustainable agriculture. At the same time, the legislation introduces transparency measures, oversight of IP implications, and continued restrictions in areas such as organic production. The proposal is currently undergoing the EU legislative process and, once adopted, is expected to reshape the governance of genome-edited plants in Europe by balancing innovation, safety, and regulatory oversight.63 The European Food Safety Authority (EFSA) regulates NGTs for food and animal feed in the EU.48
In parallel with these regulatory developments, the DETECTIVE Horizon Europe project supports European preparedness for the implementation and enforcement of the emerging NGTs framework.64 DETECTIVE focuses on strengthening detection, traceability and interpretative capacities across the EU by engaging enforcement authorities, reference laboratories, researchers and agri-food actors in a structured empowerment process. The project is developing both analytical and non-analytical approaches for the detection and traceability of NGT-derived products, addressing challenges related not only to laboratory detection methods but also to documentation, data exchange and regulatory interpretation across the agri-food value chain. Central to this effort is the application of RRI through the RRI Roadmap©™ methodology,24 which provides a systematic framework for anticipation, stakeholder inclusion, reflexive dialogue and responsive action planning. Through scenario analysis, systems mapping and co-creation workshops, DETECTIVE identified capability gaps and co-designed targeted empowerment activities aimed at strengthening European laboratories, regulatory authorities and agri-food stakeholders. In doing so, the project contributes to building coordinated European capabilities that support the effective, transparent and responsible governance of NGT-derived products under the evolving EU regulatory framework.24,64
Ethical concerns in this area include respect for consumer autonomy, the right to choose, and the implications for food sovereignty and biodiversity. The EU's cautious stance reflects broader societal values that prioritize precaution and inclusivity in innovation governance. Aligning NGTs development with RRI can help navigate these complexities by fostering dialogue and encouraging responsible innovation, while maintaining regulatory legitimacy.65–67
2.6. Sustainability and environmental responsibility
Sustainable harvesting methods and conservation are fundamental to ethical natural product R&D&I, which must promote the sustainable use and preservation of natural resources.68,69To anticipate and mitigate ecological impacts, Environmental Impact Assessments (EIAs) are designed to evaluate potential harm to ecosystems before resource extraction begins, and adaptive management strategies enable the implementation of measures, such as collection limits, protection of reproductive individuals, creation of protected buffer zones, and ongoing ecological monitoring.70,71
Given that overharvesting and environmental mismanagement drive biodiversity loss, bioprospecting should be guided by EIAs and the use of extraction methods that minimize ecological disruption and avoid long-term damage to the organisms or their habitats.69,72
Sustainable harvesting practices include rotational collection, habitat conservation, and non-destructive sampling techniques, such as compound production scale up through cell culture, tissue culture, synthesis, semisynthesis, microbial expression, or fermentation, rather than continued harvesting from wild populations.73
Alternatively, harvesting plant leaves rather than bark or roots enables regeneration and supports the long-term viability of the plant. The anticancer drug paclitaxel (marketed as Taxol®) exemplifies how sustainable sourcing strategies can be guided by the localization of bioactive compounds within the plant and the use of alternative production pathways, such as semisynthesis from leaf-derived precursors, to minimize environmental impact while ensuring a reliable supply. Taxol was originally isolated from Taxus brevifolia, but this required harvesting large amounts of slow-growing trees, leading to ecological and supply concerns.74 To address this, a precursor, 10-deacetylbaccatin III, was extracted from the leaves of Taxus baccata, and then chemically converted into Taxol via semisynthesis.75,76 Additionally, plant cell culture techniques allow the production of Taxol in bioreactors by culturing Taxus baccata cells under controlled conditions, which avoids harvesting wild trees entirely.77 These approaches ensure both environmental protection and consistent pharmaceutical production.
Nowadays, for most of commercial products derived from GR, the above-mentioned strategies constitute the primary means of sustainable production.
In marine environments, bycatch reduction strategies and habitat-sensitive collection methods are now considered best practices, reflecting a maturation of sustainability standards in marine bioprospecting.78
Evidence indicates that over-collection is now uncommon, as most bioprospecting and MGR collections are expected to be carried out using controlled and sustainable practices that result in minimal bycatch or ecological disruption; however, this was not always the case. A frequently cited example is trabectedin (ET-743, marketed as Yondelis®), a structurally complex marine-derived anticancer alkaloid originally isolated in extremely low yield (≈1–2 g per tonne of tunicate biomass) from the Caribbean tunicate Ecteinascidia turbinata, a scarcity that required processing tons of biomass for early research and clinical development, making direct extraction unsustainable, prompting aquaculture and later semi-synthetic production methods to ensure supply.79,80 The compound is approved for treating soft tissue sarcoma and ovarian cancer and was initially supplied through a semisynthesis process (e.g., from cyanosafracin B precursors though fermentation of Pseudomonas fluorescens A2-2) after total synthesis proved impractical for commercial production.81 Genomic and metagenomic analyses have revealed that trabectedin's biosynthesis in nature is attributable to a symbiotic bacterium, Candidatus Endoecteinascidia frumentensis, within the tunicate, highlighting the role of microbial symbionts in natural product biosynthesis and informing strategies for sustainable production.82,83
The Plant Treaty,84 formally known as the International Treaty on Plant Genetic Resources for Food and Agriculture (ITPGRFA), is a global agreement adopted by the Food and Agriculture Organization (FAO) in 2001 to ensure the conservation, sustainable use, and equitable sharing of benefits arising from plant GR. It covers plant GR important for food security and agriculture and facilitates access to a Multilateral System (MLS) of genetic materials for research, breeding, and training.85 Users of these resources are required to comply with benefit-sharing obligations, which may include monetary contributions, technology transfer, capacity building, and sharing of research results. The treaty also recognizes farmers' rights, including the protection of TK, support for seed safe keeping and local innovation, and participation in decision-making to ensure equitable benefits. In addition, the Plant Treaty encourages countries to conserve and sustainably use plant GR both in situ on farms and ex situ in gene banks, providing a framework for international cooperation that enhances food security, agricultural resilience, and biodiversity conservation. In essence, the Plant Treaty establishes a public, legally binding framework that balances openness and accessibility of plant GR with fair recognition, benefit-sharing, and conservation responsibilities.85
In November 2025, the governing Body of the ITPGRFA aimed to improve the MLS for Plant GR, focusing on revising the Standard Material Transfer Agreement (SMTA).86 Negotiations stalled in 2019 and were relaunched in 2022, making SMTA revision a key political priority. Only a clearly drafted and enforceable SMTA can secure future user payments and support the Treaty's objectives.86
Additionally, the EU Taxonomy Regulation,87 active since July 2020, identifies sustainable economic activities, including those affecting the marine environment, which must contribute to objectives such as climate action, circular economy, and marine conservation. Conservation scientists and managers should evaluate not only the effectiveness and efficiency of conservation efforts but also their justice, which offers a broader and more inclusive framework than equity for assessing governance outcomes.88 By 2050, the EU commits to large-scale ecosystem restoration, supported by a new European Biodiversity Framework and the new Nature Restoration Law, emphasizing shared responsibility and participatory governance, with non-governmental organization (NGOs) and judicial systems ensuring enforcement.89
Nevertheless, bioprospecting's practical impact on conservation has been limited due to weak and inconsistent economic incentives from the private sector and the scarcity of ideal market conditions that theoretical models suggest could support conservation, including high drug development costs, high royalties, and focus on major diseases.90 For example, limited attention to diseases prevalent in the Global South reduces local benefits.91 Market uncertainties, long development timelines, and pharmaceutical companies' strategic choices further diminish its conservation value, while global trade pressures often make land conversion more profitable than preservation.90 Consequently, bioprospecting activities and economic returns remain below potential, as seen in Panama, where benefits have been modest despite rich biodiversity. However, Panama's national bioprospecting program demonstrates how long-term ecological research, local capacity building, and investment in infrastructure can yield immediate and future conservation gains.92 Nevertheless, while initiatives such as the Panama bioprospecting programme have demonstrated success, they remain vulnerable to political and institutional changes, as illustrated by the closure of Costa Rica's INBio in the mid-2010s. This exemplifies the need for sustainable governance structures, long-term funding, and institutional resilience to ensure continuity of bioprospecting and benefit-sharing programmes.
Bioprospecting can contribute to conservation, but it cannot function as a standalone mechanism; broader approaches, including public funding, policy support, and international collaboration, are needed to ensure effective biodiversity protection.92,93
2.7. Traditional knowledge and biodiversity conservation
By its nature, TK is an intricate, holistic, and evolving knowledge system developed, improved and sustained across generations and often orally transmitted, holding deep cultural and spiritual significance within IPLCs, and forming part of their heritage and identity.94 It is deeply contextualized, meaning its relevance and application are specific to the local ecological and cultural setting. TK is a collective knowledge repository that allows communities to track and respond to constant environmental and societal changes, and to adapt to them in a dynamic and sustainable way.According to WIPO, TK encompasses “knowledge, know-how, skills, and practices that are developed, sustained and passed on from generation to generation within a community, often forming part of its cultural or spiritual identity”.95 TK is a broad, overarching term for the knowledge, beliefs, innovations, and practices of IPLCs globally. Consequently, it can take various forms such as stories, songs, folklore, proverbs, cultural values, beliefs, rituals, community laws, local languages, and practices. To provide clarity and standardization, the WIPO Intergovernmental Committee on Intellectual Property and Genetic Resources, Traditional Knowledge and Folklore (GRTKF) adopted a list in 2010 that offers a concise technical explanation of terms.95
Modern society is often most interested in aspects of TK related to agricultural practices, including the development of new animal breeds and plant species or varieties, as well as the utilization of natural resources, their parts, or their products in traditional treatments for various conditions and illnesses. This approach has its roots in a historical period marked by intense and largely unregulated exploitation of local biodiversity, during which bioresource-rich communities were denied equitable benefit-sharing and recognition of their rights. The economically driven value system imposed during that era disrupted traditional beliefs and customs, often resulting in the depletion, or in extreme cases, the extinction, of local species.96 There are many historical examples illustrating this pattern. One such case is the exploitation of sea turtles in Fiji.96 For millennia, sea turtles have been integral part of the spiritual and secular life of iTaukei, Fijian IPLCs. Perceived as sacred animals, their hunting was regulated by customary laws and rituals.97 Together with associated taboos and beliefs, this system ensured protection and survival of sea turtles for centuries. However, the collapse of sandalwood and sea cucumber industries in late 19th century initiated the trade of tortoise shell.97 The intense demand caused the shift from the iTaukei's custom-based hunting system to unregulated harvesting, driving the depletion of local sea turtle populations.97
IPLCs, as holders of generational knowledge and experience about biodiversity and ecosystem services, can be important agents in overcoming “the imbalances in relationships between humans and nature”. The historical observations of IPLCs regarding changes in local species populations can serve as a valuable data source for estimating and reporting on various goals and targets of the Kunming-Montreal Global Biodiversity Monitoring Framework.98–100 For example, such knowledge about the census sizes of local populations can provide data for calculating the headline indicator on genetic diversity, serving as a basis for biodiversity monitoring and management.100
2.8. Rethinking research programs for embedding RRI and ethics in the bioeconomy
Confraria et al. (2024) investigated how well national research agendas aligned with SDGs, revealing a global misalignment between national research priorities and countries' most pressing SDGs challenges, particularly in low and lower-middle income countries.101 Regions facing the greatest SDG-related needs, contributed to less than 10% of SDG-focused research, evaluated by the number of publications explicitly addressing SDG-related topics.101 Positive alignment between national research priorities and SDGs challenges were evident only in four areas: SDG1 (No poverty), SDG2 (Zero hunger), SDG6 (Clean water and sanitation), and SDG9 (Industry, innovation, and infrastructure). Particularly striking was the case of SDG12 (Responsible consumption and production), where high-income countries, despite having the most unsustainable consumption patterns, were not significantly focused on related research.101 This misalignment has direct implications for natural product R&D&I, bioprospection, and the bioeconomy. Most of the world's biodiversity, and thus the greatest potential for novel natural products, lies within underfunded, lower-income countries. However, because these countries receive a disproportionately small share of global research attention and investment, local capacity for natural product discovery is often underdeveloped.101 This is influenced by multiple factors, including local governance, the lack of priority given to the role that research can play in addressing societal needs, and resource constraints, which together shape the opportunities and challenges for advancing natural product R&D&I in these regions. It not only limits opportunities for scientific and economic advancement in these regions but also undermines sustainable development and equitable benefit-sharing, a cornerstone of sustainable bioprospecting.101 Equitable investment in local research can empower communities to lead natural product R&D&I, supporting both economic development and conservation goals.101Natural product R&D&I holds cross-sectorial potential contributing to several SDG, including SDG 1 (No Poverty), SDG 3 (Good Health and Well-Being), SDG 8 (Decent Work and Economic Growth), SDG 9 (Industry, Innovation and Infrastructure), SDG 12 (Responsible Consumption and Production), SDG 13 (Climate Action), SDG 14 (Life Below Water), and SDG 15 (Life on Land). Yet, these were precisely the goals where research misalignment was most pronounced.101 While the SDGs provide a broad global framework, their applicability depends largely on the systematic measurement and evaluation of specific indicators. Moreover, recent policy discourse and academic literature suggest a gradual decline in the prominence of the SDGs, accompanied by a shift towards more regionally focused and operationally defined strategies, particularly within the EU policy landscape. Alignment with EU Bioeconomy Strategy and the EU Biotech Act initiatives entails prioritizing sustainable, circular feedstocks and embedding life-cycle assessments, provenance tracking, and robust biosafety and dual-use controls throughout the natural product R&D&I process.4,5 Early integration of standardized data formats and regulatory templates facilitates rapid compliance with evolving EU requirements, including ABS50 obligations under the Nagoya Protocol and traceability rules, while supporting bioeconomy objectives such as resource efficiency, climate impact reduction, and competitiveness.
Under the EU Biotech Act compliance and progress are monitored by actively tracking updates and consultations, engaging stakeholders throughout the regulatory process, and structuring funding proposals around sustainability, circularity, and societal benefit.5 This proactive approach allows organizations to anticipate regulatory expectations, reduce risks during scale-up, and accelerate market access across the Single Market once the legislation comes into force. Monitoring frameworks must strike a balance between effectively identifying non-compliant actors and minimizing administrative burdens on compliant users.
To institutionalize RRI and ethics in the European bioeconomy and beyond, several strategic actions should be taken, including the development of EU and associated countries-wide ethical guidelines for bio-based innovations integrated into research funding criteria to ensure that projects align with ethical and sustainability standards.
Expanding participatory platforms that include value-chain actors, users, and policymakers will enable more democratic governance and holistic policy design, while cross-sectoral collaboration can enhance the inclusivity and responsiveness of bioeconomy strategies.
If RRI in included from the beginning of the R&D&I activities, then it contributes to the development of inclusive innovation ecosystems involving universities, SMEs, research centres, and industry, supported by regional strategies and regulatory frameworks.25 Its institutional implementation relies on tools, such as scenario planning, participatory workshops, interdisciplinary approaches, and governance mechanisms (e.g., codes of conduct, value-sensitive design) to address grand societal challenges like climate change, zero pollution, and food security. Despite its transformative potential, RRI is often applied retrospectively, particularly within academic institutions, limiting its impact.25 More proactive approaches, integrating early consideration of desirable innovation pathways and broader RRI training, are therefore recommended.25 However, there are recent examples of EU funded projects embedding RRI in R&D&I.
Like with the DETECTIVE project (Section 2.5), the GeneBEcon project “Capturing the potential of Gene editing for a sustainable BioEconomy”, also funded under the Horizon Europe, focused on NGTs-derived products for circular bio-based systems in industrial sectors along value chains and supply chains of biological feedstock.64,102 In both projects, RRI has been embedded in all work-packages, deliverables and milestones through the application of the RRI Roadmap©™ with stakeholder mapping, stakeholder needs identification, real-life scenario discussions with a wide-range of value-chain actors and policy-makers, co-creation and co-agreement activities and embedded feedback loops in all R&D&I tasks ensuring stakeholder co-created results and empowering programmes.24,64,102–104
In a society shaped by rapid scientific and technological change, individuals need the knowledge, skills, and values to make informed decisions about socio-scientific issues and to participate responsibly in research and innovation.105 Several initiatives illustrate how RRI can be integrated in education and professional training. The Higher Education Institutions and Responsible Research and Innovation (HEIRRI) project developed ten RRI training programs for different higher education levels, including a summer school and a massive open online course (MOOC).106 Pilot evaluations increased awareness of RRI principles, and valuable feedback for refining the programs.106
In addition, Responsible Research and Innovation Learning (RRIL) ERASMUS+ project develops learning materials on RRI,107 with a central focus is public engagement, understood as the active involvement of diverse stakeholders, from NGOs and companies to local communities, in shaping research and innovation. Through co-created higher education modules, the project aims to address gaps in RRI training across three domains: economics, social sciences, and energy management.107
Similar approaches should be implemented for the bioeconomy domain, extending education and training in ethical and sustainable bioeconomy practices among researchers, regulators and the general public. Building up bioeconomy literacy across these groups will support informed debate and democratic participation in shaping innovation pathways.
To complement these capacity-building efforts, the establishment of monitoring frameworks to assess the ethical, environmental, and social impacts of bio-based innovations will ensure accountability and continuous improvement. In this context, EIAs should be standardized in Horizon Europe funding evaluations and other EU research programmes.69,72
3. Ethics and RRI in natural product R&D&I and the EC's bioeconomy strategy
As the bioeconomy grows, it brings complex ethical challenges, environmental, social, and governance-related, that require integrating RRI to align technological progress with societal and ethical expectations, addressing dilemmas such as environmental degradation, social disparities, and the use of emerging technologies, while upholding the EC's Bioeconomy Strategy principles of fairness, sustainability, and inclusivity.4,108–110 Equity is another ethical dimension, particularly in ensuring that marginalized groups, such as small-scale farmers, communities in the Global South, or IPLCS can access and benefit from bioeconomy developments.The ethics dimension of RRI calls for careful consideration of who benefits from research and whether innovation aligns with societal needs. In the context of natural product R&D&I, this involves addressing concerns such biopiracy and misappropriation of IPLCs GR and associated TK, while promoting transparent practices and ethical commercialization strategies. It further encourages prioritization of global health challenges, such as neglected tropical diseases, over purely profit-driven innovation.111,112
A central principle of RRI, anticipation, complements this ethical perspective by requiring early consideration potential risks, impacts, and uncertainties. In natural product research, anticipatory reflection assessing the ecological consequences of harvesting biological material, particularly in biodiversity hotspots.113 As discussed in Section 2.6, unsustainable harvesting or poor resource management can lead to environmental degradation and species loss before their potential is fully realized.114 Through anticipatory governance, such environmental and societal risks are integrated into the innovation process from the outsets, rather than being addressed only at the stage of commercialization.
Inclusion and public engagement are core dimensions of RRI and are particularly relevant when research involves IPLCs. Many natural products originate from plants and organisms traditionally used in local healing practices, making ethical engagement essential. This requires recognition of IPLCs' knowledge systems, prior informed consent (PIC), and establishing fair benefit-sharing agreements. The Nagoya Protocol provides a legal framework that reinforces such inclusive practices, closely aligning with RRI's emphasis on co-responsibility, stakeholder involvement, and shared benefits of research processes and outcomes.25
Reflexivity is particularly important in interdisciplinary collaborations, bringing together chemists, biologists, ecologists, anthropologists, oceanographers, and business stakeholders, where differing epistemologies and interests may shape research trajectories, calls for continuously assessment on assumptions, motivations, and potential biases.20 By maintaining critical awareness of power dynamics, funding influences, and institutional pressures, reflexive practice avoid the marginalization of ethical considerations and promotes balanced decision-making.
In this regard, the RRI framework, promoted by the EC, aligns with the principles of co-responsibility and co-ownership in the European Biodiversity Framework.
Transparency and responsibility underpin ethical bioprospecting practices. Researchers and organizations must clearly communicate objectives and maintain openness throughout all stages of the research process. Even when precise outcomes cannot be fully anticipated, intentions and methodologies should be presented honestly and respectfully, supported by complete, accurate, and accessible documentation. Researchers are expected to adhere to established principles of research ethics and scientific integrity, while GR and associated TK remain the property of the IPLCs and nations from which they originate.94 These rights must be respected at every stage of research and commercialization.94
Ethical bioprospecting is therefore grounded in respect, responsibility, sustainability, and fairness, requiring strong ethical oversight, open collaborative engagement with IPLCs, and a firm commitment to safeguarding both cultural heritage and biodiversity.
Fair trade practices complement these ethical commitments by institutionalizing responsible sourcing within the natural products sector, by ensuring that suppliers, particularly IPLCs, small-scale farmers and harvesters, in biodiversity-rich regions, receive fair compensation for their materials and labor.115,116 Certification schemes, such as FairWild and Fair for Life, provide third-party verification of ethical sourcing practices, linking ethical harvesting practices to market value and consumer trust.115,116 In conjunction, these systems support both biodiversity conservation and community livelihoods, creating more sustainable and resilient supply chains.
In this context, ethics provides a framework for conducting research responsibly, ensuring respect for all stakeholders, including IPLCs, and the environment. Adherence to ethical practices enhances the credibility and acceptance of natural product R&D&I within the scientific community and among the public. Furthermore, products developed through ethical means are more likely to gain market access, as consumers awareness increasingly prioritize ethically sourced goods.117,118
Therefore, embedding ethical principles into bioeconomy policies, funding schemes, and innovation pathways is essential to creating a system that not only drives growth but also safeguards the interests of society and the planet.
3.1. Biopiracy and misappropriation issues in bioprospecting
Biopiracy and misappropriation practices undermine the rights of IPLCs and reinforce the historical marginalization of those who have preserved and transmitted TK across generations.69In a comprehensive review, Wynberg (2023) outlines several high-profile cases that have been labelled as biopiracy: Hoodia gordonii, rooibos (Aspalathus linearis), Sceletium tortuosum (kougoed), and Pelargonium sidoides.119,120 One of the most notable examples is the South African rooibos case, which involved negotiations between tea producers and San and Khoi IPLCs representatives.119,120
For over a century, the San and Khoi people used the rooibos plant for its medicinal and nutritional properties. Despite this longstanding TK, over 150 patents were filed by corporations globally, spanning applications in the food, cosmetics, and pharmaceutical industries, without any formal recognition or benefit-sharing with these communities.119 Due to the advocacy efforts of NGOs, the San and Khoi were eventually recognized as rightful knowledge holders. As a result, they now receive 1.5% of the farm gate price of unprocessed rooibos, marking a precedent-setting outcome in the fight for ethical bioprospecting and IPLCs rights.119 Demonstrating that successful outcomes rely on patience, incremental progress, honesty, trust, genuine dialogue, strong legal backing, government leadership, and unity among IPLCs, principles that can be globally applicable.121
Although considered a success case, which resulted in a historic agreement with the inclusion of benefit-sharing provisions, recognition of TK and empowerment of IPLCs,122,123 the rooibos benefit-sharing case highlights two closely linked problems in ABS systems: representation and equitable distribution.
Authority to speak on behalf of IPLCs was formally given to state-recognised bodies, mainly the South African San Council and the National Khoisan Council, because they met legal requirements under national ABS law. However, this legal authority did not necessarily reflect social legitimacy.119 Many small-scale rooibos farmers and community members with long historical connections to rooibos do not recognize these bodies as legitimate representatives of their interests, leaving unresolved questions about who truly holds and can speak for TK.119
The benefit-sharing arrangement also created a paradox for IPLCs farmers. Although the system is meant to reward TK holders, the levy applies across the value chain, meaning that IPLCs and small-scale farmers may indirectly pay into the fund themselves. At the same time, benefits are channelled through centralised structures rather than reaching these farmers directly, resulting in limited and unequal returns. Economic control over production and export largely stayed in the hands of established commercial farmers and companies, limiting transformative benefits for TK holders.119,120 Together, these issues show how ABS frameworks can recognise TK in principle while failing to ensure fair representation and meaningful benefits for those who actually hold and practise that knowledge.119
The Nagoya Protocol requires that IPLCs be consulted and grant permission before their GR and associated TK are accessed. However, these obligations are frequently disregarded in practice, excluding IPLCs from decision-making processes and rarely receiving compensation proportionate to the value derived from their resources or expertise.124
This central issue concerns the commercialization of GR and associated TK without equitable control or benefit for IPLCs. Although legal and policy frameworks such as ABS agreements were eventually put in place, the outcomes reveal persistent structural inequalities.112,119,120,125
TK can streamline drug development by guiding a reverse pharmacology approach, reducing both time and costs. It acts as a valuable “search engine,” enabling more intentional, focused, and safer research on natural products, effectively revitalizing and directing the drug discovery process.126 Even where benefits are offered, they tend to be modest, often falling short of reflecting the cultural, economic, and scientific significance of the knowledge provided, unevenly distributed, and often captured by more organized or politically connected groups. Moreover, conservation outcomes are not guaranteed, despite biodiversity being central to these products.119,120
At the same time, commercializing GR requires substantial investment (€ billions) in R&D&I, clinical trials, regulatory approvals, and marketing, making the value of TK only a portion of the total cost.127 This can create a tension in benefit-sharing arrangements: if TK contributions are undervalued relative to the extensive R&D&I investment, knowledge holders may receive minimal benefits, but if R&D&I costs are ignored in negotiations, commercialization can become economically unviable for developers.
These challenges can be addressed by making representation more inclusive and benefit sharing more targeted. Authority to speak for IPLCs should not rest only with state-recognised bodies but should include local farmers, cooperatives, and community structures, with clear accountability and meaningful participation at the community level. At the same time, benefit-sharing systems should clearly distinguish between who pays and who benefits, ensuring that IPLCs and small-scale farmers are exempt from levies or receive direct, tangible returns.119,120 More broadly, ABS frameworks need to move beyond formal legal compliance toward a justice-oriented approach that recognises historical inequalities and ensures that those who hold and practise TK genuinely benefit.119,120
To create an effective legal framework, policymakers must understand the practical realities of scientific research, including its feasibility, costs, commercialization levels, and existing practices, especially regarding non-monetary benefit-sharing.128
Proposed solutions include capacity-building for communities, mandatory TK disclosure in patents, and hybrid benefit-sharing models, like community trusts or joint ventures, that recognize both the contributions of knowledge holders and the costs of development, aiming for more equitable and sustainable ABS outcomes.
Biopiracy remains a significant global problem, while successful benefit-sharing agreements are still rare due to a combination of structural, legal, economic, and social challenges making equitable outcomes difficult to achieve. Legal frameworks are complex and unevenly enforced, with many countries lacking the capacity to monitor GR use or ensure compliance, sanctions for violations are often minimal, leaving gaps for misuse.129,130
Defining legitimate knowledge holders is difficult because IPLCs are diverse, and state-recognized institutions may not represent all interests, complicating consent processes.119 ABS fails to engage with the historical and political complexities and to address deeper power structures related to state authority, capital, race, patriarchy, land, and identity.120 Rather than transforming these inequalities, ABS can legitimize them by treating benefit sharing as a technical, apolitical compliance exercise, leaving underlying power relations and economic disparities largely unchanged.120
Economic power and global IP system imbalances favour corporations, while small-scale resource holders often lack bargaining power, legal knowledge, or resources, leading to unequal or ineffective agreements.119,131 When companies or other entities exploit IPLCs resources without authorization, it becomes extremely difficult for affected communities to understand the full scope of the exploitation or to pursue legal compensation. Large multinational corporations typically have access to significant legal and financial resources, including patent attorneys and international representation. In contrast, many biodiversity-rich countries, and the IPLCs within them, lack the institutional capacity, funding, or legal infrastructure to effectively monitor, regulate, or protect their biological heritage.119 Moreover, high administrative and transaction costs, mistrust from past exploitation, and difficulties integrating TK into formal legal and economic systems further hinder success.132
Biopiracy prevalence is difficult to quantify, as many cases are informal, unreported, or occur in legal grey areas, but ongoing misappropriation of GR is still observed, with examples including Neem, Turmeric, Hoodia, Basmati rice, among others (see Section 6 for more examples).133
Biopiracy cases can be deliberate and driven by profit motives, while others arise from unintentional violations, often linked to ignorance of, or misinterpretation of, complex ABS regulations, further compounded by inconsistent national implementation of the Nagoya Protocol or human rights regimes, weak compliance systems, and monitoring, enforcement, accessibility, and equity gaps.119,120
International and national laws on access to GR and associated TK and benefit-sharing should be reassessed to better support innovation while also recognizing and protecting IPLCs rights to use and benefit from these resources.134 ABS systems, though important, require deeper structural reform to achieve genuine equity and conservation objectives.119
Addressing biopiracy requires not only legal mechanisms but also structural reforms in IP systems, meaningful involvement of IPLCs, and global cooperation to uphold biodiversity justice.
3.2. European and national codes of conduct, guidelines, and legal frameworks for research ethics and integrity
Codes of ethics are often shaped by cultural heritage and tailored to the unique aspects of local contexts. At the same time, it is crucial to balance the universal dimension of moral values with their situational nature.In this review, we reference the European Code of Conduct for Research Integrity (ECCRI),135 several EU codes and guidelines on research ethics and integrity are of primary interest to several key European organizations, including the All European Academies (ALLEA),136 the European Network of Research Integrity Offices (ENRIO),137 the European Network for Research Ethics and Integrity (ENERI),138 and the European Network of Research Ethics Committees (EUREC),139 which bring together existing national Research Ethics Committees and associations at the European level.
To further strengthen these efforts, several EU-funded consortia have been established. For example, the ENTIRE project140 seeks to map normative frameworks governing ethics and research integrity. The currently active Pro-ethics initiative141 collaborates with research and innovation funding organizations across Europe to explore ethical methods for involving citizens and stakeholders in research and innovation processes. Another major initiative, the Path2Integrity project,142 aims to foster a culture of research integrity by developing learning paths and educational units tailored to researchers and students at various levels. Additionally, an interactive online portal for academic integrity has recently been launched to support individuals who have experienced individual or institutional malpractice or unethical behaviour, such as plagiarism, data manipulation, publication misconduct, mentorship abuse, or other breaches of integrity.143
Numerous codes, guidelines, and legal instruments related to research ethics and integrity are available at the European level, reflecting a growing commitment to responsible and ethical research practice. An overview of major European and international research ethics frameworks, outlining their scope, principles, target audiences, and legal status is presented in Table S1. It includes both non-binding guidelines, focused on research integrity, equitable partnerships, dual-use risks, and ethics self-assessment, and legally binding EU directives regulating animal welfare and biotechnological innovation. Together, these instruments demonstrate how European research governance integrates core ethical values with specific regulatory requirements, addressing researchers, institutions, funders, and industry stakeholders.
Several European countries have also developed their own national codes of conduct and research ethics or integrity guidelines.144 The national and institutional research ethics and integrity frameworks across various European countries are outlined in Table S2. It shows that most have established formal codes or guidelines promoting good scientific practice, responsible conduct, and misconduct prevention. While some operate as national frameworks, others are institutional policies, reflecting a shared European commitment to safeguarding research integrity through structured governance mechanisms.
These research ethics guidelines represent structured interpretations of widely recognized ethical norms in science, guiding researchers and students in conducting good and responsible research and are intended to support ethical reflection and decision-making in practice.
The ECCRI sets a higher standard for integrity, openness, and accountability in modern research. It reinforces core principles such as reliability, honesty, respect, and accountability, but expands them to address today's research environment.135 Researchers are now expected to design studies more rigorously, document their work transparently, and manage data responsibly in line with Findable, Accessible, Interoperable, Reusable (FAIR) principles.135 The ECCRI places stronger emphasis on open and reproducible research, clearer authorship practices, and responsible supervision and mentoring. It also broadens the definition of unacceptable practices beyond fabrication, falsification, and plagiarism to include a wider range of questionable research behaviours.135
Sector-specific guidelines further align Norway with European governance approaches. For example, medical and health research follows frameworks including the Declaration of Helsinki, complementing EU clinical trials regulations and health data initiatives. Science and technology research is guided by the Committee for Research Ethics in Science and Technology (NENT) standards and animal ethics protocols, aligning with EU directives on AI, robotics, and biotechnology.146 Social sciences, humanities, law, and theology operate under the National Committee for Research Ethics in the Social Sciences and the Humanities (NESH) guidelines and internet research ethics guidance, supporting ethical oversight in digital, cultural, and socio-legal research.147
Norway's independent National Research Ethics Committees, covering all research domains and including bodies focused on medical ethics, science and technology, social sciences, research misconduct, and human remain, provide a transparent, accountable, and interdisciplinary governance system.
Norway's comprehensive research ethics framework has long influenced European ethics initiatives, with projects such as SATORI,148 recognizing its robustness and broad applicability across cultural contexts. This influence is increasingly relevant as the EU develops new policy frameworks, including the European Research Area (ERA) Policy Agenda 2025–2027, the EU Artificial Intelligence Act, the EU Data Governance Act, and the European Strategy for RRI, aiming to ensure research and innovation uphold ethical integrity, social responsibility, transparency, and public trust principles, central to Norway's approach.23,113,149,150
These policies collectively introduce a new research environment in Europe where researchers are expected to work more openly, ethically, and collaboratively.23,113,149,150 The updated European Research Area strengthens cross-border cooperation, OS, and mission-driven research aligned with major societal challenges. The AI Act adds strict requirements for transparency, data quality, and ethical safeguards in AI-related research. The Data Governance Act expands access to public-sector and altruistically shared data while imposing clearer rules for responsible data use. The European Strategy for RRI reinforces the need for public engagement, ethical reflection, and alignment of research with societal values. Together, these policies shift research practice toward greater accountability, openness, and societal relevance.23,113,149,150
A foundational element of research ethics in the EU is the requirement for informed consent in the collection and processing of personal data, reflecting respect for individual autonomy aligning closely with the EU's General Data Protection Regulation (GDRP) and Data Governance Act.150,151 Researchers must combine expanded data opportunities with higher standards of privacy, security, and accountability.150,151 The Data Governance Act and the GDPR together create a research environment with broader access to valuable data but also stronger obligations for responsible data use.150,151 The GDPR reinforces strict rules for processing personal data, demanding clear legal bases, strong safeguards for sensitive information, and full respect for individuals' rights.151 More broadly, EU guidelines frame research ethics as encompassing scientific integrity alongside responsibilities toward participants, communities, and society, defining ethics as the values, norms, and institutional arrangements that regulate scientific activity.146,147
As the EU seeks to harmonize ethics oversight across member states, Norway's integrated model, combining normative guidance, institutional accountability, and sector-specific frameworks, offers a proven blueprint for embedding ethics into the European Research and Innovation ecosystem.
4. The Nagoya and Cartagena Protocols and other legal frameworks
The governance of biodiversity, GR, associated TK, natural products R&D&I, bioprospection, biosafety, and blue biotechnology is grounded in several key international legal instruments. Chief among these are the Nagoya Protocol and the Cartagena Protocol (addressed in Section 4.1),2,59 which function under the umbrella of the CBD, Fig. 1.1 | ||
| Fig. 1 Nagoya and Cartagena Protocols governance of biodiversity, GR and associated TK, and biosafety in biotechnology. | ||
CBD was established in response to the rapid decline of biodiversity and habitat degradation, challenges that are further intensified by climate change and require urgent action to reverse this loss.152
The Nagoya Protocol was adopted in 2010 and entered into force in 2014 to ensure the fair and equitable sharing of benefits arising from the utilization of GR and associated TK, and to incentivize conservation and sustainable use, supporting economic growth, human well-being and the bioeconomy, which are vital for preserving biodiversity, sustainable development, protecting global climate and IPLCs rights.50 Nagoya Protocol and CBD both recognize the sovereign rights of countries over their GR and associated TK.50
By July 2025, 142 countries, including UN member states and the EU, had ratified the Nagoya Protocol.2,153 The EU played a pivotal role in the negotiations, aligning the Nagoya Protocol with its Green Deal and biodiversity strategies, especially through the Strategic Plan for Biodiversity 2011–2020.154 Since the USA is not a party to the CBD, the EU represents about half of the regulated utilization of GR and TK, making it a key player along with Brazil, Norway, and several African countries in finalizing this Protocol.154 Several countries still face challenges implementing the CBD and Nagoya Protocol goals into their national governance and regulatory frameworks.155
At the heart of the Nagoya Protocol is the establishment of ABS mechanisms. These are contractual arrangements that outline the terms under which GR and TK may be accessed and how resulting benefits, monetary or non-monetary, will be shared. The ABS Clearing-House (ABSCH) serves as a central platform to facilitate transparency and data sharing on ABS activities, enabling users to access information on legal requirements, and support benefit-sharing compliance, though practical implementation can vary across countries.50
Nagoya Protocol builds upon Article 15 of the CBD, stipulating that access to GR and associated TK must be based on PIC and subject to Mutually Agreed Terms (MAT), Fig. 1. Contracting Parties must enforce regulations ensuring GR and associated TK is used within their jurisdiction are accessed under PIC, MAT and legal permits such as Material Accession Agreement (MAA), as required by the provider country. They must also establish cooperation mechanisms for addressing violations, encourage contract-based dispute resolution, enable legal recourse, and monitor GR and TK use through effective checkpoints across all stages of the value chain, from research to commercialisation.
Obtaining PIC from IPLCs requires fairness, transparency, and respect for cultural values, ensuring that consent is freely given, fully understood, and revocable at any time. True ethical engagement extends beyond formal agreements to include effective communication, cultural competence, and recognition of IPLCs rights, local practices and worldviews. In practice, political, economic, and historical inequalities often hinder meaningful participation, creating a gap between legal frameworks and their real-world application.119 Therefore, genuinely ethical consent processes demand more than legal compliance, they require long-term relationship-building, cultural humility, and a sustained commitment to justice and equity throughout all stages of research or bioprospecting.
Countries, their R&D&I community and collaborators, which are not parties to the Nagoya Protocol, such as the USA, Canada, Australia, and Russia, among others, must still comply with the Nagoya Protocol and all the provider countries' regulations, when accessing extranational GR and TK, as the Protocol allows each party to establish its own domestic rules.156
Becoming a Party to the Nagoya Protocol offers countries legal clarity, economic opportunities, and stronger environmental protection. It also enhances international cooperation, access to funding and capacity-building, and influence in global biodiversity policy. Therefore, countries that adopt an informed, cooperative, and efficient approach to implementing the Nagoya Protocol are likely to gain a significant competitive advantage in science, the bioeconomy, and future development.157
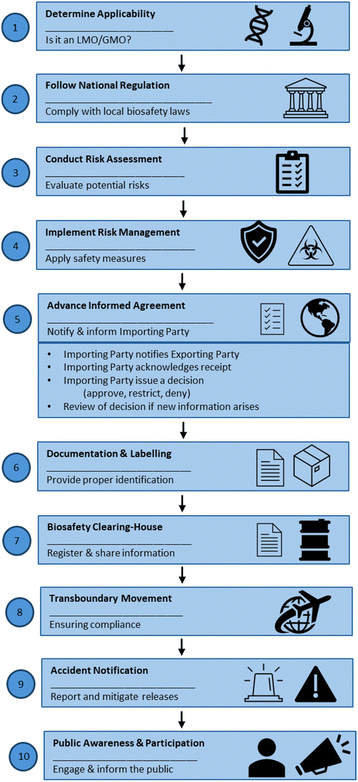
4.1. Cartagena Protocol compliance workflow
The Cartagena Protocol on Biosafety, adopted on 29 January 2000 and in force since 11 September 2003, is an international agreement under the CBD that regulates the handling, transboundary movement, and use of LMOs.59 It emphasizes the precautionary principle, risk assessment, and the sovereign right of countries to make informed decisions regarding LMOs that may affect biodiversity or human health. As of February 2026, 173 Parties have ratified the Cartagena Protocol.59 A step-by-step workflow outlining key biosafety procedures for handling LMOs or GMOs complying with the Cartagena Protocol is presented in Fig. 2.59Compliance with the Cartagena Protocol requires adherence to national biosafety regulations, since the protocol is implemented through domestic legislation.59 Researchers must therefore follow national GMOs laws, obtain approvals from institutional biosafety committees when required, and comply with relevant ethical and environmental authorization procedures. Handling LMOs follows risk assessment, containment, and documentation procedures to minimize impacts on biodiversity and human health.59 This includes determining whether research falls under the protocol, conducting scientific risk assessments, implementing appropriate risk management measures, obtaining approvals for transboundary movement, including the Advance Informed Agreement (AIA) for first-time environmental releases, maintaining traceability and proper documentation, reporting incidents of accidental release, sharing information via the Biosafety Clearing-House, and adhering to national biosafety legislation and institutional approvals, while facilitating public awareness and participation where appropriate, Fig. 2.59
4.2. Nagoya Protocol regulatory gaps and compliance barriers
Implementation of the Nagoya Protocol remains complex and fragmented due to a combination of legal, administrative, and conceptual challenges.Schneider et al. (2022) and (2023) identified several structural barriers that impede the effective implementation of RRI, the Nagoya Protocol, and other bioprospecting-related regulations, design to govern GR and associated TK, and biodiversity protection.25,124 Many researchers perceive these regulations as overly bureaucratic, time-consuming, and burdensome, a perception that contributes directly to widespread non-compliance. This challenge is compounded by a general lack of awareness and education regarding RRI, the Nagoya Protocol, the WIPO GRATK Treaty, and related legal frameworks, as well as persistent scepticism toward involving IPLCs stakeholders in research and innovation processes.25,124
Weak implementation and enforcement mechanisms further exacerbate these issues. In practice, Nagoya Protocol requirements are rarely audited, inconsistently applied, and often poorly enforced, with many signatory parties failing to operationalize monitoring checkpoints.25,124
The absence of a centralized knowledge platform to support bioprospecting-related regulatory processes also undermines compliance, leaving researchers to navigate fragmented and often inaccessible guidance. Standardized procedures remain either unavailable or difficult to obtain across countries that have ratified the Nagoya Protocol, resulting in confusion and inconsistent application.
Political challenges add another layer of complexity. In many contexts, a lack of consensus or political will continues to hinder the effective adoption and execution of the Nagoya Protocol. National focal points (NFP), which are intended to facilitate ABS processes, are frequently unresponsive due to insufficient training or limited resources.25,124
Academic programs in biodiversity and biotechnology equally tend to exclude regulatory and ethical content, leaving future professionals unprepared to engage with these frameworks.25,124
At the same time, biospecimen repositories face chronic underfunding, limiting their capacity to meet international standards. Moreover, research efforts often remain disconnected from societal needs, eroding public trust and missing critical opportunities to influence policy in constructive ways.25,124
Neumann et al. (2018) argue that one of the major obstacles to compliance is the Protocol's vague and overly broad definitions, which leaves key definitions undefined and forces users to rely on interpretation. This lack of legal clarity undermines certainty and, in practice, imposes rigid, property-based regulatory frameworks onto a scientific field that needs dynamic and rapid adaptation.158
Several ambiguous definition examples include, “access”, as posing challenges for user compliance: the CBD and the Nagoya Protocol do not explicitly define access, which can be interpreted either as obtaining ownership of GR or as accessing genetic information for research and development, corresponding to the act of taking and using GR (Art. 6 of the Nagoya Protocol).158
The CBD deliberately crafted expansive and flexible definitions of “genetic resources” and “genetic material” to encompass all biological life under legal and commercial governance. According to CBD, GR includes any material of plant, animal, microbial, or other origin containing functional units of heredity. This broad definition extends beyond DNA or RNA to living or dead organisms, cultivars, propagules, and potentially soil, drill cores, or archaeological samples depending on context, though biologically, functional units of heredity exist only in living cells.158 The transformation of nature into a tradable asset introduced significant political, legal, and scientific tensions, specially the term “functional units of heredity” has been central to debate. When first drafted in 1992, it referred to the role of genetic material in heredity and parent-to-child biological reproduction. Advances in PCR and sequencing technologies highlighted the need for a more flexible and inclusive interpretation. The term “functional” carries a dual meaning: it can refer to molecular constructs within cells or organisms functioning at a micro-organic level, or to the genetic structure and information in DNA sequences that can be digitized and function in new digital forms.158
Moreover, the definition of “material” is similarly broad, potentially including physical components, information, or both, which raises controversy over DSI, which is not comprised in the Nagoya Protocol definitions.158
The authors also raise concerns that, instead of supporting conservation and scientific progress, the lack of legal clarity, poor coordination, and overly complex regulatory frameworks obstruct the transfer of biological materials and data, posing risks to international collaboration and biodiversity research.158
PIC itself is a central challenge. While it is a core requirement under Article 15 of the CBD and Article 6 of the Nagoya Protocol, the Protocol does not provide a clear or uniform definition. This has led to inconsistent and complex national legislations, as seen in Brazil and South Africa, where requirements vary significantly.159 For example, although Korea joined the Nagoya Protocol in 2017 and established national authorities, researchers and companies continue to voice concerns over compliance complexity, especially regarding the PIC process.160
The Nagoya Protocol does not clearly define how sovereignty applies to mobile species or to GR originating from multiple jurisdictions. Both the CBD and the Nagoya Protocol frameworks apply only to GR and associated TK within national jurisdiction, typically within a country's Exclusive Economic Zone (EEZ). However, MGR found in areas beyond national jurisdiction (ABNJ), such as the high seas and extended continental shelf, as defined by the United Nations Convention on the Law of the Sea (UNCLOS) Article 76, are not covered by the Nagoya Protocol or national ABS laws.161
Furthermore, some authors claim that the applicability of the Protocol to materials collected prior to its entry into force, or before the enacted of relevant national legislation, remains ambiguous.162 However, in accordance with Article 28 of the Vienna Convention on the Law of Treaties,163 the Protocol does not apply to such pre-existing materials and governs ABS only for GR and TK accessed after it became effective. This reflects the principle of non-retroactivity in international law, which holds that a treaty does not impose obligations on acts that occurred before its entry into force.131
Additionally, the Nagoya Protocol lacks clear guidance on what constitutes full compliance, particularly in countries without comprehensive implementing legislation, such as Australia.162 Strong status-quo forces in existing institutions meant that, rather than transforming governance structures, it reinforced existing roles. However, this consolidation can foster path-dependent institutional development, even within fragmented governance systems.164
Moreover, the Nagoya Protocol applies equally to commercial and non-commercial research, raising concerns that this uniform approach could stifle collaboration between the Global North and biodiversity-rich countries in the Global South, thereby exacerbating global inequities instead of alleviating them.165
Lessa et al. (2023) also argue that the implementation of the Nagoya Protocol has created substantial bureaucratic barriers for non-commercial scientific research, particularly in biodiversity studies, unintentionally hindering non-commercial science.166 Administrative hurdles, permit delays, and regulatory uncertainty have made routine academic activities, such as sharing museum specimens, genetic materials, and tissue samples across borders, slow, difficult, or in some cases impossible.166,167 The authors note that these burdens often outweigh clearly observable benefits for academic researchers, especially when research is not commercially oriented. Increased time, costs, and coordination challenges have also complicated international collaborations and slowed global biodiversity research. While recognizing the importance of equitable benefit-sharing, Lessa et al. (2023) emphasize the need for policy refinement and proactive collaboration to better support biodiversity research.166
All these barriers highlight the systemic, educational, political, and infrastructural gaps that must be addressed to support fair and ethical use of GR and associated TK under the Nagoya Protocol.
4.3. The ABS process under the Nagoya Protocol: step-by-step workflow
The Nagoya Protocol policy-oriented legal language is often perceived as inaccessible, rendering the text laborious to read and challenging to interpret.2,168 For demystifying regulatory uncertainty and enabling researchers to navigate ABS obligations with confidence and compliance, Schneider et al. (2022) addressed the ABS requirements for GR and associated TK under the Nagoya Protocol by summarizing the process in a structured 10-step workflow, providing a guide to facilitate practical implementation.25,124 Building on this work, herein we further explore the Nagoya Protocol compliance process, clarifying key aspects and incorporating additional information into an 11-steps operational framework for access, use, monitoring, and transfer of GR and associated TK under the Nagoya Protocol,2,168 Fig. 2 and Table S3.For a clearer understanding, it is important to define key terms such as “Party”, “National Competent Authority (NCA)”, “NFP”, “Provider”, and “User”, as these are often sources of interpretive ambiguity.
“Party” is a State that has ratified the Nagoya Protocol and is therefore legally bound to implement its provisions through national legislation.2 It must create the institutional and legal framework that makes ABS work in practice, including designating NFP, NCA, and checkpoint systems for monitoring compliance. Within each Party, the NCA is the body responsible for carrying out the ABS procedures defined by national law.2 This authority receives and evaluates access requests, verifies compliance with PIC and MAT, issues permits, and often serves as the main point of contact for both users and providers. It is the operational arm of the Party's ABS system, translating the country's legal obligations into day-to-day administrative practice. Parties must formally notify the ABS Secretariat of their NFP and NCA, which are then listed in the ABSCH.
“Provider” is the entity that holds or controls the GR and associated TK.2 Depending on national law, this may be a government agency responsible for biodiversity, a research institution or biobank that maintains collections, or an IPLC with recognized rights over GR or associated TK.2 The provider is the actor that grants access, negotiates the MAT, and receives the benefits arising from utilization. Importantly, the Provider is not always the same as the NCA; it may be a community or institution empowered by the State to authorize access and participate directly in benefit-sharing.
“User” is the researcher, institution, or company seeking access to GR and/or associated TK and becomes responsible for complying with all the conditions attached to that access.2
ABS under the Nagoya Protocol relies on the provider party's sovereign consent exercised through its NCA, contractual MAT between Provider and User, user-side MAT compliance and monitoring by user party, and international cooperation supported by the ABSCH.2 The roles and responsibilities for each actor according to the Nagoya protocol are described below and illustrated in Fig. 3 and Table S3,2,168 which together frame our natural products R&D&I workflow for Nagoya Protocol compliance, including:
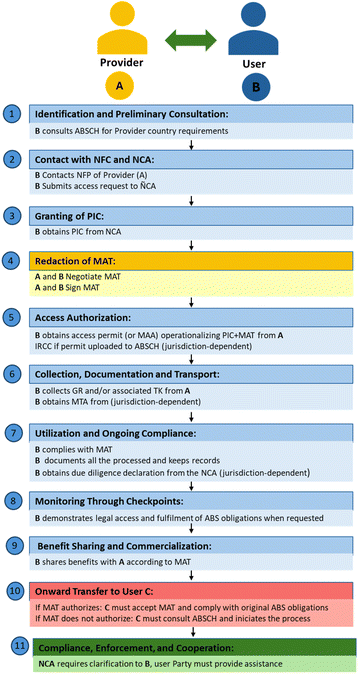 | ||
| Fig. 3 Nagoya Protocol and Access and Benefit-Sharing (ABS) compliance workflow. Adapted from Schneider et al. (2022).25 | ||
(1) Identification and preliminary consultation: the process begins when User (B) identifies GR or associated TK requiring access. Before any collection or transfer, User (B) consults the ABSCH to obtain the provider country's legal, administrative, and procedural requirements.
(2) Contact with NFP and NCA: User (B) contacts the NFP of Provider (A) to clarify access conditions. An access request is then submitted to the NCA, specifying the intended use, research activities, and whether the purpose is commercial or non-commercial.
(3) Granting of PIC: PIC is the access consent of the provider Party, exercised and formalized through its NCA, and where applicable, with the approval and involvement of IPLCs in accordance with domestic law.
(4) Redaction of MAT: following PIC granting, Provider (A) and User (B) negotiate and sign MAT. MAT define conditions of utilization, benefit-sharing obligations, including IP considerations, reporting duties, monitoring requirements, rules for third-party transfers, and procedures for changes in intended use and non-compliance.
(5) Access authorization (MAA, where applicable): Provider (A) issues an additional permit to User (B) at the time of access, which operationalizes PIC and MAT and confirms that all access conditions have been met. In some jurisdictions this is defined as MAA.
If the provider Party uses the international system, the permit is uploaded to the ABSCH and becomes an Internationally Recognized Certificate of Compliance (IRCC), which accompanies the resource throughout its lifecycle. This step is not mandatory, and if not uploaded by NCA the permit is still valid and no IRRC exists.
(6) Collection and documentation: User (B) collects or receives the GR or TK from Provider (A) and maintains complete documentation (e.g. sample number, amount, size, characteristics, GPS location, etc.) to ensure traceability and compliance.
In many provider Parties, a Material Transfer Agreement (MTA) is issued to regulate the physical transfer of samples. It requires adherence to MAT and ensures compliance during use and subsequent distribution. Although widely used, MTA is not mandated by the Protocol.
(7) Utilization and ongoing compliance: once the GR and/or TK are obtained, User (B) must use them strictly within the scope defined in MAT and comply with all reporting obligations, which may include sharing data, returning results, providing updates, or transferring technology.
EU users must formally notify their NCA that they have exercised due diligence and are complying with ABS obligations for the GR use and associated TK.
(8) Monitoring through checkpoints: after the material leaves the provider country, the user country becomes responsible for monitoring compliance through designated checkpoints. These may occur during funding applications, patent filings, journal submissions, product approvals, border controls, or institutional compliance reviews. Users must demonstrate legal access and fulfilment of benefit-sharing obligations at each checkpoint.
(9) Benefit sharing and commercialization: when benefits arise, they are shared with Provider (A) according to MAT. Benefits may include financial payments, joint publications, capacity building, training, technology transfer, or conservation contributions. If research progresses toward commercialization, User (B) must notify the Provider (A) and activate the benefit-sharing provisions, often supported by institutional technology transfer offices.
Provider Party may allocate benefits to IPLCs, biodiversity conservation, or sustainable-use initiatives, completing the benefit-sharing cycle envisioned by the Nagoya Protocol.
(10) Onward transfer to User C: if User (B) intends to transfer the resources to User (C), MAT must explicitly authorize onward transfer. If permitted, User (B) must ensure that User (C) accepts all original obligations and receives documentation proving legal access, including PIC, MAT, MAA, and any IRCC. User (C) then becomes a new user under the Protocol and must comply fully with the original terms. If MAT prohibits or does not explicitly address onward transfer, User (B) is not permitted to transfer the material. User (C) must, therefore, contact Provider A and initiate ABS process.
(11) Compliance, enforcement, and cooperation: if documentation is incomplete, the NCA may request clarification or initiate a compliance investigation. When a provider country suspects non-compliance abroad, it may request assistance from the User Party. Parties must cooperate by exchanging information, verifying documentation, and taking enforcement action when necessary.
Effective benefit sharing depends on clearly defined MAT, transparent PIC procedures, systematic monitoring through checkpoints, and the use of legal permits or IRCC to document lawful access. It is further strengthened by continuous information exchange through the ABSCH and enforcement by NCA that obligations are upheld across borders. Together, these elements create a system in which benefit sharing is properly negotiated, traceable, enforceable, and verifiable throughout the entire chain of utilization.
4.4. Biological material and digital data: legal tensions in the evolving definition of genetic resources and open access era
DSI is a policy term used in global biodiversity governance to describe molecular data derived from GR, although it still lacks a universally accepted definition.169 The Deep-Ocean Stewardship Initiative (DOSI) policy brief highlights that interpretations of DSI range from DNA and RNA sequences to proteins, metabolites, ecological interactions, and even TK.169The Nagoya Protocol primarily focused on physical genetic material and does not explicitly cover DSI. While some countries attempted to fill this gap by incorporating DSI into their national ABS laws, most did not. This omission sparked sustained debate, as genetic data are increasingly stored and used in silico, with several provider countries expressing concern that the unrestricted sharing and use of DSI enables users to circumvent ABS obligations.119,161,170–174 Early discussions emphasized the need for coordinated global governance of DSI as genomics outpaced existing ABS frameworks.175 Because digital information is non-material, infinitely replicable, and globally accessible, it cannot be regulated through traditional ABS mechanisms designed for physical samples.169 Growing fragmentation across UN processes has prompted calls for harmonized DSI benefit-sharing rules.176 These concerns ultimately led Parties to conclude that a separate, multilateral international approach was necessary, Decision 15/9, adopted in 2022 at the 15th Conference of the Parties (COP 15) on CBD, in Montreal, provided the key mandate to formally launch the process on DSI.161,177 At COP 16, held in Cali in 2024 and resumed in Rome in 2025, Parties adopted Decision 16/2 to operationalize the multilateral mechanism established under Decision 15/9. This decision established a global benefit-sharing fund, the “Cali Fund,” into which entities benefiting commercially from DSI are expected to contribute. Resources from the Fund are allocated to support biodiversity conservation and the rights and livelihoods of IPLCs, ensuring fair and equitable sharing of benefits arising from the use of DSI on GR.178,179 Decision 16/2 also called for ongoing work on the mechanism's modalities, including the potential inclusion of additional elements such as products and services, as well as the development of tools and models, such as databases, for making DSI publicly available and accessible.180,181 After COP16 established new DSI obligations, practical guidance emerged to help researchers navigate compliance.182 Subsequent analyses highlighted the implications of these evolving rules for marine natural product research and the need for clearer communication to scientists.161 Most recently, attention has turned to how the new multilateral DSI mechanism could effectively generate biodiversity finance while preserving open data access.183 The modalities of the multilateral mechanism will be further refined and elaborated at COP 17 in October 2026.
The establishment of a multilateral mechanism for benefit-sharing from DSI has significant implications for natural product R&D&I. It reduces the complexity associated with negotiating bilateral ABS agreements for DSI, providing a centralized mechanism for benefit-sharing and lowering legal uncertainty.
The mechanism introduces a formalized, predictable channel for monetary benefit-sharing, which can be integrated into institutional policies, due diligence processes, and corporate governance frameworks, simplifying obligations related to DSI. Researchers and developers, institutions and companies using GR for drug discovery, biotechnology, cosmetics, or other natural product innovations and biotechnological applications, must track the origin and use of DSI in their pipelines to ensure appropriate contributions to the Cali fund as part of their compliance obligations, while still observing national ABS requirements for physical genetic material, even when access to physical genetic material is not involved, requiring a proactive compliance management in research and development programs. Additionally, as intersessional work progresses on modalities, potentially including products, services, and database access, organizations engaged in natural product R&D&I will need to stay informed and adapt internal compliance, reporting, and benefit-sharing procedures to align with evolving international standards.
In natural products R&D&I, OA can democratize scientific knowledge, strengthen trust and collaboration between local communities and international partners, and improve transparency and reproducibility by enabling independent verification of results. OA can reduce duplication of efforts, lower projects costs, and accelerate the translation of discoveries into public benefit.184 Rapid unrestricted access to up-to-date scientific information further stimulates natural products research R&D&I by facilitating knowledge exchange across disciplines and regions. However, open dissemination of data may circumvent bilateral ABS agreements when appropriate governance mechanisms are lacking, potentially undermining fair and equitable benefit-sharing. Unrestricted access to data can also enable commercial actors to use information without proper acknowledgment or contributions to benefit-sharing mechanisms, inadvertently facilitating biopiracy, especially where local stakeholders lack strong legal or institutional protections for their resources.170
These risks are amplified by structural inequalities. OA often benefits well-resourced institutions in high-income countries while marginalizing IPLCs knowledge systems. This imbalance can narrow understandings of biodiversity and obscure local contributions. Although publication outputs from developing countries is increasing, persistent constraints in scientific capacity and research independence continue to challenge equitable ABS.171 Moreover, OA can exacerbate disparities when capacity building is not needs-based, especially in regions facing limited internet connectivity, inadequate infrastructure, or unreliable power supplies.
OA publishing also presents financial and strategic challenges. Article Processing Charges (APC) remain prohibitively high for many researchers in low- and mid-income countries, and early public disclosure of results may hinder IP strategies by limiting opportunities for patent protection if innovations are not secured prior to publication. This can restrict commercialization and proprietary innovation in natural products R&D&I.
Addressing OA limitations requires strong ethical standards, effective enforcement of benefit-sharing agreements, targeted investment in local research infrastructure, and adherence to scientific practices that ensure accurate transparent data use.
OA publications could support the Nagoya Protocol compliance with ABS frameworks, by providing a platform for documenting benefit-sharing statements and demonstrating adherence to PIC and MAT.185 In doing so, scientific journals could acknowledge IPLCs ownership and permissions over GR and TK, reinforcing IPLCs data sovereignty, collaboration, and the fair use of GR and associated TK.185 In this way, OA publications function as monitoring checkpoints that help ensure accurate and transparent data use.
By incorporating RRI principles into bioprospecting strategies, natural product R&D&I and commercialization pathways, researchers and institutions can better align their work with ecological integrity, global equity, and public trust. Achieving societal alignment requires a realistic, adaptive, and participatory approach that recognizes institutional resistance, engages innovators constructively, and adjusts expectations to match real-world innovation dynamics.186
OA genetic databases, DSI databases, have transformed scientific practice by enabling research without physical access to biological materials.177,181,187,188 This shift has generated significant legal, ethical, and policy challenges, particularly concerning fair and equitable benefit-sharing with provider countries and IPLCs, whose TK and stewardship are often linked to the biological resources underlying DSI.189,190 Examples of publicly accessible databases with genetic sequences include, ELIXIR, GenBank, BOLD, and GBIF.170,185,191
In response to these challenges, the CBD Parties have been promoting negotiations to adapt the international ABS regime to DSI.180 Proposed solutions include multilateral monetary contribution mechanisms, improved provenance tracking through standardized metadata, and digital labelling systems such as BioCultural Labels, which enhance transparency, traceability, accountability, and recognition of IPLC authority over genomic data.190,192
Centralized DSI databases support compliance through recording origin and ABS-related metadata, enabling users to identify applicable PIC and MAT obligations even for in silico use. They also facilitate standardized benefit-sharing by linking DSI use to multilateral mechanisms such as the Cali Fund, while supporting monitoring and reporting by providing clear records of DSI use and related contributions.180,190,192 By improving traceability, documentation, and transparency, they reduce legal uncertainty, support adherence to international agreements, and ensure that benefits reach provider countries and IPLCs even in digital research contexts.170,185
As DSI offers unprecedented opportunities for scientific and technological progress, its governance under the CBD requires robust, ethically grounded, and inclusive ABS mechanisms that balance OA with equity, recognize IPLC contributions, and ensure that benefits derived from DSI support biodiversity conservation and sustainable development.
4.5. The role and importance of biorepositories for Nagoya Protocol compliance
Biorepositories or biomaterial repositories (BR) are key biodiversity conservation tools and play a critical role in supporting compliance with the Nagoya Protocol. These are indispensable components of the international ABS ecosystem, by promoting fair global scientific collaborations and serving as central hubs for the collection, storage, documentation, and distribution of GR in accordance with ABS frameworks.193,194Environmental Biospecimen Repositories (EBRs) and biomaterial repositories, often referred to as Users/Depositors, facilitate this process by ensuring that GR, including DNA, proteins, enzymes, and other biological derivatives, are collected and transferred in full compliance with national and international legal obligations.195,196 Biorepositories act as compliance intermediaries, helping to formalize access through agreements such as MAT and legal permits, such as MAA. These instruments not only secure legal clarity for Users and Depositors but also ensure that benefit-sharing obligations are passed on to all downstream users.197
Infrastructures such as online platform biorepositories are increasingly vital in bridging legal and operational gaps, ensuring compliance and fairness. Biorepositories are particularly important in the Blue Biotechnology and marine research sectors, where international collaboration and cross-border sample transfers are common.198 In these cases, biorepositories offer traceability and accountability, especially where GR are accessed from countries with strict ABS regulations or from marine areas under national jurisdiction.199,200
Beyond legal compliance, biorepositories contribute to non-monetary benefit-sharing by enabling data sharing, training, and capacity-building initiatives that benefit provider countries and IPLCs. For instance, OA databases linked to biorepositories can support biodiversity monitoring, conservation planning, and equitable participation in research.184 Furthermore, IPR play a critical role in these ABS mechanisms, impacting much of the research and development involving GR and TK.197
4.6. The Nagoya Protocol and the evolving governance of marine genetic resources – BBNJ treaty
The growing emphasis on Ocean Health has been driven by global and regional frameworks, such as SDG 14 “Life below water”,201 the European Green Deal,202 One Health,203 and the EU Biodiversity Strategy for 2030.204 The EU aims to protect 30% of its marine areas through the expansion of Marine Protected Areas (MPAs), which have demonstrated economic value.205,206The Maritime Spatial Planning (MSP) Directive has led all coastal EU Member States to develop national maritime plans and cooperate across borders. MSP is increasingly seen as a key tool to support the European Green Deal, particularly for sustainable offshore energy, biodiversity protection, and maritime transport, enabling strategic coordination of sea uses while integrating environmental goals, such as those under the Marine Strategy Framework Directive and the EU Biodiversity Strategy.207 Many Member States have already planned for offshore renewables and multi-use maritime zones, especially in the North and Baltic Seas. Regional cooperation and alignment with revised national energy and climate plans are expected to grow. MSP will also help manage cumulative human impacts and support ecosystem-based approaches, guided by EU frameworks and funding, such as the European Maritime, Fisheries and Aquaculture Fund (EMFAF).208,209 While the EU leads globally in MSP, some Member States lag in implementation, prompting infringement procedures. The EC plans further support through digital tools, harmonized data models, stakeholder engagement, and funding instruments. Moreover, a Blue Forum will be launched on April 2026 to facilitate dialogue among MGR users.210
The Nagoya Protocol and ABS obligations apply to MGR and associated TK that fall within a coastal State's national jurisdiction. Under the UNCLOS, this jurisdiction begins at the coastal baseline, defined as the low-water line along the shoreline, and extends outward to encompass the territorial sea, including the EEZ or the continental shelf up to 200 nautical miles (or up to 350 in specific cases).211
Beyond a coastal State's national jurisdiction lie the high seas, an area where no State holds sovereignty and where resources are treated as part of the global commons. This boundary is crucial because the Nagoya Protocol does not apply to MGR in ABNJ.
To address this legal void, the United Nations General Assembly initiated discussions in 2006, ultimately leading in 2023 to the adoption of the Biodiversity Beyond National Jurisdiction (BBNJ) agreement, ratified on 19th September 2025. The BBNJ treaty or High Seas Treaty entered into force on 17th January 2026, establishing a comprehensive governance framework for MGR in ABNJ.
Importantly, the BBNJ Treaty is the first global ABS treaty to meaningfully involve scientists in both its design and implementation, reflecting a broader effort to align policy with scientific practice.161 At United Nations Ocean Conference (UNOC3), participants emphasized the need for coordination between the existing Nagoya Protocol frameworks, the newly established DSI multilateral mechanism under the CBD (see Section 4.4.1) and the BBNJ's ABS system for MGR, positioning the Cali Fund as a key tool to bridge these frameworks.
The BBNJ Treaty creates a global system to ensure the conservation and sustainable use of these resources, while also providing for fair and equitable benefit sharing through a newly established multilateral mechanism, a subject-matter that was also not covered by the UNCLOS.124,128,212,213
BBNJ Treaty instead of focusing on patent disclosure, adopts broader access and benefit-sharing mechanisms covering both physical resources and related DSI, supported by a dedicated ABS committee and benefit-sharing system for monetary and non-monetary benefits.161 The treaty addresses governance gaps in the high seas, moving beyond the bilateral model of the Nagoya Protocol.214 While implementation details are still evolving, BBNJ reflects a wider shift toward extending ABS norms to marine and digital genetic resources that intersect with research, innovation, and commercialization. However, its emphasis on ABJN may leave coastal TK underrepresented, highlighting the need for coordination with other ABS and IP frameworks to avoid regulatory fragmentation.
Recommendations for strengthening implementation include establishing an international body with binding dispute-settlement authority, developing a global monitoring and enforcement system supported by advanced surveillance technologies, and creating regional governance councils to enhance localized cooperation.212 Together, these measures integrate legal enforcement, technological tools, and capacity-building to address governance gaps and promote more equitable participation by developing countries.
A key priority under the BBNJ Treaty is clarifying the jurisdictional, subject matter, and temporal scope of governance, particularly how it interacts with international frameworks responsible for access, utilization, and benefit-sharing of MGR, associated TK, and DSI.215 While ambiguities still remain, practitioners and policymakers can adopt a commonsense approach to implementation.215 Part II of the treaty aims to fill governance gaps by promoting fair and equitable benefit-sharing, capacity building, knowledge generation, technological innovation, and marine technology transfer. The expansive scope agreed in negotiations, covering activities in ABNJ, physical MGR, associated TK, and DSI, and both prospective and retroactive utilization, represents a diplomatic achievement and provides a strong foundation for equity, conservation, and sustainable use of biodiversity in ABNJ.215
Although the final treaty includes limited engagement with IP, it remains central, offering both potential benefits and challenges for equitable sharing under a common heritage of humankind approach.216 IPR can be flexibly applied to support new product supply chains and benefit-sharing, but consensus on the treaty does not automatically reflect a commitment to common heritage principles.216 The absence of a dedicated IP clause may, however, provide a platform for its innovative, fair, and collaborative use in BBNJ Treaty implementation, allowing policymakers, scientists, and industry to balance commercial and societal interests.216
IP can both enable and constrain equitable benefit-sharing, reflecting broader tensions seen in WIPO and Trade-Related Aspects of Intellectual Property Rights (TRIPS) debates. Similar to the WIPO Intergovernmental Committee (IGC)'s long-standing work on GR and associated TK, the BBNJ context confronts the challenge of aligning global IP rules with the collective or common-heritage approach, while navigating the trade-oriented pressures of TRIPS.
Brown and Humphries works illuminate how BBNJ implementation can draw on WIPO-style frameworks and practical tools to operationalize benefit-sharing and mitigate North–South imbalances, paralleling the strategies debated in WIPO's GRATK negotiations.215,216 In this sense, BBNJ represents a marine-focused extension of the same global dialogue about balancing IP, development, and equity that has unfolded across WIPO, TRIPS, and CBD spaces.
Embedding IP in the dialogue around BBNJ, may influence negotiators and policy communities to consider its implications for biodiversity and benefit-sharing, and provides analytical foundations for thinking about how IP might be engaged in the implementation phase, even though the final treaty text does not directly regulate IPR.
These developments indicate growing global momentum toward more equitable, science-informed governance of both MGR and DSI. As the field evolves, ongoing engagement by researchers, legal experts, and policymakers will be essential to ensure both compliance and continued innovation, particularly in high-seas research. The next major milestone is expected in early 2027 with the first BBNJ COP, with prep work concluded in April 2026.
4.7. Towards effective Nagoya Protocol and ABS compliance mechanisms
Schneider et al. (2023) emphasize the need to strengthen awareness and implementation of the Nagoya Protocol and RRI principles among researchers and institutional administrators.Integrating ABS guidance more closely with related legal instruments and ensuring that regional and national information materials are user-friendly would reduce interpretative uncertainty. Centralized knowledge platforms consolidating PIC/MAT requirements, case law, and national regulations could reduce bureaucratic fragmentation and support informed decision-making. In this regard, improved procedural consistency, clearer guidance, and streamlined processes are critical for making implementation more practical and reducing bottlenecks.25,124
Several institutional mechanisms already contribute to this effort. Protocols and application forms made accessible through the ABSCH, simplify initial compliance steps, while research and innovation offices at universities increasingly guide researchers through application procedures. Expanding such support structures through pan-European or international regulatory knowledge platforms would further enhance coherence.25,124
At the structural level, broader stakeholder engagement, inter-ministerial coordination, and harmonized national legislation remain essential. Establishing national ABS coordinating bodies could prevent conflicting interpretations and accelerate permit issuance. Although national ABS frameworks vary considerably, the multilateral architecture of the BBNJ Agreement may provide lessons for improving coherence.124 Thus, complete harmonization is unlikely, as international environmental law deliberately allows interpretative flexibility to accommodate diverse national contexts.159
Despite its status as a legally binding instrument under the CBD, the Nagoya Protocol operates in practice through soft-law dynamics. Monitoring checkpoints implementation, patent disclosure requirements, and reporting obligations strengthen transparency, but depend heavily on domestic implementation capacity and political will.152 Capacity building, institutional support, and technical assistance, particularly in developing countries, remain indispensable for translating formal obligations into effective enforcement.
Norm-shaping actors beyond formal regulators also play an important role. Since 1994, conferences such as the International Symposium on Marine Natural Products and the European Conference on Marine Natural Products have incorporated policy discussions alongside scientific exchange, reinforcing regulatory literacy within research communities.217 Funding agencies and scientific journals increasingly function as compliance gatekeepers by requiring disclosure and monitoring, thereby embedding ABS norms into professional practice. Together with sector-specific best practices developed by networks such as the Consortium of European Taxonomic Facilities (CETAF), Botanic Gardens Conservation International (BGCI), and Global Genome Biodiversity Network (GGBN), these soft governance mechanisms complement formal legal requirements and foster trust, particularly in non-commercial research contexts.158
Technological infrastructures are increasingly presented as solutions to administrative burden. Digitally integrated biomaterial repositories that centralize metadata and incorporate approved PIC or MAT can significantly reduce transaction costs.184,218 The Leibniz Institute DSMZ-German Collection of Microorganisms and Cell Cultures (DSMZ) culture collection illustrates how standardized documentation, due diligence procedures, and traceability systems can facilitate access while maintaining compliance. Blockchain-based tools such as LUCE further promise transparent audit trails for tracking consent and licensing.219 However, concerns regarding scalability, cost-effectiveness, and energy consumption limit their viability as universal compliance solutions.
Debates surrounding DSI reveal deeper governance tensions. The Nagoya Protocol may require targeted reforms to address OA to DSI, synthetic biology, and transboundary GR. Clarifying definitions and revising annexes to address DSI explicitly could reduce interpretative ambiguity. Yet efforts to extend traceability requirements to DSI conflict with CBD Decision 15/9, which recognizes that such traceability is neither practical nor desirable given the non-linear circulation of sequence data. In this context, multilateral monetary mechanisms, such as levies on databases including the International Nucleotide Sequence Database Collaboration (INSDC), have been proposed as more workable alternatives.173,220 These approaches preserve OA while generating revenue for provider countries and IPLCs, potentially reconciling scientific openness with equitable benefit-sharing.
Market dynamics further shape ABS outcomes. Evidence that consumers are more willing to purchase products labelled with TK, particularly when IPLC contributions are clearly communicated, indicates that there are commercial incentives for Nagoya-compliant production models. While such market signals cannot replace legally benefit-sharing mechanisms, they may reinforce compliance through reputational and economic pressures.221
Ultimately, strengthening ABS implementation requires more than incremental procedural adjustments. Streamlined permitting systems, enhanced international cooperation, and sustained technical support must be paired with digitally integrated infrastructure and coherent governance strategies. Emerging paradigms such as One Health and climate-biodiversity integration highlight the need for systemic cross-sectoral solutions. Achieving the CBD's objectives of conservation, sustainable use, and fair and equitable benefit-sharing, therefore, depends on aligning legal frameworks, institutional capacity, technological innovation, and evolving scientific practice within a coordinated and adaptive governance system.158
4.8. Successes and challenges in ABS
Several case studies illustrate both successful implementation and the compliance complexities of the Nagoya Protocol.According to the ABSCH, as of February 2025, there have been 5454 IRCC granted, more than a four-fold increase compared to March 2020 (1222).222 However, only 10 are issued on the subject of TK associated with GR: Benin (1), France (1), Guyana (1), India (3), Kenya (1), Lao People's Democratic Republic (1), and Mexico (2). Only two, both from Mexico, allowed the commercial use. For ABSCH-IRCC-MX-238488-1, the provider was Mujeres y Ambiente, Sociedad de Producción Rural, de Responsabilidad Limitada de Capital Variable and the permit was issued to a Spanish company Provital, S.A., to conduct the scientific collection of forest biological resources, biotechnology modality, for commercial purposes. IRCC states that the subject matter is plant of traditional use in Mexico and associated TK, without disclosing the species name. The second IRCC, ABSCH-IRCC-MX-207343-3, is issued to the USA company BioN2, Inc., but the subject matter is confidential.222
While the total number of IRCC has increased substantially, the issuance of certificates specifically concerning TK associated with GR, particularly those permitting commercial use, remains rare and limited in scope.
The Earth BioGenome Project (EBP) and Basecamp Research illustrate complementary approaches to managing openness, control, and equity in genomic data use: a public, mission-driven model and a commercial, partnership-based one, respectively.223,224 The EBP aims to sequence all eukaryotic species and release genomic data openly to support research, conservation, and policy. By prioritizing OA and aligning with the Nagoya Protocol, it seeks to democratize genomic knowledge, while respecting the rights of provider countries and IPLCs.223 Its success depends on sustained public funding, strong international partnerships, and inclusive participation, particularly from biodiversity-rich countries with limited infrastructure.
Basecamp Research by contrast,225 links genomic sampling directly with AI-driven commercial application.226 It builds proprietary datasets through negotiated partnerships, retaining control over data access and addressing equity through explicit ABS agreements involving investment, capacity building, shared infrastructure, and longer-term economic returns.
Public initiatives like EBP can provide foundational genomic references, while commercial actors like Basecamp can support intensive sampling and data curation.
Australia illustrates the risks of fragmented governance. The absence of a unified ABS framework has led to regulatory gaps in protecting IPLCs rights, with patents suggesting possible misuse of traditional knowledge associated with native plants, such as ‘old man weed’, ‘emu bush’, and ‘Kakadu plum’.227 Ambiguous patent documentation underscores the need for an international “disclosure of origin” requirement, which Australia could support at WIPO.227 Robison and Raven (2017) criticize ongoing colonial-like treatment of IPLCs knowledge as ownerless (res nullius), despite existing legal and cultural frameworks. While joint patents with Aboriginal corporations show some progress, patents alone fail to protect the communal and culturally embedded nature of IPLCs knowledge. This could be mitigated by developing a nationally consistent ABS framework aligned with the CBD and Nagoya Protocol, including PIC, MAT, and stronger legal mechanisms such as mandatory disclosure, sui generis protections, and biocultural protocols to uphold IPLCs rights and self-determination.227
5. Intellectual property rights protection for genetic resources and traditional knowledge
Significant progress has been made in recognizing IPLCs' rights and ensuring equitable benefit-sharing from the use of GR and associated TK.228Early initiatives such as the Declaration of Belém (1988),229 the UNESCO Intangible Heritage Convention, the UN Declaration on the Rights of IPLCs Peoples (UNDRIP, 2007), and the International Covenant on Economic, Social and Cultural Rights (ICESCR) established international norms supporting the principles of free, prior and informed consent (FPIC) and cultural heritage protection.230–232
Core biodiversity governance frameworks including the CBD, the Nagoya Protocol, and ongoing negotiations under the WIPO IGC aim to strengthen legal protection and establish clearer rules for the use of GR and associated TK.233
Established in 2000, the WIPO IGC has worked to develop international legal instruments for the use of GR, TK, and Traditional Cultural Expressions, preventing misappropriation, support fair benefit-sharing and build communities capacity to navigate IP systems. This includes developing tools, such as TK databases, to reduce erroneous or wrongful patents claims.234,235 After nearly 25 years of negotiations, WIPO Member States adopted the first binding Treaty explicitly recognize IPLCs, the WIPO GRATK treaty on 24 May 2024.234 The treaty reflects decades of diplomacy aimed at rebalancing the global IP system within the boarder World Trade Organization (WTO) TRIPS framework.3 While TRIPS advances a trade-driven framework, harmonized IP regime, WIPO's Development Agenda seeks to integrate developmental, cultural, and social objectives.236
WIPO GRATK treaty is the first multilateral IP treaty to explicitly link patents with the origins of GR and associated TK. It mandates that patent applicants disclose the source country and the IPLCs providing the knowledge, aiming to enhance transparency, improve patent quality, and reduce biopiracy.233,237 By integrating TK into patent systems and complementing the Nagoya Protocol (Section 4), this treaty supports PIC, benefit-sharing, and recognition of TK custodians, ensuring that patent offices, researchers, and companies can access verified TK and reduce misappropriation.237
While the treaty may increase administrative responsibilities for patent offices, it also strengthens the ability of IPLCs to contest inappropriate patents and fosters more equitable and transparent innovation.237
Building on this momentum, gap analysis commissioned by the IGC in 2008 and 2018 identified the documenting of TK as a key prerequisite for IPR. In response, several countries and communities established TK databases that may be used as proof of prior art to defeat a patent claim based on TK.238,239 Information on such databases and registries of TK and GR is available through the WIPO, which also developed a toolkit to support TK holders in documenting and safeguarding their knowledge.240,241
Examples such as, India's Biological Diversity Act and Traditional Knowledge Digital Library (TKDL),242,243 along with community-based initiatives like the Honey Bee Network database,244 and Peru's TK registers, Law No. 27811, offer structured mechanisms for conserving and defensively protecting IPLCs,245 while balancing the interests of innovators, communities and the broader public.
WIPO's efforts highlight a central paradox: international IP law is both the cause and the potential remedy of biopiracy and misappropriation.237
The new GRATK treaty foundation is a major step forward, requiring disclosure of GR and TK origins, though its effectiveness depends on national implementation and cannot resolve deeper issues of ownership, especially for intangible, collaboratively generated data.246–249
Within the EU, human rights focused policy including the EU Biodiversity Strategy for 2030 (ref. 204) and the EU Action Plan on Human Rights and Democracy increasingly emphasized respect for IPLCs despite the absence of a single TK-specific legal instrument.250 It upholds the principle of FPIC in all decisions affecting IPLCs, and EU External Action,251 including development cooperation and trade policy, increasingly incorporates IPLCs' rights and TK, supporting initiatives for documentation, protection, and fair and equitable benefit sharing. The EU Arctic Forum also provides a platform for dialogue with IPLCs organizations, including Sámi representatives, on issues such as TK protection.252
Together, these instruments form a multilayered normative framework aimed at preventing biopiracy and misappropriation, safeguarding ownership, and securing fair benefits for IPLCs.
5.1. Global health emergencies and the evolving governance of GR and TK
The World Health Organization (WHO) Pandemic Agreement (adopted in 2025), seeks to strengthen global pandemic preparedness by promoting equitable access to pathogens, vaccines, and the benefits of related research.253,254 It overlaps with the WIPO GRATK treaty by addressing IPR during health emergencies and supporting technology transfer and benefit-sharing for innovations drawing on GR and TK. By encouraging recognition of TK contributions in patents and pathogen-sharing systems, it aims to reduce monopolies and build trust with IPLCs; however, its narrow focus on pandemics risks sidelining TK outside health contexts, and its emphasis on IPR may privilege corporate interests over communal knowledge255 When implemented alongside the WIPO GRATK Treaty, and the Nagoya Protocol, the agreement can facilitate more ethical progress from natural-product discovery through commercialization by accelerating research while ensuring that IPLCs receive recognition and benefits. Together, these instruments signal a global shift toward valuing TK in innovation, but their success depends on coordinated implementation and coherent domestic integration across IP, ABS, and public health frameworks.255The Pathogen Access and Benefit-Sharing (PABS) system is an a separate annex of the WHO pandemic agreement that is being negotiated.256 It is intended to establish a global framework governing how countries and companies share pathogen samples and genetic sequence data with equitable distribution of benefits, particularly in the context of future pandemics.257 These negotiations are highly politically sensitive due to their implications for pharmaceutical industry obligations, national sovereignty, and global equity.258 Nevertheless, once the PABS annex is adopted, the Pandemic Agreement can open for signature and ratification.256
5.2. Intellectual property rights protection tools for GR and associated TK
GR and associated TK hold immense cultural, ecological, and economic value, but often fall outside conventional IP systems, making it difficult to applying communal knowledge to individualistic patent frameworks. To address this gap, national and international legal tools (Table S4), including sui generis and hybrid IP systems, have been developed to ensure recognition, protection and equitable benefit-sharing.259 These mechanisms, include geographical indications (GIs), trademarks, certification marks, defensive protection, like India's TKDL,243,260–263 and patents (Section 5.2.2), each offering partial protection and most effective when used in complementary, context-specific combinations.264The WIPO GRATK Treaty strengthens this landscape by requiring patent applicants to disclose the origin of GR and associated TK and by supporting benefit-sharing through licensing agreements.265,266
However, some TK concerns, particularly sacred or culturally restricted knowledge cannot be disclosed without causing harm, revealing the limits of transparency-based IP and ABS frameworks.267,268 In such cases, confidentiality regimes, customary law, and the right not to disclose TK are essential.253
Taken together, these issues underscore the need to ground IP and biodiversity governance in binding international human rights principles, especially those articulated in the UNDRIP, so that self-determination, cultural integrity, FPIC and community autonomy become operational, not symbolic, within GR and TK governance.
These cases demonstrate that effective governance must include the right to withhold, restrict, or refuse access, not merely regulate disclosure or ensure benefit-sharing.269
Patenting on modified, isolated, and synthetic genes illustrates the complex role of IP in the governance of GR. For example, isolated genes like the BRCA1 and BRCA2 sequences were historically patented by Myriad Genetics, though the USA Supreme Court later ruled that naturally occurring DNA sequences are products of nature and therefore not patentable.272,273 In the EU, Directive 98/44/EC clarifies that biological material may be patentable if produced by means of a technical process, even if it previously occurred in nature.274
While natural gene sequences are not patentable, modified or synthetic genes may qualify. Biotechnological innovations including GMO, modified genes, and synthetic genes or complementary DNA (cDNA), including custom-designed sequences in synthetic biology, CRISPR techniques and diagnostic tools based on genetic data, (e.g. kits identifying disease resistance in plants), remain patentable because they are human-made and non-natural.275
Plant and animal varieties, as well as “essentially biological processes” for their production, fall outside the scope of patentability.274 Nevertheless, patents on cell lines and microbial cultures are granted for novel strains with specific industrial or therapeutic applications.275
The ethical legitimacy of patenting genetic material, especially from humans or endangered species, raises questions about commodification and the moral boundaries of ownership over life forms.275 In response, some countries have introduced legal safeguards requiring disclosure of source in patent applications and ensuring that ABS protocols are followed. Others have established patent exclusions for biological materials deemed part of common heritage or lacking sufficient human intervention to justify proprietary claims.276
Ethical constraints limit protection: inventions deemed contrary to public order or morality, such as human cloning, the use of human embryos for industrial purposes, or interventions that modify the human germline genetic identity, are strictly excluded.274
In the context of natural products R&D&I, naturally occurring substances in their raw form are not patentable, i.e., the discovery of a novel natural product is not patentable on its own.264,270 However, isolated natural products, novel or not, for an innovative use, new methods of extraction or synthesis, new synthetic derivatives, or novel processes, combinations or formulations, may qualify if the applicant can demonstrate sufficient novelty and applicability.264,270
Balancing IPR with ABS remains a persistent ethical and legal challenge. Although IPR stimulates natural products R&D&I, and economic growth by granting exclusive rights that encourage investment, they can also restrict access to essential medicines or TK-based remedies, impacts felt most acutely in low-income settings.277
A systematic review shows that IP provisions, such as data exclusivity, patent-term extensions, and secondary patenting, are associated with higher drug prices, delayed availability of generics, and increased costs for consumers and governments.277 Although TRIPS provides flexibilities through mechanisms like compulsory licensing and parallel importation, these tools remain underused in practice.277
An overview of major patent offices worldwide, listing the responsible authorities and their official websites to facilitate access to patent systems, legal information, and searchable databases, is provided in Table S5. Similarly, patent systems at the international, regional, and national levels are described in Table S6, indicating the corresponding offices or mechanisms and the geographic scope they cover.
The WIPO international patent system is centred around the Patent Cooperation Treaty (PCT), allowing a single patent application to be recognized by over 150 countries, while regional patent office's such as, the European Patent Office (EPO),278,279 the African Regional Intellectual Property Organization (ARIPO), the Organisation Africaine de la Propriété Intellectuelle (OAPI),280 the Eurasian Patent Office (EAPO),281 and the Gulf Cooperation Council Patent Office (GCCPO), offer streamlined, regional protection.282 These regional systems reduce duplication, and administrative burdens, making cross-border patenting more efficient.
At the national level, individual offices, such as the United States Patent and Trademark Office (USPTO), China National Intellectual Property Administration (CNIPA), National Intellectual Property Office of Japan (NIPO), the Japan Patent Office (JPO), and National Institute of Industrial Property (INPI), Portugal, Table S5, among others, are responsible for submitting, examining, granting and enforcing patents. Broader patent protection offers greater advantages to innovators, but carries higher filing and maintenance costs, which can burden low- and middle-income countries.
IPR grant temporary exclusivity (typically 20 years), which enables inventors to recover R&D&I investments, particularly important for natural products pharmaceuticals, cosmetics, and other biotechnological applications.264,270
Well-designed IPR systems stimulates research, attracts investment, enable technology transfer, support commercialisation, and facilitate cooperation between industry, research institutions, and IPLCs, while providing mechanisms to resolve ownership disputes. Yet an analysis of 11 million patents shows that only 4% of documented species, and less than 1% of all predicted species, appear in biodiversity-related patents, primarily in pharmaceuticals and genetic engineering.283 This limited coverage constraints the potential for broader long-term benefits, underscoring the need to expand research access, ensure fair benefit-sharing, and consider alternative models of innovation disclosure.283
5.3. Protecting traditional knowledge through IPR: case study of integration and protection
New Zealand is internationally recognised as a leader in the field of IPR of TK. The Intellectual Property Office of New Zealand (IPONZ) is the government agency responsible for the granting and registration of IPR in New Zealand.250 IPONZ manages the registration of trademarks, patents, designs, GIs, and plant variety rights in relation to mātauranga Māori.284–286 Subsection 15(3) of the Patents Act 2013 provides patent protection for the Māori peoples of New Zealand.272 It prescribes that the commissioner may seek advice from the Māori advisory committee when examining if an invention is derived from Māori TK or indigenous plants and animals, and if so, whether commercial exploitation is likely to be offensive to Māori, or contrary to Māori values.287 Māori claims of breaches of the Treaty of Waitangi by the Crown are investigated by a permanent commission of inquiry, of the Waitangi Tribunal, established in 1975.288 Although its recommendations are generally not legally binding, they carry significant political and moral weight and have been influential in addressing historical grievances, land claims, and matters concerning natural resources, cultural heritage, and TK.284–286For example, in 2019, application no. NZ 750401 was submitted for “the invention related to the manufacture and supply of consumer ready dietary supplements using monoflora and multiflora Manuka honey as the base ingredient with nutritional supplements added and the final product being in liquid, semi-liquid or creamed form”. Since Mānuka honey is produced from the nectar of the Mānuka tree (Leptospermum scoparium), species indigenous to New Zealand, a claim was reviewed under the jurisdiction of Māori Advisory Committee.289 Because the desired monopoly was not substantially constrained by any of the dependent claims, the application was denied following the committee's advice.
5.4. Global versus local IPR frameworks
While the foundational principles of IPR are shared globally, their practical implementation varies significantly between jurisdictions, which has led to ongoing debates regarding the harmonization of global and local IPR systems.IPR laws are primarily territorial in nature, meaning that protections are granted and enforced within specific national or regional boundaries, using IPR regimes often grounded in broader national policy goals.290 In this context, WIPO provides an overview of laws protecting GR, TK, and traditional cultural expressions (TCEs) at national and international levels.290
Although TRIPS sets minimum global standards for IPR protection and enforcement, national governments retain substantial flexibility in their interpretation and application.291 For example, the USA has adopted a broad view of patentable subject matter, originally allowing for the protection of genes (see Section 5.2.1),272,273 software and business methods, and has developed a litigation-intensive enforcement regime.291 In contrast, the EU applies stricter limits on patentable material, especially in areas such as biotechnology, genetics, and computer programs, while offering a centralized application process through the EPO.276,291 For example, software and business methods are not patentable in their abstract form, but they may obtain patent protection when demonstrating a genuine technical character.278 China has rapidly reformed its IPR framework to support economic modernization, strengthening legal and judicial mechanisms despite earlier enforcement challenges.292,293 India's IP regime balances protection with public interest, particularly by limiting pharmaceutical patents on incremental innovations to maintain access to affordable medicines.294
Developed countries prioritize strong IP protections to incentivize innovation, while developing nations emphasize flexibility to support access to technology, public health, and economic development, creating challenges for multinational companies seeking consistent cross-border IP protection.280,295,296
Decades of multilateral and regional efforts aim to harmonize IPR frameworks, but differences in legal systems, economic development, and public policy continue to hinder full alignment.280 Consequently, IPR governance demands flexible, context-sensitive approaches to balance global standards with national priorities.
6. Case studies in ethical and legal governance of natural product R&D&I
The discovery and development of natural products often reveal critical tensions between innovation, ethical conduct, and legal compliance. Ethical dilemmas in bioprospecting and natural product R&D&I often revolve around issues of PIC, benefit-sharing, and the use of TK without proper recognition or compensation. Case studies and real-world applications offer valuable insights into how researchers, governments, and international organizations have navigated these challenges, and provide lessons for future best practices.An early and influential example of ABS based on TK is the Kani Tribe–Jeevani case in India. The Kani community of Kerala shared knowledge about the medicinal plant Trichopus zeylanicus with scientists from the Tropical Botanic Garden and Research Institute (TBGRI), who developed and marketed Jeevani, an anti-stress drug.297 TBGRI established a benefit-sharing agreement that provided the Kani community with a share of license fees and royalties, managed through a community trust fund. Although the arrangement predated the Nagoya Protocol, it became a pioneering model for recognizing TK holders and influenced India's ABS framework.298
One widely discussed case is the Hoodia cactus in southern Africa. The San people had used Hoodia for generations as an appetite suppressant, commercial development for weight-loss products generated international interest. In the early 2000s, the South African Council for Scientific and Industrial Research (CSIR) patented an extract derived from Hoodia without initially consulting the San and Khoe IPLCs. After public criticism, a benefit-sharing agreement was reached, providing the IPLCs with a portion of the royalties.123,299 However, the most significant financial gains and decision-making power remained with research institutions and corporate actors.119,120
In contrast, the case of the Ayahuasca plant shows the consequences of unethical bioprospecting. A USA patent was granted on a strain of the plant in 1986, despite its widespread traditional use by Amazonian IPLCs. The patent was later revoked after a challenge by the Coordination Body of Indigenous Organizations of the Amazon River Basin (COICA), but the case highlighted the inadequacy of TK protection under existing patent systems.300–303 Similarly, the EPO granted a patent, in 1995, for a fungicide derived from the Neem tree (Azadirachta indica),304 which was revoked, in 2000, after a successful opposition filed by several Indian IPLCs representative organizations.305 Controversial or unresolved cases, such as the patenting of the Neem tree or quinoa varieties, highlight the challenges of defining ownership and ensuring equitable sharing in cross-border contexts, highlight the importance of transparent negotiation, legal clarity, and participatory governance in the implementation of ABS agreements.306
Sceletium tortuosum, known for its psychoactive and mood-enhancing properties, followed a comparable trajectory. Although traditional uses were acknowledged and agreements were concluded, industry actors retained dominant influence over commercialization pathways.119,120 In the case of Pelargonium sidoides, widely used in herbal remedies for respiratory infections, disputes over patents and access to GR highlighted concerns about misappropriation.119,120
The cases of Pelargonium and Baobab in southern Africa show that poorly formulated regulations originally meant to formalize trade and ensure sustainability, did unintentionally harm IPLCs, especially women and small farmers due to unclear distinction between biotrade and bioprospecting, enabling elite capture and ignoring TK systems, highlighting the need for more locally informed and context-sensitive policies.307
Even where ABS mechanisms were implemented, meaningful redistribution of power and long-term community empowerment remained limited.119,120 While formal recognition of TK and benefit-sharing agreements represent progress, they have not fundamentally altered underlying political and economic inequalities.307
These examples, among hundreds, illustrate the need for effective ethical review, inclusive stakeholder engagement, and the alignment of IPR practices with international norms, such as the CBD, the Nagoya Protocol, and the WIPO GRATK. Thus, lessons learned include the importance of PIC, MAT and access permits (e.g., MAA) from provider countries and IPLCs, the necessity of fair benefit-sharing mechanisms, and the risks of reputational and legal consequences when these are ignored.
While some natural product stakeholders argue that stringent access regulations may hinder scientific progress, others emphasize their role in ensuring ethical compliance and equitable outcomes.
A positive example, among many, is the case of the Samoa-AstraZeneca partnership for the development of the prodrug prostratin, derived from the Mamala tree (Homalanthus nutans), where a benefit-sharing agreement was negotiated in collaboration with local Samoan authorities.308,309
Conversely, rigid or unclear ABS rules, as seen in some Southeast Asian countries, have occasionally discouraged foreign researchers or led to delays in biodiversity-based innovation due to bureaucratic complexity.283 These contrasting case studies highlight the importance of legal clarity and administrative efficiency in supporting both innovation and compliance.
Well-designed legal frameworks, when paired with ethical research practices, can stimulate innovation while ensuring fairness and accountability, having a direct impact in natural product R&D&I. Best practices include establishing clear ABS protocols, involving IPLCs in research planning, and adapting IPR protections to accommodate collective TK.
International organizations such as the WHO, WIPO, and the European Group on Ethics (EGE),310 an advisory body to the EC, have issued key opinions on the ethical dimensions of patenting life and use of TK in science and new technologies. Their recommendations have informed policy debates and legislative proposals, highlighting the role of ethics in shaping IPR law, and playing a pivotal role in shaping global standards for the ethical and legal governance of natural product R&D&I.311 For instance, WIPO has led long-standing efforts to develop an international legal instrument for the protection of GR and TK. After decades of negotiations aimed at preventing misappropriation and promoting fair and equitable benefit-sharing, these efforts culminated in the adoption of the WIPO GRATK Treaty. The WHO has similarly contributed to discussions on ethical research involving traditional medicine, publishing guidelines for researchers working with IPLCs knowledge systems.312
A notable example of international cooperation is the MLS of ABS under the ITPGRFA, facilitating access to plant genetic materials from a global pool while ensuring benefit-sharing through a standardized agreement, considered a functional model that balances innovation and equity, particularly in the agricultural sector.84 The distribution of rice germplasm through the CGIAR gene bank network is a practical example of its applicability.313
The entry into force of the BBNJ Treaty transforms ocean-related R&D&I into a more coordinated, legally secure, digitally enabled, and globally inclusive system, accelerating sustainable and responsible innovation.
These case studies of international efforts demonstrate that cooperation, legal harmonization, and the integration of ethical frameworks can significantly enhance both scientific outcomes and social justice.
7. Navigating RRI, ethical, Nagoya Protocol and legal frameworks, and IPR challenges – future directions
The discovery and development of natural products across pharmaceuticals, cosmetics, agriculture, and other biotechnological sectors occupies a distinctive position at the intersection of science, ethics, law, and political economy. As global demand for bio-based innovation accelerates, actors must navigate an increasingly complex governance landscape shaped by RRI, ethical standards, ABS regimes, and IPR frameworks, Fig. 4.Sustainable and equitable progress, therefore, depends not on isolated regulatory compliance, but on integrative frameworks capable of reconciling diverse stakeholder interests while ensuring adherence to both international and local norms.
When RRI principles, ethical safeguards, law, and balanced IPR regimes are implemented cohesively and operate in a coordinated manner, they build a foundation of mutual respect and trust among researchers, IPLCs, governments, and the private sector, which enables stable partnerships, reduces legal uncertainty, and increases the chances of successful commercialization with shared benefits and biodiversity conservation objectives. These frameworks support the inclusion of legal and ethical education in scientific training, strengthening compliance and awareness. They also empower IPLCs through knowledge-sharing, allowing them to assert rights and engage as equal research partners.
Nagoya Protocol compliance in natural products R&D&I will become more harmonized, digitally integrated, and equity-oriented overtime. ABS obligations are increasingly integrated into research design through compliance-by-design approaches that incorporate PIC, MAT, and documentation from the outset. Digital traceability systems and standardized metadata play a growing role, particularly in managing DSI. Strengthened regulatory literacy, better alignment between ABS and IP systems, and greater participation of IPLCs further support transparent, meaningful, and equitable benefit-sharing.
Governance innovation itself emerges as a research frontier. Monitoring systems, compliance platforms, and digital reporting mechanisms required under evolving ABS frameworks represent new technological and institutional innovation domains. Natural products R&D&I, therefore, extends beyond metabolite discovery to encompass governance technologies that enable transparent, fair, and science-informed utilization of GR.
Marine natural products research is increasingly shaped by a dual governance framework integrating the Nagoya Protocol and the BBNJ Treaty. This configuration establishes a more structured and predictable legal environment for accessing MGR within and beyond national jurisdiction. As a result, R&D&I strategies incorporate compliance-by-design principles, embedding early jurisdictional assessment, standardized material and data tracking systems, and structured benefit-sharing mechanisms into project design. Regulatory foresight, thus, emerges as a central institutional competence in natural products research and biotechnology sectors. The emphasis on equitable participation under the BBNJ framework encourages co-development models, regional research hubs, sequencing infrastructure expansion, and training programmes embedded within research consortia. This will broaden the global scientific base contributing to natural product discovery and reduce historical North–South asymmetries in biodiversity utilization.
IP strategies will evolve toward more flexible and collaborative models. Although the BBNJ framework does not directly regulate IP, it intersects with discussions at the WIPO and within the TRIPS regime of the WHO.
IPR incentivize research and development by granting inventors temporary monopolies (e.g., patents). However, in natural product R&D&I, IPR systems should be adapted to avoid the misappropriation of GR or associated TK. Sui generis systems or hybrid IP approaches, such as structured licensing agreements incorporating benefit-sharing clauses, patent pools for pre-competitive research, and differentiated access models for developing-country partners, can ensure that collective and intergenerational community knowledge is acknowledged and protected, while also allowing innovation to flourish. Rather than purely exclusive patent-driven commercialization, natural products innovation may increasingly adopt partnership-based and equity-sensitive commercialization pathways.
Digitalization is reshaping the field. As governance frameworks expand to encompass DSI, natural products discovery relies increasingly on interoperable databases, integrated bioinformatics platforms, and traceability systems that connect physical materials with genomic data. Emerging innovation trajectories emphasize AI-assisted metabolite discovery, high-throughput screening technologies, digital bioprospecting platforms, and secure infrastructures designed to facilitate global reporting and benefit-sharing compliance. Consequently, natural products R&D&I evolves into a highly data-intensive, computational, and transnationally networked domain.
Sustainability and ecosystem-based approaches increasingly shape upstream research priorities. Natural products R&D&I moves toward biodiversity-positive innovation, integrating conservation objectives directly into discovery pipelines. This shift includes the adoption of non-destructive sampling techniques, synthetic biology alternatives to overharvesting, and nature-based solutions aligned with global biodiversity and climate agendas.
8. Conclusions
Natural products R&D&I is undergoing a structural transformation in which innovation is no longer separable from governance, ethics, and global equity. The consolidation of ABS obligations under the Nagoya Protocol and the operationalization of the BBNJ Treaty signal a decisive shift from loosely regulated bioprospecting toward a legally embedded model of biodiversity-based innovation. These frameworks redefine the conditions under which GR, associated TK, and DSI can be accessed, utilized, and commercialized.This transformation is reinforced by the growing institutionalization of RRI. Ethical reflexivity, early stakeholder engagement, environmental sustainability, and equitable partnership models are increasingly integrated into research design rather than appended as compliance formalities. In this emerging paradigm, scientific excellence and social legitimacy are mutually reinforcing. Meaningful participation of IPLCs, transparent benefit-sharing arrangements, and ecosystem-based approaches are no longer peripheral considerations but central determinants of research quality and long-term viability.
IP systems are also being recalibrated. Developments within the WIPO and broader international IP debates demonstrate a gradual alignment between patent governance and ABS principles, particularly through enhanced disclosure mechanisms and recognition of GR and associated TK. Rather than functioning solely as instruments of exclusivity, IPR increasingly operate as governance tools capable of structuring collaboration, enabling investment, and embedding benefit-sharing within commercialization pathways.
At the same time, digital transformation introduces both opportunity and complexity. The expansion of research reliant on DSI, AI-assisted discovery, and interoperable global databases demands traceability systems that safeguard equity without undermining scientific openness. Governance innovation, through metadata standards, provenance tracking, and compliance-by-design models, becomes as central to advancement as laboratory discovery itself.
Nonetheless, important challenges remain, including regulatory fragmentation across ABS and IP regimes, uneven global Nagoya Protocol implementation, administrative burdens, and persistent global asymmetries in research capacity risk slowing or distorting progress. Addressing these tensions will require sustained coordination, institutional capacity building, and continued engagement between policymakers, scientists, industry, and TK holders.
Taken together, these developments redefine natural products R&D&I as a governance-intensive, equity-oriented, and sustainability-embedded innovation domain. The field is no longer characterized primarily by resource extraction and downstream patenting, but by integrated legal foresight, ethical accountability, digital stewardship, and collaborative value creation. This marks a decisive evolution in how biodiversity-based knowledge is generated, protected, and shared within the emerging global bioeconomy.
9. Author contributions
Conceptualization, S. P. G.; methodology, S. P. G., B. K. S., L. N., C. R. and X. T. S.; data curation, S. P. G., B. K. S., L. N., C. R. and X. T. S.; writing—original draft preparation, S. P. G., B. K. S., L. N., C. R. and X. T. S.; writing—review and editing, S. P. G., B. K. S., L. N., C. R. and X. T. S.; visualization, S. P. G., B. K. S., L. N., C. R. and X. T. S; project administration, S. P. G.; funding acquisition, S. P. G. and C. R. All authors have read and agreed to the published version of the manuscript.10. Conflicts of interest
There are no conflicts to declare.11. Abbreviations
| ABNJ | Areas Beyond National Jurisdiction |
| ABS | Access and Benefit-Sharing |
| ABSCH | Access and Benefit-sharing Clearing-House |
| AI | Artificial Intelligence |
| ALLEA | All European Academies |
| ARIPO | African Regional Intellectual Property Organization |
| BBNJ | Biodiversity Beyond National Jurisdiction |
| BGCI | Botanic Gardens Conservation International |
| BR | Biomaterial Repositories |
| BSE | Bovine Spongiform Encephalopathy |
| Cas9 | CRISPR-Associated Protein 9 |
| CBD | Convention on Biological Diversity |
| cDNA | Complementary Deoxyribonucleic Acid |
| CETAF | Consortium of European Taxonomic Facilities |
| CNIPA | China National Intellectual Property Administration |
| COICA | Coordination Body of Indigenous Organizations of the Amazon River Basin |
| COP | Conference of the Parties |
| CRISPR | Clustered Regularly Interspaced Short Palindromic Repeats |
| CSIR | South African Council for Scientific and Industrial Research |
| DNA | Deoxyribonucleic Acid |
| DNSH | Do No Significant Harm |
| DOSI | Deep-Ocean Stewardship Initiative |
| DSI | Digital Sequence Information |
| EAPO | Eurasian Patent Office |
| EBP | Earth BioGenome Project |
| EBRs | Environmental Biospecimen Repositories |
| ECCRI | European Code of Conduct for Research Integrity |
| EC | European Commission |
| EEZ | Exclusive Economic Zone |
| EGE | European Group on Ethics |
| EFSA | European Food Safety Authority |
| EIAs | Environmental Impact Assessments |
| ELSA | Ethical, Legal, and Social Aspects |
| ELSI | Ethical, Legal, and Social Issues |
| EMFAF | European Maritime, Fisheries and Aquaculture Fund |
| ENERI | European Network for Research Ethics and Integrity |
| ENRIO | European Network of Research Integrity Offices |
| EPO | European Patent Office |
| ERA | European Research Area |
| EU | European Union |
| EUREC | European Network of Research Ethics Committees |
| FAO | Food and Agriculture Organization |
| FPIC | Free, Prior and Informed Consent |
| GAI | Generative Artificial Intelligence |
| GCCPO | Gulf Cooperation Council Patent Office |
| GDPR | General Data Protection Regulation |
| GGBN | Global Genome Biodiversity Network |
| GMOs | Genetically Modified Organisms |
| GR | Genetic Resources |
| GRATK | Genetic Resources and Associated Traditional Knowledge |
| GRTKF | Intergovernmental Committee on Intellectual Property and Genetic Resources, Traditional Knowledge and Folklore |
| ICESCR | International Covenant on Economic, Social and Cultural Rights |
| IGC | Intergovernmental Committee |
| INSDC | International Nucleotide Sequence Database Collaboration |
| INPI | National Institute of Industrial Property Portugal |
| IP | Intellectual Property |
| IPLCs | Indigenous Peoples and Local Communities |
| IPONZ | Intellectual Property Office of New Zealand |
| IPR | Intellectual Property Rights |
| IPR | Intellectual Property Rights |
| IRCC | Internationally Recognized Certificate of Compliance |
| ITPGRFA | International Treaty on Plant Genetic Resources for Food and Agriculture |
| JPO | Japan Patent Office |
| LLMs | Large Language Models |
| LMOs | Living Modified Organisms |
| MAA | Material Accession Agreement |
| MAT | Mutually Agreed Terms |
| MGR | Marine Genetic Resources |
| MLS | Multilateral System |
| MPAs | Marine Protected Areas |
| MSP | Maritime Spatial Planning |
| MTA | Material Transfer Agreement |
| NCA | National Competent Authority |
| NENT | Committee for Research Ethics in Science and Technology |
| NESH | National Committee for Research Ethics in the Social Sciences and the Humanities |
| NFP | National Focal Point |
| NGOs | Non-Governmental Organizations |
| NGTs | New Genomic Techniques |
| NIPO | National Intellectual Property Office of Japan |
| OA | Open Access |
| OAPI | Organisation Africaine de la Propriété Intellectuelle |
| OS | Open Science |
| PABS | Pathogen Access and Benefit-Sharing |
| PCT | Patent Cooperation Treaty |
| PIC | Prior informed consent |
| R&D&I | Research, development and innovation |
| RNA | Ribonucleic Acid |
| RRI | Responsible Research and Innovation |
| SDGs | Sustainable Development Goals |
| SMTA | Standard Material Transfer Agreement |
| TALENs | Transcription Activator-Like Effector Nucleases |
| TBGRI | Tropical Botanic Garden and Research Institute |
| TK | Traditional Knowledge |
| TKDL | Biological Diversity Act and Traditional Knowledge Digital Library |
| TRIPS | Trade-Related Aspects of Intellectual Property Rights |
| UNCLOS | United Nations Convention on the Law of the Sea |
| UNDRIP | United Nations Declaration on the Rights of IPLCs Peoples |
| UNOC | United Nations Ocean Conference |
| USA | United States of America |
| USPTO | United States Patent and Trademark Office |
| WHO | World Health Organization |
| WIPO | World Intellectual Property Organization |
| WTO | World Trade Organization |
12. Data availability
All data analysed are included in this published article and its supplementary information (SI) files. Supplementary information: Table S1. European codes of conduct, guidelines, and legal instruments related to research ethics and integrity; Table S2. National codes of conduct and research ethics or integrity guidelines; Table S3. Roles and Responsibilities of Parties, Authorities, Providers, and Users in the ABS Process under the Nagoya Protocol; Table S4. Intellectual Property Rights protection tools for Genetic Resources and Traditional Knowledge; Table S5. Major patent offices; Table S6. Patent systems overview. See DOI: https://doi.org/10.1039/d5np00072f.13. Acknowledgements
S. P. G.: National funds from FCT—Fundação para a Ciência e a Tecnologia, IP, in the scope of the project UID/04378/2025 (DOI identifier 10.54499/UID/04378/2025), and UID/PRR/04378/2025 (DOI identifier 10.54499/UID/PRR/04378/2025), of the Research Unit on Applied Molecular Biosciences – UCIBIO and the project LA/P/0140/2020 (DOI identifier 10.54499/LA/P/0140/2020) of the Associate Laboratory Institute for Health and Bioeconomy – i4HB. X. T. S.: projects Horizon Europe Grant Agreement Project 101137025 – DETECTIVE and Horizon Europe Grant Agreement Project 101061015 – GeneBEcon. C. R.: contribution supported by the Norwegian Research Council (294946, 312196, 319577; 326803), and Møreforsking AS. COST Actions CA18238 (Ocean4Biotech) and CA20106 (SEAWHEAT), supported by COST (European Cooperation in Science and Technology) program.14. References
- Convention on Biological Diversity, https://www.cbd.int/doc/legal/cbd-en.pdf, accessed October 2025 Search PubMed.
- Nagoya Protocol, https://www.cbd.int/abs/doc/protocol/nagoya-protocol-en.pdf, accessed October 2025 Search PubMed.
- The WIPO Treaty on Intellectual Property, Genetic Resources and Associated Traditional Knowledge (WIPO GRATK Treaty), https://www.wipo.int/en/web/treaties/ip/gratk/summary_gratk, accessed January 2026 Search PubMed.
- A Strategic Framework for a Competitive and Sustainable EU Bioeconomy, https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:52025DC0960, accessed October 2025 Search PubMed.
- EU Biotech Act, https://www.eu-biotech-act.com/, accessed October 2025 Search PubMed.
- EU Bioeconomy Strategy, https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:52025DC0960, accessed October 2025 Search PubMed.
- M. Burget, E. Bardone and M. Pedaste, Sci. Eng. Ethics, 2017, 23, 1–19 CrossRef PubMed.
- R. Thapa, T. Iakovleva and L. Foss, Eur. Plan. Stud., 2019, 27, 2470–2490 CrossRef.
- R. Owen, R. von Schomberg and P. Macnaghten, J. Responsible Innov., 2021, 8, 217–233 CrossRef.
- R. Owen, P. Macnaghten and J. Stilgoe, Sci. Public Policy, 2012, 39, 751–760 CrossRef.
- Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions, https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:52020DC0628&rid=1, accessed October 2025 Search PubMed.
- H. Rodríguez, E. Fisher and D. Schuurbiers, Res. Policy, 2013, 42, 1126–1137 CrossRef.
- European Governance A White Paper, 2001, https://ec.europa.eu/commission/presscorner/detail/en/doc_01_10, accessed October 2025 Search PubMed.
- R. Von Schomberg, in Technikfolgen abschätzen lehren: Bildungspotenziale transdisziplinärer Methoden, ed. M. D. R. Beecroft, Springer, Wiesbaden, 2011, pp. 39–61 Search PubMed.
- Rome Declaration on Responsible Research and Innovation in Europe, https://digital-strategy.ec.europa.eu/en/library/rome-declaration-responsible-research-and-innovation-europe, accessed October 2025 Search PubMed.
- J. Stilgoe, R. Owen and P. Macnaghten, Res. Policy, 2013, 42, 1568–1580 CrossRef.
- A. Rip, Life Sci. Soc. Policy, 2014, 10, DOI:10.1186/s40504-014-0017-4.
- S. de Saille, J. Responsible Innov., 2015, 2, DOI:10.1080/23299460.2015.1045280.
- M. van Oudheusden, J. Responsible Innov., 2014 DOI:10.1080/23299460.2014.882097.
- R. Owen, M. Pansera, P. Macnaghten and S. Randles, Res. Policy, 2021, 50, 104132 CrossRef.
- R. Vicente-Saez and C. Martinez-Fuentes, J. Bus. Res., 2018, 88, 428–436 CrossRef.
- C. Shelley-Egan, M. Gjefsen and R. Nydal, Life Sci. Soc. Policy, 2020, 16(7) DOI:10.1186/s40504-020-00103-5.
- European Strategy for Responsible Research and Innovation, https://research-and-innovation.ec.europa.eu/strategy/strategy-research-and-innovation_en#strategy-document, accessed October 2025 Search PubMed.
- RRI Roadmap, https://www.xpro-consulting.com/uploads/4/9/5/5/49557869/rri_roadmap__online_single.pdf, accessed October 2025 Search PubMed.
- X. T. Schneider, B. K. Stroil, C. Tourapi, C. Rebours, S. P. Gaudencio, L. Novoveska and M. I. Vasquez, Mar. Drugs, 2022, 20(5), 290 CrossRef PubMed.
- S. Pitt and A. Gunn, Br. J. Biomed. Sci., 2024, 81, 12366 CrossRef PubMed.
- R. Lubberink, V. Blok, J. van Ophem and O. Omta, Sustainability, 2017, 9 Search PubMed.
- K. Wakunuma, F. de Castro, T. Jiya, E. Inigo, V. Blok and V. Bryce, J. Responsible Innov., 2021, 8, 267–291 CrossRef.
- J. Kuzma and C. Cummings, Front. Polit. Sci., 2021, 3, 677003 CrossRef.
- R. Owen and M. Pansera, in Handbook on Science and Public Policy, ed. D. Simon, S. Kuhlmann, J. Stamm and W. Canzler, 2019, ch. 2, pp. 26–48 Search PubMed.
- K. R. Srinivas, in Ethics, Integrity and Policymaking: The Value of the Case Study, ed. D. O'Mathúna and R. Iphofen, Springer International Publishing, Cham, 2022, pp. 29–48 Search PubMed.
- Y. Cho, S. Wang, S. Kinoshita, Y. Ikkatai and H. M. Yokoyama, AI Ethics, 2025, 5, 6139–6150 CrossRef.
- European Commission, Directorate-General for Research and, European bioeconomy policy – Stocktaking and future developments – Report from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions, Publications Office of the European Union, 2022, https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex:52022DC0283, accessed October 2025 Search PubMed.
- A. Dwivedi, M. Moktadir, C. Jabbour and D. De Carvalho, Technol. Forecast. Soc. Change, 2022, 175, 121335 CrossRef.
- M. Mullowney, K. Duncan, S. Elsayed, N. Garg, J. van der Hooft, N. Martin, D. Meijer, B. Terlouw, F. Biermann, K. Blin, J. Durairaj, M. González, E. Helfrich, F. Huber, S. Leopold-Messer, K. Rajan, T. de Rond, J. Van Santen, M. Sorokina, M. Balunas, M. Beniddir, D. van Bergeijk, L. Carroll, C. Clark, D. Clevert, C. Dejong, C. Du, S. Ferrinho, F. Grisoni, A. Hofstetter, W. Jespers, O. Kalinina, S. Kautsar, H. Kim, T. Leao, J. Masschelein, E. Rees, R. Reher, D. Reker, P. Schwaller, M. Segler, M. Skinnider, A. Walker, E. Willighagen, B. Zdrazil, N. Ziemert, R. Goss, P. Guyomard, A. Volkamer, W. Gerwick, H. Kim, R. Müller, G. van Wezel, G. van Westen, A. Hirsch, R. Linington, S. Robinson and M. Medema, Nat. Rev. Drug Discovery, 2023, 22, 895–916 CrossRef CAS PubMed.
- V. J. Sahayasheela, M. B. Lankadasari, V. M. Dan, S. G. Dastager, G. N. Pandian and H. Sugiyama, Nat. Prod. Rep., 2022, 39, 2215–2230 RSC.
- Y. Zhu, Z. Ouyang, H. Du, M. Wang, J. Wang, H. Sun, L. Kong, Q. Xu, H. Ma and Y. Sun, Acta Pharm. Sin. B, 2022, 12, 4011–4039 CrossRef CAS PubMed.
- P. Parvatikar, S. Patil, K. Khaparkhuntikar, S. Patil, P. Singh, R. Sahana, R. Kulkarni and A. Raghu, Antiviral Res., 2023, 220, 105740 CrossRef CAS PubMed.
- M. Ramos, C. Collison and A. White, Chem. Sci., 2025, 16, 2514–2572 RSC.
- A. Gangwal and A. Lavecchia, J. Med. Chem., 2025, 68, 3948–3969 CrossRef CAS PubMed.
- P. Chunarkar-Patil, M. Kaleem, R. Mishra, S. Ray, A. Ahmad, D. Verma, S. Bhayye, R. Dubey, H. Singh and S. Kumar, Biomedicines, 2024, 12(1), 201 CrossRef CAS PubMed.
- S. P. Gaudencio and F. Pereira, Nat. Prod. Rep., 2015, 32(6), 779–810 RSC.
- N. Thomford, D. Senthebane, A. Rowe, D. Munro, P. Seele, A. Maroyi and K. Dzobo, Int. J. Mol. Sci., 2018, 19(6), 1578 CrossRef PubMed.
- D. Blakemore, L. Castro, I. Churcher, D. Rees, A. Thomas, D. Wilson and A. Wood, Nat. Chem., 2018, 10, 383–394 CrossRef CAS PubMed.
- S. P. Gaudencio, E. Bayram, L. Lukic Bilela, M. Cueto, A. R. Diaz-Marrero, B. Z. Haznedaroglu, C. Jimenez, M. Mandalakis, F. Pereira, F. Reyes and D. Tasdemir, Mar. Drugs, 2023, 21(5), 308 CrossRef CAS PubMed.
- M. Giuffrè and D. Shung, npj Digit. Med., 2023, 6, 186 CrossRef.
- C. Lawson, E. Englezos and M. Rourke, Plants People Planet, 2025, vol. 7, pp. 1485–1497 Search PubMed.
- C. Ho, Front. Public Health, 2022, 10, 768977 CrossRef PubMed.
- E. González-Esteban and P. Calvo, Heliyon, 2022, 8(2), e08946 CrossRef PubMed.
- ABS Clearing-House, https://www.cbd.int/abs/theabsch.shtml, accessed October 2025 Search PubMed.
- K. Oksman-Caldentey and D. Inzé, Trends Plant Sci., 2004, 9, 433–440 CrossRef CAS PubMed.
- H. Ikeda, Biosci. Biotechnol. Biochem., 2017, 81, 13–22 CrossRef CAS PubMed.
- A. Nemudryi, K. Valetdinova, S. Medvedev and S. Zakian, Acta Nat., 2014, 6, 19–40 CrossRef CAS.
- E. Mullins, J. Bresson, T. Dalmay, I. Dewhurst, M. Epstein, L. Firbank, P. Guerche, J. Hejatko, F. Moreno, H. Naegeli, F. Nogué, N. Rostoks, J. Serrano, G. Savoini, E. Veromann, F. Veronesi, A. Fernandez, A. Gennaro, N. Papadopoulou, T. Raffaello, R. Schoonjans and E. P. G. M. Or, EFSA J., 2022, 20(10), 7618 Search PubMed.
- D. van Bergeijk, B. Terlouw, M. Medema and G. van Wezel, Nat. Rev. Microbiol., 2020, 18, 546–558 CrossRef CAS PubMed.
- Q. Zhou, K. Hotta, Y. Deng, R. Yuan, S. Quan and X. Chen, Microorganisms, 2021, 9(12), 2551 CrossRef CAS PubMed.
- S. Bhuyan, M. Kumar, P. Devde, A. Rai, A. Mishra, P. Singh and K. Siddique, Front. Genome Ed., 2023, 5, 1272678 CrossRef PubMed.
- European Food Safety Authority (EFSA), New genomic techniques, https://www.efsa.europa.eu/en/topics/new-genomic-techniques, accessed January 2026 Search PubMed.
- Cartagena Protocol, https://bch.cbd.int/protocol, accessed January 2026 Search PubMed.
- Biosafety Information Centre, https://biosafety-info.net/articles/global-agreements-and-fora/cartagena-protocol/biodiversity-meeting-agrees-on-limited-action-on-technology-and-synthetic-biology, accessed January 2026 Search PubMed.
- A. So, M. Habets, C. Testerink and P. Macnaghten, Plants People Planet, 2024, 6, 760–773 CrossRef.
- S. Kjeldaas, T. Dassler, T. Antonsen, O. Wikmark and A. Myhr, Agric. Human Values, 2023, 40, 533–545 CrossRef.
- Provisional Agreement on NGTs for plants, https://ec.europa.eu/commission/presscorner/detail/fr/ip_25_2912, accessed February 2026 Search PubMed.
- Detective Project, https://detective-ngt.eu/, accessed February 2026 Search PubMed.
- T. Sprink, J. Metje, J. Schiemann and F. Hartung, Plant Biotechnol. Rep., 2016, 10, 345–351 CrossRef.
- D. Eriksson, R. Custers, K. Björnberg, S. Hansson, K. Purnhagen, M. Qaim, J. Romeis, J. Schiemann, S. Schleissing, J. Tosun and R. Visser, Trends Biotechnol., 2020, 38, 231–234 CrossRef CAS PubMed.
- D. Eriksson, R. Custers, K. Björnberg, S. Hansson, K. Purnhagen, M. Qaim, J. Romeis, J. Schiemann, S. Schleissing, J. Tosun and R. Visser, Trends Biotechnol., 2020, 38, 349–351 CrossRef CAS PubMed.
- K. Cartledge, F. Short, A. Hall, K. Lambert, M. Mcdonald and T. Lithgow, IUBMB Life, 2025, 77(1), e2931 CrossRef CAS PubMed.
- L. Krishna Panicker, A. Sudhakaran, M. P. Vinod, K. Swathy, A. S. Nair and O. V. Oommen, in Biodiversity and Business, ed. L. Krishna Panicker, P. Nelliyat and O. V. Oommen, Springer, 2023 Search PubMed.
- P. Mograbi, E. Archer, C. Fabricius, R. Wynberg and J. Donaldson, S. Afr. J. Sci, 2023, 119(3/4), 15739 Search PubMed.
- J. Fromentin, M. Emery, J. Donaldson, G. Balachander, E. Barron, R. Chaudhary, M. Danner, M. Gasalla, A. Hallosserie, M. Halmy, C. Hicks, D. Kieling, M. Park, B. Parlee, J. Rice, T. Ticktin and D. Tittensor, Global Environmental Change-Human and Policy Dimensions, 2023, vol. 81 Search PubMed.
- M. Habibullah, B. Din, S. Tan and H. Zahid, Environ. Sci. Pollut. Res., 2022, 29, 1073–1086 CrossRef PubMed.
- A. B. Cunningham, Applied Ethnobotany People, Wild Plant Use and Conservation, Routledge, London, 2001 Search PubMed.
- M. Wani, H. Taylor, M. Wall, P. Coggon and A. Mcphail, J. Am. Chem. Soc., 1971, 93(9), 2325–2327 CrossRef CAS PubMed.
- K. Witherup, S. Look, M. Stasko, T. Ghiorzi, G. Muschik and G. Cragg, J. Nat. Prod., 1990, 53, 1249–1255 CrossRef CAS.
- B. Gabetta, P. DeBellis, R. Pace, G. Appendino, L. Barboni, E. Torregiani, P. Gariboldi and D. Viterbo, J. Nat. Prod., 1995, 58, 1508–1514 CrossRef CAS.
- A. Zhiri, M. Jaziri, Y. Guo, R. Vanhaelenfastre, M. Vanhaelen, J. Homes, K. Yoshimatsu and K. Shimomura, Bio. Chem. Hoppe-Seyler, 1995, 376, 583–586 CrossRef CAS PubMed.
- C. Roberts, C. Bene, N. Bennett, J. Boon, W. Cheung, P. Cury, O. Defeo, G. Cleyndert, R. Froese, D. Gascuel, C. Golden, J. Hawkins, A. Hobday, J. Jacquet, P. Kemp, M. Lam, F. Le Manach, J. Meeuwig, F. Micheli, T. Morato, C. Norris, C. Nouvian, D. Pauly, E. Pikitch, F. Amargos, A. Saenz-Arroyo, U. Sumaila, L. Teh, L. Watling and B. O’Leary, npj Ocean Sustain, 2024, 3, 41 CrossRef.
- A. Nawaz, Int. J. Pharmacol. Pharm. Res., 2019, 1(1), 9–13 CrossRef.
- N. G. M. Gomes, R. Dasari, S. Chandra, R. Kiss and A. Kornienko, Mar. Drugs, 2016, 14(5), 98 CrossRef PubMed.
- C. Cuevas and A. Francesch, Nat. Prod. Rep., 2009, 26, 322–337 RSC.
- C. Rath, B. Janto, J. Earl, A. Ahmed, F. Hu, L. Hiller, M. Dahlgren, R. Kreft, F. Yu, J. Wolff, H. Kweon, M. Christiansen, K. Håkansson, R. Williams, G. Ehrlich and D. Sherman, ACS Chem. Biol., 2011, 6, 1244–1256 CrossRef CAS PubMed.
- M. Schofield, S. Jain, D. Porat, G. Dick and D. Sherman, Environ. Microbiol., 2015, 17, 3964–3975 CrossRef CAS PubMed.
- Plant Treaty, https://www.fao.org/plant-treaty/en, acessed February 2026 Search PubMed.
- C. Fowler, Genetic Resources and Crop Evolution, 2004, vol. 51, pp. 609–620 Search PubMed.
- M. Tvedt, Journal of World Intellectual Property, 2025 Search PubMed.
- EU Taxonomy Regulation, https://ec.europa.eu/info/business-economy-euro/banking-and-finance/sustainable-finance/eu-taxonomy-sustainable-activities_en, accessed October 2025 Search PubMed.
- T. Sikor, A. Martin, J. Fisher and J. He, Conserv. Lett., 2014, 7, 524–532 CrossRef.
- European Commission, Nature restoration: Council and Parliament reach agreement on new rules to restore and preserve degraded habitats in the EU, 2023, https://www.consilium.europa.eu/en/press/press-releases/2023/11/09/nature-restoration-council-and-parliament-reach-agreement-on-new-rules-to-restore-and-preserve-degraded-habitats-in-the-eu/pdf/, accessed October 2025 Search PubMed.
- R. Alam and N. Van Quyen, Environ. Econ. Policy Stud., 2025, 27, 109–138 CrossRef.
- L. Onaga, EMBO Rep., 2001, 2, 263–265 CrossRef CAS PubMed.
- T. Kursar, C. Caballero-George, T. Capson, L. Cubilla-Rios, W. Gerwick, M. Gupta, A. Ibañez, R. Linington, K. McPhail, E. Ortega-Barría, L. Romero, P. Solis and P. Coley, Bioscience, 2006, 56, 1005–1012 CrossRef.
- C. Costello and M. Ward, J. Environ. Econ. Manage., 2006, 52, 615–626 CrossRef.
- J. R. Rani, L. Krishna Panicker, V. S. Soumya, A. Jayachandran, M. P. Vinod, P. Sunitha and O. V. Oommen, in Biodiversity and Business: Bio Prospecting and Benefit Sharing, ed. L. Krishna Panicker, P. Nelliyat and O. V. Oommen, Springer Nature Switzerland, Cham, 2024, pp. 51–61 Search PubMed.
- Intergovernmental Committee on Intellectual Property and Genetic Resources Traditional Knowledge and Folklore, https://www.wipo.int/edocs/mdocs/tk/en/wipo_grtkf_ic_17/wipo_grtkf_ic_17_inf_9.pdf, accessed October2025 Search PubMed.
- R. Morgan, Hum. Organ., 2007, 66, 60–68 CrossRef.
- S. Kitolelei, A. Soderberg, N. Qaqara, S. Prakash, M. Tuiono, J. Veitayaki and S. Piovano, AMBIO, 2022, 51, 2431–2444 CrossRef PubMed.
- Community-Based Monitoring of Freshwater Turtles, https://www.forestpeoples.org/publications-resources/training-materials/article/community-based-monitoring-of-freshwater-turtles/, accessed October2025 Search PubMed.
- A. Mastretta-Yanes, S. Suárez, R. Jordan, S. Hoban, J. Silva, L. Castillo-Reina, M. Heuertz, F. Ishihama, V. Köppä, L. Laikre, A. Macdonald, J. Mergeay, I. Paz-Vinas, G. Segelbacher, A. Knapps, H. Rakoczy, A. Weiler, A. Angelica, K. Cullmann, S. Bagnato and B. Forester, Biodivers. Inform., 2024, 18, 24–27 Search PubMed.
- F. Affinito, J. Williams, J. Campbell, M. Londono and A. Gonzalez, Nat. Ecol. Evol., 2024, 8, 2163–2171 CrossRef PubMed.
- H. Confraria, T. Ciarli and E. Noyons, Res. Policy, 2024, 53, 104950 CrossRef.
- GeneBEcon, https://genebecon.eu/, accessed October 2025 Search PubMed.
- GeneBEcon Policy Brief, https://genebecon.eu/wp-content/uploads/2025/03/GeneBEcon-Policy-Brief-Promoting-Consumer-Acceptance-of-NGTs.pdf, accessed February 2026 Search PubMed.
- GeneBEcon Transition Action Plan, https://genebecon.eu/en/ngts-adoption-route-en, accessed February 2026 Search PubMed.
- M. Ariza, A. Quesada, A. Abril and F. García, Promoting Responsible Research Through Science Education. Design And Evaluation of A Teacher Training Program, in Proceedings of The INTED2016: 10th International Technology, Education And Development Conference, 2016, pp. 3941–3950 Search PubMed.
- R. Tokalic, I. Buljan, N. Mejlgaard, M. Carrió, A. Lang, G. Revuelta, A. Marusic and H. Consortium, Forensic Sci. Res., 2021, 6, 320–330 CrossRef PubMed.
- L. Esteller, A. Molas, D. Boer and K. Krüger, Year Search PubMed.
- A sustainable Bioeconomy for Europe: strengthening the connection between economy, Society and the Environment, 2018, https://www.univ-reims.fr/aebb-en/media-files/18808/ec_bioeconomy_strategy_2018.pdf, accessed October 2025 Search PubMed.
- L. Staffas, M. Gustavsson and K. McCormick, Sustainability, 2013, 5, 2751–2769 CrossRef.
- M. M. Bugge, T. Hansen and A. Klitkou, Sustainability, 2016, 8, 691 CrossRef.
- T. Efferth, M. Banerjee, M. Abu-Darwish, S. Abdelfatah, M. Böckers, D. Bhakta-Guha, V. Bolzani, S. Daak, Ö. Demirezer, M. Dawood, M. Efferth, H. El-Seedi, N. Fischer, H. Greten, S. Hamdoun, C. Hong, M. Horneber, O. Kadioglu, H. Khalid, S. Khalid, V. Kuete, N. Mahmoud, J. Marin, A. Mbaveng, J. Midiwo, H. Nakagawa, J. Nass, O. Ngassapa, D. Ochwang'i, L. Omosa, E. Ooko, N. Özenver, P. Poornima, M. Romero, M. Saeed, L. Salgueiro, E. Seo, G. Yan, Z. Yasin, E. Saeed and N. Paul, Phytomedicine, 2019, 53, 319–331 CrossRef.
- C. Hamilton, Dev. World Bioeth., 2006, 6, 158–173 CrossRef PubMed.
- European Research Area, https://european-research-area.ec.europa.eu/era-policy-agenda-2025-2027, accessed October 2025 Search PubMed.
- D. Newman, Expert Opin. Drug Discov., 2019, 14, 521–525 CrossRef PubMed.
- K. Duerbeck, F. Staubli and B. Ottens, Planta Med., 2010, 76, 1198 Search PubMed.
- FairWild and Fair for Life, https://www.fairwild.org/fairwild-certification, accessed October 2025 Search PubMed.
- A. De Witt, P. Osseweijer and R. Pierce, Public Underst. Sci., 2017, 26, 70–88 CrossRef PubMed.
- S. Sijtsema, M. Onwezen, M. Reinders, H. Dagevos, A. Partanen and M. Meeusen, NJAS Wageningen J. Life Sci., 2016, 77, 61–69 CrossRef.
- R. Wynberg, Res. Policy, 2023, 52(2), 104674 CrossRef.
- R. Wynberg, S. Ives and J. Bam, J. South. Afr. Stud., 2023, 49, 589–610 CrossRef.
- D. Schroeder, R. Chennells, C. Louw, L. Snyders and T. Hodges, Camb. Q. Healthc. Ethics, 2020, 29, 285–301 CrossRef PubMed.
- R. Wynberg, S. Afr. J. Bot., 2017, 110, 39–51 CrossRef.
- R. Wynberg, D. Schroeder and R. Chennells, Indigenous Peoples, Consent, and Benefit-Sharing: Lessons from the San-Hoodia Case, Springer, Dordrecht; New York, 2009 Search PubMed.
- X. T. Schneider, B. K. Stroil, C. Tourapi, C. Rebours, L. Novoveska, M. I. Vasquez and S. P. Gaudencio, Trends Biotechnol., 2023, 41, 1327–1331 CrossRef CAS.
- T. Efferth, M. Banerjee, N. Paul, S. Abdelfatah, J. Arend, G. Elhassan, S. Hamdoun, R. Hamm, C. Hong, O. Kadioglu, J. Nass, D. Ochwangi, E. Ooko, N. Ozenver, M. Saeed, M. Schneider, E. Seo, C. Wu, G. Yan, M. Zeino, Q. Zhao, M. Abu-Darwish, K. Andersch, G. Alexie, D. Bessarab, D. Bhakta-Guha, V. Bolzani, E. Dapat, F. Donenko, M. Efferth, H. Greten, L. Gunatilaka, A. Hussein, A. Karadeniz, H. Khalid, V. Kuete, I. Lee, L. Liu, J. Midiwo, R. Mora, H. Nakagawa, O. Ngassapa, C. Noysang, L. Omosa, F. Roland, A. Shahat, A. Saab, E. Saeed, L. Shan and S. Titinchi, Phytomedicine, 2016, 23, 166–173 CrossRef PubMed.
- B. Patwardhan, A. Vaidya and M. Chorghade, Curr. Sci., 2004, 86, 789–799 Search PubMed.
- N. Berdigaliyev and M. Aljofan, Future Med. Chem., 2020, 12, 939–947 CrossRef CAS PubMed.
- A. Broggiato, S. Arnaud-Haond, C. Chiarolla and T. Greiber, Mar. Policy, 2014, 49, 176–185 CrossRef.
- F. Rabitz, Braz. Political Sci. Rev., 2015, 9, 30–53 CrossRef.
- Y. Imran, N. Wijekoon, L. Gonawala, Y.-C. Chiang and K. R. D. De Silva, Sci. World J., 2021, 2021, 8898842 Search PubMed.
- F. Michiels, U. Feiter, S. Paquin-Jaloux, D. Jungmann, A. Braun, M. Sayoc, R. Armengol, M. Wyss and B. David, Sustainability, 2022, 14 Search PubMed.
- M. Heinrich, F. Scotti, A. Andrade-Cetto, M. Berger-Gonzalez, J. Echeverría, F. Friso, F. Garcia-Cardona, A. Hesketh, M. Hitziger, C. Maake, M. Politi, C. Spadafora and R. Spadafora, Front. Pharmacol, 2020, 11, 765 CrossRef PubMed.
- V. Sakthivel, Int. J. Legal Res. Anal., 2025, 2(7) Search PubMed.
- M. Simmonds, R. Fang, L. Wyatt, E. Bell, B. Allkin, F. Forest, R. Wynberg, M. da Silva, B. Zhang, J. Liu, Y. Qi and S. Demissew, Plants People Planet, 2020, 2, 546–556 CrossRef.
- European Code of Conduct for Research Integrity, 2023, https://allea.org/code-of-conduct/, accessed October 2025 Search PubMed.
- All European Academies, https://allea.org, accessed October 2025 Search PubMed.
- European Network of Research Integrity Offices (ENRIO), https://www.enrio.eu, accessed October 2025 Search PubMed.
- European Network for Research Ethics and Integrity (ENERI), https://eneri.eu , accessed October 2025 Search PubMed.
- European Network of Research Ethics Committees (EUREC), http://www.eurecnet.org/index.html, accessed October 2025 Search PubMed.
- ENTIRE project, http://entireconsortium.eu/en, accessed October 2025 Search PubMed.
- Pro-ethics initiative, https://eneri.eu/pro-ethics, accessed October 2025 Search PubMed.
- Path2Integrity project, https://www.path2integrity.eu, accessed October 2025 Search PubMed.
- Academic integrity, https://academicintegrity.eu/victims, accessed October 2025 Search PubMed.
- H. Desmond and K. Dierickx, Bioethics, 2021, 35, 414–428 CrossRef PubMed.
- Norway Research Ethics, https://www.forskningsetikk.no/en/resources/fostering-research-ethics/, accessed October 2025 Search PubMed.
- The Norwegian National Committee for Research Ethics in Science and Technology (NENT), https://www.forskningsetikk.no/en/about-us/our-committees-and-commission/nent/, accessed October 2025 Search PubMed.
- The Norwegian National Committee for Research Ethics in the Social Sciences and the Humanities (NESH), https://www.forskningsetikk.no/en/about-us/our-committees-and-commission/nesh/, accessed October 2025 Search PubMed.
- SATORI, https://satoriproject.eu/, accessed October 2025 Search PubMed.
- Artificial Intelligence Act, https://www.onetrust.com/blog/navigating-the-eu-ai-act/, accessed October 2025 Search PubMed.
- Data Governance Act, https://www.european-data-governance-act.com/, accessed October 2025 Search PubMed.
- EU General Data Protection Regulation, https://gdpr-info.eu/, accessed October 2025 Search PubMed.
- S. Harrop, J. Environ. Law, 2011, 23, 117–128 CrossRef.
- Nagoya Protocol Parties, https://www.cbd.int/abs/nagoya-protocol/signatories, accessed October 2025 Search PubMed.
- S. Oberthür and R. Florian, in Global Governance of Genetic Resources, ed. G. K. R. Sebastian Oberthür, Routledge, London, UK, 2014, pp. 79–95 Search PubMed.
- S. Y. Tian Zhiqi, Biodivers. Sci., 2025, 33, 24593 Search PubMed.
- K. McCluskey, K. B. Barker, H. A. Barton, K. Boundy-Mills, D. R. Brown, J. A. Coddington, K. Cook, P. Desmeth, D. Geiser, J. A. Glaeser, S. Greene, S. Kang, M. W. Lomas, U. Melcher, S. E. Miller, D. R. Nobles Jr, K. J. Owens, J. H. Reichman, M. d. Silva, J. Wertz, C. Whitworth and D. Smith, Mbio, 2017, 8(4), e00982 CrossRef PubMed.
- J. Overmann and A. Scholz, Trends Microbiol., 2017, 25, 85–88 CrossRef CAS PubMed.
- D. Neumann, A. Borisenko, J. Coddington, C. Häuser, C. Butler, A. Casino, J. Vogel, G. Haszpruna and P. Giere, Org. Divers. Evol., 2018, 18, 1–12 CrossRef.
- S.-Y. Choi, KOAJ, 2017, 41(3), 175–203 Search PubMed.
- J. Lee, M. An, J. S. LEE and Y.-H. Chang, J. Plant Biotechnol., 2021, 48, 1–11 CrossRef.
- F. Casolari, A. Westmoreland, T. Vanagt and M. Jaspars, Nat. Prod. Rep., 2025, 42, 1063–1070 RSC.
- H. Macdonald, Biopreserv. Biobank, 2025, 23, 23–30 CrossRef PubMed.
- Vienna Convention on the Law of Treaties, https://legal.un.org/ilc/texts/instruments/english/conventions/1_1_1969.pdf, accessed January 2026 Search PubMed.
- S. Oberthür and J. Pozarowska, Global Environ. Politics, 2013, 13, 100 CrossRef.
- A. Deplazes-Zemp, S. Abiven, P. Schaber, M. Schaepman, G. Schaepman-Strub, B. Schmid, K. Shimizu and F. Altermatt, Nat. Ecol. Evol., 2018, 2, 917–919 CrossRef PubMed.
- J. Colella, L. Silvestri, G. Súzan, M. Weksler, J. Cook and E. Lessa, J. Mammal., 2023, 104, 430–443 CrossRef.
- J. Koskela, B. Vinceti, W. Dvorak, D. Bush, I. Dawson, J. Loo, E. Kjaer, C. Navarro, C. Padolina, S. Bordacs, R. Jamnadass, L. Graudal and L. Ramamonjisoa, For. Ecol. Manag., 2014, 333, 22–34 CrossRef.
- Nagoya Protocol User Manual, https://bccm.belspo.be/documents/files/projects/trust/trust-handbook-march-2016.pdf, accessed October 2025 Search PubMed.
- W. Houssen, R. Sara, M. Jaspars, Deep-Ocean Stewardship Initiative (DOSI) policy brief on Digital Sequence Information on Genetic Resources: Concept, Scope and Current Use, 2020, https://www.dosi-project.org/wp-content/uploads/070-DSI-Policy-brief-V4-WEB.pdf, accessed April 2026 Search PubMed.
- A. Scholz, J. Freitag, C. Lyal, R. Sara, M. Cepeda, I. Cancio, S. Sett, A. Hufton, Y. Abebaw, K. Bansal, H. Benbouza, H. Boga, S. Brisse, M. Bruford, H. Clissold, G. Cochrane, J. Coddington, A. Deletoille, F. García-Cardona, M. Hamer, R. Hurtado-Ortiz, D. Miano, D. Nicholson, G. Oliveira, C. Bravo, F. Rohden, O. Seberg, G. Segelbacher, Y. Shouche, A. Sierra, I. Karsch-Mizrachi, J. da Silva, D. Hautea, M. da Silva, M. Suzuki, K. Tesfaye, C. Tiambo, K. Tolley, R. Varshney, M. Zambrano and J. Overmann, Nat. Commun., 2022, 13, 1086 CrossRef CAS PubMed.
- M. Leal, J. Anaya-Rojas, M. Munro, J. Blunt, C. Melian, R. Calado and M. Lürig, Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 24165–24172 CrossRef CAS PubMed.
- F. Keiper and A. Atanassova, Front. Bioeng. Biotechnol., 2020, 8, 310 CrossRef PubMed.
- J. Ambler, A. Diallo, P. Dearden, P. Wilcox, M. Hudson and N. Tiffin, Trends Biotechnol., 2021, 39, 116–125 CrossRef CAS PubMed.
- S. Laird and R. Wynberg, Nanoethics, 2016, 10, 189–200 CrossRef.
- S. Aubry, C. Frison, J. Medaglia, E. Frison, M. Jaspars, M. Rabone, A. Sirakaya, D. Saxena and E. van Zimmeren, Plants People Planet, 2022, 4, 5–12 CrossRef.
- S. Sett, W. Kress, M. Halewood, D. Nicholson, G. Nunez-Vega, D. Faggionato, M. Rouard, M. Jaspars, M. da Silva, C. Prat, D. Raposo, I. Klünker, J. Freitag, C. Tiambo, C. Ribeiro, L. Wong, H. Benbouza, J. Overmann and A. Scholz, Nat. Commun., 2024, 15, 8745 CrossRef CAS PubMed.
- D. Kharb and S. Sharma, Asian Biotechnol. Dev. Rev., 2022, 24(3), 3–22 Search PubMed.
- Conference of the Parties, https://www.cbd.int/conferences/2024/cop-16/documents, accessed October 2025 Search PubMed.
- Digital Sequence Information on Genetic Resources. https://www.cbd.int/dsi-gr/whatdone/2023-24.shtml, accessed October 2025 Search PubMed.
- B. Maia and S. Bourgeois-Gironde, Glob. Policy", 2025, 16, 751–761 CrossRef.
- M. Danielewski, M. Szalata, J. Nowak, J. Walkowiak, R. Slomski and K. Wielgus, Genes, 2025, 16(9), 1067 CrossRef CAS PubMed.
- M. Munoz-Garcia and A. Scholz, Patterns, 2025, 6(3), 101208 CrossRef PubMed.
- C. Blom, M. Jaspars, J. Rilak and A. Scholz, Nat. Biotechnol., 2025, 43, 1599–1602 CrossRef CAS PubMed.
- C. Tourapi, E. Christoforou, S. Gaudencio and M. Vasquez, Mar. Drugs, 2024, 22(9), 427 CrossRef PubMed.
- L. Liggins, M. Hudson and J. Anderson, Mol. Ecol., 2021, 30, 2477–2482 CrossRef PubMed.
- J. Kuzma and P. Roberts, J. Responsible Innov., 2018, 5, 338–346 CrossRef.
- M. Rouard, A. Scholz and M. Halewood, Trends Genet., 2025, 41, 451–455 CrossRef CAS PubMed.
- C. Seitz, Digital sequence information-legal questions for patent, copyright, trade secret protection and sharing of genomic sequencing data, in Proceedings of the 1st International Conference on Genetic Resources and Biotechnology, 2020 Search PubMed.
- A. de Lima, C. Juliao, E. da Silva, A. Dias, J. de Jesus, M. da Silva, E. Almada, J. de Andrade, N. Bensusan, S. Zank and G. Soldati, Nat. Commun., 2024, 15, 8933 CrossRef PubMed.
- J. Golan, K. Riddle, M. Hudson, J. Anderson, N. Kusabs and T. Coltman, Front. Genet., 2022, 13, 1014044 CrossRef PubMed.
- ELIXIR project, https://elixir-europe.org/platforms/data/core-data-resources, accessed October 2025 Search PubMed.
- C. Lawson, F. Humphries and M. Rourke, J. Intell. Prop. L. & Pract., 2024, 19, 337–357 Search PubMed.
- J. Strand, H. Thomsen, J. Jensen, C. Marcussen, T. Nicolajsen, M. Skriver, I. Sogaard, T. Ezaz, S. Purup, H. Callesen and C. Pertoldi, Conserv. Genet. Resour., 2020, 12, 709–725 CrossRef.
- J. Overmann, Syst. Appl. Microbiol., 2015, 38, 258–265 CrossRef PubMed.
- D. Smith, M. da Silva, J. Jackson and C. Lyal, Microbio. SGM, 2017, 163, 289–296 CrossRef CAS PubMed.
- C. Paulus, Y. Rebets, B. Tokovenko, S. Nadmid, L. P. Terekhova, M. Myronovskyi, S. B. Zotchev, C. Rueckert, S. Braig, S. Zahler, J. Kalinowski and A. Luzhetskyy, Sci. Rep., 2017, 7, 42382 CrossRef CAS PubMed.
- K. Boundy-Mills, E. Glantschnig, I. Roberts, A. Yurkov, S. Casaregola, H. Daniel, M. Groenewald and B. Turchetti, Yeast, 2016, 33, 243–260 CrossRef CAS PubMed.
- H. Harden-Davies and P. Snelgrove, Front. Mar. Sci., 2020, 7, 40 CrossRef.
- B. Santos, S. Devereaux, K. Gjerde, K. Chand, J. Martinez and L. Crowder, Front. Mar. Sci., 2022, 9, 1001240 CrossRef.
- H. Harden-Davies, D. Amon, T. Chung, J. Gobin, Q. Hanich, K. Hassanali, M. Jaspars, A. Pouponneau, K. Soapi, S. Talma and M. Vierros, Aquat. Conserv.: Mar. Freshw. Ecosyst., 2022, 32, 907–912 CrossRef.
- Sustainability and Development Goals- Goal 14, https://sdgs.un.org/goals/goal14,accessed October 2025 Search PubMed.
- European Green Deal, https://ec.europa.eu/info/strategy/priorities-2019-2024/european-green-deal_en, accessed October 2025 Search PubMed.
- One Health, https://www.who.int/health-topics/one-health#tab=tab_1, accessed October 2025 Search PubMed.
- EU Biodiversity Strategy for 2030, https://ec.europa.eu/environment/strategy/biodiversity-strategy-2030_en, accessed October 2025 Search PubMed.
- European Commission, Study on the economic benefits of marine protected areas – Literature review analysis, Publications Office, 2018, https://maritime-spatial-planning.ec.europa.eu/sites/default/files/ea0318223enn.en__0.pdf, accessed October 2025 Search PubMed.
- Global Biodiversity, https://environment.ec.europa.eu/topics/nature-and-biodiversity/global-biodiversity_en, accessed October 2025 Search PubMed.
- Maritime Spatial Planning, https://oceans-and-fisheries.ec.europa.eu/ocean/blue-economy/maritime-spatial-planning_en, accessed October 2025 Search PubMed.
- Report from the commission to the european parliament and the council outlining the progress made in implementing Directive 2014/89/EU establishing a framework for maritime spatial planning, https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX:52022DC0185, accessed October 2025 Search PubMed.
- EMFAF, https://oceans-and-fisheries.ec.europa.eu/funding-and-investment/emfaf_en, accessed October 2025 Search PubMed.
- Blue Forum, https://blueforum.org/, accessed October 2025 Search PubMed.
- L. Lallier, O. McMeel, T. Greiber, T. Vanagt, A. Dobson and M. Jaspars, Nat. Prod. Rep., 2014, 31, 612–616 RSC.
- A. Abuelenin, Biodivers. Conserv., 2025, 35, 3 CrossRef.
- High Seas Treaty, https://www.un.org/bbnjagreement/en, accessed January 2026 Search PubMed.
- K. Grorud-Colvert and J. Sullivan-Stack, Science, 2026, 391, 219 CrossRef CAS PubMed.
- F. Humphries, in Decoding Marine Genetic Resource Governance Under the BBNJ Agreement, ed. F. Humphries, Springer Nature Switzerland, Cham, 2025, pp. 55–94 Search PubMed.
- A. E. L. Brown, The Place of Intellectual Property Under the BBNJ Agreement, Springer, Cham, 2025 Search PubMed.
- R. Pedrosa, S. P. Gaudencio and V. Vasconcelos, Mar. Drugs, 2020, 18(1), 40 CrossRef PubMed.
- J. Watson, G. Clark and D. Williams, Rev. Sci. Tech. – Off. Int. Epizoot, 2021, 40, 253–259 CAS.
- V. Urovi, V. Jaiman, A. Angerer and M. Dumontier, Blockchain: Res. Appl., 2022, 3(4), 100102 Search PubMed.
- M. Bond and D. Scott, Geoforum, 2020, 117, 24–32 CrossRef PubMed.
- G. Avilés-Polanco, M. Almendarez-Hernández, L. Beltrán-Morales, I. Serrano-Fraire and A. Ortega-Rubio, Land, 2021, 10(4), 412 CrossRef.
- F. Friso, F. Mendive, M. Soffiato, V. Bombardelli, A. Hesketh, M. Heinrich, L. Menghini and M. Politi, J. Ethnopharmacol., 2020, 259, 112885 CrossRef PubMed.
- H. Lewin, G. Robinson, W. Kress, W. Baker, J. Coddington, K. Crandall, R. Durbin, S. Edwards, F. Forest, M. Gilbert, M. Goldstein, I. Grigoriev, K. Hackett, D. Haussler, E. Jarvis, W. Johnson, A. Patrinos, S. Richards, J. Castilla-Rubio, M. van Sluys, P. Soltis, X. Xu, H. Yang and G. Zhang, Proc. Natl. Acad. Sci. U. S. A., 2018, 115, 4325–4333 CrossRef CAS PubMed.
- Earth BioGenome Project, https://www.earthbiogenome.org/, accessed October 2025 Search PubMed.
- Basecamp Research, https://basecamp-research.com/, accessed January 2026 Search PubMed.
- F. Lin, GEN Edge, 2025, 7, 402–405 CrossRef.
- D. Robinson and M. Raven, Aust. Geogr., 2017, 48, 311–331 CrossRef.
- M. Heinrich and A. Hesketh, Phytomedicine, 2019, 53, 332–343 CrossRef PubMed.
- J. Richstone, Canadian Public Administration-Administration Publique du Canada, 1998, vol. 41, pp. 333–334 Search PubMed.
- A. Giunta, Int. J. Hum. Rights, 2019, 23, 149–173 CrossRef.
- A. Vrdoljak, Int. J. Cult. Prop., 2018, 25, 245–281 CrossRef.
- D. Mbwisi, Verfassung in Recht und Übersee, 2023, vol. 56, pp. 371–395 Search PubMed.
- P. Batista, J. Intell. Prop. L. & Pract., 2024, 19, 328–336 Search PubMed.
- I. Goss, Harv. Int'l L.J., 2025, 66, 35–74 Search PubMed.
- C. Oguamanam, Afr. J. Inform. Commun., 2019, 23, 1–24 Search PubMed.
- P. Drahos, Telecommun. Policy, 1997, 21, 201–211 CrossRef.
- R. Okediji, Harv. Int'l L.J., 2025, 66, 1–34 Search PubMed.
- ICG Gap Analysis, https://www.wipo.int/edocs/mdocs/tk/en/wipo_grtkf_ic_13/wipo_grtkf_ic_13_5_b_rev.pdf, accessed October 2025 Search PubMed.
- ICG Gap Analysis 2018, https://www.wipo.int/edocs/mdocs/tk/en/wipo_grtkf_ic_37/wipo_grtkf_ic_37_6.pdf, accessed October 2025 Search PubMed.
- Online Databases and Registries of Traditional Knowledge and Genetic Resources, https://www.wipo.int/en/web/traditional-knowledge/resources/db_registry, accessed October 2025 Search PubMed.
- Documenting Traditional Knowledge – A Toolkit, https://www.wipo.int/edocs/pubdocs/en/wipo_pub_1049.pdf, accessed October 2025 Search PubMed.
- India Biological Diversity Act, https://www.wipo.int/wipolex/en/legislation/details/23111?utm, accessed October 2025 Search PubMed.
- Traditional Knowledge Digital Library, https://www.tkdl.res.in, accessed October 2025 Search PubMed.
- Honey Bee Network Database, https://www.iapad.org/wp-content/uploads/2015/07/Protection-of-TK.pdf, accessed October 2025 Search PubMed.
- Law no 27811, https://www.wipo.int/wipolex/en/legislation/details/3420, accessed October 2025 Search PubMed.
- S. Balaji and A. Fish, ShermanBrad, 2025, 11, 4–5 Search PubMed.
- J. Nielsen and D. Nicol, J. Law Biosci., 2024, 11(2) DOI:10.1093/jlb/lsae024.
- D. Thaldar, Dev. World Bioeth., 2025, 25, 16–23 CrossRef PubMed.
- S. Thambisetty, SSRN Electron. J., 2023 DOI:10.2139/ssrn.4081285.
- EU Action Plan on Human Rights and Democracy, https://www.eeas.europa.eu/eeas/eu-action-plan-human-rights-and-democracy-0_en, accessed October 2025 Search PubMed.
- EU External Action. https://www.eeas.europa.eu/_en, acessed February 2025 Search PubMed.
- EU Arctic Forum. https://oceans-and-fisheries.ec.europa.eu/events/eu-arctic-forum-indigenous-peoples-dialogue-and-arctic-youth-dialogue-2025-06-26_en, acessed February 2025 Search PubMed.
- M. Hassan, P. Olliaro, E. Torreele, J. Caoili, M. Bockarie, S. Lee and A. Zumla, Int. J. Infect. Dis., 2025, 158, 107944 CrossRef PubMed.
- WHO Pandemic Agreement. https://www.who.int/health-topics/who-pandemic-agreement#tab=tab_1, acessed January 2026 Search PubMed.
- K. A. Abdel-Motaal, W. A. El Kheir-Mataria and S. Chun, Int. J. Environ. Res. Public Health, 2025, 22(12), 1807 CrossRef PubMed.
- The global agreement on pandemics in a nutshell, https://www.consilium.europa.eu/en/infographics/towards-an-international-treaty-on-pandemics/, accesssed April 2026 Search PubMed.
- Intergovernmental Negotiating Body to draft and negotiate a WHO convention, https://apps.who.int/gb/ebwha/pdf_files/WHA78/A78_10-en.pdf, accesssed April 2026 Search PubMed.
- L. Chen, China CDC Wkly, 2026, 8, 55–57 Search PubMed.
- A. Damodaran, J. Intellect. Prop. Rights, 2008, 13, 509–513 Search PubMed.
- Geographical Indications, https://www.wipo.int/geo_indications/en/, accessed October 2025 Search PubMed.
- TRIPS, Geographical Indications, https://www.wto.org/english/tratop_e/trips_e/gi_background_e.htm, accessed October 2025 Search PubMed.
- Trademarks, https://www.wipo.int/tk/en/, accessed October 2025 Search PubMed.
- Copyright, https://www.wipo.int/en/web/copyright, accessed October 2025 Search PubMed.
- Patents, https://www.wipo.int/en/web/patents, accessed October 2025 Search PubMed.
- D. Jefferson and D. Robinson, J. World Intellect. Prop., 2025, 28, 640–664 CrossRef.
- K. Geethanjali and A. Shelke, Journal of World Intellectual Property, 2025 Search PubMed.
- C. Masango, Inform. Develop., 2020, 36, 327–338 CrossRef.
- N. Kolowratnik, Andares Revista de Derechos Humanos y de la Naturaleza, 2023, 26–39 Search PubMed.
- N. Mukwevho, South Afr. J. Hum. Right, 2022, 38, 144–162 CrossRef.
- C. N. Saha and S. Bhattacharya, J. Adv. Pharm. Technol. Res., 2011, 2, 88–93 CrossRef PubMed.
- R. C. Dreyfuss, J. Nielsen and D. Nicol, J. Law Biosci., 2018, 5, 550–589 CrossRef PubMed.
- Y. Joly and P. Tonin, New Genet. Soc., 2014, 33, 167–180 CrossRef.
- J. Sherkow and H. Greely, Annu. Rev. Genet., 2015, 49, 161–182 CrossRef CAS PubMed.
- EU. Directive 98/44/EC on the legal protection of biotechnological inventions, https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:31998L0044, accessed February 2026 Search PubMed.
- T. Caulfield, E. Gold and M. Cho, Nat. Rev. Genet., 2000, 1, 227–231 CrossRef CAS PubMed.
- Patent Exclusions, https://www.wipo.int/edocs/mdocs/scp/en/scp_15/scp_15_3-annex3.pdf, accessed October 2025 Search PubMed.
- B. Tenni, H. Moir, B. Townsend, B. Kilic, A. Farrell, T. Keegel and D. Gleeson, Glob. Health, 2022, 18(1), 40 CrossRef PubMed.
- European Patent Convention, https://link.epo.org/web/EPC_17th_edition_2020_en.pdf, accessed October 2025 Search PubMed.
- European Patent Office, https://www.epo.org/en, accessed October 2025 Search PubMed.
- V. Sinaga, J. East Asia & Int'l L., 2022, 15, 331–348 Search PubMed.
- Euroasian Patente Office, https://www.eapo.org/en/about-eapo/eurasian-patent-office/, https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:31998L0044, accessed October 2025 Search PubMed.
- Gulf Cooperation Council Patent Office, https://www.gccpo.org/DefaultEn, accessed October 2025 Search PubMed.
- P. Oldham, S. Hall and O. Forero, PLoS One, 2013, 8(11), e78737 CrossRef CAS PubMed.
- T. Taiepa, P. Lyver, P. Horsley, J. Davis, M. Bragg and H. Moller, Environ. Conserv., 1997, 24, 236–250 CrossRef.
- D. Zografos, IIC Int. Rev. Intellect. Prop. Compet. Law, 2005, 36, 928–952 Search PubMed.
- D. Jefferson, J. World Intellect. Prop., 2024, 27, 91–111 CrossRef.
- New Zealand Intellectual Property Office, https://www.iponz.govt.nz/, accessed October 2025 Search PubMed.
- E. Stokes, Appl. Geogr., 1992, 12, 176–191 CrossRef.
- E. Wright and D. Robinson, J. World Intellect. Prop., 2024, 27, 222–241 CrossRef.
- WIPO Lex Database, https://www.wipo.int/wipolex/en/main/legislation, acessed February 2025 Search PubMed.
- S. Wang, Inform. Develop., 2025, 1–23 Search PubMed.
- J. Brander, V. Cui and I. Vertinsky, J. Int. Bus. Stud., 2017, 48, 908–921 CrossRef.
- S. Alshareef, J. Econ. Issues, 2023, 57, 1018–1035 CrossRef.
- R. Gabble and J. Kohler, Glob. Health, 2014, 10, 3 CrossRef PubMed.
- C. Wu, Asian J. WTO & Int'l Health L & Pol'y, 2010, 5, 141–205 Search PubMed.
- A. Shah, J. Warsh and A. Kesselheim, J. Law Med. Ethics, 2013, 41, 841–851 CrossRef PubMed.
- R. Anuradha, Econ. Polit. Wkly., 1998, 33, 1615–1619 Search PubMed.
- Sharing with the Kani, https://www.cbd.int/financial/bensharing/india-kanis.pdf, accessed January 2026 Search PubMed.
- L. Amusan, Afr. J. Tradit. Complement. Altern. Med., 2017, 14, 103–109 CrossRef PubMed.
- E. Schenberg and K. Gerber, Transcult. Psychiatry, 2022, 59, 610–624 CrossRef PubMed.
- K. Tupper, Glob. Netw., 2009, 9, 117–136 CrossRef.
- A. Tsioumanis, K. Mattas and E. Tsioumani, J. Agric. Environ. Ethics, 2003, 16, 605–616 CrossRef.
- D. A. Posey and G. Dutfield, Beyond Intellectual Property: Toward Traditional Resource Rights for Indigenous Peoples and Local Communities, International Development Research Centre Publisher, 1947, Ottawa Search PubMed.
- D. Singh, BMJ, 2003, 327, 890 Search PubMed.
- U. Hellerer and K. S. Jarayaman, Nature, 2000, 405, 267 CrossRef PubMed.
- V. Shiva, Protect Or Plunder?: Understanding Intellectual Property Rights, Zed Books, 2001 Search PubMed.
- R. Wynberg, S. Laird, J. van Niekerk and W. Kozanayi, Soc. Nat. Resour., 2015, 28, 559–574 CrossRef.
- P. Cox, H. Johnson and G. Tavana, Science, 2008, 320, 1589 CrossRef CAS PubMed.
- P. Cox, Pharm. Biol., 2001, 39, 33–40 CAS.
- European Commission, Directorate-General for Research and, European Group on Ethics in science and new technologies – General activity report – 2017-2021, Publications Office, 2021 Search PubMed.
- European Commission, European Group on Ethics in Science and New, Opinion on ethical aspects of patenting inventions involving human stem cells – Opinion No 16 – 7 May 2002, Publications Office of the European Union, 2002 Search PubMed.
- General Guidelines for Methodologies on Research and Evaluation of Traditional Medicine, https://iris.who.int/handle/10665/66783, accessed October 2025 Search PubMed.
- I. Noriega, M. Halewood, M. Abberton, A. Amri, I. Angarawai, N. Anglin, M. Blümmel, B. Bouman, H. Campos, D. Costich, D. Ellis, P. Gaur, L. Guarino, J. Hanson, V. Kommerell, L. Kumar, C. Lusty, M. Ndjiondjop, T. Payne, M. Peters, E. Popova, G. Prakash, R. Sackville-Hamilton, R. Tabo, H. Upadhyaya, M. Yazbek and P. Wenzl, Crop Sci., 2019, 59, 819–832 CrossRef.
Footnote |
| † Sui generis (latin for “of its own kind”) refers to a specially tailored form of IPP designed to protect TK or GR in ways not fully covered by conventional IP systems. |
| This journal is © The Royal Society of Chemistry 2026 |