DOI:
10.1039/D5NJ03370E
(Paper)
New J. Chem., 2026, Advance Article
A naphthalimide–benzimidazole conjugate for the light-up fluorescence detection of Ga3+ and DCP
Received
20th August 2025
, Accepted 28th January 2026
First published on 4th February 2026
Abstract
Gallium ions (Ga3+) play a crucial role in human life due to their remarkable physical, chemical, and biological properties, with applications ranging from electronic devices and solar cells to anticancer therapeutics, owing to their strong affinity for tumor tissues. However, increasing industrial and biomedical use has led to environmental accumulation, making Ga3+ exposure a concern. In this work, we report a novel naphthalimide-based reversible chelating ligand (NaHB) capable of selectively recognizing both Ga3+ ions and the nerve agent diethyl chlorophosphate (DCP) in a CH3CN/H2O medium. The binding interactions were systematically studied using UV-vis absorption, fluorescence spectroscopy, NMR, and high-resolution mass spectrometry. NaHB displays an instantaneous “switch-on” fluorescence response toward Ga3+ and DCP, with binding constants (Ka) of 5.15 × 104 M−1 and 1.2 × 105 M−1, respectively. Job's plot analysis confirmed a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry for both systems. The practical utility of NaHB was demonstrated through paper strip assays and quantitative detection of Ga3+ and DCP in real-field water samples. Furthermore, the NaHB–Ga3+ complex was explored for molecular logic gate applications, showcasing its multifunctional sensing capability.
1 stoichiometry for both systems. The practical utility of NaHB was demonstrated through paper strip assays and quantitative detection of Ga3+ and DCP in real-field water samples. Furthermore, the NaHB–Ga3+ complex was explored for molecular logic gate applications, showcasing its multifunctional sensing capability.
1. Introduction
Over the past few decades, chemosensors have gained much interest in sensing of ions and molecules that play a crucial role in the environment.1–7 Gallium is a rare element found in trace amounts as salts in various ores and soil, but it has an imperative impact on human lives.8–10 The manufacturing of integrated circuits, semiconductor devices, and optoelectronic components, including light-emitting diodes, solar cells, and photodetectors, heavily relies on the utilization of gallium salts.11–14 Gallium compounds are employed clinically as markers for diagnosis and therapeutic agents because of their capacity to target tumors. Ga3+ ions also have a great affinity for tumors, and their nitrate salts have long been used as antitumor agents. The presence of residues of these substances in the body or environment is dangerous. Their usage as chemotherapy treatment agents is a sign of their possible cell-damaging effects. Ga3+ exposure in occupational contexts and due to pollution in the environment can have adverse effects on the kidneys, the immunological system, the heart, and the testicles.15–17 As a result, the requirement for identifying the presence of Ga3+ has increased and sparked academic curiosity, not only because of its function as a pollutant, but because it can also be used to track cancer treatment responses and monitor tumors.18–20 For both health and financial reasons, it is urgently necessary to develop precise and effective technology to detect traces of gallium.
On the other hand, chemical warfare nerve agents (CWNAs) are a class of OPs (organophosphates) with a phosphate or phosphite group that is extremely poisonous. Even a trace amount of inhalation can cause death in a short time.21,22 OPs are generally utilized as pesticides and herbicides in agriculture because they have chemically reactive phosphate ester side chains.23 Sarin (GB), soman (GD), and tabun (GA) are found to be three extremely lethal organophosphate nerve agents. The reactive phosphate groups of these compounds can interact with the hydroxyl groups of the acetylcholinesterase (AChE) enzyme. Acetylcholine (ACh), a neurotransmitter found in postsynaptic membranes and neuromuscular junctions, is hydrolyzed by AChE. When acetylcholine breakdown is blocked, it causes a neurological imbalance that swiftly results in organ failure, dysfunction of the central nervous system, and death.21,24–26 Diethyl chlorophosphate (DCP), a related substance, is frequently utilized to simulate sarin for scientific studies. Comparison of DCP with real nerve agents reveals that it has a similar level of responsiveness but is somewhat less hazardous.27,28 Moreover, organophosphates are routinely employed in agriculture to control pests despite being harmful to human health.29 These compounds need to be closely watched because there is a considerable likelihood that they will accumulate within the food system as a result of agricultural usage. Hence, organophosphate contaminants found in consumer goods appear to be extremely dangerous to human health. Due to their lack of colour, flavour, and smell, these chemicals are difficult to identify.30
Research groups have already developed methodologies based on colorimetry, fluorometry, and enzymatic assays to identify organophosphates.31–34 Because of their selectivity, sensitivity, quick reaction, and on-site analysis, fluorometric approaches have gained more attention. Herein, we report the synthesis of a fluorescent probe (NaHB) incorporating a naphthalimide core conjugated with a benzimidazole unit, featuring a donor hydroxyl (–OH) group and an acceptor nitrogen atom of the benzimidazole. The system undergoes excited-state intramolecular proton transfer (ESIPT), in which the proton from the hydroxyl group in the ground-state enol form is transferred to the benzimidazole nitrogen in the excited state, generating the keto tautomer. This tautomerization process is stabilized by intramolecular hydrogen bonding, enabling distinct photophysical properties. The complexation of NaHB + Ga3+ and also the phosphorylation of NaHB in the presence of DCP restricted the ESIPT process, resulting in significant enhancement of the fluorescence signal. Hence, a probe has been designed exhibiting high sensitivity and superfast response towards Ga3+ and DCP in the liquid state, followed by quantification in real field water samples.
The NaHB chemosensor was rationally designed by fusing two photofunctional units—naphthalimide and benzimidazole—to harness the synergistic effects of intramolecular charge transfer (ICT) and excited-state intramolecular proton transfer (ESIPT) within a single molecular framework. The naphthalimide moiety serves as a robust, π-conjugated fluorophore with strong absorption in the visible region and high photostability, acting as an electron acceptor. The benzimidazole unit, on the other hand, introduces electron-donating nitrogen sites and a proton-active –NH group, which collectively enable both metal coordination and ESIPT behaviour. This hybrid structure allows the probe to interact with analytes through two orthogonal mechanisms: (i) chelation with Ga3+ ions, where coordination to the nitrogen and carbonyl oxygen atoms suppresses non-radiative PET/ESIPT processes, leading to a chelation-enhanced fluorescence (CHEF) response and (ii) electrophilic interaction with DCP, in which the phosphorylation at the –OH site perturbs the ESIPT equilibrium, producing a distinct emission modulation. The deliberate fusion of these two responsive motifs within the NaHB framework thus enables selective and differential optical responses toward Ga3+ and DCP, establishing a rational molecular design concept for future multifunctional sensing systems.
2. Experimental section
2.1 Materials and methods
Chemicals used for synthesis were purchased from Loba Chemie, Sigma-Aldrich, and Spectrochem depending upon their availability. Spectroscopic grade solvents were purchased from Spectrochem and Rankem Ltd and were used without any further purification. Reaction progress was monitored using TLC (thin layer chromatography).
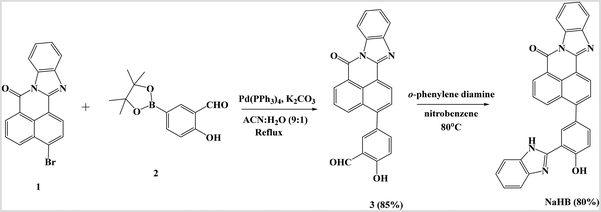
2.2 Synthesis of 3-bromo-7H-benzo[de]benzo[4,5]imidazo[2,1-a]isoquinolin-7-one (compound 3)
Compounds 1 and 2 were synthesized in accordance with the previously reported literature methods.35,36 For the synthesis of compound 3, compound 1 (200 mg, 0.575 mmol) and compound 2 (143 mg, 0.575 mmol) were dissolved in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) (10 ml) in the presence of K2CO3 (100 mg, 0.724 mmol) and Pd(PPh3)4 (6 mg, 0.005 mmol) and subjected to the Suzuki–Miyaura cross-coupling reaction (Scheme 1). The reaction mixture was refluxed for 8 h until the starting materials were completely consumed, and the reaction progress was monitored through TLC. After completion, the reaction mixture was cooled to room temperature. The excess solvent was evaporated, followed by the addition of 50 ml of cold water. The precipitates formed were filtered to obtain a crude product, which was purified by column chromatography using CHCl3
1) (10 ml) in the presence of K2CO3 (100 mg, 0.724 mmol) and Pd(PPh3)4 (6 mg, 0.005 mmol) and subjected to the Suzuki–Miyaura cross-coupling reaction (Scheme 1). The reaction mixture was refluxed for 8 h until the starting materials were completely consumed, and the reaction progress was monitored through TLC. After completion, the reaction mixture was cooled to room temperature. The excess solvent was evaporated, followed by the addition of 50 ml of cold water. The precipitates formed were filtered to obtain a crude product, which was purified by column chromatography using CHCl3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc (80
EtOAc (80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20) as an eluent to afford the pure yellow colored product 3 in 85% yield. M.pt. >300 °C. 1H NMR of compound 3 (CDCl3, 400 MHz): δ (ppm) 11.2 (d, J = 7.36 Hz, 1H, OH), 10.01 (s, 1H, CHO), 8.89 (d, J = 6.4 Hz, 1H, ArH), 8.83 (m, 1H, ArH), 8.57 (m,1H, ArH), 8.14 (d, J = 8.48 Hz, 1H, ArH), 7.89 (t, 1H, ArH), 7.75 (m, 4H, ArH), 7.50 (m, 2H, ArH), 7.20 (m, 1H, ArH) (Fig. S1). 13C NMR of compound 3 (CDCl3, 100 MHz): δ (ppm) 196.5, 191.8, 161.9, 145.6, 145.2, 135.8, 135.3, 132.3, 131.7, 130.5, 130.4, 128.1, 127.1, 120.8, 118.6 (Fig. S2). HRMS (ESI-TOF): (m/z) [M + H]+ calcd for C25H15N2O3: 391.1004, found: 391.1010.
20) as an eluent to afford the pure yellow colored product 3 in 85% yield. M.pt. >300 °C. 1H NMR of compound 3 (CDCl3, 400 MHz): δ (ppm) 11.2 (d, J = 7.36 Hz, 1H, OH), 10.01 (s, 1H, CHO), 8.89 (d, J = 6.4 Hz, 1H, ArH), 8.83 (m, 1H, ArH), 8.57 (m,1H, ArH), 8.14 (d, J = 8.48 Hz, 1H, ArH), 7.89 (t, 1H, ArH), 7.75 (m, 4H, ArH), 7.50 (m, 2H, ArH), 7.20 (m, 1H, ArH) (Fig. S1). 13C NMR of compound 3 (CDCl3, 100 MHz): δ (ppm) 196.5, 191.8, 161.9, 145.6, 145.2, 135.8, 135.3, 132.3, 131.7, 130.5, 130.4, 128.1, 127.1, 120.8, 118.6 (Fig. S2). HRMS (ESI-TOF): (m/z) [M + H]+ calcd for C25H15N2O3: 391.1004, found: 391.1010.
 |
| | Scheme 1 Synthetic route for NaHB. | |
2.3 Synthesis of 3-(3-(1H-benzo[d]imidazol-2-yl)-4-hydroxyphenyl)-7H-benzo[de]benzo[4,5]imidazo[2,1-a]isoquinolin-7-one (NaHB)
To a stirred solution of compound 3 (200 mg, 0.512 mmol) in nitrobenzene (5 ml), o-phenylenediamine (60 mg, 0.55 mmol) was added and the reaction mixture was heated to 80 °C for 6 h. After completion of the reaction, monitored by TLC, hexane (30 ml) was added to the reaction mixture, and the solid product was separated, which was then filtered off and washed with hexane and diethyl ether. After vacuum drying, the pure yellow solid of NaHB was obtained in 80% yield. M.pt. >300 °C. 1H NMR of NaHB (CDCl3, 400 MHz): δ (ppm) 8.25 (m, 2H, ArH), 7.93 (m, 2H, ArH), 7.78 (m, 1H, ArH), 7.33 (m, 3H, ArH), 7.04 (m, 2H, ArH), 6.93 (m, 2H, ArH), 6.70 (m, 4H, ArH). 13C NMR of NaHB (CDCl3, 100 MHz): δ (ppm) 200.5, 191.7, 191.5, 163.4, 145.4, 142.6, 138.4, 135.8, 135.2, 131.8, 130.8, 130.5, 129.8, 129.6, 127.1, 119.6, 119.0. HRMS (ESI-TOF): (m/z) [M + H]+ calcd for C31H19N4O2: 479.1508, found: 479.1506 (Fig. S3–S5).
2.4 Instruments
1H NMR and 13C NMR spectra were recorded on a JEOL ECS-400 MHz spectrometer at ambient temperature in CDCl3 with TMS as an internal reference. All chemical shifts were reported in ppm with respect to the reference. The mass spectra of the synthesized compounds were recorded using an XEVO G2-XS QTOF from Waters. The absorption spectra were recorded on a SHIMADZU-2600 spectrophotometer using quartz cuvettes of 1 cm path length. The fluorescence spectra were recorded on a Varian Cary Eclipse fluorescence spectrophotometer using a slit width (excitation = 20 nm, emission = 20 nm) at the stated excitation. The lifetime decay studies were executed on a DeltaFlex modular fluorescence lifetime spectrofluorimeter (HORIBA Scientific).
2.5 Spectroscopic studies
The photophysical properties of NaHB were studied by preparing a stock solution of 1 mM using dimethyl sulfoxide. UV-visible and fluorescence studies with various metal ions (Na+, K+, Mg2+, Al3+, Ca2+, Fe2+, Fe3+, Co2+, Ni2+, Cu2+, Zn2+, Ga3+, Cd2+, Ba2+, Hg2+ and Pb2+) as their perchlorate salts, anions (CH3COO−, F−, Cl−, Br−, CN−, H2PO4−, HSO4−, NO3−, AcO−, ClO−, P2O74−, and SCN− as tetra butyl ammonium salts (concentration = 10−1 M)) and nerve agents (DCP (diethyl chloro phosphate), TEP (triethyl phosphate), and N-BP (N-butyl phosphate)) were performed in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) to illustrate the ion sensing behaviour of NaHB. The stoichiometry of the complex was determined by Job's plot. Further titration studies were done to determine the detection limit and the binding constant values of the complex formed.
1) to illustrate the ion sensing behaviour of NaHB. The stoichiometry of the complex was determined by Job's plot. Further titration studies were done to determine the detection limit and the binding constant values of the complex formed.
2.6 Determination of the detection limit
The limit of detection (LOD) was calculated based on the fluorescence titrations. To determine the S/N ratio, the emission intensity of NaHB (10 µM) was measured in triplicate (n = 3). Standard deviations (SDs) were calculated from replicate data, and 95% confidence intervals were determined using Student's t-distribution. The detection limit was then calculated using eqn (1).| | |
Detection limit (LOD) = 3σ/k
| (1) |
where σ is the standard deviation of blank measurements and k is the slope of the intensity versus sample concentration calibration curve. Stability constants were determined using the Benesi–Hildebrand equation (eqn (2)).| |
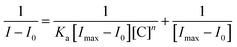 | (2) |
Here I0, I, and Imax are the absorption/emission intensities of the ligand in the absence of an analyte, at an intermediate analyte concentration, and at a concentration representing complete interaction with an analyte, respectively. Ka is the binding constant, C is the concentration of analyte, and n is the number of analytes bound per molecule.
2.7 Computational study
Geometry optimization of NaHB at the ground state (S0) was carried out using density functional theory (DFT) at the B3LYP/6-311g(d) level.37 The solvation effect of acetonitrile was used to accompany the experimental environment and was estimated using the IEFPCM model. The stability of molecular structures was confirmed with no imaginary vibration frequencies. Furthermore, the time-dependent DFT (TDDFT) method was used to calculate the excitation energies. The structural optimization and excitation energy calculation were also carried out using different basis sets at the same level of functional theory (6-311g(d) and 6-311g(d,p)), where the outcomes of B3lyp/6-311g(d) were near to observed results. Thus, for all subsequent calculations, including geometry optimization and construction of potential energy curves (PECs) for the proton transfer at S0 and the first excited state (S1), we use B3lyp/6-311g(d). All DFT and TDDFT calculations were carried out using Gaussian 16.38
2.8 Paper strip fabrication
The strips were prepared by immersing Whatman filter paper (grade 1) in a stock solution of probe NaHB (1 × 10−3 M) for 5 min, and then they were air-dried and stored under dark, airtight conditions at room temperature. The resulting changes in the colour, by dropwise addition of ions, were observed under 365 nm UV light, allowing the detection of Ga3+ and DCP. The strips retained over 90% of their fluorescence intensity after 15 days, confirming good stability.
3. Results and discussion
3.1 Photophysical studies
Photophysical characteristics of NaHB were investigated using absorption and emission spectroscopic techniques. The absorption maximum for NaHB (10 µM, CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9; v/v)) was observed at 402 nm. NaHB upon excitation at 402 nm showed emission at 520 nm with a Stokes shift of 118 nm (Fig. 1). In order to study the effect of solvents, absorption and emission spectra of NaHB were examined in a variety of solvents (Table 1). While the solvent polarity was increased, a redshift of 25 nm from 398 nm (cyclohexane) to 423 nm (water) was found in the case of absorption spectra (Fig. 2(a)). Also, NaHB extended its emission maxima region from 510 nm (cyclohexane) to 630 nm (water) as the solvent's polarity increased (Fig. 2(b)). This clearly demonstrates that upon increasing polarity, the excited state becomes more stabilized. The charge transfer could be a possible reason for the expansion of emission spectra in polar solvents. The noticeable emission colour change with solvent polarity was observed, in line with spectral shifts (Fig. 2(c)).
9; v/v)) was observed at 402 nm. NaHB upon excitation at 402 nm showed emission at 520 nm with a Stokes shift of 118 nm (Fig. 1). In order to study the effect of solvents, absorption and emission spectra of NaHB were examined in a variety of solvents (Table 1). While the solvent polarity was increased, a redshift of 25 nm from 398 nm (cyclohexane) to 423 nm (water) was found in the case of absorption spectra (Fig. 2(a)). Also, NaHB extended its emission maxima region from 510 nm (cyclohexane) to 630 nm (water) as the solvent's polarity increased (Fig. 2(b)). This clearly demonstrates that upon increasing polarity, the excited state becomes more stabilized. The charge transfer could be a possible reason for the expansion of emission spectra in polar solvents. The noticeable emission colour change with solvent polarity was observed, in line with spectral shifts (Fig. 2(c)).
 |
| | Fig. 1 Normalized absorption and emission spectra of NaHB (10 µM, CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9 H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1); λex = 402 nm). 1); λex = 402 nm). | |
Table 1 Photophysical behaviour of NaHB (10 µM) in solvents of different polarities
| Solvent |
λmax (nm) (abs) |
Molar absorptivity constant ε = M−1 cm−1 |
λem (nm) |
Stokes shift Δν (cm−1) |
Quantum yield (ϕ) |
| Cyclohexane |
394 |
5100 |
481 |
4590 |
0.13 |
| Toluene |
410 |
24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
560 |
6530 |
0.017 |
| CHCl3 |
415 |
21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
505 |
4290 |
0.014 |
| THF |
407 |
22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 600 |
501 |
4610 |
0.36 |
| IPA |
401 |
25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
461 |
3245 |
0.05 |
| Acetone |
409 |
23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
501 |
4490 |
0.27 |
| Ethanol |
407 |
23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
500 |
4570 |
0.10 |
| Methanol |
406 |
23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
516 |
5250 |
0.12 |
| DMF |
407 |
22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
501 |
4610 |
0.11 |
| CH3CN |
406 |
23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
502 |
4710 |
0.08 |
| DMSO |
390 |
23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
460 |
3900 |
0.12 |
| Glycerol |
409 |
22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
455 |
2470 |
0.28 |
| H2O |
425 |
13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 600 |
631 |
7681 |
0.14 |
 |
| | Fig. 2 (a) Normalized absorption spectra and (b) normalised emission spectra of NaHB (10 µM) in different solvents and (c) effect of different solvents on NaHB under UV light. | |
3.2 Ground state optimization and electronic charge distribution
We optimized NaHB using the B3LYP/6-311G(d) method and calculated three nearby excitations to get an insight into the nature of the absorption spectra. The S0–S1 transition was calculated from the transition contribution of 98.4% involving the highest occupied molecular orbital (HOMO) to the lowest unoccupied molecular orbital (LUMO) at 437 nm with an oscillation strength of 0.6081. Furthermore, S0–S2 and S0–S3 transitions were calculated at 399 nm (HOMO−1 → LUMO (97%); f = 0.0904) and 358 nm (HOMO−4 → LUMO (72%); f = 0.033), respectively. Among these transitions, S0–S1 transition has the highest oscillation strength and is therefore considered for further discussion. HOMO → LUMO transition shows electron density shift from the benzimidazole unit to the naphthalimide unit, indicating intramolecular charge transfer. Furthermore, the nature of electronic transitions can also be understood by natural transition orbital analysis, which also depicted that the HOMO was evenly distributed over the whole architecture, while the LUMO was distributed over the naphthalimide region only (Fig. S6). Furthermore, hole–electron distribution and intramolecular fragment charge transfer were calculated using Multiwfn.39–41 It was inferred that an electronic transition occurred with a low electron–hole overlap (Sr = 0.53092) and a charge separation of 4.3 Å (Fig. 3). Moreover, the intramolecular fragment charge transferred from the benzimidazole and fused phenylenediamine units towards the naphthalimide unit by 0.52872 and 0.08759 a.u., respectively, with an intrinsic charge transfer character of 72%. These results showed that the electronic transition has intramolecular charge transfer nature.
 |
| | Fig. 3 Optimized structures of NaHB and contributing molecular orbitals along with electron–hole distribution for S0 → S1 transition. | |
3.3 Intramolecular proton transfer or charge transfer
NaHB contains an acidic phenolic unit and a basic benzimidazole unit, which interact through small distances and could result in proton transfer at the excited state. Furthermore, we established the necessary condition of intramolecular hydrogen bonding strengthening at the S0 and S1 states to understand the behaviour of NaHB towards the ESIPT process. At the S0 state, the O28H50⋯N30 interaction has a distance of 1.728 Å and an interaction angle of 146.72°. Upon photoexcitation at the S1 state, the interaction distance decreased to 1.690 Å and the interaction angle increased to 146.84° (Fig. 4(a)). Furthermore, O34H35⋯N22 IraHB (intramolecular hydrogen bonding) exhibited a 179 cm−1 redshift from 3178 cm−1 to 2999 cm−1 upon photoexcitation to the S1 state (Fig. 4(b)). It was also noteworthy that NaHB approaches the molecular planarity by decreasing the dihedral angle between the benzimidazole unit and the naphthalimide unit from 53.68° to 43.54° at the S1 state.
 |
| | Fig. 4 (a) Optimized structures of NaHB and (b) FTIR spectra of NaHB at S0 and S1 states. | |
These factors favour the excited state intramolecular proton transfer phenomenon. Therefore, potential energy curves (PECs) were constructed with respect to the proton transfer coordinate for the NaHB enol form to the keto form. According to the PECs, the enol form is the only stable conformer in the S0 state, establishing the trend of NaHB (enol; 0 kcal mol−1) < NaHB (keto; 3.37 kcal mol−1) in the S0 state and NaHB (enol; 54.99 kcal mol−1) > NaHB (keto; 50.24 kcal mol−1) in the S1 state (Fig. 5). Furthermore, the calculated energy barrier for forward proton transfer at the S0 state is 6.64 kcal mol−1, which further decreases to 3.43 kcal mol−1 at the S1 state. On the other hand, the reverse proton transfer has an energy barrier of 3.37 kcal mol−1 at the S0 state, which increases to 8.18 kcal mol−1 at the S1 state. These observations of a low energy barrier for forward proton transfer and a high barrier for backward proton transfer at the S1 state suggest the ESIPT process for NaHB.
 |
| | Fig. 5 Energy profile of proton transfer for NaHB (the truncated view of proton transfer is shown). | |
3.4 Sensing properties of NaHB towards various metals, anions and nerve agents
The absorption and emission behaviors of NaHB were investigated using a CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) medium. NaHB exhibited absorption maxima (λmax) at 402 nm, and an emission peak was observed at 520 nm in the CH3CN
1) medium. NaHB exhibited absorption maxima (λmax) at 402 nm, and an emission peak was observed at 520 nm in the CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) solvent medium. The sensing behavior of NaHB has been explored towards various metal ions (Na+, K+, Mg2+, Ba2+, Al3+, Ca2+, Fe2+, Fe3+, Co2+, Ni2+, Cu2+, Zn2+, Ga3+, Cd2+, Hg2+ and Pb2+), anions (CH3COO−, F−, Cl−, Br−, CN−, H2PO4−, HSO4−, NO3−, AcO−, ClO−, P2O74−, and SCN−) and nerve agents (DCP, TEP (triethyl phosphate), and N-BP (N-butyl phosphate)) using UV-visible and fluorescence spectrophotometric techniques. We observed an exceptional response of NaHB towards Ga3+ ions and DCP with high sensitivity and selectivity via an enhancement in the emission band, whereas other anions and metal ions did not show any significant change in absorption and emission spectra.
1) solvent medium. The sensing behavior of NaHB has been explored towards various metal ions (Na+, K+, Mg2+, Ba2+, Al3+, Ca2+, Fe2+, Fe3+, Co2+, Ni2+, Cu2+, Zn2+, Ga3+, Cd2+, Hg2+ and Pb2+), anions (CH3COO−, F−, Cl−, Br−, CN−, H2PO4−, HSO4−, NO3−, AcO−, ClO−, P2O74−, and SCN−) and nerve agents (DCP, TEP (triethyl phosphate), and N-BP (N-butyl phosphate)) using UV-visible and fluorescence spectrophotometric techniques. We observed an exceptional response of NaHB towards Ga3+ ions and DCP with high sensitivity and selectivity via an enhancement in the emission band, whereas other anions and metal ions did not show any significant change in absorption and emission spectra.
3.4.2 Fluorescence spectral studies of NaHB towards Ga3+. When exciting NaHB (λex = 402 nm, 10 µM, CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)), a weak fluorescence intensity was observed at 520 nm. The addition of various metal ions to NaHB did not show any variation in the emission spectrum except for Ga3+. The presence of Ga3+ ions exhibited an increase in fluorescence intensity with quick response at 520 nm, displaying excellent bright green fluorescence under UV light (Fig. 6(a)).
1)), a weak fluorescence intensity was observed at 520 nm. The addition of various metal ions to NaHB did not show any variation in the emission spectrum except for Ga3+. The presence of Ga3+ ions exhibited an increase in fluorescence intensity with quick response at 520 nm, displaying excellent bright green fluorescence under UV light (Fig. 6(a)).
 |
| | Fig. 6 Fluorescence spectra of NaHB (10 µM) in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9 H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, [v/v]) (a) in the presence of various metal ions (50 equiv.) and (b) upon incremental addition of 0–50 µM Ga3+ ions. 1, [v/v]) (a) in the presence of various metal ions (50 equiv.) and (b) upon incremental addition of 0–50 µM Ga3+ ions. | |
Upon incremental addition of Ga3+ ions into the solution of NaHB, an emission enhancement was observed at the same wavelength of 520 nm (Fig. 6(b)). The maximum emission intensity (8 times) was attained with the addition of 50 µM Ga3+ ions. A good linear relationship of the fluorescence intensity was observed in the concentration range of 0–50 µM. The binding constant acquired from the titration data for Ga3+ ions, calculated from eqn 2, was found to be 5.15 × 104 ± 0.03 × 104 M−1 (95% CI: 5.07–5.22 × 104 M−1). By using eqn (1), the limit of detection (LOD) was found to be 36 ± 0.02 nM (95% CI: 35.95–36.05 nM) and the quantifiable limit range was 36–120 nM in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). The obtained LOD value was compared with reported values for chemosensors, particularly naphthalimide-based derivatives using fluorometric methods, and was found to be superior to those previously reported (Table S1). The fluorescence response of NaHB with Ga3+ and other metal ions can be seen under UV light by the naked eye (Fig. 7).
1). The obtained LOD value was compared with reported values for chemosensors, particularly naphthalimide-based derivatives using fluorometric methods, and was found to be superior to those previously reported (Table S1). The fluorescence response of NaHB with Ga3+ and other metal ions can be seen under UV light by the naked eye (Fig. 7).
 |
| | Fig. 7 Effect of different metal ions on the solution of NaHB (10 µM, CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9 H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, [v/v])) under UV light. 1, [v/v])) under UV light. | |
3.4.3 Interference study. To demonstrate the fluorescence selectivity of NaHB towards Ga3+, an interference study with some common intrusive ions (1 mM) was performed. For the selective behavior of NaHB towards Ga3+ (50 µM), the interference studies were performed in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). By consecutive addition of different analytes (1 mM) in the complex of NaHB + Ga3+, the spectral change has been measured. The study was done in 3 replicates and the standard deviations along with the p-value (probability of interference of other analytes) obtained from the t-test are incorporated in Table S2. The spectral signal does not get altered upon the addition of other metal ions, indicating no interference in the binding of NaHB + Ga3+. This signifies that NaHB possesses no response ability towards most of the analytes except Ga3+ ions (Fig. 8).
1). By consecutive addition of different analytes (1 mM) in the complex of NaHB + Ga3+, the spectral change has been measured. The study was done in 3 replicates and the standard deviations along with the p-value (probability of interference of other analytes) obtained from the t-test are incorporated in Table S2. The spectral signal does not get altered upon the addition of other metal ions, indicating no interference in the binding of NaHB + Ga3+. This signifies that NaHB possesses no response ability towards most of the analytes except Ga3+ ions (Fig. 8).
 |
| | Fig. 8 Blue bars represent the selectivity of NaHB (10 µM, CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9 H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; v/v)) at λem = 520 nm upon the addition of different cations and red bars show the competitive selectivity of NaHB in the presence of Ga3+ ions. 1; v/v)) at λem = 520 nm upon the addition of different cations and red bars show the competitive selectivity of NaHB in the presence of Ga3+ ions. | |
3.4.4 Binding behavior of NaHB towards Ga3+ ions. For determining the stoichiometry of the complex of NaHB + Ga3+, solutions of varying mole fractions of Ga3+ from 0.1 to 1.0 in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) were prepared. A graph was plotted with the emission response in each case at 520 nm and the mole fraction of Ga3+ ions (Fig. S8). Based on Job's plot, it was found that the ratio of complexation of Ga3+ ions and NaHB was 1
1) were prepared. A graph was plotted with the emission response in each case at 520 nm and the mole fraction of Ga3+ ions (Fig. S8). Based on Job's plot, it was found that the ratio of complexation of Ga3+ ions and NaHB was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Moreover, the ratio of the formation of the complex was confirmed by ESI-mass spectral analysis. The mass was taken for the mixture of NaHB and Ga3+ in CH3CN
1. Moreover, the ratio of the formation of the complex was confirmed by ESI-mass spectral analysis. The mass was taken for the mixture of NaHB and Ga3+ in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), where a new peak at m/z = 546.4014 corresponding to the complex of (NaHB + Ga3+ − H+) was observed (Fig. S9), indicating the formation of a 1
1), where a new peak at m/z = 546.4014 corresponding to the complex of (NaHB + Ga3+ − H+) was observed (Fig. S9), indicating the formation of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex.
1 complex.
3.4.5 UV-vis spectral studies of NaHB in the presence of various anions and nerve agents. UV-visible spectra of NaHB were also recorded in the presence of various anions (1 mM) like CH3COO−, F−, Cl−, Br−, I−, CN−, H2PO4−, HSO4−, NO3−, AcO−, ClO−, P2O74−, and SCN− and nerve agents such as DCP (diethyl chloro phosphate), TEP (triethyl phosphate), and N-BP (N-butyl phosphate) in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). No significant change was observed in the case of various analytes, while DCP showed a blue shift of 13 nm (at 389 nm) upon its addition to NaHB (Fig. 9(a)). The interaction of the probe with DCP was systematically examined by UV-vis titration studies in CH3CN
1). No significant change was observed in the case of various analytes, while DCP showed a blue shift of 13 nm (at 389 nm) upon its addition to NaHB (Fig. 9(a)). The interaction of the probe with DCP was systematically examined by UV-vis titration studies in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) (Fig. 9(b)). Upon incremental addition of DCP to the solution of the probe, the absorption spectra exhibited significant changes, indicating strong host–guest interaction. A gradual increase (hyperchromic effect) in the absorption band at 389 nM was observed, accompanied by a slight hypsochromic shift. A plateau was obtained after the addition of 50 µM DCP during titration, suggesting effective binding of DCP at the recognition site of the probe.
1) (Fig. 9(b)). Upon incremental addition of DCP to the solution of the probe, the absorption spectra exhibited significant changes, indicating strong host–guest interaction. A gradual increase (hyperchromic effect) in the absorption band at 389 nM was observed, accompanied by a slight hypsochromic shift. A plateau was obtained after the addition of 50 µM DCP during titration, suggesting effective binding of DCP at the recognition site of the probe.
 |
| | Fig. 9 (a) UV-visible spectra of NaHB (10 µM, CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9 H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)) in the presence of various anions (50 equiv.) and (b) upon incremental addition of DCP (0–50 µM). 1)) in the presence of various anions (50 equiv.) and (b) upon incremental addition of DCP (0–50 µM). | |
3.4.6 Fluorescence spectral studies of NaHB in the presence of anions. To check the selectivity and sensitivity of NaHB towards DCP, the fluorescence response was also studied. It was found that while complexing DCP with NaHB, the fluorescence intensity showed a remarkable 7-fold enhancement at 515 nm. No other anion or nerve agent has shown any change in the emission spectrum (Fig. 10(a)). Fluorescence titrations were carried out with 10 µM NaHB along with varying concentrations of DCP (Fig. 10(b)). A linear relation of increase in fluorescence intensity with increasing concentration of DCP in NaHB was noticed. This linear graph was used to obtain the Ka (binding constant) value using the Benesi–Hildebrand equation and it was found to be 1.2 × 105 M−1 (95% CI: 1.10–1.29 × 105 M−1). The limit of detection (LOD) obtained for DCP was 11 nM (95% CI: 10.92–11.07 nM) and the quantifiable limit was 11–37 nM. A competitive complexation study was carried out to view the possible invasion of some other analytes in the complex NaHB + DCP. The bar graph shows the variation in fluorescence intensity of the complex in the presence of different anions and nerve agents (Fig. S10(a)). It can be inferred from the graph that no other ions interfered in the complex of NaHB + DCP, signifying selectivity for DCP only. To depict the stoichiometry, Job's plot studies were done and it was observed to be 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, which can be seen from the graph (Fig. S10(b)). To further support Job's plot, the ESI-mass spectrum of NaHB in the presence of DCP was obtained. A reaction was performed between NaHB and DCP to isolate and determine the mass of the product formed. The HRMS (ESI+) of the isolated reaction material showed a prominent ion at m/z ≈ [M + 136 + H]+ (m/z = 615.1794, calcd = 615.1790, Δ = 8 ppm), indicating addition of a mass equivalent to DCP (≈+136 Da) (Fig. S11(a)). However, tandem MS (MS/MS) of this ion under collision-induced dissociation conditions reproducibly produced the parent probe ion (m/z = 479.1509) and free DCP fragments (m/z = 137) as the major products (Fig. S11(b)), even at moderate collision energies, indicating facile cleavage of the added mass. This fragmentation behavior strongly suggests that the interaction between NaHB and DCP is primarily non-covalent.
1, which can be seen from the graph (Fig. S10(b)). To further support Job's plot, the ESI-mass spectrum of NaHB in the presence of DCP was obtained. A reaction was performed between NaHB and DCP to isolate and determine the mass of the product formed. The HRMS (ESI+) of the isolated reaction material showed a prominent ion at m/z ≈ [M + 136 + H]+ (m/z = 615.1794, calcd = 615.1790, Δ = 8 ppm), indicating addition of a mass equivalent to DCP (≈+136 Da) (Fig. S11(a)). However, tandem MS (MS/MS) of this ion under collision-induced dissociation conditions reproducibly produced the parent probe ion (m/z = 479.1509) and free DCP fragments (m/z = 137) as the major products (Fig. S11(b)), even at moderate collision energies, indicating facile cleavage of the added mass. This fragmentation behavior strongly suggests that the interaction between NaHB and DCP is primarily non-covalent.
 |
| | Fig. 10 (a) Fluorescence spectra of NaHB (10 µM) in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9 H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, [v/v]) in the presence of various anions and nerve agents (1 mM) and (b) upon incremental addition of DCP ions (0–25 µM). 1, [v/v]) in the presence of various anions and nerve agents (1 mM) and (b) upon incremental addition of DCP ions (0–25 µM). | |
3.5 NMR titrations and the binding mechanism
1H NMR titrations were performed in CD3CN to investigate the binding mechanism of NaHB with Ga3+ and DCP. Upon addition of 1 equiv. of Ga3+ ions to NaHB, the protons Ha and Hb at 6.67–6.72 ppm and protons Hc and Hd at 7.02–7.06 ppm got shifted downfield to 6.78–6.86 ppm ( and
and  ) and 7.14–7.20 ppm (
) and 7.14–7.20 ppm (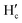 and
and 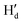 ), respectively. Moreover, the
), respectively. Moreover, the 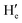 proton gets slightly deshielded from 7.76–7.78 ppm to 7.78–7.81 ppm (Fig. 11). A similar trend was seen in the case of DCP binding with NaHB. The
proton gets slightly deshielded from 7.76–7.78 ppm to 7.78–7.81 ppm (Fig. 11). A similar trend was seen in the case of DCP binding with NaHB. The 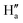 and
and 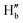 protons experienced deshielding with a shift of 0.13–0.16 ppm during the complexation of DCP with NaHB. Similarly,
protons experienced deshielding with a shift of 0.13–0.16 ppm during the complexation of DCP with NaHB. Similarly, 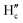 and
and 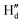 protons also shifted towards the downfield region (7.15–7.20 ppm). However, the
protons also shifted towards the downfield region (7.15–7.20 ppm). However, the 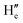 and
and 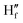 protons did not experience the shifts as these protons were far from the binding sites, but a doublet from NaHB of He got almost detached from the multiplet, thereby increasing the range from 7.76–7.78 ppm to 7.76–7.81 ppm. The results indicated that in the case of complexation with Ga3+, both the imine nitrogen and the hydroxyl group were involved, making the nearby protons deshielded. The deprotonation of the hydroxyl group was caused by the phosphate group of DCP, therefore shifting the proton signals to the downfield region. Moreover, no further change was observed with increasing equivalents of the analyte, confirming the stoichiometry as 1
protons did not experience the shifts as these protons were far from the binding sites, but a doublet from NaHB of He got almost detached from the multiplet, thereby increasing the range from 7.76–7.78 ppm to 7.76–7.81 ppm. The results indicated that in the case of complexation with Ga3+, both the imine nitrogen and the hydroxyl group were involved, making the nearby protons deshielded. The deprotonation of the hydroxyl group was caused by the phosphate group of DCP, therefore shifting the proton signals to the downfield region. Moreover, no further change was observed with increasing equivalents of the analyte, confirming the stoichiometry as 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, as obtained from Job's plot.
1, as obtained from Job's plot.
 |
| | Fig. 11 1H NMR spectra of NaHB (5 mM) in the absence and in the presence of Ga3+ ions and DCP in CD3CN (d3) (1 equiv.). | |
In order to further confirm the binding behaviour of NaHB towards Ga3+ ions, theoretical calculations have also been performed. NaHB·Ga3+ was optimized at the S0 state, and correspondingly vertical-excitations were calculated (Fig. 12). It has been noted that NaHB·Ga3+ shows S0 → S1, S0 → S2 and S0 → S3 excitations at 520 nm (HOMO → LUMO (98%); f = 0.0001), 430 nm (HOMO → LUMO+1 (97.4%); f = 0.5994) and 424 nm (HOMO−2 → LUMO (98%); f = 0.0001), respectively. Furthermore, only S0 → S2 excitation was analysed, as it contributed bright excitation, and it showed electron density shifts towards the naphthalimide unit with electron–hole overlap (Sr = 0.71310) and a charge separation of 1.715 Å. Furthermore, it can be noted that the electron–hole overlap for NaHB·Ga3+ increased relative to NaHB·DCP and NaHB, while the electron–hole centroid distance decreased, which signified that the quantity of intramolecular charge transfer decreased for NaHB·Ga3+.
 |
| | Fig. 12 Optimized structure and frontier molecular orbitals of NaHB with Ga3+ (stoichiometry 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and the electron–hole distribution of S0–S2 orbitals. 1) and the electron–hole distribution of S0–S2 orbitals. | |
Additionally, we investigated the interaction between DCP and NaHB, where the NaHB·DCP complex was formed (Fig. 13). Furthermore, we examined the vertical Franck–Condon excitation to have an insight into the absorption spectra of the complex. It was noted that NaHB·DCP shows S0 → S1 excitation at 432 nm with an oscillation strength of 0.5025 from 98.4% contribution of HOMO → LUMO transition. The preceding S0 → S2 and S0 → S3 transitions were calculated at 411 nm (HOMO−1 → LUMO (98%); f = 0.02660) and 370 nm (HOMO−3 → LUMO (94%); f = 0.03070), respectively. Furthermore, the HOMO → LUMO transition has the nature of intramolecular charge transfer that shows the electron density shifts from the benzimidazole and the connecting phenyl unit to the naphthalimide unit with low electron–hole overlap (Sr = 0.60635) and a charge separation of 4.1 Å.
 |
| | Fig. 13 Optimized structure and frontier molecular orbitals of NaHB with DCP (stoichiometry 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and the electron–hole distribution of S0–S1 orbitals. 1) and the electron–hole distribution of S0–S1 orbitals. | |
3.6 Lifetime measurements
Lifetime decay studies were also done for both probe NaHB and its complexes with Ga3+ and DCP. Upon complexation of NaHB with Ga3+ (1 equiv.) and DCP (1 equiv.), a significant change was observed in the decay profiles in both cases (Fig. 14). The average lifetime value (τav) of NaHB was calculated using three-exponential fitting and was found to be 0.177 ns (eqn (3)).| |
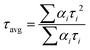 | (3) |
where αi = α1 and α2 and α3 are the normalized pre-exponential factors, and τi = τ1, τ2 and τ3 represent the excited-state lifetimes of the corresponding components. Upon complexation with Ga3+, the lifetime changed to 0.286 ns and with DCP, the value obtained was 0.231 ns (Table 2). Thus, upon complexation, the average excited lifetime of NaHB was found to be increased, and the ESIPT from the hydroxyl group of NaHB was fully inhibited; hence, in consequence, the fluorescence intensity was enhanced upon binding of NaHB with Ga3+ and DCP.
 |
| | Fig. 14 Fluorescence lifetime decay of NaHB along with Ga3+ and DCP. | |
Table 2 Fluorescence lifetime decay profile
| |
τ1 (ns) |
τ2 (ns) |
τ3 (ns) |
α1 |
α2 |
α3 |
χ2 |
τav (ns) |
| NaHB |
1.45 |
4.92 |
0.04 |
0.02 |
0.02 |
0.96 |
1.06 |
0.177 |
| NaHB + Ga3+ |
0.69 |
0.22 |
3.91 |
0.04 |
0.95 |
0.01 |
1.07 |
0.286 |
| NaHB + DCP |
0.41 |
0.11 |
3.33 |
0.30 |
0.69 |
0.01 |
1.11 |
0.231 |
3.7 Reversible studies
Another desirable property for the development of chemosensors for their application in real world is the reversibility. The experiment has been performed by sequential addition of Ga3+ (1 equiv.) and EDTA (1 equiv.) to NaHB by measuring the fluorescence intensity. When 1 equiv. of Ga3+ was added to NaHB, the fluorescence got enhanced to 8 fold, while upon addition of EDTA (1 equiv.), the fluorescence intensity got quenched. This is because of the de-complexation of the NaHB + Ga3+ complex as EDTA forms a complex with Ga3+ because of its strong affinity for metal ions, making NaHB free. The fluorescence was regained in the system of NaHB + Ga3+ + EDTA by adding another 1 equiv. of Ga3+. Hence, the addition of Ga3+ and EDTA alternatively would result in on–off–on cycles. The cycle was repeated 5 times, signifying a good reusability of NaHB towards Ga3+ sensing (Fig. 15).
 |
| | Fig. 15 The reversible and reproducible emission signal at 520 nm (λex = 402 nm, 10 µM) in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9 H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, [v/v]) upon repetitive addition of metal ions and EDTA. 1, [v/v]) upon repetitive addition of metal ions and EDTA. | |
3.8 Practical applications
3.8.1 Paper-strip based sensing of Ga3+ and DCP. Costly instrumental techniques and extensive experimental procedures are the major drawbacks in the practical usage of chemosensors. In this view, paper strips are gaining much interest in the sensing area because of their portability and cost-effectiveness. For on-site detection of Ga3+ and DCP using NaHB, Whatman-41 filter paper-based strips were prepared by immersing them in a solution of NaHB (10−3 M) in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) for 5 minutes and then air-drying for several hours. Subsequently, aqueous solutions of Ga3+ at different concentrations ranging from 10−1 M to 10−6 M were added dropwise onto the coated strips. The interaction between Ga3+ ions and NaHB on the strips was monitored visually, with particular attention paid to any observable changes such as color variation or intensity, which could indicate complex formation or sensor response (Fig. S12). Following this, different ion solutions (10−1 M) were added dropwise to the strips and then allowed to spread on the surface of the prepared coated strips in air. The fluorescence intensity of the paper strip consisting of Ga3+ and DCP was quite high when observed under a 365 nm UV lamp (Fig. 16). This implies that the fabricated paper strips of NaHB can be used for on-spot detection of Ga3+ and DCP in the presence of other metal ions, anions and nerve agents.
1) for 5 minutes and then air-drying for several hours. Subsequently, aqueous solutions of Ga3+ at different concentrations ranging from 10−1 M to 10−6 M were added dropwise onto the coated strips. The interaction between Ga3+ ions and NaHB on the strips was monitored visually, with particular attention paid to any observable changes such as color variation or intensity, which could indicate complex formation or sensor response (Fig. S12). Following this, different ion solutions (10−1 M) were added dropwise to the strips and then allowed to spread on the surface of the prepared coated strips in air. The fluorescence intensity of the paper strip consisting of Ga3+ and DCP was quite high when observed under a 365 nm UV lamp (Fig. 16). This implies that the fabricated paper strips of NaHB can be used for on-spot detection of Ga3+ and DCP in the presence of other metal ions, anions and nerve agents.
 |
| | Fig. 16 Paper strips coated with NaHB, along with (a) dropwise addition of different metal ions (10−2 M) and (b) dropwise addition of different anions (10−1 M). | |
3.8.2 Real-time monitoring in water samples. The application of chemosensors should not be limited to lab-used chemicals only; it should also be used for monitoring of analytes in real environmental samples. To evaluate the practical applicability and accuracy of the developed probe NaHB, a calibration curve was constructed using real water samples (tap, sewage, and Ghaggar river water) spiked with known concentrations of Ga3+ and DCP ions. The corresponding ion concentrations were then interpolated from the calibration curve using the measured fluorescence intensities. The fluorescence spectra of each spiked sample were recorded under identical conditions to those used in standard solution studies (Fig. 17).
 |
| | Fig. 17 Fluorescence spectra of NaHB in different concentrations of spiked (a) Ga3+ and (b) DCP from 0 to 50 µM. | |
A gradual enhancement in fluorescence intensity was observed with increasing analyte concentration, confirming that the sensor responds reliably in complex water matrices. The calibration curve, plotted as fluorescence intensity versus ion concentration, exhibited a good linear relationship within the tested range, following the regression equation (eqn (4)):
where
I represents the measured fluorescence intensity,
C is the Ga
3+ concentration,
m is the slope (sensitivity), and
b is the intercept.
The high correlation coefficient (R2 ≈ 0.99) demonstrates excellent linearity and reproducibility of the response. The close agreement between the measured and spiked concentrations, as reflected in satisfactory recovery values (Table 3) (typically 98–102%), further confirms that the probe maintains its fluorescence enhancement behavior and quantitative detection capability for Ga3+ and DCP even in real environmental water samples.
Table 3 Analyzing Ga3+ and DCP quantitatively in real water samples
| |
Ga3+ (µM) |
Found (µM) |
RSD (%) |
Recovery (%) |
DCP (µM) |
Found (µM) |
RSD (%) |
Recovery (%) |
| Tap water |
10 |
9.8 |
1.15 |
98.7 |
10 |
9.9 |
0.59 |
99.3 |
| 20 |
19.9 |
0.33 |
99.7 |
20 |
19.9 |
0.52 |
101.5 |
| 30 |
29.7 |
1.06 |
99.1 |
30 |
29.9 |
1.07 |
99.8 |
| Sewage water |
10 |
9.9 |
0.71 |
99.3 |
10 |
9.8 |
0.95 |
98.9 |
| 20 |
20.2 |
2.84 |
101.1 |
20 |
19.6 |
2.84 |
98 |
| 30 |
29.8 |
2.65 |
99.4 |
30 |
29.7 |
0.7 |
99.3 |
| Ghaggar river water |
10 |
10.1 |
3.31 |
101.3 |
10 |
10.1 |
3.09 |
101.4 |
| 20 |
19.8 |
0.63 |
99.4 |
20 |
19.9 |
0.2 |
99.9 |
| 30 |
29.8 |
0.91 |
99.3 |
30 |
30.4 |
1.83 |
101.4 |
3.8.3 Molecular logic gate. The molecular logic gate was fabricated using NaHB, Ga3+ and EDTA. The emission band at 520 nm appears due to the formation of a complex between NaHB and Ga3+ ions. When EDTA was added to the complex, the emission intensity at 520 nm was highly quenched. In this view, an ‘INHIBIT’ logic gate is made by taking Ga3+ and EDTA as chemical inputs and the emission intensity at 520 nm as the output. The inhibition is caused possibly by the decomplexation of the NaHB + Ga3+ complex and Ga3+ getting attached to EDTA, thereby giving no enhanced emission signal because of the strong binding characteristics of EDTA as a ligand. By decomplexation, the optical properties of the probe were reversed. The molecular logic gate and the truth table are shown in Fig. 18. It can be inferred from the truth table that the output comes out to be ‘1’ only when the NaHB + Ga3+ complex is formed. Once EDTA enters the solution, the output comes out to be ‘0’ in all other cases.
 |
| | Fig. 18 Logic gate and the truth table. | |
Conclusion
The fluorescence-based probe NaHB effectively enabled the selective and sensitive detection of Ga3+ and DCP, with significant emission enhancement upon analyte binding, which is clearly visible under UV light. This fluorescence response allowed for nanomolar-level detection limits of 36 nM for Ga3+ and 11 nM for DCP. The underlying binding mechanisms were further supported and confirmed by detailed NMR spectroscopy, mass spectrometry, and computational studies. The interaction of Ga3+ with both the hydroxyl and imine groups of NaHB led to a stable six-membered chelate ring, while DCP selectively engaged the hydroxyl site. Additionally, paper strip-based sensing under UV light enabled practical on-site applications, and probe NAHB performed reliably in real water samples, including tap, drinking, and river water. The NaHB–Ga3+ complex also demonstrated logic gate functionality using Ga3+ and EDTA as inputs, expanding the potential for molecular computation.
Conflicts of interest
There are no conflicts of interest to declare.
Data availability
Data shall be available upon request.
Supplementary information (SI): NMR, HRMS and photophysical studies. See DOI: https://doi.org/10.1039/d5nj03370e.
Acknowledgements
VL thanks CEEMS (Project No. TIET/CEEMS/Regular/2023/8054), VT-India, for providing funds. SAI Labs, TIET, Patiala; SAIF, PU, Chandigarh; for NMR analysis and DST-FIST (SR/FST/CS-II/2018/69) (ACALAB, TIET, Patiala) for HRMS analysis are also acknowledged.
References
- A. Mumthaj and M. Umadevi, Spectrochim. Acta, Part A, 2024, 308, 123741–123747 CrossRef CAS PubMed.
- B. Devi, A. K. Guha and A. Devi, Spectrochim. Acta, Part A, 2024, 305, 123448–123453 CrossRef CAS PubMed.
- N. A. Bumagina, A. A. Ksenofontov, E. V. Antina and M. B. Berezin, Spectrochim. Acta, Part A, 2024, 307, 123663–123675 CrossRef CAS PubMed.
- X. Gao, X. Wang, W. Lv, N. Wang, M. Yang and Q. Zhang, J. Mol. Struct., 2022, 1250, 131792–131800 CrossRef CAS.
- J. Singh, B. Mohan, A. Kumar, P. Bhardwaj and R. K. Chauhan, J. Fluoresc., 2024, 34, 149–157 CrossRef CAS PubMed.
- K. Zhang, X. Tian, P. Xu, Y. Zhu, S. Guang and H. Xu, Spectrochim. Acta, Part A, 2024, 318, 124484–124493 CrossRef CAS PubMed.
- F. Sun, L. Yang, S. Li, Y. Wang, L. Wang, P. Li, F. Ye and Y. Fu, J. Agric. Food Chem., 2021, 69, 12661–12673 CrossRef CAS PubMed.
- D. Yun, J. M. Jung and C. Kim, Inorg. Chim. Acta, 2018, 479, 154–160 CrossRef CAS.
- J. W. van Dam, M. A. Trenfield, C. Streten, A. J. Harford, D. Parry and R. A. van Dam, Ecotoxicol. Environ. Saf., 2018, 165, 349–356 CrossRef CAS PubMed.
- H. Xiang, T. Wang, S. Tang, Y. Wang and N. Xiao, Spectrochim. Acta, Part A, 2022, 267, 120510–120519 CrossRef CAS PubMed.
- J. M. Kikkawa and D. D. Awschalom, Nature, 1999, 397, 139–141 CrossRef CAS.
- H. M. Kim, Y. H. Cho, H. Lee, S. I. I. Kim, S. R. Ryu, D. Y. Kim, T. W. Kang and K. S. Chung, Nano Lett., 2004, 4, 1059–1062 CrossRef CAS.
- R. Trotta, P. Atkinson, J. D. Plumhof, E. Zallo, R. O. Rezaev, S. Kumar, S. Baunack, J. R. Schröter, A. Rastelli and O. G. Schmidt, Adv. Mater., 2012, 24, 2668–2672 CrossRef CAS PubMed.
- J. Yoon, S. Jo, I. S. Chun, I. Jung, H. S. Kim, M. Meitl, E. Menard, X. Li, J. J. Coleman, U. Paik and J. A. Rogers, Nature, 2010, 465, 329–333 CrossRef CAS PubMed.
- S. L. Yao, R. H. Wu, P. Wen, H. Liu, T. Tu and S. J. Liu, J. Mol. Struct., 2024, 1297, 136925–136931 CrossRef CAS.
- S. J. O. White and J. P. Shine, Curr. Environ. Health Rep., 2016, 3, 459–467 CrossRef CAS PubMed.
- A. Tanaka, Toxicol. Appl. Pharmacol., 2004, 198, 405–411 CrossRef CAS PubMed.
- L. Ann Burns and R. T. Abraham, Cancer Res., 1993, 53, 1862–1866 Search PubMed.
- J. Qi, Q. Yao, K. Qian, L. Tian, Z. Cheng, D. Yang and Y. Wang, Eur. J. Med. Chem., 2018, 154, 91–100 CrossRef CAS PubMed.
- Y. Zhang and B. Li, J. Mol. Struct., 2022, 1250, 131461–131467 CrossRef CAS.
- S. Zhang, B. Yang, B. Yuan, C. Zhou, M. Zhang, Y. Zhao, P. Ye, L. Li and H. Li, ACS Sens., 2023, 8, 1220–1229 CrossRef CAS PubMed.
- Y. J. Jang, K. Kim, O. G. Tsay, D. A. Atwood and D. G. Churchill, Chem. Rev., 2015, 115, 1–76 CrossRef PubMed.
- M. S. J. Khan, Y. W. Wang, M. O. Senge and Y. Peng, J. Hazard. Mater., 2018, 342, 10–19 CrossRef CAS PubMed.
- E. D. Fussell, N. D. Kline, E. Bennin, S. S. Hirschbeck and A. Darko, ACS Sens., 2024, 9, 2325–2333 CrossRef CAS PubMed.
- S. Costanzi, J. H. Machado and M. Mitchell, ACS Chem. Neurosci., 2018, 9, 873–885 CrossRef CAS PubMed.
- S. Banerjee, P. Ghosh, A. Karak, D. Banik and A. K. Mahapatra, Anal. Methods, 2025, 17, 432–439 RSC.
- P. Zheng, A. Abdurahman, G. Liu, H. Liu, Y. Zhang and M. Zhang, Sens. Actuators, B, 2020, 322, 128611–128618 CrossRef CAS.
- Z. Qiu, Y. Xiao, L. Zhang, Y. Miao, B. Zhang, X. Zhu, L. Ding, H. Peng and Y. Fang, Chem. Commun., 2024, 60, 9773–9776 RSC.
- Q. Chen, Y. Sun, S. Liu, J. Zhang, C. Zhang, H. Jiang, X. Han, L. He, S. Wang and K. Zhang, Sens. Actuators, B, 2021, 344, 130278–130300 CrossRef CAS.
- T. Qin, Y. Huang, K. Zhu, J. Wang, C. Pan, B. Liu and L. Wang, Anal. Chim. Acta, 2019, 1076, 125–130 CrossRef CAS PubMed.
- Y. Yang, H. F. Ji and T. Thundat, J. Am. Chem. Soc., 2003, 125, 1124–1125 CrossRef CAS PubMed.
- R. Subramaniam, C. Åstot, L. Juhlin, C. Nilsson and A. Östin, Anal. Chem., 2010, 82, 7452–7459 CrossRef CAS PubMed.
- K. A. Joshi, M. Prouza, M. Kum, J. Wang, J. Tang, R. Haddon, W. Chen and A. Mulchandani, Anal. Chem., 2006, 78, 331–336 CrossRef CAS PubMed.
- G. Paimard, M. Nejatian, Z. Sarlak, R. Mohammadi and M. Rouhi, Microchem. J., 2024, 198, 110173–110198 CrossRef CAS.
- M. Verma, V. Luxami and K. Paul, Eur. J. Med. Chem., 2013, 68, 352–360 CrossRef CAS PubMed.
- T. Mishra, S. Guria, J. Sadhukhan, D. Das and M. Kumar, J. Photochem. Photobiol., A, 2024, 446, 115168–115180 CrossRef CAS.
- P. M. W. Gill, B. G. Johnson, J. A. Pople and M. J. Frisch, Chem. Phys. Lett., 1992, 197, 499–505 CrossRef CAS.
- B. Das, M. Dolai, A. Dhara, A. Ghosh, S. Mabhai, A. Misra, S. Dey and A. Jana, J. Phys. Chem. A, 2021, 125, 1490–1504 CrossRef CAS PubMed.
- T. Lu and F. Chen, Multiwfn: A Multifunctional Wavefunction Analyzer, J. Comput. Chem., 2012, 33, 580–592 CrossRef CAS PubMed.
- T. Lu, A comprehensive electron wavefunction analysis toolbox for chemists, Multiwfn, J. Chem. Phys., 2024, 161, 082503 CrossRef CAS PubMed.
- G. Kumar, K. Paul and V. Luxami, New J. Chem., 2020, 44, 12866–12874 RSC.
|
| This journal is © The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article b,
Iqubal Singhc and
Vijay Luxami
b,
Iqubal Singhc and
Vijay Luxami *a
*a
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry for both systems. The practical utility of NaHB was demonstrated through paper strip assays and quantitative detection of Ga3+ and DCP in real-field water samples. Furthermore, the NaHB–Ga3+ complex was explored for molecular logic gate applications, showcasing its multifunctional sensing capability.
1 stoichiometry for both systems. The practical utility of NaHB was demonstrated through paper strip assays and quantitative detection of Ga3+ and DCP in real-field water samples. Furthermore, the NaHB–Ga3+ complex was explored for molecular logic gate applications, showcasing its multifunctional sensing capability.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) (10 ml) in the presence of K2CO3 (100 mg, 0.724 mmol) and Pd(PPh3)4 (6 mg, 0.005 mmol) and subjected to the Suzuki–Miyaura cross-coupling reaction (Scheme 1). The reaction mixture was refluxed for 8 h until the starting materials were completely consumed, and the reaction progress was monitored through TLC. After completion, the reaction mixture was cooled to room temperature. The excess solvent was evaporated, followed by the addition of 50 ml of cold water. The precipitates formed were filtered to obtain a crude product, which was purified by column chromatography using CHCl3
1) (10 ml) in the presence of K2CO3 (100 mg, 0.724 mmol) and Pd(PPh3)4 (6 mg, 0.005 mmol) and subjected to the Suzuki–Miyaura cross-coupling reaction (Scheme 1). The reaction mixture was refluxed for 8 h until the starting materials were completely consumed, and the reaction progress was monitored through TLC. After completion, the reaction mixture was cooled to room temperature. The excess solvent was evaporated, followed by the addition of 50 ml of cold water. The precipitates formed were filtered to obtain a crude product, which was purified by column chromatography using CHCl3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOAc (80
EtOAc (80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20) as an eluent to afford the pure yellow colored product 3 in 85% yield. M.pt. >300 °C. 1H NMR of compound 3 (CDCl3, 400 MHz): δ (ppm) 11.2 (d, J = 7.36 Hz, 1H, OH), 10.01 (s, 1H, CHO), 8.89 (d, J = 6.4 Hz, 1H, ArH), 8.83 (m, 1H, ArH), 8.57 (m,1H, ArH), 8.14 (d, J = 8.48 Hz, 1H, ArH), 7.89 (t, 1H, ArH), 7.75 (m, 4H, ArH), 7.50 (m, 2H, ArH), 7.20 (m, 1H, ArH) (Fig. S1). 13C NMR of compound 3 (CDCl3, 100 MHz): δ (ppm) 196.5, 191.8, 161.9, 145.6, 145.2, 135.8, 135.3, 132.3, 131.7, 130.5, 130.4, 128.1, 127.1, 120.8, 118.6 (Fig. S2). HRMS (ESI-TOF): (m/z) [M + H]+ calcd for C25H15N2O3: 391.1004, found: 391.1010.
20) as an eluent to afford the pure yellow colored product 3 in 85% yield. M.pt. >300 °C. 1H NMR of compound 3 (CDCl3, 400 MHz): δ (ppm) 11.2 (d, J = 7.36 Hz, 1H, OH), 10.01 (s, 1H, CHO), 8.89 (d, J = 6.4 Hz, 1H, ArH), 8.83 (m, 1H, ArH), 8.57 (m,1H, ArH), 8.14 (d, J = 8.48 Hz, 1H, ArH), 7.89 (t, 1H, ArH), 7.75 (m, 4H, ArH), 7.50 (m, 2H, ArH), 7.20 (m, 1H, ArH) (Fig. S1). 13C NMR of compound 3 (CDCl3, 100 MHz): δ (ppm) 196.5, 191.8, 161.9, 145.6, 145.2, 135.8, 135.3, 132.3, 131.7, 130.5, 130.4, 128.1, 127.1, 120.8, 118.6 (Fig. S2). HRMS (ESI-TOF): (m/z) [M + H]+ calcd for C25H15N2O3: 391.1004, found: 391.1010.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) to illustrate the ion sensing behaviour of NaHB. The stoichiometry of the complex was determined by Job's plot. Further titration studies were done to determine the detection limit and the binding constant values of the complex formed.
1) to illustrate the ion sensing behaviour of NaHB. The stoichiometry of the complex was determined by Job's plot. Further titration studies were done to determine the detection limit and the binding constant values of the complex formed.
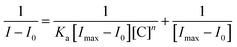
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9; v/v)) was observed at 402 nm. NaHB upon excitation at 402 nm showed emission at 520 nm with a Stokes shift of 118 nm (Fig. 1). In order to study the effect of solvents, absorption and emission spectra of NaHB were examined in a variety of solvents (Table 1). While the solvent polarity was increased, a redshift of 25 nm from 398 nm (cyclohexane) to 423 nm (water) was found in the case of absorption spectra (Fig. 2(a)). Also, NaHB extended its emission maxima region from 510 nm (cyclohexane) to 630 nm (water) as the solvent's polarity increased (Fig. 2(b)). This clearly demonstrates that upon increasing polarity, the excited state becomes more stabilized. The charge transfer could be a possible reason for the expansion of emission spectra in polar solvents. The noticeable emission colour change with solvent polarity was observed, in line with spectral shifts (Fig. 2(c)).
9; v/v)) was observed at 402 nm. NaHB upon excitation at 402 nm showed emission at 520 nm with a Stokes shift of 118 nm (Fig. 1). In order to study the effect of solvents, absorption and emission spectra of NaHB were examined in a variety of solvents (Table 1). While the solvent polarity was increased, a redshift of 25 nm from 398 nm (cyclohexane) to 423 nm (water) was found in the case of absorption spectra (Fig. 2(a)). Also, NaHB extended its emission maxima region from 510 nm (cyclohexane) to 630 nm (water) as the solvent's polarity increased (Fig. 2(b)). This clearly demonstrates that upon increasing polarity, the excited state becomes more stabilized. The charge transfer could be a possible reason for the expansion of emission spectra in polar solvents. The noticeable emission colour change with solvent polarity was observed, in line with spectral shifts (Fig. 2(c)).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000
000![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100
100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600
600![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200
200![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200
200![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200
200![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400
400![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100
100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800
800![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300
300![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500
500![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600
600

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) medium. NaHB exhibited absorption maxima (λmax) at 402 nm, and an emission peak was observed at 520 nm in the CH3CN
1) medium. NaHB exhibited absorption maxima (λmax) at 402 nm, and an emission peak was observed at 520 nm in the CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) solvent medium. The sensing behavior of NaHB has been explored towards various metal ions (Na+, K+, Mg2+, Ba2+, Al3+, Ca2+, Fe2+, Fe3+, Co2+, Ni2+, Cu2+, Zn2+, Ga3+, Cd2+, Hg2+ and Pb2+), anions (CH3COO−, F−, Cl−, Br−, CN−, H2PO4−, HSO4−, NO3−, AcO−, ClO−, P2O74−, and SCN−) and nerve agents (DCP, TEP (triethyl phosphate), and N-BP (N-butyl phosphate)) using UV-visible and fluorescence spectrophotometric techniques. We observed an exceptional response of NaHB towards Ga3+ ions and DCP with high sensitivity and selectivity via an enhancement in the emission band, whereas other anions and metal ions did not show any significant change in absorption and emission spectra.
1) solvent medium. The sensing behavior of NaHB has been explored towards various metal ions (Na+, K+, Mg2+, Ba2+, Al3+, Ca2+, Fe2+, Fe3+, Co2+, Ni2+, Cu2+, Zn2+, Ga3+, Cd2+, Hg2+ and Pb2+), anions (CH3COO−, F−, Cl−, Br−, CN−, H2PO4−, HSO4−, NO3−, AcO−, ClO−, P2O74−, and SCN−) and nerve agents (DCP, TEP (triethyl phosphate), and N-BP (N-butyl phosphate)) using UV-visible and fluorescence spectrophotometric techniques. We observed an exceptional response of NaHB towards Ga3+ ions and DCP with high sensitivity and selectivity via an enhancement in the emission band, whereas other anions and metal ions did not show any significant change in absorption and emission spectra.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) solvent revealed a blueshift from 402 nm to 396 nm, while all other metal ions did not show any significant response in the absorption spectrum. Upon incremental addition of Ga3+, both absorption bands exhibited a distinct blue shift; the absorption intensity at 396 nm was increased and attained a plateau after the addition of 60 µM Ga3+ ions (Fig. S7(b)), indicating that complex formation perturbs the electronic structure of NaHB. The hypsochromic shift suggests a reduction in conjugation and suppression of ICT as a result of coordination between Ga3+ and the donor atoms (carbonyl oxygen and imine nitrogen) or interaction with the phosphoryl center of DCP. These interactions draw electron density away from the conjugated π-system, thereby increasing the HOMO–LUMO energy gap and shifting the absorption toward shorter wavelengths.
1) solvent revealed a blueshift from 402 nm to 396 nm, while all other metal ions did not show any significant response in the absorption spectrum. Upon incremental addition of Ga3+, both absorption bands exhibited a distinct blue shift; the absorption intensity at 396 nm was increased and attained a plateau after the addition of 60 µM Ga3+ ions (Fig. S7(b)), indicating that complex formation perturbs the electronic structure of NaHB. The hypsochromic shift suggests a reduction in conjugation and suppression of ICT as a result of coordination between Ga3+ and the donor atoms (carbonyl oxygen and imine nitrogen) or interaction with the phosphoryl center of DCP. These interactions draw electron density away from the conjugated π-system, thereby increasing the HOMO–LUMO energy gap and shifting the absorption toward shorter wavelengths.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)), a weak fluorescence intensity was observed at 520 nm. The addition of various metal ions to NaHB did not show any variation in the emission spectrum except for Ga3+. The presence of Ga3+ ions exhibited an increase in fluorescence intensity with quick response at 520 nm, displaying excellent bright green fluorescence under UV light (Fig. 6(a)).
1)), a weak fluorescence intensity was observed at 520 nm. The addition of various metal ions to NaHB did not show any variation in the emission spectrum except for Ga3+. The presence of Ga3+ ions exhibited an increase in fluorescence intensity with quick response at 520 nm, displaying excellent bright green fluorescence under UV light (Fig. 6(a)).

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, [v/v]) (a) in the presence of various metal ions (50 equiv.) and (b) upon incremental addition of 0–50 µM Ga3+ ions.
1, [v/v]) (a) in the presence of various metal ions (50 equiv.) and (b) upon incremental addition of 0–50 µM Ga3+ ions.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). The obtained LOD value was compared with reported values for chemosensors, particularly naphthalimide-based derivatives using fluorometric methods, and was found to be superior to those previously reported (Table S1). The fluorescence response of NaHB with Ga3+ and other metal ions can be seen under UV light by the naked eye (Fig. 7).
1). The obtained LOD value was compared with reported values for chemosensors, particularly naphthalimide-based derivatives using fluorometric methods, and was found to be superior to those previously reported (Table S1). The fluorescence response of NaHB with Ga3+ and other metal ions can be seen under UV light by the naked eye (Fig. 7).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). By consecutive addition of different analytes (1 mM) in the complex of NaHB + Ga3+, the spectral change has been measured. The study was done in 3 replicates and the standard deviations along with the p-value (probability of interference of other analytes) obtained from the t-test are incorporated in Table S2. The spectral signal does not get altered upon the addition of other metal ions, indicating no interference in the binding of NaHB + Ga3+. This signifies that NaHB possesses no response ability towards most of the analytes except Ga3+ ions (Fig. 8).
1). By consecutive addition of different analytes (1 mM) in the complex of NaHB + Ga3+, the spectral change has been measured. The study was done in 3 replicates and the standard deviations along with the p-value (probability of interference of other analytes) obtained from the t-test are incorporated in Table S2. The spectral signal does not get altered upon the addition of other metal ions, indicating no interference in the binding of NaHB + Ga3+. This signifies that NaHB possesses no response ability towards most of the analytes except Ga3+ ions (Fig. 8).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) were prepared. A graph was plotted with the emission response in each case at 520 nm and the mole fraction of Ga3+ ions (Fig. S8). Based on Job's plot, it was found that the ratio of complexation of Ga3+ ions and NaHB was 1
1) were prepared. A graph was plotted with the emission response in each case at 520 nm and the mole fraction of Ga3+ ions (Fig. S8). Based on Job's plot, it was found that the ratio of complexation of Ga3+ ions and NaHB was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Moreover, the ratio of the formation of the complex was confirmed by ESI-mass spectral analysis. The mass was taken for the mixture of NaHB and Ga3+ in CH3CN
1. Moreover, the ratio of the formation of the complex was confirmed by ESI-mass spectral analysis. The mass was taken for the mixture of NaHB and Ga3+ in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), where a new peak at m/z = 546.4014 corresponding to the complex of (NaHB + Ga3+ − H+) was observed (Fig. S9), indicating the formation of a 1
1), where a new peak at m/z = 546.4014 corresponding to the complex of (NaHB + Ga3+ − H+) was observed (Fig. S9), indicating the formation of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex.
1 complex.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). No significant change was observed in the case of various analytes, while DCP showed a blue shift of 13 nm (at 389 nm) upon its addition to NaHB (Fig. 9(a)). The interaction of the probe with DCP was systematically examined by UV-vis titration studies in CH3CN
1). No significant change was observed in the case of various analytes, while DCP showed a blue shift of 13 nm (at 389 nm) upon its addition to NaHB (Fig. 9(a)). The interaction of the probe with DCP was systematically examined by UV-vis titration studies in CH3CN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) (Fig. 9(b)). Upon incremental addition of DCP to the solution of the probe, the absorption spectra exhibited significant changes, indicating strong host–guest interaction. A gradual increase (hyperchromic effect) in the absorption band at 389 nM was observed, accompanied by a slight hypsochromic shift. A plateau was obtained after the addition of 50 µM DCP during titration, suggesting effective binding of DCP at the recognition site of the probe.
1) (Fig. 9(b)). Upon incremental addition of DCP to the solution of the probe, the absorption spectra exhibited significant changes, indicating strong host–guest interaction. A gradual increase (hyperchromic effect) in the absorption band at 389 nM was observed, accompanied by a slight hypsochromic shift. A plateau was obtained after the addition of 50 µM DCP during titration, suggesting effective binding of DCP at the recognition site of the probe.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, which can be seen from the graph (Fig. S10(b)). To further support Job's plot, the ESI-mass spectrum of NaHB in the presence of DCP was obtained. A reaction was performed between NaHB and DCP to isolate and determine the mass of the product formed. The HRMS (ESI+) of the isolated reaction material showed a prominent ion at m/z ≈ [M + 136 + H]+ (m/z = 615.1794, calcd = 615.1790, Δ = 8 ppm), indicating addition of a mass equivalent to DCP (≈+136 Da) (Fig. S11(a)). However, tandem MS (MS/MS) of this ion under collision-induced dissociation conditions reproducibly produced the parent probe ion (m/z = 479.1509) and free DCP fragments (m/z = 137) as the major products (Fig. S11(b)), even at moderate collision energies, indicating facile cleavage of the added mass. This fragmentation behavior strongly suggests that the interaction between NaHB and DCP is primarily non-covalent.
1, which can be seen from the graph (Fig. S10(b)). To further support Job's plot, the ESI-mass spectrum of NaHB in the presence of DCP was obtained. A reaction was performed between NaHB and DCP to isolate and determine the mass of the product formed. The HRMS (ESI+) of the isolated reaction material showed a prominent ion at m/z ≈ [M + 136 + H]+ (m/z = 615.1794, calcd = 615.1790, Δ = 8 ppm), indicating addition of a mass equivalent to DCP (≈+136 Da) (Fig. S11(a)). However, tandem MS (MS/MS) of this ion under collision-induced dissociation conditions reproducibly produced the parent probe ion (m/z = 479.1509) and free DCP fragments (m/z = 137) as the major products (Fig. S11(b)), even at moderate collision energies, indicating facile cleavage of the added mass. This fragmentation behavior strongly suggests that the interaction between NaHB and DCP is primarily non-covalent.
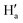 and
and 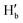 ) and 7.14–7.20 ppm (
) and 7.14–7.20 ppm (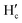 and
and 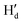 ), respectively. Moreover, the
), respectively. Moreover, the 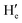 proton gets slightly deshielded from 7.76–7.78 ppm to 7.78–7.81 ppm (Fig. 11). A similar trend was seen in the case of DCP binding with NaHB. The
proton gets slightly deshielded from 7.76–7.78 ppm to 7.78–7.81 ppm (Fig. 11). A similar trend was seen in the case of DCP binding with NaHB. The  and
and  protons experienced deshielding with a shift of 0.13–0.16 ppm during the complexation of DCP with NaHB. Similarly,
protons experienced deshielding with a shift of 0.13–0.16 ppm during the complexation of DCP with NaHB. Similarly,  and
and  protons also shifted towards the downfield region (7.15–7.20 ppm). However, the
protons also shifted towards the downfield region (7.15–7.20 ppm). However, the 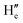 and
and 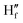 protons did not experience the shifts as these protons were far from the binding sites, but a doublet from NaHB of He got almost detached from the multiplet, thereby increasing the range from 7.76–7.78 ppm to 7.76–7.81 ppm. The results indicated that in the case of complexation with Ga3+, both the imine nitrogen and the hydroxyl group were involved, making the nearby protons deshielded. The deprotonation of the hydroxyl group was caused by the phosphate group of DCP, therefore shifting the proton signals to the downfield region. Moreover, no further change was observed with increasing equivalents of the analyte, confirming the stoichiometry as 1
protons did not experience the shifts as these protons were far from the binding sites, but a doublet from NaHB of He got almost detached from the multiplet, thereby increasing the range from 7.76–7.78 ppm to 7.76–7.81 ppm. The results indicated that in the case of complexation with Ga3+, both the imine nitrogen and the hydroxyl group were involved, making the nearby protons deshielded. The deprotonation of the hydroxyl group was caused by the phosphate group of DCP, therefore shifting the proton signals to the downfield region. Moreover, no further change was observed with increasing equivalents of the analyte, confirming the stoichiometry as 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, as obtained from Job's plot.
1, as obtained from Job's plot.


![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and the electron–hole distribution of S0–S2 orbitals.
1) and the electron–hole distribution of S0–S2 orbitals.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and the electron–hole distribution of S0–S1 orbitals.
1) and the electron–hole distribution of S0–S1 orbitals.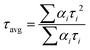

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, [v/v]) upon repetitive addition of metal ions and EDTA.
1, [v/v]) upon repetitive addition of metal ions and EDTA.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (9
H2O (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) for 5 minutes and then air-drying for several hours. Subsequently, aqueous solutions of Ga3+ at different concentrations ranging from 10−1 M to 10−6 M were added dropwise onto the coated strips. The interaction between Ga3+ ions and NaHB on the strips was monitored visually, with particular attention paid to any observable changes such as color variation or intensity, which could indicate complex formation or sensor response (Fig. S12). Following this, different ion solutions (10−1 M) were added dropwise to the strips and then allowed to spread on the surface of the prepared coated strips in air. The fluorescence intensity of the paper strip consisting of Ga3+ and DCP was quite high when observed under a 365 nm UV lamp (Fig. 16). This implies that the fabricated paper strips of NaHB can be used for on-spot detection of Ga3+ and DCP in the presence of other metal ions, anions and nerve agents.
1) for 5 minutes and then air-drying for several hours. Subsequently, aqueous solutions of Ga3+ at different concentrations ranging from 10−1 M to 10−6 M were added dropwise onto the coated strips. The interaction between Ga3+ ions and NaHB on the strips was monitored visually, with particular attention paid to any observable changes such as color variation or intensity, which could indicate complex formation or sensor response (Fig. S12). Following this, different ion solutions (10−1 M) were added dropwise to the strips and then allowed to spread on the surface of the prepared coated strips in air. The fluorescence intensity of the paper strip consisting of Ga3+ and DCP was quite high when observed under a 365 nm UV lamp (Fig. 16). This implies that the fabricated paper strips of NaHB can be used for on-spot detection of Ga3+ and DCP in the presence of other metal ions, anions and nerve agents.












