Open Access Article
Open Access ArticleLinkage isomerism in a homoleptic Fe(II) complex with BODIPY-1H-tetrazole ligands
Matthias Schöbinger†
 *a,
Martin Huber†
*a,
Martin Huber† a,
Berthold Stöger
a,
Berthold Stöger b and
Peter Weinberger
b and
Peter Weinberger a
a
aInstitute of Applied Synthetic Chemistry, TU Wien, Getreidemarkt 9/163, 1060, Vienna, Austria. E-mail: matthias.schoebinger@tuwien.ac.at; peter.e163.weinberger@tuwien.ac.at
bX-Ray Center, TU Wien, Getreidemarkt 9/164, 1060, Vienna, Austria
First published on 8th January 2026
Abstract
We examined the coordination behavior of the sterically demanding 4,4-difluoro-1,3,5,7-tetramethyl-8-[(1H-tetrazol-1-yl)methyl]-4-bora-3a,4a-diaza-s-indacene ligand L in octahedral Fe(II) coordination compounds. Using four different solvents – ClCH2CN, BrCH2CN, CH3OH, and (CH3)2CO – five unique crystal structures were obtained and characterized by single-crystal X-ray diffraction. In ClCH2CN and BrCH2CN, L coordinated equatorially through N4 and apically through its less basic N3 atom, yielding a pseudo-homoleptic [Fe(LN4)4(LN3)2]2+ architecture. This represents the first documented example of monodentate 1H-tetrazole N3/N4 linkage isomerism in an Fe(II) complex. Conversely, O-donor solvents (CH3OH and (CH3)2CO) resulted in the formation of a heteroleptic [Fe(L)4(solvent)2]2+ motif, in which the solvent molecules coordinated through their O-atoms and occupied two coordination sites. The CH3OH adduct underwent a topotactic single-crystal-to-single-crystal transformation upon air exposure, exchanging CH3OH for H2O, and was stabilized by extensive hydrogen bonding. Unlike classical [Fe(1-alkyl-1H-tetrazole)6]2+ systems, none of the investigated coordination compounds exhibited SCO properties, instead remained locked in a single spin state. These findings demonstrate that steric bulk can override the ligand's N4 coordination preference to Fe(II), either by enforcing N3 coordination or by permitting coordination of co-ligands, and thus provide guidance for the design of 1H-tetrazole-based Fe(II) materials.
Introduction
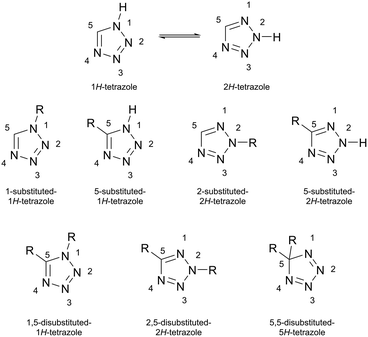
Tetrazoles (tz), five-membered heterocycles containing four adjacent nitrogen atoms, have attracted considerable attention across coordination chemistry, materials science, and medicinal chemistry. Their unique properties including stability, acidity and basicity, high dipole moment, high formation enthalpy among others make them ideal building blocks for functional materials and bioactive molecules.1–5Tz exists in two stable tautomeric forms (1H-tz and 2H-tz), each of which can be mono- or disubstituted (Scheme 1). Monosubstitution occurs at N1, N2, or C5, whereas disubstitution yields 1,5-; 2,5- or 5,5-derivatives.6 The four donor nitrogen atoms of tz enable a wide variety of coordination modes to transition metals, which strongly depend on the substitution pattern.7 Since the pioneering work of Franke et al. in 1982, who first reported an octahedral Fe(II) complex of N1-alkyltetrazoles exhibiting spin crossover (SCO) behavior,8 this ligand class has attracted growing interest for Fe(II) SCO materials, owing to their optimal ligand field strength.9 Within the N1-substituted tetrazole ligands, the N4 nitrogen is the most basic site, followed by N3.10 Consequently, N1-substituted tetrazoles coordinate exclusively in a monodentate fashion through the N4 donor atom to Fe(II).9 In contrast, C5-substituted tetrazoles exhibit a broader range of binding motifs, including bridging interactions between Fe(II) centers.3,11–14
To realize extended SCO coordination polymers from N1-substituted tetrazoles 1,ω-di(tetrazol-1-yl) derivatives have been employed to bridge Fe(II) centers, yielding 1D,15–17 2D,18 and 3D frameworks.19,20
Using N1-substituted tetrazoles as ligands with Fe(II) salts of non-coordinating anions (A; e.g., BF4−, ClO4−) typically yields homoleptic, mononuclear complexes of the general formula [Fe(R-tetrazol-1-yl)6]A2, in which six N1-substituted tetrazole ligands occupy an octahedral coordination sphere.21–26 Heteroleptic analogues are still relatively rare. Mononuclear examples featuring axial coordination by water27 or anions such as NCS−28 have been reported; however, axial coordination of organic nitrile co-ligands has so far been restricted to polymeric networks in which bridging 1,ω-di(tetrazol-1-yl)alkane ligands occupy the equatorial positions.29–31
In our previous work, we described the first heteroleptic mononuclear Fe(II) complex bearing organic nitriles (RCN; R = alkyl, alkenyl, aryl) co-ligands, [Fe(L)4(RCN)2]A2·xsolv.,32,33 where L is the tetrazol-1-yl ligand functionalized with a BODIPY (4,4-difluoro-1,3,5,7-tetramethyl-4-bora-3a,4a-diaza-s-indacene) fluorophore in N1 position of tz (Chart 1),34 A the non-coordinating anion and solv. the crystal solvates. Some coordination compounds of this family undergo a thermally induced spin transition from a low-spin (LS, S = 0) to a high-spin (HS, S = 2) state with a modest synergistic response of its photoluminescence signal. To enhance the modulation of the PL signal and probe its influence on the spin crossover (SCO) behavior, we now target a homoleptic analogue [Fe(L)6](ClO4)2 featuring six L ligands coordinated to Fe(II).
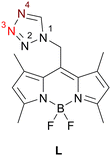 | ||
| Chart 1 Chemical structure of L with highlighted coordinating atom(s) for 1 and 2: N4 (dark red) and N3 (light red), for 3 and 4: N4 (dark red). | ||
Although strategies to selectively control the formation of heteroleptic versus homoleptic complexes have not been explored, we propose that judicious solvent selection during complexation can steer the system toward a homoleptic architecture, thereby tuning both SCO behavior and photoluminescence response.
Results and discussion
Synthesis
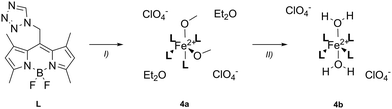
The synthesis of the ligand L and the coordination compounds 1–4 was carried out according to previously described procedures.32–34 L and Fe(ClO4)2·6H2O were mixed and stirred in different solvents (ClCH2CN (1), BrCH2CN (2), acetone (3) and CH3OH (4a)) at 40 °C overnight. The crude products were precipitated and washed with diethyl ether (Et2O) to yield 1–4a in moderate yields (Schemes 2 and 3).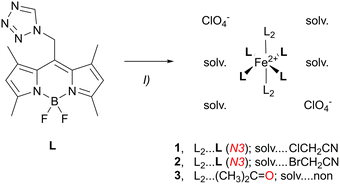 | ||
| Scheme 2 Synthesis of coordination compounds 1–3, reagents and conditions: (I) Fe(ClO4)2·6H2O, ClCH2CN (1), BrCH2CN (2), acetone (3) 40 °C, o.n., 75.3% (1), 68.9% (2), 56.6% (3). | ||
In contrast to our recent studies,32,33 where the use of nitrile solvents (RCN) led to the formation of heteroleptic complexes with RCN as co-ligands, a homoleptic coordination environment was obtained in 1. This coordination mode was reproducible with the related nitrile BrCH2CN in 2, but not when weaker coordinating solvents such as acetone or methanol were used, as in 3 and 4.
Reaction monitoring was performed using X-ray powder diffraction (XRPD) and IR spectroscopy, with the νCH(tz) band serving as an indicator of successful complexation.
Remarkably, coordination compound 4b was obtained from 4a in a single-crystal-to-single-crystal (SC-to-SC) transformation (vide infra, Scheme 3).
Crystal structures
Single crystals (SCs) of coordination compounds 1–4 were obtained via slow diffusion of Et2O into saturated solutions of the respective solvents – ClCH2CN for 1, BrCH2CN for 2, acetone for 3, and CH3OH for 4 – at room temperature (rt).Coordination compound 1 crystallizes in the triclinic P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group and features one crystallographically unique mononuclear complex located on a center of inversion. The complex exhibits a homoleptic octahedral coordination environment, consisting of six molecules of L. However, contrary to our expectations only four molecules of L coordinate via their tetrazolic N4-atom (the italicized numbered N-atoms correspond to the tetrazole numbering system in Chart 1), whereas two molecules of L coordinate via their tetrazolic N3-atom (Fig. 1 and Fig. S3). This N3-coordination mode of a monodentate N1-substituted tetrazole remains hitherto unknown since, in Fe(II) coordination compounds, these ligands coordinate preferably via their N4-atom.9 Coordination through the N3-atom is shown in literature typically only in a bidentate binding mode of N1-substituted tz with e.g. coinage metals. In the case of L such a bidentate coordination mode was previously shown in Ag coordination compounds by our group.35 To our knowledge, this is the first report of a tetrazole N3/N4 linkage isomerism36 in a Fe(II) system bearing monodentate tetrazol-1-yl ligands. Note that the N and C-Atoms of the tz rings could be unambiguously assigned owing to their different atomic form factors and the H-atom attached to C was in all cases clearly located in the difference Fourier map. The L molecules in the trans arrangement are crystallographically equivalent, whereas those in cis position are conformers (Fig. S1 and S2). These conformers differ in the orientation of the tz moiety relative to the FBF plane of the BODIPY core, as previously established by structural and theoretical studies on L.34
space group and features one crystallographically unique mononuclear complex located on a center of inversion. The complex exhibits a homoleptic octahedral coordination environment, consisting of six molecules of L. However, contrary to our expectations only four molecules of L coordinate via their tetrazolic N4-atom (the italicized numbered N-atoms correspond to the tetrazole numbering system in Chart 1), whereas two molecules of L coordinate via their tetrazolic N3-atom (Fig. 1 and Fig. S3). This N3-coordination mode of a monodentate N1-substituted tetrazole remains hitherto unknown since, in Fe(II) coordination compounds, these ligands coordinate preferably via their N4-atom.9 Coordination through the N3-atom is shown in literature typically only in a bidentate binding mode of N1-substituted tz with e.g. coinage metals. In the case of L such a bidentate coordination mode was previously shown in Ag coordination compounds by our group.35 To our knowledge, this is the first report of a tetrazole N3/N4 linkage isomerism36 in a Fe(II) system bearing monodentate tetrazol-1-yl ligands. Note that the N and C-Atoms of the tz rings could be unambiguously assigned owing to their different atomic form factors and the H-atom attached to C was in all cases clearly located in the difference Fourier map. The L molecules in the trans arrangement are crystallographically equivalent, whereas those in cis position are conformers (Fig. S1 and S2). These conformers differ in the orientation of the tz moiety relative to the FBF plane of the BODIPY core, as previously established by structural and theoretical studies on L.34
Each mononuclear complex is accompanied by two non-coordinating anion molecules (ClO4−) and four intercalated solvent molecules (ClCH2CN) (Fig. S4). These solvent molecules are disordered about two positions with an occupancy ratio of 57.6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 42.4(4) at 100 K and 53.5
42.4(4) at 100 K and 53.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46.5(11) at 300 K. The model derived from the 100 K measurement additionally features a diffuse electron density peak, which we interpret as water of crystallization. At 300 K the anion likewise becomes disordered about two positions with an occupancy ratio of 78.5
46.5(11) at 300 K. The model derived from the 100 K measurement additionally features a diffuse electron density peak, which we interpret as water of crystallization. At 300 K the anion likewise becomes disordered about two positions with an occupancy ratio of 78.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 21.5(8), which can be interconverted by rotation along the Cl1–O2 axis (Fig. S6). At both measuring temperatures (100 K and 300 K) N–Fe distances (Table 1) suggest that the compound is in LS state.37
21.5(8), which can be interconverted by rotation along the Cl1–O2 axis (Fig. S6). At both measuring temperatures (100 K and 300 K) N–Fe distances (Table 1) suggest that the compound is in LS state.37
| Compound | T/K | Spin state | N12(tz)-Fe1/Å | N18(tz)-Fe1/Å | N5(tz)-Fe1/Å | tz FBF/°g | tz FBF/°h | tz FBF/°i | ∑/°j | Fe1O1C31/° | Cl1-Fe1/Å | unit cell volume/Å3 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
a N6-Fe1.b N12-Fe1.c N17-Fe1.d N24-Fe1.e O1-Fe1.f O2-Fe1.g tz = N3N4N5N6![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) ![[1 with combining low line]](https://www.rsc.org/images/entities/char_0031_0332.gif) ![[5 with combining low line]](https://www.rsc.org/images/entities/char_0035_0332.gif) (N3 (N3![[N with combining low line]](https://www.rsc.org/images/entities/char_004e_0332.gif) ![[4 with combining low line]](https://www.rsc.org/images/entities/char_0034_0332.gif) ![[N with combining low line]](https://www.rsc.org/images/entities/char_004e_0332.gif) ![[5 with combining low line]](https://www.rsc.org/images/entities/char_0035_0332.gif) N6C15 for 2, 3, 4a and 4b), FBF= F1B1F2 and tz = N15 N6C15 for 2, 3, 4a and 4b), FBF= F1B1F2 and tz = N15![[N with combining low line]](https://www.rsc.org/images/entities/char_004e_0332.gif) ![[1 with combining low line]](https://www.rsc.org/images/entities/char_0031_0332.gif) ![[6 with combining low line]](https://www.rsc.org/images/entities/char_0036_0332.gif) ![[N with combining low line]](https://www.rsc.org/images/entities/char_004e_0332.gif) ![[1 with combining low line]](https://www.rsc.org/images/entities/char_0031_0332.gif) ![[7 with combining low line]](https://www.rsc.org/images/entities/char_0037_0332.gif) N18C45, FBF = F5B3F6 for 4b second value, underlined atoms face toward the FBF plane.h tz = N9 N18C45, FBF = F5B3F6 for 4b second value, underlined atoms face toward the FBF plane.h tz = N9![[N with combining low line]](https://www.rsc.org/images/entities/char_004e_0332.gif) ![[1 with combining low line]](https://www.rsc.org/images/entities/char_0031_0332.gif) ![[0 with combining low line]](https://www.rsc.org/images/entities/char_0030_0332.gif) ![[N with combining low line]](https://www.rsc.org/images/entities/char_004e_0332.gif) ![[1 with combining low line]](https://www.rsc.org/images/entities/char_0031_0332.gif) ![[1 with combining low line]](https://www.rsc.org/images/entities/char_0031_0332.gif) N12C30 (N9N10N11N12 N12C30 (N9N10N11N12![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) ![[3 with combining low line]](https://www.rsc.org/images/entities/char_0033_0332.gif) ![[0 with combining low line]](https://www.rsc.org/images/entities/char_0030_0332.gif) for 4a), FBF = F3B2F4 and tz = N21 for 4a), FBF = F3B2F4 and tz = N21![[N with combining low line]](https://www.rsc.org/images/entities/char_004e_0332.gif) ![[2 with combining low line]](https://www.rsc.org/images/entities/char_0032_0332.gif) ![[2 with combining low line]](https://www.rsc.org/images/entities/char_0032_0332.gif) ![[N with combining low line]](https://www.rsc.org/images/entities/char_004e_0332.gif) ![[2 with combining low line]](https://www.rsc.org/images/entities/char_0032_0332.gif) ![[3 with combining low line]](https://www.rsc.org/images/entities/char_0033_0332.gif) N24C60, FBF = F7B4F8 for 4b second value, underlined atoms face toward the FBF plane.i tz = N15 N24C60, FBF = F7B4F8 for 4b second value, underlined atoms face toward the FBF plane.i tz = N15![[N with combining low line]](https://www.rsc.org/images/entities/char_004e_0332.gif) ![[1 with combining low line]](https://www.rsc.org/images/entities/char_0031_0332.gif) ![[6 with combining low line]](https://www.rsc.org/images/entities/char_0036_0332.gif) ![[N with combining low line]](https://www.rsc.org/images/entities/char_004e_0332.gif) ![[1 with combining low line]](https://www.rsc.org/images/entities/char_0031_0332.gif) ![[7 with combining low line]](https://www.rsc.org/images/entities/char_0037_0332.gif) N18C45 (N15N16N17N18 N18C45 (N15N16N17N18![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) ![[4 with combining low line]](https://www.rsc.org/images/entities/char_0034_0332.gif) ![[5 with combining low line]](https://www.rsc.org/images/entities/char_0035_0332.gif) for 2), FBF = F5B3F6, underlined atoms face toward the FBF plane.j Sum of the deviation from 90° of all twelve cis-NFeN(O) angles.k Cl2-Fe1. for 2), FBF = F5B3F6, underlined atoms face toward the FBF plane.j Sum of the deviation from 90° of all twelve cis-NFeN(O) angles.k Cl2-Fe1. |
||||||||||||
| 1 | 100 | LS | 1.9639(17) | 1.9831(19) | 1.955(2) | 38.5(2) | 21.8(2) | 8.3(4) | 25.2 | — | 5.3649(9) | 2856.6(6)0 |
| 300 | LS | 2.007(3) | 2.022(4) | 1.993(4) | 36.0(5) | 22.3(6) | 9.9(10) | 24.0 | — | 5.389(4) | 2942(3) | |
| 2 | 100 | LS | 1.964(3) | 1.993(3)a | 1.963(4)c | 8.1(6) | 21.5(4) | 39.5(4) | 26.4 | — | 5.3834(12) | 2884.9(4) |
| 3 | 100 | HS | 2.1802(19)a | 2.1635(19)b | 2.1155(19)e | 20.2(2) | 36.4(2) | — | 34.8 | 146.79(18) | 6.0992(7) | 3810.61(12) |
| 4a | 100 | HS | 2.206(2)a | 2.182(2)b | 2.098(3)e | 23.0(3) | 27.1(3) | — | 25.0 | 136.3(6) | 5.5589(7) | 8220.1(4) |
| 4b | 100 | HS | 2.184(2) | 2.178(2) | 2.087(2)e | 40.7(3) | 38.3(3) | — | 21.4 | — | 5.5069(10) | 3637.2(3) |
| 2.186(2)a | 2.176(2)d | 2.110(3)f | 27.0(3) | 31.5(3) | 5.3619(10)k | |||||||
Coordination compound 2 is isostructural to 1 at 100 K (Fig. 2 and Fig. S7-S9). However, besides the different nitrile solvent (ClCH2CN in 1, vs. BrCH2CN in 2), 2 features no water of crystallization and the occupancy ratio of the disordered nitrile solvent molecules is different: 67.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32.5(3).
32.5(3).
Coordination compound 3 (Fig. S10–S13) crystallizes in the monoclinic P21/c space group and features acetone as co-ligand in the apical positions, which coordinates to the Fe(II) center via its O-atom (Table 1 and Fig. 3). In the equatorial plane, four molecules of L coordinate to the Fe(II) center via the tetrazolic N4-atom. Based on the symmetry of 3, like in 1, the L molecules in the trans arrangement are crystallographically equivalent, whereas those in cis position are conformers. However, no solvent molecules are incorporated into the crystal lattice. Based on the N–Fe bond lengths (Table 1), 3 is fully in the HS state at 100 K, according to literature,22,37–39 as expected from its coordination environment. Compared to the heteroleptic analogue [Fe(L)4(CH3CN)2](ClO4)2·2CH3CN, which undergoes a thermal spin transition at T1/2 = 265 K,32 3 is expected to exhibit decreased octahedral ligand field splitting (Δo) due to the replacement of CH3CN ligands with acetone. A weaker ligand field leads to the stabilization of the HS configuration.
Coordination compound 4a crystallizes in the monoclinic C2/c space group and features one crystallographically unique mononuclear complex located on a twofold rotation axis (Fig. 4 and Fig. S14–S18). The heteroleptic complex adopts an octahedral geometry with four molecules of L coordinating via their tetrazolic N4-atom and two molecules of methanol functioning as co-ligands. In contrast to 3 and earlier investigations,32,33 where the co-ligands coordinate on the apical positions, in 4a the co-ligand is coordinated by its O-atom in a cis arrangement. The C-atom (C31) of the methanol molecules is disordered about two positions with an occupancy ratio of 68.6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 31.4(3). Based on the symmetry of 4a, the complex features two pairs of crystallographically equivalent molecules of L; however, one pair of conformers is in cis arrangement and the other one in trans arrangement according to the twofold rotation symmetry. Like before, the different conformers exhibit different arrangements of the tz plane vs. the FBF plane.
31.4(3). Based on the symmetry of 4a, the complex features two pairs of crystallographically equivalent molecules of L; however, one pair of conformers is in cis arrangement and the other one in trans arrangement according to the twofold rotation symmetry. Like before, the different conformers exhibit different arrangements of the tz plane vs. the FBF plane.
Beside two non-coordinating anion molecules (ClO4−), for each mononuclear complex, there are two Et2O molecules intercalated. We attribute remaining difference electron density to additional CH3OH solvate molecules, which are realized when the C31-atom adopts its minor position. The O-atom of this molecule was modelled as disordered over four positions and the C-atom as disordered about the twofold axis.
SCs of 4a stored under ambient conditions react with air humidity in a SC-to-SC topotactic transformation to form 4b.40
4b crystallizes in the triclinic P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group and features one crystallographically unique mononuclear complex located on a general position (Fig. S20). The complex of 4b is built similar to 3, but with water as co-ligand in the apical positions and all four molecules of L are crystallographically unique (Fig. S19). The inversion (
space group and features one crystallographically unique mononuclear complex located on a general position (Fig. S20). The complex of 4b is built similar to 3, but with water as co-ligand in the apical positions and all four molecules of L are crystallographically unique (Fig. S19). The inversion (![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ) symmetry of the complex is disrupted by the ligands with the C-atoms C1 and C31. Thus, the structure can be considered as a modulated version of a hypothetic structure with halved c-axis. Again, for each mononuclear complex there are two non-coordinating anions intercalated; however, 4b is devoid of any solvent molecules and shows no disorder (Fig. S21 and S22). The two coordinated water molecules form moderate-strength hydrogen bonds41 with the ClO4− anions and the F-atoms of the L molecules in neighboring complexes. The donor–acceptor distances are listed in Table 2. The hydrogen bonds to the non-coordinating anions generate dimeric assemblies with two neighboring complexes along the [001] direction, whereas the hydrogen bonds to the F-atoms of the BODIPY moiety give rise to a one-dimensionally periodic hydrogen-bonding network extending along the [100] direction (Fig. 5 and Fig. S23-S25).
) symmetry of the complex is disrupted by the ligands with the C-atoms C1 and C31. Thus, the structure can be considered as a modulated version of a hypothetic structure with halved c-axis. Again, for each mononuclear complex there are two non-coordinating anions intercalated; however, 4b is devoid of any solvent molecules and shows no disorder (Fig. S21 and S22). The two coordinated water molecules form moderate-strength hydrogen bonds41 with the ClO4− anions and the F-atoms of the L molecules in neighboring complexes. The donor–acceptor distances are listed in Table 2. The hydrogen bonds to the non-coordinating anions generate dimeric assemblies with two neighboring complexes along the [001] direction, whereas the hydrogen bonds to the F-atoms of the BODIPY moiety give rise to a one-dimensionally periodic hydrogen-bonding network extending along the [100] direction (Fig. 5 and Fig. S23-S25).
| Donor atom (D) | H atom | Acceptor atom (A) | D–H/Å | H⋯A/Å | D⋯A/Å | D–H⋯A/° |
|---|---|---|---|---|---|---|
| O1 | H1A | O10 | 0.86(3) | 1.93(3) | 2.776(3) | 171(4) |
| O1 | H1B | O7 | 0.87(3) | 2.01(3) | 2.826(5) | 158(4) |
| O2 | H2A | O4 | 0.86(3) | 2.08(3) | 2.895(4) | 158(4) |
| O2 | H2B | F1 | 0.86(3) | 2.49(3) | 3.228(3) | 144(4) |
| O2 | H2B | F2 | 0.86(3) | 2.29(3) | 2.845(3) | 122(3) |
Similar to 3, where acetone ligands favor the HS state, the coordination environments of 4a and 4b likewise stabilize the HS configuration, with N–Fe bond distances of the L molecules confirming HS character at 100 K.
XRPD measurements
The bulk powder X-ray diffraction pattern of 1 corresponds to the calculated reflections and the bulk can therefore be considered as single phase (Fig. S33). For 2, the XRPD pattern predominately matches the calculated reflections of 2 (Fig. S34), but shows minor additional reflections, which may correspond to a heteroleptic BrCH2CN analogue of 3, although the absence of suitable SCs prevented definitive identification.The XRPD profile of 3 deviates from its simulated SC pattern, indicating incomplete phase purity or an unidentified secondary phase (Fig. S35). In contrast, the XRPD of 4 reveals an almost exclusive phase 4b (Fig. S36), consistent with an immediate SC-to-SC transformation upon air exposure.
IR spectroscopy
Upon coordination, the νCH(tz) band of L shifts from 3133 cm−1 to 3159 cm−1 in 1, to 3160 cm−1 in 2, to 3125 cm−1 in 3 and to 3144 cm−1 in 4b (Fig. S37–S40). Comparable shifts were also observed in earlier investigations involving the same ligand.32–34 The C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching mode of the coordinated acetone molecules in 3 is evident at 1691 cm−1 as a sharp band. In 4b, the broad O–H stretching band is visible at around 3370 cm−1. As previously described,32–34 the aliphatic C–H stretching vibrations (2990–2870 cm−1) of L show no noteworthy change upon complexation. Furthermore, in the fingerprint region (1600–400 cm−1), the only significant difference compared to the uncoordinated ligand in coordination compounds 1–4 is a band at 621 cm−1, corresponding to the degenerate symmetric bending mode of the ClO4− anions.
O stretching mode of the coordinated acetone molecules in 3 is evident at 1691 cm−1 as a sharp band. In 4b, the broad O–H stretching band is visible at around 3370 cm−1. As previously described,32–34 the aliphatic C–H stretching vibrations (2990–2870 cm−1) of L show no noteworthy change upon complexation. Furthermore, in the fingerprint region (1600–400 cm−1), the only significant difference compared to the uncoordinated ligand in coordination compounds 1–4 is a band at 621 cm−1, corresponding to the degenerate symmetric bending mode of the ClO4− anions.
Conclusions
In this work, we demonstrate that the steric demand of the ligand L can override the typical N4-coordination preference of monodentate 1H-tetrazoles in Fe(II) complexes. When XCH2CN (X = Br, Cl) was employed as solvent, L occupied all four equatorial positions via its N4 donor, while also binding apically through its less basic N3 atom. This resulted in a pseudo-homoleptic [Fe(LN4)4(LN3)2]2+ motif. To the best of our knowledge, this is the first example of monodentate 1H-tetrazole linkage isomerism in an Fe(II) complex. In contrast, steric crowding forced the formation of only heteroleptic [Fe(L)4(solvent)2]2+ species via O-donor coordination when CH3OH or (CH3)2CO served as solvents. Furthermore, the CH3OH derivative underwent an SC–SC topotactic transformation upon exposure to air moisture, exchanging CH3OH for H2O and forming a stabilized hydrogen bonding network in the [Fe(L)4(H2O)2](ClO4)2 compound. Remarkably, the conversion from the cis to the trans arrangement did not cause crystal destruction.Future work will focus on theoretical studies aimed at understanding why XCH2CN yields a pseudo-homoleptic [Fe(LN4)4(LN3)2]2+ complex, whereas other co-ligands (e.g. RCN,33 (CH3)2CO, CH3OH, H2O) consistently give heteroleptic compounds. In parallel, we aim to define the precise steric threshold at which a N1-substituted tetrazole shifts from forming a homoleptic [Fe(L)6]2+ structure to a heteroleptic [Fe(L)4(solvent)2]2+ motif. To this end, a series of N1-substituted tetrazoles with systematically varied substituent bulkiness is being synthesized. Comparing their Fe(II) coordination behavior, both experimentally and via DFT calculations, will reveal the critical substituent size that determines whether six 1H-tetrazole ligands can occupy all coordination sites, or if an O- or N-donor solvent must bind instead.
These insights will ultimately guide the rational design of 1H-tetrazole-based materials, whether for switchable magnetic applications, or advanced crystal engineering, in coordination chemistry and beyond.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the Supplementary information (SI). Crystallographic data for 1, 2, 3, 4a and 4b have been deposited at the CCDC under 2463767–2463772. Supplementary information: experimental protocols, crystal structure, XRPD spectra, and IR spectra. See DOI: https://doi.org/10.1039/d5nj03028e.CCDC 2463767–2463772 contain the supplementary crystallographic data for this paper.42a–f
Acknowledgements
The authors thank Werner Artner (X-ray Center, TU Wien) for his assistance with XRPD measurements. For open access purposes, P. Weinberger has applied a CC BY public copyright license to any author accepted manuscript version arising from this submission.Notes and references
- V. A. Ostrovskii, E. A. Popova and R. E. Trifonov, in Advances in Heterocyclic Chemistry, ed E. F. V. Scriven and C. A. Ramsden, Academic Press, 2017, vol. 123, pp. 1–62 Search PubMed.
- F. R. Benson, Chem. Rev., 1947, 41, 1–61 CrossRef PubMed.
- H. Zhao, Z. R. Qu, H. Y. Ye and R. G. Xiong, Chem. Soc. Rev., 2008, 37, 84–100 RSC.
- Y. Yuan, M. Li, V. Apostolopoulos, J. Matsoukas, W. M. Wolf, M. A. T. Blaskovich, J. Bojarska and Z. M. Ziora, Eur. J. Med. Chem., 2024, 279, 116870 CrossRef CAS PubMed.
- M. Nasrollahzadeh, Z. Nezafat, N. S. S. Bidgoli and N. Shafiei, Mol. Catal., 2021, 513, 111788 CAS.
- H. R. Meier and H. Heimgartner, Tetrazole, Georg Thieme Verlag, 8th edn, 1994 Search PubMed.
- P. N. Gaponik, S. V. Voitekhovich and O. A. Ivashkevich, Russ. Chem. Rev., 2006, 75, 569–603 CrossRef.
- P. L. Franke, J. G. Haasnoot and A. P. Zuur, Inorg. Chim. Acta, 1982, 59, 5–9 CrossRef CAS.
- G. Aromí, L. A. Barrios, O. Roubeau and P. Gamez, Coord. Chem. Rev., 2011, 255, 485–546 CrossRef.
- R. M. Claramunt, D. Sanz, G. Boyer, J. Catalán, J. L. G. de Paz and J. Elguero, Magn. Reson. Chem., 1993, 31, 791–800 CrossRef CAS.
- W.-T. Liu, J.-Y. Li, Z.-P. Ni, X. Bao, Y.-C. Ou, J.-D. Leng, J.-L. Liu and M.-L. Tong, Cryst. Growth Des., 2012, 12, 1482–1488 CrossRef CAS.
- Z. Yan, M. Li, H. L. Gao, X. C. Huang and D. Li, Chem. Commun., 2012, 48, 3960–3962 RSC.
- W. Zhang, F. Zhao, T. Liu, M. Yuan, Z. M. Wang and S. Gao, Inorg. Chem., 2007, 46, 2541–2555 CrossRef CAS.
- L. Sun, A. Rotaru, K. Robeyns and Y. Garcia, Ind. Eng. Chem. Res., 2021, 60, 8788–8798 CrossRef CAS.
- M. Quesada, H. Kooijman, P. Gamez, J. Sanchez Costa, P. J. van Koningsbruggen, P. Weinberger, M. Reissner, A. L. Spek, J. G. Haasnoot and J. Reedijk, Dalton Trans., 2007, 5434–5440, 10.1039/b709460d.
- A. Absmeier, M. Bartel, C. Carbonera, G. N. L. Jameson, F. Werner, M. Reissner, A. Caneschi, J. F. Létard and W. Linert, Eur. J. Inorg. Chem., 2007, 3047–3054 CrossRef CAS.
- D. Müller, C. Knoll, B. Stoger, W. Artner, M. Reissner and P. Weinberger, Eur. J. Inorg. Chem., 2013, 984–991 CrossRef.
- M. Quesada, F. Prins, O. Roubeau, P. Gamez, S. J. Teat, P. J. van Koningsbruggen, J. G. Haasnoot and J. Reedijk, Inorg. Chim. Acta, 2007, 360, 3787–3796 CrossRef CAS.
- G. N. L. Jameson, F. Werner, M. Bartel, A. Absmeier, M. Reissner, J. A. Kitchen, S. Brooker, A. Caneschi, C. Carbonera, J. F. Létard and W. Linert, Eur. J. Inorg. Chem., 2009, 3948–3959 CrossRef CAS.
- C. M. Grunert, J. Schweifer, P. Weinberger, W. Linert, K. Mereiter, G. Hilscher, M. Muller, G. Wiesinger and P. J. van Koningsbruggen, Inorg. Chem., 2004, 43, 155–165 CrossRef CAS PubMed.
- M. Tafili-Kryeziu, M. Weil, T. Muranaka, A. Bousseksou, M. Hasegawa, A. Jun and W. Linert, Dalton Trans., 2013, 42, 15796–15804 RSC.
- W. Zeni, M. Seifried, C. Knoll, J. M. Welch, G. Giester, B. Stöger, W. Artner, M. Reissner, D. Müller and P. Weinberger, Dalton Trans., 2020, 49, 17183–17193 RSC.
- A. A. Soliman, M. M. Khattab, M. Reissner, P. Weinberger, F. Werner and W. Linert, Inorg. Chim. Acta, 2007, 360, 3987–3996 Search PubMed.
- N. Hassan, P. Weinberger, K. Mereiter, F. Werner, G. Molnar, A. Bousseksou, M. Valtiner and W. Linert, Inorg. Chim. Acta, 2008, 361, 1291–1297 Search PubMed.
- N. Hassan, J. Stelzl, P. Weinberger, G. Molnar, A. Bousseksou, F. Kubel, K. Mereiter, R. Boca and W. Linert, Inorg. Chim. Acta, 2013, 396, 92–100 CrossRef CAS PubMed.
- D. Müller, C. Knoll, M. Seifried, J. M. Welch, G. Giester, M. Reissner and P. Weinberger, Chem. – Eur. J., 2018, 24, 5271–5280 Search PubMed.
- V. Braun, M. H. H. Wurzenberger, V. Weippert and J. Stierstorfer, New J. Chem., 2021, 45, 11042–11050 RSC.
- V. Smeets, M. Wolff, J. A. Wolny, V. Schünemann, M. M. Dîrtu, J. Y. Ge, J. Vanacken, V. Moshchalkov and Y. Garcia, Eur. J. Inorg. Chem., 2018, 394–413 CrossRef CAS.
- A. Tołoczko, M. Kazmierczak, M. Książek, M. Weselski, M. Siczek, J. Kusz and R. Bronisz, Dalton Trans., 2024, 53, 7163–7174 RSC.
- A. Tołoczko, M. Kaźmierczak, M. Weselski, M. Siczek, M. Książek, J. Kusz and R. Bronisz, Cryst. Growth Des., 2023, 23, 1611–1621 CrossRef.
- V. Maliuzhenko, M. Weselski, J. Gregolinski, M. Książek, J. Kusz and R. Bronisz, Inorg. Chem., 2024, 63, 17762–17773 CrossRef CAS.
- M. Huber, M. Schobinger, B. Stoger, M. Reissner and P. Weinberger, Cryst. Growth Des., 2025, 25, 8875–8885 CrossRef CAS PubMed.
- M. Huber, M. Schobinger, B. Stoger, M. Reissner and P. Weinberger, Chem. – Eur. J., 2025, e01959, DOI:10.1002/chem.202501959.
- M. Huber, M. Schöbinger, J. Cirera, B. Stöger and P. Weinberger, Eur. J. Org. Chem., 2025, e202401239 CrossRef CAS.
- M. Schöbinger, M. Huber, B. Stöger, C. Hametner and P. Weinberger, CrystEngComm, 2025, 27, 2689–2697 RSC.
- R. T. M. Fraser, Linkage Isomerism, American Chemical Society, 1967 Search PubMed.
- P. Gütlich, J. Jung and H. A. Goodwin, Spin Transitions in Iron(II) Complexes, Springer, Netherlands, Dordrecht, 1996 Search PubMed.
- A. Bialonska and R. Bronisz, Inorg. Chem., 2010, 49, 4534–4542 CrossRef CAS.
- A. Bialonska, R. Bronisz, M. F. Rudolf and M. Weselski, Inorg. Chem., 2012, 51, 237–245 CrossRef CAS PubMed.
- U. Müller and G. de la Flor, Symmetry Relationships between Crystal Structures: Applications of Crystallographic Group Theory in Crystal Chemistry, Oxford University Press, 1st edn, 2013, ch. 16, pp. 217–224 Search PubMed.
- T. Steiner, Angew. Chem., Int. Ed., 2002, 41, 49–76 Search PubMed.
- (a) CCDC 2463767: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2nprch; (b) CCDC 2463768: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2nprdj; (c) CCDC 2463769: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2nprfk; (d) CCDC 2463770: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2nprgl; (e) CCDC 2463771: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2nprhm; (f) CCDC 2463772: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2nprjn.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2026 |