 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Facile synthesis of FeS–Fe3O4 nanocomposites: highly stable & enhanced electrochemical performance in asymmetric supercapacitor applications
Junaid
Riaz
 a,
Zahra
Bayhan
b,
Ghulam
Murtaza
c,
Muhammad
Arif
*a and
Amina
Bibi
*d
a,
Zahra
Bayhan
b,
Ghulam
Murtaza
c,
Muhammad
Arif
*a and
Amina
Bibi
*d
aYunnan Key Laboratory of Optoelectronic Information Technology, School of Physics and Electronic Information, Yunnan Normal University, Kunming 650500, China. E-mail: junaidriaz1990@gmail.com
bDepartment of Physics, College of Sciences, Princess Nourah bint Abdulrahman University, P.O. Box 84428, Riyadh 11671, Saudi Arabia
cSchool of Ecology and Environmental Science, Yunnan University, Biocontrol Engineering Research Center of Crop Diseases & Pests, Kunming 650500, Yunnan Province, China
dDepartment of Physics, Hazara University, Mansehra 21300, Pakistan. E-mail: aminaamni11@gmail.com
First published on 17th February 2026
Abstract
Over the last decade, researchers have required electrode materials that have higher energy density and are highly stable to make the next generation of supercapacitors. This study developed and analyzed a FeS–Fe3O4 nanocomposite as a potential positive electrode material for asymmetric supercapacitors. Structural and morphological analyses were performed using XRD and FESEM, with EDX and EDS mapping. The FeS–Fe3O4 composite worked better than both pure FeS and pure Fe3O4 when tested with a 3 M KOH electrolyte. At 1 A g−1, it has a specific capacitance of 464.6 F g−1, and at 4 A g−1, it has a specific capacitance of 186.9 F g−1. The better performance is because FeS conducts electricity better, and Fe3O4 acts like a pseudocapacitor. These two factors work together to speed up the movement of ions and the redox processes. The supercapacitor fabricated in this work exhibits a unique configuration that has never been reported before. The negative electrode was made of activated carbon, and the positive electrode was made of FeS–Fe3O4. It can work within a voltage window of 1.4 V. The device had a power density of 699.6 W kg−1 and an energy density of 46.10 Wh kg−1. It also delivered a high power density of 3998.4 W kg−1, with a low energy density of 30.17 Wh kg−1. After 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 charge–discharge cycles, the device retained 98.6% of its capacitance and about 99.8% coulombic efficiency, which indicates good cycling stability. The results show that the FeS–Fe3O4 nanocomposite is a very good electrode material for energy storage systems that need to be stable and work well.
000 charge–discharge cycles, the device retained 98.6% of its capacitance and about 99.8% coulombic efficiency, which indicates good cycling stability. The results show that the FeS–Fe3O4 nanocomposite is a very good electrode material for energy storage systems that need to be stable and work well.
1. Introduction
In the last few decades, industrialization and fast economic growth have caused excessive use of fossil fuels, which has played a big role in global warming.1–4 It is important to create energy technologies that are both long-lasting and affordable as society moves into the next phase in order to meet the needs of modern living and industry. The rising demands of manufacturing, mechanization and the global economy are driving an increase in energy demand.5–8 This increased demand for renewable energy sources is vital for preserving global ecosystems. Without the use of renewable energy, it will be impossible to halt the depletion of the ozone layer.9–12 Developing a clean, safe, and efficient system for energy conversion and storage is crucial. Supercapacitors show great potential in addressing the energy storage requirements of electronic devices, including hybrid and electric vehicles (EVs and HEVs). Their fast charging ability, high power density, and long cycle life make them ideal for applications that require rapid energy release. Their versatility in supplementing or replacing batteries in certain situations further enhances their value in the development of advanced energy storage technologies.13–19 Supercapacitors are superior to conventional capacitors because they can store more energy, work in a wider range of temperatures, and have a higher specific capacitance. The capacitance in supercapacitors increases due to the interaction between the electrolyte and the electrode material on nickel foam, or it can arise from the faradaic electron charge generated during redox reactions or intercalation processes. Faradaic reactions make charge transfer easier.20 A lot of researchers are interested in hybrid supercapacitors (HSCs) since they perform very well when electric double-layer capacitors (EDLCs) and pseudo-capacitors (PCs) are coupled. The electrochemical characteristics and capacitance are enhanced when faradaic redox processes are combined with double layers.21–23 The type of material used for the electrode has a huge effect on how well HSCs perform, similar to batteries. This is because the energy density, power density, and cycle stability are affected.24–26 Electric double-layer capacitors (EDLCs) store charge through ion adsorption at the electrode–electrolyte interface, typically using carbon-based materials, offering high power density and long-term cycling stability. In contrast, pseudocapacitors (PCs) store energy via fast faradaic redox reactions in electroactive materials such as metal oxides or conducting polymers, providing higher capacitance. Hybrid supercapacitors combine EDLC and pseudocapacitive electrodes to achieve improved energy density and rate capability.27–29 Because of these qualities, metal sulfides and other metal compounds, such as metal oxides, are thought to be some of the best materials for electrodes in the next generation of supercapacitors.30Transition metal oxides (TMOs), such as ZnO, Fe2O3, NiO, CoAl2O4 and Fe3O4, have been widely researched as effective electrode materials for supercapacitors, offering higher capacitance than conventional electric double-layer capacitors (EDLCs).31–35 These TMOs utilize redox reactions to provide superior specific energy compared to traditional carbon-based materials, and they also show greater durability than conducting polymers.36–38 Iron oxides are well known for their excellent electrochemical redox characteristics, high capacity, environmental friendliness, abundance, non-toxicity, and low cost.39 Iron oxides can store charge across a wide voltage range because Fe3+ and Fe2+ can undergo reversible oxidation and reduction. However, iron oxides, specifically Fe3O4, have relatively low conductivity and stability, which limits their electrochemical performance and cycling stability.40
Magnetite (Fe3O4) is one of the transition metal oxides that has been found to have excellent conductivity and high electrochemical activity. This substance is appealing since it is abundant in nature, safe for people and the environment, and has little effect on the environment.41 Wang et al. examined the electrochemical performance of Fe3O4 nanoparticles exhibiting a high surface area (165.05 m2 g−1) produced via a hydrothermal process.42 Reyes et al. created Fe3O4 nanoparticles that have a specific capacitance of 36 F g−1.43 The specific capacitance of Fe3O4 is still lower than that of other transition metal oxides, though. Iron oxides are particularly important among transition metal oxides because of their environmental sustainability and abundant natural reservoirs, along with intrinsic pseudo-capacity properties, making them an outstanding choice for faradaic pseudo-capacitors. Insufficient electrical conductivity and particle agglomeration during charge/discharge cycles, on the other hand, limit Fe3O4, causing a sharp drop in capacity at elevated current densities and following extended cycling.44–48 Chang et al.49 prepared Fe3O4/graphene nanocomposites through a solvothermal process and obtained a specific capacitance of 300 F g−1 at 0.4 A g−1. Regardless of such improvements, there are still limitations to the practical application of Fe3O4-based materials including short cycle life, rate capability, and low electrical conductivity that impede their large-scale utilization.50 Iron sulfide (FeS) meets all the important requirements for supercapacitor electrode materials, such as being cheap, plentiful, and non-toxic. In the last few decades, many people have worked on making FeS nanosheets so that they can take advantage of these benefits. It is very important to make new electrode materials like FeS since they are in great demand and cheap, and have a large surface area. These factors are important for making supercapacitor technology more commercially viable.51
This study focuses on the development of hybrid electrode materials that effectively combine high electrical conductivity with significant redox activity, suggesting a promising yet complex strategy to improve supercapacitor performance. TMS and TMO have attracted significant interest due to their synergistic electrochemical capabilities; yet, their specific restrictions often hinder practical applications. To address these challenges, this study demonstrates the systematic fabrication of a FeS–Fe3O4 nanocomposite, in which the synergistic interaction between conductive FeS and pseudocapacitive Fe3O4 markedly enhances charge storage performance. The FeS–Fe3O4 nanocomposite has a high specific capacitance of 464.6 F g−1 at 1 A g−1 and preserves its outstanding rate capability because its porous structure is well-integrated. When used to create an asymmetric supercapacitor using activated carbon as the negative electrode, the device operates over a broad voltage range of 1.4 V. It has a high energy density of 46.10 Wh kg−1 and can retain 98.6% of its capacitance after 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 charge–discharge cycles, which indicates good cycling stability. These results demonstrate that FeS–Fe3O4 nanocomposites could be highly beneficial as electrode materials and represent an excellent approach to develop energy storage devices that perform well and have long-term stability.
000 charge–discharge cycles, which indicates good cycling stability. These results demonstrate that FeS–Fe3O4 nanocomposites could be highly beneficial as electrode materials and represent an excellent approach to develop energy storage devices that perform well and have long-term stability.
2. Experimental section
2.1 Materials for experiments
All materials were purchased from Sigma-Aldrich (China) and used in the experiments without further treatment or purification: ferrous sulfate heptahydrate (FeSO4·7H2O), iron(III) chloride hexahydrate (FeCl3·6H2O), thioacetamide (C2H5NS), ammonia solution (NH3·H2O), sodium hydroxide (NaOH), ethanol (C2H5OH), hydrochloric acid (HCl), N-methyl-2-pyrrolidone (NMP), polyvinylidene (PVDF), acetone and DI water.2.2 Hydrothermal synthesis of Fe3O4
To prepare Fe3O4 by using the hydrothermal method, 0.1 mmol solution of ferrous sulfate (FeSO4·7H2O) was added to 40 ml DI water. The mixture was placed on a hotplate stirrer for 3 h at room temperature. The pH of the solution was maintained at 10 by adding ammonia solution dropwise into the solution during heating at 80 °C for 40 min, due to which the color of the solution changed from bright green to black. Then the solution was transferred into an autoclave and put into a furnace at 80 °C for 4 h. After cooling naturally to room temperature, the particles were washed by centrifugation at 4000 rpm until their pH reached 7. After washing the sample many times, the sample was put into an oven for drying at 60 °C overnight to remove the water residue.2.3 Hydrothermal synthesis of FeS
For the preparation of FeS, 6.8 g of C2H5NS and 5.8 g of FeCl3·6H2O were dissolved under magnetic stirring in 15 ml of DI water and 5 ml of ethanol. After 30 minutes of magnetic stirring at room temperature, the beaker was subjected to ultrasonic treatment for the next 40 minutes for homogenous mixing. Then the prepared solution was transferred to a 30 ml stainless autoclave and heat-treated for the next 2 h at 150 °C. The thermal treatment in the autoclave facilitates good crystallinity and the desired morphology. After cooling to room temperature in an open atmosphere, the sample was washed by centrifuging many times and then put into an oven at 70 °C for the next 4 h.2.4 Preparation of FeS–Fe3O4 composites
For the preparation of FeS–Fe3O4 composites via wet chemical method, we take the weight ratio of our prepared samples, FeS, and Fe3O4, and dissolved them in 20 ml of DI water by adding sodium hydroxide using a magnetic stirrer, at 60 °C for 2 h. After that, we provide the ultrasonic treatment for 40 minutes to disperse our materials. And then put into the oven for drying at 80 °C overnight, and get the FeS–Fe3O4 composite with enhanced electrochemical performance, then pure FeS, and Fe3O4 materials.2.5 Electrode preparation process
For the preparation of Fe3O4, FeS, and FeS–Fe3O4 composite electrodes, first, we cut the Ni-foam into a 1 × 1 cm circular shape, washed it with acetone, 3 M HCl, 100% ethanol, and DI water several times, and dried it by putting it into an oven and measured its mass. Then we made slurry by using 80% active material (Fe3O4, FeS, or FeS–Fe3O4 composite), 10% activated carbon (AC), and 10% polyvinylidene (PVDF) by adding N-methyl-2-pyrrolidone (NMP) drop-wise. Then, this slurry was coated onto the Ni-foam substrate and dried in an oven for 12 h at 60 °C. To further enhance the conductivity of the porous Ni-foam, the prepared electrode was pressed under a pressure of 100 kPa for 2 min. The mass of the active material was determined by measuring the difference in weight of the Ni-foam substrate before and after electrode fabrication (for details see Fig. 1).2.6 Characterization
The successful synthesis of Fe3O4, FeS, and FeS–Fe3O4 composites was confirmed by X-ray diffraction (XRD) analysis using a Donghua TD-3500 diffract meter over a 2θ range of 20°–70°. The morphology of the samples was examined via Field Emission Scanning Electron Microscopy (FESEM) using a JEOL JSM-7800F instrument. The electrochemical performance, cyclic voltammetry (CV), galvanostatic charge discharge (GCD), and EIS, of the prepared electrodes was evaluated in a 3 M KOH aqueous electrolyte using a Donghua DH7000C workstation. A platinum wire and an Ag/AgCl electrode were employed as the counter and reference electrodes, respectively. After that an asymmetric supercapacitor were fabricated and its electrochemical performance was evaluated using FeS–Fe3O4 as the positive electrode and activated carbon (AC) as the negative electrode in 3 M KOH electrolyte and its stability was tested at 1.4 V over 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 charge–discharge cycles. Furthermore all other electrochemical parameters were calculated by using these equations.
000 charge–discharge cycles. Furthermore all other electrochemical parameters were calculated by using these equations.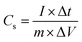 | (1) |
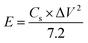 | (2) |
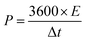 | (3) |
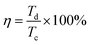 | (4) |
3. Results and discussion
3.1 Elemental analysis of prepared samples
The diffraction peaks of Fe3O4 matched with JCPDS # 01-074-33 showing peaks at positions 30°, 35°, 44°, 57°, and 64° corresponding to the respective hkl values (2 0 0), (3 1 1), (4 0 0), (5 1 1), and (4 4 0). The diffraction peaks of FeS matched with JCPDS # 75-2165, corresponding to the positions 29.9°, 34.7°, 43.2°, and 53.1°, having hkl values (1 1 0), (1 1 2), (1 0 5), and (3 0 0). The FeS–Fe3O4 composite shows all the distinct peaks of FeS, and Fe3O4, confirming the successful preparation of the composite material (Fig. 1a). Fig. 2b shows the BET surface area analysis and pore size distribution of the FeS–Fe3O4 nanocomposite. The adsorption–desorption isotherms show type IV behavior, which is typical of mesoporous materials. As the relative pressure approaches 1, the volume absorbed increases a lot. The BET surface area of the composite is predicted to be 34.21 m2 g−1, indicating a moderate surface area suitable for energy storage applications. The inset in Fig. 2b shows the pore size distribution, which is highest around 3–5 nm. This shows that the composite material has mesopores. These mesopores are useful for boosting the electrochemical performance of the FeS–Fe3O4 nanocomposite, as they provide additional surface area for charge storage. Nitrogen adsorption–desorption measurements reveal that both Fe3O4 and FeS exhibit typical type-IV isotherms with pronounced hysteresis loops, indicating their mesoporous nature. The BET surface areas of Fe3O4 (24.3 m2 g−1) and FeS (28.54 m2 g−1) confirm moderately porous structures that facilitate electrolyte penetration and ion transport; further details are provided in the SI (Fig. S3a and b). | ||
| Fig. 2 (a) X-ray diffraction of Fe3O4, FeS, and FeS–Fe3O4 composite. (b) BET analysis of the FeS–Fe3O4 composite. | ||
Fig. 3(a and a1) shows the morphology of FeS, which exhibits distributed nanoparticles with rough surfaces; the particles show agglomeration with sharp edges, and the agglomerates show strong inter-particle interactions with limited porosity within the FeS structure. Fig. 3(b and b1) display the SEM images of Fe3O4, revealing a relatively uniform morphology composed of near-spherical nanoparticles. These particles are closely packed and interconnected, forming chain-like and clustered assemblies. Compared to FeS, Fe3O4 exhibits a more homogeneous particle size distribution and smoother surfaces, indicating controlled nucleation and growth during synthesis. The interconnected nanoparticle network is beneficial for facilitating electron transport and structural stability. Fig. 3(c and c1) shows the morphology of the FeS–Fe3O4 composite, where a distinct hybrid structure can be clearly observed. The composite displays a hierarchical and porous architecture formed by the intimate integration of FeS and Fe3O4 particles. The Fe3O4 nanoparticles are uniformly anchored onto the FeS matrix, resulting in a rough and highly textured surface with abundant interparticle voids. This interconnected and porous morphology provides an increased surface area and more accessible active sites, which can effectively enhance electrolyte penetration and ion diffusion. The strong interfacial contact between FeS and Fe3O4 is expected to improve charge transfer kinetics and structural integrity during electrochemical cycling. So, the FESEM analysis confirms the successful formation of the FeS–Fe3O4 composite with a well-integrated hierarchical morphology, which is advantageous for electrochemical energy storage applications due to its enhanced surface area, improved conductivity pathways, and robust structural framework.
 | ||
| Fig. 3 Field emission scanning electron microscopy of FeS (a and a1), Fe3O4 (b and b1), and FeS–Fe3O4 composite (c and c1). | ||
Fig. 4a depicts the elemental representation of the FeS–Fe3O4 composite, with unique colors denoting all pertinent components, signifying the successful synthesis of the composite. The results are shown in dark red (Fe), purple (S), and blood red (O), showing the uniform distribution of the FeS–Fe3O4 composite. The last figure shows the EDX analysis of the FeS–Fe3O4 composite, confirming the successful formation of the FeS–Fe3O4 composite without any other additional peaks.
4. Electrochemical performance of FeS, Fe3O4, and FeS–Fe3O4 composite in a 3E system
Fig. 5a depicts a comparative cyclic voltammetry (CV) analysis of FeS, Fe3O4, and the FeS–Fe3O4 nanocomposite obtained at a scan rate of 20 mV s−1. Fig. 5(b–d) further demonstrate the CV curves of Fe3O4, FeS, and the FeS–Fe3O4 nanocomposite, respectively, obtained at various scan rates ranging from 10 to 50 mV s−1 within a potential window of 0.0–0.8 V. The FeS–Fe3O4 nanocomposite has a CV profile that is almost rectangular even at a high scan rate 50 mV s−1. This shows that it has excellent capacitive performance and fast charge transfer kinetics. In contrast, the CV curves of pure FeS and Fe3O4 electrodes exhibit strong redox peaks and obvious distortions, indicating sluggish reaction kinetics and poor electrochemical reversibility during the redox process. The composite electrode FeS–Fe3O4 shows a large area under the curve, which means its conductivity is better than that of both pure Fe3O4 and FeS electrodes. The reason for the large area under the curve is that FeS is more conductive than Fe3O4, which provides more electrons during the CV test Fig. 5a. The results demonstrate that both pure FeS and Fe3O4 electrodes have different redox peaks centered around 0.4 V, which are associated with the reversible Fe2+/Fe3+ redox processes.47 The fact that these acute redox patterns are less pronounced in the composite further supports its better capacitive properties and faster electrochemical responsiveness. The following reactions are involved in the 3 M KOH solution during the CV test.| FeS + 10OH− → Fe(OH)2 + SO4−2 + 4H2O + 8e− | (5) |
| FeS + 8OH− → Fe(OH)2 + SO3−2 + 3H2O + 6e− | (6) |
| Fe3O4 + H2O + (OH)− → 3FeO(OH)|4K+ (Oxidation) | (7) |
| 3FeO(OH) → Fe3O4 + H2O|4(OH)− + 4K+ → 4KOH (Reduction) | (8) |
 | ||
| Fig. 5 (a) Comparative CV analysis of Fe3O4, FeS, and FeS–Fe3O4 composite at 20 mV s−1, and CV analysis of (b) Fe3O4, (c) FeS, and (d) FeS–Fe3O4 nanocomposite at scan rates of 10 to 50 mV s−1. | ||
The Fe3O4 phase also plays a big role in charge storage by using iron oxy-hydroxide species in reversible oxidation–reduction processes. Fe3O4 oxidizes in the presence of water and hydroxide ions, as shown in eqn (7), forming FeO (OH) and releasing electrons. This process involves the oxidation of Fe2+ to Fe3+, which is a very important part of the faradaic charge-storage mechanism. During the cathodic scan, the reverse reaction happens, as given in eqn (8), where FeO (OH) is reduced back to Fe3O4 with the consumption of electrons and hydroxide ions, regenerating KOH in the electrolyte. This reversible transition between Fe3O4 and FeO (OH) ensures good redox reversibility and cycling stability.
The capacitive and diffusion-controlled charge storage contributions of the Fe3O4, FeS, and FeS–Fe3O4 composite electrodes were quantitatively examined at varied scan rates ranging from 10–50 mV s−1, as reported in Table S1. For the Fe3O4 electrode, the charge storage behavior is largely dominated by diffusion-controlled mechanisms at all scan rates. At a low scan rate 10 mV s−1, the diffusion contribution reaches 87%, while the capacitive contribution accounts for just 13%. With increasing scan rate, the capacitive contribution gradually improves to 39% at 50 mV s−1, indicating enhanced surface-controlled kinetics; nonetheless, diffusion-controlled behavior remains dominant, revealing the bulk redox nature of Fe3O4. In the case of the FeS electrode, a moderate increase in capacitive contribution is found compared to Fe3O4. At a scan rate of 10 mV s−1, the capacitive contribution is 22%, increasing steadily to 48% at 50 mV s−1, whereas the diffusion contribution declines from 78% to 52%. This pattern shows that FeS exhibits mixed charge storage behavior, where both surface redox reactions and diffusion-limited processes contribute significantly, perhaps because to its increased electrical conductivity relative to Fe3O4. Notably, the FeS–Fe3O4 nanocomposite electrode displays a much larger capacitive contribution across all scan rates. At 10 mV s−1, the capacitive contribution already reaches 34%, which further increases to 66% at 50 mV s−1, while the diffusion contribution similarly reduces from 66% to 34%. The relationship was utilized to precisely quantify the contributions of diffusion-controlled and capacitive processes.
| i(V) = K1v + K2v1/2 | (9) |
This equation indicates that the surface capacitive effects are represented by K1v, whereas the diffusion-controlled insertion process is denoted by K2v1/2. Rearranging the sequence of the equation:
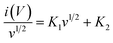 | (10) |
This dramatic shift toward capacitive-controlled behavior underlines the synergistic interaction between FeS and Fe3O4. The greater capacitive response can be due to enhanced electrical conductivity, increased electrochemically active surface area, and quicker ion transport at the hetero interface between the two phases. Overall, the FeS–Fe3O4 composite demonstrates a dominant surface-controlled charge storage mechanism at higher scan rates, which is particularly desirable for high-rate energy storage applications. The enhanced capacitive contribution verifies faster reaction kinetics and improved reversibility, explaining the superior electrochemical performance of the composite compared to the individual FeS and Fe3O4 electrodes (Fig. 6).
Fig. 7 shows the electrochemical impedance parameters found for the Fe3O4, FeS, and FeS–Fe3O4 composite electrodes. The three electrodes have nearly equal solution resistance (Rs) values. This means that the electrolyte resistance is about the same and that the electrode materials and the current collector have good electrical contact. The Rs value for the Fe3O4 electrode is 0.49 Ω, while the Rs values for FeS and the FeS–Fe3O4 composite are 0.50 Ω and 0.56 Ω, respectively. There is a considerable difference in the charge transfer resistance (Rct) between the electrodes. The Rct value of the Fe3O4 electrode is 2.34 Ω, which means that charge transfer at the point where the electrode and electrolyte come in contact is slow. The FeS electrode has a much lower Rct of 0.54 Ω, indicating higher conductivity and faster electron transport between surfaces. The Rct value of the FeS–Fe3O4 composite electrode is 0.55 Ω, which is much lower than that of pure Fe3O4 and similar to that of FeS. FeS serves as an efficient electronic pathway, ensuring rapid electron transport across the electrode, while also providing structural support that mitigates mechanical stress and suppresses agglomeration of Fe3O4 during cycling. Most importantly, the FeS–Fe3O4 composite induces interfacial polarization and electronic redistribution, creating abundant electrochemically active sites and accelerating interfacial redox reactions.52 The impedance results reveal that the creation of the FeS–Fe3O4 composite significantly boosts interfacial charge transfer while preserving low solution resistance, which leads to the higher electrochemical performance, observed for the composite electrode (Table 1).
| Electrodes | R s (Ω) | R ct (Ω) |
|---|---|---|
| Fe3O4 | 0.49 | 2.34 |
| FeS | 0.50 | 0.54 |
| FeS–Fe3O4 composite | 0.56 | 0.55 |
Fig. 8 shows the charge–discharge behavior of Fe3O4, FeS, and the FeS–Fe3O4 composite electrodes in a 3 M KOH electrolyte. At a current density of 2 A g−1 and in a potential window of (0.0–0.8) V, Fig. 8a compares the GCD curves of the three electrodes. The FeS–Fe3O4 composite has a substantially longer discharge time than the other electrodes, which means that it can store more charge than pure Fe3O4 and FeS. This enhanced performance is due to the synergistic interaction between FeS and Fe3O4, which facilitates charge transfer and ion diffusion. Fig. 8(b–d) show the GCD curves for Fe3O4, FeS, and the FeS–Fe3O4 composite at current densities of 1, 2, 3, and 4 A g−1. The charge and discharge curves of the Fe3O4 electrode (Fig. 8b) are comparable to each other. But when the current density gets higher, the discharge time gets shorter, which indicates that the rate capability is limited. The FeS electrode in Fig. 8c shows a similar pattern. When the current density is higher, the discharge time is shorter, leading to higher polarization. This illustrates that electrochemical kinetics are limited. The GCD curves for the FeS–Fe3O4 composite electrode (Fig. 8d) are clear and highly symmetrical, even at high current densities. This means that it can be readily reversed and operates better at higher rates. The lower voltage drop and longer discharge time indicate that the composite structure has better electrical conductivity and faster ion transport. Fig. 8e summarizes the specific capacitance values of all electrodes calculated from the GCD curves at different current densities; for details, see Table 2. At 1 A g−1, the FeS–Fe3O4 composite delivers a high specific capacitance of 464.6 F g−1, which is substantially higher than that of Fe3O4 (88.13 F g−1) and FeS (94.5 F g−1). Even at higher current densities of 2, 3, and 4 A g−1, the composite retains capacitance values of 449.5, 307.1, and 186.9 F g−1, respectively, demonstrating superior rate capability. In comparison, both Fe3O4 and FeS electrodes suffer from a more pronounced decline in capacitance with increasing current density. The outstanding electrochemical performance of the FeS–Fe3O4 composite can be attributed to the combined effects of enhanced conductivity from FeS and the high pseudocapacitive contribution of Fe3O4, along with improved electrolyte accessibility and structural stability.
| Current density A g−1 | Fe3O4 F g−1 | FeS F g−1 | FeS–Fe3O4 F g−1 |
|---|---|---|---|
| 1 | 88.13 | 94.5 | 464.6 |
| 2 | 81.43 | 88.3 | 449.5 |
| 3 | 72.10 | 65.5 | 307.1 |
| 4 | 44.65 | 32.1 | 186.9 |
5. Electrochemical performance of the asymmetric FeS–Fe3O4//AC supercapacitor
Fig. 9 presents the electrochemical performance of the assembled asymmetric supercapacitor device based on the FeS–Fe3O4 composite as the positive electrode and activated carbon (AC) as the negative electrode, evaluated in a 3 M KOH electrolyte. Fig. 9a shows the CV window of both AC (−0.6 to 0.0) V and FeS–Fe3O4 composite (0.0 to 0.8) V at a scan rate of 20 mV s−1 in 3 M KOH solution. The CV shape of AC is rectangular, showing its EDLC behaviour, while the FeS–Fe3O4 composite shows pseudocapacitive behaviour with a broader response due to reversible redox reactions. The suitable window for both AC and FeS–Fe3O4 composite confirms the construction of the asymmetric device for practical applications. Fig. 9b shows the CV analysis of the FeS–Fe3O4//AC device at various scan rates ranging from 10–100 mV s−1 in a potential window of 0.0 to 1.4 V. As the scan rate increases from 10–100 mV s−1, the area under the curve also increases, which shows that the electrochemical performance can be reversed easily and the transportation of charge happens quickly. The shape of the CV remains the same at high scan rates, which shows the good stability of the FeS–Fe3O4//AC device in 3 M KOH solution. Fig. 9c shows the galvanostatic charge–discharge curve at a current density of 1–5 A g−1. The CD curve remains the same even at a very small voltage drop. This shows that the device has high coulombic efficiency and excellent reversibility over repeated cycles. Fig. 9d shows the particular capacitance values that were found from the GCD curves at various current densities. At 1 A g−1, the device has a high specific capacitance of 169.29 F g−1, which slowly drops to 110.85 F g−1 at 5 A g−1 (for details see Table 3). This shows that it can operate effectively at high current densities. As the current density goes up, the energy density goes down from 46.10 to 30.17 Wh kg−1, while the power density goes up from 699.6 to 3998.4 W kg−1. This shows that the energy–power performance is very well balanced. Fig. 9e shows the coulombic efficiency of the FeS–Fe3O4//AC device at different current densities. The device has a high coulombic efficiency of about 102% at low current densities and it remains above 95% even at higher current densities. This means that it can charge and discharge quickly and doesn't lose much energy while it works. Fig. 9f shows the EIS analysis performed for the FeS–Fe3O4//AC device. The Nyquist plot shows that Rs is 1.27 ohms, which shows that the 3 M KOH electrolyte has strong ionic conductivity with low internal resistance. The low charge transfer resistance (Rct) of 3.96 ohm shows fast electron transport inside the device. So, from all the above results, it is clear that the FeS–Fe3O4//AC device is a promising device for energy storage applications.| Current density (A g−1) | Specific capacitance (F g−1) | Energy density (Wh kg−1) | Power density (W kg−1) | Coulombic efficiency (%) |
|---|---|---|---|---|
| 1 | 169.29 | 46.10 | 699.6 | 102 |
| 2 | 160.50 | 43.62 | 1401.1 | 102 |
| 3 | 145.71 | 39.59 | 2098.4 | 95.3 |
| 4 | 134.28 | 36.45 | 2801.2 | 95.3 |
| 5 | 110.85 | 30.17 | 3998.4 | 95.2 |
Fig. 10 shows the cycling stability, energy density vs. power density, and coulombic efficiency of the prepared asymmetric device (FeS–Fe3O4//AC). Fig. 10a shows the cycling stability of the asymmetric device over 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 continuous charge–discharge cycles at a current density of 7 A g−1. The device retains 98.6% of its initial capacitance after prolonged cycling, demonstrating its excellent electrochemical durability and structural stability. The high retention confirms that the FeS–Fe3O4 nanocomposite and AC maintain strong interfacial integrity during the repeated redox reactions. Fig. 10b shows the energy density and power density of the asymmetric device at a current density of 1–5 A g−1. The device attains a highest energy density of 46.10 Wh kg−1, with a power density of 699.6 W kg−1. At high current density, the energy density reaches a value of 30.17 Wh kg−1, and power density reaches 3998.4 W kg−1, highlighting its excellent rate capability. This favorable balance between energy and power densities reflects the synergistic contribution of the pseudocapacitive FeS–Fe3O4 composite and the electric double-layer behavior of the AC electrode, making the device suitable for high-power energy storage applications. Fig. 10c shows how well the FeS–Fe3O4//AC device works after 12
000 continuous charge–discharge cycles at a current density of 7 A g−1. The device retains 98.6% of its initial capacitance after prolonged cycling, demonstrating its excellent electrochemical durability and structural stability. The high retention confirms that the FeS–Fe3O4 nanocomposite and AC maintain strong interfacial integrity during the repeated redox reactions. Fig. 10b shows the energy density and power density of the asymmetric device at a current density of 1–5 A g−1. The device attains a highest energy density of 46.10 Wh kg−1, with a power density of 699.6 W kg−1. At high current density, the energy density reaches a value of 30.17 Wh kg−1, and power density reaches 3998.4 W kg−1, highlighting its excellent rate capability. This favorable balance between energy and power densities reflects the synergistic contribution of the pseudocapacitive FeS–Fe3O4 composite and the electric double-layer behavior of the AC electrode, making the device suitable for high-power energy storage applications. Fig. 10c shows how well the FeS–Fe3O4//AC device works after 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 charge–discharge cycles. During the entire cycling test, the coulombic efficiency remains rather steady at about 99.8%. This indicates that electrochemical reactions are highly reversible and involve very little energy (more details can be seen in Table S2). The asymmetric device's extraordinary efficiency further confirms its superb charge transfer kinetics and long-term stability. These findings demonstrate the FeS–Fe3O4//AC asymmetric supercapacitors' energy–power characteristics, high efficiency, and great cycle stability. This shows that it has a lot of potential for use in the future as a way to store energy in a way that doesn't harm the environment. Fig. 10d displays the XRD analysis of the FeS–Fe3O4 composite before and after the stability test. The peaks of the FeS–Fe3O4 composite matched with the characteristic planes of FeS and Fe3O4, which confirms the successful formation of the composite. Two peaks are also observed, which belong to Ni-foam. After the stability test, the intensity reduced slightly, but all the peaks confirm the good crystallinity of the FeS–Fe3O4 composite. Fig. 10(e and f) shows the SEM image of the FeS–Fe3O4 composite on Ni-foam before and after the stability test at 10 µm. The electrode before the stability test shows a comparatively smooth surface, and after the stability test, it shows a well distributed active material that provides sufficient active sites for efficient ion transfer in 3 M KOH electrolyte. The FESEM image after the stability test shows that the overall morphology of the FeS–Fe3O4 composite is well preserved. Although minor surface roughening and agglomeration of particles are observed, no structural collapse occurs. The maintained morphology after 12 K cycles further confirms the mechanical robustness and electrochemical performance of the FeS–Fe3O4 composite electrode.
000 charge–discharge cycles. During the entire cycling test, the coulombic efficiency remains rather steady at about 99.8%. This indicates that electrochemical reactions are highly reversible and involve very little energy (more details can be seen in Table S2). The asymmetric device's extraordinary efficiency further confirms its superb charge transfer kinetics and long-term stability. These findings demonstrate the FeS–Fe3O4//AC asymmetric supercapacitors' energy–power characteristics, high efficiency, and great cycle stability. This shows that it has a lot of potential for use in the future as a way to store energy in a way that doesn't harm the environment. Fig. 10d displays the XRD analysis of the FeS–Fe3O4 composite before and after the stability test. The peaks of the FeS–Fe3O4 composite matched with the characteristic planes of FeS and Fe3O4, which confirms the successful formation of the composite. Two peaks are also observed, which belong to Ni-foam. After the stability test, the intensity reduced slightly, but all the peaks confirm the good crystallinity of the FeS–Fe3O4 composite. Fig. 10(e and f) shows the SEM image of the FeS–Fe3O4 composite on Ni-foam before and after the stability test at 10 µm. The electrode before the stability test shows a comparatively smooth surface, and after the stability test, it shows a well distributed active material that provides sufficient active sites for efficient ion transfer in 3 M KOH electrolyte. The FESEM image after the stability test shows that the overall morphology of the FeS–Fe3O4 composite is well preserved. Although minor surface roughening and agglomeration of particles are observed, no structural collapse occurs. The maintained morphology after 12 K cycles further confirms the mechanical robustness and electrochemical performance of the FeS–Fe3O4 composite electrode.
6. Conclusion
In summary, a FeS–Fe3O4 nanocomposite was successfully synthesized and systematically investigated as an efficient electrode material for advanced supercapacitor applications. Structural and morphological characterization confirmed the formation of a well-integrated heterostructure composed of crystalline FeS and Fe3O4 phases, providing abundant electroactive sites and a favourable architecture for rapid ion transport. Electrochemical evaluations demonstrated that the FeS–Fe3O4 composite delivers significantly enhanced capacitive performance compared to the individual FeS and Fe3O4 electrodes, achieving a high specific capacitance of 464.6 F g−1 at 1 A g−1 and maintaining good rate capability at higher current densities. The improved electrochemical behaviour is primarily attributed to the synergistic interaction between the high electrical conductivity of FeS and the strong pseudocapacitive contribution of Fe3O4, which together facilitate fast charge transfer and efficient redox reactions. Furthermore, an asymmetric supercapacitor device assembled using the FeS–Fe3O4 nanocomposite as the positive electrode and activated carbon as the negative electrode operated stably over an extended voltage window of 1.4 V in 3 M KOH electrolyte. The device exhibited a high energy density of 46.10 Wh kg−1 at a power density of 699.6 W kg−1 and retained 30.17 Wh kg−1 at a high power density of 3998.4 W kg−1. Remarkably, the device maintained 98.6% capacitance retention and a coulombic efficiency of approximately 99.8% after 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 charge–discharge cycles, highlighting its excellent long-term stability and reversibility. Overall, this study demonstrates that the FeS–Fe3O4 nanocomposite is a promising electrode material for high-performance, durable, and practical energy storage systems.
000 charge–discharge cycles, highlighting its excellent long-term stability and reversibility. Overall, this study demonstrates that the FeS–Fe3O4 nanocomposite is a promising electrode material for high-performance, durable, and practical energy storage systems.
Author contributions
Junaid Riaz: wrote the original manuscript draft, data creation, characterization, and electrochemical analysis. Muhammad Arif: supervision, review and editing, formal analysis. Ghulam Murtaza: software analysis. Zahra Bayhan: review and editing, Amina Bibi: supervision, review and editing, characterization, and formal analysis.Conflicts of interest
The authors declare that they have no conflict of interest.Data availability
The data that have been used are confidential & will be available on request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5na01165e.
Acknowledgements
We express our gratitude to the Researchers Supporting Project (PNURSP2026R452) at Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.References
- S. Banerjee, B. Mordina, P. Sinha and K. K. Kar, Recent advancement of supercapacitors: A current era of supercapacitor devices through the development of electrical double layer, pseudo and their hybrid supercapacitor electrodes, J. Energy Storage, 2025, 108, 115075 CrossRef CAS.
- M. Arif, J. Riaz, A. Bibi, H. Yang and T. Zhu, Enhancing supercapacitor energy density by TiN–ZnS composites unveiled as a promising electrode, APL Mater., 2024, 12(7) DOI:10.1063/5.0221353.
- F. Hu, J. Wei, X. Quan and Q. Ouyang, Rational design of defect-rich MoS2-modified V2CTx electrodes for advanced flexible supercapacitor, Chem. Eng. J., 2025, 170003 CrossRef CAS.
- M. Arif, J. Riaz, H. Yang, Z. Fu, A. Bibi and T. Zhu, Synthesis of high-performance CdS/MnO composite electrode to achieve high energy and power densities for asymmetrical supercapacitors, Mater. Des., 2025, 251, 113704 CrossRef CAS.
- J. Riaz, W. Yongyuan, J. Cao, A. Bibi, D. Muhammad, H. Eman and X. Zhou, Wet-chemical synthesized TiN-CuO nanocomposite: Advancing supercapacitor technology with high energy and power density, Phys. E, 2025, 165, 116105 CrossRef CAS.
- A. F. Kassem, N. U. Hassan, M. Jelani, E. A. M. Saleh, M. M. Moharam and R. H. Althomali, et al., Superior supercapattery performance enabled by MnS/Fe2O3 nanosheets and theoretical evaluation of contributing currents, Inorg. Chem. Commun., 2025, 176, 114171 CrossRef CAS.
- J. Riaz, Y. Zhang, J. Cao, A. Bibi, M. Arif and Z. Zhang, et al., Facile synthesis of TiN nano sheets decorated Fe2O3 nanoparticles as novel cathode material for Asymmetric Supercapacitor, Surf. Interfaces, 2024, 46, 104080 CrossRef CAS.
- A. F. Kassem, N. U. Hassan, E. A. M. Saleh, R. Hasan, M. M. Moharam and R. H. Althomali, et al., Al intercalated ZnS nanosheets as anode for supercapattery application with wide operating potential window, Inorg. Chem. Commun., 2025, 171, 113588 CrossRef CAS.
- J. Riaz, J. Cao, Y. Zhang, A. Bibi and X. Zhou, Facile synthesis and electrochemical analysis of TiN-based ZnO nanoparticles as promising cathode materials for asymmetric supercapacitors, Nanoscale Adv., 2024, 6(20), 5145–5157 RSC.
- K. M. Albalawi, M. Al-Dossari, A. M. Saeedi, R. H. Althomali, G. F. Solre, M. Sadiq and S. U. Asif, Synergistic effects of scandium doping and N-rGO integration on titanium oxide and evaluating faradic/non-faradic behavior by Dunn's model for high-performance supercapattery applications, J. Energy Storage, 2024, 104, 114576 CrossRef CAS.
- J. Riaz, J. Cao, A. Bibi, M. Arif and D. Muhammad, Hydrothermal synthesis of ball-like ZnS nanospheres decorated urchin-like W18O49 nanospheres as electrode for high power and stable hybrid supercapacitor, Mater. Lett., 2024, 370, 136853 CrossRef CAS.
- J. Riaz, Y. Zhang, J. Cao, A. Bibi, Z. Zhang and X. Zhou, High-performance electrode material synthesis via wet-chemical method: A study on NbN–Fe2O3 composite, J. Mater. Sci.: Mater. Electron., 2024, 35(17), 1176 CrossRef CAS.
- J. Riaz, J. Cao, Y. Zhang, A. Bibi, M. Arif and Z. Zhang, et al., Improved performance of TiN nano buds decorated MoS2 sheets in asymmetric supercapacitors, J. Mater. Sci.: Mater. Electron., 2024, 35(17), 1142 CrossRef CAS.
- M. M. Moharam, S. U. Asif, E. A. M. Saleh, R. H. Althomali, A. F. Kassem and M. Sadiq, Tailoring electrochemical properties of zirconium oxide doped by praseodymium coated with carbon-based materials for superior supercapacitors, Inorg. Chem. Commun., 2024, 169, 113114 CrossRef CAS.
- D. Muhammad, H. Hou, K. M. Alotaibi, N. Ali, M. M. Al-Hinaai and J. Riaz, et al., Improved Asymmetric Supercapacitors Using a Novel Synthesis of Organized FeS/SnS2 Nanostructure Effective Cathode Material, J. Electron. Mater., 2025, 54(3), 1972–1984 CrossRef CAS.
- M. Sadiq, M. U. Islam, M. M. Moharam, E. A. M. Saleh and S. U. Asif, Enhanced electrochemical performance of La2S3/N-rGO/PANI nanocomposites as an efficient electrode material for supercapacitor applications, J. Mater. Sci.: Mater. Electron., 2024, 35(15), 1011 CrossRef CAS.
- D. Muhammad, X. Liu, H. Hou, X. Yu, J. Rong and J. Riaz, et al., Novel synthesis of systematized FeS/CuO nanostructure efficacious electrode material for escalating asymmetric supercapacitors, J. Alloys Compd., 2025, 1016, 178877 CrossRef CAS.
- T. Anwar, T. Manzoor, N. Hussain, S. N. Shah, S. Perveen and S. U. Asif, et al., Antimony trisulfide with graphene oxide coated titania nanotube arrays as anode material for lithium-ion batteries, J. Inorg. Organomet. Polym. Mater., 2024, 34(10), 4780–4787 CrossRef CAS.
- J. Riaz, J. Cao, N. Hakimi, A. Sikandar, F. Aslam, T. Huma and A. Bibi, Synthesis of high-performance supercapacitor electrode materials by wet-chemical route based on TiN–Al2O3 composite, J. Mater. Sci.: Mater. Electron., 2025, 36(35), 2257 CrossRef CAS.
- J. Riaz, F. Aslam, M. Arif, T. Huma and A. Bibi, First investigation of high-performance FeS-based W18O49 asymmetric supercapacitors operating at 1.6 V, Nanoscale Adv., 2025, 7(1), 231–241 RSC.
- Y. Tan, H. Lin, Z. Chen, L. Niu and H. Li, Regulating the coordination microenvironment of atomic bismuth sites in nitrogen-rich carbon nanosheets as anode for superior potassium-ion batteries, J. Energy Chem., 2024, 99, 365–374 CrossRef CAS.
- R. Wang, S. Luo, C. Xiao, Z. Chen, H. Li and M. Asif, et al., MXene-carbon nanotubes layer-by-layer assembly based on-chip micro-supercapacitor with improved capacitive performance, Electrochim. Acta, 2021, 386, 138420 CrossRef CAS.
- W. Zhu and D. Shen, Synthesis of Bi2O3/hierarchical porous carbon composites for supercapacitor application, J. Energy Storage, 2024, 79, 110118 CrossRef CAS.
- J. Zhao, L. Yang, R. Li and Y. Zhou, One-Step Synthesis of Fe-Based Metal–Organic Framework (MOF) Nanosheet Array as Efficient Cathode for Hybrid Supercapacitors, Inorganics, 2023, 11(4), 169 CrossRef CAS.
- Z. Chen, H. Lin, Y. Tan, L. Niu and H. Li, Anchoring Multi-Coordinated Bismuth Metal Atom Sites on Honeycomb-Like Carbon Rods Achieving Advanced Potassium Storage, Adv. Funct. Mater., 2024, 34(45), 2407653 Search PubMed.
- G. Qin, Y. Zhang, Z. Qi and X. He, Dynamically Reversible Gelation of Electrolyte for Efficient Wide-Temperature Adaptable Energy Storage, Adv. Funct. Mater., 2024, 34(28), 2316813 Search PubMed.
- M. Sethi, H. Bantawal, U. S. Shenoy and D. K. Bhat, Eco-friendly synthesis of porous graphene and its utilization as high performance supercapacitor electrode material, J. Alloys Compd., 2019, 799, 256–266 Search PubMed.
- M. Sethi, U. S. Shenoy, S. Muthu and D. K. Bhat, Facile solvothermal synthesis of NiFe2O4 nanoparticles for high-performance supercapacitor applications, Front. Mater. Sci., 2020, 14(2), 120–132 CrossRef.
- M. Sethi, U. S. Shenoy and D. K. Bhat, Porous graphene–NiCo2O4 nanorod hybrid composite as a high performance supercapacitor electrode material, New J. Chem., 2020, 44(10), 4033–4041 RSC.
- J. Sun, C. Xu and H. Chen, A review on the synthesis of CuCo2O4-based electrode materials and their applications in supercapacitors, J. Materiomics, 2021, 7(1), 98–126 CrossRef.
- M. Iqbal, N. G. Saykar, A. Arya, I. Banerjee, P. S. Alegaonkar and S. K. Mahapatra, High-performance supercapacitor based on MoS2@ TiO2 composite for wide range temperature application, J. Alloys Compd., 2021, 883, 160705 CrossRef CAS.
- F. Ahmad, A. Shahzad, M. Danish, M. Fatima, M. Adnan and S. Atiq, et al., Recent developments in transition metal oxide-based electrode composites for supercapacitor applications, J. Energy Storage, 2024, 81, 110430 CrossRef CAS.
- E. Sohouli, H. Teymourinia, A. Ramazani and K. Adib, Preparation of high-performance supercapacitor electrode with nanocomposite of CuO/NCNO flower-like, Sci. Rep., 2023, 13(1), 16221 CrossRef CAS PubMed.
- M. Sethi, U. S. Shenoy and D. K. Bhat, Hassle-free solvothermal synthesis of NiO nanoflakes for supercapacitor application, Phys. B, 2021, 611, 412959 CrossRef CAS.
- T. P. Ramesh, U. S. Shenoy and D. K. Bhat, Exploring the potential of CoAl2O4 nanoflakes in supercapacitor applications, J. Alloys Compd. Commun., 2025, 100111 Search PubMed.
- A. Kumar, H. K. Rathore, D. Sarkar and A. Shukla, Nanoarchitectured transition metal oxides and their composites for supercapacitors, Electrochem. Sci. Adv., 2022, 2(6), e2100187 CrossRef CAS.
- M. A. Rehman, S. Park, M. F. Khan, M. F. Bhopal, G. Nazir and M. Kim, et al., Development of directly grown-graphene–silicon Schottky barrier solar cell using co-doping technique, Int. J. Energy Res., 2022, 46(8), 11510–11522 CrossRef CAS.
- M. Sethi, U. S. Shenoy and D. K. Bhat, Iron Oxide-Functionalized Graphene Nanocomposites for Supercapacitor Application, in Iron Oxide-Based Nanocomposites and Nanoenzymes: Fundamentals and Applications, Springer International Publishing, Cham, 2024, p. 77–117 Search PubMed.
- J. Xu, D. Wang, Y. Yuan, W. Wei, L. Duan and L. Wang, et al., Polypyrrole/reduced graphene oxide coated fabric electrodes for supercapacitor application, Org. Electron., 2015, 24, 153–159 CrossRef CAS.
- T. Yamashita and P. Hayes, Analysis of XPS spectra of Fe2+ and Fe3+ ions in oxide materials, Appl. Surf. Sci., 2008, 254(8), 2441–2449 CrossRef CAS.
- X. Rui, H. Tan and Q. Yan, Nanostructured metal sulfides for energy storage, Nanoscale, 2014, 6(17), 9889–9924 RSC.
- C. Wang, S. Hu, J. Wang, S. Yao, C. Wang and Y. Wang, Fe3O4 loaded on the porous N-doped carbon spheres used as a high-performance anode material for lithium-ion batteries, Ionics, 2025, 1–14 CrossRef.
- F. Reyes-Ortega, A. V. Delgado and G. R. Iglesias, Modulation of the magnetic hyperthermia response using different superparamagnetic iron oxide nanoparticle morphologies, Nanomaterials, 2021, 11(3), 627 CrossRef CAS PubMed.
- S. Pal, S. Majumder, S. Dutta, S. Banerjee, B. Satpati and S. De, Magnetic field induced electrochemical performance enhancement in reduced graphene oxide anchored Fe3O4 nanoparticle hybrid based supercapacitor, J. Phys. D Appl. Phys., 2018, 51(37), 375501 CrossRef.
- V. S. Patil, S. S. Thoravat, S. S. Kundale, T. D. Dongale, P. S. Patil and S. A. Jadhav, Synthesis and testing of polyaniline grafted functional magnetite (Fe3O4) nanoparticles and rGO based nanocomposites for supercapacitor application, Chem. Phys. Lett., 2023, 814, 140334 CrossRef CAS.
- T. Prasankumar, B. R. Wiston, C. R. Gautam, R. Ilangovan and S. P. Jose, Synthesis and enhanced electrochemical performance of PANI/Fe3O4 nanocomposite as supercapacitor electrode, J. Alloys Compd., 2018, 757, 466–475 CrossRef CAS.
- J. Li, D. Chen and Q. Wu, α-Fe2O3 Based Carbon Composite As Pure Negative Electrode For Application As Supercapacitor, Eur. J. Inorg. Chem., 2019, 2019(10), 1301–1312 CrossRef CAS.
- S. S. Raut, L. K. Bommineedi, S. Pande and B. R. Sankapal, Prototype symmetric configured MWCNTs/Fe2O3 based solid-state supercapacitor, Synth. Met., 2021, 271, 116629 CrossRef CAS.
- J. Liao, Y. Li, Z. Wang, L. Lv and L. Chang, In-situ preparation of Fe3O4/graphene nanocomposites and their electrochemical performances for supercapacitor, Mater. Chem. Phys., 2021, 258, 123995 CrossRef CAS.
- E. Payami, A. Mohammadzadeh, K. D. Safa and R. Teimuri-Mofrad, Ferrocene surface-modified Fe3O4 nanoparticles as prominent electrode material for supercapacitor application, J. Energy Storage, 2024, 88, 111624 CrossRef CAS.
- H. Song, M. Wu, X. Tang, J. Liang, Y. Zhang and Y. Xie, et al., Synthesis of Fe3O4/FeS2 composites via MOF-templated sulfurization for high-performance hybrid supercapacitors, J. Alloys Compd., 2025, 1010, 177658 CrossRef CAS.
- Z. Zhao, W. Zhang, D. Wang, L. Li, Q. Liang and W. Li, et al., Ostwald-Ripening Induced Interfacial Protection Layer Boosts 1,000,000-Cycled Hydronium-Ion Battery, Angew. Chem., Int. Ed., 2024, 63(50), e202414420 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |







