Open Access Article
Open Access ArticleFe3O4@SiO2@taurine: a magnetically recoverable green nanocatalyst for efficient synthesis of pyrano[2,3-d]pyrimidine derivatives
Charul
Paliwal
a,
Aanchal
a,
Dharmendra
a,
P. S.
Ranawat
a,
K. L.
Ameta
b and
Chetna
Ameta
 *a
*a
aDepartment of Chemistry, M. L. Sukhadia University, Udaipur, Raj-313001, India. E-mail: chetnaameta@mlsu.ac.in; chetna.ameta@yahoo.com
bDepartment of Applied Chemistry, Central University of Gujarat, Vadodara, Gujarat-391107, India
First published on 12th February 2026
Abstract
The development of recyclable and eco-friendly catalysts for heterocyclic synthesis remains a central theme in sustainable chemistry. Here, we present a magnetically recoverable Fe3O4@SiO2@taurine catalyst, prepared by sequential silica coating and taurine functionalization of Fe3O4 nanoparticles. Structural, morphological, and magnetic characteristics were established through FT-IR, XRD, TEM, SEM-EDS, TGA, BET and ZETA analyses. This catalytic system promotes a one-pot, three-component reaction of aromatic aldehydes, malononitrile, and barbituric or thiobarbituric acid in aqueous ethanol under reflux, affording pyrano[2,3-d]pyrimidine derivatives in high yields and short reaction times. Ease of magnetic recovery, operational simplicity, and stable activity over multiple cycles highlight its practical applicability. Collectively, these findings demonstrate Fe3O4@SiO2@taurine as a green, efficient, and recyclable platform for sustainable organic transformations.
1 Introduction
Catalyzed transformations, designed as efficient, green, and sustainable protocols, have been extensively explored in both academic laboratories and the chemical industry.1 A major goal in catalysis is the development of systems with high activity, complete selectivity, low energy consumption, and long lifetime.2 Performance is dictated by parameters such as size, morphology, electron distribution, surface composition, and stability.3 With the advent of nanotechnology, nanocatalysts have demonstrated remarkable properties and are increasingly integrated into industrial processes.4,5 Their role is especially important in nanochemistry and green chemistry, where the emphasis is on safe, energy-efficient reactions with minimal waste.6,7 Heterogeneous nanocatalysts, in particular, offer high activity, simple recovery, and excellent recyclability, making them a valuable option for organic synthesis.8–10 The design and synthesis of nanocatalysts through environmentally benign approaches has become a key driving force in heterogeneous catalysis and green chemistry.11 The construction of heterocyclic scaffolds through multicomponent reactions has received great attention because of high atom economy and operational simplicity.12–15Among various nanosystems, magnetic microspheres and core–shell structures have received significant attention due to their magnetic responsivity, low cytotoxicity, good stability, and surface tunability.16–19 Core–shell magnetic silica microspheres, in particular, are attractive for bioseparation, enzyme immobilization, and catalysis.20–22 Chemical co-precipitation offers a low-temperature, cost-effective route to prepare well-dispersed Fe3O4 nanoparticles with controlled size.23 Their performance can be further improved by surface coating techniques.24 Recently, core–shell nanocomposites that integrate inorganic nanoparticles with metal–organic frameworks (MOFs) have emerged as multifunctional materials. NP@MOF hybrids show wide applications across medicine, environment, and energy.25,26 Among various MOFs, Fe-based systems stand out as heterogeneous catalysts due to their high dispersibility, abundance of active sites, and structural stability arising from strong Fe3+ carboxylate interactions, combined with their low toxicity, redox activity, and cost-effectiveness.27–29 In this context, we designed a magnetically recoverable Fe3O4@SiO2@taurine nanocatalyst for the one-pot, three-component synthesis of pyrano[2,3-d]pyrimidine derivatives, as illustrated in Scheme 1.
Magnetite (Fe3O4) is a highly attractive material for catalyst design due to its superior magnetic properties, conductivity, and biocompatibility compared to other ferrite oxides.30 However, its poor stability and dispersibility necessitate protective coatings.31 Silica serves as an excellent shell, offering chemical stability, thermal resistance, and abundant –Si–OH groups for further modification, thereby improving dispersibility, oxidation resistance, and biocompatibility.32–34 Taurine (2-aminoethanesulfonic acid), a naturally occurring β-amino sulfonic acid, acts as a green bio-organic catalyst and an effective bifunctional donor–acceptor in condensation reactions (Scheme 2).35 It has been successfully employed for the synthesis of barbituric, thiobarbituric, and pyrano[2,3-d]pyrimidine derivatives under mild aqueous conditions, delivering high yields with advantages of low cost, non-toxicity, and reusability.35,36 Considering the unique features of Fe3O4, the stabilizing role of silica, and the catalytic activity of taurine, Fe3O4@SiO2@taurine was envisioned as a robust, magnetically recoverable nanocatalyst for one-pot, three-component syntheses of pyrano[2,3-d]pyrimidine derivatives under green conditions.
2 Experimental
2.1 Materials and methods
All reagents and chemicals used in the synthesis were procured from HiMedia, Sigma-Aldrich, and Alfa Aesar, and were used as received without further purification. Melting points of the synthesized compounds were determined using open capillary tubes on a Gallen Kamp apparatus and are uncorrected. The progress of reactions was monitored by thin-layer chromatography (TLC) using silica gel plates, with a mixture of ethyl acetate and n-hexane as the mobile phase. Fourier transform infrared (FT-IR) spectra were recorded using a PerkinElmer Spectrum RX-I FTIR spectrometer in ATR mode. Proton (1H) and carbon (13C) nuclear magnetic resonance (NMR) spectra were obtained using a Jeol 400 MHz spectrometer with deuterated dimethyl sulfoxide (DMSO-d6) as the solvent. Elemental compositions were determined using an ELEMENTAR Vario EL III analyzer. Powder X-ray diffraction (XRD) patterns were recorded on a Panalytical X Pert Pro diffractometer. High-resolution transmission electron microscopy (HR-TEM) images were acquired using a Tecnai G2 20 S-TWIN [FEI] operated at 200 kV. Field emission scanning electron microscopy (FE-SEM) and energy-dispersive X-ray spectroscopy (EDX) analyses were conducted on a Nova Nano FE-SEM 450 (FEI). Thermogravimetric analysis (TGA) was carried out using a EXSTAR TG/DTA 7300 at Central University of Gujarat. BET analysis was performed by using an Autosorb IQ-XR-XR-XR (3 Stat.) Viton and Zeta potential was measured by using a LITESIZER 500.2.2 Synthesis of Fe3O4@SiO2@taurine
2.3 Framework for the synthesis of pyrano[2,3-d]pyrimidine derivatives
In a round-bottom flask, 1 mmol of an aromatic aldehyde, 1 mmol of malononitrile, 0.05 g of the catalyst, and a solvent mixture of ethanol and water (15 mL each) were combined and stirred at room temperature for 15 min. Subsequently, 1 mmol of barbituric acid or 2-thiobarbituric acid was added, and the reaction mixture was refluxed for 30 min. After completion, the mixture was cooled to room temperature, and the catalyst was separated using an external magnet. The crude product was filtered, washed with water, and recrystallized from ethanol to afford the pure pyrano[2,3-d]pyrimidines, which were finally dried in an oven at 60 °C.2.3.1.1 7-Amino-2,4-dioxo-5-phenyl-1,3,4,5-tetrahydro-2H-pyrano[2,3-d]pyrimidine-6-carbonitrile (4a). Mp. (°C): 205–206; yield: 97%; IR (νmax cm−1): 674 (C–H), 1093 (C–O–C str) 1183.58 (C–NH2 str), 1445.19 (
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH str), 1587.20 (C
CH str), 1587.20 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C str Ar), 1633 (C
C str Ar), 1633 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C alkene str), 1712.28 (C
C alkene str), 1712.28 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O str), 2191.50 (C
O str), 2191.50 (C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N str), 2831.29 (C–H str), 3063.36 (
N str), 2831.29 (C–H str), 3063.36 (![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH str), 3178.72 (NH2 str) cm−1. 1H NMR (400 MHz, DMSO-d6)/δ (ppm): 4.17 (s, 1H, C–H), 7.08 (2H, NH2, D2O exchangeable), 7.14–7.25 (m, 3H, Ar–H), 7.56–7.60 (t, J = 7.1, 2H, Ar–H), 11.04 (1H, NH, D2O exchangeable). 13C NMR (125 MHz, DMSO-d6)/δ ppm: 39.3, 82.1, 113.8, 114.7, 130, 131, 131.8, 134.9, 162.
CH str), 3178.72 (NH2 str) cm−1. 1H NMR (400 MHz, DMSO-d6)/δ (ppm): 4.17 (s, 1H, C–H), 7.08 (2H, NH2, D2O exchangeable), 7.14–7.25 (m, 3H, Ar–H), 7.56–7.60 (t, J = 7.1, 2H, Ar–H), 11.04 (1H, NH, D2O exchangeable). 13C NMR (125 MHz, DMSO-d6)/δ ppm: 39.3, 82.1, 113.8, 114.7, 130, 131, 131.8, 134.9, 162.
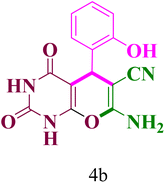
2.3.1.2 7-Amino-5-(2-hydroxyphenyl)-2,4-dioxo-1,3,4,5-tetrahydro-2-H-pyrano[2,3-d]pyrimidine-6-carbonitrile (4b). Mp. (°C):167–169; yield: 90%; IR (νmax cm−1): 1354.75 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic str), 1599.68, 1671.81 (C
C aromatic str), 1599.68, 1671.81 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O str), 2207.85 (C
O str), 2207.85 (C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N str), 2311.98 (C–H), 3043.37 (NH), 3284.21 (NH2), 3613.75 (OH), 1H NMR (400 MHz, DMSO-d6)/δ (ppm): 3.35 (s, 1H, H-5), 7.06 (d, 1H) 7.18 (2H, NH2), 7.35 (d, J = 8, 1H) 7.67 (d, J = 8, 2H, H–Ar), 8.07 (d, J = 10.6, 2H, H–Ar), 10.98 (1H, NH). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 39.4, 78.9, 103, 114.9, 117.1, 125.8, 130.3, 135.7, 153.9, 154.4, 157.2.
N str), 2311.98 (C–H), 3043.37 (NH), 3284.21 (NH2), 3613.75 (OH), 1H NMR (400 MHz, DMSO-d6)/δ (ppm): 3.35 (s, 1H, H-5), 7.06 (d, 1H) 7.18 (2H, NH2), 7.35 (d, J = 8, 1H) 7.67 (d, J = 8, 2H, H–Ar), 8.07 (d, J = 10.6, 2H, H–Ar), 10.98 (1H, NH). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 39.4, 78.9, 103, 114.9, 117.1, 125.8, 130.3, 135.7, 153.9, 154.4, 157.2.
2.3.1.3 7-Amino-5-(4-nitrophenyl)-2,4-dioxo-1,3,4,5-tetrahydro-2-H-pyrano[2,3-d]pyrimidine-6-carbonitrile (4c). Mp. (°C): 234–235; yield: 92%; IR (νmax cm−1): 821.08 (C–H), 975.02 (C–NO2 str), 1180.71 (C–O–C str), 1273.78 (C–NH2 str), 1340.95, 1516.81 (N
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O str), 1403.38, 1516.81 (C
O str), 1403.38, 1516.81 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C str Ar), 1628.48 (C
C str Ar), 1628.48 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C alkene str), 1713 (C
C alkene str), 1713 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O str), 2188.60 (C
O str), 2188.60 (C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N str), 2828.70 (C–H str), 3015.65 (
N str), 2828.70 (C–H str), 3015.65 (![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH str), 3173.26 (NH2 str). 1H NMR (400 MHz, DMSO-d6)/δ (ppm): 4.38 (s, 1H, C–H), 7.25 (2H, NH2, D2O exchangeable), 7.49 (dt, 2.5 Hz, 2H, Ar–H), 8.12 (dt, 2H, Ar–H), 11.11 (1H, NH, D2O exchangeable), 12.16 (1H, NH, D2O exchangeable). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 39.4, 58.4, 87.6, 124.5, 128.4, 146.1, 149.5, 153.4, 163.
CH str), 3173.26 (NH2 str). 1H NMR (400 MHz, DMSO-d6)/δ (ppm): 4.38 (s, 1H, C–H), 7.25 (2H, NH2, D2O exchangeable), 7.49 (dt, 2.5 Hz, 2H, Ar–H), 8.12 (dt, 2H, Ar–H), 11.11 (1H, NH, D2O exchangeable), 12.16 (1H, NH, D2O exchangeable). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 39.4, 58.4, 87.6, 124.5, 128.4, 146.1, 149.5, 153.4, 163.
2.3.1.4 7-Amino-5-(4-fluorophenyl)-2,4-dioxo-1,3,4,5- tetrahydro-2H-pyrano[2,3-d]pyrimidine-6-carbonitrile (4d). Mp. (°C): 264–265; yield: 93% IR (νmax cm−1): 837.95 (C–H), 1090.70 (C–O–C str), 1156.76 (C–F str), 1184.16 (C–NH2 str), 1506.09, 1592.94 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C str Ar), 1632.90 (C
C str Ar), 1632.90 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C alkene str), 1675.01, 1712.53 (C
C alkene str), 1675.01, 1712.53 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O str), 2190.59 (C
O str), 2190.59 (C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N str), 2828.35 (C–H str), 3000.92 (
N str), 2828.35 (C–H str), 3000.92 (![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH str), 3175.77 (NH2 str), 3382.83 (N–H str). 1H NMR (400 MHz, DMSO-d6)/δ (ppm): 4.20 (s, 1H, C–H), 6.88 (q, J = 8, 4H, Ar–H), 7.12–7.23 (m, 2H, NH2, D2O exchangeable), 11.0 (1H, NH, D2O exchangeable). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 39.4, 59.3, 89, 113.1, 114.1, 117, 128, 128, 133.8, 133.9, 140, 150, 158.1, 160.1, 162.9.
CH str), 3175.77 (NH2 str), 3382.83 (N–H str). 1H NMR (400 MHz, DMSO-d6)/δ (ppm): 4.20 (s, 1H, C–H), 6.88 (q, J = 8, 4H, Ar–H), 7.12–7.23 (m, 2H, NH2, D2O exchangeable), 11.0 (1H, NH, D2O exchangeable). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 39.4, 59.3, 89, 113.1, 114.1, 117, 128, 128, 133.8, 133.9, 140, 150, 158.1, 160.1, 162.9.
2.3.1.5 7-Amino-5-(3-bromophenyl)-2,4-dioxo-1,3,4,5- tetrahydro-2H-pyrano[2,3-d]pyrimidine-6-carbonitrile (4e). Mp. (°C): 251–254; yield: 88% IR (νmax cm−1): 791.72, 879.03 (C–H), 1069.07 (C–Br str), 1103 (C–O–C str), 1204.64 (C–NH2 str), 1470.16, 1527.18 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C aromatic str), 1658.22, 3090.93 (C
C aromatic str), 1658.22, 3090.93 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C alkene str), 1714.32 (C
C alkene str), 1714.32 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O str), 2190.26 (C
O str), 2190.26 (C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N str), 2937.96 (C–H str), 3001.83 (
N str), 2937.96 (C–H str), 3001.83 (![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH str), 3313.26, 3409.93 (NH2 str), 3735.60 (N–H str). 1H NMR (400 MHz, DMSO-d6)/δ (ppm): 4.23 (s, 1H, C–H), 7.14 (2H, NH2, D2O exchangeable), 7.19 (dt, 1H, Ar–H), 7.54 (t, J = 8, 1H, Ar–H), 7.90 (d, J = 1.8, 1H, Ar–H), 8.05 (t, J = 1.8, 1H, Ar–H), 11.04 (1H, NH, D2O exchangeable), 12.05 (1H, NH, D2O exchangeable). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 39.4, 58.7, 88.2, 114.3, 122, 127.1, 129.5, 132.1, 147.4, 150, 153, 158.2, 160.5.
CH str), 3313.26, 3409.93 (NH2 str), 3735.60 (N–H str). 1H NMR (400 MHz, DMSO-d6)/δ (ppm): 4.23 (s, 1H, C–H), 7.14 (2H, NH2, D2O exchangeable), 7.19 (dt, 1H, Ar–H), 7.54 (t, J = 8, 1H, Ar–H), 7.90 (d, J = 1.8, 1H, Ar–H), 8.05 (t, J = 1.8, 1H, Ar–H), 11.04 (1H, NH, D2O exchangeable), 12.05 (1H, NH, D2O exchangeable). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 39.4, 58.7, 88.2, 114.3, 122, 127.1, 129.5, 132.1, 147.4, 150, 153, 158.2, 160.5.
2.3.1.6 7-Amino-5-(2-chlorophenyl)-2,4-dioxo-1,3,4,5-tetrahydro-2H-pyrano[2,3-d]pyrimidine-6-carbonitrile (4f). Mp. (°C): 210–212; yield: 89% IR (νmax cm−1): 749.59 (C–H), 1039.74 (C–Cl str), 1099.42 (C–O–C str), 1210.32 (C–NH2 str), 1404.77, 1574.45 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C str Ar), 1617.82, 3038.65 (C
C str Ar), 1617.82, 3038.65 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C alkene str) 1666.73, 1706.15 (C
C alkene str) 1666.73, 1706.15 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O str), 2202.85 (C
O str), 2202.85 (C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N str) 2820.95 (C–H str), 3038.65 (
N str) 2820.95 (C–H str), 3038.65 (![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH str), 3314.60, 3167.96 (NH2 str), 3519.40 (N–H str). 1H NMR (400 MHz, DMSO-d6)/δ (ppm): 4.68 (s, 1H, C–H), 7.13 (2H, NH2, D2O exchangeable), 7.18–7.20 (m, 1H, Ar–H), 7.23 (d, J = 4, Ar–H), 7.31 (d, J = 7.4, 1H, Ar–H), 11.04 (1H, NH, D2O exchangeable), 12.08 (1H, NH, D2O exchangeable). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 39.3, 86.9, 112.7, 114.1, 128.4, 130.1, 130.3, 130.9, 134.7, 135.4, 158.6.
CH str), 3314.60, 3167.96 (NH2 str), 3519.40 (N–H str). 1H NMR (400 MHz, DMSO-d6)/δ (ppm): 4.68 (s, 1H, C–H), 7.13 (2H, NH2, D2O exchangeable), 7.18–7.20 (m, 1H, Ar–H), 7.23 (d, J = 4, Ar–H), 7.31 (d, J = 7.4, 1H, Ar–H), 11.04 (1H, NH, D2O exchangeable), 12.08 (1H, NH, D2O exchangeable). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 39.3, 86.9, 112.7, 114.1, 128.4, 130.1, 130.3, 130.9, 134.7, 135.4, 158.6.
2.3.1.7 7-Amino-5-(3a,7a-dihydro-1H-indol-3-yl)-2,4-dioxo-1,34,5-tetrahydro-2H-pyrano[2,3-d]pyridine-6-carbonitrile (4g). Mp.: > 300; yield: 91% IR (νmax cm−1): 1637.82 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C str), 2214.84 (C
C str), 2214.84 (C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N str), 3016.85 (C–H Ar), 3272.36 (N–H), 3356.09 (N–H str). 1H NMR (400 MHz, DMSO-d6)/δ (ppm): 3.35 (s, 1H, H-5), 7.21 (2H, NH2), 7.52 (d, J = 7.2, 2H, H–Ar), 8.0 (d, J = 7.2, 2H, H–Ar), 8.66 (d, J = 4.8,1H), 11.02 (1H, NH), 12.71 (1H, NH). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 39.3, 69.7, 119.5, 123.1, 127.3, 129.6, 133.9, 136.6, 140.3, 144.2, 150.9, 153, 163.8, 165.
N str), 3016.85 (C–H Ar), 3272.36 (N–H), 3356.09 (N–H str). 1H NMR (400 MHz, DMSO-d6)/δ (ppm): 3.35 (s, 1H, H-5), 7.21 (2H, NH2), 7.52 (d, J = 7.2, 2H, H–Ar), 8.0 (d, J = 7.2, 2H, H–Ar), 8.66 (d, J = 4.8,1H), 11.02 (1H, NH), 12.71 (1H, NH). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 39.3, 69.7, 119.5, 123.1, 127.3, 129.6, 133.9, 136.6, 140.3, 144.2, 150.9, 153, 163.8, 165.
2.3.1.8 7-Amino-5-(4-nitrophenyl)-4-oxo-2-thioxo-1,3,4,5-tetrahydro-2H-pyrano[2,3-d]pyrimidine-6-carbonitrile (4h). Mp.: 233–234 °C; yield: 90% IR (νmax cm−1): 694.85 (C–H), 817.17 (C–H), 962.56 (C–O str), 1120.03 (C–O–C str), 1239.78 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S str), 1350.14 (C–N str), 1404.22 (C–N str), 1472.59 (C
S str), 1350.14 (C–N str), 1404.22 (C–N str), 1472.59 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C str Ar), 1520.79 (N
C str Ar), 1520.79 (N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O2 symmetric str), 1565.46 (N
O2 symmetric str), 1565.46 (N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O2 str Ar), 1683.26 (C
O2 str Ar), 1683.26 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O str), 2198.83 (C
O str), 2198.83 (C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N str), 2887.81 (C–H str), 3108.47 (C–H str Ar), 3185.25 (N–H str. symmetric), 3361.82 (N–H str asymmetric), 1H NMR (400 MHz, DMSO-d6)/δ (ppm): 4.45 (s, 1H, H-5), 6.13 (s, 2H, NH2), 7.30 (d, J = 8, 2H, H–Ar), 8.14 (d, J = 8, 2H, H–Ar), 11.15 (s, 1H, NH), 12.50 (s, 1H, NH). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 35.8, 57.7, 92.9, 119.3, 124.1, 129.6, 147.1, 151.4, 152.4, 157.9, 161, 174.5.
N str), 2887.81 (C–H str), 3108.47 (C–H str Ar), 3185.25 (N–H str. symmetric), 3361.82 (N–H str asymmetric), 1H NMR (400 MHz, DMSO-d6)/δ (ppm): 4.45 (s, 1H, H-5), 6.13 (s, 2H, NH2), 7.30 (d, J = 8, 2H, H–Ar), 8.14 (d, J = 8, 2H, H–Ar), 11.15 (s, 1H, NH), 12.50 (s, 1H, NH). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 35.8, 57.7, 92.9, 119.3, 124.1, 129.6, 147.1, 151.4, 152.4, 157.9, 161, 174.5.
2.3.1.9 7-Amino-5-(thiophen-2-yl)-4-oxo-2-thioxo-1,3,4,5-tetrahydro-2H-pyrano[2,3-d]pyrimidine-6-carbonitrile (4i). Mp.: 280–284 °C; yield: 85% IR (νmax cm−1): 627.41 (C–S str), 734.38 (C–H str), 1138.57 (C–O str), 1217 (C–O–C str), 1259.13 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S str), 1302, 1398.83 (C–NH2 str), 1518.46 (C
S str), 1302, 1398.83 (C–NH2 str), 1518.46 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C str Ar), 1675.81 (C
C str Ar), 1675.81 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O str), 2311.94 (C
O str), 2311.94 (C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N str), 2883.24 (C–H str), 2996.53 (
N str), 2883.24 (C–H str), 2996.53 (![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) CH str), 3110.25 (C–H str Ar), 3392.53 (NH2 str). 1H NMR (500 MHz, DMSO-d6)/δ (ppm): 3.38 (s, 1H), 7.39 (d, J = 4.4, 1H, H–Ar), 8.25 (d, J = 1.89, 1H, H–Ar), 8.22 (d, J = 4.86, 1H), 12.42 (brs, 2H, NH). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 39.4, 112.2, 129.2, 137.4, 143.7, 146.9, 161.3, 162.2, 178.7.
CH str), 3110.25 (C–H str Ar), 3392.53 (NH2 str). 1H NMR (500 MHz, DMSO-d6)/δ (ppm): 3.38 (s, 1H), 7.39 (d, J = 4.4, 1H, H–Ar), 8.25 (d, J = 1.89, 1H, H–Ar), 8.22 (d, J = 4.86, 1H), 12.42 (brs, 2H, NH). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 39.4, 112.2, 129.2, 137.4, 143.7, 146.9, 161.3, 162.2, 178.7.
2.3.1.10 7-Amino-4-oxo-5-(pyren-2-yl)-2-thioxo-1,3,4,5-tetrahydro-2H-pyrano[2,3-d]pyrimidine-6-carbonitrile (4j). Mp.: 250–252 °C; yield: 80% IR (νmax cm−1): 785.04 (C–H str), 944.44 (C–O str), 1054.71 (C–O–C str), 1226.28 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S str), 1326.62, 1388.25 (C–NH2 str), 1517.14 (C
S str), 1326.62, 1388.25 (C–NH2 str), 1517.14 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C str Ar), 1545.37 (N–H bending), 1675.25 (C
C str Ar), 1545.37 (N–H bending), 1675.25 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O str), 2312.16 (C
O str), 2312.16 (C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N str), 2886.40 (C–H str Ar), 3392.76 (NH2 str). 1H NMR (500 MHz, DMSO-d6)/δ (ppm): 3.39 (s, 1H), 8.23 (m, 2H), 8.42 (m, 4H), 8.63 (m, 3H), 9.14 (s, 1H, NH), 12.42 (brs, 2H, NH). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 39.9, 121.5, 133.4, 153.6, 159.6, 162, 179.5.
N str), 2886.40 (C–H str Ar), 3392.76 (NH2 str). 1H NMR (500 MHz, DMSO-d6)/δ (ppm): 3.39 (s, 1H), 8.23 (m, 2H), 8.42 (m, 4H), 8.63 (m, 3H), 9.14 (s, 1H, NH), 12.42 (brs, 2H, NH). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 39.9, 121.5, 133.4, 153.6, 159.6, 162, 179.5.
2.3.1.11 7-Amino-5-(pyrrole-2-yl)-4-oxo-2-thioxo-1,3,4,5-tetrahydro-2H-pyrano[2,3-d]pyrimidine-6-carbonitrile (4k). Mp.: 280–283 °C; yield: 85% IR (νmax cm−1): 745.66 (C–H str), 1147.88 (C–O str), 1179.41 (C–O–C str), 1224.37 (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S str), 1315.68, 1381.39 (C–NH2 str), 1510.59 (C
S str), 1315.68, 1381.39 (C–NH2 str), 1510.59 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C str Ar), 1584.70 (N–H bending) 1655.07 (C
C str Ar), 1584.70 (N–H bending) 1655.07 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O str), 2220.82 (C
O str), 2220.82 (C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N str), 2883.89 (C–H str), 3067.04 (C–H str Ar). 1H NMR (500 MHz, DMSO-d6)/δ (ppm): 3.38 (s, 1H), 6.59 (s, 2H, C–H), 7.43 (br, s, 2H, NH2), 7.74 (s, 1H, C–H), 8.14 (s, 1H), 12.3 (s, 1H, NH), 13.3 (s, br, NH). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 106.7, 115.2, 129.7, 134.8, 141.3, 162.9, 178.1.
N str), 2883.89 (C–H str), 3067.04 (C–H str Ar). 1H NMR (500 MHz, DMSO-d6)/δ (ppm): 3.38 (s, 1H), 6.59 (s, 2H, C–H), 7.43 (br, s, 2H, NH2), 7.74 (s, 1H, C–H), 8.14 (s, 1H), 12.3 (s, 1H, NH), 13.3 (s, br, NH). 13C NMR (125 MHz, DMSO-d6)/δ (ppm): 106.7, 115.2, 129.7, 134.8, 141.3, 162.9, 178.1.
3 Results and discussion
3.1 Characterization of Fe3O4@SiO2@taurine
In curve (a), the FT-IR spectrum of Fe3O4 shows characteristic absorption bands at 618.41, 1632.15, and 3443.08 cm−1, confirming the formation of iron oxide nanoparticles. The band at 618 cm−1 corresponds to Fe–O stretching vibrations, while the peaks at 1632 and 3443 cm−1 are assigned to the bending and stretching vibrations of adsorbed water molecules (H–O–H), respectively.37,38 In curve (b), corresponding to Fe3O4@SiO2, additional absorption bands appear at 469.76, 616.11, 1108.49, 1389.05, 1634.64, and 3435.23 cm−1, confirming successful silica coating. The strong band at 1108 cm−1 is assigned to Si–O–Si asymmetric stretching, while the peak near 1389.05 cm−1 corresponds to Si–O symmetric stretching. Furthermore, the band at 469.76 cm−1 is attributed to Si–O–Si bending vibrations, providing further evidence of the silica shell.37,39
In curve (c), representing the taurine-functionalized Fe3O4@SiO2 catalyst, distinct peaks are observed at 470.32, 629.27, 745.74, 801.78, 890.86, 1101.40, 1200.05, 1390.11, 1634.84, 2069.19, and 3438.20 cm−1. The absorption at 1200 cm−1 corresponds to S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O symmetric stretching. The presence of a band at 745 cm−1 is related to the stretching vibration of S–O, while the symmetric and asymmetric stretching bands of O
O symmetric stretching. The presence of a band at 745 cm−1 is related to the stretching vibration of S–O, while the symmetric and asymmetric stretching bands of O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S
S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O appear at 801 cm−1 and 890.86 cm−1, respectively, indicating successful taurine immobilization. The band at 2069 cm−1 is attributed to ammonium ions within the structure.39 The consistent presence of Fe–O, Si–O–Si, and O–H related bands across all spectra confirms the preservation of the core–shell framework and the effectiveness of sequential surface modification. Collectively, these spectral features validate the stepwise synthesis of Fe3O4, silica encapsulation, and taurine functionalization.37,39
O appear at 801 cm−1 and 890.86 cm−1, respectively, indicating successful taurine immobilization. The band at 2069 cm−1 is attributed to ammonium ions within the structure.39 The consistent presence of Fe–O, Si–O–Si, and O–H related bands across all spectra confirms the preservation of the core–shell framework and the effectiveness of sequential surface modification. Collectively, these spectral features validate the stepwise synthesis of Fe3O4, silica encapsulation, and taurine functionalization.37,39
 | ||
| Fig. 3 X-ray diffraction (XRD) patterns of (a) the Fe3O4@SiO2@taurine nanocomposite and (b) Fe3O4 and Fe3O4@SiO2. | ||
The average crystallite size of the Fe3O4@SiO2@taurine nanoparticles was estimated using the Scherrer equation:
D = Kλ/β![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos |
The EDS spectra in Fig. 6(a)–(i) verify the elemental composition of the Fe3O4@SiO2@taurine nanocomposite. Clear signals for Fe, Si, and O correspond to the magnetite core and silica coating, while the additional peaks for sulfur (S) and nitrogen (N) originate exclusively from the sulfonic acid (–SO3H) and amino (–NH2) groups of taurine. The quantitative analysis revealed elemental proportions of approximately 0.2% Fe, 5.5% Si, 41.1% O, 1.2% S, 47.3% C, and 4.7% N. The detection of S and N peaks, which are absent in Fe3O4 and SiO2, provides direct confirmation of taurine functionalization on the silica shell and validates the successful formation of the nanocomposite.
Despite the low zeta potential, the catalyst demonstrates excellent heterogeneous catalytic activity, high yields, and easy magnetic recoverability, indicating that the active Fe sites and taurine groups remain accessible to the substrate during reactions. The near-neutral surface charge may also reduce undesired electrostatic interactions, enhancing substrate approach and facilitating recyclability. Overall, the measured zeta potential and pH confirm the chemical stability and practical applicability of Fe3O4@SiO2@taurine as an efficient, magnetically separable heterogeneous nanocatalyst.
The XRD and TEM/SEM analyses confirmed that the Fe3O4 core retains its crystalline inverse spinel structure after surface modification and that the Fe3O4@SiO2@taurine nanocatalyst possesses a well-defined spherical core–shell morphology with an average particle size of ∼22 nm. FT-IR spectroscopy and EDS analysis verified the successful immobilization of taurine on the silica surface through the appearance of characteristic sulfonic and amino functional groups, along with the presence of sulphur and nitrogen signals absent in the unmodified materials. TGA measurements demonstrated good thermal stability of the nanocomposite and further confirmed the presence of organic taurine moieties on the catalyst surface. In addition, zeta potential analysis revealed a near-neutral surface charge (+3.5 mV), consistent with the zwitterionic nature of taurine and indicative of chemical stability under reaction conditions. BET analysis showed a specific surface area of 4.203 m2 g−1, characteristic of iron oxide-based mesoporous materials, providing sufficient accessibility of active sites while maintaining structural robustness. Collectively, these results confirm the successful synthesis, surface functionalization, stability, and catalytic suitability of the Fe3O4@SiO2@taurine nanocatalyst.
3.2 The study of catalytic activity of Fe3O4@SiO2@taurine MNPs in the synthesis of pyrano[2,3-d]pyrimidines
To determine the optimum reaction conditions, a model three-component condensation reaction between an aromatic aldehyde, malononitrile, and barbituric acid was conducted using Fe3O4@SiO2@taurine as a catalyst. The reaction was examined under different reaction conditions and reflux for yield comparison. Under reflux, the transformation was completed within 30 min, affording 97% of the target product (Table 1, entry 13), whereas no product formation occurred in water at room temperature without a catalyst and solvent (Table 1, entry 1). When the reaction proceeded in the absence of a catalyst in water for 180 min at room temperature, the desired product was not formed (Table 1, entry 2). Using Fe3O4 as a catalyst in water provided only 65% yield after 120 min at room temperature (Table 1, entry 3), while SiO2 in ethanol gave 55% yield in 80 min at room temperature (Table 1, entry 4). Nano-Fe2O3 alone improved the yield to 68% in 60 min at 70 °C (Table 1, entry 5). The use of Fe3O4@SiO2 in a mixed solvent system of H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOH led to an enhanced yield of 78% under reflux in a shorter reaction time (Table 1, entry 6). Furthermore, we investigated the solvent free condition effect on the sample reaction. When 0.005 g of Fe3O4@SiO2@taurine was added in the reaction without solvent at 70 °C, the yield decreased to 50% (Table 1, entry 7). Catalyst loading studies showed that 0.005 g of catalyst resulted in 60% and 82% yield in 80 min in the presence of acetone and H2O
EtOH led to an enhanced yield of 78% under reflux in a shorter reaction time (Table 1, entry 6). Furthermore, we investigated the solvent free condition effect on the sample reaction. When 0.005 g of Fe3O4@SiO2@taurine was added in the reaction without solvent at 70 °C, the yield decreased to 50% (Table 1, entry 7). Catalyst loading studies showed that 0.005 g of catalyst resulted in 60% and 82% yield in 80 min in the presence of acetone and H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOH, respectively (Table 1, entries 8 & 9). Increasing the catalyst loading from 0.01 to 0.05 g resulted in a marked improvement in yield from 84 to 97% with a concomitant reduction in reaction time (Table 1, entries 10–13); notably, the optimized catalyst amount also showed good efficiency under solvent-free conditions, affording an 85% yield at 85 °C within 30 min. Based on these results, the optimized condition was identified as refluxing benzaldehyde, malononitrile, and barbituric acid in H2O
EtOH, respectively (Table 1, entries 8 & 9). Increasing the catalyst loading from 0.01 to 0.05 g resulted in a marked improvement in yield from 84 to 97% with a concomitant reduction in reaction time (Table 1, entries 10–13); notably, the optimized catalyst amount also showed good efficiency under solvent-free conditions, affording an 85% yield at 85 °C within 30 min. Based on these results, the optimized condition was identified as refluxing benzaldehyde, malononitrile, and barbituric acid in H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOH (1
EtOH (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) using 0.05 g of Fe3O4@SiO2@taurine (Table 1, entry 13). The use of ethanol, water, or their mixture as green solvents provides additional benefits, including environmental safety, non-toxicity, low cost, and operational simplicity.
1) using 0.05 g of Fe3O4@SiO2@taurine (Table 1, entry 13). The use of ethanol, water, or their mixture as green solvents provides additional benefits, including environmental safety, non-toxicity, low cost, and operational simplicity.
| S. no. | Catalyst | Solvent | Time (min) | Condition | Yield% |
|---|---|---|---|---|---|
| 1 | Catalyst free | Solvent free | 180 | 90 °C | NA |
| 2 | Catalyst free | H2O | 180 | R.T. | NA |
| 3 | Fe2O3 | H2O | 120 | R.T. | 65 |
| 4 | SiO2 | EtOH | 80 | R.T. | 55 |
| 5 | Fe3O4 NPs | H2O | 60 | 70 °C | 68 |
| 6 | Fe3O4@SiO2 | H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOH EtOH |
60 | Reflux | 78 |
| 7 | Fe3O4@SiO2@taurine (0.005 g) | Solvent free | 80 | 70 °C | 50 |
| 8 | Fe3O4@SiO2@taurine (0.005 g) | Acetone | 80 | Reflux | 60 |
| 9 | Fe3O4@SiO2@taurine (0.005 g) | H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOH EtOH |
80 | Reflux | 82 |
| 10 | Fe3O4@SiO2@taurine (0.01 g) | H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOH EtOH |
60 | Reflux | 84 |
| 11 | Fe3O4@SiO2@taurine (0.03 g) | H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOH EtOH |
45 | Reflux | 90 |
| 12 | Fe3O4@SiO2@taurine (0.05 g) | Solvent free | 30 | 85 °C | 85 |
| 13 | Fe3O4@SiO2@taurine (0.05 g) | H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOH EtOH |
30 | Reflux | 97 |
Table 1 summarizes the effect of various catalysts, solvents, reaction times and conditions on the yield of a chemical reaction. Notably, after functionalization of the catalyst with taurine (entries 9–13), both efficiency and reaction speed were significantly boosted. As the taurine functionalized catalyst increased from 0.005 to 0.05 g, the yield steadily enhanced from 82 to 97%, while the reaction time decreased from 80 to 30 min. These results highlight the synergistic effect of taurine modified Fe3O4@SiO2 in providing enhanced catalytic performance, likely due to increase surface activity and better interaction with the reactants under reflux in a polar protic solvent system. The 0.05 g loading represents an optimal balance of catalytic power and operational efficiency, making it the most effective condition for the synthesis of pyrano[2,3-d]pyrimidine derivatives.
Table 2 presents a comparative study of previously reported catalysts with Fe3O4@SiO2@taurine. The data in the table clearly establish that the catalyst Fe3O4@SiO2@taurine, synthesized in this work, performs comparatively better than previously reported catalysts in terms of yield and reaction time under mild conditions. It achieved an impressive 96% yield in just 30 minutes under reflux conditions in a green solvent system (H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOH). Additionally, many other catalysts require longer reaction times or give lower yields. The synthesized Fe3O4@SiO2@taurine catalyst combines high efficiency, short reaction time, excellent yield, a non-toxic, environmentally friendly and straightforward approach with simple workup and easy recovery, making it an efficient and practical catalyst for the synthesis of pyrano[2,3-d]pyrimidine derivatives.
EtOH). Additionally, many other catalysts require longer reaction times or give lower yields. The synthesized Fe3O4@SiO2@taurine catalyst combines high efficiency, short reaction time, excellent yield, a non-toxic, environmentally friendly and straightforward approach with simple workup and easy recovery, making it an efficient and practical catalyst for the synthesis of pyrano[2,3-d]pyrimidine derivatives.
| S no. | Catalyst | Solvent | Condition | Time (min) | Yield | Year/Ref |
|---|---|---|---|---|---|---|
| 1 | Et3N | DMF | MW | 10–12 | 65–70 | 2003 (ref. 41) |
| 2 | DAHP | EtOH | R.T. | 120 | 71–81 | 2008 (ref. 42) |
| 3 | L-Proline | Aq. EtOH | Reflux | 30–120 | 68–85 | 2009 (ref. 43) |
| 4 | [KAl(SO4)2] | H2O | 80 °C | 30–45 | 81–88 | 2010 (ref. 44) |
| 5 | α-Fe2O3 | EtOH | R.T. | 30 | 93 | 2011 (ref. 45) |
| 6 | Zn[(L)proline]2 | EtOH | Reflux | 30–720 | 80–90 | 2019 (ref. 46) |
| 7 | SBA-Pr-SO3H | Solvent free | 140 °C | 5–45 | 91 | 2013 (ref. 47) |
| 8 | DMA | EtOH | Reflux | 20 | 90 | 2014 (ref. 48) |
| 9 | Zn[(L)proline]2 | EtOH | Reflux | 30–720 | 80–90 | 2019 (ref. 46) |
| 10 | γ-Fe2O3@SiO2@[BisAPTES]Cl2-NPs | H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOH (2 EtOH (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
80 °C | 12 | 85 | 2019 (ref. 49) |
| 11 | Nano-Fe3O4@APTES@isatin-SO3H MNPs | EtOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O H2O |
Reflux | 7–25 | 86–95 | 2019 (ref. 50) |
| 12 | β-CD | H2O | 80 °C | 10 | 93 | 2020 (ref. 51) |
| 13 | CoFe2O4@FA-Er | H2O | 100 °C | 15 | 96 | 2021 (ref. 52) |
| 14 | Fe3O4@SiO2@taurine | H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EtOH EtOH |
Reflux | 30 | 97 | This work |
The TON and TOF values in this study were calculated on the basis of the Fe wt% present in the catalyst, which is the accepted method for heterogeneous nanocatalysts where metal atoms serve as the true catalytic sites (Table 3). Similar metal loading-based TON/TOF evaluation approaches have been reported for related magnetic and organometallic catalyst systems.53–56
3.3 Recyclability and reusability of Fe3O4@SiO2@taurine
From the perspective of green and sustainable chemistry, evaluating the recyclability and reusability of the catalyst is crucial for both environmental and economic considerations. After each reaction cycle, the nanocatalyst was conveniently separated using an external magnet and washed with ethanol. Before reuse of the catalyst in the reaction, the catalyst was dried at 60 °C. The recyclability test over five consecutive runs showed that the catalyst retained nearly the same activity, affording yields in the range of 98–90% up to four cycles but the yield of compound 4a was decreased with increase in reaction time in the fifth cycle (yield 85%) (Fig. 10). As shown in Fig. (11), the FTIR spectrum of recycled Fe3O4@SiO2@taurine after the fourth run exhibited that the catalyst could be reused numerous times without significant loss in yield%, chemical stability and structure integrity of the catalyst. The catalyst retained 85% yield after five consecutive cycles, indicating moderate recyclability.3.4 Mechanism of reaction
Mechanistic pathways involving base-mediated condensation and cyclization reactions have been described in several organic methodology studies.57 The plausible mechanism for the Fe3O4@SiO2@taurine-catalyzed synthesis of pyrano[2,3-d]pyrimidine derivatives is illustrated in Scheme 3. Initially, the nanocatalyst, acting as a bifunctional donor–acceptor, activates the carbonyl group of the aromatic aldehyde (1) to form intermediate (1a). Concurrently, malononitrile (2) undergoes a Michael addition with barbituric acid (3), followed by tautomerization to yield intermediates (2a) and (3a). A Knoevenagel condensation between (2a) and (1a) produces adduct (A), which undergoes a subsequent Michael addition with (3a) to form adduct (B). This intermediate tautomerizes (imine-enamine shift) to (C), undergoes cyclization to afford (D), and finally tautomerizes to furnish the desired pyrano[2,3-d]pyrimidine (A).The substituent effects observed in the synthesis correlate strongly with the rate-determining step of the proposed mechanism. The reaction initiates with catalyst-assisted activation of the aldehyde through hydrogen bonding with the sulfonic acid group of taurine, followed by nucleophilic attack of the malononitrile anion generated at the basic NH2/NH3+ sites. Aldehydes containing electron-withdrawing groups exhibit enhanced reactivity because their carbonyl carbons possess increased electrophilicity, facilitating rapid formation of the Knoevenagel intermediate. However, aldehydes containing electron-withdrawing substituents reacted faster with malononitrile and barbituric acid, affording the products in higher yields and shorter reaction times compared to those bearing electron-donating groups. The presence of electron-withdrawing substituents enhances the positive (electrophilic) character of the carbonyl carbon, thereby lowering the activation energy required for nucleophilic attack by the malononitrile-derived carbanion. As a result, the formation of the arylidene malononitrile intermediate occurs more rapidly. Moreover, the nucleophilic addition to this intermediate is also accelerated.
4 Conclusion
In summary, we have successfully synthesized and characterized a magnetically recoverable Fe3O4@SiO2@taurine nanocatalyst and demonstrated its excellent catalytic activity in the one-pot, three-component synthesis of pyrano[2,3-d]pyrimidine derivatives. Comprehensive analyses (FT-IR, XRD, TEM, SEM-EDS, TGA, Zeta and BET) confirmed the structural integrity, thermal stability, and effective surface functionalization of the nanocomposite. The catalyst exhibited high efficiency, affording excellent yields in short reaction times under green solvent conditions, while also offering easy magnetic separation and reusability over multiple cycles with negligible loss of activity. Compared to conventional catalysts, Fe3O4@SiO2@taurine provides a sustainable, non-toxic, and cost-effective alternative for heterocyclic synthesis. These results highlight its potential as a versatile platform for green organic transformations, paving the way for broader applications in sustainable catalysis and pharmaceutical chemistry.Conflicts of interest
The authors declare no conflict of interest, financial or otherwise.Data availability
All data supporting the findings of this study are included within the article. This comprises detailed experimental procedures and all characterization and analytical data (FTIR, XRD, SEM, TEM, VSM and NMR spectra). No additional datasets were generated or analysed during the current study.Acknowledgements
The authors are highly thankful to the Ministry of Education and SPD-RUSA Rajasthan for the support received under the RUSA-2.0 Project (F30(16)SPD/RUSA/2026/178). We sincerely thank the Department of Chemistry, Mohanlal Sukhadia University (MLSU), for providing NMR spectral facilities, and greatly appreciate the MNIT Jaipur facilities for timely assistance with FTIR, XRD, HR-TEM, and FESEM analyses. We thank SAIF/CIL Punjab University for providing BET and zeta potential analysis. We also acknowledge the Central University of Gujarat for providing the TGA facility. CA gratefully acknowledges the ANRF-PAIR-B grant (ANRF/PAIR/2025/000015/PAIR-B).References
- V. Polshettiwar and R. S. Varma, Green Chemistry by Nano-Catalysis, Green Chem., 2010, 12(5), 743, 10.1039/b921171c.
- Z. Varzi and A. Maleki, Design and Preparation of ZnS-ZnFe2 O4 : A Green and Efficient Hybrid Nanocatalyst for the Multicomponent Synthesis of 2,4,5-triaryl-1 H -imidazoles, Appl. Organomet. Chem., 2019, 33(8), e5008, DOI:10.1002/aoc.5008.
- D. K. Jambhulkar, R. P. Ugwekar, B. A. Bhanvase and D. P. Barai, A Review on Solid Base Heterogeneous Catalysts: Preparation, Characterization and Applications, Chem. Eng. Commun., 2022, 209(4), 433–484, DOI:10.1080/00986445.2020.1864623.
- S. B. Kalidindi and B. R. Jagirdar, Nanocatalysis and Prospects of Green Chemistry, ChemSusChem, 2012, 5(1), 65–75, DOI:10.1002/cssc.201100377.
- M. Duan, L. Jiang, G. Zeng, D. Wang, W. Tang, J. Liang, H. Wang, D. He, Z. Liu and L. Tang, Bimetallic Nanoparticles/Metal-Organic Frameworks: Synthesis, Applications and Challenges, Appl. Mater. Today, 2020, 19, 100564, DOI:10.1016/j.apmt.2020.100564.
- S. Vásquez-Céspedes, R. C. Betori, M. A. Cismesia, J. K. Kirsch and Q. Yang, Heterogeneous Catalysis for Cross-Coupling Reactions: An Underutilized Powerful and Sustainable Tool in the Fine Chemical Industry?, Org. Process Res. Dev., 2021, 25(4), 740–753, DOI:10.1021/acs.oprd.1c00041.
- M. Shokouhimehr, K. Hong, T. H. Lee, C. W. Moon, S. P. Hong, K. Zhang, J. M. Suh, K. S. Choi, R. S. Varma and H. W. Jang, Magnetically Retrievable Nanocomposite Adorned with Pd Nanocatalysts: Efficient Reduction of Nitroaromatics in Aqueous Media, Green Chem., 2018, 20(16), 3809–3817, 10.1039/C8GC01240G.
- S. Azad and B. B. F. Mirjalili, One-Pot Solvent-Free Synthesis of 2,3-Dihydro-2-Substituted-1H-Naphtho[1,2-e][1,3]Oxazine Derivatives Using Fe3O4@nano-Cellulose/TiCl as a Bio-Based and Recyclable Magnetic Nano-Catalyst, Mol. Divers., 2019, 23(2), 413–420, DOI:10.1007/s11030-018-9884-6.
- N. Kerru, S. V. H. S. Bhaskaruni, L. Gummidi, S. N. Maddila, P. Singh and S. B. Jonnalagadda, Efficient Synthesis of Novel Pyrazole-Linked 1,2,4-Triazolidine-3-Thiones Using Bismuth on Zirconium Oxide as a Recyclable Catalyst in Aqueous Medium, Mol. Divers., 2020, 24(2), 345–354, DOI:10.1007/s11030-019-09957-0.
- S. F. Hamzavi, S. Gerivani, S. Saeedi, K. Naghdipari and G. Shahverdizadeh, Preparation and Characterization of a Novel Spherical Cellulose–Copper(II) Oxide Composite Particles: As a Heterogeneous Catalyst for the Click Reaction, Mol. Divers., 2020, 24(1), 201–209, DOI:10.1007/s11030-019-09942-7.
- R. S. Varma, Greener and Sustainable Trends in Synthesis of Organics and Nanomaterials, ACS Sustain. Chem. Eng., 2016, 4(11), 5866–5878, DOI:10.1021/acssuschemeng.6b01623.
- H. Kiyani and F. Ghorbani, Efficient Tandem Synthesis of a Variety of Pyran-Annulated Heterocycles, 3,4-Disubstituted Isoxazol-5(4H)-Ones, and α,β-Unsaturated Nitriles Catalyzed by Potassium Hydrogen Phthalate in Water, Res. Chem. Intermed., 2015, 41(10), 7847–7882, DOI:10.1007/s11164-014-1863-7.
- H. Ostadzadeh and H. Kiyani, Multicomponent Synthesis of Tetrahydrobenzo[ b ]Pyrans, Pyrano[2,3- d ]Pyrimidines, and Dihydropyrano[3,2- c ]Chromenes Catalyzed by Sodium Benzoate, Polycycl. Aromat. Compd., 2023, 43(10), 9318–9337, DOI:10.1080/10406638.2022.2162091.
- Z. Faramarzi and H. Kiyani, Steglich's Base Catalyzed Three-Component Synthesis of Isoxazol-5-Ones, Polycycl. Aromat. Compd., 2023, 43(4), 3099–3121, DOI:10.1080/10406638.2022.2061533.
- S. Kamalifar and H. Kiyani, Facile and Efficient Synthesis of 9-Aryl-1,8-Dioxo-Octahydroxanthenes Catalyzed by Sulfacetamide, Polycycl. Aromat. Compd., 2022, 42(6), 3675–3693, DOI:10.1080/10406638.2021.1872656.
- Y. Chan, J. P. Zimmer, M. Stroh, J. S. Steckel, R. K. Jain and M. G. Bawendi, Incorporation of Luminescent Nanocrystals into Monodisperse Core–Shell Silica Microspheres, Adv. Mater., 2004, 16(23–24), 2092–2097, DOI:10.1002/adma.200400237.
- Y. H. Deng, W. L. Yang, C. C. Wang and S. K. Fu, A Novel Approach for Preparation of Thermoresponsive Polymer Magnetic Microspheres with Core–Shell Structure, Adv. Mater., 2003, 15(20), 1729–1732, DOI:10.1002/adma.200305459.
- Y. Lu, Y. Yin, B. T. Mayers and Y. Xia, Modifying the Surface Properties of Superparamagnetic Iron Oxide Nanoparticles through A Sol−Gel Approach, Nano Lett., 2002, 2(3), 183–186, DOI:10.1021/nl015681q.
- D. Yang, J. Hu and S. Fu, Controlled Synthesis of Magnetite−Silica Nanocomposites via a Seeded Sol−Gel Approach, J. Phys. Chem. C, 2009, 113(18), 7646–7651, DOI:10.1021/jp900868d.
- X. Xu, C. Deng, M. Gao, W. Yu, P. Yang and X. Zhang, Synthesis of Magnetic Microspheres with Immobilized Metal Ions for Enrichment and Direct Determination of Phosphopeptides by Matrix-Assisted Laser Desorption Ionization Mass Spectrometry, Adv. Mater., 2006, 18(24), 3289–3293, DOI:10.1002/adma.200601546.
- Y. Deng, D. Qi, C. Deng, X. Zhang and D. Zhao, Superparamagnetic High-Magnetization Microspheres with an Fe3 O4 @SiO2 Core and Perpendicularly Aligned Mesoporous SiO2 Shell for Removal of Microcystins, J. Am. Chem. Soc., 2008, 130(1), 28–29, DOI:10.1021/ja0777584.
- J. Ge, Q. Zhang, T. Zhang and Y. Yin, Core–Satellite Nanocomposite Catalysts Protected by a Porous Silica Shell: Controllable Reactivity, High Stability, and Magnetic Recyclability, Angew. Chem., 2008, 120(46), 9056–9060, DOI:10.1002/ange.200803968.
- S. Chen, J. Feng, X. Guo, J. Hong and W. Ding, One-Step Wet Chemistry for Preparation of Magnetite Nanorods, Mater. Lett., 2005, 59(8–9), 985–988, DOI:10.1016/j.matlet.2004.11.043.
- T. K. Kim, M. N. Lee, S. H. Lee, Y. C. Park, C. K. Jung and J.-H. Boo, Development of Surface Coating Technology of TiO2 Powder and Improvement of Photocatalytic Activity by Surface Modification, Thin Solid Films, 2005, 475(1–2), 171–177, DOI:10.1016/j.tsf.2004.07.021.
- H. Zou, Z. Luo, X. Yang, Q. Xie and Y. Zhou, Toward Emerging Applications Using Core–Shell Nanostructured Materials: A Review, J. Mater. Sci., 2022, 57(24), 10912–10942, DOI:10.1007/s10853-022-07328-z.
- Y. Zhao, Co-Precipitated Ni/Mn Shell Coated Nano Cu-Rich Core Structure: A Phase-Field Study, J. Mater. Res. Technol., 2022, 21, 546–560, DOI:10.1016/j.jmrt.2022.09.032.
- M. Rimoldi, A. J. Howarth, M. R. DeStefano, L. Lin, S. Goswami, P. Li, J. T. Hupp and O. K. Farha, Catalytic Zirconium/Hafnium-Based Metal–Organic Frameworks, ACS Catal., 2017, 7(2), 997–1014, DOI:10.1021/acscatal.6b02923.
- T. L. H. Doan, T. Q. Dao, H. N. Tran, P. H. Tran and T. N. Le, An Efficient Combination of Zr-MOF and Microwave Irradiation in Catalytic Lewis Acid Friedel–Crafts Benzoylation, Dalton Trans., 2016, 45(18), 7875–7880, 10.1039/C6DT00827E.
- V. T. Nguyen, H. Q. Ngo, D. T. Le, T. Truong and N. T. S. Phan, Iron-Catalyzed Domino Sequences: One-Pot Oxidative Synthesis of Quinazolinones Using Metal–Organic Framework Fe 3 O(BPDC) 3 as an Efficient Heterogeneous Catalyst, Chem. Eng. J., 2016, 284, 778–785, DOI:10.1016/j.cej.2015.09.036.
- S. Klotz, G. Steinle-Neumann, Th. Strässle, J. Philippe, Th. Hansen and M. J. Wenzel, Magnetism and the Verwey Transition in Fe 3 O 4 under Pressure, Phys. Rev. B:Condens. Matter Mater. Phys., 2008, 77(1), 012411, DOI:10.1103/PhysRevB.77.012411.
- M. Bonini, A. Wiedenmann and P. Baglioni, Synthesis and Characterization of Magnetic Nanoparticles Coated with a Uniform Silica Shell, Mater. Sci. Eng. C, 2006, 26(5–7), 745–750, DOI:10.1016/j.msec.2005.09.042.
- R. Dawn, M. Zzaman, F. Faizal, C. Kiran, A. Kumari, R. Shahid, C. Panatarani, I. M. Joni, V. K. Verma, S. K. Sahoo, K. Amemiya and V. R. Singh, Origin of Magnetization in Silica-Coated Fe3O4 Nanoparticles Revealed by Soft X-Ray Magnetic Circular Dichroism, Braz. J. Phys., 2022, 52(3), 99, DOI:10.1007/s13538-022-01102-x.
- M. H. R. Farimani, N. Shahtahmasebi, M. Rezaee Roknabadi, N. Ghows and A. Kazemi, Study of Structural and Magnetic Properties of Superparamagnetic Fe3O4/SiO2 Core–Shell Nanocomposites Synthesized with Hydrophilic Citrate-Modified Fe3O4 Seeds via a Sol–Gel Approach, Phys. E, 2013, 53, 207–216, DOI:10.1016/j.physe.2013.04.032.
- S. Sobhanardakani, A. Jafari, R. Zandipak and A. Meidanchi, Removal of Heavy Metal (Hg(II) and Cr(VI)) Ions from Aqueous Solutions Using Fe2O3@SiO2 Thin Films as a Novel Adsorbent, Process Saf. Environ. Prot., 2018, 120, 348–357, DOI:10.1016/j.psep.2018.10.002.
- N. Daneshvar, F. Shirini, M. S. N. Langarudi and R. Karimi-Chayjani, Taurine as a Green Bio-Organic Catalyst for the Preparation of Bio-Active Barbituric and Thiobarbituric Acid Derivatives in Water Media, Bioorganic Chem, 2018, 77, 68–73, DOI:10.1016/j.bioorg.2017.12.021.
- F. Shirini and N. Daneshvar, Introduction of Taurine (2-Aminoethanesulfonic Acid) as a Green Bio-Organic Catalyst for the Promotion of Organic Reactions under Green Conditions, RSC Adv., 2016, 6(111), 110190–110205, 10.1039/C6RA15432H.
- F. Ahangaran, A. Hassanzadeh and S. Nouri, Surface Modification of Fe3O4@SiO2 Microsphere by Silane Coupling Agent, Int. Nano Lett., 2013, 3(1), 23, DOI:10.1186/2228-5326-3-23.
- M. Fuentes-Pérez, M. Sotelo-Lerma, J. L. Fuentes-Ríos, E. G. Morales-Espinoza, M. Serrano and M. E. Nicho, Synthesis and Study of Physicochemical Properties of Fe3O4@ZnFe2O4 Core/Shell Nanoparticles, J. Mater. Sci. Mater. Electron., 2021, 32(12), 16786–16799, DOI:10.1007/s10854-021-06236-3.
- S. Sharifi Sharifabad, B. B. F. Mirjalili and A. Bamoniri, Nano-SiO2/Taurine as a New Natural Based Catalyst for Synthesis of Hexahydroquinolines Derivative, Iran. J. Catal., 2022, 12(3), 365–372, DOI:10.30495/ijc.2022.1959104.1936.
- S. S. Alterary and A. AlKhamees, Synthesis, Surface Modification, and Characterization of Fe3 O4 @SiO2 Core@shell Nanostructure, Green Process. Synth., 2021, 10(1), 384–391, DOI:10.1515/gps-2021-0031.
- I. Devi, B. S. D. Kumar and P. J. Bhuyan, A Novel Three-Component One-Pot Synthesis of Pyrano[2,3-d]Pyrimidines and Pyrido[2,3-d]Pyrimidines Using Microwave Heating in the Solid State, Tetrahedron Lett., 2003, 44(45), 8307–8310, DOI:10.1016/j.tetlet.2003.09.063.
- S. Balalaie, S. Abdolmohammadi, H. R. Bijanzadeh and A. M. Amani, Diammonium Hydrogen Phosphate as a Versatile and Efficient Catalyst for the One-Pot Synthesis of Pyrano[2,3-d]Pyrimidinone Derivatives in Aqueous Media, Mol. Divers., 2008, 12(2), 85–91, DOI:10.1007/s11030-008-9079-7.
- M. Bararjanian, S. Balalaie, B. Movassag and A. M. Amani, One-Pot Synthesis of Pyrano[2,3-d]Pyrimidinone Derivatives Catalyzed by L-Proline in Aqueous Media, J. Iran. Chem. Soc., 2009, 6(2), 436–442, DOI:10.1007/BF03245854.
- A. Mobinikhaledi, N. Foroughifar and M. A. Bodaghi Fard, Eco-Friendly and Efficient Synthesis of Pyrano[2,3- d ] Pyrimidinone and Tetrahydrobenzo[ b ]Pyran Derivatives in Water, Synth. React. Inorg. Met.-Org. Nano-Met. Chem., 2010, 40(3), 179–185, DOI:10.1080/15533171003629121.
- H. Nagabhushana, S. Sandeep Saundalkar, L. Muralidhar, B. M. Nagabhushana, C. R. Girija, D. Nagaraja, M. A. Pasha and V. P. Jayashankara, α-Fe2O3 Nanoparticles: An Efficient, Inexpensive Catalyst for the One-Pot Preparation of 3,4-Dihydropyrano[c]Chromenes, Chin. Chem. Lett., 2011, 22(2), 143–146, DOI:10.1016/j.cclet.2010.09.020.
- M. M. Heravi, A. Ghods, K. Bakhtiari and F. Derikvand, Zn[(L)Proline]2 : An Efficient Catalyst for the Synthesis of Biologically Active Pyrano[2,3- d ]Pyrimidine Derivatives, Synth. Commun., 2010, 40(13), 1927–1931, DOI:10.1080/00397910903174390.
- G. M. Ziarani, S. Faramarzi, S. Asadi, A. Badiei, R. Bazl and M. Amanlou, Three-Component Synthesis of Pyrano[2,3-d]-Pyrimidine Dione Derivatives Facilitated by Sulfonic Acid Nanoporous Silica (SBA-Pr-SO3H) and Their Docking and Urease Inhibitory Activity, DARU J. Pharm. Sci., 2013, 21(1), 3, DOI:10.1186/2008-2231-21-3.
- R. Ramesh and A. Lalitha, Synthesis of Pyran Annulated Heterocyclic Scaffolds: A Highly Convenient Protocol Using Dimethylamine, Res. Chem. Intermed., 2015, 41(10), 8009–8017, DOI:10.1007/s11164-014-1873-5.
- R. Karimi-Chayjani, N. Daneshvar, H. Tajik and F. Shirini, Introduction of a New Magnetic Nanocatalyst as an Organic-inorganic Hybrid Framework for the Synthesis of Pyrano[2,3- d ]Pyrimidinone(Thione)s and Pyrido[2,3- d ]Pyrimidines, ChemistrySelect, 2019, 4(4), 1205–1213, DOI:10.1002/slct.201802916.
- S. Sajjadifar and Z. Gheisarzadeh, Isatin-SO3 H Coated on Amino Propyl Modified Magnetic Nanoparticles (Fe3 O4 @APTES@isatin-SO3 H) as a Recyclable Magnetic Nanoparticle for the Simple and Rapid Synthesis of Pyrano[2,3-d] Pyrimidines Derivatives, Appl. Organomet. Chem., 2019, 33(1), e4602, DOI:10.1002/aoc.4602.
- F. Mohamadpour, Supramolecular β-Cyclodextrin as a Biodegradable and Reusable Catalyst Promoted Environmentally Friendly Synthesis of Pyrano[2,3- d ]Pyrimidine Scaffolds via Tandem Knoevenagel–Michael–Cyclocondensation Reaction in Aqueous Media, Polycycl. Aromat. Compd., 2022, 42(5), 2805–2814, DOI:10.1080/10406638.2020.1852274.
- S. Sorkhabi, R. Mozafari and M. Ghadermazi, New Advances in Catalytic Performance of Erbium-folic Acid-coated CoFe2 O4 Complexes for Green One-pot Three-component Synthesis of Pyrano[2,3- d ]Pyrimidinone and Dihydropyrano[3,2- c ]Chromenes Compounds in Water, Appl. Organomet. Chem., 2021, 35(7), e6225, DOI:10.1002/aoc.6225.
- M. Nikpassand, L. Z. Fekri, R. S. Varma, L. Hassanzadi and F. S. Pashaki, Green Synthesis of Novel 5-Amino-Bispyrazole-4-Carbonitriles Using a Recyclable Fe3 O4 @SiO2 @vanillin@thioglycolic Acid Nano-Catalyst, RSC Adv., 2022, 12(2), 834–844, 10.1039/D1RA08001F.
- H. N. Akolkar, S. G. Dengale, K. K. Deshmukh, B. K. Karale, N. R. Darekar, V. M. Khedkar and M. H. Shaikh, Design, Synthesis and Biological Evaluation of Novel Furan & Thiophene Containing Pyrazolyl Pyrazolines as Antimalarial Agents, Polycycl. Aromat. Compd., 2022, 42(5), 1959–1971, DOI:10.1080/10406638.2020.1821231.
- F. Ghorbani, H. Kiyani and S. A. Pourmousavi, Facile and Expedient Synthesis of α,β-Unsaturated Isoxazol-5(4H)-Ones under Mild Conditions, Res. Chem. Intermed., 2020, 46(1), 943–959, DOI:10.1007/s11164-019-03999-7.
- S. Aslanpour and H. Kiyani, Rapid Synthesis of Fully Substituted Arylideneisoxazol-5(4H)-One Using Zinc Oxide Nanoparticles, Res. Chem. Intermed., 2023, 49(10), 4603–4619, DOI:10.1007/s11164-023-05059-7.
- A. Azimi-Roshan, H. Kiyani and M. Mamaghani, Green Pseudo-Seven Component Synthesis of Tris-Cyclohexylamino-Oxo-Benzene- 1,3,5-Tricarboxylates via Passerini Reaction under Aqueous Conditions, Curr. Org. Chem., 2025, 29(13), 1057–1065, DOI:10.2174/0113852728269992241030054239.
| This journal is © The Royal Society of Chemistry 2026 |