 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Navigating the Nanoverse: how emerging nanomaterials are transforming bioscience and society
Asia
Saorin
 a,
Ahmed
Subrati
a,
Ahmed
Subrati
 b,
Gloria
Saorin
b,
Gloria
Saorin
 c,
Giulia Yuri
Moscatiello
c,
Giulia Yuri
Moscatiello
 d,
Carmina
Natale
d,
Carmina
Natale
 d,
Roger
Bellido-Peralta
d,
Roger
Bellido-Peralta
 e,
Giulia
Cazzador
e,
Giulia
Cazzador
 f,
Mélina
Guérin
f,
Mélina
Guérin
 g,
Michele
Crozzolin
g,
Michele
Crozzolin
 f,
Montserrat
Vallet-Buisan
f,
Montserrat
Vallet-Buisan
 h,
Anita
Salmaso
h,
Anita
Salmaso
 d,
Benjamin
Punz
d,
Benjamin
Punz
 i,
Sara
Catalini
i,
Sara
Catalini
 jk,
Annalisa
Morelli
jk,
Annalisa
Morelli
 d and
Alberto
Martinez-Serra
d and
Alberto
Martinez-Serra
 *l
*l
aDepartment of Chemistry, Royal College of Surgeons in Ireland (RCSI), D02 YN77, Dublin, Ireland
bCenter for Cooperative Research in Biomaterials (CIC biomaGUNE), Basque Research and Technology Alliance (BRTA), Donostia, San Sebastian 20014, Spain
cDepartment of Chemistry, University of Basel, Mattenstrasse 24a, BPR-1096, 4058 Basel, Switzerland
dDepartment of Biochemistry and Molecular Pharmacology, Istituto di Ricerche Farmacologiche Mario Negri IRCCS, Via Mario Negri 2, 20156, Milan, Italy
eNational Graphene Institute, The University of Manchester, Manchester, UK
fDepartment of Molecular Sciences and Nanosystems, Ca' Foscari University of Venice, Via Torino 155 Mestre 30172, Venezia, Italy
gUniversity of Angers, INSERM, CNRS, MINT, SFR ICAT, 49000 Angers, France
hNuffield Department of Women's & Reproductive Health, Institute of Reproductive Sciences, University of Oxford, Oxford, OX3 9DU, UK
iDepartment of Biosciences & Medical Biology, Paris Lodron University of Salzburg, Hellbrunnerstrasse 34, 5020 Salzburg, Austria
jEuropean Laboratory for Non-Linear Spectroscopy, Via Nello Carrara 1, 50019 Sesto Fiorentino, Florence, Italy
kDepartment of Chemistry “Ugo Schiff”, University of Florence, Via della Lastruccia, 3-13, 50019 Sesto Fiorentino, Florence, Italy
lBarcelona Supercomputing Center (BSC), Plaça Eusebi Güell 1-3, 08034, Barcelona, Spain. E-mail: alberto.martinez@bsc.es
First published on 20th April 2026
Abstract
The rapid expansion of nanomaterials research has given rise to the concept of the Nanoverse, a multidisciplinary shared domain where increasingly sophisticated nanoscale architectures address societal challenges while introducing new safety and sustainability questions. This review writes up recent advances across diverse nanomaterial classes, including carbon-based nanomaterials, two-dimensional membranes, chromopeptide nanoarchitectures, microfluidic drug delivery platforms, extracellular vesicles, and antimicrobial nanomaterials. We trace the evolution of nanomaterial definitions, noting the widening scope to include particles up to micrometer size, and highlight how unique physicochemical properties govern biological uptake, intracellular fate, and environmental behavior. Using micro- and nanoplastics as a case study, we emphasize the need for realistic exposure assessments, biomolecular corona characterization, and ethical models for toxicological analysis such as C. elegans. By integrating material science, biotechnology, toxicology, and environmental science, we map the Nanoverse as both a technological frontier and a regulatory challenge. We believe that realizing its full potential requires harmonized characterization protocols, scalable manufacturing, and sustainable design principles to ensure that transformative nanomaterial applications advance responsibly from the labs to society.
1 Introduction
The European Union originally defined nanomaterials (NMs) in 2011 as natural, incidental, or manufactured materials containing isolated, aggregated, or agglomerated particles, where 50% or more of them in the number-based size distribution have one or more external dimensions between 1–100 nm.1 This definition was updated on June 10, 2022, introducing stricter criteria: at least 50% of particles must meet one of the following conditions – (i) one or more external dimensions between 1–100 nm; (ii) elongated shape (e.g., rod, fiber, tube) with two dimensions <1 nm and the third >100 nm; or (iii) plate-like shape with one dimension <1 nm and the other being >100 nm.2 This demonstrates that the concept of NMs is evolving along with the progress of NMs development and the increasing concern for their possible effect on human health and environment. Beside this definition, researchers nowadays commonly use the term NMs to refer to particles with size up to micrometer scale which can have a very wide range of composition, size and shape.3 This has led in 2015 to the use of the term ‘nanoParticle Zoo’ for describing the state of the field.4 However, in the last ten years, NMs and nanoparticles (NPs) have evolved and spread further moving from scientific curiosities into foundational elements of modern technology, shaping innovations across medicine, energy, electronics, and environmental science. Therefore, we enter what can be called the Nanoverse, a rapidly growing dimension where increasingly tailored NMs address societal challenges such as vaccine development and energy production, while also becoming widespread in everyday products, leading to their presence in the environment. Indeed, the unique physicochemical properties of NMs, arising from their size, surface area, quantum effects,5 and structural tunability,6 have unlocked previously unimaginable capabilities, from single-molecule biosensing to ultra-efficient energy storage.7,8 Emerging manufacturing strategies now combine precision molecular design with scalable, high-throughput production, focusing on multifunctional hybrids and engineered materials capable of addressing industrial, environmental, and biomedical challenges.9–13Among the many classes of NMs, a broad distinction can be made between organic, inorganic, and multifunctional nanomaterials, each offering unique properties and application potential (Fig. 1). Inorganic NMs encompass a wide range of materials, among which carbon-based NMs (CBNs) such as graphene, carbon nanotubes, and carbon nanodots have demonstrated extraordinary versatility due to their exceptional electrical conductivity, mechanical strength, and functionalization potential.14 Simultaneously, two-dimensional (2D) materials have opened new frontiers in molecular separation,15 enabling precision filtration16 and environmentally responsive membranes.17–19 Organic NMs, including lipid- or polymer-based systems, have attracted increasing attention due to their biocompatibility and capacity for functional design. For example, chromopeptide nanoarchitectures, i.e. self-assembled structures combining optical and structural functionality, have emerged as intelligent platforms for light-driven diagnostics and therapies.20 Multifunctional nanomaterials are engineered systems that combine two or more distinct physicochemical, biological, or technological functionalities within a single nanoscale platform.21,22 Examples within the biomedical field include inhalable nanoformulations for respiratory and cardiovascular therapies,23,24 macrophage-targeting smart nanocarriers for precision medicine,25,26 extracellular vesicles as naturally derived platforms for targeted drug delivery,27–29 engineered NMs with antimicrobial properties capable of circumventing traditional resistance pathways,30 as well as the development of nanostructured theranostic platforms and wearable biosensors, offering enhanced sensitivity and specificity for real-time, personalized healthcare monitoring.31 All this influenced by technological advancements, such as the development of microfluidic platforms that enable continuous, scalable, and precisely controlled fabrication of nanomedicines with clinical relevance.32,33 However, the rise of these technologies brings along the critical need for rigorous toxicological assessment,34 including advanced characterization of NMs under realistic environmental and biological conditions,35,36 and investigations into the interactions of micro- and nanoplastics with living systems.37,38 Organisms like Caenorhabditis elegans (C. elegans) are increasingly recognized as ethically and biologically relevant in vivo models for evaluating the safety of NMs, offering insight into long-term exposure effects and sub-toxic responses that remain undetectable in simpler systems.39
 | ||
| Fig. 1 Classification of common NMs across different research fields by molecule nature: organic, inorganic, and multicomponent. Created with BioRender.com (https://www.biorender.com/). | ||
Despite this momentum, the full potential of NMs remains largely untapped. Key challenges persist, including the lack of standardized descriptors, insufficient cross-domain data integration, and limited scalability of many promising lab-scale innovations.40–43 As we map the Nanoverse, it becomes essential not to only catalogue the materials and their functions but also to understand the systemic interconnections that allow nanotechnology to transform society at scale.
This review provides a cross-sectional analysis of emerging NMs and technology, focusing on their novel architectures, mechanisms of action, and translational applications. By synthesizing insights across material science, biotechnology, and toxicology, we aim to offer a comprehensive view of how these nano-scale constructs are driving macro-scale change—and what lies ahead in the continued exploration of the Nanoverse.
2 Transforming nanomaterial development for real-world impact
NM innovation is accelerating through the fusion of precision design and advanced manufacturing. Emerging approaches leverage molecular-level control, structural customization, and hybrid architectures to unlock new functionalities while enabling seamless translation from laboratory synthesis to industrial production. The next frontier lies in integrating adaptive design strategies with continuous, high throughput manufacturing platforms, paving the way for NMs tailored to meet the evolving demands of technology, medicine, and sustainable industries.2.1 From novel carbon nanomaterials to breakthrough nanotechnologies
Carbon has garnered the attention of various technological and biomedical fields with its exceptional chemical, physical, and electronic properties. Nanoscaling carbon has opened applied research horizons for abovementioned fields by introduction of carbon-based NMs (CBNs) such as graphene, fullerene, carbon nanotube, carbon nanodot, carbon onion, carbon nanodisk, carbon nanofiber, carbon quantum dot, activated carbon, nanodiamond, and carbon nanoribbon, as shown in Fig. 2(a and b).44–50 Superior electron mobility, tunable surface chemistry, high tensile strength, exceptional thermal and chemical stability, high porosity, tunable density and nature of defects, high surface area, and extremely low density are some of the properties that characterize CBNs and drive applied research in technological and biomedical fields. CBNs have been implemented in adsorption,49 drug delivery,51 wound healing,52 biomedical scaffolds,53 tissue engineering,54 prosthesis engineering,55 biosensing,56 photodynamic therapy,57 bioimaging,47 anticancer treatment,51 transistors,58 energy storage – such as batteries (lithium-ion batteries, lithium–sulfur ion batteries),59 (super-)capacitors,60 fuel cells61 and photovoltaics,62 quantum computing,63 spintronics,64 aviation fabrications,65 sensing,66 catalysis,67 and coating.68 Mention must be made of the possibility of discovery of new (sub-)classes of CBNs. Defect engineering in all CBNs holds great potentials for corresponding applications and is direct consequence of the tunable chemistry of CBNs.69 For instance, defective oxygen-functionalized domains were spatially clustered by action of mild thermal annealing (80 °C) of nanobodies-coupled graphene oxide to enhance cell capture from whole blood, as illustrated in Fig. 2(c).70 The ability of oxygen functionalities to migrate and cluster holds great potential not only in biological applications, but also in energy applications.71 Shaping the sp2 percolation network in all CBNs is theoretically predicted to significantly alter electronic properties, as demonstrated in Fig. 2(d). Defects in carbon materials, such as that in Fig. 2(e), can spatially and chemically localize sp2 bonds.72 Oxygen-clustering was evidenced to play a pivotal role in defining nitrogen doping configurations, which essentially impacts redox capabilities of nitrogen-doped carbon derivatives.71 Therefore, one can envisage that functionalization of CBNs with heteroatoms is not analogous to attaching stationary atoms. Heteroatoms, particularly oxygen, are mobile and their distribution can be tuned by action of mild thermal treatment. This step, often a post-processing step, is simple yet very deterministic and reflects orders of magnitude of potential new insights into already existing research utilizing CBNs. Thereby, there are still unexplored possibilities for useful and safe applications of CBNs. While many CBNs have been explored, their full potential remains untapped due to a lack of systematic archival reporting of their properties. The main obfuscated challenge from novel carbon NMs to breakthrough nanotechnologies is the establishment of universally accepted descriptors, numerical descriptors not only for physico-chemical characteristics, but also for application related parameters. Emerging efforts for grouping and read-across are already in place for few candidate CBNs.73 However, there is a pressing need for similar efforts for all explored CBNs. Established platforms that archive peer-reviewed research focusing on novel CBNs with corresponding physico-chemical descriptors and associated application related characteristics will facilitate human-readable comparisons as well as hastened accessibility, improved visibility, and recognition.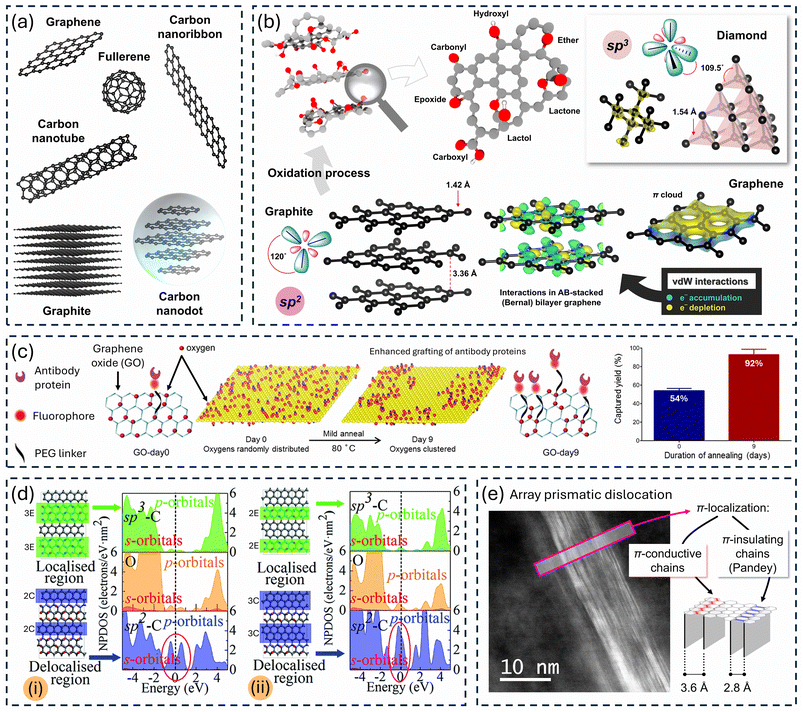 | ||
| Fig. 2 (a) Few examples of carbon-based materials. Structures were constructed from CIF files obtained from the Crystallography Open Database (COD)74–76 using Vesta.77 (b) Structures of graphene, graphite, graphite oxide, and diamond. Oxygen functional groups result from the oxidation of graphite endowing the layer material with multi-node hydrogen bonding. van der Waals (vdW) interactions are visualized for the graphene bilayer. Reproduced from Subrati et al.78 with permission from Wiley-VCH, Copyright (2024). (c) Oxygen-clustering in graphene oxide drives phase transformation separating graphitic sp2 and oxidized sp3 domains reshaping the percolation network on the carbon backbone reflected in reduced resistance while preserving the oxygen content. Antibody grafting is augmented in the oxygen-clustered derivative, and this leads to an almost two-fold improvement in cell capture efficiency. Reproduced from Bardhan et al.70 with permission from American Chemical Society, Copyright (2017). (d) Theoretical investigation on confinement of π-conjugated domains within oxidized domains on two models reveal strong impact on electronic nature of partially oxidized graphene. The two models vary in the width of the chains of epoxy groups (E, green) and the π-conjugated zigzag carbon chains (C, blue). DFT-calculated normalised projected density of states of partially oxidized graphene models: (i) semiconductor E3C2 and (ii) semi-metal E2C3. The carbon atoms enclosed in the blue and green rectangles contain delocalized and localized electrons, respectively. The partially delocalized sp2 carbons make up most of the electronic states near the Fermi level. Such states contribute immensely to quantum capacitance. These states facilitate partial charge transfer between electrolyte ions and redox centers via quantum capacitance leading to specific ionic adsorption and consequently fast reversible faradaic processes in partially oxidized graphene. Reproduced from Li et al.79 with permission from The Royal Society of Chemistry, Copyright (2017). (e) Experimental evidence of defects confining chains of electron delocalization (localized π-orbitals). Dark field – scanning transmission electron microscopy image of an array prismatic dislocation found in carbon ravioli of pyrolyzed graphite oxide framework. Abundance of such defects was increased by heating framework at 80 °C for 24 h in air prior to pyrolysis. This confinement of electron delocalizations augmented redox activity sizeably. Reproduced from Subrati et al.71 with permission from Elsevier, Copyright (2024). | ||
2.2 2D materials membrane design for molecular sieving purposes
Current environmental issues range from atmospheric pollution to water contamination. There seem to be many solutions to these problems, but nanotechnology is constantly in the spotlight, more precisely 2D materials and their applications.80 There are multiple ways to use bidimensional structures to achieve different purposes such as drug delivery, anti-rust coatings or liquid filtration.81,82 The latter was discovered upon realising that self-assembled graphene oxide (GO) membranes were permeable to water but able to block other elements such as argon.83 In the past decade, efforts have been made to expand the range of materials that allow self-assembling membrane manufacture, e.g. graphene, graphene-related 2D materials (GR2M, like graphene oxide), transition metal dichalcogenides (TMD), MXenes, clays, MOFs, COFs and 2D heterostructures (alternate stacking of different 2D materials). A wide availability of materials translates into an also wide range of properties, which allows to devise all kinds of filtration and mixture separation procedures.84–86 Taking advantage of its laminar structure, stability and self-assembly properties, they can be used as filters to purify water,87 separate mixtures88 and even as molecular sieves.89 The performance and properties of membranes paired up with its facile manufacture allowed to scale up processes and are currently being tested for industrial applications.90 However, much research remains to be done, for example regarding molecular sieving properties. Membranes made from 2D materials assemble themselves by piling up nanoflakes, the filo pastry of nanotechnology, so to say. These structures have the capability to present different interlayer spaces (the distance between each layer),91 which in turn regulates the size of the molecules that can permeate. For example, water molecules flow with no constraint through graphene oxide (GO) structures, while contaminants can potentially be retained. However, what if we wanted water to be retained, while extracting larger molecules? Or how about extracting certain elements from a multi-component mixture? The solution seems to point towards polymers and how to appropriately fuse them with bidimensional materials. An easy and promising method is to cross-link polymeric chains to the layers of the membranes, thus keeping the laminar structure intact (Fig. 3).92 Using this assembly, we can force an interplay between the properties of both materials, obtaining new phenomena that are non-characteristic to any of them separately. This new type of structure could be the next step in the field of nanofluidics, as we can selectively tune the properties we desire in the resulting outcome. Composite membranes could be the answer to greener and more efficient biofuel manufacturing, less costly water filtering procedures or advances in the food industry. Be as it may, nanotechnology (and self-assembled membranes in this particular case) can lead the field further towards developments that make a difference. | ||
| Fig. 3 Schematics of a self-assembled 2D material membrane. Embedding polymeric chains between the nanoflakes conditions the properties of the final structure, e.g., the interlayer space. | ||
2.3 Chromopeptide nanoarchitectures for the development of next-generation photonic devices
The emergence of NMs has marked a significant milestone in the development of various scientific fields, particularly medicine93 and photonics.94 Their impact stems from unique chemical and physical properties that arise at the nanoscale,95 enabling the creation of functional materials with capabilities not present in their monomeric forms.96 Within this context, supramolecular chemistry has proven to be a powerful bottom-up strategy for constructing highly organized, dynamic nanoarchitectures from molecular building blocks.97–99 These systems often exhibit emergent properties such as optical, magnetic, and electronic, resulting from precise molecular organization rather than individual component features.97–99 Notably, nanostructures assembled from small molecules can show intrinsic biological activity, even without incorporating additional bioactive agents.100 Among these, self-assembling peptides have attracted considerable interest for biomedical applications due to their ability to encode diverse bioactive functions.95 However, high production and purification costs often limit their practical use, prompting the search for short, cost-effective sequences that still retain self-assembling capacity.95 A prominent example is derived from amyloid beta (Aβ) peptide, associated with Alzheimer's disease. Efforts to identify minimal sequences responsible for Aβ′s self-assembly have highlighted the pentapeptide Lys-Leu-Val-Phe-Phe, recognized as the shortest natural L-peptide capable of forming amyloids in physiological conditions without modification.101 Earlier, in 2003, a breakthrough identified diphenylalanine (Phe–Phe) as a minimal motif that forms stable nanotubes in aqueous media.102 This foundational discovery led to the development of a new class of minimalistic peptide-based nanostructures.103,104 Advancing from this groundwork, researchers have begun exploring hybrid systems that combine peptide self-assembly with photonic functionality. At this intersection lie chromopeptide nanoarchitectures, i.e. self-assembled structures composed of peptides conjugated with chromophores. These materials offer molecular precision, tunable morphologies, and intrinsic biocompatibility, along with the capacity to tailor light–matter interactions.20 When chromophores, molecules that absorb and emit light, are covalently linked to peptides, the resulting assemblies acquire both optical and structural roles.105 This combination enables the engineering of photonic NMs with versatile properties, including tunable spectral features,106 anisotropic emission,107 and nonlinear optical responses.108 These features position chromopeptides as promising candidates for use in photonic crystals, waveguides, and light-harvesting platforms.109 Crucially, the peptide sequence and hydrophobicity can be modulated to control the spacing and orientation of chromophores, thereby adjusting electronic coupling and optical bandgaps. Such molecular-level tunability is essential for the development of next-generation photonic devices. One particularly promising characteristic of chromopeptides is their responsiveness to environmental stimuli. Variations in pH, temperature, or ionic strength can alter their optical signatures, such as fluorescence and refractive index, making them well-suited for biosensing and real-time monitoring applications.110 Beyond sensing, these nanoarchitectures also offer significant potential for energy management by mimicking natural photosynthetic processes. Their ability to facilitate Förster resonance energy transfer (FRET) and exciton migration opens new avenues for artificial photosynthesis and related light-driven technologies.20 Despite these promising attributes, several challenges persist. Key among them is achieving long-range structural order, enhancing quantum efficiency, and maintaining stability under operational conditions. Overcoming these hurdles will require integrated efforts across disciplines such as peptide chemistry, material science, and photonics. Advanced tools, including computational modelling and high-resolution characterization, will be critical to unlocking the full potential of chromopeptide nanoarchitectures.1113 Innovating nanomedicine for a healthier tomorrow
Nanomedicine is a growing field which includes imaging, drug delivery, diagnosis, vaccine development, nanoscale scaffolds for tissue engineering and it has obtained consistent investments both from private and public authorities.112 Around 80 nano-based pharmaceutical products have been approved by EMA and FDA since 1989.113,114 Most of these approved products are organic particles, namely nanocrystals, lipid-based NPs, polymer-based NPs, dendrimer based NPs, protein based NPs, while only 9 products are based on inorganic NPs, of which 6 to 9 are iron NPs for iron deficiency treatment.115 More recent advances in this field are biologically derived approaches, such as the use of extracellular vesicles or the development of smart nanocarriers which exploit the function of living cells, harnessing their natural functions for targeted delivery, therapeutic modulation, and improved biocompatibility. However, inorganic NPs may play a crucial role in areas where other approaches fall short, such as the development of novel antimicrobial systems, the development of biosensors, medical and industrial imaging, and as active transducers in photodynamic therapies for cancer treatment. In this section we report several cutting-edge examples of nanomedicine applications, from precision delivery to disease treatment and health monitoring, focusing in drug delivery, innovative treatments, disease detection, the role of microfluidics and further technological applications.3.1 Nanoformulations for pulmonary drug delivery
The emergence of the COVID-19 pandemic incentivized the rapid development and implementation of nanoformulations, particularly as innovative platforms for vaccine delivery and antiviral therapeutics. The success of COVID-19 vaccines highlighted nanotechnology's role as a versatile and promising approach for both preventive medicine and the therapeutic management of infectious diseases.116–118 Recently, pulmonary diseases have gained increasing prominence in biomedical research, not only because of the impact of the pandemic, but also due to the global burden of lung cancer, cystic fibrosis and bacterial infections.119–124 Conventional treatments for respiratory disorders are mainly administered systemically, often requiring high doses to achieve effective local drug concentration. However, systemic administration can cause off-target effects and systemic toxicity, limiting therapeutic efficacy and patient compliance.125 Yet, pulmonary and nasal routes of administration for nanomedicines represent a great opportunity to decrease the drug administrated dose, increase the local concentration on the disease site and decreasing the healthy organs exposure.122,123,126,127The lungs, together with the heart, are vital organs working in close coordination to ensure oxygenation of the body through blood circulation. The first ensures the gas exchanges between air and blood, while the second is the pump allowing the blood to circulate in the body. The key to this natural connection between the environment and the systemic circulation is the high vascularization of the lungs as well as the alveoli thin epithelium, also called the air–blood barrier, and the direct proximity of the heart with lungs.128–130 Not only can gas cross the air–blood barrier, but studies have also shown that nano- and ultrafine pollution particles can reach the heart via oxygenated blood from the lungs, as well as inhaled NMs.131,132 This gateway to systemic circulation can be turned into an advantage for medical research, offering a strategic entry point for delivering nanomedicines to the body and particularly for targeting the heart.130,133 Traditional drug delivery routes face significant challenges: oral administration is hindered by gastrointestinal barriers, while intravenous or intramyocardial injections are invasive and carry risks of side effects and myocardial injury.128,129 Although intranasal administration provides a more direct route than systemic administration, it still requires passage through multiple anatomical barriers and defence mechanisms within the respiratory tract.134 NPs offer a promising strategy to protect and selectively deliver drugs to pulmonary tissues, enhancing therapeutic outcomes. The lungs' large surface area and rich blood supply enable the absorption of NPs, allowing fast drug accumulation, with reduced systemic exposure and fewer side effects129,135 (Fig. 4).
 | ||
| Fig. 4 (a) TEM image of lipid NPs used in the mRNA-1273 platform developed by Moderna (Cambridge, MA, USA). Reproduced from Fongaro et al.142 with permission from Elsevier, Copyright (2023). (b) Schematic of LNP structure composed of lipids encapsulating therapeutic cargo. (c) Illustration of inhalable NPs for nasal, lung or systemic administration, highlighting a key challenge: how different formulations interact with pulmonary fluids like mucus and surfactants as well as macrophages, influencing the uptake. | ||
NPs can vectorize both conventional drugs and biomolecules such as siRNA, miRNA, and other nucleic acids, which are otherwise unstable and difficult to administer.136 Nanocarriers protect these labile agents from degradation, permitting their therapeutic use in both infectious diseases and genetic disorders. Additionally, functionalizing NPs with specific proteins or ligands facilitates targeted interaction and drug release at specific cells or tissues, advancing personalized medicine that addresses individual patient needs.137,138 Aside from pulmonary diseases, current treatments for heart diseases suffer from poor cardiac specificity and low bioavailability. They only tend to accumulate slightly in cardiac tissue and exhibit short retention times, requiring high doses that lead to toxicity and side effects. Designing and administering inhaled nanomedicines could help overcome these limitations, offering a more targeted, efficient, and safer therapeutic strategy for cardiovascular diseases.128,129 Indeed, nanomedicines offer unique physicochemical properties, compatible with alveolar deposition and uptake through the air–blood barrier. Their surfaces can be functionalized, for example, with cardiac-targeting peptides to enhance heart-specific accumulation.128,129,131,133 Several studies are currently exploring this strategy, using polymeric, liposomal, inorganic, and especially calcium-phosphate NPs, which are currently among the most studied.128,131,139
Looking ahead, future research is focused on the development of smart nanocarriers capable of recognizing biochemical changes, cellular alterations, or tumour markers, and consequently triggering targeted drug release.140 In this context, careful selection of NP composition is essential to ensure biocompatibility and avoid pulmonary toxicity. Inhaled nanomedicine represents a promising approach to overcome the limitations of current cardiovascular therapies by improving specificity, efficacy, and safety. Nanotechnology presents vast opportunities in the treatment of pulmonary diseases; however, these are accompanied by significant challenges and unanswered questions that must be addressed to ensure both safety and efficacy.141 It is crucial to understand how NPs interact within complex biological systems to avoid their recognition as harmful foreign materials or undesired accumulation that could trigger inflammatory or degenerative responses. Therefore, the development of new nanoformulations should be carried out by multidisciplinary teams capable of thoroughly characterizing NPs from both physicochemical and biological perspectives.
3.2 Smart nanocarriers and macrophages for precision medicine
Advances in technology and medicine generated the ‘smart’ nanocarriers, able to bypass physiological barriers with high accuracy, selectivity, and sensitivity. They can exploit the unique characteristics of the biological environment, targeting specific factors involved in the onset and progression of diseases, such as macrophages, and releasing drugs in a controlled manner.143,144Macrophages are immune cells that play a crucial role in surveillance, inflammation, and tissue repair. They are part of the mononuclear phagocyte system (MPS), which also includes cells like monocytes. Known for their ability to ingest foreign material, including pathogens, debris, and NPs, macrophages represent one of the major challenges in nanomedicine.145 When nanocarriers are introduced into the organism, these cells often recognize and engulf them through phagocytosis. The clearance can reduce their effectiveness and prevent them from reaching targeted areas, such as tumors or inflamed tissue.146 This limitation may present an opportunity in precision medicine.
For instance, macrophages can be targeted as delivery vehicles for nanocarriers, releasing therapeutic agents directly to disease sites during their infiltration. Another strategy is represented by macrophage polarization into the M1 phenotype to promote immune activation against diseases like cancer, or into the M2 phenotype for promoting tissue repair and wound healing in conditions like inflammatory diseases.147 They are designed with surface modifications (like ligands or antibodies) that recognize specific receptors on target cells. When these nanocarriers reach their target, they release the cargo in response to specific stimuli (pH, temperature, light, or enzyme activity), ensuring that the drug acts only at the intended site, reducing systemic side effects.148 Smart nanocarriers can not only deliver drugs, but also agents that modulate macrophage activity, promoting the desired immune response.149 An emerging strategy involves using RNA molecules to treat genetic disorders and cancer. Nanocarriers can be used to deliver these unstable molecules to cells, including macrophages, where they can modulate gene expression to reprogram immune responses or fight disease150 (Fig. 5). Their unique properties and versatility make them applicable in various biomedical fields, for treating cancer,149 chronic inflammation,151 neurological disorders,152 and promoting tissue regeneration.153
Smart nanocarriers hold potential for precision medicine. By exploiting macrophages' natural functions, these systems enhance drug delivery, modulate immune responses, and provide effective treatments for various disorders.154 The integration of artificial intelligence and machine learning could allow the design of more responsive, adaptive nanocarriers specific for individual patient profiles. Moreover, advances in RNA therapeutics and gene editing may lead to even more precise modulation of macrophage function, unlocking new possibilities for treating genetic and immune-related disorders. Innovative delivery platforms capable of multi-functional payload release, real-time in vivo monitoring, or self-regulating therapeutic responses are emerging. As research improves and interdisciplinary collaborations grow, smart nanocarriers may become central to the next generation of personalized therapies.
3.3 Extracellular vesicles as a next-generation therapeutic platform
Extracellular vesicles (EVs) are membrane-bound NPs naturally released by cells that carry proteins, lipids, and nucleic acids from their cell of origin. Once thought to be cellular waste, EVs are now recognized as key mediators of intercellular communication, and interest in their therapeutic potential has grown rapidly.155 Their role in diverse physiological and pathological processes, including cancer, neurodegeneration, and inflammation, has made EVs attractive as both biomarkers and therapeutic delivery systems. Several characteristics make EVs especially well-suited for drug delivery: they are stable in circulation, can naturally target specific cell types, and can be engineered to deliver a variety of therapeutic molecules, from small RNAs to proteins and small-molecule drugs.156,157 Furthermore, unlike synthetic NPs, EVs have an inherent ability to evade immune detection, cross biological barriers (including the blood–brain barrier), and deliver complex bioactive molecules to target tissues.158 And because they are derived from cells, they offer a level of biocompatibility and safety that is hard to match with current delivery systems. Their potential uses are remarkably broad: from treating cancer (by delivering drugs or serving as cancer vaccines), to regenerating injured tissues (by delivering growth factors or RNAs), to modulating the immune system in autoimmune diseases, to even crossing into the brain to treat neurological conditions.Nevertheless, despite rapid progress, several hurdles remain before EV therapies can become mainstream. Scalable production is a major bottleneck: current isolation methods are not easily adapted to clinical-grade, large-scale manufacturing. EV heterogeneity also complicates characterization and quality control, making it difficult to link therapeutic effects to specific cargo. The field still lacks standardized protocols for isolation, dosing, and potency assessment, though guidelines like MISEV2023 are helping address this.159 Regulatory frameworks are also evolving; EVs do not fit neatly into existing drug categories, and approval pathways remain unclear. Finally, biodistribution and targeting remain challenging, with many EVs accumulating in off-target organs. Addressing these challenges through better analytics, standardization, and regulatory dialogue will be essential to advancing EVs as a clinically validated delivery platform.
Even so, progress is well underway, and in recent years EV-based therapeutics have begun to move from the bench to the clinic. Most preclinical studies have focused on inflammation, cancer, tissue regeneration, and neurological disorders, where EVs, often from stem or immune cells, are used to deliver bioactive molecules. Clinical translation, however, remains in its early stages. As of 2024, only a few dozen clinical trials have tested EV-based therapies in humans.160 These have mostly been small-scale pilot studies, with wide variation in disease targets, EV sources, doses, and administration routes – making it difficult to draw sound conclusions or compare outcomes across trials. Still, initial results are promising: a recent systematic review and meta-analysis reported a very low incidence of serious adverse events (∼0.7%), supporting the idea that EVs are generally safe.161 Such outcomes, while preliminary, reinforce the view that EVs hold genuine therapeutic promise. In the coming years, larger Phase II/III trails with more uniform protocols will be key to determining how EVs are effective across different diseases.
3.4 Nanomaterials as broad-spectrum antimicrobial alternatives in the fight against infectious diseases
The increasing prevalence of infectious diseases, coupled with the accelerating threat of antimicrobial resistance (AMR), has prompted urgent global efforts to identify effective alternatives to conventional antibiotics. The therapeutic efficacy of many frontline antibiotics is rapidly diminishing, and clinicians are often left with limited options to contain the spread of resistant pathogens. In this context, engineered NMs have emerged as promising broad-spectrum antimicrobial agents with versatile applicability beyond the scope of traditional antibiotics.162 Both organic and inorganic NPs exhibit antimicrobial properties, albeit with distinct advantages and limitations. Organic nanomaterials, most notably CBNs, are primarily valued for their high biocompatibility, structural versatility, and strong potential as carrier platforms, while still expressing intrinsic antimicrobial activity. In addition, their biodegradability is considered advantageous in mitigating long-term environmental concerns. However, for applications outside of biological systems, industrial development has predominantly focused on inorganic NPs. A key reason for this preference is the central role of ion release as a highly potent antimicrobial mode of action, which is characteristic of many inorganic NMs but is not a dominant mechanism in organic nanomaterials.163 Metallic and metal oxide NPs, in particular, exhibit intrinsic antimicrobial properties through multiple mechanisms of action, including ion release, disruption of microbial membranes, and the generation of reactive oxygen species (ROS).164 These effects depend primarily on three factors: (i) the chemical composition of the NM, (ii) its physicochemical properties (e.g., size, surface charge, solubility165), and (iii) surface modifications or functionalizations.166 Such multifaceted mechanisms reduce the likelihood of resistance development and can be readily tailored by modifying NP characteristics, enabling precise design strategies for targeted applications. A key advantage of NMs over conventional antibiotics lies in their broad-spectrum activity. While antibiotics are typically species-specific and limited to bacterial infections, NPs such as ZnO, Ag, and CuO have demonstrated efficacy against a wide range of pathogens, including bacteria, fungi, and viruses.167 This expanded spectrum of activity makes them particularly attractive as preventive agents in both clinical and non-clinical environments. Given their tunability, NMs are increasingly being considered for incorporation into everyday objects to mitigate the spread of infections. Potential applications include embedding antimicrobial coatings into high-contact surfaces such as mobile phones, keyboards, handrails, and doorknobs, where pathogen transmission is common. In healthcare settings, surfaces of medical devices and workstations could be coated with biocidal NMs to minimize nosocomial infections. The scalability and cost-effectiveness of such preventive measures could significantly reduce the incidence of infectious diseases in public and clinical environments. Beyond healthcare and consumer safety, NMs are also being explored in agricultural and environmental contexts. For example, nano-enabled crop coatings could reduce fungal infestations and mitigate crop losses, while the integration of antimicrobial NMs into wastewater treatment systems offers a route to inactivate pathogenic organisms and improve sanitation.168 Therefore, antimicrobial NMs represent a versatile and potent platform with far-reaching potential to combat pathogens across sectors. Their unique physicochemical properties, tunable antimicrobial activity, and broad applicability position them as promising candidates in a multi-pronged strategy to address the global challenge of infectious diseases and AMR.3.5 Nanoparticles for radiological theranostic
In the field of medical diagnostics, the detection of ionizing radiation (essential for CT and PET imaging) faces a constant demand for higher sensitivity, better resolution, and easier processability.169 These requirements rely critically on scintillators, materials that convert high-energy radiation into visible light for signal detection. However, conventional bulk scintillators often involve trade-offs between performance, cost, and fabrication complexity.170 Similarly, in Photodynamic Therapy (PDT), limited light penetration hinders the treatment of deep-seated tumors. Nanomaterials address these challenges through X-ray Photodynamic Therapy (X-PDT), which utilizes X-rays to reach deep tissues.171Unlike bulk scintillators, which require expensive high-temperature procedures like the Czochralski method and suffer from light scattering, nanoscintillators are produced via cost-effective wet chemical methods.170 Their nanoscale nature enables tunable emissions (from UV to NIR-II) and custom core–shell designs to optimize performance.172 Among tunable luminescent platforms, the NaREF4 (RE = rare-earth element) system stands out as an example of structural and optical customization. Furthermore, quantum confinement effects allow for enhanced light yield and ultrafast scintillation kinetics, with decay times reaching the sub-nanosecond regime173 due to modified electronic relaxation processes.174
Nanoscintillators also improve spatial resolution: while excited regions in bulk materials extend over tens of micrometers, nanoscintillators reduce this to a few micrometers, allowing more precise localization of scintillation events.174 Additionally, heterostructured approaches combine complementary properties—such as high atomic number for attenuation and fast response for timing—to optimize detector performance.173
In X-PDT, scintillating nanoparticles act as internal light sources, bridging the gap between deep X-ray penetration and photosensitizer activation.175 High-Z elements within these particles, such as LnF3, amplify X-ray absorption and ROS generation.176 This enhancement allows effective tumor control with significantly lower radiation doses (e.g., 4 Gy with nanocarriers vs. 12 Gy for radiotherapy alone), reducing systemic side effects.
Nanocarriers further improve photosensitizer stability and delivery through the enhanced permeability and retention (EPR) effect or active targeting with specific ligands.176 Encapsulation ensures controlled release (a remarkable example are Gd2O2S:Tb nanocapsules) and protects agents from degradation.177 Finally, incorporating oxygen-generating nanoagents (e.g., catalase-loaded materials) addresses tumor hypoxia.178 Collectively, nanocarriers transform X-PDT into a high-precision modality, maximizing therapeutic impact while minimizing toxicity.
3.6 Nanomaterials for biosensors monitoring of health
The increasing research interest in personalized medicine has led wearable sensors to be great candidates to improve health care and continuous control of the quality of life, giving information about the effectiveness of the care, an early detection of disease and real-time monitoring.179,180 Since wearable biosensors must be selective and sensible, NMs are a good choice to enhance interaction with biological targets, thanks to their high surface-to-volume ratio, improving sensitivity in the detection.A wide range of NMs – including carbon-based structures (graphene,181 graphene oxide,182 carbon nanotubes,183 fullerene184), metal and metal oxide NPs (gold,185 platinum,186 silver,187 copper188), and metal–organic frameworks189 – has been integrated into wearable devices. For instance, graphene-based sensors have demonstrated high sensitivity and flexibility, making them ideal for integration into epidermal patches190 and smart textiles.191 Gold NPs and quantum dots are widely used for optical sensing due to their tunable fluorescence and plasmonic properties.192
Moreover, the functionalization193 of NMs with biomolecules (e.g., enzymes, aptamers, antibodies, DNAzyme194) allows for high specificity toward target analytes, enabling personalized and precision medicine approaches (Fig. 6). Recent advances in nanofabrication195 and microfluidics196 have also facilitated the development of multiplexed platforms capable of monitoring multiple biomarkers simultaneously, improving the accuracy and reliability of health assessments.
 | ||
| Fig. 6 Schematic illustration of wearable biosensors based on nanomaterials and their functionalization with common bioreceptors. Created with BioRender.com (https://www.biorender.com/). | ||
Despite these advancements, several challenges remain before large-scale clinical translation can be achieved.197–199 These include long-term biocompatibility, sustainability and device stability. Nonetheless, the integration of NMs with wireless communication systems and artificial intelligence200 is opening new avenues for continuous and remote patient monitoring, with the potential to shift healthcare from reactive to proactive models.
3.7 Microfluidics in drug delivery: bridging precision and innovation
Microfluidics is a technology that enables precise manipulation and control of fluid flow within channels characterized by at least one dimension smaller than 1 µm. This miniaturization allows access to fluidic behaviours not observable at the macroscale, making microfluidics a highly versatile platform. It is widely applied across diverse disciplines, including fluid dynamics, synthetic and analytical chemistry, biology, and medicine. Key applications include drug toxicity assessment, the development and characterization of drug delivery systems, regenerative medicine, and single-cell analysis.201 Microfluidics presents significant advantages in the development of drug delivery systems (DDS), primarily due to its miniaturized scale, which reduces material consumption. Its precise control over experimental conditions enables the fabrication of particles with clinically relevant properties, eliminating the need for additional post-production processing (such as extrusion) reducing the risk of cargo unloading or degradation.202 Furthermore, microfluidic platforms allow for the rapid and systematic testing of multiple parameters with processes that are inherently scale-independent. Therefore, microfluidics enables rapid optimization of DDS characteristics using a design-of-experiments approach, followed by the seamless translation of formulation and processing parameters from laboratory development to industrial scale with a continuous process. Continuous manufacturing offers several general advantages, including reduced capital expenditure, a smaller manufacturing footprint, and lower cost of goods. In the context of DDS development, continuous manufacturing enables the integration of critical processing steps such as purification, concentration adjustment, and sterilization, directly within the continuous flow, enhancing efficiency and process control.203 These advantages have been proved by Pfizer's mRNA COVID-19 vaccine production. Indeed, the vaccine's lipid NPs, encapsulating the mRNA, were produced using an impingement jet mixer (IJM). IJMs are passive mixing devices, firstly reported in 2003,204 known for their ability to achieve rapid and efficient mixing at microscale dimensions while enabling high throughput205 (schematic workflow in Fig. 7). Instead of scaling up mixer size, Pfizer scaled out by parallelizing 100 IJMs, enabling continuous synthesis and achieving a production rate of 100 million doses per month. Automation ensured precise control over flow and pressure.32 This approach demonstrated the scalability, efficiency, and versatility of microfluidics in producing nanomedicines for large-scale drug delivery. Various approaches can be employed to develop DDS using microfluidics, primarily differing in the method of flow mixing used to induce particle self-assembly. Microfluidic technologies for DDS development have largely focused on the production of organic NPs, particularly lipid-based nanomedicines. This class of NPs has seen substantial growth, as evidenced by a rising number of clinical trials and FDA approvals and now represents a central component of modern therapeutic strategies.206 New microfluidic systems are continually being developed to produce DDS, increasingly leveraging advancements in 3D printing technologies. However, these innovations to reach industrial application, must undergo rigorous regulatory evaluation and receive approval from agencies such as the European Medicines Agency (EMA) or the Food and Drug Administration (FDA). This process, particularly for novel microfluidic devices, demands extensive clinical testing and demonstration of long-term safety and efficacy, which can be both time-consuming and costly.207 Conversely, commercially available off-the-shelf microfluidic kits, designed with scalability in mind, offer faster route to therapeutic development. Their production flexibility makes them especially valuable for personalized medicine, enabling patient-specific dosing and rapid adaptation to individual treatment needs.208 | ||
| Fig. 7 Schematic representation of nanoparticle preparation for drug delivery using an impingement jet mixer. | ||
4 Nanomaterial characterization and ethical testing for responsible nanosafety assessment
NMs are expected to play a central role in the transition to greener technologies and improved healthcare, but their full potential remains limited by the lack of practical, standardized methods for evaluating their safety and interactions in complex systems. Advancing nanosafety demands strategies that reflect how NMs truly behave in relevant biological and environmental media. The future lies in developing new protocols that capture dynamic transformations, and realistic exposure profiles, while embracing animal-free, high-throughput models such as C. elegans in line with the 3Rs (Replacement, Reduction, and Refinement) principles.209 Integrating these approaches will provide more predictive, ethical, and standardized assessments, ensuring safer design and deployment of next-generation NMs but also proper assessment of impact of emerging NM pollutants.2104.1 Advancing characterization from standard nanomaterials to industrially relevant and environmental contaminants
A major challenge in nanosafety assessment lies in the insufficient characterization of these materials under realistic exposure conditions, especially when tested in biological or environmental media. Once introduced into such media, NMs undergo dynamic physicochemical transformations that alter their size, aggregation state, surface properties, and most importantly, lead to the formation of a biomolecular corona.211 This corona is a layer of proteins, lipids, and other biomolecules that adsorbs onto the NM surface and it fundamentally redefines the biological identity of the particle and governs its interactions with cells and tissues.212 Significant efforts have been done to mitigating these transformations through surface functionalization with biocompatible polymers,213 such as poly(ethylene glycol) (PEG),214–216 poly(ethylenimine) (PEI),217 poly(amidoamine) (PAMAM) dendrimers,218,219 chitosan,220,221 and related coatings. While these modifications improve colloidal stability and quench biomolecular corona formation, even functionalized nanomaterials undergo dynamic transformations under realistic biological and environmental conditions.222 And despite its importance, the corona is still often overlooked in in vitro studies, particularly in standard toxicity tests that do not consider the species origin of serum components or the effect of preconditioning protocols.223,224 Therefore, a thorough characterization of the corona, especially the hard corona that persists and dictates interactions,225 is essential for reliable hazard assessment. In particular beside proteomic, the workhorse of corona characterization, the role of other biomolecules is emerging such as glycans or lipids.226,227 Moreover, it is also important to be aware of the advantages and limits of the established protocols such as for proteomics analysis.228 In addition to corona profiling, the colloidal stability and state of dispersion of NMs in test media are critical factors.147,229 Many NMs, such as graphene or some kind of quantum dots, are not initially designed to be water-dispersible and often require mechanical or chemical dispersion to be evaluated in vitro.230 Improper dispersion leads to uncontrolled agglomeration and sedimentation, which in turn results in inaccurate dose delivery.231 However, dosimetry remains one of the most underappreciated yet impactful factors in nanotoxicology. Indeed, the actual dose delivered to cells is not simply the concentration administered but a function of time-dependent transformations such as agglomeration, sedimentation, and diffusion, all of which are influenced by particle size, shape, surface chemistry, and effective density. Large agglomerates sediment faster, increasing the local concentration at the cell surface, while buoyant particles may float away, preventing contact entirely. Moreover, agglomeration decreases the number of bioavailable particles and reduces total surface area, a key determinant of nano–bio interactions. Accurate dosimetric modeling, incorporating these transformations and validated by in vitro to in vivo extrapolation, is crucial to resolving the discrepancies that frequently arise between.231 The importance of the assessment of the real delivered dose is also fundamental for low-density materials such as plastics like polyethylene and polypropylene, which are buoyant and do not settle onto the cell layer in traditional assays. These materials, while poorly represented in current studies, are the most abundant in human exposure scenarios, including ingestion and environmental contamination.232–234 Their irregular shapes, broad size distributions, and resistance to aqueous dispersion present unique challenges for both characterization and testing. To capture realistic exposure profiles, specialized models such as inverse cell culture systems are necessary, especially for floating particles.235 Without such adaptations, results may systematically underestimate or misrepresent toxicity.In conclusion, as the diversity of NMs increases, with the emergence of complex composites, 2D materials with high surface areas, non-aqueous dispersible particles, and environmentally derived NMPs, there is an urgent need for robust and adaptable characterization strategies. These materials often exhibit unique behaviours that are not adequately captured by standard protocols.230,236 Their heterogeneity, non-spherical morphology, and interactions with environmental or biological macromolecules challenge conventional assumptions and necessitate the use of orthogonal techniques to fully assess size, aggregation, surface chemistry, and interaction potential in relevant media.
4.2 Influence of surface charge of plastic nanomaterials on interaction with biological models
Plastic industry is one of the main revolutions of the last century. Production of plastic has increased more than 100% per year.232 Also, the COVID-19 pandemic has further increased the demand for single-use plastic materials for daily usage.237 Although the effect of macroplastics has been extensively investigated,238,239 the interaction between micro and nanoplastics and biological targets needs further clarifications.240,241 Understanding how NP features, such as the external surface charge, impact the uptake by cells, the intracellular fate and, on a larger scale, the environment and health is needed to improve sustainable plastics production. To this aim, an integrated platform including physical and chemical characterization, cellular activity studies, internalization and intracellular path and fate of NPs, also suitable and tunable for many different NMs, has been developed (Fig. 8). | ||
| Fig. 8 Development of a platform suitable for different NMs, to study features of NPs-biological targets interaction. | ||
Human cell lines and C. elegans are biological models commonly employed to study the cytotoxicity of various materials. Recent studies revealed that positive and negative polystyrene nanoplastics (PS-NPs) act differently in the interaction with cells and animal models. Positively charged NPs tend to be more internalized than negatively charged ones, also causing a dose-dependent decrease in cell growth and viability. The z-potential of PS-NPs also affected their toxic effect in vivo, where only positive NPs are able to cause a dose-related decrease of C. elegans viability and defects in motility, pharyngeal function, reproduction, and development. Investigating the route through which the NPs enter the cells result crucial to better understand their fate once internalized. Latest findings confirmed that cells primarily exploit clathrin-mediated endocytosis to uptake PS-NPs, depending also on the NPs size and z-potential.242 In particular, highly positively charged NMs, get attached to the negative charge of the cell membrane because of electrostatic attraction and then penetrate the membranes. For the same reason, a charge repulsion between negative PS-NPs and the cell membrane could lead to a minor internalization rate. Furthermore, the surface charge of the PS-NPs impacts the formation and the composition of the protein corona once the particles interact with biological fluids. Experiments based on the incubation of PS-NPs with serum show a lower rate of internalization of both positively and negatively charged PS-NPs into cells, broadening the research field of the protein corona.243
The concern about these nanometric particles is their fate once internalized into the cells. Usually, when internalized, NPs are rapidly entrapped into the early endosomes starting the degradation path through the late endosomes and lysosomes. However, some particles including ionizable lipid NPs (such as mRNA based nanovaccines) are able to escape from the endosomes, through the endosomal escape, and release their cargo into the cytoplasm.142 Nanomedicine often takes advantage of this feature for its therapeutic aims. In the case of PS-NPs, the issue could be related to the long-term effects of the accumulation in various districts of our body. Thus, the evidence that after 24 hours a high percentage of NPs are already entrapped into the lysosomes suggest an efficient degradation of the particles. The effect of the accumulated particles over time is still unknown and deserves a deeper investigation, for human, animal and environmental health.
4.3 C. elegans as an in vivo model for toxicological studies on nanomaterials
Caenorhabditis elegans (C. elegans) is a free-living, non-parasitic nematode that is approximately 1 mm in length and is widely used as a model organism in biomedical research. It has a well-characterized anatomy consisting of around 1000 somatic cells, including a fully mapped nervous system with 302 neurons.244 Despite being evolutionarily distant from vertebrates, 65% of its genes have a homolog in humans, and many pathways involved in development, cell death, metabolism, and stress responses are conserved.245C. elegans offers several technical advantages: a short lifecycle, rapid reproduction, a transparent body for in vivo imaging, and well-established genetic manipulation techniques, such as RNAi and the development of transgenic worms.245,246 These characteristics make it a valuable tool for toxicological studies, particularly in the context of NMs.Exposure studies can be conducted via ingestion, dermal absorption, or environmental exposure in liquid or agar-based media. Key endpoints include lethality, reproductive output, developmental progression, behavioural phenotypes, and molecular biomarkers such as oxidative stress genes or heat-shock proteins246,247 (Fig. 9). C. elegans is also helpful in assessing the chronic effects of low-dose NM exposure. Together, this application renders C. elegans a valuable model for high-throughput assays.248
Another advantage of using C. elegans is that this nematode is ubiquitous worldwide.249 It inhabits both soil and water at varying temperatures, thus serving as an excellent environmental indicator that provides insight into the impact of pollution on the health of living organisms. Ultimately, C. elegans offers a biologically relevant and ethically advantageous alternative to vertebrate models, aligning with the principles of the 3Rs and facilitating the early-phase screening of NM toxicology, thereby promoting the safe development of new NMs.
Several studies have been published using C. elegans as an animal model to evaluate the toxicity of various NMs.243,247,250,251 For instance, the relationship between the physicochemical properties of PS-NPs, particularly the surface charge, and their toxicity in vitro and in vivo was recently studied. The in vivo findings obtained from C. elegans supported those acquired from cells, providing further insights into the sub-toxic effects of PS-NPs, which are impossible to detect in simpler systems, such as cell culture.243
Significant challenges persist in the field of nanotoxicity. Chief among these is translating the effects observed in C. elegans to human health risk assessment. This nematode lacks certain tissues, such as a circulatory system, making it difficult to fully understand the biodistribution of NMs. Additionally, the absence of organs like the liver, brain, and lungs obstructs the study of organ-specific effects. A clear need exists for collaboration among various laboratories to validate findings from C. elegans through studies on more complex vertebrate models. It is essential to develop standardized protocols for the physicochemical characterization of NMs and for assessing their toxicity in C. elegans. Generating comparable and reliable data will foster acceptance within the scientific community and among nanotoxicologists regarding the use of C. elegans as a model for preclinical analysis of NM toxicity.
In conclusion, looking to a future in which the development of NMs for medical or industrial use takes over, C. elegans represents a promising and versatile in vivo model for the early-stage toxicological evaluation of NMs, not only for the analysis of classical endpoints, such as survival, reproduction, and locomotion, but also by its integration into high-throughput assay, and from an ethics point of view. However, to fully realize its potential, strengthening collaboration between scientists and harmonizing methodologies will be critical steps toward integrating C. elegans into regulatory frameworks and preclinical analysis, ultimately advancing the safe and responsible development of nanomedicines.
5 Summary and perspective
This review has explored the rapidly expanding Nanoverse, a conceptual framework capturing the diversity and interconnectivity of NMs across disciplines. As reported in this review, these advances collectively illustrate how precision molecular design, hybrid architectures, and scalable manufacturing strategies are enabling NMs to address current biomedical, environmental, and industrial challenges.At the same time, we have highlighted the critical importance of realistic characterization and ethically responsible toxicological assessment. The behaviour of NMs in complex biological and environmental matrices is governed not only by their intrinsic physicochemical parameters but also by dynamic transformations such as biomolecular corona formation. Understanding these processes, particularly under realistic exposure conditions, remains a prerequisite for accurate risk assessment. In this regard, novel models such as C. elegans offer a biologically relevant and ethically sound complement to conventional in vitro systems, enabling high-throughput, whole-organism toxicology while aligning with the principles of the 3Rs. We also highlight how this is also crucial, not only to properly assess the impact of manufactured NMs, but also for the study of emerging NM contaminants, such as micro- and nanoplastics. Indeed, these NMs further highlight the need for adaptable protocols capable of addressing materials with irregular morphologies, broad size distributions, and atypical dispersion behaviours.
Despite all the reported advances, the Nanoverse is still in its early stages. Major challenges persist, including the absence of universally accepted descriptors, limited cross-domain data integration, and difficulties in scaling promising lab-scale innovations to industrial production. Meeting these challenges will require the integration of advanced computational tools into NM research pipelines. Molecular simulations, from quantum-level modelling of defect chemistry to mesoscale dynamics of corona evolution, can now be coupled with Quantitative Structure–Activity Relationships (QSAR) and machine learning algorithms to predict NM behaviour across multiple environments and time scales.252–254 Artificial intelligence (AI) can accelerate discovery by enabling inverse design approaches, identifying optimal structural parameters for targeted functions, and guiding adaptive manufacturing processes.255–257 These tools are equally crucial for safety-by-design, allowing the early identification of potential hazards before materials enter biological systems or the environment.
Looking ahead, the evolution of the Nanoverse will depend on a synergistic interplay between experimental innovation, computational prediction, and regulatory adaptation. International collaboration will be essential for developing standardized characterization frameworks, ensuring data interoperability, and facilitating the translation of novel NMs into safe, high-performance applications. By merging advances in material science, bioengineering, AI-driven modelling, and nanosafety assessment, we can accelerate the responsible expansion of the Nanoverse, unlocking its potential to transform our society while safeguarding human and planetary health.
Author contributions
A. Sao.: conceptualization, investigation, visualization, project administration, writing – original draft, writing – review & editing; A. Su.: investigation, visualization, writing – original draft, writing – review & editing; G. S.: investigation, visualization, writing – original draft, writing – review & editing; G.-Y. M.: investigation, visualization, writing – original draft, writing – review & editing; C. N.: investigation, visualization, writing – original draft, writing – review & editing; R. B.-P.: investigation, visualization, writing – original draft, writing – review & editing; G. C.: investigation, visualization, writing – original draft, writing – review & editing; M. G.: investigation, visualization, writing – original draft, writing – review & editing; M. C.: investigation, writing – original draft, writing – review & editing; M. V.-B.: investigation, writing – original draft, writing – review & editing; A. Sal.: investigation, writing – original draft, writing – review & editing; B. P.: investigation, writing – original draft, writing – review & editing; S. C.: investigation, writing – original draft, writing – review & editing; A. M.: investigation, visualization, writing – original draft, writing – review & editing; A. M.-S.: conceptualization, supervision, visualization, project administration, writing – original draft, writing – review & editing.Conflicts of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
A. Sao., A. Su. and C. N. acknowledge the support from the European Union's Research and Innovation Program no. 101092901 (POTENTIAL). G.-Y. M. thanks the EU funding within the NextGenerationEU-MUR PNRR Extended Partnership initiative on Emerging Infectious Diseases (Project no. PE00000007, INF-ACT). A. M. thanks the grant from the Italian Ministry of Health (BANDO RICERCA FINALIZZATA 2021, RF-2021-12374476). A. M.-S. acknowledges the AI4S fellowship within the “Generación D” initiative by Red.es, Ministerio para la Transformación Digital y de la Función Pública, for talent attraction (C005/24-ED CV1), funded by NextGenerationEU through PRTR.References
- Commission Recommendation of 18 October 2011 on the Definition of Nanomaterial, Off. J. Eur. Union Search PubMed.
- Commission Recommendation of 10 June 2022 on the Definition of Nanomaterial, Off. J. Eur. Union Search PubMed.
- J. Jeevanandam, A. Barhoum, Y. S. Chan, A. Dufresne and M. K. Danquah, Review on Nanoparticles and Nanostructured Materials: History, Sources, Toxicity and Regulations, Beilstein J. Nanotechnol., 2018, 9, 1050–1074, DOI:10.3762/bjnano.9.98.
- C. J. Moore, H. Montón, R. O'Kennedy, D. E. Williams, C. Nogués, C. Crean (Née Lynam) and V. Gubala, Controlling Colloidal Stability of Silica Nanoparticles during Bioconjugation Reactions with Proteins and Improving Their Longer-Term Stability, Handling and Storage, J. Mater. Chem. B, 2015, 3(10), 2043–2055, 10.1039/C4TB01915F.
- K. S. Novoselov, A. K. Geim, S. V. Morozov, D. Jiang, M. I. Katsnelson, I. V. Grigorieva, S. V. Dubonos and A. A. Firsov, Two-Dimensional Gas of Massless Dirac Fermions in Graphene, Nature, 2005, 438(7065), 197–200, DOI:10.1038/nature04233.
- S. Cerveny, F. Barroso-Bujans, Á. Alegría and J. Colmenero, Dynamics of Water Intercalated in Graphite Oxide, J. Phys. Chem. C, 2010, 114(6), 2604–2612, DOI:10.1021/jp907979v.
- A. Martinez-Serra, J. Cheeseman, A. Saorin, M. G. Soliman, M. Dobricic, D. I. R. Spencer and M. P. Monopoli, Multiple Exposures of Plasma to Nanoparticles: A Novel Tool to Personalize Biomolecular Coronas and Fractionate Fluids, Anal. Chem., 2025, 97(27), 14132–14141, DOI:10.1021/acs.analchem.4c05573.
- H. Mohammed, M. F. Mia, J. Wiggins and S. Desai, Nanomaterials for Energy Storage Systems—A Review, Molecules, 2025, 30(4), 883, DOI:10.3390/molecules30040883.
- V. Harish, D. Tewari, M. Gaur, A. B. Yadav, S. Swaroop, M. Bechelany and A. Barhoum, Review on Nanoparticles and Nanostructured Materials: Bioimaging, Biosensing, Drug Delivery, Tissue Engineering, Antimicrobial, and Agro-Food Applications, Nanomaterials, 2022, 12(3), 457, DOI:10.3390/nano12030457.
- M. A. VandenBerg, X. Dong, W. C. Smith, G. Tian, O. Stephens, T. F. O'Connor and X. Xu, Learning from the Future: Towards Continuous Manufacturing of Nanomaterials, AAPS Open, 2025, 11(1), 7, DOI:10.1186/s41120-025-00111-9.
- R. K. Mishra, K. Verma and D. S. Singh, Defect Engineering in Nanomaterials: Impact, Challenges, and Applications, Smart Mater. Manuf., 2024, 2, 100052, DOI:10.1016/j.smmf.2024.100052.
- N. Zavanelli, J. Kim and W.-H. Yeo, Recent Advances in High-Throughput Nanomaterial Manufacturing for Hybrid Flexible Bioelectronics, Materials, 2021, 14(11), 2973, DOI:10.3390/ma14112973.
- C. A. Charitidis, P. Georgiou, M. A. Koklioti, A.-F. Trompeta and V. Markakis, Manufacturing Nanomaterials: From Research to Industry, Manuf. Rev., 2014, 1, 11, DOI:10.1051/mfreview/2014009.
- Z. Xia, Y. Wen and M. Jian, Introduction to Carbon Nanomaterials for Smart Applications, Nanoscale Adv., 2025, 7(6), 1487–1488, 10.1039/D5NA90013A.
- C. Fang, H. Wu, S.-Y. Lee, R. L. Mahajan and R. Qiao, The Ionized Graphene Oxide Membranes for Water-Ethanol Separation, Carbon, 2018, 136, 262–269, DOI:10.1016/j.carbon.2018.04.077.
- C. Y. Hu, A. Achari, P. Rowe, H. Xiao, S. Suran, Z. Li, K. Huang, C. Chi, C. T. Cherian, V. Sreepal, P. D. Bentley, A. Pratt, N. Zhang, K. S. Novoselov, A. Michaelides and R. R. Nair, pH-Dependent Water Permeability Switching and Its Memory in MoS2 Membranes, Nature, 2023, 616(7958), 719–723, DOI:10.1038/s41586-023-05849-4.
- N. Parvin, V. Kumar, S. W. Joo and T. K. Mandal, Emerging Trends in Nanomedicine: Carbon-Based Nanomaterials for Healthcare, Nanomaterials, 2024, 14(13), 1085, DOI:10.3390/nano14131085.
- Y. Li, J. Zhao, J. Zhang, X. Gong, J. Zhou, N. Zhang and Y. Su, Smart and Solvent-Switchable Graphene-Based Membrane for Graded Molecular Sieving, Nat. Commun., 2025, 16(1), 5363, DOI:10.1038/s41467-025-60680-x.
- A. He, Z. Jiang, Y. Wu, H. Hussain, J. Rawle, M. E. Briggs, M. A. Little, A. G. Livingston and A. I. Cooper, A Smart and Responsive Crystalline Porous Organic Cage Membrane with Switchable Pore Apertures for Graded Molecular Sieving, Nat. Mater., 2022, 21(4), 463–470, DOI:10.1038/s41563-021-01168-z.
- R. Chang, L. Zhao, R. Xing, J. Li and X. Yan, Functional Chromopeptide Nanoarchitectonics: Molecular Design, Self-Assembly and Biological Applications, Chem. Soc. Rev., 2023, 52(8), 2688–2712, 10.1039/D2CS00675H.
- R. Shakeel, A. Ahmad, H. Mushtaq, T. Haider, M. Javeria, A. Asif, K. Zia, H. Zafar, B. Ishaque and W. Z. Khan, Multifunctional Nanomaterials in Materials Chemistry: A Cross-Disciplinary Review of Applications in Energy, Environment, Catalysis, and Biomedicine, Scholars J. Eng. Technol., 2025, 13(08), 673–707, DOI:10.36347/sjet.2025.v13i08.007.
- M. Srinivasan, M. Rajabi and S. Mousa, Multifunctional Nanomaterials and Their Applications in Drug Delivery and Cancer Therapy, Nanomaterials, 2015, 5(4), 1690–1703, DOI:10.3390/nano5041690.
- N. Gulati, D. K. Chellappan, R. MacLoughlin, K. Dua and H. Dureja, Inhaled Nano-Based Therapeutics for Inflammatory Lung Diseases: Recent Advances and Future Prospects, Life Sci., 2021, 285, 119969, DOI:10.1016/j.lfs.2021.119969.
- D. E. Mahmoud, S. H. Hosseini, H. A. Rathore, A. M. Alkilany, A. Heise and A. Elhissi, Inhalable Nanotechnology-Based Drug Delivery Systems for the Treatment of Inflammatory Lung Diseases, Pharmaceutics, 2025, 17(7), 893, DOI:10.3390/pharmaceutics17070893.
- H. Liu, H. Lv, X. Duan, Y. Du, Y. Tang and W. Xu, Advancements in Macrophage-Targeted Drug Delivery for Effective Disease Management, Int. J. Nanomed., 2023, 18, 6915–6940, DOI:10.2147/IJN.S430877.
- Q. Guo and Z.-M. Qian, Macrophage Based Drug Delivery: Key Challenges and Strategies, Bioact. Mater., 2024, 38, 55–72, DOI:10.1016/j.bioactmat.2024.04.004.
- S. Du, Y. Guan, A. Xie, Z. Yan, S. Gao, W. Li, L. Rao, X. Chen and T. Chen, Extracellular Vesicles: A Rising Star for Therapeutics and Drug Delivery, J. Nanobiotechnol., 2023, 21(1), 231, DOI:10.1186/s12951-023-01973-5.
- T. Chen, D. Chen, W. Su, J. Liang, X. Liu and M. Cai, Extracellular Vesicles as Vital Players in Drug Delivery: A Focus on Clinical Disease Treatment, Front. Bioeng. Biotechnol., 2025, 13, 1600227, DOI:10.3389/fbioe.2025.1600227.
- S. Moghassemi, A. Dadashzadeh, M. J. Sousa, H. Vlieghe, J. Yang, C. M. León-Félix and C. A. Amorim, Extracellular Vesicles in Nanomedicine and Regenerative Medicine: A Review over the Last Decade, Bioact. Mater., 2024, 36, 126–156, DOI:10.1016/j.bioactmat.2024.02.021.
- J. M. V. Makabenta, A. Nabawy, C.-H. Li, S. Schmidt-Malan, R. Patel and V. M. Rotello, Nanomaterial-Based Therapeutics for Antibiotic-Resistant Bacterial Infections, Nat. Rev. Microbiol., 2021, 19(1), 23–36, DOI:10.1038/s41579-020-0420-1.
- S. Gulati, R. Yadav, V. Kumari, S. Nair, C. Gupta and M. Aishwari, Nanosensors in Healthcare: Transforming Real-Time Monitoring and Disease Management with Cutting-Edge Nanotechnology, RSC Pharm., 2025, 2(5), 1003–1018, 10.1039/D5PM00125K.
- Y. Liu, G. Yang, Y. Hui, S. Ranaweera and C. Zhao, Microfluidic Nanoparticles for Drug Delivery, Small, 2022, 18(36), 2106580, DOI:10.1002/smll.202106580.
- S. Gimondi, H. Ferreira, R. L. Reis and N. M. Neves, Microfluidic Devices: A Tool for Nanoparticle Synthesis and Performance Evaluation, ACS Nano, 2023, 17(15), 14205–14228, DOI:10.1021/acsnano.3c01117.
- F. Lebre, N. Chatterjee, S. Costa, E. Fernández-de-Gortari, C. Lopes, J. Meneses, L. Ortiz, A. R. Ribeiro, V. Vilas-Boas and E. Alfaro-Moreno, Nanosafety: An Evolving Concept to Bring the Safest Possible Nanomaterials to Society and Environment, Nanomaterials, 2022, 12(11), 1810, DOI:10.3390/nano12111810.
- P. Jagadeesh, S. M. Rangappa and S. Siengchin, Advanced Characterization Techniques for Nanostructured Materials in Biomedical Applications, Adv. Ind. Eng. Polym. Res., 2024, 7(1), 122–143, DOI:10.1016/j.aiepr.2023.03.002.
- A. Leudjo Taka, C. M. Tata, M. J. Klink, X. Y. Mbianda, F. M. Mtunzi and E. B. Naidoo, A Review on Conventional and Advanced Methods for Nanotoxicology Evaluation of Engineered Nanomaterials, Molecules, 2021, 26(21), 6536, DOI:10.3390/molecules26216536.
- N. Ali, J. Katsouli, E. L. Marczylo, T. W. Gant, S. Wright and J. Bernardino De La Serna, The Potential Impacts of Micro-and-Nano Plastics on Various Organ Systems in Humans, EBioMedicine, 2024, 99, 104901, DOI:10.1016/j.ebiom.2023.104901.
- S. Alqahtani, S. Alqahtani, Q. Saquib and F. Mohiddin, Toxicological Impact of Microplastics and Nanoplastics on Humans: Understanding the Mechanistic Aspect of the Interaction, Front. Toxicol., 2023, 5, 1193386, DOI:10.3389/ftox.2023.1193386.
- P. Gubert, G. Gubert, R. C. D. Oliveira, I. C. O. Fernandes, I. C. Bezerra, B. D. Ramos, M. F. D. Lima, D. T. Rodrigues, A. F. N. D. Cruz, E. C. Pereira, D. S. Ávila and D. H. Mosca, Caenorhabditis Elegans as a Prediction Platform for Nanotechnology-Based Strategies: Insights on Analytical Challenges, Toxics, 2023, 11(3), 239, DOI:10.3390/toxics11030239.
- N. Desai, Challenges in Development of Nanoparticle-Based Therapeutics, AAPS J., 2012, 14(2), 282–295, DOI:10.1208/s12248-012-9339-4.
- X. Gao and G. V. Lowry, Progress towards Standardized and Validated Characterizations for Measuring Physicochemical Properties of Manufactured Nanomaterials Relevant to Nano Health and Safety Risks, NanoImpact, 2018, 9, 14–30, DOI:10.1016/j.impact.2017.09.002.
- M. Hachhach, S. Bayou, A. El Kasmi, M. Z. Saidi, H. Akram, M. Hanafi, O. Achak, C. El Moujahid and T. Chafik, Towards Sustainable Scaling-Up of Nanomaterials Fabrication: Current Situation, Challenges, and Future Perspectives, Eng, 2025, 6(7), 149, DOI:10.3390/eng6070149.
- M. Faria, M. Björnmalm, K. J. Thurecht, S. J. Kent, R. G. Parton, M. Kavallaris, A. P. R. Johnston, J. J. Gooding, S. R. Corrie, B. J. Boyd, P. Thordarson, A. K. Whittaker, M. M. Stevens, C. A. Prestidge, C. J. H. Porter, W. J. Parak, T. P. Davis, E. J. Crampin and F. Caruso, Minimum Information Reporting in Bio–Nano Experimental Literature, Nat. Nanotechnol., 2018, 13(9), 777–785, DOI:10.1038/s41565-018-0246-4.
- Waris, M. S. Chaudhary, A. H. Anwer, S. Sultana, P. P. Ingole, S. A. A. Nami and M. Z. Khan, A Review on Development of Carbon-Based Nanomaterials for Energy Storage Devices: Opportunities and Challenges, Energy Fuels, 2023, 37(24), 19433–19460, DOI:10.1021/acs.energyfuels.3c03213.
- N. Jayaprakash, K. Elumalai, S. Manickam, G. Bakthavatchalam and P. Tamilselvan, Carbon Nanomaterials: Revolutionizing Biomedical Applications with Promising Potential, Nano Mater. Sci., 2024, S2589965124001673, DOI:10.1016/j.nanoms.2024.11.004.
- K.-M. Lyra, I. Tournis, M. Subrati, K. Spyrou, A. Papavasiliou, C. Athanasekou, S. Papageorgiou, E. Sakellis, M. A. Karakassides and Z. Sideratou, Carbon Nanodisks Decorated with Guanidinylated Hyperbranched Polyethyleneimine Derivatives as Efficient Antibacterial Agents, Nanomaterials, 2024, 14(8), 677, DOI:10.3390/nano14080677.
- L. Cardo, L. Martínez-Parra, M. Cesco, B. M. Echeverría-Beistegui, M. Martínez-Moro, N. Herrero-Álvarez, M. Cabrerizo, S. Carregal-Romero, P. Ramos-Cabrer, J. Ruiz-Cabello and M. Prato, Luminescent Carbon Nanodots Doped with Gadolinium (III): Purification Criteria, Chemical and Biological Characterization of a New Dual Fluorescence/MR Imaging Agent, Small, 2023, 19(31), 2206442, DOI:10.1002/smll.202206442.
- Z.-L. Xu, S. Lin, N. Onofrio, L. Zhou, F. Shi, W. Lu, K. Kang, Q. Zhang and S. P. Lau, Exceptional Catalytic Effects of Black Phosphorus Quantum Dots in Shuttling-Free Lithium Sulfur Batteries, Nat. Commun., 2018, 9(1), 4164, DOI:10.1038/s41467-018-06629-9.
- A. Subrati, S. Mondal, M. Ali, A. Alhindi, R. Ghazi, A. Abdala, D. Reinalda and S. Alhassan, Developing Hydrophobic Graphene Foam for Oil Spill Cleanup, Ind. Eng. Chem. Res., 2017, 56(24), 6945–6951, DOI:10.1021/acs.iecr.7b00716.
- E. Sturabotti, B. Sierra-Serrano, S. M. Apresto, M. Cesco, L. Comparini, L. Cardo and M. Prato, Carbon Dots as Multi-Modal Contrast Agents: Opportunities and Open Challenges for in Vivo Bioimaging, Adv. Drug Delivery Rev., 2025, 224, 115659, DOI:10.1016/j.addr.2025.115659.
- P. Zygouri, G. Tsiodoulos, M. Angelidou, E. Papanikolaou, A.-M. Athinodorou, Y. V. Simos, K. Spyrou, M. Subrati, A. Kouloumpis, A. S. Kaloudi, G. Asimakopoulos, K. Tsamis, D. Peschos, P. Vezyraki, V. Ragos and D. P. Gournis, Graphene Oxide and Oxidized Carbon Nanodiscs as Biomedical Scaffolds for the Targeted Delivery of Quercetin to Cancer Cells, Nanoscale Adv., 2024, 6(11), 2860–2874, 10.1039/D3NA00966A.
- J. Wang, Z. Wen, Y. Xu, X. Ning, D. Wang, J. Cao and Y. Feng, Procedural Promotion of Wound Healing by Graphene-Barium Titanate Nanosystem with White Light Irradiation, Int. J. Nanomed., 2023, 18, 4507–4520, DOI:10.2147/IJN.S408981.
- J. K. Wychowaniec, J. Litowczenko and K. Tadyszak, Fabricating Versatile Cell Supports from Nano- and Micro-Sized Graphene Oxide Flakes, J. Mech. Behav. Biomed. Mater., 2020, 103, 103594, DOI:10.1016/j.jmbbm.2019.103594.
- J. Ordoño, M. Islam, A. D. Lantada, M. Echeverry-Rendón and D.-Y. Wang, Evaluation of Laser-Induced Graphene for Skeletal Muscle Tissue Engineering Applications, Biomater. Sci., 2025, 13(12), 3242–3251, 10.1039/D5BM00174A.
- A. Subrati, T. A. Pacheco, Í. A. Costa, V. C. Mello, J. F. R. Marroquin, M. Kościński, L. A. G. Pinho, A. P. Silveira, J. F. Felix, M. Cunha-Filho, M. J. Poças-Fonseca, R. B. D. Azevedo, S. N. Báo, R. P. Avelar, L. G. Paterno, S. Moya and J. P. F. Longo, Graphene Oxide Composites with Ultrahigh-Molecular-Weight Polyethylene for Innovative Prostheses for Arthroplasty, ACS Appl. Bio Mater., 2026, 9(1), 81–95, DOI:10.1021/acsabm.5c01289.
- A. Silvestri, F. Wang, X. Feng, A. L. Cortajarena and M. Prato, Protein-Based (Bio)Materials: A Way toward High-Performance Graphene Enzymatic Biosensors, J. Mater. Chem. C, 2022, 10(14), 5466–5473, 10.1039/D2TC00483F.
- X. Wu, M. Xu, S. Wang, K. Abbas, X. Huang, R. Zhang, A. C. Tedesco and H. F. Bi, N-Doped Carbon Dots as Efficient Type I Photosensitizers for Photodynamic Therapy, Dalton Trans., 2022, 51(6), 2296–2303, 10.1039/D1DT03788A.
- A. Silvestri, J. Zayas-Arrabal, M. Vera-Hidalgo, D. Di Silvio, C. Wetzl, M. Martinez-Moro, A. Zurutuza, E. Torres, A. Centeno, A. Maestre, J. M. Gómez, M. Arrastua, M. Elicegui, N. Ontoso, M. Prato, I. Coluzza and A. Criado, Ultrasensitive Detection of SARS-CoV-2 Spike Protein by Graphene Field-Effect Transistors, Nanoscale, 2023, 15(3), 1076–1085, 10.1039/D2NR05103F.
- D. Park, P. C. Sherrell, F. Xie and A. V. Ellis, Improved Lithium-Ion Battery Cathode Rate Performance via Carbon Black Functionalization, J. Mater. Chem. A, 2024, 12(8), 4884–4892, 10.1039/D3TA05093A.
- H. Park, R. K. Bera, H. Yoon and K. Kim, 3D Graphene-like Microporous Carbon for Ultralong-Life Lithium-Ion Capacitors, ACS Appl. Energy Mater., 2025, 8(13), 9489–9496, DOI:10.1021/acsaem.5c01138.
- H. Wu, J. Li, H. Wang, X. Li, J. Xu, Z. Yu, H. Zhang, Y. Zhao, X. Gong and L. Zhao, High-Strength and Dual Thermal-Electrical Conductive Modified Carbon Paper Based on MWCNTs-CFs Hierarchical Architecture for Fuel Cells, Chem. Eng. J., 2026, 529, 172598, DOI:10.1016/j.cej.2026.172598.
- C. M. Ibarra-Barreno, S. Chowdhury, M. Crosta, T. Zehra, F. Fasano, P. Kundu, J. Verstraelen, S. Bals, M. Subrati, D. Bonifazi, R. D. Costa and P. Rudolf, Bottom-Up Fabrication of BN-Doped Graphene Electrodes from Thiol-Terminated Borazine Molecules Working in Solar Cells, ACS Appl. Mater. Interfaces, 2025, 17(15), 23062–23075, DOI:10.1021/acsami.4c23116.
- J. Furtado, A. C. A. Ramos, J. E. G. Silva, R. Bachelard and A. C. Santos, Encoding Quantum Bits in Bound Electronic States of a Graphene Nanotorus, Ann. Phys., 2025, 472, 169862, DOI:10.1016/j.aop.2024.169862.
- T. S. Ghiasi, A. A. Kaverzin, A. H. Dismukes, D. K. De Wal, X. Roy and B. J. Van Wees, Electrical and Thermal Generation of Spin Currents by Magnetic Bilayer Graphene, Nat. Nanotechnol., 2021, 16(7), 788–794, DOI:10.1038/s41565-021-00887-3.
- N. Fantuzzi, A. Dib, S. Babamohammadi, S. Campigli, D. Benedetti and J. Agnelli, Mechanical Analysis of a Carbon Fibre Composite Woven Composite Laminate for Ultra-Light Applications in Aeronautics, Compos., Part C: Open Access, 2024, 14, 100447, DOI:10.1016/j.jcomc.2024.100447.
- A. Silvestri, A. Criado, F. Poletti, F. Wang, P. Fanjul-Bolado, M. B. González-García, C. García-Astrain, L. M. Liz-Marzán, X. Feng, C. Zanardi and M. Prato, Bioresponsive, Electroactive, and Inkjet-Printable Graphene-Based Inks, Adv. Funct. Mater., 2022, 32(2), 2105028, DOI:10.1002/adfm.202105028.
- Q. Hanniet, M. Boussmen, J. Barés, V. Huon, I. Iatsunskyi, E. Coy, M. Bechelany, C. Gervais, D. Voiry, P. Miele and C. Salameh, Investigation of Polymer-Derived Si–(B)–C–N Ceramic/Reduced Graphene Oxide Composite Systems as Active Catalysts towards the Hydrogen Evolution Reaction, Sci. Rep., 2020, 10(1), 22003, DOI:10.1038/s41598-020-78558-x.
- M. Yang, H. Liu, C. Qiu, I. Iatsunskyi, E. Coy, S. Moya, Z. Wang, W. Wu, X. Zhao and G. Wang, Electron Transfer Correlated Antibacterial Activity of Biocompatible Graphene Nanosheets-TiO2 Coatings, Carbon, 2020, 166, 350–360, DOI:10.1016/j.carbon.2020.05.036.
- Z. Zhao, H. Chen, W. Zhang, S. Yi, H. Chen, Z. Su, B. Niu, Y. Zhang and D. Long, Defect Engineering in Carbon Materials for Electrochemical Energy Storage and Catalytic Conversion, Mater. Adv., 2023, 4(3), 835–867, 10.1039/D2MA01009G.
- N. M. Bardhan, P. V. Kumar, Z. Li, H. L. Ploegh, J. C. Grossman, A. M. Belcher and G.-Y. Chen, Enhanced Cell Capture on Functionalized Graphene Oxide Nanosheets through Oxygen Clustering, ACS Nano, 2017, 11(2), 1548–1558, DOI:10.1021/acsnano.6b06979.
- A. Subrati, B. Gurzęda, P. Jeżowski, M. Kościński, G. Nowaczyk, M. Kempiński, P. Florczak, B. Peplińska, M. Jarek, Y. Al Wahedi, W. Kempiński, L. Smardz and P. Krawczyk, The Impact of Oxygen-Clustering on the Transformation of Electrochemically-Derived Graphite Oxide Framework, Carbon, 2024, 217, 118641, DOI:10.1016/j.carbon.2023.118641.
- J. G. McHugh, P. Mouratidis, A. Impellizzeri, K. Jolley, D. Erbahar and C. P. Ewels, Prismatic Edge Dislocations in Graphite, Carbon, 2022, 188, 401–419, DOI:10.1016/j.carbon.2021.11.072.
- Platform Optimisation To Enable NanomaTerIAL safety assessment for rapid commercialisation (POTENTIAL), https://www.potential-eu.eu/.
- P. Trucano and R. Chen, Structure of Graphite by Neutron Diffraction, Nature, 1975, 258(5531), 136–137, DOI:10.1038/258136a0.
- C. R. Groom, I. J. Bruno, M. P. Lightfoot and S. C. Ward, The Cambridge Structural Database, Acta Crystallogr., Sect. B: Struct. Sci., Cryst. Eng. Mater., 2016, 72(2), 171–179, DOI:10.1107/S2052520616003954.
- D. L. Dorset and M. P. McCourt, Disorder and the Molecular Packing of C60 Buckminsterfullerene: A Direct Electron-Crystallographic Analysis, Acta Crystallogr., Sect. A: Found. Crystallogr., 1994, 50(3), 344–351, DOI:10.1107/S0108767393012607.
- K. Momma and F. Izumi, VESTA 3 for Three-Dimensional Visualization of Crystal, Volumetric and Morphology Data, J. Appl. Crystallogr., 2011, 44(6), 1272–1276, DOI:10.1107/S0021889811038970.
- A. Subrati, E. Gutiérrez-Pineda and S. E. Moya, To-Pack or Not-to-Pack: Towards Improved Graphite Electrochemical Oxidation Through Electrode Design, ChemPlusChem, 2024, 89(12), e202400265, DOI:10.1002/cplu.202400265.
- J. Li, J. O'Shea, X. Hou and G. Z. Chen, Faradaic Processes beyond Nernst's Law: Density Functional Theory Assisted Modelling of Partial Electron Delocalisation and Pseudocapacitance in Graphene Oxides, Chem. Commun., 2017, 53(75), 10414–10417, 10.1039/C7CC04344A.
- R. K. Ibrahim, M. Hayyan, M. A. AlSaadi, A. Hayyan and S. Ibrahim, Environmental Application of Nanotechnology: Air, Soil, and Water, Environ. Sci. Pollut. Res., 2016, 23(14), 13754–13788, DOI:10.1007/s11356-016-6457-z.
- D. Prasai, J. C. Tuberquia, R. R. Harl, G. K. Jennings and K. I. Bolotin, Graphene: Corrosion-Inhibiting Coating, ACS Nano, 2012, 6(2), 1102–1108, DOI:10.1021/nn203507y.
- J. Liu, L. Cui and D. Losic, Graphene and Graphene Oxide as New Nanocarriers for Drug Delivery Applications, Acta Biomater., 2013, 9(12), 9243–9257, DOI:10.1016/j.actbio.2013.08.016.
- R. R. Nair, H. A. Wu, P. N. Jayaram, I. V. Grigorieva and A. K. Geim, Unimpeded Permeation of Water Through Helium-Leak–Tight Graphene-Based Membranes, Science, 2012, 335(6067), 442–444, DOI:10.1126/science.1211694.
- G. Liu, W. Jin and N. Xu, Two-Dimensional-Material Membranes: A New Family of High-Performance Separation Membranes, Angew. Chem., Int. Ed., 2016, 55(43), 13384–13397, DOI:10.1002/anie.201600438.
- J. Luo, R. Qiao and B. Ding, Enhancement of Ion Selectivity and Permeability in Two-Dimensional Material Membranes, Matter, 2024, 7(10), 3351–3389, DOI:10.1016/j.matt.2024.07.006.
- P. Liu, J. Hou, Y. Zhang, L. Li, X. Lu and Z. Tang, Two-Dimensional Material Membranes for Critical Separations, Inorg. Chem. Front., 2020, 7(13), 2560–2581, 10.1039/D0QI00307G.
- J. Abraham, K. S. Vasu, C. D. Williams, K. Gopinadhan, Y. Su, C. T. Cherian, J. Dix, E. Prestat, S. J. Haigh, I. V. Grigorieva, P. Carbone, A. K. Geim and R. R. Nair, Tunable Sieving of Ions Using Graphene Oxide Membranes, Nat. Nanotechnol., 2017, 12(6), 546–550, DOI:10.1038/nnano.2017.21.
- X. Chen, S. Mohammed, G. Yang, T. Qian, Y. Chen, H. Ma, Z. Xie, X. Zhang, G. P. Simon and H. Wang, Selective Permeation of Water through Angstrom-Channel Graphene Membranes for Bioethanol Concentration, Adv. Mater., 2020, 32(33), 2002320, DOI:10.1002/adma.202002320.
- L. Huang, Y. Li, Q. Zhou, W. Yuan and G. Shi, Graphene Oxide Membranes with Tunable Semipermeability in Organic Solvents, Adv. Mater., 2015, 27(25), 3797–3802, DOI:10.1002/adma.201500975.
- K. Goh, L. Setiawan, L. Wei, R. Si, A. G. Fane, R. Wang and Y. Chen, Graphene Oxide as Effective Selective Barriers on a Hollow Fiber Membrane for Water Treatment Process, J. Membr. Sci., 2015, 474, 244–253, DOI:10.1016/j.memsci.2014.09.057.
- W. Wang, N. Onofrio, E. Petit, B. A. Karamoko, H. Wu, J. Liu, J. Li, K. Qi, Y. Zhang, C. Gervais, L. Lajaunie, C. Salameh, P. Miele, Z. Zeng and D. Voiry, High-Surface-Area Functionalized Nanolaminated Membranes for Energy-Efficient Nanofiltration and Desalination in Forward Osmosis, Nat. Water, 2023, 1(2), 187–197, DOI:10.1038/s44221-023-00036-1.
- Y. Wang, H. Yan, M. Ma, C. Zhou, Q. Hou, Z. Xu, M. Xia, H. Ni, Y. Jia and P. Ye, Graft-crosslinked Graphene Oxide Nanofiltration Membrane with pH -responsive Nanochannels, Polym. Adv. Technol., 2024, 35(1), e6250, DOI:10.1002/pat.6250.
- D. Iglesias and S. Marchesan, Short peptide self-assembled nanostructures for therapeutics innovative delivery, Nanostructures for Novel Therapy, Elsevier, 2017, pp. 227–250, DOI:10.1016/B978-0-323-46142-9.00009-8.
- J. Lv, R. Sun and X. Gao, Emerging Devices Based on Chiral Nanomaterials, Nanoscale, 2025, 17(7), 3585–3599, 10.1039/D4NR03998J.
- S. Marchesan and M. Prato, Nanomaterials for (Nano)Medicine, ACS Med. Chem. Lett., 2013, 4(2), 147–149, DOI:10.1021/ml3003742.
- N. Bäumer, S. Yamada, S. Ogi and S. Yamaguchi, Interfacing Flexible Design and Social Self-Sorting Enables Comprehensive Control over Photophysical and Self-Assembly Properties of Supramolecular Polymers, J. Am. Chem. Soc., 2025, 147(10), 8300–8311, DOI:10.1021/jacs.4c15766.
- J. Ren, M. Peng, C. Ye and F. Xin, Single Atom Copper Catalysts on Sulfated Zirconia for the Catalytic Synthesis of Dimethoxymethane, ACS Appl. Nano Mater., 2024, 7(23), 27436–27446, DOI:10.1021/acsanm.4c05422.
- H. Xie, Y. Wang, N. Zhang, S. Li, J. Li and X. Xin, Solvent-Induced Self-Assembly of Silver Nanoclusters for White-Light-Emitting Diodes and Temperature Sensing, ACS Appl. Nano Mater., 2024, 7(1), 1009–1018, DOI:10.1021/acsanm.3c05006.
- Z. Xie, P. Sun, Z. Wang, H. Li, L. Yu, D. Sun, M. Chen, Y. Bi, X. Xin and J. Hao, Metal–Organic Gels from Silver Nanoclusters with Aggregation-Induced Emission and Fluorescence-to-Phosphorescence Switching, Angew. Chem., Int. Ed., 2020, 59(25), 9922–9927, DOI:10.1002/anie.201912201.
- M. J. Krysmann, V. Castelletto, A. Kelarakis, I. W. Hamley, R. A. Hule and D. J. Pochan, Self-Assembly and Hydrogelation of an Amyloid Peptide Fragment, Biochemistry, 2008, 47(16), 4597–4605, DOI:10.1021/bi8000616.
- Y. Kuang, J. Shi, J. Li, D. Yuan, K. A. Alberti, Q. Xu and B. Xu, Pericellular Hydrogel/Nanonets Inhibit Cancer Cells, Angew. Chem., Int. Ed., 2014, 53(31), 8104–8107, DOI:10.1002/anie.201402216.
- M. Reches and E. Gazit, Casting Metal Nanowires Within Discrete Self-Assembled Peptide Nanotubes, Science, 2003, 300(5619), 625–627, DOI:10.1126/science.1082387.
- X. Yan, P. Zhu and J. Li, Self-Assembly and Application of Diphenylalanine-Based Nanostructures, Chem. Soc. Rev., 2010, 39(6), 1877, 10.1039/b915765b.
- S. Catalini, F. Bagni, S. Cicchi, M. Di Donato, A. Iagatti, A. Lapini, P. Foggi, C. Petrillo, A. Di Michele, M. Paolantoni, G. Schirò, L. Comez and A. Paciaroni, Multiple Length-Scale Control of Boc-Protected Diphenylalanine Aggregates through Solvent Composition, Mater. Adv., 2024, 5(9), 3802–3811, 10.1039/D4MA00018H.
- S. Catalini, F. Mancusi, S. Cicchi, M. Di Donato, A. Iagatti, A. Lapini, P. Foggi, C. Petrillo, A. Di Michele, M. Paolantoni, L. Comez and A. Paciaroni, Self-Assembly of Boc-Protected Diphenylalanine Functionalized with Boron-Dipyrromethane, Spectrochim. Acta, Part A, 2025, 340, 126362, DOI:10.1016/j.saa.2025.126362.
- E. Nikoloudakis, K. Mitropoulou, G. Landrou, G. Charalambidis, V. Nikolaou, A. Mitraki and A. G. Coutsolelos, Self-Assembly of Aliphatic Dipeptides Coupled with Porphyrin and BODIPY Chromophores, Chem. Commun., 2019, 55(94), 14103–14106, 10.1039/C9CC06125H.
- S. Bera, Umesh and S. Bhattacharya, Enhanced Circularly Polarized Luminescence Attained via Self-Assembly of Heterochiral as Opposed to Homochiral Dipeptides in Water, Chem. Sci., 2024, 15(34), 13987–13997, 10.1039/D4SC01631A.
- S. Krekic, T. Zakar, Z. Gombos, S. Valkai, M. Mero, L. Zimányi, Z. Heiner and A. Dér, Nonlinear Optical Investigation of Microbial Chromoproteins, Front. Plant Sci., 2020, 11, 547818, DOI:10.3389/fpls.2020.547818.
- H. Zheng and S. Ravaine, Bottom-Up Assembly and Applications of Photonic Materials, Crystals, 2016, 6(5), 54, DOI:10.3390/cryst6050054.
- K. Patel, S. K. H. Shah and P. Prabhakaran, Aggregation-Induced Emission Materials for Protein Fibrils Imaging, in Progress in Molecular Biology and Translational Science, Elsevier, 2021, vol. 185, pp. 113–136, DOI:10.1016/bs.pmbts.2021.06.011.
- J. Min, X. Rong, J. Zhang, R. Su, Y. Wang and W. Qi, Computational Design of Peptide Assemblies, J. Chem. Theory Comput., 2024, 20(2), 532–550, DOI:10.1021/acs.jctc.3c01054.
- F. Kurul, H. Turkmen, A. E. Cetin and S. N. Topkaya, Nanomedicine: How Nanomaterials Are Transforming Drug Delivery, Bio-Imaging, and Diagnosis, Next Nanotechnol., 2025, 7, 100129, DOI:10.1016/j.nxnano.2024.100129.
- E. Karahmet Sher, M. Alebić, M. Marković Boras, E. Boškailo, E. Karahmet Farhat, A. Karahmet, B. Pavlović, F. Sher and L. Lekić, Nanotechnology in Medicine Revolutionizing Drug Delivery for Cancer and Viral Infection Treatments, Int. J. Pharm., 2024, 660, 124345, DOI:10.1016/j.ijpharm.2024.124345.
- A. A. Halwani, Development of Pharmaceutical Nanomedicines: From the Bench to the Market, Pharmaceutics, 2022, 14(1), 106, DOI:10.3390/pharmaceutics14010106.
- B. Pelaz, C. Alexiou, R. A. Alvarez-Puebla, F. Alves, A. M. Andrews, S. Ashraf, L. P. Balogh, L. Ballerini, A. Bestetti, C. Brendel, S. Bosi, M. Carril, W. C. W. Chan, C. Chen, X. Chen, X. Chen, Z. Cheng, D. Cui, J. Du, C. Dullin, A. Escudero, N. Feliu, M. Gao, M. George, Y. Gogotsi, A. Grünweller, Z. Gu, N. J. Halas, N. Hampp, R. K. Hartmann, M. C. Hersam, P. Hunziker, J. Jian, X. Jiang, P. Jungebluth, P. Kadhiresan, K. Kataoka, A. Khademhosseini, J. Kopeček, N. A. Kotov, H. F. Krug, D. S. Lee, C.-M. Lehr, K. W. Leong, X.-J. Liang, M. Ling Lim, L. M. Liz-Marzán, X. Ma, P. Macchiarini, H. Meng, H. Möhwald, P. Mulvaney, A. E. Nel, S. Nie, P. Nordlander, T. Okano, J. Oliveira, T. H. Park, R. M. Penner, M. Prato, V. Puntes, V. M. Rotello, A. Samarakoon, R. E. Schaak, Y. Shen, S. Sjöqvist, A. G. Skirtach, M. G. Soliman, M. M. Stevens, H.-W. Sung, B. Z. Tang, R. Tietze, B. N. Udugama, J. S. VanEpps, T. Weil, P. S. Weiss, I. Willner, Y. Wu, L. Yang, Z. Yue, Q. Zhang, Q. Zhang, X.-E. Zhang, Y. Zhao, X. Zhou and W. J. Parak, Diverse Applications of Nanomedicine, ACS Nano, 2017, 11(3), 2313–2381, DOI:10.1021/acsnano.6b06040.
- C. Xu, C. Lei, S. Hosseinpour, S. Ivanovski, L. J. Walsh and A. Khademhosseini, Nanotechnology for the Management of COVID-19 during the Pandemic and in the Post-Pandemic Era, Natl. Sci. Rev., 2022, 9(10), nwac124, DOI:10.1093/nsr/nwac124.
- M. Papi, M. De Spirito and V. Palmieri, Nanotechnology in the COVID-19 Era: Carbon-Based Nanomaterials as a Promising Solution, Carbon, 2023, 210, 118058, DOI:10.1016/j.carbon.2023.118058.
- K. R. Chaudhary, S. Kujur and K. Singh, Recent Advances of Nanotechnology in COVID 19: A Critical Review and Future Perspective, OpenNano, 2023, 9, 100118, DOI:10.1016/j.onano.2022.100118.
- I. Rose, M. Greenwood, M. Biggart, N. Baumlin, R. Tarran, S. L. Hart and D. L. Baines, Adenine Base Editing of CFTR Using Receptor Targeted Nanoparticles Restores Function to G542X Cystic Fibrosis Airway Epithelial Cells, Cell. Mol. Life Sci., 2025, 82(1), 144, DOI:10.1007/s00018-025-05587-y.
- G. Chauhan, A. A. Shaik, S. S. Sawant, R. Diwan, M. Mokashi, M. Goyal, S. K. Shukla, N. K. Kunda and V. Gupta, Continuously Producible Aztreonam-Loaded Inhalable Lipid Nanoparticles for Cystic Fibrosis-Associated Pseudomonas Aeruginosa Infections – Development and in-Vitro Characterization, Biomater. Adv., 2025, 166, 214027, DOI:10.1016/j.bioadv.2024.214027.
- M. H. Alyami, M. Z. Ahmad, J. Ahmad, B. A. Abdel-Wahab and K. Pathak, Advances in Lipid-Based Nanoformulations for Inhaled Antibiotic Therapy in Respiratory Infections, Drug Discovery Today, 2025, 30(6), 104380, DOI:10.1016/j.drudis.2025.104380.
- W. Peng, W. Tai, B. Li, H. Wang, T. Wang, S. Guo, X. Zhang, P. Dong, C. Tian, S. Feng, L. Yang, G. Cheng and B. Zheng, Inhalable Nanocatalytic Therapeutics for Viral Pneumonia, Nat. Mater., 2025, 24(4), 637–648, DOI:10.1038/s41563-024-02041-5.
- L. Guyon, A. Ladaycia, A. Bosio, L. Lemaire, F. Franconi, B. Lelièvre, N. Lautram, P. Pigeon, G. Jaouen, C. Passirani and E. Lepeltier, Self-Assemblies of Cell-Penetrating Peptides and Ferrocifens: Design and Biological Evaluation of an Innovative Platform for Lung Cancer Treatment, Nanoscale, 2025, 17(15), 9232–9244, 10.1039/D5NR00643K.
- S. Yasmin-Karim, G. Richards, A. Fam, A.-M. Ogurek, S. Sridhar and G. M. Makrigiorgos, Aerosol Delivery of Hesperetin-Loaded Nanoparticles and Immunotherapy Increases Survival in a Murine Lung Cancer Model, Nanomaterials, 2025, 15(8), 586, DOI:10.3390/nano15080586.
- M. Govender and Y. E. Choonara, Drug Delivery Systems for Respiratory Diseases: Insights into the Therapeutic Innovations for Pulmonary Administration, Pharmaceutics, 2025, 17(5), 539, DOI:10.3390/pharmaceutics17050539.
- D. Ge, S. Ma, T. Sun, Y. Li, J. Wei, C. Wang, X. Chen and Y. Liao, Pulmonary Delivery of Dual-Targeted Nanoparticles Improves Tumor Accumulation and Cancer Cell Targeting by Restricting Macrophage Interception in Orthotopic Lung Tumors, Biomaterials, 2025, 315, 122955, DOI:10.1016/j.biomaterials.2024.122955.
- S. N. Kassaee, D. Richard, G. A. Ayoko and N. Islam, Lipid Polymer Hybrid Nanoparticles against Lung Cancer and Their Application as Inhalable Formulation, Nanomedicine, 2024, 19(25), 2113–2133, DOI:10.1080/17435889.2024.2387530.
- C. Liu, L. Chen, Y. Ma, K. Hu, P. Wu, L. Pan, H. Chen, L. Li, H. Hu and J. Zhang, Pulmonary Circulation-Mediated Heart Targeting for the Prevention of Heart Failure by Inhalation of Intrinsically Bioactive Nanoparticles, Theranostics, 2021, 11(17), 8550–8569, DOI:10.7150/thno.61875.
- H. Weng, W. Zou, F. Tian, H. Xie, A. Liu, W. Liu, Y. Liu, N. Zhou, X. Cai, J. Wu, Y. Zheng and X. Shu, Inhalable Cardiac Targeting Peptide Modified Nanomedicine Prevents Pressure Overload Heart Failure in Male Mice, Nat. Commun., 2024, 15(1), 6058, DOI:10.1038/s41467-024-50312-1.
- A. Alogna, F. P. Lo Muzio and D. Catalucci, Cardiovascular Inhalation for Targeted Drug Delivery in Cardiac Disease, Heart Fail. Rev., 2025, 30(5), 1103–1111, DOI:10.1007/s10741-025-10527-w.
- M. Miragoli, P. Ceriotti, M. Iafisco, M. Vacchiano, N. Salvarani, A. Alogna, P. Carullo, G. B. Ramirez-Rodríguez, T. Patrício, L. D. Esposti, F. Rossi, F. Ravanetti, S. Pinelli, R. Alinovi, M. Erreni, S. Rossi, G. Condorelli, H. Post, A. Tampieri and D. Catalucci, Inhalation of Peptide-Loaded Nanoparticles Improves Heart Failure, Sci. Transl. Med., 2018, 10(424), eaan6205, DOI:10.1126/scitranslmed.aan6205.
- H. Xu, Y. Zhu, L. Zhu, D. Wang, S. Lv, X. Li, C. Guo and Y. Li, Warning on the Inhalation of Silica Nanoparticles: Experimental Evidence for Its Easy Passage through the Air-Blood Barrier, Resulting in Systemic Distribution and Pathological Injuries, Chem.-Biol. Interact., 2025, 409, 111423, DOI:10.1016/j.cbi.2025.111423.
- M. Guérin and E. Lepeltier, Nanomedicines via the Pulmonary Route: A Promising Strategy to Reach the Target?, Drug Deliv. Transl. Res., 2024, 14(8), 2276–2297, DOI:10.1007/s13346-024-01590-1.
- A. J. Plaunt, T. L. Nguyen, M. R. Corboz, V. S. Malinin and D. C. Cipolla, Strategies to Overcome Biological Barriers Associated with Pulmonary Drug Delivery, Pharmaceutics, 2022, 14(2), 302, DOI:10.3390/pharmaceutics14020302.
- S. Anderson, P. Atkins, P. Bäckman, D. Cipolla, A. Clark, E. Daviskas, B. Disse, P. Entcheva-Dimitrov, R. Fuller, I. Gonda, H. Lundbäck, B. Olsson and J. Weers, Inhaled Medicines: Past, Present, and Future, Pharmacol. Rev., 2022, 74(1), 48–118, DOI:10.1124/pharmrev.120.000108.
- S. Dong, H. Fang, J. Zhu, Z. Wu, Y. Liu, J. Zhu, B. Ma, Q. Chen and Y. Yang, Inhalable siRNA Targeting IL-11 Nanoparticles Significantly Inhibit Bleomycin-Induced Pulmonary Fibrosis, ACS Nano, 2025, 19(2), 2742–2758, DOI:10.1021/acsnano.4c15130.
- B. B. Mendes, J. Conniot, A. Avital, D. Yao, X. Jiang, X. Zhou, N. Sharf-Pauker, Y. Xiao, O. Adir, H. Liang, J. Shi, A. Schroeder and J. Conde, Nanodelivery of Nucleic Acids, Nat. Rev. Methods Primers, 2022, 2(1), 24, DOI:10.1038/s43586-022-00104-y.
- A. Wietrzyk, A. Hutny, L. Jonderko, K. Biliński, M. Makieła, J. Kalinin, B. Jachimska and J. Kulbacka, Protein-Functionalized Nanoparticles: Emerging Strategies in Drug Delivery, Biomed. Pharmacother., 2025, 189, 118337, DOI:10.1016/j.biopha.2025.118337.
- Y. Santin, K. Formoso, F. Haidar, M. D. P. O. Fuentes, F. Bourgailh, N. Hifdi, K. Hnia, Y. Doghri, J. Resta, C. Champigny, S. Lechevallier, M. Détrait, G. Cousin, M. Bisserier, A. Parini, F. Lezoualc’h, M. Verelst and J. Mialet-Perez, Inhalation of Acidic Nanoparticles Prevents Doxorubicin Cardiotoxicity through Improvement of Lysosomal Function, Theranostics, 2023, 13(15), 5435–5451, DOI:10.7150/thno.86310.
- X. Liu, F. He and M. Liu, New Opportunities of Stimulus-Responsive Smart Nanocarriers in Cancer Therapy, Nano Mater. Sci., 2024, S2589965124001612, DOI:10.1016/j.nanoms.2024.10.013.
- R. K. Dey, B. Jana and D. G. Dastidar, Applications of Nanotechnology in the Treatment of Pulmonary Diseases, Vessel Plus, 2023, 7–17, DOI:10.20517/2574-1209.2023.06.
- B. Fongaro, B. Campara, G. Y. Moscatiello, A. De Luigi, D. Panzeri, L. Sironi, P. Bigini, G. Carretta, G. Miolo, G. Pasut and P. Polverino De Laureto, Assessing the Physicochemical Stability and Intracellular Trafficking of mRNA-Based COVID-19 Vaccines, Int. J. Pharm., 2023, 644, 123319, DOI:10.1016/j.ijpharm.2023.123319.
- M. D. Park, A. Silvin, F. Ginhoux and M. Merad, Macrophages in Health and Disease, Cell, 2022, 185(23), 4259–4279, DOI:10.1016/j.cell.2022.10.007.
- S. R. Hwang, K. Chakraborty, J. M. An, J. Mondal, H. Y. Yoon and Y. Lee, Pharmaceutical Aspects of Nanocarriers for Smart Anticancer Therapy, Pharmaceutics, 2021, 13(11), 1875, DOI:10.3390/pharmaceutics13111875.
- F. Ginhoux and M. Guilliams, Tissue-Resident Macrophage Ontogeny and Homeostasis, Immunity, 2016, 44(3), 439–449, DOI:10.1016/j.immuni.2016.02.024.
- H. H. Gustafson, D. Holt-Casper, D. W. Grainger and H. Ghandehari, Nanoparticle Uptake: The Phagocyte Problem, Nano Today, 2015, 10(4), 487–510, DOI:10.1016/j.nantod.2015.06.006.
- J. Fernandez Alarcon, P. Perez Schmidt, N. Panini, F. Caruso, M. B. Violatto, N. G. Sukubo, A. Martinez-Serra, C. B. Ekalle-Soppo, A. Morelli, G. Y. Moscatiello, C. Grasselli, A. Corbelli, F. Fiordaliso, J. Kelk, L. Petrosilli, G. d'Orazio, R. Mateu Ferrando, A. Verdaguer Ferrer, C. Fornaguera, L. Lay, S. Fumagalli, S. Recchia, M. P. Monopoli, L. Polito, P. Bigini and G. Sitia, Functional Polarization of Liver Macrophages by Glyco Gold Nanoparticles, Adv. Sci., 2025, 12(16), 2407458, DOI:10.1002/advs.202407458.
- S. S. Das, P. Bharadwaj, M. Bilal, M. Barani, A. Rahdar, P. Taboada, S. Bungau and G. Z. Kyzas, Stimuli-Responsive Polymeric Nanocarriers for Drug Delivery, Imaging, and Theragnosis, Polymers, 2020, 12(6), 1397, DOI:10.3390/polym12061397.
- L. Sun, H. Liu, Y. Ye, Y. Lei, R. Islam, S. Tan, R. Tong, Y.-B. Miao and L. Cai, Smart Nanoparticles for Cancer Therapy, Signal Transduction Targeted Ther., 2023, 8(1), 418, DOI:10.1038/s41392-023-01642-x.
- L. H. Rhym and D. G. Anderson, Nanoscale Delivery Platforms for RNA Therapeutics: Challenges and the Current State of the Art, Med, 2022, 3(3), 167–187, DOI:10.1016/j.medj.2022.02.001.
- S. Li, J. Su, W. Cai and J. Liu, Nanomaterials Manipulate Macrophages for Rheumatoid Arthritis Treatment, Front. Pharmacol, 2021, 12, 699245, DOI:10.3389/fphar.2021.699245.
- J. Chu, W. Zhang, Y. Liu, B. Gong, W. Ji, T. Yin, C. Gao, D. Liangwen, M. Hao, C. Chen, J. Zhuang, J. Gao and Y. Yin, Biomaterials-Based Anti-Inflammatory Treatment Strategies for Alzheimer's Disease, Neural Regen. Res., 2024, 19(1), 100–115, DOI:10.4103/1673-5374.374137.
- A. Joorabloo and T. Liu, Recent Advances in Nanomedicines for Regulation of Macrophages in Wound Healing, J. Nanobiotechnol., 2022, 20(1), 407, DOI:10.1186/s12951-022-01616-1.
- N. G. Sukubo, P. Bigini and A. Morelli, Nanocarriers and Macrophage Interaction: From a Potential Hurdle to an Alternative Therapeutic Strategy, Beilstein J. Nanotechnol., 2025, 16, 97–118, DOI:10.3762/bjnano.16.10.
- M. A. Kumar, S. K. Baba, H. Q. Sadida, S. Al. Marzooqi, J. Jerobin, F. H. Altemani, N. Algehainy, M. A. Alanazi, A.-B. Abou-Samra, R. Kumar, A. S. Al-Shabeeb Akil, M. A. Macha, R. Mir and A. A. Bhat, Extracellular Vesicles as Tools and Targets in Therapy for Diseases, Signal Transduction Targeted Ther., 2024, 9(1), 27, DOI:10.1038/s41392-024-01735-1.
- D. Gupta, O. P. B. Wiklander, M. J. A. Wood and S. El-Andaloussi, Biodistribution of Therapeutic Extracellular Vesicles, Extracell. Vesicles Circ. Nucleic Acids, 2023, 4(2), 170–190, DOI:10.20517/evcna.2023.12.
- X. Liang, D. Gupta, J. Xie, E. Wonterghem, L. Hoecke, J. Hean, Z. Niu, O. Wiklander, W. Zheng, R. Wiklander, R. He, D. Mammad, J. Bost, G. Zhou, H. Zhou, S. Roudi, A. Zickler, A. Gorgens, D. Hagey, O. G. De Jong, A. Uy, Y. Zong, I. Mager, C. Perez, T. Roberts, P. Vader, A. Fougerolles, M. Wood, R. Vandenbroucke, J. Nordin and S. El-Andaloussi, Engineering of Extracellular Vesicles for Efficient Intracellular Delivery of Multimodal Therapeutics Including Genome Editors, Nat. Commun., 2025, 16, 4028, DOI:10.1038/s41467-025-59377-y.
- I. K. Herrmann, M. J. A. Wood and G. Fuhrmann, Extracellular Vesicles as a Next-Generation Drug Delivery Platform, Nat. Nanotechnol., 2021, 16(7), 748–759, DOI:10.1038/s41565-021-00931-2.
- M. Samuels and G. Giamas, MISEV2023: Shaping the Future of EV Research by Enhancing Rigour, Reproducibility and Transparency, Cancer Gene Ther., 2024, 31(5), 649–651, DOI:10.1038/s41417-024-00759-7.
- C. Fusco, G. De Rosa, I. Spatocco, E. Vitiello, C. Procaccini, C. Frigè, V. Pellegrini, R. La Grotta, R. Furlan, G. Matarese, F. Prattichizzo and P. De Candia, Extracellular Vesicles as Human Therapeutics: A Scoping Review of the Literature, J. Extracell. Vesicles, 2024, 13(5), e12433, DOI:10.1002/jev2.12433.
- M. Van Delen, J. Derdelinckx, K. Wouters, I. Nelissen and N. Cools, A Systematic Review and Meta-analysis of Clinical Trials Assessing Safety and Efficacy of Human Extracellular Vesicle-based Therapy, J. Extracell. Vesicles, 2024, 13(7), e12458, DOI:10.1002/jev2.12458.
- 2021 Antibacterial Agents in Clinical and Preclinical Development: an Overview and Analysis, World Health Organization, Geneva, 1st edn, 2022 Search PubMed.
- Q. Xin, H. Shah, A. Nawaz, W. Xie, M. Z. Akram, A. Batool, L. Tian, S. U. Jan, R. Boddula, B. Guo, Q. Liu and J. R. Gong, Antibacterial Carbon-Based Nanomaterials, Adv. Mater., 2019, 31(45), 1804838, DOI:10.1002/adma.201804838.
- N. Santos, J. Fernandez Alarcon and M. Ahumada, Understanding the Toxicity of Metal-Based Nanomaterials: Current Insights, Challenges, and Future Directions, ACS Mater. Lett., 2025, 7(7), 2547–2564, DOI:10.1021/acsmaterialslett.5c00616.
- R. Mills-Goodlet, L. Johnson, I. J. Hoppe, C. Regl, M. Geppert, M. Schenck, S. Huber, M. Hauser, F. Ferreira, N. Hüsing, C. G. Huber, H. Brandstetter, A. Duschl and M. Himly, The Nanotopography of SiO2 Particles Impacts the Selectivity and 3D Fold of Bound Allergens, Nanoscale, 2021, 13(48), 20508–20520, 10.1039/D1NR05958K.
- B. Punz, L. Johnson, M. Geppert, H.-H. Dang, J. Horejs-Hoeck, A. Duschl and M. Himly, Surface Functionalization of Silica Nanoparticles: Strategies to Optimize the Immune-Activating Profile of Carrier Platforms, Pharmaceutics, 2022, 14(5), 1103, DOI:10.3390/pharmaceutics14051103.
- S. Wolfgruber, J. Rieger, O. Cardozo, B. Punz, M. Himly, A. Stingl, P. M. A. Farias, P. M. Abuja and K. Zatloukal, Antiviral Activity of Zinc Oxide Nanoparticles against SARS-CoV-2, Int. J. Mol. Sci., 2023, 24(9), 8425, DOI:10.3390/ijms24098425.
- B. Punz, C. Christ, A. Waldl, S. Li, Y. Liu, L. Johnson, V. Auer, O. Cardozo, P. M. A. Farias, A. C. D. S. Andrade, A. Stingl, G. Wang, Y. Li and M. Himly, Nano-Scaled Advanced Materials for Antimicrobial Applications – Mechanistic Insight, Functional Performance Measures, and Potential towards Sustainability and Circularity, Environ. Sci.: Nano, 2025, 12(3), 1710–1739, 10.1039/D4EN00798K.
- M. K. Singh, A Review of Digital PET-CT Technology: Comparing Performance Parameters in SiPM Integrated Digital PET-CT Systems, Radiography, 2024, 30(1), 13–20, DOI:10.1016/j.radi.2023.10.004.
- L. Lei, Y. Wang, A. Kuzmin, Y. Hua, J. Zhao, S. Xu and P. N. Prasad, Next Generation Lanthanide Doped Nanoscintillators and Photon Converters, eLight, 2022, 2(1), 17, DOI:10.1186/s43593-022-00024-0.
- W. Sun, Z. Zhou, G. Pratx, X. Chen and H. Chen, Nanoscintillator-Mediated X-Ray Induced Photodynamic Therapy for Deep-Seated Tumors: From Concept to Biomedical Applications, Theranostics, 2020, 10(3), 1296–1318, DOI:10.7150/thno.41578.
- M. Wang, C. Hu and Q. Su, Luminescent Lifetime Regulation of Lanthanide-Doped Nanoparticles for Biosensing, Biosensors, 2022, 12(2), 131, DOI:10.3390/bios12020131.
- N. Babayevska, M. Jancelewicz, I. Iatsunskyi, M. Jarek, I. Yakymenko, A. Padmanaban, O. Viahin, G. Terragni, C. Lowis, E. Auffray and O. Sidletskiy, BGO@ZnO Heterostructures for Ultrafast Scintillation Detectors, Crystals, 2025, 15(9), 820, DOI:10.3390/cryst15090820.
- N. V. Klassen, V. N. Kurlov, S. N. Rossolenko, O. A. Krivko, A. D. Orlov and S. Z. Shmurak, Scintillation Fibers and Nanoscintillators for Improving the Spatial, Spectrometric, and Time Resolution of Radiation Detectors, Bull. Russ. Acad. Sci. Phys., 2009, 73(10), 1369–1373, DOI:10.3103/S1062873809100141.
- A.-L. Bulin, C. Truillet, R. Chouikrat, F. Lux, C. Frochot, D. Amans, G. Ledoux, O. Tillement, P. Perriat, M. Barberi-Heyob and C. Dujardin, X-Ray-Induced Singlet Oxygen Activation with Nanoscintillator-Coupled Porphyrins, J. Phys. Chem. C, 2013, 117(41), 21583–21589, DOI:10.1021/jp4077189.
- A. Kamkaew, F. Chen, Y. Zhan, R. L. Majewski and W. Cai, Scintillating Nanoparticles as Energy Mediators for Enhanced Photodynamic Therapy, ACS Nano, 2016, 10(4), 3918–3935, DOI:10.1021/acsnano.6b01401.
- G. Li, C. Wang, B. Jin, T. Sun, K. Sun, S. Wang and Z. Fan, Advances in Smart Nanotechnology-Supported Photodynamic Therapy for Cancer, Cell Death Discov., 2024, 10(1), 466, DOI:10.1038/s41420-024-02236-4.
- L. M. Ailioaie, C. Ailioaie and G. Litscher, Fighting Cancer with Photodynamic Therapy and Nanotechnologies: Current Challenges and Future Directions, Int. J. Mol. Sci., 2025, 26(7), 2969, DOI:10.3390/ijms26072969.
- M. Chandran, M. Veerapandian, B. Dhanasekaran, S. Govindaraju and K. Yun, Advanced Nanomaterials for Health Monitoring and Diagnostics in Next-Generation Wearable Sensors, Mater. Sci. Eng. R Rep., 2025, 165, 101015, DOI:10.1016/j.mser.2025.101015.
- Y. Yang and W. Gao, Wearable and Flexible Electronics for Continuous Molecular Monitoring, Chem. Soc. Rev., 2019, 48(6), 1465–1491, 10.1039/C7CS00730B.
- J. Xie, Q. Chen, H. Shen and G. Li, Review—Wearable Graphene Devices for Sensing, J. Electrochem. Soc., 2020, 167(3), 037541, DOI:10.1149/1945-7111/ab67a4.
- S. Tang, Z. Liu and X. Xiang, Graphene Oxide Composite Hydrogels for Wearable Devices, Carbon Lett., 2022, 32(6), 1395–1410, DOI:10.1007/s42823-022-00402-1.
- G. Misia, C. Evangelisti, J. P. Merino, E. Pitzalis, A. M. Abelairas, J. Mosquera, A. Criado, M. Prato and A. Silvestri, Design and Optimization of an Electrochemical Sensor Based on Carbon Nanotubes for the Reliable Voltammetric Detection of Serotonin in Complex Biological Fluids, Carbon, 2024, 229, 119550, DOI:10.1016/j.carbon.2024.119550.
- U. Kilimci, B. Öndeş, Ç. Sunna, M. Uygun and D. Aktaş Uygun, Development of Label-Free Immunosensors Based on AuNPs-Fullerene Nanocomposites for the Determination of Cancer Antigen 125, Bioelectrochemistry, 2025, 163, 108863, DOI:10.1016/j.bioelechem.2024.108863.
- M. R. Kumalasari, R. Alfanaar and A. S. Andreani, Gold Nanoparticles (AuNPs): A Versatile Material for Biosensor Application, Talanta Open, 2024, 9, 100327, DOI:10.1016/j.talo.2024.100327.
- S. Xiao, K. Shang, W. Li and X. Wang, An Efficient Biosensor Based on the Synergistic Catalysis of Helicobacter Pylori Urease b Subunit and Nanoplatinum for Urease Inhibitors Screening and Antagonistic Mechanism Analyzing, Sens. Actuators, B, 2022, 355, 131284, DOI:10.1016/j.snb.2021.131284.
- J. Qin, H. Tang, G. Qu, K. Pan, K. Wei, J. Lv and J. Li, Preparation of Molecularly Imprinted Electrochemical Sensors for Selective Detection of Hydroxyl Radicals Based on Reduced Graphene Oxide Nanosilver (rGO/AgNPs) Composites, Microchem. J., 2024, 199, 110006, DOI:10.1016/j.microc.2024.110006.
- G. Sriramulu, R. Verma, K. R. Singh, P. Singh, C. S. Chakra, S. Mallick, R. P. Singh, K. Sadhana and J. Singh, Self-Assembled Copper Oxide Nanoflakes for Highly Sensitive Electrochemical Xanthine Detection in Fish-Freshness Biosensors, J. Mol. Struct., 2024, 1304, 137640, DOI:10.1016/j.molstruc.2024.137640.
- L. Wang, Y. Wei, X. Liu, J. Chen, C. Mao and B. Jin, Electrochemiluminescence Biosensor Based on Gold Nanoparticles Modulated Cathode AIE-Activated Metal-Organic Frameworks for the Ultrasensitive Detection of CA15-3, Biosens. Bioelectron., 2025, 282, 117465, DOI:10.1016/j.bios.2025.117465.
- X. Li, D. Su, Y. Gu, J. Zhang, S. Li, Y. Xiao, J. He, W. Wang and D. Li, Laser Fabrication of Epidermal Paper-Based Graphene Sensors, Appl. Mater. Today, 2024, 36, 102051, DOI:10.1016/j.apmt.2023.102051.
- F. Poletti, A. Scidà, B. Zanfrognini, A. Kovtun, V. Parkula, L. Favaretto, M. Melucci, V. Palermo, E. Treossi and C. Zanardi, Graphene-Paper-Based Electrodes on Plastic and Textile Supports as New Platforms for Amperometric Biosensing, Adv. Funct. Mater., 2022, 32(7), 2107941, DOI:10.1002/adfm.202107941.
- L. Fu, Q. Chen and L. Jia, Carbon Dots and Gold Nanoclusters Assisted Construction of a Ratiometric Fluorescent Biosensor for Detection of Gram-Negative Bacteria, Food Chem., 2022, 374, 131750, DOI:10.1016/j.foodchem.2021.131750.
- K. E. Sapsford, W. R. Algar, L. Berti, K. B. Gemmill, B. J. Casey, E. Oh, M. H. Stewart and I. L. Medintz, Functionalizing Nanoparticles with Biological Molecules: Developing Chemistries That Facilitate Nanotechnology, Chem. Rev., 2013, 113(3), 1904–2074, DOI:10.1021/cr300143v.
- Y. Wu, S.-F. Torabi, R. J. Lake, S. Hong, Z. Yu, P. Wu, Z. Yang, K. Nelson, W. Guo, G. T. Pawel, J. Van Stappen, X. Shao, L. M. Mirica and Y. Lu, Simultaneous Fe2+/Fe3+ Imaging Shows Fe3+ over Fe2+ Enrichment in Alzheimer's Disease Mouse Brain, Sci. Adv., 2023, 9(16), eade7622, DOI:10.1126/sciadv.ade7622.
- A. Hatamie, Advances and New Perspectives in Micro-Nanofabricated Sensors for Bioanalysis, Biosensors, 2024, 14(2), 87, DOI:10.3390/bios14020087.
- M. B. Kulkarni, N. H. Ayachit and T. M. Aminabhavi, Biosensors and Microfluidic Biosensors: From Fabrication to Application, Biosensors, 2022, 12(7), 543, DOI:10.3390/bios12070543.
- S. L. Mekuria, G. Li, Z. Wang, W. M. Girma, A. Li, M. He, H. Wang, M. M. A. Hameed, M. EL-Newehy, X. Shi and M. Shen, Dendrimer Nanoclusters Loaded with Gold Nanoparticles for Enhanced Tumor CT Imaging and Chemotherapy via an Amplified EPR Effect, J. Mater. Chem. B, 2024, 12(38), 9524–9532, 10.1039/D4TB01747A.
- S. Fiori, A. Scroccarello, F. Della Pelle, M. Del Carlo, E. Cozzoni and D. Compagnone, Integrating Electrochemical Sensors in Circular Economy: Biochar-Film Sensors Based on Paper Industry Waste for Agri-Food by-Product Valorization, Green Anal. Chem., 2025, 13, 100277, DOI:10.1016/j.greeac.2025.100277.
- E. Demir, K. K. Kırboga and M. Işık, Chapter 8 – An Overview of Stability and Lifetime of Electrochemical Biosensors, in Novel Nanostructured Materials for Electrochemical Bio-Sensing Applications, ed. J. G. Manjunatha, Elsevier, 2024, pp. 129–158, DOI:10.1016/B978-0-443-15334-1.00022-5.
- B. A. Taha, N. M. Ahmed, R. K. Talreja, A. J. Haider, Y. Al Mashhadany, Q. Al-Jubouri, A. B. Huddin, M. H. H. Mokhtar, S. Rustagi, A. Kaushik, V. Chaudhary and N. Arsad, Synergizing Nanomaterials and Artificial Intelligence in Advanced Optical Biosensors for Precision Antimicrobial Resistance Diagnosis, ACS Synth. Biol., 2024, 13(6), 1600–1620, DOI:10.1021/acssynbio.4c00070.
- G. Saorin, I. Caligiuri and F. Rizzolio, Microfluidic Organoids-on-a-Chip: The Future of Human Models, Semin. Cell Dev. Biol., 2023, 144, 41–54, DOI:10.1016/j.semcdb.2022.10.001.
- A. Saorin, G. Saorin, F. Duzagac, P. Parisse, N. Cao, G. Corona, E. Cavarzerani and F. Rizzolio, Microfluidic Production of Amiodarone Loaded Nanoparticles and Application in Drug Repositioning in Ovarian Cancer, Sci. Rep., 2024, 14(1), 6280, DOI:10.1038/s41598-024-55801-3.
- S. Shah, V. Dhawan, R. Holm, M. S. Nagarsenker and Y. Perrie, Liposomes: Advancements and Innovation in the Manufacturing Process, Adv. Drug Delivery Rev., 2020, 154–155, 102–122, DOI:10.1016/j.addr.2020.07.002.
- B. K. Johnson and R. K. Prud'homme, Flash NanoPrecipitation of Organic Actives and Block Copolymers Using a Confined Impinging Jets Mixer, Aust. J. Chem., 2003, 56(10), 1021, DOI:10.1071/CH03115.
- C. Devos, S. Mukherjee, P. Inguva, S. Singh, Y. Wei, S. Mondal, H. Yu, G. Barbastathis, T. Stelzer, R. D. Braatz and A. S. Myerson, Impinging Jet Mixers: A Review of Their Mixing Characteristics, Performance Considerations, and Applications, AIChE J., 2025, 71(1), e18595, DOI:10.1002/aic.18595.
- S. Mehraji and D. L. DeVoe, Microfluidic Synthesis of Lipid-Based Nanoparticles for Drug Delivery: Recent Advances and Opportunities, Lab Chip, 2024, 24(5), 1154–1174, 10.1039/D3LC00821E.
- D. Singh, Patented Technologies of Microfluidic Devices for Targeted Drug Delivery: A Revolution in Optimization, Microfluid. Nanofluid., 2025, 29(6), 39, DOI:10.1007/s10404-025-02814-7.
- S. E. Alavi, S. Alharthi, S. F. Alavi, S. Z. Alavi, G. E. Zahra, A. Raza and H. Ebrahimi Shahmabadi, Microfluidics for Personalized Drug Delivery, Drug Discovery Today, 2024, 29(4), 103936, DOI:10.1016/j.drudis.2024.103936.
- W. T. Poh and J. Stanslas, The New Paradigm in Animal Testing – “3Rs Alternatives.”, Regul. Toxicol. Pharmacol., 2024, 153, 105705, DOI:10.1016/j.yrtph.2024.105705.
- K. Rasmussen, P. Sayre, A. Kobe, M. Gonzalez and H. Rauscher, 25 Years of Research and Regulation: Is Nanotechnology Safe to Commercialize?, Front. Toxicol., 2025, 7, 1629813, DOI:10.3389/ftox.2025.1629813.
- M. G. Soliman, A. Martinez-Serra, G. Antonello, M. Dobricic, T. Wilkins, T. Serchi, I. Fenoglio and M. P. Monopoli, Understanding the Role of Biomolecular Coronas in Human Exposure to Nanomaterials, Environ. Sci.: Nano, 2024, 11(11), 4421–4448, 10.1039/D4EN00488D.
- D. Walczyk, F. B. Bombelli, M. P. Monopoli, I. Lynch and K. A. Dawson, What the Cell “Sees” in Bionanoscience, J. Am. Chem. Soc., 2010, 132(16), 5761–5768, DOI:10.1021/ja910675v.
- M. Barz, W. J. Parak and R. Zentel, Concepts and Approaches to Reduce or Avoid Protein Corona Formation on Nanoparticles: Challenges and Opportunities, Adv. Sci., 2024, 11(34), 2402935, DOI:10.1002/advs.202402935.
- J. S. Suk, Q. Xu, N. Kim, J. Hanes and L. M. Ensign, PEGylation as a Strategy for Improving Nanoparticle-Based Drug and Gene Delivery, Adv. Drug Delivery Rev., 2016, 99, 28–51, DOI:10.1016/j.addr.2015.09.012.
- L. Spinelli, P. D'Anna, E. Morretta, C. Cassiano, V. Piccolo, M. De Rosa, R. Amico, P. De Cicco, D. Brancaccio, C. Conte, A. Zampella, F. Quaglia and M. C. Monti, PEGylation-Driven Remodeling of the Protein Corona on PLGA Nanoparticles: Implications for Macrophage Recognition, Biomacromolecules, 2025, 26(12), 8522–8534, DOI:10.1021/acs.biomac.5c01369.
- K. Partikel, R. Korte, N. C. Stein, D. Mulac, F. C. Herrmann, H.-U. Humpf and K. Langer, Effect of Nanoparticle Size and PEGylation on the Protein Corona of PLGA Nanoparticles, Eur. J. Pharm. Biopharm., 2019, 141, 70–80, DOI:10.1016/j.ejpb.2019.05.006.
- D. Zhu, H. Yan, Z. Zhou, J. Tang, X. Liu, R. Hartmann, W. J. Parak, N. Feliu and Y. Shen, Detailed Investigation on How the Protein Corona Modulates the Physicochemical Properties and Gene Delivery of Polyethylenimine (PEI) Polyplexes, Biomater. Sci., 2018, 6(7), 1800–1817 RSC.
- A. Åkesson, M. Cárdenas, G. Elia, M. P. Monopoli and K. A. Dawson, The Protein Corona of Dendrimers: PAMAM Binds and Activates Complement Proteins in Human Plasma in a Generation Dependent Manner, RSC Adv., 2012, 2(30), 11245, 10.1039/c2ra21866f.
- B. Wang, Y. Sun, T. P. Davis, P. C. Ke, Y. Wu and F. Ding, Understanding Effects of PAMAM Dendrimer Size and Surface Chemistry on Serum Protein Binding with Discrete Molecular Dynamics Simulations, ACS Sustain. Chem. Eng., 2018, 6(9), 11704–11715, DOI:10.1021/acssuschemeng.8b01959.
- A. Almalik, H. Benabdelkamel, A. Masood, I. O. Alanazi, I. Alradwan, M. A. Majrashi, A. A. Alfadda, W. M. Alghamdi, H. Alrabiah, N. Tirelli and A. H. Alhasan, Hyaluronic Acid Coated Chitosan Nanoparticles Reduced the Immunogenicity of the Formed Protein Corona, Sci. Rep., 2017, 7(1), 10542, DOI:10.1038/s41598-017-10836-7.
- A. E. Caprifico, P. J. S. Foot, E. Polycarpou and G. Calabrese, Overcoming the Protein Corona in Chitosan-Based Nanoparticles, Drug Discovery Today, 2021, 26(8), 1825–1840, DOI:10.1016/j.drudis.2021.04.014.
- R. Rampado, S. Crotti, P. Caliceti, S. Pucciarelli and M. Agostini, Recent Advances in Understanding the Protein Corona of Nanoparticles and in the Formulation of “Stealthy” Nanomaterials, Front. Bioeng. Biotechnol., 2020, 8, 166, DOI:10.3389/fbioe.2020.00166.
- V. Castagnola, V. Tomati, L. Boselli, C. Braccia, S. Decherchi, P. P. Pompa, N. Pedemonte, F. Benfenati and A. Armirotti, Sources of Biases in the in Vitro Testing of Nanomaterials: The Role of the Biomolecular Corona, Nanoscale Horiz., 2024, 9(5), 799–816, 10.1039/D3NH00510K.
- A. Martinez-Serra, A. Saorin, A. Serrano-Lotina, A. Subrati, M. G. Soliman, D. Hristozov, M. A. Bañares, P. Demokritou and M. P. Monopoli, Dispersion Protocols Have Minimal Impact on the Biomolecular Corona of Advanced Nanomaterials in Cell Culture Assays, NanoImpact, 2025, 38, 100560, DOI:10.1016/j.impact.2025.100560.
- O. Vilanova, A. Martinez-Serra, M. P. Monopoli and G. Franzese, Characterizing the Hard and Soft Nanoparticle-Protein Corona with Multilayer Adsorption, Front. Nanotechnol., 2025, 6, 1531039, DOI:10.3389/fnano.2024.1531039.
- C. Braccia, V. Castagnola, E. Vázquez, V. J. González, F. Loiacono, F. Benfenati and A. Armirotti, The Lipid Composition of Few Layers Graphene and Graphene Oxide Biomolecular Corona, Carbon, 2021, 185, 591–598, DOI:10.1016/j.carbon.2021.09.052.
- S. Wan, P. M. Kelly, E. Mahon, H. Stöckmann, P. M. Rudd, F. Caruso, K. A. Dawson, Y. Yan and M. P. Monopoli, The “Sweet” Side of the Protein Corona: Effects of Glycosylation on Nanoparticle–Cell Interactions, ACS Nano, 2015, 9(2), 2157–2166, DOI:10.1021/nn506060q.
- K. Faserl, A. J. Chetwynd, I. Lynch, J. A. Thorn and H. H. Lindner, Corona Isolation Method Matters: Capillary Electrophoresis Mass Spectrometry Based Comparison of Protein Corona Compositions Following On-Particle versus In-Solution or In-Gel Digestion, Nanomaterials, 2019, 9(6), 898, DOI:10.3390/nano9060898.
- A. Saorin, A. Martinez-Serra, G. J. Paparoni-Bruzual, M. Crozzolin, V. Lombardi, M. Back, P. Riello, M. P. Monopoli and F. Rizzolio, Understanding the Impact of Silica Nanoparticles in Cancer Cells through Physicochemical and Biomolecular Characterizations, Mater. Adv., 2024, 5(12), 5106–5117, 10.1039/D4MA00084F.
- A. Saorin, A. Subrati, A. Martinez-Serra, B. Alonso, M. Henry, P. Meleady, S. E. Moya and M. P. Monopoli, A Redefined Protocol for Protein Corona Analysis on Graphene Oxide, ACS Nanosci. Au, 2025, 5c00052, DOI:10.1021/acsnanoscienceau.5c00052.
- G. M. DeLoid, J. M. Cohen, G. Pyrgiotakis and P. Demokritou, Preparation, Characterization, and in Vitro Dosimetry of Dispersed, Engineered Nanomaterials, Nat. Protoc., 2017, 12(2), 355–371, DOI:10.1038/nprot.2016.172.
- Plastics – The facts. An Analysis of European Plastics Production, Demand and Waste Data, 2017.
- R. Geyer, J. R. Jambeck and K. L. Law, Production, Use, and Fate of All Plastics Ever Made, Sci. Adv., 2017, 3(7), e1700782, DOI:10.1126/sciadv.1700782.
- P. Schwabl, S. Köppel, P. Königshofer, T. Bucsics, M. Trauner, T. Reiberger and B. Liebmann, Detection of Various Microplastics in Human Stool: A Prospective Case Series, Ann. Intern. Med., 2019, 171(7), 453–457, DOI:10.7326/M19-0618.
- V. Forest and J. Pourchez, Can the Impact of Micro- and Nanoplastics on Human Health Really Be Assessed Using in Vitro Models? A Review of Methodological Issues, Environ. Int., 2023, 178, 108115, DOI:10.1016/j.envint.2023.108115.
- M. G. Soliman, A. Martinez-Serra, M. Dobricic, D. N. Trinh, J. Cheeseman, D. I. R. Spencer and M. P. Monopoli, Protocols for Isolation and Characterization of Nanoparticle Biomolecular Corona Complexes, Front. Toxicol., 2024, 6, 1393330, DOI:10.3389/ftox.2024.1393330.
- S. Ali, D. A. Bukhari and A. Rehman, Call for Biotechnological Approach to Degrade Plastic in the Era of COVID-19 Pandemic, Saudi J. Biol. Sci., 2023, 30(3), 103583, DOI:10.1016/j.sjbs.2023.103583.
- I. Brandts, J. Lima, I. Garcia-Meilán, A. R. Khansari, M. Llorca, M. Farré, A. Tvarijonaviciute, J. C. Balasch and M. Teles, Bioaccumulation and Homeostatic Alterations in Trout Exposed to a Sublethal Dose of Polystyrene Nanoplastics, J. Hazard. Mater. Adv., 2025, 19, 100836, DOI:10.1016/j.hazadv.2025.100836.
- G. F. Schirinzi, I. Pérez-Pomeda, J. Sanchís, C. Rossini, M. Farré and D. Barceló, Cytotoxic Effects of Commonly Used Nanomaterials and Microplastics on Cerebral and Epithelial Human Cells, Environ. Res., 2017, 159, 579–587, DOI:10.1016/j.envres.2017.08.043.
- G. Polo, F. Lionetto, M. E. Giordano and M. G. Lionetto, Interaction of Micro- and Nanoplastics with Enzymes: The Case of Carbonic Anhydrase, Int. J. Mol. Sci., 2024, 25(17), 9716, DOI:10.3390/ijms25179716.
- M. Viana, F. S. Tonin and C. Ladeira, Assessing the Impact of Nanoplastics in Biological Systems: Systematic Review of In Vitro Animal Studies, J. Xenobiot., 2025, 15(3), 75, DOI:10.3390/jox15030075.
- M. Sousa De Almeida, E. Susnik, B. Drasler, P. Taladriz-Blanco, A. Petri-Fink and B. Rothen-Rutishauser, Understanding Nanoparticle Endocytosis to Improve Targeting Strategies in Nanomedicine, Chem. Soc. Rev., 2021, 50(9), 5397–5434, 10.1039/D0CS01127D.
- G. Y. Moscatiello, C. Natale, M. Inserra, A. Morelli, L. Russo, N. Battajini, L. Sironi, D. Panzeri, A. Corbelli, A. De Luigi, F. Fiordaliso, G. Candiani, P. Bigini and L. Diomede, The Surface Charge Both Influences the Penetration and Safety of Polystyrene Nanoparticles despite the Protein Corona Formation, Environ. Sci.: Nano, 2025, 12(5), 2857–2870, 10.1039/D4EN00962B.
- N. Chaffey, B. Alberts, A. Johnson, J. Lewis, M. Raff, K. Roberts and P. Walter, Molecular Biology of the Cell, 4th edn, 2003 Search PubMed.
- T. Kaletta and M. O. Hengartner, Finding Function in Novel Targets: C. Elegans as a Model Organism, Nat. Rev. Drug Discov., 2006, 5(5), 387–399, DOI:10.1038/nrd2031.
- L. Gonzalez-Moragas, A. Roig and A. C. Laromaine, Elegans as a Tool for in Vivo Nanoparticle Assessment, Adv. Colloid Interface Sci., 2015, 219, 10–26, DOI:10.1016/j.cis.2015.02.001.
- F. Côa, L. D. S. Bortolozzo, D. S. Ávila, A. G. Souza Filho and D. S. T. Martinez, Toxicology of Carbon Nanomaterials in the Caenorhabditis Elegans Model: Current Status, Characterization, and Perspectives for Testing Harmonization, Front. Carbon, 2023, 2, 1241637, DOI:10.3389/frcrb.2023.1241637.
- N. A. Swierczek, A. C. Giles, C. H. Rankin and R. A. Kerr, High-Throughput Behavioral Analysis in C. Elegans, Nat. Methods, 2011, 8(7), 592–598, DOI:10.1038/nmeth.1625.
- L. Frézal and M.-A. C. Félix, Elegans Outside the Petri Dish, eLife, 2015, 4, e05849, DOI:10.7554/eLife.05849.
- G. Sitia, F. Fiordaliso, M. B. Violatto, J. F. Alarcon, L. Talamini, A. Corbelli, L. M. Ferreira, N. L. Tran, I. Chakraborty, M. Salmona, W. J. Parak, L. Diomede and P. Bigini, Food-Grade Titanium Dioxide Induces Toxicity in the Nematode Caenorhabditis Elegans and Acute Hepatic and Pulmonary Responses in Mice, Nanomaterials, 2022, 12(10), 1669, DOI:10.3390/nano12101669.
- Y. Li, L. Zhong, L. Zhang, X. Shen, L. Kong and T. Wu, Research Advances on the Adverse Effects of Nanomaterials in a Model Organism, Caenorhabditis Elegans, Environ. Toxicol. Chem., 2021, 40(9), 2406–2424, DOI:10.1002/etc.5133.
- T. Puzyn, A. Gajewicz, D. Leszczynska and J. Leszczynski, Nanomaterials – the Next Great Challenge for Qsar Modelers, in Recent Advances in QSAR Studies: Methods and Applications, ed. T. Puzyn, J. Leszczynski and M. T. Cronin, Springer Netherlands, Dordrecht, 2010, pp. 383–409, DOI:10.1007/978-1-4020-9783-6_14.
- G. Tsiliki, P. Nymark, P. Kohonen, R. Grafström and H. Sarimveis, Enriching Nanomaterials Omics Data: An Integration Technique to Generate Biological Descriptors, Small Methods, 2017, 1(11), 1700139, DOI:10.1002/smtd.201700139.
- M. R. Findlay, D. N. Freitas, M. Mobed-Miremadi and K. E. Wheeler, Machine Learning Provides Predictive Analysis into Silver Nanoparticle Protein Corona Formation from Physicochemical Properties, Environ. Sci.: Nano, 2018, 5(1), 64–71, 10.1039/C7EN00466D.
- X.-Q. Han, X.-D. Wang, M.-Y. Xu, Z. Feng, B.-W. Yao, P.-J. Guo, Z.-F. Gao and Z.-Y. Lu, AI-Driven Inverse Design of Materials: Past, Present, and Future, Chin. Phys. Lett., 2025, 42(2), 027403, DOI:10.1088/0256-307X/42/2/027403.
- B. Sanchez-Lengeling and A. Aspuru-Guzik, Inverse Molecular Design Using Machine Learning: Generative Models for Matter Engineering, Science, 2018, 361(6400), 360–365, DOI:10.1126/science.aat2663.
- J. Wang, Y. Wang and Y. Chen, Inverse Design of Materials by Machine Learning, Materials, 2022, 15(5), 1811, DOI:10.3390/ma15051811.
| This journal is © The Royal Society of Chemistry 2026 |



