Open Access Article
Open Access ArticleElectropolymerization of Au nanoparticle incorporated poly(dopamine) thin-films at a micro liquid|liquid interface
Leila
Nazari
a,
Fanqi
Kong
 b,
E. Bradley
Easton
b,
E. Bradley
Easton
 b and
Talia Jane
Stockmann
b and
Talia Jane
Stockmann
 *a
*a
aDepartment of Chemistry, Core Science Facility, Memorial University of Newfoundland, 45 Artic Ave, St. John's, NL A1C 5S7, Canada. E-mail: tstockmann@mun.ca
bElectrochemical Materials Lab, Faculty of Science (Chemistry), Ontario Tech University, 2000 Simcoe Street North, Oshawa, ON L1G 0C5, Canada
First published on 14th January 2026
Abstract
Dopamine (DA) is an important biomolecule of the central nervous system (CNS) as well as a monomer incorporated into melanin, a macromolecule often associated with skin pigmentation. Herein, we have investigated the electropolymerization of DA to form polydopamine (PDA) at a micro interface between two immiscible electrolyte solutions (micro-ITIES) between water|1,2-dichloroethane (w|DCE) under external, electrochemical control. DA oxidation to form PDA was achieved via the use of an ionic liquid composed of trihexyltetradecylphosphonium (P66614) paired with AuCl4− and dissolved in the DCE phase. AuCl4− acts as a strong oxidizer, accepting electrons from DA to form Au nanoparticles (NPs) that become incorporated into the growing PDA matrix. Au NP/PDA electrosynthesis was found to improve with increasing aqueous phase pH and was discovered to create a delicate, free-standing film. Au NP/PDA was used to modify a glassy-carbon electrode (GCE) and employed as a platform for DA detection. This proof-of-concept DA-biosensor demonstrated quasi-reversible DA oxidation and a good limit-of-detection (LOD) and linear-dynamic-range of 0.27 µM and 0.2–20 µM, respectively, using differential pulse voltammetry (DPV).
Introduction
Dopamine (DA) is ubiquitous in nature, often found as a neurotransmitter that participates in most cognitive or movement control events in humans1 within the central nervous system (CNS). It can also be found outside the CNS and is the chemical basis of melanin, including eumelanin, pheomelanin, and neuromelanin.2,3 DA is synthesized by the body from tyrosine, which is first converted to L-3,4-dihydroxyphenylalanine, a.k.a. Levodopa (L-DOPA), before being transformed to DA. Melanin is a heterogeneous polymer that can appear dark brown to black or even yellow.2 Eumelanin is composed of monomers of 5,6-dihydroxyindole (DHI) and 5,6-dihydroxyindole-2-carboxylic acid (DHICA).4 The formation of DHI is achieved through an intramolecular 1,4-Michael addition of the amine towards the catechol ring within DA. DHICA is similarly generated, however, from L-DOPA. All of these are derivatives of DA; thus, melanin, a complex macromolecule is partially composed of polydopamine (PDA). Polymerization off of the aromatic, catechol ring can generate a conductive polymer. Neuromelanin, a heteropolymer composed partly of the monomer DHI, has been found in the substantia nigra of the brain, and its loss or degradation has been linked to Parkinson's5 and Alzheimer's6 diseases. Melanin coordinates well to metal ions, including Fe, Cu, and some heavy metals like Pb or Hg,7 through the hydroxyl groups of the catechol moiety. It has been proposed that neuromelanin may act as a protective barrier for the brain by coordinating to these metal ions that are toxic to neurons. However, as neuromelanin breaks down, this protection is lost, potentially leaking toxic metal ions into neural tissue.7Outside the CNS, different variations of melanin are employed throughout the animal kingdom as skin pigmentation8 owing to their broad optical absorption cross-section that inhibits radiative cell damage. Since the discovery of PDA as an adhesive coating on mussels,9 it has been increasingly employed in anti-corrosion, materials science studies,10,11 and even as an energy storage material.12 Meanwhile, the detection of DA for neuroscience investigations has been of critical importance for decades.13 In the 1980s, Wightman and Ewing developed carbon fiber microelectrodes14–16 as in vivo electroanalytical probes to directly measure DA. This has since been extended to the nanoscale, particularly for the real-time monitoring of chemical communication through fundamental understanding of exocytosis.17
A number of in vitro analytical methods have arisen for DA quantification, including exploiting the inherent fluorescence of PDA,18,19 or via liquid chromatography-mass spectroscopy,20 as well as many other approaches that often combine two or more sensing platforms.21 Nevertheless, electrochemical detection is the only present method for real-time monitoring in vivo. Shao's group demonstrated that DA could be detected at an interface between two immiscible electrolyte solutions (ITIES) through facilitated ion transfer reactions employing 18-crown-6 ether, while ensuring that DA was in a protonated, charged state.22 More recently, Shen's group23 employed a nano-ITIES between 2-nitrophenyl octyl ether (NPOE) and water for the detection of another neurotransmitter, acetylcholine, through simple ion transfer. The ITIES was considered advantageous for two predominant reasons. First, on unmodified electrodes, DA oxidation is often irreversible and can cause fouling of the electrode surface through the formation of PDA – this cannot happen at a liquid|liquid interface when DA undergoes simple or facilitated ion transfer.24 Second, DA oxidation occurs in roughly the same potential region as other common biomolecules, particularly ascorbic acid (AA) and uric acid (UA), that can interfere with its quantification;25–27 this type of interference is overcome at the liquid|liquid interface since the charge transfer mechanism is fundamentally different to a redox process. Research has continued at the solid/electrolyte interface using modified electrodes. Electrodes incorporating conjugated, aromatic systems such as graphene or graphene oxide have been shown to improve the electrochemical reversibility of DA oxidation as well as enhance the selectivity and sensitivity of the electrochemical sensing platform.27–30 This can also be achieved via inclusion of metal nanoparticles (NPs) such as Au.31 These platforms often rely on modifying glassy carbon electrodes (GCEs). GCEs are advantageous as they have very low background currents and are poor electrocatalysts; this provides a good baseline, since any enhancement is brought about by the modification itself. For in vivo continuous neurotransmitter monitoring, however, this is not ideal since hard, rigid objects like the GCE or Pt ultramicroelectrodes, such as the ones used in deep-brain-stimulation (DBS) in the treatment of Parkinson's disease, could be isolated from the body by scar tissue.32 This has the potential to limit the long-term efficacy of DBS.32,33 So, research into soft polymer electrodes for bioimplantation has become a focus.34
Electropolymerization of conductive polymer films at a liquid|liquid interface offers several advantages over solid/electrolyte ones. Any film formed at a solid electrode will be influenced by its baseline surface morphology, and it cannot be removed without significantly damaging the film. This is not the case at a liquid|liquid interface; whereby, films are easily removed and used as free-standing electrodes.34–41 Moreover, the liquid|liquid interface is molecularly sharp and smooth, so this likely exhibits little morphological influence on the film. Electrochemical control is achieved by externally manipulating the Galvani potential difference (ΔwDCEϕ) across the water|oil (w|o) interface, ϕw – ϕo = ΔwDCEϕ, using an electrode immersed in either phase.42 Lehane et al.34 demonstrated the electropolymerization of 3,4-ethyldioxythiophene (EDOT) at a large (cm scale) water|α,α,α-trifluorotoluene (w|TFT) interface using Ce4+(aq) as the electron acceptor, which generated a free-standing PEDOT, conductive polymer film. Our group has explored the electrogeneration of nanocomposites through the simultaneous formation of Au and Cu metal NPs within a growing conductive polymer matrix using monomers such as 9-vinylcarbazole,39 2,2′:5′,2′′-terthiophene (TT),41,43 as well as more exotic molecules such as dithiafulvenyl-functionalized pyrenes.40
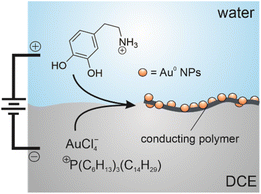
Herein, we investigated the electrogeneration of Au NP-incorporated PDA at a micro-ITIES. In this system, DA is dissolved in water and injected into the back of a specialized pipette holder. The pipette is composed of borosilicate glass with a pulled end that incorporates a ∼500 µm long microchannel that is 25 µm in diameter.40,44 The tip is then submerged inside a 1,2-dichloroethane (DCE) solution containing a hydrophobic Au salt, P66614AuCl4 (trihexyltetradecylphosphonium tetrachloroaurate) while the ITIES is maintained at the end of the microchannel. AuCl4− acts as an excellent oxidizing agent, accepting electrons from DA in a heterogeneous electron transfer reaction across the ITIES (see Scheme 1). The Au NP/PDA film was successfully generated using cyclic voltammetry; however, it was also quite soft and fragile. It was removed from the interface by ejecting a droplet from the tip of the pipette using a syringe on the back of the specialized holder.40,41 This was used to deposit the nanocomposite on a GCE to investigate its use as a DA biosensor. Here it showed a good limit-of-detection and linear range using differential pulse voltammetry (DPV).
Experimental
All chemicals were used as received unless described otherwise with aqueous solutions prepared using ultrapure water obtained from a MilliQ system (18.2 MΩ cm). Dopamine hydrogen chloride (≥98%), potassium tetrachloroaurate (KAuCl4, >98%), 1,2-dichloroethane (DCE, ≥99.0%), trihexyltetradecylphosphonium bromide (P66614Br, >95%) trioctylphosphine (>97%), bromooctane (99%), hydrochloric acid solution (HCl, >37%), potassium hydroxide (≥85%), potassium phosphate monobasic (≥99.0%), ferrocene methanol (FcCH2OH, 97%), and potassium phosphate dibasic (≥98%) were sourced from Sigma-Aldrich. Potassium chloride (KCl, 99.0%) was bought from ACP. Tetrakis(pentafluorophenyl)borate lithium etherate (LiTB, ≥99%) was obtained from Boulder Scientific. The ionic liquid (IL) P8888TB (tetraoctylphosphonium tetrakis(pentafluorophenyl)borate) was synthesized as described previously45 and used as supporting electrolyte in the DCE phase.A second IL, trihexyltetradecylphosphonium tetrachloroaurate (P66614AuCl4), was prepared via a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 metathesis reaction between P66614Br and KAuCl4 dissolved in CH2Cl2. The reactants were allowed to stir overnight, and the reaction mixture was filtered under reduced pressure through Whatman filter paper, removing the solid KBr precipitate. The filtrate containing P66614AuCl4 was washed 3× with MilliQ water, then excess CH2Cl2 was removed on a roto-evaporator followed by drying on a Schlenk line under vacuum.
1 metathesis reaction between P66614Br and KAuCl4 dissolved in CH2Cl2. The reactants were allowed to stir overnight, and the reaction mixture was filtered under reduced pressure through Whatman filter paper, removing the solid KBr precipitate. The filtrate containing P66614AuCl4 was washed 3× with MilliQ water, then excess CH2Cl2 was removed on a roto-evaporator followed by drying on a Schlenk line under vacuum.
CHI6059 potentiostat from CH Instruments was used throughout for electrochemical measurements. Micro-interfacial experiments were conducted in 2-electrode mode using a micropipette installed within a specialized holder described previously by us.40,41 The working electrode (WE) of the potentiostat was connected to an Ag wire (Delta Scientific) via a modified SMA connector integrated within the holder. The Ag wire was immersed in the aqueous solution which was back filled into the holder/capillary. An Au wire (Delta Scientific) was used as the counter/reference electrode (CE/RE) in the DCE phase. Unless otherwise indicated, the micro-ITIES was 25 µm in diameter and positioned at the tip of the micropipette. Scheme 2 describes the electrolytic cell employed at the micro liquid|liquid interface.
Glassy-carbon electrodes (GCE, CH Instruments) 4 mm outer diameter were employed for large, solid/solution electrode experiments in 3-electrode mode, whereby a Pt-wire counter and Ag/AgCl reference electrodes completed the cell. GCEs were tested with and without modification by the Au NP/PDA nanocomposite.
X-ray photoelectron spectroscopy was performed using a Thermo Scientific Nexsa instrument equipped with a monochromatic Al Kα X-ray source (1486.7 eV) and a 180° double focusing hemispherical analyser with a 128-channel detector with effective charge compensation. The instrument was equipped with a flood gun, which provides a steady flow of low-energy electrons to compensate plausible charging. The adventitious carbon peak at 284.8 eV was used to calibrate the binding energies.
A Varian 6000i UV-Vis-NIR and Bruker INVENIO-R spectrometers were used for UV-vis and FTIR studies, respectively.
Scanning electrochemical microscopy (SECM) was performed using an Elproscan (Heka Electroniks) in 3-electrode mode; whereby, an inlaid disc carbon fibre ultramicroelectrode (UME) served as the working electrode, Pt wire as counter, paired with an Ag/AgCl reference electrode.
Results and discussion
Scheme 1 depicts the electrosynthetic configuration; whereby, DA and P66614AuCl4 were dissolved in the aqueous and DCE phases, respectively. Owing to the intermediate hydrophobicity/hydrophilicity of AuCl4−,39,40,44,48–51 it can partition from DCE to water and undergo homogeneous electron transfer, initiating bulk polymerization of DA. This partitioning is likely mitigated somewhat by the small interfacial size and experimental configuration that limits mixing during setup.52 Liquid|liquid interfacial electropolymerization of DA was first explored voltammetrically. Fig. 1 shows the cyclic voltammogram (CV) recorded using Cell 1 without DA added to the aqueous phase. The pulse was initiated at roughly −0.15 V and swept towards positive potentials. A sigmoidal wave was observed with a half-wave potential (ΔwDCEϕ1/2) of 0.185 V which is the simple ion transfer of AuCl4− from DCE → w.44,48,50 The sharp increase in the current at ∼0.6 V is the limit of the polarizable potential window (PPW) that is associated with the simple ion transfer of the supporting electrolyte; in this case, K+ transfer from w → DCE as well as TB− from DCE → w.42,53 During the reverse pulse towards negative potentials, a peak-shaped wave was recorded with a peak-potential (ΔwDCEϕp) ≈ 0.126 V which is AuCl4− transfer back from w → DCE. If one assumes Randles-Ševčík-like behaviour,54 then ΔwDCEϕ1/2 ≈ ΔwDCEϕp ± (0.0285 V) × z−1 ≈ 0.154 V. Here, z is the charge of the ion undergoing transfer. The asymmetric ion transfer profile (peak-shaped from w → DCE, and sigmoidal from DCE → w) is due to the pipette's unique geometry, which results in linear diffusion inside and hemispherical diffusion outside.55 Thus, the former resembles the signal profile common at larger electrodes, while the latter is more analogous to an inlaid-disc ultramicroelectrode (UME). Continuing to scan negatively, the other edge of the PPW is reached at roughly −0.3 V with an increase in the negative current signal associated with the ion transfer of the other supporting electrolyte components, i.e., P8888+/P66614+ from DCE → w and Cl− from w → DCE. These voltammetric results for simple ion transfer agree well with previous reports in which the micro-ITIES is positioned at the tip of a pulled glass capillary.23,39,43,44,48,50,51 All voltammograms recorded at the liquid|liquid interface have been referenced using the AuCl4− ion transfer potential which was assumed to be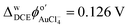 .44
.44
 | ||
| Fig. 1 Cyclic voltammogram (CV) obtained using Cell 1 with no DA added to the aqueous phase, i.e., a blank CV, measured at 0.020 V s−1. | ||
AuCl4− can undergo ligand speciation with increasing pH above 2 forming AuCl4−γ(OH)γ−; whereby, γ is the stoichiometric equivalents of Cl− ligands replaced with OH− which increases concomitantly with pH.39,44,49,56 The presence of AuCl4−γ(OH)γ−, however, results in a second ion transfer signal at slightly more negative potentials relative to that for AuCl4−. This was not observed at pH 2; however, as the pH was increased a second peak emerges at pH 8 and 10, while a third appears at pH 11.5, see Fig. S1 of the SI. These have been tentatively labelled as AuCl3OH− and AuCl2(OH)2−, respectively. Because AuCl4− is present initially in the DCE phase, it will only undergo ligand speciation when it transfers into the water phase. The local pH in the vicinity of the ITIES may vary from the bulk. In the work of Luty-Błocho et al.,57 AuCl4−, AuCl3OH−, AuCl2(OH)2−, and AuCl(OH)3− were calculated to be the dominant species at pH < 5, pH 6, pH 7, and pH > 9, respectively. Since only 3 peaks were observed over the course of increasing the pH, it was decided to label them according to the first Au-ligand species described by Luty-Błocho et al. This is speculative and awaits future work incorporating tandem spectroscopy to confirm with certainty which peak – beyond that of AuCl4− – belongs to which species.
Next, DA was added to the aqueous phase starting at a concentration of 20 mM while adjusting the pH of the aqueous phase via additions of HCl or KOH. Scheme 3 shows the 3-step deprotonation mechanism (reaction [1]) whose three pKa's are 9.05, 10.58, and 12.07 as described by Sanchez-Riviera et al.47 Thus, at pH < 9, H3DA+ (see Scheme 3) should be the dominant species, while at roughly pH 9–10, 10–13, and >13, H2DA, HDA−, and DA2−, respectively, are the dominant protonation states. The remaining hydroxyl-proton is likely stabilized via hydrogen bonding to the adjacent –O− for the H2DA and HDA− forms.46 Also in Scheme 3 are reactions [2] and [3] which show the oxidation of DA to dopamine quinone (DAQ) as well as the intramolecular 1,4-Michael addition forming the indole,58 respectively. Polymerization of DA is complex and still under scrutiny;59,60 however, it likely occurs off of the 2, 3, 4, and 7 positions of the indole, i.e., DHI and I56D, as well as the 3 and 6 positions of DA or DAQ.
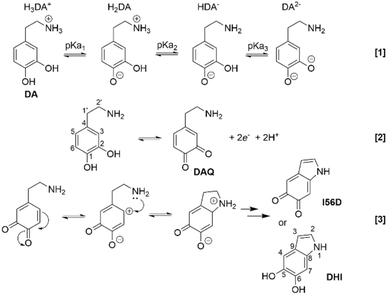 | ||
| Scheme 3 Reaction [1] depicts the dopamine (DA) deprotonation mechanism with changing pH; whereby, pKa's 1–3 are 9.05, 10.58, and 12.07 as reported by Sanchez-Riviera et al.46,47 and the different protonated states have been labeled H3DA+ to DA2−. Reaction [2] shows the 2-electron concerted oxidation of DA to dopamine quinone (DAQ) as well as the carbon numbering scheme within the DA molecule. Enolate formation and subsequent intramolecular 1,4-Micheal addition to form the indole has been drawn in reaction [3]. The numbering scheme for the indole is also given. Abbreviations: DHI = 5,6-dihydroxyindole; I56D: indole-5,6-dione. | ||
Fig. 2 depicts the voltammograms recorded at 0.020 V s−1 while changing the pH of the aqueous phase from 2 to 11.5. Fig. 2A–C and G–I show the first voltammetric scan, while Fig. 2D–F and J–L plot each subsequent 5th scan up to the 20th. At pH's greater than 8 and in the presence of oxygen, it was observed that DA polymerized spontaneously in the aqueous phase forming a black precipitate that filled and clogged the pipette. This is in good agreement with previous reports of DA stability at high pH that has been exploited to coat surfaces in the past.8,12,46,59,60 Thus, to ensure that PDA was not forming spontaneously,46 the aqueous phase was purged with N2 gas before adding DA and injecting the solution into the micropipette when operating at pH ≥ 5 just to err on the side of caution.
Examining Fig. 2A with the aqueous pH = 2, during the first voltammetric scan, the simple ion transfer signal for AuCl4− transfer has been replaced by a sigmoidal wave in both positive and negative potential sweep directions at ΔwDCEϕ1/2 ≈ 0.09 V. After the first CV cycle, a peak-shaped wave grows with ΔwDCEϕp = 0.11 V during the scan from positive to negative potentials. The peak-shaped wave quickly increases in current intensity achieving a magnitude of ∼4 nA relative to the baseline current. This is consistent with the transfer of a negative charge from w → DCE. Thus, we hypothesize the initial steady-state voltammetric response is likely owing to heterogeneous electron transfer from DA(aq) to AuCl4−(DCE). It is also possible that the lack of a peak-shaped wave during the negative potential scan is owing to the homogeneous consumption of AuCl4− through uncontrolled electropolymerization with DA after it transfers from DCE → w. After the initial scan, and owing to sluggish polymerization kinetics at low pH, the simple ion transfer wave for AuCl4− re-emerges. If one assumes that DA donates 2 electrons and AuCl4− accepts 3, then a general first step could be described using reaction 4 (see Scheme 4); whereby, 8Cl− and 6H+ are effectively released to the DCE and w phases, respectively. The presence of 4 equivalents of Cl− for each AuCl4− reduced in the vicinity of the ITIES on the DCE side should result in a positive current offset in the voltammetric response; however, this was not observed (see Fig. 2). Thus, it is likely that liberated Cl− ions are either incorporated within the polymer matrix as a dopant or present at the surface of the formed Au NPs as a capping agent and do not undergo simple ion transfer from DCE → w – at least not at appreciable, detectable levels. It is also possible that AuCl4− reacts with DA homogeneously within the aqueous phase, and the lack of a peak-shaped wave when scanning negatively is owing to an absence of AuCl4− to transfer back.
At pH = 4 (Fig. 2B and E), a sigmoidal wave was again observed during both scan directions of the first cycle; however, unlike at pH 2, it retains its s-shaped profile even with repeated CV cycling. The current intensity of the signal occurring during the scan from positive to negative potentials does increase subtly from 2.00 to 2.50 nA between the 1st and 20th scan. The CV profile at pH 6 undergoes a similar transition (Fig. 2F); however, by the 15th scan a peak-shaped wave has emerged at −0.11 V (Fig. 2J). Again, this is consistent with interfacial electron transfer from DA(aq) → AuCl4−(DCE) that is diffusion limited by DA(aq).
In 2015, Drye and Uehara's groups50 characterized the Brust–Schiffrin mechanism of Au NP synthesis via AuCl4− reduction at a w|DCE interface; whereby, they revealed the importance of the polymeric [Au(I)SR] species which is Au1+-thiol species. This means that Au(III) was first reduced to Au(I). The presence of AuCl2− or Au1+-ligand coordinated species cannot be ruled out; however, XPS data (detailed below) seems to indicate the presence of only Au3+ and Au0 species in the polymer film.
The sigmoidal waves during forward and reverse sweeps are present in the first CV scan up until pH 10, while the intensity of the peak current signal continues to increase with pH up to pH 11.5. At pH 11.5, the first CV already contains a peak-shaped wave during the initial scan from positive to negative potentials. These data suggest that the kinetics and thermodynamics of film formation improve concomitantly with increasing pH. As shown in Scheme 3, DA has 3 protonation/deprotonation steps;46 moreover, polymerization of either the indole or catecholamine requires proton loss and is thus facilitated at more basic pHs. Examining the CVs shown in Fig. 2 altogether, it is likely that at pH 2 the kinetics of film formation are poor and improve concomitantly with increasing pH. Thus, these data agree with the generally accepted PDA formation mechanism and the need for a Brønsted base.8,12,46,59,60
After 25 CV cycles, a sample was ejected from the micropipette using the syringe incorporated into the back of the specialized holder and deposited onto a GCE and carefully rinsed to remove any residual, unreacted material. The modified GCE surface was then imaged using SEM; whereby, the images captured of films formed at pH equal to 4, 8, 10, and 11.5 have been given in Fig. 3A–D, respectively. These images demonstrate that the Au NP incorporated PDA nanocomposite are formed via the w|DCE interfacial reaction depicted in Scheme 1. Because the nanocomposite was ejected out of the pipette tip, it is likely that the side facing up in the SEM images (see also Fig. S1 of the SI) is the aqueous side of the film. Polymer films generated at the ITIES have been shown to have Janus-type34,35,61 properties since they grow differently on either side of the interface. Nishi et al.61 demonstrated that Au NP/polythiophene films generated at a water|ionic liquid (w|IL) interface demonstrated long polymer strands on the IL side containing the hydrophobic monomer, while Au NP growth continued on the water side which contained the KAuCl4 hydrophilic salt. Any AuCl4− that partitioned from w to IL formed Au nanofibers. Similarly, Scanlon's group34,35 electrogenerated poly(3,4-ethylenedioxythiophene) (PEDOT) films at a large w|TFT interface; whereby, a cerium sulfate salt in the aqueous phase acted as the electron acceptor while EDOT was dissolved in the TFT phase and behaved as the electron donor. In that case, the aqueous side was very smooth, while the oil side was rough due to the continued polymer growth. Because the micro-ITIES was employed herein, it was not possible to flip the film over and image the portion of the film formed on the DCE side of the ITIES. Based on these previous reports, it is expected that there would be morphological differences between the aqueous and oil sides of the nanocomposite. The film generated at pH 4 had more prominent Au NPs across its surface (Fig. 3A) and was revealed by EDX mapping (Fig. S1). However, at higher pHs the film was much thicker, and these NPs were difficult to observe and likely encapsulated within the polymer matrix. Fig. S2 of the SI shows EDX mapping images of the modified GCE surface for Au and O distributions. At low pH, Au is distributed evenly; however, as the pH increases Au seems to be pushed outwards forming a ring towards the outside edge of the GCE disc. This may be owing to Au NP encapsulation; whereby, they cannot be detected by EDX. However, at this stage, it is still poorly understood and will be the focus of future investigations. All Au NP/PDA films were electrogenerated using only 1 series of 25 CVs at a micro-ITIES (25 µm in diameter) as depicted in Fig. 2 before being deposited onto the GCE; nevertheless, the nanocomposite did spread to cover all the GCE surface (4 mm diameter). The film coverage of the GCE can be seen in the SEM images presented in Fig. S2A–C. When films were deposited, they were gently rinsed with MilliQ water to remove any excess supporting electrolyte or unreacted starting material. The fragile, soft nature of the film may explain how this coverage was achieved. It may also indicate that the Au NPs are only loosely bound with the polymer matrix; thus, the concentration of Au NPs towards the outer edge of the GCE disc may be a ‘coffee-ring’ effect and a result of ejecting the film onto the solid electrode surface from the micropipette. Another possible reason for Au NP concentration towards the outside ring of the film could be due to hemispherical diffusion of AuCl4− towards the w|DCE interface. This would result in the outer edge of the micropipette-supported ITIES receiving a higher flux of AuCl4− to the outside rim of the interface relative to its centre possibly generating a higher number of Au NPs in this region. This higher flux is often observed at ultramicroelectrodes;62 thus, these data would agree with those reports.
 | ||
| Fig. 3 Scanning electron microscopy (SEM) images of Au NP/poly(DA) films deposited onto glassy-carbon electrodes (GCE). Films were generated as described in Fig. 2 with the aqueous pH adjusted to (A) 4, (B) 8, (C) 10, and (D) 11.5. | ||
Electrogenerated Au NP/PDA films were also deposited onto ultra-thin lacey carbon Au TEM grids and imaged. Fig. 4A, C and E depict the TEM micrographs captured of films formed at pH 4, 8, and 10, respectively. The Au NPs were sized using ImageJ software and Fig. 4B, D and F show the respective histograms compiled (green, bar plots) as well as the Gaussian curve fitting (red traces) performed using Igor Pro (version 9). NPs were measured to be 25 ± 12, 5.8 ± 1.7, and 15.2 ± 2.4 nm for nanocomposites formed at pH's of 4, 8, and 10, respectively. The error in the NP sizing was taken from the half-width-at-half-height from the Gaussian fitting. The Au NPs within the nanocomposite film generated at pH 4 (Fig. 4A and B) were observed inside spherical dark spots on the TEM grid which are hypothesized to be the polymer matrix. At pH 8 (Fig. 4C and D), NPs are well distributed within the polymer film which seemed to cover the TEM grid. Finally, at pH 10 (Fig. 4E and F) polymer nanocrystals/needles where also observed within the nanocomposite and Au NPs where not as evenly distributed as observed at lower pH's. At low pH, the film was much thinner and more fragile making it difficult to manipulate by hand. The trend in Au NP size is surprising. Since the electropolymerization kinetics are likely slow at low pH our initial hypothesis was that Au NP size would increase with increasing pH; however, this was not the case. It likely means that the interplay between Au NP formation and PDA polymerization is much more complex and in need of further investigation.
 | ||
| Fig. 4 TEM images (A, C and E) taken of Au NP/poly(DA) films formed as described in Fig. 2 and deposited on TEM grids with the aqueous pH at 4 (A and B), 8 (C and D), and 10 (E and F). (B, D and F) Histograms of Au NP diameters measured using ImageJ software of the accompanying image. The red, solid trace shows the Gaussian curve fitting. Inset are the Gaussian peak maximums plus/minus the half-width-at-half-height. | ||
Combining the voltammetric as well as SEM and TEM image observations, it is likely that the kinetics of film formation are slow at lower aqueous pH and increase concomitantly with alkalinity. At lower pH, the Au NPs are more exposed on the surface, while as the pH increases, they become occluded by the polymer matrix. As described above, this agrees well with earlier investigations of PDA formation.12,60 Indeed, alkaline aqueous phases are often exploited to spontaneously generate PDA which can be dip-coated onto surfaces for anti-corrosion or electrochemical applications.46,59 These results also agree with the electropolymerization of thiophene-containing monomers at the ITIES.34,35,39–41,43 Our group recently demonstrated that the aqueous pH could be exploited as a means to control the electropolymerization of 2,2′:5′,2″-terthiophene (TT).43 Exchanging DCE for another solvent, e.g., TFT, or an IL, e.g., tetraoctylphosphonium tetrakis(pentafluorophenyl)borate (P8888TB), can be used to alter the nanocomposite morphology of Au NP/poly(9-vinylcarbazole).39 Both TT and 9-vinylcarbazole polymerization rely on a deprotonation step to form a conducting polymer similar to PDA formation. Nevertheless, these data show that the aqueous phase pH plays a key role in electropolymerization thermodynamics and kinetics.
XPS spectra were also collected for the films generated at each pH regime. Au NP/PDA films were deposited onto Al foil and then rinsed with MilliQ water before acquisition of the spectra. Fig. 5A depicts the XPS survey spectra obtained using the Au NP/PDA film electrogenerated at pH 2. The F-1s, O-1s, and C-1s signals are present at roughly 690, 534, and 287 eV, respectively. F-1s was only observed in films generated at pH's 2, 6, and 8. This may indicate that B(C6F5)− is acting as a dopant in the likely p-type PDA matrix. O-1s, N-1s, and C-1s signals were present in all spectra (see also Fig. S6 of the SI).
Fig. 5B–F show high-resolution XPS spectra for films generated at pH 2, 4, 8, 10, and 11.5, respectively, within the binding energy domain expected for Au-4f. The Au0 4f7/2 and 4f5/2 orbitals are expected to appear at roughly 83.8 and 87.5 eV, respectively.63–66 Multi-peak fitting reveals two additional peaks at 84.4 and 88.1 eV slightly positively shifted relative to those for Au0 which are likely Au3+ or AuCl4−.67 Thus, either some unreacted AuCl4− salt remains, or Au3+ species have coordinated to the catechol moiety of DA or the PDA backbone. While the sample was rinsed prior to XPS analysis, residual P66614AuCl4 from the DCE phase may be possible. The XPS spectra collected from the film formed at pH 6 showed no detectable Au-4f signal (data not shown). It is possible that Au NPs/P66614AuCl4 salt was dislodged during sampling and rinsing; however, it may be that Au NPs were not incorporated into the film. Because Au NPs were evidenced using TEM and SEM, including EDX mapping (see Fig. S2), the former is the more likely scenario.
Fig. S6C shows high-resolution XPS spectra collected in the Cl-2p region. Cl-2p3/2 has organic character towards 200 eV and metallic when shifted to 198.5–199 eV (red vertical bar in Fig. S6C).68 Interestingly, Cl-2p is present at all pH's and gains metallic character with increasing pH, i.e., signal shifts towards 198.5 eV. At low pH, Cl− is acting as a dopant, while as the pH increases and the kinetics of polymerization improve, likely more P66614AuCl4 salt is incorporated into the polymer matrix. Nevertheless, Au0 signal is still strong in these pH regions, so Au NPs are still present. A signal for P-2s can also be seen in some pH's which may indicate the presence of the P8888+ (tetraoctylphosphonium) or P66614+ (trihexyltetradecylphosphonium) cation from the DCE supporting electrolyte or P66614AuCl4 IL.
Water contact angle (WCA) measurements (data not shown) demonstrated that the film was very hydrophilic. Indeed, a 2 µL water droplet deposited on the surface of films electrogenerated at any pH spread so rapidly it could not be captured in a single photo by the CCD camera.
Au NP/PDA samples were also collected and analysed using FTIR and compared with DA, see Fig. S7. The peak at 3338 cm−1 which is associated with –NH2 stretch and disappears after electropolymerization. Meanwhile, the peak shift from 1618 to 1598 cm−1 from DA to PDA is likely a result of the formation of the π-conjugated indole. Thus, it is likely DA is forming PDA but first undergoes intramolecular cyclization to form DHI.
A series of shake-flask experiments at increasing pH were performed in 20 mL scintillation vials using 20 mM DA(aq) and 1 mM P66614AuCl4(DCE); whereby, both phases were purged with N2 gas for ∼30 min before being combined, sealed, shaken, then left to stand for 24 hours. Fig. 6A depicts photographs taken afterwards, while Fig. 6B and C show UV/Vis spectra recorded of the aqueous phase before and after 24 hours, respectively. At pH 2 and 4, there is no observable formation of black PDA, while at pH 6 a slight reddish hue can be seen in the aqueous phase. This likely points to AuCl4− spontaneous ion transfer followed by homogeneous reaction with DA generating Au NPs. pH's 8–11.5 show increasing amounts of a black precipitate, likely PDA. Within the UV/Vis spectra before interfacing the aqueous and DCE phases there is a peak at roughly 280 nm that shifts to 300 nm as the pH increases. Additionally, a signal grows at ∼435 nm. These are consistent with π–π* electronic transitions owing to the presence of the aromatic ring with the catechol moiety and agree well with the work of Sanchez-Rivera et al.47 After the two phases are combined and mixed for 24 hours (Fig. 6C), the signals at 280 and 435 nm broaden and there appears to be a shoulder above 435 nm at pH 10 and 11.5 that may indicate the presence of the Au NP localized-surface-plasmon-band (LSPR) which should appear at roughly 520 nm.69 These data also agree with the thermodynamic calculations surrounding Scheme 4 discussed below.
Using scanning electrochemical microscopy (SECM), probe-approach-curves (PACs) were measured towards a Au coated Si wafer modified with Au NP/PDA nanocomposite electrogenerated at a micro-ITIES at increasing pH in a solution of 0.9 mM ferrocene methanol (FcCH2OH) as redox mediator, 0.1 M phosphate buffer solution (PBS), and 0.1 M KCl (see Fig. 7) as well as using a carbon fibre UME. The UME was fabricated using 7 µm diameter carbon fibre and its ratio of outer glass radius (rg) to electroactive inlaid disc radius (ra), or Rg was confirmed to be ∼7 by comparing it to a simulated PAC of an insulating substrate (experimental: dashed, black trace and simulated: ○ marker curve in Fig. 7). Simulated PACs were generated using Comsol Multiphysics (version 6.3) as described in detail elsewhere.39 PACs were performed by biasing the UME at potential within the steady state oxidation current (iss) of FcCH2OH. PACs have been plotted using the normalized current (i/iss) and tip-to-substrate distance (L = d/ra); whereby, d is the actual tip-to-substrate distance.
 | ||
| Fig. 7 Probe approach curves (PACs) recorded using a carbon fibre ultramicroelectrode (UME), ra = 3.5 µm and an Rg ≈ 7, towards Au coated Si wafers modified with Au NP/PDA composites electrogenerated at the pH indicated inset (line traces). The UME was moved at a rate of 0.5 µm s−1. Overlaid are simulated PACs using generated using Comsol Multiphysics software at the substrate rate constant (k) indicated inset. Comsol code has been described in detail previously by us.39 | ||
The rate of the redox mediator re-reduction at the substrate surface (k) decreased concomitantly with increasing pH. For example, at pH = 2 or 4, the simulated curve with k = 3.4 × 10−3 cm s−1 showed the best overlap, while k = 1.25 × 10−3 cm s−1 generated the simulated PAC with the best overlap for the film generated at pH 11.5. The decrease in k as pH increases likely points to decreased conductivity of the film.
Au NP/PDA composites deposited on GCEs and carefully rinsed were then investigated as sensors for DA detection in a 0.1 M phosphate buffer solution (PBS). These experiments were performed in 3-electrode mode using a Pt mesh counter (CE) and an Ag/AgCl reference electrode (RE). All solid/electrolyte potentials have been plotted versus Ag/AgCl unless otherwise indicated. Fig. 8A shows CVs recorded at 0.050 V s−1; whereby, the red, solid and dashed curves were recorded using a bare GCE with and without 0.5 mM DA, respectively. Next, an Au NP/PDA film electrogenerated at pH 4 using Cell 1 with 20 mM of DA in the aqueous phase and 25 CV cycles was used to measure the black, dotted trace. Finally, the same nanocomposite film modified GCE was employed to record a CV with 0.3 mM of DA added to the electrolyte solution (blue, dashed plot). The CV obtained at the bare GCE without DA added is featureless in both forward and reverse scans with a modest capacitive current of ∼60 nA. With DA added, the bare GCE contains an irreversible electron transfer signal for DA oxidation with a peak potential (Ep) at ∼0.5 V.
After modification with the nanocomposite, a redox couple was observed with a half-wave potential (E1/2) at 0.208 V (vs. Ag/AgCl) and a peak-to-peak separation (ΔE) of ∼0.058 V. This is likely the oxidation/reduction of residual, unreacted DA within the nanocomposite. However, this signal persists even after several CV cycles; thus, the black, dotted curve in Fig. 8A is considered at a steady state condition. The narrow ΔE indicates that the heterogeneous electron transfer of DA across the nanocomposite/electrolyte interface is at least quasi-reversible for a 2-electron transfer as described by reaction 2 (Scheme 3). This assumes the criteria for reversibility is ΔE ≈ 0.057 V n−1, where n is the number of electrons transferred.54,70 If 0.3 mM of DA is added to the solution (blue, dashed curve), then the signal at E1/2 = 0.208 V increases with a final anodic peak current (ip,a) of 7.05 × 10−6 A. ΔE increased as well to 0.078 V, while the ratio of ip,a to the cathodic peak current (ip,c), ip,a/ip,c was ∼1.56. This slight increase in ΔE may be owing to a modest resistivity in the conductive nanocomposite film or uncompensated solution resistance, i.e., iR-drop. Nevertheless, this highlights the good electron transfer reactivity of the nanocomposite modified GCE electrode and it's significant improvement versus the bare GCE in DA oxidation performance. While the oxidation potential of DA is pH dependent,71E1/2 is in good agreement with previous reports.71,72 One can also intuit that the conductivity of the film is quite good.
Fig. 8B depicts i–E curves measured under similar conditions with [DA] = 0.2 mM while changing the pH. E1/2 for the DA redox signal shifts towards negative potentials as the pH increases from 4.1 to 9.0. This demonstrates the enhanced thermodynamic driving force of DA oxidation with increasing pH. A plot of E1/2vs. pH is presented in Fig. 8C while a linear regression fitting is shown as the solid, red trace; the linear fitting equation is provided inset. This trend can be used to calculate the thermodynamics of electropolymerization at the liquid|liquid interface as well. Fig. S3A and B of the SI show the trends in ip,a, ip,c, ip,a/ip,c and ΔE with changing pH. There is a slight decrease in ip,a and ip,c with increasing pH, while their ratio remains fairly close to ∼1.0. Simultaneously, ΔE shows a sharp increase at pH 7; however, at the other pH's it is less than 0.06 V and drops as low as 0.043 V at pH 9.0. Thus, it is likely that DA oxidation is at least quasi-reversible at the Au NP/PDA modified electrode.
The Ag/AgCl RE employed has an operational range from roughly pH 6 to 11.73 While the pH 4.1 and 5.0 experiments may be outside this region, the Ag/AgCl RE has been employed for convenience and the error in E1/2 is likely small.
Using the pH dependence shown in Fig. 8C, which is in good agreement with previous reports,74,75 one can establish the standard redox potential of DA, 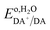 (versus SHE), such that it can be described by,
(versus SHE), such that it can be described by,
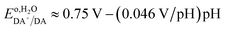 | (1) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, then this slope should be −0.059 V-per-pH. It is interesting, however, that in multiple reported cases, including this one, it was found to be close to that shown in eqn (1).74,75
1, then this slope should be −0.059 V-per-pH. It is interesting, however, that in multiple reported cases, including this one, it was found to be close to that shown in eqn (1).74,75
AuCl4− is a strong oxidizing agent with a standard reduction potential in water, 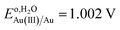 ;76 however, this is shifted far more negatively in organic solvents.77,78 To establish the redox potential of AuCl4− in DCE, AuCl4−/Au0 was measured in the DCE phase using a carbon fiber UME, r = 3.5 µm, while referencing the potential versus the ferrocene (Fc) redox couple, assuming
;76 however, this is shifted far more negatively in organic solvents.77,78 To establish the redox potential of AuCl4− in DCE, AuCl4−/Au0 was measured in the DCE phase using a carbon fiber UME, r = 3.5 µm, while referencing the potential versus the ferrocene (Fc) redox couple, assuming 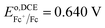 .79,80 CVs were measured in solutions containing 5 mM of P8888TB as supporting electrolyte as well as 1 mM of either Fc or P66614AuCl4 and are provided in Fig. S4A and B, respectively, of the SI. A solution of 1 mM Fc and P66614AuCl4 plus supporting electrolyte was mixed in DCE resulting in the generation of a purple Au NP suspension. Fc has been shown to react with gold salts donating electrons and spontaneously generating Au NPs with ferrocenium (Fc+) acting as the capping agent.44,81 Nevertheless, voltammetric response measurements in this mixture (Fig. S4C of the SI) show that residual AuCl4− and Fc are present even after they are allowed to react for 5–10 min. This was sufficient to estimate Eo,DCEAu(III)/Au as −0.920 V. A steady-state response was also observed for what is believed to be, AuCl4− + 2e− → AuCl2− + 2Cl−, with Eo,DCEAu(III)/Au(I) calculated to be −0.20 V. These values are in good agreement with those measured and calculated from first principles at a microelectrode by Gründer et al.77 of Eo,DCEAu(III)/Au = −0.9 V and Eo,DCEAu(III)/Au(I) = −0.27 V, respectively.
.79,80 CVs were measured in solutions containing 5 mM of P8888TB as supporting electrolyte as well as 1 mM of either Fc or P66614AuCl4 and are provided in Fig. S4A and B, respectively, of the SI. A solution of 1 mM Fc and P66614AuCl4 plus supporting electrolyte was mixed in DCE resulting in the generation of a purple Au NP suspension. Fc has been shown to react with gold salts donating electrons and spontaneously generating Au NPs with ferrocenium (Fc+) acting as the capping agent.44,81 Nevertheless, voltammetric response measurements in this mixture (Fig. S4C of the SI) show that residual AuCl4− and Fc are present even after they are allowed to react for 5–10 min. This was sufficient to estimate Eo,DCEAu(III)/Au as −0.920 V. A steady-state response was also observed for what is believed to be, AuCl4− + 2e− → AuCl2− + 2Cl−, with Eo,DCEAu(III)/Au(I) calculated to be −0.20 V. These values are in good agreement with those measured and calculated from first principles at a microelectrode by Gründer et al.77 of Eo,DCEAu(III)/Au = −0.9 V and Eo,DCEAu(III)/Au(I) = −0.27 V, respectively.
With these values in hand, the thermodynamics of the heterogeneous liquid|liquid electron transfer reaction including the interfacial electron transfer potential (ΔwDCEϕET) were calculated for the first step in the polymerization reaction if one assumes that the stoichiometric ratio of AuCl4− to DA is 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, through following formula,44,80
3, through following formula,44,80
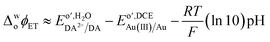 | (2) |
Considering an alternative mechanism, whereby AuCl4− partitions and undergoes homogeneous electron transfer with DA in the aqueous phase, then an alternate equation is needed as shown below,
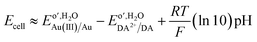 | (3) |
Using eqn (3), large negative ΔG values are calculated indicating a spontaneous reaction pathway that may be a driving force for AuCl4− transfer to water. This would manifest as a relatively large positive current offset that was not observed; therefore, it is likely the small interfacial size limits this reaction pathway. The shake-flask experiments shown in Fig. 6A at a large ITIES also demonstrate that AuCl4− transfer followed by homogeneous reaction in the water phase is not favoured at low pH; however, it may be a factor as pH increases.
Fig. S5A depicts the i–E curves recorded at an Au NP/PDA modified GCE while changing the scan rate, while Fig. S5B contains plots of ip,a and ip,cversus ν1/2. Linear curve fittings of the latter show good correlation with R2 values of ∼0.999. The number of electrons transferred (n) was calculated using the Randles-Ševčík equation70,82 (eqn (4)) and assuming a DA diffusion coefficient (D) equal to 0.54 × 10−5 cm2 s−1 as described by Corona-Avendaño et al.83
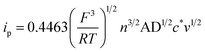 | (4) |
In this way, n ≈ 1.87 and 2.00 were calculated for ip,a and ip,cversus ν1/2, respectively. This agrees well with the 2-electron DA oxidation generally to DAQ (Scheme 3). The redox chemistry of DA has been studied extensively at the GCE/electrolyte interface.83 Zhang and Dryhurst84 investigated DA electrochemistry at a pyrolytic graphite electrode that showed reversible DA oxidation. They also identified several intermediate species using HPLC and proposed that DA is oxidized to DAQ which can then undergo the intramolecular cyclization to form I56D or DHI (see Scheme 3). DHI has a much lower oxidation potential than DA84 and thus, readily polymerizes to form PDA and melanin.
Fig. 9A shows differential pulse voltammograms (DPVs) measured in 3-electrode mode using Au NP/PDA modified GCEs while changing [DA]. Fig. 9B depicts the calibration curve generated using the peak current values for DA oxidation from Fig. 9A. The Au NP/PDA film was electrogenerated using Cell 1 at pH 4.1, 20 mM of DA, and 25 CV cycles. This pH was chosen as the Au NPs appeared more visually exposed in the SEM images. Other film generation conditions were investigated; however, these are showcased here as a proof-of-concept. The ipversus [DA] plot has a linear region spanning roughly between 0.1–20 µM; afterwards, the curve deviates.
Next, the influence of interferents such as AA and UA were investigated using a 0.1 M PBS buffer solution containing 0.1 M KCl electrolyte at pH 5.5–6 (see Fig. 10A). Even with no DA added, a DA oxidation wave was observed at 0.17 V like that seen using cyclic voltammetry (see dashed, black trace in Fig. 8A). No peak for AA was observed even at concentrations of 50 or 100 µM (data not shown), and its oxidation may be inhibited at the modified electrode interface. A UA oxidation peak was observed with a peak potential of ∼0.37 V. As [DA] increased from 0.3 to 24 µM, the UA anodic peak current decreased. Simultaneously, the UA peak potential shifts slightly by ∼0.016 V; however, at higher [DA] it stabilizes. This may be owing to the Ag/AgCl reference electrode, which is on the lower end of its operational range,73 or it may be caused by DA inhibiting UA oxidation. Nevertheless, using these DA peak signals a new calibration curve was constructed with both AA and UA interferents present (see Fig. 10B). There is a sharp decrease in the slope of the linear correlation curve which indicates a loss in sensitivity. The R2 value is still high at 0.979 showing a good fitting.
 | ||
| Fig. 10 (A) DPVs recorded under the same conditions as Fig. 9A, however, with different concentrations of DA, AA, and UA added, as indicated inset, as interferents. DA and UA oxidation waves are labelled inset. (B) Calibration curve constructed using the ip data from A. Red trace is the linear curve fitting while the red, dashed traces are lines-of-best-fit at a 99.9% confidence interval as described in Section 4 of the SI. Linear regression equation and R2 values are written inset. (C) Chemical structures of ascorbic and uric acid. | ||
A limit-of-detection (cm) was calculated using the average of 3 blank DPV signal profiles and the following two equations,85
Sm = ![[S with combining macron]](https://www.rsc.org/images/entities/i_char_0053_0304.gif) blk + kmσblk blk + kmσblk | (5) |
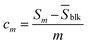 | (6) |
Whereby, ![[S with combining macron]](https://www.rsc.org/images/entities/i_char_0053_0304.gif) blk is the mean blank signal, σblk is the standard deviation of the blank signal, km is the variation in the blank, and m is the slope of the calibration curve. Using a km value of 3, cm was determined to be 0.27 and 0.43 µM for the calibration curves shown in Fig. 9B and 10B, respectively. Thus, there is a slight loss in sensitivity when AA and UA are present. The Au NP/PDA modified electrode is an order of magnitude less sensitive than some of the highest sensitivity platforms reported.86,87 However, these platforms often rely on complex enzyme- or antibody-based modifications that are challenging to achieve,87 or are not practical for in vivo implementation. Karim et al.88 recently wrote a comprehensive review of dopamine electrochemical sensors. Table 1 provides a few select electrode modifications for comparison with the work presented herein.
blk is the mean blank signal, σblk is the standard deviation of the blank signal, km is the variation in the blank, and m is the slope of the calibration curve. Using a km value of 3, cm was determined to be 0.27 and 0.43 µM for the calibration curves shown in Fig. 9B and 10B, respectively. Thus, there is a slight loss in sensitivity when AA and UA are present. The Au NP/PDA modified electrode is an order of magnitude less sensitive than some of the highest sensitivity platforms reported.86,87 However, these platforms often rely on complex enzyme- or antibody-based modifications that are challenging to achieve,87 or are not practical for in vivo implementation. Karim et al.88 recently wrote a comprehensive review of dopamine electrochemical sensors. Table 1 provides a few select electrode modifications for comparison with the work presented herein.
| Electrode functionalization material | Technique | c m µM−1 | Linear range µM−1 | Ref. |
|---|---|---|---|---|
| Laccase-carbon dots | DPV | 0.08 | 0.25–76.81 | 89 |
| PEDOT–GO–polyphenol oxidase | Amperometry | 8 × 10−9 | 5 × 10−5 – 0.35 | 90 |
| Au NPs + polymelamine | DPV | 0.067 | 0.1–11 | 91 |
| rGO-Fe3O4/AgNPs | DPV | 0.004 | 0.015–100 | 92 |
| FeMoSe2–GO | DPV | 0.002 | 0.04–40 | 93 |
| Au NP/PDA–GCE | DPV | 0.27 | 0.1–24 | This work |
Thus, this proof-of-concept electrochemical sensing device, with the potential of generating a free-standing, conductive PDA electrode is promising and comparable to other biosenors in the field in terms of cm and limit-of-linearity (see Table 1).
Conclusions
In summary, Au NP incorporated PDA films were electrogenerated at a micro liquid|liquid interface (25 µm in diameter) under external, potential control. DA was dissolved in the aqueous phase, while a P66614AuCl4 ionic liquid was added to the DCE phase. The influence of aqueous pH on film formation was observed voltammetrically which manifested as a more prominent heterogeneous electron transfer wave as pH increased. Moreover, the film morphology was shown to change using SEM imaging. At low pH, the film seemed thinner with the Au NPs more exposed on the surface; however, as the pH was adjusted to become more alkaline the film thickness seemed to increase and NPs were more encapsulated. The ease of film formation at higher pH's agreed with thermodynamic calculations and previous DA as well as PDA studies.8,12,46,59,60 The need for a Brønsted base to enhance the thermodynamics of electropolymerization also agreed with other liquid|liquid electrochemical reports.34,35,39–41,43,48Au NP/PDA films deposited onto a large (mm scale) GCEs were then used as a proof-of-concept platform for DA detection with and without the presence of interfering species such as AA and UA. DA oxidation was shown to be quasi-reversible with ΔE varying from roughly 0.04–0.08 V with increasing pH; simultaneously, the pH dependence of 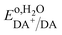 was found to agree quite well with previous reports.74,75 A good linear dynamic range was discovered for DA detection using the Au NP/PDA modified GCE from 0.2 to 20 µM. Moreover, the limit-of-detection (LOD) was quantified with and without the addition of other biomolecules that act as interferents, specifically AA and UA. The LOD changed slightly from 0.27 to 0.43 µM; thus, there was a modest decrease in the overall sensitivity of the biosensor. Future investigations will include modifying microelectrodes as well as using the Au NP/PDA composite as a free-standing electrode. The latter proved challenging in the present work as the film was quite fragile. While this may be disadvantageous for an in vitro biosensor device, the nanocomposite materials softness may prove to be quite advantageous for bioimplantation and avoiding rejection by the host.
was found to agree quite well with previous reports.74,75 A good linear dynamic range was discovered for DA detection using the Au NP/PDA modified GCE from 0.2 to 20 µM. Moreover, the limit-of-detection (LOD) was quantified with and without the addition of other biomolecules that act as interferents, specifically AA and UA. The LOD changed slightly from 0.27 to 0.43 µM; thus, there was a modest decrease in the overall sensitivity of the biosensor. Future investigations will include modifying microelectrodes as well as using the Au NP/PDA composite as a free-standing electrode. The latter proved challenging in the present work as the film was quite fragile. While this may be disadvantageous for an in vitro biosensor device, the nanocomposite materials softness may prove to be quite advantageous for bioimplantation and avoiding rejection by the host.
The liquid|liquid interface is often employed as an analogue or approximation of the biological membranes. This work showcases how it can be exploited to generate complex nanocomposites as well as large biomolecules such as PDA. We hope this work will be a potential springboard for novel studies of nature's approach to molecular synthesis based on the ITIES.
Author contributions
L. N.: formal analysis; data curation; formal analysis; investigation; writing – review & editing. F. K.: analysis; data curation; formal analysis; investigation; E. B. E.: conceptualization; funding acquisition; supervision; methodology; project administration; formal analysis; T. J. S.: conceptualization; funding acquisition; supervision; methodology; project administration; formal analysis; writing – original draft; writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
All data from this research will be made available on the Borealis Dataverse database repository https://borealisdata.ca/dataverse/memorial.Supplementary information (SI): EDX mapping of Au NP/PDA thin-films, trends in DA redox properties, and additional voltammetric data. See DOI: https://doi.org/10.1039/d5na00744e.
Acknowledgements
T. J. S. is grateful to the Natural Sciences and Engineering Research Council (NSERC) for a Discovery Grant (#006074-2019) as well as the Canadian Foundation for Innovation (CFI) and Memorial University (MUN) for infrastructure funding. L. N. is thankful for the Dr Liqin Chen Scholarship fund and MUN CREAIT network for access to instrumentation. E. B. E. gratefully acknowledges financial support from the Natural Sciences and Engineering Research Council of Canada (NSERC) through a Discovery Grant (RGPIN-2020-05152) and from Ontario Tech University through the Research Excellence Chairs program. F. K. acknowledges scholarship support from the NSERC Canada Graduate Scholarship–Master’s (CGS M) program and the Ontario Graduate Scholarship (OGS) program.Notes and references
- R. Franco, I. Reyes-Resina and G. Navarro, Biomedicines, 2021, 9, 109 CrossRef CAS PubMed.
- I.-E. Pralea, R.-C. Moldovan, A.-M. Petrache, M. Ilieş, S.-C. Hegheş, I. Ielciu, R. Nicoară, M. Moldovan, M. Ene, M. Radu, A. Uifălean and C.-A. Iuga, Int. J. Mol. Sci., 2019, 20, 3943 CrossRef CAS PubMed.
- T. Ligonzo, M. Ambrico, V. Augelli, G. Perna, L. Schiavulli, M. A. Tamma, P. F. Biagi, A. Minafra and V. Capozzi, J. Non-Cryst. Solids, 2009, 355, 1221–1226 CrossRef CAS.
- A. B. Mostert, B. J. Powell, F. L. Pratt, G. R. Hanson, T. Sarna, I. R. Gentle and P. Meredith, Proc. Natl. Acad. Sci. U. S. A., 2012, 109, 8943–8947 CrossRef CAS PubMed.
- Q. Liu, P. Wang, C. Liu, F. Xue, Q. Wang, Y. Chen, R. Hou and T. Chen, BMC Neurol., 2023, 23, 301 Search PubMed.
- X. Pan, A. C. Kaminga, S. W. Wen, X. Wu, K. Acheampong and A. Liu, Front. Aging Neurosci., 2019, 11, 2019 Search PubMed.
- F. A. Zucca, E. Basso, F. A. Cupaioli, E. Ferrari, D. Sulzer, L. Casella and L. Zecca, Neurotox. Res., 2014, 25, 13–23 CrossRef CAS PubMed.
- Y. Liu, K. Ai and L. Lu, Chem. Rev., 2014, 114, 5057–5115 CrossRef CAS PubMed.
- H. Lee, S. M. Dellatore, W. M. Miller and P. B. Messersmith, Science, 2007, 318, 426–430 CrossRef CAS PubMed.
- F. Yu, S. Chen, Y. Chen, H. Li, L. Yang, Y. Chen and Y. Yin, J. Mol. Struct., 2010, 982, 152–161 CrossRef CAS.
- J. Jeong, J. Do and S. M. Kang, Adv. Mater. Interfaces, 2024, 11, 2300871 CrossRef CAS.
- H. Khezraqa, S.-A. Safavi-Mirmahalleh, H. Roghani-Mamaqani and M. Salami-Kalajahi, J. Energy Storage, 2024, 79, 110170 CrossRef.
- C. Zhang, X. Du and Z. Zhang, Sens. Actuators Rep., 2025, 10, 100348 CrossRef.
- R. M. Wightman, L. J. May and A. C. Michael, Anal. Chem., 1988, 60, 769A–770A CrossRef CAS.
- A. G. Ewing, J. C. Bigelow and R. M. Wightman, Science, 1983, 221, 169–171 Search PubMed.
- W. G. Kuhr, A. G. Ewing, W. L. Caudill and R. M. Wightman, J. Neurochem., 1984, 43, 560–569 CrossRef CAS PubMed.
- C. Gu and A. G. Ewing, Chem. Sci., 2021, 12, 7393–7400 RSC.
- X. Wei, Z. Zhang and Z. Wang, Microchem. J., 2019, 145, 55–58 CrossRef CAS.
- R. Pramanik, Anal. Method., 2025, 17, 4251–4292 RSC.
- A. A. El-Sherbeni, M. R. Stocco, F. B. Wadji and R. F. Tyndale, J. Chromatogr. A, 2020, 1627, 461403 CrossRef CAS PubMed.
- Y. Zhang, S. Qi, Z. Liu, Y. Shi, W. Yue and C. Yi, Mater. Sci. Eng. C, 2016, 61, 207–213 Search PubMed.
- D. P. Zhan, S. N. Mao, Q. Zhao, Z. Chen, H. Hu, P. Jing, M. Q. Zhang, Z. W. Zhu and Y. H. Shao, Anal. Chem., 2004, 76, 4128–4136 Search PubMed.
- H. D. Jetmore, C. B. Milton, E. S. Anupriya, R. Chen, K. Xu and M. Shen, Anal. Chem., 2021, 93, 16535–16542 CrossRef CAS PubMed.
- A. Lytvynenko, S. Baluchová, J. Zima, J. Krůšek and K. Schwarzová-Pecková, Bioelectrochemistry, 2024, 158, 108713 CrossRef CAS.
- M. Sajid, M. K. Nazal, M. Mansha, A. Alsharaa, S. M. S. Jillani and C. Basheer, Trends Anal. Chem., 2016, 76, 15–29 CrossRef CAS.
- M. Bilal, Z. U. Rehman, F. K. Butt, Y. Cao, K. Zheng, J. A. Jrar, Z. Zhang and J. Hou, ACS Appl. Nano Mater., 2025, 8, 9711–9719 CrossRef CAS.
- F. Weyand, S. Gianvittorio, F. Longo, J. Wang and A. Lesch, Electrochim. Acta, 2025, 526, 146174 CrossRef CAS.
- A. Pandikumar, G. T. Soon How, T. P. See, F. S. Omar, S. Jayabal, K. Z. Kamali, N. Yusoff, A. Jamil, R. Ramaraj, S. A. John, H. N. Lim and N. M. Huang, RSC Adv., 2014, 4, 63296–63323 RSC.
- M. Mallesha, R. Manjunatha, C. Nethravathi, G. S. Suresh, M. Rajamathi, J. S. Melo and T. V. Venkatesha, Bioelectrochemistry, 2011, 81, 104–108 CrossRef CAS.
- A. Cecilia Rossi Fernández, L. Alejandra Meier and N. Jorge Castellani, Comput. Theor. Chem., 2022, 1212, 113705 CrossRef.
- E. A. Khudaish and J. A. Rather, J. Electroanal. Chem., 2016, 776, 206–212 CrossRef CAS.
- J. Evers, K. Sridhar, J. Liegey, J. Brady, H. Jahns and M. Lowery, J. Neural Eng., 2022, 19, 046004 CrossRef PubMed.
- P. Limousin and T. Foltynie, Nat. Rev. Neurol., 2019, 15, 234–242 CrossRef PubMed.
- R. A. Lehane, A. Gamero-Quijano, S. Malijauskaite, A. Holzinger, M. Conroy, F. Laffir, A. Kumar, U. Bangert, K. McGourty and M. D. Scanlon, J. Am. Chem. Soc., 2022, 144, 4853–4862 CrossRef CAS PubMed.
- R. A. Lehane, A. Gamero-Quijano, J. A. Manzanares and M. D. Scanlon, J. Am. Chem. Soc., 2024, 146, 28941–28951 CrossRef CAS PubMed.
- K. Lepková, J. Clohessy and V. J. Cunnane, Electrochim. Acta, 2008, 53, 6273–6277 CrossRef.
- R. Knake, A. W. Fahmi, S. A. M. Tofail, J. Clohessy, M. Mihov and V. J. Cunnane, Langmuir, 2005, 21, 1001–1008 CrossRef CAS PubMed.
- U. Evans-Kennedy, J. Clohessy and V. J. Cunnane, Macromolecules, 2004, 37, 3630–3634 CrossRef CAS.
- L. Nazari and T. J. Stockmann, Langmuir, 2024, 40, 24494–24506 CrossRef CAS PubMed.
- R. Moshrefi, K. Ryan, E. P. Connors, J. C. Walsh, E. Merschrod, G. J. Bodwell and T. J. Stockmann, Nanoscale, 2023, 15, 5834–5842 RSC.
- R. Moshrefi, H. Przybyła and T. J. Stockmann, Sci. Rep., 2023, 13, 1201 CrossRef CAS PubMed.
- M. D. Scanlon, E. Smirnov, T. J. Stockmann and P. Peljo, Chem. Rev., 2018, 118, 3722–3751 CrossRef CAS PubMed.
- R. Moshrefi, E. P. Connors, E. Merschrod and T. J. Stockmann, Electrochim. Acta, 2022, 426, 140749 CrossRef CAS.
- R. Moshrefi, A. Suryawanshi and T. J. Stockmann, Electrochem. Commun., 2021, 122, 106894 CrossRef CAS.
- T. J. Stockmann, P. D. Boyle and Z. Ding, Catal. Today, 2017, 295, 89–94 CrossRef CAS.
- S. Schindler and T. Bechtold, J. Electroanal. Chem., 2019, 836, 94–101 CrossRef CAS.
- A. E. Sánchez-Rivera, S. Corona-Avendaño, G. Alarcón-Angeles, A. Rojas-Hernández, M. T. Ramirez-Silva and M. A. Romero-Romo, Spectrochim. Acta, Part A, 2003, 59, 3193–3203 CrossRef PubMed.
- R. Moshrefi and T. J. Stockmann, Nanomaterials, 2022, 12, 2748 CrossRef CAS PubMed.
- A. Uehara, S.-Y. Chang, S. G. Booth, S. L. M. Schroeder, J. F. W. Mosselmans and R. A. W. Dryfe, Electrochim. Acta, 2016, 190, 997–1006 CrossRef CAS.
- A. Uehara, S. G. Booth, S. Y. Chang, S. L. M. Schroeder, T. Imai, T. Hashimoto, J. F. W. Mosselmans and R. A. W. Dryfe, J. Am. Chem. Soc., 2015, 137, 15135–15144 CrossRef CAS PubMed.
- A. Uehara, T. Hashimoto and R. A. W. Dryfe, Electrochim. Acta, 2014, 118, 26–32 CrossRef CAS.
- T. J. Stockmann, L. Angelé, V. Brasiliense, C. Combellas and F. Kanoufi, Angew. Chem., Int. Ed., 2017, 56, 13493–13497 CrossRef CAS PubMed.
- T. J. Stockmann, R. Guterman, P. J. Ragogna and Z. Ding, Langmuir, 2016, 32, 12966–12974 CrossRef CAS PubMed.
- A. J. Bard and L. R. Faulkner, Electrochemical Methods: Fundamentals and Applications, John Wiley, New York, 2nd edn, 2001 Search PubMed.
- S. Liu, Q. Li and Y. Shao, Chem. Soc. Rev., 2011, 40, 2236–2253 RSC.
- A. Usher, D. C. McPhail and J. Brugger, Geochim. Cosmochim. Acta, 2009, 73, 3359–3380 CrossRef CAS.
- M. Luty-Błocho, K. Pacławski, M. Wojnicki and K. Fitzner, Inorg. Chim. Acta, 2013, 395, 189–196 CrossRef.
- Y. Li, M. Liu, C. Xiang, Q. Xie and S. Yao, Thin Solid Films, 2006, 497, 270–278 CrossRef CAS.
- J. H. Ryu, P. B. Messersmith and H. Lee, ACS Appl. Mater. Interfaces, 2018, 10, 7523–7540 CrossRef CAS PubMed.
- M. d'Ischia, A. Napolitano, V. Ball, C.-T. Chen and M. J. Buehler, Acc. Chem. Res., 2014, 47, 3541–3550 CrossRef.
- N. Nishi, I. Yajima, K.-i. Amano and T. Sakka, Langmuir, 2018, 34, 2441–2447 CrossRef CAS PubMed.
- K. Aoki, Electroanalysis, 1993, 5, 627–639 CrossRef CAS.
- M. Brust, M. Walker, D. Bethell, D. J. Schiffrin and R. Whyman, J. Chem. Soc. Chem. Commun., 1994, 801–802, 10.1039/C39940000801.
- O. F. Odio, L. Lartundo-Rojas, P. Santiago-Jacinto, R. Martínez and E. Reguera, J. Phys. Chem. C, 2014, 118, 2776–2791 CrossRef CAS.
- Z. Tang, B. Xu, B. Wu, M. W. Germann and G. Wang, J. Am. Chem. Soc., 2010, 132, 3367–3374 CrossRef CAS PubMed.
- S. Peters, S. Peredkov, M. Neeb, W. Eberhardt and M. Al-Hada, Surf. Sci., 2013, 608, 129–134 CrossRef CAS.
- S. Wang, K. Qian, X. Bi and W. Huang, J. Phys. Chem. C, 2009, 113, 6505–6510 CrossRef CAS.
- J. R. Araujo, B. S. Archanjo, K. R. de Souza, W. Kwapinski, N. P. S. Falcão, E. H. Novotny and C. A. Achete, Biol. Fertil. Soils, 2014, 50, 1223–1232 CrossRef CAS.
- K. M. Mayer and J. H. Hafner, Chem. Rev., 2011, 111, 3828–3857 CrossRef CAS PubMed.
- R. S. Nicholson and I. Shain, Anal. Chem., 1964, 36, 706–723 CrossRef CAS.
- H.-S. Jang, D. Kim, C. Lee, B. Yan, X. Qin and Y. Piao, Inorg. Chem. Commun., 2019, 105, 174–181 CrossRef CAS.
- H. Chen, H. Chen and R. Hong, Catalysts, 2019, 9, 653 CrossRef CAS.
- S. Anantharaj, P. J. J. Sagayaraj, M. S. Yesupatham, R. Arulraj, K. Eswaran, K. Sekar and S. Noda, J. Mater. Chem. A, 2023, 11, 17699–17709 RSC.
- H. Yang, D. Liu, X. Zhao, J.-H. Yang, H. Chang, R. Xing and S. Liu, Anal. Method., 2017, 9, 3191–3199 RSC.
- C. Rajkumar, B. Thirumalraj, S.-M. Chen and H.-A. Chen, J. Colloid Interface Sci., 2017, 487, 149–155 CrossRef CAS PubMed.
- P. Vanýsek, in CRC Handbook of Chemistry and Physics, W. M. Haynes, CRC Press/Taylor, Boca Raton, FL, 2021, pp. 20–29 Search PubMed.
- Y. Grunder, H. L. T. Ho, J. F. W. Mosselmans, S. L. M. Schroeder and R. A. W. Dryfe, Phys. Chem. Chem. Phys., 2011, 13, 15681–15689 RSC.
- J. Anderson, S. Sawtelle and C. McAndrews, Inorg. Chem., 1990, 29, 2627–2633 CrossRef CAS.
- D. J. Fermin and R. Lahtinen, in Liquid Interfaces in Chemical, Biological and Pharmaceutical Applications, A. G. Volkov, Marcel Dekker, Inc., 2001, vol. 8 Search PubMed.
- M. A. Méndez, R. Partovi-Nia, I. Hatay, B. Su, P. Y. Ge, A. Olaya, N. Younan, M. Hojeij and H. H. Girault, Phys. Chem. Chem. Phys., 2010, 12, 15163–15171 RSC.
- R. Ciganda, J. Irigoyen, D. Gregurec, R. Hernández, S. Moya, C. Wang, J. Ruiz and D. Astruc, Inorg. Chem., 2016, 55, 6361–6363 CrossRef CAS PubMed.
- G. A. Mabbott, J. Chem. Educ., 1983, 60, 697 CrossRef CAS.
- S. Corona-Avendaño, G. Alarcón-Angeles, M. T. Ramírez-Silva, G. Rosquete-Pina, M. Romero-Romo and M. Palomar-Pardavé, J. Electroanal. Chem., 2007, 609, 17–26 CrossRef.
- F. Zhang and G. Dryhurst, Bioorg. Chem., 1993, 21, 392–410 CrossRef CAS.
- D. A. Skoog, F. J. Holler and S. Crouch, Principles of Instrumental Analysis, Thomson Higher Education, Belmont, CA, 6th edn, 2007 Search PubMed.
- K. Zhang, H. Wu and Y. Zhang, Int. J. Electrochem. Sci., 2022, 17, 220662 CrossRef CAS.
- X. Liu and J. Liu, VIEW, 2021, 2, 20200102 CrossRef.
- A. Karim, M. Yasser, A. Ahmad, H. Natsir, A. Wahid Wahab, S. Fauziah, P. Taba, I. Pratama, Rosalin, A. Rajab, A. Nur Fitriani Abubakar, T. Widayati Putri, R. Munadi, A. Fudhail Majid, A. Nur Fadliah, A. Rifai and M. Syahrir, J. Electroanal. Chem., 2024, 959, 118157 CrossRef CAS.
- R. Wu, S. Yu, S. Chen, Y. Dang, S.-H. Wen, J. Tang, Y. Zhou and J.-J. Zhu, Anal. Chim. Acta, 2022, 1229, 340365 CrossRef CAS PubMed.
- P. Ramu, S. P. Vimal, P. Suresh, A. Sanmugam, U. Saravanakumar, R. S. Kumar, A. I. Almansour, N. Arumugam and D. Vikraman, RSC Adv., 2022, 12, 15575–15583 RSC.
- M. Harsini, B. A. Widyaningrum, E. Fitriany, D. R. A. Paramita, A. N. Farida, A. Baktir, F. Kurniawan and S. C. W. Sakti, Chin. J. Anal. Chem., 2022, 50, 100052 Search PubMed.
- A. R. M. Rosli, M. S. Noorashikin and F. Yusoff, J. Electrochem. Soc., 2021, 168, 087512 CrossRef CAS.
- Y. Zhang and S. Luo, J. Electrochem. Soc., 2025, 172, 057501 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |