 Open Access Article
Open Access ArticleElectromagnetic milling-promoted steel rod-enabled Sonogashira coupling reactions of aryl halides and terminal alkynes
Xinyao Liua,
Yanyan Chena,
Qiuhong Ma*b,
Meng Yuc,
Wenjie Guod,
Baoju Wanga,
Qiang Liua,
Rujun Yu*a,
Qing Liu*a and
Hui Liu *a
*a
aSchool of Chemistry and Chemical Engineering, Shandong University of Technology Zibo, Shandong 255049, China. E-mail: yu21cen@163.com; qingliu0315@163.com; huiliu1030@163.com
bDepartment of Clinical Laboratory, Zibo Central Hospital Affiliated to Binzhou Medical University, Shandong 255049, China. E-mail: shui0717@163.com
cZibo Construction Project Environmental Review Service Center, Shandong 255049, China
dShandong Diam Chemical Co., Ltd, Shandong 256800, China
First published on 14th April 2026
Abstract
Nickel-catalyzed Sonogashira reactions between aryl halides and terminal alkynes have become a powerful method for constructing C(sp2)–C(sp) bonds. Conventional approaches typically required copper co-catalysts, solvents, inert atmospheres, and external heating. Herein, we report an alternative and streamlined reductive C(sp2)–C(sp) cross-coupling protocol that employs an inexpensive nickel catalyst, eliminating the need for Ni/Cu dual catalysts. This electromagnetic milling (EMM) promoted strategy effectively circumvents the constraints associated with traditional nickel-catalyzed Sonogashira reactions. Mechanistic studies reveal that the Ni(II) precursor in NiCl2 is activated under a magnetic field and in the presence of DABCO to produce Ni(0) as the active catalyst, and EMM plays a critical role in promoting reductive elimination from the NiII complex.
Introduction
As versatile building blocks in organic chemistry, alkynes are widely used in fields from biochemistry to materials science and drug discovery.1–9 Consequently, significant efforts are focused on developing efficient methods to incorporate alkyne moieties into molecules for synthesizing substituted internal alkynes. The Sonogashira reaction is widely regarded as the leading C–C(sp) cross-coupling method, traditionally pairing terminal alkynes and electrophiles using a palladium catalyst with or without a copper co-catalyst (Fig. 1A).10–15 Additionally, recent research has focused on developing Sonogashira-type reactions using precious transition metals like Co, Ir and Rh, as alternatives to traditional palladium catalysts.16–22 Meanwhile, nickel catalysts, as less toxic and readily commercially available transition metal catalysts, attracted great interest in the development of Sonogashira reactions.23,24 However, most existing nickel-catalyzed Sonogashira cross-coupling reactions of aryl halides with terminal alkynes still depend on copper co-catalysts (Fig. 1B).25 In the solution phase, Liao's team achieved nickel-catalyzed Sonogashira coupling without copper co-catalysts, albeit requiring 48 hours of reaction time in DMAc solvent at 60 °C (Fig. 1C).26Mechanochemistry, as a representative technology in green synthesis, enables efficient homogenization of reactants through mechanical energy input, significantly reducing or eliminating the use of organic solvents.27–43 However, Sonogashira reactions under mechanochemical conditions have predominantly relied on palladium catalysts44–47 or noble metal alloys (e.g., Pd–Ni) (Fig. 1D). In 2024, the Borchardt research group reported the first instance of mechanocatalytic Sonogashira reactions using a Pd–Ni alloy containing 20% palladium in a ball mill, achieving over 80% yield within one hour under solvent-free conditions without external heating.48 In summary, to date, a mechanochemical Sonogashira reaction using a pure nickel catalyst without copper co-catalysts under solvent-free and externally unheated conditions has not been reported.
As an innovative milling device, the electromagnetic mill (EMM) employs ferromagnetic particles as the milling medium, utilizing an axially rotating electromagnetic field to drive ferromagnetic rods at high speed, achieving efficient mixing and energy transfer of solid reactants while offering advantages such as high energy efficiency and excellent controllability.49–51 Our previous research has successfully applied electromagnetic milling technology to organic synthesis, achieving palladium/copper dual-catalyzed Sonogashira cross-coupling reactions under solvent-free and externally unheated conditions.52 Building on this foundation, this paper reports a Sonogashira cross-coupling reaction utilizing pure nickel catalysis under EMM conditions, achieved without solvents, external heating, or copper co-catalysts, thereby providing a more environmentally friendly and sustainable strategy for constructing C(sp)–C(sp2) bonds (Fig. 1E).
Results and discussion
The research commenced by exploring the reaction between p-tert-butyliodobenzene 1a and phenylacetylene 2a in the presence of [NiCl2(PPh3)2], 1,4-diazabicyclo [2.2.2] octane (DABCO), K3PO4, 4,4′-di-tert-butyl-2,2′-bipyridine (dtbbpy) and steel rods using EMM at a frequency of 50 Hz, which afforded the desired product 3a in 83% yield (Table 1, entry 1). Several nickel catalysts were initially examined, including nickel powder, NiBr2, NiCl2, Ni(dppf)2 and Ni(COD)2. Among these, the reaction with NiCl2 exhibited the best performance, achieving a 90% yield (Table 1, entries 2–6). Further investigation of the reaction components showed that the absence of the ligand dtbbpy resulted in a reduced yield (entry 7), while the elimination of DABCO or K3PO4 led to almost no reaction (entries 8–10). Based on these findings, the influence of various ligands and bases was explored, and it was determined that dtbbpy and K3PO4 were the optimal choices (entries 11–17). When DABCO was replaced with Et3N and DBU, the reaction yields decreased to 80% and 85%, respectively (entries 18 and 19). In contrast, the strongly reducing nature of Zn powder led to preferential reduction of the alkyne to the alkane, rendering the coupling reaction nearly inoperative and resulting in a substantial decrease in the reaction yield (entry 20).The optimal conditions for the reaction were Nicl2 as the catalyst, dtbbpy as the ligand, K3PO4 as the base, DABCO as the additive, and steel rods under EMM at 50 Hz.| Entry | Ni | Additive | Base | Ligand | Rods | Yield (%) |
|---|---|---|---|---|---|---|
| a Conditions: 1a (0.6 mmol, 2 equiv.), 2a (0.3 mmol, 1 equiv.), catalyst (10 mol%), ligand (10 mol%), DABCO (1.5 equiv.), K3PO4 (2.0 equiv.), steel rods (0.3 mm × 5.0 mm, 5 g), 0.15 T, 50 Hz, 3 h. | ||||||
| 1 | NiCl2(PPh3)2 | DABCO | K3PO4 | L1 | Steel rods | 83% |
| 2 | Ni powder | DABCO | K3PO4 | L1 | Steel rods | 15% |
| 3 | NiBr2 | DABCO | K3PO4 | L1 | Steel rods | 85% |
| 4 | NiCl2 | DABCO | K3PO4 | L1 | Steel rods | 90% |
| 5 | Ni(dppf)Cl2 | DABCO | K3PO4 | L1 | Steel rods | 84% |
| 6 | Ni(COD)2 | DABCO | K3PO4 | L1 | Steel rods | 75% |
| 7 | NiCl2 | DABCO | K3PO4 | \ | Steel rods | 12% |
| 8 | NiCl2 | — | K3PO4 | \ | Steel rods | ND |
| 9 | NiCl2 | — | K3PO4 | L1 | Steel rods | ND |
| 10 | NiCl2 | DABCO | — | L1 | Steel rods | ND |
| 11 | NiCl2 | DABCO | K3PO4 | L2 | Steel rods | 60% |
| 12 | NiCl2 | DABCO | K3PO4 | L3 | Steel rods | 74% |
| 13 | NiCl2 | DABCO | K3PO4 | L4 | Steel rods | 75% |
| 14 | NiCl2 | DABCO | K3PO4 | L5 | Steel rods | 76% |
| 15 | NiCl2 | DABCO | K2CO3 | L1 | Steel rods | 73% |
| 16 | NiCl2 | DABCO | Na2CO3 | L1 | Steel rods | 79% |
| 17 | NiCl2 | DABCO | Cs2CO3 | L1 | Steel rods | 85% |
| 18 | NiCl2 | Et3N | K3PO4 | L1 | Steel rods | 80% |
| 19 | NiCl2 | DBU | K3PO4 | L1 | Steel rods | 85% |
| 20 | NiCl2 | Zn powder | K3PO4 | L1 | Steel rods | 33% |
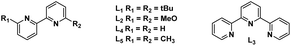 |
||||||
After determining the initial reaction conditions, the equipment parameters were further optimized (Fig. 2). The magnetic field strength was adjusted (0.07–0.15 T) by changing the position of the reaction tube, leading to a proportional increase in reaction efficiency (Fig. 2A). Increasing the frequency from 10 to 50 Hz also enhanced the yields (Fig. 2B). Screening of the ferromagnetic rods identified a diameter of 0.3 mm and a length of 5 mm as optimal, likely due to their ability to maximize collision speed and intensity (Fig. 2C). Finally, a loading mass of 5 g was found to be optimal, as other amounts caused non-uniform agitation (Fig. 2D).
With the optimal conditions established, the substrate scope of the coupling reactions was explored (Table 2). Both electron-donating (tBu, OEt, and H) and electron-withdrawing (Ph, F, and Cl) groups at the para-position of aryl iodides were tolerated, resulting in the desired products 3aa–3fa in moderate to high yields. Ortho-substituted groups (2-Me, 2-Et and 2-Cl) were also tolerated, yielding products 3ga–3ia. Disubstituted substrates at the para- and meta- or ortho-positions of the benzene ring of aryl iodides also afforded the desired products (3ja–3la). Notably, 2-iodofluorene was successfully coupled, providing the desired product 3ma in 61% yield. The scope of arylacetylene substrates was also investigated. Under the optimized reaction conditions, both electron-donating (4-OMe and 4-Me) and electron-withdrawing (4 F, 4-Cl, 2-Cl, and 4-Br) substituted arylacetylenes reacted smoothly with various functionalized aromatic iodides, such as iodobenzene, 4-tert-butyl iodobenzene, and 4-fluoroiodobenzene, affording the corresponding products in acceptable yields (3cb–3ah). The poorly soluble substrate polyaryl halide (1-iodobiphenyl) underwent reactions with phenylacetylene and p-methoxyphenylacetylene, respectively, affording the target products in excellent yields (3da, 81% and 3dd, 58%). Additionally, bromobenzene was also examined under the standard conditions with phenylacetylene, giving the coupling product 3ca in 20% yield.
| a Reaction conditions: 1 (0.6 mmol, 2 equiv.), 2 (0.3 mmol, 1 equiv.), NiCl2 (10 mol%), dtbbpy (10 mol%), DABCO (0.45 mmol ,1.5 equiv.), K3PO4 (0.6 mmol, 2 equiv.), steel rods (0.3 mm × 5.0 mm, 5 g), 0.15 T, 50 Hz, 3 h.b 1 (0.3 mmol, 1 equiv.), 2 (0.6 mmol, 2 equiv.).c 1 (0.3 mmol, 1 equiv.), 2 (0.3 mmol, 1 equiv.). |
|---|
 |
Elemental analysis confirmed the presence of key metallic elements (i.e., Fe, Ni, Cr) in the steel rods. To elucidate the reaction mechanism and determine the origin of the catalytically active nickel, a series of mechanistic and control experiments were conducted (Fig. 3). Initially, the reaction was carried out using Fe rods instead of steel rods, in the absence of externally added NiCl2. No desired product 3ab was detected, confirming that the coupling reaction could not proceed under these conditions (Fig. 3A). Consequently, Fe rods were used to replace steel rods in all subsequent experiments to eliminate potential interference from other elements in the steel.
Subsequently, different loadings of nickel salts (NiCl2(PPh3)2, NiCl2, and NiBr2) were individually combined with Fe rods for the reaction. The results showed that all three nickel salts effectively facilitated the formation of the desired product, with the yield progressively increasing as the dosage of the nickel catalyst was raised (Fig. 3B). Notably, NiCl2 delivered the highest yield among the tested salts, suggesting its superior catalytic efficiency under identical conditions. The linear correlation between catalyst loading and product yield further supports a well-defined catalytic cycle rather than stoichiometric participation. Additionally, control experiments without nickel salts afforded no detectable product, underscoring the indispensable role of the nickel species in enabling this transformation.
To further rule out the contribution of nickel derived from the steel rods, the steel rods were directly milled under EMM coupling conditions, and the resulting trace powder was introduced into the model reaction. No target product 3ab was detected in this case. Meanwhile, control experiments using Ni rods alone or Fe rods combined with Ni powder also failed to yield the desired product (Fig. 3C). Additionally, to exclude any possible synergistic effect between nickel (released from milled steel rods) and Fe/Cr, combined reactions of Ni rods with Fe powder and Cr powder were carried out, but no target product was obtained either (Fig. 3D).
To elucidate the role of DABCO in the reaction, three sets of control experiments were conducted (Fig. 3E). Using NiCl2 as the Ni(II) source with 1.5 equiv. of DABCO delivered the desired product in 74% yield. However, no reaction was detected in the absence of DABCO. These results indicated that Ni(II) could not initiate the coupling reaction, but DABCO could convert it to the active nickel species. Furthermore, using Ni(COD)2 as the Ni(0) source could produce the corresponding product in 32% yield, establishing the intrinsic activity benchmark for Ni(0) catalysts in this system. The addition of 10% of DABCO increased the yield to 38%, demonstrating that DABCO might play an important role in the nickel catalysis cycle. Collectively, these findings unambiguously establish that nickel released from milled steel rods is catalytically inactive in the reaction system, and NiCl2 serves as the true catalytic component for the Sonogashira coupling reaction.
Based on the XPS analysis results (Fig. 4), the following summary can be made: in the reference spectrum of NiCl2, the Ni 2p3/2 peak exhibits a distinct Ni2+ main peak (∼855 eV) accompanied by its characteristic satellite peaks, consistent with an octahedral coordination environment, indicating that nickel exists in a stable divalent state (Fig. 4A). In complex 1, the Ni2+ signal intensity is significantly diminished, with the main peak and satellite peaks nearly submerged by noise, suggesting that under the combined action of mechanical grinding and DABCO, Ni2+ is reduced to a low-valent Ni0 species. Owing to the high electron cloud density and enhanced shielding effect of Ni0, the XPS signal intensity is substantially reduced and the peak shape is broadened, marking the successful generation of the catalytically active zero-valent nickel species (Fig. 4B). In contrast, the spectrum of the sample subjected only to stirring closely resembles that of pure NiCl2, retaining strong and well-defined Ni2+ characteristic signals with only minor changes in peak shape (Fig. 4C), indicating that dtbbpy and DABCO couldn't reduce Ni2+ without mechanical grinding. Furthermore, the mixture of Ni and dtbbpy subjected to mechanical grinding in the absence of DABCO showed no evidence of reduction (Fig. 4D). These results demonstrate that DABCO is the key reductant enabling the Ni2+ → Ni0 transformation, and its reducing capability is dependent on the energy input provided by mechanical grinding.
To assess the practicality and scale-up potential, the reaction was conducted on a 2 mmol scale for gram-scale synthesis (Fig. 5). The transformation proceeded smoothly, delivering the target product 3aa in 80% yield (0.37 g), thereby establishing a foundation for its potential industrial application.
Based on the combination of the above experiments and reported literature, a plausible catalytic cycle is delineated in Fig. 6. Ni(II) in NiCl2 is reduced under a magnetic field and in the presence of DABCO, forming a Ni0L2 complex with the ligand dtbbpy. Subsequently, the oxidative addition of [dtbbpyNi0] (4) with aryl iodides gave the aryl-Ni(II)-iodide complex 5. The coordination and subsequent transfer of the alkyne to 5 yielded intermediate 6 in the presence of K3PO4 and DABCO. Finally, reductive elimination resulted in the formation of the target product 3 and the regeneration of the Ni0 catalyst under the magnetic field. Generally, reductive elimination from a Ni(II) complex is a thermodynamically unfavorable process. In this strategy, the promoted reductive elimination from complex 6 may be attributed to the magnetic field.
Conclusions
In summary, an electromagnetic mill-promoted Sonogashira coupling reaction of aryl halides and terminal alkynes was developed, utilizing NiCl2 as the nickel catalyst source. This method demonstrated significant advantages by eliminating the need for additional precious transition metal catalysts, solvents, inert-gas atmospheres, and external heating. The easily operable methodology also showed excellent tolerance to various substitutions and a wide substrate scope. Mechanistic investigations revealed that the Ni(II) precursor in NiCl2 is activated to form a Ni(0) catalyst under the influence of the magnetic field and DABCO, facilitating the coupling reaction. The electromagnetic milling enhances the transfer of alkynes to the Ni(II) species. This strategy is expected to provide a viable alternative for alkyne synthesis.Author contributions
All authors have reviewed the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information (SI): all supporting experimental data, including copies of 1H, 19F and 13C NMR spectra. See DOI: https://doi.org/10.1039/d6mr00022c.References
- K. C. Nicolaou, C. W. Hummel, E. N. Pitsinos, M. Nakada, A. L. Smith, K. Shibayama and H. Saimoto, J. Am. Chem. Soc., 1992, 114, 10082–10084 CrossRef CAS.
- J. Taunton, J. L. Wood and S. L. Schreiber, J. Am. Chem. Soc., 1993, 115, 10378–10379 CrossRef CAS.
- Y. Koyama, M. J. Lear, F. Yoshimura, I. Ohashi, T. Mashimo and M. Hirama, Org. Lett., 2005, 7, 267–270 CrossRef CAS PubMed.
- I. Paterson and T. Paquet, Org. Lett., 2010, 12, 2158–2161 CrossRef CAS PubMed.
- R. Chinchilla and C. Nájera, Chem. Soc. Rev., 2011, 40, 5084–5121 RSC.
- B. De Carné-Carnavalet, A. Archambeau, C. Meyer, J. Cossy, B. Folléas, J. L. Brayer and J. P. Demoute, Chem. Eur. J., 2012, 18, 16716–16727 CrossRef PubMed.
- B. Vaz, L. Otero, R. Álvarez and Á. R. de Lera, Chem. Eur. J., 2013, 19, 13065–13074 CrossRef CAS PubMed.
- M. Esmaeilpour, A. Sardarian and J. Javidi, Catal. Sci. Technol., 2016, 6, 4005–4019 RSC.
- J. S. Cyniak, Ł. Kocobolska, N. Bojdecka, A. Gajda-Walczak, A. Kowalczyk, B. Wagner, A. Nowicka, H. Sakurai and A. Kasprzak, Dalton Trans., 2023, 52, 3137 RSC.
- K. Sonogashira, Y. Tohda and N. Hagihara, Tetrahedron Lett., 1975, 16, 4467–4470 CrossRef.
- M. S. M. Ahmed and A. Mori, Org. Lett., 2003, 5, 3057 CrossRef CAS PubMed.
- T. Lauterbach, M. Livendahl, A. Rosellón, P. Espinet and A. M. Echavarren, Org. Lett., 2010, 12, 3006–3009 CrossRef CAS PubMed.
- M. Trunk, A. Herrmann, H. Bildirir, A. Yassin, J. Schmidt and A. Thomas, Chem. Eur. J., 2016, 22, 7179–7183 CrossRef CAS PubMed.
- D. D. Vo and M. Elofsson, ChemistrySelect, 2017, 2, 6245–6248 CrossRef CAS.
- H. Yanık, S. Yesilot and M. Durmus, Dyes Pigm., 2017, 140, 157–165 CrossRef.
- C. M. R. Volla and P. Vogel, Tetrahedron Lett., 2008, 49, 5961–5964 CrossRef.
- M. Carril, A. Correa and C. Bolm, Angew. Chem., Int. Ed., 2008, 47, 4862–4865 CrossRef CAS PubMed.
- H. Huang, H. Jiang, K. Chen and H. Liu, J. Org. Chem., 2008, 73, 9061–9064 CrossRef CAS PubMed.
- M. Karak, L. C. A. Barbosa and G. C. Hargaden, RSC Adv., 2014, 4, 53442–53466 RSC.
- L. Jin, W. F. Hao, J. N. Xu, N. Sun, B. X. Hu, Z. Shen, W. Mo and X. Hu, Chem. Commun., 2017, 53, 4124–4127 RSC.
- A. Hazra, M. T. Lee, J. F. Chiu and G. Lalic, Angew. Chem., Int. Ed., 2018, 57, 5492–5496 CrossRef CAS PubMed.
- J. Y. Song, X. Zhou, H. Song, Y. Liu, H. Y. Zhao, Z. Z. Sun and W. Y. Chu, ChemCatChem, 2018, 10, 758–762 CrossRef CAS.
- D. Gallego, A. Brueck, E. Irran, F. Meier, M. Kaupp, M. Driess and J. F. Hartwig, J. Am. Chem. Soc., 2013, 135, 15617–15626 CrossRef CAS PubMed.
- N. Hussain, P. Gogoi, P. Khare and M. R. Das, RSC Adv., 2015, 5, 103105–103115 RSC.
- D. L. Zhu, R. J. Xu, Q. Wu, H. Y. Li, J. P. Lang and H. X. Li, J. Org. Chem., 2020, 85, 12457–12465 Search PubMed.
- H. Chen, L. Yao, L. Guo, Y. A. Liu, B. Tian and X. Liao, Cell Rep. Phys. Sci., 2023, 4, 101573 CrossRef CAS.
- G.-W. Wang, Chem. Soc. Rev., 2013, 42, 7668–7700 RSC.
- K. Kubota, Y. Pang, A. Miura and H. Ito, Science, 2019, 366, 1500–1504 CrossRef CAS PubMed.
- J. Zhang, P. Zhang, L. Shao, R. H. Wang, Y. Ma and M. Szostak, Angew. Chem., Int. Ed., 2022, 61, e202114146 CrossRef CAS PubMed.
- R. R. A. Bolt, S. E. Raby-Buck, K. Ingram, J. A. Leitch and D. Browne, Angew. Chem., Int. Ed., 2022, 61, e202210508 CrossRef CAS PubMed.
- X. Yang, C. Wu, W. Su and J. Yu, Eur. J. Org Chem., 2022, 6, e202101440 CrossRef.
- X. Yang, H. Wang, Y. Zhang, W. Su and J. Yu, Green Chem., 2022, 24, 4108–4116 RSC.
- C. Wu, T. Ying, H. Fan, C. Hu, W. Su and J. Yu, Org. Lett., 2023, 25, 2531–2536 CrossRef CAS PubMed.
- A. R. R. Hernandez, I. Trakakis, J. Cuccia and T. Friščić, Eur. J. Org Chem., 2023, 26, e202300374 CrossRef.
- T. Seo, K. Kubota and H. Ito, J. Am. Chem. Soc., 2023, 145, 6823–6837 CrossRef CAS PubMed.
- C. Patel, E. André-Joyaux, J. Leitch, X. Irujo-Labalde, F. Ibba, J. Struijs, M. Ellwanger, R. Paton, D. Browne, G. Pupo, S. Aldridge, M. Hayward and V. Gouverneur, Science, 2023, 381, 302–306 CrossRef CAS PubMed.
- S. Pan, F. F. Mulks, P. Wu, K. Rissanen and C. Bolm, Angew. Chem., Int. Ed., 2024, 63, e202316702 CrossRef CAS PubMed.
- X. Wang, X. Zhang, X. He, G. Guo, Q. Huang, F. Q. Wang, R. Qu, F. Zhou and Z. Lian, Angew. Chem., Int. Ed., 2024, 63, e202410334 CrossRef CAS PubMed.
- R. Qu, S. Wan, X. Zhang, X. Wang, L. Xue, Q. Wang, G. Cheng, L. Dai and Z. Lian, Angew. Chem., Int. Ed., 2024, 63, e202400645 CrossRef CAS PubMed.
- H. Lei, B. Wang, Y. Yang, S. Fan, S. Wang and X. Wei, Org. Lett., 2024, 26, 7688–7694 CrossRef CAS PubMed.
- M. Hribersek, C. Méndez-Gálvez, M. Huber, P. Gates, P. Shakari, A. Samanta and L. Pilarski, Green Chem., 2023, 25, 9138–9145 RSC.
- S. Hutsch, A. Leonard, S. Graetz, M. Höfler, T. Gutmann and L. Borchardt, Angew. Chem., Int. Ed., 2024, 63, e202403649 CrossRef CAS PubMed.
- V. Chantrain, T. Rensch, W. Pickhardt, S. Grätz and L. Borchardt, Chem. Eur. J., 2024, 30, e202304060 CrossRef CAS PubMed.
- D. A. Fulmer, W. C. Shearouse, S. T. Mendonza and J. Mack, Green Chem., 2009, 11, 1821 RSC.
- R. Thorwirth, A. Stolle and B. Ondruschka, Green Chem., 2010, 12, 985 RSC.
- Y. Gao, C. Feng, T. Seo, K. Kubota and H. Ito, Chem. Sci., 2022, 13, 430–438 RSC.
- W. Pickhardt, E. Siegfried, S. Fabig, M. F. Rappen, M. Etter, M. Wohlgemuth, S. Grätz and L. Borchardt, Angew. Chem., Int. Ed., 2023, 62, e202301490 CrossRef CAS PubMed.
- W. Pickhardt, F. J. L. Kraus, M. Wohlgemuth, M. Etter, M. Spallek, M. Schwensow, Z. Genç, S. Grätz and L. Borchardt, ChemCatChem, 2024, 16, e202400491 CrossRef CAS.
- M. Wołosiewicz-Głąb, S. Ogonowski and D. Foszcz, IFAC-PapersOnLine, 2016, 49, 67 CrossRef.
- M. Wołosiewicz-Głąb, D. Foszcz and S. Ogonowski, IFAC-PapersOnLine, 2017, 50, 14964 CrossRef.
- M. Pawelczyk, Z. Ogonowski, S. Ogonowski, D. Foszcz, D. Saramak, T. Gawenda and D. Krawczykowski, Minerals, 2018, 8, 138 Search PubMed.
- X. Zhang, Q. Liu, J. Gao, L. Zhang, F. Yan, S. Zhou and H. Liu, Adv. Synth. Catal., 2025, 367, e202500100 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |







