Open Access Article
Open Access ArticleLithium-mediated mechanochemical annulative dimerization of diarylacetylenes for synthesis of 1,4-dihydrodinaphthopentalenes
Koya M.
Hori†
 a,
Yoshifumi
Toyama†
a,
Yoshifumi
Toyama†
 a,
Takato
Mori
a,
Takato
Mori
 a,
Takumu
Nakamura
b,
Yohei
Ono
b and
Hideto
Ito
a,
Takumu
Nakamura
b,
Yohei
Ono
b and
Hideto
Ito
 *a
*a
aGraduate School of Science, Nagoya University, Chikusa, Nagoya 464-8602, Japan. E-mail: ito.hideto.p4@f.mail.nagoya-u.ac.jp
bTokyo Research Center, Advanced Materials Research Laboratory, Advanced Integration Research Center, Research Devision, Tosoh Corporation, 2743-1 Hayakawa, Ayase, Kanagawa 252-1123, Japan
First published on 5th February 2026
Abstract
1,4-Dihydrodiarenopentalenes, the reduced derivatives of diarenopentalene, have attracted attention owing to their rigid planar π-conjugated ladder framework, photophysical properties, C2-chiral structure, and potential applications in physical organic chemistry and materials science. Unlike the established methods for parent diarenopentalenes, the synthetic method for 1,4-dihydrodiarenopentalenes is less established, and the constructed structures are highly limited to dihydrodibenzopentalenes with trialkylsilyl substituents. Herein, we report a novel method for synthesizing 1,4-dihydrodinaphthopentalene derivatives by mechanochemical annulative dimerization of 2-(arylethynyl)naphthalenes with a lithium(0) wire. This reaction is easy to handle—even in an ambient atmosphere at room temperature—and requires a minimum amount of tetrahydrofuran for efficient grinding, resulting in the formation of various 1,4-diaryl-1,4-dihydrodinaphthopentalenes within 15 min. Furthermore, structural and theoretical analyses, as well as photophysical and chiro-optical measurements, reveal untapped properties of chiral 1,4-dihydrodinaphthopentalene.
Introduction
Dibenzo[a,e]pentalene or indenoindene is a distinctive example of a ladder-type polycyclic π-conjugated hydrocarbon, characterized by a fused 6-5-5-6-membered ring system (Fig. 1A).1 This molecule and its derivatives have received marked interest owing to their narrow highest occupied molecular orbital (HOMO)-lowest unoccupied molecular orbital (LUMO) gaps2 and anti-aromatic 8π-electron system stabilized by two benzo-fusions. Therefore, various stoichiometric and catalytic preparation methods have been developed so far.3 The hydrogenated homologues of dibenzo[a,e]pentalenes, namely, dihydrodibenzopentalenes or dihydroindenoindene, have also garnered interest as π-conjugated ladder platforms.4,5 Dihydrodibenzopentalene possesses a fixed, rigid, and planar C2-symmetric stilbene structure. Depending on the substituents at the benzylic positions (R and R′), it possesses a planar chirality. This framework serves as a synthetic precursor for chiral dibenzocyclooctatetraene, a so-called “chiral diene” ligand, for asymmetric catalysis.6a,b Incorporating the multi-aryldihydropentalene moiety into an oligo- or polyarylene is known as an effective strategy to elongate and kinetically stabilize the π-conjugated system in the design of organic electronic materials.7 Classical stepwise synthetic routes to dihydrodibenzopentalene include derivatization of the dihydropentalene-1,4-dione derivative,8 dimerizative annulation of ethyl phenylacetate,4,5 and transannulation of dibenzocyclooctatetraene derivatives.6 Tsuji et al. reported the synthesis of dihydrodibenzopentalenes and its oligomers through a tandem cyclization reaction in ortho-substituted diphenylacetylene derivatives.9 Saito and Kuwabara reported a one-step synthesis of disilylated dibenzo[a,e]pentalenes from 1-phenyl-2-(trialkylsilyl)acetylenes using bulk lithium or sodium dispersion in the solution state. Further transformation into tri- and tetra-substituted dihydrodibenzopentalenes via the stepwise addition of MeLi and electrophiles has also been demonstrated.10,11 Furthermore, Xi12 also reported a one-pot synthesis of similar disilylated dihydrodibenzopentalene via dimerizative dilithiation of 1-phenyl-2-silylacetylenes with lithium,10,11 followed by reaction with Ba[N(SiMe3)2]2 and quenching with H2O (Fig. 1B). Although these alkyne dimerization methods are attractive in terms of step economy and availability of the starting alkynes, the scope of alkynes is highly limited to 1-phenyl-2-(trialkylsilyl)acetylenes, and alkyne dimerization using diarylacetylene or examples of diarenodihydropentalenes other than dibenzo-fused structures have not been reported to date. This could be due to the stability and reactivity of 1,4-dilithio-1,3-butadiene bearing α-silyl groups,10,12,13 which is easily generated in situ by the reaction of 1-phenyl-2-silylacetylenes with lithium in tetrahydrofuran (THF). To expand the scope of the available diarenodihydropentalenes, the development of alternative and/or more feasible methods is urgently required. | ||
| Fig. 1 (A) Dibenzo[a,e]pentalene and dihydrodibenzopentalene. (B) Previous reports on synthesis of dihydrodibenzopentalenes. (C) Mechanochemical synthesis of dihydrodinaphthopentalenes (this work). | ||
In this regard, we and the Ito/Kubota group have recently developed lithium-mediated mechanochemical transformations of aromatic compounds, such as anionic cyclodehydrogenation,13a Birch reduction,14a Birch reductive arylation,13a and related transformations14b using bulk lithium. Despite the fact that bulk lithium sources (e.g., lithium wire) generally exhibit low reactivity in the solution state owing to their higher atomization enthalpy and melting point than those of sodium and potassium,13a direct vigorous mixing by ball-milling pieces of lithium(0) wire and aromatic compounds with stoichiometric amounts of additives forms aromatic anionic species even in air, realizing highly efficient, quick transformations13a,14 and unprecedented reactions.13b Over the course of our mechanochemistry investigation,15,16 we surprisingly found that mechanochemical grinding of 2-(arylethynyl)naphthalene 1 with lithium(0) wire and aqueous work-up resulted in unexpected formation of 1,4-diaryl-1,4-dihydrodinaphthopentalene 2 (Fig. 1C). To the best of our knowledge, there is no example of annulative dimerization of bare diarylacetylene with alkali metals, whereas 1-phenyl-2-(trialkylsilyl)acetylenes can undergo annulation with lithium(0) or potassium(0) in the solution state using THF to afford dibenzo[a,e]pentalene.10–12 The dihydrodinaphthopentalene core itself is unprecedented, and its fixed planar π-conjugation system, crystallographic packing, photophysical properties, and potential as a novel chiral platform are of interest to the physical organic chemistry community. Herein, we report the synthesis of dihydrodinaphthopentalenes with two dangling aryl substituents via lithium(0)-mediated mechanochemical annulative dimerization of 2-(arylethynyl)naphthalenes. Unlike typical solution-state reactions using alkali metals, the developed mechanochemical reaction can be performed in air at room temperature, without solvents for solubilizing the substrates. The prepared compounds exhibit novel dihydrodinaphthopentalene skeletons, and their crystallographic structures, photophysical and chiroptical properties are characterized.
Results and discussion
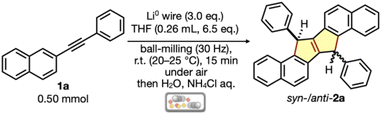
Through exhaustive optimization of the reaction conditions, we found that mechanochemical annulative dimerization of 2-(phenylethynyl)naphthalene (1a) (0.50 mmol, 1.0 eq.) proceeded using liquid-assisted grinding (LAG)17 at room temperature (r.t.: 20–25 °C) under air by ball milling with lithium wire pieces (3 eq.), a small amount of THF (0.26 mL, 6.5 eq.) and a 7-millimeter-diameter stainless-steel ball in a 1.5 mL stainless–steel jar (Table 1, entry 1). After ball milling for 15 min, the jar was opened to air, and the deep-black slurry was quenched by adding H2O. Aqueous workup using sat. NH4Cl aq. afforded dihydrodinaphthopentalene 2a as the major product, with a 23% 1H-NMR yield as a 7.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 syn-/anti-mixture. In this reaction, various side products, such as fully aromatized dinaphthopentalene and alkyne reduction products, appear to form, which lowers the yield and mass balance of the reaction. Purification by size-exclusion chromatography (SEC) enabled the isolation of syn-2a in 16% yield. The isolated yield of syn-2a was unsatisfactory, likely owing to the difficulty in separating it from other isomers and impurities during SEC purification. The relative configuration of syn-2a was determined by the X-ray diffraction (XRD) analysis of a single crystal (see Fig. 4A). The use of 4 eq. of lithium(0) also afforded 2a in 21% NMR yield, with increased syn-selectivity (syn/anti = 22
1.0 syn-/anti-mixture. In this reaction, various side products, such as fully aromatized dinaphthopentalene and alkyne reduction products, appear to form, which lowers the yield and mass balance of the reaction. Purification by size-exclusion chromatography (SEC) enabled the isolation of syn-2a in 16% yield. The isolated yield of syn-2a was unsatisfactory, likely owing to the difficulty in separating it from other isomers and impurities during SEC purification. The relative configuration of syn-2a was determined by the X-ray diffraction (XRD) analysis of a single crystal (see Fig. 4A). The use of 4 eq. of lithium(0) also afforded 2a in 21% NMR yield, with increased syn-selectivity (syn/anti = 22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0). Reducing the amount of lithium(0) from 3 equiv. to 2 or 1 eq. decreased the yield of 2a and syn-selectivity (10% with syn/anti = 3.3
1.0). Reducing the amount of lithium(0) from 3 equiv. to 2 or 1 eq. decreased the yield of 2a and syn-selectivity (10% with syn/anti = 3.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0; 6% with syn/anti = 3.0
1.0; 6% with syn/anti = 3.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0) (Table 1, entries 2 and 3). In controlled experiments, the use of other LAG additives, such as 1,4-dioxane, 1,2-dimethoxyethane (DME), cyclopentyl methyl ether (CPME), N,N,N′,N′-tetramethylethylenediamine (TMEDA), and hexane resulted in low yields or no formation of 2a (Table 1, entries 5–7). The addition of 8.0 eq. of THF yielded 2a in an amount comparable to that of the reaction under standard conditions (entry 8), whereas the reactions conducted with 4.6 or 16 eq. of THF were ineffective (entries 9 and 10). Compared with mechanochemical grinding, no reaction occurred when lithium naphthalenide (4.0 eq.) was added to THF (10 mL) at 0 °C (entry 11). Mechanochemical grinding and direct heating using a heat gun (70 °C preset temperature) did not improve the yield of 2a (20%, syn/anti = 4.5
1.0) (Table 1, entries 2 and 3). In controlled experiments, the use of other LAG additives, such as 1,4-dioxane, 1,2-dimethoxyethane (DME), cyclopentyl methyl ether (CPME), N,N,N′,N′-tetramethylethylenediamine (TMEDA), and hexane resulted in low yields or no formation of 2a (Table 1, entries 5–7). The addition of 8.0 eq. of THF yielded 2a in an amount comparable to that of the reaction under standard conditions (entry 8), whereas the reactions conducted with 4.6 or 16 eq. of THF were ineffective (entries 9 and 10). Compared with mechanochemical grinding, no reaction occurred when lithium naphthalenide (4.0 eq.) was added to THF (10 mL) at 0 °C (entry 11). Mechanochemical grinding and direct heating using a heat gun (70 °C preset temperature) did not improve the yield of 2a (20%, syn/anti = 4.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) (Table 1, entry 12). By using a 5 mL stainless–steel jar and a 10-millimeter-diameter stainless–steel ball instead of a 1.5 mL jar and a 7 mm ball, the yield of 2a dramatically dropped to 1% (entry 13). Although we tested lower-frequency ball-milling at 20 Hz to prevent overreaction and lowering the product yield (entry 14),19 the yield of 2a rather decreased to 9.7% (syn/anti = 5.4
1) (Table 1, entry 12). By using a 5 mL stainless–steel jar and a 10-millimeter-diameter stainless–steel ball instead of a 1.5 mL jar and a 7 mm ball, the yield of 2a dramatically dropped to 1% (entry 13). Although we tested lower-frequency ball-milling at 20 Hz to prevent overreaction and lowering the product yield (entry 14),19 the yield of 2a rather decreased to 9.7% (syn/anti = 5.4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) upon the full consumption of starting material. Notably, the reaction under argon atmosphere did not improve the yield of 2a (entry 15). These results indicate that the mechanochemical reaction is highly specific in the solid or slurry state, and that the amount of THF additive, the size of the jar/ball and mixing frequencies greatly influence the reaction progress.
1) upon the full consumption of starting material. Notably, the reaction under argon atmosphere did not improve the yield of 2a (entry 15). These results indicate that the mechanochemical reaction is highly specific in the solid or slurry state, and that the amount of THF additive, the size of the jar/ball and mixing frequencies greatly influence the reaction progress.
| Entry | Deviation from the standard conditions | Yield of 2ab | Ratio of syn-/anti-2a |
|---|---|---|---|
| a Reactions were conducted in a 1.5 mL stainless–steel jar using a stainless–steel ball with a diameter of 7 mm by ball milling using a Retsch MM400 ball mill machine. The reactions were conducted under air at r.t. (20–25 °C). b 1H-NMR yield using Cl2CHCHCl2 as an internal standard. c Isolated yield of syn-2a by SEC. d The reaction was carried out in a two-neck round-bottom flask using a magnetic stirring bar and a stirrer under N2. | |||
| 1 | None | 23% (syn-2a: 16%c) | 7.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 1.0 |
| 2 | Li (4.0 eq.) instead of 3.0 eq. | 21% | 22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 1.0 |
| 3 | Li (2.0 eq.) instead of 3.0 eq. | 10% | 3.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 1.0 |
| 4 | Li (1.0 eq.) instead of 3.0 eq. | 6% | 3.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 1.0 |
| 5 | 1,4-Dioxane was used instead of THF | 3% | 2.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 1.0 |
| 6 | DME instead of THF | 2% | 1.4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 1.0 |
| 7 | CPME, TMEDA, or hexane instead of THF | 0% | — |
| 8 | 0.32 mL (8.0 eq.) of THF | 22% | 5.9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 1.0 |
| 9 | 0.19 mL (4.6 eq.) of THF | 0% | — |
| 10 | 0.64 mL (16 eq.) of THF | 5% | 1.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.1 1.1 |
| 11 | Li naphthalenide (4.0 eq.), THF (10 mL), 0 °C, N2, 3 h | 0% | — |
| 12 | Heating by heat gun at a preset temperature of 70 °C | 20% | 4.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 1.0 |
| 13 | Reaction conducted in a 5.0 mL stainless–steel jar using a stainless–steel ball with a diameter of 10 mm | 1% | 1.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 1.0 |
| 14 | Ball-milling was conducted at 20 Hz | 9.7% | 5.4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 1.0 |
| 15 | Reaction under Ar | 23% | 4.9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.0 1.0 |
Next, we performed control experiments to elucidate the reaction profile of the mechanochemical alkyne dimerization. In the deuteration experiments, the addition of D2O instead of H2O followed by sat. NH4Cl aq. in the quenching step produced a mixture of 2a and deuterated product (2a-D) in 11% isolated yield with a 97% deuteration ratio (Fig. 2A). In contrary, additional grinding for 60 min after adding D2O alone resulted in dramatical drop of isolated yield (2.8%) along with almost completion of deuteration at the two benzylic positions (98%D). These indicates that the bis(cyclopentadienyl anion) species E (Fig. 2K) is relatively stable and remained intact even after the addition of D2O, but is rapidly protonated with sat. NH4Cl aq. In addition, extending the mixing time under basic conditions can induce significant degradation. Then we demonstrated trapping this dianion species by other electrophiles. The addition of iodomethane10 and extra grinding for 60 min afforded the dimethylated product 2a-Me in 16% yield as a syn- and anti-mixture (Fig. 2B). Based on our previous findings of the Birch reductive arylation of polycyclic aromatic hydrocarbons with fluoroarenes,13b we found that a similar arylation occurred in the presence of 2-fluoropyridine, affording pyridylated dihydrodinaphthopentalene (2a-Py) in 28% yield (Fig. 2C). Expecting that quenching with I2/THF as an oxidant can afford dinaphthopentalene 3a as well demonstrated in the synthesis of silyl-substituted dibenzo[a,e]pentalene,10 we tried the similar reaction as shown in Fig. 2D. Although the formation of 3a, which was previously synthesized by Takimiya and Kawase20 using an alternative synthetic method, was certainly detected by 1H and 13C NMR spectra and EI-MS, it was obtained as an inseparable mixture of other side products (see Fig. S3 and S4 for details). Interestingly, 1-(phenylethynyl)naphthalene (1b) did not afford the corresponding dihydrodinaphthopentalene 2b (Fig. 2E). Furthermore, the use of diphenylacetylene (1c) afforded a completely different product, 1,2,3-triphenylnaphthalene (4), in 31% yield without any dihydropentalene derivative 2c (Fig. 2F). The similar reaction was also recently reported by Kuwabara, who carried out the reaction in the solution state using lithium(0) and NaOtBu in THF.11a The further early report by Smith and Hoehn also clarified that the reaction of 1c with a stoichiometric amount of lithium(0) in Et2O affords 1,4-dilithio-1,2,3,4-tetraphenyl-1,3-butadiene (A in Fig. 2J), and an excess amount of lithium(0) further promotes the cyclization to not the five-membered-ring intermediate B, but the six-membered-ring product 4.11b In fact, the mechanochemical reaction of 1c with lithium(0) and Et2O for 15 min and aqueous work-up also gave (1E,3E)-1,2,3,4-tetraphenyl-1,3-butadiene (5) in 33% yield (Fig. 2G), which implies that dilithio intermediate A formed and the subsequent cyclization is slower with Et2O than that with THF. To trap the similar intermediate, the mechanochemical reaction of 1a was examined with Et2O for 15 min, resulting in the formation of complex mixture including a major compound having 458 of m/z as a mass peak in APCI-MS analysis (Fig. 2H). While the isolation of the major compound was failed due to the complexity of crude mixture and the presence of various rotamers, two possible structures 6 and 7 can be considered by judging from 1H NMR and MS analysis (Fig. 2I). Particularly, butadiene 6 seem to be contained because the singlet olefinic proton peak in 1H NMR at δ = 6.8 ppm in ClCD2CD2Cl is very resemble to that observed in compound 5 (δ = 6.75–6.73 ppm in CDCl3). This result would be indirect evidence for the initial formation of 1,4-dilithio-1,3-butadiene species derived from 1a.
 | ||
| Fig. 2 (A–H) Control experiments for elucidation of reaction mechanism. (I) Plausible structures for m/z = 458. (J) Proposed reaction pathway to 4 from 1c based on ref. 12b. (K) Proposed reaction pathway in the reaction of 1a with lithium(0). | ||
Based on these control experiments and our expectation, we summarized the proposed mechanism to afford 2c and 4 from 1a and 1c in (Fig. 2J and K). In the reaction of 1c with lithium(0), 1,4-dilithio-1,2,3,4-tetraphenyl-1,3-butadiene (A) was preferentially forms as a s-cis form.10,12b It is well known that there is an equilibrium between s-cisA and s-transA, which vias to the s-cis form.10,12 While the cyclization of s-transA is expected difficult because it accompanies energetically unfavorable dearomatization of a benzene ring. In the case of the reaction of 1a, the formation of similar 1,4-dilithio-1,3-butadiene C can initially form, and its s-trans form can be allowed to cyclize in a fashion of 5-endo-trig to afford allyl lithium species D. Then, the formation of bis(cyclopentadienyl) dilithium E10via a second cyclization and elimination of benzylic hydrogen atoms followed by quenching with electrophiles affords dihydrodinaphthopentalene derivatives. We rationalize that 1b and 1c did not afford the corresponding dihydropentalenes because the stabilities of the anions corresponding to B were relatively low, that is, the π-benzyl-allyl anion D derived from 1a is more stable than those generated from 1b and 1c. The low yield of 2a could also be due to the lower stability of bis(cyclopentadienyl) dianion E than the corresponding disilylated anion derived from 2-trialkylsilylphenylacetylene,10,12 and the active anion species degraded much more easily in the present reaction.
Next, we explored the substrate scope of various 2-(arylethynyl)naphthalenes 1d–1j. All experiments were performed on a 0.50 mmol scale under air at r.t. (Fig. 3). The use of 2-[{4-(t-butyl)phenyl}ethynyl]naphthalene (1d) afforded a mixture of syn-/anti-2d in 79% yield, and syn-2d was isolated in 18% yield after purification by SEC. The reaction of 2-[(4-methoxyphenyl)ethynyl]naphthalene (1e) proceeded, affording the corresponding diarylated pentalenes syn-/anti-2e in 29% yield. However, isolation by SEC resulted in 4.6% (syn-2e) and 1.5% (anti-2e) yields owing to significant degradation of products in purification. In addition, the reaction of naphthylacetylene with p-(diphenylamino)phenyl group 1f afforded a mixture of syn-/anti-2f in 33% yield, and syn-/anti-2f was isolated by SEC in 8.0% and 6.6% yields, respectively. On the other hand, phenylethynylnaphthalenes containing electron-withdrawing groups, such as 1-[(4-fluorophenyl)ethynyl]naphthalene (1g) and 1-[(3-cyanophenyl)ethynyl]naphthalene (1h), were found to be ineffective in this mechanochemical dimerization. The reaction of 1g underwent to give trace amount of 2g along with the major formation of defluorinated product 2a which can be due to lithium-halogen exchanging.16f In contrast, majority of 1h remained intact after the reaction. Then, 1-(arylethynyl)naphthalenes bearing naphthalen-2-yl (1i) and triphenylene-2-yl (1j) moieties were transformed into a syn-/anti-mixture of 2i (25%) and 2j (14%), and SEC separation afforded syn-2i in 4.6% yields. While the final isolated yields of syn and/or anti isomers were unsatisfactory owing to the difficulties in SEC separation and the significant degradation in SEC, the combined yields of syn-/anti-2 at the silica gel column chromatography were moderate. Notably, a series of compounds are relatively unstable under air and basic conditions probably due to the presence of reactive benzylic/allylic protons on cyclopentadiene structures, and the products having electro-donating aryl groups 2e and 2f show lower stabilities than others. This mechanochemical reaction protocol provides a rapid, simple one-pot synthesis of novel dihydrodinaphthopentalenes.
Because the obtained dihydrodinaphthopentalene has a novel structural motif similar to that of dihydropentalene and can be regarded as a fixed planar stilbene derivative with a C2-symmetric chiral structure, its structural, electronic, photophysical, and chiro-optical properties are of interest. First, we obtained a single crystal of syn-2a and determined its X-ray crystallographic structure (Fig. 4A). It exhibited a slightly bent but almost planar bismethylene-bridged dinaphthylethene core with two dangling phenyl groups whose benzene rings were parallel to each other. In the packing structure, two identical enantiomers were paired and stacked on sterically less-hindered faces through weak CH–π interactions between benzylic protons and aromatic sp2-carbons. This pair (green molecules in Fig. 4A, bottom right) and the opposite enantiomeric pairs (orange molecules in Fig. 4A, bottom right) were alternately aligned. In this packing structure, π–π stacking was not apparent between each molecule.
Next, we measured the UV-vis absorption spectrum of a solution of syn-2a in CH2Cl2 (c = 2.0 × 10−5 M) (Fig. 4B). A sharp absorption maximum at λabs = 284 nm and broad absorption bands containing absorption maxima at λabs = 377 and 396 nm were observed. Compared with a major absorption peak at 333 nm in the longest-wavelength region (300–390 nm) of the absorption spectrum of (E)-1,2-di(naphthalen-2-yl)ethene (8) (see ESI and ref. 18), which features a partial structural motif of syn-2a, the absorption band of syn-2a was red-shifted to 320–430 nm, with a maximum at 377 nm. The molar absorptivity (ε) at the peak maximum diminished from 8.1 × 104 M−1 cm−1 (8 in CH2Cl2; see ESI) to 9.3 × 103 M−1 cm−1 (syn-2a). The red shift of the peak maximum can be attributed to the effect of π-extension derived from a fixed planarity by bis-methylene bridging. The decreased absorptivity was also rationalized by the suppressed allowed transitions caused by the breakdown of molecular symmetry by molecular vibrations and bond rotations.
In the frontier molecular orbitals (MOs) calculated at the B3LYP/6-31G+(d) level of theory, the HOMO and LUMO were mainly delocalized across the dihydrodinaphthopentalene core (Fig. 4C). Time-dependent density functional theory (TD-DFT) calculations at the B3LYP/6-311 + G(d,p)//B3LYP/6-31 + G(d) level also revealed that the experimental absorption maximum at λabs = 396 nm is well reproduced by the allowed HOMO→LUMO transition (S0→S1: λ = 409 nm, oscillator strength (f) = 0.37). By contrast, the second transition, arising from HOMO→LUMO+1 excitations (S0→S2: λ = 372 nm, f = 0.0002) are essentially forbidden (see ESI for details of other transitions). Furthermore, syn-2a exhibited fluorescence upon excitation with 380 nm light, and a broad emission band from 390 to 570 nm with a maximum at λem = 434 nm was observed (Fig. 4B). The emission maximum was also significantly red-shifted compared with that of compound 5 (λem = 384 nm; see ESI for details).
After optimizing the separation conditions, each enantiomer in racemic syn-2a was successfully resolved by high-performance liquid chromatography (HPLC) using a chiral column (DAICEL CHIRALPAK IF) and n-hexane/CH2Cl2 (80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20) as the eluent. In the circular dichroism (CD) spectra, the earlier- and later-eluted fractions show levo (green line in Fig. 5A) and dextro (orange line in Fig. 5A) optical rotations in the 230–300 nm range, and the two spectra were almost mirror images. In addition, opposite-sign Cotton effects were observed at ca. 300 nm, which were attributed to the phenyl rings around the chiral carbon centers. However, significant optical rotations were not observed in the range of 330–400 nm, which corresponds to the absorption of the dihydrodinaphthopentalene core. The CD spectrum of (7R,14R)-syn-2a simulated by TD-DFT (Fig. 5B) was identical to the experimentally obtained spectrum of the earlier-eluted fraction (green line in Fig. 5A).
20) as the eluent. In the circular dichroism (CD) spectra, the earlier- and later-eluted fractions show levo (green line in Fig. 5A) and dextro (orange line in Fig. 5A) optical rotations in the 230–300 nm range, and the two spectra were almost mirror images. In addition, opposite-sign Cotton effects were observed at ca. 300 nm, which were attributed to the phenyl rings around the chiral carbon centers. However, significant optical rotations were not observed in the range of 330–400 nm, which corresponds to the absorption of the dihydrodinaphthopentalene core. The CD spectrum of (7R,14R)-syn-2a simulated by TD-DFT (Fig. 5B) was identical to the experimentally obtained spectrum of the earlier-eluted fraction (green line in Fig. 5A).
Conclusions
In summary, we developed a novel mechanochemical dimerization of 2-(arylethynyl)naphthalenes and a synthetic method for dihydrodibenzopentalene analogs. By directly mixing lithium wires, diarylacetylenes, and a stoichiometric THF additive in air using a ball mill, diarylated dihydrodibenzopentalenes formed quickly, even in air. To clarify the potential of syn-2a as a novel scaffold for π-conjugated ladder molecules with face chirality and fixed planarity, we determined the structural features of syn-2a by XRD analysis, photophysical properties by UV-vis absorption/emission spectroscopy, electronic properties by DFT calculations, and chiroptical properties by CD measurements. Further studies using established methods and synthesized molecules are ongoing in our laboratory.Author contributions
Y. T. preliminarily discovered the reaction, and K. M. H. mainly developed the reaction, synthesized, analyzed, and characterized all compounds and performed theoretical calculations with partial support from Y. T., T. N. and Y. O. T. M. conducted XRD analysis. H. I. directed the project, designed the target compounds, supervised the experiments, and supported the data analyses. The draft manuscript was written by Y. T. and H. I., and all authors finalized the manuscript through proofreading. All authors approved the final version of the manuscript. K. M. H. and Y. T. equally contributed to this paper.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2495836 (syn-2a) contains the supplementary crystallographic data for this paper21.Experimental and characterization data, including crystallo-graphic data, photophysical measurements, and NMR spectra, as well as computational investigations have been included as part of the electronic supplementary information (SI). Supplementary information: syntheses, NMR, UV-vis absorption, emission, CD spectra, DFT calculations, and crystallographic table. See DOI: https://doi.org/10.1039/d5mr00145e.
Acknowledgements
This study was supported by JSPS KAKENHI (JP25K01764 and JP25H01408 to H. I.), Sumitomo Foundation (2300884 to H. I.), Kondo Memorial Foundation (2022-03 to H. I.), NAGAI Foundation of Science & Technology (to H. I.), and Mazda Foundation (to H. I.). Y. T. thanks the Interdisciplinary Frontier Next-Generation Researcher Program of the Tokai Higher Education and Research System for the fellowship. We also thank Mr Takato Mori (Nagoya University) for assistance with XRD analysis. Computations were performed at the Research Center for Computational Science, Okazaki, Japan (Project No. 24-IMS-C059, and 25-IMS-C061).Notes and references
- K. Brand, Ber. Dtsch. Chem. Ges., 1912, 45, 3071 Search PubMed.
- (a) T. Kawase and J. I. Nishida, Chem. Rec., 2015, 15, 1045 CrossRef PubMed; (b) M. Rabinovitz, I. Willner and A. Minsky, Acc. Chem. Res., 1983, 16, 298 CrossRef CAS.
- (a) M. Saito, Symmetry, 2010, 2, 950 Search PubMed; (b) P. J. Mayer, B. B. Botlik, E. Meiszter, K. Németh, C. J. Schürmann, T. Holczbauer and G. London, Comput. Theor. Chem., 2023, 1224, 114113 Search PubMed; (c) M. Ballester, J. Castaner, J. Riera and O. Armet, J. Org. Chem., 1986, 51, 1100 CrossRef CAS; (d) H. Zhang, T. Karasawa, H. Yamada, A. Wakamiya and S. Yamaguchi, Org. Lett., 2009, 11, 3076 CrossRef CAS PubMed; (e) Z. U. Levi and T. D. Tilley, J. Am. Chem. Soc., 2009, 131, 2796 CrossRef CAS PubMed; (f) T. Kawase, A. Konishi, Y. Hirao, K. Matsumoto, H. Kurata and T. Kubo, Chem. – Eur. J., 2009, 15, 2653 CrossRef CAS PubMed; (g) F. Xu, L. Peng, A. Orita and J. Otera, Org. Lett., 2012, 14, 3970 CrossRef CAS PubMed; (h) L. J. Jeffrey and R. Sarpong, Tetrahedron Lett., 2009, 50, 1969 CrossRef PubMed; (i) A. S. K. Hashmi, M. Wieteck, I. Braun, P. Nösel, L. Jongbloed, M. Rudolph and F. Rominger, Adv. Synth. Catal., 2012, 354, 555 Search PubMed; (j) T. Maekawa, Y. Segawa and K. Itami, Chem. Sci., 2013, 4, 2369 RSC.
- W. Liu, J. Zhang, Z. Zhou, D. Zhang, Y. Zhang, S. Xu and X. Zhu, Adv. Mater., 2018, 30, 1800403 CrossRef PubMed.
- S. Suhee, Y. Jin, S. H. Kim, J. Moon, K. Kim, J. Y. Kim, S. H. Park, K. Lee and H. Suh, Macromolecules, 2008, 41, 7296 CrossRef.
- (a) M. Tasić, A. Ruiz-Soriano and D. Strand, J. Org. Chem., 2022, 87, 7501 CrossRef PubMed; (b) D. Hellwinkel, H.-J. Hasselbach and F. Lämmerzahl, Angew Chem. Int. Ed. Engl., 1984, 23, 705 Search PubMed.
- X. Zhu, C. Mitsui, H. Tsuji and E. Nakamura, J. Am. Chem. Soc., 2009, 131, 13596 Search PubMed.
- C. T. Blood and R. P. Linstead, J. Chem. Soc., 1952, 2263 RSC.
- (a) P. Mayorga Burrezo, N.-T. Lin, K. Nakabayashi, S.-I. Ohkoshi, E. M. Calzado, P. G. Boj, M. A. Díaz García, C. Franco, C. Rovira, J. Veciana, M. Moos, C. Lambert, J. T. López Navarrete, H. Tsuji, E. Nakamura and J. Casado, Angew. Chem., Int. Ed., 2017, 56, 2898 CrossRef PubMed; (b) X. Zhu, H. Tsuji, A. Yella, A.-S. Chauvin, M. Grätzel and E. Nakamura, Chem. Commun., 2013, 49, 582 Search PubMed; (c) X. Zhu, H. Tsuji, J. T. López Navarrete, J. Casado and E. Nakamura, J. Am. Chem. Soc., 2012, 134, 19254 CrossRef CAS PubMed; (d) H. Tsuji, A. Ichimura, M. Kudo, J. Sukegawa and E. Nakamura, Chem.–Asian J., 2019, 14, 1672 Search PubMed; (e) Y. Zhang, H. Tsuji, S. F. Martin, S. Ogoshi, T. Yoshikawa and S. Kawaguchi, Chem. Lett., 2014, 43, 699 CrossRef; (f) J. Sukegawa, C. Schubert, X. Zhu, H. Tsuji, D. M. Guldi and E. Nakamura, Nat. Chem., 2014, 6, 899 Search PubMed; (g) C. Ouyang, K. Hashimoto, H. Tsuji, E. Nakamura and Y. Majima, ACS Omega, 2018, 3, 5125 Search PubMed; (h) H. Tsuji and E. Nakamura, Acc. Chem. Res., 2019, 52, 2939 CrossRef CAS PubMed; (i) P. Mayorga Burrezo, X. Zhu, S.-F. Zhu, Q. Yan, J. T. López Navarrete, H. Tsuji, E. Nakamura and J. Casado, J. Am. Chem. Soc., 2015, 137, 3834 CrossRef PubMed.
- (a) M. Saito, M. Nakamura and T. Tajima, Chem. – Eur. J., 2008, 14, 6062 Search PubMed; (b) M. Saito, M. Nakamura and T. Tajima, Angew. Chem., Int. Ed., 2007, 46, 1504 Search PubMed; (c) T. Kuwabara, K. Ishimura, T. Sasamori, N. Tokitoh and M. Saito, Chem. – Eur. J., 2014, 20, 7571 CrossRef CAS PubMed.
- (a) A. Sato and T. Kuwabara, J. Org. Chem., 2025, 90, 9258 Search PubMed; (b) L. I. Smith and H. H. Hoehn, J. Am. Chem. Soc., 1941, 63, 1184 CrossRef CAS.
- (a) H. Li, B. Wei, L. Xu, W. X. Zhang and Z. Xi, Angew. Chem., Int. Ed., 2013, 52, 10822 Search PubMed; (b) Z. Xi, Acc. Chem. Res., 2010, 43, 1342 CrossRef CAS PubMed; (c) Q. Luo, C. Wang, L. Gu, W.-X. Zhang and Z. Xi, Chem.–Asian J., 2010, 5, 1120 CrossRef CAS PubMed; (d) K. Münster, S. Kudo, T. Kuwabara, E. Shimamura, S. Furukawa, Y. Yoshida, S. Ishida, T. Iwamoto, K. Tanifuji, Y. Ohki, M. Minoura and M. Saito, Dalton Trans., 2025, 54, 4030 RSC.
- (a) K. Fujishiro, Y. Morinaka, Y. Ono, T. Tanaka, L. T. Scott, H. Ito and K. Itami, J. Am. Chem. Soc., 2023, 145, 8163 CrossRef CAS PubMed; (b) Y. Toyama, A. Yagi, K. Itami and H. Ito, Nat. Commun., 2025, 16, 5044 CrossRef CAS PubMed.
- (a) Y. Gao, K. Kubota and H. Ito, Angew. Chem., Int. Ed., 2023, 62, e202217723 CrossRef CAS PubMed; (b) K. Kondo, K. Kubota and H. Ito, Nat. Synth., 2025, 4, 744 CrossRef CAS.
- Recent reviews on mechanochemistry and mechanochemical transformations: (a) S. L. James, C. J. Adams, C. Bolm, D. Braga, P. Collier, T. Friščić, F. Grepioni, K. D. M. Harris, G. Hyett, W. Jones, A. Krebs, J. Mack, L. Maini, A. G. Orpen, I. P. Parkin, W. C. Shearouse, J. W. Steed and D. C. Waddell, Chem. Soc. Rev., 2012, 41, 413 RSC; (b) G.-W. Wang, Chem. Soc. Rev., 2013, 42, 7668 RSC; (c) J.-L. Do and T. Friščić, ACS Cent. Sci., 2017, 3, 13 CrossRef CAS PubMed; (d) J. G. Hernández and C. Bolm, J. Org. Chem., 2017, 82, 4007 CrossRef PubMed; (e) J. L. Howard, Q. Cao and D. L. Browne, Chem. Sci., 2018, 9, 3080 Search PubMed; (f) J. Andersen and J. Mack, Green Chem., 2018, 20, 1435 RSC; (g) D. Tan and T. Friščić, Eur. J. Org Chem., 2018, 2018, 18 CrossRef CAS; (h) D. Tan and F. García, Chem. Soc. Rev., 2019, 48, 2274 RSC; (i) C. Bolm and J. G. Hernández, Angew. Chem., Int. Ed., 2019, 58, 3285 CrossRef CAS PubMed; (j) T. Friščić, C. Mottillo and H. M. Titi, Angew. Chem., Int. Ed., 2020, 59, 1018 CrossRef PubMed; (k) K. Kubota and H. Ito, Trends Chem., 2020, 2, 1066 CrossRef CAS; (l) K. J. Ardila-Fierro and J. G. Hernández, ChemSusChem, 2021, 14, 2145 CrossRef CAS PubMed; (m) R. R. A. Bolt, J. A. Leitch, A. C. Jones, W. I. Nicholson and D. L. Browne, Chem. Soc. Rev., 2022, 51, 4243 RSC; (n) F. Cuccu, L. De Luca, F. Delogu, E. Colacino, N. Solin, R. Mocci and A. Porcheddu, ChemSusChem, 2022, 15, e202200362 CrossRef CAS PubMed; (o) M. T. J. Williams, L. C. Morrill and D. L. Browne, ChemSusChem, 2022, 15, e202102157 CrossRef CAS PubMed; (p) S. Hwang, S. Grätz and L. Borchardt, Chem. Commun., 2022, 58, 1661 RSC.
- Recent examples of mechanochemical transformation: (a) K. Kubota, Y. Pang, A. Miura and H. Ito, Science, 2019, 366, 1500 CrossRef CAS PubMed; (b) T. Seo, K. Kubota and H. Ito, J. Am. Chem. Soc., 2020, 142, 9884 CrossRef CAS PubMed; (c) T. Seo, N. Toyoshima, K. Kubota and H. Ito, J. Am. Chem. Soc., 2021, 143, 6165 Search PubMed; (d) R. Takahashi, A. Hu, P. Gao, Y. Gao, Y. Pang, T. Seo, S. Maeda, J. Jiang, H. Takaya, K. Kubota and H. Ito, Nat. Commun., 2021, 12, 6691 CrossRef CAS PubMed; (e) T. Seo, K. Kubota and H. Ito, J. Am. Chem. Soc., 2023, 145, 6823 CrossRef CAS PubMed; (f) K. Kondo, K. Kubota and H. Ito, Nat. Synth., 2025, 4, 744 CrossRef CAS; (g) Y. Toyama, T. Nakamura, Y. Horikawa, Y. Morinaka, Y. Ono, A. Yagi, K. Itami and H. Ito, Chem. Sci., 2025, 16, 9117 RSC; (h) Q. Cao, J. L. Howard, E. Wheatley and D. L. Browne, Angew. Chem., Int. Ed., 2018, 57, 11339 CrossRef CAS PubMed; (i) M. Mayer, M. Wohlgemuth, A. Salomé Straub, S. Grätz and L. Borchardt, Angew. Chem., Int. Ed., 2025, 64, e202424139 CrossRef CAS PubMed; (j) A. W. J. Bowles, J. A. Quirk, Y. Liu, G. H. Morritt, M. Freitag, G. F. S. Whitehead, A. W. Woodward, A. Brookfield, C. A. P. Goodwin, D. Collison, F. Tuna, C. L. McMullin, J. A. Dawson, E. Lu and F. Ortu, J. Am. Chem. Soc., 2024, 146, 28914 CrossRef CAS PubMed; (k) N. Davison, P. G. Waddell and E. Lu, J. Am. Chem. Soc., 2023, 145, 17007 CrossRef CAS PubMed.
- (a) T. Friščić, A. V. Trask, W. Jones and W. D. S. Motherwell, Angew. Chem., Int. Ed., 2006, 45, 7546 CrossRef PubMed; (b) T. Friščić, L. Fábián and W. Jones, Cryst. Growth Des., 2007, 7, 2417 Search PubMed; (c) T. Friščić, S. L. Childs, S. A. A. Rizvi and W. Jones, CrystEngComm, 2009, 11, 418 RSC; (d) T. Friščić, C. Mottillo and H. M. Titi, Angew. Chem., Int. Ed., 2020, 59, 1018 CrossRef PubMed.
- M. T. Quick, M. Quick, I. N. Ioffe, C. Richter, R. Mahrwald, S. Druzhinin and S. A. Kovalenko, J. Phys. Chem. B, 2020, 124, 1049 CrossRef CAS PubMed.
- (a) L. Vugrin, C. Chatzigiannis, E. Colacino and I. Halasz, RSC Mechanochem., 2025, 2, 482 RSC; (b) K. Kubota, R. Hisazumi, T. Seo and H. Ito, RSC Mechanochem., 2024, 1, 250 RSC.
- T. Kawase, T. Fujiwara, C. Kitamura, A. Konishi, Y. Hirao, K. Matsumoto, H. Kurata, T. Kubo, S. Shinamura, H. Mori, E. Miyazaki and K. Takimiya, Angew. Chem., Int. Ed., 2010, 49, 7728 CrossRef CAS PubMed.
- CCDC 2495836: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2ps3vg.
Footnote |
| † Equal contribution. |
| This journal is © The Royal Society of Chemistry 2026 |