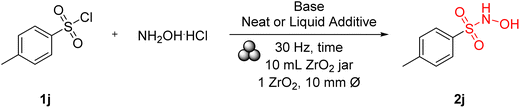Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Reimagining sulfohydroxamic acid formation through mechanochemical solid-state activation
Muhammad Irfan†
,
Pietro Caboni ,
Andrea Porcheddu
,
Andrea Porcheddu * and
Francesco Basoccu†
* and
Francesco Basoccu†
Dipartimento di Scienze Chimiche e Geologiche, Università Degli Studi di Cagliari, Cittadella Universitaria, SS554 bivio per Sestu, 09042-Monserrato, CA, Italy. E-mail: porcheddu@unica.it
First published on 1st April 2026
Abstract
Sulfohydroxamic acids (–SO2–NHOH), including classical Piloty's acid derivatives, are key NO/HNO donors and valuable building blocks, but their synthesis still relies on hazardous hydroxylamine solutions and solvent-intensive protocols. Here, we report a simple, solvent-free mechanochemical route based on milling sulfonyl chlorides with hydroxylamine hydrochloride in the presence of an inorganic base. Magnesium oxide (MgO) emerges as an ideal promoter, combining sufficient basicity with low nucleophilicity to release hydroxylamine in situ while preserving both the sulfonyl chloride and the sulfohydroxamic product. Under optimised conditions, a broad range of aromatic sulfonyl chlorides bearing electron-donating and electron-withdrawing substituents are converted into the corresponding sulfohydroxamic acids. The method efficiently produces sulfohydroxamic acids with yields ranging from 18% to 81% under mild conditions and is adaptable to standard milling equipment, enabling scalable synthesis. The protocol can be readily extended to O- and N,O-substituted hydroxylamine derivatives by using appropriately functionalized hydroxylamine salts. This mechanochemical strategy drastically reduces solvent consumption and streamlines work-up, providing a safer, cleaner, and scalable approach to sulfohydroxamic acids and their NO-donor analogues.
Introduction
Sulfohydroxamic acids constitute a unique and enduring class of compounds that have achieved significant status at the confluence of synthetic, medicinal, and biological chemistry.1–6 Their structural motif, defined by the N–O–S linkage, imparts a remarkable combination of stability and reactivity, facilitating their participation in diverse chemical transformations and controlled decomposition into bioactive nitrogen species.6–10The prototypical member of this family, benzenesulfohydroxamic acid, commonly known as Piloty's acid, was first reported by Oskar Piloty in the late 19th century.11 His groundbreaking research not only introduced a novel functional group into the chemist's repertoire but also provided a straightforward synthetic framework that remains the primary method for producing these derivatives over a century later. Piloty's initial synthesis, which involved reacting a sulfonyl chloride dissolved in methanol with an aqueous solution of hydroxylamine, was sophisticated and effective. This transformation elucidates the fundamental reactivity of sulfonyl electrophiles with nitrogen nucleophiles, culminating in a stable N–O–S linkage amenable to further manipulation under mild conditions. The simplicity and broad applicability of this method have facilitated its swift adoption and adaptation, catalysing extensive research into the reactivity and coordination behavior of sulfohydroxamic acids and their derivatives (Fig. 1).12–16 Over the subsequent decades, this class of compounds has become integral to both synthetic methodology and biological chemistry, owing to their ability to serve as donors of reactive nitrogen species, such as nitric oxide (NO) and nitroxyl (HNO).13,17–19
The biological significance of these two species is profound. Nitric oxide, a small, diffusible free radical, is integral to numerous physiological processes, including vasodilation, neurotransmission, and immune modulation.20 Consequently, the controlled release of NO from chemical precursors, such as sulfohydroxamic acids, has attracted sustained interest from medicinal chemists seeking reliable and tunable NO donors for therapeutic and diagnostic applications. In contrast, the one-electron-reduced form of NO, HNO, demonstrates a distinct and complementary reactivity profile, particularly in redox and metalloprotein chemistry.20–22
Sulfohydroxamic acids capable of generating either NO or HNO under mild conditions have thus emerged as potent chemical tools for investigating biological redox processes and as versatile intermediates for the synthesis of nitrogen-containing compounds.18
Despite its enduring significance, the synthesis of sulfohydroxamic acids has undergone minimal evolution since Piloty's era.8,13,23,24 The traditional solution-phase conversion of sulfonyl chlorides with hydroxylamine remains the predominant methodology; however, it suffers from several inherent limitations. The high reactivity and limited stability of both reagents and products frequently result in competing side reactions, leading to reduced yields and complex purification processes.25–27 Furthermore, hydroxylamine presents significant safety concerns owing to its volatility, toxicity, and propensity for explosive decomposition, particularly in concentrated or heated solutions.28–32 These hazards are amplified when reactions are scaled up, posing challenges to industrial applications.32 Even contemporary modifications of the original method, such as those utilizing mixed solvent systems (e.g., MeOH–H2O–THF) and solid bases like magnesium oxide, have not completely overcome these challenges. They continue to rely on substantial volumes of organic solvents, generate considerable waste, and necessitate the careful handling of hazardous reagents, all of which contravene the principles of green and sustainable chemistry.33–35
In response to these challenges, mechanochemistry has emerged as a highly promising alternative platform for the synthesis of sulfohydroxamic acids. Mechanochemistry is characterized by the utilization of mechanical energy, typically through grinding, milling, or shearing, to drive chemical reactions, signifying a genuine shift in synthetic philosophy.36–41 Unlike conventional solution-based approaches, mechanochemical methods operate under solvent-free or solvent-minimized conditions, significantly reducing chemical waste generation and enhancing the overall process safety.42–49 Beyond these ecological advantages, mechanochemical reactions often exhibit accelerated reaction rates, improved selectivity, and distinct mechanistic behaviours compared to their solution-phase counterparts. In numerous instances, novel products or reactivity patterns emerge that are otherwise inaccessible in traditional media, highlighting the fundamentally unique solid-state environment.8,50–56
Mechanochemistry has already demonstrated a transformative impact in diverse fields, including organic synthesis, coordination chemistry, and materials science.57–63 Its advantages, such as minimal solvent use, simplified work-up, and often superior atom economy, render it particularly attractive for the preparation of sensitive or unstable compounds. These features are especially valuable in the context of sulfohydroxamic acids due to the inherent instability of both hydroxylamine and sulfohydroxamic intermediates under aqueous or alcoholic conditions, which suggests that a solvent-free or solid-assisted approach could significantly enhance both safety and efficiency. Furthermore, the mechanical activation of sulfonyl chlorides in the presence of solid hydroxylamine salts offers the prospect of controlled and selective conversion under mild conditions, eliminating the need for hazardous liquid reagents and cumbersome solvent systems.
Building on our previous work on the synthesis of hydroxamic acids, we sought to extend this methodology to the preparation of the corresponding sulfonic derivatives by identifying an alternative to carbonyl diimidazole (CDI).36 The use of CDI proved unsuccessful, as the higher acidity of sulfonic acids, together with the intrinsic instability of the sulfonyl intermediate, resulted in no productive activation and led to the exclusive recovery of the starting material. Therefore, we opted for the direct milling of sulfonyl chlorides with hydroxylamine hydrochloride in the presence of a benign inorganic base, such as magnesium oxide, which enables the clean and selective formation of sulfohydroxamic acids at room temperature. Importantly, this method not only enhances laboratory safety and practicality but also exemplifies how mechanochemical activation can fundamentally reshape established reactivities. By bridging Piloty's pioneering work on sulfohydroxamic acids with the principles of contemporary green chemistry, the present methodology redefines a century-old reaction through the lens of safety and environmental stewardship. The results underscore the capacity of solid-state reactivity to deliver practical, scalable, and inherently safer synthetic routes to valuable NO-donor compounds, thereby advancing both synthetic methodology and medicinal chemistry applications.
Results and discussion
The initial approach for synthesizing sulfohydroxamic acids involved using equimolar quantities of p-tosyl chloride (1j) and hydroxylamine hydrochloride as the hydroxylamine source. Various bases were assessed under neat conditions; however, the reactivity was notably limited when only one equivalent of the base was employed (Table 1, entries 1–5). To overcome this limitation, a liquid component was incorporated into the reaction mixture. This adjustment resulted in a significant enhancement in the formation of target sulfohydroxamic acid 2j (Table 1, entries 6–7), although an undefined impurity was also detected under these conditions. We postulated that this byproduct may arise from secondary transformations of nascent sulfohydroxamic acid, indicative of its inherent reactivity.64| Entry | p-Tosyl chloride (eq.) | NH2OH·HCl (eq.) | Base | Base (eq.) | LAG | Time (h) | 2j (%) |
|---|---|---|---|---|---|---|---|
| a The reactions were performed on a 1.0 mmol scale using a Retsch MM500 ball milling machine. The conversion rates were calculated using 1H-NMR analysis. The amount of liquid additive had an η of 0.3 µL mg−1, which represents the ratio of the liquid volume to the weight of the reaction mixture. | |||||||
| 1 | 1 | 1 | NaHCO3 | 1 | — | 2 | 14% |
| 2 | 1 | 1 | Na2CO3 | 1 | — | 2 | 27% |
| 3 | 1 | 1 | NaOH | 1 | — | 2 | 11% |
| 4 | 1 | 1 | Na3PO4 | 1 | — | 2 | 29% |
| 5 | 1 | 1 | MgO | 1 | — | 2 | 25% |
| 6 | 1 | 1 | Na2CO3 | 1 | MeOH | 2 | 40% |
| 7 | 1 | 1 | Na3PO4 | 1 | H2O | 2 | 27% |
| 8 | 1 | 2 | Na2CO3 | 2 | MeOH | 2 | 51% |
| 9 | 1 | 2 | MgO | 2 | — | 2 | 49% |
| 10 | 1 | 2 | MgO | 4 | — | 2 | 65% |
| 11 | 1 | 3 | Mg(OH)2 | 3 | — | 2 | 66% |
| 12 | 1 | 3 | MgO | 3 | — | 2 | 68% |
| 13 | 1 | 3 | Li2CO3 | 3 | iPrOH | 3 | 63% |
| 14 | 1 | 3 | MgO | 3 | — | 3 | 66% |
To enhance process efficiency, the methodology was refined by increasing the equivalents of both hydroxylamine hydrochloride and the base. This modification led to a significant improvement in the conversion to 2j (Table 1, entries 8 and 9). Notably, doubling the amount of the base further increased the reactivity, achieving a conversion rate of 65% (Table 1, entry 10). However, subsequent efforts to optimize the outcome, such as using three equivalents of hydroxylamine and base along with extended reaction times of up to 3 h, did not yield additional improvements in conversion (Table 1, entries 11–14). Collectively, these findings suggest that the most efficient conditions are attained with two equivalents of hydroxylamine hydrochloride and four equivalents of MgO, balancing reactivity and process simplicity. These optimized parameters were adopted as standard conditions for subsequent investigations.
With the optimized conditions established, we proceeded to evaluate the generality and robustness of the protocol by extending its application to a wide array of sulfonyl chlorides, thereby facilitating the synthesis of a diverse library of sulfohydroxamic acids (Scheme 1). The methodology demonstrated considerable versatility, accommodating a broad range of substituents with notable efficiencies. Substrates containing halogen substituents exhibited consistently good reactivity, allowing these moieties to be to potentially used as valuable handles for subsequent synthetic elaboration. Conversely, the incorporation of either electron-withdrawing groups or electron-donating groups adversely affected the reaction outcome, indicating that, instead of specific electronic balance, other factors such as the rheology of the mixture might negatively affect the protocol. Notably, substrates containing an additional aromatic ring were particularly suitable for the transformation, as the extended conjugation appeared to facilitate the reaction pathway, resulting in the production of the corresponding sulfohydroxamic acids in commendable yields. In addition to synthesizing primary sulfohydroxamic acids, we further broadened the method's applicability to encompass the synthesis of structurally diverse derivatives featuring alkyl substituents on either the oxygen or nitrogen atoms. This was accomplished by substituting hydroxylamine hydrochloride with appropriately functionalized hydroxylamine derivatives, pre-installed with the desired substituents on the hydroxylamine scaffold (Scheme 1).
This strategic modification of the original protocol facilitated the efficient preparation of a diverse range of O- and N, O-substituted sulfohydroxamic acids, exemplified by O-benzylhydroxylamine hydrochloride and N, O-dimethyl-hydroxylamine hydrochloride. Notably, the overall efficiency of the process was markedly enhanced, which was attributed to both the increased stability of the resulting scaffolds and the absence of collateral pathways that affect the formation of the final product. The ability to access a diverse range of sulfohydroxamic acid derivatives not only illustrates the versatility of the methodology but also highlights its potential utility in the synthesis of tailored molecules relevant to both synthetic and medicinal chemistry applications. For completeness, the synthesis of 2j was also performed on a gram scale, resulting in the final 60% conversion of the starting material to the corresponding sulfohydroxamic acid.
To examine the reaction mechanism, it is essential to analyse the characteristics of this process under mechanochemical conditions. The transformation occurs through two sequential steps: an acid–base reaction between hydroxylamine hydrochloride and the chosen base, followed by the nucleophilic attack of the liberated hydroxylamine on the sulfonyl chloride (Scheme 2).
 | ||
| Scheme 2 General reaction mechanism concerning the release of gaseous hydroxylamine and its reaction with sulfonyl chloride present in the reaction environment. | ||
The selection of the base and the potential inclusion of a liquid additive significantly impact the transformation outcome.65 Sulfonyl chlorides and the resultant sulfohydroxamic acids are inherently sensitive to nucleophilic species and strongly basic environments. Consequently, inappropriate conditions can readily compromise the stability of both the starting material and product, leading to degradation pathways or competing side reactions. Therefore, the selected base (and, if applicable, the liquid additive) must meet a crucial dual criterion: it must possess sufficient basicity to effectively deprotonate hydroxylamine hydrochloride, thereby enabling the in situ release of free hydroxylamine within the milling jar to initiate the reaction. To exclude the formation of gaseous hydroxylamine or volatile by-products, the mass loss during milling of hydroxylamine hydrochloride and magnesium oxide was analysed (see the SI for details). Under the optimized conditions, no significant mass loss was detected, indicating that free hydroxylamine predominantly operates without developing any gaseous species. Although the reaction mixture remained macroscopically solid at the end of milling, a transient liquid phase arising from the low melting point of the free base (33 °C) cannot be completely ruled out. Concurrently, it must not exhibit excessive reactivity to avoid undesirable interactions with sulfonyl chloride or alterations to the newly formed sulfohydroxamic acid.
Among all the bases examined, magnesium oxide (MgO) emerged as the most reliable promoter, uniquely combining adequate Brønsted basicity with a mild nucleophilic profile. Under neat solvent-free milling conditions, MgO enables the controlled in situ liberation of hydroxylamine from its hydrochloride salt, while simultaneously maintaining a reaction environment that preserves as much as possible both the integrity of the sulfonyl chloride and the stability of the newly formed sulfohydroxamic acid (Fig. 2, a further analysis of the stability of sulfonyl chlorides can be found in the SI). Beyond its fundamental chemical role, the solid-state characteristics of MgO, including its high surface area, heterogeneous distribution of basic sites, and mechanical robustness, are ideally suited for mechanochemical activation.66,67
Notably, alternative bases either failed to initiate the reaction or induced undesirable side processes, underscoring the unique dual functionality of MgO as both a mild base and a structural moderator of the milling medium.
Lastly, the green metrics of the mechanochemical methodology were calculated, revealing an improvement in process mass intensity (PMI) of more than 20% (see the SI for additional details). Furthermore, the elimination of toxic volatile organic compounds (VOCs), such as THF, further enhances the overall safety of the process.
Conclusions
This study establishes mechanochemistry as a robust, user-friendly, and inherently safe method for synthesizing sulfohydroxamic acids. The straightforward, solvent-free milling of sulfonyl chlorides with hydroxylamine hydrochloride in the presence of a benign inorganic base, such as MgO, facilitates the production of target compounds under mild conditions. This approach avoids hazardous, waste-intensive organic solvents and simplifies the workup.In addition to operational simplicity, mechanochemical activation directly addresses the persistent challenges in this area of chemistry. This reduces the handling risks associated with volatile hydroxylamines and mitigates the instability of sensitive substrates and products, which are often problematic under solution conditions. Consequently, this method enhances practical access to sulfohydroxamic scaffolds, which are significant in both synthetic and medicinal chemistry applications.
Overall, these findings position mechanochemistry as a versatile and sustainable platform for the preparation of sulfohydroxamic acids, aligning with green chemistry principles by combining safety, efficiency, and environmental sustainability. We anticipate that this approach will be readily applicable to a diverse range of sulfonyl chloride frameworks and related N-functionalization reactions. Furthermore, scaling up using contemporary enabling technologies, such as RAM or extrusion, is expected to facilitate its adoption in research and development.
Author contributions
Conceptualisation: A. P.; investigation: M. I. and F. B.; data curation: M. I.; supervision: A. P. and P. C.; writing – original draft: M. I. and A. P.; writing – review and editing: all authors. F. B. and M. I. have contributed equally to this work.Conflicts of interest
The authors declare that they have no conflicts of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). All experimental data relevant to the manuscript's preparation and formulation of theories and/or hypotheses, including conclusions, etc. are provided in the SI. Supplementary information is available. See DOI: https://doi.org/10.1039/d5mr00141b.Acknowledgements
The authors acknowledge financial support under the National Recovery and Resilience Plan (NRRP), Mission 4, Component 2, Investment 1.1, Call for Tender No. 1409 published on 14. 9. 2022 by the Italian Ministry of University and Research (MUR), funded by the European Union – NextGenerationEU-Project Title: The Second Life of Posidonia Oceanica: Sustainability-guided Isolation and Valorisation of its Main Constituents – CUP J53D23014680001-Grant Assignment Decree No. 1064 adopted on 18. 07. 2023 by the Italian Ministry of Ministry of University and Research (MUR). The financial support of the European Union NextGenerationEU under the National Recovery and Resilience Plan (NRRP) of Ministero dell'Università e della Ricerca (MUR) (Project code PE0000021, CUP no. F53C22000770007, Network 4 Energy Sustainable Transition, NEST) is acknowledged. Fondazione Banco di Sardegna (FDS) also supported the project financially, with grant number F73C23001600007.Notes and references
- A. Y. Jeng, M. Chou and D. T. Parker, Bioorg. Med. Chem. Lett., 1998, 8, 897–902 CrossRef CAS PubMed.
- Z. Huang, C. Velázquez, K. Abdellatif, M. Chowdhury, S. Jain, J. Reisz, J. DuMond, S. B. King and E. Knaus, Org. Biomol. Chem., 2010, 8, 4124–4130 RSC.
- F. N. Shirota, E. G. DeMaster, M. J. C. Lee and H. T. Nagasawa, Nitric Oxide, 1999, 3, 445–453 CrossRef CAS PubMed.
- Z. Huang, C. A. Velázquez, K. R. A. Abdellatif, M. A. Chowdhury, J. A. Reisz, J. F. DuMond, S. B. King and E. E. Knaus, J. Med. Chem., 2011, 54, 1356–1364 CrossRef CAS PubMed.
- Y. Long, Z. Xia, A. M. Rice and S. B. King, Molecules, 2022, 27, 5305 CrossRef CAS PubMed.
- M. N. Hughes and R. Cammack, in Methods in Enzymology, Academic Press, 1999, vol. 301, pp. 279–287 Search PubMed.
- M. A. Alam, Curr. Org. Chem., 2019, 23, 978–993 CrossRef CAS PubMed.
- A. Porcheddu, L. De Luca and G. Giacomelli, Synlett, 2009, 2009, 2149–2153 CrossRef.
- R. Smulik-Izydorczyk, J. Pięta, R. Michalski, M. Rola, K. Kramkowski, A. Artelska, J. Zielonka and A. B. Sikora, Redox Biochem. Chem., 2024, 8, 100031 CrossRef CAS.
- A. Citarella, D. Moi, L. Pinzi, D. Bonanni and G. Rastelli, ACS Omega, 2021, 6, 21843–21849 CrossRef CAS PubMed.
- O. Piloty, Ber. Dtsch. Chem. Ges., 1896, 29, 1559–1567 CrossRef CAS.
- P. C. Wilkins, H. K. Jacobs, M. D. Johnson and A. S. Gopalan, Inorg. Chem., 2004, 43, 7877–7881 CrossRef CAS PubMed.
- K. Sirsalmath, S. A. Suárez, D. E. Bikiel and F. Doctorovich, J. Inorg. Biochem., 2013, 118, 134–139 CrossRef CAS PubMed.
- K. Aizawa, H. Nakagawa, T. Suzuki and N. Miyata, Free Radic. Biol. Med., 2012, 53, S141 CrossRef.
- J. Polaczek, H. Subedi, Ł. Orzeł, L. S. Lisboa, R. B. Cink, G. y. Stochel, N. E. Brasch and R. Van, Adv. Inorg. Chem., 2021, 60, 2964–2975 CrossRef CAS PubMed.
- A. M. Confer, A. C. Vilbert, A. Dey, K. M. Lancaster and D. P. Goldberg, J. Am. Chem. Soc., 2019, 141, 7046–7055 CrossRef CAS PubMed.
- S. Mondal and S. Malakar, Tetrahedron, 2020, 76, 131662 CrossRef CAS.
- X.-R. Huang, Y.-M. Zhang, T.-B. Wan, P. Zhang, X.-X. Zhang, F.-M. Wang, D. Xu, M.-H. Shen and H.-D. Xu, Tetrahedron, 2019, 75, 130336 CrossRef.
- R. Zamora, A. Grzesiok, H. Weber and M. Feelisch, Biochem. J., 1995, 312, 333–339 CrossRef CAS PubMed.
- D. Sanna, G. Rocchitta, M. Serra, M. Abbondio, P. A. Serra, R. Migheli, L. De Luca, E. Garribba and A. Porcheddu, Pharmaceuticals, 2017, 10, 74 CrossRef PubMed.
- H. Subedi and N. E. Brasch, Dalton Trans., 2016, 45, 352–360 RSC.
- J. F. DuMond and S. B. King, Antioxid. Redox Signaling, 2011, 14, 1637–1648 CrossRef CAS PubMed.
- T. Nakano, N. Kousaka, A. Nakayama, Y. Kato, K. Takashima, G. Tanabe and M. Yoshimatsu, Org. Lett., 2024, 26, 1753–1757 CrossRef CAS PubMed.
- F. J. Geffers, F. R. Kurth, P. G. Jones and D. B. Werz, Chem.–Eur. J., 2021, 27, 14846–14850 CrossRef CAS PubMed.
- M. Medeiros, E. H. Wanderlind, J. R. Mora, R. Moreira, A. J. Kirby and F. Nome, Org. Biomol. Chem., 2013, 11, 6272–6284 RSC.
- J. B. Domingos, E. Longhinotti, T. A. S. Brandão, C. A. Bunton, L. S. Santos, M. N. Eberlin and F. Nome, J. Org. Chem., 2004, 69, 6024–6033 CrossRef CAS PubMed.
- Y. Zeng, Y. Zhao, L. Yang and Y. Jiang, ChemistrySelect, 2023, 8, e202303345 CrossRef CAS.
- C. T. A. Evelo, A. A. M. G. Spooren, R. A. G. Bisschops, L. G. M. Baars and J. M. Neis, Blood Cell Mol. Dis., 1998, 24, 280–295 CrossRef CAS PubMed.
- S. C. Gad, in Encyclopedia of Toxicology, ed. P. Wexler, Academic Press, Oxford, 3rd edn, 2014, pp. 982–983 Search PubMed.
- J. M. Desesso and G. C. Goeringer, Toxicol. Ind. Health, 1990, 6, 109–121 CrossRef CAS PubMed.
- L. A. Long, Process Saf. Prog., 2004, 23, 114–120 CrossRef CAS.
- F. N. A. f. t. E. a. E. M. (ADEME), Hydroxylamine Explosion in a Chemical Plant - ARIA Accident Report No. 14952, https://www.aria.developpement-durable.gouv.fr/accident/14952_en/?lang=en.
- I. T. Horvath and P. T. Anastas, Chem. Rev., 2007, 107, 2169–2173 CrossRef CAS PubMed.
- M. Tobiszewski, M. Marć, A. Gałuszka and J. Namieśnik, Molecules, 2015, 20, 10928–10946 CrossRef CAS PubMed.
- S. Trakhtenberg and J. C. Warner, Chem. Rev., 2007, 107, 2174–2182 CrossRef CAS PubMed.
- R. Mocci, L. D. Luca, F. Delogu and A. Porcheddu, Adv. Synth. Catal., 2016, 358, 3135–3144 CrossRef CAS.
- F. Basoccu, L. De Luca and A. Porcheddu, Eur. J. Org Chem., 2024, 27, e202400425 CrossRef CAS.
- N. Fantozzi, J.-N. Volle, A. Porcheddu, D. Virieux, F. García and E. Colacino, Chem. Soc. Rev., 2023, 52, 6680–6714 RSC.
- S. L. James and T. Friščić, Chem. Soc. Rev., 2013, 42, 7494–7496 RSC.
- J.-L. Do and T. Friščić, ACS Cent. Sci., 2017, 3, 13–19 CrossRef CAS PubMed.
- L. H. Joseph, Q. Cao and D. L. Browne, Chem. Sci., 2018, 9, 3080–3094 RSC.
- T. Friščić, C. Mottillo and H. M. Titi, Angew. Chem., 2020, 132, 1030–1041 CrossRef.
- J. Batteas, K. G. Blank, E. Colacino, F. Emmerling, T. Friščić, J. Mack, J. Moore, M. E. Rivas and W. Tysoe, RSC Mechanochem., 2025, 2, 10–19 RSC.
- K. Floyd, L. Gonnet, T. Friščić and J. Batteas, RSC Mechanochem., 2024, 1, 289–295 RSC.
- J. G. Hernández and C. Bolm, J. Org. Chem., 2017, 82, 4007–4019 CrossRef PubMed.
- G. N. Hermann, P. Becker and C. Bolm, Angew. Chem., 2015, 127, 7522–7525 CrossRef.
- A. Gallego, N. Pétry, J. Pinaud, O. Giani and F. Lamaty, ChemistryEurope, 2025, e202500232 Search PubMed.
- M. Perez-Venegas and E. Juaristi, ACS Sustain. Chem. Eng., 2020, 8, 8881–8893 CrossRef CAS.
- S. L. James, C. J. Adams, C. Bolm, D. Braga, P. Collier, T. Friščić, F. Grepioni, K. D. Harris, G. Hyett and W. Jones, Chem. Soc. Rev., 2012, 41, 413–447 RSC.
- T. Nakano, N. Kousaka, A. Nakayama, Y. Kato, K. Takashima, G. Tanabe and M. Yoshimatsu, Org. Lett., 2024, 26, 1753–1757 CrossRef CAS PubMed.
- G. Kirby, G. Prestat and F. Berhal, J. Org. Chem., 2023, 88, 4720–4729 CrossRef CAS PubMed.
- G. Giacomelli, A. Porcheddu and M. Salaris, Org. Lett., 2003, 5, 2715–2717 CrossRef CAS PubMed.
- I. S. Sow, Eur. J. Chem., 2024, 15, 345–354 CrossRef CAS.
- E. Juaristi and C. G. Avila-Ortiz, Synthesis, 2023, 55, 2439–2459 CrossRef CAS.
- F. Basoccu, 2025.
- K. Kubota and H. Ito, Trends Chem., 2020, 2, 1066–1081 CrossRef CAS.
- F. Basoccu, F. Cuccu and A. Porcheddu, ChemSusChem, 2024, 17, e202301034 CrossRef CAS PubMed.
- F. Fischer, K.-J. Wenzel, K. Rademann and F. Emmerling, Phys. Chem. Chem. Phys., 2016, 18, 23320–23325 RSC.
- A. A. Michalchuk and F. Emmerling, Angew. Chem., Int. Ed., 2022, 61, e202117270 CrossRef CAS PubMed.
- P. P. Mazzeo, M. Prencipe, T. Feiler, F. Emmerling and A. Bacchi, Cryst. Growth Des., 2022, 22, 4260–4267 CrossRef CAS PubMed.
- A. M. Constantin, F. Mele, M. Lanzi, G. Maestri, R. Maggi, N. Della Ca’ and L. Capaldo, ChemSusChem, 2025, 2500211 Search PubMed.
- D. E. Crawford, S. L. James and T. McNally, ACS Sustain. Chem. Eng., 2018, 6, 193–201 CrossRef CAS.
- A. Krusenbaum, S. Grätz, G. T. Tigineh, L. Borchardt and J. G. Kim, Chem. Soc. Rev., 2022, 51, 2873–2905 RSC.
- It was not possible to fully elucidate the exact chemical structures of the impurities detected in the 1H NMR spectra of para-methylbenzenesulfohydroxamic acid, even with the use of additional analytical techniques. This area remains underexplored in the literature, as impurities arising from sulfohydroxamic acid synthesis are rarely characterized. Consequently, this aspect of our study remains an ongoing area of investigation.
- F. Basoccu, S. Piermarini, T. Angelini, M. Mari, E. Mariani, A. Falchi and A. Porcheddu, ChemSusChem, 2025, 18, e202500457 CrossRef CAS PubMed.
- C. Sousa, J. Mejias, G. Pacchioni and F. Illas, Chem. Phys. Lett., 1996, 249, 123–129 CrossRef CAS.
- S. Ardizzone, C. Bianchi and B. Vercelli, Appl. Surf. Sci., 1998, 126, 169–175 CrossRef CAS.
Footnote |
| † F. B. and M. I. contributed equally to this study. |
| This journal is © The Royal Society of Chemistry 2026 |