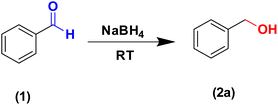Open Access Article
Open Access ArticleSolvent-free continuous flow reduction of aldehydes and ketones via mechanochemistry in a jacketed screw reactor
Mukesh Purohitab,
Jagannath Padhia and
Amol Kulkarni *ab
*ab
aChemical Engineering & Process Development, CSIR-National Chemical Laboratory Pune, 411008, India. E-mail: aa.kulkarni@ncl.res.in; mn.purohit@ncl.res.in
bAcademy of Scientific and Innovative Research (AcSIR), Ghaziabad, 201002, India
First published on 9th April 2026
Abstract
We present a simple and efficient solvent-free method for the continuous flow reduction of aldehydes and ketones using sodium borohydride as a reducing agent at room temperature. By employing a jacketed screw reactor, this process achieves nearly complete conversion with residence times ranging from 2 to 10 minutes. The method has been demonstrated on ten representative aldehydes and ketones, highlighting its broad applicability and sustainability. This approach utilizes readily available starting materials, showcasing the potential of mechanochemistry for green and scalable chemical synthesis.
Introduction
In organic synthesis, reduction is one of the most fundamental reactions. The two most commonly used reducing agents are sodium borohydride (NaBH4) and lithium aluminium hydride (LiAlH4). NaBH4 is widely used in industry due to its selectivity, safety, and versatility. The global sodium borohydride production reached around 826k tons in 2023.1 It efficiently reduces aldehydes and ketones to alcohols under mild conditions, with minimal side reactions. It is also safer and more stable than LiAlH4, making it easy to handle and compatible with protic and aprotic solvents. Industries use NaBH4 in pharmaceutical synthesis,2 paper bleaching,3 dye reduction,4 and hydrogen generation.5 It is cost-effective, aligns with green chemistry principles, and produces non-toxic by-products. Use of NaBH4 along with catalysts can be extended further for enabling reductions of esters and nitriles. These features make it indispensable in pharmaceuticals, chemicals, textiles, and energy applications. Traditionally, flammable solvents such as ethanol, isopropanol (2-propanol), and methanol are used for reduction of aldehydes using NaBH4.6 Use of solvents such as methanol, ethanol, and tetrahydrofuran (THF) is known for reducing aldehydes and ketones using sodium borohydride. However, solvolysis can make aldehydes unstable in methanol or ethanol. Reduction in methanol needs a significant excess of NaBH4.In a recent study, Ishoni et al. have utilized a twin-screw reactor to reduce aldehydes in the presence of aqueous caustic solution as solvent.7 For synthesizing 18.76 grams of alcohol from an aldehyde, approximately 100 mL solvent was used,8 which needs to be neutralized later if implemented on a large scale. The literature also highlights that aldehydes can be directly reduced to alcohols in the presence of Lewis acids.
In 2022, Tanemura et al. demonstrated NaBH4 reductions of aldehydes and ketones in methanol and ethanol with 99% conversion in 10–60 minutes.9 Despite this efficiency, conventional methods still require extended reaction times, large solvent volumes, and energy-intensive reflux. The reductions are strongly exothermic (ΔH ≈ −530 kJ mol−1), often necessitating semi-batch addition of NaBH4 at low temperatures, which prolongs production.10 Over the past decade, various solvent-minimised strategies viz. grinding with solid acids, ball milling with liquid-assisted grinding, and sonication have been explored.11–21 While these methods exemplify that high reactivity can be used for accelerating the reaction, they suffer from limited scalability, variable reproducibility, and high energy demands (Table 1). Continuous processing offers clear advantages in handling exothermic reactions, as shown in systems employing Luche reagent or NaBH4-packed columns22–24 but these often rely on solvents or sacrificial media. In contrast, solvent-free methods reduce waste, conserve energy, and often enhance selectivity and yield,25–29 though efficient handling of solid materials remains challenging. Screw reactors address this limitation and have been successfully applied to OLED materials, amides, esters, dyes, and APIs.30–36 Building on these advances, we explore their potential for continuous, solvent-free NaBH4 mediated reductions.
| Ref. | Solvent [additive] | Reactor type | Substrate(s) | Key remarks |
|---|---|---|---|---|
| 7 | Caustic solution (aq) |  |
4-Chlorobenzaldehyde | Continuous flow, aqueous caustic solution used |
| 8 | MeOH |  |
Benzylideneacetone | Uses solvent, requires distillation for recovery |
| 9 | Solvent-free |  |
Benzaldehyde, vanillin, furfural | Scale-up via ball milling is not safe or feasible |
| 10 | Solvent-free |  |
Furfural, 2-naphthaldehyde | Mechanochemical approach in batch |
| 11 | MeOH, EtOH, THF |  |
Benzophenone, acetophenone | Solvent necessary for reaction |
| This work | Solvent-free [boric acid for ketones] |  |
Aldehydes and ketones | Solvent-free, continuous, safer heat management |
In this communication, we discuss solvent-free solid–solid and solid–liquid reactions for reduction of aldehyde and ketone functional groups in a PTFE single continuous glass-jacketed screw reactor. This approach facilitates the efficient processing of soft solid materials or pastes.
Reaction setup
Initially, the solvent-free synthesis was done in batch mode using a mortar and pestle. Subsequently, the experiments were performed in continuous mode using a single PTFE (polytetrafluoroethylene) screw reactor confined in a glass jacket (Fig. 1). All reactants were in solid or liquid forms, and the residence time was controlled by varying the speed of a calibrated motor and aligning the screw at different angles (vertical and horizontal). The glass jacket has a 39.6 mm outer diameter and an 18 mm inner diameter. It houses a 315 mm long PTFE screw with a diameter of 17.8 mm, which leaves a gap of only 0.2 mm between the jacket wall and the screw threads.During the experiment, initially, the motor connected to the screw reactor was turned on followed by the motors for the two screws that fed the reactants. The reaction was observed to initiate immediately after the reactants came into contact with each other and the reaction continued to proceed from the inlet to the outlet. The speed was managed with a variable frequency drive motor (Remi, India). The extruder outlet discharges directly into a continuous stirred tank reactor (CSTR-1) with intense mixing, where water is continuously added to quench the alkoxide intermediate. The reaction mass was facilitated to exit the CSTR via overflow directly into CSTR-2, where ethyl acetate helped continuous extraction, followed by phase separation using a conventional layer separator and a Zyput membrane separator. The system was designed in-house and manufactured locally (Alpro Industries Pvt. Ltd, India).
Results & discussion
Initial batch experiments were carried out solely to confirm the feasibility of sodium borohydride-mediated reduction of carbonyl compounds and to identify suitable stoichiometric ranges. These reactions were performed using a mortar and pestle at room temperature. In some cases, methanol was used to assist mixing and monitoring; however, these batch experiments were not intended to directly define the conditions for continuous processing. Instead, they served as qualitative screening studies to understand the influence of sodium borohydride equivalents and substrate electronic effects on reaction time. It was observed that one equivalent of NaBH4 typically resulted in complete aldehyde reduction within 15–30 min, whereas reducing the NaBH4 loading to 0.5 or 0.25 equivalents significantly increased reaction time to 0.5–1 hour. On the other hand, electron-withdrawing substituents on the aromatic ring facilitated reduction even at lower NaBH4 equivalents. Since this approach lacks scalability and offers inefficient mixing as well as heat transfer, all further experiments were performed using a continuous mechanochemical approach using a single screw reactor.Benzaldehyde was selected as the model substrate, and equimolar quantities of benzaldehyde and sodium borohydride were continuously fed into the reactor at room temperature. The screw rotation speed was systematically varied to control the residence time, while all other parameters were held constant. A hydride transfer reaction takes place in the screw reactor forming the corresponding benzyl alkoxide intermediate. Upon completion of the reduction, the extruder outlet discharges directly into a CSTR, where water is continuously introduced to quench the alkoxide intermediate, converting it to the corresponding alcohol. This reduction step is exothermic; heat is generated inside the screw reactor. Continuous stirring ensures efficient mixing and complete quenching. The quenched mixture exits the CSTR through an overflow outlet and is continuously transferred to CSTR-2, where ethyl acetate is added via (P-2). This mixture is then pumped by (P-3) to a liquid–liquid membrane separator for downstream phase separation occurs, and the organic layer containing the product is collected, while the aqueous layer is removed. This integrated setup enables uninterrupted continuous operation without the need to stop the extruder during downstream processing.
As summarized in Table 2, high screw speeds (180 rpm, 30 s residence time) resulted in incomplete conversion, affording only 25% isolated yield of benzyl alcohol (2a), due to the short residence time. Progressive reduction of the screw speed increased residence time and led to higher conversion: 42% yield at 120 rpm (45 s), 60% at 90 rpm (60 s), and 87% at 60 rpm (100 s). Optimal performance was achieved at 50 rpm, corresponding to a residence time of 120 s, yielding benzyl alcohol (2a) in 99% isolated yield. These conditions were selected as the optimized parameters for further studies. To monitor reaction progress and determine the optimal residence time, samples were collected directly from the extruder outlet at 30 s, 45 s, 60 s, 90 s, and 120 s residence times (as governed by the screw speed and feed rate). Each collected sample was immediately quenched with water and extracted with ethyl acetate, and the organic phase was analyzed by ATR-FTIR spectroscopy (Fig. 2). The disappearance of the characteristic aldehyde C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching band (∼1700 cm−1) and the appearance of O–H (∼3350 cm−1) stretching bands confirmed the progressive conversion of benzaldehyde to benzyl alcohol. This systematic sampling allowed correlation of residence time with reaction completion and enabled identification of the optimal operating conditions. In conventional batch reactors, this often results in localized overheating or requires slow reagent addition. In contrast, the jacketed single-screw reactor used in our study provides continuous and efficient heat management. The screw reactor is equipped with an external jacket that allows the circulation of cooling fluid for temperature control. During operation, a modest exothermic increase of approximately 5–7 °C was observed if operated in adiabatic mode. This exotherm can be efficiently dissipated by circulating cooling water through the reactor jacket to maintain the desired operating temperature. No hydrogen evolution was observed during extrusion. Hydrogen generation occurred only during the downstream aqueous quench, indicating complete formation of the alkoxide intermediate inside the screw reactor. ATR-FTIR spectra of material collected directly from the extruder further confirmed the disappearance of the aldehyde C
O stretching band (∼1700 cm−1) and the appearance of O–H (∼3350 cm−1) stretching bands confirmed the progressive conversion of benzaldehyde to benzyl alcohol. This systematic sampling allowed correlation of residence time with reaction completion and enabled identification of the optimal operating conditions. In conventional batch reactors, this often results in localized overheating or requires slow reagent addition. In contrast, the jacketed single-screw reactor used in our study provides continuous and efficient heat management. The screw reactor is equipped with an external jacket that allows the circulation of cooling fluid for temperature control. During operation, a modest exothermic increase of approximately 5–7 °C was observed if operated in adiabatic mode. This exotherm can be efficiently dissipated by circulating cooling water through the reactor jacket to maintain the desired operating temperature. No hydrogen evolution was observed during extrusion. Hydrogen generation occurred only during the downstream aqueous quench, indicating complete formation of the alkoxide intermediate inside the screw reactor. ATR-FTIR spectra of material collected directly from the extruder further confirmed the disappearance of the aldehyde C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O band prior to quenching (see the SI).
O band prior to quenching (see the SI).
Optimization table
Optimization of reaction conditions for reduction of aldehyde compounds.In a vertical single-screw reactor, under equimolar feed conditions (i.e. the flow rate is 2.40 mL min−1 for benzaldehyde and 0.88 g min−1 for NaBH4 at room temperature) at a residence time of 60 s, it showed incomplete conversion on TLC, while for a residence time of 2 minutes, 99% yield of benzyl alcohol (2a) was obtained. The substrate scope for reducing aldehyde derivatives was investigated using the optimized conditions. However, the additional reaction time of 60 s ensured complete conversion, resulting in a 99% yield of benzyl alcohol (2a) after workup.
Using the optimized conditions, a wide range of aldehydes were subjected to a reduction reaction. Electron-rich aldehydes such as 3,4,5-trimethoxybenzaldehyde got reduced rapidly, achieving complete conversion within approximately 1 min and affording the corresponding alcohol (2c) in 99% isolated yield. Electron-deficient substrates, including nitro- and halogen-substituted benzaldehydes, also underwent efficient reduction within 1–2 min, providing products (2d–2e, 2h) in 96–99% yields.
In the case of bulky substrates such as 9-anthracenecarboxaldehyde, movement of the reaction mass inside the screw reactor was slow due to its sticky nature. Under these circumstances, the addition of a minimal amount (0.2 to 0.4 mL) of methanol for 9 g material was required solely to improve flow behavior, allowing complete conversion within 5 min and yielding 1-pyrenemethanol (2f) in 99% yield after workup. This solvent was not required for the chemical transformation itself. Ketones were found to be less reactive than aldehydes under identical conditions. To address this limitation, boric acid (30 mol%) was co-fed with the ketone substrate and sodium borohydride into the screw reactor, where it acts as a Lewis acid to activate the carbonyl group toward hydride transfer. Under these conditions, ketones such as acetophenone derivatives and trifluoromethyl ketones were successfully reduced within 6–8 min residence time, affording the corresponding alcohols (2i–2k) in 97–98% isolated yields.
Overall, the residence time required for complete conversion ranged from 1 to 10 min, depending on the substrate structure and reactivity, consistently delivering high isolated yields (90–99%). Increasing the NaBH4 loading further accelerated the reaction but was not pursued due to economic considerations.
This process eliminates the need for column chromatography, as it achieves 100% conversion with the formation of a single product.
The scaled 50 mL screw reactor was operated continuously for 2 h under optimized conditions to evaluate operational stability. Benzaldehyde was continuously fed at a flow rate of 16.15 mL min−1, while sodium borohydride was introduced at 5.9 g min−1 to maintain the required stoichiometric ratio. During this period, consistent conversion (≈99%) was achieved without observable fouling, clogging, or performance decline. Based on the experimentally measured substrate throughput and isolated yield, approximately 2.06 kg of benzyl alcohol was produced during the 2 hour continuous operation, demonstrating the system's high productivity and stable performance.
The approach was also applicable to synthesis of various alcohol derivatives from aldehydes under optimized reaction conditions, i.e., furfural alcohol (2m) and 1-hexanol (2l). The quantitative yield of all products was obtained following the straightforward workup procedure. The products were subsequently characterized using 1H NMR and 13C NMR spectroscopy, confirming their structural integrity and purity (Fig. 3).
 | ||
| Fig. 3 Scope of derivatives for the reduction of aldehydes and ketones in a continuous flow mechanochemical process using a single screw reactor. | ||
The heat of the reduction reaction of any organic substrate using sodium borohydride varies in the range of −65 kJ mol−1 to −100 kJ mol−1. This implies that for the given scale of operation in the laboratory, the overall heat duty will vary from 0.243 kW to 1.122 kW, which is relatively manageable. However, upon scaling-up such a system by 100 times, one has to take care of 100 times higher heat duty as well as very steep heat generation rates. In such a situation, scale-up of screw reactors to a certain extent followed by numbering-up can be a realistic approach. We are exploring such approaches and the results will be reported separately to evaluate realistic scale-up options. In such a case, heat from this reaction can be effectively used for some useful purpose. Similarly, excess NaBH4 would release hydrogen from the CSTR during its quenching, which can be compressed and used for other reduction reactions.
Conclusions
A simple and efficient solvent-free method for the continuous flow reduction of aldehydes and ketones using sodium borohydride as a reducing agent at room temperature in a single screw reactor is reported. Nearly complete conversion was achieved in 2 to 10 minutes for ten representative aldehydes and ketones. More work on scale-up is in progress and will be reported in an appropriate engineering journal. Using a single screw reactor accelerates early stage developments due to its simplicity and compactness.Conflicts of interest
The authors have filed a patent based on part of the information in this paper.Data availability
The data that support the findings of this study are available in the supplementary information (SI) of this article. Supplementary information is available. See DOI: https://doi.org/10.1039/d5mr00127g.Acknowledgements
The authors acknowledge funding from DSIR (CRTDH (DSIR/CRTDH/NMCP/NCL/2021) at CSIRNCL) and SERB (DST, Govt. of India) (CRG/2023/001897-G) for this work. MP acknowledges financial support through Prime minister's PhD Fellowship. The authors extend sincere gratitude to Abhishek Patil for helping build a reaction setup and Dr Jayesh Sonawane for his help during the preparation of the manuscript.References
- A. I. P. Ltd, Global Sodium Borohydride Market to Hit Sales of USD 2,606 Million by 2032, at 5.89% CAGR Growth, https://www.globenewswire.com/news-release/2024/03/25/2851764/0/en/Global-Sodium-Borohydride-Market-to-Hit-Sales-of-USD-2-606-Million-by-2032-at-5-89-CAGR-Growth-Astute-Analytica.html, accessed April 12, 2025 Search PubMed.
- J. Magano and J. R. Dunetz, Org. Process Res. Dev., 2012, 16, 1156–1184 CrossRef CAS.
- M. Hey, Pap. Conserv., 1977, 2, 10–23 CrossRef.
- K. Naseem, Z. H. Farooqi, R. Begum and A. Irfan, J. Cleaner Prod., 2018, 187, 296–307 CrossRef CAS.
- H. N. Abdelhamid, Int. J. Hydrogen Energy, 2021, 46, 726–765 CrossRef CAS.
- C. Hardacre, H. Huang, S. L. James, M. E. Migaud, S. E. Norman and W. R. Pitner, Chem. Commun., 2011, 47, 5846–5848 RSC.
- V. Isoni, K. Mendoza, E. Lim and S. K. Teoh, Org. Process Res. Dev., 2017, 21, 992–1002 CrossRef CAS.
- T. Fukumoto and A. Yamamoto, Jpn. Pat., JP4760196, 1988.
- K. Tanemura, Results Chem., 2022, 4, 100486 CrossRef CAS.
- R. E. Davis and J. Carter, Tetrahedron, 1966, 22, 495–504 CrossRef CAS.
- K. Tanaka and F. Toda, Chem. Rev., 2000, 100, 1025–1074 CrossRef CAS PubMed.
- G. Kaupp, CrystEngComm, 2009, 11, 388–403 RSC.
- D. Tan, L. Loots and T. Friščić, Chem. Commun., 2016, 52, 7760–7781 RSC.
- G. Rothenberg, A. P. Downie, C. L. Raston and J. L. Scott, J. Am. Chem. Soc., 2001, 123, 8701–8708 CrossRef CAS PubMed.
- B. Rodríguez, T. Rantanen and C. Bolm, Angew. Chem., Int. Ed. Engl., 2006, 45, 6924 CrossRef PubMed.
- K. Tanemura, Tetrahedron Lett., 2021, 82, 153391 CrossRef CAS.
- C. B. Aakeröy and P. D. Chopade, Org. Lett., 2011, 13, 1–3 CrossRef PubMed.
- A. R. Hajipour, M. Mostafavi and A. E. Ruoho, Catal. Commun., 2007, 8, 1825–1828 CrossRef CAS.
- S. Kuntikana, C. Bhat, M. Kongot, S. I. Bhat and A. Kumar, ChemistrySelect, 2016, 1, 1723–1728 CrossRef CAS.
- M. Pattarawarapan, S. Jaita and W. Phakhodee, Tetrahedron Lett., 2016, 57, 3171–3174 CrossRef CAS.
- M. Firdaus, N. Handayani and L. T. Marfu’ah, Indones. J. Chem., 2016, 16, 229–232 CrossRef CAS.
- M. B. Plutschack, B. Pieber, K. Gilmore and P. H. Seeberger, Chem. Rev., 2017, 117, 11796–11893 CrossRef CAS PubMed.
- V. Bernardin, R. Philippe, L. Vanoye, C. De Bellefon and A. Favre-Réguillon, Ind. Eng. Chem. Res., 2024,(16), 6829–6836 CAS.
- K. Gilmore, S. Vukelić, D. T. McQuade, B. Koksch and P. H. Seeberger, Org. Process Res. Dev., 2014, 18, 1771–1776 CrossRef CAS.
- D. Bedi, A. Brar and M. Findlater, Green Chem., 2020, 22, 1125–1128 RSC.
- Z.-B. Xie, N. Wang, M.-Y. Wu, T. He, Z.-G. Le and X.-Q. Yu, Beilstein J. Org. Chem., 2012, 8, 534–538 CrossRef CAS PubMed.
- R. de S. Martins, M. P. Pereira, P. P. de Castro and F. I. Bombonato, Tetrahedron, 2020, 76, 130855 CrossRef CAS.
- M. Jeganathan, K. Kanagaraj, A. Dhakshinamoorthy and K. Pitchumani, Tetrahedron Lett., 2014, 55, 2061–2064 CrossRef CAS.
- S. Thiyagarajan, V. Krishnakumar and C. Gunanathan, Chem.–Asian J., 2020, 15, 518–523 CrossRef CAS PubMed.
- S. Spinella, M. Ganesh, G. L. Re, S. Zhang, J.-M. Raquez, P. Dubois and R. A. Gross, Green Chem., 2015, 17, 4146–4150 RSC.
- G. Moad, Prog. Polym. Sci., 2011, 36, 218–237 CrossRef CAS.
- R. S. Atapalkar and A. A. Kulkarni, Chem. Commun., 2023, 59, 9231–9234 RSC.
- M. Lavayssiere and F. Lamaty, Chem. Commun., 2023, 59, 3439–3442 RSC.
- M. Klimakow, P. Klobes, A. F. Thünemann, K. Rademann and F. Emmerling, Chem. Mater., 2010, 22, 5216–5221 CrossRef CAS.
- M. Banerjee, A. A. Bhosle, A. Chatterjee and S. Saha, J. Org. Chem., 2021, 86, 13911–13923 CrossRef CAS PubMed.
- R. R. A. Bolt, H. R. Smallman, J. A. Leitch, G. W. Bluck, F. Barreteau, A. V. Iosub, D. Constable, O. Dapremont, P. Richardson and D. L. Browne, Angew. Chem., 2024, 136, e202408315 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |