Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Force-driven architectonics of inorganic nanomaterials: pathways to smart and functional interfaces
Mohammed Ali
Dheyab
 *ab,
Wesam
Abdullah
*ab,
Wesam
Abdullah
 a,
Sara
Abdulwahab
a,
Sara
Abdulwahab
 a,
Sadeen Metib
Alsarayreh
a,
Sadeen Metib
Alsarayreh
 a,
Mothana Hussein
Tarawneh
a,
Mothana Hussein
Tarawneh
 a,
Mutaz Mohammad
Alsardi
a,
Mutaz Mohammad
Alsardi
 a,
Mansour A.
Alanazi
a,
Mansour A.
Alanazi
 c and
Azlan
Abdul Aziz
ab
c and
Azlan
Abdul Aziz
ab
aSchool of Physics, Universiti Sains Malaysia, 11800 Pulau Pinang, Malaysia. E-mail: mdali@usm.my; lan@usm.my
bNano-Biotechnology Research and Innovation (NanoBRI), Institute for Research in Molecular Medicine (INFORMM), Universiti Sains Malaysia, 11800, Pulau Pinang, Malaysia
cDepartment of Physics, College of Science, Northern Border University, Arar P. O. Box 1321, Saudi Arabia
First published on 7th January 2026
Abstract
The deliberate structuring of inorganic nanomaterials through mechanical forces offers a powerful alternative to conventional synthesis, enabling solvent-free, energy-efficient, and scalable design strategies. Rather than serving only as a synthetic shortcut, force-driven processing is increasingly recognized as an architectonic tool as a means of directing matter into well-defined architectures that integrate top-down shaping with bottom-up assembly. This review develops a conceptual framework of architectonics under mechanical activation, treating external force as a design parameter that dictates structure formation across multiple length scales. Methodological platforms such as ball milling, extrusion, and hybrid force–stimuli systems are systematically assessed, alongside mechanistic insights spanning multiscale reaction pathways, computational modeling, and AI-enabled predictions. The potential of this approach to generate smart and functional interfaces is highlighted through applications in catalytic and energy conversion processes, biomedical nanomedicine, and electronic or sensing devices. Finally, we discuss current limitations particularly gaps in mechanistic understanding, predictive control, and scalability and outline future opportunities to advance force-driven architectonics as a foundation for next-generation functional inorganic nanomaterials.
1. Introduction
Mechanochemistry, as defined by the International Union of Pure and Applied Chemistry (IUPAC) Compendium of Chemical Terminology, refers to chemical reactions induced by mechanical energy,1 while Heinicke further described it as encompassing chemical and physico-chemical transformations of substances in all states of aggregation under mechanical force.2 Building on this foundation, Takacs et al.3 demonstrated that mechanical force produces qualitatively distinct outcomes compared to thermal or photochemical treatments, establishing an early cornerstone for the field. Over recent decades, mechanochemistry has evolved from a scientific curiosity into a versatile methodology enabling solvent-free, reproducible, and scalable transformations aligned with the principles of green chemistry.4–6Ball milling in particular has re-emerged as a powerful solid-state technique, providing the necessary mechanical energy to drive reactions rapidly and under environmentally benign conditions.7,8 Initially considered a top-down comminution process for producing micro- and nanoscale powders,2,9 milling is now innovatively adapted for bottom-up nanoparticle synthesis through chemical reactions.10,11 Mechanochemical grinding of oxides exemplifies this dual role, where simultaneous physical and chemical processes occur in coordinated patterns, yielding superior fine powders compared to conventional routes.8,12 Despite its simplicity, mechanical milling often induces amorphization and surface defects, as shown by Šepelak's observation of 2–5 nm amorphous surface layers on oxide nanoparticles,13,14 yet these very features underpin the phenomenon of mechanical activation. More broadly, mechanochemical approaches such as mechanical milling, alloying, reactive extrusion, and induced solid-state reactions offer efficient, solvent-free, and sustainable routes for producing nanocrystalline materials and functional composites.15–17 Recognized as a pillar of green chemistry, mechanochemistry has been named by IUPAC among the top ten chemical innovations shaping sustainable science,18 underscoring its relevance for modern material design. Compared with conventional wet-chemistry syntheses that generate solvent waste, involve prolonged treatment times, and require multistep drying or surfactant-assisted procedures,8,19,20 mechanosynthesis bypasses solubility limitations and reduces hazardous waste while lowering energy input and reaction time.21–24 These advantages have enabled the scalable, solvent-free synthesis of diverse nanomaterials, including oxides (TiO2, CeO2, Gd2O3) and metals such as Fe,25 as well as perovskites, catalytic systems, and superhydrophobic surfaces.15,16,20,26
In recent years, applications of mechanochemical synthesis have expanded into pharmaceuticals, ligand design, drug synthesis, graphene production, and the surface modification of nanomaterials.23,27 Importantly, soft mechanochemical strategies employing solid acids, bases, hydrated compounds, or salts have opened new opportunities for tailoring inorganic nanostructures,28 while dry synthesis routes minimize solvents or surfactants, providing significant ecological and functional benefits.29–32 The performance of noble metal nanoparticles, governed by size, shape, composition, and elemental distribution, further illustrates the critical role of mechanochemistry in advancing functional nanomaterials, as their catalytic, electronic, optical, and biomedical properties extend far beyond the aesthetic appeal that first inspired their study.33,34 Collectively, these advances position mechanochemistry as a transformative platform for nanomaterials synthesis, bridging multiscale pathways of activation and assembly with the design of smart and functional interfaces.
Nonetheless, despite these achievements, previous academic research on mechanochemical synthesis has primarily emphasized scalability, solvent-free operation, and sustainability benefits, while leaving critical gaps unaddressed. Notably, Fiss et al.35 highlighted the advantages of mechanochemical strategies in inorganic redox and ion-exchange processes, yet their analysis lacks mechanistic depth and broader sustainability metrics beyond nanoparticles and hybrid frameworks such as metal–organic frameworks (MOFs). While MOFs are classified as organic–inorganic hybrid systems, their inclusion in mechanochemical discussions is common due to their highly crystalline architectures and force-responsive porosity, which often parallel the behaviors of inorganic nanomaterials. Likewise, Auvray and Friščić6 offered valuable mechanistic insights and a systematic classification for hard and high-melting inorganic materials, but their work overlooks opportunities in nanomedicine and hybrid nanostructures. Similarly, Xu et al.16 underscored the simplicity and catalytic benefits of mechanochemical synthesis as a green alternative to conventional routes, although their discussion remains confined to catalysis, neglecting wider applications in energy storage and biomedical interfaces. In light of these deficiencies, our review aims to address the unmet need for a cross-disciplinary framework in mechanochemical synthesis of inorganic nanomaterials. We provide a coherent architectonic perspective that goes beyond catalysis and selected inorganic systems, integrating methodological platforms, mechanistic insights, and smart interface engineering. By systematically linking force-driven design to functional outcomes in catalysis, energy storage, and biomedicine, we establish mechanochemistry as a versatile architectonic tool rather than a niche synthetic route. This exclusive focus on designing smart and functional interfaces aligns with global priorities for sustainable, scalable, and multifunctional nanomaterials, offering a roadmap for future advances across chemistry, materials science, and nanomedicine.
2. Mechanochemical architectonics: conceptual framework
2.1. Defining “architectonics” in mechanochemistry
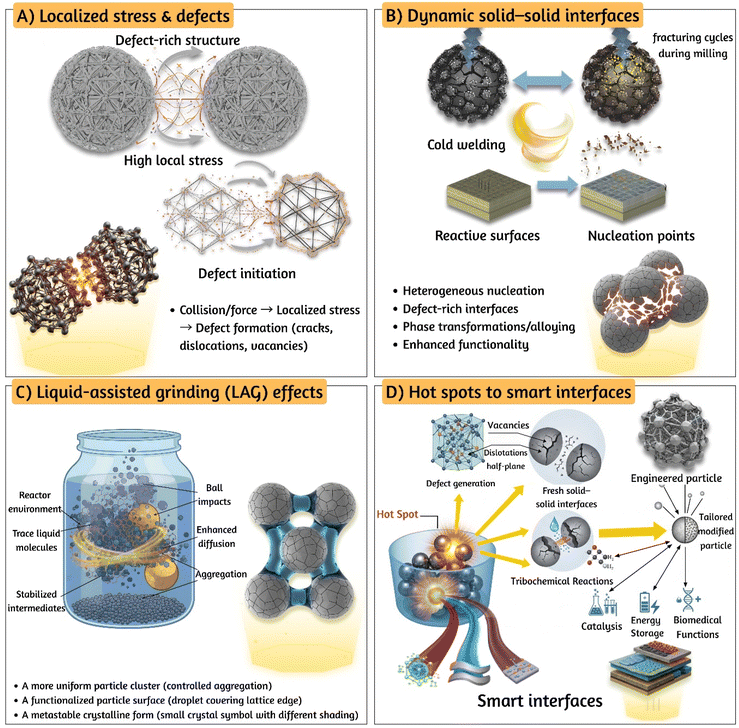
Mechanochemical architectonics reframes the synthesis of inorganic nanomaterials as a deliberate, multiscale strategy that couples mechanical energy input with interfacial design principles to yield functional architectures rather than merely discrete particles. In this framework, architectonics denotes the purposeful construction of structure function relationships across length scales ranging from atomic arrangements and defect structures to particle morphology and interfacial connectivity. Mechanical actions such as impact, shear, or extrusion serve as orthogonal variables that control crystallinity, defect chemistry, surface coordination, and assembly pathways. Unlike conventional thermal or solution-based routes, mechanochemical reactions (i) activate otherwise inaccessible transformations by generating high-energy defect states and transient solid–solid contacts, (ii) impose nonequilibrium mixing that favors metastable polymorphs or alloying, and (iii) induce tribochemical processes that produce unique interfacial chemistries. These attributes establish mechanochemistry as a versatile lever for architectonic control.36,37The power of mechanochemical architectonics comes from three interconnected processes (Fig. 1). First, the intense local stresses and collisions during milling distort the crystal lattice and create defect-rich areas. These defects make it easier for ions to move and redox reactions to happen, allowing solids like metals and oxides to transform quickly at the atomic level.38 For instance, when making metal oxide nanoparticles like Fe2O3, milling causes repeated breaking and welding of particles, which increases defects and speeds up reactions that would normally require high heat, enabling them to occur near room temperature.39 Second, the constant cycle of cold welding and fracturing during milling keeps generating fresh, reactive surfaces and dynamic interfaces between solids. These tiny surfaces act like small reactors where the chemistry occurs how many of these nucleation spots form and their chemical nature ultimately shape the final product. A good example is making mixed oxides like titanium dioxide (TiO2) and cerium dioxide (CeO2) by milling: the process disperses these oxides evenly and mixes them at the nanoscale, resulting in catalysts with unique properties without needing to heat them in bulk.40 Mechanochemical milling of TiO2–CeO2 at 350 rpm for 100 min yields nanoscale composites with particle sizes ∼5 µm and enhanced oxygen sensitivity (up to 8.2 at 320 °C), demonstrating force-driven architectonics without bulk heating. Third, adding small amounts of liquid during milling (called liquid-assisted grinding) changes how energy moves and how surfaces interact. This helps guide how particles aggregate or bond on the surface, offering precise control over the assembly and functionalization of particles.38 This combination of mechanical stress, dynamic surface creation, and liquid effects makes mechanochemical architectonics a powerful method for designing and synthesizing advanced materials efficiently and in environmentally friendly ways.
Shi et al.41 demonstrate a form of nano architectonics by embedding Pt single-atoms and differently-sized Pt nanoparticles (1.5, 3.0, and 7.0 nm) onto manganese-oxide nanosheets. Although the synthesis was not purely mechanochemical, the principles of architectonics namely, vacancy filling, atomic dispersion, and interface-driven functionality are central to the material's catalytic behavior. The manganese-oxide nanosheets serve as a defect-rich scaffold, with oxygen vacancies acting as anchoring sites for Pt species. The interaction between Pt and MnO2 is not merely compositional but architectonic: it alters the oxidation state of manganese, modulates the oxygen vacancy concentration, and tunes the catalytic activity of the hybrid nanozyme. This reflects the essence of mechanochemical architectonics where structure and function co-evolve through controlled energy input and spatial design.
Despite its promise, several limitations constrain the broader adoption of mechanochemical architectonics. Reproducibility and parameter standardization remain persistent challenges: milling frequency, ball-to-powder ratio, filling factor, and jar geometry introduce nonlinear effects on energy input and product distribution, complicating cross-study comparisons. Contamination from milling media, together with uncontrolled localized heating, may obscure mechanistic interpretation and compromise functional performance. Scale-up is particularly problematic, as laboratory-scale ball mills generate heterogeneous energy distributions that are not easily replicated in continuous or industrial platforms. Furthermore, mechanistic insights are often inferred from ex situ characterizations, which risk conflating transient intermediates with end products.42
Several strategies have been proposed to address these limitations and strengthen the mechanochemical toolkit. Standardized reporting of energy metrics (e.g., J g−1 or specific impact energy) would enable quantitative comparison across studies. Media wear and contamination can be minimized through careful selection of ceramic liners and inert milling components. The systematic use of Liquid-Assisted Grinding (LAG), rather than as an ad hoc additive, allows controlled tuning of interfacial chemistry. Hybrid activation methods combining mechanical milling with thermal annealing, sonication, or photonic inputs may further decouple defect generation from morphological evolution. Most critically, real-time monitoring through acoustic emission, synchrotron X-ray scattering, Raman spectroscopy, and operando temperature–pressure probes offer the potential to resolve transient intermediates and establish direct correlations between energy input and physicochemical outcomes. The development of scalable, continuous mechanochemical reactors such as twin-screw extruders and reactive ion-milling devices, validated against batch systems with conserved energy metrics, represents an essential step toward industrial translation.43
A central mechanistic underpinning is the generation of localized, transient high-energy sites commonly referred to as “hot spots” which store strain energy and enable bond cleavage and atomic rearrangements without the need for bulk thermal activation.44 The stochastic nature of collisions and nonequilibrium energy distributions during milling open pathways inaccessible to conventional methods, resulting in accelerated reaction rates, selective product formation, and stabilization of otherwise unstable intermediates. This capacity to create dynamically evolving “smart” interfaces, where chemical state, morphology, and function are continuously reconfigured under mechanical stress, provides a rational basis for enhanced catalytic, electronic, and optical performance.45
Recent studies illustrate these dynamics with increasing clarity. For instance, the mechanochemical synthesis of MOFs such as MOF-74 proceeds through rapid formation of short-lived intermediates, with liquid additives accelerating coordination and structural reorganization. In situ powder X-ray diffraction (PXRD) has revealed that these intermediates adopt distinct coordination motifs or polymorphic forms, highlighting the structural dynamism inherent to mechanochemical pathways.46 Klimakow et al.47 highlight mechanochemical architectonics is exemplified through the solvent-free synthesis of MOFs, specifically HKUST-1 and MOF-14, using ball milling techniques. The authors demonstrate that mechanical energy can be harnessed not only to drive chemical reactions but also to sculpt the pore architecture and surface properties of the resulting materials. This approach yields high specific surface areas up to 1713 m2 g−1 comparable to the best values reported via conventional solvothermal methods, yet achieved without solvents or extensive post-synthetic treatments. Similarly, mechanochemical anion-exchange in lead(II) coordination polymers demonstrates crystal-to-crystal transformations that preserve crystallinity while enabling functional modulation, underscoring the potential of architectonics to deliver solvent-minimized, highly efficient material design strategies.48
The objectives of architectonics are realized by intentionally guiding how simple molecular or nanoscale units organize into more complex and functional architectures. Rather than relying on spontaneous assembly, architectonic design emphasizes control over interfacial interactions, defect formation, and assembly pathways, allowing structure to evolve in a predictable manner across length scales. In mechanochemical systems, mechanical force serves not only as an energy source but also as a regulating element that influences contact dynamics, surface reactivity, and kinetic pathways. Through careful adjustment of processing conditions and chemical environments, this approach enables a direct connection between architectural design and material performance.49,50 This architectonic perspective provides the foundation for treating mechanical force not merely as an energy source, but as a tunable design parameter.
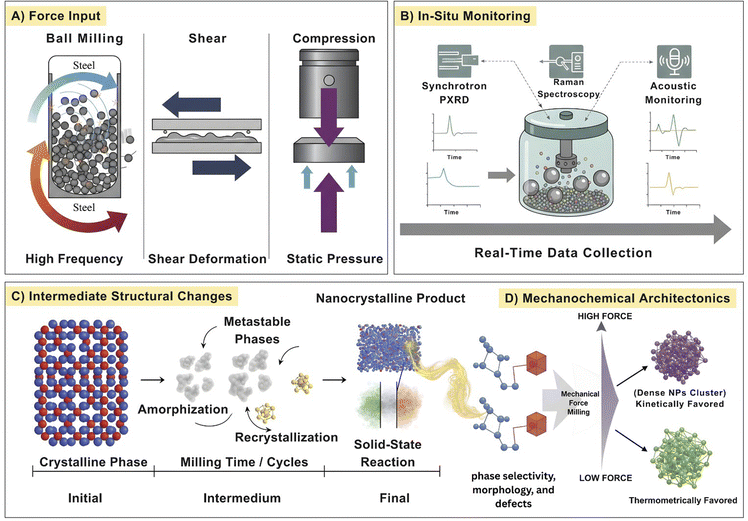
2.2. Force as a design parameter
Mechanochemical force is not simply an energetic input it is a tunable design parameter, finely controllable and capable of directing reaction pathways, structural outcomes, and interfacial functionality. In contrast to thermal or solution-based methods, mechanical force in milling is discrete, spatially heterogeneous, and temporally dynamic, offering unparalleled opportunity for architectonic control.51 At the microscopic level, force defines the nature and density of reactive sites. Higher milling frequencies and greater impact energies induce localized lattice distortions, shear bands, and defect clusters that dramatically reduce activation barriers for ionic migration or redox transformations. For example, increasing frequency from 15 to 30 Hz in a ball mill amplifies single-collision energies nearly fourfold (0.49 mJ to 1.96 mJ), translating directly into faster conversions in C–C coupling reactions and enhanced solid-state diffusion in alloy formation. Here, single collision energy refers to the mechanical energy transferred during an individual impact event between the milling media and the material. The energy values reported in mJ correspond to milli-Joules (10−3 J).52 This correlation underscores why force tuning enables mechanochemical pathways inaccessible to conventional routes: the creation of transient, high-energy states that bias metastable phase formation, alloying, or selective bond scission. To consolidate force as a mechanistic design variable, Fig. 2 couples applied mechanical input with experimentally validated structural evolution pathways in mechanochemical systems. Increasing collision energy and frequency drive force-dependent transitions from lattice distortion and defect nucleation to metastable intermediate formation, amorphization, or selective recrystallization, as directly observed by in situ synchrotron PXRD through time-resolved changes in phase fraction and peak broadening.53 Complementary operando Raman spectroscopy captures force-induced bond reorganization and intermediate lifetimes, confirming that mechanical force reshapes reaction energy landscapes rather than merely accelerating kinetics. The figure further highlights intrinsic force heterogeneity, where localized high-energy “hot spots” coexist with under-energized regions, providing a physical basis for simultaneous crystalline, amorphous, and defect-rich domains within a single milling environment.51Additionally, force controls how solid–solid interactions collectively evolve. The balance between fracturing and cold welding, controlled by milling intensity and ball-to-powder ratio (BPR), regulates the frequency of interface renewal and the density of nucleation sites. High BPRs accelerate composite synthesis, as demonstrated in molybdenum disilicide (MoSi2) and titanium carbide (TiC) systems where a 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio completed conversion in 10 h compared to incomplete reaction at 5
1 ratio completed conversion in 10 h compared to incomplete reaction at 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 even after 30 h.54 Similarly, denser milling media such as zirconium dioxide (ZrO2) or tungsten carbide (WC) deliver sharper impacts that promote rapid defect generation but can also over-fragment sensitive phases, producing amorphization or unintended contaminations.54 These trade-offs illustrate the duality of force: it can either enable rapid architectonic assembly or induce destructive over-milling.
1 even after 30 h.54 Similarly, denser milling media such as zirconium dioxide (ZrO2) or tungsten carbide (WC) deliver sharper impacts that promote rapid defect generation but can also over-fragment sensitive phases, producing amorphization or unintended contaminations.54 These trade-offs illustrate the duality of force: it can either enable rapid architectonic assembly or induce destructive over-milling.
Despite its centrality, quantitative control of force in mechanochemical synthesis remains underdeveloped. Most studies report milling parameters such as frequency, ball size, or jar material but these serve as indirect proxies rather than true measures of force. Without standardized reporting of impact energy (J g−1), collision frequency, or energy density, reproducibility across laboratories suffers, reducing mechanistic comparisons to qualitative speculation at best. For example, Belenguer et al.55 emphasized that variations in jar size, shape, ball size, and materials can dramatically influence reaction outcomes even when nominal conditions remain constant highlighting the sensitive dependency of mechanochemical reactions on experimental setup specifics. Adding to this complexity, Jafter et al.56 developed a kinematic modeling framework that demonstrates how even minor changes in planetary mill geometry (such as jar radius or sun wheel dimensions) can cause a 2–3× variation in impact energy, despite identical milling speed and ball load (Fig. 3A(a)). This variability stems from the way geometric parameters influence effective impact velocity, which directly affects energy transfer. Moreover, force distribution within the milling jar is inherently heterogeneous. Fig. 3A(b) illustrates how only a subset of particles experience sufficient impact energy to overcome activation barriers, creating localized (hot spots) while adjacent regions remain under-energized. This spatial disparity aligns with granular media models predicting wide force fluctuations due to varied contact angles and bead pack dynamics. Additionally, Fig. 3A(c) highlights how ball material density alters the filling factor even at constant mass affecting the total volume occupied and thus the energy delivery profile. These insights are particularly relevant for inorganic mechanochemical reactions, such as the synthesis of metal oxides (e.g., TiO2, ZnO) and intermetallics (e.g., MoSi2–TiC), where defect formation, phase transitions, and interface engineering are highly sensitive to localized energy input.
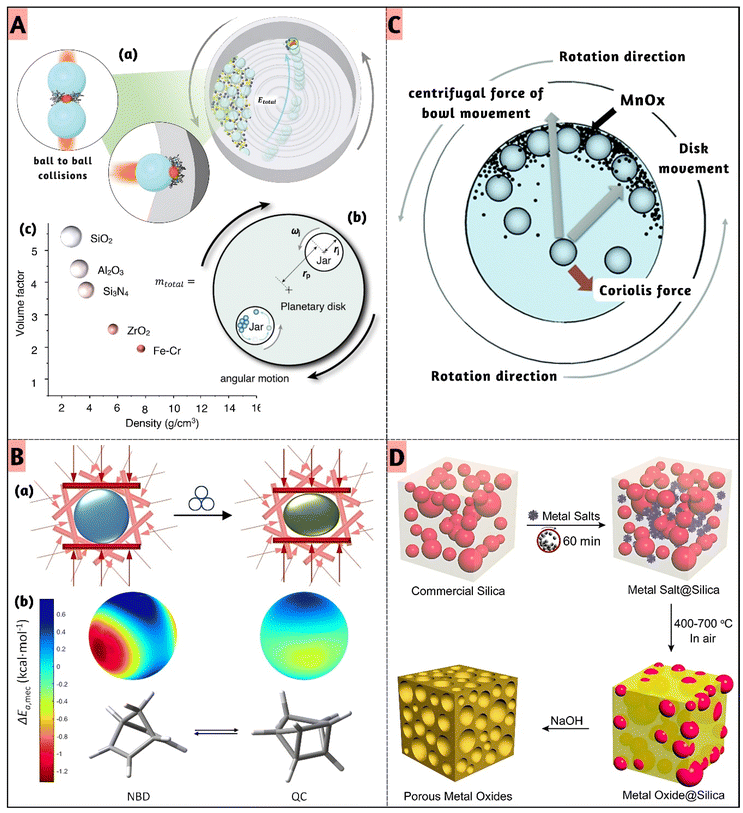 | ||
| Fig. 3 (A) (a) Ball collisions during circular motion deliver mechanical energy directly to the reactant molecules. (b) The mill's illustration and the pertinent factors that affect the effective impact velocity and (c) balls have particular properties that have a direct impact on the energetics. If all ball types had the same mass, the size of the balls in the illustrations would match the relative size. Reduced with permission.56 Copyright 2024, Wily. (B) (a) Considering all directions, random collisions drive mechanochemical reactivity to form the product. (b) (Left) Activation energy drops by ∼1.2 kcal mol−1 under 2 GPa, indicating a “hot” mechanochemical species. (Right) Minimal response reflects a “cold” species; isomer orientations differ for clarity. Reduced with permission.58 Copyright 2025, American chemical society. (C) Milling-map of the samples, displaying the grounding process with different ball-impact and cumulative energy which is dependent on the rotational speed. Reduced with permission.60 Copyright 2020, Royal Society of chemistry. (D) Mechanochemical nano casting method for porous metal oxide. Adapted with permission.63 Copyright 2018, American chemical society. | ||
Moreover, force distribution within the milling environment is far from homogeneous. Particle trajectories and impact geometries create regions of intense energy concentration (hot spots) and adjacent under-energized areas (cold zones), all within the same jar. This spatial heterogeneity complicates mechanistic interpretation, as only a subset of the material may undergo defect-rich transformations while the remainder remains largely unaltered. This behavior aligns with earlier theoretical models of granular media that predict wide force fluctuations due to varied contact angles and force networks even in static bead packs.57 De Armas et al.58 quantifies how directional compressive forces mimicking ball-milling impacts affect activation energy across molecular orientations. Mechanical activation energy varied by up to 1.2 kcal mol−1 depending on impact direction under 2 GPa pressure, confirming the presence of “hot spots” where reactivity is enhanced. Conversely, the reverse reaction showed negligible mechanical response, illustrating “cold zones” with minimal activation. This anisotropic behavior validates granular media models predicting force heterogeneity due to variable contact angles and impact geometries (Fig. 3B).
A further limitation arises from the interplay between mechanical and thermal effects. At high frequencies, energy dissipation as heat can blur the distinction between mechanochemical and thermally assisted pathways, confounding mechanistic models. In polymer mechanochemistry, excessive force accelerates chain scission, undermining the very architectonic control sought in synthesis. Such examples highlight that more force does not necessarily mean better outcomes; instead, there exists an optimal force window where activation is maximized while degradation is minimized.59 In a detailed investigation of oxide nanoparticle formation, Ochirkhuyag and co-workers reported that increasing the milling speed from 200 to 600 rpm raised the average collision energy from 0.1 to 0.5 J per impact, resulting in a nearly fourfold increase in defect density and a corresponding reduction in crystallite size from 45 nm to 12 nm (Fig. 3C). In the mechanochemical synthesis, that Raman bands at ∼640 cm−1 (MnO2), ∼580 cm−1 (Mn2O3), and ∼490 cm−1 (Mn3O4) evolved systematically with milling time, where extending milling from 1 h to 6 h shifted the spectrum toward the Mn3O4-dominant profile, quantitatively confirming the collision-driven reduction pathway induced by high-energy ball milling.60 Similarly, Zhao et al.61 demonstrated that applying higher milling frequency (30 Hz vs. 15 Hz) increased the lattice strain in Fe2O3 nanoparticles from 0.12% to 0.36%, which directly correlated with a 2.7× increase in catalytic activity for CO oxidation. These results show why controlled force input is central to defect engineering and catalytic optimization. Force modulation also governs interface formation. In the mechanochemical synthesis of TiO2-based composites, Bharath and colleagues observed that varying the ball-to-powder ratio (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 20
1 to 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) accelerated reaction completion from 30 h down to 10 h, as more frequent high-energy collisions generated fresh interfaces and increased nucleation site density. In their solvent-free mechanochemical synthesis used Raman spectroscopy to track structural disorder and reduction, where the characteristic D and G bands provided direct evidence of defect generation and graphitic restoration under high-energy milling mirroring how increased collision intensity in other mechanochemical systems accelerates interface formation and reaction progression.62 Importantly, these fresh interfaces were chemically distinct from bulk surfaces, hosting under-coordinated atoms that enabled selective photocatalytic activity under visible light.
1) accelerated reaction completion from 30 h down to 10 h, as more frequent high-energy collisions generated fresh interfaces and increased nucleation site density. In their solvent-free mechanochemical synthesis used Raman spectroscopy to track structural disorder and reduction, where the characteristic D and G bands provided direct evidence of defect generation and graphitic restoration under high-energy milling mirroring how increased collision intensity in other mechanochemical systems accelerates interface formation and reaction progression.62 Importantly, these fresh interfaces were chemically distinct from bulk surfaces, hosting under-coordinated atoms that enabled selective photocatalytic activity under visible light.
Nevertheless, excessive force can be detrimental. Xiao et al.63 reported that over-milling at 800 rpm caused amorphization in zinc oxide (ZnO) nanostructures, lowering photocatalytic efficiency by >35% compared to optimally milled samples. This underscores the need to define an optimal force window, where sufficient defect generation enhances reactivity but excessive impacts lead to structural collapse and loss of crystallinity (Fig. 3D). Operando and in situ studies now provide direct experimental validation of force-driven mechanisms. In situ synchrotron PXRD during MOF-74 ball milling reveals rapid formation of short-lived intermediates with distinct coordination motifs, where liquid-assisted grinding accelerates structural reorganization under controlled collision energies. Operando acoustic spectroscopy monitors mechanochemical reactions in real-time, tracking acoustic emission changes that correlate with reaction progress and energy input during ball milling.64 The lack of standardized force quantification in mechanochemistry remains a persistent challenge, with most studies relying on indirect parameters such as rpm, ball size, or filling ratio. Li et al.65 addressed this limitation by introducing the concept of specific energy dose (J g−1), correlating mechanical input with reaction yield across different milling platforms. In their synthesis of defective molybdenum trioxide (MoO3), TiO2, and ZnO, they demonstrated that a specific energy dose of approximately 1200 J g−1 was sufficient to induce significant oxygen vacancy formation and hydrogen doping in MoO3, yielding localized surface plasmon resonance properties. In the archetypal mechanochemical synthesis of the metal–organic framework ZIF-8 from ZnO and 2-methylimidazole, in situ synchrotron PXRD measurements revealed that the distinctive Bragg reflection intensity of the (011) plane a proxy for product crystallinity evolves markedly with milling time, showing initial formation of ZIF-8 within minutes followed by significant amorphization over ca. 30–40 min of continued milling (with complete amorphization observed at ∼30 min under certain liquid additive conditions) and retention of crystalline features up to ∼55 min with higher liquid content, demonstrating force-dependent structural kinetics directly tied to milling parameters such as liquid volume and additive concentration.66 This time-resolved intensity profiles provide quantitative evidence that prolonged mechanical impact does not simply accelerate reactions but actively alters phase stability and pathway, consistent with mechanochemical architectonic control. Beyond diffraction, in situ coupled PXRD–Raman spectroscopic studies have been used to track mechanochemical reactions in real time, providing correlated structural and vibrational data that reveal the formation and consumption of intermediate phases with clear kinetic signatures as a function of mechanical input.67
3. Methodological platforms for mechanochemical inorganics
3.1. Ball milling and grinding architectures
Among the various mechanochemical platforms including manual grinding, liquid-assisted grinding, extrusion-based processing, high-pressure compaction, and shear-induced activation ball milling has emerged as the most widely adopted and systematically investigated architecture for inorganic synthesis, as its comminution, impact, and mixing dynamics efficiently overcome activation barriers in solid-state reactions and enable reactivity inaccessible to conventional solution-based methods.68 Evolving from manual grinding to advanced planetary, vibratory, and Simoloyer mills, as well as scalable extrusion systems, these architectures provide enclosed, controllable environments that ensure reproducible, efficient, and adaptable synthesis across organic and inorganic frameworks.69–71 Characteristic advantages include short reaction times, high yields, precise stoichiometric control, and the ability to proceed without bulk solvent irrespective of reactant solubility.23,72,73 Automated mills such as shaker, planetary, and attritor devices allow programmable control of frequency and impact, while the selection of jar and media materials critically shapes frictional dynamics and mechanochemical reactivity.32,74 Notably, LAG, defined by the η-parameter, expands the reaction scope by enhancing reactivity and selectivity.75 This versatility is evident in the successful in situ preparation of hydrides such as LiBH4,76 lithium-mediated ammonia-free Birch reductions,77 and scandium complexes unobtainable in solution.78 Beyond molecular transformations, mechanochemical milling has proven effective in driving gas–solid reactions under ambient conditions, Lou et al.79 achieved urea synthesis at a yield rate of 41.61 mg L−1 h−1 using ZrO2 jars and balls, as schematically depicted in Fig. 4A, with earlier studies reporting ammonia generation up to 82.5 vol% and nitrogen fixation of 2.432 mg L−1 h−1, underscoring the sustainable potential of ball–powder collisions for nitrogen conversion.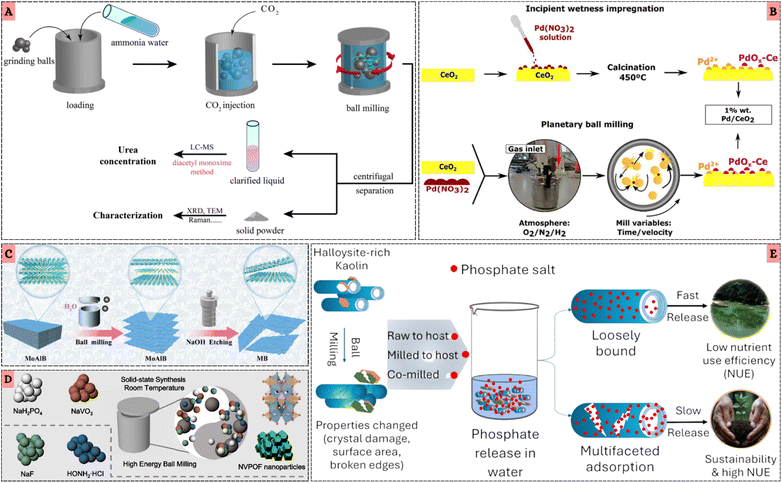 | ||
| Fig. 4 (A) Schematic illustration of the urea synthesis process using ZrO2 milling balls and a milling jar. Reproduced with permission.72 Copyright 2025, American chemical society. (B) Illustration of the two methods used to prepare Pd/CeO2 samples: incipient wetness impregnation and planetary ball milling under controlled atmosphere. Reproduced with permission.74 Copyright 2025, American chemical society. (C) Synthesis flowchart of ZIF-90PN@MB. Reproduced with permission.61 Copyright 2025, American chemical society. (D) Schematic illustration of the mechanochemical synthesis of Na3(VOPO4)2F nanoparticles starting from NaVO3, depicting the typical synthetic process of NVPFs. Reproduced with permission.75 Copyright 2021, Nature. (E) Schematic illustration of the mechanochemical activation of halloysite nanotube-rich kaolin clay and its application as a carrier for sustainable slow-release phosphate fertilizer. Reproduced with permission.76 Copyright 2025, American chemical society. | ||
At the architectural scale, Dhokale et al.80 demonstrated the critical role of milling parameters, where 7 mm balls resulted in 30% yield, while optimizing with 34 smaller 4 mm balls improved efficiency to 44% within only 99 minutes. Similarly, Marín et al.81 highlighted the impact of planetary ball milling forces on catalytic activity, showing that an optimum velocity of 250 rpm maximized metal–support interactions compared to both lower- and higher-energy regimes, while the reaction atmosphere further modulated activity. Their comparative mechanistic insights are illustrated in Fig. 4B, contrasting incipient wetness impregnation with atmosphere-controlled ball milling in the preparation of Pd/CeO2 catalysts. In another example, Zhang et al.68 processed MoAlB through intensive milling at 1000 rpm for 12 h, generating amorphous phases and thinner nanosheets, followed by NaOH etching to obtain well-dispersed MBene (MB) sheets. The mechanistic sequence of lattice disruption and selective Al removal is shown in Fig. 4C, underscoring how milling can tailor nanoscale inorganic architectures.
In the context of energy storage, Shen et al.82 synthesized Na3(VOPO4)2F nanoparticles via high-energy ball milling, achieving ∼5 nm crystallites, in situ carbon-active composites, and ultralong cycling stability (98% over 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles). This industrially scalable process (2 kg batches, 26
000 cycles). This industrially scalable process (2 kg batches, 26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 650 cells) is visualized in Fig. 4D, highlighting the multiscale transformation pathways accessed during solid-state milling. To establish that mechanochemical routes extend well beyond this single system, several additional force-driven syntheses have demonstrated similarly robust performance enhancements across diverse electrode chemistries.83 For example, solvent-free mechanochemical activation has proven highly effective for Na-ion Prussian blue analogues, where reducing reaction activation energy and promoting solid-ion diffusion trigger low-temperature solid-state transformations. He et al.84 showed that a Na-rich monoclinic NaMHCF cathode prepared entirely by milling exhibits high specific capacities, superior rate capability, and fully reversible multi-phase evolution during Na+ (de)insertion representing one of the most compelling demonstrations of mechanochemistry-enabled Na-ion storage. Recent advances in all-solid-state lithium batteries provide yet another compelling line of evidence, where mechanochemical synthesis has enabled the scalable preparation of Li3PS4-based sulfide electrolytes with room-temperature ionic conductivities above 10−4 S cm−1 and favorable mechanical deformability that enhances solid–solid interfacial contact. Importantly, aliovalent ZnO substitution yields glass-ceramic Li3PS4 with significantly improved conductivity (1.12 × 10−3 S cm−1) and increased stability against both air and lithium metal, illustrating how structural distortion and defect engineering introduced by force-driven processing can be leveraged to optimize ion-transport pathways and long-term cycling reliability.85 Complementarily, Biswas et al.86 demonstrated that planetary milling of halloysite-rich kaolin shortened tubular morphologies by ∼44% within 1 h and ∼53% within 2 h, where dry milling produced nanofragments while wet milling preserved tubular structures but still tripled surface area. As highlighted in Fig. 4E, such modifications critically regulate phosphate adsorption and release, advancing kaolin as a sustainable slow-release fertilizer carrier.
650 cells) is visualized in Fig. 4D, highlighting the multiscale transformation pathways accessed during solid-state milling. To establish that mechanochemical routes extend well beyond this single system, several additional force-driven syntheses have demonstrated similarly robust performance enhancements across diverse electrode chemistries.83 For example, solvent-free mechanochemical activation has proven highly effective for Na-ion Prussian blue analogues, where reducing reaction activation energy and promoting solid-ion diffusion trigger low-temperature solid-state transformations. He et al.84 showed that a Na-rich monoclinic NaMHCF cathode prepared entirely by milling exhibits high specific capacities, superior rate capability, and fully reversible multi-phase evolution during Na+ (de)insertion representing one of the most compelling demonstrations of mechanochemistry-enabled Na-ion storage. Recent advances in all-solid-state lithium batteries provide yet another compelling line of evidence, where mechanochemical synthesis has enabled the scalable preparation of Li3PS4-based sulfide electrolytes with room-temperature ionic conductivities above 10−4 S cm−1 and favorable mechanical deformability that enhances solid–solid interfacial contact. Importantly, aliovalent ZnO substitution yields glass-ceramic Li3PS4 with significantly improved conductivity (1.12 × 10−3 S cm−1) and increased stability against both air and lithium metal, illustrating how structural distortion and defect engineering introduced by force-driven processing can be leveraged to optimize ion-transport pathways and long-term cycling reliability.85 Complementarily, Biswas et al.86 demonstrated that planetary milling of halloysite-rich kaolin shortened tubular morphologies by ∼44% within 1 h and ∼53% within 2 h, where dry milling produced nanofragments while wet milling preserved tubular structures but still tripled surface area. As highlighted in Fig. 4E, such modifications critically regulate phosphate adsorption and release, advancing kaolin as a sustainable slow-release fertilizer carrier.
Expanding further, mechanochemical strategies have enabled solvent-free C–C bond formation using Cu-complex-functionalized polyoxoniobates with high recyclability and gram-scale applicability,87 selective mechanosynthesis of quinone derivatives with yields up to 98% through frequency and additive control,88 and the sustainable conversion of coal-derived graphene oxide into turbostratic graphene sheets with improved purity and carbon content.70 Ball milling has also been applied to metal–organic framework synthesis, where Plant-Collins et al.89 reported optimal crystallinity and hierarchical porosity in MOF-505 within 80 minutes under reduced solvent input.
Mechanochemical synthesis of MOFs has been systematically established through neat grinding (NG), LAG, and ion- and liquid-assisted grinding (ILAG). James and Friščić90 highlighted that direct milling of metal salts with organic linkers enables framework formation via force-driven solid–solid reactions, while LAG alters reaction kinetics and phase outcomes by enhancing interfacial mobility. Subsequent ILAG studies demonstrated that inorganic salt additives further regulate nucleation and polymorphism during mechanochemical MOF formation.91Operando diffraction investigations later confirmed that these force-driven routes govern MOF crystallization under solvent-minimized conditions.92
Environmental applications benefit as well: ball-milled MoS2 exhibits enhanced peroxymonosulfate activation for antibiotic degradation due to defect engineering,93 while ultrafine biochar/Fe3O4 composites efficiently remove pharmaceutical contaminants. In the energy domain, mechanochemical synthesis underpins cathode, anode, and electrolyte development for lithium- and sodium-ion batteries, offering improved electrochemical stability and performance.94,95 Importantly, the sustainable production of ammonia under mild conditions via ball milling achieving yields of 82.5% vol. at 45 °C and 1 bar positions this platform as a compelling alternative to the Haber–Bosch process.96 Collectively, ball milling enables the synthesis of diverse inorganic nanoparticles, including metal oxides, sulfides, carbides, perovskites, and layered compounds. Compared with liquid-assisted mechanochemistry and conventional wet-chemical routes, ball-milled nanoparticles typically exhibit nanocrystalline cores, high lattice strain, and defect-rich or partially amorphous shells, imparting unique properties such as enhanced catalytic activity, accelerated ion transport, and superior interfacial reactivity.
Taken together, ball milling and grinding architectures transcend conventional solution-based approaches by coupling tunable impact forces, scalable design, and solvent-free conditions to activate multiscale pathways across molecular, crystalline, and hierarchical levels. By enabling sustainable nitrogen conversion, energy-relevant nanophase design, catalytic interface engineering, and environmental remediation, these mechanochemical strategies collectively exemplify how controlled impact dynamics create smart and functional interfaces, establishing ball milling as a cornerstone of mechanochemical architectonics.
3.2. Extrusion and high-pressure mechanochemistry
Extrusion-based mechanochemistry has emerged as a transformative platform for continuous and scalable solid-state synthesis, offering distinct advantages over batch milling in terms of reproducibility, process intensification, and industrial translation.5,97–99 Its efficiency is exemplified by ZIF-8 production, which achieved a space-time yield of 144 × 103 kg per m3 per day, far surpassing spray-cast and electrochemical routes.5 In extrusion, ZIF-8 is prepared by feeding basic zinc carbonate and 2-methylimidazole (Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ligand = 2
ligand = 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) as a dry mixture into a twin-screw extruder, where alternating conveying and kneading zones impose intense shear and compression, driving rapid solvent-free coordination between the components. Residence times of only seconds to minutes are sufficient to produce a crystalline extrudate, while controlled barrel heating (up to 200 °C) enables a melt-phase route in which the molten ligand accelerates diffusion and framework formation, yielding ZIF-8 at throughputs of 1–4 kg h−1. Beyond throughput, extrusion enhances porosity and surface area compared to conventional methods,97,98 while life-cycle assessment of ZIF-67 confirmed orders-of-magnitude lower environmental impact relative to solvothermal synthesis, underscoring its green and sustainable character.78 Twin-screw extrusion (TSE), originally developed for polymer and food processing, has been successfully adapted to mechanochemical organic synthesis, providing a robust alternative to batch methods.4,100 Pharmaceutical applications illustrate its power: direct amidation of esters eliminates the need for stoichiometric activating agents,101 and Bolt et al.102 achieved a 100-fold scale-up of this transformation by translating from batch ball milling to continuous extrusion.
3) as a dry mixture into a twin-screw extruder, where alternating conveying and kneading zones impose intense shear and compression, driving rapid solvent-free coordination between the components. Residence times of only seconds to minutes are sufficient to produce a crystalline extrudate, while controlled barrel heating (up to 200 °C) enables a melt-phase route in which the molten ligand accelerates diffusion and framework formation, yielding ZIF-8 at throughputs of 1–4 kg h−1. Beyond throughput, extrusion enhances porosity and surface area compared to conventional methods,97,98 while life-cycle assessment of ZIF-67 confirmed orders-of-magnitude lower environmental impact relative to solvothermal synthesis, underscoring its green and sustainable character.78 Twin-screw extrusion (TSE), originally developed for polymer and food processing, has been successfully adapted to mechanochemical organic synthesis, providing a robust alternative to batch methods.4,100 Pharmaceutical applications illustrate its power: direct amidation of esters eliminates the need for stoichiometric activating agents,101 and Bolt et al.102 achieved a 100-fold scale-up of this transformation by translating from batch ball milling to continuous extrusion.
This work further demonstrated solvent-minimized synthesis of 36 amides via TSE, with optimization of rheology, grinding auxiliaries, and residence time enabling long-term operation. A 7-hour run yielded ∼500 g (1.3 mol) of amide at 80% yield, and the dynamics of both small-scale and extended campaigns are vividly depicted in Fig. 5, confirming extrusion's scalability and process stability.102
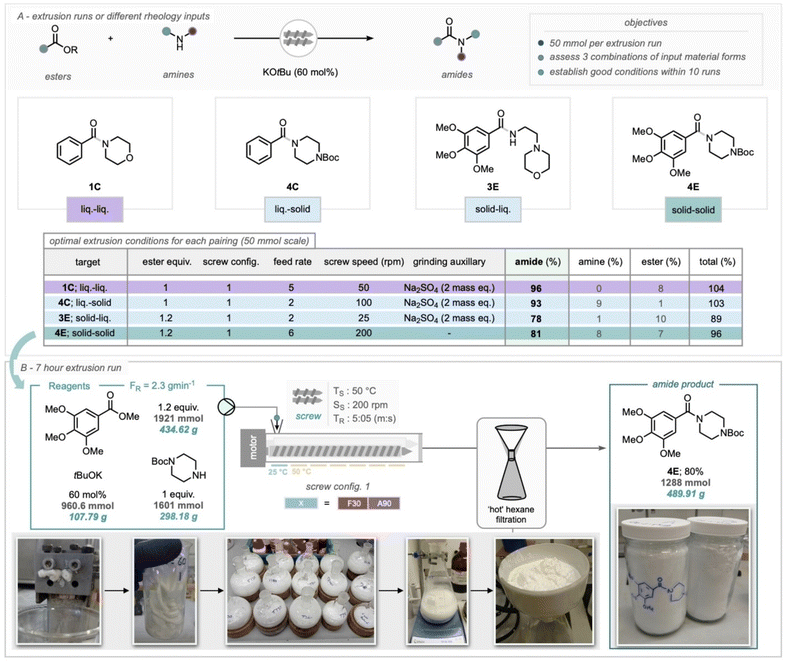 | ||
| Fig. 5 (A) Optimal extrusion runs of amides on a 50 mmol scale. (B) A 7-hour solvent-minimized continuous extrusion synthesis of an amide, with 30-minute batches of the extrudate collected to monitor performance over time (see SI for detailed breakdown). Reproduced with permission.102 Copyright 2024, wiley. | ||
Song et al.103 extended these principles through a liquid-assisted spiral gas–solid flow (LA-S-GSF) system, enabling rapid synthesis of HP-HKUST-1 at 96% yield within 11 minutes, achieving a space–time yield of 6.9 × 104 kg per m3 per day, significantly outperforming solvothermal methods. Similarly, mechanochemical cocrystallization has produced stable pharmaceutical solids, such as ligustrazine cocrystals (TMP–MG, TMP–MG–H2O, TMP–EG, TMP–PG), with reduced hygroscopicity and reversible solid-state interconversion, highlighting their sustainable pharmaceutical potential.104
Parallel developments extend into polymer and composite systems, where mechanochemical rearrangements under extrusion and high pressure preserve nanostructural porosity (>98%) and enable scalable solar water evaporation, producing 9 L per day with a 1 m2 prototype.105 Water-assisted gel-like extrusion of PVA achieved remarkable toughness (elongation at break 504.9%) and ultralow oxygen permeability after biaxial stretching,106 while incorporation of NiO nanofillers into PP/PE composites enhanced tensile strength, elongation, and thermal stability.107 Similarly, integration of caffeine-loaded UiO-66 capsules into PA6 and PLA composites via extrusion produced matrices with ∼2.5 wt% caffeine loading and significantly improved durability relative to surface post-treatments.108
Complementing extrusion, high-pressure mechanochemistry exerts profound influence on atomic arrangements in solids, driving unique chemical and physical transformations.109,110 A persistent challenge lies in controlling which mechanical strains induce specific nuclear distortions and corresponding electronic effects.111 Recent strategies aim to tune crystal flexibility, thereby directing mechanochemical reactivity toward selective stress-responsive pathways.112 Together, extrusion and high-pressure approaches open new frontiers for transitioning mechanochemical processes from laboratory to industry, particularly for continuous solid-state synthesis.113,114 By coupling throughput, structural control, and environmental compatibility, these methodologies serve as critical enablers of multiscale mechanochemical pathways that underpin the design of smart and functional inorganic interfaces.
3.3. Multimodal stimuli-coupled mechanochemistry
Smart interfaces in advanced inorganic and hybrid systems are defined as adaptive material–bio interfaces that integrate stimuli responsiveness, bio-recognition, and feedback-controlled behavior, enabling dynamic modulation of interfacial structure and reactivity under external cues.115 In this section, polymeric components are discussed only when they function as interfacial matrices or hybrid partners that directly enable the architectonic control and functional performance of inorganic nanomaterials under mechanochemical activation.Hybrid mechanochemistry, wherein mechanical forces are synergistically combined with external stimuli such as temperature, light, ultrasound, plasma, or electric fields has emerged as a transformative strategy for accessing reaction pathways inaccessible to conventional solid-state processes.116,117 These thermo-, sono-, photo-, and electro-mechanochemical platforms significantly broaden the chemical reactivity landscape, enabling both enhanced performance and novel functionality.117,118 Plasma-assisted milling, for example, introduces defects into MXenes, improving their electrochemical behavior, while strain engineering under mechanochemical conditions via biaxial compression or out-of-plane tension has been shown to boost lithium-ion diffusion kinetics.118,119 Hybrid protocols also facilitate in situ functionalization of MXenes, where stimuli-activated dopants or surface groups expand their utility in smart interfaces.120,121
To contextualize these differences, Table 1 provides a comparative overview of representative mechanochemical platforms in terms of energy input characteristics, scalability, and defect control. This mechanistic shift toward stimuli-activated and dynamically functionalized surfaces highlights a fundamental contrast with conventional inorganic interfaces, which respond to mechanical loading through passive pathways dominated by defect generation, microplasticity, or frictional dissipation. Traditional interfaces rely on irreversible deformation and defect-mediated reactivity characteristic of classical ball-milling or mechanically enhanced adhesion where mechanical force merely perturbs existing bonds. In contrast, smart interfaces incorporate force-responsive motifs such as mechanophores or reorganizable coordination environments that transduce mechanical, thermal, photonic, or acoustic inputs into reversible chemical rearrangements or controlled activation events. This transition from passive defect accumulation to programmable, stimuli-coupled mechanochemical behavior underpins the adaptive reactivity observed in modern thermo-, sono-, and photo-mechanochemical systems.122,123 The fusion of ball milling with sonochemistry or photochemistry unlocks novel hybrid nanomaterials with adaptive properties.43,124 Hwang et al.125 demonstrated a mechano–chemical coupling system by integrating phospholipid copolymer vesicles within alginate hydrogels, enabling stress-induced ethylene glycol tetraacetic acid (EGTA) release at only 33–55 kPa and achieving controlled degradation. On a quantum scale, Wang et al.126 and Mittal et al.127 used the constrained geometries simulate external force (CoGEF) method, where force application narrowed HOMO–LUMO gaps and lowered reaction barriers, highlighting the predictive power of mechanochemical simulations. Similarly, Ghose et al.128 and Zhao et al.129 illustrated how external solvation and ultrasound-induced piezoelectricity, respectively, modulate mechanical rupture thresholds and reactive oxygen species (ROS) generation in Zn–bis-terpyridine and Mn–ZnO hybrids. Remarkably, Qu et al.130 reported that ball milling enabled MXene/PAbz hybridization via electrostatic and hydrogen bonding interactions, drastically reducing peak heat release rate (−69.57%) and total heat release (−54.38%), showcasing the fire-retardant promise of hybrid mechanochemistry.
| Platform | Energy input | Scalability | Defect control |
|---|---|---|---|
| Ball milling | High local energy, but heterogeneous | Moderate (batch-limited) | High defect density, limited precision |
| Extrusion | Efficient continuous energy input | High (industrial-scale) | Moderate, more uniform |
| Hybrid systems | Variable, stimulus-dependent | Moderate | Tunable and interface-specific |
Inorganic–organic hybrids can also be rapidly synthesized through silane-functionalized ball milling routes with over 90% efficiency, as reported by Amrute et al.8 The solid-state synthesis of D4SB crystals via thermally responsive grinding,131 and reversible emission switching, further underscores force–stimuli cooperation in tuning optoelectronic features. Structurally responsive systems such as MOFs display dynamic mechanochemical behaviors including bond rearrangement,132 phase transitions,133 and amorphization134 that depend on framework topology,135 defect density, and pore distribution.136 Real-time tools like high-pressure crystallography and nanoindentation elucidate these force–structure couplings,137,138 while milling experiments reveal rapid coordination bond cleavage and energy storage modulation.139 Overall, hybrid mechanochemical systems not only offer a versatile toolbox for manipulating structure–function relationships at multiple scales but also underpin the rational design of smart and responsive interfaces for advanced inorganic materials.
4. Mechanistic insights
4.1. Multiscale reaction pathways
Mechanochemical transformations operate through inherently multiscale pathways in which atomic, interfacial, and bulk processes converge to create smart and functional interfaces. At the atomic level, mechanical input perturbs orbital overlaps, destabilizes crystal lattices, and initiates bond cleavage, generating radicals and defects that substantially lower activation barriers and activate non-thermal routes.16,140 Ball milling exemplifies these dynamics, where localized high-energy collisions induce solid-state reactions by breaking covalent and redox bonds, decomposing surface contaminants into small molecules, and triggering vibrational excitation at transient hot spots.141–145 Such events coalesce into a three-step progression comprising atomic-scale mixing, bond-level reorganization, and the nucleation of nanocrystalline or microcrystalline products.4 Repeated deformation and fracture further intensify defect accumulation at grain boundaries, enabling radical generation and product nucleation under ambient conditions.2,39 Yang et al.146 extended this concept by showing that press-and-rotate mechanical inputs can reconfigure self-assembled inorganic nanorod superlattices into chiral architectures (Fig. 6A), illustrating how macroscale grinding induces nanoscale symmetry breaking with emergent optical properties.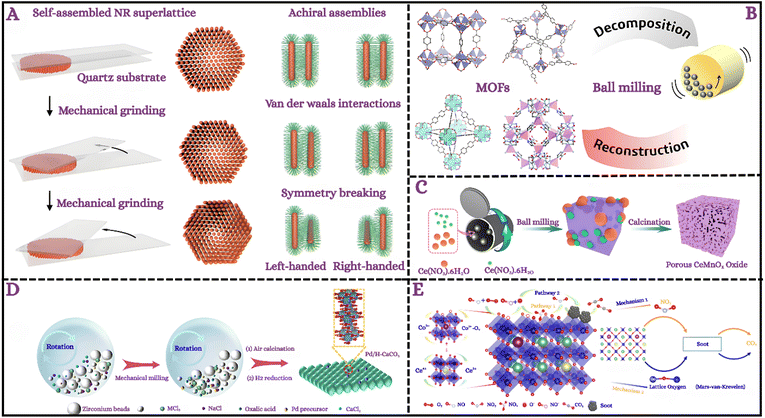 | ||
| Fig. 6 (A) Schematic illustration of chirality generation in self-assembled NR superlattices via a press-and-rotate mechanical force. Reproduced with permission.146 Copyright 2022, Nature. (B) Schematic of MOF transformation via ball milling, showing framework decomposition followed by structural reconstruction. Reproduced with permission.156 Copyright 2021, American chemical society. (C) Schematic illustration of mechanochemical nanocasting synthesis of porous CeMnOx catalysts. Reproduced with permission.160 Copyright 2019, American chemical society. (D) Process of the preparation of MCO3.161 Copyright 2023, American chemical society. (E) Schematic of La (Ce,K)CoO3 lattice and redox cycles, highlighting soot oxidation via Langmuir–Hinshelwood and Mars–van Krevelen mechanisms. Reproduced with permission.162 Copyright 2022, American chemical society. | ||
Progressive energy input drives crystalline–amorphous transitions, where bulk structural transformations arise from bond disruption, densification, and framework collapse.2,147–149 Localized shell formation provides both opportunities and challenges: for instance, neat milling generates nanometer-thick amorphous shells on ZnO surfaces, enhancing interfacial reactivity but also limiting further conversion by encapsulating unreacted cores.150,151 Liquid-assisted grinding modulates this balance, as demonstrated for ZnO systems where hydrated intermediates gradually reorganize into crystalline frameworks with decreasing water content.152 Under high pressure, Zn-imidazolate frameworks undergo selective bond cleavage and structural reorganization, collapsing pore networks while preserving short-range order.149,153 Such cases underscore that mechanochemical reactivity does not simply replicate thermal pathways; rather, it introduces novel transformation routes linking defect accumulation, metastable polymorph formation, and bulk structural evolution.2,154
Mechanochemistry also provides unique avenues for coupling metal oxides and organic linkers into hybrid nanostructures. For example, solid-state reactions between ZnO and Hmim yield crystalline ZIF-8 shells encapsulating Pd nanoparticles,155 while iron oxide precursors undergo stepwise transformations from maghemite to MOF-integrated composites via neat and liquid-assisted grinding.19 Similar pathways enable metal–organic recoordination and even reconstruction of decomposed frameworks: Lee et al.156 showed that ball-milling restores crystallinity and porosity in degraded MOFs such as MOF-5, MOF-177, UiO-67, and ZIF-65 through cycles of defect generation and interfacial recoordination (Fig. 6B). These insights highlight the role of mechanical force in bridging atomic-scale bond cleavage with bulk-scale structural recovery.
Beyond oxides and MOFs, mechanochemical methods have unlocked reaction channels unattainable in solution. The generation of Grignard reagents from fluoronaphthalenes,157 the activation of benzene through Mg(I) dimers,69 and the stabilization of unusual η3-allyl–Mg coordination modes158 exemplify bond activation pathways arising directly from mechanical inputs. Likewise, lignin depolymerization under milling produces stable phenoxy radicals that reduce metal ions and initiate unconventional bond transformations.16 At higher scales, metal alloys and nitrides undergo progressive grain refinement, defect accumulation, and hot-spot-assisted diffusion, as shown for Fe–Cr–Si alloys and TiN formation.6,159 These cases emphasize that interfacial charge gradients, radical fluxes, and nanoscale strain collectively govern reactivity in ways decoupled from conventional thermal dynamics.39,97,142
Hybrid mechanochemistry further expands this framework by integrating force with external stimuli, enabling hierarchical transformations across multiple scales. Cheng et al.160 reported a nanocasting route in which lattice-level Mn incorporation into CeO2 generated mesostructured CeMnOx catalysts, achieving nearly complete NOx conversion at 150 °C and highlighting the solvent-free scalability of this process (Fig. 6C). Zhang et al.161 demonstrated that salt-assisted milling engineers hierarchical porosity in carbonates, where lattice perturbations, NaCl templating, and pore evolution converge to confine Pd nanoparticles within CaCO3 matrices (Fig. 6D). Yu et al.162 advanced the concept by designing alkali/cerium co-modified La–Co perovskites that exploit coupled atomic redox cycles, interfacial oxygen vacancy dynamics, and bulk lattice oxygen participation. In situ DRIFTS and DFT revealed synergistic Langmuir–Hinshelwood and Mars–van Krevelen mechanisms, with (Fig. 6E) illustrating how bifunctional effects in soot combustion catalysis arise from coordinated atomic, interfacial, and bulk processes.
These multiscale pathways reveal that mechanochemistry does not merely replicate thermal dynamics but opens unconventional reaction channels. However, the lack of a comprehensive quantitative framework limits predictive control over outcomes. The critical challenge remains to bridge atomic and interfacial events with bulk transformations through integrated modeling for rational design of smart interfaces.
4.2. Computational modeling and AI integration
Computational modeling has become indispensable for unraveling mechanochemical pathways, yet the diversity and complexity of applied forces render atomistic simulations inherently challenging. Classical frameworks such as Bell's model have long provided first approximations to reaction energy modifications,163 while more advanced strategies such as the CoGEF method quantify force-dependent energy variations by relaxing perturbed systems between pulling points.164 These efforts underscore that truncated model risk overlooking stereochemical contributions, since computational predictions of activation lengths for E- and Z-alkene isomers (1.65 Å and 1.24 Å) closely match experimental values (1.67 ± 0.05 and 1.20 ± 0.05 Å), affirming the cusp model's utility in describing reaction potential energy surfaces despite qualitative assumptions.164 Recent developments, such as steepest-descent pathways (SDPs), further refine predictive capabilities by offering efficient yet experimentally validated approximations of mechanochemical reaction rates.165 Bunno et al.166 elegantly demonstrated this principle by employing DFT within the push–pull protonated atomic distance (PAD) framework to predict proton conductivity in acid–base hybrids. As shown in Fig. 7A, incorporation of TiO2 shortened PAD values, reorganized hydroxyl groups, and promoted proton hopping cascades, thereby guiding the rational design of proton-conducting inorganic–organic hybrids. Parallel work by Wu and Wang167 extended this paradigm to hybrid organic–inorganic perovskites (HOIPs), where a progressive machine learning framework integrated enriched input databases with DFT validation. As presented in Fig. 7B, their models spanned diverse A-site cations, B-site metals, and X-site anions, with band-gap distributions confirming robust predictive accuracy across metals, semiconductors, and insulators.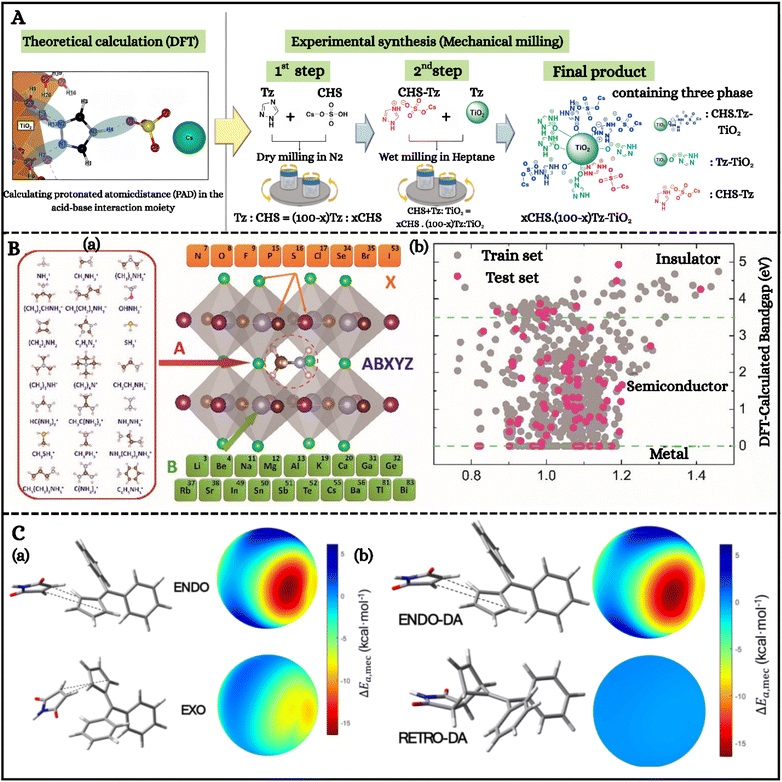 | ||
| Fig. 7 (A) Schematic representation of the procedure for developing proton-conducting inorganic–organic hybrids, guided by computational modeling. Reproduced with permission.166 Copyright 2023, American chemical society. (B) (a) Dataset construction for HOIP prediction, comprising 21 monovalent organic molecular cations (A-site), 19 metallic cations, and 9 representative anions across the periodic table. (b) Visualization of training (gray dots) and test (red dots) data based on tolerance factor and DFT-calculated band gap (PBE), illustrating the coverage of metals, semiconductors, and insulators within the dataset. Reproduced with permission.167 Copyright 2020, American chemical society. (C) (a) (Top) Endo adduct and its spherical representation of ΔEa,mec as a function of wall-type force directions. Some impact directions reduce the activation energy by up to −16.5 kcal mol−1, with an average 〈ΔEa,mec〉 of −3.88 kcal mol−1. (Bottom) Exo case, showing a maximum reduction of −8.9 kcal mol−1 and an average 〈ΔEa,mec〉 of −2.89 kcal mol−1. (C) (b) Comparison of activation energy variations for the endo Diels–Alder reaction (top) and the retro-DA reaction from the endo product (bottom). The mechanical effect on the retro-DA reaction is nearly negligible, as reflected in the ΔEa,mec scale. (C and D) Reproduced with permission.58 Copyright 2025, American chemical society. | ||
Complementary to these approaches, quantum chemistry calculations have revealed molecular deformation during activation, with the extreme-pressure polarizable continuum model tracking cavity volume changes under hydrostatic stress,168 while density functional theory (DFT) demonstrated how mechanical stress deforms C–C bonds, enabling thermally forbidden electrocyclic reactions.169 At the mesoscale, computational studies show that forces applied orthogonally to the reaction coordinate can significantly alter transition-state curvature,170 though no current model fully quantifies the mechanical work exerted during ball milling.
Nonetheless, recent work by Pladevall et al.171 has shown that standard DFT combined with continuum solvation and microkinetic modeling can quantitatively describe ball-milling reactions. By treating mechanochemical conditions as solution-like reactions with modified effective concentrations and dielectric constants, they reproduced experimentally observed reaction times for Diels–Alder cycloadditions and N-sulfonylguanidine formation under ball milling, and concluded that, at least for these benchmark systems, the mechanisms under milling and in solution are essentially identical and amenable to DFT-based analysis. Moreover, dispersion-corrected DFT has been directly applied to ball-milling conditions themselves, as demonstrated by Rojas-Chávez et al.,172 who computed ligand–surface interactions, surface-energy shifts, and facet stabilization in PbTe nanoparticles during high-energy milling.
Molecular dynamics further illuminate surface and interfacial pathways, exemplified by alkyl thiolate interfaces on coinage metals, where shear generates distinct metal-containing species and governs stress-driven surface-to-bulk transport.173 Fusaro et al.174 advanced this direction by developing a DFT-based framework incorporating electrostatics and solvent effects, successfully predicting ∼20 kcal mol−1 binding energies for biomolecule–gold interactions, consistent with experiments. Importantly, such mechanistic insights extend directly to nanoscale biointerfaces, as demonstrated by Triatmaja et al.,175 whose DFT-optimized docking models clarified the antibacterial mechanism of GO/ZnO/eugenol composites, revealing strong binding (−11.65 kcal mol−1) to DNA gyrase and effective ATP blockade.
To capture milling dynamics more holistically, discrete element method (DEM) and structural phase-field-crystal (XPFC) models have been employed. By incorporating ballistic terms, XPFC simulations reproduced nanoparticle nucleation and growth trends under room-temperature milling, with ligand chain length modulating the free energy landscape and predicting reduced particle sizes for longer ligands.176 Further refinement of reaction selectivity under mechanical stress has been achieved through mechanochemical algorithms. For example, diphenylfulvene/maleimide systems under isotropic wall-type forces showed preferential endo-pathway stabilization, with 〈ΔEa,mec〉 reductions of −3.88 kcal mol−1 compared to −2.89 kcal mol−1 for the exo case, offering a microscopic rationale for experimentally observed endo selectivity in mechanochemical Diels–Alder reactions (Fig. 7C(a)).58 Strikingly, retro-Diels–Alder processes remained unaltered, confirming that force biases are pathway-specific (Fig. 7C(b)). Collectively, these computational strategies from CoGEF and SDPs to DEM and XPFC demonstrate how stress can reshape potential energy landscapes, drive bond rupture (C–C, Si–O, S–Au), and rationalize transformations inaccessible by purely thermal means.
The integration of artificial intelligence (AI) has further accelerated progress, enabling optimization of mechanochemical conditions and predictive modeling with unprecedented precision. Machine learning potentials now reconcile the accuracy of quantum methods with the efficiency of classical simulations, resolving long-standing trade-offs.177 Algorithms such as XGBoost, coupled with dimensionality reduction, achieve up to 80% predictive accuracy in mechanochemical cocrystallization, outperforming conventional approaches and highlighting electrostatics and structural compatibility as AI-derived guidelines for rational solvent-free design.178 Artificial neural networks (ANNs) extend this capability, with models trained on process parameters and formulations achieving R2 > 0.99 for predicting pharmaceutical cocrystallization during ball milling. Such predictive robustness motivates hybrid models that fuse mechanistic frameworks with AI, enhancing accuracy in simulating complex multi-component systems.179 Beyond organics, computational chemistry paired with AI has successfully predicted defect energetics, binding affinities, and electronic behaviors in inorganic nanomaterials ranging from metal oxides to sulfur-based systems,159,180 underscoring cross-material generality. Together, computational modeling and AI not only decode multiscale mechanistic pathways but also enable rational control of smart functional interfaces, establishing a predictive foundation for the design of next-generation inorganic nanomaterials.
5. Unlocking smart and functional interfaces
5.1. Catalysis and energy interfaces
Mechanochemical architectonics offers a revolutionary approach to the design of catalytic and energy interfaces, wherein structural evolution-characterized by flaws, porosity, and dopant distribution-occurs directly under mechanical force. Mechanochemical processing, unlike traditional synthesis, works in solvent-free or low-solvent environments. This lets you quickly get to nonequilibrium states with high defect concentrations, nanocrystallinity, and close contact between components-things that are frequently hard to get using traditional methods.181 This force-driven environment fosters oxygen migration, accelerates redox cycling, and enhances selective adsorption, leading to the development of “active microreactors” on the material surface that improve catalytic responsiveness. Crucially, mechanochemical routes allow direct tuning of active site structures-vacancy-rich surfaces, hierarchical pores, and atomically dispersed dopants. This means that solvents, templates, or high-temperature procedures are not needed, which is in line with the ideas of green chemistry and scalable synthesis.23,182Siniard et al.183 indicated that incorporating CuCoFeNiMnOx into a CeO2 lattice using ultrasound-driven lattice engineering generates numerous oxygen vacancies, well-dispersed cations, and enhanced Ce3+/Cu+ redox centers. These characteristics facilitated ultralow-temperature CO oxidation (T50 = 100 °C, T100 = 150 °C), exceeding traditional dense HEOs. Fig. 8A demonstrates that the catalytic cycle involves lattice oxygen migration, vacancy formation, and fast oxygen replenishment, exemplifying a Mars–van Krevelen mechanism that imparts remarkable activity and stability to the HEO–CeO2 interface. Similarly, Zhang et al.161 devised a salt-assisted mechanochemical method to synthesize porous CaCO3, SrCO3, and BaCO3 in few minutes, attaining unprecedented surface areas (up to 172 m2 g−1). The Pd-loaded hierarchical CaCO3 catalyst achieved nearly total nitrobenzene hydrogenation (≥99% yield, TON = 1.3 × 104 h−1) and shown exceptional reusability throughout 11 cycles. Fig. 8B illustrates that NaCl templating during milling facilitates pore development, resulting in stable, high-surface-area interfaces conducive to effective catalysis. Furthermore, Xiao et al.184 developed an innovative method for extracting gold from anode slime without the use of solvents. This process involves KMnO4 and NH4Cl generating reactive chlorine species during milling, which oxidize metallic Au into soluble AuCl3. This approach was highly selective and yielded numerous results without employing toxic cyanide or solvent-intensive methods. This illustrates how mechanochemistry may benefit the environment through resource reutilization. Fig. 8C illustrates that the mechanical activation of KMnO4 and NH4Cl in conjunction results in the release of chlorine, facilitating the direct conversion of gold at ambient temperature. Likewise, Zhan et al.185 described a template-free mechanochemical method for synthesizing mesoporous CuOx–CeO2, with surface areas of up to 122 m2 g−1 with interconnected pores measuring 2–5 nm. The sponge-like structure maintained atomic-scale Cu2+ dispersion and improved Cu2+/Cu+–Ce3+/Ce4+ redox cycling, facilitating complete CO oxidation at around 98 °C, as illustrated in (Fig. 8D). Yang et al.186 developed a solvent-free mechanochemical approach for synthesizing Fe3O4 nanoparticles, distinguished by many oxygen vacancies and Fe2+/Fe3+ redox centers, which promote improved chemoselective reduction of nitroarenes. This catalyst, produced using ball milling and characterized by numerous flaws, exhibited over 99% selectivity, commendable turnover efficiency, and excellent reusability after 10 cycles. The engineered contact inhibited sintering, preserved the crystals, and facilitated electron mobility among them. All of them are crucial for energy-related catalytic applications. This study demonstrates how mechanochemistry can produce catalytic surfaces that are both physically robust and functionally superior.
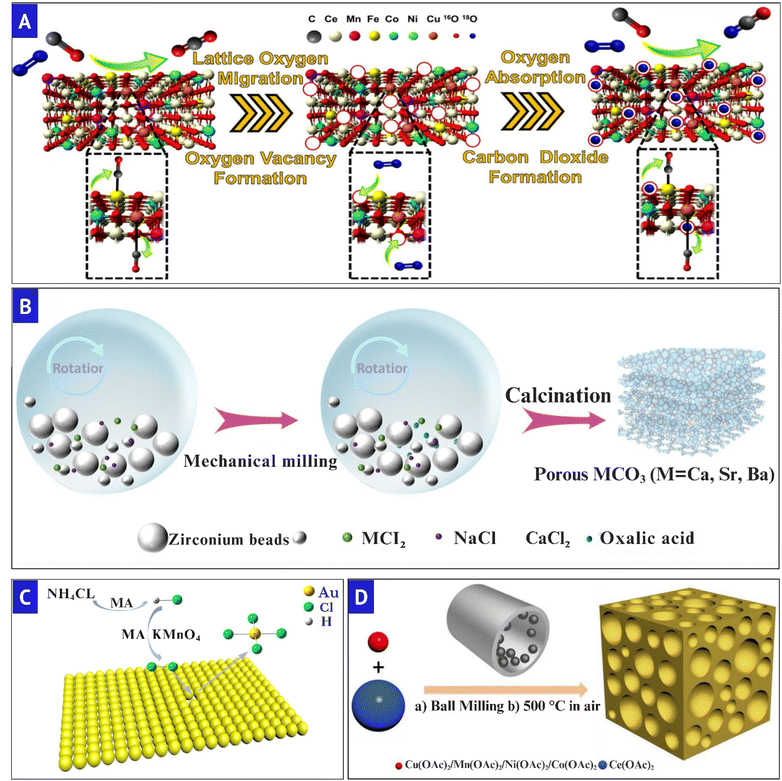 | ||
| Fig. 8 (A) Illustration of the synergistic HEO-assisted Mars–van-Krevelen mechanism for 0.25-HEO–CeO2. Reduced with permission.183 Copyright 2024, American chemical society. (B) Process of the preparation of MCO3via salt-assisted mechanochemical milling and calcination. Reduced with permission.161 Copyright 2023, American chemical society. (C) Mechanism of gold conversion using KMnO4–NH4Cl through the mechanochemistry approach (MA). Reduced with permission.184 Copyright 2019, American chemical society. (D) Illustration of mechanochemical synthesis of mesoporous CuOx–CeO2 and transition-metal-doped ceria catalysts. Reduced with permission.185 Copyright 2017, American Chemical Society. | ||
5.2. Biomedical interfaces
Mechanochemical architectonics is gaining prominence as a method for constructing biomedical interfaces, where stability, biocompatibility, and multifunctionality are paramount. Conventional synthetic methods sometimes encounter issues such as limited dispersibility, cytotoxicity, or loss of functionality under physiological conditions, complicating the application of nanomaterials in clinical environments. By contrast, mechanochemistry enables direct control over surface functionalization and hybridization, producing defect-rich, coated, or host–guest-modified nanostructures without the need for harsh solvents or complex templating.187 Beyond improving safety profiles, mechanochemical engineering also unlocks smart functionalities at the bio-nano interface, ranging from enhanced antioxidant performance to controlled release and protective exoskeletons. Hybrid coatings and supramolecular assemblies formed via mechanical activation can safeguard sensitive biomolecules and impart them with innovative abiotic characteristics such as magnetism, fluorescence, or UV resistance. These attributes are essential for bioimaging, biosensing, and therapeutic systems. These advancements demonstrate that mechanochemistry transcends conventional boundaries of nanoparticle coatings, extending into living cell encapsulation, drug delivery, and biomedical protective technologies.2,188Here, biomedical interfaces are approached from an inorganic-centered perspective, where organic and supramolecular elements are introduced as functional enablers to tailor surface chemistry, protection, and biointeractions of nanomaterials rather than as standalone material systems. Krupiński et al.189 introduced a mechanochemical strategy to produce sub-10 nm ZnO nanocrystals stabilized with urea ligands, demonstrating how surface chemistry can be tuned during synthesis to achieve stable organic–inorganic interfaces. A subsequent modification of β-cyclodextrin (β-CD) conferred water solubility and enhanced bioprocessability, essential conditions for the biomedical application of ZnO nanoparticles. Fig. 9A demonstrates the sequential process-from urea precursors to ZnO–urea nanocrystals and ultimately β-CD functionalization-demonstrating how mechanochemistry facilitates the synthesis of nanostructures specifically designed for biomedical interfaces, where dispersibility and compatibility are crucial. Similarly, Ma et al.190 engineered solid SiO2-sealed mesoporous silica nanoparticles to co-encapsulate TiO2 and the organic UV filter DHHB, attaining broad-spectrum protection while inhibiting ROS release and skin penetration. The coating method demonstrated stability and safety in sunscreen applications, as illustrated in Fig. 9B. Likewise, Mazaheri et al.191 illustrated a solvent-free mechanochemical method for encapsulating urea within metal–phenolic networks (MPNs), wherein tannic acid coordinated with Ti4+ during grinding to create stable hybrid matrices. As illustrated in (Fig. 9C), the resultant coatings demonstrated adjustable stability and regulated release, underscoring how mechanochemistry facilitates biocompatible and protective interfaces, akin to β-cyclodextrin-modified ZnO nanocrystals and silica-encapsulated UV filter systems.
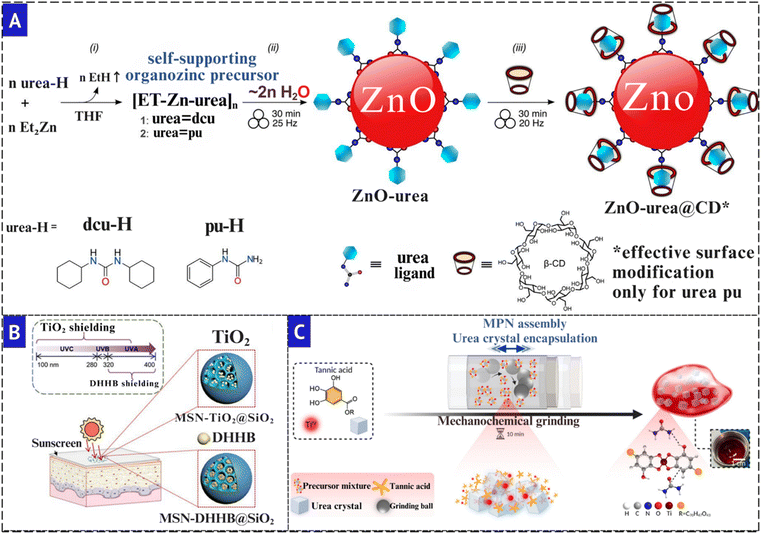 | ||
| Fig. 9 (A) Mechanochemical synthesis of ZnO nanocrystals from ethylzinc–ureate precursors and postsynthetic β cyclodextrin (β-CD) modification for water solubility. Reduced with permission.189 Copyright 2021, American chemical society. (B) Schematic structures and advantages of solid SiO2-sealed filter-entrapped MSNs (MSN-TiO2@SiO2 and MSN-DHHB@SiO2) and their UV protection mechanism. Reduced with permission.190 Copyright 2023, American chemical society. (C) Schematic of the mechanochemical, coordination-driven assembly of MPNs for urea crystal encapsulation. Reduced with permission.191 Copyright 2023, American chemical society. | ||
To ensure consistency with the catalysis and energy sections, this section now emphasizes direct mechanism–function relationships at the bio–nano interface. In particular, mechanochemically induced surface defects, ligand coordination modes, and hybrid architectures are explicitly correlated with biological performance metrics such as biocompatibility, oxidative stress modulation, and cellular uptake behavior. A comparative overview of mechanochemical and conventional synthesis routes is provided in Table 2.
| Aspect | Mechanochemical synthesis | Conventional synthesis | Biological mechanism–function correlation | References |
|---|---|---|---|---|
| Surface structure | Defect-rich, force-activated interfaces with tunable coordination | Thermodynamically relaxed surfaces | Controlled defects reduce cytotoxicity | 189 |
| ROS behavior | Defect passivation and solid-state ligand binding suppress ROS | Unpassivated surfaces promote ROS formation | Lower oxidative stress improves cell compatibility | 192 |
| Surface chemistry | Strong solvent-free ligand anchoring | Weak post-synthetic coatings | Enhanced dispersion and stability | 193 |
| Colloidal stability | Clean interfaces minimize aggregation | Residual solvents increase agglomeration | Reduced immune activation | 97 and 194 |
| Biocompatibility & hemocompatibility | Cleaner surfaces (no solvents), strong ligand anchoring → higher compatibility | Residual solvents, surfactants → potential cytotoxicity | Mechanochemical coatings significantly improve biocompatibility and reduce cellular stress | 195–197 |
| Drug encapsulation & release profiles | Solid-state assembly of metal–phenolic networks (MPNs) yield sustained release | Sol–gel and emulsion routes often show burst release | Mechanochemical MPNs provide controlled and stable release profiles | 191 |
The clinical relevance depends on a clearer understanding of their in vivo behavior, safety profile, and regulatory positioning. Recent studies demonstrate that solvent-free architectures, including metal–phenolic networks and ligand-passivated oxide nanostructures, display enhanced serum stability, reduced aggregation, and predictable degradation pathways, resulting in improved biodistribution and biocompatibility in vivo.198,199 The stability and safety attributes correspond closely with the current expectations of the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA), especially for the reduction of organic solvent residues, impurity control, and reproducibility in nanomaterial production.200,201 Mechanochemistry, being a solvent-free and energy-efficient process, automatically meets numerous regulatory standards, making it a compelling option for future nanomedical production pipelines.202
Beyond conventional nanocarriers, mechanochemistry is accelerating the emergence of drug–nanomaterial composite strategies that hold significant value for pharmaceutical and clinical translation. Mechanochemical co-crystallization has enabled the formation of drug–excipient pharmaceutical co-crystals that improve solubility, dissolution kinetics, and physicochemical stability.203–205 Moreover, solid-state amorphization via high-energy milling generates amorphous drug phases that exhibit improved bioavailability and regulated release characteristics, thereby obviating the necessity for organic solvents or high temperatures.206 Collectively, these advancements underscore the capacity of mechanochemistry to produce biocompatible, stable, and therapeutically pertinent solid-state drug–nanomaterial hybrids, hence enhancing the translational potential of the mechanochemical platforms outlined in this section.
5.3 Electronics and sensing interfaces
Mechanochemical strategies are increasingly redefining the design of electronic and sensing materials by enabling solvent-free, force-driven construction of functional interfaces with atom-level precision. These technologies provide sustainable, scalable approaches to develop heterojunctions, doped frameworks, and responsive structures that are meticulously optimized for enhanced charge transfer, selective analyte interaction, and signal transduction. Mechanochemically synthesized devices have demonstrated potential in the creation of self-powered gas sensors, vibrational energy harvesters, and chemiresistive and luminous detectors, which are crucial for applications spanning environmental monitoring to smart wearables. As the demand for miniaturized, flexible, and intelligent sensing modalities grows, mechanochemical interface engineering stands out as a transformative platform capable of unlocking next-generation transduction technologies.116,207,208Yang et al.209 devised a more environmentally friendly mechanochemical method for producing CoAl2O4 composite pigments through the ball-milling of sea sand, CaCO3, and metal nitrates, succeeded by calcination. Fig. 10A illustrates that the CoAl2O4 nanoparticles were affixed to quartz surfaces, augmenting blue-light reflectance (∼430 nm) via interface-induced scattering. This sustainable technique diminished cobalt use while attaining superior optical stability, indicating prospective applications in colorimetric sensors, NIR-reflective coatings, and functional inks. In another study by Sarfudeen et al.210 engineered a mechanochemically manufactured triboelectric nanogenerator (TENG) utilizing ZTF-8 (a zeolitic tetrazolate framework) combined with carbon, resulting in a flexible, layered interface for energy harvesting and sensing. Fig. 10B illustrates that the TENG incorporates layers of PTFE, aluminum, ZTF-8, and carbon, which cyclically accumulate and discharge surface charge during mechanical contact and separation. This arrangement facilitates alternating electrostatic equilibrium and electron flow, yielding an output of 65 V and 4.2 µA, demonstrating exceptional responsiveness to human motion. Qin et al.211 fabricated Se-doped MoS2/ZnO heterostructures by a mechanochemical approach, incorporating both substitutional and interstitial selenium atoms to improve NH3 detection at ambient temperature. The dual-site doping enhanced charge transfer, surface reactivity, and heterojunction integrity, yielding a sixfold increase in sensitivity, a detection limit of 25 ppb, and expedited reaction and recovery times. Hidalgo-Rosa et al.212 employed multiconfigurational ab initio calculations to elucidate the luminescence quenching mechanism in nitroaromatic-responsive lanthanide MOFs, demonstrating that strong host–guest electronic coupling perturbs the antenna-to-metal energy-transfer pathway. Theoretical analysis revealed that π–π interactions and analyte-induced destabilization of the ligand triplet state suppress Tb3+ sensitization, resulting in efficient turn-off luminescence behavior and high sensing selectivity toward nitro compounds.
 | ||
| Fig. 10 (A) Schematic diagram of the preparation of CoAl2O4 composite pigments via a CaCO3-assisted mechanochemistry method followed by calcination. Reduced with permission.209 Copyright 2022, American chemical society. (B) (a) Schematic diagram of the proposed ZTF-TENG device architecture, in which ZTF-8 acts as the triboelectric positive material and PTFE as the negative counterpart, layered with carbon and aluminum electrodes for flexible operation. (b) Schematic of the contact–separation mechanism, illustrating charge generation and electron flow during device operation. Reduced with permission.210 Copyright 2024, American chemical society. | ||
6. Challenges and future perspectives
6.1. Current limitations
Mechanochemistry, though eco-friendly and capable of reducing solvent use, still faces critical challenges that constrain its broader application in designing smart and functional interfaces. Nanostructured oxides obtained through mechanochemical routes often contain a high fraction of structurally disordered interfaces, which complicates the separation of bulk and surface contributions,213 while diffraction techniques lose much of their resolving power in such nanoscale and disordered systems, making atomic-scale resolution particularly difficult. Mechanically induced phase transformations strongly depend on milling dynamics, rendering reproducibility and kinetic control difficult,151 and subsequent thermal relaxation alters functional properties, thereby compromising reliable performance in practical applications. Achieving consistent and reproducible reactions is further hindered by variable parameters such as temperature, pressure, and milling intensity,144 while localized heating at impact points often triggers unintended side reactions or precursor decomposition.214 Reports on nanoparticle synthesis must also be interpreted cautiously, as XRD frequently underestimates particle sizes compared to catalytically relevant surface features.The inherent simplicity of mechanochemistry masks persistent issues: processes often proceed on a trial-and-error basis due to the absence of a comprehensive predictive framework,9 and current models including hot spot and magma–plasma theories remain qualitative, failing to integrate mechanical energy into thermodynamic formalisms. Existing nanoparticle growth models from dilute solutions cannot be directly applied to solid-state systems,176 and no current theory adequately describes the spatiotemporal evolution of nucleation or nanocrystalline precipitates. Consequently, rational design remains limited, and reaction selectivity is frequently undermined by unintended side reactions.215 These challenges are compounded by the restricted scope of mechanochemical transformations, as reactions requiring precise temperatures, pressures, or solvent participation often fall beyond the reach of mechanical activation.144,215
Practical synthesis also faces substantial barriers. Multistep and solvent-intensive methods raise toxicity concerns and limit suitability for biomedical applications,19,216 while poor control over size, shape, and surface chemistry continues to limit reproducibility and targeted functionality.217 Magnetic nanoparticle synthesis exemplifies these challenges, as hazardous multistep methods, difficulties in reducing poorly soluble oxides, aggregation, and reduced Curie temperatures compromise reproducibility and nanoscale stability.218 Similarly, AgNP synthesis suffers from agglomeration, contamination, and poor stability, reducing antimicrobial activity.29 For magnetic framework composites, limited studies and scarce biomedical evaluations further restrict comprehensive assessment of their potential.19 Strontium-based semiconductors also remain hindered by low precursor solubility and stable adduct formation, which compromise their targeted electronic properties.69 Highly soluble oxides such as B2O3 require sublimation or physical separation techniques due to poor by-product removal using conventional washing.219 More broadly, end products are frequently contaminated by milling media, jars, or container abrasion,181,220 which, alongside difficulties in tuning porosity, morphology, and uniformity, restricts the optimization of functional interfaces.
From a processing perspective, power ball milling has been widely employed for iron oxide synthesis,221 yet limitations in particle size control, post-processing, and contamination remain acute. Even in successful demonstrations, such as magnetite nanoparticle synthesis via prolonged milling,222 metallic impurities persisted. Knowledge on salt precursor reduction into noble metal nanoparticles remains scarce,33 and diffusion-limited mass transfer further constrains reproducibility. Moreover, mechanochemical grafting often leads to reduced surface area, pore diameter, and pore volume, while increasing silane loadings diminish silylation efficiency and limit uniform functionalization.8 Although polymer mechanochemistry holds promise, its extension to regulate heterogeneous catalysis a domain critical for industrial relevance has not yet been realized.223
Scaling laboratory processes to industrial production introduces further complexities. High-energy milling requires extensive optimization to balance quality, efficiency, and safety, while uncontrolled variables such as dust generation, noise, and equipment hazards demand stringent safety protocols.144 The energy-intensive nature of high mechanical forces also raises concerns over sustainability, operational costs, and environmental impact when reliant on non-renewable sources.215 Although mechanochemistry has advanced industrial use in pigmentation,224 catalysis,225 and wastewater treatment,226 its broader adoption remains limited. Moreover, systematic evaluation using sustainability metrics such as atom economy and waste reduction remains underutilized, with only a few applications in MOFs and nanoparticles.35 From a pharmaceutical manufacturing perspective, GMP compatibility of mechanochemical processes is platform-dependent. Continuous technologies, particularly twin-screw extrusion, are inherently more GMP-aligned due to enclosed operation, improved process control, and established pharmaceutical validation, whereas batch ball milling remains challenged by contamination risks and batch-to-batch variability, motivating a shift toward inert, enclosed, and continuous or hybrid reactors.
Taken together, these limitations underscore the dual challenge mechanochemistry faces: fundamental gaps in understanding multiscale reaction pathways227 and practical obstacles in reproducibility, scalability, and interface tuning. Without bridging the divide between atomic-scale mechanisms and process-level control, the full potential of mechanochemical architectonics in constructing smart and functional interfaces will remain unrealized.
6.2. Future opportunities
Mechanochemistry offers a scalable, solvent-free, and energy-efficient platform to design advanced nanomaterials, presenting significant opportunities for sustainable catalytic technologies beyond the limitations of conventional synthesis.16 Expanding mechanochemistry into green and sustainable domains offers promising avenues to replace hazardous reagents, valorize biomass, and reduce energy inputs across inorganic material synthesis.35 In this context, future strategies envision the integration of renewable feedstocks, such as biomass-derived reducing agents or plant extracts, to fabricate nanomaterials with enhanced catalytic performance, antimicrobial activity, and improved environmental compatibility.29,228,229 Impressively, the material cost of this mechanochemical method has been reported to be less than 5% of those prepared using conventional sol–gel techniques,230 highlighting its economic advantage. Mechanochemistry thus offers significant opportunities for producing nanoparticles from renewable and low-cost precursors while minimizing toxic chemicals and energy use.231The future of mechanochemical research will also benefit from hybrid strategies that couple mechanical energy with thermal, light-based, or electromagnetic inputs, thereby enabling greater control over nanostructure formation and reaction selectivity.159 Similarly, integrating mechanochemical methods with colloidal synthesis holds promise for overcoming current shape-control limitations, particularly in noble metal nanostructures, where anisotropy and tunability are crucial for applications in catalysis, sensing, and biomedicine.33,232 The next phase of research should therefore prioritize precise size, shape, and composition control during synthesis, supported by deeper investigation into parameters such as ball-to-powder ratio, milling time, and post-milling heat treatments, including the influence of salt matrices and ion–surface interactions.34,39,233 Developing strategies for soft-mechanochemical processing remains particularly critical for controlling size, shape, and agglomeration compared with more established non-soft approaches.39
In parallel with these broader technological advances, mechanochemistry enables clearly defined opportunities in nanomedicine and hybrid architectures. Force-driven synthesis supports solvent-free fabrication of inorganic–organic and biohybrid interfaces with enhanced stability, defect-mediated bioactivity, and controlled surface chemistry, enabling systematic development of multimodal bioimaging platforms, therapeutic nanozymes, and adaptive biomedical interfaces.
Beyond nanoparticles, mechanochemistry provides outstanding opportunities to access stoichiometric variations of inorganic compounds and synthesize ordered frameworks such as MOFs with tunable porosity, thereby advancing crystal engineering, adsorption, and separation technologies.97,234,235 Mechanochemical methodologies have also demonstrated the ability to generate inorganic complexes and reactive species inaccessible by solution routes, with notable examples including kilogram-scale synthesis of Al(III) and In(III) salen and salophen complexes, which underscores the potential for industrial upscaling.13,14 Such approaches can dramatically reduce reaction times from days to minutes, further enhancing energy efficiency for complex main-group and transition-metal compounds.236 Recent advances in solvent-free C–C and C–N couplings exemplify how mechanochemistry can reshape sustainable industrial and pharmaceutical production,69 while the growing exploration of bioconjugates involving proteins and monosaccharides points to future applications in supramolecular assembly and biointerfaces.237
The integration of real-time in situ monitoring with computational modeling, coupled with advanced synchrotron X-ray diffraction and Raman spectroscopy, provides new opportunities to unravel mechanochemical mechanisms with unprecedented resolution.35,227 Such efforts will be complemented by the emerging concept of reversible mechanochemical reactions, which enables the exploration of equilibrium processes under mechanically driven conditions.238 Life cycle assessment methodologies tailored to mechanochemistry offer a further opportunity to benchmark environmental performance against conventional routes, facilitating industrial adoption.239 At the same time, advancing theoretical models and simulations to describe mass and energy transfer across different scales remains a pressing need for bridging laboratory insights with industrial process design.240 Critically, the industrial translation of mechanochemistry requires optimization of equipment design, milling rates, and energy inputs, ensuring scalability while maintaining sustainability advantages.144,241,242 Mechanochemical routes have already demonstrated significant potential to scale catalyst synthesis toward industrial levels while simultaneously reducing hazardous waste generation and energy consumption.242 Future studies should therefore exploit mechanochemistry to design nanostructured catalysts with enhanced stability and selectivity, enabling applications in renewable energy and environmental remediation beyond traditional chemical production.144,181 The emerging practice of integrating mechanochemistry with complementary electrochemical and photochemical methods further expands the scope of multifunctional pathways, opening new directions for energy storage, conversion, and sustainable environmental technologies.144,243
Altogether, these opportunities establish mechanochemistry not merely as a cost-effective and sustainable synthetic tool, but as a transformative paradigm for designing multiscale inorganic architectures and smart functional interfaces. By uniting green chemistry, computational modeling, real-time monitoring, and hybrid processing strategies, mechanochemistry is poised to shape the next generation of advanced nanomaterials with unprecedented precision, functionality, and scalability.
7. Conclusion
Mechanochemistry has evolved into a transformative paradigm in the synthesis of inorganic nanomaterials, enabling the deliberate construction of smart and functional interfaces through scalable, solvent-free, and energy-efficient routes. This review has articulated the conceptual foundation of mechanochemical architectonics, wherein mechanical force serves not merely as an energetic input but as a precise design parameter that orchestrates structural, interfacial, and functional evolution across multiple length scales. By bridging top-down comminution with bottom-up assembly, mechanochemical platforms ranging from ball milling and extrusion to hybrid force-coupled strategies unlock reaction pathways inaccessible via conventional methods, fostering defect engineering, dynamic interface regeneration, and the creation of responsive nanostructures.Case studies spanning catalysis, energy systems, biomedicine, and electronics affirm mechanochemistry's potential to deliver application-specific nanomaterials with superior performance, tunability, and sustainability. Nonetheless, challenges persist in achieving reproducibility, real-time mechanistic insight, and precise control over size, composition, and morphology. Future progress will hinge on integrating in situ diagnostics, multiscale computational modeling, and AI-guided synthesis to establish predictive control. Furthermore, coupling mechanochemical methods with electrochemical, photochemical, and bioinspired pathways promises to expand functionality while adhering to principles of green chemistry. Altogether, mechanochemical architectonics emerges not merely as a synthetic strategy but as a forward-looking materials design philosophy, poised to shape the next generation of inorganic nanotechnologies for sustainable and multifunctional applications.
Conflicts of interest
There are no conflicts of interest.Abbreviations
| AI | Artificial Intelligence |
| ANNs | Artificial Neural Networks |
| BPR | Ball-to-Powder Ratio |
| CoGEF | Constrained Geometries Simulate External Force |
| DEM | Discrete Element Method |
| DFT | Density Functional Theory |
| GO | Graphene Oxide |
| HER | Hydrogen Evolution Reaction |
| HKUST | Hong Kong University of Science and Technology (MOF family) |
| LAG | Liquid-Assisted Grinding |
| MOFs | Metal–Organic Frameworks |
| MXenes | Two-Dimensional Transition Metal Carbides/Nitrides |
| NMR | Nuclear Magnetic Resonance |
| NR | Nanorods |
| PAD | Push–Pull Protonated Atomic Distance |
| PLA | Polylactic Acid |
| PVA | Polyvinyl Alcohol |
| ROS | Reactive Oxygen Species |
| TSE | Twin-Screw Extrusion |
| WC | Tungsten Carbide |
Data availability
No new data were generated or analyzed in this study. Data sharing is not applicable to this article as it is a review of previously published literature.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5mr00116a.
Acknowledgements
This work was supported by a Universiti Sains Malaysia, Bridging Grant with Project No.: R501-LR-RND003-0000002097-0000.References
- A. D. McNaught and A. D. McNaught, Compendium of Chemical Terminology, Blackwell Science Oxford, vol. 1669, 1997 Search PubMed.
- P. Baláž, et al., Hallmarks of mechanochemistry: from nanoparticles to technology, Chem. Soc. Rev., 2013, 42(18), 7571–7637 Search PubMed.
- L. Takacs, M. Carey Lea, the first mechanochemist, J. Mater. Sci., 2004, 39(16), 4987–4993 Search PubMed.
- D. E. Crawford, et al., Organic synthesis by Twin Screw Extrusion (TSE): continuous, scalable and solvent-free, Green Chem., 2017, 19(6), 1507–1518 Search PubMed.
- D. Crawford, et al., Synthesis by extrusion: continuous, large-scale preparation of MOFs using little or no solvent, Chem. Sci., 2015, 6(3), 1645–1649 RSC.
- T. Auvray and T. Friščić, Shaking things from the ground-up: A systematic overview of the mechanochemistry of hard and high-melting inorganic materials, Molecules, 2023, 28(2), 897 CrossRef CAS PubMed.
- N. Celik, et al., Mechanochemical coupling of alkylsilanes to nanoparticles for solvent-free and rapid fabrication of superhydrophobic materials, ACS Appl. Nano Mater., 2023, 6(16), 14921–14930 CrossRef CAS.
- A. P. Amrute, B. Zibrowius and F. Schüth, Mechanochemical grafting: A solvent-less highly efficient method for the synthesis of hybrid inorganic–organic materials, Chem. Mater., 2020, 32(11), 4699–4706 CrossRef CAS.
- C. Suryanarayana, Mechanical alloying and milling, Prog. Mater Sci., 2001, 46(1–2), 1–184 CrossRef CAS.
- L. Russo, et al., A mechanochemical approach to porous silicon nanoparticles fabrication, Materials, 2011, 4(6), 1023–1033 Search PubMed.
- M. J. Rak, et al., Mechanosynthesis of ultra-small monodisperse amine-stabilized gold nanoparticles with controllable size, Green Chem., 2014, 16(1), 86–89 RSC.
- G. Karagedov and N. Lyakhov, Mechanochemical grinding of inorganic oxides, Kona Powder Part. J., 2003, 21, 76–87 CrossRef CAS.
- M. J. Rak, T. Friščić and A. Moores, Mechanochemical synthesis of Au, Pd, Ru and Re nanoparticles with lignin as a bio-based reducing agent and stabilizing matrix, Faraday Discuss., 2014, 170, 155–167 Search PubMed.
- V. Šepelák, et al., High-resolution 27Al MAS NMR spectroscopic studies of the response of spinel aluminates to mechanical action, J. Mater. Chem., 2011, 21(23), 8332–8337 RSC.
- K. Ralphs, C. Hardacre and S. L. James, Application of heterogeneous catalysts prepared by mechanochemical synthesis, Chem. Soc. Rev., 2013, 42(18), 7701–7718 RSC.
- C. Xu, et al., Mechanochemical synthesis of advanced nanomaterials for catalytic applications, Chem. Commun., 2015, 51(31), 6698–6713 RSC.
- T. Tsuzuki and P. G. McCormick, Mechanochemical synthesis of nanoparticles, J. Mater. Sci., 2004, 39(16), 5143–5146 CrossRef CAS.
- F. Gomollón-Bel, Ten Chemical Innovations That Will Change Our World: IUPAC identifies emerging technologies in Chemistry with potential to make our planet more sustainable, Chem. Int., 2019, 41(2), 12–17 Search PubMed.
- M. Bellusci, et al., Magnetic metal–organic framework composite by fast and facile mechanochemical process, Inorg. Chem., 2018, 57(4), 1806–1814 CrossRef CAS.
- S. Kumar, et al., All-inorganic CsPbBr3 nanocrystals: gram-scale mechanochemical synthesis and selective photocatalytic CO2 reduction to methane, ACS Appl. Energy Mater., 2020, 3(5), 4509–4522 CrossRef CAS.
- Y. Sun, et al., Synthesis and formation mechanism of cubic ZrN nanopowders by mechanochemical reaction of ZrCl4 and Li3N, J. Alloys Compd., 2009, 479(1–2), 599–602 CrossRef CAS.
- R. N. Baig and R. S. Varma, Alternative energy input: mechanochemical, microwave and ultrasound-assisted organic synthesis, Chem. Soc. Rev., 2012, 41(4), 1559–1584 RSC.
- J.-L. Do and T. Friščić, Mechanochemistry: a force of synthesis, ACS Cent. Sci., 2017, 3(1), 13–19 CrossRef CAS PubMed.
- B. G. Fiss, et al., Solvent-free mechanochemical synthesis of ultrasmall nickel phosphide nanoparticles and their application as a catalyst for the hydrogen evolution reaction (HER), ACS Sustain. Chem. Eng., 2020, 8(32), 12014–12024 CrossRef CAS.
- A. Vílchez, et al., Mechanochemical synthesis of TiO2 nanoparticles and their self-organization at interfaces to produce emulsion-templated photocatalytic porous polymers, J. Inorg. Organomet. Polym. Mater., 2021, 31(5), 1912–1930 CrossRef.
- J. Chen, K. Shen and Y. Li, Greening the processes of metal–organic framework synthesis and their use in sustainable catalysis, ChemSusChem, 2017, 10(16), 3165–3187 CrossRef CAS PubMed.
- J. Andersen and J. Mack, Mechanochemistry and organic synthesis: from mystical to practical, Green Chem., 2018, 20(7), 1435–1443 RSC.
- T. D. Isfahani, et al., Mechanochemical synthesis of zirconia nanoparticles: Formation mechanism and phase transformation, Int. J. Refract. Met. Hard Mater., 2012, 31, 21–27 CrossRef CAS.
- M. Kováčová, et al., Sustainable one-step solid-state synthesis of antibacterially active silver nanoparticles using mechanochemistry, Nanomaterials, 2020, 10(11), 2119 CrossRef.
- D. Becker, M. Klos and G. Kickelbick, Mechanochemical synthesis of Mn3O4 nanocrystals and their lithium intercalation capability, Inorg. Chem., 2019, 58(22), 15021–15024 CrossRef CAS.
- S. L. James, et al., Mechanochemistry: opportunities for new and cleaner synthesis, Chem. Soc. Rev., 2012, 41(1), 413–447 RSC.
- O. Lapshin, E. Boldyreva and V. Boldyrev, Role of mixing and milling in mechanochemical synthesis, Russ. J. Inorg. Chem., 2021, 66(3), 433–453 CrossRef CAS.
- P. F. de Oliveira, et al., Challenges and opportunities in the bottom-up mechanochemical synthesis of noble metal nanoparticles, J. Mater. Chem. A, 2020, 8(32), 16114–16141 RSC.
- Y. Xia, et al., Shape-controlled synthesis of metal nanocrystals: simple chemistry meets complex physics?, Angew. Chem., Int. Ed., 2009, 48(1), 60–103 CrossRef CAS.
- B. G. Fiss, et al., Mechanochemical methods for the transfer of electrons and exchange of ions: inorganic reactivity from nanoparticles to organometallics, Chem. Soc. Rev., 2021, 50(14), 8279–8318 RSC.
- F. Cuccu, et al., Mechanochemistry: New Tools to Navigate the Uncharted Territory of “Impossible” Reactions, ChemSusChem, 2022, 15(17), e202200362 CrossRef CAS.
- D. Singh and S. Dilip Saoji, Nanoarchitectonics in Macromolecular Science: Integrating Molecular Dynamics with Smart Materials, J. Macromol. Sci., Part B: Phys., 2024, 1–10 Search PubMed.
- T. Tsuzuki, Mechanochemical synthesis of metal oxide nanoparticles, Commun. Chem., 2021, 4, 143, DOI:10.1038/s42004-021-00582-3.
- T. Tsuzuki, Mechanochemical synthesis of metal oxide nanoparticles, Commun. Chem., 2021, 4(1), 143 CrossRef CAS.
- J. N. Stevanović, et al., Mechanochemical Synthesis of TiO(2)-CeO(2) Mixed Oxides Utilized as a Screen-Printed Sensing Material for Oxygen Sensor, Sensors, 2023, 23(3), 1313 CrossRef.
- Q. Shi, et al., Nano-architectonics of Pt single-atoms and differently-sized nanoparticles supported by manganese-oxide nanosheets and impact on catalytic and anti-biofilm activities, J. Colloid Interface Sci., 2024, 672, 224–235 CrossRef CAS PubMed.
- E. Colacino, F. Delogu and T. Hanusa, Advances in Mechanochemistry, ACS Sustain. Chem. Eng., 2021, 9(32), 10662–10663 CrossRef CAS.
- X. Liu, et al., A review on mechanochemistry: approaching advanced energy materials with greener force, Adv. Mater., 2022, 34(46), 2108327 CrossRef CAS PubMed.
- J.-L. Do and T. Friščić, Mechanochemistry: A Force of Synthesis, ACS Cent. Sci., 2017, 3(1), 13–19 CrossRef CAS.
- P. F. M. de Oliveira, et al., Challenges and opportunities in the bottom-up mechanochemical synthesis of noble metal nanoparticles, J. Mater. Chem. A, 2020, 8(32), 16114–16141 RSC.
- J. Beamish-Cook, et al., Insights into the Mechanochemical Synthesis of MOF-74, Cryst. Growth Des., 2021, 21(5), 3047–3055 CrossRef CAS PubMed.
- M. Klimakow, et al., Mechanochemical Synthesis of Metal−Organic Frameworks: A Fast and Facile Approach toward Quantitative Yields and High Specific Surface Areas, Chem. Mater., 2010, 22(18), 5216–5221 CrossRef CAS.
- G.-Y. Yan, et al., Mechanochemical solid state architectonics on Lead(II) coordination polymer by anion-exchange, J. Solid State Chem., 2021, 304, 122592 CrossRef CAS.
- M. Avinash and T. Govindaraju, Architectonics: design of molecular architecture for functional applications, Acc. Chem. Res., 2018, 51(2), 414–426 CrossRef CAS PubMed.
- R. Huang, et al., Molecular design and architectonics towards film-based fluorescent sensing, Chem. Soc. Rev., 2024, 53(13), 6960–6991 RSC.
- L. Dong, et al., Mechanochemistry: Fundamental Principles and Applications, Adv. Sci., 2025, 12(24), 2403949 CrossRef CAS PubMed.
- S. Hwang, S. Grätz and L. Borchardt, A guide to direct mechanocatalysis, Chem. Commun., 2022, 58(11), 1661–1671 RSC.
- T. Stauch and A. Dreuw, Advances in quantum mechanochemistry: electronic structure methods and force analysis, Chem. Rev., 2016, 116(22), 14137–14180 CrossRef CAS PubMed.
- M. Zakeri, M. Ramezani and A. Nazari, Effect of ball to powder weight ratio on the mechanochemical synthesis of MoSi2-TiC nanocomposite powder, Mater. Res., 2012, 15, 891–897 Search PubMed.
- A. M. Belenguer, G. I. Lampronti and J. K. M. Sanders, Reliable Mechanochemistry: Protocols for Reproducible Outcomes of Neat and Liquid Assisted Ball-mill Grinding Experiments, J. Visualized Exp., 2018, 131, 56824 Search PubMed.
- O. F. Jafter, et al., Navigating Ball Mill Specifications for Theory-to-Practice Reproducibility in Mechanochemistry, Angew. Chem., Int. Ed., 2024, 63(48), e202409731 CrossRef CAS PubMed.
- L. K. Wei, et al., Producing Metal Powder from Machining Chips Using Ball Milling Process: A Review, Materials, 2023, 16(13), 4635 CrossRef CAS PubMed.
- R. De Armas, M. Temprado and L. M. Frutos, Computational Model to Predict Reactivity under Ball-Milling Conditions, J. Chem. Theory Comput., 2025, 21(20), 10353–10361 Search PubMed.
- A. Krusenbaum, et al., The mechanochemical synthesis of polymers, Chem. Soc. Rev., 2022, 51(7), 2873–2905 Search PubMed.
- A. Ochirkhuyag, et al., One-pot mechanochemical ball milling synthesis of the MnOx nanostructures as efficient catalysts for CO2 hydrogenation reactions, Phys. Chem. Chem. Phys., 2020, 22(25), 13999–14012 RSC.
- B. Zhao, et al., Multifunctional Iron Oxide Nanoflake/Graphene Composites Derived from Mechanochemical Synthesis for Enhanced Lithium Storage and Electrocatalysis, ACS Appl. Mater. Interfaces, 2015, 7(26), 14446–14455 CrossRef CAS PubMed.
- G. Bharath, et al., Solvent-free mechanochemical synthesis of graphene oxide and Fe3O4–reduced graphene oxide nanocomposites for sensitive detection of nitrite, J. Mater. Chem. A, 2015, 3(30), 15529–15539 Search PubMed.
- W. Xiao, et al., Facile Synthesis of Highly Porous Metal Oxides by Mechanochemical Nanocasting, Chem. Mater., 2018, 30(9), 2924–2929 Search PubMed.
- V. Oskoei, M. Mathesh and W. Yang, Mechanochemical Nanoarchitectonics for the Synthesis of Enzyme-Based Hydrogen-Bonded Organic Frameworks, Chem. Mater., 2025, 37(18), 7206–7213 Search PubMed.
- R. Li, et al., Mechanochemical Synthesis of Defective Molybdenum Trioxide, Titanium Dioxide, and Zinc Oxide at Room Temperature, ACS Sustain. Chem. Eng., 2019, 7(14), 11985–11989 Search PubMed.
- A. D. Katsenis, et al., In situ X-ray diffraction monitoring of a mechanochemical reaction reveals a unique topology metal-organic framework, Nat. Commun., 2015, 6(1), 6662 CrossRef CAS PubMed.
- H. Kulla, et al., In Situ Investigations of Mechanochemical One-Pot Syntheses, Angew Chem. Int. Ed. Engl., 2018, 57(20), 5930–5933 Search PubMed.
- G. Zhang, et al., Fabrication of MBene-Based ZIF Flame Retardant Epoxy Resin via Green and Feasible Mechanochemical Method, ACS Sustain. Chem. Eng., 2025, 13(27), 10573–10586 Search PubMed.
- J. F. Reynes, F. Leon and F. García, Mechanochemistry for organic and inorganic synthesis, ACS Org. Inorg. Au, 2024, 4(5), 432–470 CrossRef CAS PubMed.
- D. Sarmah, A. Hazarika and B. K. Saikia, Graphene Architectures in Coal-Derived Carbon: Turbostratic Twist and Supercapacitor Promise, Energy Fuels, 2025, 39(30), 14724–14737 Search PubMed.
- V. Boldyrev and K. Tkáčová, Mechanochemistry of solids: past, present, and prospects, J. Mater. Synth. Process., 2000, 8(3), 121–132 Search PubMed.
- T. Friščić, C. Mottillo and H. M. Titi, Mechanochemistry for synthesis, Angew. Chem., 2020, 132(3), 1030–1041 CrossRef.
- L. Takacs, The historical development of mechanochemistry, Chem. Soc. Rev., 2013, 42(18), 7649–7659 RSC.
- C. G. Vogt, et al., Bronze age of direct mechanocatalysis: how alloyed milling materials advance coupling in ball mills, Adv. Energy Sustainability Res., 2021, 2(5), 2100011 CrossRef CAS.
- P. A. Julien and T. Friscic, Methods for monitoring milling reactions and mechanistic studies of mechanochemistry: a primer, Cryst. Growth Des., 2022, 22(9), 5726–5754 CrossRef CAS.
- J. Mack, et al., The first solvent-free method for the reduction of esters, Green Chem., 2007, 9(10), 1041–1043 RSC.
- Y. Gao, K. Kubota and H. Ito, Mechanochemical Approach for Air-Tolerant and Extremely Fast Lithium-Based Birch Reductions in Minutes, Angew. Chem., 2023, 135(21), e202217723 CrossRef.
- H. U. Escobar-Hernandez, et al., Life cycle assessment of metal–organic frameworks: sustainability study of zeolitic imidazolate framework-67, ACS Sustain. Chem. Eng., 2023, 11(10), 4219–4225 CrossRef CAS.
- Y. Lou, et al., Mechanochemical Urea Synthesis Using Ammonia–Water and Carbon Dioxide Under Mild Conditions: An Experimental and Theoretical Study, ACS Sustain. Chem. Eng., 2025, 13(1), 151–164 CrossRef CAS.
- B. Dhokale, et al., Mechanochemistry for the Sustainable Synthesis of Organic Hole Transport Materials in Perovskite Solar Cells, Cryst. Growth Des., 2025, 25(8), 2402–2408 CrossRef CAS PubMed.
- E. Marín, X. Vendrell and J. Llorca, Pd Supported on CeO2 Nanostructures Prepared by Planetary Ball Milling under a Modified Atmosphere for Catalytic Oxidation of CO, ACS Appl. Nano Mater., 2025, 8(23), 12151–12163 Search PubMed.
- X. Shen, et al., Rapid mechanochemical synthesis of polyanionic cathode with improved electrochemical performance for Na-ion batteries, Nat. Commun., 2021, 12(1), 2848 CrossRef CAS PubMed.
- R. Schlem, et al., Energy storage materials for solid-state batteries: design by mechanochemistry, Adv. Energy Mater., 2021, 11(30), 2101022 Search PubMed.
- S. He, et al., Solvent-free mechanochemical synthesis of Na-rich Prussian white cathodes for high-performance Na-ion batteries, Chem. Eng. J., 2022, 428, 131083 Search PubMed.
- G. Liu, et al., High air-stability and superior lithium ion conduction of Li3+ 3xP1-xZnxS4-xOx by aliovalent substitution of ZnO for all-solid-state lithium batteries, Energy Storage Mater., 2019, 17, 266–274 Search PubMed.
- B. Biswas, et al., Mechanochemically Activated Halloysite Nanotube-Rich Kaolin Clay As a Carrier for Slow-Release Phosphate Fertilizer, ACS Sustain. Chem. Eng., 2025, 13(23), 8711–8721 Search PubMed.
- W. Cai, et al., Mechanochemically Driven C–C Bond Formation via Cu-Complex-Functionalized Polyoxoniobate under Solvent-Free Conditions, Cryst. Growth Des., 2025, 25(5), 1636–1643 CrossRef CAS.
- I. Sande, et al., Mechanochemical Synthesis of 2-Amino-1, 4-naphthoquinones and Telescopic Synthesis of Lawsone, ACS Omega, 2025, 10(40), 46369–46383 CrossRef CAS PubMed.
- T. Plant-Collins, et al., Beyond Solution Chemistry: Mechanochemistry Enables Clustered Defects in Metal–Organic Frameworks, Inorg. Chem., 2025, 64(34), 17436–17447 CrossRef CAS PubMed.
- S. L. James and T. Friščić, Mechanochemistry, Chem. Soc. Rev., 2013, 42(18), 7494–7496 RSC.
- T. Friščić, et al., Ion-and liquid-assisted grinding: improved mechanochemical synthesis of metal–organic frameworks reveals salt inclusion and anion templating, Angew. Chem., Int. Ed., 2010, 49(4), 712–715 CrossRef PubMed.
- Y. Filinchuk, et al., Porous and dense magnesium borohydride frameworks: synthesis, stability, and reversible absorption of guest species, Angew. Chem., 2011, 123(47), 11358–11362 CrossRef.
- R. Zhou, et al., Cu-MOF@ Pt 3D nanocomposites prepared by one-step wrapping method with peroxidase-like activity for colorimetric detection of glucose, Colloids Surf., B, 2022, 216, 112601 CrossRef CAS PubMed.
- J. Alić, et al., Meeting the UN sustainable development goals with mechanochemistry, Angew. Chem., Int. Ed., 2024, 63(50), e202414745 CrossRef PubMed.
- M. Mohamed, et al., Mechanochemical synthesis of Li-rich (Li 2 Fe) SO cathode for Li-ion batteries, Green Chem., 2023, 25(10), 3878–3887 RSC.
- G.-F. Han, et al., Mechanochemistry for ammonia synthesis under mild conditions, Nat. Nanotechnol., 2021, 16(3), 325–330 CrossRef CAS PubMed.
- S. Głowniak, et al., Mechanochemistry: Toward green synthesis of metal–organic frameworks, Mater. Today, 2021, 46, 109–124 CrossRef.
- A. M. Fidelli, et al., Green and rapid mechanosynthesis of high-porosity NU-and UiO-type metal–organic frameworks, Chem. Commun., 2018, 54(51), 6999–7002 RSC.
- S. Bonciolini, A. Pulcinella and T. Noël, Tech-Enhanced Synthesis: Exploring the Synergy between Organic Chemistry and Technology, J. Am. Chem. Soc., 2025, 147(32), 28523–28545 CrossRef CAS PubMed.
- D. E. Crawford, et al., Solvent-free, continuous synthesis of hydrazone-based active pharmaceutical ingredients by twin-screw extrusion, ACS Sustain. Chem. Eng., 2020, 8(32), 12230–12238 CrossRef CAS.
- M. Lavayssiere and F. Lamaty, Amidation by reactive extrusion for the synthesis of active pharmaceutical ingredients teriflunomide and moclobemide, Chem. Commun., 2023, 59(23), 3439–3442 RSC.
- R. R. Bolt, et al., Solvent Minimized Synthesis of Amides by Reactive Extrusion, Angew. Chem., 2024, 136(41), e202408315 CrossRef.
- Y. Song, et al., Continuous and Large-Scale Preparation of Hierarchical Porous HKUST-1 via the “Nanofusion” Mechanism Using Liquid-Assisted Mechanosynthesis, Inorg. Chem., 2025, 64(11), 5579–5585 Search PubMed.
- Y. Xie, et al., Hygroscopicity Reduction and Interconversion of Ligustrazine in Ligusticum chuanxiong Hort. via Cocrystallization and Mechanochemical Technology, Cryst. Growth Des., 2025, 25(16), 6686–6696 CrossRef CAS.
- J. Wang, et al., Self-Correcting Assembly via Mechanochemical Polymer Rearrangement for Scalable Nanostructure Manufacturing, ACS Mater. Lett., 2024, 6(10), 4443–4451 Search PubMed.
- S. Zeng, et al., Green Preparation of High-Performance Poly (Vinyl Alcohol) Film by Combining Gel-Like Extrusion and Biaxial Stretching, Ind. Eng. Chem. Res., 2024, 63(19), 8622–8632 CrossRef CAS.
- N. Kurbanova, S. Ragimova and T. Guliyeva, Nickel-Containing Nanocomposites Based on Isotactic Polypropylene and High-Pressure Polyethylene, Inorg. Mater. Appl. Res., 2024, 15(5), 1350–1354 CrossRef.
- C. Pina-Vidal, et al., Mechanochemical encapsulation of caffeine in UiO-66 and UiO-66-NH2 to obtain polymeric composites by extrusion with recycled polyamide 6 or polylactic acid biopolymer, Polymers, 2024, 16(5), 637 CrossRef CAS PubMed.
- B. A. Steele, et al., Mechanochemical synthesis of glycine oligomers in a virtual rotational diamond anvil cell, Chem. Sci., 2020, 11(30), 7760–7771 RSC.
- J. A. Ciezak-Jenkins and T. A. Jenkins, Mechanochemical induced structural changes in sucrose using the rotational diamond anvil cell, J. Appl. Phys., 2018, 123(8), 085901 CrossRef.
- G. De Bo, Mechanochemistry of the mechanical bond, Chem. Sci., 2018, 9(1), 15–21 RSC.
- H. Yan, et al., Sterically controlled mechanochemistry under hydrostatic pressure, Nature, 2018, 554(7693), 505–510 CrossRef CAS PubMed.
- L. Zhu, et al., Mechanochemistry, solvent-free and scale-up: Application toward coupling of acids and amines to amides, Results Chem., 2023, 5, 100882 Search PubMed.
- C. Espro and D. Rodriguez-Padron, Re-thinking organic synthesis: Mechanochemistry as a greener approach, Curr. Opin. Green Sustainable Chem., 2021, 30, 100478 CrossRef CAS.
- B. Cecen, et al., Smart biomaterials in biomedical applications: current advances and possible future directions, Macromol. Biosci., 2024, 24(3), 2200550 Search PubMed.
- S. Iravani, et al., Advancements in MXenes and mechanochemistry: exploring new horizons and future applications, Mater. Adv., 2024, 5(21), 8404–8418 RSC.
- V. Martinez, et al., Advancing mechanochemical synthesis by combining milling with different energy sources, Nat. Rev. Chem., 2023, 7(1), 51–65 CrossRef CAS PubMed.
- X. Wang, et al., Defect Engineering to Boost the Lithium-Ion Storage Performance of Ti3C2T x MXene Induced by Plasma-Assisted Mechanochemistry, ACS Appl. Energy Mater., 2021, 4(9), 10280–10289 CrossRef CAS.
- J. Wang, et al., Mechanochemistry-induced biaxial compressive strain engineering in MXenes for boosting lithium storage kinetics, Nano Energy, 2021, 87, 106053 CrossRef CAS.
- S. Zhang, et al., An MXene/CNTs@ P nanohybrid with stable Ti–O–P bonds for enhanced lithium ion storage, J. Mater. Chem. A, 2019, 7(38), 21766–21773 RSC.
- X. Liu, et al., Solid-state mechanochemistry advancing two dimensional materials for lithium-ion storage applications: A mini review, Nano Mater. Sci., 2023, 5(2), 210–227 CrossRef CAS.
- J. Batteas, et al., Moving mechanochemistry forward, RSC Mechanochem., 2025, 2(1), 10–19 Search PubMed.
- M. Senna and A. A. Michalchuk, Reassessing mechanochemical processes in polyatomic systems for smart fabrication of nanocomposites, RSC Mechanochem., 2025, 2(3), 351–369 RSC.
- W. A. Ali, S. E. Richards and R. H. Alzard, Unlocking the potential of ball milling for nanomaterial Synthesis: An overview, J. Ind. Eng. Chem., 2025, 149, 63–93 Search PubMed.
- S. W. Hwang, et al., Hybrid Vesicles Enable Mechano-Responsive Hydrogel Degradation, Angew. Chem., 2023, 135(41), e202308509 Search PubMed.
- C. Wang, et al., How external forces affect the degradation properties of perfluorooctanoic acid in mechanochemical degradation: a DFT study, RSC Mechanochem., 2025, 2, 692–705 RSC.
- S. Mittal, et al., Molecular dynamics model of mechanophore sensors for biological force measurement, Heliyon, 2025, 11(1), e41178 Search PubMed.
- S. Ghose, et al., Response of a Tethered Zn-Bis-Terpyridine Complex to an External Mechanical Force: A Computational Study of the Roles of the Tether and Solvent, J. Phys. Chem. A, 2025, 129(15), 3423–3434 CrossRef CAS PubMed.
- X. Zhao, et al., Hybrid Spike-Facilitated Capture and Biofilm Destruction Co-Enhances Ultrasound-Mediated Bactericidal Therapy, ACS Nano, 2025, 19(35), 31720–31739 CrossRef CAS PubMed.
- J. Qu, et al., Mechanochemically synthesised MXene@ PAbz hybrids endow EP coatings with excellent fire safety performance, Chem. Eng. J., 2025, 167688 CrossRef CAS.
- M. Rok, et al., Secret agent in the secret service: Utilization of Sb (iii)-based complexes' emission properties for the study of forgery and document authenticity, J. Mater. Chem. C, 2025, 13(33), 17241–17250 RSC.
- E. C. Spencer, et al., Pressure-Induced Bond Rearrangement and Reversible Phase Transformation in a Metal–Organic Framework, Angew. Chem., 2014, 126(22), 5689–5692 CrossRef.
- A. V. Neimark, et al., Structural transitions in MIL-53 (Cr): view from outside and inside, Langmuir, 2011, 27(8), 4734–4741 CrossRef CAS PubMed.
- T. D. Bennett, et al., Reversible pressure-induced amorphization of a zeolitic imidazolate framework (ZIF-4), Chem. Commun., 2011, 47(28), 7983–7985 RSC.
- A. U. Ortiz, et al., Anisotropic Elastic Properties of Flexible Metal-Organic Frameworks:<? format?> How Soft are Soft Porous Crystals?, Phys. Rev. Lett., 2012, 109(19), 195502 Search PubMed.
- J. McCarron, B. Turner and L. N. McHugh, Hybrid Framework Materials: Next-Generation Engineering Materials, Adv. Eng. Mater., 2025, 27(9), 2402554 Search PubMed.
- E. C. Spencer, et al., Pressure-induced cooperative bond rearrangement in a zinc imidazolate framework: a high-pressure single-crystal X-ray diffraction study, J. Am. Chem. Soc., 2009, 131(11), 4022–4026 Search PubMed.
- S. Henke, W. Li and A. K. Cheetham, Guest-dependent mechanical anisotropy in pillared-layered soft porous crystals–a nanoindentation study, Chem. Sci., 2014, 5(6), 2392–2397 Search PubMed.
- T. D. Bennett, et al., Connecting defects and amorphization in UiO-66 and MIL-140 metal–organic frameworks: a combined experimental and computational study, Phys. Chem. Chem. Phys., 2016, 18(3), 2192–2201 Search PubMed.
- K. Uzarevic, I. Halasz and T. Friscic, Real-time and in situ monitoring of mechanochemical reactions: A new playground for all chemists, J. Phys. Chem. Lett., 2015, 6(20), 4129–4140 CrossRef CAS PubMed.
- W. Wang, et al., Metal–organic framework composites from a mechanochemical process, Mol. Syst. Des. Eng., 2023, 8(5), 560–579 RSC.
- E. Boldyreva, Mechanochemistry of inorganic and organic systems: what is similar, what is different?, Chem. Soc. Rev., 2013, 42(18), 7719–7738 Search PubMed.
- B. Szczęśniak, et al., Mechanochemical synthesis of highly porous materials, Mater. Horiz., 2020, 7(6), 1457–1473 RSC.
- A. B. Chetry, Mechanochemistry: A new frontier in chemical synthesis, J. Chem. Res., 2025, 49(3), 17475198251339299 CrossRef CAS.
- Y. Wu, K. Lin and J. Ruan, Control the Mechanochemical Energy of Ball Milling To Remove Surface Organic Contamination without Damaging the Integrity of the Glass, ACS Sustain. Chem. Eng., 2023, 11(41), 15083–15090 CrossRef CAS.
- Z. Yang, et al., Chiral superstructures of inorganic nanorods by macroscopic mechanical grinding, Nat. Commun., 2022, 13(1), 5844 CrossRef CAS PubMed.
- S. Cao, et al., Amorphization of the prototypical zeolitic imidazolate framework ZIF-8 by ball-milling, Chem. Commun., 2012, 48(63), 7805–7807 RSC.
- M. Taheri, et al., Green full conversion of ZnO nanopowders to well-dispersed zeolitic imidazolate framework-8 (ZIF-8) nanopowders via a stoichiometric mechanochemical reaction for fast dye adsorption, Cryst. Growth Des., 2020, 20(4), 2761–2773 CrossRef CAS.
- Y.-R. Miao and K. S. Suslick, Mechanochemical reactions of metal-organic frameworks, in Advances in Inorganic Chemistry, Elsevier, 2018, pp. 403–434 Search PubMed.
- S. Tanaka, et al., Mechanochemical dry conversion of zinc oxide to zeolitic imidazolate framework, Chem. Commun., 2013, 49(72), 7884–7886 Search PubMed.
- V. Šepelák, S. Bégin-Colin and G. Le Caer, Transformations in oxides induced by high-energy ball-milling, Dalton Trans., 2012, 41(39), 11927–11948 Search PubMed.
- J. M. Marrett, et al., Mechanochemistry for Metal–Organic Frameworks and Covalent–Organic Frameworks (MOFs, COFs): Methods, Materials, and Mechanisms, Adv. Mater., 2025, 2418707 CrossRef CAS PubMed.
- X. Zhou, et al., Mechanochemistry of metal–organic frameworks under pressure and shock, Acc. Chem. Res., 2020, 53(12), 2806–2815 Search PubMed.
- J. De Bellis, et al., Surface and bulk chemistry of mechanochemically synthesized tohdite nanoparticles, J. Am. Chem. Soc., 2022, 144(21), 9421–9433 CrossRef CAS PubMed.
- X. Li, et al., Mechanochemistry-assisted encapsulation of metal nanoparticles in MOF matrices via a sacrificial strategy, J. Mater. Chem. A, 2019, 7(24), 14504–14509 Search PubMed.
- H. K. Lee, J. H. Lee and H. R. Moon, Mechanochemistry as a reconstruction tool of decomposed metal–organic frameworks, Inorg. Chem., 2021, 60(16), 11825–11829 CrossRef CAS PubMed.
- I. R. Speight and T. P. Hanusa, Exploration of mechanochemical activation in solid-state fluoro-Grignard reactions, Molecules, 2020, 25(3), 570 CrossRef CAS PubMed.
- N. C. Boyde, et al., Symmetric Assembly of a Sterically Encumbered Allyl Complex: Mechanochemical and Solution Synthesis of the Tris (Allyl) Beryllate, K [BeA′ 3](A′= 1, 3-(SiMe3) 2C3H3), Inorganics, 2017, 5(2), 36 Search PubMed.
- H. Hasan, F. Arshad and M. P. Sk, Mechanochemical conversion of elemental sulfur into functional sulfur nanomaterials for promising applications, RSC Mechanochem., 2025, 2(6), 786–801 RSC.
- H. Cheng, et al., Mechanochemical synthesis of highly porous CeMnO x catalyst for the removal of NO x, Ind. Eng. Chem. Res., 2019, 58(36), 16472–16478 CrossRef CAS.
- B. Zhang, et al., Embedding hierarchical pores by mechanochemistry in carbonates with superior chemoselective catalysis and stability, Inorg. Chem., 2023, 62(32), 12920–12930 CrossRef CAS PubMed.
- D. Yu, et al., Alkali metals and cerium-modified La–Co-based perovskite catalysts: facile synthesis, excellent catalytic performance, and reaction mechanisms for soot combustion, ACS Catal., 2022, 12(24), 15056–15075 Search PubMed.
- G. I. Bell, Models for the specific adhesion of cells to cells: a theoretical framework for adhesion mediated by reversible bonds between cell surface molecules, Science, 1978, 200(4342), 618–627 CrossRef CAS PubMed.
- N. Hopper, et al., Exploring mechanochemical reactions at the nanoscale: Theory versus experiment, Phys. Chem. Chem. Phys., 2023, 25(23), 15855–15861 RSC.
- R. Rana, et al., Anisotropy of shear-induced mechanochemical reaction rates of surface adsorbates; implications for theoretical models, J. Phys. Chem. C, 2022, 126(28), 11585–11593 CrossRef CAS.
- T. Bunno, et al., Ternary Triazole-Based Organic–Inorganic Proton-Conducting Hybrids Based on Computational Models for HT-PEMFC Application, ACS Omega, 2023, 8(46), 44172–44182 Search PubMed.
- T. Wu and J. Wang, Deep mining stable and nontoxic hybrid organic–inorganic perovskites for photovoltaics via progressive machine learning, ACS Appl. Mater. Interfaces, 2020, 12(52), 57821–57831 Search PubMed.
- B. Chen, R. Hoffmann and R. Cammi, The effect of pressure on organic reactions in fluids—a new theoretical perspective, Angew. Chem., Int. Ed., 2017, 56(37), 11126–11142 CrossRef CAS PubMed.
- M. T. Ong, et al., First principles dynamics and minimum energy pathways for mechanochemical ring opening of cyclobutene, J. Am. Chem. Soc., 2009, 131(18), 6377–6379 CrossRef CAS PubMed.
- W. Sakai, et al., Origin of stereoselectivity in a mechanochemical reaction of diphenylfulvene and maleimide, J. Phys. Chem. A, 2023, 127(28), 5790–5794 Search PubMed.
- B. S. Pladevall, A. de Aguirre and F. Maseras, Understanding ball milling mechanochemical processes with DFT calculations and microkinetic modeling, ChemSusChem, 2021, 14(13), 2763–2768 CrossRef CAS PubMed.
- H. Rojas-Chávez, et al., A Comparative DFT Study on Process Control Agents in the Mechanochemical Synthesis of PbTe, Int. J. Mol. Sci., 2022, 23(19), 11194 Search PubMed.
- H. Adams, Modeling mechanochemical reaction mechanisms, ACS Appl. Mater. Interfaces, 2017, 9(31), 26531–26538 Search PubMed.
- M. Fusaro, et al., Computational modeling of gold nanoparticle interacting with molecules of pharmaceutical interest in water, Molecules, 2023, 28(20), 7167 CrossRef CAS PubMed.
- K. Triatmaja, et al., Development of Graphene Oxide-Based Antibacterial Agents via ZnO/Eugenol Modification: Experimental and Computational Analysis, Indones. J. Chem., 2025, 25(2), 359 Search PubMed.
- L. Yang, et al., Thermodynamics model for mechanochemical synthesis of gold nanoparticles: Implications for solvent-free nanoparticle production, ACS Appl. Nano Mater., 2021, 4(2), 1886–1897 Search PubMed.
- H. T. Ta, et al., Ab initio informed machine learning potential for tribochemistry and mechanochemistry: Application for eco–friendly gallate lubricant additive, Comput. Mater. Today, 2024, 1, 100005 Search PubMed.
- J. R. Gröls and B. Castro-Dominguez, Mechanochemical co-crystallization: Insights and predictions, Comput. Chem. Eng., 2021, 153, 107416 CrossRef.
- R. Shaikh, S. Shirazian and G. M. Walker, Application of artificial neural network for prediction of particle size in pharmaceutical cocrystallization using mechanochemical synthesis, Neural Comput. Appl., 2021, 33(19), 12621–12640 CrossRef.
- M. U. Rahman, et al., Computational chemistry unveiled: a critical analysis of theoretical coordination chemistry and nanostructured materials, Chem. Prod. Process Model., 2024, 19(4), 473–515 Search PubMed.
- A. P. Amrute, et al., Mechanochemical synthesis of catalytic materials, Chem.–Eur. J., 2021, 27(23), 6819–6847 CrossRef CAS PubMed.
- F. García, M. Senna and V. Šepelák, Moving mechanochemistry forward: reimagining inorganic chemistry through mechanochemistry, RSC Mechanochem., 2025, 2, 499–502 RSC.
- K. M. Siniard, et al., Precision structure engineering of high-entropy oxides under ambient conditions, ACS Catal., 2024, 14(19), 14807–14818 CrossRef CAS.
- L. Xiao, et al., A novel, solvent-free mechanochemistry approach for gold extraction from anode slime, ACS Sustain. Chem. Eng., 2019, 7(13), 11415–11425 CrossRef CAS.
- W. Zhan, et al., Incorporating rich mesoporosity into a ceria-based catalyst via mechanochemistry, Chem. Mater., 2017, 29(17), 7323–7329 CrossRef CAS.
- Q. Yang, et al., Mechanochemistry Strategy in Metal/Fe3O4 with High Stability for Superior Chemoselective Catalysis, ACS Appl. Mater. Interfaces, 2024, 16(48), 66219–66229 CrossRef CAS PubMed.
- W. Zhu, et al., SupraCells: living mammalian cells protected within functional modular nanoparticle-based exoskeletons, Adv. Mater., 2019, 31(25), 1900545 CrossRef PubMed.
- F. H. Silver, Mechanosensing and Mechanochemical Transduction in Extracellular Matrix: Biological, Chemical, Engineering, and Physiological Aspects, Springer, 2006 Search PubMed.
- P. Krupiński, et al., From Uncommon Ethylzinc Complexes Supported by Ureate Ligands to Water-Soluble ZnO Nanocrystals: A Mechanochemical Approach, ACS Sustain. Chem. Eng., 2021, 9(4), 1540–1549 CrossRef.
- Q. Ma, et al., Solid SiO2-sealed mesoporous silica for synergistically combined use of inorganic and organic filters to achieve safe and effective skin protection from all-band UV radiation, ACS Appl. Mater. Interfaces, 2023, 15(9), 12209–12220 Search PubMed.
- O. Mazaheri, et al., Solid-state encapsulation of urea via mechanochemistry-driven engineering of metal–phenolic networks, Chem. Mater., 2023, 35(18), 7800–7813 Search PubMed.
- Q. Zhang, et al., Zinc oxide nanorods for light-activated gas sensing and photocatalytic applications, ACS Appl. Nano Mater., 2023, 6(19), 17445–17456 CrossRef CAS.
- Y. Liu, et al., Guest release from coordination assemblies in the solid state, Chem, 2024, 10(5), 1502–1515 CAS.
- L. Zheng, et al., Unveiling the electronic interaction in ZnO/PtO/Pt nanoarrays for catalytic detection of triethylamine with ultrahigh sensitivity, ACS Appl. Mater. Interfaces, 2020, 12(41), 46267–46276 Search PubMed.
- R. M. Dragoman, et al., Surface-engineered cationic nanocrystals stable in biological buffers and high ionic strength solutions, Chem. Mater., 2017, 29(21), 9416–9428 CrossRef CAS PubMed.
- G. Zhang, et al., Influence of anchoring ligands and particle size on the colloidal stability and in vivo biodistribution of polyethylene glycol-coated gold nanoparticles in tumor-xenografted mice, Biomaterials, 2009, 30(10), 1928–1936 CrossRef CAS PubMed.
- R. Dubadi, S. D. Huang and M. Jaroniec, Mechanochemical synthesis of nanoparticles for potential antimicrobial applications, Materials, 2023, 16(4), 1460 CrossRef CAS PubMed.
- H. Wang, et al., Applications of metal–phenolic networks in nanomedicine: a review, Biomater. Sci., 2022, 10(20), 5786–5808 RSC.
- J. Chen, et al., Metal-phenolic networks as broad-spectrum antioxidant coatings for hemoglobin nanoparticles working as oxygen carriers, Chem. Mater., 2022, 34(20), 9200–9211 CrossRef CAS.
- C. L. Ventola, Progress in nanomedicine: approved and investigational nanodrugs, Pharm. Ther., 2017, 42(12), 742 Search PubMed.
- S. Soares, et al., Nanomedicine: principles, properties, and regulatory issues, Front. Chem., 2018, 6, 360 Search PubMed.
- G. Lin, et al., Designing metal–phenolic networks in biomedicine, Appl. Mater. Today, 2025, 45, 102822 Search PubMed.
- Y. Xiao, et al., Mechanochemical synthesis of cocrystal: From mechanism to application, Cryst. Growth Des., 2023, 23(6), 4680–4700 Search PubMed.
- N. Schultheiss and A. Newman, Pharmaceutical cocrystals and their physicochemical properties, Cryst. Growth Des., 2009, 9(6), 2950–2967 Search PubMed.
- A. Chettri, et al., Pharmaceutical co-crystals: A green way to enhance drug stability and solubility for improved therapeutic efficacy, J. Pharm. Pharmacol., 2024, 76(1), 1–12 Search PubMed.
- A. Delori, T. Friščić and W. Jones, The role of mechanochemistry and supramolecular design in the development of pharmaceutical materials, CrystEngComm, 2012, 14(7), 2350–2362 Search PubMed.
- Z. Wu, T. Cheng and Z. L. Wang, Self-powered sensors and systems based on nanogenerators, Sensors, 2020, 20(10), 2925 CrossRef CAS PubMed.
- D. Zhang, et al., Diversiform gas sensors based on two-dimensional nanomaterials, Nano Res., 2023, 16(10), 11959–11991 CrossRef.
- H. Yang, et al., Utilization of sea sand for preparation of high-performance CoAl2O4 composite pigments via a cleaner mechanochemistry route, ACS Sustain. Chem. Eng., 2022, 10(29), 9553–9564 CrossRef CAS.
- S. Sarfudeen, et al., A Novel Mechano-Synthesized Zeolitic Tetrazolate Framework for a High-Performance Triboelectric Nanogenerator and Self-Powered Selective Neurochemical Detection, ACS Appl. Mater. Interfaces, 2024, 16(19), 24851–24862 CrossRef CAS PubMed.
- Y. Qin, et al., Interstitial/substitutional Se doping in nanosheet heterostructures for sensing NH3: Experimental and theoretical study, ACS Appl. Nano Mater., 2023, 6(20), 19290–19299 Search PubMed.
- Y. Hidalgo-Rosa, et al., Expanding the knowledge of the selective-sensing mechanism of nitro compounds by luminescent terbium metal–organic frameworks through multiconfigurational ab initio calculations, J. Phys. Chem. A, 2022, 126(39), 7040–7050 Search PubMed.
- V. Šepelák, et al., Mechanochemical reactions and syntheses of oxides, Chem. Soc. Rev., 2013, 42(18), 7507–7520 Search PubMed.
- M. Sopicka-Lizer, High-energy Ball Milling: Mechanochemical Processing of Nanopowders, Elsevier, 2010 Search PubMed.
- A. S. Burange, Z. A. Alothman and R. Luque, Mechanochemical design of nanomaterials for catalytic applications with a benign-by-design focus, Nanotechnol. Rev., 2023, 12(1), 20230172 Search PubMed.
- Y. Nailwal and A. Rapid, et al., Sustainable, One-step Mechanochemical Strategy for Synthesizing Gold Nanoparticle-Doped Covalent Organic Frameworks, Chem.–Eur. J., 2025, 31(22), e202500339 CrossRef CAS PubMed.
- J. E. Ogbezode, et al., A narrative review of the synthesis, characterization, and applications of iron oxide nanoparticles, Discover Nano, 2023, 18(1), 125 CrossRef CAS PubMed.
- V. Chaudhary, et al., Mechanochemical synthesis of iron and cobalt magnetic metal nanoparticles and iron/calcium oxide and cobalt/calcium oxide nanocomposites, ChemistryOpen, 2018, 7(8), 590–598 CrossRef CAS PubMed.
- O. Y. Posudievsky, et al., Facile mechanochemical preparation of nitrogen and fluorine co-doped graphene and its electrocatalytic performance, Carbon, 2019, 152, 274–283 CrossRef CAS.
- T. Tsuzuki, Commercial scale production of inorganic nanoparticles, Int. J. Nanotechnol., 2009, 6(5–6), 567–578 CrossRef CAS.
- A. Ali, et al., Synthesis, characterization, applications, and challenges of iron oxide nanoparticles, Nanotechnol. Sci. Appl., 2016, 49–67 Search PubMed.
- J. De Carvalho, et al., Synthesis of magnetite nanoparticles by high energy ball milling, Appl. Surf. Sci., 2013, 275, 84–87 Search PubMed.
- S. Wu, T. Wang and H. Xu, Regulating heterogeneous catalysis of gold nanoparticles with polymer mechanochemistry, ACS Macro Lett., 2020, 9(9), 1192–1197 CrossRef CAS PubMed.
- A. Drmota, et al., Microemulsion method for synthesis of magnetic oxide nanoparticles, Microemulsions—An Introduction to Properties and Applications, ed. Najjar, R., 2012, pp. 191–214 Search PubMed.
- I. Mitar, et al., Rapid microwave method for synthesis of iron oxide particles under specific conditions, Crystals, 2021, 11(4), 383 CrossRef CAS.
- M. J. Rivera-Chaverra, et al., Synthesis of oxide iron nanoparticles using laser ablation for possible hyperthermia applications, Nanomaterials, 2020, 10(11), 2099 Search PubMed.
- S. Pagola, Outstanding advantages, current drawbacks, and significant recent developments in mechanochemistry: A perspective view, Crystals, 2023, 13(1), 124 Search PubMed.
- R. Luque, et al., Evaluation of biomass-derived stabilising agents for colloidal silver nanoparticles via nanoparticle tracking analysis (NTA), RSC Adv., 2013, 3(19), 7119–7123 Search PubMed.
- Z. Shalabayev, et al., Sulfur-mediated mechanochemical synthesis of spherical and needle-like copper sulfide nanocrystals with antibacterial activity, ACS Sustain. Chem. Eng., 2019, 7(15), 12897–12909 CrossRef CAS.
- T. Iwasaki, et al., Synthesis of titanosilicate TS-1 crystals via mechanochemical route using low cost materials, Microporous Mesoporous Mater., 2012, 150, 1–6 CrossRef CAS.
- S. L. Tang, R. L. Smith and M. Poliakoff, Principles of green chemistry: PRODUCTIVELY, Green Chem., 2005, 7(11), 761–762 Search PubMed.
- P. F. de Oliveira, et al., A mechano-colloidal approach for the controlled synthesis of metal nanoparticles, Chem. Commun., 2019, 55(95), 14267–14270 Search PubMed.
- Y. Xia, X. Xia and H.-C. Peng, Shape-controlled synthesis of colloidal metal nanocrystals: thermodynamic versus kinetic products, J. Am. Chem. Soc., 2015, 137(25), 7947–7966 CrossRef CAS PubMed.
- M. Klimakow, et al., Mechanochemical synthesis of metal− organic frameworks: a fast and facile approach toward quantitative yields and high specific surface areas, Chem. Mater., 2010, 22(18), 5216–5221 CrossRef CAS.
- J. Shen and R. Blachnik, Mechanochemical syntheses of antimony selenide, tin selenides and two tin antimony selenides, Thermochim. Acta, 2003, 399(1–2), 245–246 Search PubMed.
- Y. X. Shi, et al., The first synthesis of the sterically encumbered adamantoid phosphazane P4 (NtBu) 6: enabled by mechanochemistry, Angew. Chem., 2016, 128(41), 12928–12932 CrossRef.
- M. J. Muñoz-Batista, et al., Mechanochemistry: Toward Sustainable Design of Advanced Nanomaterials for Electrochemical Energy Storage and Catalytic Applications, ACS Publications, 2018 Search PubMed.
- A. M. Belenguer, et al., Solid-state dynamic combinatorial chemistry: reversibility and thermodynamic product selection in covalent mechanosynthesis, Chem. Sci., 2011, 2(4), 696–700 RSC.
- S. Arfelis, et al., Linking mechanochemistry with the green chemistry principles, Heliyon, 2024, 10(14), e34655 CrossRef CAS PubMed.
- T. Friščić, et al., Highlights from Faraday discussion 170: Challenges and opportunities of modern mechanochemistry, Montreal, Canada, 2014, Chem. Commun., 2015, 51(29), 6248–6256 RSC.
- H. Mio, J. Kano and F. Saito, Scale-up method of planetary ball mill, Chem. Eng. Sci., 2004, 59(24), 5909–5916 CrossRef CAS.
- R. A. Buyanov, V. V. Molchanov and V. V. Boldyrev, Mechanochemical activation as a tool of increasing catalytic activity, Catal. Today, 2009, 144(3–4), 212–218 CrossRef CAS.
- P. Y. Butyagin, Kinetics and nature of mechanochemical reactions, Russ. Chem. Rev., 1971, 40(11), 901 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |