 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Multi-scale structural design of electromagnetic wave absorbers: pathways to improved performance
Yang Guanabc,
Yongpeng Wua,
Jianyu Zhoua,
Kai Yaoa,
Ruolin Liua,
Yang Yang*a and
Wei Lu *a
*a
aShanghai Key Lab. of D&A for Metal-Functional Materials, School of Materials Science & Engineering, Tongji University, Shanghai 201804, China. E-mail: yangyang_@tongji.edu.cn; weilu@tongji.edu.cn
bShanghai Institute of Ceramics, Chinese Academy of Sciences, Shanghai 200050, China
cUniversity of the Chinese Academy of Sciences, Beijing 100049, China
First published on 26th March 2026
Abstract
With the rapid advancement of wireless communication technologies and the proliferation of Internet of Things (IoT) devices over the past decade, electromagnetic interference (EMI) has emerged as a significant challenge. EMI can degrade the performance, stability, and lifespan of advanced electronics and even poses potential health risks. One of the most effective strategies to mitigate this issue is the development of highly efficient electromagnetic wave absorbers (EMAs), a field that has garnered considerable attention. However, achieving the necessary efficiency for widespread adoption remains a challenge. The internal structure of EMAs plays a pivotal role in determining their absorption performance. This review explores the fundamental principles of electromagnetic wave absorption and highlights recent advancements in optimizing internal structures across various length scales, from the atomic to the macroscopic level. We examine design strategies, structure–performance relationships, and fabrication methods for tuning absorption efficiency, either independently or in combination. Finally, we address key challenges and future directions for the next generation of EMAs, offering insights into their potential for practical applications.
Wider impactThis review focuses on the significant progress in the multi-scale structural design of electromagnetic wave absorbers (EMAs), highlighting the breakthroughs in enhancing their absorption performance. The research covers the atomic/molecular scale, nanoscale, microscale, and macroscale. Multi-scale structural design plays a decisive role in determining the electromagnetic parameters, impedance matching characteristics, and loss mechanisms of materials, providing a broad design space for breaking through the physical limits of traditional materials in terms of “thinness, lightness, wideband, and strength” performance. These breakthroughs are crucial for advancing the research on intelligent and integrated EMAs and offer innovative solutions to address the increasingly severe demands of electromagnetic interference (EMI) and radar stealth. Through the study of multi-scale structural design of EMAs, this review has made important contributions to the development of advanced EMAs to alleviate electromagnetic pollution. |
1. Introduction
With the rapid development of 6G mobile communication technology, the Internet of Things (IoT) and artificial intelligence (AI) technology, the extensive application of electromagnetic waves (EMWs) in wireless communication, radar detection and electronic devices has made the problem of electromagnetic interference (EMI) increasingly prominent. EMI adversely impacts the reliable functioning of electronic devices, resulting in diminished efficiency, and also presents possible health hazards to humans.1,2 Meanwhile, in the military field, the application of precision-guided technology has driven the rapid development of radar stealth technology.3 Therefore, the development and application of new multi-functional EMAs have become some of the important study directions in current scientific studies across various countries.4 Excellent EMAs must fulfill criteria of having minimal thickness, light weight, robust absorption, and broad frequency band, while also ensuring anti-oxidation, corrosion resistance, high-temperature resistance and environmental friendliness.5,6 In recent years, to enhance the overall performance of EMAs, researchers have concentrated on the structural design of these materials and conducted significant investigations into the correlation between the structure and performance.7,8Over the past several decades, tremendous progress has been achieved in the research of EMAs, and the related technology development has shown a vigorous trend.9,10 On the basis of the various loss mechanisms of EMAs, traditional EMAs can mainly be classified into three types: dielectric loss type,11,12 resistive loss type,13 and magnetic loss type materials.14,15 Dielectric loss type EMAs include ceramic materials (e.g., SiC, BaTiO3) and polymer composites.16,17 These materials exhibit superior high-frequency absorption performance, outstanding high-temperature resistance and excellent chemical stability.18 However, they suffer from certain limitations, including difficulties in dielectric constant regulation, and for some ceramic materials, notable brittleness and poor processability.19 Resistive loss type EMAs primarily include graphene,20,21 and carbon nanotubes (CNTs).22,23 These materials demonstrate promising broadband absorption capabilities, widely tunable electrical conductivity and excellent potential for functionalization modifications.24 However, they are prone to severe agglomeration and frequently demonstrate unsatisfactory impedance matching performances.25 Magnetic loss type EMAs primarily consist of ferrites, metallic magnetic powders and their alloys.26 These materials typically exhibit strong absorption intensity and low-cost advantages, but their practical applications are significantly limited by high density and susceptibility to corrosion. However, as the requirements for thin, light, wide, and strong absorption become increasingly strict, material design based solely on chemical composition modulation has become increasingly unable to meet the needs of practical applications.27,28 The fundamental challenge lies in the inherent trade-off between impedance matching and attenuation capacity, which is difficult to decouple within a single chemical composition modulation. Therefore, shifting the design paradigm from composition-driven to structure-driven through multi-scale structural design is no longer just an option, but a necessity.29,30
Multi-scale structural designs of EMAs include atomic/molecular scale, nanoscale, microscale and macroscale designs.31 Through the design of multi-scale structures, EMAs can exhibit unique structures and properties, effectively regulating EMW absorption, and providing an effective approach for constructing broadband EMAs.32 In particular, atomic/molecular scale design includes an atomic dispersion system,33 and defect engineering.34,35 At this scale, it is possible to precisely control the intrinsic electromagnetic parameters of materials, and by designing defects and sites, one can actively create the desired physical mechanisms. In addition, nanoscale design encompasses the regulation of nanostructure morphology,36 and the design of low-dimensional nanomaterials.37 By constructing a hollow structure,38 porous structure,39 and core–shell structure,40 the morphology of nanostructures can be controlled, and this can enrich the attenuation mechanisms of EMAs. Low-dimensional nanomaterials can enhance the dielectric loss capacity of EMAs. Furthermore, the microscale design includes designs such as aerogels,41 porous foams,42 fiber network materials,43 layered structures,44 and oriented arrangement structures. This scale solves the problems of traditional materials' tendency to agglomerate and their single functionality, and endows the materials with lightweight and high-loss properties. Additionally, the macroscale design includes periodic arrays,158 frequency-selective surfaces (FSSs),160 and impedance gradient regulation.162 Macroscale design no longer merely focuses on the chemical composition of the material itself; instead, it constructs macroscopic physical structures to manage and control EMWs, thereby achieving breakthroughs in EMW absorption performance.
This work systematically reviews the latest progress in the multi-scale structural design of EMAs. This paper initially introduces the basic principles of EMW absorption, then provides a comprehensive overview of EMAs with different scale structure types; layer by layer, it analyzes the structural design strategies from the atomic scale to the macro scale and how their regulation affects performance; finally, in view of the challenges faced by EMAs in some emerging application scenarios, the future development direction of broadband EMAs is discussed. This review aims to provide theoretical guidance for the development of the next generation of intelligent tunable, wideband and efficient EMAs (Fig. 1).
 | ||
| Fig. 1 Multi-scale structural design of EMAs, including design strategies at atomic/molecular scale, nanoscale, microscale, and macroscale. Images for atomic/molecular scale: reproduced with permission from ref. 93. Copyright (2021) John Wiley and Sons; reproduced with permission from ref. 103. Copyright (2026) Elsevier; reproduced with permission from ref. 109. Copyright (2023) Elsevier; reproduced with permission from ref. 114. Copyright (2024) John Wiley and Sons. Images for nanoscale: reproduced with permission from ref. 117. Copyright (2020) Elsevier; reproduced with permission from ref. 123. Copyright (2025) Elsevier; reproduced with permission from ref. 137. Copyright (2023) Elsevier; reproduced with permission from ref. 150. Copyright (2023) Springer Nature. Images for microscale: reproduced with permission from ref. 153. Copyright (2023) Elsevier; reproduced with permission from ref. 155. Copyright (2025) Elsevier; reproduced with permission from ref. 157. Copyright (2025) Elsevier; reproduced with permission from ref. 163. Copyright (2025) Elsevier. Images for macroscale: reproduced with permission from ref. 168. Copyright (2024) John Wiley and Sons; reproduced with permission from ref. 169. Copyright (2017) Elsevier; reproduced with permission from ref. 174. Copyright (2023) Elsevier; reproduced with permission from ref. 176. Copyright (2024) Donghua University. | ||
2. EMW absorption theory
The core of EMW absorption theory lies in investigating how to maximize the conversion of incident EMW energy into other forms of energy through specific absorbers, thereby reducing or eliminating the reflection and transmission of EMWs.45 The performance of EMAs is directly dictated by the degree of impedance matching between the absorber and free space, as well as the absorber's own electromagnetic loss capability.46When EMWs interact with EMAs, it can be described based on Maxwell's equations: when the EM field oscillates with the EMAs, a relative response is generated, leading to energy dissipation of the EMAs.47 Maxwell's equations reveal the constitutive relationship between EMAs and EMWs. Their response characteristics are jointly described by complex permittivity (εr) and complex permeability (μr), and these complex parameters comprehensively manifest the electromagnetic energy storage and dissipation characteristics of EMAs. The formulas for εr and μr are as follows:48,49
| εr = ε′ − jε″ | (1) |
| μr = μ′ − jμ″ | (2) |
2.1. Electromagnetic absorption performance evaluation
Compared to electromagnetic shielding materials, EMAs generate less secondary pollution when processing EMWs. The key indicator for evaluating the performance of EMAs is reflection loss (RL).51 A more negative RL value indicates a stronger absorption capability. Typically, RL ≤ −10 dB (which refers to a 90% absorption rate) is considered effective absorption, while RL ≤ −20 dB (99% absorption rate) indicates exceptional performance.52 The effective absorption bandwidth (EAB), which corresponds to the frequency range where RL ≤ −10 dB, serves as another crucial metric for evaluating the performance of EMAs. Modern applications urgently require EMAs to possess a wider EAB. According to transmission line theory, RL can be calculated using the following formulas:53,54
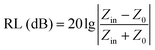 | (3) |
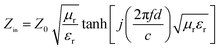 | (4) |
The recently proposed delta function method in research offers an innovative solution for the assessment of electromagnetic impedance matching degree, and the formula is as follows:57
| |Δ| = |sinh2(Kfd) − M| | (5) |
2.2. EMW attenuation mechanisms
The attenuation mechanism of EMAs is the core physical basis. The attenuation mechanism can mainly be divided into two categories: dielectric loss and magnetic loss.59 The detailed attenuation mechanisms of EMW absorption are shown in Fig. 2. The dielectric loss tangent (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δe = ε″/ε′) and the magnetic loss tangent (tan
δe = ε″/ε′) and the magnetic loss tangent (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δm = μ″/μ′) can be used to evaluate the dielectric loss and magnetic loss capabilities, respectively.60 Different attenuation mechanisms determine the way in which EMAs convert the energy of EMWs into other forms of energy and dissipate it.
δm = μ″/μ′) can be used to evaluate the dielectric loss and magnetic loss capabilities, respectively.60 Different attenuation mechanisms determine the way in which EMAs convert the energy of EMWs into other forms of energy and dissipate it.
 | ||
| Fig. 2 Schematic illustration of possible EMW absorption mechanisms. Reproduced with permission from ref. 85. Copyright (2023) Royal Society of Chemistry; reproduced with permission from ref. 105. Copyright (2025) Elsevier; reproduced with permission from ref. 137. Copyright (2023) Elsevier; reproduced with permission from ref. 139. Copyright (2022) Elsevier. | ||
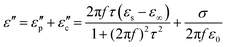 | (6) |
Conductive loss refers to the process where the charge carriers within the material undergo directional migration under the alternating electric field of the incident EMWs, forming an electric current. The material's own resistance causes Joule heating during the current flow, thereby converting the electromagnetic energy into thermal energy.63 High conductivity EMAs have a high electrical conductivity, so the ε″ value is also high, indicating strong dielectric loss. However, high conductivity can lead to poor impedance matching and a decline in EMW absorption performance.64,65
Polarization loss refers to the phenomenon where the electric dipoles in a material will shift and rearrange under the influence of an alternating electric field, attempting to keep up with the changes in the direction of the electric field.66 However, this polarization process lags behind the changes in the electric field, and this lag results in the dissipation of energy in the form of heat.67 Compared to conductive loss, polarization loss has more manifestations, specifically including interface polarization, dipole polarization, electron polarization and ionic polarization.68 However, electron polarization and dipole polarization usually occur in the high-frequency region (103–106 GHz), and their relaxation times are very short (10−12–10−16 s). Therefore, in most cases, electron polarization and dipole polarization are not applicable, and dipole polarization and interface polarization are the primary attenuation mechanisms of polarization loss.69,70 Dipole polarization usually corresponds with the doping of non-metallic elements and defects. Interface polarization usually occurs at heterojunctions. Space charges accumulate at the heterojunction, forming a structure similar to a capacitor, thereby generating interface polarization and enhancing the ability to attenuate incident waves. Cole–Cole semicircles have been defined by Debye's theory to clarify the relationship between ε″ and ε′ to characterize the relaxation process:71,72
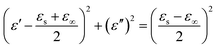 | (7) |
Generally speaking, magnetic losses include various types, such as domain wall resonance, hysteresis loss, natural resonance, and eddy current loss.76 Domain wall resonance exists in multiferroic materials. The domain walls are forced to vibrate under weak alternating magnetic fields. When the external field frequency approaches the intrinsic vibration frequency of the domain walls, resonance occurs, resulting in energy dissipation. Domain wall resonance mainly occurs in the low-frequency range and has a relatively weak contribution in the high-frequency range, so it can be ignored.75 Hysteresis loss is attributed to the irreversible movement of magnetic domains or the irreversible displacement of domain walls. It usually occurs under weak magnetic fields. Hysteresis loss is typically very weak and is not the main loss mechanism.75 Natural resonance is the most significant magnetic loss mechanism in ferroelectric and other strongly anisotropic magnetic crystalline materials. Under the influence of an applied alternating magnetic field, the magnetic moment undergoes precession around its magnetic anisotropy field, and its resonance frequency satisfies the following formula:77
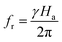 | (8) |
According to Faraday's law of electromagnetic induction, an alternating magnetic field will induce a closed ring-shaped current, known as eddy currents, within the conductor. When these eddies flow through materials with a certain resistivity, they generate Joule heat, thereby converting magnetic energy into thermal energy. The formula for calculating the eddy current loss coefficient (C0) is as follows:79
| C0 ≈ 2πμ0μ′2σD2f = μ″(μ′)−2f−1 | (9) |
2.3. EMW absorption influence factors
It is widely known that advanced EMAs can achieve good impedance matching and efficient EMW attenuation. Therefore, how to coordinate impedance matching and EMW attenuation is the core issue in practical applications. The attenuation constant (α) can be calculated by the following formula:80
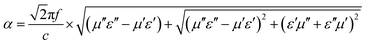 | (10) |
Dissipating reflected EMWs through the interference of EMAs is another key factor. The quarter-wavelength (λ/4) matching mechanism is described as follows:83
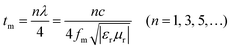 | (11) |
3. Multi-scale structural design of EMAs
As we discussed above, EMAs modulated by a single chemical component struggle to satisfy the stringent requirements of high-performance EMAs. In recent years, researchers have gradually realized that the design of high-efficiency EMAs is a complex systematic form of engineering that must span multiple structural scales from the atomic to the macro level, and requires hierarchical and integrated detailed design of the EMAs. This work focuses on the design of the following four types of scale structures: (i) atomic/molecular scale, (ii) nanoscale, (iii) microscale and (iv) macroscale.3.1. Atomic/molecular scale design
The atomic/molecular scale is the cornerstone of material design, determining the intrinsic electromagnetic parameters of EMAs. Design and control at this scale are crucial for the realization of ultrathin, lightweight, wideband and highly EMAs in modern technology.N-doped hollow carbon spheres (NHCS@Co-SAs) with atom-dispersed Co-SAs were designed by Li et al. using the template method (Fig. 3(a)). Multiple techniques for characterization, especially high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) and X-ray absorption fine structure (XAFS), were employed to jointly confirm that the Co atoms existed in the carbon framework in an atomic monomer form and coordinated with four N atoms (Co–N4 configuration). When the filling loading was 8 wt% and the thickness was 2 mm, NHCS@Co-SAs exhibited outstanding EMW absorption performance, with a minimum reflection loss (RLmin) of −44.96 dB and a corresponding EAB of 5.25 GHz. The introduction of Co-SAs regulated the electron structure of the carbon matrix, thereby enhancing the electrical conductivity; the Co–N4 sites and the high content of pyridine N/graphite N act as polarization centers, significantly enhancing dipole and interface polarization. Density functional theory (DFT) simulations indicate that the Co–N4 sites and the surrounding N doping may significantly increase the density of states near the Fermi level and cause significant charge rearrangement, resulting in a dipole moment. This, in turn, enhances both conduction loss and polarization loss simultaneously. The interaction mechanism between the HCSs and incident EMW was investigated under actual application conditions (Fig. 3(b)–(f)). By simulating the surface current density and power loss density of three samples, the reasons for the enhanced EMW absorption properties of NHCS@Co-SAs were explained. This work elucidates the dual mechanism by which M-SAs optimize impedance matching and enhance dielectric loss (conductive loss and polarization loss) by regulating the electronic structure of the carbon matrix. It also deepens understanding of the EMW absorption mechanism at the atomic scale.88 Sun et al. have successfully constructed multi-void composite microplates (CoNxC), which exhibited excellent EMW absorption performance, with a RLmin of −63.4 dB. In the CoNxC, the stable coexistence of Co nanoparticles and Co-SAs has been successfully achieved, forming rich heterogeneous interfaces and SA sites (Fig. 3(h)–(k)). The charges accumulate and rearrange at these heterogeneous interfaces, resulting in intense interface polarization, which promotes electron transport and enhances conductive loss. By controlling the N doping content, various cobalt–nitrogen coordination structures (such as CoN2, CoN3 and CoN4) can be obtained (Fig. 3(l) and (m)). These structures break the regular charge distribution of the graphite surface, forming electric dipole moments and significantly enhancing dipole polarization. Theoretical calculations and experimental results reveal that the exceptional properties of CoNxC are related to the synergistic effect of the heterogeneous interface and the SA configuration.89 Xu et al. achieved precise anchoring of Er single atoms (Er-N6) on N-doped hollow carbon nanofibers. Both theoretical and experimental results showed that the high-coordination Er-N6 configuration improved conductivity and polarization loss, resulting in significantly enhanced EMW absorption performance.90 Zhang et al. successfully dispersed Ni-SAs uniformly throughout the N-doped hierarchical porous carbon flower-like structure (Ni-SA/HPCF). This research successfully achieved the homogeneous dispersion of Ni-SAs within the carbon framework while preserving the hierarchical porous flower-like structure. The EAB reached 5 GHz, and the RLmin was −53.2 dB. The excellent EMW absorption performance of Ni-SA/HPCF is attributed to its outstanding dielectric loss capability. Its conductivity and polarization loss are significantly higher than those of the control samples. DFT calculations indicate that the Ni–N4 sites can reduce the energy band gap, increase the density of states (DOS), and enhance the conductance loss; at the same time, they cause the redistribution of charges, forming electric dipoles, and enhancing the polarization loss.91 In practical applications under extreme conditions, the development of lightweight and acid–base resistant EMAs has attracted much attention. Lou et al. successfully synthesized N-doped porous carbon (NC) composites with hierarchical pore structures, and achieved the confined anchoring of Zn-SAs during the high-temperature carbonization process. NC-1000 exhibited outstanding EMW absorption performance and outstanding chemical stability. Its RLmin was −50.5 dB, and the EAB was 5.1 GHz. After being immersed in strong acid and strong alkali solutions, the attenuation of its EMW absorption performance was less than 15%. The analyses confirmed that Zn exists in a monatomic form with a coordination number of 4, and the length of the Zn–N bond is approximately 2.01 Å (Fig. 3(n)–(p)). Zn-SAs can regulate the local electronic structure, providing abundant polarization sites, thereby enhancing polarization loss and electrical conduction loss.92
 | ||
| Fig. 3 (a) Illustration of the synthesis of NHCS@Co-SAs. (b)–(d) The surface current density, and (e)–(g) the power loss density of NHCS@Co-SAs, NHCS@Co3O4 and NHCS. Reproduced with permission from ref. 88. Copyright (2023) John Wiley and Sons. (h)–(k) Charge density distribution on the CoN2 surface, CoN3 surface, CoN4 surface and Co/C interface models, (l) Bader charges of the Co atom and neighboring N and C atoms in each model (gray: carbon, blue: carbon, and purple: cobalt), (m) a rough estimate of the quasi-electric dipole moment of various bonding configurations from the corresponding models. Reproduced with permission from ref. 89. Copyright (2022) Elsevier. (n) Normalized XANES spectra of NC-1000, (o) Fourier transform (FT)-EXAFS spectra of Zn foil and NC-1000, and (p) EXAFS fitting curves of NC-1000 in the R-space. Reproduced with permission from ref. 92. Copyright (2024) Tsinghua University Press. | ||
Huang et al. successfully prepared broadband EMAs with magnetic–electrostatic synergy by anchoring Zn-SAs onto the N-doped carbon shell derived from a MOF and constructing a core–shell structure (Fe3O4@Zn–N–carbon, FZNC) with a magnetic Fe3O4 core (Fig. 4(a)). Its RLmin was −61.9 dB, and the EAB was 11.5 GHz. The dispersion states of Zn species at the atomic scale were investigated by aberration-corrected HAADF-STEM. The intensity profiles further confirmed well-dispersed atomic Zn species in the N-doped carbon materials (Fig. 4(b)–(g)). The interface polarization is maximized by the dielectric Zn–N doped carbon layer, and the dielectric dissipation is maximized by both the interface polarization effect and the Zn-SA doping effect.93 The SA strategy has been proven to be effective in regulating the EMW absorption performance. However, the clear correlation connection between the electronic configuration of M-SAs and the physical loss mechanisms has not yet been established, especially at the atomic scale. Liang et al. synthesized Ni-SAs/N-doped carbon model (Ni-SAsx/NC) EMAs through a ligand polymerization strategy (Fig. 4(h)). The HAADF-TEM analysis results confirmed the existence of isolated single-Ni sites in the structure. The addition ratio of the Ni salts also affected the loading amount of Ni-SAsx in the N-doped carbon materials. The individual Ni-SAs dispersed at the atomic level induce polarization loss through the unpaired electrons in the excess Ni 3d orbitals, significantly enhancing the EMW absorption performance (Fig. 4(i)). The EAB of the SA absorber (Ni-SAs3/NC) reached 7.08 GHz, covering the entire Ku band. This research proposes a novel idea for in-depth design of EMAs at the atomic scale.94
 | ||
| Fig. 4 (a) Schematic preparation process of Fe3O4@Zn–N–carbon composites. (e) TEM image, (b) HRTEM image, (c) enlarged HAADF-STEM image and (d) intensity profiles of FZNC-2 obtained in the dotted rectangle regions 1# and 2# in (c). (f) Reconstructed phase images from (e), and (g) the corresponding charge density distribution line profiles in the region of the green arrow for the FZNC-2 composites. Reproduced with permission from ref. 93. Copyright (2021) John Wiley and Sons. (h) Schematic illustration of the carbon trapping of metal atoms. (i) Continuous evolution mechanism of microscopic electromagnetic loss based on single-atom model absorbers. Reproduced with permission from ref. 94. Copyright (2022) John Wiley and Sons. (j) Schematic flow diagram of the stepwise process for the preparation of DA absorbers. (k) Experimental and fitting EXAFS curves for Ni/Cu-NC. (l) Structural models of Ni/Cu-NC. Reproduced with permission from ref. 96. Copyright (2024) John Wiley and Sons. | ||
Metal dual atoms (M-DAs), characterized by flexible structural tunability and high atomic utilization, show significant promise for atom-level coordination engineering.94 He et al. proposed an efficient strategy to enhance the EMW absorption performance of M-DA dispersed carbon-based composites, and successfully prepared EMAs (ZnCo-NC@rGO) with atom dispersion. Zn- and Co-SAs could function as effective polarization centers, effectively promoting the polarization of dipoles. The increased heterogeneous interfaces, increased point defects, and formation of capacitor-like structures induced by the bimetallic Zn/Co sites can effectively suppress excessive conductive loss and enhance magnetic loss, thereby achieving perfect impedance matching.95 Liang et al. successfully fabricated a high-density Ni–Cu pair DA coupled EMAs (Ni/Cu-NC) on a N-rich carbon substrate (NC) using a bottom-up stepwise annealing method (Fig. 4(j)). The Ni/Cu-NC achieved an EAB of 7.8 GHz and a RLmin of −70.96 dB. The results obtained from HAADF-STEM and XAFS analysis indicate that a N3–Ni1–Cu1–N3 coordination spatial structure with coupled DA pairs was successfully synthesized in Ni/Cu-NC, which breaks the traditional D4h symmetry configuration (Fig. 4(k) and (l)). DFT calculations indicate that the cooperative effect of the Ni/Cu pairs’ diatomic sites leads to the transfer of electrons from the N site to the Cu site, resulting in local charge redistribution, thereby generating strong asymmetric polarization loss and significantly enhancing the EMW absorption performance. This paper provides a theoretical basis for understanding the polarization loss mechanism at the atomic scale.96 The above research on atomic dispersion materials has paved the way for the rational design of lightweight and high-performance EMAs at the atomic scale.
Doping engineering, as an innovative strategy for optimizing material properties by regulating crystal structures and physical–chemical properties, has received extensive attention in recent years.97 This technique, through heteroatom doping, can effectively trigger interatomic charge transfer and local charge redistribution at the atomic scale, enhance electronic interactions, and thereby significantly improve EMW absorption performance.108 The S and P-doped graphitic carbon nitride (g-C3N4) called SP-PCN prepared by Lv et al. exhibited excellent electromagnetic absorption performance under high-temperature conditions. At 150 °C, the RLmin was −50.7 dB, and the EAB was 6 GHz.98 Tao et al. used the existing in situ N and O doping models to achieve exogenous S doping through external thermal driving. During this process, the introduced S atoms preferentially replace the pyrrole N in the material, while also causing some O atoms to be eliminated and generating O vacancies. These changes induce a dielectric polarization of 3–6 times greater than the intensity of conduction loss, and significantly improve the EMW absorption performance. The DFT analysis demonstrated the catfish effect connecting anion species and vacancy defects, proving that S doping is more likely to trigger vacancy formation. The results showed that vacancies have stronger electronic absorption capabilities, have greater influence on the charge shifts of neighboring atoms, and may even gather electrons from neighboring S regions (Fig. 5(a)–(c)). The EAB of this EMA was as high as 9.03 GHz, and the RLmin was −64.05 dB.99 Tian et al. first proposed a unique doping engineering method, using the unsaturated S doping process, to prepare S-doped CoxP@C composites with 6-coordination and 9-coordination environments. Theoretical calculations indicated that the S–C and S–Co6 configurations in S–CoP@SC interact cooperatively in the 6-coordination environment, effectively improving the charge transport properties and changing the charge distribution, thereby enhancing the conductivity and polarization loss, and effectively improving the EMW absorption performance of S–CoP@SC composites (Fig. 5(d)).100 Huang et al. successfully developed surface-modified NdCo composites based on N-doped carbon nanotubes (CNTs). Due to its heterostructure and doping defects, as well as the constructed robust conductive network, the N-doped CNTs significantly enhanced the dielectric loss performance. The presence of magnetic NdCo further provided magnetic loss for the attenuation of EMW.101 Fan et al. successfully synthesized Cl−-doped PPy nanotubes (NTs) via a soft template-assisted oxidation method (Fig. 5(e)). Theoretical results indicated that the band structure of PPy could be adjusted and its conductivity enhanced by modulating the Cl− doping level. There are many lattice defects in Cl−-doped PPy nanotubes (PPy-M-4), which act as dipole centers, enhancing dipole/defect polarization and improving EMW absorption performance (Fig. 5(f)). PPy-M-4 exhibits excellent EMW absorption (3.32 GHz mm−1) and radar cross section (RCS) reduction performance (33.45 dB m2). Furthermore, by precisely controlling the Cl− doping level and nanotube structure, Cl−-doped PPy NT/silicone membranes show high thermal conductivity (3.597 W m−1 K−1) (Fig. 5(g)).102 Wang et al. successfully fabricated mesoporous N-doped mesoporous hollow nanospheres (HNSs) by using a template-assisted carbonization-etching process combined with the N doping strategy. The prepared N-doped mesoporous HNSs exhibit advanced EMW absorption performance and good corrosion resistance. The intrinsic defects and functional group defects of amorphous carbon, combined with those generated by N doping, collectively form a large number of dipoles, which leads to strong dipole polarization and enhanced EMW dissipation performance (Fig. 5(h)).103 Lu et al. successfully prepared nitrogen-doped biomass-derived carbon (NBC) with a porous structure through a two-step process of hydrothermal treatment and carbonization. The excellent EMW absorption capacity of this material stems from its abundant N doping and porous structure. The N-doped atoms and defect structures can promote dipole polarization under alternating electromagnetic fields.104 Guan et al. successfully prepared N-doped reduced graphene oxide/zinc ferrite@polypyrrole (NRGO/ZnFe2O4@PPy) composite aerogels through a three-step process of solvothermal, in situ polymerization and hydrothermal method. This N-doped composite achieved a RLmin of −77.8 dB and the minimum RCS value reached −47.9 dB m2. Furthermore, successfully introducing N atoms into the graphene lattice would disturb the originally complete sp2 hybridization structure of C atoms, resulting in an enormous number of disordered sites. Due to the substantial difference in electronegativity between C and N elements, numerous C–N electric dipoles can be formed, dissipating electromagnetic energy via the dipole polarization effect.105 He et al. prepared ordered mesoporous M-type barium ferrite (M-BaM) by using sodium dodecyl sulfate (SLS) and cetyltrimethylammonium bromide (CTAB) as dual templating agents through the sol–gel method. Subsequently, M-BaM was combined with GO through the hydrothermal method, simultaneously achieving the reduction and N doping of GO, forming the M-BaM/N-RGO composites. The surface of N-RGO contains a large number of oxygen-containing functional groups, intrinsic defects, and N-doped defects, which are conducive to defect polarization and dipole polarization relaxation (Fig. 5(i)). The RLmin of the composites arrived at −62.6 dB, and the EAB was extended to 4.2 GHz.106
 | ||
| Fig. 5 (a) Preparation route of S-doped porous carbon micro-flakes. (b) Schematic diagram of the catfish effect. (c) Comprehensive ability index of crab-derived absorbers. Reproduced with permission from ref. 99. Copyright (2022) John Wiley and Sons. (d) Diagram of 9 and 6-coordination configurations and differential charges. Reproduced with permission from ref. 100. Copyright (2023) John Wiley and Sons. (e) Growth mechanism of Cl−-doped PPy NTs. (f) Defect/dipole polarization. (g) Infrared images. Reproduced with permission from ref. 102. Copyright (2025) John Wiley and Sons. (h) EMW absorption mechanism of N-doped mesoporous HNSs. Reproduced with permission from ref. 103. Copyright (2026) Elsevier. (i) Schematic diagram of the EMW absorption mechanisms for the composites. Reproduced with permission from ref. 106. Copyright (2023) John Wiley and Sons. | ||
Vacancy engineering provides a key strategy for optimizing material properties. By controlling the formation, type, and concentration of vacancies, it is possible to precisely modulate the electronic structure of materials, enhance the interaction between electrons, and improve charge transfer efficiency.109 Zhao et al. successfully prepared a unique nested doll structure of MoS2/V2O3 composites. By regulating the concentration of S2−, the lattice spacing and interface vacancies of molybdenum disulfide nanosheets can be simultaneously adjusted. A higher concentration of S2− will cause the lattice spacing of molybdenum disulfide to expand and the interface vacancies to increase, thereby precisely controlling the ordered stacking structure of the MoS2–V2O3–MoS2 layers and inducing an interface polarization effect (Fig. 6(a)). The introduction of vacancies effectively reduces the band gap of molybdenum disulfide, lowers the energy barrier for electron transition, and simultaneously enhances the dielectric loss capacity (Fig. 6(b) and (b1)). These changes contribute greatly to the material's EMW absorption capability.107 Jia et al. successfully achieved local charge regulation in dielectric CoSe2 through Se vacancy engineering, thereby enhancing the EMW absorption ability. The experimental results demonstrated that the concentration of Se vacancies may be efficiently regulated by controlling the re-annealing temperature. The formation of Se vacancies significantly improves the conductivity of CoSe2, thereby enhancing the material's conductive loss capacity. Moreover, the DFT calculations further reveal that the Se vacancies disrupt the original charge balance distribution in CoSe2, inducing a strong dipole polarization effect, which further promotes the polarization loss.108 Gong et al. were the first to propose the introduction of Se vacancies at the heterogeneous interface formed by Fe–Se compounds (Fig. 6(c)). It is noteworthy that the formation of Se vacancies significantly promotes the electron transfer from Fe atoms to Se atoms, thereby generating a large number of dipoles and enhancing the polarization loss ability of the material. In addition, the Se vacancies can act as binding centers, capturing free electrons around them, and reducing the overall conductivity of the material (Fig. 6(d)). This behavior not only weakens the skin effect but also helps to optimize the impedance matching characteristics. By precisely controlling the Se vacancies in the Fe–Se compound, excellent EMW absorption performance was successfully achieved at an ultrathin thickness. The EAB at 1.5 mm was 5.42 GHz.109 Hao et al. successfully achieved efficient EMW absorption by constructing abundant oxygen vacancies in Co3O4/C/N-doped carbon nanonets (NCNs). The introduction of oxygen-rich vacancies enhanced the polarization effect and conductivity, resulting in a RLmin of −78.1 dB.110 Su et al. successfully integrated ultrafine vacancy-rich Nb2O5 semiconductors into carbon nanosheets. The O 1s XPS spectra revealed the existence of O vacancies in Nb2O5, and EPR analysis also verified the existence of O vacancy defect sites (Fig. 6(e) and (f)). When there are two oxygen vacancies, electrons will delocalize at the vacancies and flow into the nearby O atoms, thereby causing the separation of charges, generating electric dipoles, and forming electron dipole polarization, significantly increasing the dielectric loss ability of the material (Fig. 6(g)).111 Zhang et al. successfully developed a highly efficient EMA composed of one-dimensional carbon fibers and cation-doped NiCo-MOF (CF@NiCoMOF-800). By adjusting the O vacancies distribution of the NiCo-MOF through different radii of cation atoms, the resulting EMAs exhibited excellent EMW absorption performance, with a RLmin of −80.63 dB and an EAB of up to 8.01 GHz.112
 | ||
| Fig. 6 (a) Differential charge 3D plots and 2D cut-away plots of O-vacancy V2O3 and S-vacancy MoS2. (b) and (b1) HR-TEM images of the MoS2/V2O3-5 absorber. Reproduced with permission from ref. 107. Copyright (2024) John Wiley and Sons. (c) Evolution diagram of the Se vacancies. (d) Enlarged HR-TEM image. Reproduced with permission from ref. 109. Copyright (2023) Elsevier. (e) High-resolution O 1s XPS spectra and (f) EPR spectra of a-Nb2O5/CNS and ov-Nb2O5/CNS. (g) Charge density distribution of ov-Nb2O5 (yellow and blue regions depict aggregation and dispersion of electrons). Reproduced with permission from ref. 111. Copyright (2023) Springer Nature. (h) Atomic-resolution HAADF-STEM image of the 8HEB (8-cation HEB) samples. Reproduced with permission from ref. 113. Copyright (2025) Elsevier. (i) Schematic diagram of induced lattice distortion to enhance impedance matching and electromagnetic loss mechanism to achieve the regulation of FeCoNiCuCx absorption performance. Reproduced with permission from ref. 114. Copyright (2024) John Wiley and Sons. | ||
Lattice distortion refers to the deviation of the periodic arrangement of atoms in a crystal from their ideal positions, resulting in structural defects, stress or distortion. This change in structure significantly affects the electronic structure and electromagnetic parameters of the absorber, and the electromagnetic parameters directly determine the material's EMW absorption performance. Gu et al. successfully prepared high-entropy diborides (HEB) through lattice distortion engineering. By adjusting the composition of the metal elements, they rationally designed the lattice distortion structure of the HEB, achieving superior EMW absorption performance with an EAB of 7.2 GHz (Fig. 6(h)).113 Qiu et al. precisely controlled the lattice distortion of the FeCoNiCu high entropy alloys through doping and annealing processes, successfully observing significant lattice distortion in the composites (Fig. 6(i)). The EAB of the composites was 7.99 GHz. Its outstanding EMW absorption capability was attributed to the optimized impedance matching and enhanced polarization loss114 (Table 1).
| Absorbers | RLmin (dB) | EAB (GHz) | Thickness (mm) | Ref. |
|---|---|---|---|---|
| NHCS@Co-SAs | −44.96 | 5.25 | 2 | 88 |
| CoN20C | −63.4 | — | 2.78 | 89 |
| HCNF-Er-based film | −57.8 | 6 | — | 90 |
| Ni-SA/HPCF | −53.2 | 5 | — | 91 |
| NC-1000 | −50.5 | 5.1 | 1.9 | 92 |
| Fe3O4@Zn–N–carbon | −61.9 | 11.5 | 2.5 | 93 |
| Ni-SAs3/NC | — | 7.08 | 2.5 | 94 |
| ZnCo-NC@rGO | −59.63 | 4.52 | — | 95 |
| Ni/Cu-NC | −70.96 | 7.8 | — | 96 |
| SP-PCN | −50.7 | 6 | 1.8 | 98 |
| S3PC | −64.05 | 9.03 | — | 99 |
| S-CoP@SC | −66 | 5 | 2.2 | 100 |
| NdCo@C/CNTs-2 | −66.31 | 4.74 | 1.34 | 101 |
| PPy-M-4 | — | 6.64 | 2 | 102 |
| HNSs | −51.39 | 7 | 1.96 | 103 |
| NBC | −53 | 4.22 | 1.7 | 104 |
| NRGO/ZnFe2O4@PPy | −77.8 | 6.4 | — | 105 |
| MBG-2 | −62.6 | 4.2 | 1.4 | 106 |
| MoS2/V2O3-5 | −69.65 | 7.68 | 3 | 107 |
| CoSe2-600 | −30.73 | 4 | 1.8 | 108 |
| Fe3Se4/Fe7Se8 | −42.4 | 5.42 | — | 109 |
| NCM-350 | −78.1 | 6.83 | — | 110 |
| ov-Nb2O5/CNS | −80.8 | — | 2.76 | 111 |
| CF@NiCoMOF-800 | −80.63 | 8.01 | 2.59 | 112 |
| 9HEB | −42.6 | 7.2 | — | 113 |
| FeCoNiCuC0.37 | — | 7.99 | 1.95 | 114 |
3.2. Nanoscale design
Rational nanoscale design is one of the key strategies for constructing lightweight and efficient EMAs. Through nanoscale design, unique physical and chemical properties can be endowed to EMAs, such as size effect, large specific surface area, multiple loss synergies and precisely controllable microstructures.The hollow structure, with its abundant cavities, can effectively regulate the conductive properties, promote multiple reflections and scattering, and improve impedance matching, thereby considerably improving the EMW absorption capacity.117,118 Xu et al. successfully prepared silicon oxycarbonitride (SiOCN) ceramics with a hollow bowl-like morphology via a template-free pyrolysis strategy (Fig. 7(a)–(d)). The resulting material exhibited excellent EMW absorption performance, achieving a RLmin of −52.93 dB and an EAB of 3.88 GHz. Besides, the RCS decreased by more than 15 dB m2. This enhanced attenuation performance was attributed to the unique hollow architecture, which facilitates multiple scattering events and optimizes impedance matching.115 Zhang et al. successfully synthesized a Co–CoFe2O4@mesoporous hollow carbon sphere (PCHMs) nanocomposite using a hard template and subsequent pyrolysis process. Owing to the unique hollow structure of the Co–CoFe2O4@PCHMs, via adjusting the content of Co–CoFe2O4 nanoparticles, the electromagnetic parameters of the composites could be flexibly regulated, effectively controlling the EMW absorption performance of the Co–CoFe2O4@PCHMs. When 0.1 mmol Co(NO3)2·6H2O and 0.2 mmol Fe(NO3)3·6H2O were added, the RLmin of Co–CoFe2O4@PCHMs reached −65.31 dB, and the EAB was 8.48 GHz.116 Xiao et al. successfully fabricated a kind of hollow silicon carbide (SiC) microtubes with a special structure through the de-templating method (Fig. 7(e) and (f)). The carbon element was evenly distributed within the hollow SiC microtubes, which helps to improve the conductive loss of the composites. Furthermore, the SiC microtubes' unique hollow structure facilitated multiple EMW reflections and scattering. For the SiC-T2 sample, prepared with an initial Si/C molar ratio of 0.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, the RLmin reached −25.7 dB and the EAB was 5 GHz.117 Qiu et al. successfully synthesized a nickel metal–organic framework (Ni-MOF) using a mixed solvent through the solvothermal method, and then obtained hollow nickel/carbon (Ni/C) microspheres by pyrolysis of Ni-MOF. This study found that by adjusting the ratio of N,N-dimethylformamide (DMF) and H2O in the solvent, the morphology of Ni-MOF could be effectively controlled. When the ratio of DMF to H2O in the solvent was 2
1, the RLmin reached −25.7 dB and the EAB was 5 GHz.117 Qiu et al. successfully synthesized a nickel metal–organic framework (Ni-MOF) using a mixed solvent through the solvothermal method, and then obtained hollow nickel/carbon (Ni/C) microspheres by pyrolysis of Ni-MOF. This study found that by adjusting the ratio of N,N-dimethylformamide (DMF) and H2O in the solvent, the morphology of Ni-MOF could be effectively controlled. When the ratio of DMF to H2O in the solvent was 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, the RLmin of the hollow Ni/C-20 composites was −57.25 dB, and the EAB was 5.1 GHz. The unique hollow structure and the synergistic effect between C nanoparticles and Ni nanoparticles all led to the excellent EMW absorption capability.118 Sun et al. successfully coated MoS2 on a MOF-derived hollow MoS2@CNS@CoS2 composite (HCNSs) through tannic acid-protected etching, carbonization and hydrothermal techniques (Fig. 7(g)–(j)). The special hollow structure of HCNSs brought excellent performance to the material. When the carbonization temperature was 700 °C, the HCNS700 exhibited a RLmin of −63.63 dB and the EAB extended to 6.69 GHz. In addition, HCNS700 also has excellent radar stealth performance.119 Liu et al. used hollow carbon microcapsules (HC) as the core and synthesized several mutually intersecting nanosheets of MoS2 on the surface of the HC (HCM). The hollow structure with strong dielectric loss capability could promote multiple reflections of incident waves. Additionally, the construction of the MoS2 shell layer increased the heterogeneous interfaces and enhanced interface polarization. The EAB was 6.1 GHz.120 Chen et al. successfully prepared 1D magnetic nickel–carbon matrix nanotube composites (nNi-HMCNTs-800, where n represents the mmol amount of Ni(NO3)2·6H2O, and 800 refers to the calcination temperature) by mixing hydrogen bond organic frameworks (HOFs) and MOFs (Fig. 7(k) and (l)). This synthesis involved a transformation from HOF-to-MOF, characterized by the penetration of Ni ions into the HOF structure via a competitive coordination reaction, thereby forming the hollow structure of Ni-HMNTs. The air/CNT in the hollow structure play an effective role in regulating impedance matching. The RLmin of the 1.2Ni-HMCNTs-800 composites was −56.9 dB, and the corresponding EAB was 5.95 GHz.121 Wang et al. proposed an internal-to-external Ostwald ripening and phase evolution technique, successfully synthesizing uniform-sized hollow N-doped carbon spheres with Ni/Ni2P heterojunctions (Ni/Ni2P/CNs). When the addition amount of NaH2PO2 was 1000 mg, Ni/Ni2P/CNs-2 exhibited outstanding EMW absorption performance, with a RLmin of −72.2 dB and the corresponding EAB was 5.8 GHz. The experimental results and simulation analysis indicated that the large-sized internal cavities of Ni/Ni2P/CNs-2 microspheres played a crucial role in the EMW absorption performance (Fig. 7(m) and (n)).122
1, the RLmin of the hollow Ni/C-20 composites was −57.25 dB, and the EAB was 5.1 GHz. The unique hollow structure and the synergistic effect between C nanoparticles and Ni nanoparticles all led to the excellent EMW absorption capability.118 Sun et al. successfully coated MoS2 on a MOF-derived hollow MoS2@CNS@CoS2 composite (HCNSs) through tannic acid-protected etching, carbonization and hydrothermal techniques (Fig. 7(g)–(j)). The special hollow structure of HCNSs brought excellent performance to the material. When the carbonization temperature was 700 °C, the HCNS700 exhibited a RLmin of −63.63 dB and the EAB extended to 6.69 GHz. In addition, HCNS700 also has excellent radar stealth performance.119 Liu et al. used hollow carbon microcapsules (HC) as the core and synthesized several mutually intersecting nanosheets of MoS2 on the surface of the HC (HCM). The hollow structure with strong dielectric loss capability could promote multiple reflections of incident waves. Additionally, the construction of the MoS2 shell layer increased the heterogeneous interfaces and enhanced interface polarization. The EAB was 6.1 GHz.120 Chen et al. successfully prepared 1D magnetic nickel–carbon matrix nanotube composites (nNi-HMCNTs-800, where n represents the mmol amount of Ni(NO3)2·6H2O, and 800 refers to the calcination temperature) by mixing hydrogen bond organic frameworks (HOFs) and MOFs (Fig. 7(k) and (l)). This synthesis involved a transformation from HOF-to-MOF, characterized by the penetration of Ni ions into the HOF structure via a competitive coordination reaction, thereby forming the hollow structure of Ni-HMNTs. The air/CNT in the hollow structure play an effective role in regulating impedance matching. The RLmin of the 1.2Ni-HMCNTs-800 composites was −56.9 dB, and the corresponding EAB was 5.95 GHz.121 Wang et al. proposed an internal-to-external Ostwald ripening and phase evolution technique, successfully synthesizing uniform-sized hollow N-doped carbon spheres with Ni/Ni2P heterojunctions (Ni/Ni2P/CNs). When the addition amount of NaH2PO2 was 1000 mg, Ni/Ni2P/CNs-2 exhibited outstanding EMW absorption performance, with a RLmin of −72.2 dB and the corresponding EAB was 5.8 GHz. The experimental results and simulation analysis indicated that the large-sized internal cavities of Ni/Ni2P/CNs-2 microspheres played a crucial role in the EMW absorption performance (Fig. 7(m) and (n)).122
 | ||
| Fig. 7 (a) SEM, (b) and (c) TEM, and (d) HRTEM, IFFT images and line profiles of the hollow SiOCN bowl. Reproduced with permission from ref. 115. Copyright (2024) Royal Society of Chemistry. (e) The SEM and (f) magnification images of SiC-T2. Reproduced with permission from ref. 117. Copyright (2020) Elsevier. TEM images of (g) ZIF-67-TA, (h) HCN700, and (i) HCNS700. (j) Partial enlarged view of (i). Reproduced with permission from ref. 119. Copyright (2025) American Chemical Society. (k) SEM and (l) TEM images of 1.2Ni-HMCNTs-800. Reproduced with permission from ref. 121. Copyright (2024) John Wiley and Sons. (m) and (n) TEM images of Ni/Ni2P/CNs-2. Reproduced with permission from ref. 122. Copyright (2023) Royal Society of Chemistry. | ||
The porous structure can not only improve the impedance matching of EMAs, increase the efficiency of EMW penetration through the materials, and extend the propagation path, but can also improve the interface polarization effect.123 Shi et al. successfully prepared porous FeNi powder by using the spray pyrolysis reduction method and adjusting the concentration of nitrate (Fig. 8(a)). The porous structure provided abundant heterogeneous interfaces, enhancing the interface effect and promoting further dissipation of EMW energy. The RLmin of the FeNi powder was −60.2 dB, and a general EAB reached 3.38 GHz.123 Lv et al. reported a method for synthesizing graphene using a nanoparticle template with monodisperse and staggered circular nanopores. This structure realizes the electromagnetic-heat-direct current conversion pathway (Fig. 8(b)). Through experiments and theoretical analysis, it was confirmed that this staggered nanoporous structure alters the electronic and phonon properties of graphene by co-coordinately regulating the nanoscale structures within the layers and the interlayer interactions.124 Li et al. successfully prepared homogenous Co/nanoporous carbon (NPC) nano-islands via effective co-precipitation along with in situ pyrolysis (Fig. 8(c)). By adjusting the pyrolysis temperature, they achieved effective control over the EMW absorption capacity. At 800 °C heating temperature, Co/NPC-800 exhibited excellent EMW absorption performance, with a RLmin of −73.87 dB and a broad EAB extended to 6.64 GHz. The porous nanostructure of Co/NPC-800 not only enhanced multiple reflections but also improved the interface polarization effect.125 Liu et al. prepared a MOF derived Fe/Fe3Co7/Co/C composite with high porosity. They systematically studied the impedance and attenuation characteristics of single-pore and gradient-pore derivatives. At a thickness of 2.5 mm, the RLmin of the Fe/Fe3Co7/Co/C composites reached −64.7 dB, and the EAB was as wide as 5.8 GHz.126 Wang et al. successfully constructed a three-dimensional ZnOCo@C material with a sub-millimeter porous structure and rich heterogeneous interfaces by introducing Co elements for co-coordination during the preparation of Zn-based MOFs, followed by pyrolysis. The sub-millimeter-sized pore structure can effectively absorb EMW (Fig. 8(d)). The composites prepared with a Zn![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co ratio of 5
Co ratio of 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 was labeled as ZnOCo@C-3. ZnOCo@C-3 exhibited a wide EAB of 6.12 GHz and a RLmin of −46.77 dB.127 Li et al. constructed a porous graphene magnetic composite foam (GMF) loaded with Fe2O3 (Fig. 8(e)). Thanks to the construction of micro-to nano-sized pores, the incident EMW could undergo multiple scattering and dissipate as fully as possible after entering the foam (Fig. 8(f)). The GMF had a RLmin of −60.13 dB and a EAB extended to 6.23 GHz.128 Lv et al. constructed a unique two-dimensional graphene superlattice by stacking graphene sub-lattices with monodisperse, nanometer-sized, square-shaped pores and strategically doped elements at the pore edges. The resulting graphene superlattice exhibited significant quantum phase correlations at both the electronic and phononic levels, thereby enabling various functions such as energy harvesting, optoelectronics, and thermoelectric effects.129 Tian et al. successfully prepared the macro-ordered porous carbon nanocomposites composed of MWCNTs and CHI (MOPCN-x/y, where x/y donates the corresponding mass ratio of multi-walled carbon nanotubes (MWCNTs) to chitosan (CHI) in the precursor solution) by using the freeze casting technique. By adjusting the freezing temperature gradient, they prepared an axial (A) honeycomb-like pore array (Fig. 8(g) and (h)). The findings from the experiments revealed that the RLmin of the MOPCN-1/2-A composites reached −72.9 dB.130 Jiao et al. developed a porous carbon fiber/polyacrylamide (CP) composite frame that simultaneously exhibited high temperature resistance and high strength (Fig. 8(i)). The sample with a mass fraction of CF in monomer of 3% (CP-3) exhibited an ultra-high EAB of 14.1 GHz at room temperature and could also cover the X-band at 473 K. The superior EMW absorption performance was due to the porous structure of the composites, which could optimize impedance matching and achieve multiple reflections.131 Su et al. precisely constructed the structure and defects through controlled migration of S atoms, and conducted experimental verification based on the Kirkendall effect and heterogeneous atomic S doping. The study showed that low-concentration sulfur vapors mainly formed a hollow structure, while excessive vapors will simultaneously produce both a hollow structure and point defects (Fig. 8(j) and (k)). When the amount of sulfur powder added was 70 mg, the RLmin of the cobalt sulfide/S-doped carbon (CSC-70) aerogel was −52.82 dB, and it achieved a broad EAB of 8.82 GHz. In addition, the aerogels also possessed superhydrophobic properties and excellent thermal insulation performance.132 Zhao et al. designed molybdenum disulfide-modified/molybdenum carbide/porous nitrogen-doped carbon (MS/MC/PNC) composites with a three-dimensional porous structure through the salt melt synthesis (SMS) method (Fig. 8(l) and (m)). By changing the mass ratio of molybdenum/sulfur sources in the composites, the EMW absorption capability of the MS/MC/PNC composites could be effectively controlled. When the mass ratio of molybdenum/sulfur sources was 0.52 g/0.9 g, the RLmin of MS/MC/PNC-2 was −55.3 dB, and the EAB was 7.6 GHz.133
1 was labeled as ZnOCo@C-3. ZnOCo@C-3 exhibited a wide EAB of 6.12 GHz and a RLmin of −46.77 dB.127 Li et al. constructed a porous graphene magnetic composite foam (GMF) loaded with Fe2O3 (Fig. 8(e)). Thanks to the construction of micro-to nano-sized pores, the incident EMW could undergo multiple scattering and dissipate as fully as possible after entering the foam (Fig. 8(f)). The GMF had a RLmin of −60.13 dB and a EAB extended to 6.23 GHz.128 Lv et al. constructed a unique two-dimensional graphene superlattice by stacking graphene sub-lattices with monodisperse, nanometer-sized, square-shaped pores and strategically doped elements at the pore edges. The resulting graphene superlattice exhibited significant quantum phase correlations at both the electronic and phononic levels, thereby enabling various functions such as energy harvesting, optoelectronics, and thermoelectric effects.129 Tian et al. successfully prepared the macro-ordered porous carbon nanocomposites composed of MWCNTs and CHI (MOPCN-x/y, where x/y donates the corresponding mass ratio of multi-walled carbon nanotubes (MWCNTs) to chitosan (CHI) in the precursor solution) by using the freeze casting technique. By adjusting the freezing temperature gradient, they prepared an axial (A) honeycomb-like pore array (Fig. 8(g) and (h)). The findings from the experiments revealed that the RLmin of the MOPCN-1/2-A composites reached −72.9 dB.130 Jiao et al. developed a porous carbon fiber/polyacrylamide (CP) composite frame that simultaneously exhibited high temperature resistance and high strength (Fig. 8(i)). The sample with a mass fraction of CF in monomer of 3% (CP-3) exhibited an ultra-high EAB of 14.1 GHz at room temperature and could also cover the X-band at 473 K. The superior EMW absorption performance was due to the porous structure of the composites, which could optimize impedance matching and achieve multiple reflections.131 Su et al. precisely constructed the structure and defects through controlled migration of S atoms, and conducted experimental verification based on the Kirkendall effect and heterogeneous atomic S doping. The study showed that low-concentration sulfur vapors mainly formed a hollow structure, while excessive vapors will simultaneously produce both a hollow structure and point defects (Fig. 8(j) and (k)). When the amount of sulfur powder added was 70 mg, the RLmin of the cobalt sulfide/S-doped carbon (CSC-70) aerogel was −52.82 dB, and it achieved a broad EAB of 8.82 GHz. In addition, the aerogels also possessed superhydrophobic properties and excellent thermal insulation performance.132 Zhao et al. designed molybdenum disulfide-modified/molybdenum carbide/porous nitrogen-doped carbon (MS/MC/PNC) composites with a three-dimensional porous structure through the salt melt synthesis (SMS) method (Fig. 8(l) and (m)). By changing the mass ratio of molybdenum/sulfur sources in the composites, the EMW absorption capability of the MS/MC/PNC composites could be effectively controlled. When the mass ratio of molybdenum/sulfur sources was 0.52 g/0.9 g, the RLmin of MS/MC/PNC-2 was −55.3 dB, and the EAB was 7.6 GHz.133
 | ||
| Fig. 8 (a) TEM images of porous FeNi powders prepared with 1.50 mol L−1 KNO3. Reproduced with permission from ref. 123. Copyright (2025) Elsevier. (b) TEM image of monolayer graphene with circular pores with a diameter of 6 ± 1 nm. Reproduced with permission from ref. 124. Copyright (2023) Springer Nature. (c) SEM image of Co/NPC-800. Reproduced with permission from ref. 125. Copyright (2023) John Wiley and Sons. (d) SEM image of ZnOCo@C-2. Reproduced with permission from ref. 127. Copyright (2023) Elsevier. (e) SEM image of GMF. (f) Part of the EMW absorption mechanisms in GMF foam. Reproduced with permission from ref. 128. Copyright (2023) Elsevier. SEM images of the MOPCN samples: (g) and (h) top view of the MOPCN-x/y-A specimen with a honeycomb pattern. Reproduced with permission from ref. 130. Copyright (2023) Elsevier. (i) SEM image of CP-3. Reproduced with permission from ref. 131. Copyright (2022) Springer Nature. (j) SEM image of CSC-70 and (k) EDS mapping of CSC-70 with the elements C, S and Co. Reproduced with permission from ref. 132. Copyright (2024) John Wiley and Sons. (l) and (m) SEM images of MS/MC/PNC-2. Reproduced with permission from ref. 133. Copyright (2024) Tsinghua University Press. | ||
The core–shell structure design is one of the most common design strategies in nanoscale design. This structure can usually protect the core layer from external interference, and the core and shell layers can also work in synergy to attenuate incident waves. Furthermore, by adjusting the material kind, thickness, and morphology of the shell and core layers, multiple loss mechanisms can be achieved, dielectric properties can be modulated, and impedance matching can be optimized.134,135 Shi et al. successfully prepared novel core–shell structured Fe3O4@C microspheres through polydopamine (PDA) confined pyrolysis (Fig. 9(a)). The core–shell design endowed the Fe3O4@C microspheres with strong magnetic and dielectric losses, and formed abundant heterogeneous interfaces, enhancing the interfacial effect. The RLmin of the Fe3O4@C microspheres reached −55.4 dB, with an EAB as high as 9.5 GHz (8.5–18 GHz).134 Wang et al. coated a layer of PDA onto the outside of a FeCo Prussian blue analog (FeCo PBAs), and then under an argon atmosphere, core–shell FeCo@C nanoparticles encased within carbon nanocages derived from PDA (FeCo@C@CNGs) were obtained via pyrolysis at 600 °C for 2 h (Fig. 9(b)). By adjusting the proportion of DA to the FeCo PBAs, the EMW absorption capacity could be effectively improved. When the mass proportion of DA to FeCo PBAs approached 0.75, the RLmin of FeCo@C@CNGs-0.75 was −67.8 dB, and the EAB was 5.3 GHz.135 To address the imbalance of impedance issue owing to the high electrical conductivity of Ti3C2Tx MXene, Jiang et al. successfully fabricated SiO2@MXene@MoS2 nanospheres with a core–shell structure by combining electrostatic self-assembly with hydrothermal processing (Fig. 9(c)). The core–shell structure prevented the aggregation of MXene and MoS2. The presence of SiO2 enabled the SiO2@MXene@MoS2 nanospheres to have an appropriate dielectric constant, effectively regulating the impedance matching (Fig. 9(d) and (e)). The RLmin of the SiO2@MXene@MoS2-2 (sample with amounts of 0.484 g Na2MoO4·2H2O and 0.608 g C2H5NS) was −52.11 dB, and the corresponding EAB was 5.44 GHz.136 By employing in situ polymerization of pyrrole combined with hydrazine vapor reduction, Dong et al. successfully synthesized reduced graphene oxide (rGO) nanosheets decorated with core–shell Fe3O4@polypyrrole (FP) microspheres. This synthesis strategy effectively mitigated rGO aggregation, resulting in a homogeneous dispersion of FP particles across the rGO surface, as depicted in Fig. 9(f)–(j). For the as-prepared Fe3O4@PPy/rGO (FPG) composites, a RLmin value of −61.20 dB was achieved at 1.89 mm. Additionally, the material demonstrated an EAB of 5.26 GHz at a matching thickness of 1.71 mm.137 Liu et al. developed a dual-pathway optimization method to improve the magnetic and dielectric loss effects in the core–shell Fe3O4@C composites. By adjusting the chemical composition from Fe3O4 to metallic Fe, the magnetic loss capacity of the FC-x (x referred to the reduction temperature) was enhanced. Additionally, by anchoring MoS2 nanosheets on the surface of the carbon shell, interface polarization and dipole polarization could be provided for the Fe/Fe3O4@C@MoS2 (FC-x-M) composites, thereby increasing the dielectric loss effect (Fig. 9(k) and (l)). The RLmin of the FC-500-M was −36.1 dB, and the EAB was 5.4 GHz.138 Zhang et al. used Co-nitrilotriacetic acid chelate nanowires (Co-NTAC) as the template and coated a layer of PDA on the surface of the Co-NTAC via self-polymerization. Then, the template was removed by the hydrothermal method and Co@PDA@MoS2 was obtained simultaneously. Subsequently, Co@PDA@MoS2 was subjected to annealing treatment, converting PDA into NC, and a multilayer core–shell structure Co@NC@MoS2 was successfully prepared (Fig. 9(m)). Thanks to the multilayer core–shell design, the impedance matching and the synergistic effect of dielectric loss and magnetic loss were optimized. Co@NC@MoS2-1 (sample with 80 mg of Na2MoO4·2H2O and 160 mg of thiourea) exhibited an excellent EMW absorption capacity, with a RLmin of −61.97 dB and an EAB of 5.6 GHz.139 Wu et al. were the first to use sustainable biomass cotton as the template (Fig. 9(n)). By constructing layered core–shell tubular carbon/Co nanoparticle composites (CMT@CNT/Co), they achieved a significant improvement in EMW absorption performance, with a RLmin of −52.3 dB and an EAB of 5.1 GHz.140 Guo et al. designed a hollow multilayer core–shell construction for magnetic FeNi@SiO2@PPy (FNSP) nanorods (Fig. 9(o)). SEM and TEM investigations revealed that FNSP was successfully synthesized (Fig. 9(p)–(s)). The results showed that the excellent EMW absorption performance of FNSP originated from the magnetic loss in the inner layer, the enhanced conductive loss in the outermost layer of PPy, and the improved interface polarization in the middle layer of SiO2. The RLmin of FNSP was −76.38 dB, and the EAB was 5.42 GHz.141 Jiang et al. utilized skin collagen fibers as templates to construct a novel multicore-shell Fe2N–carbon framework anode configuration – this configuration was composed of layered nitrogen-doped carbon nanofiber bundles embedded with Fe2N nanoparticles (Fe2N@N-CFBs), and achieved good electronic connection through the long-range carbon nanofiber framework, providing a favorable conductive path for electron/ion transmission.142 The above research results have provided a new idea for constructing lightweight and highly efficient EMAs at the nanoscale.
 | ||
| Fig. 9 (a) TEM image of the core–shell Fe3O4@C microsphere. Reproduced with permission from ref. 134. Copyright (2022) Royal Society of Chemistry. (b) TEM image of FeCo@C@CNGs-0.50. Reproduced with permission from ref. 135. Copyright (2019) Elsevier. (c) Schematic diagram of the process of the preparation of core–shell structured SiO2@MXene@MoS2 nanospheres. TEM images of (d) SiO2@MXene and (e) SiO2@MXene@MoS2-2. Reproduced with permission from ref. 136. Copyright (2024) Wenzhou University and John Wiley & Sons. SEM images of (f) FP, and (g) and (h) FPG-4 (the FPG composites prepared using 4 mg mL−1 GO). (i) TEM and (j) HAADF images of FPG-4. Reproduced with permission from ref. 137. Copyright (2023) Elsevier. SEM images of (k) FC-500 and (l) FC-500-M. Reproduced with permission from ref. 138. Copyright (2023) Elsevier. (m) TEM image of Co@NC@MoS2-1. Reproduced with permission from ref. 139. Copyright (2022) Elsevier. (n) SEM image of CMT@CNT/Co. Reproduced with permission from ref. 140. Copyright (2019) John Wiley and Sons. (o) Schematic diagram of the preparation of FNSP and the construction of dielectric-magnetic synergies. TEM images of (p) and (q) FN (FeNi–C), (r) FNS (FeNi@SiO2) and (s) FNSP. Reproduced with permission from ref. 141. Copyright (2025) Elsevier. | ||
Yan et al. proposed a growth regulation strategy for CNTs. By altering the differential catalytic effects of Co salts and Fe salts, the growth kinetics of CNTs could be effectively controlled, and the impedance matching of the composites could be improved to produce optimal EMW absorption (Fig. 10(a)–(a3)). The RLmin of NFCJ2 (the obtained black powder, F indicates FeCl2·4H2O, C indicates Co(NO3)2·6H2O, and 2 represents the proportion of g-C3N4 added) at 1.76 mm was −55.85 dB, and the corresponding EAB was 6.35 GHz.144 Huang et al. successfully synthesized CNT array hybrid materials with controllable lengths on silicon carbide fibers (SiCf) using a floating catalyst chemical vapor deposition (CVD) method. As the duration of the CVD reaction was extended from 2 to 15 min, the lengths and densities of the CNTs changed significantly. When the CVD reaction duration was 5 min, the CNTs presented a vertical array formation on the SiCf surface, with an average length of 15 µm (Fig. 10(b)–(g)). The RLmin of SiCf-CNTs at 3 mm was −56.2 dB, and the EAB was 4.3 GHz.145 The above research studies provided a new reference solution for the controllable regulation of CNTs.
 | ||
| Fig. 10 (a) Material morphology changes induced by metal component, (a1) NCJ2, (a2) NFCJ2 and (a3) NFJ2. Reproduced with permission from ref. 144. Copyright (2024) John Wiley and Sons. (b) SEM image of desized SiCf. SEM images of the CNT morphology on SiCf after various CVD reaction durations: (c) 2 min, (d) 5 min, (e) 10 min and (f) 15 min, respectively. (g) TEM images of CNTs on SiCf for 10 min. Reproduced with permission from ref. 145. Copyright (2022) Elsevier Ltd and Techna Group S.r.l. (h) and (i) FESEM graphs of SiCnws grown on the surface of CF. Reproduced with permission from ref. 146. Copyright (2017) American Chemical Society. (j) and (k) SEM images of the SiCnws array. Reproduced with permission from ref. 147. Copyright (2025) Elsevier. (l) Schematic depicting the variations in layer spacing and surface functional groups during MXene synthesis and alkali treatment. Reproduced with permission from ref. 149. Copyright (2023) John Wiley and Sons. Schematic illustration of structural units demonstrating variation with the GO/MXene ratio for different microspheres: (m) GMX1 and GMX-MFe1, (n) GMX2 and GMX-MFe2, (o) GMX3 and GMX-MFe3 and (p) GMX4 and GMX-MFe4. Reproduced with permission from ref. 150. Copyright (2023) Springer Nature. | ||
The orientation design of the nanowire array is an advanced structural design strategy. By precisely controlling the arrangement direction of one-dimensional nanomaterials in three-dimensional space, it actively regulates the propagation path of EMW and their energy conversion process, thereby achieving high-performance, wide-bandwidth, thin thickness, and low-density electromagnetic absorption. Yan et al. successfully constructed a core-sheath heterostructured silicon carbide nanowires (SiCnws) array on the surface of carbon fibers (CF) through the one-step chemical vapor infiltration method (Fig. 10(h) and (i)). Compared to CF, the RLmin of SiCnws-CF increased from −5.58 dB to −45.98 dB. Additionally, a conductive PPy layer was in situ coated on the surface of SiCnws-CF (SiCnws-CF/PPy), further increasing the RLmin to −50.19 dB, and the EAB could reach 6.2 GHz. This study provided novel insight into the orientation design and fabrication of the nanowires array.146 Deng et al. drew inspiration from biology, and using the CVD method prepared a high-oriented SiCnws array. By observing the cross-sectional images, it could be more clearly seen that the SiCnws grow radially outward along the carbon fiber cloth axis. The highly oriented needle-like nanowires are strikingly similar to the needle-like structures of the arctic woolly bear caterpillar (Fig. 10(j) and (k)). The research indicated that the SiCnws array constructed with Co(NO3)2 as the catalyst exhibited good EMW absorption performance. Its RLmin was −37.92 dB, and the corresponding EAB was 4.59 GHz. The orientation design of these two nanowire arrays has opened up new avenues for lightweight and efficient EMA design.147
The control of interlayer spacing in graphene/MXene is a revolutionary structural design strategy. By precisely adjusting the internal spatial structure of the materials at the nanoscale, it fundamentally optimizes its electromagnetic parameters, thereby solving the two core problems faced by two-dimensional materials as absorbers: the high conductivity causing impedance mismatch and the single loss mechanism leading to insufficient absorption efficiency.150 Su et al. successfully loaded thin boron nitride (BN) onto the surface of graphene through a simple process. The addition of BN might significantly increase the lamellar spacing, thus improving the interface polarization. The graphene/BN/iron/BN (graphene/BN/Fe/BN) composites benefited from the wave-transmitting property of BN, reducing the attenuation constant of the composites and optimizing the impedance matching. The absorption ability of the EMAs was effectively enhanced, with an RLmin of −44.53 dB and an EAB of 4.4 GHz.148 Du et al. successfully regulated the interlayer spacing of Ti3C2Tx MXenes through different alkali treatments (Fig. 10(l)). Different alkali treatments have various influences on the electrical and electromagnetic properties of Ti3C2Tx. The interlayer spacing of Ti3C2Tx treated with KOH expanded to 13.7 Å, showing a significant intercalation effect; while ammonia can provide more precise regulation. Ti3C2Tx treated with ammonia can effectively balance the interlayer resistance, achieving efficient EMW absorption, with an RLmin of −49.1 dB and an EAB of 3.9 GHz.149 Wang et al. successfully synthesized rGO/MXene/TiO2/Fe2C composites with a periodic intercalation structure through spray-freeze-drying and microwave radiation techniques. By varying the portion of GO to MXenes, the intercalation degree of two-dimensional nanosheets in the heterostructures could be precisely controlled to obtain excellent EMW absorption capacity. The samples were named GMX according to the mass ratio of GO to MXenes in the composites. The RLmin of GMX-MFe3 obtained by microwave treatment of GMX3 was −67.4 dB at 3.1 mm, and the EAB was 5.47 GHz at 2 mm.150 The above research provided new ideas for the controllable assembly of low-dimensional nanomaterials and their application in the field of EMW absorption (Table 2).
| Absorbers | RLmin (dB) | EAB (GHz) | Thickness (mm) | Ref. |
|---|---|---|---|---|
| SiOCN | −52.93 | 3.88 | — | 115 |
| Co–CoFe2O4@PCHMs | −65.31 | 8.48 | — | 116 |
| SiC-T2 | −25.7 | 5 | 2 | 117 |
| Ni/C-20 | −57.25 | 5.1 | 1.8 | 118 |
| HCNS700 | −63.63 | 6.69 | — | 119 |
| HCM-2 | −34.2 | 6.1 | 2 | 120 |
| 1.2Ni-HMCNTs-800 | −56.9 | 5.95 | 2.4 | 121 |
| Ni/Ni2P/CNs-2 | −72.2 | 5.8 | 1.7 | 122 |
| Porous FeNi | −60.2 | 3.38 | 1.8 | 123 |
| Co/NPC-800 | −73.87 | 6.64 | — | 125 |
| Fe/Fe3Co7/Co/C | −64.7 | 5.8 | 2.5 | 126 |
| ZnOCo@C | −46.77 | 6.12 | 4.6 | 127 |
| GMF | −60.13 | 6.23 | — | 128 |
| MOPCN-1/2-A | −72.9 | — | 4.4 | 130 |
| CP | −20.6 | 14.1 | 4 | 131 |
| CSC-70 | −52.82 | 8.82 | — | 132 |
| MS/MC/PNC-2 | −55.3 | 7.6 | — | 133 |
| Fe3O4@C | −55.4 | 9.5 | 2 | 134 |
| FeCo@C@CNGs-0.75 | −67.8 | 5.3 | — | 135 |
| SiO2@MXene@MoS2 | −52.11 | 5.44 | 2.4 | 136 |
| FPG | — | 5.26 | 1.71 | 137 |
| FC-500-M | — | 5.4 | 1.8 | 138 |
| Co@NC@MoS2 | −61.97 | 5.6 | — | 139 |
| CMT@CNT/Co | −52.3 | 5.1 | 2 | 140 |
| FeNi@SiO2@PPy | −76.38 | 5.42 | — | 141 |
| NFCJ2 | −55.85 | 6.35 | 1.76 | 144 |
| SiCf-CNTs | −56.2 | 4.3 | 3 | 145 |
| SiCnws-CF/PPy | −50.19 | 6.2 | 2 | 146 |
| SiCnws array | — | 4.59 | 1.35 | 147 |
| Graphene/BN/Fe/BN | −44.53 | — | 1.5 | 148 |
| NH3·H2O–Ti3C2Tx | −49.1 | 3.9 | 1.7 | 149 |
| GMX-MFe3 | −67.4 | — | 3.1 | 150 |
3.3. Microscale design
Microscale design can meticulously construct the geometric shape, spatial arrangement and interface relationship of materials, and regulate the propagation and conversion process of EMW at the physical level. Microscale design is the key to breaking through the performance bottlenecks of traditional absorbers. It shifts material design from being dominated by composition to being dominated by structure, and is the core research direction of the new generation of high-performance absorbers.An aerogel can introduce a large number of pores into materials, making it more similar to the impedance of air, thereby reducing the reflection on the material's surface and allowing a greater amount of EMWs to enter the material's interior. Yang et al. successfully fabricated MoS2/MXene aerogels using a specific porous template and atomic layer deposition technology (Fig. 11(a)). The aerogels maintained their porous structure, increased the paths for EMW reflection and scattering, and optimized the dielectric loss (Fig. 11(b)). The MoS2/MXene hybrid aerogels prepared by this method exhibited a strong RLmin of −61.65 dB.152 Cao et al. have developed an ultra-light Ni-MOF-rGO aerogel that possesses advantages such as a wide frequency band, strong absorption, and lightweight thin-layer matching thickness (Fig. 11(c) and (d)). By adjusting the mass ratio of the MOF to GO, the final product exhibited a RLmin of −51.19 dB and an EAB of 6.32 GHz.153 Wu et al. incorporated MXene nanosheets into polyacrylonitrile (PAN) nanofibers and then carried out carbonization to obtain MXene/C aerogels (MC) with a three-dimensional network structure (Fig. 11(e)). The three-dimensional network structure of the aerogels enhanced the electronic transmission efficiency, increased the conductive loss, and improved the impedance matching. The MC-2 (PAN![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ti3C2Tx of 2
Ti3C2Tx of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) aerogel exhibited a RLmin of −53.02 dB and an EAB of 5.3 GHz.154
1) aerogel exhibited a RLmin of −53.02 dB and an EAB of 5.3 GHz.154
 | ||
| Fig. 11 (a) Schematic illustration of the preparation process for the MoS2/MXene hybrid aerogel. (b) SEM image of the transverse section of the Ti3C2Tx aerogel. Reproduced with permission from ref. 152. Copyright (2022) John Wiley and Sons. (c) and (d) SEM images of the 600Ni-rGO aerogel. Reproduced with permission from ref. 153. Copyright (2023) American Chemical Society. (e) SEM image of the MC-2 aerogel. Reproduced with permission from ref. 154. Copyright (2023) Springer Nature. SEM images of the (f) CF sample, (g) and (h) CCF sample, and (i) FCCF-2 sample. Reproduced with permission from ref. 156. Copyright (2025) Springer Nature. (j) SEM image of SC3-F. Reproduced with permission from ref. 157. Copyright (2025) Elsevier. (k) and (l) SEM images of Cu9S5/C-40. Reproduced with permission from ref. 158. Copyright (2024) Donghua University. | ||
The porous foam possesses low density, high porosity, excellent electrical conductivity and a large number of interfaces, making it an ideal EMA with integrated structural and functional properties.156 Zhou et al. successfully prepared a novel kind of MoS2/CNT carbon foam (MC@CF). By altering the portion of MoS2/CNT in MC@CF, the EMW absorption performance could be significantly regulated. When the mass fraction of MoS2/CNT is 3%, the RLmin of MC@CF was −68.7 dB. Its excellent EMW absorption capability was attributed to the three-dimensional interconnected porous architecture and multiple heterogeneous interface effects.155 Zhang et al. successfully synthesized carbon nanowires (CNC) on three-dimensional carbon foam (CF), and then mixed them with FeNi/NiFe2O4 to form a new type of FeNi/NiFe2O4/CNC/CF (FCCF) ternary composite foams (Fig. 11(f)–(i)). The RLmin of the FCCF-2 (a precursor solution with a concentration of 1.5 times that of the original solution) composite foam reached −53 dB, and the broad EAB was 10 GHz. Its remarkable EMW absorption performance was attributed to the synergistic effect of its dielectric property, chirality, and magnetism.156
The fiber network structure is more likely to form an effective three-dimensional conductive network at a lower filling content, thereby achieving efficient and controllable conductive loss. Meanwhile, many fibers inherently exhibit excellent conductive loss and magnetic loss capabilities, making them potential materials for constructing advanced EMAs. Xiang et al. successfully fabricated SiC nanofibers (SC) through electrospinning and carbonization treatment. Subsequently, magnetic component Fe3Si was introduced into the SC. By varying the addition amount of polycarbosilane (PCS), the EMW absorption ability of the SiC@Fe3Si (SC-F) was significantly enhanced. When the content of PCS was 5.3 wt%, the SC3-F demonstrated remarkable EMW absorption ability, with a RLmin of −73.75 dB. The formation of a three-dimensional conducting system and an increase in magnetic loss had a significant impact on enhancing of the EMW absorption ability (Fig. 11(j)).157 Wu et al. successfully constructed MOF-derived Cu9S5/C nanocomposite fibers through a three-step process of electrospinning, carbonization, and sulfidation. The study of the SEM images revealed that Cu9S5/C was uniformly distributed within the carbon fibers, forming a unique three-dimensional network structure (Fig. 11(k) and (l)). The three-dimensional conductive network played a crucial part in improving the electrical conductivity of the MOF derivative and optimizing the conductive loss of the composites. When the loading amount of Cu-MOF was 40 wt%, the RLmin of Cu9S5/C-40 reached −69.6 dB, and the EAB reached 5.81 GHz.158 The above research has revealed that the design of the three-dimensional conductive network provides a theoretical basis for the construction of optimal EMAs.
Layered structure materials possess large specific surface areas and interlayer interfaces. EMW will undergo multiple reflections and scattering at the gaps or interfaces between layers, thereby increasing the chances of EMW being absorbed. Gu et al. successfully prepared MXene/CNTs/polyimide (MCP) aerogels with a multilayered structure via a bidirectional freeze-casting method. Morphological results indicated that the MCP3 (MXene![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CNT
CNT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) polyamic acid =3
polyamic acid =3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6) aerogel was composed of a layered structure with parallel-aligned multilayered components (Fig. 12(a)–(c)). This unique structure could significantly reduce the heat transfer between layers, promote heat dissipation, and achieve an ultralow thermal conductivity of 15.3 mW m−1 K−1. Meanwhile, the RLmin of this aerogel was −75.8 dB, and the EAB was 7.14 GHz.159 Deng et al. constructed SiOC/ZrB2 reinforced ZrO2/SiO2 layered composites. The (ZrO2–SiO2)f/ZrB2–SiOC composites had a continuous multi-layer structure and exhibited excellent oxidation resistance and thermal insulation properties, indicating their potential application value in complex environments (Fig. 12(d)). The RLmin of the composites was −59.34 dB, and the EAB was 4.2 GHz.160 Zhang et al. utilized the solution self-assembly strategy and employed ionic liquids (IL) as cross-linking agents to successfully prepare an MXene-IL aerogel featuring an internal layered structure in the form of a wrinkled shape (Fig. 12(e) and (f)). The results showed that the microscopic layered folds in the aerogel may be precisely regulated by adjusting the alkyl side chain length of the ionic liquid. The EAB of the aerogel reached 6.12 GHz. This study achieved the controllable regulation of the microscopic structure of MXenes.161
6) aerogel was composed of a layered structure with parallel-aligned multilayered components (Fig. 12(a)–(c)). This unique structure could significantly reduce the heat transfer between layers, promote heat dissipation, and achieve an ultralow thermal conductivity of 15.3 mW m−1 K−1. Meanwhile, the RLmin of this aerogel was −75.8 dB, and the EAB was 7.14 GHz.159 Deng et al. constructed SiOC/ZrB2 reinforced ZrO2/SiO2 layered composites. The (ZrO2–SiO2)f/ZrB2–SiOC composites had a continuous multi-layer structure and exhibited excellent oxidation resistance and thermal insulation properties, indicating their potential application value in complex environments (Fig. 12(d)). The RLmin of the composites was −59.34 dB, and the EAB was 4.2 GHz.160 Zhang et al. utilized the solution self-assembly strategy and employed ionic liquids (IL) as cross-linking agents to successfully prepare an MXene-IL aerogel featuring an internal layered structure in the form of a wrinkled shape (Fig. 12(e) and (f)). The results showed that the microscopic layered folds in the aerogel may be precisely regulated by adjusting the alkyl side chain length of the ionic liquid. The EAB of the aerogel reached 6.12 GHz. This study achieved the controllable regulation of the microscopic structure of MXenes.161
 | ||
| Fig. 12 (a) Schematic diagram of the MXene/CNTs/PI aerogel. (b) and (c) Anisotropic structure of MCP3. Reproduced with permission from ref. 159. Copyright (2024) John Wiley and Sons. (d) Cross-section SEM images of the (ZrO2–SiO2)f/ZrB2–SiOC composites. Reproduced with permission from ref. 160. Copyright (2024) Elsevier. (e) Schematic diagram of MXene-IL aerogels with different alkyl chain lengths. (f) SEM image of the MXene–[C4VIm]Br hybrid aerogel. Reproduced with permission from ref. 161. Copyright (2025) Elsevier. (g) SEM image of the synthesized HCNF/HEA composite. Reproduced with permission from ref. 162. Copyright (2024) American Chemical Society. (h) Schematic diagram and SEM image of FeC/C nanofibers with a bionic coral-like network structure. Reproduced with permission from ref. 163. Copyright (2025) Elsevier. (i) SEM image of FeCo@AAO-2. Reproduced with permission from ref. 164. Copyright (2025) Elsevier Ltd and Techna Group S.r.l. (j) SEM and (k) TEM images of the Fe3O4@void@mSiO2 composites. (l) SEM and (m) TEM images of the Fe3O4@void@mSiO2@MnO2 composites. Reproduced with permission from ref. 165. Copyright (2021) Elsevier. | ||
Biological structures in nature have evolved many efficient energy management structures (including light, sound, heat and force) for survival and adaptation to the environment. Biomimetic structure design precisely provides solutions to problems such as high density, thick thickness and poor mechanical properties. Introducing honeycomb, coral, etc. structures into EMAs can form a good conductive network and significantly reduce the material's density, meeting the strict lightweight requirements in various fields.162,163 Wang et al. used honeycomb-like porous nanofibers (HCNF) as the base materials and successfully combined the FeCoNiCuMn high-entropy alloys (HEA) with HCNF through an electrospinning and Joule heating method to fabricate the HEA/C materials derived from HCNF/HHA (Fig. 12(g)). Thanks to its honeycomb structure and lattice distortion effect, the composites exhibited excellent EMW absorption capacity, with a RLmin of −65.8 dB and an EAB of 7.68 GHz.162 Hu et al. drew inspiration from the structure of corals and successfully fabricated FeC/C nanofibers with a coral-like multi-branch network structure through electrospinning combined with a carbonization process (Fig. 12(h)). The RLmin of the composite nanofibers was −67.24 dB, and the EAB was 5.24 GHz. The excellent EMW absorption performance was attributed to the bionic coral structure, which optimized the multiple scattering paths and impedance matching of EMWs.163
The oriented structure arrangement can precisely control the electromagnetic loss path through the orderly design of the microstructure, thereby breaking through the performance bottleneck of disordered composites. Zhu et al. successfully fabricated a three-electrode electrochemical deposition method assisted by a template to produce the three-dimensional nanowire arrays of FeCo@anodic aluminum oxide (AAO). Morphological analysis revealed that the nanowires grew directionally within the pores of the AAO template (Fig. 12(i)). By altering the Fe/Co atomic ratio in the composites, the electromagnetic parameters could be precisely controlled, achieving advanced EMW absorption capabilities with a RLmin of −61.9 dB.164 Qiao et al. effectively created a chain-like structure of Fe3O4@void@mSiO2@MnO2 composites. The cross-linked chain structure provided favorable conditions for the multiple scatterings and reflection of EMW (Fig. 12(j)–(m)). Compared with the Fe3O4@void@mSiO2 composites, the introduction of the MnO2 shell layer helped to increase the porosity. Besides, the BET specific surface area increased from 125.45 m2 g−1 to 201.50 m2 g−1, and the RLmin was −45.76 dB165 (Table 3).
| Absorbers | RLmin (dB) | EAB (GHz) | Filler ratio (wt%) | Ref. |
|---|---|---|---|---|
| MoS2/MXene | −61.65 | 5.9 | — | 152 |
| 600Ni-rGO | −51.19 | 6.32 | 2 | 153 |
| MXene/C | −53.02 | 5.3 | 15 | 154 |
| MoS2/CNT | −68.7 | 14.9 | — | 155 |
| FCCF-2 | −53 | 10 | — | 156 |
| SC3-F | −73.75 | 4.4 | 30 | 157 |
| Cu9S5/C-40 | −69.6 | 5.81 | 20 | 158 |
| MXene/CNTs/PI | −75.8 | 7.14 | 2.4 | 159 |
| (ZrO2–SiO2)f/ZrB2–SiOC | −59.34 | 4.2 | — | 160 |
| MXene-IL | −42.05 | 6.12 | 1.38 | 161 |
| HCNF/HEA | −65.8 | 7.68 | 2 | 162 |
| FeC/C | −67.24 | 5.24 | 30 | 163 |
| FeCo@AAO | −61.9 | 4 | 80 | 164 |
| Fe3O4@void@mSiO2@MnO2 | −45.76 | 5.13 | 40 | 165 |
3.4. Macroscale design
Macroscale design is the core approach for EMAs to achieve practical engineering applications. It no longer focuses solely on the chemical composition of the materials themselves, but instead manages and controls EMWs by constructing macroscopic physical structures, thereby achieving breakthroughs in EMW absorption performance. Moreover, macroscale design elevates EMAs from simple functional coatings to functional devices, addressing stringent engineering demands for lightweight construction, high strength, and environmental stability. Typical macroscale designs include periodic structures, frequency selective surfaces and impedance gradient structures, etc. | ||
| Fig. 13 (a) A schematic representation of the fabrication and deformation processes of the elastic EMAs featuring macro-dome array structures. Reproduced with permission from ref. 166. Copyright (2025) Elsevier. (b) PCCF absorber. (c) Geometry of the EMW absorption structure using PPCF with design variables. Reproduced with permission from ref. 167. Copyright (2020) Elsevier. (d) Model diagram of MDSGM consisting of a GTFA unit and oriented FCI film. (e) Images of MDSGM. Reproduced with permission from ref. 168. Copyright (2024) John Wiley and Sons. (f) Schematic structure of the FSS unit cell. C is the period of the FSS, R is the diameter of the circle, a and b are the side lengths of the square including in the first and the second layer FSS, respectively. t1, t2 and t are the thicknesses of the first and second layer FSS, and the coating, respectively. Reproduced with permission from ref. 169. Copyright (2017) Elsevier. (g) Unit-cell structure of the proposed MMA. (h) Schematic illustration of the FSS. Reproduced with permission from ref. 170. Copyright (2025) Elsevier. (i)–(k) Designed absorbing materials model. Reproduced with permission from ref. 171. Copyright (2019) Elsevier. | ||
The construction of FSS is like an electromagnetic sieve, whose characteristics determine that it can only reflect or transmit waves within specific frequency bands. Combined with the underlying absorber structure, it is possible to design intelligent and highly efficient EMAs for specific frequency bands. Yang et al. designed and fabricated a double-layer ultra-thin broadband radar absorber structure (RAS), with the FSS on the top layer and a dielectric ceramic coating on the bottom layer (Fig. 13(f)). This study systematically revealed the influence of key design parameters such as the size and period of the FSS on the EMW absorption performance of the RAS. The results showed that by optimizing the size of the FSS, the EMW absorption capability of the RAS can be significantly enhanced, ultimately extending the EAB to 7 GHz.169 Zhang et al. successfully designed multi-layer FSS broadband EMAs. This absorber featured two layers of FSS (Fig. 13(g) and (h)). By collaboratively optimizing the materials parameters and geometric configurations of the surface, the absorber exhibited an absorption rate exceeding 90% within the frequency range of 3.1–18 GHz.170 Yang et al. successfully prepared Cr2O3–TiO2 (CrTiO) composite coatings with different Cr2O3 contents using high-temperature solid-phase reaction and plasma spraying technology (Fig. 13(i)–(k)). The study confirmed that by controlling the content of Cr2O3 and the overall thickness of the coating, efficient EMW attenuation could be achieved. Furthermore, it was found that constructing the FSS composed of circular and square elements on the surface of the CrTiO coating could significantly optimize its EMW absorption performance.171
 | ||
| Fig. 14 (a) Simulated electrical fields of pure MXene, MrG (with 80 wt% rGO content) and pure rGO films. Reproduced with permission from ref. 172. Copyright (2023) Elsevier. (b) The equivalent impedance at various heights of the substrate of the moth eye biomimetic materials with the top structure height of 8 mm. Reproduced with permission from ref. 173. Copyright (2023) John Wiley and Sons. Schematic diagram of the (c) pyramid-shaped impedance gradient absorber and (d) Salisbury screen absorber. Reproduced with permission from ref. 174. Copyright (2023) Elsevier. (e) Schematic of the gradient honeycomb sandwich structure. (f) Gradient honeycomb sandwich architecture. Reproduced with permission from ref. 175. Copyright (2025) Elsevier. (g) Digital image of MAAH. Reproduced with permission from ref. 176. Copyright (2024) Donghua University. (h) The EMW absorption property test image of the material at room temperature. (i) The digital image of the material. Reproduced with permission from ref. 177. Copyright (2026) Elsevier. | ||
Wu et al. prepared a dual-response synergistic gradient honeycomb sandwich structure (GHSS) which exhibited excellent broadband absorbing performance. The gradient structure optimized the impedance matching between the honeycomb structure and the air, and the dual-response synergistic mechanism further widened the bandwidth (Fig. 14(e) and (f)). Together, these two mechanisms ensure comprehensive and reliable absorption across the 2–18 GHz wide frequency range.175 The EMW absorption aramid honeycomb (MAAH) composed of MXene nanosheets and aramid nanofibers (ANFs) was successfully developed by Sun et al. (Fig. 14(g)). With the RLmin of −38.5 dB, MAAH's EMW absorption performance was improved by the formation of a three-dimensional conductive network and the combined action of the honeycomb multi-level structure. In the meantime, MAAH's compressive strength was also improved by the dense network structure.176 A frustum pyramid structure SiCf/SiBCN composite was devised and successfully manufactured by Huang et al. (Fig. 14(h) and (i)). The composite showed a great synergistic effect of EMW absorption at both room temperature and high temperatures following structural optimization and geometric parameter adjustment. The EAB was 34.8 GHz (5.2–40 GHz) at ambient temperature and remained at 12.5 GHz (5.5–18 GHz) at 1100 °C177 (Table 4).
| Absorbers | RLmin (dB) | EAB (GHz) | Thickness (mm) | Ref. |
|---|---|---|---|---|
| NiCo chains/CNTs-OH | −34.93 | 5.16 | 1.55 | 166 |
| PPCF | −27 | 4.2 | — | 167 |
| MDSGM | −40.24 | — | — | 168 |
| RAS | −27.5 | ∼7 | — | 169 |
| MMA | — | 14.9 | 8.45 | 170 |
| CrTiO | — | 4.2 | 1.9 | 171 |
| MXene/rGO | −44.3 | 4.84 | — | 172 |
| CIP/PU | — | 6.94 | 3 | 173 |
| Polyamide@C/Fe3O4 | −47.51 | 5.2 | — | 174 |
| GHSS | −29 | 16 | 21 | 175 |
| MAAH | −38.5 | — | 1.9 | 176 |
| SiCf/SiBCN | −33.4 | 12.5 | — | 177 |
4. Conclusion and outlook
Multi-scale structural design and optimization of EMAs play a crucial role in enhancing their performance. This paper systematically reviews recent research advances in improving EMA performance through multi-scale structural design, providing a theoretical basis for optimizing the properties of different structural materials. Research indicates that material structure ranging from atomic arrangement to macroscopic configuration, has become the core approach for pushing beyond the performance limits of traditional EMAs. Structural design plays a decisive role in determining their electromagnetic parameters, impedance matching characteristics and loss mechanisms. At the atomic/molecular scale, atomic dispersion and defect engineering significantly enhance dielectric loss and charge transport capabilities by precisely optimizing the intrinsic electromagnetic parameters of materials, such as electronic structure and localized polarization. At the nanoscale, the construction of hollow, porous, core–shell structures and low-dimensional nanomaterials not only achieves lightweight design, but also significantly enhances the electromagnetic energy dissipation efficiency through multiple reflections, interface polarization and the synergistic effect of magnetism and dielectricity. At the microscale, a three-dimensional network, layered structure and biomimetic designs effectively coordinate impedance matching and attenuation performance by regulating EMW propagation paths and the distribution of a conductive network. At the macroscale, the building of periodic structures, FSS and gradient structures has further enabled the active management of EMW propagation and broadband absorption, accelerating the development of EMAs towards intelligence, device integration, and system-level integration. These structural design strategies, through the synergistic effect of multiple levels and multiple mechanisms, significantly enhance the comprehensive performance of the materials, providing a reliable path for achieving high-performance EMAs with properties such as lightweight, wide bandwidth, and robust absorption.Although multi-scale structural design has achieved significant results in the area of EMAs, the development of advanced EMAs still faces numerous challenges and opportunities to meet the diverse demands of next-generation electromagnetic protection technologies.
(1) Current structures predominantly feature static designs. Future development must focus on creating smart materials (such as phase-change materials and liquid crystal composites) capable of responding to environmental stimuli including temperature, electric fields and stress. This will enable dynamic control over absorption bandwidth and intensity, meeting the demands of complex electromagnetic environments and adaptive stealth requirements.
(2) The current preparation techniques still have limitations in the precise control of cross-scale structures and large-scale production. In the future, advanced methods such as atomic manufacturing (e.g., scanning probe technology), 4D printing and template self-assembly should be combined to achieve controllable preparation from atomic to macro structures. At the same time, the application of multi-physics field simulation, machine learning and AI in material design should be strengthened. Through cross-scale modeling and big data analysis, the discovery and optimization of new material systems can be accelerated.
(3) The materials should develop towards a direction of structural–function integration, maintaining efficient absorption performance while integrating functions such as thermal management, load-bearing, and corrosion resistance. Especially in fields like aerospace and flexible electronics, it is necessary to focus on solving the stability and durability issues of the materials under extreme conditions (high temperature, corrosion and mechanical load).
(4) While striving for high performance, the full life-cycle impact of materials must also be taken into account. Developing environmentally friendly, biocompatible, and even biodegradable green absorbers, as well as exploring low-energy consumption and low-pollution sustainable production processes, will be inevitable requirements for the long-term healthy development of this field.
Author contributions
Yang Guan: conceptualization, information collection, and writing original draft. Yongpeng Wu, Jianyu Zhou, Kai Yao, and Ruolin Liu: information collection and writing original draft. Yang Yang and Wei Lu: conceptualization, writing (editing), supervision, and project administration.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was supported by the National Key Research and Development Program of China (2024YFE0100600), the National Natural Science Foundation of China (52373303), the Shanghai Municipal Science and Technology Major Project (2021SHZDZX0100), the Fundamental Research Funds for the Central Universities, and the Interdisciplinary Joint Research and Development Project of Tongji University (2024-4-ZD-03).Notes and references
- X. Zhang, X. Tian, N. Wu, S. Zhao, Y. Qin, F. Pan, S. Yue, X. Ma, J. Qiao, W. Xu, W. Liu, J. Liu, M. Zhao, K. Ostrikov and Z. Zeng, Sci. Adv., 2024, 10, eadl6498 CrossRef CAS PubMed.
- M. Ying, R. Zhao, X. Hu, Z. Zhang, W. Liu, J. Yu, X. Liu, X. Liu, H. Rong, C. Wu, Y. Li and X. Zhang, Angew. Chem., Int. Ed., 2022, 61, e202201323 CrossRef CAS PubMed.
- F. Pan, Y. Shi, Y. Yang, H. Guo, L. Li, H. Jiang, X. Wang, Z. Zeng and W. Lu, Adv. Mater., 2024, 36, 2311135 CrossRef CAS PubMed.
- L. Liang, W. Gu, Y. Wu, B. Zhang, G. Wang, Y. Yang and G. Ji, Adv. Mater., 2022, 34, 2106195 CrossRef CAS PubMed.
- K. Yao, L. Song, X. Zhang, L. Li, F. Pan, Y. Yang, J. Liang, J. Wang, B. Yuan and W. Lu, Sci. Adv., 2025, 11, eaea0362 CrossRef CAS PubMed.
- Z. Zhao, Y. Qing, L. Kong, H. Xu, X. Fan, J. Yun, L. Zhang and H. Wu, Adv. Mater., 2024, 36, 2304182 CrossRef CAS PubMed.
- Z. Wu, H. Cheng, C. Jin, B. Yang, C. Xu, K. Pei, H. Zhang, Z. Yang and R. Che, Adv. Mater., 2022, 34, 2107538 CrossRef CAS PubMed.
- Z. Wei, Z. Li, D. Chen, J. Liang and J. Kong, Small Struct., 2025, 6, 2400615 CrossRef.
- Y. Liu, J. Zhou, C. Li, H. Zhang, Y. Wang, Y. Yan, L. Duan, Z. Cheng, Y. Ma and Z. Yao, Nat. Commun., 2025, 16, 202 CrossRef CAS PubMed.
- N. Qu, H. Sun, Y. Sun, M. He, R. Xing, J. Gu and J. Kong, Nat. Commun., 2024, 15, 5642 CrossRef CAS PubMed.
- J. Yan, Y. Huang, X. Liu, X. Zhao, T. Li, Y. Zhao and P. Liu, Polym. Rev., 2021, 61, 3 Search PubMed.
- L. Kong, S. Zhang, Y. Liu, H. Wu, X. Fan, Y. Cao and J. Huang, Carbon, 2023, 207, 198–206 CrossRef CAS.
- Y. Liu, X. Qi, X. Deng, J. Ye, Y. Fu and S. Fu, ACS Appl. Mater. Interfaces, 2023, 15, 28491–28502 CrossRef CAS PubMed.
- S. Hou, Y. Wang, F. Gao, H. Yang, F. Jin, L. Ren, Q. Wu, H. Ge and Y. Wang, Chem. Eng. J., 2023, 471, 144779 CrossRef CAS.
- M. Qin, L. Zhang, X. Zhao and H. Wu, Adv. Funct. Mater., 2021, 31, 2103436 CrossRef CAS.
- Y. Guo, K. Ruan, G. Wang and J. Gu, Sci. Bull., 2023, 68, 1195–1212 CrossRef CAS PubMed.
- C. Liang, Z. Gu, Y. Zhang, Z. Ma, H. Qiu and J. Gu, Nano-Micro Lett., 2021, 13, 181 CrossRef CAS PubMed.
- Z. Li, W. Yang, B. Jiang, C. Wang, C. Zhang, N. Wu, C. Zhang, S. Du, S. Li, H. Bai, X. Wang and Y. Li, ACS Appl. Mater. Interfaces, 2023, 15, 7578–7591 CrossRef CAS PubMed.
- X. Liu, M. Ji and J. Shao, RSC Adv., 2021, 11, 26056 RSC.
- R. Shu, Y. Guan and B. Liu, J. Mater. Sci. Technol., 2025, 214, 16–26 CrossRef CAS.
- Y. Guo, Y. Duan, X. Liu, Y. Shi, Z. Fan, H. Pang and L. Pan, J. Mater. Sci. Technol., 2025, 237, 288–297 CrossRef CAS.
- M. Zhang, S. Fang, A. A. Zakhidov, S. B. Lee, A. E. Aliev, C. D. Williams, K. R. Atkinson and R. H. Baughman, Science, 2005, 309, 1215–1219 CrossRef CAS PubMed.
- B. Huang, X. Chen, Y. Wang, Y. Liu, J. Zhou, Y. Liu and J. Yue, Ceram. Int., 2023, 49, 36715–36723 CrossRef CAS.
- J. Xu, X. Zhang, H. Yuan, S. Zhang, C. Zhu, X. Zhang and Y. Chen, Carbon, 2020, 159, 357–365 CrossRef CAS.
- Y. Wu, Y. Zhao, M. Zhou, S. Tan, R. Peymanfar, B. Aslibeiki and G. Ji, Nano-Micro Lett., 2022, 14, 171 CrossRef CAS PubMed.
- X. Luo, H. Xie, J. Cao, Y. Lu, S. Tao, Z. Meng, L. Pu, L. Sun, P. He and Z. Liu, RSC Adv., 2024, 14, 10687 RSC.
- J. Shu, M. Cao, Y. Zhang, Y. Wang, Q. Zhao, X. Fang, S. Yang, Y. Qin and J. Yuan, Adv. Funct. Mater., 2023, 33, 2212379 CrossRef CAS.
- L. Cai, H. Jiang, F. Pan, H. Liang, Y. Shi, X. Wang, J. Cheng, Y. Yang, X. Zhang, Z. Shi, H. Wu and W. Lu, Small, 2024, 20, 2306698 CrossRef CAS PubMed.
- K. Yao, F. Pan, H. Liang, X. Zhang, L. Li, L. Song, Y. Yang, B. Yuan and W. Lu, Adv. Funct. Mater., 2025, 35, 2413639 CrossRef CAS.
- L. Li, F. Pan, H. Guo, H. Jiang, X. Wang, K. Yao, Y. Yang, B. Yuan, I. Abdalla, R. Che and W. Lu, Small, 2024, 20, 2402564 CrossRef CAS PubMed.
- H. Wang, J. Bi, J. Wang, Y. Sha, Z. Liu, C. Wang and L. Qian, Mater. Today Phys., 2025, 58, 101852 CrossRef CAS.
- J. Li, S. Chen, R. Fan, X. Gong, H. Zhao, L. Yan and Y. Zhou, Carbon, 2024, 224, 119058 CrossRef CAS.
- P. Zhang, X. Zhang, B. Li, L. Xu, F. Dang and B. Li, Adv. Compos. Hybrid Mater., 2021, 4, 1292–1301 CrossRef CAS.
- Y. Shi, H. Liu, N. Tian and C. You, J. Mater. Sci. Technol., 2026, 258, 22–44 CrossRef.
- W. Yu and G. Shao, J. Colloid Interface Sci., 2023, 640, 680–687 CrossRef CAS PubMed.
- J. Xiao, X. Qi, L. Wang, T. Jing, J. Yang, X. Gong, Y. Chen, Y. Qu, Q. Peng and W. Zhong, Nano Res., 2023, 16, 5756–5766 CrossRef CAS.
- Q. Li, D. Liang, D. Wang, L. Ling, L. Jiang, F. Qiu and M. Qian, Mater. Today Commun., 2024, 38, 107862 CrossRef CAS.
- H. Ding, W. Song, Y. Chen, Y. Wang, Y. Wang, C. Tian and Y. Du, Carbon, 2025, 244, 120621 CrossRef CAS.
- R. Xu, D. Xu, Z. Zeng and D. Liu, Chem. Eng. J., 2022, 427, 130796 CrossRef CAS.
- J. Liu, J. Dong, M. Tang, Y. Si, Y. Wang and R. Sun, Mater. Des., 2025, 257, 114420 CrossRef.
- C. Gao, D. Gou, G. Huang, Z. Zhang, J. Wei, F. Gao, Y. Zhang, M. Terrones, X. Chen and Y. Wang, Nano Energy, 2025, 138, 110863 CrossRef CAS.
- Z. He, Y. Shi, H. Wang and Y. Zhu, Compos. Sci. Technol., 2024, 257, 110826 CrossRef CAS.
- Y. Zhang, Y. Xie, W. Yang, G. Wu, S. Chen and Y. Wang, Colloids Surf., A, 2024, 696, 134270 CrossRef CAS.
- T. Liu, Y. Mu, X. Geng, S. Han and S. Huang, J. Alloys Compd., 2025, 1035, 181399 CrossRef CAS.
- Q. An, D. Li, W. Liao, T. Liu, D. Joralmon, X. Li and J. Zhao, Adv. Mater., 2023, 35, 2300659 CrossRef CAS PubMed.
- Z. Ma, K. Yang, D. Li, H. Liu, S. Hui, Y. Jiang, S. Li, Y. Li, W. Yang, H. Wu and Y. Hou, Adv. Mater., 2024, 36, 2314233 CrossRef CAS PubMed.
- Y. Ma, S. Wei, R. Liu, L. Sun and W. Wang, J. Mater. Chem. C, 2024, 12, 9068 RSC.
- X. Zhang, S. Guo, G. Wei, H. Zhang, X. Hao, S. Tan, K. Liu and G. Ji, Composites, Part B, 2025, 306, 112798 CrossRef CAS.
- I. Hussain and S. Yesmin, Mater. Chem. Phys., 2023, 309, 128337 CrossRef CAS.
- H. Jiang, B. Yuan, H. Guo, F. Pan, F. Meng, Y. Wu, X. Wang, L. Ruan, S. Zheng, Y. Yang, Z. Xiu, L. Li, C. Wu, Y. Gong, M. Yang and W. Lu, Nat. Commun., 2024, 15, 6138 CrossRef CAS PubMed.
- M. Yuan, A. H. Weible, F. Azadi, B. Li, J. Cui, H. Lv, R. Che and X. Wang, Mater. Horiz., 2025, 12, 1033–1057 RSC.
- M. Yuan, B. Li, Y. Du, J. Liu, X. Zhou, J. Cui, H. Lv and R. Che, Adv. Mater., 2025, 37, 2417580 CrossRef CAS PubMed.
- Y. Zhao, R. Liu, K. Yao, S. Ye, Y. Guan, Y. Wu, X. Wang, Y. Rao, B. Yuan, W. Lu and Y. Yang, Adv. Funct. Mater., 2025, e26207 CrossRef.
- X. Guan, M. Jiang, Z. Shen, S. Tan, G. Ji and Z. J. Xu, Adv. Funct. Mater., 2025, e27596 CrossRef.
- S. Cheng, D. Sheng, S. Mukherjee, W. Dong, Y. Huang, R. Cao, A. Xie, R. A. Fischer and W. Li, Nat. Commun., 2024, 15, 9077 CrossRef CAS PubMed.
- Y. Yang, Z. Xiu, F. Pan, H. Liang, H. Jiang, H. Guo, X. Wang, L. Li, B. Yuan and W. Lu, Adv. Funct. Mater., 2025, 35, 2406133 CrossRef CAS.
- X. Wang, F. Pan, Z. Xiang, Q. Zeng, K. Pei, R. Che and W. Lu, Carbon, 2020, 157, 130–139 CrossRef CAS.
- J. Liu, S. Tan, X. Yang, J. Zhu, X. Yan, T. Chen and G. Ji, Nano-Micro Lett., 2026, 18, 115 CrossRef PubMed.
- P. Yi, X. Zhang, L. Jin, P. Chen, J. Tao, J. Zhou and Z. Yao, Chem. Eng. J., 2022, 430, 132879 CrossRef CAS.
- Y. Wu, S. Tan, T. Zhang, M. Zhou, G. Fang and G. Ji, Nano Res., 2023, 16, 8522–8532 CrossRef CAS.
- H. Zhao, Y. Cheng, Z. Zhang, J. Yu, J. Zheng, M. Zhou, L. Zhou, B. Zhang and G. Ji, Composites, Part B, 2020, 196, 108119 CrossRef CAS.
- G. Peng, J. Zhou, J. Yao, L. Lu, J. Liu, K. Zou, J. Tao, Y. Liu and Z. Yao, Carbon, 2024, 229, 119549 CrossRef CAS.
- H. Guan, Q. Wang, X. Wu, J. Pang, Z. Jiang, G. Chen, C. Dong, L. Wang and C. Gong, Composites, Part B, 2021, 207, 108562 CrossRef CAS.
- W. Li, Z. Yu, Q. Wen, Y. Feng, B. Fan, R. Zhang and R. Riedel, Int. Mater. Rev., 2023, 68, 487–520 CrossRef CAS.
- J. Qiao, X. Zhang, C. Liu, L. Lyu, Y. Yang, Z. Wang, L. Wu, W. Liu, F. Wang and J. Liu, Nano-Micro Lett., 2021, 13, 75 CrossRef PubMed.
- H. Wu, J. Liu, H. Liang and D. Zang, Chem. Eng. J., 2020, 393, 124743 CrossRef CAS.
- W. Song, M. Cao, Z. Hou, J. Yuan and X. Fang, Scr. Mater., 2009, 61, 201–204 CrossRef CAS.
- S. Wang, D. Li, Y. Zhou and L. Jiang, ACS Nano, 2020, 14, 8634–8645 CrossRef CAS PubMed.
- D. Li, H. Liao, H. Kikuchi and T. Liu, ACS Appl. Mater. Interfaces, 2017, 9, 44704–44714 CrossRef CAS PubMed.
- X. Zhang, Y. Rao, J. Guo and G. Qin, Carbon, 2016, 96, 972–979 CrossRef CAS.
- Y. Li, X. Liu, X. Nie, W. Yang, Y. Wang, R. Yu and J. Shui, Adv. Funct. Mater., 2019, 29, 1807624 CrossRef.
- Z. Lou, Q. Wang, U. I. Kara, R. S. Mamtani, X. Zhou, H. Bian, Z. Yang, Y. Li, H. Lv, S. Adera and X. Wang, Nano-Micro Lett., 2022, 14, 11 CrossRef CAS PubMed.
- J. Chen, J. Zheng, Q. Huang, G. Wang and G. Ji, J. Mater. Sci. Technol., 2021, 94, 239–246 CrossRef CAS.
- X. Liu, Y. Chen, X. Cui, M. Zeng, R. Yu and G. Wang, J. Mater. Chem. A, 2015, 3, 12197 RSC.
- M. Wu, Y. D. Zhang, S. Hui, T. D. Xiao, S. Ge, W. A. Hines, J. I. Budnick and G. W. Taylor, Appl. Phys. Lett., 2002, 80, 4404–4406 CrossRef CAS.
- A. Houbi, Z. A. Aldashevich, Y. Atassi, Z. B. Telmanovna, M. Saule and K. Kubanych, J. Magn. Magn. Mater., 2021, 529, 167839 CrossRef CAS.
- B. Wang, Q. Wu, Y. Fu and T. Liu, J. Mater. Sci. Technol., 2021, 86, 91–109 CrossRef CAS.
- Y. Liu, T. Cui, T. Wu, Y. Li and G. Tong, Nanotechnology, 2016, 27, 165707 CrossRef PubMed.
- M. Huang, L. Wang, K. Pei, B. Li, W. You, L. Yang, G. Zhou, J. Zhang, C. Liang and R. Che, Adv. Funct. Mater., 2024, 34, 2308898 CrossRef CAS.
- J. Hua, Z. Jiao, X. Han, J. Liu, M. Ma, J. Jiang, Y. Hou, X. Wang, C. Feng and Y. Ma, J. Mater. Sci. Technol., 2024, 175, 141–152 CrossRef.
- Y. Li, K. Yun, X. Yi, C. Wang, X. Ding, S. Kang, D. Kong, J. Xing, K. Gao, B. Wang, K. Tian and R. Shu, Carbon, 2025, 244, 120719 CrossRef CAS.
- J. Wang, H. Qu, X. Ye, S. Tan, X. Yan, P. Li, W. Tian and G. Ji, J. Adv. Ceram., 2025, 14, 9221191 CrossRef CAS.
- Q. Liu, Q. Cao, H. Bi, C. Liang, K. Yuan, W. She, Y. Yang and R. Che, Adv. Mater., 2016, 28, 486–490 CrossRef CAS PubMed.
- Z. Li, X. Li, Y. Zong, G. Tan, Y. Sun, Y. Lan, M. He, Z. Ren and X. Zheng, Carbon, 2017, 115, 493–502 CrossRef CAS.
- R. Shu, J. Zhang, S. Liu and Z. Luo, J. Mater. Chem. C, 2023, 11, 17012–17021 RSC.
- Z. Fang, L. Peng, Y. Qian, X. Zhang, Y. Xie, J. J. Cha and G. Yu, J. Am. Chem. Soc., 2018, 140, 5241–5247 CrossRef CAS PubMed.
- K. Cao, T. Zoberbier, J. Biskupek, A. Botos, R. L. McSweeney, A. Kurtoglu, C. T. Stoppiello, A. V. Markevich, E. Besley, T. W. Chamberlain, U. Kaiser and A. N. Khlobystov, Nat. Commun., 2018, 9, 3382 CrossRef PubMed.
- B. Li, Z. Ma, J. Xu, X. Zhang, Y. Chen and C. Zhu, Small, 2023, 19, 2301226 CrossRef CAS PubMed.
- Z. Sun, Z. Yan, Z. Guo, H. Liu, L. Zhao and L. Qian, Chem. Eng. J., 2023, 452, 139430 CrossRef CAS.
- H. Xu, M. Liu, L. Huang, X. Zhang, Z. Ma, C. Zhu, F. Cao and Y. Chen, Adv. Funct. Mater., 2025, 35, 2502952 CrossRef CAS.
- X. Zhang, B. Li, J. Xu, X. Zhang, Y. Shi, C. Zhu, X. Zhang and Y. Chen, Adv. Funct. Mater., 2023, 33, 2210456 CrossRef CAS.
- Y. Lou, J. Li, X. Li, Z. Zhu, Z. Shi and B. Xu, Nano Res., 2024, 17, 6785–6794 CrossRef CAS.
- M. Huang, L. Wang, W. You and R. Che, Small, 2021, 17, 2101416 CrossRef CAS PubMed.
- H. Liang, G. Chen, D. Liu, Z. Li, S. Hui, J. Yun, L. Zhang and H. Wu, Adv. Funct. Mater., 2023, 33, 2212604 CrossRef CAS.
- J. He, G. Zhang, Y. Xiao, C. Wang and N. Song, J. Mater. Sci.: Mater. Electron., 2024, 35, 354 CrossRef CAS.
- H. Liang, S. Hui, L. Zhang, K. Tao, Q. Chen, W. Lu and H. Wu, Small, 2025, 21, 2408396 CrossRef CAS PubMed.
- R. Peymanfar, Z. S. Ershad, E. Selseleh-Zakerin and S. H. Tavassoli, Ceram. Int., 2022, 48, 16461–16476 CrossRef CAS.
- H. Lv, X. Zhou, G. Wu, U. I. Kara and X. Wang, J. Mater. Chem. A, 2021, 9, 19170 Search PubMed.
- J. Tao, L. Xu, C. Pei, Y. Gu, Y. He, X. Zhang, X. Tao, J. Zhou, Z. Yao, S. Tao and H. Wu, Adv. Funct. Mater., 2023, 33, 2211996 CrossRef CAS.
- S. Tian, Z. Sun, H. Ding, Z. Guo, P. Wang, Y. Qiu, B. Du, J. Bi, G. Li and L. Qian, Adv. Funct. Mater., 2024, 34, 2310475 CrossRef CAS.
- G. Huang, S. Zhu, J. He, Q. Wu, Z. Jing, Y. Shen and N. Song, App. Surf. Sci., 2025, 713, 164308 CrossRef CAS.
- B. Fan, R. Ji, B. Huang, Y. Yu, C. Xie, Q. Lin, G. Tong, L. Xie and T. Wu, Adv. Funct. Mater., 2025, 35, e11825 CrossRef CAS.
- H. Wang, J. Xiao, X. Qi, X. Gong, J. Ding, Y. Qu, J. Yang and W. Zhong, J. Mater. Sci. Technol., 2026, 247, 55–63 CrossRef.
- Y. Lu, X. Zhao, Q. Tian, Y. Lin, P. Li, Y. Tao, Z. Wang, J. Ma, H. Xu and Y. Liu, Carbon, 2024, 224, 119083 CrossRef CAS.
- Y. Guan, R. Shu and B. Liu, Chem. Eng. J., 2025, 505, 159819 CrossRef CAS.
- F. He, W. Zhao, L. Cao, Z. Liu, L. Sun, Z. Zhang, H. Zhang and T. Qi, Small, 2023, 19, 2205644 CrossRef CAS PubMed.
- J. Zhao, Z. Wang, H. Wang, P. Liu and R. Che, Adv. Funct. Mater., 2025, 35, 2418282 CrossRef CAS.
- H. Jia, Y. Duan, N. Wu, S. Gu, M. Wang, J. Di and J. Ma, Chem. Eng. J., 2024, 496, 153783 CrossRef CAS.
- C. Gong, J. Ding, C. Wang, Y. Zhang, H. Cong, H. Liu, Y. Guo, K. Song, C. Shi and F. He, Chem. Eng. J., 2024, 480, 147793 CrossRef CAS.
- Z. Hao, S. Shi, B. Wang, P. Liu, Q. Gao, K. Xu, Y. Ni, Z. Tian, W. Xiao, C. Zhu, G. Xu, H. Zhang and F. Liu, J. Alloys Compd., 2025, 1024, 180206 CrossRef CAS.
- Z. Su, S. Yi, W. Zhang, X. Xu, Y. Zhang, S. Zhou, B. Niu and D. Long, Nano-Micro Lett., 2023, 15, 183 CrossRef CAS PubMed.
- F. Zhang, N. Li, J. Shi, Y. Wang, D. Yan and Z. Li, Small, 2024, 20, 2312135 CrossRef CAS PubMed.
- F. Gu, W. Wang, H. Meng, Y. Liu, L. Zhuang, H. Yu and Y. Chu, Matter, 2025, 8, 102004 CrossRef CAS.
- Z. Qiu, X. Liu, T. Yang, J. Wang, Y. Wang, W. Ma and Y. Huang, Adv. Funct. Mater., 2024, 34, 2400220 CrossRef CAS.
- R. Xu, J. Zhou, W. Huang, G. Yu, L. Guo, X. Huang and G. Shao, J. Mater. Chem. C, 2024, 12, 4640–4647 RSC.
- H. Zhang, Z. Jia, B. Wang, X. Wu, T. Sun, X. Liu, L. Bi and G. Wu, Chem. Eng. J., 2021, 421, 129960 CrossRef CAS.
- T. Xiao, J. Kuang, H. Pu, Q. Zheng, Y. Lu, W. Liu and W. Cao, J. Alloys Compd., 2021, 862, 158032 CrossRef CAS.
- Y. Qiu, Y. Lin, H. Yang, L. Wang, M. Wang and B. Wen, Chem. Eng. J., 2020, 383, 123207 CrossRef CAS.
- Y. Sun, Y. Wang, D. Liu, H. Jiang, B. Ding, J. Guo and S. Dai, ACS Appl. Mater. Interfaces, 2025, 17, 14278–14290 CrossRef CAS PubMed.
- Y. Liu, F. Wang, Y. Wang, B. Hu, P. Xu, X. Han and Y. Du, Composites, Part B, 2024, 273, 111244 CrossRef CAS.
- L. Chen, J. Pan, T. Wang, W. Xia, J. He and K. Zhang, Adv. Funct. Mater., 2025, 35, 2409432 CrossRef CAS.
- B. Wang, H. Wu, W. Hou, Z. Fang, H. Liu, F. Huang, S. Li and H. Zhang, J. Mater. Chem. A, 2023, 11, 23498–23510 RSC.
- D. Shi, J. Zheng, Y. Qian, J. Liu, J. Zheng, J. Lv, W. Cai, L. Qiao, Y. Ying, J. Yu, J. Li and S. Che, J. Alloys Compd., 2025, 1037, 182377 CrossRef CAS.
- H. Lv, Y. Yao, S. Li, G. Wu, B. Zhao, X. Zhou, R. L. Dupont, U. I. Kara, Y. Zhou, S. Xi, B. Liu, R. Che, J. Zhang, H. Xu, S. Adera, R. Wu and X. Wang, Nat. Commun., 2023, 14, 1982 CrossRef CAS PubMed.
- S. Li, Y. Sun, K. Zhang, X. Jiang and H. Yu, Small, 2024, 20, 2306990 CrossRef CAS PubMed.
- G. Liu, P. Zhu, J. Teng, R. Xi, X. Wang, X. Wang, M. Yan and C. Wu, Adv. Funct. Mater., 2025, 35, 2413048 CrossRef CAS.
- J. Wang, Y. Wang, R. Jiang, S. Chen, Q. He and G. Wu, Mater. Today Phys., 2023, 35, 101126 CrossRef CAS.
- S. Li, T. Ma, Z. Chai, Z. Zhang, M. Zhu, X. Tang, X. Zhao, Y. Lu, Q. Lan, Z. Wang, F. He, Z. Wang and T. Liu, Carbon, 2023, 207, 105–115 CrossRef CAS.
- H. Lv, Y. Yao, M. Yuan, G. Chen, Y. Wang, L. Rao, S. Li, U. I. Kara, R. L. Dupont, C. Zhang, B. Chen, B. Liu, X. Zhou, R. Wu, S. Adera, R. Che, X. Zhang and X. Wang, Nat. Commun., 2024, 15, 1295 CrossRef CAS PubMed.
- Y. Tian, D. Estevez, G. Wang, M. Peng and F. Qin, Carbon, 2024, 218, 118614 CrossRef CAS.
- Z. Jiao, W. Huyan, F. Yang, J. Yao, R. Tan, P. Chen, X. Tao, Z. Yao, J. Zhou and P. Liu, Nano-Micro Lett., 2022, 14, 173 CrossRef CAS PubMed.
- X. Su, J. Wang, T. Liu, Y. Zhang, Y. Liu, B. Zhang, Y. Liu, H. Wu and H. Xu, Adv. Funct. Mater., 2024, 34, 2403397 CrossRef CAS.
- T. Zhao, D. Lan, Z. Jia, Z. Gao and G. Wu, Nano Res., 2024, 17, 9845–9856 CrossRef CAS.
- X. Shi, Z. Wu, Z. Liu, J. Lv, Z. Zi and R. Che, J. Mater. Chem. A, 2022, 10, 8807–8816 RSC.
- F. Wang, N. Wang, X. Han, D. Liu, Y. Wang, L. Cui, P. Xu and Y. Du, Carbon, 2019, 145, 701–711 CrossRef CAS.
- X. Jiang, Q. Wang, L. Song, H. Lu, H. Xu, G. Shao, H. Wang, R. Zhang, C. Wang and B. Fan, Carbon Energy, 2024, 6, e502 CrossRef CAS.
- F. Dong, B. Dai, H. Zhang, Y. Shi, R. Zhao, X. Ding, H. Wang, T. Li, M. Ma and Y. Ma, J. Colloid Interface Sci., 2023, 649, 943–954 CrossRef CAS PubMed.
- Y. Liu, C. Tian, F. Wang, B. Hu, P. Xu, X. Han and Y. Du, Chem. Eng. J., 2023, 461, 141867 CrossRef CAS.
- Y. Zhang, Y. Li, M. Wei, D. Yang, Q. Zhang and B. Zhang, J. Mater. Sci. Technol., 2022, 128, 148–159 CrossRef CAS.
- Z. Wu, K. Pei, L. Xing, X. Yu, W. You and R. Che, Adv. Funct. Mater., 2019, 29, 1901448 CrossRef.
- Q. Guo, F. Zhai, H. Wang, Y. Zhuang, X. Shao, T. Li, K. Xie and Y. Ma, Chem. Eng. J., 2025, 520, 165714 CrossRef CAS.
- H. Jiang, L. Huang, Y. Wei, B. Wang, H. Wu, Y. Zhang, H. Liu and S. Dou, Nano-Micro Lett., 2019, 11, 56 CrossRef CAS PubMed.
- Y. He, Q. Su, D. Liu, L. Xia, X. Huang, D. Lan, Y. Liu, Y. Huang and B. Zhong, Chem. Eng. J., 2024, 491, 152041 CrossRef CAS.
- Y. Yan, J. Zhou, J. Tao, L. Duan, Y. Liu, Z. Cheng, Y. Wang, Z. Liu, Z. Ning, X. Wang, X. Tao, P. Liu and Z. Yao, Small, 2025, 21, 2410799 CrossRef CAS PubMed.
- B. Huang, J. Yue, B. Fan, Y. Liu and X. Huang, Ceram. Int., 2022, 48, 19375–19381 Search PubMed.
- L. Yan, C. Hong, B. Sun, G. Zhao, Y. Cheng, S. Dong, D. Zhang and X. Zhang, ACS Appl. Mater. Interfaces, 2017, 9, 6320–6331 CrossRef CAS PubMed.
- J. Deng, H. Liu, Q. Cui, X. Zhang, L. Han, Q. He, Y. Wang, X. Yin and H. Li, Carbon, 2025, 244, 120630 CrossRef CAS.
- Q. Su, D. Liu, C. Wang, L. Xia, X. Huang and B. Zhong, ACS Appl. Nano Mater., 2022, 5, 15902–15913 CrossRef CAS.
- Y. Du, Z. Yan, W. You, Q. Men, G. Chen, X. Lv, Y. Wu, K. Luo, B. Zhao, J. Zhang and R. Che, Adv. Funct. Mater., 2023, 33, 2301449 CrossRef CAS.
- G. Wang, C. Li, D. Estevez, P. Xu, M. Peng, H. Wei and F. Qin, Nano-Micro Lett., 2023, 15, 152 CrossRef CAS PubMed.
- S. Liu, Y. Lian, Y. Zhao, H. Hou, J. Ren, E. R. Elsharkawy, S. M. El-Bahy, Z. M. El-Bahy and N. Wu, Adv. Compos. Hybrid Mater., 2025, 8, 144 CrossRef CAS.
- J. Yang, J. Wang, H. Li, Z. Wu, Y. Xing, Y. Chen and L. Liu, Adv. Sci., 2022, 9, 2101988 CrossRef CAS PubMed.
- K. Cao, X. Yang, R. Zhao and W. Xue, ACS Appl. Mater. Interfaces, 2023, 15, 9685–9696 CrossRef CAS PubMed.
- F. Wu, P. Hu, F. Hu, Z. Tian, J. Tang, P. Zhang, L. Pan, M. W. Barsoum, L. Cai and Z. Sun, Nano-Micro Lett., 2023, 15, 194 CrossRef CAS PubMed.
- C. Zhou, M. Yuan, X. Di, S. Qiu, M. Liang, Y. Luo and H. Zou, Chem. Eng. J., 2025, 512, 162574 CrossRef CAS.
- H. Zhang, K. Kuang, Y. Zhang, C. Sun, T. Yuan, R. Yin, Z. Fan, R. Che and L. Pan, Nano-Micro Lett., 2025, 17, 133 CrossRef CAS PubMed.
- D. Xiang, Q. He, C. Fan, Y. Wang, X. Yin and C. Wang, Mater. Today Nano, 2025, 30, 100644 CrossRef CAS.
- S. Wu, C. Wang, Y. Tang, J. Jiang, H. Jiang, X. Xu, B. Cui, Y. Jiang and Y. Wang, Adv. Fiber Mater., 2024, 6, 430–443 CrossRef CAS.
- H. Gu, L. Tian, Q. Zhang, X. You, M. Wang, S. Dong and J. Yang, Small, 2024, 20, 2402423 CrossRef CAS PubMed.
- Y. Deng, B. Ren, Y. Jia, Q. Wang and H. Li, J. Mater. Sci. Technol., 2024, 196, 50–59 CrossRef CAS.
- J. Zhang, X. Zhou, H. Wang, S. Li, H. Xi, P. Zhong, Z. Wang, J. Liu and H. Wu, Carbon, 2025, 244, 120723 CrossRef CAS.
- S. Wang, Q. Liu, S. Li, F. Huang and H. Zhang, ACS Nano, 2024, 18, 5040–5050 CrossRef CAS PubMed.
- J. Hu, J. Jiang, Q. Li, J. Cao, X. Sun, S. Huo, Z. Qin and Y. Pan, Composites, Part A, 2025, 198, 109068 CrossRef CAS.
- S. Zhu, J. He, G. Huang, Y. Wang, P. Li, X. Wu, Y. Shang and N. Song, Ceram. Int., 2025, 51, 55897–55908 CrossRef CAS.
- M. Qiao, J. Li, D. Wei, X. He, X. Lei, J. Wei and Q. Zhang, Microporous Mesoporous Mater., 2021, 314, 110867 CrossRef CAS.
- C. Liu, L. Zhu, S. Zheng, L. Xu, L. Yan, Y. Zhang and G. Ji, Acta Mater., 2025, 296, 121255 CrossRef CAS.
- W. H. Choi, B. S. Kwak, J. H. Kweon and Y. W. Nam, Compos. Struct., 2020, 238, 111953 CrossRef.
- W. Ma, X. Liu, T. Yang, J. Wang, Z. Qiu, Z. Cai, P. Bai, X. Ji and Y. Huang, Adv. Funct. Mater., 2025, 35, 2314046 CrossRef CAS.
- Z. Yang, F. Luo, W. Zhou, H. Jia and D. Zhu, J. Alloys Compd., 2017, 699, 534–539 CrossRef CAS.
- D. Zhang, Y. Chen and Q. Zhou, Results Phys., 2025, 73, 108261 CrossRef.
- Z. Yang, W. Ren, L. Zhu, Y. Qing, Z. Huang, F. Luo and W. Zhou, J. Alloys Compd., 2019, 803, 111–117 CrossRef CAS.
- X. Li, D. Xu, D. Zhou, S. Pang, C. Du, M. A. Darwish, T. Zhou and S. Sun, Carbon, 2023, 208, 374–383 CrossRef CAS.
- Y. Duan, B. Ma, L. Huang, X. Ma and M. Wang, Adv. Mater. Technol., 2023, 8, 2202172 CrossRef CAS.
- X. Deng, S. Gao, X. Qi, H. Dai, S. Fu, Q. Ni and Y. Fu, Chem. Eng. J., 2023, 473, 145040 CrossRef CAS.
- N. Wu, Y. Duan, W. Chen, Y. Shi, H. Jia, J. Ma and H. Pang, Mater. Today Phys., 2025, 56, 101766 CrossRef CAS.
- H. Sun, B. Yang and M. Zhang, Adv. Fiber Mater., 2024, 6, 1122–1137 CrossRef CAS.
- B. Huang, Y. Cao, F. Ye, J. Liang, C. Li and X. Fan, J. Mater. Sci. Technol., 2026, 249, 203–213 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |
