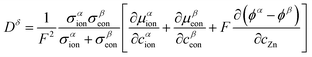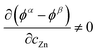Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Interface storage of vanadium based materials in zinc-ion batteries
Jie
Chen
 a,
Yuhang
Dai
a,
Yuhang
Dai
 b,
Hang
Yang
b,
Hang
Yang
 a and
Guanjie
He
a and
Guanjie
He
 *a
*a
aDepartment of Chemistry, University College London, 20 Gordon Street, London, WC1H 0AJ, UK. E-mail: g.he@ucl.ac.uk
bDepartment of Engineering Science, University of Oxford, Parks Road, OX1 3PJ, Oxford, UK
First published on 6th February 2026
Abstract
In the study of aqueous zinc-ion batteries, vanadium-based materials, as typical insertion-type cathode materials, present an inherent contradiction in simultaneously achieving high energy density (deep-level insertion) and high power density (fast kinetics), a phenomenon referred to as the “Ragone conflict”. While constructing artificial interfaces has been shown to enhance both capacity and kinetics, the underlying mechanisms of these improvements primarily rely on qualitative understanding. In this brief focus article, we elucidate the interface storage model of vanadium-based materials, providing a more quantitative approach to describing interface kinetics and specific capacity. In some reports involving vanadium-based heterostructures, there is evidence suggesting that zinc storage may rely on interfacial storage in specific materials, accompanied by reversible interfacial bond rearrangement (interfacial breathing) and decoupled ionic/electronic transport (job-sharing), thereby achieving more and faster zinc ion storage, providing an effective solution to the Ragone conflict.
Introduction
The rise of aqueous zinc-ion battery technology stems from the advantages of aqueous electrolytes (inherent safety, high ionic conductivity) and the characteristics of zinc anodes (low cost, high theoretical capacity, and moderate potential, enabling zinc metal to directly serve as the anode).1–3 Vanadium-based materials, as representatives of insertion-type cathode materials, have been extensively studied in terms of characterization techniques and regulation strategies from a technical perspective. However, from Ragone plots illustrating the relationship between power density and energy density across various systems, achieving the ideal region (simultaneously realizing high energy density and high power density) presents an inherent contradiction. This is because fully utilizing the vanadium-based cathode can achieve high energy density, but deep-level insertion inevitably leads to slower solid-state transport, resulting in reduced power density, which is known as the “Ragone conflict”.4Artificially constructing heterostructure interfaces is a widely used strategy to improve both rate capability and capacity in vanadium-based zinc-ion cathodes. Yet the dominant explanations, such as “built-in fields, lowered diffusion barriers, and more active sites”, remain largely qualitative and often internally ambiguous.5–8 In particular, two questions recur across the literature: How is the directional built-in electric field at the heterostructure interface related to the rapid diffusion of zinc ions, and why do some heterostructures exhibit apparent capacities exceeding expectations from the active vanadium phase while the conductive substrates themselves are largely non-faradaic in aqueous zinc electrolytes?9–11 These gaps motivate a interface-storage framework that treats the interface as an emergent storage/transport region to give more quantitative understanding to explain changes in capacity and kinetics through a clearer interface storage model (Fig. 1).
The interfacial storage model of vanadium-based materials
Intuitively, the interface is clearly distinct from the bulk phase. Defects at the heterostructure interface and exposed active sites facilitate ion storage and rapid migration, leading to preferential ion storage at the interface. In 2003, Maier et al. proposed a special interface storage model, which posits that ions and electrons decouple at the heterophase interface and concentrate in the space charge regions to function at the interface, enabling ultra-fast kinetics and abnormally high specific capacity.12 That framework has been developed largely in Li-ion batteries. For vanadium-based cathodes in aqueous zinc-ion batteries, the unresolved issue is more specific: when does the interface become the primary site for storing and transmitting charge, rather than just a label that intuitively enhances performance? This article delineates this boundary through several well-supported examples and clearly points out what additional evidence is needed to support the interface storage model.Some reports based on vanadium-based heterostructures have provided important references for the development of interface storage mechanisms. Despite different substrates (rGO, MXene, or RuO2), these systems share three common features: (i) a mixed two-phase architecture and the two-phase interface is believed to have a significant impact on both the ion transport rate and storage capacity, (ii) chemically specific interfacial bonding (e.g., V–O–C, V–O–Ti, V–O–Ru) that can respond to electrochemical driving, and (iii) interface-sensitive evidence that Possess reversible interface reconstruction behaviour. For example, VOx–rGO interfaces were reported to concentrate Zn storage at the interface with reversible V–O–C bond reconfiguration (“interface breathing”) and unusually fast kinetics.13 Similarly, V–O–Ti bonding in Ti3C2-based heterostructures was linked to electrochemically driven interfacial bond changes and high apparent capacity (surpassing the theoretical capacity of vanadium-based materials).14 More recently, X-ray Absorption Fine Structure (XAFS) evidence for reversible V–O–Ru bond evolution further supports the notion that dynamic interfacial bonding can be a decisive element in interfacial storage.15 The development of interface storage concepts and revelant schematic of the job-sharing are shown in Fig. 2a and b. A brief comparison of these representative studies and their key supporting evidence is summarised in Table 1.
 | ||
Fig. 2 Conceptual pathway of vanadium-based heterostructure interface storage in zinc-ion batteries. (a) The development of interface storage concepts: from Maier's theory to the discovery of interface storage and transport mechanisms in vanadium-based materials, (b) schematic of the job-sharing scenario in a two-phase conductor, illustrating decoupled ionic and electronic transport channels connected through an interfacial space-charge region, where the left blue-gray spheres represent excess M+ ions (metal-ion charge carriers, such as Li+ or Zn2+) located in interstitial positions (Mi˙), gray spheres represent M+ vacancies 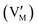 , the right blue spheres represent excess electrons (e′), and blue-gray spheres represent electron holes (h˙). The vertical axis is a logarithmic concentration axis, and the curves reflect the chemical equilibrium of defects, (c) working picture for vanadium-based heterostructures in aqueous Zn-ion batteries based on interfacial storage, highlighting interfacial Zn accumulation/transfer, built-in fields, and reversible interfacial bond reconfiguration (“interface breathing”) as a potential origin of fast and apparently high-capacity storage. Fig. 2b is redrawn based on ref. 4. © Copyright.4 , the right blue spheres represent excess electrons (e′), and blue-gray spheres represent electron holes (h˙). The vertical axis is a logarithmic concentration axis, and the curves reflect the chemical equilibrium of defects, (c) working picture for vanadium-based heterostructures in aqueous Zn-ion batteries based on interfacial storage, highlighting interfacial Zn accumulation/transfer, built-in fields, and reversible interfacial bond reconfiguration (“interface breathing”) as a potential origin of fast and apparently high-capacity storage. Fig. 2b is redrawn based on ref. 4. © Copyright.4 | ||
| System | Interface bonding | Main interface claim | Characterization evidence |
|---|---|---|---|
| VOx-rGO | V–O–C | Zinc primarily stored at interface; fast kinetics linked to interfacial transport | Raman/EELS/XPS DFT simulation; |
| HVO@Ti3C2 | V–O–Ti | Dynamic interface coupling contributes to extra capacity and pseudocapacitive kinetics | Raman/XPS DFT simulation; |
| ZVO/RuO2 | V–O–Ru | Improved rate and cycling linked to reversible heterostructures | FT-IR/XPS/XAFS DFT simulation; |
Based on our previous report, the interfacial storage model of vanadium oxide is briefly described here, as shown in Fig. 2c. For the “job sharing” mechanism, ion transport and electron transport are carried by different phases within a heterostructured cathode. The ion-conducting phase primarily supplies mobile Zn species, whereas the electron-conducting phase supplies electronic carriers. The transmission process transitions from bulk mixed conduction of ion and electron to decoupled transmission, with these two fluxes being correlated through the interface within the space charge region. Under these conditions, the Zn flux can be written in a Maier-type form as:13
This equation follows Fick's first law, where the diffusion flux is proportional to the concentration gradient, thus reflecting that the diffusion coefficient Dδ is represented by the following equation:
In a homogeneous bulk where ions and electrons move within the same mixed-conducting matrix, the aforementioned additional term does not exist, whereas in a heterostructure it can remain finite and can dominate the effective interfacial transport. The research conducted by Maier et al. in 2016 showed that due to the “job-sharing” mechanism, the built-in electric field at the interface can lead to the additional third term that is not zero.:16
For vanadium oxides, high resolution transmission electron microscope (HRTEM) observations reveal that the “job-sharing” mechanism leads to the enrichment of zinc ions at the two-phase interface of heterostructure.13 From this perspective, the relationship between the interfacial electric field and zinc ion diffusion depends on the change in zinc ion flux within the interfacial storage, rather than the intuitively understood acceleration of zinc ions insertion the bulk of vanadium-based materials.
Apparent capacity depends on the maximum number of zinc ions that can be accommodated to achieve a reversible “interfacial breathing” effect. DFT simulations provide a scheme for the quantification of theoretical limit capacity, which can calculate the formation energy Eb of zinc ions insertion at the interface:
| Eb = Esubstrate+nZn − Esubstrate − EnZn |
Maintaining structural stability requires partial fracture of interfacial bonding. Under this condition, the maximum number of zinc ion intercalation reflects the theoretical limit capacity. For VOx–G hetero-materials, the reversible bond-breaking zinc ion intercalation limit is 7, and its theoretical limit capacity is 717.06 mAh g−1 based on vanadium-based materials (V8O16 is the adopted here to calculated the specific capacity) in heterostructure constructed from DFT models, which far exceeds the theoretical capacity of pure VO2 undergoing traditional “lattice breathing” effect.13 However, as the apparent capacity of aqueous zinc-ion batteries does not solely originate from zinc ion intercalation, unusually high apparent capacities should be discussed with explicit accounting of (i) mass normalisation (active vanadium oxide or total composite), (ii) contributions from surface redox/pseudocapacitance, (iii) possible proton co-insertion or electrolyte participation, and (iv) parasitic reactions.
So far, we have quantified the changes in kinetics and specific capacity. However, it is important to note that the above discussion is based on the ideal case of complete interface storage. Additionally, our recent work has shown that even for the same interface bonding, the occurrence of “interface breathing” is selective.5 The V–O–Ti artificial interface constructed using in situ growth exhibits no significant changes during the charging and discharging process (unlike electrostatic adsorption strategies). Although the introduction of extremely low MXene (5.62%) results in fewer interfaces, which amplifies the noise signal in characterization, considering that the bonding strength of the interface in the in situ growth strategy is considered to be much stronger than that of electrostatic adsorption, it seems to suggest that a moderate bonding strength of the interface may be a prerequisite for the “interface breathing” effect. A “moderate” interface bond strength is essentially a balance. If the interface is bonded too strongly, it can become locked in place during cycling, so the structure has little freedom to rearrange and any breathing-like reversibility is hard to realise. If the bonding is too weak, the two phases tend to lose intimate contact over time, and the pathways for ion and electron transfer deteriorate, which accelerates fading. What seems most favourable is an interface that holds the phases together and preserves fast charge transfer, while still allowing small, reversible rearrangements at the bond level during cycling.
It is also worth separating interface-dominated storage from the more general ideas of pseudocapacitance. A pseudocapacitive response, such as b values close to 1, can simply reflect fast reactions near the surface and does not by itself imply a distinct interfacial transport mode. By contrast, when we invoke interface storage and job sharing, the claim is that ions and electrons mainly move through different parts of the heterostructure and establish a connection at the interface at the interface, with the interface acting as an active region rather than a passive contact. So if “interface breathing” is proposed, it should be backed by structural characterisation that tracks reversible changes in interfacial bonding during cycling.
In conclusion, we emphasise that job-sharing and interface breathing are not expected to be universal across vanadium cathodes. They require (i) a sufficiently large and connected interfacial area, ideally in the form of a standard 2D/2D heterostructure (ii) ionic and electronic pathways that remain connected through interfacial charge transfer, and (iii) interfacial bonding that is strong enough to maintain contact yet labile enough to undergo reversible reconstruction under electrochemical driving. When these conditions are not met, the electrode is more appropriately described as interface-assisted bulk insertion rather than interface-dominated storage.
Conclusion
Interface engineering is often invoked to explain why vanadium-based cathodes can charge faster and sometimes deliver unusually high capacities in aqueous Zn-ion batteries. The key message of this Focus Article is that “heterostructures” should not be used as a generic label for improved electrochemical performance. When the interface is regarded as an active area capable of carrying charges and mediating transmission, rather than a passive region that merely achieves the construction of an internal electric field, the interface storage mechanism may bring unexpected performance to material properties. In that context, the job-sharing mechanism offers a concrete way to think about how ionic and electronic pathways cooperate across two coupled phases, and why transport behaviour can deviate from conventional bulk insertion.At the same time, interface-dominated behaviour is still supported by only a small number of well documented cases, and many reports likely reflect a mixture of interfacial effects and bulk insertion. For that reason, the wording needs to stay disciplined: when interface storage is claimed, it should be accompanied by clear mass normalisation, and a discussion of alternative charge-storage contributions that are common in aqueous zinc-ion batteries. Moving forward, it is an important research direction to directly link electrochemical performance changes with structurally sensitive interface characterization, which can track the interface binding during cycling processes. From a design standpoint, a central challenge is to build interfaces that preserve fast charge transfer yet remain able to reorganise reversibly under operation, as interfaces that are either too rigid or too brittle may not be capable of achieving effective interface storage, thereby limiting the advantages that heterostructures can provide.
Conflicts of interest
The authors declare no conflict of interest.Data availability
The data that support the findings of this study are available from the authors upon reasonable request.Acknowledgements
Jie Chen thanks the funding support from University College London and UK Research and Innovation (UKRI) for the CDT scholarship. The authors would like to acknowledge Engineering and Physical Sciences Research Council (EP/V027433/2) and UK Research and Innovation (UKRI) under the UK government's Horizon Europe funding guarantee (101077226; EP/Y008707/1) for funding support.References
- X. Jia, C. Liu, Z. G. Neale, J. Yang and G. Cao, Chem. Rev., 2020, 120, 7795–7866 CrossRef CAS PubMed.
- H. Yang, T. Zhang, D. Chen, Y. Tan, W. Zhou, L. Li, W. Li, G. Li, W. Han, H. J. Fan and D. Chao, Adv. Mater., 2023, 35, e2300053 Search PubMed.
- Y. Liu, X. Lu, F. Lai, T. Liu, P. R. Shearing, I. P. Parkin, G. He and D. J. L. Brett, Joule, 2021, 5, 2845–2903 CrossRef CAS.
- C.-C. Chen and J. Maier, Nat. Energy, 2018, 3, 102–108 Search PubMed.
- J. Chen, Y. Liu, B. Xiao, J. Huang, H. Chen, K. Zhu, J. Zhang, G. Cao, G. He, J. Ma and S. Peng, Angew. Chem., Int. Ed., 2024, 63, e202408667 CrossRef CAS PubMed.
- S. Liu, L. Kang, J. M. Kim, Y. T. Chun, J. Zhang and S. C. Jun, Adv. Energy Mater., 2020, 10, 2000477 Search PubMed.
- Z. Xing, G. Xu, J. Han, G. Chen, B. Lu, S. Liang and J. Zhou, Trends Chem., 2023, 5, 380–392 CrossRef CAS.
- C. Guo, S. Yi, R. Si, B. Xi, X. An, J. Liu, J. Li and S. Xiong, Adv. Energy Mater., 2022, 12, 2202039 Search PubMed.
- Y. Li, J. Zhang, Q. Chen, X. Xia and M. Chen, Adv. Mater., 2021, 33, e2100855 Search PubMed.
- J. Chen, Y. Ding, D. Yan, J. Huang and S. Peng, SusMat, 2022, 2, 293–318 Search PubMed.
- Y. Dai, C. Zhang, X. Zhang, P. Jiang, J. Chen, W. Zong, S. Zheng, X. Gao, T. J. Macdonald and G. He, Energy Environ. Sci., 2025, 18, 9018–9030 RSC.
- J. Jamnik and J. Maier, Phys. Chem. Chem. Phys., 2003, 5, 5215–5220 RSC.
- Y. Dai, X. Liao, R. Yu, J. Li, J. Li, S. Tan, P. He, Q. An, Q. Wei, L. Chen, X. Hong, K. Zhao, Y. Ren, J. Wu, Y. Zhao and L. Mai, Adv. Mater., 2021, 33, e2100359 Search PubMed.
- B. Xiao, J. Chen, C. Hu, L. Mou, W. Yang, W. He, Z. Lu, S. Peng and J. Huang, Adv. Funct. Mater., 2023, 33, 2211679 Search PubMed.
- D. H. Liu, A. Wang, Y. Liu, F. Xu, D. Luo, J. Zheng, M. Song, C. Xu and Z. Chen, Adv. Mater., 2025, e2501624, DOI:10.1002/adma.202501624.
- C. C. Chen, L. Fu and J. Maier, Nature, 2016, 536, 159–164 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |