Integrating intrinsic charge-trapping sites in an insulated MOF nanoparticle-based dielectric layer for effective photo/synaptic transistors
Abstract
The design of charge-trapping sites is the basic and critical factor to explore organic photomultiplier phototransistors and synaptic transistors. There are few examples of charge trapping achieved directly by constructing dielectric materials, as the traditional view holds that their wide band gaps make charge trapping and release difficult. This work employs the liquid phase epitaxy method to construct a two-dimensional confined dielectric MOF-199 island-like film with a 20 nm diameter as the dielectric layer of bulk heterojunction-based photo/synaptic transistors. Diverse energy level structures of metal complex nanostructures integrated intrinsic charge-trapping sites with multi-channel electron and energy exchange to the active layer. The multi-channel exchange combines the response advantages of machines and the human brain, while the intrinsic charge trapping avoids interface charge quenching. It demonstrates simultaneous high photocurrent and good long-term plasticity, a high phototransistor performance of photo-responsivity R = 650.1 A W−1/specific detectivity 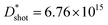 Jones, and efficient photonic synaptic transistors with maximum paired-pulse facilitation index of 142% and single-pulse remaining ratio of 65%. All these results point to the organic phototransistors for emulating the functions of biological synapses, which indicate their potential as the building blocks of bionic electronic circuit systems.
Jones, and efficient photonic synaptic transistors with maximum paired-pulse facilitation index of 142% and single-pulse remaining ratio of 65%. All these results point to the organic phototransistors for emulating the functions of biological synapses, which indicate their potential as the building blocks of bionic electronic circuit systems.

- This article is part of the themed collection: Celebrating 30 years of materials science and engineering at South China University of Technology


 Please wait while we load your content...
Please wait while we load your content...