 Open Access Article
Open Access ArticleClean production of chlorine (Cl2) and hypochlorous acid (HOCl) from photocatalytic and photoelectrochemical seawater splitting
Rohul H.
Adnan
 *ab and
Yun Hau
Ng
*ab and
Yun Hau
Ng
 *ab
*ab
aCenter for Renewable Energy and Storage Technologies (CREST), Physical Science and Engineering (PSE) Division, King Abdullah University of Science and Technology (KAUST), Thuwal 23955-6900, Saudi Arabia. E-mail: rohul.adnan@kaust.edu.sa; yunhau.ng@kaust.edu.sa
bClean Energy Research Platform (CERP), Physical Science and Engineering (PSE) Division, King Abdullah University of Science and Technology (KAUST), Thuwal 23955-6900, Saudi Arabia
First published on 5th February 2026
Abstract
Seawater splitting has emerged as a promising alternative to overall water splitting because it eliminates the kinetically sluggish oxygen evolution reaction (OER), which is a bottleneck in water splitting, and avoids the low economic value of O2. Moreover, in seawater splitting, H2 evolution coupled with the oxidation of chloride (Cl−) to value-added chlorine (Cl2) and/or hypochlorous acid (HOCl) can simultaneously benefit the energy and environmental sectors. Cl2 and HOCl are widely used for bleaching, disinfection, sanitisation and sterilisation in the medical sector and for purifying drinking water and water in swimming pools owing to their strong oxidising and antibacterial properties. Mainstream industrial production employs the chlor-alkali electrolysis of sodium chloride (NaCl), which requires significant energy input and releases enormous amounts of CO2. To achieve the sustainable production of Cl2 and HOCl while reducing energy consumption and environmental impacts, photocatalytic (PC) and photoelectrochemical (PEC) technologies have been employed as green alternatives. Importantly, PC and PEC enable the on-site production of Cl2/HOCl in remote areas, which can circumvent their instability (decomposition), storage and transport issues. This article reviews the recent progress in the PC and PEC production of Cl2/HOCl, along with the catalytic materials used and their designs and photocatalytic performance. The applications of in situ HOCl production in anti-bacterial treatment, ammonia removal, the selective oxidation and conversion of organic compounds, and CO2 conversion are discussed. We also address the challenges in this area and highlight prospects for future research directions. Overall, we demonstrate that the PC and PEC production of Cl2/HOCl serves as a green and sustainable alternative to the chlor-alkali process. This research area is still in its infancy, and we hope that this review article will garner the attention of researchers to contribute to this area, leading to a step closer toward practical applications.
Wider impactThe chlor-alkali process has been the predominant industrial production process for chlorine gas (Cl2) via the electrolysis of seawater since 1892. Despite its industrial success, the large energy input, high capital costs, complex separation and purification processes, and toxic cell components (mercury in mercury cells and asbestos in diaphragm cells) have led to serious energy and environmental concerns. The most straightforward path to clean and sustainable production of Cl2/HOCl is by employing a solar-to-chemical conversion mechanism. Progress in photocatalytic (PC) and photoelectrochemical (PEC) technologies has achieved maturity, yet their application in Cl2/HOCl production is still in its infancy. PC and PEC technologies harvesting solar energy and using seawater in the presence of suitable photocatalysts are expected to reduce energy input and environmental impacts. Moreover, these technologies enable on-site production of Cl2/HOCl in remote areas using portable PC and PEC cells, which can overcome the instability, transport, and storage issues of Cl2/HOCl. This review discusses the recent progress in the PC and PEC production of Cl2/HOCl from seawater splitting in the past decade. Importantly, the oxidation of chloride (Cl−) at the anode (or anodic site) can be coupled with hydrogen production or CO2 reduction at the cathode (or cathodic site) for the simultaneous production of fuels and useful chemicals as a type of artificial photosynthesis. |
1. Introduction
The two most pressing long-standing global challenges are the energy crisis due to the depletion of fossil fuels and environmental pollution resulting from human activities, industrial chemical processes and power plants. Therefore, there is an urgency to implement renewable energy strategies, including wind energy, photovoltaic solar cells and H2 production, to mitigate these issues. H2 production via photocatalytic water splitting has been dominating the photocatalysis research for the last 50 years since the groundbreaking work by Honda and Fujishima.1,2 Despite extensive research efforts, photocatalytic water splitting is far from practical realisation due to the challenging issues like the reverse reaction of H2 and O2, slow kinetics of O2 evolution reaction (OER), and low solar-to-hydrogen (STH) efficiency (<10%).2 A recently proposed strategy is to couple the H2 evolution reaction (HER) with more kinetically favourable oxidation reactions that produce high value-added compounds to circumvent the issues of the slow kinetics of the OER and the low economic value of O2.3–5 Sayama has pioneered the photoelectrochemical oxidation of several high value-added chemicals, including H2O2, HOCl, S2O82− and Ce4+, paving the way towards the effective utilization of solar light energy and oxidative power of photogenerated holes.6 Chloride (Cl−) oxidation to Cl2 and/or HOCl proceeds smoothly in a seawater solution, allowing the use of abundant and low-cost seawater/saltwater. To the best of our knowledge, photocatalytic seawater splitting has not been extensively discussed in the literature, and therefore, it is timely to present a comprehensive review on photocatalytic (PC) and photoelectrochemical (PEC) seawater splitting.Hypochlorous acid (HOCl) and sodium hypochlorite (NaOCl) are widely used in a plethora of applications in industrial processes and commercial applications, including pulp and paper bleaching, sanitization, and wound care.7 For use in bleaching and disinfection, 40% of the application goes into drinking water and wastewater treatments, and the remaining 60% is used in household applications, including pool sanitization, laundry bleaching and surface disinfection, in food processing and healthcare facilities. The most common type of drinking water disinfection is chlorination, which is highly effective in killing bacteria and other microorganisms to ensure that the water is safe for drinking. The World Health Organization (WHO) has set the recommended Cl2 concentration to 2 mg L−1 for effective household water treatment and the residual free Cl2 to 0.2–0.5 mg L−1.8 For convenience, Cl2, HOCl and ClO− are classified as reactive chlorine species (RCS) and are used throughout this article although sometimes they are termed HOCl in other articles.9,10 RCS are produced commercially on a large-scale using the chlor-alkali electrochemical process in a brine (NaCl) solution. However, due to the uphill thermodynamic process and formation of unwanted species, the electricity consumption in the chlor-alkaline process is huge, which leads to enormous CO2 emissions, thus resulting in a significant reduction in energy consumption and carbon footprint using green and sustainable alternatives.11 In light of energy-efficient processes and reduced environmental impacts, photocatalytic (PC) and photoelectrochemical (PEC) technologies pave the way towards a green and sustainable strategy for the production of RCS.
In water splitting, O2 is produced along with H2 (eqn (1)). However, the oxygen evolution reaction (OER) has become a kinetic bottleneck due to its large overpotential. Moreover, from a practical point of view, O2 has a negligible economic value. In saltwater/seawater, Cl− ions are present in the majority, and the Cl− oxidation reaction (COR) via a two-electron process is more kinetically favourable than the OER due to the lower overpotential. The use of seawater can dramatically reduce the operating costs of PC or PEC water splitting, where pure water is very scarce and expensive. In the chlor-alkali process, Cl− is oxidized at the anode to form Cl2 (eqn (3)), and dissolved Cl2 in water forms hypochlorous acid (HOCl) via a disproportion reaction (eqn (4)) or is mixed with sodium hydroxide (NaOH) to form NaOCl. The distribution of Cl2, HOCl and OCl− in aqueous solution depends on the pH of the solution. Under acidic, neutral and basic conditions, Cl− is oxidized to Cl2, HOCl and OCl−, respectively (Fig. 1a).12 HOCl exists in equilibrium with molecular Cl2 (eqn (6)). If Cl2 is allowed to escape, the equilibrium shifts to the left, reducing the amount of HOCl. Therefore, the selectivity of RCS for Cl− oxidation depends on the complexity of the pH-dependent equilibria of the solution. Fig. 1b shows the Pourbaix diagram of the chlorinated species across all pH values. Nonetheless, it is difficult to maintain the stability of HOCl because of its rapid decomposition even in the dark. The decomposition of HOCl follows two disproportionate pathways, producing either chlorate and/or oxygen (refer to Section 2 for details).13 Such rapid decomposition makes on-site production of HOCl via PC or PEC even more compelling to avoid the need for long-distance transport and long-term storage.
| 2H+ + 2e− → H2 E = 0 V vs. RHE | (1) |
| O2 + 4H+ + 4e− → 2H2O E = −1.23 V vs. RHE | (2) |
| 2Cl−(aq) → Cl2(g) + 2e− E = 1.36 V vs. RHE | (3) |
| Cl− + H2O → HOCl + H+ + 2e− E = 1.49 vs. RHE | (4) |
| Cl− + H2O → OCl− + 2H+ + 2e− E = 1.72 V vs. RHE | (5) |
| Cl2 + H2O ⇌ H+ + Cl− + HOCl | (6) |
| HOCl ⇌ H+ + OCl− | (7) |
 | ||
| Fig. 1 (a) Distribution of reactive chlorine species (RCS) of Cl2, HOCl, and ClO− across a wide pH range from acidic to neutral and basic. Reproduced from ref. 14. Copyright 2020, Elsevier. (b) Pourbaix diagram of the reactive chlorine species at all pH values. Reproduced from ref. 15. Copyright 2025, Springer. | ||
A photocatalytic process requires a suitable photocatalyst, a Cl-saturated solution and light to produce Cl2 and/or HOCl. The sources of Cl− include a brine solution, aqueous HCl, and seawater/saltwater. Seawater is an ideal and practical reaction solution due to its abundance, cost-effectiveness, and unlimited availability compared to pure water, which incurs an additional cost for its high-purity resource. Most ions in seawater are Cl− (55%), followed by sulfate (SO42−, 7.7%) and carbonate (CO32−, 0.33%). For this reason, a seawater solution for photocatalytic HOCl or Cl2 production is often used at 0.5–0.6 M (or 3.5 wt%) aqueous NaCl to mimic natural seawater.16 Therefore, the photocatalytic Cl− oxidation reaction (COR) brings about four merits: (i) the use of renewable solar energy, (ii) a low-cost and low carbon footprint process, (iii) on-demand production for remote areas, and (iv) no requirement for facilities for storage, handling and transport of RCS. Notably, the simultaneous PC production of H2 and HOCl avoids the use of an expensive membrane due to self-separation by phase (gaseous H2vs. liquid HOCl). Additionally, a seawater solution can be an ideal, low-cost electrolyte for operating PEC cells.
For Cl− to be oxidized to Cl2 by a photogenerated hole, the valence band (VB) must be more positive than 1.36 V (vs. RHE). This redox potential is not significantly different from that of water oxidation to O2 potential (1.23 V vs. RHE); therefore, Cl− oxidation usually competes with water oxidation. In seawater or seawater solution, Cl− oxidation is preferred to water oxidation due to a large overpotential (∼0.25 V) and thus sluggish kinetics of the latter.17 As an example, Johnston's group recorded a current density in HClO4 and NaCl/HClO4 electrolytes and observed a twice higher current density for Cl− oxidation compared to that of water oxidation between 0.5 and 2.0 V (Fig. 2a).18 Moreover, it has been shown that Cl− can quench a photogenerated hole, giving an advantage of selective HOCl formation over water oxidation.19,20 In some work, it has been shown that O2 evolution in saltwater/seawater originates from the decomposition of HOCl, providing a kinetically easier route for O2 evolution than direct 4-electron water oxidation.9 Notably, Huang et al. unravelled that adding a small amount of Cl− in water or pre-adsorbing Cl− on TiO2 lowered the activation barrier, thereby boosting the H2 production rate by 3-fold and achieving a stoichiometric ratio of H2 and O2 evolution in water splitting (Fig. 2b and c).17 Similarly, enhanced H2 production using a p-GaN-based nanowire in seawater (cf. pure water) is attributed to Cl− oxidation and better conductivity of seawater.21 These works also suggest that seawater splitting is a feasible strategy for improving water splitting for a high production rate of H2. Although this article focuses exclusively on the production of Cl2/HOCl via PC and PEC seawater splitting, readers interested in photocatalytic H2 production from seawater splitting are encouraged to refer to related review articles elsewhere.22–24
 | ||
| Fig. 2 (a) Photocurrent density of water (black) and Cl− (red) oxidation. Reproduced from ref. 18. Copyright 2023, the Royal Society of Chemistry. (b) Mechanism of seawater splitting into H2 and O2 through the formation of an HOCl intermediate in the presence of Cl−. (c) Free energy diagram of seawater splitting. Reproduced from ref. 17. Copyright 2014, the Royal Society of Chemistry. | ||
With growing attention in this research area over the past decade, it is timely to present a comprehensive and critical discussion on the development of the effective production of Cl2/HOCl. This review article aims to discuss the recent progress in PC and PEC of Cl2/HOCl production and review the photocatalytic materials and designs, as well as their photocatalytic performance. First, we present an overview of the challenges in the effective production of Cl2 and HOCl. Next, we review the photocatalytic materials designed for this purpose, including metal oxides, metal chloride and oxychlorides. In addition, we highlight the applications of photocatalytic HOCl production in other related areas, including anti-bacterial treatment, degradation of ammonia–nitrogen, selective oxidation and conversion of organic compounds, and photocatalytic CO2 reduction. Given the rapid progress in this area, it is appropriate and meaningful to summarise the developments in PC and PEC material designs for Cl2/HOCl production. We believe that this timely review will guide catalytic researchers, materials scientists and computational chemists in catalytic materials designs and developments for industrially relevant processes.
2. Challenges in Cl2 and HOCl production
Low Cl2 and HOCl yields
Low yields and selectivity of Cl2 and HOCl formation result from a few factors. First, further oxidation of Cl2 or HOCl to higher chlorine oxo species, like ClO2−, ClO3−, and ClO4−, under UV irradiation, which are usually toxic, is shown in eqn (8)–(10).25,26 Formation of higher chlorine oxo species over a prolonged reaction time leads to the reduction of faradaic efficiency (FE).| Cl− + 2H2O → ClO2− + 4H+ + 4e− E0 = 0.76 vs. RHE | (8) |
| Cl− + 3H2O → ClO3− + 6H+ + 6e− E0 = 1.45 vs. RHE | (9) |
| Cl− + 4H2O → ClO4− + 8H+ + 8e− E0 = 1.39 vs. RHE | (10) |
Second, Cl2 dissolves in water to produce HOCl and HCl (refer to eqn (6)). HOCl has a limited stability and forms radicals, like ˙OH and ˙Cl, upon exposure to UV light irradiation (eqn (11)).27–29 In the presence of ˙OH, it reacts with HOCl to generate ˙OCl and consequently to reduce the HOCl yield.30 HOCl can also decompose to yield O2 (eqn (12)) as discussed in the previous section. As a result, the use of wide band gap semiconductors like ZnO and TiO2, which absorb UV light, poses a problem for Cl2/HOCl formation. Notably, the deep valence band maximum for some semiconductor photocatalysts underscores their strong oxidising capability to decompose Cl2 and HOCl.31 In addition, there is evidence of HOCl or OCl− decomposition to ClO3− and Cl− under UV (365 nm LED) irradiation, as reported by Okada et al., reducing the overall yield of HOCl (eqn (13)).32 As a matter of fact, Koo et al. showed that the formation of toxic ClO3− can be inhibited using visible light instead of UV light in PEC systems.30
Decomposition of HOCl
 | (11) |
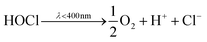 | (12) |
 | (13) |
For immediate applications, such as pollutant degradation or oxidative organic reactions using in situ generated Cl2 and/or HOCl, rapid decomposition to radicals is highly favoured because they are responsible for oxidising pollutants. Similarly, in situ-generated O2 from HOCl decomposition might serve as an oxidant in oxidation reactions. Although short-lived HOCl seems like a drawback owing to its high-yield production, its use in wastewater or sewage treatment and organic transformation is a merit. A discussion on the photodegradation of pollutants and bacteria, and the photocatalytic conversion of organic compounds, is provided in Section 7.
The need for O2 supply
It has been found that the presence of O2 for ORR is required to achieve a high production rate of HOCl in Cl− oxidation. Molecular O2 acts as an electron scavenger to suppress the recombination of charge carriers so that holes can be consumed by Cl− ions for the chloride oxidation reaction (COR). For some photocatalysts, like BiOCl, molecular O2 is required to prevent self-reduction of lattice Bi3+ to lower oxidation states, e.g. Bi metal. Nonetheless, the need for a constant supply of O2 may restrict the feasibility of the green strategy in photocatalytic Cl2/HOCl production. Additionally, CO2 has also been found to quench electrons, leading to the coupling of the photoreduction of CO2 and the photooxidation of Cl− ions as a means of producing fuels and oxidants in a single photocatalytic system. Therefore, the use of natural seawater containing a decent amount of dissolved CO2 might be an advantage for COR.Photocorrosion and dissolution of catalytic materials
Another issue is the dissolution and/or photocorrosion of photocatalytic materials in a neutral seawater solution or seawater. The production of Cl2 and HOCl leads to local acidification, which changes the pH of the seawater solution to become acidic (refer to eqn (6)). Jadwiszczak et al. calculated the pH of 0.5 M NaCl to be 2.5 upon Cl2 evolution and dissolution.33 Although some photocatalytic materials, like WO3, TiO2 and BiVO4, are chemically stable in an acidic medium, other materials such as Cu2O and CdS undergo photocorrosion upon exposure to light for a prolonged duration. Furthermore, although metal co-catalysts dramatically enhance the production rate and yield of Cl2/HOCl, their use in acidic media has limited stability. For instance, Okada et al. observed a partial dissolution of Pt co-catalyst in acidic seawater solution at pH 1 after one hour of photocatalysis; it must be noted that the dissolution of Pt only occurs under photoirradiation.32 Therefore, modifications to photocatalysts and photoanodes are often required to preserve stability in the presence of Cl− ions under highly oxidising conditions (Cl2 and HOCl) and/or under photoirradiation. For example, a MoOx layer is inserted between Co2(OH)3Cl and BiVO4 to suppress the photocorrosion of Co2(OH)3Cl by photogenerated holes of BiVO4.34Complex mixture of natural seawater
Natural seawater is an ideal reaction medium owing to its abundance and free availability. Although natural seawater contains Cl− ions as a majority, the presence of other cations (Na+, Ca2+, Mg2+, and Ba2+) and anions (HCO3−, CO32−, and SO42−) interferes with the surface of photocatalysts by selective adsorption and reaction, thereby reducing the overall production yield and rate of HOCl. Some of these ions, like CO32−, SO42− and PO43−, are effective hole scavengers that compete with Cl− oxidation, thereby reducing its selectivity.35 Additionally, HCO3−, CO32−, and PO43− have been shown to consume Cl2/HOCl.36 Furthermore, the adsorption of these ions on the surface of photocatalysts might block the diffusion and interaction of Cl− with the surface. Therefore, the deleterious effects of these ions are inevitable. As a result, designing photocatalytic materials that can perform efficiently in seawater containing various ions, as opposed to pure seawater solutions, is a major challenge in this research area. The average composition of natural seawater is summarized in Table 1.| Ion | Composition (%) |
|---|---|
| Cl− | 55 |
| Na+ | 31 |
| SO42− | 7.7 |
| Mg2+ | 3.7 |
| Ca2+ | 1.2 |
| K+ | 1.1 |
| HCO3− | 0.3 |
| Br− | 0.2 |
| CO32− | 0.03 |
| Sr2+ | 0.03 |
| Other ions | 0.09 |
3. Mechanism of Cl2 formation
Although Cl− oxidation to Cl2 looks like a simple 2-electron oxidation process, this mechanism is still under debate. Three mechanistic reaction pathways have been proposed based on theoretical calculations and experimental electrochemical studies: Volmer–Tafel, Volmer–Heyrovsky and Krishatalik.39Volmer–Tafel
| 2Cl− + 2* → 2Cl* + 2e− | (14) |
| Cl* + Cl* → 2* + Cl2 | (15) |
Volmer–Heyrovsky
| 2Cl− + * → Cl* + Cl− + e− | (16) |
| Cl− + Cl* → * + Cl2 + e− | (17) |
Krishatalik
| 2Cl− + * → Cl* + Cl− + e− | (18) |
| Cl− + Cl* → Cl*+ + Cl− + e− | (19) |
| Cl− + Cl*+ → * + Cl2 | (20) |
Three possible reaction pathways have been proposed based on theoretical computations, kinetic data and experimental verifications as plausible mechanisms. The elementary steps in COR are Volmer, Tafel, and Heyrovsky. The adsorption of Cl− on the active site of an electrode is termed the Volmer step. The Tafel step includes a combination of two adsorbed Cl* species forming a Cl2 product. The formation and desorption of a product following a reaction between an adsorbed species (Cl*) and a reactant (Cl−) is called the Heyrovsky step. In the Krishtalik step, a second electron transfer occurs between a reactant and an adsorbed species prior to product formation. The three mechanistic pathways are illustrated in Fig. 3.
 | ||
| Fig. 3 Scheme of the mechanistic pathways for chloride oxidation according to the mechanisms of Volmer–Heyrovsky, Volmer–Tafel, and Krishtalik. Reproduced from ref. 40. Copyright 2023, Wiley-VCH. | ||
Herein, we discuss the mechanism of the chlorine evolution reaction (CER) using a RuO2 model system, one of the best performing catalysts for the chlor-alkali process. The mechanistic reaction pathway is often proposed based on kinetic experimental data. However, kinetic data alone do not provide the nature of the active sites of a catalyst and intermediates. Moreover, the nature of the active sites has not been elucidated by in situ spectroscopy. Exner et al. performed ab initio atomistic thermodynamic calculations on the model catalyst O-covered RuO2(110).41 Electrochemical adsorption (Volmer step) of Cl− on the fully O-covered RuO2(110) surface at electrode potential, U = 0 V, costs a Gibbs energy change of 1.49 eV. In the Volmer–Heyrovsky mechanism, a direct reaction between an adsorbed chlorine (Cl*) and a chloride (Cl−) from the solution to yield gaseous Cl2 requires a Gibbs energy change of 1.23 and −0.13 eV at U = 0 V and 1.36 V, respectively. The net Gibbs energy loss in the Volmer–Heyrovsky mechanism is 0.13 eV. Meanwhile, the second Cl− adsorption in the Volmer–Tafel mechanism requires a Gibbs energy change of 1.59 and 0.23 eV at U = 0 V and 1.36 V, respectively. Therefore, the Gibbs energy loss is 0.23 eV in the Volmer–Tafel mechanism. The formation of a Cl+ ion in the Krishtalik mechanism results in a Gibbs energy loss of 0.85 eV, which is much higher than the Volmer–Heyrovsky and Volmer–Tafel mechanisms. Hence, the Krishtalik mechanism can be ruled out for CER on the fully O-covered RuO2(110) surface while favouring Volmer–Heyrovsky as the most plausible mechanism based on thermodynamic considerations. Additionally, doping a monolayer of PtO2 in RuO2(110) further lowers the Gibbs energy loss of 0.08 eV. Importantly, it should be noted that while the Volmer–Heyrovsky mechanism is the most plausible and preferred pathway, all proposed mechanistic pathways are feasible on the RuO2(110) surface.39 The simplified mechanism of Volmer-Heyrovsky on metal oxides (MOs) is given by
| M–O + Cl− → M–OCl + e− | (21) |
| M–OCl + Cl− → M–O + Cl2 + e− | (22) |
In a previous study, Khrishtalik proposed a three-elementary step mechanism following a combination of electrochemical measurements and electrokinetic data on RuO2–TiO2.42 A combination of a low coverage of intermediate and a low slope of the anodic polarisation curve provides a strong indication that the transfer of the first electron is reversible and the second electron transfer is a slow step. A total of two electrons per one Cl− is consumed in the slow step, suggesting that a positive Cl (Cl+) should react with a chloride (Cl−) to form Cl2. Khristalik then proposed a mechanism with three elementary steps (eqn (18)–(20)) for CER. Although this proposed mechanism based on electrokinetic data and electrochemical measurements seems plausible, it certainly does not provide a correlation with the physical and structural characteristics of electrocatalysts, a limitation that must be addressed to gain a correct understanding of CER selectivity on the electrode surface. Furthermore, the microscopic molecular level of insights is not captured and elucidated by electrokinetic data, which often requires computational calculations, such as ab initio DFT with thermodynamic considerations/frameworks. DFT calculations by Hansen et al. on chlorine evolution disclosed the possibility of Cl− adsorption on two oxygen atoms in RuO2(110), forming ClO2cc (cc denotes two coordinatively unsaturated sites).43 This result, in which ClO2cc is more positively charged than O2cc, is consistent with the Khrishtalik mechanism.
4. Detection and determination of Cl2 and HOCl concentrations
The photocatalytic activity in Cl− oxidation is assessed using the production rate and/or concentration of Cl2 or HOCl. The concentration of Cl2 in seawater splitting can be quantified using iodometric titration. The iodide-starch test is usually employed where iodide is oxidised to iodine in the presence of Cl2 as an oxidising agent, following eqn (23). The generated iodine is titrated with sodium thiosulfate using starch as a colour indicator to determine the concentration of iodine (eqn (24)), and thus Cl2 indirectly.44| Cl2 + 2I− → I2 + 2Cl− | (23) |
| I2 + 2S2O32− → 2I− + S4O62− | (24) |
The evolved Cl2 can also be detected using the N,N′-bis(2,4-di-sulfobenzyl)tolidine tetrasodium method. Briefly, Cl2 is purged from a photoreactor under an inert gas flow and trapped in a 4 M aqueous NaOH solution to form hypochlorite (ClO−) ions, and the amount of ClO− is determined using N,N′-bis(2,4-di-sulfobenzyl)tolidine tetrasodium (SBT).32 The oxidized form of SBT following a reaction with chlorine has a characteristic absorption at 675 nm at pH 5.2, which allows a spectroscopic detection.45 The concentration of HOCl is commonly determined spectroscopically using the N,N-diethyl-p-phenylenediamine (DPD) method. In the presence of HOCl, DPD is oxidized to a red species with characteristic absorption at 515 or 552 nm.
The selectivity for Cl− oxidation to Cl2 or HOCl in PEC systems is often evaluated in terms of faradaic efficiency (FE), which is the ratio of the measured HOCl/Cl2 concentration to the theoretical one when passed through current. Hence, the sum of FE for HOCl and O2 should ideally be unity (100%). The FE can be calculated based on the following formula (eqn (25)):
 | (25) |
5. Photocatalytic Cl2/HOCl production
Photocatalytic systems are much simpler than their photoelectrocatalytic counterparts and can be more readily scaled up. One advantage of nanoparticulate powders used in photocatalytic systems is gram-scale preparation. Furthermore, the former does not need a specialized set up and can be used in a simple test tube or beaker in a slurry reactor without an external bias (or applied potential). Photocatalytic systems run with a single-compartment reactor where both oxidation and reduction occur in the same compartment, unlike PEC systems, which have dual compartments containing an anode and a cathode for both half-reactions. This, in turn, often requires the use of an electron scavenger, e.g. molecular O2, to enhance the formation of Cl2 because both oxidation and reduction take place on the same photocatalytic system. Photocatalytic materials can be prepared using various wet chemistry methods, such as sol–gel, hydrothermal/solvothermal and colloidal synthesis. These photocatalytic materials can be annealed further at high temperatures to improve crystallinity and enhance photocatalytic performance.Metal oxides are commonly employed as photocatalysts owing to their chemical stability, suitable band gap, low toxicity, low-cost and facile preparation, as well as a highly positive valence band that is suitable for chloride oxidation reaction (COR). TiO2, WO3, Bi2WO6, and BiVO4 are the most extensively studied metal oxide photocatalysts. WO3 exhibits a moderate band gap (2.5–2.7 eV) and low toxicity and possesses suitable band edge positions for water and Cl− oxidation (Fig. 2). It is well known that photoactivity depends on the size, morphology, and defect chemistry of photocatalysts. Lu et al. synthesized various WO3 morphologies, including nanorods, nanowires, nanotube bundles, nanoplates and hollow microspheres, loaded with 1 wt% Pt as a co-catalyst and studied the structure-activity relationships in HOCl synthesis.46 Pt/WO3 hollow microspheres show the highest HOCl concentrations in 0.5 M NaCl and seawater at 14.52 µM (1.24 mg L−1) and 4.34 µM (0.354 mg L−1), respectively, with good stability up to four recycle tests. The author attributed the highest photoactivity to the hollow structure of WO3 that facilitates charge separation. This work achieved the recommended free chlorine (0.2–2 mg L−1) set by the WHO, highlighting its practical feasibility.
A systematic study on the effects of photocatalysts (BiVO4, TiO2, H2WO4, and Bi2WO6) and metal co-catalysts (Au, Ag, Cu, Rh, Pd and Pt) by Pang et al. disclosed that 1 wt% Pt/WO3 exhibited the highest photoactivity with 17.6 µM HOCl produced in 1 hour while suppressing water oxidation (Fig. 4a).10 A Pt co-catalyst enhances the catalytic activity by serving as an electron sink to capture photoexcited electrons from the conduction band (CB) of WO3 and reduce O2 to superoxide O2˙− or H2O. The same group also modified Pt/WO3 by co-loading with various other metals as a co-catalyst. The Pt–M (M = Ni, Cr, Mn, Co, and Zn) co-catalyst improves the selectivity toward Cl− oxidation to HOCl and promotes the decomposition of H2O2 (a product of ORR) to prevent its reaction with formed HOCl (Fig. 4b); Pt–Mn/WO3 shows the highest HOCl production (37 µmol h−1), which is twice that of Pt/WO3 in an earlier work.47 However, the dissolution of Mn presents a problem for recyclability and long-term usage of Pt–Mn/WO3. Moreover, precise control must be taken to avoid the formation of MnOx, which favours the oxidation of water over Cl− oxidation.48 Interestingly, the formation rate of HOCl is suppressed severely over Pt–M (M = Fe, Cu, and Ag). To ease recyclability in heterogeneous photocatalysis, a magnetic material, like Fe3O4, is typically incorporated into catalytic material designs for magnetic separation. Wang et al. constructed a multishell quaternary Pt/WO3/SiO2/Fe3O4 photocatalyst that achieved 6.31 µM HOCl in 2 hours.49 The SiO2 intermediate layer prevents an electron transfer from WO3 to Fe3O4, inhibiting the loss of activity in HOCl generation.
 | ||
| Fig. 4 Schematic of the Cl− oxidation mechanism over (a) Pt/WO3; reproduced from ref. 10. Copyright 2020, the American Chemical Society; and (b) Pt-Mn/WO3 photocatalysts; reproduced from ref. 47. Copyright 2022, the Royal Society of Chemistry. | ||
A family of metal tungstate (MWO4, where M is a bivalent metal) has emerged as a new type of photocatalyst owing to its low toxicity, low-cost and earth-abundant starting materials.50 Wide band gap ZnWO4 nanorods show a hypochlorite (OCl−) yield of 1.01 µg mL−1 in seawater.51 The production yield increases 2.5-fold when ZnWO4 is loaded with an IrO2 co-catalyst due to higher selectivity of Cl− oxidation on IrO2. More recently, Han et al. reported that MnWO4 with a band gap of 2.83 eV could produce 12.6 µM h−1 HOCl in 0.5 M NaCl with good stability and recyclability of up to four runs, paving the way toward promising noble metal-free photocatalysts.52 In another work, the same group constructed a Z-scheme WO3/CdS photocatalyst. An excited electron in the CB of CdS serves to reduce O2 to ˙O2− and a photogenerated hole in the VB of WO3 oxidises Cl− to free Cl2 (3.54 mg L−1),53 with a much higher activity than Pt/WO3 (1.24 mg L−1 free Cl2).46 This work demonstrates the feasibility of designing promising noble metal-free photocatalysts for HOCl synthesis. Notably, noble metals, like Pt and Au, are not involved in Cl− oxidation but are used to quench the excited electrons by capturing them from the VB of the photocatalyst. A similar quenching role can be applied through a Z-scheme mechanism or other types of photocatalyst design, thereby reducing the material cost in catalyst development.
In 2020, Li et al. investigated the origin of elevated Cl2 in the atmosphere using KCl-coated TiO2 to understand atmospheric Cl2 chemistry in urban areas.54 Although this work is not directly related to COR via seawater splitting, it shows a strong correlation between the amount of surface-bound Cl− and the amount of Cl2 evolved. TiO2 is widely used in photocatalytic research due to its low cost, low toxicity and high chemical stability in a wide pH range.55 Nonetheless, due to the fast charge carrier recombination and low photocatalytic activity, metal nanoparticles are often required to boost photoactivity. Okada et al. systematically studied the effects of pH on seawater splitting over Pt/TiO2 (0.1 wt% Pt) and demonstrated a near stochiometric evolution of H2 and Cl2 with a minor O2 evolution from direct water oxidation for over 14 hours (Fig. 5a and b); the selectivity toward Cl2 formation is 81.1%, highlighting preferential oxidation of Cl− over water oxidation.32 High Cl2 evolution was detected in an acidic (pH 1) seawater solution driven by the change in the VB maximum of TiO2, contributing to stronger Cl− oxidation at low pH. In another work, Jadwiszczak et al. found that PEC Cl2/HOCl synthesis in acidic environments can suppress their decomposition to chlorates and oxygen, thus favouring acidic conditions for promoting high Cl2/HOCl yields.33 Nonetheless, the Cl2 evolution could not be detected in neutral and basic conditions in Okada's work. The reason for inactivity is attributed to the rapid decomposition of HOCl to ClO3− and Cl− ions. Additionally, the competing reactions between Cl− and OH− oxidation at high pH, which favours the latter, could be another reason for inactivity.56,57 More recently, the same group studied the effects of Pt loading on the production rate of H2 and Cl2.58 The rate of gas evolution increases as the Pt loading increases and reaches a maximum at 1.0 wt% (Fig. 5c). Beyond 1.0 wt% of Pt loading, the photoactivity decreases most likely due to the light-shading effects of Pt. Notably, it is worth noting that Pt dissolution in an acidic seawater solution is triggered by photoirradiation, with two plausible mechanisms of dissolution. First, photogenerated electrons reduce PtOx to Pt, and PtOx reacts with Cl− to form PtCl62− species, which are soluble in an aqueous solution. In the second mechanism, photogenerated electrons and holes reduce PtOx and oxidize Cl− to Cl2, respectively, where Cl2 then reacts with Pt to form soluble PtCl62−.
 | ||
| Fig. 5 Photocatalytic seawater splitting (0.5 M NaCl) over Pt/TiO2 (a) at different pH values (1, 6, 13) and (b) time course reaction over 14 hours. Reproduced from ref. 32. Copyright 2024, the Royal Society of Chemistry. (c) Time course reaction with different Pt loadings (wt%) on TiO2. Reproduced from ref. 58. Copyright 2025, Elsevier. | ||
Rh/Cr2O3 and RhCrOx alloy are used extensively as a co-catalyst for the hydrogen evolution reaction (HER) in photocatalytic water splitting.59–62 Importantly, CrOx overlayers have been shown to act as a protective layer to prevent unwanted side reactions and undesirable species in water splitting.63 Nonetheless, their use in seawater splitting is relatively unexplored; thus, the knowledge about their roles and performance in seawater splitting is limited. Okunaka et al. investigated the role of co-catalyst (RhOx, CrOx, and RhCrOx) loaded on SrTiO3:Al for HOCl production in aqueous NaCl solution.64 It was found that only RhCrOx shows a significant HOCl production along with H2 evolution, where the Rh species promotes H+ reduction and CrOx suppresses HOCl decomposition. Additionally, the role of RhCrOx is to enhance the charge separation between the co-catalyst and the photocatalyst.
The extraction and insertion of surface lattice atoms/ions via self-oxidation of lattice Cl− in Cl-containing photocatalysts (e.g. AgCl and BiOCl) to Cl2 by a photogenerated hole provide smooth oxidation, often followed by a structural change during a catalytic cycle. The lost/oxidized lattice Cl− is then replaced by Cl− ions in the bulk seawater solution, creating a chlorine cycle. The participation of lattice atoms/ions as active species in chemical reactions is known as the Mars–van Krevelen (MvK) mechanism. Hirai's group reported that the compensation of Cl− ions in seawater solution suppresses photocatalyst deactivation.65 In light of this characteristic, AgCl and BiOCl have advantages for photocatalytic Cl− oxidation in seawater splitting. Although wide band gap AgCl scarcely exhibits photocatalytic activity, loading Au NPs onto AgCl drastically boosts Cl− oxidation with a HOCl formation yield of 21.4 µmol, as reported by Shiraishi et al.66 The localised surface plasmon resonance (LSPR) in Au NPs enables visible light (>420 nm) absorption to create hot electrons (ehot−) and holes (hhot+), where the latter oxidises lattice Cl− in AgCl to Cl2 (Fig. 6). Although the redox potential of Cl−/Cl2 (1.36 V vs. RHE) is more positive than the VB of Au NP, the photogenerated holes in the VB of Au NP can oxidize the lattice Cl in AgCl, circumventing the insufficient redox potential. The oxidation of lattice Cl− was confirmed by XPS analysis and control experiments.
 | ||
| Fig. 6 Band energy diagrams of (a) AgCl and (b) Au/AgCl. Reproduced from ref. 66. Copyright 2023, the American Chemical Society. | ||
Another promising photocatalyst that has garnered attention is the non-toxic, inexpensive, and wide band gap BiOCl (3.1–3.3 eV). The structure is made of [Bi2O2]2+ slabs interleaved between double slabs of [Cl]− along the [001] direction, which creates an internal electric field (IEF) along the crystal orientation perpendicular to the [Bi2O2]2+ and [Cl]− slabs that promote charge separation and migration (Fig. 7a–c).52 Atoms in the [Bi2O2]2+ layers are held by a covalent bond, while the interaction between the [Cl–Bi–O–Bi–Cl] stacks is held together by a non-covalent van der Waals force.67 Therefore, the removal of Cl− ions from the crystal structure of BiOCl is much easier owing to the weaker van der Waals interaction, which is beneficial for Cl− lattice extraction and insertion, as demonstrated in chloride ion batteries,68 which may have implications for photocatalytic Cl− oxidation.
 | ||
| Fig. 7 Simplified surface of BiOCl with (a) different facets. (b) Side view of the surface along the [001] direction and (c) top view of the surface along the perpendicular [110] direction. TEM images of BiOCl (d) before reaction and (e) after reaction. DFT calculations of the free energy for the Cl2 evolution reaction on the (f) (001) surface and (g) (110) surface. Reproduced from ref. 56. Copyright 2021, Elsevier. | ||
Wang et al. demonstrated that the insertion/extraction of Cl− proceeded on the {110} facet, as evidenced by the photocorroded {110} facet of the BiOCl microplates after several catalytic runs (Fig. 8d and e).56 The finding is reinforced by DFT calculations, which suggest that the open channel {110} facet exhibits lower overpotential and easier lattice Cl− transport than the {001} facet, giving simultaneous advantages of Cl− extraction and oxidation (Fig. 8f and g). In another work, Zhou et al. systematically investigated the effects of different facets {001} and {110}, on the formation rate and yield of HOCl. The highest production rate of RCS (95.9 µg L−1 min−1) was achieved with the largest {001}/{110} ratio owing to the largest IEF, as verified by transient photocurrent response, EIS and PL spectroscopy; the lowest {100}/{110} ratio of BiOCl yields 75.6 µg L−1 min−1.36 The Cl− lattice oxidation and extraction contribute to the weakening of Bi–Cl vibrations, the emergence of metallic Bi (reduction of Bi3+ to Bi0), and the formation of oxygen vacancies, as confirmed by Raman spectroscopy, XRD and XPS.36,69 The presence of molecular O2 helps to prevent self-oxidation of lattice Bi3+ and regulate the electronic structure of BiOCl by adsorbing to the oxygen vacancies (V0), usually leading to a higher photoactivity in Cl− oxidation.
 | ||
| Fig. 8 (A) Transient photocurrent measurements for the as-deposited WO3 electrodes at (i) 0.8 V, (ii) 0.5 V, and (iii) 0.2 V vs. SCE and (iv) annealed WO3 electrode at 0.5 V vs. SCE. Illustration of charge behaviour (B) when irradiated with light, (C) under photostationary conditions, and (D) when light is off. Reproduced from ref. 79. Copyright 2013, the American Chemical Society. | ||
Doping foreign elements (non-metals or metals) into a host material has been a straightforward strategy for improving charge transport properties. In 2016, Li et al. doped Bi3O4Cl with carbon to boost the IEF by 126 times and achieved a bulk charge separation efficiency of 80% that enabled photocatalytic water oxidation without noble metals or electron scavengers.70 Motivated by their work, Xu et al. reported that carbon-doped BiOCl (C-BiOCl) was found to enhance IEF and improve charge separation and migration as well as preference for Cl− oxidation over water oxidation in photocatalytic Cl2 formation.71 A 2-fold enhancement in IEF in C-BiOCl leads to a 2-fold increase in the production rate of Cl2 (0.23 mg L−1 min−1) compared to that of pristine BiOCl (0.11 mg L−1 min−1), underscoring a strong correlation between IEF and photoactivity. Nitrogen doping in BiOCl has the advantage of providing basic Lewis sites for reduction reactions. Shi et al. attempted the thermal decomposition of ammonium oxalate that delaminated bulk BiOCl into atomic-layer nanosheets and simultaneously introduced N dopants and O vacancies (N–OV associate). The N–OV associate reduces the exciton binding energy (from 199 to 46 meV) and lifetime (107.5 to 25.5 ms) to facilitate dissociation of an exciton into free charge carriers, leading to a HOCl production rate of up to 87.2 µmol g−1 h−1 (Table 2).72
| Photocatalyst | Light source | Reaction solution | [HOCl]a/µM | Time /h | AQY (%) | Ref. |
|---|---|---|---|---|---|---|
| a The default product measured was set to HOCl as determined using the DPD method. However, some studies reported the yield of Cl2. b SCC refers to solar-to-chemical conversion. | ||||||
| 0.1% Pt/TiO2 | LED 365 nm | 0.5 M NaCl (pH 1) | Cl2: 10.3 | 1 | ∼0.65% (365 nm) | 32 |
| 1% Pt/TiO2 | LED 365 nm | 0.5 M NaCl (pH 1) | Cl2: 70 µmol h−1 | — | 4.3% (365 nm) | 58 |
| 1% Pt/WO3 | AM 1.5 G | 0.5 M NaCl | 17.6 | 1 | 2.3% (420 nm) | 10 |
| Pt–Mn/WO3 | 300 W Xe (>420 nm) | 0.5 M NaCl | 37 | 1 | — | 47 |
| 1% Pt/WO3 hollow microsphere | Visible light | 0.5 M NaCl | 14.52 | 2 | — | 46 |
| Seawater | 4.34 | 1 | — | |||
| Pt/WO3/SiO2/Fe3O4 | White LED | 0.5 M NaCl | 6.31 | 2 | — | 49 |
| MnWO4 | Visible light | 0.5 M NaCl | 12.6 | 1 | SCCb 0.15% | 52 |
| IrO2/ZnWO4 | UV 254 nm | Seawater | 2.49 µg mL−1 | 1 | — | 51 |
| CdS/WO3 | LED (380–780 nm) | 0.5 M NaCl | 3.54 mg L−1 | 2 | — | 53 |
| 0.1% RhCrOx/SrTiO3:Al | AM 1.5 G | 1 M NaCl | ∼1 µmol | 1 | — | 64 |
| Au/AgCl | Xe lamp (>420 nm) | 0.55 M NaCl | 21.4 µmol | 24 | SCCb ∼0.03% | 66 |
| BiOCl | UV lamp | 6 g L−1 NaCl | Cl2: 0.125 mg L−1 min−1 | 1 | — | 69 |
| BiOCl | UV 254 nm | 6 g L−1 NaCl | 9.8 µmol | 0.5 | — | 56 |
| BiOCl | UV 254 nm | 6 g L−1 NaCl | 95.9 µg L−1 min−1 | — | — | 36 |
| C-BiOCl | UV 254 nm | 6 g L−1 NaCl | 0.023 mg L−1 min−1 | 1 | — | 71 |
| BiOCl–N–Ov | 300 W Xe (>400 nm) | 0.5 M NaCl with CO2 saturation | 87.2 µmol g−1 h−1 | 4 | 0.69% (400 nm) | 72 |
| BiOCl | UV lamp | 0.51 M NaCl | 19.8 µmol | 0.5 | — | 73 |
The ocean absorbs around 30% of the CO2 that is emitted to the atmosphere, which consequently decreases the pH of the ocean, also known as ocean acidification. The dissolution of CO2 in water forms weak carbonic acid (H2CO3), which exists in equilibrium with (H+) and bicarbonate (HCO3−) ions. A recent work revealed that dissolved CO2 in a NaCl solution boosted the formation rate of Cl2 on BiOCl by over 3-fold and 2-fold compared to N2-rich and O2-rich atmospheres, respectively.73 SEM images of BiOCl before and after photocatalysis disclose interlaminar deformation and distortion, which are probably caused by the insertion of CO2 into the interlayers and anion exchange. The proposed mechanism of CO2-mediated formation of Cl2 in synthetic seawater is given by
| HCO3− → CO32− + H+ | (26) |
| CO32− → ˙CO3− + e− | (27) |
| ˙CO3− + H2O + h+ → HCO4− + H+ | (28) |
| HCO4− + 2Cl− + 2H+ → HCO3− + Cl2 + H2O | (29) |
6. Photoelectrochemical (PEC) Cl2/HOCl production
In a photoelectrocatalytic system, HOCl formation occurs at the photoanode, and other reduction reactions proceed at the photocathode, including H2 evolution, H2O2 production or CO2 reduction (in the presence of CO2). Advantages of photoelectrocatalysis are relatively simple electrode construction with tailored properties, such as controlled thickness, porosity, defects (or vacancies) and other surface properties. Photoelectrode performance is highly dependent on the interface, morphology and thickness of the thin film electrode. The thickness affects the light absorption (up to several µm), photocurrent generation and charge carrier diffusion length (up to several hundred nm). These properties are easily optimized to maximize photocurrent responses and charge transport efficiency. Moreover, it allows the use of various techniques, like layer-by-layer deposition, electrodeposition, pulse-laser deposition (PLD), and successive ionic layer adsorption and reaction (SILAR). Additionally, catalytic materials can be prepared using common methods, such as sol–gel and hydrothermal, and successively deposited on conducting substrates (ITO, FTO) by doctor-blading, drop-coating and spin-coating.WO3 exhibits high chemical stability in acidic solution (pH < 4), which is beneficial for photoelectrochemical Cl2 synthesis (eqn (4)) but prone to dissolution in neutral or basic media.60 An acidic seawater solution due to the presence of formed HOCl is advantageous for WO3 because it ensures the WO3 stability. At a higher pH, WO3 forms WO42− by reacting with OH−, leading to its dissolution in alkaline media. As early as 1987, Desilvestro and Gratzel reported ∼76% FE of Cl2 production over polycrystalline WO3 in 1 M HCl.74 It has also been demonstrated that porous structures of WO3 favour Cl− oxidation over water oxidation. Recently, highly porous WO3 nanosheets with a specific surface area of 100 m2 g−1 and a thickness between 10 and 20 nm prepared by chemical bath deposition achieved 100% FE.75 However, a rapid decrease in FE as the charge increased was observed most likely due to the formation of higher chlorine oxo species. Hill and Choi showed that WO3 in PEC favoured the oxidation of Cl− (with 95% consumption of photogenerated holes) and completely suppressed water oxidation to O2 and peroxo species in aqueous NaCl solution.76 A similar observation of suppression of water oxidation was reported by Mi et al. using a 0.1 M HCl aqueous solution.77 These studies demonstrated WO3 thin films as a promising photoanode for Cl2 evolution.
Shi et al. reported a stable WO3 film prepared by 25 cycles of deposition-annealing process that produced 1.95 mA cm−2 at 1.23 V (vs. RHE) in natural seawater. However, the FE of Cl2 evolution was quite low (56%).78 Thick, nanoporous WO3ca. 3 µm with particle size of 20–30 nm provides a large internal surface area that allows contact with electrolytes, achieving large saturation photocurrents of 4.5 mA cm−2 at 0.76 V (vs. Ag/AgCl), although the FE is moderate (70%) due to competing O2 evolution.33
Annealing of thin films is usually performed to remove excess solvent, improve the crystallinity, control the structural phase, and create a strong interface contact between the sample and a substrate (ITO and FTO). A “roll-on” process was employed to deposit commercial nano-WO3 on ITO. The annealed WO3 film at 500 °C shows a much higher transient photocurrent and Cl2 concentration due to enhanced interparticle charge transport by hopping between aggregates compared to the as-made WO3 film (Fig. 8).79 The influences of annealing temperature on the compositional phases and PEC performance of WO3 electrodes were reported by Jo et al. At the annealing temperature of 500 °C, the WO3 contained predominantly monoclinic (87%) and orthorhombic (13%) phases, which gave the highest photocurrent of 2.7 mA cm−2 and FE of 93% at 1.0 V (vs. Ag/AgCl).80 The fraction of monoclinic increases as the annealing temperature increases, which induces more defects and increases donor density for enhanced PEC performance. However, too large a concentration of defects can be detrimental to the PEC performance of WO3 in Cl− oxidation because defects could serve as a recombination centre.75
Recently, Johnston and co-workers employed photoinduced absorption spectroscopy (PIAS) and transient photocurrent (TC) measurements to unravel the reaction order of Cl− oxidation on a WO3 photoanode.18 It has been found that the rate of Cl2 formation has a first-order dependence on the surface holes, [hs+], albeit the faradaic efficiency is not unity (62%). The remaining 38% is speculated to be due to the formation of higher oxo species, such as ClO4− or Cl2, trapped in the PEC cell, as no O2 was detected in the study. Such a high selectivity of Cl− oxidation over water oxidation reinforces the lower overpotential and higher exchange current density of the former reaction.77,81
Proton intercalation can modify the electronic structure and optical properties of WO3, rendering HxWO3 a promising photocatalyst due to increased electron mobility and high conductivity.82 Remarkably, Bartlett's group unravelled that fast electron injection from Cl− ions into the VB of HxWO3 leads to 100% FE for Cl− oxidation.83 Such fast electron injection also suppresses oxidation of W5+ by holes, giving high photostability and stable photocurrent during Cl− oxidation in 0.5 M NaCl at pH 4.
BiVO4 has a moderate band gap (2.4–2.5 eV) and a deep valence band suitable for many oxidation reactions; therefore, it has been widely employed as a photoanode in water oxidation under visible light irradiation.84,85 Nonetheless, the fast charge carrier recombination, low electron mobility, poor charge transfer and short hole diffusion length (∼70 nm) become obstacles to achieving its theoretical maximum STH (9.2%) and current efficiency (7.5 mA cm−2).86 Doping BiVO4 with other atoms or ions is a straightforward and efficient strategy that increases charge carrier density, improves charge separation and transfer efficiencies, introduces surface states, modifies the electronic band structure and enhances IPCE of BiVO4.87–89 Mo-doped BiVO4 exhibits enhanced IPCE, charge accumulation and FE of 78% with ∼7.5 µmol ClO− produced at ∼2.1 C compared to the undoped BiVO4 owing to the increased donor density by Mo dopants in BiVO4.90 Similarly, Luo et al. reported that Mo-doped BiVO4 showed a 6-fold increase in photocurrent and 42% IPCE in the wavelength range of 365–440 nm compared to pure BiVO4 (10% IPCE).91 The Mo6+ substitution of V5+ in BiVO4 enhances electron donation and creates more carrier density, and further loading with the RhO2 layer results in improved photostability in seawater.
The use of a multilayer strategy to improve charge separation and transport efficiency, as well as catalytic activity, is ubiquitous in designing efficient photoanodes.86 In the construction of a multilayer photoanode, WO3 is first deposited onto an FTO glass prior to BiVO4 deposition, in which the WO3 layer serves as an electron transport layer (ETL) and a hole mirror to block the transfer of holes to the FTO substrate, leading to an enhanced charge transfer. It was reported that a multilayer BiVO4/WO3 photoanode exhibited a low applied bias (0.5 V) to achieve high current efficiency (∼80%) and HOCl concentration (660 µM) in the 2000 s under optimised conditions.9 Without the WO3 underlayer, the formation yield and current efficiency of HOCl are lower and require a much higher external bias. A similar BiVO4/WO3 photoanode has been extended to the PEC production of HOBr.92
Surface passivation with metal oxide overlayers has several benefits. The overlayer can serve as a protective layer to prevent photocorrosion or dissolution, as a co-catalyst to enhance activity and selectivity in Cl− oxidation, as a hole (or electron) transport layer to boost charge transport, and as a passivating layer to quench surface states.93 Okunaka et al. explored several metal oxides (MOx; M = Mn, Co, Ni, Fe, Pt, and Rh) as a surface passivator on BiVO4/WO3 photoanodes to investigate the selectivity (O2 evolution vs. HOCl production) of oxidation half-reaction.48 All metal oxides except MnOx favour the formation of HOCl owing to their lower overpotential for Cl− oxidation (Fig. 9a). However, it should be noted that an accurate determination of each (O2 and HOCl) overpotential in these photoanodes is difficult because both water and Cl− oxidation occur with different selectivities.
 | ||
| Fig. 9 (a) Faradaic efficiency of the HClO and O2 formation on various metal oxide overlayers. Reproduced from ref. 48. Copyright 2020, Elsevier. (b) Cl2 production rate and faradaic efficiency with different CoOx thicknesses. Reproduced from ref. 94. Copyright 2023, the American Chemical Society. (c) HOCl concentrations with varying CoOx precursor concentrations. (d) Effects of calcination temperature on the HOCl concentration and faradaic efficiency. Reproduced from ref. 95. Copyright 2021, the Royal Society of Chemistry. | ||
A unique feature of monoclinic BiVO4 is spatial charge separation by different crystallographic facets. BiVO4 is known to accumulate photogenerated holes at the {110} facet for oxidation reactions and to direct photoexcited electrons to the {010} facet for reduction reactions.76 Hence, selective deposition of cobalt borate (CoBi) as a co-catalyst on the WO3/BiVO4 {110} facets has been observed to enhance the charge transfer kinetics, photocurrent response and conversion of photogenerated holes to redox species at the anodic sites for improved Cl2 production.96
A thin layer coating with amorphous cobalt oxide (CoOx) prepared by pulsed laser deposition is demonstrated to act as a co-catalyst and to protect BiVO4 against corrosion. The thickness of CoOx overlayer from 2 to 5 nm is controlled using the number of laser pulses, with the optimum thickness of 3 nm achieving a current density of 2.5 mA cm−2 at 1.2 V (vs. Ag/AgCl) and 60% FE in 3 M KCl solution at pH 7 (Fig. 9b).94 By studying various CoOx precursor concentrations, Sayama's group achieved near 100% FE of HOCl formation with 0.01 M CoOx precursor concentration (Fig. 9c).95 Such an enhancement in FE is attributed to improved charge separation, efficient capture of photogenerated holes by CoOx, and possibly the presence of active sites for Cl− adsorption on CoOx.97 These studies showed that beyond a certain CoOx thickness (>3 nm) and loading (>0.03 M), the FE and current density decrease due to increased charge carrier recombination, thus requiring optimisation studies to correlate to reach the right conclusion.
Sayama's work also showed that the calcination temperature of CoOx precursor should be optimized at 400 °C because a higher temperature (>500 °C) promotes Co doping in BiVO4, which may favour recombination of charge carriers, thus lowering the FE (Fig. 9d).95 However, this finding seems to contradict Chauhan's work, which demonstrates that Co doping in BiVO4 reduces the charge transfer resistance, facilitates charge carrier lifetime and improves the Cl− oxidation kinetics.98 The authors investigated the effect of Co dopant concentration in BiVO4 and found that 0.05 mol% Co gives the highest photocurrent of 0.190 mA cm−2 at 1.1 V vs. RHE and 92% FE, which exhibited excellent stability against photocorrosion in acidic (pH 2.3) medium.98 Other Co dopant concentrations yield much lower photocurrent and higher onset potential. The PEC performance is highly dependent on dopant concentrations; therefore, a systematic study of Co dopant concentration and its effect on the PEC performance must be optimized to achieve maximum FE and current density, as well as to resolve conflicting findings with other works.
The instability of BiVO4 in electrolytes and acidic medium poses a problem for PEC reactions because it causes the dissolution of V5+ ions, leading to compositional and structural changes.99,100 Chen and Wang theoretically demonstrated that BiVO4 undergoes photocorrosion in an acidic medium (pH = 0) to form BiCl3.101 To prevent photocorrosion, a 20 nm-thick amorphous WO3 layer is applied as a coating on BiVO4 owing to its high chemical and electrochemical stability in acidic media and high selectivity for the COR over the WOR (Fig. 10a–c).102 This strategy achieved a photocurrent density of 2.5 mA cm−2 at 1.42 V (vs. RHE) and FE of 85% (Fig. 10d). The photocurrent is remarkably stable for up to three hours.
 | ||
| Fig. 10 (a) Illustration of the BiVO4/WO3 photoanode, (b) energy level diagram of the BiVO4/WO3 photoanode, (c) TEM image of BiVO4/WO3, and (d) photocurrent density across the applied potential. Reproduced from ref. 102. Copyright 2019, Springer Nature. | ||
Pristine TiO2 suffers from fast charge carrier recombination and separation. Modification strategies, such as doping, defect creation, and structural transformation are often performed to improve charge separation and transfer. 1D TiO2 nanostructures, such as nanorods and nanotubes, are favourable photocatalysts owing to their high specific surface area and improved charge transport properties. TiO2 nanotubes demonstrate a higher FE of 60.6% at 0.2 V (vs. Ag/AgCl) than TiO2 nanoparticles prepared by sol–gel for Cl2 formation.103 Ye et al. introduced oxygen vacancies (Vo) in TiO2 nanotubes (TiO2-Vo) and achieved 37.1 µmol h−1 cm−2 of Cl2 with FE of 73.9%.44 The improved PEC performance of oxygen-deficient TiO2 nanotubes by 1.5-fold compared to bare TiO2 nanotubes is attributed to the enhanced utilization of photogenerated charge carriers promoted by oxygen vacancies rather than selective COR over OER. Another study also showed an improved photocurrent in rutile TiO2 with Vo and stability of Ti3+ surface states over 5 hours in PEC conditions.57 It was suggested that Ti3+ sites allow Cl− ions to bind and react with photogenerated holes; however, no concrete spectroscopic evidence was provided in the study. Another study by Wu et al. combined DFT calculations and EPR trapping experiments to reveal that the activation of Cl− on TiO2-Vo occurs spontaneously (negative Gibbs free energy).104 In summary, these findings highlight that oxygen vacancies (Vo) are highly beneficial for enhanced Cl− adsorption and oxidation, leading to enhanced PEC performance in the Cl2/HOCl formation (Table 3).104,105
| Photoanode | [HOCl] or [Cl2] | Photocurrent (mA cm−2) | Potential (V) | FE (%) | Ref. |
|---|---|---|---|---|---|
| a Referenced to SCE. b Referenced to Ag/AgCl. c Referenced to RHE. d Two-electrode cell system. e The steady current was set to 2 mA on a 1.5 × 5 cm2 electrode and potential (vs. SHE). f At 2.3 V vs. Ag/AgCl. | |||||
| WO3 | — | 0.5–0.6 | 0.5–0.6a | 76 | 74 |
| WO3 | — | ∼0.8 | 1.6b | 100 | 75 |
| WO3 | — | 3–3.5 | 0.9b | 100 | 76 |
| WO3 | 140 µmol | 1.95 | 1.23c | 56 | 78 |
| WO3 | — | 4.5 | 0.76b | 70 | 33 |
| WO3 | — | 2.7 | 1.0b | 93 | 80 |
| WO3 | — | ∼0.62 | 1.0b | 62 | 18 |
| WO3 | — | ∼0.21 | 1.6d | 30 | 81 |
| WO3 | — | ∼0.60 | 1.23c | 100 | 83 |
| Mo:BiVO4 | — | ∼0.05 | 1.2b | 78 | 90 |
| RhO2/Mo–BiVO4 | ∼1.25 µmol h−1 | 2.16 | 1.0c | — | 91 |
| BiVO4/WO3 | 60 µM (500 s) | ∼3.0 | 0.22d | 81 | 9 |
| 660 µM (2000 s) | 0.50d | ∼80 | |||
| CoOx/BiVO4/WO3 | — | 2 | 0.68e | 88 | 48 |
| CoOx/BiVO4/WO3 | 12.5 µmol h−1 cm−2 | 2.5 | 1.2b | ∼60 | 94 |
| CoOx/BiVO4/WO3 | ~160 μM | ∼2.3 | 1c | 100 | 95 |
| CoBi/BiVO4/WO3 | 39.2 mg L−1f | 0.38 | 1.2b | — | 96 |
| Co:BiVO | — | 0.19 | 1.1c | 92 | 98 |
| WO3/BiVO4 | 6 µmol | 2.5 | 1.42c | 85 | 102 |
| TiO2-NT | — | ∼0.5 | 0.2b | 60.6 | 103 |
| TiO2-NT/Ti | 37.1 µmol h−1 cm−2 | 3.21 | 1.23c | 73.2 | 44 |
| TiO2 (rutile) | — | 8 | 1.0a | — | 57 |
| TiO2-Vo | 48.7 mg L−1 h−1 | ∼2.5 | 0.5a | — | 104 |
7. Scalability challenges
PC and PEC seawater splitting demonstrates the feasible production of Cl2/HOCl. Both the systems have merits and challenges in efficiently producing Cl2/HOCl. The advantages, limitations and challenges of both the systems are summarised in Table 4.| System | Photocatalysis (PC) | Photoelectrochemistry (PEC) |
|---|---|---|
| a The recommended Cl2 concentration for water treatment is a maximum of 4 mg L−1 for drinking water, as recommended by US EPA, and 0.2–0.5 mg L−1 for residual disinfection of free chlorine, as recommended by WHO. | ||
| Efficiency | Produced Cl2/HOCl concentration has already reached the benchmark value for water treatment set by WHO.a However, the AQY and SCC efficiencies are below 10% and 0.1%, respectively | Much higher Cl2/HOCl concentration obtained than the benchmark value set by WHO, giving higher SCC efficiencies than PC |
| The moderate operating potential (<1.2 vs. RHE) for most photoanodes is an advantage for higher efficiency | ||
| Selectivity | High selectivity of Cl2/HOCl is usually obtained. In many works, minimal O2 evolution from water oxidation has been observed due to its sluggish kinetics | Faradaic efficiency is usually high (>90%) for most metal oxide photoanodes, and the use of applied voltage is rather moderate |
| Scalability | Construction of large-scale PC panels or planar reactors is straightforward (without electrical wiring and connectivity) and cost-effective | Complex configuration, including external electrical bias integration of PEC cells, makes up-scaling difficult |
| Photocatalyst films usually have lower SCC efficiencies than photocatalyst nanopowders | Photoelectrode thin films often exhibit non-uniformity for large electrodes, reducing the current density and efficiencies | |
| The leaching of immobilised photocatalysts from the films might be a problem | High Cl2/HOCl concentrations obtained might be a merit for small-scale demonstration of water treatment using small portable devices | |
| Issues with oxygen flow input, mass transfer, and separation of oxidation (Cl2/HOCl) and reduction (H2) products | Product separation is readily available in two-compartment PEC cells | |
| Cost | Construction of panels or planar reactors is relatively cost-effective | Complex configuration for large-scale PEC cells makes it less cost-effective than PC reactors |
| Material cost is relatively high due to the use of noble metals (Pt, Au, and Rh) as a co-catalyst and electron sink | Material cost is low because many performing photoanodes are transition metal oxides | |
| The recovery of leached photocatalysts adds an extra cost | Electrical energy input is required to provide the applied potential | |
Industrial and commercial practical applications require the high-performance, cost-effective, and long-term stability of photocatalytic materials (photocatalysts and photoelectrodes) and innovative large-scale reactor designs. These factors constitute scalability challenges for the large-scale production of Cl2/HOCl. The efficiencies of photocatalytic materials are usually expressed using apparent quantum yield (AQY) and/or solar-to-chemical conversion (SCC). Many excellent photocatalysts with high AQY are wide-band gap semiconductors, such as TiO2, WO3, and BiOCl, which restrict the efficient utilization of visible light in the solar spectrum. Therefore, a search for other promising photocatalysts or modifications of wide-band gap photocatalysts is highly desirable.
The choice of photocatalytic materials is essential for achieving a high production rate and yield of Cl2/HOCl. Single-component photocatalysts are often restricted by low photon absorption, fast charge carrier recombination and slow charge transfer, which contribute to low SCC and AQY in the visible light range. A typical and convenient method to improve charge carrier separation and transfer is to load a noble metal (Pt, Au, and Rh) co-catalyst onto a photocatalyst. Metal co-catalysts serve as an electron sink and a catalytic site for reduction reactions. This, however, places an additional material cost on the system. Many works have attempted to limit the usage of such noble metal to below 1% wt. to achieve a compromise between high photocatalytic efficiency and material costs. Another promising strategy is to construct heterojunction photocatalysts, like Z-scheme or S-scheme, in photocatalysis to enhance carrier separation and transfer, as well as retain strong redox potentials for chloride oxidation.
The long-term stability of photocatalytic materials presents another challenge. The issues are corrosion in an acidic medium due to the presence of Cl2/HOCl, and photocorrosion by charge carriers (electrons and holes) under light irradiation. Sayama's group demonstrated photocorrosion and dissolution of Pt co-catalysts, forming soluble PtCl62− when reacting with Cl− in seawater. One simple strategy is to create a core–shell Pt-MOX (MOX = metal oxide) co-catalyst where a MOX shell protects the Pt core from photocorrosion and dissolution (Fig. 11a). A few stable and robust metal oxides have been commercially employed in the chlor-alkali process. Notably, TiO2, RuO2, and IrO2 are widely used as coatings on anodes owing to their excellent chemical resistance in a wide range of pH. Forming this oxide coating can suppress the photocorrosion and dissolution of the metal core. For thin film photoanodes, such metal oxides can be deposited as a thin overlayer to protect the photoactive semiconductor (Fig. 11b). The direct deposition of an overlayer can be easily performed using photodeposition or electrodeposition, which creates robust thin films. For instance, Xiao et al. constructed hollow, mesoporous TiO2 containing Pt/Cu2O that maintained high photoactivity in seawater for 30 h.106
 | ||
| Fig. 11 Illustration of (a) oxide shell protecting the metal core and (b) overlayer protecting the photocatalyst from photocorrosion and dissolution. | ||
Reactor design is the most critical aspect of large-scale practical applications. When designing reactors, considerations include the reactor geometry, amount of light passing through the solution and reaching the photocatalyst, mass transfer process, leaching of photocatalysts from films or electrodes, accumulation of sodium salt, and separation and collection of Cl2/HOCl. Domen's group constructed a scalable panel reactor with 100 m2 in size that consists of photocatalyst sheets (Fig. 12a).107,108 The photocatalyst sheets were prepared by applying a spray method on frosted glass sheets. A similar design for photocatalytic seawater can be adopted, where seawater flows through the film panel and the gases evolved can be collected at the top. Because H2 is the expected reduction product, there is a need for a dedicated separation of Cl2 and H2 gases. Fortunately, HOCl (resulting from the dissolution of Cl2 in water) can be collected in a liquid phase, thereby easing the separation of H2 and HOCl. Unlike pure water splitting, two further considerations must be taken into account, namely, the removal of accumulated sodium salt and oxygen flow input (where oxygen acts as an electron scavenger). The accumulated sodium salt on the film panel needs to be adsorbed, precipitated and filtered out for removal. Considering that the salinity of seawater is usually 3.5% (∼0.6 M) NaCl, which is relatively low compared to the one used in the industrial chlor-alkali process, precipitation of sodium salt is much less, and thus its removal is relatively easier. It should also be noted that the solution of aqueous NaCl is temperature dependent; therefore, the temperature of the seawater solution must be controlled to avoid salt precipitation during a photocatalytic reaction.
 | ||
| Fig. 12 (a) Scalable reactor design for a large-scale 100-m2 panel reactor with 33 × 33 cm2 films for photocatalytic water splitting. Reproduced from ref. 108. Copyright 2018, Cell Press. (b) Schematic diagram of large-scale panel experiments and (c) photograph of the 25 cm2 photoreactor for the photoreforming of plastics, biomass and mixed waste. Reproduced from ref. 109. Copyright 2020, Wiley-VCH. | ||
Another consideration is designing moderate-scale configurations for photocatalytic seawater splitting with a similar design to flow cells with a low-cost LED light source externally mounted to the cell. Uekert et al. designed a 25 cm2 CNx/Ni2P panel for a real-world demonstration under low sunlight and seawater conditions (Fig. 12c and d).109 The authors reported a loss of 50% efficiency in an upscaled (25 cm2, 30 mL solution volume), custom-built flow reactor under real-world conditions. With this design, it is also possible to stack the cells into a larger configuration to boost the production rate and yield of Cl2/HOCl. Considering that a tiny amount of Cl2/HOCl (4 mg L−1) is required for drinking water, this technology brings us a step closer toward real-world applications for on-site, on-demand production of Cl2/HOCl for disinfection in remote areas.
Upscaling photochemical (PEC) reactors remains challenging because of several problems associated with PEC cells. However, PEC cells already have built-in compartments to collect H2 and Cl2/HOCl separately. The identified challenges for large-scale PEC cells include high cell efficiency (>10% STH), long life cycles and cost-effective fabrication process. Another issue is the incorporation of external bias to apply potential to the system. To overcome this issue, a research direction can be aimed at developing unassisted stand-alone PEC devices where one compartment carries out Cl− oxidation using a photoanode and another carries out proton reduction using a photocathode. Large-sized photoelectrode fabrication presents another bottleneck that ensures homogeneous current density distribution. Dilger et al. successfully upscaled a PEC cell dimension from the typical 1 cm2 to 40 cm2 using LaTiO2N coated with co-catalysts.110 The authors reported that the reduced activity and thus photocurrent density (by 54%) were attributed to the variation in the spatial distribution of potential and local photocurrent density. Overall, while fabricating homogeneous large-size photoelectrodes, controlling the optimised thickness of photoactive semiconductors and loading of co-catalysts, optimising cell geometry (including the distance of the light path), and inducing fast mass transfer dynamics remain challenging for upscaled PEC cells, notably when the electrode is larger than 10 cm by length, as remarked by Hankin et al.111
Very recently, Rodríguez-Gutiérrez designed a 3D-printed reactor and assembled ten such reactors to form a 100 cm2 module for moderate upscaling with stable photocurrents for over 120 hours (Fig. 13).112 This modular PEC reactor design paves the way for scalable outdoor applications. To minimize ohmic losses when photoelectrodes are in contact with an electrolyte, and to obtain efficient product separation in individual compartments, Tsampas's group constructed a zero-gap PEC device in which an anion exchange membrane (AEM) was sandwiched directly between a photoanode and a cathode.113 The up-scaled device with an electrode area of 100 cm2 preserves ∼98% of the photocurrent density exhibited by the 1 cm2 photoanodes, demonstrating the practical feasibility and high performance of scalable PEC devices. More recently, a scalable PEC cell for H2 and H2O2 production with a 144 cm2 array panel has been reported.114 Precipitation of electrolyte (NaHCO3) on the photoanode films can be removed by rinsing with deionized water to recover the initial photocurrent. A similar design and post-treatment of photoanode films can be adopted for PEC chloride oxidation from seawater splitting; sodium salts can be removed by rinsing with water to recover the photoanode films.
 | ||
| Fig. 13 (a) Illustration of the design of a modular PEC cell, (b) photograph of a real reactor with hematite photoanodes, and (c) outdoor demonstration for upscaled solar hydrogen production. Reproduced from ref. 112. Copyright 2025, the American Chemical Society. | ||
8. Emerging photoactive semiconducting materials
Perovskite photocatalysts have garnered attention in water and seawater splitting. Metal halide perovskites (MHPs) adopt the crystal structure of ABX3, where A and B are cations and X is a halide (Cl, Br, and I), with a well-known example of CsPbBr3. The A site can also be occupied by organic cations, such as methylammonium and formamidinium in MAPbI3 and FAPbI3, respectively. MHPs are interesting and attractive because they exhibit high optical absorption coefficient, tunable optical properties via compositional engineering, defect-tolerant properties and large carrier diffusion lengths (∼100 nm–∼1 µm).115 Due to the toxicity of Pb, several other metal cations have been explored, such as CsSnI3, CsGeI3, Cs3Bi2I9 and CsNaBiCl6. For perovskite oxides, piezoelectric properties present an advantage in applying mechanical vibration to enhance photoactivity. Although the work on Cl2 /HOCl production from seawater splitting using MHPs has been insignificant, the potential for Cl2 /HOCl production is desirable because the inherent Cl− ions participate in the chlorine evolution reaction (CER) through the Mars–van Krevelen mechanism.Perovskite-type oxynitrides AB(N)3 (A = La, Ca, Sr, and Ba; B = Ti, Nb, and Ta) have recently emerged as promising photocatalysts due to their narrow band gap, which can harness visible light for photoredox processes (Fig. 14). SrNbO2N exhibits a narrow band gap (1.8–2.0 eV), a maximum current density of ∼20.5 mA cm−2 and suitable band positions for seawater splitting. Trinh and Seo reported that porous SrNbO2N/Nb photoanodes exhibited a photocurrent density of 2.6 mA cm−2 at 1.36 VRHE, and three-fold selectivity for CER than OER.116 The same group also reported that Co(OH)x-loaded BaTaO2N promoted CER and subsequently decomposed Cl2 back to O2 and Cl−, creating a self-sustaining steady-state Cl− concentration in the bulk electrolyte.117 This work paves the way towards sustainable H2 production at the cathode without replenishing the NaCl electrolyte.
 | ||
| Fig. 14 Band gap and redox potentials of several perovskite oxynitrides. Reproduced from ref. 118. Copyright 2024, the Royal Society of Chemistry. | ||
Perovskite oxides are another class of emerging photocatalytic materials with unique features, such as piezoelectric and ferroelectric properties. Moreover, perovskite oxides display superior stability to metal halide perovskites and perovskite-type oxynitride.119,120 La2NiO4 achieves high activity for H2 and Cl2 evolution in coupled piezo-photocatalytic seawater splitting without any sacrificial agent.121 Inducing mechanical vibration via ultrasonication improves the charge separation and transfer, which reduces the need for noble metal co-catalysts.122 A self-generated Na0.5Bi0.5TiO3/Na0.5Bi4.5Ti4O15 heterojunction also exhibits a decent photoactivity in seawater splitting albeit the HOCl concentration was not quantified.123
Porous coordination polymers, including metal–organic frameworks (MOFs) and covalent organic frameworks (COFs), are porous and crystalline materials with large surface areas, high porosity, diverse structures and topologies, and tunable electronic and optical properties through molecular engineering. These remarkable features render them promising in photocatalysis, electrocatalysis and photoelectrocatalysis. The catalytic activity and selectivity in MOFs are dictated by the unsaturated metal nodes and functional organic groups.124 Moreover, the existence of charge transfer (linker-to-linker, metal-to-ligand, and ligand-to-metal) provides photoactive characteristics to MOFs.125 Additionally, porous solid materials exhibiting different properties than the original MOFs can be prepared by pyrolysis of MOFs.
MOFs have been widely investigated for their promising applications in gas and liquid separation, membrane filtration and molecular sieving. The high-surface area and porosity offer a high capacity for ion adsorption, including chloride (Cl−). Although the direct use of MOFs for the production of Cl2/HOCl via seawater splitting is still limited and at an early stage of development, electrocatalytic and photocatalytic H2 and O2 evolution in seawater splitting have been demonstrated.126,127 Jothi et al. demonstrated that bimetallic CuV-MOF showed significant CER activity albeit the faradaic efficiency was not quantified.128 High durability and stability of several MOFs under harsh and corrosive conditions of seawater offer potential for the design of robust co-catalysts or electrocatalysts to produce Cl2/HOCl. For example, crystalline-amorphous ferrocene-based Ni-BDC-aNiOOH MOFs exhibit superior durability with ∼90% retention of current density for over 400 hours.129
Bao et al. recently reported the selectivity of Ni-BDC/NM88B(Fe) MOF-on-MOF heterojunction in seawater oxidation.130 Although the intermediate in CER generally has a lower Gibbs free energy and a lesser number of intermediates at both Fe and Ni sites, the lower adsorption free energy of OH− governs the selectivity, thereby favouring OER over CER. The formation of the NiFeOOH protective layer defends Ni-BDC/NM88B(Fe) against dissolution by Cl2, leading to an excellent stability of 200 hours. Ligand defect engineering has proven to be a viable strategy for improving durability in seawater splitting. The electronic structure of NH2-MIL-88B(Fe) has been modified by introducing ligand vacancies using acetic acid, resulting in the suppression of Cl− corrosion.131
Few studies have applied porous coordination networks to PC and PEC seawater splitting. The major focus in seawater splitting is the H2 evolution in the presence of a sacrificial agent, notably a hole scavenger.132 For example. Li et al. employed Au-loaded thioether-functionalised COF in PC seawater splitting and observed no formation of Cl2 because the presence of triethanolamine effectively scavenges photogenerated holes.133 Attempts have been made to test PC seawater splitting using a variety of COFs without hole scavengers; however, little or no photoactivity was observed.134 Crown ethers discontinuously conjugated polymers (DCPs) showed reduced photoactivity in a narrow range of NaCl concentrations (0–0.6 M) in synthetic seawater.135 The finding implies that the presence of Na+ and Cl− ions negatively impacts the photocatalytic performance. The crown ether moiety adsorbs ions, leaving only the proton to interact with the active sites. In contrast, 2,4,6-trihydroxy-1,3,5-benzenetricarbaldehyde (TPBD) COF shows enhanced photoactivity in seawater due to the strong polorisation effect induced by C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O groups, leading to better charge separation and transfer.136 Similarly, thieno[3,4-c]pyrrole-4,6-dione (TPD) COFs exhibit higher photoactivity in seawater owing to their increased hydrophilicity and enhanced charge carrier separation and transfer.137
O groups, leading to better charge separation and transfer.136 Similarly, thieno[3,4-c]pyrrole-4,6-dione (TPD) COFs exhibit higher photoactivity in seawater owing to their increased hydrophilicity and enhanced charge carrier separation and transfer.137
The limited studies in photocatalytic seawater splitting particularly chloride oxidation using COFs originates from the low charge mobility and rapid charge carrier recombination that often requires a hole scavenger which precludes oxidation of chloride.138 Additionally, certain organic functional groups in COFs readily adsorb ions, preventing Cl− from reaching the actual active sites for CER. Future work should be directed to investigating and controlling the hydrophilicity/hydrophobicity, improving charge mobility and charge transfer, and creating selective active sites for Cl− adsorption and oxidation.
9. PC and PEC applications of Cl2 and HOCl
The advanced oxidation process (AOP) based on reactive chlorine species (RCS) provides a feasible strategy for the removal of organic contaminants and pollutants. HOCl undergoes photolysis under UV light irradiation to form highly reactive radical species, like Cl˙, ˙OH, and ˙OCl, which are strong oxidants. In fact, as discussed below, RCS exerts a much more powerful and selective oxidising ability than reactive oxygen species (ROS) in some cases, leading to more efficient degradation performance. As a result, in situ generated oxidants from HOCl have been utilised to drive oxidative reactions, and organic pollutant degradation and removal. A well-established chlorination process has been employed for over 100 years in drinking water treatment, swimming pool disinfection and medical disinfection/sterilization, underscoring a profound and ubiquitous use of Cl2/HOCl in bacteria deactivation and pollutant degradation. It is therefore imperative to discuss the relevant applications of Cl2/HOCl in various chemical processes, considering the impacts of PC and PEC production of Cl2/HOCl.(a) Bacteria deactivation and inhibition
Chlorination is a well-established and effective means of disinfecting drinking water, swimming pools, and wastewater treatment. Bacterial deactivation is utterly important for marine antifouling, wastewater treatment and drinking water disinfection. Photocatalytic antibacterial properties are usually tested using Gram-negative bacteria, such as Escherichia coli (E. coli) and Pseudoalteromonas, and Gram-positive bacteria, such as Staphylococcus aureus (S. aureus) and Bacillus sp. E. coli is a common Gram-negative bacterium found in contaminated water sources (lakes, rivers, and reservoirs) and food that can be pathogenic and cause poisoning, stomach pains and cramps, watery diarrhea, vomiting and fever. Thus, E. coli is commonly used as a model bacterium for bacterial deactivation.Koo et al. showed the complete deactivation of E. coli in four hours over WO3 photoanodes due to the in situ generation of HOCl as an active species.30 In another work by Juodkazyte et al., the complete deactivation of E. coli and Bacillus sp. was demonstrated using a WO3 photoanode and was ascribed to chlorine radicals (Cl˙ and Cl2˙−).81 It is worth noting that ˙OH radicals are less effective in bacteria deactivation compared to RCS, highlighting the superiority of the latter. For example, Sun et al. disclosed that IrO2/ZnWO4 displayed a higher sterilisation efficiency against E. coli. owing to a higher production yield of RCS (HOCl, ClO−) than ZnWO4, which generated mainly ˙OH.51 Moreover, the quenching experiment verified that scavenging of ˙OH led to a slight decrease in sterilisation efficiency, suggesting a minor role of ˙OH compared to RCS. More recently, carbon-doped BiOCl (C-BiOCl) displays impressive E. coli deactivation in 40 minutes (Fig. 15).71
 | ||
| Fig. 15 Inactivation of E. coli over C-BiOCl with two different RCS concentrations. Reproduced from ref. 71. Copyright 2024, Elsevier. | ||
A quaternary Pt/Fe3O4@SiO2@WO3 photocatalyst shows good antibacterial effects on a broad spectrum of bacteria, including E. coli, Pseudoalteromonas, S. aureus, and Bacillus, with antibacterial rates of 90%, 83%, 86%, and 88%, respectively, in three hours.49 When the same photocatalyst was applied for algae inhibition on Dicrateria inornata and Chlorella under visible light irradiation for 12 hours, the inhibition rates reached 93% and 95%, respectively. Lu et al. reported an impressive bacterial deactivation (>97%) in two hours and algae inactivation (>90%) in 8 hours using Pt/WO3 hollow microspheres owing to high in situ generated free chlorine (1.24 mg L−1).46 More recently, a Z-scheme WO3/CdS photocatalyst exhibits higher deactivation efficiencies (>94%) on E. coli, Pseudoalteromonas, S. aureus, and Bacillus in three hours.53 The inhibition rate of Dicrateria inornata and Chlorella achieved >90% in 12 hours.
In conclusion, these works demonstrate the practical feasibility of using in situ generated RCS for bacteria deactivation and algae inhibition. A potential future direction is to design portable photoreactors for PC or PEC systems that are suitable for use in remote areas for on-site sanitization and water treatments for small water sources or storage facilities, like wells, ponds and tanks. A small amount of RCS from PC or PEC seawater splitting is beneficial for sanitizing surfaces in the kitchen and lounge in rural houses.
(b) Ammonia removal
Ammonia is a key fertilizer for plant growth and nutrition in agriculture because it supplies nitrogen to improve crop quality. Although it is crucial for agriculture, excess ammonia in soil has negative impacts on human health and the environment, behaving as a pollutant in the atmosphere that causes acid rains, depositing in the soil and being dissolved in water (lakes, rivers), and changing species composition through soil acidification and direct toxic damage. Indirect discharge into wastewater containing ammonia nitrogen (NH4+–N) remains a challenging issue in agriculture and thus becomes a problem of environmental concern. Chlorine disinfectants have been used to remove NH4+–N by converting it to N2. Utilizing photocatalytic Cl2/HOCl production may eliminate the need for external chlorine sources. For example, Xiao et al. showed that RCS (Cl2/HOCl) plays a critical role in NH4+–N degradation on TiO2 nanotube photoanodes, where the degradation rates increase with NaCl concentration across a wide pH range.139 Zhu's group demonstrated the practical feasibility of NH4+–N removal using landfill leachate and real wastewater as examples.36,71The overall reaction for NH4+–N degradation proceeds according to eqn (30). The mechanism of NH4+–N degradation over a BiVO4/WO3 photoanode has been proposed to proceed via chloramine intermediates (NH2Cl and NHCl2).140 COR produces Cl˙, which abstracts H from NH4+ to form NH2˙; this radical reacts with HOCl to yield NH2Cl (eqn (31)–(33)). A further reaction leads to an NHCl2 and eventually N2 evolution as described below (eqn (34)–(36)). NH4+–N can also be oxidized to NO2−–N and NO3−–N species (eqn (37) and (38)), thereby presenting competitive steps for selective N2 formation in NH4+–N degradation.141 The formation of NO3− is proposed to include reactive oxygen species (ROS), such as ˙OH, O2˙−, and H2O2.142 Therefore, it is expected that COR in seawater solution would favour the selective formation of N2 in the degradation of NH4+–N.
| 2NH4+ + 3HOCl → N2 + 3HCl + 3H2O | (30) |
| Cl− + h+ → Cl˙ | (31) |
| NH4+ + Cl˙ → NH2˙ + 2H+ + Cl− | (32) |
| NH2˙ + HOCl → NH2Cl + OH˙ | (33) |
| NH2Cl + Cl˙ → NHCl˙ + H+ + Cl− | (34) |
| NHCl˙ + HOCl → NHCl2 + OH˙ | (35) |
| NHCl2 + NH2Cl → N2 + 3H+ + 3Cl− | (36) |
| NH2˙ + OH˙ → NH2OH | (37) |
| NH2OH + OH˙ → NO2−→NO3− | (38) |
| 2NH4+ + 2ClO˙ → N2 + 4H+ + 2Cl− + 2H2O | (39) |
An early work by Ji et al. using a WO3 photoanode disclosed that the complete degradation of NH4+–N to a major N2 and minor NO3− occurred in 90 min and followed the pseudo first-order kinetics.143 However, the degradation of NH4+–N over BiOCl was shown to follow the zeroth-order kinetics with almost complete degradation and 97% selectivity toward N2 over BiOCl in 80 minutes.69 Zhu's group reported that BiOCl with dominant {001} facets exhibited the degradation efficiency of 91.9% with stoichiometric Cl− (in NH4Cl) ions and near unity with extra Cl− (from NaCl) in the reaction solution within 150 minutes.36 Without Cl− in the reaction solution, only 25.9% degradation was observed, highlighting the crucial role of RCS (Cl2, HOCl, and Cl˙) for promoting NH4+–N degradation. The higher degradation efficiency with increasing Cl− content is attributed to the higher amount of RCS formed. Using a landfill leachate from a landfill in Zhongshan City as a real wastewater for treatment, the authors showed a degradation efficiency of 85.4% in 30 hours and 95.6% in 40 hours of treatment over BiOCl and C-BiOCl, respectively.36,71 Nonetheless, Zhu and co-workers realised that the degradation efficiency under the Xe lamp is far inferior to that of the UV lamp.71 The cause might be attributed to the less efficient photolysis of HOCl that creates radicals under the Xe lamp compared to that of the UV lamp.
Yan et al. disclosed that OCl˙ radicals are responsible for achieving the complete degradation of NH4+-N in 120 min and boosting 100% selectivity towards N2 in acidic NaCl solution (pH ≤ 6), as shown in Fig. 16a and b.144 By scavenging and ESR trapping experiments, the authors disclosed that OCl˙ radicals resulted from photolysis of HOCl under UV irradiation; the mechanism NH4+–N degradation is illustrated in Fig. 16c. Importantly, other works have also attributed the active species for NH4+–N degradation to ClO˙ radicals.141,145 It is worth noting that ˙OH radicals are relatively inert to NH4+–N and thus are less effective in the degradation process. Similarly, the imperative role of RCS (Cl2, HOCl, Cl˙, and ClO˙) in promoting the degradation of NH4+-N was reinforced by Zhang et al. using a CoBi/BiVO4/WO3 photoanode, whereas ˙OH radicals play a negligible role.96 Notably, it has been revealed that ˙OH reacts with HOCl to form ˙OCl (and H2O), showing an indirect participation of ˙OH to form OCl˙ active species.104 Although OCl˙ radicals are regarded as the primary active species in NH4+–N degradation,146 HOCl plays a role in completing the cyclic chlorine species between Cl− and Cl˙ (or Cl2), indicating complex reaction pathways (refer to eqn (29)–(37), and Scheme 1).
 | ||
| Fig. 16 (a) Time-course degradation of NH4+–N, (b) conversion of NH4+–N to different oxidised species (N2, NO2−–N, and NO3−–N), and (c) a proposed mechanism of NH4+–N degradation in acidic water over BiOCl. Reproduced from ref. 144. Copyright 2021, Elsevier. | ||
 | ||
| Scheme 1 NH4+–N degradation pathways to N2 through chloramine intermediates and radicals. Reproduced from ref. 96. Copyright 2023, Elsevier. | ||
In summary, these reports show that RCS resulting from COR plays a predominant and significant role in degrading NH4+–N through chloramine intermediates and radicals, which are much more efficient than reactive oxygen species (ROS). Despite a lower oxidation potential of Cl˙ (2.47 V vs. NHE) than ˙OH (2.80 V vs. NHE), Cl˙ is more selective and reacts faster with electron-rich moieties, such as NH4+.143,147 The high selectivity towards N2 formation renders PC and PEC degradation of NH4+–N using in situ generated RCS appealing, green and sustainable.
(c) Conversion of alkenes and alcohols
Due to the strong oxidising ability of HOCl in bacterial deactivation and pollutant degradation in wastewater, several works have extended its utility in the oxidation of alkenes and alcohols using in situ generated HOCl. HOCl behaves as a redox mediator and strong oxidant to oxidize organic compounds with a lower applied potential than the direct oxidation of such organic compounds by adding molecular O2, H2O2 or t-BuOOH. For example, Yang et al. reported almost complete conversion and selectivity in the epoxidation of water-soluble alkenes on a Co2(OH)3Cl/BiVO4 photoanode (Fig. 17a and b).13 The mechanism of such epoxidation involves the formation of a secondary alcohol and terminal alkyl chloride as intermediates resulting from the reaction between in situ generated HOCl and alkenes (Fig. 17c). Such high photoactivity in epoxidation can potentially replace the use of meta-chloroperbenzoic acid (m-CPBA), which possesses a high risk of explosion and limits its large-scale utilization in alkene epoxidation.148 | ||
| Fig. 17 (a) Alkenes used in the epoxidation reaction, (b) conversion and selectivity in epoxidation over CoOCl/BVO, and (c) proposed mechanism of epoxidation via in situ generated HOCl. Reproduced from ref. 34. Copyright 2024, Wiley-VCH. (d) Proposed mechanism of the selective oxidation of methane over BiOCl-OV. Reproduced from ref. 149. Copyright 2023, Elsevier. | ||
Oxidation of ethanol was demonstrated by in situ PEC HOCl generation on HxWO3, without the addition of external oxidants. Ethanol oxidation to acetaldehyde and acetic acid was mediated by HOCl serving as a strong oxidant – most likely through ˙OH and Cl˙ from the photolysis of HOCl – rather than direct oxidation on the HxWO3 electrode as confirmed by linear sweep voltammetry (LSV).149 The EtOH conversion achieves 62% in 8 hours with 50% and 40% selectivity to acetaldehyde and acetic acid, respectively. The proposed reaction pathways are described by eqn (40)–(42). Such oxidation can be extended to secondary alcohols, like 2-propanol, with 92% selectivity to acetone.
| Cl− + H2O → HOCl + H+ + 2e− | (40) |
| CH3CH2OH + HOCl → CH3CHO + H+ + Cl− + H2O | (41) |
 | (42) |
The selective photocatalytic oxidation of methane to methanol is highly desirable to simultaneously mitigate greenhouse gases and to produce fuels, but it is very challenging due to a large energy barrier of C–H activation and bond energy (439 kJ mol−1).150 Fortunately, active chlorine species can be used to abstract H from CH4, which can then be oxidised to CH3OH with 80% selectivity in the presence of H2O2. H2O2 undergoes decomposition and supplies ˙OH and O2˙− radicals. Cl− oxidation to a ˙Cl radical offers an advantage in carrying out CH4 oxidation in a seawater solution. Wang et al. showed that both Cl˙ and ˙OH are crucial for the conversion of CH4 to CH3OH over BiOCl with oxygen vacancies (BiOCl–OV), where oxygen vacancies (Vo) serve as an active site for Cl− adsorption (Fig. 17d).149 Cl˙ is required to form a ˙CH3 intermediate, which is oxidised by ˙OH to yield CH3OH. Interestingly, pristine BiOCl (without oxygen vacancies) produces Cl2, which is responsible for halogenated compounds (CH2Cl2 and CHCl3), while BiOCl-Vo yields Cl˙, which is responsible for H abstraction from CH4. In another work, the same group also reported the formation of diethyl ether (C4H10O) as the majority and other chlorinated products such as CH2Cl2, CHCl3 and C2H4Cl2O in synthetic seawater.56
Overall, in situ generated RCS are key for the oxidation and conversion of organic compounds. Importantly, RCS shows high selectivity towards particular products, which renders COR advantageous and promising for selective oxidation.
(d) Oxidation and chlorination of amines and sulfides
A strong oxidising capability of reactive chlorine species (Cl2 and HOCl) renders it suitable for various organic transformations, including the oxidation of amines and sulfides. Notably, HOCl can initiate oxidative fragmentation without incorporating Cl atoms into the final products. For example, Hull et al. reported the mechanism of triethylenediamine oxidation by HOCl to produce piperazine, which can be further oxidised to ethylenediamine and formaldehyde.151 The first step usually involves hydrogen abstraction and the inclusion of a chlorine atom in amines. Aldehydes seem to be a common product of amine oxidation by HOCl, as also observed in the reaction of aminoalcohols with HOCl. Interestingly, kinetic studies have shown that tertiary amines react much faster with HOCl than primary and secondary amines.152Reactions between organic amines and RCS often yield the corresponding chlorinated amine (N–Cl) compounds, which are significant in biochemistry and environmental chemistry. Organic amines, like amino acids, are often present in wastewater and sewage sludge, which are treated with HOCl. Reactions between amino acids and peptides with HOCl proceed readily. The importance of the reaction between RCS and amino acids in enzymes in bacteria is paramount to understanding bacterial deactivation and inactivation in wastewater treatment. A free amino group in amino acids reacts with HOCl to yield unstable chloroamine intermediates (RNHCl/RNH2+Cl), which subsequently decompose to NH3, CO2, nitriles, and aldehydes.153–156 Notably, N-chloro derivatives form rapidly from the reaction of amino acids and HOCl and decompose slowly in multisteps (Scheme 2). Although the products of amine oxidation by HOCl are well known and well characterised, the mechanism remains elusive and less understood. Hawkins and Davies verified the formation of radical intermediates from the decomposition of chloroamines and chloroamides by electron paramagnetic resonance (EPR).157 However, several dipeptides were found to be resistant to HOCl at room temperature.
 | ||
| Scheme 2 Mechanistic pathways of the oxidation of amino acids by HOCl with the liberation of CO2 and formation of aldehyde and nitrile. Reproduced from ref. 158. Copyright 2017, the American Chemical Society. | ||
N-chlorination reactions render it suitable for the application of in situ generated RCS by photocatalytic and photoelectrochemical means. Ding et al. demonstrated enantioselective chlorofunctionalization of N-substituted amides using in situ generated HOCl.159 The role of HOCl is identified as a proton donor and a hypochlorite source. It is therefore expected that in situ generated RCS from seawater splitting facilitates organic transformations of amine compounds.
Hydrogen sulfide (H2S) is a weak acid and toxic gas, which is produced as an emission gas from chemical, oil and gas industries. Chemical scrubbing is a typical method for removing H2S emissions using a gas–liquid contactor containing an oxidant, such as HOCl or H2O2.160 The oxidation of H2S is a fast reaction, follows a complex mechanism, and is strongly pH dependent. The nucleophilic characteristics of HS− and S2− (sulfide) make it reactive to electrophilic HOCl. Sulfide in H2S is oxidised by HOCl to yield elemental sulfur (S) and chloride (Cl−).161 Reacting emitted H2S with in situ generated HOCl may pave the way toward efficient H2S removal with low capital and operating costs. Moreover, the reactivity between H2S and HOCl helps us to understand the role of HOCl in neurodegenerative diseases associated with the H2S concentration in the brain.162
The oxidation products of organic sulfur compounds depend on the type of sulfur. The reaction rate for a few sulfur compounds was established in the following order: thiol > thioether > sulfoxide. Sulfides are oxidised to sulfoxides, and overoxidation in excess of chlorine yields sulfones, while thiols form disulfides and gradually transform to sulfonic acids.163 Meanwhile, thioethers are oxidised to sulfoxides.164 Sulfur-containing compounds, such as thiourea, thiosulfate and thioglycolate, are reactive towards oxidation by HOCl. For instance, Stanbury's group reported that the reaction rate of these sulfur compounds with HOCl is much more rapid than using H2O2 and [Pt(CN)4Cl2]2− oxidants, highlighting the former's superior oxidative capability owing to the highly electrophilic HOCl.165 Moreover, the solubility of HOCl in polar organic solvents, such as CH2Cl2, renders it advantageous in various organic reactions because no phase transfer catalysts are required for the oxidation process.
(e) Photocatalytic CO2 reduction
Photocatalytic CO2 reduction (PCR) represents a sustainable carbon cycling system and a clean route to mitigate the CO2 level in the atmosphere and produce high value-added chemicals and fuels, like CO, CH4, and CH3OH. However, research in PCR generally employs water as an electron donor; thus, the non-trivial role of other photooxidation half-reactions is often overlooked. It has been shown that Cl− oxidation enhances activity and selectivity toward specific products in PCR. Coupling CO2 reduction to CO and Cl− oxidation to Cl2 (or HOCl) is kinetically favoured because both half-reactions involve a stoichiometric amount of charge carriers, i.e. two electrons and two holes. Layered-double hydroxides (LDHs) with remarkable adsorption capacities, diverse compositions and ions, and tunable optical and photoelectrochemical properties are widely used for CO2 capture and gaining attention for photocatalytic CO2 reduction.166 Ni–Al LDHs exhibit a selective formation of CO (86%) from photoreduction of CO2 with almost twice the CO2 yield in aqueous NaCl (0.1 M) compared to pure water; the remaining 14% is H2 evolution, and no other products, such as CH4, CH3OH and HCOOH, are detected in the reaction.19 However, the mechanism and the underlying reason the addition of Cl− ions favours higher activity and selectivity are not addressed in the work, except that Cl− acts as a hole scavenger to form HOCl and thus remains elusive.Recently, surface Lewis basicity has been introduced by doping the {001} facets in BiOCl with nitrogen (N-BiOCl) to enhance CO2 adsorption and activation for PCR in natural seawater (The Yellow Sea, China, was used as an example).72 The near stochiometric production rates of HOCl and CO under visible light are 83.2 and 92.8 µmol g−1 h−1, respectively, with product selectivity above 90% for both HOCl and CO (Fig. 18a and b). The reaction pathway attributes CO2 reduction on the {001} facets, where photoelectrons are accumulated, and HOCl formation to be most likely on the lateral {110}/{010} facets via self-oxidation of lattice Cl− ions. In another work, Wang et al. observed that the CO32− intermediate in PCR may participate in the BiOCl interlayers, causing distortion and exchange of Cl− ions in BiOCl as evidenced by XRD patterns, Raman and XPS measurements (Fig. 18c–f).73 However, no reduction products (e.g. CO, CH4, and CH3OH) were quantified in the study.
 | ||
| Fig. 18 Photocatalytic formation of (a) CO and (b) HOCl over N-BiOCl. Reproduced from ref. 72. Copyright 2023, Wiley-VCH. Characterizations of the BiOCl photocatalysts before and after reactions: (c) XRD patterns, (d) Raman spectra, (e) C 1s XP spectra, and (f) Bi 4f XP spectra. Reproduced from ref. 73. Copyright 2023, Wiley-VCH. | ||
Notably, the findings from photocatalytic COR improving the photoactivity and selectivity in PCR have led researchers to design novel Cl-modified photocatalysts through surface modification, grafting or doping with Cl− ions.167–169 Although these works are not related to PC or PEC seawater splitting or Cl2/HOCl formation, they highlight how the progress in seawater splitting has impacted other photocatalytic reactions and photocatalyst designs.
10. Challenges and future direction
The competing reactions between oxygen evolution reaction (OER) and chlorine evolution reaction (CER) determine the selectivity in photo(electro)catalytic seawater splitting. A considerable number of factors govern the selectivity of CER, including the Cl− concentration, pH of the electrolyte, electrocatalyst, and applied potential. In acidic and near neutral media, CER dominates over OER, while OER is preferred at high pH. Thus, acidification is often performed to enhance CER selectivity. In addition, the free energy difference in the –OCl and –OOH intermediates is used as the selectivity descriptor to predict the selectivity of CER and OER. For example, the stabilisation energy of OClad is used as an activity descriptor to evaluate the effectiveness of CER electrocatalysts and is further employed to explain the superior activity of RuO2 than IrO2.170 DFT calculations by Hansen et al. disclosed that Cl− is adsorbed on the O-covered rutile surfaces spontaneously forming an –OCl intermediate.43 Moreover, CER only involves a single intermediate compared to OER that proceeds via three intermediates, giving an advantage to the former reaction. Similarly, ab initio density functional theory (DFT) was performed using different RuO2 crystal facets to investigate the overpotential of CER and OER.171 RuO2(101) was found to promote CER, and RuO2(001) preferentially catalysed OER.Zeradjanin et al. proposed a dynamic catalytic descriptor based on vibrational frequencies of the crystal lattice from in situ Raman spectroscopy and electrochemical methods.172 The Raman shifts of metal–oxide bonds form a volcano plot, where the most active electrocatalysts with low overpotential for CER are RuO2 and IrO2, located close to the Cl–O bond (725 cm−1) found in HOCl (Fig. 19a). The volcano plot rationalises the established knowledge that RuO2 and IrO2 are the best performing electrocatalysts in dimensionally stable anodes (DSAs) for the chlor-alkali process. The authors also concluded that CER is a resonance phenomenon in which the catalyst surface, intermediates and products exhibit vibrational bonds with a similar frequency. It is therefore possible to predict and screen the best performing catalysts based on Raman spectroscopy and electrochemical studies where the vibrational modes of the metal–oxide (M–O) bond are close to those of the Cl–O bond. Notably, the Raman frequency and the length of the M–O bond are inversely proportional, which uses X-ray absorption spectroscopy (EXAFS/XANES) in determining the bond lengths useful for predicting suitable co-catalysts for CER.173
 | ||
| Fig. 19 (a) Overpotential of CER as a function of Raman shift for several metal oxides and (b) resistivity of several metal oxides. Reproduced from ref. 40. Copyright 2023, Wiley-VCH. | ||
Another key parameter for efficient and robust electrocatalysts for CER is high conductivity to survive anodic conditions. The two best-performing and most employed electrocatalysts in the chlor-alkali process, RuO2 and IrO2, are metallic oxides (Fig. 19b). Rutile RuO2 has partially filled 4d orbitals and therefore exhibits metallic conductivity.174 For IrO2, a large 5d orbitals overlap which is comparable to the Ir–O bond distance renders it metallic oxide.175 Furthermore, low CER overpotential was observed for perovskite oxides with metallic conductivity, such as La0.5Sr0.5CoO3 and LaNiO3. It should be noted that mixed metal oxides (MMO) usually contain a TiO2 semiconductor to improve the stability of DSAs while contributing little to their activity and selectivity. Metallic conductivity serves as a useful guide for exploring and designing suitable co-catalysts for photoanodes in CER.
A reasonable direction for exploring novel photocatalysts and photoanodes would involve the utilisation of in situ spectroscopy and electrochemical measurements to probe the vibrational frequencies and correlate them with the corresponding CER overpotential. This is possible for coordination polymers, such as metal–organic frameworks (MOFs) and covalent organic frameworks (COFs). The presence of metal nodes in MOFs and organic functional groups in COFs opens a new avenue for establishing similar overpotential-based volcano plots. Moreover, in situ Raman and infrared spectroscopy have proved advantageous in identifying intermediates and their binding with specific metal nodes or functional groups. Currently, the lack of such data restricts the prediction and knowledge-based design of effective novel photocatalysts for CER.
Computational studies provide a means of describing phenomena on the surface of photocatalysts at the molecular level, which can complement and help understand experimental studies. DFT with thermodynamic considerations was utilised to establish a correct selectivity descriptor for CER. Exner unravelled that the formation free-energy of oxygen with respect to hydroxide, ΔGO–OH, provides a correct description of the activity (or selectivity) trend for CER than the free-energy of binding energy of oxygen (ΔGO), which is typically used in the oxidation activity of metal oxides.176 Importantly, the ΔGO–OH descriptor has reproduced the activity trends of CER and OER for RuO2(110) and IrO2(110) accurately. Because OER and CER compete in an anodic process, both reactions must be considered to reproduce activity trends. The ΔGO-OH descriptor is capable of describing both OER and CER activity trends correctly, whereas the ΔGO cannot reproduce OER activity correctly. A prime advantage of using the ΔGO–OH descriptor is that the values can be computed by ab initio calculations and verified by cyclic voltammetry.177
Besides a direct Cl− adsorption to an oxygen atom in rutile metal oxides, Cl− can also bind to two oxygen atoms, forming ClO2− in CER.43 Exner included this possibility of ClO2− in his unifying material-screening framework, which includes overpotential, kinetics and electrochemical-step symmetry index, to comprehend the selectivity issues in CER.178 Based on this thermodynamic consideration, the formation of –Cl intermediate (i.e. OcCl, where Oc is a coordinatively unsaturated oxygen atom in metal oxides) should be promoted over –OCl adsorbate (i.e. Oc2Cl) to enhance the Cl2/HOCl selective formation. The implication of this finding is to search for and design novel photocatalysts and photoelectrodes that can stabilise a Cl− intermediate for enhanced Cl2 selectivity.
In many scenarios, the active sites that bind to the Cl* intermediate also bind to the OH* intermediate, i.e. they both share a common active. Hence, the scaling relationship correlates with CER and OER in the chlor-alkali process. To break this scaling relationship, active sites with larger adsorption energy for OH* must be created to promote selectivity towards CER.179 Doping RuO2 that results in an increase in the free energy of the OH* formation leads to an improved selectivity toward CER. For example, DFT calculations reveal that replacing the outermost Ru atoms with Ti atoms through the growth of a TiO2 monolayer on RuO2 enhances the CER selectivity by several orders of magnitude.180 Experimental work has also confirmed that doping is an effective strategy for enhancing CER selectivity.39,181,182
Although most computational studies on CER are directed at rutile-based metal oxides, there is limited work on emerging materials, such as covalent-organic frameworks (COFs) and metal–organic frameworks (MOFs). Future direction should be dedicated to computing the free-energy of binding energy of Cl− with N, S, and O atoms in organic functional groups and establishing the corresponding overpotential-based volcano plots because the findings will provide a knowledge-based guideline for rational design of efficient COFs and MOFs for CER. Furthermore, the findings from computational work can be validated by experimental techniques, such as infrared, Raman and nuclear magnetic resonance spectroscopy.
Photocatalytic Cl2/HOCl production from seawater splitting is still at an early stage of development. In what historically started as a hole scavenger in photocatalytic water splitting using seawater or aqueous seawater solution, the oxidation of Cl− to Cl2/HOCl has emerged as a new direction in photocatalysis research for the past decade. Early works attempted to enhance the selectivity of oxidation half-reaction towards water oxidation, neglecting the potential of Cl− oxidation to RCS (Cl2/HOCl). The challenging issues in overall water splitting, low economic value of molecular O2 and scarcity of pure/fresh water have urged researchers to assess other oxidation products that are kinetically favoured and have a high value-added.6 More importantly, PC and PEC seawater splitting have the potential to replace the energy-intensive conventional, industrial chlor-alkaline process in Cl2/HOCl production.
PC and PEC Cl2/HOCl production coupled with H2 evolution at the anode is a novel strategy for simultaneously generating clean energy and commodity chemicals from widely abundant seawater or saltwater. In natural seawater, dissolved CO2 could be an advantage in consuming photoexcited electrons, thereby suppressing the recombination of charge carriers and avoiding the continuous supply of O2 as an electron scavenger. However, dissolved CO2 forms carbonic acid (H2CO3) and HCO3−, in which the carbonate anion (CO32−) is a good hole scavenger, as reported by Hiroaki Tada.183 Therefore, there is a competing Cl− and CO32− oxidation in PC or PEC systems. Further work should explore PC or PEC Cl2/HOCl oxidation in natural seawater/saltwater to investigate the effects of other ions and factors affecting the selectivity in Cl− oxidation. Importantly, such work using natural and/or synthetic seawater will bring us closer toward a practical application.
Given that HOCl easily decomposes under exposure to UV light, the use of wide band gap photocatalysts should be minimized to achieve a high production rate and yield of HOCl. Nonetheless, good photocatalysts for COR are still dominated by wide band gap photocatalysts, like TiO2 and BiOCl (band gap > 3.0 eV), which render visible-light-driven photocatalysts less explored. Many earth-abundant, visible light photocatalysts have appropriate valence bands (>1.40 V vs. RHE), such as g-C3N4, CdS, TaON, MoS2 and Fe2O3, or other polymeric photocatalysts that remain relatively unexplored for COR and thus open a window of opportunity for catalyst development. Furthermore, photocatalysts for Cl− oxidation should consist of few (precious) metals because the acidic medium, resulting from the formation of HOCl, tends to dissolve metals under photoirradiation. Sayama's group observed a dissolution of Pt co-catalyst, forming soluble PtCl62− through a reaction with evolved Cl2, during photoirradiation in seawater splitting.58 The same group also reported a dissolution of Mn co-catalysts under photocatalytic conditions.
Except for limited photocatalytic materials, such as BiOCl and BiVO4, the facet-dependent reactivity and selectivity of Cl/HOCl formation remain underexplored. Different crystal facets have different surface energetics and accumulate different charge carriers caused by the internal electric field (IEF).184 However, the selectivity of oxidation-favoured facets is complicated by competitive water and chloride oxidation reactions. Theoretical simulations of specific facets can reveal the relative activation energy or overpotential for Cl− oxidation. Moreover, the intermediates in COR have not yet been fully clarified, thus presenting an opportunity for further theoretical work.
Currently, despite numerous investigations on PC and PEC of Cl2/HOCl formation using various photocatalysts and co-catalysts, the knowledge about the active sites remains elusive. Except for a few photocatalytic materials, like BiOCl and AgCl as well as Co2(OH)3Cl co-catalyst, where participation of the lattice Cl− in COR via the Mars–van Krevelen mechanism is verified, the active sites in many other catalytic materials are unknown. Moreover, the postulate of the existence of Co–Cl interaction when using a CoOx co-catalyst and/or overlayer has not been successfully verified by X-ray photoelectron spectroscopy.95 CoOx is a well-known electrocatalyst in oxygen evolution reaction (OER);185–187 therefore, understanding the nature of the catalytically active sites and interaction with Cl− is of paramount importance to tune the selectivity between OER and COR. Additionally, it has been reported that Cl− can stabilise oxygen vacancies in TiO2 and BiOCl, thereby promoting the formation of RCS or Cl˙ radicals for photocatalytic oxidation and degradation.149,188 It has also been suggested that Cl− interacts favourably with the TiO2 surface although this interaction has not been verified by physical or electrochemical characterizations.139 Future work should disclose the interaction and/or adsorption of Cl− with the surface of photocatalysts. Currently, such limitations impede the knowledge-driven design and optimization of selective, active, and stable photocatalysts for COR.
We highlight that PC or PEC seawater splitting represents another type of artificial solar photosynthesis where H2 evolution at the cathode can be coupled with Cl2/HOCl formation at the photoanode.33,44,64 More importantly, the separation of the evolution of H2 and Cl2 in PEC systems enables a direct collection of both gases for further utilisation or storage (Fig. 20). Such a strategy potentially circumvents the low-economic value of O2 and sluggish kinetics of O2 evolution in water-splitting. Therefore, PC and PEC seawater splitting present a feasible green alternative and economical method for renewable energy and CO2 emission mitigation compared to the traditional chlor-alkaline process.
 | ||
| Fig. 20 Schematic of the simultaneous Cl2 and H2 evolution at a TiO2 photoanode and a Pt cathode, respectively. Reproduced from ref. 44. Copyright 2023, Elsevier. | ||
Finally, we offer a perspective on coupled redox reactions involving a COR half-reaction. The other half-reduction reactions are not limited to H2 evolution or CO2 reduction, as discussed above. Very recently, Chen and Zhang constructed In2S3/MnIn2S4-based photoanode and photocathode for simultaneous production of HOCl and H2O2, respectively, which highlighted a promising approach for the production of fine chemicals via PEC.189 N2 reduction to NH3 has been another intense area of photocatalysis research for producing energy carriers. Shiraishi et al. demonstrated photocatalytic N2 fixation to produce NH3 in a seawater solution over oxygen-deficient BiOCl–Ov with solar-to chemical conversion of 0.05% HOCl.65 Moreover, halogenation of alkanes into organic halides – important industrial and medical chemicals – has been reported using a Vo-rich TiO2 photoanode.188 These findings open an avenue for broad, significant impacts of Cl2/HOCl production, photodegradation of pollutants, production of high value-added chemicals, and artificial photosynthesis involving the production of clean energy. Progress in COR via seawater splitting is still in its infancy and offers much room for development in the field.
More importantly, for remote areas that lack water treatment facilities, PC and PEC production of Cl2/HOCl using natural seawater/saltwater is a sustainable method for on-site and rapid production of Cl2/HOCl for treating drinking water and sanitizing purposes. It is undoubtedly that PC and PEC seawater splitting to produce Cl2/HOCl addresses at least four SDGs’ goals: Good Health and Well-being (#3), Clean Water and Sanitization (#6), Affordable and Clean Energy (#7), and Climate Action (#13).
Conflicts of interest
There are no conflicts to declare.Abbreviation
| AQY | Apparent quantum yield |
| COR | Chloride (Cl−) oxidation reaction |
| DFT | Density-functional theory |
| DPD | N,N-diethyl-p-phenylenediamine |
| DSA | Dimensionally stable anode |
| EIS | Electrochemical impedance spectroscopy |
| ETL | Electron transport layer |
| FE | Faradaic efficiency |
| HTL | Hole transport layer |
| IEF | Internal electric field |
| IET | Interfacial electron transfer |
| IPCE | Incident photon conversion efficiency |
| LDH | Layered double hydroxide |
| LSPR | Localised surface plasmon resonance |
| LSV | Linear sweep voltammetry |
| MSI | Metal-support interaction |
| OER | Oxygen evolution reaction |
| PL | Photoluminescence |
| P25 | TiO2 with anatase (70%–80%) and rutile (20%–30%) phases |
| PCR | Photocatalytic CO2 reduction |
| PWS | Photocatalytic water splitting |
| RCS | Reactive chlorine species |
| ROS | Reactive oxygen species |
| SCC | Solar-to-chemical conversion |
| STH | Solar-to-hydrogen |
| Vo | Oxygen vacancies |
Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
RHA thanks Greg Metha (Adelaide University) and Gunther Andersson (Flinders University) for the fruitful discussions on Sayama's paper (https://doi.org/10.1021/acsenergylett.8b00318), which motivated this work on photocatalytic production of high value-added chemicals beyond water splitting. This project was financially supported by KAUST (BAS/1/1433-01-01 and FCC/1/5937-19-01).References
- A. Fujishima and K. Honda, Nature, 1972, 238, 37–38 CrossRef CAS PubMed.
- R. Marschall, Eur. J. Inorg. Chem., 2021, 2435–2441 CrossRef CAS.
- C. R. Lhermitte and K. Sivula, ACS Catal., 2019, 9, 2007–2017 Search PubMed.
- Z. Guo, Y. Tian, J. He and H. Song, ChemCatChem, 2024, e202301595 CrossRef CAS.
- S. Cao, T. Sun, Y. Peng, X. Yu, Q. Li, F. L. Meng, F. Yang, H. Wang, Y. Xie, C.-C. Hou and Q. Xu, Small, 2024, 20, 2404285 CrossRef CAS PubMed.
- K. Sayama, ACS Energy Lett., 2018, 3, 1093–1101 CrossRef CAS.
- W. A. Rutala and D. J. Weber, Clin. Microbiol. Rev., 1997, 10, 597–610 CrossRef CAS PubMed.
- World Health Organization, Guidelines for drinking-water quality, 4th edition, incorporating the 1st addendum, World Health Organization, 2017.
- S. Iguchi, Y. Miseki and K. Sayama, Sustainable Energy Fuels, 2018, 2, 155–162 RSC.
- R. Pang, Y. Miseki, S. Okunaka and K. Sayama, ACS Sustainable Chem. Eng., 2020, 8, 8629–8637 CrossRef CAS.
- R. K. B. Karlsson and A. Cornell, Chem. Rev., 2016, 116, 2982–3028 CrossRef CAS PubMed.
- Z. Chen, J. J. Concepcion, N. Song and T. J. Meyer, Chem. Commun., 2014, 50, 8053–8056 RSC.
- M. Busch, N. Simic and E. Ahlberg, Phys. Chem. Chem. Phys., 2019, 21, 19342–19348 RSC.
- M. Luna-Trujillo, R. Palma-Goyes, J. Vazquez-Arenas and A. Manzo-Robledo, J. Electroanal. Chem., 2020, 878, 114661 Search PubMed.
- C. Zhao, Z. Ding, K. Zhang, Z. Du, H. Fang, L. Chen, H. Jiang, M. Wang and M. Wu, Nano-Micro Lett., 2025, 17, 113 CrossRef CAS PubMed.
- K. Mase, M. Yoneda, Y. Yamada and S. Fukuzumi, Nat. Commun., 2016, 7, 11470 CrossRef CAS PubMed.
- L. Huang, R. Li, R. Chong, G. Liu, J. Han and C. Li, Catal. Sci. Technol., 2014, 4, 2913–2918 Search PubMed.
- J. Johnston, C. O’Rourke and A. Mills, Phys. Chem. Chem. Phys., 2023, 25, 31825–31835 RSC.
- S. Iguchi, K. Teramura, S. Hosokawa and T. Tanaka, Phys. Chem. Chem. Phys., 2015, 17, 17995–18003 RSC.
- G. Hippargi, P. Mangrulkar, A. Chilkalwar, N. Labhsetwar and S. Rayalu, Int. J. Hydrogen Energy, 2018, 43, 6815–6823 Search PubMed.
- X. Guan, F. A. Chowdhury, N. Pant, L. Guo, L. Vayssieres and Z. Mi, J. Phys. Chem. C, 2018, 122, 13797–13802 CrossRef CAS.
- V.-H. Dang, T.-A. Nguyen, M.-V. Le, D. Q. Nguyen, Y. H. Wang and J. C.-S. Wu, Chem. Eng. J., 2024, 484, 149213 CrossRef CAS.
- F. Dingenen and S. W. Verbruggen, Renewable Sustainable Energy Rev., 2021, 142, 110866 CrossRef CAS.
- M. S. Yesupatham, A. Augustin, N. Agamendran, B. Honnappa, M. Shanmugam, P. J. J. Sagayaraj, G. Thennarasu, N. C. Sagaya Selvam and K. Sekar, Sustainable Energy Fuels, 2023, 7, 4727–4757 RSC.
- B. Rao, N. Estrada, S. McGee, J. Mangold, B. Gu and W. A. Jackson, Environ. Sci. Technol., 2012, 46, 11635–11643 CrossRef CAS PubMed.
- S. Dresp, F. Dionigi, M. Klingenhof and P. Strasser, ACS Energy Lett., 2019, 4, 933–942 Search PubMed.
- R. Vogt and R. N. Schindler, J. Photochem. Photobiol., A, 1992, 66, 133–140 CrossRef CAS.
- J. Fang, Y. Fu and C. Shang, Environ. Sci. Technol., 2014, 48, 1859–1868 CrossRef CAS PubMed.
- X. Li, M. Kan, T. Wang, Z. Qin, T. Zhang, X. Qian, Y. Kuwahara, K. Mori, H. Yamashita and Y. Zhao, Appl. Catal., B, 2021, 296, 120387 Search PubMed.
- M. S. Koo, X. Chen, K. Cho, T. An and W. Choi, Environ. Sci. Technol., 2019, 53, 9926–9936 CrossRef CAS PubMed.
- M. Szabó, N. Lihi, N. Simic and I. Fábián, Chem. Eng. Res. Des., 2021, 169, 97–102 Search PubMed.
- T. Okada, M. Kodera, Y. Miseki, H. Kusama, T. Gunji and K. Sayama, Chem. Commun., 2024, 60, 3299–3302 RSC.
- M. Jadwiszczak, K. Jakubow-Piotrowska, P. Kedzierzawski, K. Bienkowski and J. Augustynski, Adv. Energy Mater., 2020, 10, 1903213 CrossRef CAS.
- Y. Yang, X. Yuan, Q. Wang, S. Wan, C. Lin, S. Lu, Q. Zhong and K. Zhang, Angew. Chem., Int. Ed., 2024, 63, e202314383 Search PubMed.
- Y. Wang, Y. Wang, J. Zhao and Y. Xu, Appl. Catal., B, 2021, 299, 120676 CrossRef CAS.
- Y. Zhou, Z. Xu, L. Tang, J. Qin, G. Lu, H. Dong, Z. Bian and M. Zhu, Environ. Sci. Technol., 2024, 58, 6049–6057 CrossRef CAS PubMed.
- J. N. Hausmann, R. Schlögl, P. W. Menezes and M. Driess, Energy Environ. Sci., 2021, 14, 3679–3685 RSC.
- J. Yu, B.-Q. Li, C.-X. Zhao and Q. Zhang, Energy Environ. Sci., 2020, 13, 3253–3268 Search PubMed.
- Y. Wang, Y. Liu, D. Wiley, S. Zhao and Z. Tang, J. Mater. Chem. A, 2021, 9, 18974–18993 RSC.
- S. Choi, W. I. Choi, J.-S. Lee, C. H. Lee, M. Balamurugan, A. D. Schwarz, Z. S. Choi, H. Randriamahazaka and K. T. Nam, Adv. Mater., 2023, 35, 2300429 Search PubMed.
- K. S. Exner, J. Anton, T. Jacob and H. Over, Electrocatalysis, 2015, 6, 163–172 CrossRef CAS.
- L. I. Krishtalik, Electrochim. Acta, 1981, 26, 329–337 Search PubMed.
- H. A. Hansen, I. C. Man, F. Studt, F. Abild-Pedersen, T. Bligaard and J. Rossmeisl, Phys. Chem. Chem. Phys., 2010, 12, 283–290 Search PubMed.
- Y. Ye, B. Liao, M. Li, M. Mai, L. Zhang, L. Ma, D. Lin, J. Zhao, D. Chen and X. Ma, J. Alloys Compd., 2023, 947, 169480 Search PubMed.
- R. Sakamoto, D. Horiguchi, T. Ikegami, M. Ishiyama, M. Shiga, K. Sasamoto and Y. Katayama, Anal. Sci., 2003, 19, 1445–1447 CrossRef CAS PubMed.
- Q. Lu, Y. Wang, D. Zhang and H. Cong, Res. Chem. Intermed., 2022, 48, 29–47 Search PubMed.
- R. Pang, Y. Miseki and K. Sayama, Catal. Sci. Technol., 2022, 12, 2935–2942 RSC.
- S. Okunaka, Y. Miseki and K. Sayama, iScience, 2020, 23, 101540 CrossRef CAS PubMed.
- Y. Wang, Q. Lu, S. Han, D. Zhang, H. Cong and C. Chen, Mater. Res. Bull., 2023, 166, 112347 CrossRef CAS.
- J. Ke, M. Adnan Younis, Y. Kong, H. Zhou, J. Liu, L. Lei and Y. Hou, Nano-Micro Lett., 2018, 10, 69 Search PubMed.
- J. Sun, Y. Chi, W. Wang, S. Zhan and F. Zhou, React. Kinet., Mech. Catal., 2024, 137, 1239–1253 CrossRef CAS.
- S. Han, Y. Wang, D. Zhang and H. Cong, Chem. Commun., 2023, 59, 6857–6860 RSC.
- Y. Wang, S. Han, Q. Lu, D. Zhang, J. Wang and P. He, ChemSusChem, 2024, e202400795 Search PubMed.
- Y. Li, W. Nie, Y. Liu, D. Huang, Z. Xu, X. Peng, C. George, C. Yan, Y. J. Tham, C. Yu, M. Xia, X. Fu, X. Wang, L. Xue, Z. Wang, Z. Xu, X. Chi, T. Wang and A. Ding, Environ. Sci. Technol. Lett., 2020, 7, 70–75 Search PubMed.
- A. Fujishima, X. Zhang and D. A. Tryk, Surf. Sci. Rep., 2008, 63, 515–582 Search PubMed.
- H. Wang, Z. Han, Y. Zhou, X. Liu, D. Zeng, W. Wang, D. Sarker, L. Zhang and W. Wang, Appl. Catal., B, 2021, 297, 120436 CrossRef CAS.
- T. R. Harris-Lee, Y. Zhang, C. R. Bowen, P. J. Fletcher, Y. Zhao, Z. Guo, J. W. F. Innocent, S. A. L. Johnson and F. Marken, Electrocatalysis, 2021, 12, 65–77 CrossRef CAS.
- T. Okada, M. Kodera, Y. Miseki, T. Gunji and K. Sayama, J. Catal., 2025, 443, 115969 Search PubMed.
- K. Maeda, K. Teramura, D. Lu, N. Saito, Y. Inoue and K. Domen, Angew. Chem., Int. Ed., 2006, 45, 7806–7809 Search PubMed.
- K. Maeda, K. Teramura, D. Lu, N. Saito, Y. Inoue and K. Domen, J. Phys. Chem. C, 2007, 111, 7554–7560 CrossRef CAS.
- M. Yoshida, K. Takanabe, K. Maeda, A. Ishikawa, J. Kubota, Y. Sakata, Y. Ikezawa and K. Domen, J. Phys. Chem. C, 2009, 113, 10151–10157 CrossRef CAS.
- T. Takata, J. Jiang, Y. Sakata, M. Nakabayashi, N. Shibata, V. Nandal, K. Seki, T. Hisatomi and K. Domen, Nature, 2020, 581, 411–414 CrossRef CAS PubMed.
- M. Qureshi, T. Shinagawa, N. Tsiapis and K. Takanabe, ACS Sustain. Chem. Eng., 2017, 5, 8079–8088 CrossRef CAS.
- S. Okunaka, T. Nakamura, T. Ikeda, K. Tsuruda and H. Tokudome, Sustainable Energy Fuels, 2024, 8, 745–751 RSC.
- Y. Shiraishi, M. Hashimoto, K. Chishiro, K. Moriyama, S. Tanaka and T. Hirai, J. Am. Chem. Soc., 2020, 142, 7574–7583 CrossRef CAS PubMed.
- Y. Shiraishi, Y. Shimabukuro, K. Shima, S. Ichikawa, S. Tanaka and T. Hirai, JACS Au, 2023, 3, 1403–1412 CrossRef CAS PubMed.
- J. Li, Y. Yu and L. Zhang, Nanoscale, 2014, 6, 8473–8488 RSC.
- X. Zhao, Z. Zhao-Karger, D. Wang and M. Fichtner, Angew. Chem., Int. Ed., 2013, 52, 13621–13624 CrossRef CAS PubMed.
- Y. Zhou, H. Wang, X. Liu, D. Zeng, K. Wang, L. Zhang and W. Wang, Appl. Catal., B, 2021, 294, 120265 CrossRef CAS.
- J. Li, L. Cai, J. Shang, Y. Yu and L. Zhang, Adv. Mater., 2016, 28, 4059–4064 Search PubMed.
- Z. Xu, L. Tang, Y. Zhou, G. Lu, H. Dong and M. Zhu, Chem. Eng. J., 2024, 493, 152589 CrossRef CAS.
- Y. Shi, H. Shou, H. Li, G. Zhan, X. Liu, Z. Yang, C. Mao, J. Cheng, X. Zhang, Y. Jiang, S. Zhao, J. Wang, X. Liu, L. Song, H. Sun and L. Zhang, Angew. Chem., Int. Ed., 2023, 62, e202302286 CrossRef CAS PubMed.
- J. Wang, L. Zhang, R. Li, D. Zeng, W. Wang and W. Wang, Chem. – Eur. J., 2023, 29, e202301457 Search PubMed.
- J. Desilvestro and M. Grätzel, J. Electroanal. Chem. Interfacial Electrochem., 1987, 238, 129–150 Search PubMed.
- M. Parvin, I. Savickaja, S. Tutliene, A. Naujokaitis, R. Ramanauskas, M. Petruleviciene and J. Juodkazyte, J. Electroanal. Chem., 2024, 954, 118026 Search PubMed.
- J. C. Hill and K.-S. Choi, J. Phys. Chem. C, 2012, 116, 7612–7620 Search PubMed.
- Q. Mi, A. Zhanaidarova, B. S. Brunschwig, H. B. Gray and N. S. Lewis, Energy Environ. Sci., 2012, 5, 5694–5700 RSC.
- Y. Shi, Y. Li, X. Wei, J. Feng, H. Li and W. Zhou, J. Electron. Mater., 2017, 46, 6878–6883 CrossRef CAS.
- S. Ahmed, I. A. I. Hassan, H. Roy and F. Marken, J. Phys. Chem. C, 2013, 117, 7005–7012 Search PubMed.
- S. Y. Jo, J. Y. Bae, J. Y. Park, M. K. Kim, W.-S. Chae, Y.-I. Kim and K. M. Nam, ACS Appl. Mater. Interfaces, 2024, 16, 54864–54872 Search PubMed.
- J. Juodkazytė, M. Petrulevičienė, M. Parvin, B. Šebeka, I. Savickaja, V. Pakštas, A. Naujokaitis, J. Virkutis and A. Gegeckas, J. Electroanal. Chem., 2020, 871, 114277 CrossRef.
- S. J. Calero, P. Ortiz, A. F. Oñate and M. T. Cortés, Int. J. Hydrogen Energy, 2016, 41, 4922–4930 Search PubMed.
- A. G. Breuhaus-Alvarez, Q. Cheek, J. J. Cooper, S. Maldonado and B. M. Bartlett, J. Phys. Chem. C, 2021, 125, 8543–8550 Search PubMed.
- D. Chen, Z. Xie, Y. Tong and Y. Huang, Energy Fuels, 2022, 36, 9932–9949 Search PubMed.
- H. Shi, H. Guo, S. Wang, G. Zhang, Y. Hu, W. Jiang and G. Liu, Energy Fuels, 2022, 36, 11404–11427 Search PubMed.
- J. H. Kim and J. S. Lee, Adv. Mater., 2019, 31, 1806938 CrossRef PubMed.
- X. Zhao, J. Hu, S. Chen and Z. Chen, Phys. Chem. Chem. Phys., 2018, 20, 13637–13645 RSC.
- M. Chen, X. Chang, C. Li, H. Wang and L. Jia, J. Colloid Interface Sci., 2023, 640, 162–169 CrossRef CAS PubMed.
- S. Sriwichai, R. Irani, F. Xi, D. Friedrich, C. Höhn, I. Y. Ahmet, N. Wetchakun and F. F. Abdi, Solar RRL, 2021, 5, 2100268 CrossRef CAS.
- M. Petruleviciene, I. Savickaja, A. Griguceviciene, A. Naujokaitis, R. Ramanauskas and J. Juodkazyte, J. Photochem. Photobiol., A, 2023, 443, 114842 CrossRef CAS.
- W. Luo, Z. Yang, Z. Li, J. Zhang, J. Liu, Z. Zhao, Z. Wang, S. Yan, T. Yu and Z. Zou, Energy Environ. Sci., 2011, 4, 4046–4051 RSC.
- S. Iguchi, H. Tateno, S. Takasugi, Y. Miseki and K. Sayama, Sustainable Energy Fuels, 2019, 3, 3441–3447 RSC.
- P. Chen, B. Kang, P. Liu, X. Cheng, S. Zhong, X. Wang and B. Fang, J. Power Sources, 2025, 628, 235860 CrossRef CAS.
- Z. Xi, C. Zhou, K. Kisslinger, T. Nanayakkara, F. Lu, X. Tong and M. Liu, ACS Appl. Mater. Interfaces, 2023, 15, 49281–49288 CrossRef CAS PubMed.
- S. Okunaka, Y. Miseki and K. Sayama, Catal. Sci. Technol., 2021, 11, 5467–5471 RSC.
- Y. Zhang, J. Cui and Y. Pei, Chem. Eng. J., 2023, 464, 142652 CrossRef CAS.
- Q. Xiong, X. Zhang, Q. Cheng, G. Liu, G. Xu, J. Li, X. Ye and H. Gao, Nano Res., 2021, 14, 1443–1449 CrossRef CAS.
- I. Chauhan, K. K. Patra, H. Bajpai, N. B. Mhamane, K. N. Salgaonkar and C. S. Gopinath, Dalton Trans., 2023, 52, 2051–2061 RSC.
- S. Zhang, I. Ahmet, S.-H. Kim, O. Kasian, A. M. Mingers, P. Schnell, M. Kölbach, J. Lim, A. Fischer, K. J. J. Mayrhofer, S. Cherevko, B. Gault, R. van de Krol and C. Scheu, ACS Appl. Energy Mater., 2020, 3, 9523–9527 CrossRef CAS PubMed.
- S. Zhang, M. Rohloff, O. Kasian, A. M. Mingers, K. J. J. Mayrhofer, A. Fischer, C. Scheu and S. Cherevko, J. Phys. Chem. C, 2019, 123, 23410–23418 CrossRef CAS.
- S. Chen and L.-W. Wang, Chem. Mater., 2012, 24, 3659–3666 Search PubMed.
- A. M. Rassoolkhani, W. Cheng, J. Lee, A. McKee, J. Koonce, J. Coffel, A. H. Ghanim, G. A. Aurand, C. Soo Kim, W. Ik Park, H. Jung and S. Mubeen, Commun. Chem., 2019, 2, 57 CrossRef.
- C. Boyle, N. Skillen, H. Q. N. Gunaratne, P. K. Sharma, J. A. Byrne and P. K. J. Robertson, Mater. Sci. Semicond. Process., 2020, 109, 104930 Search PubMed.
- J. Wu, Y. Tao, C. Zhang, Q. Zhu, D. Zhang and G. Li, J. Hazard. Mater., 2023, 443, 130363 Search PubMed.
- Z. Hu, H. Wu, S. Li, C. Zhang, R. Liang and M. Zhou, Chem. Eng. J., 2024, 498, 155308 CrossRef CAS.
- S.-T. Xiao, S.-M. Wu, L. Wu, Y. Dong, J.-W. Liu, L.-Y. Wang, X.-Y. Chen, Y.-T. Wang, G. Tian, G.-G. Chang, M. Shalom, P. Fornasiero and X.-Y. Yang, ACS Nano, 2023, 17, 18217–18226 CrossRef CAS PubMed.
- H. Nishiyama, T. Yamada, M. Nakabayashi, Y. Maehara, M. Yamaguchi, Y. Kuromiya, Y. Nagatsuma, H. Tokudome, S. Akiyama, T. Watanabe, R. Narushima, S. Okunaka, N. Shibata, T. Takata, T. Hisatomi and K. Domen, Nature, 2021, 598, 304–307 CrossRef CAS PubMed.
- Y. Goto, T. Hisatomi, Q. Wang, T. Higashi, K. Ishikiriyama, T. Maeda, Y. Sakata, S. Okunaka, H. Tokudome, M. Katayama, S. Akiyama, H. Nishiyama, Y. Inoue, T. Takewaki, T. Setoyama, T. Minegishi, T. Takata, T. Yamada and K. Domen, Joule, 2018, 2, 509–520 CrossRef CAS.
- T. Uekert, M. A. Bajada, T. Schubert, C. M. Pichler and E. Reisner, ChemSusChem, 2021, 14, 4190–4197 CrossRef CAS PubMed.
- S. Dilger, M. Trottmann and S. Pokrant, ChemSusChem, 2019, 12, 1931–1938 CrossRef CAS PubMed.
- A. Hankin, F. E. Bedoya-Lora, C. K. Ong, J. C. Alexander, F. Petter and G. H. Kelsall, Energy Environ. Sci., 2017, 10, 346–360 RSC.
- I. Rodríguez-Gutiérrez, L. R. P. Peregrino, G. H. Morais, F. Coa, D. S. Teodoro Martinez, R. V. Gonçalves and F. L. Souza, ACS Energy Lett., 2025, 10, 4769–4776 CrossRef.
- P. P. Kunturu, S. Bera, H. Johnson and M. N. Tsampas, Artif. Photosynth., 2025, 1, 106–116 CrossRef CAS.
- Y. An, C. Lin, C. Dong, R. Wang, J. Hao, J. Miao, X. Fan, Y. Min and K. Zhang, ACS Energy Lett., 2024, 9, 1415–1422 Search PubMed.
- K. Bienkowski, R. Solarska, L. Trinh, J. Widera-Kalinowska, B. Al-Anesi, M. Liu, G. K. Grandhi, P. Vivo, B. Oral, B. Yılmaz and R. Yıldırım, ACS Catal., 2024, 14, 6603–6622 CrossRef CAS PubMed.
- V.-H. Trinh and J. Seo, ACS Sustainable Chem. Eng., 2023, 11, 1655–1665 CrossRef CAS.
- J. Seo, S. Mun and V.-H. Trinh, Inorg. Chem. Front., 2026, 13, 51–62 RSC.
- J. Seo and K. Domen, Mater. Chem. Front., 2024, 8, 1451–1479 RSC.
- A. Kumar, A. Kumar and V. Krishnan, ACS Catal., 2020, 10, 10253–10315 CrossRef CAS.
- X. Li, H. Zhao, J. Liang, Y. Luo, G. Chen, X. Shi, S. Lu, S. Gao, J. Hu, Q. Liu and X. Sun, J. Mater. Chem. A, 2021, 9, 6650–6670 RSC.
- X. Ma, J. Qu, L. Zhang, J. Ma, Y. Cao, K. Nie, Q. Ji, C. Wang, L. Ma and D. Jing, Int. J. Hydrogen Energy, 2024, 55, 441–454 CrossRef CAS.
- X. Ma, Y. Gao, B. Yang, X. Lou, J. Huang, L. Ma and D. Jing, Mater. Today Nano, 2023, 21, 100289 CrossRef CAS.
- Y. Jiang, S. Zhou, S. S. Mofarah, R. Niu, Y. Sun, A. Rawal, H. Ma, K. Xue, X. Fang, C. Y. Toe, W.-F. Chen, Y.-S. Chen, J. M. Cairney, R. Rahman, Z. Chen, P. Koshy, D. Wang and C. C. Sorrell, Nano Energy, 2023, 116, 108830 CrossRef CAS.
- S. Navalón, A. Dhakshinamoorthy, M. Álvaro, B. Ferrer and H. García, Chem. Rev., 2023, 123, 445–490 CrossRef PubMed.
- K. Sun, Y. Qian and H.-L. Jiang, Angew. Chem., Int. Ed., 2023, 62, e202217565 CrossRef CAS PubMed.
- B. Singh and S. Verma, Mater. Chem. Front., 2024, 8, 3136–3149 RSC.
- W.-Y. Chen, Y.-A. Yang and I.-W. P. Chen, Chem. Eng. J., 2025, 520, 166045 CrossRef CAS.
- M. Jothi, P. Gnanasekar, B. S. Ooi and J. Kulandaivel, J. Phys. Chem. C, 2024, 128, 12799–12807 CrossRef CAS.
- H. Liu, P. Fu, D. Cui, A. Yuan, H. Zhang, J. Du, Y. Cheng, G. Wang, X. Yang and L. Chen, J. Colloid Interface Sci., 2026, 703, 139234 CrossRef CAS PubMed.
- Y. Bao, H. Ru, Y. Wang, K. Zhang, R. Yu, Q. Wu, A. Yu, D.-S. Li, C. Sun, W. Li and J. Tu, Adv. Funct. Mater., 2024, 34, 2314611 CrossRef CAS.
- D. Xie, J. Wang, B. Huang, Y. Yang, D. Lin, C. Xu and F. Xie, CrystEngComm, 2025, 27, 678–686 RSC.
- J. Li, S. Sun, N. Gao, H. Li, K. Liang, J. Hai, S. He, X. Mu and B. Wang, Nanoscale Adv., 2023, 5, 3527–3535 RSC.
- L. Li, Z. Zhou, L. Li, Z. Zhuang, J. Bi, J. Chen, Y. Yu and J. Yu, ACS Sustainable Chem. Eng., 2019, 7, 18574–18581 Search PubMed.
- Q. Yue, G. Li, P. Fu, B. Meng, F. Ma, Y. Zhou and J. Wang, Nano Res., 2023, 16, 6251–6259 CrossRef CAS.
- C.-L. Chang, T.-F. Huang, W.-C. Lin, L.-Y. Ting, C.-H. Shih, Y.-H. Chen, J.-J. Liu, Y.-T. Lin, Y.-T. Tseng, Y.-H. Wu, Y.-E. Sun, M. H. Elsayed, C.-W. Chen, C.-H. Yu and H.-H. Chou, Adv. Energy Mater., 2023, 13, 2300986 CrossRef CAS.
- Q. Yue, Z. Zhang, X. Liu, C. Zhu, Y. Wen, P. Fu, Q. Hu, X. Qu, Y. Zhou and J. Wang, Chem. Eng. J., 2025, 507, 160481 CrossRef CAS.
- X. Zhang, M. Chang, D. Wang, L. Wang, X. Yang, Z. Ben, Q. Zhang and Y. Lu, J. Colloid Interface Sci., 2025, 682, 1151–1163 CrossRef CAS PubMed.
- J. Zhang, X. Li, J. Chang, P. She, H. Li, S. Qiu and Q. Fang, Angew. Chem., Int. Ed., 2025, e19752 Search PubMed.
- S. Xiao, D. Wan, K. Zhang, H. Qu and J. Peng, J. Environ. Sci., 2016, 50, 103–108 CrossRef CAS PubMed.
- Y. Zhang, Y. Ji, J. Li, J. Bai, S. Chen, L. Li, J. Wang, T. Zhou, P. Jiang, X. Guan and B. Zhou, J. Hazard. Mater., 2021, 402, 123725 CrossRef CAS PubMed.
- F. Li, L. Sun, Y. Liu, X. Fang, C. Shen, M. Huang, Z. Wang and D. D. Dionysiou, J. Hazard. Mater., 2020, 400, 123246 CrossRef PubMed.
- X. Zhang, W. Li, X. Gong, W. Fan and P. Ren, Desalin. Water Treat., 2015, 54, 1003–1012 CrossRef CAS.
- Y. Ji, J. Bai, J. Li, T. Luo, L. Qiao, Q. Zeng and B. Zhou, Water Res., 2017, 125, 512–519 CrossRef CAS PubMed.
- Z. Yan, Z. Dai, W. Zheng, Z. Lei, J. Qiu, W. Kuang, W. Huang and C. Feng, Water Res., 2021, 205, 117678 CrossRef CAS PubMed.
- Y. Zhang, J. Li, J. Bai, L. Li, S. Chen, T. Zhou, J. Wang, L. Xia, Q. Xu and B. Zhou, Environ. Sci. Technol., 2019, 53, 6945–6953 CrossRef CAS PubMed.
- Z. Shen, Y. Zhang, C. Zhou, J. Bai, S. Chen, J. Li, J. Wang, X. Guan, M. Rahim and B. Zhou, Water Res., 2020, 170, 115357 CrossRef CAS PubMed.
- J. E. Grebel, J. J. Pignatello and W. A. Mitch, Environ. Sci. Technol., 2010, 44, 6822–6828 CrossRef CAS PubMed.
- X. Zhang, A. Hu, C. Pan, Q. Zhao, X. Wang and J. Lu, Org. Process Res. Dev., 2013, 17, 1591–1596 CrossRef CAS.
- J. Wang, R. Li, D. Zeng, W. Wang, Y. Zhang, L. Zhang and W. Wang, Chem. Eng. J., 2023, 452, 139505 CrossRef CAS.
- S. J. Freakley, N. Dimitratos, D. J. Willock, S. H. Taylor, C. J. Kiely and G. J. Hutchings, Acc. Chem. Res., 2021, 54, 2614–2623 CrossRef CAS PubMed.
- L. A. Hull, W. P. Giordano, D. H. Rosenblatt, G. T. Davis, C. K. Mann and S. B. Milliken, J. Phys. Chem., 1969, 73, 2147–2152 CrossRef CAS.
- W. H. Dennis, L. A. Hull and D. H. Rosenblatt, J. Org. Chem., 1967, 32, 3783–3787 CrossRef CAS.
- E. E. van Tamelen, V. B. Haarstad and R. L. Orvis, Tetrahedron, 1968, 24, 687–704 CrossRef CAS PubMed.
- W. E. Pereira, Y. Hoyano, R. E. Summons, V. A. Bacon and A. M. Duffield, Biochim. Biophys. Acta, Gen. Subj., 1973, 313, 170–180 CrossRef CAS PubMed.
- V. C. Hand, M. P. Snyder and D. W. Margerum, J. Am. Chem. Soc., 1983, 105, 4022–4025 CrossRef CAS.
- T. Stelmaszyńska and J. M. Zgliczynski, Eur. J. Biochem., 1978, 92, 301–308 CrossRef PubMed.
- C. L. Hawkins and M. J. Davies, J. Chem. Soc., Perkin Trans. 2, 1998, 1937–1946 RSC.
- Z. T. How, K. L. Linge, F. Busetti and C. A. Joll, Environ. Sci. Technol., 2017, 51, 4870–4876 CrossRef CAS PubMed.
- T. Ding, L. Long, M.-J. Yuan, W. Xu, S.-Y. Wong, H. Gao, Y.-H. Wang, Y.-Y. Yeung and X. Jiang, J. Am. Chem. Soc., 2025, 147, 16225–16236 CrossRef CAS PubMed.
- P.-F. Biard, A. Couvert, C. Renner and J.-P. Levasseur, Can. J. Chem. Eng., 2010, 88, 682–687 CrossRef CAS.
- M. Azizi, P.-F. Biard, A. Couvert and M. Ben Amor, Chem. Eng. Res. Des., 2015, 94, 141–152 CrossRef CAS.
- M. Whiteman, N. S. Cheung, Y.-Z. Zhu, S. H. Chu, J. L. Siau, B. S. Wong, J. S. Armstrong and P. K. Moore, Biochem. Biophys. Res. Commun., 2005, 326, 794–798 CrossRef CAS PubMed.
- X. L. Armesto, M. Canle, M. I. Fernandez, M. V. Garcia and J. A. Santaballa, Tetrahedron, 2000, 56, 1103–1109 CrossRef CAS.
- J. Ra, J. Tolu, D. Rentsch, T. Manasfi and U. von Gunten, Water Res., 2025, 270, 122806 CrossRef CAS PubMed.
- Y. Hu, G. Xie and D. M. Stanbury, Inorg. Chem., 2017, 56, 4047–4056 CrossRef CAS PubMed.
- G. Ding, C. Li, Y. Ni, L. Chen, L. Shuai and G. Liao, EES. Catal., 2023, 1, 369–391 RSC.
- Y.-Y. Li, J.-S. Fan, R.-Q. Tan, H.-C. Yao, Y. Peng, Q.-C. Liu and Z.-J. Li, ACS Appl. Mater. Interfaces, 2020, 12, 54507–54516 CrossRef CAS PubMed.
- L. Hao, L. Kang, H. Huang, L. Ye, K. Han, S. Yang, H. Yu, M. Batmunkh, Y. Zhang and T. Ma, Adv. Mater., 2019, 31, 1900546 CrossRef PubMed.
- W. Zhang, W. Ma, Y. Ma, P. Chen, Q. Ye, Y. Wang, Z. Jiang, Y. Ou and F. Dong, Nano Mater. Sci., 2024, 6, 235–243 CrossRef CAS.
- D.-Y. Kuo, H. Paik, J. N. Nelson, K. M. Shen, D. G. Schlom and J. Suntivich, J. Chem. Phys., 2019, 150, 041726 CrossRef PubMed.
- S. Saha, P. Gayen and V. K. Ramani, ChemCatChem, 2020, 12, 4922–4929 CrossRef CAS.
- A. R. Zeradjanin, N. Menzel, P. Strasser and W. Schuhmann, ChemSusChem, 2012, 5, 1897–1904 CrossRef CAS PubMed.
- I. E. Wachs, Catal. Today, 1996, 27, 437–455 CrossRef CAS.
- Y. Krockenberger, J. Ruiters, H. Irie, Y. Taniyasu and H. Yamamoto, J. Phys. Chem. C, 2025, 129, 18157–18163 CrossRef CAS.
- Y. Ping, G. Galli and W. A. I. Goddard, J. Phys. Chem. C, 2015, 119, 11570–11577 CrossRef CAS.
- K. S. Exner, ChemElectroChem, 2020, 7, 1448–1455 CrossRef CAS.
- D.-Y. Kuo, H. Paik, J. Kloppenburg, B. Faeth, K. M. Shen, D. G. Schlom, G. Hautier and J. Suntivich, J. Am. Chem. Soc., 2018, 140, 17597–17605 CrossRef CAS PubMed.
- K. S. Exner, Phys. Chem. Chem. Phys., 2020, 22, 22451–22458 RSC.
- K. S. Exner, J. Phys. Chem. C, 2019, 123, 16921–16928 CrossRef CAS.
- K. S. Exner, J. Anton, T. Jacob and H. Over, Angew. Chem., Int. Ed., 2014, 53, 11032–11035 CrossRef CAS PubMed.
- C. E. Finke, S. T. Omelchenko, J. T. Jasper, M. F. Lichterman, C. G. Read, N. S. Lewis and M. R. Hoffmann, Energy Environ. Sci., 2019, 12, 358–365 RSC.
- H. Gong, B. Zhu, D. Zhang, T. Liu, P. Kuang and J. Yu, Appl. Catal., B, 2024, 349, 123892 CrossRef CAS.
- M. Teranishi, R. Hoshino, S. Naya and H. Tada, Angew. Chem., Int. Ed., 2016, 55, 12773–12777 CrossRef CAS PubMed.
- S. Sun, L. He, M. Yang, J. Cui and S. Liang, Adv. Funct. Mater., 2022, 32, 2106982 CrossRef CAS.
- D. Chen, C.-L. Dong, Y. Zou, D. Su, Y.-C. Huang, L. Tao, S. Dou, S. Shen and S. Wang, Nanoscale, 2017, 9, 11969–11975 RSC.
- Y. Yan, C. Liu, H. Jian, X. Cheng, T. Hu, D. Wang, L. Shang, G. Chen, P. Schaaf, X. Wang, E. Kan and T. Zhang, Adv. Funct. Mater., 2021, 31, 2009610 CrossRef CAS.
- F. T. Haase, A. Bergmann, T. E. Jones, J. Timoshenko, A. Herzog, H. S. Jeon, C. Rettenmaier and B. R. Cuenya, Nat. Energy, 2022, 7, 765–773 CrossRef CAS.
- Z. Li, L. Luo, M. Li, W. Chen, Y. Liu, J. Yang, S.-M. Xu, H. Zhou, L. Ma, M. Xu, X. Kong and H. Duan, Nat. Commun., 2021, 12, 6698 CrossRef CAS PubMed.
- Y. Chen and L. Zhang, Appl. Catal., B, 2024, 347, 123768 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |
