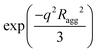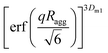Inverse vulcanization enabled self-motivated polysulfide silane: an ultra-efficient interfacial architecture for silica-filled elastomer hybrids
Dong
Wang
a,
Yun
Duan
a,
Jialiang
Cheng
a,
Yiting
Zhang
a,
Zhenghai
Tang
*a,
Panchao
Yin
 b,
Baochun
Guo
b,
Baochun
Guo
 *a and
Liqun
Zhang
*a and
Liqun
Zhang
 ac
ac
aInstitute of Emergent Elastomers, Guangdong Basic Research Center of Excellence for Energy & Information Polymer Materials, School of Materials Science and Engineering, South China University of Technology, Guangzhou 510640, China. E-mail: mszhtang@scut.edu.cn; psbcguo@scut.edu.cn
bState Key Laboratory of Luminescent Materials and Devices, South China Advanced Institute for Soft Matter Science and Technology, South China University of Technology, Guangzhou 510640, China
cSchool of Chemical Engineering and Technology, Xi’an Jiao Tong University, Xi'an 710049, China
First published on 15th October 2025
Abstract
Incorporating sulfur-containing silane coupling agents (SSCAs) into silica-filled rubber composites is essential to improve the compatibility between silica and the rubber matrix. However, the synthesis of SSCAs requires multi-step processes and organic solvent usage, and the silanization efficiency is rather low. Herein, we presented the facile synthesis of a self-motivated polysulfide silane (PSTG) via one-pot sequential inverse vulcanization of sulfur, methoxy polyethylene glycol acrylate and triethoxyvinylsilane and utilized it as a new-brand interfacial modifier for rubber composites. The polyethylene glycol chains in PSTG direct its ethoxysilyl groups onto the silica surface via hydrogen bonds and subsequently promote the silanization reaction, and the polysulfide fragments couple with rubber chains during vulcanization. The interactions of PSTG with silica and hierarchical structures of the composites were comprehensively studied. The incorporation of PSTG greatly strengthens interfacial interactions and improves silica dispersion in rubber composites, resulting in remarkably high static mechanical properties and ultra-low hysteresis loss when compared to the most widely used SSCA bis[γ-(triethoxysilyl)propyl] tetrasulfide at the same ethoxysilyl group content.
New conceptsSulfur-containing silane coupling agents (SSCAs) are indispensable for optimizing the interfacial compatibility in the fabrication of silica-filled rubber composites, yet their reputation is tarnished by two persistent challenges: cumbersome synthesis processes and low silanization efficiency. Herein, we report an entirely new molecular-design paradigm of SSCAs by engineering polyethylene glycol chains into a polysulfide silane (PSTG) that was facilely synthesized by one-pot sequential inverse vulcanization. The polyethylene glycol chains in PSTG direct its ethoxysilyl groups onto the silica surface via hydrogen bonds and promote the condensation reaction, leading to a remarkably improved coupling efficiency in the composites. The interactions of PSTG with silica and hierarchical structures of the composites were comprehensively studied. Compared to previously reported SSCA-modified composites, PSTG-mediated composites exhibit ultra-low hysteresis loss, lowering energy consumption and decreasing temperature rise in tire applications. |
Introduction
“Green tires”, formulated from silica-reinforced rubber composites, have become the mainstream of passenger car tires because of the remarkable benefits they offer in lowering rolling resistance (improving the fuel economy) and enhancing wet traction (improving driving safety).1 It is well acknowledged that a 20% reduction in rolling resistance can lead to a 5% decrease in fuel consumption, and the global-scale replacement of conventional tires with “green tires” will conserve approximately 20 billion liters of fuel annually.2 Note that the abundance of silanol groups on the silica surface makes it thermodynamically immiscible with non-polar rubber matrices, resulting in poor silica dispersion and weak interfacial interaction and consequently compromising composites' properties.3 To maximize the potential of silica, sulfur-containing silane coupling agents (SSCAs) such as bis[γ-(triethoxysilyl)propyl] tetrasulfide (TESPT) are commonly incorporated to create molecular bridges between silica and rubber, with the alkoxysilyl groups undergoing condensation with silanol groups on the silica surface and the polysulfide moieties coupling with rubber chains.4 Nevertheless, two enduring challenges persist in the implementation of SSCAs: firstly, SSCAs are generally synthesized via the substitution reaction of sodium polysulfide with 3-chloropropyltriethoxysilane, which involves multi-step procedures and requires organic solvent usage;5–7 secondly, the silanization efficiency in rubber composites is rather low as the high viscosity of rubber matrices impedes diffusion of SSCAs onto the silica surface. Thus far, a high-temperature mixing process and excessive SSCA dosage have been considered essential to achieve satisfactory silanization.8 However, it inevitably leads to premature scorch and an uncontrollable interface structure due to the self-condensation of alkoxysilyl groups.To address the aforementioned dilemmas, two primary methodologies have been developed: the addition of catalysts and the exploration of new coupling chemistries. In the catalytic approach, nucleophiles such as triphenylborane compounds, quinuclidine, ionic liquids, etc. have been incorporated into SSCA-modified composites to promote silane hydrolysis and the subsequent silanization reaction, which can reduce the amount of SSCAs while maintaining the composites' properties.9–11 However, these catalysts often have potential toxicity and pose a risk of leakage due to the polarity difference with respect to rubber matrices. In terms of coupling chemistries, newly emerging compounds carrying two functional sites, including unsaturated oligomers functionalized with hydroxyl or amino groups12,13 and di- or poly-sulfides bearing carboxyl groups,14 have been explored to modify the silica–rubber interface. The polar groups can readily interact with silica via hydrogen bonds, and the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C or S–S bonds are grafted into the rubber network by participating in the crosslinking reaction, thereby enhancing the compatibility between silica and the rubber matrix. However, the interfacial chemistry essentially involves non-covalent interactions, which tend to dissociate under external force. As a result, the resultant composites exhibit inferior comprehensive properties, particularly a high hysteresis loss, when compared to those modified with TESPT. Although the reactive groups, including epoxy,15 Si–H,16 and propane-1,3-diyl,17 have been integrated into modifiers to create covalent linkages across the interface, their reactivity with silica is even lower than that of alkoxysilyl groups. Collectively, it is highly desirable to seek a new interface engineering strategy that can provide robust covalent interactions while having high efficiency.
C or S–S bonds are grafted into the rubber network by participating in the crosslinking reaction, thereby enhancing the compatibility between silica and the rubber matrix. However, the interfacial chemistry essentially involves non-covalent interactions, which tend to dissociate under external force. As a result, the resultant composites exhibit inferior comprehensive properties, particularly a high hysteresis loss, when compared to those modified with TESPT. Although the reactive groups, including epoxy,15 Si–H,16 and propane-1,3-diyl,17 have been integrated into modifiers to create covalent linkages across the interface, their reactivity with silica is even lower than that of alkoxysilyl groups. Collectively, it is highly desirable to seek a new interface engineering strategy that can provide robust covalent interactions while having high efficiency.
Inverse vulcanization,18,19 conceptualized by Pyun et al. in 2013, presents an innovative approach to synthesize sulfur-rich polymers, achieved by directly adding olefins to polysulfide radicals generated from the ring-opening of sulfur (S8) at elevated temperatures. Inverse vulcanized polysulfides (IVPs) demonstrate a high refractive index and in some cases transparency to MWIR and LWIR light, intriguing electrochemical activity and thermomechanical properties, rendering them highly promising for applications in cathodic materials for Li–S batteries,18,20–22 windows and lenses for infrared imaging,23–26 and heavy metal adsorbents.27–30 Owing to its atom-economic and solvent-free advantages, this approach has witnessed rapid growth and diversification. Recently, substantial efforts have been directed toward altering the molecular structure and functionality of IVPs by exploring new olefinic compounds31 and polymerization techniques,32 aiming to regulate their properties and broaden the application areas.19,33 Of particular interest is the synthesis of IVPs containing functional groups, which can endow IVPs with novel properties34 and serve as versatile platforms for post-modification35 and polymerization.36 Very recently, by engineering functional groups that can interact with fillers into IVPs, our group pioneered the use of IVPs as highly efficient interfacial modifiers for rubber composites.37–40 For example, a polysulfide silane was synthesized and utilized as an interfacial modifier for silica-filled rubber composites.38 Notably, although a high coupling efficiency was achieved due to the multiple reactive sites, two critical issues remain unaddressed: (1) similar to traditional SSCAs, the access of IVPs onto the silica surface is impeded and (2) the function mechanism of polysulfide silane and structure–property relationship in the composites have not been clearly elucidated.
In this work, a self-motivated polysulfide silane was synthesized via a one-pot sequential inverse vulcanization process involving S8, methoxy polyethylene glycol acrylate (mPEG) and triethoxyvinylsilane (TEVS). The synthesized polysulfide silane (Poly(Sulfur-TEVS-mPEG), abbreviated as PSTG) architecture possesses three functional components: polysulfide moieties grafted onto rubber chains, ethoxysilyl groups chemically coupled with silanol groups on the silica surface, and polyethylene glycol chains creating a synergistic effect on accelerating the silanization reaction by driving ethoxysilyl groups toward the silica surface to shorten the distance with silanol groups via hydrogen bonding with silica.41 As a proof of concept, incorporating PSTG into silica-filled rubber composites leads to a significantly improved interfacial interaction, resulting in higher static mechanical properties and ultra-low hysteresis loss relative to the TESPT-modified counterpart.
Results and discussion
Synthesis and characterization of PSTG
As illustrated in Fig. 1a, the inverse vulcanization of S8 and mPEG was first conducted at 170 °C; then the reaction temperature was lowered to 150 °C, and the low-boiling-point TEVS was added for copolymerization to produce PSTG. Note that an initial high temperature of 170 °C, above the floor temperature (159 °C) of inverse vulcanization, is essential to cleave the S8 ring for initiating copolymerization with mPEG. | ||
| Fig. 1 (a) Schematic illustration of the synthesis of PSTG by sequential inverse vulcanization of S8 with mPEG and TEVS. (b) FTIR spectra and (c) 1H NMR spectra of mPEG, TEVS and PSTG. | ||
After that, the as-synthesized polysulfide is readily cleaved to generate sulfur radicals at a lower temperature of 150 °C, accommodating the incorporation of TEVS into polysulfide backbones.
The successful synthesis of PSTG was confirmed by FTIR and NMR measurements. Fig. 1b displays the FTIR spectra of mPEG, TEVS and PSTG. Compared with mPEG and TEVS, the absorptions attributed to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibrations at around 1600 cm−1 are absent in PSTG, and a new absorption peak assigned to C–S stretching vibration at 696 cm−1 is observed,22 indicating the addition of C
C stretching vibrations at around 1600 cm−1 are absent in PSTG, and a new absorption peak assigned to C–S stretching vibration at 696 cm−1 is observed,22 indicating the addition of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds to sulfur to form C–S bonds. Additionally, the characteristic absorptions, such as C
C bonds to sulfur to form C–S bonds. Additionally, the characteristic absorptions, such as C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibrations and C–O–C rocking vibrations from mPEG, and Si–O stretching vibrations from TEVS, are inherited into PSTG. The formation of C–S bonds and the preservation of the polyethylene glycol chains and ethoxysilyl groups in PSTG are further verified by 13C NMR analysis (Fig. S1). Furthermore, in the 1H NMR spectra of PSTG (Fig. 1c), the signals related to the protons in the polyethylene glycol chains from mPEG (from 3.35 to 4.20 ppm) and the ethoxysilyl groups from TEVS (at 1.23 and 3.82 ppm) are retained, while the signals attributed to the vinyl groups completely disappeared. In addition, PSTG shows new signals in the range of 1.8–3.2 ppm, which are assigned to the protons in S–CH fragments.18 According to the integral area of the protons in –O–CH3 and –CH3, the molar ratio of mPEG to TEVS units in PSTG is determined to be 1/3.
O stretching vibrations and C–O–C rocking vibrations from mPEG, and Si–O stretching vibrations from TEVS, are inherited into PSTG. The formation of C–S bonds and the preservation of the polyethylene glycol chains and ethoxysilyl groups in PSTG are further verified by 13C NMR analysis (Fig. S1). Furthermore, in the 1H NMR spectra of PSTG (Fig. 1c), the signals related to the protons in the polyethylene glycol chains from mPEG (from 3.35 to 4.20 ppm) and the ethoxysilyl groups from TEVS (at 1.23 and 3.82 ppm) are retained, while the signals attributed to the vinyl groups completely disappeared. In addition, PSTG shows new signals in the range of 1.8–3.2 ppm, which are assigned to the protons in S–CH fragments.18 According to the integral area of the protons in –O–CH3 and –CH3, the molar ratio of mPEG to TEVS units in PSTG is determined to be 1/3.
DSC measurement demonstrates that PSTG exhibits a glass transition temperature of 0.3 °C without the crystalline peak of mPEG (Fig. S2). The number-average molecular weight and polymer dispersity index of PSTG determined by GPC are 1005 g mol−1 and 1.5, respectively (Fig. S3), manifesting as an oligomer. It should be noted that the molecular weights of IVPs determined by GPC analysis are generally undervalued,18 due to the possible presence of loop structures42 and cleavage of polysulfides in solvent. Elemental analysis confirms that the sulfur content in PSTG is 29.7 wt%. Accordingly, the mass fractions of mPEG and TEVS units in PSTG are calculated to be 32.1 and 38.2 wt%, respectively. Assuming that each vinyl group forms two C–S bonds,43 the average S rank in PSTG is estimated to be 3.5. The chemical compositions of the as-synthesized PSTG and the commonly used silane TESPT are listed in Table 1. Compared with TESPT, PSTG has a similar S rank but contains only about half the ethoxysilyl group content.
| Samples | –Si–O–C2H5 (mmol g−1) | mPEG (mmol g−1) | S rank | S mass fraction (%) |
|---|---|---|---|---|
| PSTG | 6.02 | 0.67 | 3.5 | 29.7 |
| TESPT | 11.13 | — | 3.9 | 23.7 |
Interactions between PSTG and silica
The state-of-the-art approach for modifying silica-filled rubber composites is to incorporate SSCAs; however, they suffer from rather low silanization efficiency as the high-viscosity rubber matrices prevent SSCAs from approaching the silica surface.8 In this work, we envisage that PSTG can readily migrate onto the silica surface through hydrogen bonds between polyethylene glycol chains and silanol groups on silica, which shortens the distance of ethoxysilyl groups with silanol groups and facilitates the silanization reaction.41 The interaction between PSTG and silica is schematically illustrated in Fig. 2a; however, the molecular structure of PSTG in the composites cannot be precisely defined due to the complexity of the chemistry during processing. | ||
| Fig. 2 (a) Diagram showing interactions between PSTG and silica. (b) Adsorption energy and its components over 200 ps determined by molecular dynamics simulations. | ||
To prove this notion, molecular dynamics (MD) simulations are conducted to calculate the adsorption energy (Eadsorption) of SSACs (PSTG or TESPT) on the silica surface, as expressed by the following equation:
| Eadsorption = EA + EB − Etotal |
The silanization reactivity was investigated by treating silica with PSTG or TESPT, followed by TGA measurement (Fig. S5). Compared with neat silica, both PSTG- and TESPT-modified silica exhibit a larger mass loss between 150 and 650 °C, which is attributed to the decomposition of the grafted PSTG or TESPT on the silica surface.44 Accordingly, the grafting efficiency of PSTG on silica is calculated to be 52.9%, higher than that of TESPT (44.4%), implying that the hydrogen bonds between PSTG and silica facilitate the silanization reaction.
To gain insights into the interactions between PSTG and silica, the temperature-variable FTIR spectra of an equimass PSTG/silica mixture were recorded from 40 to 120 °C. As shown in Fig. 3a, with increasing temperature, the absorption intensity at 1730 cm−1 attributed to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O interaction with silica via hydrogen bonds decreases, while the absorption of the free C
O interaction with silica via hydrogen bonds decreases, while the absorption of the free C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O at 1750 cm−1 increases, which is due to the dissociation of hydrogen bonds at high temperatures.45,46 In Fig. 3c, the perturbation-correlation moving window (PCMW, Note S1) spectra of the C
O at 1750 cm−1 increases, which is due to the dissociation of hydrogen bonds at high temperatures.45,46 In Fig. 3c, the perturbation-correlation moving window (PCMW, Note S1) spectra of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching region reveal two distinct transition temperatures: the transition at 65 °C is due to the transformation of H-bonded C
O stretching region reveal two distinct transition temperatures: the transition at 65 °C is due to the transformation of H-bonded C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O into free C
O into free C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, and the transition at 95 °C may be due to the desorption of polyethylene glycol chains from silica. Note that the C–O moiety in polyethylene glycol chains may also form hydrogen bonds with silica; however, the related absorptions are spectroscopically unresolved (Fig. S6).
O, and the transition at 95 °C may be due to the desorption of polyethylene glycol chains from silica. Note that the C–O moiety in polyethylene glycol chains may also form hydrogen bonds with silica; however, the related absorptions are spectroscopically unresolved (Fig. S6).
In addition, the absorption intensity evolution for the stretching vibrations of –CH3 at 2976 cm−1 and Si–OH at 957 cm−1 is illustrated in Fig. 3b. As temperature increases, these two absorptions gradually decline, which is because the silanization reaction consumes ethoxysilyl and silanol groups. Furthermore, two-dimensional correlation analysis of the FTIR results was conducted, as depicted in Fig. 3d. The synchronous spectra reveal a positive cross-peak Φ (2976, 957), manifesting a strong correlation between –CH3 and Si–OH. In the asynchronous spectra, the cross-peak Ψ (2976, 957) is negative. Based on Noda′s rule (Note S2),47,48 the intensity change of Si–OH precedes that of –CH3, demonstrating that the silanization reaction follows a two-step pathway: initial deprotonation of Si–OH on silica, followed by nucleophilic substitution with the ethoxysilyl groups of PSTG.9
Silica dispersion and interfacial adhesion in SBR/PSTG-x composites
As a proof of the proposed concept, silica-filled rubber composites modified with varied PSTG dosages were prepared and compared with that modified with TESPT (see the SI). Fig. 4a presents the dependence of shear storage modulus (G′) on the strain amplitude for the composites. All the composites exhibit a nonlinear decrease in G′ with increasing strain, known as the “Payne effect”, which is caused by the collapse of filler agglomerates under oscillatory shear. The “Payne effect” magnitude (ΔG′), expressed as the difference between G′ at small and large strains, can serve as a quantitative indicator of silica dispersion. The smaller the ΔG′, the better the silica dispersion. Compared to SBR/blank, the ΔG′ of SBR/PSTG-x is significantly lower and consistently decreases with increasing PSTG dosage (Fig. S7), clearly indicating improved silica dispersion upon PSTG modification. Particularly, although the ethoxysilyl molar content in SBR/PSTG-5 is similar to that in SBR/TESPT-3 (Table S1), SBR/PSTG-5 exhibits a smaller ΔG′, i.e., better silica dispersion. In addition, SEM and TEM images visually show that silica agglomerates are greatly suppressed and the rubber–silica boundaries become blurred in the PSTG-modified composite (Fig. S8 and S9), confirming improved silica dispersion and strengthened interfacial adhesion. These observations highlight a higher modification efficiency of PSTG, which is attributed to the synergistic mechanism of PSTG: the polyethylene glycol chains direct ethoxysilyl groups onto the silica surface via hydrogen bonds and subsequently promote the condensation reactions among ethoxysilyl groups and silanol groups.41Bound rubber (BR) is a rubber that is either tightly adsorbed or chemically grafted onto the filler surface and cannot be extracted using a good solvent prior to vulcanization. BR content is frequently used to assess interfacial adhesion in rubber composites.49 As shown in Fig. 4b, the BR content in SBR/PSTG-x shows a substantial enhancement from 11% to 70% with increasing PSTG dosage. This enhancement arises from two complementary mechanisms: (1) PSTG functions as a bridge between silica and rubber to enhance their interactions and (2) the improved silica dispersion exposes more surface accessible sites for interaction with the SBR matrix. Notably, SBR/PSTG-3 exhibits a higher BR content than SBR/TESPT-3 despite containing only approximately half the ethoxysilyl group concentration. This exceptional outcome stems from the hydrogen bonds between PSTG and silica, which direct ethoxysilyl groups onto the silica surface and concurrently facilitate the silanization reaction. Furthermore, the crosslink density (Ve) of SBR/PSTG-x consistently increases with PSTG dosage (Fig. S10), as the polysulfide fragments in PSTG can donate activated sulfur radicals and contribute to Ve. Additionally, SBR/PSTG-3 exhibits comparable Ve to SBR/TESPT-3 due to a similar S rank and mass content in PSTG and TESPT.
The interfacial adhesion was further assessed by measuring the freezing point depression of cyclohexane in the swollen composites. As shown in the DSC cooling profiles (Fig. S11), all composites exhibit two exothermic peaks corresponding to cyclohexane crystallization: one peak at approximately 3.0 °C is related to the crystallization of free cyclohexane, and the other peak at a lower temperature is ascribed to the crystallization of confined cyclohexane trapped within the composite. The depressed freezing point of confined cyclohexane arises from the dimensional restrictions of the polymer mesh, which inhibits its crystallization kinetics.50 Therefore, the difference between these two temperatures (ΔT) can be adopted to estimate interfacial adhesion.51 Notably, SBR/PSTG-x demonstrates significantly larger ΔT values than SBR/blank, with ΔT increasing as the PSTG content rises (Fig. 4b). This is primarily because the strengthened interfacial interaction diminishes the vacuoles in the interfacial phase, thereby decreasing the solvent cage size for crystal nuclei formation. Additionally, SBR/PSTG-3 exhibits a greater ΔT than SBR/TESPT-3, evidencing stronger interfacial adhesion in SBR/PSTG-3. This is consistent with the observations of the BR content, collectively confirming the higher effectiveness of PSTG.
The hierarchical structure across multiple length scales in the composites was investigated using ultra-small-angle X-ray scattering (USAXS) measurements. The scattering profiles of SBR/blank and SBR/PSTG-5 are shown in Fig. 4c. Each scattering curve can be segmented by a cutoff 1/Ragg into one form-factor profile (Guinier regime) and two power-law profiles with the power exponents of –Dm1 and −Dm2.52,53 Here, Ragg corresponds to the gyration radius of silica aggregates, and Dm1 and Dm2 are the mass-fractal dimensions of silica aggregates and their agglomerates, respectively. Both SBR/blank and SBR/PSTG-5 exhibit an identical Dm1 value of 2.6, consistent with a theoretical study on the fractality of objects.54 In addition, the Dm2 value for SBR/blank is 2.0, which is lower than 2.3 for SBR/PSTG-5. A higher Dm2 value for SBR/PSTG-5 arises from the PSTG-mediated coupling between silica aggregates, which increases inter-aggregate connectivity within agglomerates.53
Furthermore, the scattering profiles are analyzed using the Beaucage Unified equation to quantitatively determine Ragg:55
at qRagg ≥ 1; the second term corresponds to the Guinier scattering profile of silica aggregates with gyration radius Ragg; and the third term describes a power-law decay I(q)∼q−Dm1 modulated by a damping factor of
at qRagg ≤ 1. Assuming spherical silica aggregates with radius Ra, the relationship between Ragg and Ra is given by:
The calculated Ra values are provided in the inset of Fig. 4c. Compared with SBR/blank, SBR/PSTG-5 exhibits a lower Ra, indicating that PSTG incorporation favors the dispersion of silica in the rubber matrix and leads to the formation of smaller silica aggregates.
Time-resolved USAXS measurements were performed on SBR/blank and SBR/PSTG-5 to probe the hierarchical structure evolution during uniaxial stretching (Fig. S12). The scattering curves parallel to the stretching direction are fitted based on the Beaucage Unified equation to extract the aggregate radius Ra (Fig. S13). As displayed in Fig. 4d, Ra values for both composites increase upon stretching, attributed to the separation of primary silica particles. In the low-strain regime (< 50%), the Ra value of SBR/blank grows faster than that of SBR/PSTG-5, as weakly bound aggregates in SBR/blank are more easily pulled apart under strain. When the strain exceeds 50%, the Ra value of SBR/blank nearly levels off. This behavior arises from the poor interfacial interaction in SBR/blank, which impedes stress transfer to silica aggregates, causing rubber chains to detach from the silica surface and consequently preventing aggregates from being further pulled apart. In contrast, Ra values of SBR/PSTG-5 gently and continuously increase with strain, because the stronger interfacial adhesion maintains efficient stress transfer that enables the continuous deformation of the tightly bound aggregates in the high-strain regime.
Static mechanical properties and hysteresis loss of the composites
The representative stress–strain curves of the composites are presented in Fig. 5a, with characteristic mechanical properties summarized in Table S2. Notably, SBR/PSTG-x exhibits a higher modulus (stress at 300% strain) and greater ultimate strength compared to SBR/blank. Moreover, increasing PSTG dosage leads to a progressive increase in the modulus while concurrently decreasing the breaking strain. Two mechanisms can account for such outcomes: (1) improved silica dispersion and strengthened interfacial adhesion promote the adsorption of rubber chains onto the silica surface, which facilitates stress transfer and restricts chain mobility and (2) PSTG releases activated sulfur to increase crosslink density, resulting in a more constrained network. In addition, at a comparable crosslink density, SBR/PSTG-3 exhibits a higher modulus and ultimate strength than SBR/TESPT-3, despite having only approximately half the ethoxysilyl group concentration of the latter, highlighting the advantages of synergistic mechanisms of PSTG in improving silica dispersion and enhancing interfacial adhesion.Rubber composites are frequently subjected to dynamic loading during service, where their viscoelastic properties are critical in determining energy transfer and dissipation. In the predominant application in tires, rolling resistance, a key performance metric referring to the mechanical energy converted into heat per unit distance, is commonly quantified by the tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ value at 7% strain of rubber composites.56,57
δ value at 7% strain of rubber composites.56,57
As depicted in Fig. 5b, incorporating PSTG remarkably decreases hysteresis loss of the composites, indicating higher elasticity and lower energy dissipation in the composites upon PSTG modification.Particularly, compared to SBR/TESPT-3, the tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ value at 7% strain of SBR/PSTG-5 decreases by 23%. The hysteresis loss mainly stems from the destruction–reformation of silica agglomerates and interfacial friction between silica and rubber chains under repeated deformations.58,59 Here, the reduced hysteresis loss in SBR/PSTG-x is undoubtedly attributed to two aspects: the improved silica dispersion suppresses the self-aggregation, reducing interparticle friction; the enhanced interfacial adhesion restrains rubber chains from slipping onto the silica surface, thereby alleviating interfacial friction.
δ value at 7% strain of SBR/PSTG-5 decreases by 23%. The hysteresis loss mainly stems from the destruction–reformation of silica agglomerates and interfacial friction between silica and rubber chains under repeated deformations.58,59 Here, the reduced hysteresis loss in SBR/PSTG-x is undoubtedly attributed to two aspects: the improved silica dispersion suppresses the self-aggregation, reducing interparticle friction; the enhanced interfacial adhesion restrains rubber chains from slipping onto the silica surface, thereby alleviating interfacial friction.
As substantiated above, the hydrogen bonding interactions facilitate the adsorption of PSTG onto the silica surface and promote the silanization reaction. To further elucidate the mechanisms of the self-motivated PSTG, another reference SBR/(TESPT + mPEG) was prepared by modifying the composite with a mixture of 3.0 phr TESPT and 1.0 phr mPEG (Table S1). Compared with SBR/TESPT-3, the additional incorporation of mPEG improves silica dispersion (Fig. S14) and moderately enhances interfacial interaction (Fig. S15) due to the shielding of silanol groups by mPEG. However, because the mere physical coating of mPEG cannot promote the proximity of TESPT to the silica surface for the silanization reaction, the interfacial interaction in SBR/(TESPT + mPEG) is significantly weaker when compared to SBR/PSTG-5, having similar contents of ethoxysilyl groups and polyethylene glycol chains. As a result, compared with SBR/TESPT-3, SBR/(TESPT + mPEG) exhibits a slightly higher modulus and lower hysteresis loss, but the improvements are much less pronounced than those in SBR/PSTG-5 (Fig. S16). In addition, compared to our previously reported polysulfide silane without polyethylene glycol chains,38 the PSTG-modified composite shows a markedly lower hysteresis loss, reflected by an about 10 °C decrease in the heat build-up. This further highlights the pivotal role of the integrated polyethylene glycol chain in promoting the coupling efficiency.
To assess the practical efficacy of PSTG, solid rubber wheels were fabricated from PSTG-modified (Tread/PSTG) and TESPT-modified (Tread/TESPT) tread composites based on “green tire” formulation (Table S3). The energy loss and temperature increment of the wheels were measured using a rotary power loss machine, with the setup illustrated in Fig. S17. As shown in Fig. 5c, compared with the tread/TESPT wheel, the energy loss of the tread/PSTG wheel demonstrates a 27% reduction (from 2.46 to 1.79 J r−1), and the surface temperature increment decreases by 19.6 °C (from 41.8 to 22.2 °C). After the test, infrared thermography visually confirms a lower temperature on the tread/PSTG wheel due to the reduced energy dissipation (Fig. 5d). These results demonstrate that PSTG-modified composites offer substantial advantages in improving the fuel economy and safety in tire applications.
Conclusions
In summary, we reported a new-brand polysulfide silane (PSTG) as a highly efficient interfacial architecture for silica-filled rubber composites. PSTG was synthesized via a sequential inverse vulcanization technique, as evidenced by 1H NMR and FTIR spectra. The PCMW technique, 2D correlation spectroscopy and MD calculations reveal that the polyethylene glycol chains in PSTG can interact with silica via hydrogen bonds, directing ethoxysilyl groups onto the silica surface and promoting the silanization reaction. Leveraging a higher silanization efficiency, PSTG-mediated composites exhibit greatly strengthened interfacial interactions and improved silica dispersion when compared to TESPT-modified composites despite containing only approximately half the ethoxysilyl group concentration.Particularly, the hierarchical structures, including the silica aggregate size and interfacial interaction in the composites, are quantitatively disclosed by USAXS measurements. Accordingly, the incorporation of PSTG leads to prominently improved quasi-static mechanical properties and remarkably decreased hysteresis loss of the composites. Based on a green tire formula, the energy loss and temperature rise of PSTG-modified tread wheels are significantly decreased when compared with that of tread/TESPT wheels, demonstrating the significant advantages of PSTG in enhancing the fuel economy and improving safety for tire applications. Overall, this work presents a novel paradigm founded on inverse vulcanization to develop an ultra-efficient interfacial modifier for facile synthesis of silica-filled rubber composites.Author contributions
Z. T., B. G. and L. Z. conceived the project, revised the manuscript, and supervised the research. D. W. and Y. D. conducted the experiments, performed the data analysis, and drafted the manuscript. J. C. Y. Z. and P. Y. participated in some of the experiments and the data analysis.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the Supplementary information (SI). Supplementary information: Details for the Raw materials; synthesis of PSTG; preparation of PSTG-modified composites, PSTG-modified silica model compound and PSTG-modified tread composites; characterizations; the model and method of molecular dynamics simulation; the rules of Noda and PCMW; 13C NMR spectrum of PSTG. DSC curves of PSTG and mPEG; GPC trace for PSTG; interfacial model of silica in combination with PSTG/TESPT; TGA curves of neat silica, T-silica and P-silica; temperature-variable FTIR spectra of the mixture of PSTG and silica; ΔG′ for SBR/blank, SBR/TESPT-3 and SBR/PSTG-x; SEM and TEM images of SBR/blank, SBR/TESPT-3 and SBR/PSTG-5; crosslinking density of the composites; DSC cooling curves of the composites swollen in cyclohexane; USAXS diagrams and scattering curves of SBR/blank and SBR/SPVE-5 under different strains; dependence of G′ on the strain, bound rubber content and freezing point depression for SBR/TESPT-3, SBR/(TESPT + mPEG) and SBR/PSTG-5; tensile profiles and dependence of tan δ on strain (@60 °C, 10 Hz) for SBR/TESPT-3, SBR/(TESPT + mPEG) and SBR/PSTG-5; RSS-II model rolling resistance testing machine; formulations for SBR/blank, SBR/TESPT-3, SBR/(TESPT + mPEG), SBR/PSTG-x and tread composites; mechanical properties of SBR/blank, SBR/TESPT-3, SBR/(TESPT + mPEG) and SBR/PSTG-x. See DOI: https://doi.org/10.1039/d5mh01179e.Acknowledgements
This work was funded by the National Natural Science Foundation of China (52322303 and 52130305), the Guangdong Basic and Applied Basic Research Foundation (2023B1515020085), the Postdoctoral Fellowship Program of China Postdoctoral Science Foundation (GZC20250091) and the Key R&D Program of Shandong Province, China (2024CXGC010402). We thank USAXS Beamline BL10U1 at SSRF (Shanghai Synchrotron Radiation Facility, China) for providing the beamtime.Notes and references
- R. J. Dhanorkar, S. Mohanty and V. K. Gupta, Ind. Eng. Chem. Res., 2021, 60, 4517–4535 CrossRef CAS
.
- X. Qin, B. Han, J. Lu, Z. Wang, Z. Sun, D. Wang, T. P. Russell, L. Zhang and J. Liu, Nano Energy, 2018, 48, 180–188 CrossRef CAS
.
- X. Zhang, L. Cai, A. He, H. Ma, Y. Li, Y. Hu, X. Zhang and L. Liu, Compos. Sci. Technol., 2021, 203, 108601 CrossRef CAS
.
- J. T. Byers, Rubber Chem. Technol., 2002, 75, 527–548 CAS
.
- K. W. Park, Z. Zujovic and E. M. Leitao, Macromolecules, 2022, 55, 2280–2289 CrossRef CAS
.
- T. Sokolnicki, A. Franczyk, B. Janowski and J. Walkowiak, Adv. Synth. Catal., 2021, 363, 5493–5500 CrossRef CAS
.
- J. Park, J. Lim and J. Y. Lee, Sci. China Mater., 2023, 66, 1997–2003 CrossRef CAS
.
- W. Kaewsakul, K. Sahakaro, W. K. Dierkes and J. W. M. Noordermeer, Rubber Chem. Technol., 2012, 85, 277–294 CAS
.
- Z. Tang, J. Huang, X. Wu, B. Guo, L. Zhang and F. Liu, Ind. Eng. Chem. Res., 2015, 54, 10747–10756 CrossRef CAS
.
- D. Frasca, D. Schulze, M. Böhning, B. Krafft and B. Schartel, Rubber Chem. Technol., 2016, 89, 316–334 CAS
.
- C. Hayichelaeh, L. A. E. M. Reuvekamp, W. K. Dierkes, A. Blume, J. W. M. Noordermeer and K. Sahakaro, Rubber Chem. Technol., 2017, 90, 651–666 CAS
.
- S. Kong, S. Wu, Z. Tang and B. Guo, Compos. Commun., 2023, 37, 101413 CrossRef
.
- C. Sun, S. Wen, H. Ma, Y. Li, L. Chen, Z. Wang, B. Yuan and L. Liu, Ind. Eng. Chem. Res., 2019, 58, 1454–1461 CrossRef CAS
.
- S. Yu, Z. Tang, S. Wu and B. Guo, Compos. Sci. Technol., 2022, 227, 109624 CrossRef CAS
.
- N. Ye, J. Zheng, X. Ye, J. Xue, D. Han, H. Xu, Z. Wang and L. Zhang, Composites, Part B, 2020, 202, 108301 CrossRef CAS
.
- C. Zhang, Z. Tang, B. Guo and L. Zhang, Compos. Sci. Technol., 2019, 169, 217–223 CrossRef CAS
.
- D. Locatelli, A. Bernardi, L. R. Rubino, S. Gallo, A. Vitale, R. Bongiovanni, V. Barbera and M. Galimberti, ACS Sustainable Chem. Eng., 2023, 11, 2713–2726 CrossRef CAS
.
- W. J. Chung, J. J. Griebel, E. T. Kim, H. Yoon, A. G. Simmonds, H. J. Ji, P. T. Dirlam, R. S. Glass, J. J. Wie, N. A. Nguyen, B. W. Guralnick, J. Park, Á. Somogyi, P. Theato, M. E. Mackay, Y.-E. Sung, K. Char and J. Pyun, Nat. Chem., 2013, 5, 518–524 CrossRef CAS
.
- T. Lee, P. T. Dirlam, J. T. Njardarson, R. S. Glass and J. Pyun, J. Am. Chem. Soc., 2022, 144, 5–22 CrossRef CAS
.
- P. T. Dirlam, R. S. Glass, K. Char and J. Pyun, J. Polym. Sci., Part A: Polym. Chem., 2017, 55, 1635–1668 CrossRef CAS
.
- F. Zhao, Y. Li and W. Feng, Small Methods, 2018, 2, 1800156 CrossRef
.
- I. Gomez, D. Mecerreyes, J. A. Blazquez, O. Leonet, H. Ben Youcef, C. Li, J. L. Gómez-Cámer, O. Bondarchuk and L. Rodriguez-Martinez, J. Power Sources, 2016, 329, 72–78 CrossRef CAS
.
- J. Pyun and R. A. Norwood, Prog. Polym. Sci., 2024, 156, 101865 CrossRef CAS
.
- T. S. Kleine, N. A. Nguyen, L. E. Anderson, S. Namnabat, E. A. LaVilla, S. A. Showghi, P. T. Dirlam, C. B. Arrington, M. S. Manchester, J. Schwiegerling, R. S. Glass, K. Char, R. A. Norwood, M. E. Mackay and J. Pyun, ACS Macro Lett., 2016, 5, 1152–1156 CrossRef CAS PubMed
.
- J. Molineux, T. Lee, K. J. Kim, K.-S. Kang, N. P. Lyons, A. Nishant, T. S. Kleine, S. W. Durfee, J. Pyun and R. A. Norwood, Adv. Opt. Mater., 2024, 12, 2301971 CrossRef CAS
.
- S. J. Tonkin, J. Pople, L. N. Pham, D. Lewis, B. Aljubran, et al., ChemRxiv, 2025, preprint, chemrxiv-2025-wd224-v2 DOI:10.26434/chemrxiv-2025-wd224-v2.
- L.-A. Ko, Y.-S. Huang and Y. A. Lin, ACS Appl. Polym. Mater., 2021, 3, 3363–3372 CrossRef CAS
.
- H.-K. Lin, Y.-S. Lai and Y.-L. Liu, ACS Sustainable Chem. Eng., 2019, 7, 4515–4522 CrossRef CAS
.
- M. P. Crockett, A. M. Evans, M. J. H. Worthington, I. S. Albuquerque, A. D. Slattery, C. T. Gibson, J. A. Campbell, D. A. Lewis, G. J. L. Bernardes and J. M. Chalker, Angew. Chem., Int. Ed., 2016, 55, 1714–1718 CrossRef CAS PubMed
.
- M. J. H. Worthington, R. L. Kucera, I. S. Albuquerque, C. T. Gibson, A. Sibley, A. D. Slattery, J. A. Campbell, S. F. K. Alboaiji, K. A. Muller, J. Young, N. Adamson, J. R. Gascooke, D. Jampaiah, Y. M. Sabri, S. K. Bhargava, S. J. Ippolito, D. A. Lewis, J. S. Quinton, A. V. Ellis, A. Johs, G. J. L. Bernardes and J. M. Chalker, Chem. – Eur. J., 2017, 23, 16219–16230 CrossRef CAS PubMed
.
- C. M. Marshall, J. Molineux, K.-S. Kang, V. Kumirov, K.-J. Kim, R. A. Norwood, J. T. Njardarson and J. Pyun, J. Am. Chem. Soc., 2024, 146, 24061–24074 CrossRef CAS
.
- J. Jia, J. Liu, Z.-Q. Wang, T. Liu, P. Yan, X.-Q. Gong, C. Zhao, L. Chen, C. Miao, W. Zhao, S. Cai, X.-C. Wang, A. I. Cooper, X. Wu, T. Hasell and Z.-J. Quan, Nat. Chem., 2022, 14, 1249–1257 CrossRef CAS
.
- Y. Zhang, R. S. Glass, K. Char and J. Pyun, Polym. Chem., 2019, 10, 4078–4105 RSC
.
- J. M. Scheiger, C. Direksilp, P. Falkenstein, A. Welle, M. Koenig, S. Heissler, J. Matysik, P. A. Levkin and P. Theato, Angew. Chem., Int. Ed., 2020, 59, 18639–18645 CrossRef CAS
.
- Y. Zhang, T. S. Kleine, K. J. Carothers, D. D. Phan, R. S. Glass, M. E. Mackay, K. Char and J. Pyun, Polym. Chem., 2018, 9, 2290–2294 RSC
.
- K.-S. Kang, A. Phan, C. Olikagu, T. Lee, D. A. Loy, M. Kwon, H.-J. Paik, S. J. Hong, J. Bang, W. O. Parker Jr., M. Sciarra, A. R. de Angelis and J. Pyun, Angew. Chem., Int. Ed., 2021, 60, 22900–22907 CrossRef CAS PubMed
.
- D. Wang, Z. Tang, R. Huang, Y. Duan, S. Wu, B. Guo and L. Zhang, Chem. Mater., 2023, 35, 764–772 CrossRef CAS
.
- D. Wang, Z. Tang, R. Huang, H. Li, C. Zhang and B. Guo, Macromolecules, 2022, 55, 8485–8494 CrossRef CAS
.
- D. Wang, Z. Tang, Z. Wang, L. Zhang and B. Guo, Polym. Chem., 2022, 13, 485–491 RSC
.
- D. Wang, Z. Tang, Y. Liu and B. Guo, Green Chem., 2020, 22, 7337–7342 RSC
.
- K. Ma, D. Zhang, Y. Cong and U. Wiesner, Chem. Mater., 2016, 28, 1537–1545 CrossRef CAS
.
- Y. Zhang, J. J. Griebel, P. T. Dirlam, N. A. Nguyen, R. S. Glass, M. E. Mackay, K. Char and J. Pyun, J. Polym. Sci., Part A: Polym. Chem., 2017, 55, 107–116 CrossRef CAS
.
- D. H. Kim, W. Jang, K. Choi, J. S. Choi, J. Pyun, J. Lim, K. Char and S. G. Im, Sci. Adv., 2020, 6, eabb5320 CrossRef CAS PubMed
.
- S. Livi, A. A. Silva, J. Pereira, T. K. L. Nguyen, B. G. Soares, M. B. Cardoso, J.-F. Gérard and J. Duchet-Rumeau, Chem. Eng. J., 2014, 241, 103–111 CrossRef CAS
.
- L. Hou and P. Wu, Carbohydr. Polym., 2019, 205, 420–426 CrossRef CAS PubMed
.
- C. Wong, S. Wang, L. Ren, J.-B. Xu, X. Zeng and R. Sun, Adv. Func. Mater., 2024, 34, 2401157 CrossRef CAS
.
- S. Morita, H. Shinzawa, I. Noda and Y. Ozaki, Appl. Spectrosc., 2006, 60, 398–406 CrossRef CAS PubMed
.
- S.-t Sun and P.-y Wu, Chin. J. Polym. Sci., 2017, 35, 700–712 CrossRef CAS
.
- V. M. Litvinov and P. A. M. Steeman, Macromolecules, 1999, 32, 8476–8490 CrossRef CAS
.
- M. A. López-Manchado, J. L. Valentín, B. Herrero and M. Arroyo, Macromol. Rapid Commun., 2004, 25, 1309–1313 CrossRef
.
- J. L. Valentín, I. Mora-Barrantes, J. Carretero-González, M. A. López-Manchado, P. Sotta, D. R. Long and K. Saalwächter, Macromolecules, 2010, 43, 334–346 CrossRef
.
- H. Kishimoto, Y. Shinohara, Y. Amemiya, K. Inoue, Y. Suzuki, A. Takeuchi, K. Uesugi and N. Yagi, Rubber Chem. Technol., 2008, 81, 541–551 CAS
.
- S. Mihara, R. N. Datta, W. K. Dierkes, J. W. M. Noordermeer, N. Amino, Y. Ishikawa, S. Nishitsuji and M. Takenaka, Rubber Chem. Technol., 2014, 87, 348–359 CAS
.
- S. Tolman and P. Meakin, Phys. Rev. A, 1989, 40, 428–437 CrossRef CAS PubMed
.
- T. Koga, T. Hashimoto, M. Takenaka, K. Aizawa, N. Amino, M. Nakamura, D. Yamaguchi and S. Koizumi, Macromolecules, 2008, 41, 453–464 CrossRef CAS
.
- P. Zhang, M. Morris and D. Doshi, Rubber Chem. Technol., 2016, 89, 79–116 CAS
.
- Société de technologie Michelin, The tyre: rolling resistance and fuel savings, M. Clermont-Ferrand, 2003.
- M.-J. Wang, Rubber Chem. Technol., 1998, 71, 520–589 CAS
.
- M.-J. Wang, S. Wolff and J.-B. Donnet, Rubber Chem. Technol., 1991, 64, 714–736 CAS
.
| This journal is © The Royal Society of Chemistry 2026 |