Open Access Article
Open Access ArticleRecent advances towards BACE1 drug discovery and therapeutics design
Saim Imran ab,
Meera Patela,
Meissam Noroozifarc and
Kagan Kerman
ab,
Meera Patela,
Meissam Noroozifarc and
Kagan Kerman *ac
*ac
aDepartment of Chemistry, University of Toronto, 80 St. George Street, Toronto, ON M5S 3H6, Canada. E-mail: kagan.kerman@utoronto.ca
bTemerty Faculty of Medicine, University of Toronto, 1 King's College Cir, Toronto, ON M5S 3K3, Canada
cDepartment of Physical and Environmental Sciences, University of Toronto Scarborough, 1265 Military Trail, Toronto, ON M1C 1A4, Canada
First published on 9th April 2026
Abstract
Alzheimer's disease (AD) is a progressive neurodegenerative disorder characterized by cognitive decline and memory loss. A key feature of AD is the accumulation of amyloid beta (Aβ) peptides in the form of extracellular plaques. The amyloid cascade hypothesis suggests that the pathogenesis of AD is initiated by the cleavage of amyloid precursor protein (APP) by β-site amyloid precursor protein cleaving enzyme 1 (BACE1). Numerous therapeutic approaches have been pursued to target BACE1 due to its crucial role in AD. However, the complexity of AD and the localization of BACE1 in the brain have posed challenges, leading to the failure of clinical trials and, in some cases, even exacerbating disease progression. Specifically, the blood–brain barrier (BBB) prevents the entry of many molecules, making BACE1 a difficult target to approach. Recent advancements in BACE1 therapy have shifted the focus from traditional enzyme inhibitor-based therapeutics to modulators, antibody therapy, and gene therapy. These approaches offer several advantages, including the ability to efficiently cross the BBB and provide targeted treatment. In this review, we explore the latest developments in modulators, antibody therapy, and gene therapy targeting BACE1 to combat AD. These approaches offer a promising avenue to mitigate the progression of AD and provide a novel therapeutic strategy.
1. Introduction
1.1. Alzheimer's disease
Alzheimer's disease (AD) is a progressive debilitating neurodegenerative disorder that causes memory loss, progressive loss of cognitive function, and decline in language. AD, first reported in 1906 by German neuropathologist, Alois Alzheimer, now affects over 50 million people globally and is anticipated to rise to 152 million by the World Health Organization (WHO) estimates.1,2 The most prevalent cause of dementia in older individuals, it is initially characterized by memory decline, followed by deterioration of cognitive function with increasing age. In addition to affecting mood and behaviour, AD significantly impairs a person's daily functioning, making living with the disease highly disabling, posing challenges for patients and their families. Furthermore, the Global Economic Impact of AD in the United States (UA) is 818 billion US dollars (about 1.1% of GDP) and is projected to rise to two trillion US dollars by 2030.3,4 The cognitive symptoms of AD arise from the damage to neuronal cells responsible for brain function, which can extend to different areas of the brain and lead to additional adverse symptoms that may ultimately be fatal.5 The development of AD is thought to be influenced by multiple factors, including age, lifestyle, environmental factors, and family history, making it a complex disease.6The accumulation of insoluble Aβ oligomers through the amyloid cascade hypothesis has been proposed as a central mechanism in the development of senile plaques.7,8 Aβ peptides are derived upon cleavage of the larger Amyloid Precursor Peptide (APP). This cleavage occurs by secretases – β-secretase, α-secretase, and γ-secretase – which form various Aβ isoforms differing based on their size, from which the major isoforms are Aβ1–38, Aβ1–40, and Aβ1–42.9 Both Aβ1–40, and Aβ1–42 are hydrophobic, fibrillogenic, and are prone to oligomerize and form plaques in the brain.10 While previously it was thought that only Aβ plaques were the neurotoxic species, it has recently been shown that oligomeric forms of Aβ are toxic as well, causing synaptic dysfunction and neuronal disorganization.11 While the physiological function of Aβ is still under debate, it is well established as being linked to AD.12
From the secretases, the α-secretase preferentially cleaves APP outside the central nervous system (CNS), and is found to have a protective role as it cleaves the Aβ sequence itself, producing soluble amyloid precursor protein-α (sAPPα) and preventing Aβ peptide formation.13 However, β-secretase and γ-secretase are of concern as they cleave the APP at sites allowing for the extracellular release of Aβ.14 In particular, this cleavage is initiated by β-secretase, also known as β-site APP cleaving enzyme 1 (BACE1), which cleaves the extracellular domain of APP to produce a soluble extracellular fragment (sAPPβ) and a cell membrane-bound fragment called C99.8 Cleavage of C99 inside its transmembrane domain by γ-secretase results in the releases the intracellular domain of APP and the formation of Aβ peptides. This APP cleavage is mediated by γ-secretase and determines whether Aβ40 or Aβ42 is produced. While Aβ40 is the more common product, Aβ42 is most found in senile plaques as it is more prone to aggregation (β amyloid plaques).14 A summary of various cleavage sites is shown on Fig. 1. Upon Aβ release, it initially forms oligomers, followed by fibrils and finally plaques. Based on the cascade of events, inhibition of β-secretase and γ-secretase reduces released Aβ, slowing plaque formation of and preventing development of AD.15 Alternatively, cleavage of APP by α-secretase can also occur through the non-amyloidogenic pathway leading to the release of sAPPα and C83.8 C83 is further processed to P3 by γ-secretase. P3 is associated with Alzheimer's disease however its role is still unclear.14
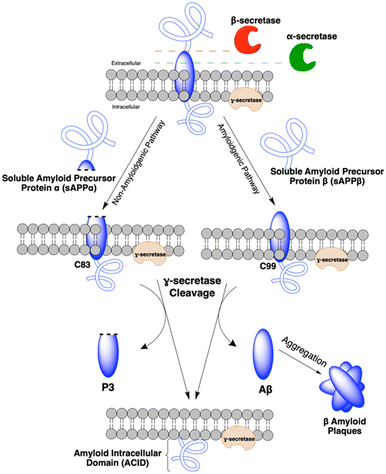 | ||
| Fig. 1 Schematic of the various cleavage sites of secretase enzymes on APP. This figure was drawn by the authors using ChemDraw XML (version 22.2). | ||
Furthermore, plaque formation appears to promote tau hyperphosphorylation, which results in neurofibril tangles.14 Tau is a major microtubule associated protein (MAP) present in adult neurons that plays a role in microtubule network formation and stabilization.16 Activity of tau is mediated by its degree of phosphorylation. In AD patients, tau is in its hyperphosphorylated state about four-fold higher as compared to a healthy brain.16,17 Hyperphosphorylated tau tend to polymerize to form oligomers, causing neurofibrillary tangles which have toxic effects such as amplifying loss of normal tau function and axonal transport defects, resulting in neurodegeneration.18 Initially, these symptoms of plaques and tangles manifest in the hippocampus, the memory centre of the brain, affecting memory formation. As the disease progresses, it spreads to the cortical gray matter, damaging brain cells and compromising their functions, ultimately leading to the various symptoms of dementia.8
Due to its widespread socio-economic impact, AD is considered a global priority to find a cure against, currently for which there still exists none.4 Currently, research is focusing on early stages of disease, attempting to modify drugs to inhibit enzymes involved in the accumulation of Aβ and tau protein. Inhibiting the function of these enzymes can help to reduce plaque formation and either slow down the development of AD and associated dementia or prevent it altogether.19
While this review highlights emerging modalities and delivery strategies for BACE1-targeted therapy, it is important to emphasize that improved BBB penetration or innovative molecular formats alone cannot overcome the fundamental biological constraint amyloid-lowering is most effective before widespread neurodegeneration has occurred. Lessons from large phase II/III trials of small-molecule BACE1 inhibitors consistently show that robust reductions in Aβ production in patients with mild to moderate Alzheimer's disease did not translate into clinical benefit and in some cases were associated with cognitive worsening, underscoring the importance of disease stage when evaluating BACE1 as a therapeutic target.20 These data support a distinction between target validity and clinical timing: BACE1 remains a mechanistically valid target for modulating Aβ biology, but its window of therapeutic opportunity is likely confined to preclinical or very early symptomatic phases, rather than established symptomatic Alzheimer's disease.1,2
1.2. Current therapies
Currently, AD is only treated symptomatically, as there is no existing prevention or cure against it. The U.S. Food and Drug Administration (USFDA) has approved six treatments against AD, of which four are small molecule inhibitor therapies (Fig. 2).5,20,21 Of these, three – donepezil, galantamine, and rivastigmine – target acetylcholinesterase (AChE). AChE is an enzyme involved in the cleavage and breakdown of acetylcholine (ACh) in the synapses. ACh is a neurotransmitter involved in learning and memory related functions. Patients with AD show reduced concentrations of ACh in the brains, impeding memory and learning based tasks.22 Donepezil, galantamine, and rivastigmine all act as inhibitors of AChE, thus allowing for greater accumulation of ACh in the brain, which helps improve cholinergic function. Despite some differences in the pharmacokinetics of the three drugs, all show similar efficacy and tolerability among patients who use them.5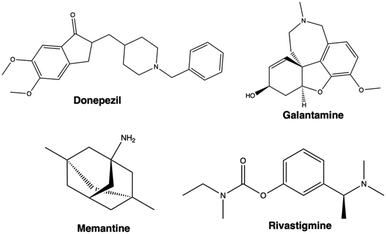 | ||
| Fig. 2 Chemical structures of the four currently widely prescribed Alzheimer's disease drugs. This figure was drawn by the authors using ChemDraw XML (version 22.2). | ||
The fourth small molecule inhibitor is memantine, a modulator that inhibits N-methyl-D-aspartate (NMDA) receptor activity.23 The NMDA receptor is acted on by glutamate, whose overexcitation can lead to neurotoxicity due to glutamate's excitatory properties. Higher levels of glutamate may cause neuronal cell dysfunction and eventual cell death, as observed in AD. By inhibiting the function of the NMDA receptor, memantine can help counteract receptor overexcitation.23 Furthermore, combination therapy with AChE inhibitors has shown to have benefits compared to non-combination treatment.24
Although these drugs are still used to treat Alzheimer's disease, they provide limited benefits in managing AD symptoms and do not prevent the steady decrease in cognition associated with the disease.5 As a result, further strategies have been investigated.
Beyond the designs of small molecule inhibitors, recent developments have been made towards antibody-based methods for the treatment of AD.20,21 Biogen has produced two antibody therapies, aducanumab and lecanemab, for this purpose. Both therapies have been approved under accelerated approval from the FDA, with the contingency of verifying clinical benefit in a confirmatory trial to determine their safety and efficacy.25 Aducanumab, which received approval in 2021, is a monoclonal immunoglobulin G1 (IgG1) antibody that is administered through intravenous infusion and targets Aβ plaques. It selectively targets and binds to soluble aggregated oligomers and insoluble fibril conformations of Aβ plaques (Fig. 3). It is proposed, that the antibody-Aβ complex is then cleared from the brain through fragment crystallizable γ receptor (FcγR)-mediated phagocytosis. It was designed for the treatment of patients with mild cognitive impairment or mild dementia stage of the disease. However, aducanumab was met with controversy for its approval as the clinical trial results were ambiguous regarding the actual efficacy of the drug, its high cost, the need for continuous use of the treatment for lifetime, as well as serious side effects correlated with it such as brain swelling and intracerebral haemorrhage.26 This resulted in limited efficacy and accessibility, and low sales for the drug.
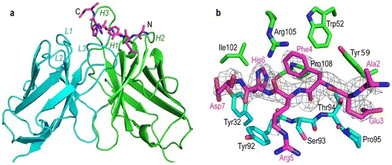 | ||
| Fig. 3 (a) Structure of aducanumab (heavy chains shown in green, light chains shown in cyan) with bound Aβ1–11 peptide (magenta). Nitrogen and oxygen atoms are shown in blue and red, respectively. (b) Various amino acid interactions between aducanumab and Aβ1–11 which give rise to its specific binding. This figure has been adapted from ref. 27 with permission from Nature Portfolio, copyright 2018. | ||
In 2023, lecanemab received accelerated approval.20 Like aducanumab, it is an IgG1 monoclonal antibody, however, lecanemab reduces Aβ plaques by recognizing protofibrils rather than oligomers or fibrils. Additionally, it is priced lower than aducanumab and comparatively shows fewer side effects. Despite the improvements, both therapies remain expensive and are associated with adverse effects that must be considered in therapeutic design.25
Therefore, despite the current FDA-approved therapies for AD, their limited efficacy and various side effects make them inadequate for effective treatment. Consequently, significant efforts are being directed towards finding new and innovative therapies that can modify the course of the disease.28–30 As of December 24, 2024, a search on https://ClinicalTrials.gov for active interventional studies in Phases I to III revealed 249 ongoing trials. This demonstrates the extensive research being conducted to find new and effective treatments for AD. Moreover, aducanumab and lecanemab demonstrate promising emerging strategies, such as the use of antibodies to treat AD. These novel approaches highlight the potential for developing more effective AD therapies.
1.3. β-Secretase
BACE1, a type 1 transmembrane protein, has an open reading frame of 501 residues of which distinct domains are of great interest for research.31 The catalytic domain (residue 46–451), transmembrane domain (residue 452–483), and cytoplasmic domain (residues 484–501) are critical as they contribute to its functionality (Fig. 4a). As a member of the aspartic protease family, its mechanism for cleavage occurs through two aspartic acid residues (Fig. 4b).31 When encountering a substrate peptide, one catalytic aspartic acid initiates attack on the nucleophilic carbonyl carbon with the assistance of a water molecule, while the other activates the carbonyl oxygen resulting in the formation of a tetrahedral intermediate. This intermediate subsequently breaks, leading to the reformation of the carbonyl group and the cleavage of the amide bond generating two products. This is driven to cleave APP, in particular, BACE1 recognizes the methionine 671 – aspartic acid 672 (Met671–Asp672) bond in APP.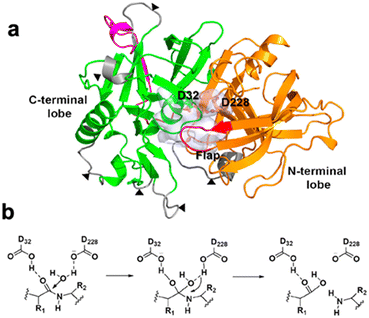 | ||
| Fig. 4 Overview of the catalytic domain of BACE1 and its mechanism on the substrate. (a) The 3D structure of the catalytic domain is shown, with the orange and green representing the two lobes. The magenta color highlights the extension region that is not present in human pepsin, and the red showcases the flap covering the ligand binding pocket. (b) The general catalytic mechanism of BACE1 is mainly implemented by two aspartic acids. This figure has been adapted from ref. 31 with permission from American Chemical Society, copyright 2019. | ||
BACE1 shares approximately 60% sequence identity and has similar domain architecture with its isoform, θ-secretase or BACE2.32 While BACE1 is primarily found in neurons and is widely expressed in the brain, BACE2 does not exhibit organ-specific expression.32 Additionally, BACE1 is a critical therapeutic target in treating AD due to its role as the initial enzyme responsible for initiating the amyloid cascade. In contrast, despite having a similar sequence and domain, BACE2 acts more like α-secretase and cleaves APP at a different site.32,33 Moreover, it shares a similar sequence with various other proteases such as pepsin, renin and cathepsin.31 Due to its similarities to various proteins and proteases, it is critical when designing therapeutics to consider the potential targets that the inhibitors may affect, and to ensure selectivity for BACE1. This consideration is necessary to prevent cross-reactivity, which could potentially lead to negative side effects.
BACE1 has been extensively linked to AD.31,34 Elevated expression of BACE1 in AD patients leads to increased production and accumulation of Aβ. While the activity of BACE1 is positively correlated with extractable Aβ and AD cases, other similar proteins such as BACE2, show activity independent of Aβ concentrations and does not show significant changes in AD.35,36 Additionally, elevated BACE1 expression is associated with reduced protein kinase A activity, which plays a crucial role in memory generation. Therefore, BACE1 represents a critical therapeutic target for regulating AD.
BACE1 also acts on other targets such as Neuregulin1 type III (NRG1). BACE1 cleaved NRG1 acts as a ligand for the erythroblastic oncogene B (ErbB) receptor family. Binding of the ligand to the receptor promotes intercellular signaling to mediate myelin sheath formation.37 Hence, inhibition or significant modulation of BACE1 can impair myelin sheath formation, potentially worsening neurological function. This raises the question of how to achieve the correct level of BACE1 inhibition without compromising its beneficial effects.
Various studies have approached the selective inhibition of BACE1, with animal studies demonstrating a reduction in brain Aβ levels, thereby mitigating the formation of Aβ aggregates with BACE1 inhibition.8,19,38,39 Moreover, knockout mice deficient in the BACE1 gene exhibit lower Aβ production, without exhibiting adverse effects on health, fertility, or phenotype in adulthood. However, the effective inhibition of BACE1 is hindered by the enzyme's location in the brain and therefore, the need to cross the blood–brain barrier (BBB).40 Thus, therapy designs against BACE1 need to have physiochemical properties that allow for crossing the BBB.
Furthermore, the gene that encodes BACE1 is located on chromosome 11 and undergoes complex alternative splicing to produce various protein isoforms with different enzymatic activities.30,41 Understanding the genomic basis of BACE1 expression can help guide the development of effective gene therapies against BACE1.
1.4. Current status of BACE1 therapeutics
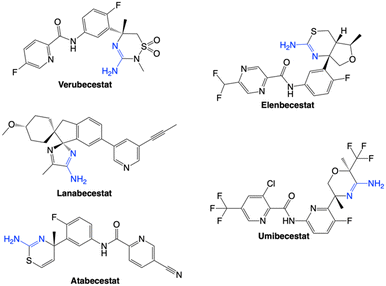
As the BACE1 enzyme plays a critical role in the development of AD, many drugs have been designed to inhibit BACE1 in the hope of reducing Aβ generation and slowing the progression of AD. However, only five of the various BACE1 inhibitors developed have reached phase III clinical trials: verubecestat, lanabecestat, atabecestat, elenbecestat, and umibecestat.5,30 These inhibitors all contain a conserved 2-aminoheterocycle motif embedded into a cyclic scaffold which can interact with the catalytic aspartates (Fig. 5).42 The cyclic scaffold was designed to increase selectivity of BACE1 over other protein targets. All five inhibitors underwent long-term clinical trials and were found to show a significant reduction in Aβ in the cerebrospinal fluid (∼50–85%). Although they showed great reduction in Aβ levels, they failed to provide cognitive or functional benefits, and in some cases, worsened cognitive outcomes.5,30 Lanabecestat and elenbecestat did not improve cognitive or functional abilities while verubecestat, atabecestat and umibecestat were associated with increased adverse effects, worsening cognition, and an overall deterioration of the condition. A potential reason for the adverse effects is the on-target consequence of BACE1 inhibition (i.e., adverse effects due to strong inhibition of BACE1), as observed in of the case of umibecestat, rather than due to off-targets effects.42 Overall, this highlights the complex challenges in creating effective strategies for BACE1 modulation. While BACE1 modulation can help immensely in reducing AD progression, the side targets and level of modulation needs to be considered to design the most effective strategies.This literature review will focus on novel strategies developed in the last 7 years (2018–2024) that aim to inhibit or modulate BACE1 activity through direct or indirect means. The discussion will not include clinical trials as they have been elegantly summarized in other literature reviews.5,6,30,43,44 Instead, this review will examine upcoming strategies in small molecule modulation of BACE1 activity. Novel antibody and gene therapy strategies will also be discussed to showcase new directions in AD research. Firstly, however, a discussion about the BBB and the challenges it poses to designing BACE1 therapeutics will be considered. This is critical as around 98% of small molecule drugs and virtually all large molecule drugs, such as gene drugs and peptides, encounter significant difficulties in penetrating the BBB.45 BACE1 localization in the brain, requires an adequate understanding of the BBB to facilitate BACE1 therapeutic design.
Overall, the following sections will explore the exciting new directions in BACE1 research, highlighting the innovative strategies being developed to modulate BACE1 activity and potentially provide new treatment options for patients with AD.
2. Blood–brain barrier (BBB)
A key consideration in designing therapeutics against BACE1 is to overcome the BBB that controls the entry of material into the brain. As BACE1 is localized within neurons, it is critical for therapeutics to be able to overcome the barrier to access and act on BACE1.462.1. Structure of the BBB
The BBB represents the distinct characteristics of the microvasculature of the central nervous system (CNS). The blood vessels in CNS are continuous non-fenestrated vessels with characteristics that enable it to tightly control the passage of molecules, ions, and cells between the blood and the CNS (Fig. 6).47 BBB transport can occur via two mechanisms: paracellular transport, which involves solutes passing between cells based on concentration gradients, and transcellular transport, which involves molecules passing through cells.47 Lipophilic molecules, in most cases, passively diffuse through specific receptors via the transcellular pathway. On the contrary, hydrophilic molecules such as proteins and peptides require specific transport mechanisms that enter the brain. For example, glucose transporter-1 (GLUT-1) is responsible for glucose uptake, while other specific receptors, such as insulin receptor (IR) and transferrin receptor (TfR) are responsible for the transport of molecules insulin or transferrin, respectively.48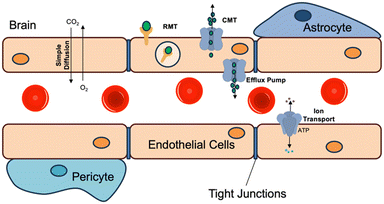 | ||
| Fig. 6 Diagram of a vessel in the brain lined by BBB. The BBB is multicellular composed of various cells such as endothelial cells, pericytes and astrocytes in direct contact with brain tissue. The endothelial cells are connected through tight junctions preventing the movement of molecules between the blood and the brain. As a result, movement of molecules can occur through simple diffusion, receptor mediated transport (RMT), carrier mediated transport (CMT), efflux pump or ion transport through various proteins. This figure has been reproduced from ref. 47 with permission from Cold Spring Harbor Laboratory Press, copyright 2015. | ||
While the characteristics of the BBB are primarily expressed in the endothelial cells (ECs) that create the walls of the blood vessels, they are induced and sustained through vital interactions with various other cell types, such as mural cells, immune cells, glial cells, and neural cells, which work together in the neurovascular unit. The distinctive properties of CNS ECs compared to those of ECs in other tissues enable them to tightly control the passage of molecules. They are connected through tight junctions that greatly restrict the paracellular flux of solutes.49 Moreover, compared to peripheral ECs, CNS ECs exhibit significantly lower rates of transcytosis, which limits the vesicle-mediated transcellular transport of solutes. Due to the tight paracellular and transcellular barrier, ECs are polarized and have distinct abluminal (basolateral) and luminal (apical) surfaces with efflux and influx transporters and receptors that play a critical role in regulating the movement of substances between the blood and brain.47,50
Therefore, the BBB functions as a highly selective interface between the blood and the brain, maintaining an ideal environment for proper CNS function and homeostasis. Without the BBB, the CNS would be vulnerable to harmful toxins, pathogens, ion imbalances, etc., potentially resulting in neuronal damage and dysfunction.47 However, this essential barrier becomes a major hurdle when attempting to deliver various therapeutics to the CNS. For instance, after therapeutic antibodies were administered parenterally, their levels in the brain were found to be approximately 0.01–0.1% of the levels found in the plasma.51 Therefore, a major challenge hindering the success of various therapeutics for AD is the difficulty in crossing the BBB and delivering therapeutics to the CNS.52
2.2. Strategies for improving the transport of therapeutic agents through the BBB
Delivery techniques such as intracranial, intrathecal, or intraventricular injections, as well as chemical disruption of the BBB, may enhance the concentration of certain drugs; however, these methods are invasive and cannot be used in repeated dosing schedules.53 Thus, there are ongoing efforts to increase the effectiveness of delivering therapeutics to the brain with minimal invasion by mitigating the challenges posed by the BBB (Fig. 7).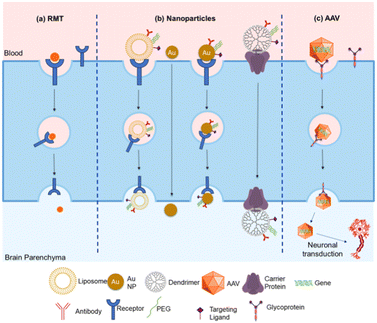 | ||
| Fig. 7 Strategies to transport therapeutic agents across the BBB. (a) In receptor-mediated transcytosis (RMT), a peptide-based molecule binds to a receptor's specific domain on the luminal side of the BBB cells, inducing an endocytic event that transports the vesicles in the cells and delivers the loaded cargo to the brain. (b) Nanoparticle-based strategies use nanoparticles such as liposomes, gold nanoparticles, or dendrimers to transport loaded cargo through the BBB. They can be functionalized with targeting ligands, antibodies, polyethylene glycol (PEG), etc. to enhance their targeting efficiency. These nanoparticles cross the BBB through mechanisms such as RMT, transmembrane diffusion, and carrier-mediated transport. (c) Neurotropic viruses such as Adeno-Associated Virus (AAV) can carry gene-based therapeutics and facilitate their transfer through glyco-protein mediated endocytosis. This figure has been adapted from ref. 69 with permission from MDPI (Basel), copyright 2021. | ||
One such approach is to leverage endogenous processes such as receptor mediated endocytosis, where significant advancements have been made to non-invasively deliver peptide and antibody based biotherapeutics to the brain.54 As mentioned earlier, the BBB endothelium expresses various specific receptors for transporting large molecules like selective peptides, proteins, and other macromolecules. This process can occur via receptor-mediated transcytosis (RMT), which is the primary route for crossing the BBB and entering the brain.55 RMT across the BBB is a multistage endogenous process, that is initiated when specific biomolecules attach to corresponding receptors on brain ECs and trigger endocytosis of the molecule. The internalized vesicles are then transported intracellularly and fused with the abluminal membrane of the BBB, allowing delivery of their contents to the brain parenchyma (Fig. 7A). Therefore, by designing protein therapeutics that target these receptors, RMT can be utilized to deliver treatments to the CNS. To achieve optimal delivery of a therapeutic agent through RMT, it is crucial that the target receptor or carrier protein is highly expressed in the endothelial cells of the BBB. There has been a heavy focus on ubiquitous targets such as TfR, IR, members of the low-density lipoprotein receptor (LDLR) family, and so on.56 For instance, the iron binding protein transferrin (Tf) is the natural receptor for TfR. It was initially observed that upon the binding of Tf to TfR on the luminal surface of the BBB cells, transcytosis was triggered, delivering the Tf to the brain. Later studies discovered that the fusion of a mouse monoclonal antibody (OX26) to rat TfR also initiates transcytosis, leading transport of that antibody into the brain.51 Using a similar approach, bispecific antibodies consisting of anti-Aβ antibody fused with an anti-TfR antibody have been designed to trigger receptor-mediated transport across the BBB via the endogenous TfR expressed on the BBB.57,58 Additionally, there is also a growing amount of research focusing on developing diverse nanotechnology-based platforms to address the BBB challenge for delivering therapeutics to the CNS. Nanoparticles have great potential for use in drug delivery due to their ability to be modified and designed to carry a wide range of molecules, including peptides, proteins, nucleic acids, and antibodies. They can be customized in various shapes and sizes, as well as different levels of hydrophobicity, surface charge, and chemistry, enabling them to facilitate controlled drug release, site-specific targeting, and protecting the drug from specific environmental and biological factors.59 Moreover, they have high biocompatibility, non-toxicity, and prolonged blood circulation, making them ideal drug delivery carriers.60
Nanoparticles are broadly categorized into three categories: lipid-based nanoparticles, polymer-based nanoparticles, and non-polymeric nanoparticles.61 These nanotechnology-based platforms are mostly used in combination with other drug delivery strategies. For instance, poly(lactic-co-glycolic acid) (PLGA) nanoparticles functionalized with anti-TfR antibodies have been shown to enhance delivery of iAβ5, an inhibitor of Aβ aggregate formation.62 Similarly, gold nanoparticles conjugated with monovalent antibodies targeting TfR and BACE1 can effectively cross BBB through TfR mediated transcytosis.63
Another strategy that is being explored for overcoming the challenge of crossing the BBB involves the utilization of neurotropic viruses for gene therapy-based applications.64 As these viruses tend to infect nerve tissues, they have developed diverse techniques for entering the brain. Adeno-associated viral vectors (AAV) and lentiviral vectors (LV) are currently considered the two most beneficial gene therapy vectors for the CNS and have demonstrated successful expression of foreign genes in diverse neural cell types and brain regions.65 For example, using LV expressing siRNA targeting BACE1, lower levels of BACE1 and amyloid production were observed in APP transgenic mice, along with improvements in behavioural and neurodegenerative deficits.66 Viral-based delivery methods offer several advantages such as ability to transfer genes to differentiated cells, expression of the gene for long-term, and relatively low immunogenicity. Nevertheless, viral vectors are not without their challenges as they are prone to issues such as generation of neutralizing antibodies against vectors, lack of cell-specific targeting, and difficulty related to producing high doses on a large scale.67 Similarly, antioxidant strategies, including curcumin and resveratrol, have shown to reduce oxidative stress and Aβ plaques, with nanoparticle systems improving their delivery across the BBB.68
To sum up, various drug delivery strategies are currently being researched with encouraging results to address the challenges posed by the BBB. Given this intricate nature of the BBB, it is crucial thoroughly consider it while designing and delivering different BACE1-targeting therapeutics AD patients.
3. Inhibitors and modulators
The most common approach to therapy against BACE1, is through inhibition of BACE1 using inhibitors and modulators that can act either directly at the active site (also known as orthosteric sites) or at secondary binding sites (also known as allosteric sites).30,70 Inhibitors directly targeting the orthosteric site may provide great clinical results as they can cause the complete inhibition of the enzyme, this can have cascading effects on various biochemical pathways, leading to unfavourable side effects.71 Allosteric modulators offer a more nuanced approach by allowing modulation of the enzyme's activity. Binding to allosteric sites on BACE1 can cause changes in its three-dimensional structure and consequently, its function by either decreasing or enhancing its activity based on whether an agonist or antagonist is bound.8 Small molecule drugs that reduce BACE1 binding and hence, cleavage to APP, are referred to as negative allosteric modulators (NAMs) and are the sought-after design for BACE1 therapy. However, not all allosteric binding sites are suitable targets to induce favourable conformational changes.71 Only a few compounds can effectively induce changes to the active site and affect the enzyme's binding ability. Thus, designing effective allosteric modulators requires identifying the allosteric sites and molecules that can bind to these sites and cause the desired conformational changes in the biomolecule of interest.Moreover, indirect BACE1 modulation can also provide a viable alternative, as BACE1 activity can be reduced without complete inhibition. BACE1 activity and expression is controlled by interactions with various proteins and compounds in the cell, as a result this provides a large selection base to design modulators against.72
The field of BACE1 inhibition has seen significant advancements in the development of novel inhibitors and allosteric modulators. While designing molecules that can fit into the active site and inhibit BACE1 activity is a classic strategy, there is a growing trend towards the development of allosteric modulators that offer a more nuanced approach of reducing enzyme activity through direct or indirect means without complete inhibition.
Earlier research on developing BACE1 inhibitors has concentrated on peptide-based compounds that serve as transition-state mimics.31,42 These compounds were designed based on the amino acid sequences present at the cleavage sites of APP for BACE1. Although these peptidomimetics showed BACE1 inhibitory activity at nanomolar concentrations, they suffered from low bioavailability, high hydrophilicity, large molecular weight, as well as low BBB penetration owing to their low binding affinity for transport proteins. These factors have posed significant barriers to their therapeutic applications as BACE1 inhibitors.73,74 Therefore, BACE1 inhibitors have progressed from substrate-inspired peptidomimetics to more recent non-peptidics with a variety of reactive functional groups that target the catalytic aspartic dyad and have enhanced CNS drug-like properties.75
Moreover, when designing BACE1 inhibitors to reduce Aβ accumulation, it is important to ensure that there are little to no side effects. An important factor to consider is having selectivity for BACE1 over its close homologue, BACE2. Inhibition of BACE2 has been linked to disrupted glucose homeostasis and melanosome morphology that leads to a depigmentation phenotype. Several non-selective BACE1/2 inhibitors have shown similar fur and skin pigment loss in mice or dog models, similar to the findings for BACE2 knockout mice.76–79 Using structure–activity relationships (SARs) of different inhibitors for selectivity of BACE1 over BACE2, Pettus et al.80 modified a previously existing cyclopropylthizaine BACE1 inhibitor (compound 1) to increase BACE1 selectivity, resulting in formation of compound AM-6494 (compound 2; Fig. 8). Compound 1, with two fluorine atoms at C4 and C5 of the phenyl ring in the P1 region demonstrated high BACE1 inhibition potency with IC50 values of 12 nM. However, it had even higher potency for BACE2, with an IC50 value of 1.8 nM. Through molecular modelling, it was found that the S1′ and S3 pockets of BACE1 protein were largely unoccupied by compound 1. The depth of S3 pocket in BACE1 is greater than in BACE2, which is attributed to the greater flexibility of the 10 s loop (Gly69–Gly74) in BACE1 compared to BACE2. Therefore, Pettus et al.80 attempted to optimize this through different structural modifications, resulting in the eventual formation of compound 2, where the IC50 of BACE1 is 0.4 nM while the IC50 of BACE2 is 18.6 nM. These results demonstrate the promising potential of SAR principles in the design of BACE1 modulators. This approach has garnered significant attention and popularity among researchers in the field.81–84
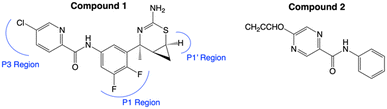 | ||
| Fig. 8 Inhibitors against BACE1, compound 1 indicates various P regions that were modified to reach compound 2, improving BACE1 selective inhibition over BACE2. This figure has been reproduced from ref. 85 with permission from American Chemical Society, copyright 2020. | ||
Research efforts are also being directed towards finding natural compounds with potential to act as BACE1 inhibitors. For instance, Ali et al.86 conducted a study to evaluate the inhibitory effect of flavanone naringenine glycosylated derivatives, didymin, prurin, and poncirin (Fig. 9), against the BACE1 enzyme. Flavonoids and phenolic compounds that are prevalent in plant-derived foods, contain C6–C3–C6 backbone skeletons. Their kinetic data relieved that didymin, prunin, and poncirin are mixed-type inhibitors of BACE1, indicating that these flavonoids have stronger affinity either with the free enzyme or enzyme–substrate complex, or to both the allosteric site of the free enzyme and the enzyme–substrate complex. The IC50 values of didymin and poncirin, both of which contain two disaccharides, were found to be 2.31 ± 0.07 and 3.96 ± 0.09 μM, respectively. On the contrary, a single monosaccharide glucose containing prunin and aglycone naringenin had IC50 values of 13.41 and 38.06 μM, respectively. These results suggest that the inhibitory activity of BACE1 is significantly influenced by the number and position of sugar moieties, as well as the position of the glycosidic linkage. Moreover, the data from their docking studies suggested that hydrogen bonds between the flavonoids and major active site residue of the enzyme are vital for enzyme inhibition.86 Another study examined natural protein hydrolysates from gliadin, whey, and casein prepared with bromelain, papain, and thermolysin. It found that bromelain-hydrolyzed gliadin (G-Bro) showed the strongest BACE1 inhibition with an IC50 of 0.408. In a murine neuroblastoma cell model (N2a/PS/APP), G-Bro reduced BACE1 expression and APP processing, suggesting its potential to reduce Aβ aggregation.87 This shows the notability of natural compounds in BACE1 therapy.
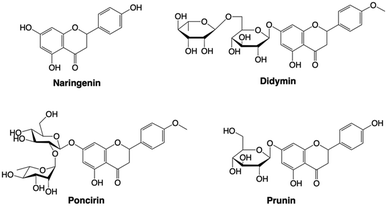 | ||
| Fig. 9 Various flavonoids derivatives utilized for BACE1 inhibition. This figure has been reproduced from ref. 86 with permission from Elsevier, copyright 2019. | ||
Natural compounds offer distinct advantages, including structural complexity and a wide range of scaffold diversity, which contributes to their diverse chemical structures. Furthermore, these compounds may possess favourable properties such as biocompatibility and reduced adverse effects.88,89 Despite the advantages, natural compound were phased out of drug discovery due to complexity in their extraction, characterization and screening. In recent years, however, there has been a resurgence of interest in exploring natural compounds for drug discovery, thanks to significant technological and scientific advancements.89 This renewed focus is also shown for investigation of BACE1 modulators, as various other researchers have also researched natural products for BACE1 modulator design.90–94
As mentioned earlier, a major challenge in developing modulators for AD is crossing the BBB. An innovative strategy being investigated to address this includes the use of chimeric peptides to develop selective BACE1 inhibitors. Resende et al.95 developed BACE1 inhibitors by creating chimeric peptides based on BACE1 substrate, that consisted of a sequence associated with the human Swedish mutant form of APP (APPswe) conjugated with the Tat carrier peptide. They created two chimeric peptide versions: The L and D-retroinverso forms. In the D-retroinverso form, D-amino acids were used, which are resistant to proteases and therefore increase the proteolytic stability of the peptides. As a result, these peptides are expected to be less immunogenic. Furthermore, the Tat carrier peptide facilitates the cell membrane permeation and BBB crossing through the endocytic pathway, which is advantageous as the cargo transported to endosomal compartments. This is particularly useful as BACE1 is predominantly localized in acidic compartments such as the endosome, allowing for selective inhibition of APP-β cleavage while avoiding the proteolysis of other BACE1 substrates. Both the chimeric peptides were able to inhibit the activity of recombinant BACE1 and reduce Aβ production in cellular (Neuro-2a; N2A) and animal (3xTg-AD mice) models of AD. Additionally, in N2A cells, these chimeric peptides did not inhibit processing of other endogenous substrates of BACE1, such as the close homologue of L1 (CHL1) and seizure-related gene 6 (SEZ6). Therefore, these chimeric peptides have the potential to be a promising selective disease-modifying therapy for AD.95
In conclusion, the use of modulators remains a popular and promising strategy in the design of BACE1 therapies. The growing fields of SAR, natural compounds, and chimeric peptides indicate the evolving nature of research in this area, with a focus on addressing crucial aspects such as selectivity, modulation levels, and delivery. These advances are support by technological improvements. Such as the integration of computational methods to aid in screening and BBB penetration assessment adds a new dimension to the development of effective BACE1 modulators.96,97 Overall, these advancements highlight the exciting progress being made towards designing effective BACE1 modulators.
4. Antibody therapy
The use of antibody is a rapidly expanding drug class that offers significant health benefits. Antibody therapy leverages the ability of antibodies to target endogenous proteins with high specificity, triggering the immune system for antibody-mediated clearance.98,99As mentioned previously, although BACE-1 is a promising target for reducing Aβ production, it also has biological functions substrates other than APP such as β-subunit of voltage-gated sodium channels and NRG-1 in myelination, resulting in detrimental effects like impaired synaptic function and hypomyelination.100 For instance, Evans et al.,101 used a monoclonal antibody 2B3, that specifically targets the BACE1 cleavage site of APP. This approach decreased BACE1-mediated APP cleavage through steric hindrance, without influencing other BACE1 substrates. They evaluated the effects of 2B3 administration on associative memory dysfunction in aged transgenic platelet-derived growth factor promotor (PDAPP) mice, which overexpress a mutant human APP and serve as preclinical models for autosomal dominant AD.102 They found that 2B3 administration reduced soluble Aβ40 and β-amyloid derived C-terminal fragment (βCTF) in the hippocampus and improved associated memory dysfunction in aged PDAPP mice, and no change in APP levels (Fig. 10). Furthermore, longitudinal peripheral administration of 2B3 prevented the onset of associative recognition memory impairment and decline in spatial working memory in PDAPP mice.101 Moreover, 2B3 administration also normalized the deficit in NMDA receptor NRDB subunit phosphorylation and subsequent extracellular-signalling. Overproduction of Aβ has been shown to result in changes in the dynamic properties of synaptic plasticity in the hippocampus, hindering the induction of long-term potentiation (LTP) while promoting long-term depression (LTD). This shift in plasticity dynamics is associated with various mechanisms that involve NMDA receptors and calcium signalling.103 SRC kinase Fyn and striatal-tyrosine-enriched phosphatase (STEP) play significant roles in regulating NR2B phosphorylation. Excess Aβ production can activate Fyn kinase, leading to phosphorylation of NR2B at tyrosine 1472 (Y1472) and stabilizing the receptor's expression on the membrane surface. However, this Aβ mediated activation of Fyn kinase is countered by an increase in STEP, a tyrosine phosphatase that dephosphorylates NR2B at Y1472 and deactivates Fyn kinase. This increased expression of NR2B seen with Aβ is believed to ultimately contribute to the loss of synaptic connections. Administration of 2B3 in PDAPP mice reversed the phosphorylation of NMDA-NR2B pY1472 in hippocampal synaptosomes and decreased total STEP levels. Thus, their data suggests that 2B3-mediated changes in NR2B synaptic signalling processes contributed to the improvement in associative recognition memory in aged 2B3 PDAPP mice.101
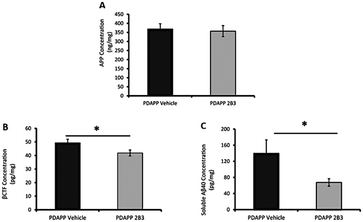 | ||
| Fig. 10 Effect of intracerebroventricular administration of monoclonal antibody 2B3 on reducing Aβ production compared to PDAPP vehicle. (A) no significant changes were observed in APP levels; however, (B) βCTF and (C) soluble Aβ40 levels were significantly reduced. This figure has been adapted from ref. 101 with permission from Elsevier, copyright 2019. | ||
There has also been a growing focus in combining different treatments to increase efficiency of antibody-based therapeutics in reducing Aβ levels. Recognizing that BACE1 has multiple substrates other than APP, He et al.,106 also focused on developing iBesc1, an antibody that would selectively block BACE-1 activity towards the APP cleavage site, while leaving its activity towards other substrates unaffected. However, in addition, He et al.106 further tested another scFv antibody, Asec1a, that was designed to act like α-secretase, enhancing α-secretase cleavage of APP. To further increase the efficiency in addressing the accumulation of Aβ, they developed a bispecific antibody (Diab), that combined both iBsec1 and Asec1a constructs. This Diab antibody effectively increased levels of sAPPα, reduced Aβ deposits, and increased overall neuronal health in a mouse model of AD, APP/PS1.
Although both scFv-Diab and scFv-Bsec decreased Aβ deposits in the cortex and hippocampus of APP/PS1 transgenic mice, only scFv-Diab showed significant improvement in neurogenesis, as measured by tracking doublecortin (DCX) which is a conventional marker of immature neurons. Moreover, compared to mice with scFV-Bsec administration, mice with scFV-Diab showed strongly higher survival rates that were similar to the wildtype.
Thus, they successfully demonstrated that through combining two different antibodies, scFv-Diab, amyloidogenic processing can be reduced through simultaneous inhibition of β-secretase and stimulation of α-secretase activity to promote neuroprotective processing. This led to a higher decrease in plaque formation and inflammation, restoration of neuronal health, and a significant increase in survival rates of the AD mice models, compared to BACE-1 inhibition only.106 Moreover, to address the BBB challenge, He et al.106 genetically added an apolipoprotein B (ApoB), a low-density lipoprotein (LDL), binding domain peptide to the diabody. At the BBB, the LDL receptor (LDLR) would recognize and bind to the ApoB and be transformed to an endosome. This process allows for the transcytosis of the complex to the abluminal side of the BBB, where the ApoB can be released upon a decrease in pH and taken up by neurons and/or astrocytes. Meanwhile, the receptor is returned to the cell surface through recycling.107
Furthermore, the accumulation of Aβ peptides is not only due to the production of Aβ, but due to an imbalance between their production and clearance. Therefore, several approaches being investigated to reduce cerebral Aβ levels include combination of BACE1 inhibition to prevent Aβ production and passive immunotherapy for promoting Aβ clearance. Janssens et al.108 designed BAMB31, a new antibody with a high affinity and specificity against Aβ peptides containing a pyroglutamate residue at the third position (3pE), an Aβ species abundantly found in the plaque deposits of AD brains.
Upon BAMB31 administration, a lower total and 3pE-modified amyloid accumulation was observed in mouse models. According to the authors, this can be attributed to phagocytosis induced by antibodies when they attach to the 3pE-modified Aβ, leading to a clearance of plaques and likely combined with prevention of creation of new aggregates. Since nucleation is crucial for new protein aggregation on the surface of existing amyloid fibrils of the same protein, the clearance of AB deposits through 3pE-specific antibodies is also expected to reduce formation of new aggregates.108 More importantly, combination treatment of the 3pE Aβ specific BAMB31 antibody and a chronic BACE1 inhibitor (BACEi), Atabecestat, was tested. While chronic treatment with BACEi alone maintained the total amyloid levels similar to the baseline, combination treatment with the antibody resulted in a significant lowering of total amyloid levels up to 51%, compared to isotype control animals through clearance of pre-existing deposits (Fig. 11A and B). This demonstrates the enhanced efficacy of combination treatment of BACE inhibitor and anti-Aβ antibody treatment over individual treatments, which has been shown by several other studies as well.109,110
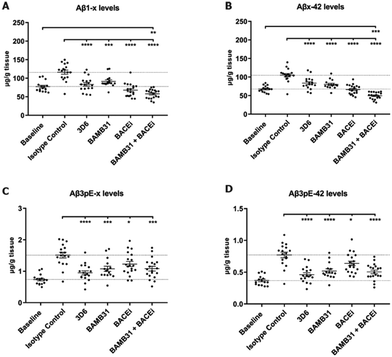 | ||
| Fig. 11 Biochemical analysis showcasing the efficiency of different treatments in reducing levels of (A and B) Total amyloid and (C and D) 3pE-modified amyloid levels in AD model mice brain after chronic treatment. 3D6 is a standard anti-Aβ antibody, BAMB31 is a new anti-3pEAβ antibody, and BACEi is a BACE1 inhibitor. This figure has been adapted from ref. 108 with permission from Elsevier, copyright 2021. | ||
For 3pE-modified amyloid, while BAMB31 led to a significant reduction compared to isotype control group, the combination treatment also only reduced levels to similar extent, with no significant reduction below the baseline observed (Fig. 11C and D), suggesting that further work is needed to enhance the antibody's efficiency for clearing 3pE-modified amyloid. The most effective antibody therapy would ideally achieve plaque elimination without triggering any adverse reactions and this is often reliant on the antibody properties. A recognized possible side effect of passive Aβ immunotherapy is the occurrence of Amyloid-related imaging abnormalities (ARIA) in the brain, which includes microhemorrhages and vasogenic edema.111 Chronic treatment with 3D6, another anti-Aβ antibody, was shown to increase the number of hemosiderin-positive cells in the cortex, meninges, and hippocampus, suggesting a marked increase in microhemorrhages. Several other studies with transgenic mice have also shown a rise in microhemorrhages subsequent to the administration of 3D6.112 Conversely, the administration of BAMB31, BACEi, or the combination therapy did not result in significant development of microhemorrhages in these brain areas, while they did lead to a decrease in amyloid accumulation as mentioned earlier, showcasing enhanced efficacy along with higher safety.108
While novel antibodies against BACE-1 and Aβ are of great interest for clearing Aβ load, several undesired side effects such as meningoencephalitis, angioedema, microhemorrhages, etc. and ineffectiveness of anti-Aβ antibodies are often observed. According to Robert and Wark,104 these side effects are attributed to the fragment crystallizable (Fc) region of the full antibody that intensively activates microglia-mediated immune reactions. Through removal of the Fc region of the antibody, the Fcγ receptors are not activated, avoiding the above-mentioned side effects. Thus, Hu et al.105 focused on developing a novel single-chain variable (scFv) monoclonal anti-Aβ antibodies, eliminating the Fc region, to effectively reduce the Aβ burden. They initially demonstrated that the 31–35 sequence of the Aβ molecule is the shortest active center responsible for Aβ neurotoxicity. Thus, they designed a novel scFv monoclonal anti-Aβ31–35 (scFv17) antibody, which specifically recognized extracellular Aβ and was effective in reducing pathological impairments in APP/PS1 transgenic mice. As compared to 6E10, a commonly used anti-amyloid monoclonal antibody (used as a positive control here), scFv17 showed a similar Aβ-clearing ability and a higher clearing ability of Aβ oligomers. Additionally, scFv17 also resulted in higher anti-inflammatory responses with a significant increase in IL-10 and TGF-β, two major anti-inflammatory cytokines. Moreover, while 6E10 decreased BACE1 levels, scFv17 functioned by significantly enhancing levels of the secreted precursor protein-α (sAPPα), a vital neurotrophin from APP generated by α-secretase, suggesting different modes of functionality for the two antibodies. The authors suggest that this difference is due to the different binding sites of the two antibodies, where 6E10 binds to Aβ1–16 and scFv17 binds to Aβ31–35. Thus, this study highlighted the importance of considering the binding regions when designing antibodies for therapy.105
As mentioned before, despite the advantages of using antibody-based treatments for CNS disorders, their usage is complicated mainly due to the BBB that restricts the access of antibodies to the CNS from systemic circulation. The commonly used approach to overcome BBB for antibody-based delivery relies on RMT. The majority methods for RMT-based delivery methods utilize either a bispecific antibody that has one Fab region that binds to the RMT receptor and another that binds to a therapeutic target, or an antibody that has both Fabs that bind to a therapeutic target and a domain that attaches to the RMT through a peptide linker, added to either the heavy or light chain terminus. However, these methods either eliminate the possibility of bivalent and bispecific targeting of therapeutic agents or require the addition of an artificial fragment to the immunoglobulin G (IgG) scaffold.113 Therefore, using directed evolution, Kariolis et al.53 created a highly modular transport vehicle (TV) to traverse the BBB, that consisted of a human IgG1 Fc fragment designed to bind to the RMT targets expressed on brain endothelial cell surface, instead of depending on the antibody variable domain binding. In this study, the receptor target was TfR, a highly expressed receptor on the brain endothelial cells that experiences constitutive ligand-independent endocytosis. Using recombinant expression of TVs fused with anti-BACE1 Fabs, an antibody transport vehicle (ATV) with native IgF structure and stability was developed.
After administration, TfR-targeted ATV:BACE1 variants demonstrated brain IgG concentrations in rodents that were 10- to 40-fold higher than those of standard anti-BACE1 antibodies 24 to 72 h later. In non-human primates, ATV:BACE1 variants led to IgG concentration in multiple regions of the brain that were over 30-fold higher than those of a standard anti-BACE1 antibody 48 h after administration. Furthermore, a significantly better CNS uptake and sustained pharmacodynamic effect that reduced CNS Aβ production was also observed. Moreover, this TV platform is highly adaptable and can be used to create numerous configurations, including bispecific antibodies and protein fusions, demonstrating a proof-of-concept for making a versatile CNS delivery platform (Fig. 12A and B).53 Kariolis et al.53 further developed a TV configuration fused with Fab fragments for BACE1 and Tau, enabling bispecific targeting. Using biolayer interferometry, they demonstrated the successful binding of this bispecific ATV:BACE1/Tau molecule to hTfR, BACE1, and Tau simultaneously. Moreover, when primary human cortical neurons were treated with ATV:BACE1/Tau, there was a significant decrease of approximately 50% in Aβ levels compared to untreated neurons. However, IgG-treated controls and anti-Tau treated cells retained similar levels of Aβ. This suggests that adding the bispecific ATV:BACE1/Tau did not affect its anti-BACE1 activity (Fig. 12C).
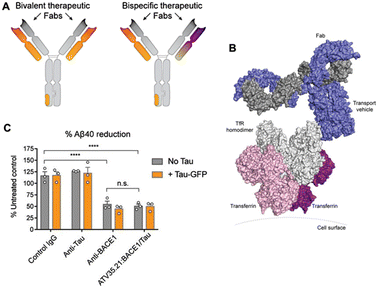 | ||
| Fig. 12 Efficacy of an Fc fragment transport vehicle platform as AD therapeutic. (A) Different possible configurations of transport vehicle platform demonstrating its high modularity. The TV is a transferrin receptor (TfR) binding Fc polypeptide, enabling it to have high versatility for delivering proteins to the brain across the BBB. It can be modified by attaching Fab arms to enable bivalent (ATV:BACE1) or bispecific (ATV:BACE1/Tau) targeting. (B) Surface model of TV with human TfR complex overlaid with human IgG1 and transferrin: TfR complex. (C) Percent reduction in Aβ40 in cultured human neurons expressing hTfR and BACE1 after incubation treatment with ATV35.21:BACE1/Tau. The data suggests that the anti-BACE1 activity of the ATV bispecific was unaffected by addition of Tau. This figure has been adapted from ref. 53 with permission from American Association for the Advancement of Science, copyright 2020. | ||
Another approach to address the BBB challenge, gene-transfer approaches using AAV-based vectors that can cross the BBB to transport antibodies directly to the CNS are being studied.114 However, AAV-based vectors have limited cargo capacity, and therefore, are ideal for delivering single variable domain antibodies (VHHs) as a substitute for monoclonal antibodies.115 While the high selectivity of monoclonal antibodies provides a distinct advantage for targeting key proteins, they can potentially lead to Fc receptor-mediated immunogenicity facilitated by microglia, causing the occurrence of vasogenic edema and cerebral microhemorrhage. The distinct antigen binding domain structure and smaller size of VHHs enables them to access epitopes that are typically not accessible to traditional antibodies. Moreover, due to them lacking the Fc region, they have lower immunogenic profiles compared to traditional antibodies. Marino et al.114 utilized the AAV approach deliver a VHH targeting BACE1, VHH-B9, and found that administering a single systemic dose of AAV-VHH-B9 resulted in a beneficial long-term effect on amyloid accumulation, neuroinflammation, synaptic functioning, and cognitive performance in mouse models of AD.
5. Gene therapy
Gene therapy aims to modify gene expression of the cell to address the underlying disease.116 This can involve the introduction of a new gene to aid in treating the condition, inactivation of a disease-causing gene, or replacement of a faulty gene with a healthy one. Furthermore, these modifications can be targeted at the DNA, RNA, or protein expression levels.AD has been shown to have a genetic basis.117,118 The gene ApoE has been extensively studied and found to play a crucial role in AD. Individuals with the apolipoprotein E4 (ApoE4) isoform have been shown to experience faster disease progression, while those with the ApoE2 isoform exhibit protective effects.119 Having two copies of ApoE4 is also correlated with the highest genetic risk factor for developing AD. In addition to ApoE, the expression of other genes in the neuron can also influence AD progression. For instance, presence of the Swedish mutation in APP has been shown to increase cleavage by BACE1, thereby exacerbating AD progression.117 Given the genetic basis of AD, numerous therapeutic strategies have aimed to silence genes involved in the disease or target their mRNA for degradation at the translational level.
BACE1 gene silencing is of particular interest as silencing the gene's expression can provide greater control over AD progression and greatest therapeutic results. To this end, many strategies have focused on silencing BACE1 transcription. MicroRNAs (miRNAs) are a class of non-coding RNAs that regulate gene expression at the post-transcriptional level (Fig. 13).120 They bind to the 3′ untranslated region (3′ UTR) of target mRNAs, marking them for degradation. They can interact with multiple different targets, resulting in widespread regulation within the cell. Moreover, miRNAs may interact with other regions in the gene or mRNA, thereby controlling transcription and translation processes. This regulatory mechanism helps maintain appropriate cellular protein concentrations. Imbalance of either can lead to widespread effects in the homeostasis of the cell. For instance, alteration of levels of miR-124, a miRNA with regulatory functions in the neuronal cells, can lead to AD.121,122 Patients with AD show a ∼100-fold lower concentration of miR-124 as compared to patients without AD. Previous studies have shown miR-124 to be responsible in regulating BACE by binding to BACE1 5′UTR region.122,123 Restoring miR-124 abundance has shown to slow down plaque progression and hence, AD. Additional papers have reported the effect of miRNA in modulating BACE1.124–126
 | ||
| Fig. 13 BACE1 gene control and expression. Diagram of the pathway of BACE1 gene expression. BACE1 gene (shown in green) undergoes transcription to the BACE1 mRNA, which is translated to the BACE1 protein. BACE1 protein then cleaves the amyloid precursor peptide to C99, followed by further processing to Aβ by γ-secretase, which is release to the extracellular environment where it aggregates (pathway shown in black arrows). Various miRNAs and factors can influence BACE1 transcription and translation to increase (shown in green arrows) or to decrease (shown in red arrows with flat head). This figure has been reproduced from ref. 120 with permission from Cell Press, copyright 2005. | ||
Moreover, BACE1 is also controlled by BACE1 antisense (BACE1-AS), a long noncoding RNA sequence which binds to BACE1 mRNA increasing its stability of the mRNA and as a result increase the cleavage and production of Aβ.127 Previous studies have shown upregulation of BACE1-AS can lead to increased BACE1 mRNA expression.128 HIV infection, and in particular the viral Tat protein, have been shown to induce amyloidosis via BACE1-AS control of BACE1.129 Tat protein causes the upregulation of hypoxia-inducible factor (HIF-1α), which binds to the BACE1-AS leading to its greater stability. This results in further stability of BACE1 mRNA and hence, the accumulation of BACE1 protein and AD progression.130 As a result, BACE1-AS regulation is of great interest for various gene therapy strategies.
Small interfering RNAs (siRNAs) are similar to miRNAs in that they regulate protein expression at the translation level.131 However, unlike miRNA, they are not expressed within mammals and as such are introduced via cellular uptake. Additionally, they provide a few advantages over miRNAs in their greater target specificity and comparably higher stability due to their double-stranded nature.131,132 Due these advantages, siRNA have been approached as great targets for gene therapy. For example, Wang et al.133 designed PEGylated poly(2-(N,N-dimethylamino) ethyl methacrylate) (PEG-PDMAEMA) nanocarriers loaded with siRNA to silence BACE1 mRNA expression. They observed a significant reduction of BACE1 mRNA in transgenic mice, suggesting effective gene silencing. Moreover, the transgenic mice showed a reduction in amyloid plaques, and phosphorylated tau protein levels, while promoting neurogenesis highlighting the potential of siRNA administration as an effective strategy for treating Alzheimer's disease.
Gene therapy using indirect protein control is another strategy for BACE1 regulation. SPONDIN1 (SPON1) is a peptide that is known to bind to BACE1 binding site and hence regulate its cleavage of APP.134 Expression of SPON1 is reduced in AD, and hence, reintroduction of SPON1 can regulate BACE1. Park et al.135 engineered induced neural stem cells (iNSCs) to express SPON1 (iNSC-SPON1) and injected SPON1 protein to show reduced BACE1 activity. Moreover, the authors also used a lentiviral vector to infect cells and introduce the SPON1 into HEK 293T cells expressing the human APP transgene, which showed reduced Aβ generation.
Despite the potential advantages of gene therapies, there are several drawbacks that hinder their development as therapeutics. One significant issue is RNA instability, as RNAs are prone to degradation and have a short half-life.119,136 Additionally, RNAs face the same challenges as other therapeutics in crossing the blood–brain barrier (BBB). Consequently, many approaches aim to overcome these hurdles by improving RNA stability, encapsulation, transport across the BBB, and duration of effect.
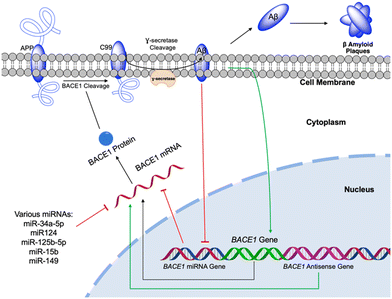
Nanocarrier strategies have shown great potential for gene therapy, as they can encapsulate the gene of interest within a nano-sized carrier that is often decorated with features allowing it to cross the BBB and target specific cells.40 These nanocarriers can increase the stability of the genetic material and be triggered for release once they approach the targeted cell, thereby improving the efficacy of gene therapy. Ouyang et al.122 demonstrated the potential of this strategy using a DNA nanoflower (DF) delivery system to silence BACE1. The DF was loaded with miR-124 chimera and Rutin, a small inhibiting molecule that interacts with BACE1 and can synergize the inhibition effect of miR-124 on BACE1. Additionally, the DF was decorated with rabies virus glycoprotein 29 peptide motif (RVG29), which is a ligand that binds specifically with α7 nicotinic acetylcholine receptor (nAchRs), a receptor highly expressed in the BBB, allowing for penetration and entry across the BBB.137 The DF was synthesized through rolling circular amplification followed by a DNA template containing miR-124 chimeric loading block, Rutin loading block, and RVG29 functional group loading block. Following this, miR-124 chimera, Rutin, and RVG29 were added and allowed to hybridize with the DF. A summary of this fabrication is shown on Fig. 14. The author assessed the pharmokinetics of this strategy and found that compared to drug-free controls, the DF loaded with miR-124 and Rutin showed a greater suppression of BACE1, showing the greatest suppression with both present, thus demonstrating the synergistic potential of both drugs in controlling BACE1 expression.122 Moreover, the DF was found to greatly increase the half-life of the miRNA as compared to free miRNA and showed greater brain targeting and uptake across the BBB as compared to controls, making it a novel and promising strategy for delivering therapeutics across the BBB and controlling BACE1 expression in AD.
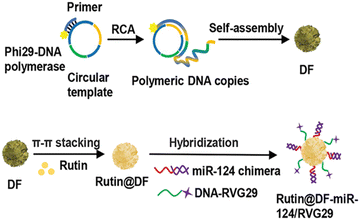 | ||
| Fig. 14 The fabrication of DNA nanoflower delivery vesicles using rolling circle amplification of polymeric DNA. This results in the self-assembly of the DNA into a nanoflower shape, which can then be associated with Rutin using π–π interactions and hybridized with the miR-124 chimera, enabling targeted delivery of the therapeutic cargo. This figure has been adapted from ref. 122 with permission from Wiley-VCH, copyright 2022. | ||
Lv et al.138 showed another strategy using BACE1-AS small hairpin RNA (shRNA) and epigallocatechin-3-gallate (EGCG) loaded into polymeric micelles (Fig. 15). shRNA is similar to siRNA in its function in silencing BACE1 expression; however, it differs in the presence of the hairpin loop which provides greater stability.139 EGCG is a strong antioxidant which has been shown to inhibit other features of AD such as tau and Aβ aggregation.138 The polymeric micelles were fabricated with poly(ethylene glycol) (PEG) PLGA nanoparticles. The PEGylated PLGA provided greater stability for the nanoparticles, allowing for greater circulation. The authors conjugated RVG29 to PEG-PLGA nanoparticles to allow for entry across the BBB via the nAchRs. The authors reported a loading efficiency of 88.5 ± 1.45% for EGCG and almost 100% loading of BACE1-AS shRNA. The pharmokinetic performance and were able to demonstrate greater stability of the micelle as compared to free EGCG, as well as greater accumulation in the brain at 2.5 ± 0.29 ng mg−1 following the encapsulation in nanoparticles 24 hours after intravenous administration. Moreover, when assessing the area under the curve (AUC) obtained from the plot of plasma concentration against time after dosage, information about the clearance rate can be derived as its inversely proportional to the AUC value. In this study, the micelle exhibited a higher AUC value compared to free EGCG, indicating its greater stability and prolonged presence within the system. Furthermore, western blot analysis showed reduced BACE1 expression taken from hippocampus of APP/PS1 transgenic mice. Showing ∼60% reduction from saline control in BACE1 using the nanoparticles. And a ∼50% reduction in Aβ. Overall this strategy presents another novel method to deliver gene therapy against AD.
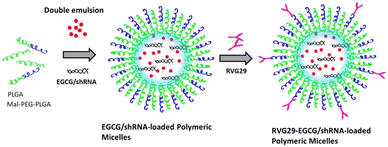 | ||
| Fig. 15 Schematic of loading and preparation of PEG-PLGA NPs. Loading of shRNA gene and epigallocatechin-3-gallate (EGCG) into PLGA/PLGA-PEG polymeric nanoparticles. The nanoparticles were chemically conjugated with RVG29 as a targeting ligand to allow for receptor mediated entry across the blood brain barrier. This figure has been adapted from ref. 138 with permission from Wiley-VCH, copyright 2020. | ||
Micelle-based gene therapy delivery has also been explored by various researchers. Guo et al.140 demonstrated an innovative approach for siRNA delivery by utilizing PEGylated poly(2-(N,N-dimethylamino)ethyl methacrylate) (PEG-PDMAM) decorated with CGN peptide (a BBB targeted peptide) and QSH peptide (an Aβ binding peptide). Using SPR, they showed that QSH-decorated nanoparticles had a binding affinity constant (KD) of 30 times less than that of CGN decorated nanoparticles, indicating greater affinity of the nanoparticles to Aβ binding. Furthermore, combination of QSH and CGN decorated nanoparticles showed a further reduction in the KD, suggesting the presence of CGN does not hinder QSH affinity and a combination of both can lead to higher uptakes. Their approach resulted in increased accumulation of nanoparticles at Aβ sites, leading to more targeted suppression of BACE1 and better clinical outcomes. In vivo studies using APP/PS1 transgenic mice showed that the nanocomplexes successfully down-regulated BACE1 at the mRNA and protein levels and reduced the presence of Aβ.140
Similarly, Zhou et al.141 developed glycosylated polymeric siRNA nanocarriers (Gal-nanoparticles@siRNA) that can cross the BBB via Glut1-mediated transport due to the presence of D-galactose on the nanoparticle surface (Fig. 16). The Gal-nanoparticles@siRNA showcased superior stability over 14 days and lower BACE1 levels in APP/PS1 transgenic AD mouse models, and pharmacokinetic studies showed greater stability of siRNA with a half-life of 39.2 min, which is significantly higher than the 8 min half-life of free siRNA. This study highlights the potential of siRNA-loaded nanocarriers for targeted gene therapy, which has been explored further in various papers.142,143 In a similar study, Jiang et al.144 developed exosome-liposome hybrid nanovesicles that co-deliver BACE1 siRNA and TREM2 plasmid (pTREM2) DNA. These nanovesicles are coated with angiopep-2 (Ang2)-conjugated exosomes, allowing them to cross the BBB. The nanovesicles enhance TREM2 expression, which reprograms microglia from M1 pro-inflammation to M2 anti-inflammation phenotype, thereby improving their ability to clear Aβ deposits and support neural repair. Simultaneously, the BACE1 siRNA reduces BACE1 expression, further decreasing Aβ production. This multifaceted strategy showed in in vivo studies with APP/PS1 mice demonstrated reduced Aβ accumulation, reprogrammed microglia, mitigated neuroinflammation, and improved cognitive function.
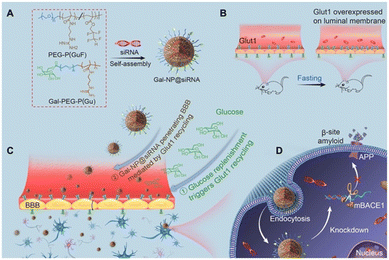 | ||
| Fig. 16 Fabrication method and delivery of glycosylated “triple-interaction” stabilized siRNA nanomedicine (Gal-Nanoparticles@siRNA) and the mechanism to AD pathology in APP/PS1 transgenic mice. (A) Schematic fabrication of Gal-Nanoparticles@siRNA. (B and C) Mechanism of BBB penetration and brain accumulation via Glut1-mediated transport. (D) Gal-Nanoparticles@siRNA-mediated knockdown of BACE1 mRNA. This figure has been adapted from ref. 141 with permission from American Association for the Advancement of Science, copyright 2020. | ||
Nanocarriers offer a potential solution for overcoming the BBB and delivering genes directly to neurons. However, a drawback of this approach is the necessity of pre-loading the particles and maintaining continuous gene delivery for effective long-term therapy.40,136 Nevertheless, with advancements in technology, ongoing efforts are being made to tackle these challenges.
Similarly, viral-based strategies are also amongst the most researched techniques for gene therapy.145 Many of these strategies use lentiviruses to introduce the gene of interest into the genome, allowing for gene and protein expression at various levels. This type of genetic modification offers a permanent cure for genetic disorders as the genes can be continuously read.40,146 However, long-term safety remains an important consideration. Viral vectors may integrate into the host genome, raising concerns regarding insertional mutagenesis and unintended alterations in gene regulation. In addition, viral delivery systems may induce immune responses against the vector or the expressed transgene, which could limit therapeutic durability or lead to inflammatory reactions in the central nervous system.
Zhang et al.147 used an siRNA to knock down BACE1-AS and used a lentivirus to deliver the siRNA in vivo to SAMP8 mice. Western blot analysis showed a significant reduction in BACE1 in the siRNA-treated mice compared to controls. ELISA analysis also showed lower Aβ concentrations compared to controls. Although they successfully demonstrated infection and expression of the siRNA in vitro and in vivo, the long-term effects of delivery were not studied. In addition to this strategy, other strategies have also used lentiviruses for delivery.148,149
Despite its advantages, viral carriers require direct brain delivery such as neurosurgical infusion, making it less favourable due to its invasive nature.40 Less invasive routes, such as intracerebroventricular or intrathecal injections, have been explored for viral delivery, but these have the drawbacks of high off-target effects, immunogenicity, and toxicity concerns. In particular, immune responses triggered by viral capsid proteins or vector components may lead to inflammation, reduced therapeutic efficacy, or the development of neutralizing antibodies that limit repeated administration. Long-term safety is also an important consideration, as viral integration into the host genome may pose risks of insertional mutagenesis or unintended gene activation.
In contrast to viral delivery systems, nanocarrier-based platforms typically avoid genomic integration and therefore present a lower risk of long-term genetic alterations or vector-induced immune responses. Additionally, nanocarriers can be engineered to control release kinetics, improve RNA stability, and enhance targeting across the blood–brain barrier. However, these systems may require repeated administration due to the transient nature of gene expression.
Due to these limitations, viral strategies have become less favoured compared to non-viral strategies in recent years.40 However, due to their ability to integrate the gene into the genome, they remain the most efficient designs for achieving a long-lasting cure. Therefore, despite their current limitations, they have high therapeutic potential.
Remaining challenges & future directions
Based on the strategies discussed above, it is clear that considerable progress has been made in targeting BACE1; however, several key limitations remain that must be addressed to enable successful therapeutic development. Early diagnosis will likely play a critical role in the successful implementation of BACE1-targeted therapies. Amyloid plaque accumulation can begin years, or even decades, before the onset of clinical symptoms, emphasizing the importance of detecting AD at its earliest stages. Advances in diagnostic technologies may enable earlier identification of at-risk individuals and facilitate timely therapeutic intervention.Recent studies have also highlighted the growing role of computational approaches in drug discovery. Varma et al.150 conducted a comprehensive analysis of over 9000 reported BACE1 inhibitors to identify the interaction fingerprints associated with effective binding along with key physicochemical properties.
Zaken et al.151 performed a virtual screening of drugs against 11 protein targets associated with AD. Recent advances in computational drug discovery have significantly improved the efficiency of virtual screening pipelines, enabling the rapid evaluation of large compound libraries against multiple disease-relevant targets. These approaches typically integrate molecular docking, pharmacophore modelling, and molecular dynamics simulations to evaluate ligand–protein interactions and prioritize compounds with favourable binding profiles. In parallel, in silico pharmacokinetic prediction tools, such as ADMET (absorption, distribution, metabolism, excretion, and toxicity) modelling are increasingly used during early stages of drug discovery to assess key properties such as BBB permeability, metabolic stability, and potential toxicity. For AD, ADMET prediction is particularly important because candidate molecules must demonstrate sufficient brain penetration while minimizing systemic toxicity. Computational tools can therefore help eliminate unsuitable compounds early in the screening process, improving the efficiency of lead identification and optimization. With improved virtual screening methodologies and ADMET prediction models, it is possible to identify compounds that may target early pathological events such as the accumulation of amyloid plaques in the brain. These computational approaches complement advances in early diagnostic technologies, including positron emission tomography imaging, biomarker detection, and cognitive assessments. Together, early diagnosis and computationally guided therapeutic discovery may enable earlier intervention strategies aimed at slowing the progression of AD.
Conclusions
BACE1 plays a crucial role in the amyloid cascade and therefore, strategies targeting BACE1 continue to evolve for building an effective therapeutic strategy against AD. Despite this, clinical trials have yet to yield a successful therapeutic against BACE1. BACE1 inhibition has proven to be challenging due to diverse factors such as its involvement in other important physiological functions and crossing the BBB.152,153Moreover, it is uncertain whether BACE1 inhibition would be effective at later stages of AD, where significant amyloid accumulation has already occurred. As BACE1 is not directly involved in Aβ clearance, individuals who are at later stages of AD may find little benefit or use through BACE1. Hence, BACE1 therapeutics might be better suited for early treatment of AD prior to the accumulation of Aβ, or in junction with other therapies which can facilitate the removal of plaques. Thus, the importance of early diagnosis cannot be overstated, especially since the accumulation of amyloid plaques can start years, if not decades, before the onset of clinical symptoms. Taken together, these considerations suggest that BACE1-targeted modulators, antibodies, and gene therapies should be viewed not as symptomatic treatments for established Alzheimer's disease, but as potential components of prevention or very-early-intervention strategies implemented in biomarker-defined at-risk populations.
Despite several barriers, recent progress in BACE1-mediated therapeutic development has generated optimism for potential AD therapies. Nevertheless, determining the appropriate level of BACE1 inhibition and the stage of AD for optimal efficacy remain challenging. Previous clinical trials have yielded limited success, but ongoing studies provide valuable insights into where the field is headed. In conclusion, targeting BACE1 remains a promising avenue for AD therapy, and the field is constantly growing.
Author contributions
Saim Imran: conceptualization, writing draft, visualization; Meera Patel: conceptualization, data curation, investigation, writing draft, visualization; Meissam Noroozifar: conceptualization, data curation, investigation, writing–review & editing, project administration, supervision; Kagan Kerman: conceptualization, funding acquisition, project administration, supervision, writing–review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was supported by the Canada Research Chair Tier-2 award to Dr. K. Kerman for “Bioelectrochemistry of Proteins” (Project no. 950–231116), the Ontario Ministry of Research and Innovation (Project no. 35272), Discovery Grant (Project no. RGPIN-2020–07164) from the Natural Sciences and Engineering Research Council of Canada (NSERC), and the Canada Foundation for Innovation (Project no. 35272).References
- S. Liu, M. Zhao, Y. Liu, X. Yang, H. Yan, H. Xu, Y. Wu and Y. Xu, Front. Neurosci., 2025, 19, 1656906 CrossRef PubMed.
- P. Scheltens, K. Blennow, M. M. B. Breteler, B. de Strooper, G. B. Frisoni, S. Salloway and W. M. van der Flier, Lancet, 2016, 388, 505–517 CrossRef CAS.
- O. Hansson, R. M. Edelmayer, A. L. Boxer, M. C. Carrillo, M. M. Mielke, G. D. Rabinovici, S. Salloway, R. Sperling, H. Zetterberg and C. E. Teunissen, Alzheimer's Dementia, 2022, 18, 2669–2686 CrossRef CAS PubMed.
- C. Lynch, Alzheimer's Dementia, 2020, 16, e038255 CrossRef.
- N. M. Moussa-Pacha, S. M. Abdin, H. A. Omar, H. Alniss and T. H. Al-Tel, Med. Res. Rev., 2020, 40, 339–384 CrossRef PubMed.
- R. Vassar, Alzheimers Res. Ther., 2014, 6, 89 CrossRef PubMed.
- C. Sala Frigerio, L. Wolfs, N. Fattorelli, N. Thrupp, I. Voytyukt, I. Schmidt, R. Mancuso, W.-T. Chen, M. E. Woodbury, G. Srivastava, T. Moller, E. Hudry, S. Das, T. Saido, E. Karran, B. Hyman, V. H. Perry, M. Fiers and B. De Strooper, Cell Rep., 2019, 27, 1293–1306 CrossRef CAS.
- R. Vassar, D. M. Kovacs, R. Yan and P. C. Wong, J. Neurosci., 2009, 29, 12787–12794 CrossRef CAS.
- B. De Strooper, R. Vassar and T. Golde, Nat. Rev. Neurol., 2010, 6, 99–107 CrossRef CAS PubMed.
- M. P. Murphy and H. LeVine III, J. Alzheimers Dis., 2010, 19, 1–17 Search PubMed.
- L. Mucke and D. J. Selkoe, Cold Spring Harbor Perspect. Med., 2012, 2, 1–17 Search PubMed.
- Md. Yousof Ali, S. Zaib, S. Jannat and I. Khan, Bioorg. Chem., 2022, 129, 106137 CrossRef CAS PubMed.
- M. Nistor, M. Don, M. Parekh, F. Sarsoza, M. Goodus, G. E. Lopez, C. Kawas, J. Leverenz, E. Doran, I. T. Lott, M. Hill and E. Head, Neurobiol. Aging, 2007, 28, 1493–1506 CrossRef CAS PubMed.
- R. MacLeod, E.-K. Hillert, R. T. Cameron and G. S. Baillie, Future Sci. OA, 2015, 1, FSO11 CrossRef PubMed.
- D. Bozyczko-Coyne and M. Williams, in Comprehensive Medicinal Chemistry II, ed. J. B. Taylor and D. J. Triggle, Elsevier, Oxford, 2007, pp. 193–228 Search PubMed.
- K. Iqbal, F. Liu, C.-X. Gong and I. Grundke-Iqbal, Curr. Alzheimer Res., 2010, 7, 656–664 CrossRef CAS PubMed.
- J. W. Vogel, Y. Iturria-Medina, O. T. Strandberg, R. Smith, E. Levitis, A. C. Evans and O. Hansson, Nat. Commun., 2020, 11(1), 2612 CrossRef CAS PubMed.
- C. Ballatore, V. M.-Y. Lee and J. Q. Trojanowski, Nat. Rev. Neurosci., 2007, 8, 663–672 CrossRef CAS.
- G. Bahn, J.-S. Park, U. J. Yun, Y. J. Lee, Y. Choi, J. S. Park, S. H. Baek, B. Y. Choi, Y. S. Cho, H. K. Kim, J. Han, J. H. Sul, S.-H. Baik, J. Lim, N. Wakabayashi, S. H. Bae, J.-W. Han, T. V. Arumugam, M. P. Mattson and D.-G. Jo, Proc. Natl. Acad. Sci. U. S. A., 2019, 116, 12516–12523 CrossRef CAS PubMed.
- C. H. van Dyck, C. J. Swanson, P. Aisen, R. J. Bateman, C. Chen, M. Gee, M. Kanekiyo, D. Li, L. Reyderman, S. Cohen, L. Froelich, S. Katayama, M. Sabbagh, B. Vellas, D. Watson, S. Dhadda, M. Irizarry, L. D. Kramer, T. Iwatsubo and N. Engl, J. Med., 2023, 388, 9–21 CAS.
- R. R. Tampi, B. P. Forester and M. Agronin, Drugs Context, 2021, 10 DOI:10.7573/dic.2021-7-3.
- T. M. Rees and S. Brimijoin, Drugs Today (Barc), 2003, 39, 75–83 CrossRef CAS PubMed.
- J. Folch, O. Busquets, M. Ettcheto, E. Sánchez-López, R. D. Castro-Torres, E. Verdaguer, M. L. Garcia, J. Olloquequi, G. Casadesús, C. Beas-Zarate, C. Pelegri, J. Vilaplana, C. Auladell and A. Camins, J. Alzheimers Dis., 2018, 62(3), 1223–1240 CrossRef CAS PubMed.
- M. Vaz and S. Silvestre, Eur. J. Pharmacol., 2020, 887, 173554 CrossRef CAS PubMed.
- M. Shi, F. Chu, F. Zhu and J. Zhu, Front. Aging Neurosci., 2022, 14, 870517 CrossRef CAS PubMed.
- G. C. Alexander, S. Emerson and A. S. Kesselheim, JAMA, 2021, 325, 1717–1718 CrossRef PubMed.
- J. W. Arndt, F. Qian, B. A. Smith, C. Quan, K. P. Kilambi, M. W. Bush, T. Walz, R. B. Pepinsky, T. Bussière, S. Hamann, T. O. Cameron and P. H. Weinreb, Sci. Rep., 2018, 8, 6412 CrossRef PubMed.
- Y. Jin, Q. Du, M. Song, R. Kang, J. Zhou, H. Zhang and Y. Ding, J. Controlled Release, 2024, 375, 346–365 CrossRef CAS PubMed.
- X. Guo, L. Yan, D. Zhang and Y. Zhao, Ageing Res. Rev., 2024, 94, 102192 CrossRef CAS.
- F. H. Bazzari and A. H. Bazzari, Molecules, 2022, 27, 8823 CrossRef CAS PubMed.
- H. Hu, Z. Chen, X. Xu and Y. Xu, ACS Chem. Neurosci., 2019, 10, 880–889 CrossRef CAS PubMed.
- M. Farzan, C. E. Schnitzler, N. Vasilieva, D. Leung and H. Choe, Proc. Natl. Acad. Sci. U. S. A., 2000, 97, 9712–9717 CrossRef CAS PubMed.
- F. Mancini, A. De Simone and V. Andrisano, Anal. Bioanal. Chem., 2011, 400, 1979–1996 CrossRef CAS PubMed.
- R. Yan, Q. Fan, J. Zhou and R. Vassar, Neurosci. Biobehav. Rev., 2016, 65, 326–340 CrossRef CAS PubMed.
- H. Hampel, R. Vassar, B. De Strooper, J. Hardy, M. Willem, N. Singh, J. Zhou, R. Yan, E. Vanmechelen, A. De Vos, R. Nisticò, M. Corbo, B. P. Imbimbo, J. Streffer, I. Voytyuk, M. Timmers, A. A. Tahami Monfared, M. Irizarry, B. Albala, A. Koyama, N. Watanabe, T. Kimura, L. Yarenis, S. Lista, L. Kramer and A. Vergallo, Biol. Psychiatry, 2021, 89, 745–756 CrossRef CAS PubMed.
- J. Fan, X. Wei, H. Dong, Y. Zhang, Y. Zhou, M. Xu and G. Xiao, Crit. Rev. Anal. Chem., 2022, 1–13 Search PubMed.
- L. Zhou, M. L. Fisher, R. D. Cole, T. J. Gould, V. Parikh, P. I. Ortinski and J. R. Turner, Neuropsychopharmacol., 2018, 43, 1343–1354 CrossRef CAS PubMed.
- P. C. May, R. A. Dean, S. L. Lowe, F. Martenyi, S. M. Sheehan, L. N. Boggs, S. A. Monk, B. M. Mathes, D. J. Mergott, B. M. Watson, S. L. Stout, D. E. Timm, E. S. LaBell, C. R. Gonzales, M. Nakano, S. S. Jhee, M. Yen, L. Ereshefsky, T. D. Lindstrom, D. O. Calligaro, P. J. Cocke, D. G. Hall, S. Friedrich, M. Citron and J. E. Audia, J. Neurosci., 2011, 31, 16507–16516 CrossRef CAS PubMed.
- M. T. Heneka, M. P. Kummer, A. Stutz, A. Delekate, S. Schwartz, A. Vieira-Saecker, A. Griep, D. Axt, A. Remus, T.-C. Tzeng, E. Gelpi, A. Halle, M. Korte, E. Latz and D. T. Golenbock, Nature, 2013, 493, 674–678 CrossRef CAS PubMed.
- S. Arora, D. Sharma, B. Layek and J. Singh, Mol. Pharmaceutics, 2021, 18, 4237–4255 CrossRef CAS PubMed.
- F. Mancini, A. De Simone and V. Andrisano, Anal. Bioanal. Chem., 2011, 400, 1979–1996 CrossRef CAS PubMed.
- R. Machauer, R. Lueoend, K. Hurth, S. J. Veenstra, H. Rueeger, M. Voegtle, M. Tintelnot-Blomley, J.-M. Rondeau, L. H. Jacobson, G. Laue, K. Beltz and U. Neumann, J. Med. Chem., 2021, 64, 15262–15279 CrossRef CAS PubMed.
- C. E.-H. Moussa, Expert Opin. Invest. Drugs, 2017, 26, 1131–1136 CrossRef CAS PubMed.
- C.-C. Hsiao, F. Rombouts and H. J. M. Gijsen, Bioorg. Med. Chem. Lett., 2019, 29, 761–777 CrossRef CAS PubMed.
- Q. Ouyang, Y. Meng, W. Zhou, J. Tong, Z. Cheng and Q. Zhu, J. Drug Targeting, 2022, 30, 61–81 CrossRef CAS PubMed.
- W. M. Pardridge, Front. Aging Neurosci., 2020, 11, 373 CrossRef PubMed.
- R. Daneman and A. Prat, Cold Spring Harbor Perspect. Biol., 2015, 7, a020412 CrossRef PubMed.
- Y. Persidsky, S. H. Ramirez, J. Haorah and G. D. Kanmogne, J. Neuroimmune Pharmacol., 2006, 1, 223–236 CrossRef PubMed.
- H. Kadry, B. Noorani and L. Cucullo, Fluids Barriers CNS, 2020, 17, 69 CrossRef PubMed.
- E. Zenaro, G. Piacentino and G. Constantin, Neurobiol. Dis., 2017, 107, 41–56 CrossRef CAS.
- G. C. Terstappen, A. H. Meyer, R. D. Bell and W. Zhang, Nat. Rev. Drug Discovery, 2021, 20, 362–383 CrossRef CAS PubMed.
- W. M. Pardridge, Pharmaceuticals, 2020, 13, 394 CrossRef CAS PubMed.
- M. S. Kariolis, R. C. Wells, J. A. Getz, W. Kwan, C. S. Mahon, R. Tong, D. J. Kim, A. Srivastava, C. Bedard, K. R. Henne, T. Giese, V. A. Assimon, X. Chen, Y. Zhang, H. Solanoy, K. Jenkins, P. E. Sanchez, L. Kane, T. Miyamoto, K. S. Chew, M. E. Pizzo, N. Liang, M. E. K. Calvert, S. L. DeVos, S. Baskaran, S. Hall, Z. K. Sweeney, R. G. Thorne, R. J. Watts, M. S. Dennis, A. P. Silverman and Y. J. Y. Zuchero, Sci. Transl. Med., 2020, 12, eaay1359 CrossRef CAS PubMed.
- V. M. Pulgar, Front. Neurosci., 2019, 12, 1019 CrossRef PubMed.
- J. Barar, M. A. Rafi, M. M. Pourseif and Y. Omidi, BioImpacts, 2016, 6, 225–248 CrossRef CAS PubMed.
- W. Zhang, Q. Y. Liu, A. S. Haqqani, S. Leclerc, Z. Liu, F. Fauteux, E. Baumann, C. E. Delaney, D. Ly, A. T. Star, E. Brunette, C. Sodja, M. Hewitt, J. K. Sandhu and D. B. Stanimirovic, Fluids Barriers CNS, 2020, 17, 47 CrossRef CAS PubMed.
- R. K. Sumbria, E. K.-W. Hui, J. Z. Lu, R. J. Boado and W. M. Pardridge, Mol. Pharmaceutics, 2013, 10, 3507–3513 CrossRef CAS PubMed.
- J. Niewoehner, B. Bohrmann, L. Collin, E. Urich, H. Sade, P. Maier, P. Rueger, J. O. Stracke, W. Lau, A. C. Tissot, H. Loetscher, A. Ghosh and P.-O. Freskgård, Neuron, 2014, 81, 49–60 CrossRef CAS PubMed.
- Z. Yang, Z. W. Liu, R. P. Allaker, P. Reip, J. Oxford, Z. Ahmad and G. Ren, J. R. Soc., Interface, 2010, 7, S411–S422 CrossRef CAS PubMed.
- J. Ahlawat, G. G. Barroso, S. M. Asil, M. Alvarado, I. Armendariz, J. Bernal, X. Carabaza, S. Chavez, P. Cruz, V. Escalante, S. Estorga, D. Fernandez, C. Lozano, M. Marrufo, N. Ahmad, S. Negrete, K. Olvera, X. Parada, B. Portillo, A. Ramirez, R. Ramos, V. Rodriguez, P. Rojas, J. Romero, D. Suarez, G. Urueta, S. Viel and M. Narayan, ACS Omega, 2020, 5, 12583–12595 CrossRef CAS PubMed.
- A. A. Yetisgin, S. Cetinel, M. Zuvin, A. Kosar and O. Kutlu, Molecules, 2020, 25, 2193 CrossRef CAS.
- J. A. Loureiro, B. Gomes, G. Fricker, M. A. N. Coelho, S. Rocha and M. C. Pereira, Colloids Surf., B, 2016, 145, 8–13 CrossRef CAS PubMed.
- K. B. Johnsen, M. Bak, P. J. Kempen, F. Melander, A. Burkhart, M. S. Thomsen, M. S. Nielsen, T. Moos and T. L. Andresen, Theranostics, 2018, 8, 3416–3436 CrossRef CAS PubMed.
- R. Bajracharya, A. C. Caruso, L. J. Vella and R. M. Nisbet, Pharmaceutics, 2021, 13, 2014 CrossRef CAS PubMed.
- H. An, D.-W. Cho, S. E. Lee, Y.-S. Yang, S.-C. Han and C. J. Lee, Exp. Neurobiol., 2016, 25, 48–54 CrossRef PubMed.
- O. Singer, R. A. Marr, E. Rockenstein, L. Crews, N. G. Coufal, F. H. Gage, I. M. Verma and E. Masliah, Nat. Neurosci., 2005, 8, 1343–1349 CrossRef CAS PubMed.
- D. L. Puhl, A. R. D'Amato and R. J. Gilbert, Brain Res. Bull., 2019, 150, 216–230 CrossRef CAS PubMed.
- B. S. Daraban, A. S. Popa and M. S. Stan, Molecules, 2024, 29, 4056 CrossRef CAS PubMed.
- R. Bajracharya, A. C. Caruso, L. J. Vella and R. M. Nisbet, Pharmaceutics, 2021, 13, 2014 CrossRef CAS PubMed.
- S. C. Ugbaja, I. A. Lawal, B. H. Abubakar, A. G. Mushebenge, M. M. Lawal and H. M. Kumalo, Molecules, 2022, 27, 4372 CrossRef CAS.
- R. Nussinov and C.-J. Tsai, Curr. Pharm. Des., 2012, 18, 1311–1316 CrossRef CAS PubMed.
- L. Zhou, M. L. Fisher, R. D. Cole, T. J. Gould, V. Parikh, P. I. Ortinski and J. R. Turner, Neuropsychopharmacol., 2018, 43, 1343–1354 CrossRef CAS.
- A. K. Ghosh, N. Kumaragurubaran, L. Hong, H. Lei, K. A. Hussain, C.-F. Liu, T. Devasamudram, V. Weerasena, R. Turner, G. Koelsch, G. Bilcer and J. Tang, J. Am. Chem. Soc., 2006, 128, 5310–5311 CrossRef CAS PubMed.
- J. E. Kang, J. K. Cho, M. J. Curtis-Long, H. W. Ryu, J. H. Kim, H. J. Kim, H. J. Yuk, D. W. Kim and K. H. Park, Molecules, 2012, 18, 140–153 CrossRef PubMed.
- J. R. M. Coimbra, D. F. F. Marques, S. J. Baptista, C. M. F. Pereira, P. I. Moreira, T. C. P. Dinis, A. E. Santos and J. A. R. Salvador, Front. Chem., 2018, 6, 178 CrossRef PubMed.
- L. Rochin, I. Hurbain, L. Serneels, C. Fort, B. Watt, P. Leblanc, M. S. Marks, B. De Strooper, G. Raposo and G. van Niel, Proc. Natl. Acad. Sci. U. S. A., 2013, 110, 10658–10663 CrossRef CAS PubMed.
- G. Cebers, T. Lejeune, B. Attalla, M. Soderberg, R. C. Alexander, S. Budd Haeberlein, A. R. Kugler, E. W. Ingersoll, S. Platz and C. W. Scott, J. Prev. Alzheimers Dis., 2016, 3, 202–218 CAS.
- U. Neumann, M. Ufer, L. H. Jacobson, M. Rouzade-Dominguez, G. Huledal, C. Kolly, R. M. Lüönd, R. Machauer, S. J. Veenstra, K. Hurth, H. Rueeger, M. Tintelnot-Blomley, M. Staufenbiel, D. R. Shimshek, L. Perrot, W. Frieauff, V. Dubost, H. Schiller, B. Vogg, K. Beltz, A. Avrameas, S. Kretz, N. Pezous, J. Rondeau, N. Beckmann, A. Hartmann, S. Vormfelde, O. J. David, B. Galli, R. Ramos, A. Graf and C. Lopez Lopez, EMBO Mol. Med., 2018, 10, e9316 CrossRef PubMed.
- S. Filser, S. V. Ovsepian, M. Masana, L. Blazquez-Llorca, A. Brandt Elvang, C. Volbracht, M. B. Müller, C. K. E. Jung and J. Herms, Biol. Psychiatry, 2015, 77, 729–739 CrossRef CAS PubMed.
- L. H. Pettus, M. P. Bourbeau, J. Bradley, M. D. Bartberger, K. Chen, D. Hickman, M. Johnson, Q. Liu, J. R. Manning, A. Nanez, A. C. Siegmund, P. H. Wen, D. A. Whittington, J. R. Allen and S. Wood, J. Med. Chem., 2020, 63, 2263–2281 CrossRef CAS PubMed.
- A. Kumar, A. Tiwari and A. Sharma, Curr. Neuropharmacol., 2018, 16, 726–739 CrossRef CAS PubMed.
- M. Yazdani, N. Edraki, R. Badri, M. Khoshneviszadeh, A. Iraji and O. Firuzi, Mol. Diversity, 2020, 24, 641–654 CrossRef CAS PubMed.
- C. Wei, J. Fan, X. Sun, J. Yao, Y. Guo, B. Zhou and Y. Shang, Free Radical Biol. Med., 2020, 150, 96–108 CrossRef CAS PubMed.
- R. Ghobadian, M. Mahdavi, H. Nadri, A. Moradi, N. Edraki, T. Akbarzadeh, M. Sharifzadeh, S. N. A. Bukhari and M. Amini, Eur. J. Med. Chem., 2018, 155, 49–60 CrossRef CAS PubMed.
- L. H. Pettus, M. P. Bourbeau, J. Bradley, M. D. Bartberger, K. Chen, D. Hickman, M. Johnson, Q. Liu, J. R. Manning, A. Nanez, A. C. Siegmund, P. H. Wen, D. A. Whittington, J. R. Allen and S. Wood, J. Med. Chem., 2020, 63, 2263–2281 CrossRef CAS PubMed.
- M. Y. Ali, S. Jannat, N. Edraki, S. Das, W. K. Chang, H. C. Kim, S. K. Park and M. S. Chang, Chem.-Biol. Interact., 2019, 309, 108707 CrossRef CAS PubMed.
- C.-Y. Lin, C.-H. Hsieh, P.-Y. Lai, C.-W. Huang, Y.-H. Chung, S.-M. Huang and K.-C. Hsu, Int. J. Mol. Sci., 2024, 25, 13212 CrossRef CAS.
- K. Dzobo, Compr. Pharmacol., 2022, 408–422 Search PubMed.
- A. G. Atanasov, S. B. Zotchev, V. M. Dirsch and C. T. Supuran, Nat. Rev. Drug Discov., 2021, 20, 200–216 CrossRef CAS PubMed.
- Y. Hou, W. Zhao, H. Yu, F. Zhang, H.-T. Zhang and Y. Zhou, Phytomedicine, 2022, 100, 154056 CrossRef PubMed.
- U. A. More, M. N. Noolvi, D. Kumar and A. Tripathi, Curr. Drug Discovery Technol., 2023, 20, e290323215095 CrossRef CAS.
- S. Das, S. Datta, A. Ghosal, N. R. Chaudhuri, G. Sundaram and S. Basu, Neurosci. Lett., 2023, 792, 136965 CrossRef CAS PubMed.
- M. Naushad, S. S. K. Durairajan, A. K. Bera, S. Senapati and M. Li, Planta Med., 2019, 85, 1316–1325 CrossRef CAS PubMed.
- S. H. Seong, M. T. Ha, B. S. Min, H. A. Jung and J. S. Choi, Life Sci., 2018, 210, 20–28 CrossRef CAS PubMed.
- R. Resende, M. Ferreira-Marques, P. Moreira, J. R. M. Coimbra, S. J. Baptista, C. Isidoro, J. A. R. Salvador, T. C. P. Dinis, C. F. Pereira and A. E. Santos, J. Alzheimers Dis., 2020, 76, 1317–1337 CAS.
- J. Chen, J. Wang, B. Yin, L. Pang, W. Wang and W. Zhu, ACS Chem. Neurosci., 2019, 10, 4303–4318 CrossRef CAS PubMed.
- J. Han, Y. Ji, K. Youn, G. Lim, J. Lee, D. H. Kim and M. Jun, Nutrients, 2019, 11, 2694 CrossRef CAS PubMed.
- M. Arbel and B. Solomon, Trends Immunol., 2007, 28, 511–513 CrossRef CAS PubMed.
- K. Rygiel, Indian J. Pharmacol., 2016, 48, 629–636 CrossRef CAS PubMed.
- M. L. Hemming, J. E. Elias, S. P. Gygi and D. J. Selkoe, PLoS One, 2009, 4, e8477 CrossRef PubMed.
- C. E. Evans, R. S. Thomas, T. J. Freeman, M. Hvoslef-Eide, M. A. Good and E. J. Kidd, Neurobiol. Aging, 2019, 75, 136–149 CrossRef CAS PubMed.
- D. L. Brody and D. M. Holtzman, Exp. Neurol., 2006, 197, 330–340 CrossRef CAS PubMed.
- R. Yan, Q. Fan, J. Zhou and R. Vassar, Neurosci. Biobehav. Rev., 2016, 65, 326–340 CrossRef CAS PubMed.
- R. Robert and K. L. Wark, Arch. Biochem. Biophys., 2012, 526, 132–138 CrossRef CAS PubMed.
- M. Hu, J. Zhang, J. Yang, Y. Cao and J. Qi, J. Mol. Neurosci., 2018, 66, 1–9 CrossRef CAS PubMed.
- P. He, W. Xin, P. Schulz and M. R. Sierks, Mol. Neurobiol., 2019, 56, 7420–7432 CrossRef CAS PubMed.
- Z. Zou, S. Shao, R. Zou, J. Qi, L. Chen, H. Zhang, Q. Shen, Y. Yang, L. Ma, R. Guo, H. Li, H. Tian, P. Li, M. Yu, L. Wang, W. Kong, C. Li, Z. Yu, Y. Huang, L. Chen, Q. Shao, X. Gao, X. Chen, Z. Zhang, J. Yan, X. Shao, R. Pan, L. Xu, J. Fang, L. Zhao, Y. Huang, A. Li, Y. Zhang, W. Huang, K. Tian, M. Hu, L. Xie, L. Wu, Y. Wu, Z. Luo, W. Xiao, S. Ma, J. Wang, K. Huang, S. He, F. Yang, S. Zhou, M. Jia, H. Zhang, H. Lu, X. Wang and J. Tan, Alzheimers Dement., 2019, 5, 717–731 Search PubMed.
- J. Janssens, B. Hermans, M. Vandermeeren, E. Barale-Thomas, M. Borgers, R. Willems, G. Meulders, C. Wintmolders, D. Van Den Bulck, A. Bottelbergs, L. Ver Donck, P. Larsen, D. Moechars, W. Edwards, M. Mercken and B. Van Broeck, Neurobiol. Dis., 2021, 154, 105365 CrossRef CAS PubMed.
- H. Jacobsen, L. Ozmen, A. Caruso, R. Narquizian, H. Hilpert, B. Jacobsen, D. Terwel, A. Tanghe and B. Bohrmann, J. Neurosci., 2014, 34, 11621–11630 CrossRef CAS PubMed.
- R. DeMattos, P. May, M. Racke, J. Hole, J. Tzaferis, F. Liu, C. DeLong, T. Day, Z. Yang, L. Boggs, S. Monk, D. Mergott, Y. Tang, J. Lu, M. Hutton, C. Nordstedt, W. Anderson and P. Iverson, Alzheimer's Dementia, 2014, 10, P149 Search PubMed.
- R. A. Sperling, C. R. Jack, S. E. Black, M. P. Frosch, S. M. Greenberg, B. T. Hyman, P. Scheltens, M. C. Carrillo, W. Thies, M. M. Bednar, R. S. Black, H. R. Brashear, M. Grundman, E. R. Siemers, H. H. Feldman and R. J. Schindler, Alzheimer's Dementia, 2011, 7, 367–385 CrossRef PubMed.
- R. B. DeMattos, J. Lu, Y. Tang, M. M. Racke, C. A. DeLong, J. A. Tzaferis, J. T. Hole, B. M. Forster, P. C. McDonnell, F. Liu, R. D. Kinley, W. H. Jordan and M. L. Hutton, Neuron, 2012, 76, 908–920 CrossRef CAS PubMed.
- A. R. Jones and E. V. Shusta, Pharm. Res., 2007, 24, 1759–1771 CrossRef CAS PubMed.
- M. Marino, L. Zhou, M. Y. Rincon, Z. Callaerts-Vegh, J. Verhaert, J. Wahis, E. Creemers, L. Yshii, K. Wierda, T. Saito, C. Marneffe, I. Voytyuk, Y. Wouters, M. Dewilde, S. I. Duqué, C. Vincke, Y. Levites, T. E. Golde, T. C. Saido, S. Muyldermans, A. Liston, B. De Strooper and M. G. Holt, EMBO Mol. Med., 2022, 14, 201809824 Search PubMed.
- I. Trapani, P. Colella, A. Sommella, C. Iodice, G. Cesi, S. Simone, E. Marrocco, S. Rossi, M. Giunti, A. Palfi, G. J. Farrar, R. Polishchuk and A. Auricchio, EMBO Mol. Med., 2014, 6, 194–211 CrossRef CAS PubMed.
- G. A. R. Gonçalves and R. de M. A. Paiva, Einstein, 2017, 15, 369–375 CrossRef.
- C. M. Karch and A. M. Goate, Biol. Psychiatry, 2015, 77, 43–51 CrossRef CAS PubMed.
- X.-P. Wang and H.-L. Ding, Neurosci. Bull., 2008, 24, 105–109 CrossRef CAS PubMed.
- S. Alves, R. Fol and N. Cartier, Hum. Gene Ther., 2016, 27, 100–107 CrossRef CAS PubMed.
- C. M. Croce and G. A. Calin, Cell, 2005, 122, 6–7 CrossRef CAS PubMed.
- S. Ghafouri-Fard, H. Shoorei, Z. Bahroudi, A. Abak, J. Majidpoor and M. Taheri, Biomed. Pharmacother., 2021, 135, 111198 CrossRef CAS PubMed.
- Q. Ouyang, K. Liu, Q. Zhu, H. Deng, Y. Le, W. Ouyang, X. Yan, W. Zhou and J. Tong, Small, 2022, 18, 2107534 CrossRef CAS PubMed.
- Y.-P. Liang, Q. Liu, G.-H. Xu, J. Zhang, Y. Chen, F.-Z. Hua, C.-Q. Deng and Y.-H. Hu, J. Bioenerg. Biomembr., 2019, 51, 381–392 CrossRef CAS PubMed.
- B.-L. Zhu, Y. Long, W. Luo, Z. Yan, Y.-J. Lai, L.-G. Zhao, W.-H. Zhou, Y.-J. Wang, L.-L. Shen, L. Liu, X.-J. Deng, X.-F. Wang, F. Sun and G.-J. Chen, Brain, 2019, 142, 176–192 CrossRef PubMed.
- X. Tan, Y. Luo, D. Pi, L. Xia, Z. Li and Q. Tu, Curr. Neurovasc. Res., 2020, 17, 86–92 CrossRef CAS PubMed.
- D.-B. Li, J.-L. Liu, W. Wang, X.-M. Luo, X. Zhou, J.-P. Li, X.-L. Cao, X.-H. Long, J.-G. Chen and C. Qin, Front. Aging Neurosci., 2018, 10, 24 CrossRef PubMed.
- A. Sayad, S. Najafi, B. M. Hussen, S. T. Abdullah, A. Movahedpour, M. Taheri and M. Hajiesmaeili, Front. Aging Neurosci., 2022, 14, 853180 CrossRef CAS.
- F. Li, Y. Wang, H. Yang, Y. Xu, X. Zhou, X. Zhang, Z. Xie and J. Bi, BMC Mol. Biol., 2019, 20, 23 CrossRef PubMed.
- S. Sil, G. Hu, K. Liao, F. Niu, S. Callen, P. Periyasamy, H. S. Fox and S. Buch, PLoS Biol., 2020, 18, e3000660 CrossRef CAS PubMed.
- M. A. Faghihi, F. Modarresi, A. M. Khalil, D. E. Wood, B. G. Sahagan, T. E. Morgan, C. E. Finch, G. St. Laurent III, P. J. Kenny and C. Wahlestedt, Nat. Med., 2008, 14, 723–730 CrossRef CAS PubMed.
- K. Gavrilov and W. M. Saltzman, Yale J. Biol. Med., 2012, 85, 187–200 CAS.
- B. Hu, L. Zhong, Y. Weng, L. Peng, Y. Huang, Y. Zhao and X.-J. Liang, Signal Transduction Targeted Ther., 2020, 5, 1–25 Search PubMed.
- P. Wang, X. Zheng, Q. Guo, P. Yang, X. Pang, K. Qian, W. Lu, Q. Zhang and X. Jiang, J. Controlled Release, 2018, 279, 220–233 CrossRef CAS PubMed.
- A. Ho and T. C. Südhof, Proc. Natl. Acad. Sci. U. S. A., 2004, 101, 2548–2553 CrossRef CAS.
- S. Y. Park, J. Y. Kang, T. Lee, D. Nam, C.-J. Jeon and J. B. Kim, Cell, 2020, 9, 1275 CrossRef CAS PubMed.
- B. E. Deverman, B. M. Ravina, K. S. Bankiewicz, S. M. Paul and D. W. Y. Sah, Nat. Rev. Drug Discovery, 2018, 17, 641–659 CrossRef CAS PubMed.
- C. Gong, X. Li, L. Xu and Y.-H. Zhang, Biomaterials, 2012, 33, 3456–3463 CrossRef CAS PubMed.
- L. Lv, F. Yang, H. Li and J. Yuan, IUBMB Life, 2020, 72, 1819–1829 CrossRef CAS.
- D. D. Rao, J. S. Vorhies, N. Senzer and J. Nemunaitis, Adv. Drug Delivery Rev., 2009, 61, 746–759 CrossRef CAS PubMed.
- Q. Guo, X. Zheng, P. Yang, X. Pang, K. Qian, P. Wang, S. Xu, D. Sheng, L. Wang, J. Cao, W. Lu, Q. Zhang and X. Jiang, Acta Pharm. Sin. B, 2019, 9, 590–603 CrossRef CAS PubMed.
- Y. Zhou, F. Zhu, Y. Liu, M. Zheng, Y. Wang, D. Zhang, Y. Anraku, Y. Zou, J. Li, H. Wu, X. Pang, W. Tao, O. Shimoni, A. I. Bush, X. Xue and B. Shi, Sci. Adv., 2020, 6, eabc7031 CrossRef CAS PubMed.
- J. Shen, Z. Lu, J. Wang, Q. Hao, W. Ji, Y. Wu, H. Peng, R. Zhao, J. Yang, Y. Li, Z. Shi and X. Zhang, Adv. Mater., 2021, 33, 2101993 CrossRef CAS.
- P. Wang, X. Zheng, Q. Guo, P. Yang, X. Pang, K. Qian, W. Lu, Q. Zhang and X. Jiang, J. Controlled Release, 2018, 279, 220–233 CrossRef CAS PubMed.
- S. Jiang, G. Cai, Z. Yang, H. Shi, H. Zeng, Q. Ye, Z. Hu and Z. Wang, ACS Nano, 2024, 18, 11753–11768 CrossRef CAS PubMed.
- Lentivirus Gene Engineering Protocols, ed. M. Federico, Humana Press, Totowa, NJ, 2010, vol. 614 Search PubMed.
- C. Lundberg, T. Bjorklund, T. Carlsson, J. Jakobsson, P. Hantraye, N. Deglon and D. Kirik, Curr. Gene Ther., 2008, 8, 461–473 CrossRef CAS PubMed.
- W. Zhang, H. Zhao, Q. Wu, W. Xu and M. Xia, Exp. Ther. Med., 2018, 16, 2080–2086 Search PubMed.
- A. T. Barros-Viegas, V. Carmona, E. Ferreiro, J. Guedes, A. M. Cardoso, P. Cunha, L. Pereira de Almeida, C. Resende de Oliveira, J. Pedro de Magalhães, J. Peça and A. L. Cardoso, Mol. Ther. Nucleic Acids, 2020, 19, 1219–1236 CrossRef CAS PubMed.
- O. Singer, R. A. Marr, E. Rockenstein, L. Crews, N. G. Coufal, F. H. Gage, I. M. Verma and E. Masliah, Nat. Neurosci., 2005, 8, 1343–1349 CrossRef CAS PubMed.
- T. Varma, P. Kamble and P. Garg, Drug Dev. Res., 2025, 86, e70169 CrossRef CAS PubMed.
- K. B. Zaken, S. Mesfin, N. Bloch and A. O. Samson, J. Alzheimers Dis., 2025, 108, 1088–1103 CrossRef.
- S. Lopez, C. Del Percio, G. Forloni, A. Frasca, W. H. Drinkenburg, R. Lizio, G. Noce, R. Ferri, A. Soricelli, F. Stocchi, L. Vacca, R. Bordet, J. C. Richardson, C. Babiloni and on behalf of PharmaCog Consortium, Int. J. Mol. Sci., 2020, 21, 9072 CrossRef CAS PubMed.
- M. Vnencak, M. L. Schölvinck, S. W. Schwarzacher, T. Deller, M. Willem and P. Jedlicka, Brain Struct. Funct., 2019, 224, 1279–1290 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |