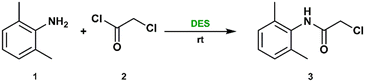Open Access Article
Open Access ArticleIntegrating green synthesis and liquid–liquid extraction of lidocaine in deep eutectic solvents
Assunta
Perri†
a,
Leonardo C.
Parrotta†
a,
Stefania
Gencarelli
a,
Roberta
Sole
a,
Oana M.
Dragostin
b,
Gabriela
Guillena
 c and
Maria L.
Di Gioia
c and
Maria L.
Di Gioia
 *a
*a
aDepartment of Pharmacy, Health and Nutritional Sciences, University of Calabria, 87036 Arcavacata of Rende, Italy. E-mail: ml.digioia@unical.it
bResearch Centre in the Medicinal-Pharmaceutical Field, Faculty of Medicine and Pharmacy, “Dunarea de Jos” University of Galati, 800010 Galati, Romania
cDepartment of Organic Chemistry, Institute of Organic Synthesis (ISO), University of Alicante, 03080 Alicante, Spain
First published on 18th February 2026
Abstract
The development of greener and more sustainable processes for active pharmaceutical ingredients (APIs) remains a major challenge for the chemical industry. Here we present a fully integrated process for the synthesis of lidocaine carried out directly in a deep eutectic solvent, followed by a eutectic-to-eutectic liquid–liquid extraction into a hydrophobic DES for product recovery. The first step of the reaction affords the intermediate α-chloro-2,6-dimethylacetanilide in 92% isolated yield in 20 min; the subsequent SN2 displacement with diethylamine yields lidocaine in excellent yield under mild conditions. To further enhance process sustainability, the crude reaction mixture was subjected to a liquid–liquid extraction using a hydrophobic DES composed of menthol and decanoic acid (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio). This innovative work-up step avoids the use of conventional organic solvents and enables direct transfer of the synthesized lidocaine into a biocompatible eutectic phase already reported for lidocaine-based formulations and delivery systems. GC-MS and FT-IR analyses confirmed exclusive partitioning of lidocaine into the hydrophobic DES. Green metrics (CHEM21) demonstrate a reduced process mass intensity (PMI) and solvent burden compared to classical routes. This study demonstrates how DESs can act simultaneously as reaction media, extractants, and formulation matrices, providing a practical blueprint for sustainable API manufacturing.
1 molar ratio). This innovative work-up step avoids the use of conventional organic solvents and enables direct transfer of the synthesized lidocaine into a biocompatible eutectic phase already reported for lidocaine-based formulations and delivery systems. GC-MS and FT-IR analyses confirmed exclusive partitioning of lidocaine into the hydrophobic DES. Green metrics (CHEM21) demonstrate a reduced process mass intensity (PMI) and solvent burden compared to classical routes. This study demonstrates how DESs can act simultaneously as reaction media, extractants, and formulation matrices, providing a practical blueprint for sustainable API manufacturing.
1. Introduction
The pharmaceutical industry, a cornerstone of global healthcare, is also one of the most resource-intensive sectors, with a significant environmental footprint. The production of Active Pharmaceutical Ingredients (APIs) often involves multistep synthetic routes that generate large quantities of hazardous waste, consume substantial amounts of energy, and rely heavily on toxic solvents.1,2The environmental burden is exacerbated by the high E-factor values (kilograms of waste per kilogram of product) of the industry, which can range from 25 to over 100 for many pharmaceutical synthesis processes. This issue is particularly critical in light of the ongoing global push towards sustainability, with stricter environmental regulations and rising concerns over resource depletion, pollution, and the health impacts of industrial waste.3–8 This model aligns with current EMA and ICH trends toward solvent minimization, process efficiency, and circular pharmaceutical manufacturing, encouraging the replacement of volatile organic compounds with safer, recyclable media. The emergence of green chemistry (GC) has provided a transformative framework to redesign chemical processes, offering more sustainable alternatives that minimize environmental impact.9–11 This approach is particularly relevant for the pharmaceutical industry, where the adoption of benign reaction media, catalytic efficiency, and energy-saving methodologies, leading to cleaner, safer, and more sustainable production routes.12–14 Solvent-related waste, particularly from volatile organic compounds (VOCs), poses a considerable environmental risk, as many of these solvents are toxic, hazardous, and difficult to dispose of.15,16
In recent years, deep eutectic solvents (DESs) have emerged as a promising class of eco-friendly solvents for pharmaceutical synthesis and formulations. DESs are mixtures of a hydrogen-bond acceptor (HBA) and a hydrogen-bond donor (HBD) that form a eutectic system with a melting point far below that of the individual components. Typically composed of natural, renewable compounds such as choline chloride, urea, glycerol, or organic acids, DESs are non-volatile, tunable in polarity, biodegradable and often biocompatible. Their application in pharmaceutical science has expanded rapidly, enabling green synthetic routes, drug solubilization, and novel formulation systems.17–20 For example, natural DESs have been used to enhance the solubility and stability of poorly water-soluble drugs including ibuprofen, dapsone, nifedipine, and curcumin.21–24 Among widely used APIs, lidocaine (2-(diethylamino)-N-(2,6-dimethylphenyl)acetamide; Fig. 1), represents an ideal candidate for developing greener synthetic processes. A potent local anesthetic and antiarrhythmic agent, lidocaine is widely employed in diverse clinical applications, ranging from dental anesthesia to chronic pain management.25–28
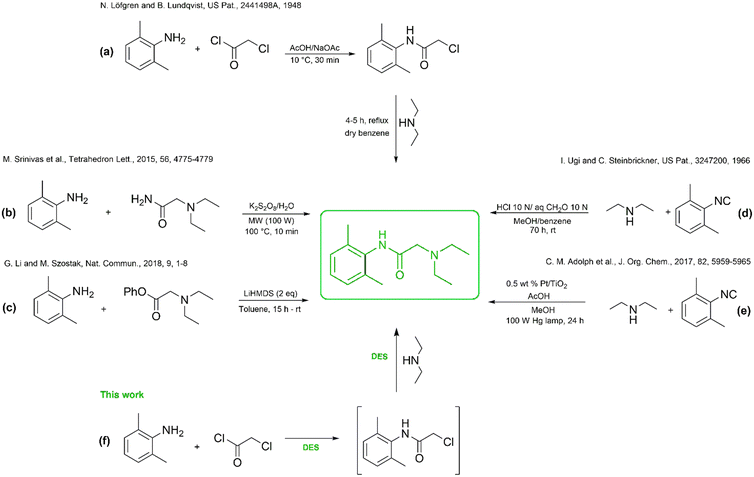
It was first synthesized and patented by Astra in 1948, following a two-step route involving the acylation of 2,6-dimethylaniline with chloroacetyl chloride and subsequent amidation with diethylamine (Scheme 1(a)). This process, however, relies on toxic solvents such as glacial acetic acid and benzene, and involves multi-step workup procedures employing hydrochloric acid and sodium hydroxide.29 Alternative routes, including transamidation (Scheme 1(b)) and amidation-based syntheses (Scheme 1(c)), simplified certain steps but still required chromatographic purification.30,31 The multicomponent Ugi reaction proposed by Ugi et al. (1966) (Scheme 1(d)) offered conceptual novelty yet entailed large solvent volumes (aqueous formaldehyde, benzene) and long reaction times.32 More recently, a Pt/TiO2-catalyzed Ugi-type protocol under UV irradiation (Scheme 1(e)) further improved selectivity but remained solvent-intensive and operationally complex.33 Despite incremental progress, all reported strategies continue to rely on hazardous organic media such as hexane or N-methyl-2-pyrrolidone (NMP), limiting their environmental and industrial viability.34,35
To the best of our knowledge, no DES-mediated syntheses of lidocaine have been reported beyond preliminary solubilization studies, and no integrated eutectic-to-eutectic synthesis–extraction approaches exist in the literature. In contrast, all reported methodologies rely on conventional organic solvents for both reaction and isolation. This gap highlights the opportunity for a continuous, DES-based route that avoids VOCs and chromatographic purification while integrating synthesis and recovery within eutectic media.
Incorporating DESs into lidocaine synthesis offers a compelling opportunity to develop a safer, waste-minimized, and solvent-free process while enhancing reaction performance through hydrogen-bond-assisted activation. Moreover, DESs have demonstrated the ability to improve drug solubility and bioavailability, suggesting that their function can extend beyond synthesis to formulation.36–40
Building on our previous experience in DES-promoted organic transformations,41 the present study aims to bridge the gap between green synthesis and pharmaceutical formulation by designing the first integrated process that combines synthesis and recovery of lidocaine entirely within DES media. The approach couples a hydrophilic choline chloride-urea DES for the two-step synthesis with a hydrophobic menthol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) decanoic acid DES for direct eutectic-to-eutectic extraction. This dual-DES strategy eliminates conventional VOC-based work-up and enables the direct use of the resulting eutectic phase as a potential drug-delivery matrix42 offering a VOC-free reaction and isolation of lidocaine on laboratory scale.
decanoic acid DES for direct eutectic-to-eutectic extraction. This dual-DES strategy eliminates conventional VOC-based work-up and enables the direct use of the resulting eutectic phase as a potential drug-delivery matrix42 offering a VOC-free reaction and isolation of lidocaine on laboratory scale.
2. Results and discussion
2.1 DES screening and optimization of the first step in lidocaine synthesis
The multi-step synthesis of lidocaine is traditionally low-yielding, mainly due to the first step of formation of α-chloro-2,6-dimethylacetanilide (3) which involves the acyl nucleophilic substitution between 2,6-dimethylaniline (1) and chloroacetyl chloride (2). In the patented industrial process, this reaction is typically carried out in glacial acetic acid, a corrosive and environmentally hazardous solvent.29,43 To align the process with green chemistry principles, we investigated deep eutectic solvents (DESs) as alternative, safer media for this transformation (Table 1).| Entrya | DES | Molar ratio | Time (min) | Conversionb (%) | Yieldc (%) |
|---|---|---|---|---|---|
| a Reaction conditions: 2,6-dimethylaniline (1 mmol), chloroacetyl chloride (1 mmol), DES (2 g), 25 °C. b Conversions were determined by GC-MS. c Isolated yields after precipitation induced by water addition, followed by vacuum filtration. | |||||
| 1 |
ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) U U
|
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 2 2
|
20 | 100 | 92 |
| 2 | ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EG EG |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
120 | 35 | 30 |
| 3 | ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Gly Gly |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
20 | 80 | 77 |
| 4 | ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA LA |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
120 | 0 | — |
| 5 |
L-Men![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LA LA |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
120 | 0 | — |
| 6 | ChCl | — | 120 | 0 | — |
A series of choline chloride (ChCl)-based DESs were screened, using common hydrogen-bond donors (HBDs) such as urea (U), ethylene glycol (EG), glycerol (Gly), and lactic acid (LA). Among them, the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) U mixture (1
U mixture (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 molar ratio, also known as reline) afforded the best results, with 95% conversion and 92% isolated yield after only 20 min at room temperature.
2 molar ratio, also known as reline) afforded the best results, with 95% conversion and 92% isolated yield after only 20 min at room temperature.
The addition of a small amount of water at the end of the reaction disrupted the eutectic network, inducing the precipitation of compound 3, which was readily isolated by vacuum filtration. The product identity was confirmed by GC-MS and NMR spectroscopy. The performance of ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) U can be attributed to the strong hydrogen-bond network, which enhances medium polarity and facilitates the nucleophilic attack of the amine on the acyl chloride. The ability of urea to form directional hydrogen bonds with both reactants likely lowers the activation barrier, promoting acylation under mild conditions. Similar catalytic effects of ChCl
U can be attributed to the strong hydrogen-bond network, which enhances medium polarity and facilitates the nucleophilic attack of the amine on the acyl chloride. The ability of urea to form directional hydrogen bonds with both reactants likely lowers the activation barrier, promoting acylation under mild conditions. Similar catalytic effects of ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) U DES have been reported in other amide- and ester-forming reactions.44–47
U DES have been reported in other amide- and ester-forming reactions.44–47
In contrast, polyol-based DESs such as ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethylene glycol (1
ethylene glycol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) and ChCl
2) and ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) glycerol (1
glycerol (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) provided lower conversions, with isolated yields of 30% and 77%, respectively (Table 1, entries 2–3). No product formation was observed when employing acidic DESs such as ChCl
2) provided lower conversions, with isolated yields of 30% and 77%, respectively (Table 1, entries 2–3). No product formation was observed when employing acidic DESs such as ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) lactic acid (1
lactic acid (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and L-menthol
1) and L-menthol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) lactic acid (1
lactic acid (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) (Table 1, entries 4–5), most likely due to protonation of the amine, which suppresses its nucleophilicity and prevents acylation.
1) (Table 1, entries 4–5), most likely due to protonation of the amine, which suppresses its nucleophilicity and prevents acylation.
Literature evidence suggests that the performance differences among DESs arise from the nature of the HBD–HBA hydrogen-bond network: ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) urea exhibits a more integrated and cooperative H-bond network with the choline chloride components compared to ChCl
urea exhibits a more integrated and cooperative H-bond network with the choline chloride components compared to ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) EG and ChCl
EG and ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Gly, where the H-bond interactions largely preserve the bulk structure of the polyol HBD. This more cooperative network in reline can better stabilize reactive intermediates and facilitate nucleophilic substitution processes.48
Gly, where the H-bond interactions largely preserve the bulk structure of the polyol HBD. This more cooperative network in reline can better stabilize reactive intermediates and facilitate nucleophilic substitution processes.48
Finally, to confirm the essential role of the eutectic hydrogen-bonding network, the reaction was performed in pure choline chloride (Table 1, entry 6). Under identical conditions, no reaction occurred, and the starting materials were fully recovered. This control experiment unequivocally demonstrates that the cooperative interactions between the HBD and HBA components in the DES, rather than the presence of the salt alone, are responsible for the observed reactivity enhancement.
Overall, these results highlight the dual role of the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) U (1
U (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) system, acting as both a green solvent and a mild promoter for nucleophilic acyl substitution. The optimized procedure operates efficiently at room temperature, without added catalysts or bases, and avoids the use of volatile organic solvents. The promising results obtained in this step established a robust foundation for extending the DES-mediated approach to the subsequent amine alkylation step.
2) system, acting as both a green solvent and a mild promoter for nucleophilic acyl substitution. The optimized procedure operates efficiently at room temperature, without added catalysts or bases, and avoids the use of volatile organic solvents. The promising results obtained in this step established a robust foundation for extending the DES-mediated approach to the subsequent amine alkylation step.
2.2 Optimization of the second step in lidocaine synthesis
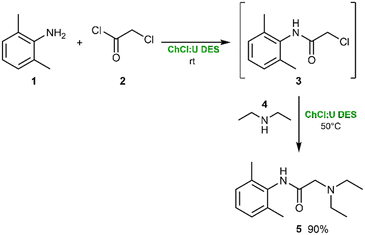
With the efficient formation of the α-chloro-2,6-dimethylacetanilide intermediate 3 established in reline, our attention next focused on the nucleophilic substitution step leading to lidocaine (5).The SN2 displacement of chloride by diethylamine (4) was carried out in the same DES medium without isolation of 3, thus implementing a one-pot process (Scheme 2). Diethylamine acted as both nucleophile and acid scavenger, forming Et2NH2Cl in situ; therefore, a slight excess of amine was initially employed to drive the reaction to completion.
When 1.0 equivalent of diethylamine was added to the reline solution of 3 at room temperature, only trace amounts of lidocaine were detected by GC-MS after 1 h, with unreacted starting material still predominant. Increasing the amount of amine to 2.0 equivalents led to complete conversion within 4 h, affording lidocaine as the sole detectable product. To further reduce the excess amine, the reaction was repeated at 50 °C: under these conditions, full conversion was achieved with only 1.5 equiv. of diethylamine, confirming that a modest increase in temperature effectively compensates for the lower nucleophile concentration (Scheme 2). After reaction completion, an acid–base extraction removed excess amine and residual α-chloro-2,6-dimethylacetanilide, and basification of the aqueous layer afforded the desired lidocaine in 90% isolated yield, without requiring chromatographic purification.
This DES-based protocol markedly differs from traditional methods, which typically require refluxing in organic solvents, use of strong bases and multi-step purification. The ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) U medium enables efficient conversion under mild, metal-free conditions, minimizes waste and eliminates the need for volatile or toxic solvents thus representing a significant advancement in the green synthesis of lidocaine.
U medium enables efficient conversion under mild, metal-free conditions, minimizes waste and eliminates the need for volatile or toxic solvents thus representing a significant advancement in the green synthesis of lidocaine.
The high efficiency of both reaction steps in reline can be rationalized by the cooperative hydrogen-bond network intrinsic to the DES medium. In Scheme 3 we report a plausible mechanistic hypothesis where the cooperative hydrogen-bond network provided by the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) U DES is proposed to facilitate both acylation and SN2 substitution steps. As illustrated, the DES does not merely act as a reaction medium but also exerts a catalytic role by activating the acyl chloride, stabilizing developing charges in the transition state, and promoting nucleophilic attack. The dual activation and solvation effects provided by the DES allow the smooth progression of both transformations under mild, metal-free and operationally simple conditions.
U DES is proposed to facilitate both acylation and SN2 substitution steps. As illustrated, the DES does not merely act as a reaction medium but also exerts a catalytic role by activating the acyl chloride, stabilizing developing charges in the transition state, and promoting nucleophilic attack. The dual activation and solvation effects provided by the DES allow the smooth progression of both transformations under mild, metal-free and operationally simple conditions.
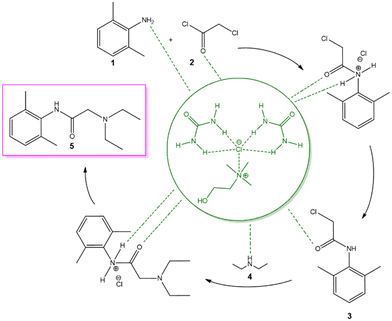 | ||
Scheme 3 Hypothesis of the reaction mechanism in ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) U DES. Dashed lines indicate hydrogen-bonding interactions with DES components. U DES. Dashed lines indicate hydrogen-bonding interactions with DES components. | ||
To evaluate the recyclability of the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) U (1
U (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) DES, the reaction mixture obtained after lidocaine synthesis was extracted with ethyl acetate (3 × 10 mL) to remove the organic products and residual diethylamine. The combined organic phases were washed with water, dried, and evaporated to isolate the crude product. The remaining DES phase was then dried under vacuum (80 °C, <10 mbar) to remove residual volatile components, including traces of ethyl acetate and water. Fresh reagents were subsequently added directly to the same regenerated DES, and the next reaction cycle was carried out under identical conditions. The ChCl
2) DES, the reaction mixture obtained after lidocaine synthesis was extracted with ethyl acetate (3 × 10 mL) to remove the organic products and residual diethylamine. The combined organic phases were washed with water, dried, and evaporated to isolate the crude product. The remaining DES phase was then dried under vacuum (80 °C, <10 mbar) to remove residual volatile components, including traces of ethyl acetate and water. Fresh reagents were subsequently added directly to the same regenerated DES, and the next reaction cycle was carried out under identical conditions. The ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) U DES could be efficiently reused for three consecutive cycles, affording lidocaine in 90%, 88%, 77% yield, respectively, although a decrease in yield was observed after the third cycle.
U DES could be efficiently reused for three consecutive cycles, affording lidocaine in 90%, 88%, 77% yield, respectively, although a decrease in yield was observed after the third cycle.
2.3 Eutectic-to-eutectic liquid–liquid extraction with hydrophobic DES
To further enhance process sustainability and extend the utility of DESs from synthesis to formulation, the recovery of lidocaine was explored through liquid–liquid extraction using a hydrophobic DES (HDES).42 HDES, typically composed of long-chain carboxylic acids combined with natural terpenes or amides, represent an emerging class of environmentally benign media that combine high extraction efficiency with tunable polarity and biocompatibility.49 They have been successfully employed for the selective extraction of bioactive compounds and pharmaceuticals, offering an eco-efficient alternative to conventional volatile organic solvents.50,51Interestingly, lidocaine itself has been reported as a hydrogen-bond acceptor in HDES with menthol, thymol, decanoic acid and dodecanoic acid.52 A recent work by Syed et al.53 demonstrated that the solubility of lidocaine in menthol-based HDES is at least 15 times higher than in water or traditional organic solvents, underscoring the strong affinity of this drug for the hydrophobic eutectic environment.
Building on this knowledge, we hypothesized that the work-up of lidocaine could be simplified by directly extracting the product from the hydrophilic DES reaction medium into a hydrophobic DES, thus achieving solvent-free isolation in a biocompatible eutectic phase.
After replicating our protocol for lidocaine synthesis in ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) U DES, the reaction mixture was diluted with 30% water to break the continuous hydrogen bond network defining the DES structure. The HDES composed of menthol and decanoic acid (1
U DES, the reaction mixture was diluted with 30% water to break the continuous hydrogen bond network defining the DES structure. The HDES composed of menthol and decanoic acid (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio) was then added. The mixture was subjected to vigorous magnetic stirring for 30 minutes at 25 °C. Then, phase separation occurred spontaneously, yielding a hydrophobic top layer and a hydrophilic bottom phase (Fig. 2).
1 molar ratio) was then added. The mixture was subjected to vigorous magnetic stirring for 30 minutes at 25 °C. Then, phase separation occurred spontaneously, yielding a hydrophobic top layer and a hydrophilic bottom phase (Fig. 2).
 | ||
Fig. 2 Schematic representation of the eutectic-to-eutectic liquid–liquid extraction of lidocaine between ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) U and menthol U and menthol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) decanoic acid DES. decanoic acid DES. | ||
NMR and FT-IR analyses confirmed the exclusive presence of lidocaine in the hydrophobic DES (see SI file), demonstrating the efficiency of the eutectic-to-eutectic extraction and its potential as a direct, solvent-free route to API formulation within biocompatible DES.
2.4 Comparative green metrics study
To quantitatively evaluate the sustainability of the developed process, we calculated the key green chemistry metrics recommended by the CHEM21 consortium:54 atom economy (AE), reaction mass efficiency (RME), optimum efficiency (OE), and process mass intensity (PMI), including its derivatives PMIRRC (reactants, reagents, and catalysts) and PMIsolv (solvents). Detailed calculations are provided in the SI.As summarized in Table 2, the DES-mediated synthesis of lidocaine displays significantly improved green metrics compared to both conventional and recently reported procedures. The data reveal that our DES-mediated route exhibits a substantially lower PMI (6.4 g g−1) compared with both the classical (15–16 g g−1) and modern methodologies (up to 816 g g−1). This improvement arises from the dual solvent–catalyst function of ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) U, which eliminates auxiliary reagents, minimizes solvent waste, and enables near-quantitative conversion. Although the atom economy (76%) is inherently limited by the stoichiometric nature of the amide formation, the reaction mass efficiency (61%) and optimum efficiency (80.4%) clearly surpass those of the benchmark processes (Table 2). The reduced PMI_solv (4.8) emphasizes the minimal solvent load, as no volatile organic compounds (VOCs) are used or emitted (Table 2) and employs eutectic mixtures in both reaction and extraction steps. By comparing the values of this process with those of the classical approaches reported, our DES-based systems offer operational simplicity, full conversion under mild conditions, and direct product isolation fulfilling the key CHEM21 criteria for green synthesis.
U, which eliminates auxiliary reagents, minimizes solvent waste, and enables near-quantitative conversion. Although the atom economy (76%) is inherently limited by the stoichiometric nature of the amide formation, the reaction mass efficiency (61%) and optimum efficiency (80.4%) clearly surpass those of the benchmark processes (Table 2). The reduced PMI_solv (4.8) emphasizes the minimal solvent load, as no volatile organic compounds (VOCs) are used or emitted (Table 2) and employs eutectic mixtures in both reaction and extraction steps. By comparing the values of this process with those of the classical approaches reported, our DES-based systems offer operational simplicity, full conversion under mild conditions, and direct product isolation fulfilling the key CHEM21 criteria for green synthesis.
| Entry | AE (%) | RME (%) | OE (%) | PMI (g g−1) | PMIRRC (g g−1) | PMISOLV (g g−1) | Solvents | Catalyst/enzyme | Energy | Work-up | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ideal value | 100 | 100 | 100 | 1 | 1 | 1 |
|
|
|
|
|
| This work | 76 | 61 | 80.4 | 6.4 | 1.6 | 4.8 |
|
|
|
|
|
| Ref. 29 | First step | 84 | 60 | 71.5 | 15.4 | 1.7 | 13.8 |
|

|

|

|
| Second step | 87 | 51 | 58.4 | 6.1 | 2.0 | 4.1 |

|
|

|

|
|
| Ref. 30 | 93 | 89 | 95 | 11.9 | 3 | 9.0 |
|

|
|
|
|
| Ref. 31 | 71 | 65 | 91 | 816.2 | 3.1 | 813.1 |
|

|
|

|
3. Conclusions
This study reports, for the first time, a fully DES-mediated one-pot synthesis of lidocaine, integrating both the reaction and the purification steps within sustainable eutectic media. The process combines the acylation and alkylation reactions in a choline chloride-urea DES with a subsequent reactive liquid–liquid extraction into a menthol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) decanoic acid hydrophobic DES. This dual-DES strategy eliminates conventional solvent-demanding purification procedures, enables direct API transfer into a formulation-compatible medium, and establishes a new paradigm for solvent-integrated green pharmaceutical synthesis at the laboratory scale. Compared to classical lidocaine syntheses (PMI 11.9–816 g g−1), the DES-integrated route achieves a substantially reduced PMI of 6.4 and a PMI_solv of 4.8, reflecting the absence of VOCs and the minimal solvent burden of the eutectic-based system. This improvement underscores the practical environmental benefits of using DESs as dual solvent–catalyst media, fulfilling CHEM21 criteria for reduced solvent usage, operational simplicity and greener work-up. Our approach demonstrates how eutectic systems can simultaneously act as reaction media, separation tools, and formulation platforms, paving the way toward more sustainable and circular pharmaceutical manufacturing. Looking forward, structurally related amide-type local anesthetics such as prilocaine and mepivacaine, that share similar pKa, lipophilicity and solubility characteristics, are expected to benefit from an analogous DES-integrated synthesis–extraction approach, making them ideal candidates for future development.
decanoic acid hydrophobic DES. This dual-DES strategy eliminates conventional solvent-demanding purification procedures, enables direct API transfer into a formulation-compatible medium, and establishes a new paradigm for solvent-integrated green pharmaceutical synthesis at the laboratory scale. Compared to classical lidocaine syntheses (PMI 11.9–816 g g−1), the DES-integrated route achieves a substantially reduced PMI of 6.4 and a PMI_solv of 4.8, reflecting the absence of VOCs and the minimal solvent burden of the eutectic-based system. This improvement underscores the practical environmental benefits of using DESs as dual solvent–catalyst media, fulfilling CHEM21 criteria for reduced solvent usage, operational simplicity and greener work-up. Our approach demonstrates how eutectic systems can simultaneously act as reaction media, separation tools, and formulation platforms, paving the way toward more sustainable and circular pharmaceutical manufacturing. Looking forward, structurally related amide-type local anesthetics such as prilocaine and mepivacaine, that share similar pKa, lipophilicity and solubility characteristics, are expected to benefit from an analogous DES-integrated synthesis–extraction approach, making them ideal candidates for future development.
4. Experimental
4.1 General information
All reagents were purchased from Sigma-Aldrich (Merck KGaA, Darmstadt, Germany) and used as received without further purification, unless otherwise specified. Reactions were monitored by thin-layer chromatography (TLC) on silica gel 60 F254 plates (Sigma-Aldrich) and by gas chromatography-mass spectrometry (GC-MS). GC-MS analyses were performed on a Thermo Scientific Trace 1310 GC system coupled with an ISQ QD single quadrupole mass spectrometer (Thermo Fisher Scientific, Waltham, MA, USA) equipped with a TG-5MS capillary column (30 m × 0.25 mm i.d., 0.25 μm film thickness). Electron impact (EI) ionization at 70 eV was employed, and spectra were recorded in the m/z range 40–650 with relative intensities (%) reported for key fragment ions. 1H NMR spectra were recorded at 500 MHz and 13C NMR spectra at 125 MHz on a Bruker Avance III 500 spectrometer (Bruker BioSpin, Milan, Italy). Chemical shifts (δ) are reported in parts per million (ppm) relative to residual solvent signals, and coupling constants (J) are given in Hertz (Hz). Spectra were acquired at 20 °C in CDCl3 or DMSO-d6, as indicated, using standard Bruker TopSpin acquisition and processing parameters. The HR mass spectra was obtained using the following instrument: Orbitrap Exploris 120 (Thermo Fisher Scientific, San José, CA, USA). The instrument was composed of a heated electrospray ionization (H-ESI II) probe. The mass spectrometer was coupled with a Vanquish system consisting of HPLC pump and autosampler (Thermo Fisher Scientific, San José, CA, USA). The chromatographic separation was achieved employing a C18 reversed-phase column (Hypersil GOLD, 2.6 μm, 100 × 2.1 mm, Thermo Fisher Scientific, San José, CA, USA).4.2 Experimental procedure for DES preparation
The DES used were prepared following literature-reported procedures, using the heating and stirring method.55Before preparing the DES used in this study, choline chloride was dried during 72 h to remove water absorbed from ambient humidity. All DES were prepared by employing choline chloride as the hydrogen bond acceptor (HBA) and urea, glycerol, and ethylene glycol as hydrogen bond donors (HBD) in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 molar ratios, respectively. The components were placed in a heated glass flask at approximately 70–80 °C and stirred using a heating plate equipped with a magnetic stirrer, for 2 h. The obtained DES were used without any further purification. The prepared DES was stored in a desiccator for 24 hours before further use.
2 molar ratios, respectively. The components were placed in a heated glass flask at approximately 70–80 °C and stirred using a heating plate equipped with a magnetic stirrer, for 2 h. The obtained DES were used without any further purification. The prepared DES was stored in a desiccator for 24 hours before further use.
4.3 General procedure for the synthesis of the α-chloro-2,6-dimethylacetanilide intermediate (3) in ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) U DES
U DES
In a 25 mL round-bottom flask equipped with a magnetic stir bar, 2.05 g of choline chloride![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) urea (1
urea (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) DES were introduced and equilibrated at 25 °C. 2,6-Dimethylaniline (0.250 mL, 1.0 equiv.) was added under stirring until complete dissolution. Subsequently, chloroacetyl chloride (0.161 mL, 1.0 equiv.) was added dropwise over 2–3 minutes while maintaining the temperature at 25 °C. The reaction mixture was stirred magnetically for an additional 20 minutes. The reaction progress was monitored by TLC and GC-MS to confirm the complete consumption of 2,6-dimethylaniline. Upon completion, the reaction mixture was diluted with water (5 mL), leading to the immediate formation of a white precipitate corresponding to the desired α-chloro-2,6-dimethylacetanilide (3). The solid was collected by filtration, washed with cold water, and dried under reduced pressure to give the pure product in 92% isolated yield, without any need for further purification.
2) DES were introduced and equilibrated at 25 °C. 2,6-Dimethylaniline (0.250 mL, 1.0 equiv.) was added under stirring until complete dissolution. Subsequently, chloroacetyl chloride (0.161 mL, 1.0 equiv.) was added dropwise over 2–3 minutes while maintaining the temperature at 25 °C. The reaction mixture was stirred magnetically for an additional 20 minutes. The reaction progress was monitored by TLC and GC-MS to confirm the complete consumption of 2,6-dimethylaniline. Upon completion, the reaction mixture was diluted with water (5 mL), leading to the immediate formation of a white precipitate corresponding to the desired α-chloro-2,6-dimethylacetanilide (3). The solid was collected by filtration, washed with cold water, and dried under reduced pressure to give the pure product in 92% isolated yield, without any need for further purification.
4.4 General procedure for the synthesis of lidocaine in ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) U DES
U DES
In a 25 mL round-bottom flask containing 2.05 g of the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) U DES, 0.250 mL (1.0 equiv.) of 2,6-dimethylaniline were added, and the mixture was stirred magnetically at room temperature (25 °C). Chloroacetyl chloride (0.161 mL, 1.0 equiv.) was then added dropwise and the reaction mixture was further stirred at room temperature for 20 minutes. The reaction progress was monitored using TLC or GC-MS to monitor consumption of the aniline. After completion of the acylation (formation of the α-chloro-2,6-dimethylacetanilide intermediate), 0.315 mL (1.5 equiv.) of diethylamine were added to the reaction mixture which was further subjected to magnetic stirring and heated to a 50 °C temperature. The progress of the reaction was monitored by TLC and GC-MS. Upon completion, the reaction mixture was quenched by the addition of water (2 mL) and extracted with ethyl acetate (3 × 2 mL). The combined organic layers were washed with water to remove residual DES components. The aqueous phase obtained after extraction was then adjusted to pH = 4 using citric acid (1 M), leading to the protonation of lidocaine and its transfer into the aqueous layer. The acidic aqueous phase was subsequently basified to pH = 10 with saturated sodium bicarbonate solution, liberating the free-base form of lidocaine, which was then extracted with ethyl acetate (2 × 3 mL). The combined organic extracts were dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure to afford crude lidocaine as a white solid in 90% overall yield and characterized by 1H NMR, 13C NMR spectroscopy and GC-MS, showing full agreement with literature data.
U DES, 0.250 mL (1.0 equiv.) of 2,6-dimethylaniline were added, and the mixture was stirred magnetically at room temperature (25 °C). Chloroacetyl chloride (0.161 mL, 1.0 equiv.) was then added dropwise and the reaction mixture was further stirred at room temperature for 20 minutes. The reaction progress was monitored using TLC or GC-MS to monitor consumption of the aniline. After completion of the acylation (formation of the α-chloro-2,6-dimethylacetanilide intermediate), 0.315 mL (1.5 equiv.) of diethylamine were added to the reaction mixture which was further subjected to magnetic stirring and heated to a 50 °C temperature. The progress of the reaction was monitored by TLC and GC-MS. Upon completion, the reaction mixture was quenched by the addition of water (2 mL) and extracted with ethyl acetate (3 × 2 mL). The combined organic layers were washed with water to remove residual DES components. The aqueous phase obtained after extraction was then adjusted to pH = 4 using citric acid (1 M), leading to the protonation of lidocaine and its transfer into the aqueous layer. The acidic aqueous phase was subsequently basified to pH = 10 with saturated sodium bicarbonate solution, liberating the free-base form of lidocaine, which was then extracted with ethyl acetate (2 × 3 mL). The combined organic extracts were dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure to afford crude lidocaine as a white solid in 90% overall yield and characterized by 1H NMR, 13C NMR spectroscopy and GC-MS, showing full agreement with literature data.
4.5 Preparation of (L-menthol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) decanoic acid) hydrophobic DES
decanoic acid) hydrophobic DES
In a 50 mL round-bottom flask equipped with a magnetic stir bar, L-menthol (1.00 g, 6.53 mmol) and decanoic acid (1.14 g, 6.53 mmol) were combined and heated to 80 °C under stirring until a homogeneous clear liquid formed (60 min). The HDES was cooled to room temperature and stored under dry conditions. 1H and 13C NMR spectra and assignment are reported in the SI file. NMR and FT-IR analysis of the HDES were in accordance with the literature data.56,57
FTIR νmax: 3342, 2940, 2928, 2850, 1690, 1320, 1043 cm−1.
4.6 L–L work-up using L-menthol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) decanoic acid (1
decanoic acid (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) 1) HDES
1) HDES
After confirming reaction completion, the excess of diethylamine was removed by evaporation under reduced pressure. Water was added to the hydrophilic DES to a final water content of approximately 25–30 wt%. Then, the HDES (L-menthol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) decanoic acid, 1
decanoic acid, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) in a volume or mass corresponding to the mass of the hydrophilic DES (2.0 g) was added. The two-phase DES-system was magnetically stirred vigorously for 30 min at 25 °C to promote transfer of organic components into the HDES phase. The HDES (top) layer was then separated using a pipette. Both phases were analyzed by FT-IR and NMR to confirm partitioning of lidocaine into the HDES. Typical recovery in the HDES phase was quantitative within analytical uncertainty.
1) in a volume or mass corresponding to the mass of the hydrophilic DES (2.0 g) was added. The two-phase DES-system was magnetically stirred vigorously for 30 min at 25 °C to promote transfer of organic components into the HDES phase. The HDES (top) layer was then separated using a pipette. Both phases were analyzed by FT-IR and NMR to confirm partitioning of lidocaine into the HDES. Typical recovery in the HDES phase was quantitative within analytical uncertainty.
4.7 Recycling of ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/h3_char_2009.gif) U DES
U DES
Upon completion of the L–L work-up, the ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) U (1
U (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) DES was dried under vacuum (80 °C, <10 mbar) to remove water. Fresh reagents were subsequently added directly to the regenerated DES and the next reaction cycle was carried out under identical conditions. The ChCl
2) DES was dried under vacuum (80 °C, <10 mbar) to remove water. Fresh reagents were subsequently added directly to the regenerated DES and the next reaction cycle was carried out under identical conditions. The ChCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) U DES could be efficiently reused for three consecutive cycles, affording lidocaine in 90%, 88%, 77% yield, respectively, although a decrease in yield was observed after the third cycle.
U DES could be efficiently reused for three consecutive cycles, affording lidocaine in 90%, 88%, 77% yield, respectively, although a decrease in yield was observed after the third cycle.
Author contributions
Assunta Perri and Leonardo C. Parrotta: chemical synthesis; Stefania Gencarelli and Roberta Sole: formal analysis; Oana M. Dragostin: data curation, manuscript preparation; Gabriela Guillena: methodology, manuscript preparation; Maria Luisa Di Gioia: idea generation, review, editing and overall supervision.Conflicts of interest
There are no conflicts to declare.Data availability
All data has been incorporated in supplementary information (SI) file.Supplementary information is available. See DOI: https://doi.org/10.1039/d5md00922g.
Notes and references
- C. Capello, U. Fischer and K. Hungerbühler, Green Chem., 2007, 9, 927–934 RSC.
- S. Ahmad, R. Jaiswal, R. Yadav and S. Verma, Sust. Chem. One World, 2024, 4, 100029 CrossRef.
- D. Ott, D. Kralisch, I. Denčić, V. Hessel, Y. Laribi, P. D. Perrichon, C. Berguerand, L. Kiwi-Minsker and P. Loeb, ChemSusChem, 2014, 7, 3521–3533 Search PubMed.
- F. P. Byrne, S. Jin, G. Paggiola, T. H. Petchey, J. H. Clark, T. J. Farmer, A. J. Hunt, C. R. McElroy and J. Sherwood, Sustainable Chem. Processes, 2016, 4, 7 CrossRef.
- R. A. Sheldon, Green Chem., 2023, 25, 1704–1728 RSC; R. A. Sheldon, ACS Sustainable Chem. Eng., 2018, 6, 32–48 CrossRef CAS.
- O. O. Ogbuagu, A. O. Mbata, O. D. Balogun, O. Oladapo, O. O. Ojo and M. Muonde, IRE Journals, 2024, 8, 761–767 Search PubMed.
- S. Sharma, F. Gallou and S. Handa, Green Chem., 2024, 26, 6289–6317 Search PubMed.
- C. Falcini and G. D. Gonzalo, Catalysts, 2024, 14, 120 CrossRef CAS.
- P. T. Anastas and J. C. Warner, Green chemistry: Theory and Practice, Oxford University Press, New York (USA), 2000 Search PubMed.
- P. Anastas and N. Eghbali, Chem. Soc. Rev., 2010, 39, 301–312 RSC.
- J. B. Zimmerman, P. T. Anastas, H. C. Erythropel and W. Leitner, Science, 2020, 367, 397–400 CrossRef CAS PubMed.
- C. J. Li and B. M. Trost, Proc. Natl. Acad. Sci. U. S. A., 2008, 105, 13197–13202 CrossRef CAS PubMed.
- S. Kar, H. Sanderson, K. Roy, E. Benfenati and J. Leszczynski, Chem. Rev., 2021, 122, 3637–3710 CrossRef PubMed.
- J. H. Clark, T. J. Farmer, A. J. Hunt and J. Sherwood, Int. J. Mol. Sci., 2015, 16, 17101–17159 Search PubMed.
- C. J. Clarke, W. C. Tu, O. Levers, A. Brohl and J. P. Hallett, Chem. Rev., 2018, 118, 747–800 CrossRef CAS PubMed.
- V. Hessel, N. N. Tran, M. R. Asrami, Q. D. Tran, N. V. D. Long, M. Escribà-Gelonch, J. O. Tejada, S. Linke and K. Sundmacher, Green Chem., 2022, 24, 410–437 RSC.
- A. F. Quivelli, F. V. Rossi, P. Vitale, J. Garcia-Alvarez, F. M. Perna and V. Capriati, ACS Sustainable Chem. Eng., 2022, 10, 4065–4072 CrossRef CAS; L. Cicco, A. Yousaf, P. Vitale, F. M. Perna and V. Capriati, RSC Sustainability, 2025, 3, 4079–4086 RSC.
- A. Prabhune and R. Dey, J. Mol. Liq., 2023, 379, 121676 CrossRef CAS; D. J. Ramón and G. Guillena, Deep Eutectic Solvents: Synthesis, Properties, and Applications, John Wiley & Sons, 2020 Search PubMed.
- P. A. Shah, V. Chavda, D. Hirpara, V. S. Sharma, P. S. Shrivastav and S. Kumar, J. Mol. Liq., 2023, 390, 123171 CrossRef CAS.
- E. Chevé-Kools, Y. H. Choi, C. Roullier, G. Ruprich-Robert, R. Grougnet, F. Chapeland-Leclerc and F. Hollmann, Green Chem., 2025, 27, 8360 RSC.
- S. Sangiorgi, B. Albertini, S. Bertoni and N. Passerini, Pharmaceutics, 2025, 17, 300 CrossRef CAS PubMed.
- S. Trombino, C. Siciliano, D. Procopio, F. Curcio, A. S. Laganà, M. L. Di Gioia and R. Cassano, Pharmaceutics, 2022, 14, 333 CrossRef CAS PubMed.
- M. R. Ruesgas Ramon, E. Durand, K. Garcia-Sosa and L. M. Peña-Rodríguez, PeerJ Anal. Chem., 2023, 5, e28 CrossRef.
- N. V. P. Veríssimo, C. U. Mussagy, H. B. S. Bento, J. F. B. Pereira and V. C. Santos-Ebinuma, Biotechnol. Adv., 2024, 71, 108316 CrossRef PubMed.
- M. H. Holmdahl, Acta Anaesthesiol. Scand., 1998, 42, 8–12 CrossRef PubMed.
- J. A. Wildsmith and J. R. Jansson, Eur. J. Anaesthesiol., 2015, 32, 143–146 CrossRef PubMed.
- R. Karnina, S. K. Arif, M. Hatta and A. Bukhari, Ann. Med. Surg., 2021, 69, 102733 Search PubMed.
- F. Hasanpour, M. Budai-Szűcs, A. Kovács, R. Ambrus, O. Jójárt-Laczkovich, M. Cseh, Z. Geretovszky, F. Ayaydin and S. Berkó, Int. J. Pharm., 2024, 660, 124377 CrossRef CAS PubMed.
- N. Löfgren and B. Lundqvist, US Pat., 2441498A, 1948 Search PubMed.
- M. Srinivas, A. D. Hudwekar, V. Venkateswarlu, G. L. Reddy, K. A. A. Kumar, R. A. Vishwakarma and S. D. Sawant, Tetrahedron Lett., 2015, 56, 4775–4779 CrossRef CAS.
- G. Li and M. Szostak, Nat. Commun., 2018, 9, 1–8 Search PubMed.
- I. Ugi and C. Steinbrickner, US Pat., 3247200, 1966 Search PubMed.
- C. M. Adolph, J. Werth, R. Selvaraj, E. C. Wegener and C. Uyeda, J. Org. Chem., 2017, 82, 5959–5965 CrossRef CAS PubMed.
- J. C. M. Monbaliu, T. Stelzer, E. Revalor, N. Weeranoppanant, K. F. Jensen and A. S. Myerson, Org. Process Res. Dev., 2016, 20, 1347–1353 CrossRef CAS.
- A. Adamo, R. L. Beingessner, M. Behnam, J. Chen, T. F. Jamison, K. F. Jensen, J. C. M. Monbaliu, A. S. Myerson, E. M. Revalor and D. R. Snead, Science, 2016, 352, 61–67 CrossRef CAS PubMed.
- L. Cicco, G. Dilauro, F. M. Perna, P. Vitale and V. Capriati, Org. Biomol. Chem., 2021, 19, 2558–2577 RSC.
- S. Emami and A. Shayanfar, Pharm. Dev. Technol., 2020, 25, 779–796 CrossRef CAS PubMed.
- S. Javed, B. Mangla, M. H. Sultan, Y. Almoshari, D. Sivadasan, S. S. Alqahtani, O. A. Madkhali and W. Ahsan, Heliyon, 2024, 10, e29783 CrossRef CAS PubMed.
- F. Oyoun, A. Toncheva, L. Castillo Henríquez, R. Grougnet, F. Laoutid, N. Mignet, K. Alhareth and Y. Corvis, ChemSusChem, 2023, 16, e202300669 CrossRef CAS PubMed.
- S. B. Ganorkar, P. M. Hadole, M. R. Patil, C. V. Pardeshi, P. S. Bobade, A. A. Shirkhedkar and Y. Vander Heyden, Int. J. Pharm., 2025, 672, 125278 CrossRef CAS PubMed.
- D. Procopio, C. Siciliano, A. Perri, G. Guillena, D. J. Ramón and M. L. Di Gioia, Int. J. Mol. Sci., 2024, 25, 6677 CrossRef CAS PubMed; V. Algieri, L. Maiuolo, D. Procopio, P. Costanzo, F. P. Nicoletta, S. Trombino, M. L. Di Gioia and A. De Nino, Polymers, 2024, 16, 757 CrossRef PubMed; D. Procopio, X. Marset, G. Guillena, M. L. Di Gioia and D. J. Ramón, Adv. Synth. Catal., 2024, 366, 870 Search PubMed; D. Procopio, C. Siciliano and M. L. Di Gioia, Org. Biomol. Chem., 2024, 22, 1400–1408 RSC; D. Procopio, C. Siciliano, R. De Rose, S. Trombino, R. Cassano and M. L. Di Gioia, Catalysts, 2022, 12, 1480 CrossRef; M. L. Di Gioia, R. Cassano, P. Costanzo, N. Herrera Cano, L. Maiuolo, M. Nardi, F. P. Nicoletta, M. Oliverio and A. Procopio, Molecules, 2019, 24, 2885 CrossRef PubMed; M. L. Di Gioia, M. Nardi, P. Costanzo, A. De Nino, L. Maiuolo, M. Oliverio and A. Procopio, Molecules, 2018, 23, 1891 CrossRef PubMed.
- A. G. Pekel, E. Kurtulbaş, İ. Toprakçı and S. Şahin, Biomass Convers. Biorefin., 2022, 12, 1249–1256 CrossRef CAS.
- W. Doles, G. Wilkerson, S. Morrison and R. G. Richmond, Hosp. Pharm., 2015, 50, 304–309 CrossRef CAS PubMed.
- H. Tavakol and P. Shafieyoon, J. Mol. Liq., 2025, 428, 127510 CrossRef CAS.
- E. Guzmán, ChemCatChem, 2025, 17, e202500522 CrossRef.
- A. R. Alcántara and G. de Gonzalo, Catalysts, 2025, 15, 586 CrossRef.
- M. S. Álvarez, M. A. Longo, A. Rodríguez and F. J. Deive, J. Ind. Eng. Chem., 2024, 132, 36–49 CrossRef.
- R. Stefanovic, M. Ludwig, G. B. Webber, R. Atkin and A. J. Page, Phys. Chem. Chem. Phys., 2017, 19, 3297–3306 RSC.
- R. Kanzaki, Anal. Sci., 2023, 39, 1021–1022 CrossRef CAS PubMed.
- C. Florindo, L. C. Branco and I. M. Marrucho, ChemSusChem, 2019, 12, 1549–1559 CrossRef CAS PubMed.
- M. Devi, R. Moral, S. Thakuria, A. Mitra and S. Paul, ACS Omega, 2023, 8, 9702–9728 CrossRef CAS PubMed.
- D. J. G. P. van Osch, S. Zubeir, A. van den Bruinhorst, M. A. A. Rocha and M. C. Kroon, Chem. Commun., 2015, 51, 15954–15957 Search PubMed.
- U. T. Syed, J. Calzada, G. Mendoza, M. Arruebo, E. Piacentini, L. Giorno, J. G. Crespo, C. Brazinha and V. Sebastian, ACS Appl. Mater. Interfaces, 2025, 17, 4075–4086 CrossRef CAS PubMed.
- C. R. McElroy, A. Constantinou, L. C. Jones, L. Summerton and J. H. Clark, Green Chem., 2015, 17, 3111–3121 RSC.
- I. M. Aroso, A. Paiva, R. L. Reis and A. R. C. Duarte, J. Mol. Liq., 2017, 241, 654–661 CrossRef CAS.
- D. O. Adeoye, Z. S. Gano, O. U. Ahmed, S. M. Shuwa, A. Y. Atta, S. A. Iwarere, B. Y. Jubril and M. O. Daramola, Chemistry Proceedings, 2023, 14, 98 Search PubMed.
- J. M. Silva, C. V. Pereira, F. Mano, E. Silva, V. I. B. Castro, I. Sá-Nogueira, R. L. Reis, A. Paiva, A. A. Matias and A. R. C. Duarte, ACS Appl. Bio Mater., 2019, 2, 4346–4355 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |