 Open Access Article
Open Access ArticleRelationship between enthalpy fluctuation and nonexponential relaxation in glass-forming liquids
Wataru Takeda and
Pierre Lucas
and
Pierre Lucas *
*
Department of Materials Science and Engineering, University of Arizona, Tucson, AZ 85712, USA. E-mail: Pierre@arizona.edu
First published on 29th April 2026
Abstract
A relationship between the equilibrium fictive temperature fluctuation  and the nonexponentiality βKWW is derived by extending the Adam–Gibbs equation within Donth's fluctuation–dissipation theorem to quantitatively account for dynamic heterogeneity in supercooled liquids. The framework is validated against three structurally, chemically, and kinetically distinct glass-forming systems, namely B2O3, Pd43Cu27Ni10P20, and PVAc, without any adjustable fitting parameters. A condition for time-temperature superposition (TTS) is further derived as a balance between the fictive temperature fluctuation rate and a configurational term composed of the configurational entropy and heat capacity difference between the glass and liquid. The present framework reveals the interplay between thermodynamic quantities, enthalpy fluctuations, and dynamic heterogeneity in glass-forming liquids.
and the nonexponentiality βKWW is derived by extending the Adam–Gibbs equation within Donth's fluctuation–dissipation theorem to quantitatively account for dynamic heterogeneity in supercooled liquids. The framework is validated against three structurally, chemically, and kinetically distinct glass-forming systems, namely B2O3, Pd43Cu27Ni10P20, and PVAc, without any adjustable fitting parameters. A condition for time-temperature superposition (TTS) is further derived as a balance between the fictive temperature fluctuation rate and a configurational term composed of the configurational entropy and heat capacity difference between the glass and liquid. The present framework reveals the interplay between thermodynamic quantities, enthalpy fluctuations, and dynamic heterogeneity in glass-forming liquids.
I. Introduction
Dynamic heterogeneity is a fundamental characteristic of glass-forming liquids.1,2 As they approach the glass transition, distinct spatial regions relax at different rates, giving rise to a broad spectrum of relaxation times and the characteristic nonexponential relaxation response.3 This nonexponential behavior is commonly described by the Kohlrausch–Williams–Watts (KWW) function,4,5 whose stretching exponent βKWW reflects the width of the underlying distribution of relaxation times, where βKWW = 1 corresponds to Debye relaxation and decreasing values of βKWW correspond to an increasingly broader spectrum of relaxation times. Each spatial region may relax exponentially or may have an intrinsic nonexponentiality β0.6 The global relaxation is then understood to be a superposition of local relaxation processes weighted by a distribution of relaxation times X(ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) τ):
τ):
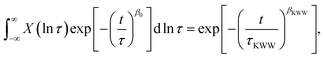 | (1) |
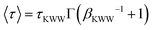 , where Γ is the gamma function. Eqn (1) links βKWW directly to the underlying dynamic heterogeneity: in the homogeneous limit where all subsystems share a single relaxation time, X(ln
, where Γ is the gamma function. Eqn (1) links βKWW directly to the underlying dynamic heterogeneity: in the homogeneous limit where all subsystems share a single relaxation time, X(ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) τ) → δ(ln
τ) → δ(ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) τ − ln
τ − ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) τ0), where δ denotes Dirac's delta function, and the macroscopic exponent reduces to the intrinsic value, βKWW → β0. A key observation is that βKWW is not a material constant but depends on the temperature.7–10
τ0), where δ denotes Dirac's delta function, and the macroscopic exponent reduces to the intrinsic value, βKWW → β0. A key observation is that βKWW is not a material constant but depends on the temperature.7–10
According to Angell, glass-forming liquids can be classified as strong or fragile depending on the temperature dependence of their relaxation time or, equivalently, their viscosity, and can be characterized by the fragility index m, defined as the slope d![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log10
log10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) τ/d(Tg/T) evaluated at the glass transition temperature Tg.11 Fragility has been correlated with several dynamic and thermodynamic properties, including the configurational entropy Sc12 the heat capacity difference between the liquid and glass,13,14 the stretching exponent at Tg βKWW (Tg),15 and the temperature dependence of βKWW.16,17 When βKWW remains constant over a range of temperatures, the relaxation-time spectrum preserves its shape, a condition known as time–temperature superposition (TTS). Dielectric studies17,18 and shear mechanical spectroscopy16 across a wide range of glass-forming liquids have shown a clear trend where more fragile liquids tend to maintain TTS over a broader range of relaxation times. However, no unified thermodynamic framework has been established that quantitatively predicts the conditions under which TTS holds or breaks down.
τ/d(Tg/T) evaluated at the glass transition temperature Tg.11 Fragility has been correlated with several dynamic and thermodynamic properties, including the configurational entropy Sc12 the heat capacity difference between the liquid and glass,13,14 the stretching exponent at Tg βKWW (Tg),15 and the temperature dependence of βKWW.16,17 When βKWW remains constant over a range of temperatures, the relaxation-time spectrum preserves its shape, a condition known as time–temperature superposition (TTS). Dielectric studies17,18 and shear mechanical spectroscopy16 across a wide range of glass-forming liquids have shown a clear trend where more fragile liquids tend to maintain TTS over a broader range of relaxation times. However, no unified thermodynamic framework has been established that quantitatively predicts the conditions under which TTS holds or breaks down.
Understanding the temperature dependence of βKWW requires examining how relaxation time fluctuations arise from thermodynamic fluctuations. As established by Donth's fluctuation-dissipation theorem (DFDT),19,20 the distribution of relaxation times maps onto the fictive temperature fluctuation 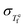 , which, since the equilibrium fictive temperature is proportional to the enthalpy, is fundamentally rooted in the distribution of local enthalpies.21,22 A distribution of enthalpy in supercooled liquids has been observed in both experiments and simulations.22–25 While the connection between enthalpy fluctuations, the distribution of relaxation times, and βKWW is physically well motivated, a quantitative relationship linking the dynamic heterogeneity characterized by nonexponential relaxation to the fictive temperature fluctuation through rigorously defined thermodynamic quantities, such as the configurational entropy and heat capacity, has not yet been developed.
, which, since the equilibrium fictive temperature is proportional to the enthalpy, is fundamentally rooted in the distribution of local enthalpies.21,22 A distribution of enthalpy in supercooled liquids has been observed in both experiments and simulations.22–25 While the connection between enthalpy fluctuations, the distribution of relaxation times, and βKWW is physically well motivated, a quantitative relationship linking the dynamic heterogeneity characterized by nonexponential relaxation to the fictive temperature fluctuation through rigorously defined thermodynamic quantities, such as the configurational entropy and heat capacity, has not yet been developed.
In this work, we expand on a phenomenological model, the Takeda–Lucas (TL) model,26 to develop a thermodynamic framework that connects the Adam–Gibbs relation27 to dynamic fluctuations. While the TL model accounts for both equilibrium and nonequilibrium states, the present generalization focuses on the equilibrium supercooled liquid, where the thermodynamic quantities are well defined. This yields a parameter-free prediction connecting 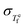 and βKWW, through Sc(T) and ΔCp(T). We validate the framework against three chemically, structurally, and kinetically distinct glass-forming systems: B2O3 (network oxide, m = 3628), Pd43Cu27Ni10P20 (metallic alloy, m = 6929), and PVAc (polymer, m = 9515). Furthermore, we derive a condition for TTS in terms of a balance between the fictive temperature fluctuation rate and a configurational term, revealing the interplay between the thermodynamic quantities Sc(T), ΔCp(T), the enthalpy fluctuation captured by
and βKWW, through Sc(T) and ΔCp(T). We validate the framework against three chemically, structurally, and kinetically distinct glass-forming systems: B2O3 (network oxide, m = 3628), Pd43Cu27Ni10P20 (metallic alloy, m = 6929), and PVAc (polymer, m = 9515). Furthermore, we derive a condition for TTS in terms of a balance between the fictive temperature fluctuation rate and a configurational term, revealing the interplay between the thermodynamic quantities Sc(T), ΔCp(T), the enthalpy fluctuation captured by 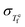 , and the temperature dependence of nonexponentiality in glass-forming liquids.
, and the temperature dependence of nonexponentiality in glass-forming liquids.
This paper is organized as follows. In Section II, we briefly review the TL model of dynamic heterogeneity. In Section III, we present the heterogeneous extension of Adam–Gibbs (AG) theory with DFDT formalism and derive the key equations. In Section IV, we validate the framework against three glass-forming systems. In Section V, we verify the predicted condition for TTS, in Section VI we discuss the implications of the developed framework, and Section VII concludes the paper.
II. TL model of supercooled liquids and glasses
The TL model accounts for both enthalpy fluctuation and temperature-dependent βKWW by introducing a distribution of equilibrium fictive temperatures that maps to a distribution of relaxation times via DFDT.19,26,30,31 The enthalpy fluctuation is modeled by the fictive temperature fluctuation, using the consideration that fictive temperature Tf is proportional to enthalpy H, Tf ∝ H. The model accounts for the heterogeneity of the supercooled liquid by introducing a distribution of equilibrium fictive temperatures Tfe and for the temperature dependence of nonexponentiality by introducing a temperature dependent distribution of relaxation times. This is achieved using Cangialosi et al.'s formulation of dynamic heterogeneity, which relates the distribution X of relaxation times to a distribution Y of Vogel temperatures32 and further relates it to a distribution We of equilibrium fictive temperatures Tfe as:
Xd![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) τ = YdTv = WedTfe, τ = YdTv = WedTfe,
| (2) |
The detailed equations of the TL model (discretized domain weights, relaxation time expressions, and skewness parameters) are provided in ref. 26. The key result relevant to the present work is the connection between the fluctuation of fictive temperatures, i.e. the width of the distribution or the variance of fictive temperatures and the fluctuation of relaxation times through the DFDT formalism:19,26,30,31
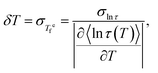 | (3) |
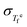 is the fluctuation (variance) of equilibrium fictive temperatures and σln
is the fluctuation (variance) of equilibrium fictive temperatures and σln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) τ is the fluctuation of the relaxation time in equilibrium (see the detailed derivation of eqn (3) in the supplementary information, SI). Furthermore, the relaxation time for each domain i of the distribution is determined by the local Vogel temperature Tv,i using the Adam–Gibbs equation modified by Hodge (AGH)33,34 as
τ is the fluctuation of the relaxation time in equilibrium (see the detailed derivation of eqn (3) in the supplementary information, SI). Furthermore, the relaxation time for each domain i of the distribution is determined by the local Vogel temperature Tv,i using the Adam–Gibbs equation modified by Hodge (AGH)33,34 as
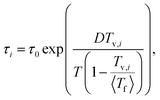 | (4) |
In equilibrium, 〈Tf〉 = T, and eqn (4) adopts the Vogel–Fulcher–Tammann (VFT) form:
 | (5) |
 | (6) |
 | ||
| Fig. 1 Schematics of the temperature dependence of the discrete distribution for (a) the Vogel temperatures, (b) the equilibrium fictive temperatures, and (c) the relaxation time. | ||
Eqn (6) effectively makes the distribution of Tfe temperature-dependent.
Indeed, it is important to recall that the distribution of local fictive temperatures is determined by both the weights Wie and the relative values of Tf,ie. In our framework, the weights Wie are temperature-independent, so the temperature dependence of the distribution width is determined entirely by the relative positions of Tf,ie, which evolve with temperature at rates defined by their respective Tv,i per eqn (6). Hence, the width of the distribution is temperature-dependent (Fig. 1(b)). This accounts for the temperature dependence of the distribution of enthalpy fluctuations.
Finally, a discretized distribution of weight Xi for τi is obtained from the weights Yi of the Vogel temperatures Tv,i according to:
 | (7) |
Consequently, according to eqn (6) and (7), both the shape and the width of the distribution of relaxation times are temperature dependent, which accounts for the temperature dependence of nonexponentiality (Fig. 1(c)).
III. Heterogeneous Adam–Gibbs theory applied to the TL model
The TL model is based on the AGH equation (eqn (4)). This form of the AG equation approximates the configurational entropy Sc by assuming the temperature dependence of the difference in heat capacity between the glass Cpg and the liquid Cpl to be inversely proportional to temperature as:
 | (8) |
 , the configurational entropy Sc, and ΔCp.
, the configurational entropy Sc, and ΔCp.
Let us begin by restating the original AG equation as
 | (9) |
 | (10) |
 | (11) |
A. Enthalpy fluctuation and nonexponentiality
The DFDT framework establishes that fluctuations in relaxation time map to fluctuations in fictive temperature through eqn (3).From eqn (11), the denominator in eqn (3) becomes
 | (12) |
Thus, by inserting eqn (12) into eqn (3), σln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) τ can be expressed in terms of
τ can be expressed in terms of  as
as
 | (13) |
Richert and Richert show that the fluctuation (variance) of relaxation time can be expressed in terms of nonexponential parameter βKWW and intrinsic nonexponentiality β0 as:36
 | (14) |
We can then obtain an expression for βKWW by inserting eqn (13) into eqn (14), and solving for βKWW gives:
 | (15) |
 | (16) |
Thus, eqn (15) relates the dynamic heterogeneity βKWW, the fictive temperature fluctuation  , and the configurational entropy Sc(T), all of which are experimentally accessible. In most cases, the absolute value of Sc is not experimentally available, thus, one may use the approximation Sc ≈ Sex, where Sex is the excess entropy obtained by integrating ΔCp(T)/T.
, and the configurational entropy Sc(T), all of which are experimentally accessible. In most cases, the absolute value of Sc is not experimentally available, thus, one may use the approximation Sc ≈ Sex, where Sex is the excess entropy obtained by integrating ΔCp(T)/T.
B. A condition for time-temperature-superposition (TTS)
One of the hallmark questions in glass science is in which systems, and over what temperature ranges, does time–temperature superposition (TTS) apply in glasses and glass-forming liquids. Based on eqn (15), we can determine the conditions under which βKWW increases, remains constant, or decreases with increasing temperature by taking the temperature derivative, as follows:
 | (17) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/i_char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Λ/dT = 0, and solving for the equilibrium fluctuation term in eqn (16) as:
Λ/dT = 0, and solving for the equilibrium fluctuation term in eqn (16) as:
 | (18) |
We introduce two dimensionless parameters: the fictive-temperature fluctuation rate R(T), and configurational term Ξ(T), defined as
 | (19) |
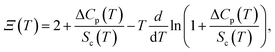 | (20) |
| Condition | Temperature dependence of βKWW |
|---|---|
| R(T) < Ξ(T) | βKWW increases upon heating |
| R(T) = Ξ(T) | βKWW is constant (TTS condition) |
| R(T) > Ξ(T) | βKWW decreases upon heating |
IV. Validation
In this section, we validate the derived heterogeneous AG model with the DFDT formalism using three systems: B2O3, Pd43Cu27Ni10P20, and PVAc. We do so by using experimentally determined values of βKWW, , and Sc to confirm the validity of the relation expressed in eqn (15) and (16) with no adjustable fitting parameters. These systems were chosen based on the availability of accurate experimental heat capacity data, temperature-dependent nonexponentiality values, and equilibrium fictive temperature fluctuations. Additionally, they span a wide range of fragility, specifically 36,28 69,29 and 95,15 for B2O3, Pd43Cu27Ni10P20, and PVAc, respectively, and are structurally and chemically distinct.
, and Sc to confirm the validity of the relation expressed in eqn (15) and (16) with no adjustable fitting parameters. These systems were chosen based on the availability of accurate experimental heat capacity data, temperature-dependent nonexponentiality values, and equilibrium fictive temperature fluctuations. Additionally, they span a wide range of fragility, specifically 36,28 69,29 and 95,15 for B2O3, Pd43Cu27Ni10P20, and PVAc, respectively, and are structurally and chemically distinct.
In all the analyses below, we first need to obtain ΔCp(T) and Sex(T) to compute Sc(T).
To estimate configurational entropy for B2O3, we calculate the excess entropy as
 | (21) |
For Pd43Cu27Ni10P20, we follow the procedure of Gallino et al.40 using the temperature at which the viscosity is 1 Pa s as the reference. Details of these calculations are provided in the SI. For PVAc, where no melting temperature exists, the configurational entropy is obtained by integrating the heat capacity difference from the Kauzmann temperature Tk, estimated using the Vogel temperature. Thus, we assume that ΔCp(T) values in three glass-forming systems are purely configurational in origin in all analyses.41
The heat capacity data for B2O3, Pd43Cu27Ni10P20, and PVAc are taken from ref. 42–44, respectively. The heat capacity of crystalline Pd43Cu27Ni10P20 was used for the heat capacity of a solid following the procedure of Gallino et al.34
The other necessary parameters for determining Sc(T) for each system are ΔSm for B2O3, Sc(Tr) for Pd43Cu27Ni10P20, and Tk for PVAc, and the values and corresponding references are compiled in Table 2. Fig. 2 shows the heat capacity data and corresponding fits for B2O3 as an example. The heat capacity data and their fits for Pd43Cu27Ni10P20 and PVAc, as well as the details of the fitting procedures, are shown in the SI.
 | ||
| Fig. 2 Heat capacities of B2O3. The data are taken from ref. 42. Cooling and heating in the legend indicate that the data were collected during constant cooling and heating. The semiempirical equation for the heat capacity of the solid introduced by Tsao et al.45 was used for Cpg, and the Cpl was assumed to be constant. The details of the fitting are given in the SI. | ||
The calculated Sc and the ratio ΔCp/Sc as a function of the reduced temperature T/Tg for the three glass-forming systems are shown in Fig. 3(a) and (b).
Next, 〈A〉 and β0 must be determined to establish the correlation between βKWW and  using eqn (15) and (16).
using eqn (15) and (16).
First, 〈A〉 is derived based on the AG equation in eqn (11) as
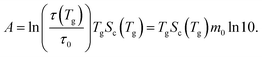 | (22) |
A is determined from eqn (22) with m0 = 16 for B2O3 and with τ0 obtained by dielectric spectroscopy for PVAc. The experimentally determined A from viscosity34 is used for Pd43Cu27Ni10P20. Second, β0 is set as 1 for B2O3, 0.76 for Pd43Cu27Ni10P20 taken from ref. 39, and 0.67 for PVAc taken from ref. 37. The resulting parameters are summarized in Table 2.
Using the experimentally determined equilibrium fictive temperature fluctuation 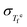 and independently evaluated thermodynamic quantities ΔCp and Sc, we now test the quantitative validity of eqn (15) and (16) by comparing the predicted temperature dependence of the nonexponentiality parameter βKWW with reported experimental values. Importantly, this comparison involves no adjustable fitting parameters.
and independently evaluated thermodynamic quantities ΔCp and Sc, we now test the quantitative validity of eqn (15) and (16) by comparing the predicted temperature dependence of the nonexponentiality parameter βKWW with reported experimental values. Importantly, this comparison involves no adjustable fitting parameters.
Panels (a)–(c) in Fig. 4 show the equilibrium fictive temperature fluctuation  for B2O3, Pd43Cu27Ni10P20, and PVAc, respectively. The reported
for B2O3, Pd43Cu27Ni10P20, and PVAc, respectively. The reported  values for B2O3 are measured via DSC and modulated-DSC.46
values for B2O3 are measured via DSC and modulated-DSC.46
 | ||
Fig. 4 Equilibrium fictive-temperature fluctuations  in (a) B2O3, (b) Pd43Cu27Ni10P20, and (c) PVAc. in (a) B2O3, (b) Pd43Cu27Ni10P20, and (c) PVAc.  for B2O3 is taken from ref. 46; for Pd43Cu27Ni10P20, it is taken from ref. 47; for PVAc, it is calculated from the reported ΔTβ in ref. 48 (see the SI). The solid line in panel (b) shows the fit used to obtain the smoothed for B2O3 is taken from ref. 46; for Pd43Cu27Ni10P20, it is taken from ref. 47; for PVAc, it is calculated from the reported ΔTβ in ref. 48 (see the SI). The solid line in panel (b) shows the fit used to obtain the smoothed 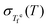 data for calculating the fictive temperature fluctuation rate R(T) in eqn (19). Panels (d–f) show the corresponding βKWW predicted from eqn (15) and (16) (red circles) compared with experimental values (black markers). For B2O3 in (d), data labeled a is taken from ref. 49 and (b)–(g) from ref. 50. For Pd43Cu27Ni10P20 in (e), the βKWW values reported by Liu et al.29 are shown as a solid line. For PVAc in (f), black circles represent data from Alegria et al.37 and black squares represent data from Sasabe et al.51 data for calculating the fictive temperature fluctuation rate R(T) in eqn (19). Panels (d–f) show the corresponding βKWW predicted from eqn (15) and (16) (red circles) compared with experimental values (black markers). For B2O3 in (d), data labeled a is taken from ref. 49 and (b)–(g) from ref. 50. For Pd43Cu27Ni10P20 in (e), the βKWW values reported by Liu et al.29 are shown as a solid line. For PVAc in (f), black circles represent data from Alegria et al.37 and black squares represent data from Sasabe et al.51 | ||
For Pd43Cu27Ni10P20, we use  reported in ref. 47 measured via Flash-DSC. For PVAc,
reported in ref. 47 measured via Flash-DSC. For PVAc,  is obtained from the reported glass transition temperature width ΔTβ measured via Flash-DSC in ref. 48 using Schawe's vitrification function κ31 as
is obtained from the reported glass transition temperature width ΔTβ measured via Flash-DSC in ref. 48 using Schawe's vitrification function κ31 as
 | (23) |
For  , the independently reported values by Tombari et al.40 and Hempel et al.46 are used. This was done because we found the deviation of the reported
, the independently reported values by Tombari et al.40 and Hempel et al.46 are used. This was done because we found the deviation of the reported 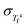 in ref. 48 from the values measured via M-DSC.
in ref. 48 from the values measured via M-DSC.
Using these 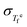 values as inputs, eqn (15) and (16) yield predictions for βKWW. The results are shown as red circles in Panels (d)–(f) of Fig. 4 and are compared with experimental βKWW values obtained from various techniques such as dielectric spectroscopy, mechanical spectroscopy, and ultrasonic sound-velocity measurements compiled from the literature (see Fig. 4 caption for details and the values listed in the SI). Despite the distinct differences in chemistry, structure, and kinetics (fragility) among the three systems, the predicted βKWW values show quantitative agreement with the experiment across the accessible temperature range.
values as inputs, eqn (15) and (16) yield predictions for βKWW. The results are shown as red circles in Panels (d)–(f) of Fig. 4 and are compared with experimental βKWW values obtained from various techniques such as dielectric spectroscopy, mechanical spectroscopy, and ultrasonic sound-velocity measurements compiled from the literature (see Fig. 4 caption for details and the values listed in the SI). Despite the distinct differences in chemistry, structure, and kinetics (fragility) among the three systems, the predicted βKWW values show quantitative agreement with the experiment across the accessible temperature range.
V. Time–temperature superposition
The assumption of time–temperature superposition (TTS) for analyzing relaxation dynamics in supercooled liquids and glasses works remarkably well near the standard Tg,17,18,52 where the relaxation time is on the order of 100 seconds. However, numerous studies have reported that this assumption breaks down at temperatures far from Tg,32,53,54 leading to what is commonly referred to as thermorheologically complex behavior.The analysis developed in Section III.B suggests that the validity or breakdown of TTS is not simply a consequence of the temperature range considered, but instead reflects a balance between the fictive temperature fluctuation rate R(T) and the configurational term Ξ(T), as defined in eqn (19) and eqn (20), respectively. To validate these derived conditions, smoothed 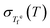 data for Pd43Cu27Ni10P20, obtained by fitting the two linear regimes with a sigmoid function (solid line in Fig. 4(c)), are used (see the SI for the detailed method). This system is chosen because the available
data for Pd43Cu27Ni10P20, obtained by fitting the two linear regimes with a sigmoid function (solid line in Fig. 4(c)), are used (see the SI for the detailed method). This system is chosen because the available 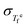 data exhibit comparatively small experimental uncertainty.
data exhibit comparatively small experimental uncertainty.
As summarized in Table 1, when R(T) − Ξ(T) = 0, the system exhibits TTS, when R(T) − Ξ(T) < 0, βKWW increases upon heating, and when R(T) − Ξ(T) > 0, βKWW decreases upon heating. In Fig. 5, the difference between the calculated R(T), evaluated using eqn (19), and Ξ(T) evaluated using eqn (20) is plotted as a function of the predicted βKWW. When R(T) − Ξ(T) = 0, the system exhibits TTS and when R(T) − Ξ(T) < 0, βKWW increases upon heating in agreement with the predictions in Table 1 established from eqn (18)–(20). The horizontal error bars represent 95% confidence from the linear fits to  in Fig. 4(b), while the vertical error bars are from the experimental uncertainty in
in Fig. 4(b), while the vertical error bars are from the experimental uncertainty in 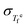 shown in Fig. 4(e). The figure shows that when βKWW remains approximately constant near Tg, the quantity R(T) − Ξ(T) approaches zero, thereby confirming the internal consistency of the present derivation.
shown in Fig. 4(e). The figure shows that when βKWW remains approximately constant near Tg, the quantity R(T) − Ξ(T) approaches zero, thereby confirming the internal consistency of the present derivation.
 | ||
Fig. 5 Difference R(T) − Ξ(T) evaluated using eqn (19) and (20), plotted as a function of the predicted βKWW for Pd43Cu27Ni10P20. Error bars represent 95% confidence intervals from linear fits’ in Fig. 4(b), and the uncertainty in βKWW arising from the experimental  . The color indicates temperature (see color bar). The dot-dashed line denotes the experimental βKWW near Tg, and the vertical dashed line indicates the TTS condition R(T) = Ξ(T) summarized in Table 1. . The color indicates temperature (see color bar). The dot-dashed line denotes the experimental βKWW near Tg, and the vertical dashed line indicates the TTS condition R(T) = Ξ(T) summarized in Table 1. | ||
Discussion
The successful application of the proposed framework was demonstrated for three chemically, structurally, and kinetically distinct glass-forming systems. The agreement between predictions and measurements, obtained without any adjustable fitting parameters, demonstrates that the correlations between the nonexponentiality parameter βKWW and the fictive temperature fluctuation , established in eqn (15) and (16) are physically consistent and applicable to a broad range of glass-forming systems.
, established in eqn (15) and (16) are physically consistent and applicable to a broad range of glass-forming systems.
The current approach does not rely on a particular functional form for ΔCp(T), Sc(T), βKWW, and  . Furthermore, extending the Adam–Gibbs equation to account for dynamic fluctuations (eqn (11)) through the DFDT relationship allows us to investigate not only the relationship between the configurational entropy Sc(T), and the average dynamics 〈τ〉 as in the standard AG equation (eqn (9)), but also the fluctuations of the dynamic and thermodynamic quantities, σln
. Furthermore, extending the Adam–Gibbs equation to account for dynamic fluctuations (eqn (11)) through the DFDT relationship allows us to investigate not only the relationship between the configurational entropy Sc(T), and the average dynamics 〈τ〉 as in the standard AG equation (eqn (9)), but also the fluctuations of the dynamic and thermodynamic quantities, σln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) τ and
τ and  . Since the equilibrium fictive temperature is proportional to the enthalpy,
. Since the equilibrium fictive temperature is proportional to the enthalpy,  directly reflects the magnitude of local enthalpy fluctuations in the supercooled liquid. The framework thus provides a quantitative link between the enthalpy fluctuation and the nonexponential relaxation spectrum through the configurational entropy and heat capacity.
directly reflects the magnitude of local enthalpy fluctuations in the supercooled liquid. The framework thus provides a quantitative link between the enthalpy fluctuation and the nonexponential relaxation spectrum through the configurational entropy and heat capacity.
The model further yields a thermodynamic condition for time–temperature superposition (TTS), requiring a balance between the temperature dependence of  , defined as the fictive temperature fluctuation rate R(T), and the configurational term Ξ(T). Wang and Richert17 found that βKWW remains constant from Tg up to a crossover temperature Tc (TTS regime), then rises steeply toward 1 at high T. They plot βKWW against log10
, defined as the fictive temperature fluctuation rate R(T), and the configurational term Ξ(T). Wang and Richert17 found that βKWW remains constant from Tg up to a crossover temperature Tc (TTS regime), then rises steeply toward 1 at high T. They plot βKWW against log10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) νmax, the peak frequency of the α-relaxation loss spectrum at different temperatures. The range of log10
νmax, the peak frequency of the α-relaxation loss spectrum at different temperatures. The range of log10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) νmax over which βKWW stays flat, expands systematically with fragility m: strong liquids show no TTS at all, while fragile liquids maintain TTS from Tg all the way to Tc, covering up to ∼8 decades. Above Tc, all systems converge to Debye relaxation (β = 1) by νmax ∼ 10 GHz regardless of m. Similar behavior was observed in shear mechanical spectroscopy measurements of inorganic systems by Sen and Lovi.16 In the current framework, the TTS condition (eqn (18)) is:
νmax over which βKWW stays flat, expands systematically with fragility m: strong liquids show no TTS at all, while fragile liquids maintain TTS from Tg all the way to Tc, covering up to ∼8 decades. Above Tc, all systems converge to Debye relaxation (β = 1) by νmax ∼ 10 GHz regardless of m. Similar behavior was observed in shear mechanical spectroscopy measurements of inorganic systems by Sen and Lovi.16 In the current framework, the TTS condition (eqn (18)) is:
It has been previously established that ΔCp(T)/Sc(T) and the rate of change of Sc(T) are directly correlated to fragility.12,13 Indeed, for the three systems investigated, both the ratio ΔCp(T)/Sc(T) and the magnitude of its slope are larger for more fragile systems, as shown in Fig. 3(b). This implies that the right-hand side of eqn (18) (shown above) will be larger for more fragile systems. Consequently, for TTS to hold, the rate at which the equilibrium fictive temperature fluctuation increases must also be larger. The empirical observation that fragile systems maintain TTS over a wider range of relaxation times thus indicates that the temperature dependence of the fictive temperature fluctuation is stronger in fragile systems. However, the fictive temperature fluctuation rate is itself temperature-dependent, as seen in the analysis of Pd43Cu27Ni10P20 in Fig. 5, supporting the eventual breakdown of TTS even for fragile liquids at high temperature, as observed by Wang and Richert.17
Conclusion
This work developed a framework that connects thermodynamic fluctuations and dynamic heterogeneity in supercooled liquids by extending the Adam–Gibbs relation through DFDT formalism. This approach establishes a direct relationship between the equilibrium fictive temperature fluctuation , which reflects the underlying distribution of local enthalpies, and the nonexponential parameter βKWW through thermodynamic quantities: the configurational entropy Sc(T) and the heat capacity difference between the glass and liquid ΔCp(T). The successful application of the framework was demonstrated for three chemically, structurally, and kinetically distinct glass-forming systems. The agreement between predictions and measurements, obtained without any adjustable fitting parameters, indicates that the derived expression in eqn (15) is physically consistent and applicable to a broad range of glass-forming systems. The model further yields a thermodynamic condition for time–temperature superposition (TTS), which requires a balance between the temperature dependence of
, which reflects the underlying distribution of local enthalpies, and the nonexponential parameter βKWW through thermodynamic quantities: the configurational entropy Sc(T) and the heat capacity difference between the glass and liquid ΔCp(T). The successful application of the framework was demonstrated for three chemically, structurally, and kinetically distinct glass-forming systems. The agreement between predictions and measurements, obtained without any adjustable fitting parameters, indicates that the derived expression in eqn (15) is physically consistent and applicable to a broad range of glass-forming systems. The model further yields a thermodynamic condition for time–temperature superposition (TTS), which requires a balance between the temperature dependence of  defined as the fictive temperature fluctuation rate R(T), and the configurational term Ξ(T), related to Sc(T), and ΔCp(T), and their temperature dependence. These results provide a thermodynamic connection to dynamic heterogeneity and TTS in supercooled liquids. Further validation would require a more integrated examination of the relationships among the temperature dependence of fictive temperature fluctuations (enthalpy fluctuation), configurational entropy, and dynamic heterogeneity in glass-forming systems. Future work combining experiments and atomistic simulations may further clarify the structural origins of these relationships.
defined as the fictive temperature fluctuation rate R(T), and the configurational term Ξ(T), related to Sc(T), and ΔCp(T), and their temperature dependence. These results provide a thermodynamic connection to dynamic heterogeneity and TTS in supercooled liquids. Further validation would require a more integrated examination of the relationships among the temperature dependence of fictive temperature fluctuations (enthalpy fluctuation), configurational entropy, and dynamic heterogeneity in glass-forming systems. Future work combining experiments and atomistic simulations may further clarify the structural origins of these relationships.
Conflicts of interest
There are no conflicts to declare.Data availability
This study does not report any new experimental data. The data reported in this study are modeled from experimental data available in the cited literature. All modeled data are included in the manuscript and the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6ma00442c.Acknowledgements
The authors acknowledge financial support from NSF-DMR under grant#: 1832817.References
- M. D. Ediger, Spatially Heterogeneous Dynamics in Supercooled Liquids, Annu. Rev. Phys. Chem., 2000, 51, 99–128, DOI:10.1146/annurev.physchem.51.1.99.
- P. G. Debenedetti and F. H. Stillinger, Supercooled liquids and the glass transition, Nature, 2001, 410, 259–267, DOI:10.1038/35065704.
- R. Richert, Heterogeneous dynamics in liquids: fluctuations in space and time, J. Phys.: Condens. Matter, 2002, 14, R703, DOI:10.1088/0953-8984/14/23/201.
- R. Kohlrausch, Theorie des elektrischen Rückstandes in der Leidener Flasche, Ann. Phys. Chem., 1854, 167, 179–214, DOI:10.1002/andp.18541670103.
- G. Williams and D. C. Watts, Non-symmetrical dielectric relaxation behaviour arising from a simple empirical decay function, Trans. Faraday Soc., 1970, 66, 80–85, 10.1039/TF9706600080.
- R. Richert, Homogeneous dispersion of dielectric responses in a simple glass, J. Non. Cryst. Solids, 1994, 172–174, 209–213, DOI:10.1016/0022-3093(94)90436-7.
- K. L. Ngai, Temperature dependence of the stretched exponent in structural relaxation of fragile glass-forming molecular liquids, J. Non-Cryst. Solids, 1991, 131–133, 80–83, DOI:10.1016/0022-3093(91)90277-D.
- K. L. Ngai and S. W. Martin, Correlatioin between the activation enthalpy and Kohlrausch exponent for ionic conductivity in oxide glasses, Phys. Rev. B:Condens. Matter Mater. Phys., 1989, 40, 10550–10556, DOI:10.1103/PhysRevB.40.10550.
- P. Zhang, J. J. Maldonis, Z. Liu, J. Schroers and P. M. Voyles, Spatially heterogeneous dynamics in a metallic glass forming liquid imaged by electron correlation microscopy, Nat. Commun., 2018, 9, 1129, DOI:10.1038/s41467-018-03604-2.
- P. K. Dixon and S. R. Nagel, Frequency-Dependent Specific Heat and Thermal Conductivity at the Glass Transition in o-Terphenyl Mixtures, Phys. Rev. Lett., 1988, 61, 341–344, DOI:10.1103/PhysRevLett.61.341.
- C. A. Angell, Relaxation in liquids, polymers and plastic crystals—strong/fragile patterns and problems, J. Non-Cryst. Solids, 1991, 131–133, 13–31, DOI:10.1016/0022-3093(91)90266-9.
- L. M. Martinez and C. A. Angell, A thermodynamic connection to the fragility of glass-forming liquids, Nature, 2001, 410, 663–667, DOI:10.1038/35070517.
- L. M. Wang, C. A. Angell and R. Richert, Fragility and thermodynamics in nonpolymeric glass-forming liquids, J. Chem. Phys., 2006, 125, 074505/1–074505/8, DOI:10.1063/1.2244551.
- V. Lubchenko and P. G. Wolynes, Theory of aging in structural glasses, J. Chem. Phys., 2004, 121, 2852–2865, DOI:10.1063/1.1771633.
- R. Böhmer, K. L. Ngai, C. A. Angell and D. J. Plazek, Nonexponential relaxations in strong and fragile glass formers, J. Chem. Phys., 1993, 99, 4201–4209, DOI:10.1063/1.466117.
- S. Sen and J. Lovi, Trends in the temperature dependence of dynamical heterogeneity in strong and fragile supercooled liquids, J. Non-Cryst. Solids, 2024, 638, 123060, DOI:10.1016/j.jnoncrysol.2024.123060.
- L. M. Wang and R. Richert, Primary and secondary relaxation time dispersions in fragile supercooled liquids, Phys. Rev. B: Condens. Matter Mater. Phys., 2007, 76, 1–8, DOI:10.1103/PhysRevB.76.064201.
- N. B. Olsen, T. Christensen and J. C. Dyre, Time-temperature superposition in viscous liquids, Phys. Rev. Lett., 2001, 86, 1271–1274, DOI:10.1103/PhysRevLett.86.1271.
- E. Donth, General derivation of the WLF equation from a fluctuation approach to the glass transition, Acta Polym., 1979, 30, 481–485, DOI:10.1002/actp.1979.010300804.
- E. W. Fischer, E. Donth and W. Steffen, Temperature dependence of characteristic length for glass transition, Phys. Rev. Lett., 1992, 68, 2344–2346, DOI:10.1103/PhysRevLett.68.2344.
- J. C. Mauro and R. J. Loucks, Selenium glass transition: A model based on the enthalpy landscape approach and nonequilibrium statistical mechanics, Phys. Rev. B: Condens. Matter Mater. Phys., 2007, 76, 174202, DOI:10.1103/PhysRevB.76.174202.
- J. C. Mauro, R. J. Loucks, J. Balakrishnan and S. Raghavan, Monte Carlo method for computing density of states and quench probability of potential energy and enthalpy landscapes, J. Chem. Phys., 2007, 126, 194103, DOI:10.1063/1.2733674.
- C. T. Moynihan and J. Schroeder, Non-exponential structural relaxation, anomalous light scattering and nanoscale inhomogeneities in glass-forming liquids, J. Non-Cryst. Solids, 1993, 160, 52–59, DOI:10.1016/0022-3093(93)90283-4.
- J. Schroeder, M. Lee, S. K. Saha, J. H. Whang and C. T. Moynihan, Independently relaxing nanoscale inhomogeneities as a model for structural relaxation: Light scattering around the glass transition region, J. Non-Cryst. Solids, 1996, 203, 186–191, DOI:10.1016/0022-3093(96)00500-5.
- M. Lee, S. K. Saha, C. T. Moynihan and J. Schroeder, Non-exponential structural relaxation, anomalous light scattering and nanoscale inhomogeneities in glasses, J. Non. Cryst. Solids, 1997, 222, 369–375, DOI:10.1016/S0022-3093(97)90138-1.
- W. Takeda and P. Lucas, A model of heterogeneous undercooled liquid and glass accounting for temperature-dependent nonexponentiality and enthalpy fluctuation, J. Chem. Phys., 2024, 160, 174504, DOI:10.1063/5.0196812.
- G. Adam and J. H. Gibbs, On the Temperature Dependence of Cooperative Relaxation Properties in Glass-Forming Liquids, J. Chem. Phys., 1965, 43, 139–146, DOI:10.1063/1.1696442.
- K. J. Crowley and G. Zografi, The use of thermal methods for predicting glass-former fragility, Thermochim. Acta, 2001, 380, 79–93, DOI:10.1016/S0040-6031(01)00662-1.
- C. Liu, E. Pineda, J. Qiao and D. Crespo, Modeling of the Sub-Tg Relaxation Spectrum of Pd42.5Ni7.5Cu30P20 Metallic Glass, J. Phys. Chem. B, 2016, 120, 2838–2844, DOI:10.1021/acs.jpcb.5b11754.
- E. Donth, Characteristic length of the glass transition, J. Polym. Sci. B Polym. Phys., 1996, 34, 2881–2892, DOI:10.1002/(SICI)1099-0488(199612)34:17<2881::AID-POLB3>3.0.CO;2-U.
- J. E. K. Schawe, Vitrification in a wide cooling rate range: The relations between cooling rate, relaxation time, transition width, and fragility, J. Chem. Phys., 2014, 141, 184905, DOI:10.1063/1.4900961.
- D. Cangialosi, A. Alegría and J. Colmenero, On the temperature dependence of the nonexponentiality in glass-forming liquids, J. Chem. Phys., 2009, 130, 124902, DOI:10.1063/1.3098904.
- I. M. Hodge, Adam-Gibbs Formulation of Nonlinearity in Glassy-State Relaxations, Macromolecules, 1986, 19, 936–938, DOI:10.1021/ma00157a082.
- I. Gallino, J. Schroers and R. Busch, Kinetic and thermodynamic studies of the fragility of bulk metallic glass forming liquids, J. Appl. Phys., 2010, 108, 063501, DOI:10.1063/1.3480805.
- P. K. Gupta and J. C. Mauro, Two factors governing fragility: Stretching exponent and configurational entropy, Phys. Rev. E:Stat., Nonlinear, Soft Matter Phys., 2008, 78, 1–3, DOI:10.1103/PhysRevE.78.062501.
- R. Richert and M. Richert, Dynamic heterogeneity, spatially distributed stretched-exponential patterns, and transient dispersions in solvation dynamics, Phys. Rev. E:Stat. Phys., Plasmas, Fluids, Relat. Interdiscip. Top., 1998, 58, 779–784, DOI:10.1103/PhysRevE.58.779.
- A. Alegría, J. Colmenero, P. O. Mari and I. A. Campbell, Dielectric investigation of the temperature dependence of the nonexponentiality of the dynamics of polymer melts, Phys. Rev. E:Stat. Phys., Plasmas, Fluids, Relat. Interdiscip. Top., 1999, 59, 6888–6895, DOI:10.1103/PhysRevE.59.6888.
- B. Y. J. C. Southard, The Thermal Properties of Crystalline and Glassy Boron Trioxide, J. Am. Ceram. Soc., 1941, 63, 3147–3150, DOI:10.1021/ja01856a073.
- A. Meyer, R. Busch and H. Schober, Time-temperature superposition of structural relaxation in a viscous metallic liquid, Phys. Rev. Lett., 1999, 83, 5027–5029, DOI:10.1103/PhysRevLett.83.5027.
- E. Tombari, C. Ziparo, G. Salvetti and G. P. Johari, Vibrational and configurational heat capacity of poly(vinyl acetate) from dynamic measurements, J. Chem. Phys., 2007, 127, 014905, DOI:10.1063/1.2747596.
- H. L. Smith, C. W. Li, A. Hoff, G. R. Garrett, D. S. Kim, F. C. Yang, M. S. Lucas, T. Swan-Wood, J. Y. Y. Lin, M. B. Stone, D. L. Abernathy, M. D. Demetriou and B. Fultz, Separating the configurational and vibrational entropy contributions in metallic glasses, Nat. Phys., 2017, 13, 900–905, DOI:10.1038/nphys4142.
- S. B. Thomas and G. S. Parks, Studies on Glass. VI. Some Specific Heat Data on Boron Trioxide, J. Phys. Chem., 1931, 35, 2091–2102, DOI:10.1021/j150325a016.
- M. Kuno, L. A. Shadowspeaker, J. Schroers and R. Busch, Thermodynamics of the Pd43Ni10Cu27P 20bulk metallic glass forming alloy, Mater. Res. Soc. Symp. Proc., 2003, 806, 227–232, DOI:10.1557/proc-806-mm5.2.
- U. Gaur, B. B. Wunderlich and B. Wunderlich, Heat Capacity and Other Thermodynamic Properties of Linear Macromolecules. VII. Other Carbon Backbone Polymers, J. Phys. Chem. Ref. Data, 1983, 12, 29–63, DOI:10.1063/1.555677.
- J. Y. Tsao, Two semiempirical expressions for condensed-phase heat capacities, J. Appl. Phys., 1990, 68, 1928–1930, DOI:10.1063/1.346588.
- E. Hempel, G. Hempel, A. Hensel, C. Schick and E. Donth, Characteristic Length of Dynamic Glass Transition near Tg for a Wide Assortment of Glass-Forming Substances, J. Phys. Chem. B, 2000, 104, 2460–2466, DOI:10.1021/jp991153f.
- J. E. K. Schawe, M. K. Kwak, M. Stoica, E. S. Park and J. F. Löffler, The Cooperativity of Atomic Fluctuations in Highly Supercooled Glass-Forming Metallic Melts, J. Phys. Chem. Lett., 2025, 16, 948–954, DOI:10.1021/acs.jpclett.4c03275.
- K. Hallavant, M. Mejres, J. E. K. Schawe, A. Esposito and A. Saiter-Fourcin, Influence of Chemical Composition and Structure on the Cooperative Fluctuation in Supercooled Glass-Forming Liquids, J. Phys. Chem. Lett., 2024, 15, 4508–4514, DOI:10.1021/acs.jpclett.4c00632.
- D. Sidebottom, R. Bergman, L. Börjesson and L. M. Torell, Two-step relaxation decay in a strong glass former, Phys. Rev. Lett., 1993, 71, 2260–2263, DOI:10.1103/PhysRevLett.71.2260.
- G. M. Bartenev and V. A. Lomovskoi, Relaxation time spectra and the peculiarities of the process of boron anhydride glass transition, J. Non-Cryst. Solids, 1992, 146, 225–232, DOI:10.1016/S0022-3093(05)80495-8.
- H. Sasabe and C. T. Moynihan, Structural Relaxation in Poly(Vinyl Acetate), J Polym Sci Polym Phys. Ed., 1978, 16, 1447–1457, DOI:10.1002/pol.1978.180160810.
- J. Zhao and G. B. McKenna, Temperature divergence of the dynamics of a poly(vinyl acetate) glass: Dielectric vs. mechanical behaviors, J. Chem. Phys., 2012, 136, 154901, DOI:10.1063/1.3701736.
- G. W. Scherer, Volume Relaxation Far from Equilibrium, J. Am. Ceram. Soc., 1986, 69, 374–381, DOI:10.1111/j.1151-2916.1986.tb04764.x.
- R. F. Lancelotti, E. D. Zanotto and S. Sen, Kinetics of physical aging of a silicate glass following temperature up- and down-jumps, J. Chem. Phys., 2024, 160, 034504, DOI:10.1063/5.0185538.
| This journal is © The Royal Society of Chemistry 2026 |


