 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Tuning magnetic order and spin–exciton interactions in MnPSe3 via Cu substitution
Mohamed Nawwar a,
Alexander Blackston
a,
Alexander Blackston a,
Sogol Lotfi
a,
Sogol Lotfi a,
Ziling Deng
a,
Ziling Deng a,
Alexander Reifsnydera,
David McComb
a,
Alexander Reifsnydera,
David McComb a,
Wolfgang Windlab,
Vicky Doan-Nguyen Trigg
a,
Wolfgang Windlab,
Vicky Doan-Nguyen Trigg a and
Roberto C. Myers
a and
Roberto C. Myers *abc
*abc
aDepartment of Materials Science and Engineering, The Ohio State University, Columbus, OH 43210, USA. E-mail: myers.1079@osu.edu
bDepartment of Physics, The Ohio State University, Columbus, OH 43210, USA
cDepartment of Electrical and Computer Engineering, The Ohio State University, Columbus, OH 43210, USA
First published on 23rd April 2026
Abstract
We report a comprehensive investigation of Cu-substituted MnPSe3 [Mn1−xCu2xPSe3 (x = 0–0.2)], a layered van der Waals magnetic semiconductor, revealing a unique interplay between chemical substitution, magnetism, and excitonic behavior. Structural analyses demonstrate that Cu(I) ions substitute Mn(II) at distinct crystallographic sites, producing anisotropic lattice expansion. With increasing Cu content, magnetic susceptibility measurements show a systematic suppression of antiferromagnetic order and a transition to a paramagnetic state beyond x ≥ 0.15. Concurrently, thermal conductivity decreases due to enhanced phonon scattering. Photoluminescence spectroscopy uncovers three sub-bandgap excitons, two of which show pronounced redshifts as a function of temperature, indicating strong spin–exciton coupling. This work establishes chemical substitution as a powerful tool to tune magnetism and spin–exciton interactions in 2D materials, opening new design strategies for spintronic and quantum optoelectronic platforms.
1 Introduction
Two-dimensional (2D) van der Waals (vdW) magnetic materials offer a rich platform for controlling spin interactions, topological properties, and collective excitations in layered systems. Their atomic thinness and intrinsic anisotropy enable magnetic properties to be tuned via electrostatic gating, strain, doping, and heterostructuring—making them compelling candidates for next-generation spintronic and magnonic devices. In recent years, intrinsic magnetism has been discovered in vdW compounds such as MnPSe3, MnPS3, FePS3, CrI3, CrCl3, Cr2Ge2Te6, VSe2, and MnSe2.1–8 In many of these systems, magnetic order and Berry curvature can be modulated by strain, electric, and magnetic fields.9–14 The MPX3 (M = Cr, Mn, Fe, Co, Ni, Zn, Cd; X = S, Se) family of materials has shown great potential thanks to the many discoveries and predictions of topological magnons, magnon–phonon hybridization, excitonic insulators and magneto-electric coupling.13,15–19Elemental doping has shown great potential in tuning various physical properties in different families of materials.20–23 Among various approaches to tune magnetism in vdW magnets, atomic substitution has emerged as a powerful route to systematically perturb exchange interactions and spin–lattice coupling. The MPX3 family consists of layered materials that crystallize in monoclinic (C2/m) or trigonal (R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) ) structures with transition metal cations forming honeycomb sublattices.24,25 Substitution of magnetic ions with other magnetic species has been widely explored in compounds such as MnxFe1−xPS3, MnxNi1−xPS3, Mn1−xFexPSe3, Ni1−xFexPS3, and Fe0.5Ni0.5PS3, leading to modified anisotropies, ordering temperatures, and magnetic structures.26–34 In contrast, the effects of substituting magnetic ions with non-magnetic species remain comparatively unexplored. Such substitutions offer a route to investigate dilute or frustrated magnetic orderings, and may shed light on how non-magnetic impurities influence spin coherence, transport, and exciton–spin interactions in layered magnets.
) structures with transition metal cations forming honeycomb sublattices.24,25 Substitution of magnetic ions with other magnetic species has been widely explored in compounds such as MnxFe1−xPS3, MnxNi1−xPS3, Mn1−xFexPSe3, Ni1−xFexPS3, and Fe0.5Ni0.5PS3, leading to modified anisotropies, ordering temperatures, and magnetic structures.26–34 In contrast, the effects of substituting magnetic ions with non-magnetic species remain comparatively unexplored. Such substitutions offer a route to investigate dilute or frustrated magnetic orderings, and may shed light on how non-magnetic impurities influence spin coherence, transport, and exciton–spin interactions in layered magnets.
MnPSe3, a member of the MPX3 family, crystallizes in the trigonal R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) structure, with Mn(II) ions forming an antiferromagnetic honeycomb lattice in the ab plane (Fig. 1a and b).35 Prior work has shown that monovalent Cu(I) can coexist with divalent Mn(II) in MnPS3 forming Mn0.87Cu0.26PS3.36,37 In the selenide family, Cu2PSe3 is known to exist in both monoclinic (P21/c) and triclinic (P
structure, with Mn(II) ions forming an antiferromagnetic honeycomb lattice in the ab plane (Fig. 1a and b).35 Prior work has shown that monovalent Cu(I) can coexist with divalent Mn(II) in MnPS3 forming Mn0.87Cu0.26PS3.36,37 In the selenide family, Cu2PSe3 is known to exist in both monoclinic (P21/c) and triclinic (P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ) structures.38 These findings suggest that Cu(I) may be incorporated into MnPSe3 as a structural and electronic dopant, potentially modulating magnetic order while preserving crystallinity.
) structures.38 These findings suggest that Cu(I) may be incorporated into MnPSe3 as a structural and electronic dopant, potentially modulating magnetic order while preserving crystallinity.
In this work, we demonstrate successful Cu substitution in MnPSe3 across the solid solution Mn1−xCu2xPSe3 (x = 0–0.2), and investigate its effects on structure, magnetism, and excitonic optical response. Cu(I) is found to lie in trigonal coordination with Se, leading to anisotropic lattice expansion and progressive suppression of antiferromagnetic order. At substitution levels x ≥ 0.15, the material becomes paramagnetic down to 2 K. Photoluminescence spectra for x = 0, 0.05, and 0.2 reveal three sub-bandgap excitonic features, two of which redshift with temperature—an effect that is most pronounced in compositions with stronger antiferromagnetic order. These results suggest that excitons in MnPSe3 couple to the spin lattice and that Cu substitution weakens this coupling by disrupting magnetic correlations. Together, our findings highlight the use of non-magnetic atomic substitution as a versatile tool to tune magnetic order and spin–exciton interactions in 2D vdW materials.
2 Experimental
2.1 Synthesis and crystal growth
Polycrystalline powder samples of the solid solution Mn1−xCu2xPSe3 (x = 0, 0.05, 0.1, 0.15, 0.2 and 0.25) were synthesized using conventional solid-state synthesis technique in an inert environment. Stoichiometric amounts of Mn (Sigma Aldrich 99.9%), Cu (Sigma Aldrich 99.5%), P (Sigma Aldrich, red, 99.99%), and Se (Sigma Aldrich, 99.5%) were ground using an agate mortar and pestle inside an Ar-filled glove box (<5 ppm H2O, O2). Chemicals were used without purification with a mass loading of 1.5 g. Powder elemental precursors were loaded into a 7 mm inner diameter fused silica tube and sealed under vacuum (<1 torr). The samples were then placed in a box furnace at 600 °C for 6 days with a ramp rate of 5 °C min−1 and a cooling rate of 10 °C min−1. Products were then ground using an agate mortar and pestle and stored under ambient conditions. No degradation or oxidation was observed over time.Single crystals of the alloys used for the thermal conudctivity and optical studies were grown using Chemical Vapor Transport (CVT) method. Resulting polycrystalline powdered samples were mixed with 0.03 g of iodine (transport gas) and then loaded into a fused silica tube and sealed under vacuum (<1 Torr). During the sealing, the precursors were immersed in liquid nitrogen to prevent the evaporation of iodine. The sample was then placed in a two-zone furnace with Thot at 650 °C and Tcold at 600 °C for one week with a ramp rate of 5 °C min−1 and cooling rate of 0.1 °C min−1.
2.2. Synchrotron X-ray diffraction (XRD) and pair distribution functions (PDF)
XRD and PDF data were obtained at Argonne National Lab's Advanced Photon Source, beamline 17-BM-B, with λ = 0.24012 Å at sample-to-detector distances of 700 mm and 200 mm, respectively. Data processing and Rietveld refinement were conducted using PDFgetX339 and GSAS-II.402.3. X-ray photoelectron spectroscopy (XPS)
XPS measurement was conducted using a Kratos Axis Ultra XPS instrument with a monochromatic Al X-ray source. XPS spectra were fit by Voigt functions with Shirley baselines using CasaXPS.41 Adventitious C 1s was used for calibration at 284.5 eV.2.4. Magnetic susceptibility
Magnetic susceptibility measurements were done using a Quantum Design MPMS3 SQUID with a 7T magnet. Both field-cooled (FC) and zero field-cooled (ZFC) measurements were done under an applied magnetic field of 1T.2.5. Thermal conductivity
Samples were mounted at the edge of a copper block that acts as a heat sink. A 120 Ω resistor was mounted at the opposite side of the sample and used as a heater to send a heat flux across the sample. Two type T thermocouples were placed on one side of the sample to measure the temperature difference. All parts were adhered using silver epoxy. The samples were then placed in a vacuumed environment inside a Lakeshore cryostat. The voltages across the thermocouples were read using a 2182A Keithley nanovoltmeter. The cryostat temperature was controlled via a 331 Lakeshore temperature controller. The temperature was allowed to stabilize for 20 minutes before the sample heater was turned on. The temperature gradient was allowed to stabilize for 5 minutes before measuring.2.6. Photoluminescence spectroscopy
Spectrally resolved photoluminescence measurements were taken in the reflection geometry using a tunable Ti-sapphire laser (Coherent Chameleon Ultra II) in conjunction with a SHG and THG generation system (Coherent Vue) as the excitation source. Focus was obtained using a 150 mm focal length achromatic lens. An excitation wavelength of 250 nm (4.96 eV) was used with a power of 1 mW leading to a power density of 0.13 W cm−2. PL measurements were conducted on different sample locations and no degradation in PL spectra were detected even after long (>1 h) laser exposure periods mirroring previous studies that found no laser degradation in MnPSe3 at similar power densities.16The reflected beam and photoluminescence was sent through a 265 nm long pass filter to filter out elastically scattered light and then analyzed using a 1200 g mm−1, 500 nm blaze reflective diffraction grating and Teledyne Vision Solutions Pixis 400 CCD camera housed inside of a Princeton Instruments Acton Series SP-2500i spectrometer. All photoluminescence measurements were carried out in a closed-loop helium cryostat (Montana Instruments Cryostation S50) that could be cooled to 8 K.2.7. Absorption measurement
The beam from a halogen lamp (Ocean Optics DH-2000) is collimated and then focused through the flakes, which were less than 2 mm wide and exfoliated onto glass slides, with a 40× reflective objective. A second 40× reflective objective was used to re-collimate the transmitted light. A diffraction grating spectrometer then spectrally resolves the light intensity. The transmitted spectrum is then spectrally analyzed using a 1200 g mm−1, 500 nm blaze reflective diffraction grating and Teledyne Vision Solutions Pixis 400 CCD camera housed inside of a Princeton Instruments Acton Series SP-2150i spectrometer. Transmission along the beam path is measured without the flake in the beam path to collect a reference spectrum. All measurements were completed at room temperature.3 Results and discussion
3.1. Structural characterization
The XPS spectrum for x = 0.15 depicts Cu 2p3/2 peak with FWHM of 1.13, centered at 932.52 eV and separated from Cu 2p1/2 (FWHM of 1.56) by 19.80 eV (Fig. 1(d)), consistent with previous reports for Cu2O.42 Cu(II)acetylacetonate and Cu(I)Br were used as standards for comparison. The shift in the peak center of Cu 2p3/2 and the absence of the shake-up peaks around 940 eV and 945 eV (marked with red asterisks in Fig. 1(d)) confirm that Cu exists as monovalent. In addition, the Cu LMM auger peak for this sample appears at 569.90 eV binding energy, which is 916.70 eV kinetic energy. The Cu 2p3/2 XPS peak meanwhile is at 932.52 eV binding. The summation of the auger kinetic and the XPS binding energies is 1849.22 eV. The intersection of these three numbers on an auger parameters chart confirms that Cu is monovalent.43
![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) . Increasing Cu substitution amounts, however, had a major impact on the (00l) and (11l) planes. Increasing x shifts the (006) peak to lower Q values, indicating lattice expansion along the c-axis (Fig. 2(b)). Rietveld refinement of the PXRD data showed that the lattice parameter, c increased from 20.051(1) Å to 20.141(2) Å by increasing x from 0.0 to 0.10. Unlike the peak shift of the (006) peak, the (113) plane showed negligible shift with a major peak broadening, indicating increasing lattice strain along this plane(Fig. 2(c)). Rietveld refinement confirmed that the lattice parameters (Table S1) increased linearly with Cu substitution (Fig. 2(d)). Lattice expansion was found to be highly anisotropic where the c-axis expansion rate with respect to Cu substitution is 10 times that of the a-axis.
. Increasing Cu substitution amounts, however, had a major impact on the (00l) and (11l) planes. Increasing x shifts the (006) peak to lower Q values, indicating lattice expansion along the c-axis (Fig. 2(b)). Rietveld refinement of the PXRD data showed that the lattice parameter, c increased from 20.051(1) Å to 20.141(2) Å by increasing x from 0.0 to 0.10. Unlike the peak shift of the (006) peak, the (113) plane showed negligible shift with a major peak broadening, indicating increasing lattice strain along this plane(Fig. 2(c)). Rietveld refinement confirmed that the lattice parameters (Table S1) increased linearly with Cu substitution (Fig. 2(d)). Lattice expansion was found to be highly anisotropic where the c-axis expansion rate with respect to Cu substitution is 10 times that of the a-axis.
By increasing Cu substitution, we observe peak broadening and an increase in peak intensity in the P–Se peak (r = 2.24 Å) suggesting that Cu(I) is substituted onto a site with a similar interatomic distance (Fig. 3(b)). A noticeable increase in the peak intensity was also observed at r = 3.99 Å, indicating the existence of Cu(I) at this interatomic distance as well (Fig. 3(d)).
Mn–Se and Mn–Mn interatomic distances (r = 2.71 Å and r = 3.69 Å, respectively) were not shifted by Cu substitution (Fig. 3(c) and (d)). Only a decrease in the peak intensity was observed. The decrease in the peak intensity in these peaks is a result of introducing Mn vacancies with higher substitution levels.
| dij = Rij0 − Blnvij | (1) |
To achieve this trigonal coordination and maintain the monovalent oxidation state of Cu, we found that Cu(I) is located at (0, 0, 0.083) position above Mn (Fig. 4(a and b)) with bond distances 2.19 Å and 4.06 Å for Cu–Se and Cu–Mn, respectively, in agreement with PDF and XRD analyses. Another possible location for Cu with trigonal coordination is above phosphorous. However, this results in Cu bonded to the divalent (PSe3)2−, contradicting the XPS results. This leaves the Cu atom at the Wyckoff site, 6c in the R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) space group.46 However, because of the valency difference between Mn(II) and Cu(I) ions, we are obliged to replace each Mn vacancy with 2 Cu(I) ions not one. Since the R
space group.46 However, because of the valency difference between Mn(II) and Cu(I) ions, we are obliged to replace each Mn vacancy with 2 Cu(I) ions not one. Since the R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) space group doesn't have a Wyckoff site with a 12 multiplicity,46 we need to introduce another Cu atom with the same interatomic distances and trigonal coordination. Such site is available when we look at a mirror symmetry across the Mn plane, where Mn site is at
space group doesn't have a Wyckoff site with a 12 multiplicity,46 we need to introduce another Cu atom with the same interatomic distances and trigonal coordination. Such site is available when we look at a mirror symmetry across the Mn plane, where Mn site is at 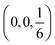 . The second Cu site, therefore is at
. The second Cu site, therefore is at 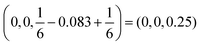 .
.
The final structure shown in Fig. 4(a) maintains the same symmetry elements as the pure MnPSe3 and is similar to the previously studied Mn0.87Cu0.26PS3,37 where each Mn(II) ion is replaced by two Cu(I) ions above and below it, and Cu(I) ions are in trigonal coordination with Se. Our DFT calculations further confirm our predicted structure, where it shows negative formation energy for substituting Mn with two Cu atoms, while substitution with a single Cu atom showed positive formation energy and thus is energetically unfavorable (Fig. S8). HAADF-STEM image of the crystal shown in Fig. 4(c) confirms the crystallinity and purity of the grown material.
3.2. Magnetic and thermal characterization
FC and ZFC magnetic susceptibility data of Mn1−xCu2xPSe3 shows that the phases x = 0, 0.05, and 0.1 exhibit antiferromagnetic (AFM) behavior (Fig. 5(a)). The broadness of the AFM transition peak is due to short-range spin correlation.47 The Néel temperature was, therefore, determined by the maximum point in the dM/dT graph (Fig. S2). The Néel temperature found for MnPSe3 was 68 K, in agreement with previous reports.1 Néel temperatures for x = 0.05 and 0.1 decreased to 64 K and 58 K respectively. Higher Cu substitution levels (x = 0.15, and 0.2) did not exhibit any magnetic ordering as further confirmed by M vs. H hystersis loops (Fig. S3). In addition, no separation between FC and ZFC was observed.PDF indicates that Mn–Mn on the honeycomb lattice and Mn-Se bonds on the octahedral coordination environment are not affected by Cu substitution. Therefore, the intralayer Mn–Mn magnetic direct exchange interactions (J1, J2, and J3) are not expected to be affected at low substitution levels. Yet, the AFM order is affected by Cu substitution, indicating that other factors play a significant role in the magnetism of Mn1−xCu2xPSe3.
XRD confirmed that Cu substitution causes lattice expansion along the c-axis, and PDF analysis confirmed that Mn–Se distances remain constant with Cu substitution indicating the layer's width is constant as well. As a result, the vdW gap increases as a function of Cu substitution, affecting the interlayer magnetic exchange interaction (Jc). Even though this exchange parameter is usually small and negligible in vdW materials, neutron studies on MnPSe3 showed that the magnitude of Jc (0.031 meV) is similar to that of J2 (0.03 meV).48 Therefore, by varying Jc through increasing the interlayer spacing, a significant impact on the AFM ordering of MnPSe3 is expected. Additionally, by increasing substitution values, more vacancies are introduced onto the Mn site. Magnetic susceptibility confirmed that magnetic order is lost at 15% of Mn vacancies, which is equivalent to one Mn(II) vacancy per honeycomb lattice. The introduction of Mn vacancies is confirmed from ICP and PDF data (Fig. 3(c) and (d)), where the peak intensities decrease by increasing substitution levels. Therefore, establishing a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 correspondence between (x) and the Mn vacancy concentration This would highly affect the intralayer magnetic exchange parameters and contribute to the loss of magnetic ordering.
1 correspondence between (x) and the Mn vacancy concentration This would highly affect the intralayer magnetic exchange parameters and contribute to the loss of magnetic ordering.
The effect of introducing Mn vacancies on the magnetic structure is quite similar to substituting non-magnetic ions such as Zn on the honeycomb lattice. This non-magnetic substitution, known also as magnetic dilution, has been previously studied on MnPS3, a sister compound to MnPSe3. It was found that the long-range antiferromagnetic order was maintained up to 50% substitution.49 However, in our copper substitution, the long-range order was lost at only 15%. This illustrates that magnetic dilution on the honeycomb lattice cannot alone explain this, and that the vdW gap increase must play a significant role. Further neutron diffraction and EXAFS studies could be conducted to further confirm the proposed crystal structure and obtain more robust information about the magnetic structure.
Fig. 5(b) shows the thermal conductivity, κxx of the solid solution, Mn1−xCu2xPSe3 for x = 0, 0.15, and 0.2 from 160 K up to 300 K. Thermal conductivity was measured across the ab plane on single crystals of the alloys. In the pure phase, MnPSe3, κxx decreases from 9.5 W mK−1 at 160 K to 7 W mK−1 at room temperature. This consistent decrease across all samples is due to phonon–phonon scattering that's dominant at high temperatures. By increasing the Cu substitution amounts, κxx decreases consistently. As x increases, more defects are introduced into the material; this will enhance phonon-defect scattering, and thus lowers the overall thermal conductivity, κxx.
3.3. Opto-electronic simulation and characterization
![[D with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0044_20d1.gif) ij|2) between transitions involving initial (i) and final (j) states between bands at high symmetry points are calculated using the equation,
ij|2) between transitions involving initial (i) and final (j) states between bands at high symmetry points are calculated using the equation,
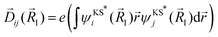 | (2) |
![[R with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0052_20d1.gif) I is the position of the ions taken from previous neutron diffraction studies35 and
I is the position of the ions taken from previous neutron diffraction studies35 and 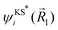 and
and 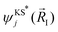 are the Kohn–Sham wave functions of the initial and final electronic states.51,52 The most probable transitions, i.e. the transitions with the highest
are the Kohn–Sham wave functions of the initial and final electronic states.51,52 The most probable transitions, i.e. the transitions with the highest ![[D with combining right harpoon above (vector)]](https://www.rsc.org/images/entities/i_char_0044_20d1.gif) ij, are included in Table 1.
ij, are included in Table 1.
 | ||
| Fig. 6 DFT band structure identifies spin-resolved optical transitions in MnPSe3. DFT calculated spin-resolved band structure of MnPSe3. Highly probable transitions outlined in Table 1 are shown with arrows and labeled. | ||
| Label | Transition | Ej − Ei (eV) | |Dij|2 |
|---|---|---|---|
| S01 | S0-35 → S0-27 | 2.644 | 2.798 |
| H01 | H0-25 → H0-26 | 2.960 | 0.601 |
| S21 | S2-35 → S2-38 | 3.535 | 23.5 |
| S02 | S0-35 → S0-31 | 3.891 | 9.457 |
| H21 | H2-35 → H2-41 | 4.430 | 6.857 |
| H22 | H2-35 → H2-42 | 4.803 | 5.624 |
The three exciton peak structure and the spacing of the measured exciton energies are strikingly similarly to exciton energies previously measured in CrI3 “Eex–A ≈ 1.50 eV, Eex–B ≈ 1.85 eV, and Eex–C ≈ 2.20 eV”, a van der Waals magnetic material that has a similar crystal structure to MnPSe3.55–58 Furthermore, in our measurements, an anomalously large temperature induced red-shift in exciton energy is observed for excitons B and C (XB and XC. In the pure MnPSe3 sample, between 8 K and 300 K, the energy of excitons B and C decrease by a magnitude of 35.2 meV and 29.4 meV, respectively (Fig. 7(d)). These energy shifts are significantly higher than that of exciton A, whose energy decreases by merely 7.3 meV with the same increase in temperature.
A reduction in exciton energy with increasing temperature is generally expected across all materials, primarily due to enhanced interactions with phonons at elevated temperatures. However, in magnetic materials, temperature induced changes in magnetic ordering can also affect exciton energy. Intriguingly, the magnitude of the energy shift for excitons B and C decreases with increasing Cu concentration (Fig. 7(d)). This trend suggests that these excitons' interaction with the magnetic ordering of the material may be responsible for the large temperature induced redshift. Accordingly, the differences in the magnitude of the redshift between the different exciton peaks could be explained by differences in the different excitons' tendency to interact with the ordered Mn(II) 3d5 spins that give rise to the AFM order in MnPSe3. Specifically, differences in the orbital character and/or spin state of the excitons would lead to significant differences in how strongly they couple to the magnetic order.59 The large temperature-dependent redshift of excitons B and C requires extensive further work to explore. Namely, continuous temperature-dependent PL and polarization-resolved, magnetic-field-dependent PL to observe the Zeeman splitting of exciton states and further probe the coupling of exciton states to the magnetic Mn ions. Such experiments should be the subject of a separate focused study to sufficiently exclude trivial lattice expansion effects and quantitatively correlate the redshift with M(T). Fig. 8 summarizes the structural and functional changes in MnPSe3 with Cu substitution.
 | (3) |
A close fit of the absorption spectra is obtained utilizing Gaussian functions to fit peaks centered at 2.5 eV and 3.0 eV, energies close to the predicted band-to-band transitions S01 and H01 (Fig. 9). These energies agree reasonably well with peaks observed in the PL spectra. The energy of the S01 peak is lower in the absorption measurement due to band tail defect absorption states that lead to the broadening of the absorption band edge. Exciton peaks were fit with Gaussian functions. A peak centered at 1.86 eV, corresponding to the energy at which exciton A was measured in the PL measurements is observed for MnPSe3 and Mn0.95Cu0.1PSe3 samples. Additionally, a close fit to the absorption spectrum of the Mn0.8Cu0.4PSe3 sample could only be acquired by accounting for peaks associated with the B and C excitons. The reason for the lack of absorption related to the B and C excitons in the MnPSe3 and Mn0.95Cu0.1PSe3 samples is unclear. It is possible that the interference effects observed in these two samples hamper our ability to accurately measure absorption associated with those excitons. Lastly, the optical band gap “Eg-opt” of the MnPSe3 and Mn0.95Cu0.1PSe3 was approximated using the Tauc method to be 2.295 eV and 2.329 eV, respectively (supplementary). This value of Eg-opt for MnPSe3 lines up with previous experimental reports where the Eg-opt was reported to be 2.27 eV.60 Unfortunately, the strong exciton absorption in the Mn0.8Cu0.4PSe3 negated the use of the Tauc method. Aligning with the increase of the optical band gap with Cu-composition, the lowest energy SO1 bulk energy band transition peak is also observed to increase in energy with Cu-composition in both the MnPSe3 and Mn0.95Cu0.1PSe3 samples and continues to increase in the Mn0.8Cu0.4PSe3 sample.
4 Conclusion
We have synthesized and characterized a series of Mn1−xCu2xPSe3 solid solutions (x = 0, 0.05, 0.1, 0.15, and 0.2), demonstrating that Cu substitution introduces systematic structural and functional changes in MnPSe3. X-ray diffraction confirms phase-pure R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) symmetry across the series. Through our structural analysis, we identified that Cu(I) ions are located in a trigonal coordination with Se.
symmetry across the series. Through our structural analysis, we identified that Cu(I) ions are located in a trigonal coordination with Se.
Magnetic susceptibility measurements reveal a continuous suppression of the Néel temperature with increasing Cu content, culminating in a transition from antiferromagnetic order (x ≤ 0.1) to a paramagnetic state down to 2 K for x ≥ 0.15. A modulation of the magnetic exchange parameters is expected due to the observed Cu-substitution-induced lattice expansion, as well as the introduction of Mn vacancies on the honeycomb lattice. A detailed theoretical study is required to elucidate the fundamental mechanism behind these effects.
Photoluminescence spectroscopy identifies three sub-bandgap excitonic features, two of which (B and C) exhibit a redshift with increasing temperature. This redshift is most pronounced in the pristine and x = 0.05 samples, where magnetic ordering remains intact, suggesting strong exciton–spin coupling. Absorption spectroscopy further confirms the sub-bandgap features.
Our results demonstrate that non-magnetic Cu substitution in MnPSe3 can be used to tune both structural and magnetic behavior. This work highlights atomic substitution as a robust design strategy to control coupled spin–optical phenomena in layered magnetic semiconductors, opening new opportunities for tailoring 2D materials for spintronic and quantum optoelectronic applications.
Author contributions
R.C.M. and V.D.N. conceived the project; M.N. and S.L. grew and characterized the samples with help from V.D.N.; M.N. conducted magnetic and thermal measurements; A.B. conducted and analyzed photoluminescence and absorption measurements with help from R.C.M.; X.D. conducted DFT calculations with help from W.W.; A.R. conducted HAADF-STEM measurement with help from D.M.; M.N., A.B. and R.C.M. wrote the manuscript with input from all coauthors.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information (available online) contains additional XRD and magnetometry data, a description of the fitting and analysis of interference fringes in the optical transmission spectra, and details of the electronic structure calculations. Data is available from the corresponding author upon reasonable request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d6ma00397d.
Acknowledgements
This work was supported by the Center for Emergent Materials, a National Science Foundation (NSF) MRSEC, grant number DMR 2011876. S.L. was supported by OSU start-up funds. We thank Prof. Joseph P. Heremans for inspirational guidance and highly fruitful discussions.References
- Z. Ni, A. Haglund, H. Wang, B. Xu, C. Bernhard, D. Mandrus, X. Qian, E. Mele, C. Kane and L. Wu, Imaging the Néel vector switching in the monolayer antiferromagnet MnPSe3 with strain-controlled Ising order, Nat. Nanotechnol., 2021, 16, 782–787 CrossRef CAS PubMed.
- J.-U. Lee, S. Lee, J. H. Ryoo, S. Kang, T. Y. Kim, P. Kim, C.-H. Park, J.-G. Park and H. Cheong, Ising-Type Magnetic Ordering in Atomically Thin FePS3, Nano Lett., 2016, 16, 7433–7438 CrossRef CAS PubMed.
- X. Wang, K. Du, Y. Y. Fredrik Liu, P. Hu, J. Zhang, Q. Zhang, M. H. S. Owen, X. Lu, C. K. Gan, P. Sengupta, C. Kloc and Q. Xiong, Raman spectroscopy of atomically thin twodimensional magnetic iron phosphorus trisulfide (FePS3) crystals, 2D Mater., 2016, 3, 031009 CrossRef.
- B. Huang, G. Clark, E. Navarro-Moratalla, D. R. Klein, R. Cheng, K. L. Seyler, D. Zhong, E. Schmidgall, M. A. McGuire, D. H. Cobden, W. Yao, D. Xiao, P. Jarillo-Herrero and X. Xu, Layer-dependent ferromagnetism in a van der Waals crystal down to the monolayer limit, Nature, 2017, 546, 270–273 CrossRef CAS PubMed.
- A. Bedoya-Pinto, J.-R. Ji, A. K. Pandeya, P. Gargiani, M. Valvidares, P. Sessi, J. M. Taylor, F. Radu, K. Chang and S. S. P. Parkin, Intrinsic 2D-XY ferromagnetism in a van der Waals monolayer, Science, 2021, 374, 616–620 CrossRef CAS PubMed.
- C. Gong, L. Li, Z. Li, H. Ji, A. Stern, Y. Xia, T. Cao, W. Bao, C. Wang, Y. Wang, Z. Q. Qiu, R. J. Cava, S. G. Louie, J. Xia and X. Zhang, Discovery of intrinsic ferromagnetism in twodimensional van der Waals crystals, Nature, 2017, 546, 265–269 CrossRef CAS PubMed.
- M. Bonilla, S. Kolekar, Y. Ma, H. C. Diaz, V. Kalappattil, R. Das, T. Eggers, H. R. Gutierrez, M.-H. Phan and M. Batzill, Strong room-temperature ferromagnetism in VSe2 monolayers on van der Waals substrates, Nat. Nanotechnol., 2018, 13, 289–293 CrossRef CAS PubMed.
- R. Basnet, A. Wegner, K. Pandey, S. Storment and J. Hu, Highly sensitive spin-flop transition in antiferromagnetic van der Waals material MPS3 (M= Ni and Mn), Phys. Rev. Mater., 2021, 5, 064413 CrossRef CAS.
- E. C. Ahn, 2D materials for spintronic devices, npj 2D Mater. Appl., 2020, 4, 1–14 Search PubMed.
- Z. Jia, M. Zhao, Q. Chen, Y. Tian, L. Liu, F. Zhang, D. Zhang, Y. Ji, B. Camargo, K. Ye, R. Sun, Z. Wang and Y. Jiang, Spintronic Devices upon 2D Magnetic Materials and Heterojunctions, ACS Nano, 2025, 19, 9452–9483 CrossRef CAS PubMed.
- M. J. Karaki, X. Yang, A. J. Williams, M. Nawwar, V. Doan-Nguyen, J. E. Goldberger and Y.-M. Lu, An efficient material search for room-temperature topological magnons, Sci. Adv., 2023, 9, eade7731 CrossRef CAS PubMed.
- M. J. Karaki, A. E. Fahmy, A. J. Williams, S. Haravifard, J. E. Goldberger and Y.-M. Lu, High-throughput search for topological magnon materials, arXiv, 2024, preprint, arXiv:2410.18873 DOI:10.48550/arXiv.2410.18873.
- M. Nawwar, R. R. Neumann, J. Wen, A. Mook, I. Mertig and J. P. Heremans, Large Thermal Hall Effect in MnPS3, Rep. Prog. Phys., 2025, 88, 080503 CrossRef CAS PubMed.
- C. Xu, H. Zhang, C. Carnahan, P. Zhang, D. Xiao and X. Ke, Thermal Hall effect in the van der Waals ferromagnet CrI3, Phys. Rev. B, 2024, 109, 094415 CrossRef CAS.
- Q. Meng, X. Li, J. Liu, L. Zhao, C. Dong, Z. Zhu, L. Li and K. Behnia, Thermodynamic origin of the phonon Hall effect in a honeycomb antiferromagnet, arXiv, 2024, preprint, arXiv:2403.13306 DOI:10.48550/arXiv.2403.13306.
- T. T. Mai, K. F. Garrity, A. McCreary, J. Argo, J. R. Simpson, V. Doan-Nguyen, R. V. Aguilar and A. R. H. Walker, Magnonphonon hybridization in 2D antiferromagnet MnPSe3, Sci. Adv., 2021, 7, eabj3106 CrossRef CAS PubMed.
- J. Luo, S. Li, Z. Ye, R. Xu, H. Yan, J. Zhang, G. Ye, L. Chen, D. Hu and X. Teng, et al., Evidence for topological magnon–phonon hybridization in a 2D antiferromagnet down to the monolayer limit, Nano Lett., 2023, 23, 2023–2030 CrossRef CAS PubMed.
- J. H. Zhang, L. Lin, S. H. Zheng, P. Z. Chen, L. Huang, G. Z. Zhou, W. J. Zhai, F. Yu, B. Yu, X. M. Cui, M. F. Liu, Y. S. Tang, Z. B. Yan and J.-M. Liu, Weak ferromagnetism and magnetoelectric coupling in van der Waals antiferromagnet MnPSe3, Appl. Phys. Lett., 2024, 124, 172903 CrossRef CAS.
- M. Rybak, P. E. Faria Junior, T. Wozniak, P. Scharoch, J. Fabian and M. Birowska, Magneto-optical anisotropies of two-dimensional antiferromagnetic PX3 from first principles, Phys. Rev. B, 2024, 109, 054426 CrossRef CAS.
- B. Zheng, J. Fan, B. Chen, X. Qin, J. Wang, F. Wang, R. Deng and X. Liu, Rare-earth doping in nanostructured inorganic materials, Chem. Rev., 2022, 122, 5519–5603 CrossRef CAS PubMed.
- D. Liang, F. Peng, Q. Du, Y. Zhao and W. Ma, Gadolinium and Europium Doped Chiral Perovskite Films with Magnetic Circular Dichroism and Circularly Polarized Luminescence for Information Encryption Applications, Angew. Chem., Int. Ed., 2025, 64, e202510754 CrossRef CAS PubMed.
- J. A. Schiemer, R. L. Withers, Y. Liu and M. A. Carpenter, Cadoping of BiFeO3: the role of strain in determining coupling between ferroelectric displacements, magnetic moments, octahedral tilting, and oxygen-vacancy ordering, Chem. Mater., 2013, 25, 4436–4446 CrossRef CAS PubMed.
- Z. A. VanOrman, M. Cardenes Wuttig, A.-P. M. Reponen, T.-S. Kim, C. E. Casaday, D. Cui, T. Deshpande, H. J. Jobsis, P. Schouwink and E. Oveisi, et al., Metal Doping of Strongly Confined Halide Perovskite Nanocrystals under Ambient Conditions, J. Am. Chem. Soc., 2025, 147, 16536–16544 CrossRef CAS PubMed.
- F. Wang, T. A. Shifa, P. Yu, P. He, Y. Liu, F. Wang, Z. Wang, X. Zhan, X. Lou and F. Xia, et al., New frontiers on van der Waals layered metal phosphorous trichalcogenides, Adv. Funct. Mater., 2018, 28, 1802151 CrossRef.
- M. A. Susner, M. Chyasnavichyus, M. A. McGuire, P. Ganesh and P. Maksymovych, Metal thio-and selenophosphates as multifunctional van der Waals layered materials, Adv. Mater., 2017, 29, 1602852 CrossRef PubMed.
- D. J. Goossens, A. J. Studer, S. J. Kennedy and T. J. Hicks, The impact of magnetic dilution on magnetic order in MnPS3, J. Phys.: Condens. Matter, 2000, 12, 4233 CrossRef CAS.
- Y. He, Y.-D. Dai, H. Huang, J. Lin and Y. Hsia, The ordering distribution of the metal ions in the layered cation-mixed phosphorus trisulfides MnxFe1xPS3, J. Alloys Compd., 2003, 359, 41–45 CrossRef CAS.
- Y. Takano, A. Arai, Y. Takahashi, K. Takase and K. Sekizawa, Magnetic properties and specific heat of new spin glass Mn0.5Fe0.5PS3, J. Appl. Phys., 2003, 93, 8197–8199 CrossRef CAS.
- T. Masubuchi, H. Hoya, T. Watanabe, Y. Takahashi, S. Ban, N. Ohkubo, K. Takase and Y. Takano, Phase diagram, magnetic properties and specific heat of Mn1xFexPS3, J. Alloys Compd., 2008, 460, 668–674 CrossRef CAS.
- D. J. Goossens, S. Brazier-Hollins, D. R. James, W. D. Hutchison and J. R. Hester, Magnetic structure and glassiness in Fe0.5Ni0.5PS3, J. Magn. Magn. Mater., 2013, 334, 82–86 CrossRef CAS.
- J. N. Graham, M. J. Coak, S. Son, E. Suard, J.-G. Park, L. Clark and A. R. Wildes, Local nuclear and magnetic order in the two-dimensional spin glass Mn0.5Fe0.5PS3, Phys. Rev. Mater., 2020, 4, 084401 CrossRef CAS.
- A. Bhutani, J. L. Zuo, R. D. McAuliffe, C. R. dela Cruz and D. P. Shoemaker, Strong anisotropy in the mixed antiferromagnetic system Mn1-xFexPSe3, Phys. Rev. Mater., 2020, 4, 034411 CrossRef CAS.
- S. Lee, J. Park, Y. Choi, K. Raju, W.-T. Chen, R. Sankar and K.-Y. Choi, Chemical tuning of magnetic anisotropy and correlations Ni1-xfexPS3, Phys. Rev. B, 2021, 104, 174412 CrossRef CAS.
- R. Basnet, T. Patel, J. Wang, D. Upreti, S. K. Chhetri, G. Acharya, M. R. U. Nabi, J. Sakon and J. Hu, Understanding and Tuning Magnetism in Layered Ising-Type Antiferromagnet FePSe3 for Potential 2D Magnet, Adv. Electron. Mater., 2024, 10, 2300738 CrossRef CAS.
- A. Wiedenmann, J. Rossat-Mignod, A. Louisy, R. Brec and J. Rouxel, Neutron diffraction study of the layered compounds MnPSe3 and FePSe3, Solid State Commun., 1981, 40, 1067–1072 CrossRef CAS.
- Y. Mathey, R. Clement, J. P. Audiere, O. Poizat and C. Sourisseau, Structural, vibrational and conduction properties of a new class of layer-type MPS3 compounds: MnII1xMI2xPS3 (MI = Cu, Ag), Solid State Ionics, 1983, 9–10, 459–465 CrossRef CAS.
- Y. Mathey, A. Michalowicz, P. Toffoli and G. Vlaic, Resolution of a structural disorder through apparently inconsistent x-ray diffraction and EXAFS data: structure of the new layered system manganese copper phosphorus sulfide (Mn1-xCu2x)PS3 (x = 0.13), Inorg. Chem., 1984, 23, 897–902 CrossRef CAS.
- A. Kuhn, L. M. Schoop, R. Eger, I. Moudrakovski, S. Schwarzmüller, V. Duppel, R. K. Kremer, O. Oeckler and B. V. Lotsch, Copper Selenidophosphates Cu4P2Se6, Cu4P3Se4, Cu4P4Se3, and CuP2Se, Featuring Zero-, One-, and Two-Dimensional Anions, Inorg. Chem., 2016, 55, 8031–8040 CrossRef CAS PubMed.
- P. Juhás, T. Davis, C. L. Farrow and S. J. Billinge, PDFgetX3:a rapid and highly automatable program for processing powder diffraction data into total scattering pair distribution functions, Appl. Crystallogr., 2013, 46, 560–566 CrossRef.
- B. H. Toby and R. B. Von Dreele, GSAS-II: the genesis of a modern open-source all purpose crystallography software package, Appl. Crystallogr., 2013, 46, 544–549 CrossRef CAS.
- N. Fairley, V. Fernandez, M. Richard-Plouet, C. Guillot- Deudon, J. Walton, E. Smith, D. Flahaut, M. Greiner, M. Biesinger and S. Tougaard, et al., Systematic and collaborative approach to problem solving using X-ray photoelectron spectroscopy, Appl. Surf. Sci. Adv., 2021, 5, 100112 CrossRef.
- T. Fleisch and G. Mains, Reduction of copper oxides by UV radiation and atomic hydrogen studied by XPS, Appl. Surf. Sci., 1982, 10, 51–62 CrossRef CAS.
- J. Chastain and R. C. King Jr, Handbook of X-ray photoelectron spectroscopy, PerkinElmer Corporation, 1992, 40, 25.
- P. M. Woodward, P. Karen, J. S. Evans and T. Vogt, Solid state materials chemistry, Cambridge University Press, 2021 Search PubMed.
- I. D. Brown, The chemical bond in inorganic chemistry: the bond valence model, Oxford university press, 2016, vol. 27 Search PubMed.
- C. P. Brock, T. Hahn, H. Wondratschek, U. Müller, U. Shmueli, E. Prince, A. Authier, V. Kopsky, D. Litvin, E. Arnold et al., International tables for crystallography volume A: Space-group symmetry, Wiley Online Library, 2016.
- P. Jeevanandam and S. Vasudevan, Magnetism in MnPSe3:a layered 3d5 antiferromagnet with unusually large XY anisotropy, J. Phys.: Condens. Matter, 1999, 11, 3563 CrossRef CAS.
- S. Calder, A. V. Haglund, A. I. Kolesnikov and D. Mandrus, Magnetic exchange interactions in the van der Waals layered antiferromagnet Mn P Se 3, Phys. Rev. B, 2021, 103, 024414 CrossRef CAS.
- D. J. Goossens and T. J. Hicks, The magnetic phase diagram of, J. Phys.:Condens. Matter, 1998, 10, 7643 CrossRef CAS.
- P. E. Blöchl, Projector augmented-wave method, Phys. Rev. B:Condens. Matter Mater. Phys., 1994, 50, 17953–17979 CrossRef PubMed.
- L. Kronik, T. Stein, S. Refaely-Abramson and R. Baer, Excitation Gaps of Finite-Sized Systems from Optimally Tuned Range-Separated Hybrid Functionals, J. Chem. Theory Comput., 2012, 8, 1515–1531 Search PubMed.
- D. J. Vogel and D. S. Kilin, First-Principles Treatment of Photoluminescence in Semiconductors, J. Phys. Chem. C, 2015, 119, 27954–27964 CrossRef CAS.
- M. M. Ugeda, A. J. Bradley, S.-F. Shi, F. H. da Jornada, Y. Zhang, D. Y. Qiu, W. Ruan, S.-K. Mo, Z. Hussain, Z.-X. Shen, F. Wang, S. G. Louie and M. F. Crommie, Giant bandgap renormalization and excitonic effects in a monolayer transition metal dichalcogenide semiconductor, Nat. Mater., 2014, 13, 1091–1095 CrossRef CAS PubMed.
- J. Huang, T. B. Hoang and M. H. Mikkelsen, Probing the origin of excitonic states in monolayer WSe2, Sci. Rep., 2016, 6, 22414 CrossRef CAS PubMed.
- I. Pollini and G. Spinolo, Intrinsic Optical Properties of CrCl3, Phys. Status Solidi B, 1970, 41, 691–701 CrossRef CAS.
- V. M. Bermudez and D. S. McClure, Spectroscopic studies of the two-dimensional magnetic insulators chromium trichloride and chromium tribromide—I, J. Phys. Chem. Solids, 1979, 40, 129–147 CrossRef CAS.
- K. L. Seyler, D. Zhong, D. R. Klein, S. Gao, X. Zhang, B. Huang, E. Navarro-Moratalla, L. Yang, D. H. Cobden, M. A. McGuire, W. Yao, D. Xiao, P. Jarillo-Herrero and X. Xu, Ligand-field helical luminescence in a 2D ferromagnetic insulator, Nat. Phys., 2018, 14, 277–281 Search PubMed.
- W. Jin, H. H. Kim, Z. Ye, G. Ye, L. Rojas, X. Luo, B. Yang, F. Yin, J. S. A. Horng, S. Tian, Y. Fu, G. Xu, H. Deng, H. Lei, A. W. Tsen, K. Sun, R. He and L. Zhao, Observation of the polaronic character of excitons in a two-dimensional semiconducting magnet CrI3, Nat. Commun., 2020, 11, 4780 Search PubMed.
- N. J. Brennan, C. A. Noble, J. Tang, M. E. Ziebel and Y. J. Bae, Important Elements of Spin-Exciton and Magnon-Exciton Coupling, ACS Phys. Chem. Au, 2024, 4, 322–327 Search PubMed.
- V. Grasso and L. Silipigni, Optical absorption and reflectivity study of the layered MnPSe3 seleniophosphate, J. Opt. Soc. Am. B, 1999, 16, 132–136 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |








