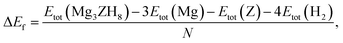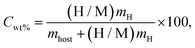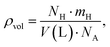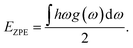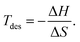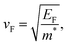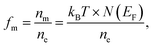Open Access Article
Open Access ArticleExploring structural, electronic, mechanical, and hydrogen storage properties of Mg3ZH8 (Z = Fe, Co): a density functional theory study
Md. Hasan Mia *a,
Md. Zahid Hasan
*a,
Md. Zahid Hasan b,
A. Arunkumar*c and
S. AlFaifyd
b,
A. Arunkumar*c and
S. AlFaifyd
aDepartment of Computer and Communication Engineering, International Islamic University Chittagong, Kumira, Chittagong 4318, Bangladesh. E-mail: mdhasan111.ru@gmail.com
bMaterials Research and Simulation Lab, Department of Electrical and Electronic Engineering, International Islamic University Chittagong, Kumira, Chittagong 4318, Bangladesh
cDepartment of Condensed Matter Physics, Saveetha School of Engineering, Saveetha Institute of Medical and Technical Sciences, SIMATS, Chennai, Tamil Nadu-602105, India. E-mail: arunkumara.phys@gmail.com
dDepartment of Physics, College of Science, King Khalid University, Post Box 960, Abha 61421, AlQura'a, Saudi Arabia
First published on 5th May 2026
Abstract
The development of efficient hydrogen storage materials is crucial for practical hydrogen energy utilization. This study uses first-principles density functional theory (DFT) to examine the structural, electronic, mechanical, optical, and hydrogen storage properties of Mg3ZH8 (Z = Fe, Co) complex hydrides. Both compounds are thermodynamically and dynamically stable, as confirmed by negative formation energies and phonon spectra free of imaginary modes. Mg3FeH8 exhibits a ferromagnetic metallic ground state, while Mg3CoH8 shows non-magnetic metallic behavior, with transition-metal d states playing a key role in their electronic and magnetic properties. The calculated gravimetric hydrogen storage capacities of 5.90 wt% for Mg3FeH8 and 5.76 wt% for Mg3CoH8, along with high volumetric densities of 195.6 and 186.6 g H2 per L, respectively, surpass current U.S. Department of Energy targets. Dehydrogenation thermodynamics show moderate desorption temperatures, with Mg3FeH8 demonstrating better hydrogen release behavior. Mechanical analysis indicates ductility with positive Cauchy pressure, high Pugh's ratios, and moderate Vickers hardness, suggesting resistance to hydrogen embrittlement. Optical and transport properties reveal metallic conductivity, which enhances hydrogen desorption kinetics. These findings position Mg3ZH8 (Z = Fe, Co) as a promising hydrogen storage material and provide a solid theoretical basis for future experimental work.
1. Introduction
Hydrogen energy has come to light as a possible way to solve both the world's energy crisis and pollution problems. Hydrogen is a clean energy carrier that has a high gravimetric energy density and does not release any carbon when used. It is seen as a good replacement for traditional fossil fuels. The increasing demand for clean and sustainable energy has intensified interest in hydrogen as a promising energy carrier due to its high gravimetric energy density and environmentally benign combustion products.1–3 Despite significant progress in hydrogen production and utilization technologies, the lack of safe, efficient, and reversible hydrogen storage materials operating under near-ambient conditions remains a major obstacle to the large-scale deployment of hydrogen-based energy systems. Among the available storage strategies, solid-state hydrogen storage using metal and complex hydrides is considered one of the most promising approaches because of its high volumetric density, enhanced safety, and compactness compared to compressed or liquefied hydrogen. To guide the development of practical hydrogen storage materials, the U.S. Department of Energy (DOE)4 has established performance targets for onboard storage systems, including a gravimetric hydrogen capacity exceeding 5.5 wt%, a volumetric capacity above 40 g H2 per L, operating temperatures in the range of 233–333 K, and moderate pressures below 10 MPa. Achieving these targets simultaneously remains challenging, as many known hydrides exhibit either excessively strong metal–hydrogen bonding that leads to high desorption temperatures or insufficient thermodynamic stability that compromises reversibility and cycling performance.In recent years, complex and perovskite-type hydrides have emerged as an important class of hydrogen storage materials because their flexible crystal chemistry enables systematic tuning of structural, electronic, and thermodynamic properties. Numerous categories of complex hydrides, such as borohydrides, amides, and alanates, have undergone extensive research for hydrogen storage purposes. Alkali alanates MAlH4 (M = Li, Na, K) have garnered significant attention owing to their relatively high hydrogen density and reversible hydrogen absorption/desorption characteristics.5 First-principles investigations of MgVH6 and MgSiH6 have demonstrated chemical stability, pressure-dependent phase behaviour, and notable electronic properties, such as metallic characteristics and superconductivity under extreme conditions, underscoring the adaptability of hexahydride systems.6,7 Concurrently, double perovskite hydrides have become attractive options for multipurpose uses. For example, KNaMg2F6−xHx and KNaAe2H6 (Ae = Be, Mg, Ca) show semiconducting behaviour with ultraviolet absorption capacity and favourable desorption temperatures.8 Furthermore, large hydrogen capacities and favourable formation energetics have been anticipated for a number of hexahydrides, such as Q2FeH6 (Q = Mg, Ca, Sr),9 Mg2XH6 (X = Cr, Mn),10 A2LiCuH6 (A = Be, Mg, Ca, Sr),11 KNaX2H6 (X = Mg, Ca, and X2CaCdH6 (X = Rb, Cs).12 Similarly, indirect band gap semiconductors with the capacity to store hydrogen have been found as A2BH6 (A = K, Rb; B = Ge, Sn) hydrides.13 Additionally, it has been demonstrated that compositional tweaking, such as lithium doping in Na-based perovskites, improves photocatalytic activity and optoelectronic response.14 Numerous first-principles studies have demonstrated promising hydrogen storage performance in alkaline-earth hydrides systems. For example, DFT investigations of alkali and alkaline-earth hydrides, including AMAlH4 (AM = Li, Na, K and Rb),15 XNH6 (X = Li, Na, K),16 NaXH3 (X = Be, Mg, Ca, Sr),17 and RbXH3 (X = Mg, Ca, Sr, Ba),18 have reported high gravimetric hydrogen capacities and favorable formation energetics. These investigations also emphasize the critical role of lattice environment, electronic structure, and bonding characteristics in determining hydrogen stability and release behavior. First-principles studies of Mg3XH8 (X = Ca, Sc, Ti, V, Cr, Mn) hydrides have shown that transition-metal substitution strongly affects lattice stability, hydrogen binding strength, and mechanical behavior.19 These modifications lead to favorable gravimetric hydrogen densities and moderate desorption temperatures.
Despite these advances, Mg3ZH8 (Z = Fe, Co) hydrides containing late transition metals such as Fe and Co have not yet been systematically explored. Compared with early transition metals, Fe and Co possess more localized d electrons, which can significantly modify magnetic behavior, electronic conductivity, bonding character, and metal–hydrogen interactions. Understanding how these factors affect hydrogen storage thermodynamics, kinetics, and mechanical durability is essential for expanding the design space of Mg-based complex hydrides. Moreover, experimental data for Mg3ZH8 (Z = Fe, Co) systems are currently unavailable, underscoring the need for reliable theoretical benchmarks. In this work, we present a comprehensive first-principles investigation of Mg3ZH8 (Z = Fe, Co) complex hydrides. Structural stability, electronic and magnetic properties, mechanical and thermophysical behavior, optical response, and hydrogen storage characteristics are systematically examined. Multiple dehydrogenation pathways are analyzed to evaluate reaction energetics and hydrogen desorption temperatures. By directly comparing Fe- and Co-based systems, the role of late transition-metal substitution in tailoring hydrogen storage performance is clarified. The present study provides the first detailed theoretical benchmark for Mg3ZH8 (Z = Fe, Co) hydrides and offers guidance for future experimental synthesis and optimization.
2. Computational details
The hydrogen storage properties, thermo-mechanical, opto-electronic, and magnetic nature of Mg3ZH8 (Z = Fe, Co) hydrides were investigated based on density functional theory (DFT) using the CASTEP code.20 Vanderbilt ultrasoft pseudopotentials were employed to describe the interaction between valence electrons and ionic cores. The exchange–correlation energy was treated using the generalized gradient approximation (GGA) with the Perdew–Burke–Ernzerhof (PBE) functional.21 Mg3ZH8 (Z = Fe, Co) hydrides crystallize in a cubic structure with space group Pm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m (no. 221), belonging to the m
m (no. 221), belonging to the m![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m point group. In the optimized structures, the transition metal atom (Fe or Co) occupies the 1a Wyckoff position at (1/2, 1/2, 0). Mg atoms are located at the 3c site with fractional coordinates (0, 0, 0). H atoms occupy the 8g Wyckoff position at (0.243175, 0.243175, 0.756825). These atomic positions were fully relaxed during geometry optimization. The optimized lattice parameters were found to be a = b = c = 4.09 Å for Mg3FeH8 and 4.15 Å for Mg3CoH8, corresponding to unit cell volumes of 68.39 Å3 and 71.68 Å3, respectively as shown in Table 1. Geometry optimizations were performed using the BFGS minimization algorithm.22 The convergence criteria were set to 5 × 10−6 eV per atom for total energy, 0.01 eV Å−1 for maximum ionic force, 5 × 10−4 Å for maximum atomic displacement, and 0.02 GPa for maximum stress. All calculations were carried out at 0 K and zero external pressure. A Monkhorst–Pack k-point mesh23 of 6 × 6 × 6 was used for Brillouin zone integration. The plane-wave cutoff energy was fixed at 630 eV. A smearing width of 0.5 eV was applied to improve electronic convergence. Elastic constants were evaluated to determine the mechanical properties. The elastic moduli of the polycrystalline systems were derived using the Voigt–Reuss–Hill approximation.24,25 Electronic band structures and optical properties were calculated in reciprocal space using fully optimized crystal geometries. Optical properties were obtained within the CASTEP code through evaluation of the complex dielectric function. Phonon dispersion calculations for Mg3ZH8 (Z = Fe, Co) were performed using the finite-displacement method with ultrasoft pseudopotentials. A 4 × 4 × 4 Monkhorst–Pack k-point mesh was employed to ensure accurate force-constant determination and phonon convergence. Plane-wave energy cutoffs of 850 eV for Mg3FeH8 and 950 eV for Mg3CoH8 were used, and all computational parameters were carefully tested to obtain converged phonon spectra without imaginary frequencies.
m point group. In the optimized structures, the transition metal atom (Fe or Co) occupies the 1a Wyckoff position at (1/2, 1/2, 0). Mg atoms are located at the 3c site with fractional coordinates (0, 0, 0). H atoms occupy the 8g Wyckoff position at (0.243175, 0.243175, 0.756825). These atomic positions were fully relaxed during geometry optimization. The optimized lattice parameters were found to be a = b = c = 4.09 Å for Mg3FeH8 and 4.15 Å for Mg3CoH8, corresponding to unit cell volumes of 68.39 Å3 and 71.68 Å3, respectively as shown in Table 1. Geometry optimizations were performed using the BFGS minimization algorithm.22 The convergence criteria were set to 5 × 10−6 eV per atom for total energy, 0.01 eV Å−1 for maximum ionic force, 5 × 10−4 Å for maximum atomic displacement, and 0.02 GPa for maximum stress. All calculations were carried out at 0 K and zero external pressure. A Monkhorst–Pack k-point mesh23 of 6 × 6 × 6 was used for Brillouin zone integration. The plane-wave cutoff energy was fixed at 630 eV. A smearing width of 0.5 eV was applied to improve electronic convergence. Elastic constants were evaluated to determine the mechanical properties. The elastic moduli of the polycrystalline systems were derived using the Voigt–Reuss–Hill approximation.24,25 Electronic band structures and optical properties were calculated in reciprocal space using fully optimized crystal geometries. Optical properties were obtained within the CASTEP code through evaluation of the complex dielectric function. Phonon dispersion calculations for Mg3ZH8 (Z = Fe, Co) were performed using the finite-displacement method with ultrasoft pseudopotentials. A 4 × 4 × 4 Monkhorst–Pack k-point mesh was employed to ensure accurate force-constant determination and phonon convergence. Plane-wave energy cutoffs of 850 eV for Mg3FeH8 and 950 eV for Mg3CoH8 were used, and all computational parameters were carefully tested to obtain converged phonon spectra without imaginary frequencies.
| Compounds | a = b = c | V | ΔEfor | ρ(g H2 per L) | Cwt% | Tdes (K) |
|---|---|---|---|---|---|---|
| Mg3FeH8 | 4.09 | 68.39 | −0.264 | 195.6 | 5.90 | 194.74 |
| Mg3CoH8 | 4.15 | 71.68 | −0.220 | 186.6 | 5.76 | 162.57 |
| Mg3ScH8 (ref. 19) | 4.73 | — | −0.240 | 197 | 6.36 | |
| Mg3TiH8 (ref. 19) | 4.61 | — | −0.250 | 218 | 6.21 | |
| Mg3VH8 (ref. 19) | 4.54 | — | −0.230 | 234 | 6.07 | |
| Mg3CaH8 (ref. 19) | 4.90 | — | −0.100 | 171 | 6.61 | |
| Mg3CrH8 (ref. 19) | 4.50 | — | −0.280 | 242 | 6.02 | |
| Mg3MnH8 (ref. 19) | 4.47 | — | −0.190 | 252 | 5.89 | |
| Mg3MnH7 (ref. 26) | — | — | — | — | 5.2expt. | 530–640expt. |
| Mg2FeH6 (ref. 27) | — | — | — | — | 5.5expt. | 603expt. |
| MgH2 (ref. 28) | — | — | — | — | 7.1expt. | 623–673expt. |
3. Results and discussion
3.1 Phase stability and gravimetric properties
The optimized crystal structures and dynamical stability profiles of the Mg-based hydrides Mg3ZH8 (Z = Fe, Co) are shown in Fig. 1(a)–(c). Both compounds crystallize in a highly hydrogen-rich framework, where the choice of transition-metal cation at the Z site plays a decisive role in determining lattice geometry and stability. Table 1 summarizes the optimized structural parameters, volumetric and gravimetric hydrogen storage capacities of Mg3ZH8 (Z = Fe, Co) in comparison with previously reported Mg3ZH8-type hydrides. The calculated lattice parameters for Mg3FeH8 (a = 4.09 Å) and Mg3CoH8 (a = 4.15 Å) are smaller than those of Mg3ScH8, Mg3TiH8, and Mg3CaH8, reflecting the reduced atomic radii of Fe and Co and the resulting lattice contraction.19 This compact crystal structure directly contributes to enhanced volumetric hydrogen storage performance. | ||
| Fig. 1 (a)–(c) The phonon dispersion with three-dimensional structure of Mg based Mg3ZH8 (Z = Fe, Co) hydrides. | ||
Thermodynamic stability was evaluated by calculating the formation enthalpy (ΔEf), defined as,
Dynamical stability was further assessed through phonon dispersion calculations, as shown in Fig. 1(a) and (b). The phonon characteristics reveal how hydrogen-rich stoichiometry and metal–hydrogen bonding collectively stabilize the lattice while enabling high gravimetric hydrogen capacity. The phonon spectra of both hydrides exhibit three acoustic branches and multiple optical modes, with no imaginary frequencies observed throughout the Brillouin zone. This confirms that Mg3ZH8 (Z = Fe, Co) are dynamically stable and free from lattice instabilities. The optical phonon modes at higher frequencies are dominated by hydrogen vibrations, reflecting the strong metal–hydrogen bonding and hydrogen-rich nature of the lattice. A pronounced frequency gap between acoustic and optical phonon branches is observed in both compounds. This gap originates from the large mass contrast between light hydrogen atoms and heavier metal atoms (Mg and Z), which leads to well-separated low-frequency lattice translations and high-frequency hydrogen-dominated vibrations. The wider acoustic–optical gap observed in Mg3FeH8 compared to Mg3CoH8 reflects stronger Fe–H bonding and a stiffer local hydrogen environment, consistent with its more negative formation energy and higher lattice rigidity.
The ab initio molecular dynamics (AIMD) simulations for Mg3FeH8 and Mg3CoH8 were performed using a 2 × 2 × 2 supercell with a timestep of 1 fs in the NVT ensemble at 300 K, after an initial equilibration phase. The simulations ran for 5000 fs, providing sufficient sampling of the atomic dynamics. The Fig. 2(a) and (b) show the temperature evolution of both hydrides during the simulations, with the temperature fluctuating around 300 K. The temperature was analyzed using the kinetic energy relation: 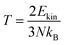 ; where Ekin is the average kinetic energy, N is the number of atoms, and kB is Boltzmann's constant. In these simulations, the temperature fluctuations around 300 K indicate dynamic stability for both hydrides, as neither system exhibits significant deviations from the target temperature. The stable behavior of the temperature profiles suggests that both Mg3FeH8 and Mg3CoH8 maintain their structural integrity and dynamic stability at room temperature, making them suitable candidates for hydrogen storage applications under practical conditions. The time evolution of the total energy for Mg3FeH8 and Mg3CoH8 systems is presented in Fig. 2(c) and (d). In both cases, the energy exhibits a slight upward drift over the simulation time of 5000 fs. For Mg3FeH8, the energy increases from −3695.85376 eV to −3691.13808 eV, corresponding to a relative variation of approximately 0.128%. Similarly, Mg3CoH8 shows an increase from −4229.13344 eV to −4224.47209 eV, yielding a relative variation of about 0.110%. Despite these small increases, the overall fluctuations remain minimal compared to the total energy scale, indicating good stability of the simulations. The slightly lower variation observed in Mg3CoH8 suggests marginally improved energy conservation compared to Mg3FeH8.
; where Ekin is the average kinetic energy, N is the number of atoms, and kB is Boltzmann's constant. In these simulations, the temperature fluctuations around 300 K indicate dynamic stability for both hydrides, as neither system exhibits significant deviations from the target temperature. The stable behavior of the temperature profiles suggests that both Mg3FeH8 and Mg3CoH8 maintain their structural integrity and dynamic stability at room temperature, making them suitable candidates for hydrogen storage applications under practical conditions. The time evolution of the total energy for Mg3FeH8 and Mg3CoH8 systems is presented in Fig. 2(c) and (d). In both cases, the energy exhibits a slight upward drift over the simulation time of 5000 fs. For Mg3FeH8, the energy increases from −3695.85376 eV to −3691.13808 eV, corresponding to a relative variation of approximately 0.128%. Similarly, Mg3CoH8 shows an increase from −4229.13344 eV to −4224.47209 eV, yielding a relative variation of about 0.110%. Despite these small increases, the overall fluctuations remain minimal compared to the total energy scale, indicating good stability of the simulations. The slightly lower variation observed in Mg3CoH8 suggests marginally improved energy conservation compared to Mg3FeH8.
In this study, the hydrogen storage potential of Mg3ZH8 (Z = Fe, Co) hydrides was evaluated by calculating the gravimetric hydrogen capacity using,29
 | ||
| Fig. 3 (a) Gravimetric capacity of Mg3ZH8 (Z = Fe, Co), and (b) comparison of gravimetric hydrogen storage capacities of Mg3ZH8 (Z = Fe, Co) with previously reported complex hydrides. | ||
Since there are no previously reported experimental or theoretical gravimetric hydrogen storage capacities for Mg3ZH8 (Z = Fe, Co), Fig. 3(b) presents a comparative analysis with a range of well-established complex hydrides. Both Mg3FeH8 and Mg3CoH8 exhibit among the highest gravimetric hydrogen storage capacities, surpassing or closely matching widely studied Mg-based complex hydrides such as Mg2NiH6 (5.32 wt%),31 Mg2CoH6 (5.29 wt%),31 Mg2CrH6 (5.60 wt%),10 and Mg2VH6 (5.72 wt%).32 Their performance also exceeds that of several alkali- and alkaline-earth-stabilized hydrides, including, Mn3NaH8 (4.12 wt%),33 Mn3KH8 (3.80 wt%),33 K2LiAlH6 (5.08 wt%),34 Ca2VH6 (4.41 wt%),32 Rb2NaGaH6 (1.48 wt%),35 and KNaMg2H6 (5.19 wt%).8 This comparison demonstrates that Mg3ZH8 (Z = Fe, Co) hydrides combine high hydrogen content with a lightweight Mg-based framework, positioning them among the most competitive solid-state hydrogen storage materials reported to date and underscoring their scientific relevance and potential for practical hydrogen energy applications. Our study aims to provide foundational insights into the hydrogen storage capacities of Mg3FeH8 and Mg3CoH8 at the material level. System-level performance would require experimental validation and simulations that account for the integration of these materials into practical storage systems, which is a necessary next step in future research.
In addition to gravimetric hydrogen storage capacity, the volumetric hydrogen storage capacity (ρvol) is a critical parameter for evaluating the practical applicability of solid-state hydrogen storage materials, particularly for space-constrained applications such as onboard storage systems. The volumetric capacity quantifies the amount of hydrogen that can be stored per unit volume of the material and is expressed in g H2 per L. Following the methodology adopted in the referenced hydrogen-storage studies, the volumetric hydrogen storage capacity is calculated using the relation:29
A key parameter for hydrogen storage applications is the hydrogen desorption temperature, Tdes, which defines the thermal conditions required for hydrogen release. Following the approach proposed by Ikeda et al.,36 the hydrogen decomposition reaction for Mg3ZH8 (Z = Fe, Co) hydrides is expressed as: Mg3ZH8 → 3Mg + Z + 4H2.
Based on this reaction, the hydrogen decomposition enthalpy (ΔH) is calculated as:
| ΔH = 3HMg + HZ + 4HH2 − HMg3ZH8 |
| H = Eele + EZPE |
where h, ω, and g(ω) are Planck's constant, phonon frequency, and phonon density of states, respectively.
The hydrogen desorption temperature (Tdes) is estimated using:
The hydrogen diffusion behavior in Mg3FeH8 and Mg3CoH8 is analyzed using the activation energy profiles shown in Fig. 4(a) and (b). In hydrogen storage materials, atomic migration is governed by the activation energy barrier, where lower values facilitate easier hydrogen transport and improved absorption/desorption kinetics. The diffusion pathways were evaluated using the nudged elastic band (NEB) method, which determines the minimum energy path and corresponding barriers.37 Two possible diffusion pathways (path 1 and path 2) are considered for both compounds. For Mg3FeH8, the activation energies are 0.45 eV for path 1 and 1.69 eV for path 2, indicating that hydrogen migration is more favorable along path 1. In contrast, Mg3CoH8 exhibits lower barriers of 0.20 eV for path 1 and 1.39 eV for path 2, suggesting enhanced hydrogen mobility compared to Mg3FeH8. The relatively low activation energy along the preferred pathway, particularly in Mg3CoH8, indicates efficient hydrogen diffusion within the lattice. Overall, these results confirm that hydrogen transport is not kinetically hindered and support the suitability of these materials for hydrogen storage applications.
 | ||
| Fig. 4 Hydrogen diffusion energy profiles for (a) Mg3FeH8 and (b) Mg3CoH8 along two possible migration pathways (path 1 and path 2) calculated using the NEB method. | ||
3.2 Electronic properties
Hydrogen absorption and desorption kinetics in solid-state hydrides are strongly influenced by their electronic structure, as the availability of electronic states near the Fermi level governs charge transfer, metal–hydrogen bonding, and hydrogen diffusion processes. Perovskite and complex hydrides have therefore attracted significant attention as promising hydrogen storage materials. In this section, the electronic properties of Mg3ZH8 (Z = Fe, Co) complex hydrides are investigated through a detailed analysis of their electronic band structures, total density of states (TDOS), and partial density of states (PDOS). Fig. 5(a) and 6(a) display the calculated electronic band structures of Mg3FeH8 and Mg3CoH8, where the horizontal dashed line represents the Fermi level (EF). The band structures were obtained within the GGA-PBE framework over an energy window from −6.0 to 6.0 eV, with EF set to 0 eV. The Kohn–Sham eigenvalues were plotted along the high-symmetry directions X–R–M–Γ–R of the first Brillouin zone. Both Mg3FeH8 and Mg3CoH8 exhibit multiple electronic bands crossing the Fermi level, indicating the absence of a band gap and confirming their metallic ground state. The band dispersion near EF provides further insight into their electronic transport behavior. Several bands crossing EF display steep slopes along specific high-symmetry directions, reflecting strong orbital overlap and enhanced electronic delocalization. Mg3FeH8 shows relatively steeper band curvature near EF compared to Mg3CoH8, suggesting a smaller carrier effective mass and higher carrier mobility. This behavior is primarily attributed to stronger Fe (3d)–H (1s) hybridization, whereas Mg3CoH8 exhibits slightly flatter bands due to comparatively weaker Co-3d contributions. | ||
| Fig. 5 (a) and (b) The electronic band structure with partial and total density of states of Mg3FeH8 complex hydride. | ||
 | ||
| Fig. 6 (a) and (b) The electronic band structure with partial and total density of states of Mg3CoH8 complex hydride. | ||
The steep dispersion of electronic bands near EF implies a reduced carrier effective mass and, consequently, a higher Fermi velocity, given by38,39
where nm is the number of thermally excited electrons, ne is the total number of valence electrons, and N(EF) is the TDOS at the Fermi level. The calculated values of fm for Mg3ZH8 (Z = Fe, Co) are shown in Table 2. Since, fm is directly proportional to N(EF), Mg3FeH8 exhibits a higher metallic fraction at room temperature than Mg3CoH8, implying a larger population of mobile charge carriers. The metallic conductivity, enhanced carrier mobility, and appreciable metallic fraction of Mg3ZH8 (Z = Fe, Co) hydrides have important implications for hydrogen storage applications. Metallic electronic behavior facilitates efficient charge transfer and heat dissipation during hydrogen absorption and desorption cycles, reducing kinetic barriers and localized thermal buildup.
| Compound | TDOS at EF (states per eV) | Metallic fraction fm (300 K) | Metallic strength | Dominant orbital at EF |
|---|---|---|---|---|
| Mg3FeH8 | ∼5.43 | ∼7.3 × 10−3 | Strong metallic | Fe-3d with H-1s hybridization |
| Mg3CoH8 | ∼3.91 | ∼4.4 × 10−3 | Moderate metallic | Co-3d with H-1s contribution |
Fig. 5(b) and 6(b) display the partial and total density of states of Mg3FeH8 and Mg3CoH8 hydrides. The total and partial density of states (TDOS and PDOS) of Mg3ZH8 (Z = Fe, Co) clarify the orbital contributions governing the valence and conduction regions. For both Mg3FeH8 and Mg3CoH8, the TDOS shows a finite value at the Fermi level, confirming their metallic nature. States near EF are dominated by transition-metal d orbitals, with Fe-3d states contributing most strongly in Mg3FeH8 and Co-3d states in Mg3CoH8. This dominant d-state presence explains the band crossings at EF and the high carrier density. In the valence band region, extending from about −6 eV up to the Fermi level, the DOS is mainly composed of hybridized metal d and hydrogen 1s states, indicating strong metal–hydrogen bonding. The sharp d-orbital peaks just below EF reflect localized d electrons, while the broader H-1s contribution at lower energies signifies covalent interaction with the metal framework. Magnesium s and p states contribute weakly in this region, acting mainly as a charge donor to the metal–hydrogen network. In the conduction band region above EF, the DOS is again dominated by metal d states with minor contributions from Mg-p orbitals, indicating that electronic transport is primarily governed by the transition-metal sublattice. The similarity in DOS distribution for Fe- and Co-based systems suggests that Z-site substitution mainly tunes the intensity and energy position of d-state peaks rather than altering the overall electronic character. This d-H hybridization near the Fermi level is crucial for metallic conductivity and supports efficient charge transfer and lattice screening, which are favorable for hydrogen diffusion and desorption kinetics.
Further, the electronic band structures of Mg3FeH8 and Mg3CoH8 were calculated using the HSE06 functional. As shown in Fig. 7(a) and (b), both compounds exhibit metallic behavior. This is confirmed by the presence of several energy bands crossing the Fermi level. This indicates the absence of a band gap and confirms the availability of free charge carriers in both systems. In Mg3FeH8, the bands near the Fermi level show relatively stronger dispersion compared to Mg3CoH8, suggesting enhanced electronic conductivity due to more pronounced hybridization between Fe 3d-states and H 1s-states. In contrast, Mg3CoH8 displays slightly flatter bands in certain regions, indicating comparatively localized electronic states. The overall metallic nature of both compounds is beneficial for hydrogen storage applications, as it promotes efficient charge transfer and heat dissipation, which are essential for improving hydrogen absorption and desorption kinetics.
 | ||
| Fig. 7 Electronic band structures of (a) Mg3FeH8 and (b) Mg3CoH8 calculated using the HSE06 hybrid functional. The Fermi level is set at 0 eV (green dashed line). | ||
3.3 Magnetic properties
To clarify the magnetic characteristics of Mg3ZH8 (Z = Fe, Co), spin-polarized electronic band structure calculations were performed within the PBE–GGA framework, as presented in Fig. 8(a) and (b). The band structures are resolved into spin-up (red) and spin-down (blue) channels. Fig. 8(a) shows the spin-polarized electronic band structure of Mg3FeH8, where a pronounced asymmetry between the spin-up and spin-down bands is observed near the Fermi level. Several Fe-derived bands cross EF in only one spin channel, indicating strong exchange splitting of the Fe 3d states. This spin-dependent band dispersion leads to unequal occupation of the two spin channels and results in a finite net magnetic moment, confirming the ferromagnetic ground state of Mg3FeH8. The presence of magnetism in Mg3FeH8 is associated with stronger Fe–H hybridization, which can influence hydrogen bonding strength and thermodynamic stability. In contrast, the spin-polarized band structure of Mg3CoH8 in Fig. 8(b) exhibits nearly identical spin-up and spin-down bands throughout the Brillouin zone. No exchange splitting is observed at the Fermi level, and both spin channels show the same band crossings. This spin degeneracy indicates zero net spin polarization, confirming that Mg3CoH8 adopts a non-magnetic ground state.Fig. 9(a)–(c) shows the spin-polarized PDOS of Mg3FeH8 hydride. A clear asymmetry between the spin-up and spin-down channels is observed, most prominently in the Fe-3d states near the Fermi level (EF). This exchange splitting leads to unequal occupation of the two spin channels and gives rise to a finite net magnetic moment, confirming the ferromagnetic nature of the compound. The Mg-s and p states exhibit nearly symmetric spin-up and spin-down distributions around the Fermi level, indicating a negligible contribution to the magnetic moment and a primarily ionic role. In contrast, the Fe-s and p states exhibit clear spin asymmetry near EF, induced by exchange splitting from Fe-d orbitals, confirming the ferromagnetic nature of the compound. The H-s states display weak induced spin polarization due to Fe–H hybridization, reflecting an indirect contribution to the overall ferromagnetic ground state. Fig. 10(a)–(c) presents the spin-polarized PDOS of Mg3CoH8. The spin-up and spin-down components of the Mg (s, p), Co (s, p, d), and H (s) orbitals are nearly identical across the entire energy range, and no noticeable exchange splitting is observed at the Fermi level (EF). This spin symmetry results in zero net spin polarization, confirming that Mg3CoH8 adopts a non-magnetic ground state, in agreement with the spin-polarized band structure shown in Fig. 8(b). The absence of magnetism can be attributed to the nearly filled and weakly spin-polarized Co-3d states, which are insufficient to support exchange-driven magnetic ordering.
The presence of ferromagnetism in Mg3FeH8 is associated with stronger Fe–H hybridization and enhanced bonding anisotropy, which can influence hydrogen stability, whereas the non-magnetic character of Mg3CoH8 reflects more uniform Co–H bonding and comparatively weaker exchange-driven stabilization.
3.4 Optical properties
The optical response of Mg3ZH8 (Z = Fe, Co) hydrides provides insight into their electronic behavior and its relevance to hydrogen storage. The dielectric function,40 ε(ω) = ε1(ω) + iε2(ω), reveals strongly negative ε1(ω) at low photon energies, confirming Drude-like metallic behavior and efficient free-carrier screening (Fig. 11(a)). This facilitates rapid charge redistribution and heat dissipation during hydrogen absorption–desorption cycles. Mg3FeH8 exhibits a slightly stronger response than Mg3CoH8, consistent with enhanced carrier mobility and stronger d-H hybridization. The large ε2(ω) values in the low-energy region indicate strong absorption arising from interband transitions between transition-metal d states and H-1s orbitals (Fig. 11(a)). The absorption spectra further show strong absorptivity across the UV-visible range, with Mg3FeH8 displaying slightly higher absorption (Fig. 11(b)). The presence of absorption from low photon energies reflects efficient electronic excitations that support charge transfer and improve hydrogen desorption kinetics. Prominent UV peaks (∼5 eV) originate from valence–conduction band transitions, indicating active electronic participation in hydrogen-related processes. The optical conductivity confirms metallic transport behavior, with high σ1(ω) values indicating efficient charge transport and enhanced carrier dynamics (Fig. S1). The dispersive σ2(ω) behavior reflects strong carrier screening, characteristic of metallic systems (Fig. S1). These features are beneficial for facilitating hydrogen diffusion and improving reversibility during cycling. High reflectivity at low energies further supports the metallic nature and strong free-carrier response (Fig. 11(c)). Its decrease at higher energies corresponds to interband transitions, consistent with the absorption behavior. The refractive index n(ω) and extinction coefficient k(ω) of Mg3FeH8 and Mg3CoH8 indicate strong light–matter interaction and metallic behavior (Fig. S2). High n(ω) at low energies and finite k(ω) confirm efficient charge transfer, supporting hydrogen desorption kinetics. The increase of k(ω) in the visible region reflects enhanced absorption and improved heat management during hydrogen cycling. Overall, the combined dielectric, absorption, conductivity, and refractive index responses confirm strong electronic activity and metallic behavior in Mg3ZH8 (Z = Fe, Co), which promote efficient charge transfer, thermal transport, and hydrogen desorption kinetics. These features reinforce the suitability of both hydrides for stable and efficient hydrogen storage. | ||
| Fig. 11 Optical properties of Mg3ZH8 (Z = Fe, Co): (a) dielectric function, (b) absorption coefficient, and (c) reflectivity as a function of photon energy. | ||
3.5 Thermo-mechanical and anisotropic nature
Elastic constants provide fundamental insight into the mechanical stability, lattice rigidity, and hydrogen accommodation capability of solid-state hydrogen storage materials. In hydrogen-rich hydrides, appropriate elastic behavior is essential to tolerate repeated hydrogen absorption–desorption cycles, which induce volumetric expansion and local lattice distortion. Insufficient stiffness may lead to structural degradation, while excessively rigid lattices can hinder hydrogen diffusion and release kinetics. The mechanical stability of cubic Mg3ZH8 (Z = Fe, Co) hydrides was evaluated using the Born–Huang stability criteria,41 expressed as C11 − C12 > 0, C44 > 0, C11 > 0, and C11 + 2C12 > 0. The calculated elastic constants summarized in Table 3 satisfy all stability conditions, confirming that both Mg3FeH8 and Mg3CoH8 are mechanically stable.| Compound | Elastic constants | Elastic moduli | Elastic anisotropy indices | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| C11 | C12 | C44 | B | G | E | A | AB | AG | AU | |
| Mg3FeH8 | 296.53 | 150.80 | 58.22 | 199.38 | 63.69 | 172.69 | 0.799 | 0.000 | 0.006 | 0.061 |
| Mg3CoH8 | 182.34 | 49.57 | 36.01 | 93.82 | 46.12 | 118.87 | 0.542 | 0.000 | 0.044 | 0.463 |
| Mg7TiH16 (ref. 44) | 125.81 | 18.55 | 16.33 | 82.31 | 31.45 | 84.27 | — | — | — | — |
| Mg7FeH16 (ref. 44) | 137.92 | 28.3 | 22.71 | 89.76 | 35.82 | 95.41 | — | — | — | — |
| K2LiAlH6 (ref. 34) | 60.2 | 18.5 | 19.2 | 32.18 | 13.98 | 37.86 | — | — | — | — |
| LiCaCoH6 (ref. 45) | 107.02 | 29.05 | 39.12 | 67.54 | 39.12 | 101.02 | — | — | — | — |
| K2LiTiH6 (ref. 46) | 43.68 | 18.12 | 28.18 | 26.84 | 17.89 | 45.76 | — | — | — | — |
| Ca2LiTiH6 (ref. 46) | 108.83 | 23.40 | 34.92 | 71.63 | 35.44 | 92.13 | — | — | — | — |
Table 3 indicates Mg3FeH8 exhibits significantly higher stiffness than Mg3CoH8, with C11 = 296.53 GPa and C12 = 150.80 GPa, compared to C11 = 182.34 GPa and C12 = 49.57 GPa for Mg3CoH8. This indicates a much stronger resistance to longitudinal compression in the Fe-based lattice. The shear response follows the same trend, with C44 = 58.22 GPa for Mg3FeH8 and 36.01 GPa for Mg3CoH8, suggesting greater tolerance against shape distortions during hydrogen insertion and removal. Furthermore, the tetragonal shear parameter,42 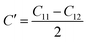 , is positive for both compounds (≈72.87 GPa for Mg3FeH8 and ≈66.39 GPa for Mg3CoH8), confirming stability against shear-driven lattice instabilities.
, is positive for both compounds (≈72.87 GPa for Mg3FeH8 and ≈66.39 GPa for Mg3CoH8), confirming stability against shear-driven lattice instabilities.
As there are no previously reported experimental or theoretical elastic data for Mg3ZH8 (Z = Fe, Co) hydrides, a comparison with other complex and double-perovskite hydrides is provided in Table 3. Reported A2BH6-type and double-perovskite hydrides typically exhibit moderate stiffness, with C11 values in the range of ∼44–138 GPa (e.g., K2LiAlH6, LiCaCoH6, Mg7TiH16-based systems). In contrast, Mg3ZH8 (Z = Fe, Co) shows substantially higher elastic stiffness, with C11 reaching nearly 300 GPa for Mg3FeH8, indicating an unusually rigid hydrogen-rich framework. This distinct combination of high elastic resilience and high hydrogen content differentiates Mg3ZH8 (Z = Fe, Co) from previously reported complex and perovskite hydrides,34,43–45 underscoring its scientific novelty and suitability for durable solid-state hydrogen storage under repeated cycling conditions.
The elastic response of Mg3ZH8 (Z = Fe, Co) is closely linked to its lattice vibrational behavior, providing a direct connection between mechanical stiffness and phonon dynamics. Higher elastic constants, particularly C11 and C44, correspond to increased sound velocities and stiffer acoustic phonon branches, which enhance lattice stability and suppress low-frequency soft modes. In Mg3FeH8, the larger elastic stiffness is therefore consistent with its wider acoustic–optical phonon separation and the absence of imaginary frequencies in the phonon dispersion (Fig. 1(a)), confirming robust dynamical stability. Conversely, the comparatively lower elastic constants of Mg3CoH8 lead to softer acoustic phonons, which facilitate lattice flexibility and hydrogen-induced vibrational motion during absorption and desorption (Fig. 1(b)).
Elastic anisotropy describes the directional dependence of mechanical response, which is highly relevant for hydrogen-rich solids because hydrogen uptake and release generate directional lattice strain and non-uniform stress fields. In practical hydrogen storage, anisotropy affects microcrack initiation, diffusion pathways, and long-term cycling durability. In this work, the anisotropic behavior of Mg3ZH8 (Z = Fe, Co) is evaluated using the Zener anisotropy index 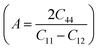 , the bulk and shear anisotropy indices
, the bulk and shear anisotropy indices 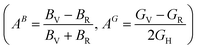 , and the universal anisotropy index
, and the universal anisotropy index 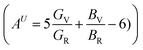 .46 For cubic systems, isotropy is indicated by A = 1, while AB = AG = AU = 0 corresponds to fully isotropic behavior.46 For Mg3FeH8, the Zener anisotropy index is A = 0.799, which deviates from unity and therefore confirms elastic anisotropy. Mg3CoH8 shows a stronger deviation with A = 0.542, indicating that the Co-based hydride is more anisotropic than the Fe-based phase. This trend is consistent with their elastic constants: Mg3CoH8 has a much smaller C12 relative to C11, which increases the directional contrast between shear and longitudinal deformation and enhances anisotropy. The tetragonal shear stability parameter C′ remains positive, confirming that the anisotropy does not arise from instability but from intrinsic bonding asymmetry within the H-rich framework. The bulk anisotropy index is essentially zero for both materials (AB ≈ 0.000), indicating that the compressibility is nearly isotropic. This is beneficial for hydrogen storage because volumetric expansion during hydrogen loading is less likely to concentrate stress along a single axis, reducing the probability of catastrophic lattice failure. In contrast, the shear anisotropy index shows a clear difference: Mg3FeH8 exhibits a very small value (AG = 0.006), while Mg3CoH8 has a much larger shear anisotropy (AG = 0.044). The same conclusion is supported by the universal anisotropy index, which is low for Mg3FeH8 (AU = 0.061) but significantly higher for Mg3CoH8 (AU = 0.463). These results indicate that Mg3FeH8 is closer to isotropic mechanical behavior, whereas Mg3CoH8 exhibits stronger directional sensitivity, mainly governed by shear deformation.
.46 For cubic systems, isotropy is indicated by A = 1, while AB = AG = AU = 0 corresponds to fully isotropic behavior.46 For Mg3FeH8, the Zener anisotropy index is A = 0.799, which deviates from unity and therefore confirms elastic anisotropy. Mg3CoH8 shows a stronger deviation with A = 0.542, indicating that the Co-based hydride is more anisotropic than the Fe-based phase. This trend is consistent with their elastic constants: Mg3CoH8 has a much smaller C12 relative to C11, which increases the directional contrast between shear and longitudinal deformation and enhances anisotropy. The tetragonal shear stability parameter C′ remains positive, confirming that the anisotropy does not arise from instability but from intrinsic bonding asymmetry within the H-rich framework. The bulk anisotropy index is essentially zero for both materials (AB ≈ 0.000), indicating that the compressibility is nearly isotropic. This is beneficial for hydrogen storage because volumetric expansion during hydrogen loading is less likely to concentrate stress along a single axis, reducing the probability of catastrophic lattice failure. In contrast, the shear anisotropy index shows a clear difference: Mg3FeH8 exhibits a very small value (AG = 0.006), while Mg3CoH8 has a much larger shear anisotropy (AG = 0.044). The same conclusion is supported by the universal anisotropy index, which is low for Mg3FeH8 (AU = 0.061) but significantly higher for Mg3CoH8 (AU = 0.463). These results indicate that Mg3FeH8 is closer to isotropic mechanical behavior, whereas Mg3CoH8 exhibits stronger directional sensitivity, mainly governed by shear deformation.
To connect single-crystal stiffness to polycrystalline mechanical performance, the Voigt and Reuss bounds for the bulk and shear moduli were evaluated and subsequently averaged using the Voigt–Reuss–Hill (VRH) scheme. For cubic symmetry, the moduli are given by:24,25
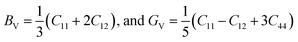 | (1) |
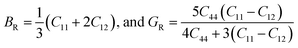 | (2) |
The Hill averages were obtained as
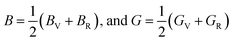 | (3) |
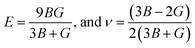 | (4) |
The calculated elastic moduli listed in Table 3 are consistent with those reported for comparable complex hydrides, confirming the reliability of the present calculations.34,43–45
The bulk modulus B reflects resistance to volumetric compression and is directly relevant to lattice stability during hydrogen uptake. Mg3FeH8 exhibits a high bulk modulus (B = 199.38 GPa), which is significantly larger than those reported for typical complex hydrides such as Mg7TiH16 (∼82 GPa),43 Mg7FeH16 (∼90 GPa),43 and K2LiAlH6 (∼32 GPa),34 indicating strong resistance to hydrogen-induced lattice expansion and favorable cycling stability. Mg3CoH8 shows a moderate bulk modulus (B = 93.82 GPa), comparable to Mg7FeH16,43 and LiCaCoH6,44 suggesting a more compliant lattice that may facilitate hydrogen diffusion while maintaining mechanical integrity.
The shear modulus G governs resistance to shape deformation and plays a key role in suppressing lattice distortion and crack formation during cycling. Mg3FeH8 exhibits a higher shear modulus (G = 63.69 GPa) than Mg3CoH8 (G = 46.12 GPa), and exceeds values reported for Mg7TiH16 (∼31 GPa)43 and K2LiAlH6 (∼14 GPa).34 This enhanced shear resistance supports greater mechanical durability in the Fe-based hydride, while the lower G of Mg3CoH8 may facilitate lattice flexibility favorable for hydrogen diffusion.
Young's modulus E, which characterizes overall elastic stiffness, follows the same trend. Mg3FeH8 shows a high Young's modulus (E = 172.69 GPa), substantially exceeding those of Mg7TiH16 (∼84 GPa),43 Mg7FeH16 (∼95 GPa),43 and K2LiAlH6 (∼38 GPa).34 Mg3CoH8 exhibits a moderate value (E = 118.87 GPa), still higher than most reported complex hydrides. Collectively, these comparisons indicate that Mg3ZH8 (Z = Fe, Co) combines high elastic stiffness with hydrogen-rich chemistry, highlighting its mechanical robustness and reinforcing the reliability of the VRH-based elastic analysis.
Poisson's ratio (ν), Pugh's ratio (B/G), and Cauchy pressure (CP = C12 − C44) provide insight into bonding character, ductility, and mechanical adaptability, all of which directly influence hydrogen cycling durability. In general, ν values near 0.33 indicate metallic bonding, values around 0.25 suggest ionic bonding, and values near 0.1 are characteristic of covalent bonding, while ν > 0.26 and B/G > 1.75 are commonly associated with ductile behavior.47,48 Positive Cauchy pressure further reflects ionic bonding and enhanced plasticity, whereas negative values indicate covalent bonding and brittleness.49 The mechanical indicators are listed in Table 4 further clarify the ductility, bonding character, and hydrogen-cycling resilience of Mg3ZH8 (Z = Fe, Co). The Cauchy pressure (CP) is positive for both compounds, confirming dominant metallic bonding. However, Mg3FeH8 exhibits a markedly larger value (92.58 GPa) than Mg3CoH8 (13.56 GPa), indicating superior ductility and resistance to hydrogen-induced embrittlement. This enhanced ductility is also reflected in the Pugh's ratio (B/G), where Mg3FeH8 (B/G = 3.13) and Mg3CoH8 (B/G = 2.03) both exceed the brittle–ductile threshold (≈1.75), with the Fe-based hydride showing a stronger ductile character. These results suggesting enhanced mechanical resilience during repeated hydrogen absorption–desorption cycles. The Poisson's ratio (ν) further supports this trend, with values of 0.356 for Mg3FeH8 and 0.289 for Mg3CoH8, consistent with metallic bonding and good plastic deformability during repeated hydrogen absorption–desorption cycles. The Vickers hardness50 (HV), estimated using HV = (1 − 2ν)Y/[6(1 + ν)], quantifies resistance to plastic deformation and is therefore directly relevant to hydrogen cycling durability. Despite differences in ductility, both compounds exhibit comparable Vickers hardness (HV ≈ 6–6.5 GPa), suggesting sufficient resistance to surface deformation without excessive brittleness.
| Compound | CP | B/G | ν | HV | μm | ρ | vt | vl | vm | θD | Tm | kph | kmin |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mg3FeH8 | 92.58 | 3.13 | 0.356 | 6.13 | 3.42 | 5.89 | 3.29 | 6.05 | 3.70 | 746.4 | 2305 | 3.39 | 2.35 |
| Mg3CoH8 | 13.56 | 2.03 | 0.289 | 6.49 | 2.61 | 5.76 | 2.83 | 5.19 | 3.16 | 627.3 | 1631 | 3.57 | 1.94 |
The calculated hardness values of Mg3FeH8 and Mg3CoH8 (≈6–6.5 GPa) are comparable to those reported for Mg3CrH8 (6.21 GPa) and Mg3MnH8 (5.37 GPa), and slightly higher than Mg3TiH8 and Mg3VH8, as reported in the literature.19 This indicates that the Fe- and Co-based hydrides possess sufficient resistance to surface deformation while retaining ductile characteristics, which is favorable for suppressing crack formation and mechanical degradation during hydrogen cycling.
When compared with other hydride perovskites such as MgCuH3 (0.77 GPa),51 RbNiH3 (0.64 GPa),52 BeGaH3 (3.02 GPa),53 and BeInH3 (3.17 GPa),53 both Mg3FeH8 and Mg3CoH8 exhibit relatively high hardness values, confirming their favorable mechanical stability among hydrogen-rich hydrides. The machinability index 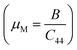 provides insight into both intrinsic plasticity and industrial workability.54 Finally, the higher machinability index (μm = 3.42) of Mg3FeH8 compared to Mg3CoH8 (2.61) implies improved processability and structural tolerance, which are advantageous for fabricating durable hydrogen storage components. Collectively, these parameters demonstrate that Mg3ZH8 (Z = Fe, Co) hydrides combine metallic ductility with mechanical robustness, reinforcing their suitability for stable and kinetically efficient solid-state hydrogen storage.
provides insight into both intrinsic plasticity and industrial workability.54 Finally, the higher machinability index (μm = 3.42) of Mg3FeH8 compared to Mg3CoH8 (2.61) implies improved processability and structural tolerance, which are advantageous for fabricating durable hydrogen storage components. Collectively, these parameters demonstrate that Mg3ZH8 (Z = Fe, Co) hydrides combine metallic ductility with mechanical robustness, reinforcing their suitability for stable and kinetically efficient solid-state hydrogen storage.
The Debye temperature (ΘD) is a key thermophysical parameter that governs lattice vibrational behavior, heat capacity, lattice stability, and thermal conductivity, all of which are directly relevant to hydrogen storage kinetics and reversibility. In solid-state hydrides, ΘD reflects the strength of interatomic bonding and the ability of the lattice to withstand repeated hydrogen absorption–desorption cycles without structural degradation. A higher ΘD generally indicates stronger metal–hydrogen bonding and enhanced lattice stability, while moderate reductions in ΘD can facilitate hydrogen diffusion and release. The Debye temperature was calculated using the following relations:55
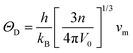 | (5) |
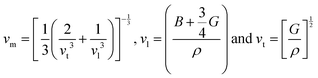 | (6) |
As summarized in Table 4, Mg3FeH8 exhibits higher sound velocities and a larger average phonon velocity than Mg3CoH8. Consequently, Mg3FeH8 shows a higher Debye temperature (ΘD = 746.4 K) compared to Mg3CoH8 (ΘD = 627.3 K), indicating stronger interatomic bonding and enhanced lattice rigidity. When compared with previously reported single, complex, and double hydrides, the thermo-mechanical characteristics of Mg3FeH8 and Mg3CoH8 fall within a favorable range for durable hydrogen storage. Typical single perovskite hydrides such as MgXH3 (X = Ga, Tl),29 MgCuH3,56 MBeH3 (M = Li, Na, and K),57 and XFeH3 (X = Ca, Sr, Ba)58 exhibit Debye temperatures generally below ∼400 K, reflecting softer lattices that are more susceptible to hydrogen-induced vibrational instability during cycling. In contrast, several complex and double hydrides, including Cs2NaInH6,59 Sr2LiCuH6,11 and BaXH4 (X = Mn, Re, or Tc)60 display Debye temperatures in the range of 300–550 K, indicating improved lattice stiffness and phonon stability. The Debye temperatures of Mg3FeH8 and Mg3CoH8 are comparable to or higher than those of many established similar type hydrides,19 suggesting similarly robust vibrational stability under repeated hydrogen absorption–desorption cycles. This insight provides a rational design principle for developing thermally stable, high-capacity solid-state hydrogen storage materials suitable for stationary and high-temperature hydrogen energy applications. However, the absence of imaginary phonon modes, high Debye temperatures, ductile mechanical behavior, and moderate reaction enthalpies suggest favorable lattice resilience and potential hydrogen mobility.
The melting temperature (Tm) is a key indicator of mechanical robustness and thermal endurance, which are essential for solid-state hydrogen storage materials operating under repeated absorption–desorption cycles. A sufficiently high Tm ensures that the host lattice maintains its structural integrity at elevated temperatures required for hydrogen release, while also preventing mechanical degradation during long-term cycling. Consequently, Tm is closely linked to hydrogen storage kinetics, safety, and operational reliability. The melting temperature was estimated using the empirical relation:61
| Tm = 553 + 5.91C11 (±300 K) | (7) |
Using this relation, Mg3FeH8 exhibits a significantly higher melting temperature (Tm ≈ 2305 K) compared to Mg3CoH8 (Tm ≈ 1631 K). This difference directly correlates with their elastic stiffness, as Mg3FeH8 possesses a much larger C11 and bulk modulus than Mg3CoH8, indicating stronger interatomic bonding and greater resistance to lattice deformation. From a hydrogen storage perspective, the higher Tm of Mg3FeH8 implies superior thermal stability, making it suitable for high-temperature hydrogen storage and stationary energy systems, where structural durability is critical. In contrast, the lower Tm of Mg3CoH8 reflects a comparatively softer lattice, which can facilitate lattice breathing and atomic rearrangements during hydrogen desorption. Such behavior is advantageous for enhancing hydrogen release kinetics at reduced temperatures, highlighting a trade-off between thermal robustness and kinetic accessibility. A comparison with previously reported hydrides further clarifies the thermal robustness of Mg3FeH8 and Mg3CoH8. Single perovskite hydrides such as MgCuH3,56 CsAH3 (A = Fe, Cu and Tl),62 and XFeH3 (X = Ca, Sr, Ba)58 generally exhibit lower estimated melting temperatures, which limits their resistance to thermally induced lattice degradation during hydrogen cycling. In contrast, complex and double hydrides, including Cs2NaInH6,59 Cs2AlInH6,59 Cs2AlTlH6,59 and X2MgH4 (X = K, Rb, Cs),63 display moderate melting temperatures, reflecting improved thermal endurance and cycling stability. This combination of high melting temperature, strong mechanical stability, and hydrogen-rich composition highlights the scientific novelty of Mg3ZH8 (Z = Fe, Co) hydrides.
The lattice thermal conductivity (κph) was evaluated using Slack's model, which correlates the Debye temperature, Grüneisen parameter (γ), and other lattice descriptors:64
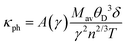 | (8) |
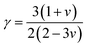 | (9) |
In addition, the minimum lattice thermal conductivity (kmin) was estimated using Clarke's model:65
 | (10) |
The calculated κph and kmin values listed in Table 4 indicate that both Mg3FeH8 and Mg3CoH8 hydrides possess moderate phonon heat transport at 300 K. Mg3FeH8 shows κph = 3.39 W m−1 K−1 and kmin = 2.35 W m−1 K−1, whereas Mg3CoH8 exhibits a slightly higher κph = 3.57 W m−1 K−1 but a lower kmin = 1.94 W m−1 K−1. The larger kmin in Mg3FeH8 is consistent with its higher sound velocities and stronger lattice stiffness, implying a higher intrinsic phonon transport floor, while the comparatively lower kmin of Mg3CoH8 reflects a softer lattice that can enhance phonon scattering. From a hydrogen-storage perspective, these thermal transport characteristics are beneficial because efficient heat dissipation can mitigate local temperature gradients during absorption–desorption cycling, supporting stable kinetics and improved cycling durability.
4. Conclusion
First-principles calculations provide a powerful framework for the discovery and rational design of advanced hydrogen storage materials. In this study, transition-metal substitution into magnesium-based hydrides was explored as an effective strategy to tailor hydrogen storage thermodynamics and kinetics. The structural, electronic, mechanical, optical, and hydrogen storage properties of Mg3ZH8 (Z = Fe, Co) were systematically investigated using density functional theory to assess their suitability for solid-state hydrogen storage applications. Both compounds are found to be thermodynamically, mechanically, and dynamically stable, as confirmed by negative formation energies, elastic stability criteria, and phonon spectra free of imaginary modes. Mg3FeH8 exhibits a ferromagnetic metallic ground state, whereas Mg3CoH8 is non-magnetic, highlighting the role of transition-metal d states in governing magnetic and electronic behavior. The hydrogen storage performance of Mg3ZH8 (Z = Fe, Co) is promising, with gravimetric capacities of 5.90 wt% for Mg3FeH8 and 5.76 wt% for Mg3CoH8, exceeding the U.S. DOE minimum target. Their volumetric hydrogen densities (≈195–187 g H2 per L) are substantially higher than current DOE benchmarks, indicating excellent space efficiency. The calculated desorption temperatures fall within a moderate range, with Mg3FeH8 consistently showing lower Tdes values than Mg3CoH8, suggesting more favorable hydrogen release kinetics. Mechanical analysis reveals that both hydrides are ductile, with positive Cauchy pressures, high Pugh's ratios, and moderate Vickers hardness values, implying good resistance to hydrogen-induced embrittlement during cycling. Optical and electronic analyses further confirm their metallic nature, with strong free-carrier response and interband transitions that correlate with their hydrogen desorption behavior. Overall, the balanced combination of high hydrogen density, mechanical resilience, moderate desorption temperature, and electronic activity positions Mg3ZH8 (Z = Fe, Co), particularly Mg3FeH8, as a promising candidate for next-generation hydrogen storage materials and provides a robust theoretical benchmark for future experimental exploration.Author contributions
Md. Hasan Mia: conceptualization; methodology; writing manuscript – reviewing and editing; data curation; validation; supervision; Md. Zahid Hasan: formal analysis; validation; review – editing. A. Arunkumar: formal analysis; validation; review – editing; and supervision. S. AlFaify: formal analysis; validation; review – editing.Conflicts of interest
The authors declare that they have no known conflicting financial interests or personal ties that may have seemed to affect the work presented in this study.Data availability
Supplementary information: the figures in the supplementary information represent the optical conductivity and refractive properties of Mg3ZH8 (Z = Fe, Co) hydrides. See DOI: https://doi.org/10.1039/d6ma00353b.Relevant data from this study are available from the corresponding author upon a reasonable request.
Acknowledgements
The authors are grateful to the Materials Research and Simulation lab, Department of Electrical Electronics and Engineering, International Islamic University Chittagong, Chittagong-4358, Bangladesh for providing the computing facilities for this work.References
- L. Zhou, Renewable Sustainable Energy Rev., 2005, 9, 395–408 CrossRef CAS.
- E. Fakioğlu, Y. Yürüm and T. N. Veziroğlu, Int. J. Hydrogen Energy, 2004, 29, 1371–1376 CrossRef.
- S. Niaz, T. Manzoor and A. H. Pandith, Renewable Sustainable Energy Rev., 2015, 50, 457–469 CrossRef CAS.
- V. Kudiiarov, J. Lyu, O. Semyonov, A. Lider, S. Chaemchuen and F. Verpoort, Appl. Mater. Today, 2021, 25, 101208 CrossRef.
- R. Zhou, X. Mo, Y. Huang, C. Hu, X. Zuo, Y. Ma, Q. Wei and W. Jiang, Batteries, 2023, 9, 179 CrossRef CAS.
- J. Zheng, W. Sun, X. Dou, A.-J. Mao and C. Lu, J. Phys. Chem. C, 2021, 125, 3150–3156 CrossRef CAS.
- Y. Ma, D. Duan, Z. Shao, H. Yu, H. Liu, F. Tian, X. Huang, D. Li, B. Liu and T. Cui, Phys. Rev. B:Condens. Matter Mater. Phys., 2017, 96, 144518 CrossRef.
- T. Tang and Y. Tang, Int. J. Hydrogen Energy, 2024, 61, 13–24 CrossRef CAS.
- A. Hosen, D. Dahliah, N. F. AlShaikh Mohammad, A. A. Mousa and M. S. Abu-Jafar, Int. J. Hydrogen Energy, 2025, 102, 348–359 CrossRef CAS.
- G. M. Mustafa, B. Younas, H. D. Alkhaldi, A. Mera, A. K. Alqorashi, J. Hakami, S. A. Mahmoud, I. Boukhris and Q. Mahmood, Int. J. Hydrogen Energy, 2024, 95, 300–308 CrossRef CAS.
- A. Ayyaz, M. A. Ullah, M. Zaman, N. D. Alkhaldi, Q. Mahmood, I. Boukhris, M. S. Al-Buriahi and M. M. Al-Anazy, Int. J. Hydrogen Energy, 2025, 102, 1329–1339 CrossRef CAS.
- W. Azeem, S. Hussain, M. K. Shahzad, F. Azad, G. Khan, V. Tirth, H. Alqahtani, A. Algahtani, T. Al-Mughanam and Y. H. Wong, Int. J. Hydrogen Energy, 2024, 79, 514–524 CrossRef CAS.
- Q. Dai, T.-Y. Tang, Q.-Q. Liang, Z.-Q. Chen, Y. Wang and Y.-L. Tang, Int. J. Hydrogen Energy, 2024, 92, 769–778 CrossRef CAS.
- T. Tang and Y. Tang, Mater. Chem. Phys., 2024, 316, 129099 CrossRef CAS.
- Y. Pan and Y. Zhu, J. Alloys Compd., 2025, 1021, 179661 CrossRef CAS.
- H. Murtaza, Q. Ain, R. A. Alshgari, T. Akhter and J. Munir, J. Power Sources, 2025, 641, 236788 CrossRef CAS.
- D. Tufail, U. Ahmed, M. Haleem, B. Amin and M. Shafiq, RSC Adv., 2025, 15, 337–347 RSC.
- M. M. Al-Anazy, G. M. Mustafa, O. Zayed, B. Younas, T. M. Al-Daraghmeh, N. D. Alkhaldi, A. S. Alofi, A. K. Alqorashi and Q. Mahmood, Int. J. Hydrogen Energy, 2024, 78, 927–937 CrossRef CAS.
- T. Tang and Y. Tang, Int. J. Hydrogen Energy, 2024, 74, 372–383 CrossRef CAS.
- M. D. Segall, P. J. D. Lindan, M. J. Probert, C. J. Pickard, P. J. Hasnip, S. J. Clark and M. C. Payne, J. Phys.: Condens. Matter, 2002, 14, 2717 CrossRef CAS.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865 CrossRef CAS PubMed.
- P. E. Gill, W. Murray and M. A. Saunders, SIAM Rev., 2005, 47, 99–131 CrossRef.
- P. Wisesa, K. A. McGill and T. Mueller, Phys. Rev. B:Condens. Matter Mater. Phys., 2016, 93, 155109 CrossRef.
- W. Voigt, Lehrbuch der Kristallphysik (Textbook of crystal physics), BG Teubner, Leipzig und Berlin, 1928 Search PubMed.
- A. Reuß, Z. Angew. Math. Mech., 1929, 9, 49–58 CrossRef.
- M. Bortz, B. Bertheville, K. Yvon, E. A. Movlaev, V. N. Verbetsky and F. Fauth, J. Alloys Compd., 1998, 279 Search PubMed.
- L. Yi, Trans. Nonferrous Met. Soc. China, 2010, 20, 2281–2288 CrossRef.
- E. Evard, I. Gabis and V. A. Yartys, Int. J. Hydrogen Energy, 2010, 35, 9060–9069 CrossRef CAS.
- M. H. Mia, M. S. Parves, O. Alsalmi and M. Z. Hasan, ACS Appl. Eng. Mater., 2025, 3, 4218–4232 CrossRef.
- S. Al, Int. J. Hydrogen Energy, 2019, 44, 1727–1734 CrossRef CAS.
- T. Tang and Y. Tang, Ceram. Int., 2024, 50, 52270–52283 CrossRef CAS.
- S. Zhang, S. Chen, Y. Chen, J. Hou, D. Xu, W. Luo and Z. Shi, Int. J. Hydrogen Energy, 2025, 166, 150903 CrossRef CAS.
- Z. El Fatouaki, A. Tahiri, A. Jabar and M. Idiri, Int. J. Hydrogen Energy, 2025, 175, 151433 CrossRef CAS.
- H. Murtaza, M. A. Habib, Q. Ain, A. Kumar, J. Munir, A. B. M. Ibrahim, A. D. Oza and M. S. S. Al-Salimi, Sci. Rep., 2025, 16, 2019 CrossRef PubMed.
- M. Caid, E. Deligöz, H. Ozisik, H. Rached, D. Rached, H. Mansour and Y. Rached, Int. J. Hydrogen Energy, 2025, 182, 151797 CrossRef CAS.
- T. Ikeda, Y. Mikami and T. Haruki, J. Phys. Chem. C, 2007, 111, 8389–8396 CrossRef CAS.
- Y. Didi, A. Tahiri, R. Touti, R. Ahfir, M. Naji, H. Naïli, H. Nebdi, C. Hajjaj and A. Rjeb, Int. J. Hydrogen Energy, 2026, 225, 154319 CrossRef CAS.
- M. W. Qureshi, X. Ma, G. Tang and R. Paudel, Materials, 2020, 13, 5148 CrossRef CAS PubMed.
- M. H. Mia, M. A. Khatun and M. Rahman, Next Mater., 2025, 7, 100434 CrossRef CAS.
- V. Lucarini, K. E. Peiponen, J. J. Saarinen and E. M. Vartiainen, Kramers-Kronig relations in optical materials research, Springer Berlin Heidelberg, Berlin, Heidelberg, 2005, vol. 110, pp. 27–28. Search PubMed.
- F. Mouhat and F.-X. Coudert, Phys. Rev. B:Condens. Matter Mater. Phys., 2014, 90, 224104 CrossRef.
- M. H. Mia, F. Parvin, A. K. M. A. Islam and M. A. Khatun, Int. J. Mod. Phys. B, 2025, 39, 2550118 CrossRef CAS.
- S. Berri and Ç. Yamçıçıer, Int. J. Hydrogen Energy, 2025, 177, 151559 CrossRef CAS.
- A. Hosen, E. N. Kande, H. D. Alkhaldi, A. A. Mousa, A. Akremi, I. Boukhris and M. S. Abu-Jafar, J. Mater. Res. Technol., 2025, 36, 8688–8697 CrossRef CAS.
- M. A. Ullah, M. Kaleem, A. Nasir, Z. Sarfraz, M. M. A. Iqbal, M. Rizwan, K. N. Riaz and M. Tanzeel, RSC Adv., 2025, 15, 38714–38728 RSC.
- J. K. Rony, M. Islam, M. Saiduzzaman, K. M. Hossain, S. Alam, A. Biswas, M. H. Mia, S. Ahmad and S. K. Mitro, J. Mater. Res. Technol., 2024, 29, 897–909 CrossRef CAS.
- I. N. Frantsevich, Elastic Constants and Elastic Moduli of Metals and Insulators, 1982 Search PubMed.
- S. F. Pugh, Philos. Mag., 1954, 45, 823–843 CAS.
- D. G. Pettifor, Mater. Sci. Technol., 1992, 8, 345–349 CrossRef CAS.
- Y. Tian, B. Xu and Z. Zhao, Int. J. Refract. Met. Hard Mater., 2012, 33, 93–106 CrossRef CAS.
- Z. U. Rehman, M. A. Rehman, B. Rehman, S. Sikiru, S. Qureshi, E. M. Ali and M. Awais, Environ. Sci. Pollut. Res., 2023, 30, 113889–113902 CrossRef CAS PubMed.
- W. Khan and M. K. Masood, Mater. Sci. Semicond. Process., 2024, 173, 108149 CrossRef CAS.
- W. Khan, Mater. Sci. Semicond. Process., 2024, 174, 108221 CrossRef CAS.
- Z. Sun, D. Music, R. Ahuja and J. M. Schneider, Phys. Rev. B:Condens. Matter Mater. Phys., 2005, 71, 193402 CrossRef.
- P. Debye, Ann. Phys., 1912, 344, 789–839 CrossRef.
- A. Koufi, Y. Ziat, H. Belkhanchi and A. Bouzaid, Comput. Condens. Matter, 2025, e01010 CrossRef.
- D. Guendouz, Z. Charifi, H. Baaziz, T. Ghellab, N. Arikan, Ş. Uğur and G. Ö. K. A. Y. Uğur, Can. J. Phys., 2016, 94, 865–876 CrossRef CAS.
- R. Song, N. Xu, Y. Chen, S. Chen, J. Zhang, W. Dai and W. Zhang, Chin. J. Phys., 2024, 89, 1152–1163 CrossRef CAS.
- M. Tarekuzzaman, M. S. Parves, M. Z. Rahman and S. S. Hasan, Solid State Commun., 2025, 116043 CrossRef CAS.
- E. Ededet, H. Louis, U. G. Chukwu, T. O. Magu, A. E. Udo, S. A. Adalikwu and A. S. Adeyinka, J. Electron. Mater., 2024, 53 Search PubMed.
- M. E. Fine, L. D. Brown and H. L. Marcus, Scr. Metall., 1984, 18, 951–956 CrossRef CAS.
- M. H. Mia, M. Rasheduzzaman, O. Alsalmi and M. Z. Hasan, Int. J. Hydrogen Energy, 2026, 209, 153607 CrossRef CAS.
- Ç. Yamçıçıer, Int. J. Hydrogen Energy, 2023, 48, 39930–39943 CrossRef.
- D. T. Morelli and G. A. Slack, High Thermal Conductivity Materials, Springer, New York, 2006, pp. 37–68 Search PubMed.
- D. R. Clarke, Surf. Coat. Technol., 2003, 163, 67–74 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |