 Open Access Article
Open Access ArticleDevelopment of a supramolecular cobalt(II) metallohydrogel from L-serine-based low molecular weight gelator for dual antimicrobial and semiconducting applications
Sangita Somea,
Aiswarya Mohanb,
Rajlakshmi Lahac,
Subhendu Dhibar*l,
Priya Kaithd,
Aditi Trivedie,
Subham Bhattacharjee f,
Timothy O. Ajiboye
f,
Timothy O. Ajiboye g,
Lebea N. Nthunya
g,
Lebea N. Nthunya h,
Ashok Bera
h,
Ashok Bera d,
Asit Kumar Das
d,
Asit Kumar Das i,
Sumit Kumar Panja
i,
Sumit Kumar Panja j,
Somasri Dam*c,
Padmanabhan Predeep*k and
Bidyut Saha
j,
Somasri Dam*c,
Padmanabhan Predeep*k and
Bidyut Saha *a
*a
aColloid Chemistry Laboratory, Department of Chemistry, The University of Burdwan, Golapbag, Burdwan-713104, West Bengal, India. E-mail: bsaha@chem.buruniv.ac.in; Tel: +91 9476341691
bLaboratory for Molecular Photonics and Electronics (LAMP), Department of Physics, National Institute of Technology Calicut, Calicut-673603, Kerala, India
cDepartment of Microbiology, The University of Burdwan, Burdwan-713104, West Bengal, India. E-mail: sdam@microbio.buruniv.ac.in
dDepartment of Physics, Indian Institute of Technology Jammu, J&K-181221, India
eNational Institute of Science Education and Research (NISER), Bhubaneswar, Odisha 752050, India
fDepartment of Chemistry, Kazi Nazrul University, Asansol-713303, West Bengal, India
gFaculty of Natural and Agricultural Sciences, North-West University, Mmabatho-2735, South Africa
hInstitute for Nanotechnology and Water Sustainability, University of South Africa, Florida Science Campus, 1709 Roodepoort, South Africa
iDepartment of Chemistry, Murshidabad University, Berhampore-742101, West Bengal, India
jTarsadia Institute of Chemical Science, Uka Tarsadia University, Surat-394350, Gujrat, India
kSchool of Nanoscience and Nanotechnology, Mahathma Gandhi University, Kottayam-686560, Kerala, India. E-mail: predeep@mgu.ac.in
lThe Institute for Solid State Physics (ISSP), University of Tokyo, 5-1-5 Kashiwanoha, Kashiwa City, Chiba Prefecture 277-8581, Japan. E-mail: sdhibar@g.ecc.u-tokyo.ac.jp
First published on 30th April 2026
Abstract
A Co(II)-metallohydrogel was synthesized by combining cobalt(II) nitrate and L-serine in the presence of potassium hydroxide in an aqueous medium at room temperature. The resulting hydrogel demonstrated excellent mechanical strength, as confirmed through rheological studies. Field emission scanning electron microscopy (FESEM) and energy-dispersive X-ray (EDX) mapping revealed a hierarchical microstructure and confirmed the presence of essential elements such as Co, C, N, O, and K. FT-IR spectroscopy shed light on the supramolecular interactions involved in gel formation, while powder X-ray diffraction (PXRD) provided information on the material's crystalline features. Optical absorption analysis confirmed the semiconducting nature of the gel, showing notably high electron mobility compared to similar materials. Furthermore, the metallohydrogel exhibited strong antibacterial activity against both Gram-positive (B. subtilis, S. aureus) and Gram-negative (E. coli, P. aeruginosa) pathogens. These multifunctional properties underscore the material's potential for future applications in both flexible electronics and biomedical technology.
1. Introduction
Gels represent a unique subset of soft materials, notable for their capacity to incorporate large volumes of liquid within a three dimensional network structure.1 This retention of fluid is enabled by gelator molecules that assemble into a robust framework via various cross linking mechanisms.2,3 The combination of solid like mechanical strength with the dynamic mobility of a liquid endows gels with remarkable physical and chemical properties. These characteristics make them suitable for diverse applications across biomedical engineering, pharmaceuticals, food processing, cosmetic formulations, and advanced materials development.4Gels are generally classified into two major types based on how their networks are formed: covalently bonded chemical gels and physically assembled supramolecular gels. Chemical gels rely on permanent covalent linkages that produce rigid, often irreversible structures.5 Conversely, supramolecular gels are formed by the self-assembly of small gelator molecules6,7-typically under 3000 Daltons-held together by noncovalent interactions8–13
The molecular design of the gelator plays a pivotal role in the gelation process.14–16 An extensive variety of LMWGs have been employed to create supramolecular gels, including dicarboxylic acids,14 urea derivatives,15 modified amino acids,16 fatty acids,5 sorbitol,17 dendrimers,17 carbohydrates,17 and many more. Equally crucial to the gelation process is the role of the solvent, which not only acts as the medium but also directly affects molecular assembly.17,18 The choice of solvent can significantly impact gel morphology, fiber dimensions, and overall gel stability. Parameters such as polarity, viscosity, hydrogen-bonding capability, and coordination tendencies of the solvent influence the strength and nature of the interactions among the gelator molecules.14–17 Solvents commonly used ingelation studies include water,18–23 alcohols,24 dimethylformamide (DMF),25 dimethyl sulfoxide (DMSO),26 acetonitrile,27 acetone,28 are among the most effective solvents for initiating supramolecular gel formation.
Metallohydrogels represent a novel and rapidly evolving category within the field of supramolecular gels. These systems are characterized by the integration of metal ions or metal-based complexes that coordinate with organic ligands, facilitating the formation of self-organized three-dimensional networks.29 Typically, such gels arise from the coordination interactions between low molecular weight gelators (LMWGs) and various transition metal ions, giving rise to hybrid materials with superior functionalities. Transition metals including copper(II),30 nickel(II),31,32 cobalt(II),33 have frequently been employed to construct metallogels. The presence of metal centers not only reinforces the structural integrity of the gel matrix but also introduces diverse functional characteristics such as redox behavior,34 catalytic activity,18,19 magnetic properties,35 and the ability to serve as templates for nanoparticle formation.36
Selecting an appropriate ligand is crucial in metallohydrogel synthesis. Ligands possessing multiple functional groups, such as amino, hydroxyl, or carboxylate moieties, are especially effective, with amino acids being prime examples due to their versatile coordination capabilities.37 Among them, L serine stands out as a promising candidate for constructing metallohydrogels. This naturally derived amino acid features a side chain that includes both amine and hydroxyl groups, enabling strong interactions with metal ions and promoting the assembly of stable supramolecular frameworks. Additionally, its natural abundance and biocompatibility make L-serine highly suitable for applications in both biomedical and environmental fields.
Metallogels exhibit antibacterial properties through several distinct mechanisms. One primary mode of action involves the disruption of bacterial cell membranes, which leads to the leakage of vital intracellular contents.
Metallohydrogel-based Schottky diodes are promising for advanced electronics due to their tunable electronic properties, stability, and compatibility with various fabrication techniques. By adjusting metal–ligand combinations, device performance can be optimized. Their integration with other components and environmental durability make them ideal for multifunctional electronic and optoelectronic device applications. In a significant advancement, Dhibar and collaborators developed an efficient and straightforward approach for synthesizing metallogels, eliminating the need for harsh conditions while retaining material functionality.18,32 Inspired by their strategy, We report the synthesis of a novel L-serine–derived metallohydrogel, in which L-serine acts as a tridentate (N, O, O) ligand.38 The gel formation occurs through coordination of Co(II) ions with the amino nitrogen, caboxylate oxygen, and hydroxyl oxygen groups of the ligand under alkaline conditions (pH ≥ 10).39 This gel forms swiftly in an aqueous environment, yielding a stable blue coloured material. The gel network is predominantly stabilized through coordination interactions between Co(II) ions and serine, complemented by intermolecular hydrogen bonding among gelator units. The resulting hydrogel demonstrates remarkable stability, uniform structure, and reproducibility, positioning it as a strong candidate for future functional materials. However, systematic studies on cobalt-L serine based metallohydrogels remain limited, particularly regarding coordination driven gelation mechanisms and their influence on multifunctional properties.40
This Co(II) based metallohydrogel displays excellent electrical conductivity and notable optoelectronic behaviour, along with pronounced antibacterial efficacy against various pathogenic strains. Comprehensive analyses of its rheological performance and internal morphology have been carried out. Designed around a metal semiconductor junction, the material shows efficient charge transport properties, highlighting its applicability in semiconducting devices.
2. Experimental
2.1. Materials
Cobalt(II) nitrate hexahydrate (Sigma-Aldrich, USA, 99.99% trace metals basis), L-serine (Sigma-Aldrich, USA, 99%), and Potassium hydroxide (Merck, 98%) were utilized as received. Dry DMF solvent was employed throughout the study. Tryptone, D-(+)-Glucose anhydrous, and Yeast Extract Powder were obtained from Himedia.2.2. Apparatus and measurements
2.3. Synthesis of Co(II)-L-serine based metallohydrogel (Co-LS)
An aqueous solution of bright pink cobalt(II) nitrate hexahydrate (0.291 g, 1 mmol, ∼1 mL) was swiftly combined with a clear aqueous solution (∼1 mL) containing L-serine (0.105 g, 1 mmol) and 0.5 equivalents of potassium hydroxide at ambient temperature. This immediate mixing led to the rapid formation of a deep blue cobalt(II)-based metallohydrogel, referred to as Co-LS. A schematic illustration of the proposed Co-LS network and an inverted vial image showcasing its gel state stability are shown in Fig. 1. The successful gelation and mechanical integrity of the Co-LS metallohydrogel were initially confirmed via the inverted vial method (Fig. 1) and further substantiated by rheological investigations (detailed below). To determine the minimum gelation threshold, the minimum gelation concentration (MGC) of the Co-LS system was evaluated by systematically varying the concentrations of Co (NO3)2·6H2O and L-serine from 20 to 150 mg mL−1, keeping their mass ratio constant at 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. At 150 mg mL−1 concentration of each component in water, a uniform and stable deep blue gel was consistently formed. Thermal stability of the gel was assessed using a digital melting point apparatus. The Co-LS metallohydrogel retained its structure until a transition into the sol phase occurred at approximately 80 ± 2 °C, indicating a high thermal resistance. Additionally, the pH of the Co-LS gel was measured using a Mettler Toledo FP20 pH meter, revealing that the gel system forms and remains stable at pH values of 10 or higher.
1. At 150 mg mL−1 concentration of each component in water, a uniform and stable deep blue gel was consistently formed. Thermal stability of the gel was assessed using a digital melting point apparatus. The Co-LS metallohydrogel retained its structure until a transition into the sol phase occurred at approximately 80 ± 2 °C, indicating a high thermal resistance. Additionally, the pH of the Co-LS gel was measured using a Mettler Toledo FP20 pH meter, revealing that the gel system forms and remains stable at pH values of 10 or higher.
2.4. Antimicrobial activity of Co-LS
Quantitative assessment of antimicrobial effect of Co-Ls was carried out with a series of concentrations of the metallogel on different bacterial strains such as Escherichia coli, Bacillus subtilis, Pseudomonas aeruginosa, and Staphylococcus aureus. Among them, Pseudomonas aeruginosa and Escherichia coli are Gram-negative in nature, whereas Staphylococcus aureus and Bacillus subtilis are Gram-positive. The concentrations of Co-Ls used for the experiment were 20 mg mL−1, 40 mg mL−1, 60 mg mL−1, 80 mg mL−1, and 100 mg mL−1. 100 µL of inoculum from log phase culture of each bacterial strain was spread uniformly on TGA (1% tryptone, 1% glucose, 1% yeast extract and pH 6.5) agar plate with the help of sterile cotton swabs. Later, 10 µL of metallogel suspension from each concentration was spotted on the agar surface with bacterial inoculum spread on it. Broad-spectrum antibiotic Streptomycin was used as positive control for this experiment. The positive control was spotted too on each plate. The plates were incubated at 37 °C for 24 hours. All the experiments are performed in triplicate.3. Results and discussion
3.1. Rheological analysis
To evaluate the mechanical strength and viscoelastic behaviour of the Co-LS metallohydrogel, rheological measurements were carried out using a rheometer. Both angular frequency sweep and strain-sweep tests were conducted on the sample prepared at the minimum gelation concentration (MGC) of 150 mg mL−1 for Co(NO3)2·6H2O and L-serine. The gel-like nature of the material was confirmed by the dominance of the storage modulus (G′) over the loss modulus (G″), indicating its semi-solid, elastic characteristics. Specifically, the storage modulus (G′) exhibited significantly higher values than the loss modulus (G″) across the frequency range tested, a typical signature of viscoelastic gels (Fig. 2a). The average G′ was observed to be greater than 102 Pa, suggesting a mechanically robust network stabilized by Co(II)-serine coordination and non-covalent interactions. Furthermore, strain-sweep experiments were performed at a constant angular frequency of 6.283 rad s−1 to assess the structural integrity of the hydrogel under increasing strain. As shown in Fig. 2b, the gel maintained its mechanical integrity over a broad range of strain values before yielding, reflecting its high strain tolerance. These findings demonstrate that the Co-LS metallohydrogel possesses excellent mechanical stability, making it a strong candidate for practical applications in soft materials and flexible electronic devices. | ||
| Fig. 2 (a) Angular frequency measurements vs. G′ and G″ of Co-LS metallohydrogel; (b) Strain-sweep measurements of Co-LS metallohydrogel performed at a constant frequency of 6.283 rad s−1. | ||
3.2. Study of morphology
Field emission scanning electron microscopy (FESEM) analysis of the Co-LS metallohydrogel reveals a well-defined hierarchical flake-like network structure (Fig. 3a). This intricate morphology results from the coordination of Co(NO3)2·6H2O with L-serine in an aqueous medium. The observed microstructure is attributed to strong supramolecular interactions, which play a vital role in stabilizing the gel framework. The fibrous morphology of the gel facilitates the charge transfer mechanisms. The porous network enhances the surface area availability which helps for interaction with microbial cells. These properties contribute to the observed semiconducting behaviour and antibacterial activity of the metallogel. f Elemental mapping performed on a selected region confirms the uniform distribution of key components, including cobalt, L-serine, and potassium hydroxide. EDX further supports these findings, detecting the presence of carbon (C), nitrogen (N), oxygen (O), potassium (K), and cobalt (Co) elements within the metallohydrogel matrix (Fig. 3b–g). These results collectively validate the successful incorporation of the gel's primary constituents into a stable, interconnected supramolecular network, highlighting the material's compositional integrity and structural uniformity. | ||
| Fig. 3 (a) FESEM analysis reveals the microstructural features of the Co-LS metallohydrogel, (b–g) the elemental mapping of Co-LS metallohydrogel showing the presence of C, N, O, K and Co elements. | ||
3.3. FT-IR analysis of Co-LS metallohydrogel
Fourier Transform Infrared spectroscopy is an essential tool for investigating the functional groups and molecular interactions in metallogels. Fig. 4 shows the comparative FTIR spectra of Cobalt nitrate hexahydrate, L-serine and Cobalt-L Serine metallohydrogel. The spectrum of cobalt nitrate appears at ∼3400 cm−1 assigned to coordinated water molecules along with band around 1370 cm−1 attributed to stretching mode of nitrate ion. The spectrum of L-Serine exhibits a broad band around 3200–3400 cm−1 assigned to the overlapping of –NH2 and –OH stretching vibrations while asymmetric and symmetric stretching mode of carboxylate (COO–) group appear at 1600 and 1400 cm−1 respectively. After metallogel formation the asymmetric stretching band of carboxylate group shifts to 1570 cm−1 and symmetric band 1370 cm−1. The change in (ΔνCOO−) i.e., (νas–νs), indicate the coordination of carboxylate oxygen atom of L serine with Co2+ ion. On the other hand, the broad –NH/–OH become less intense and broader that implies the involvement of these groups in H bonding and metal ion coordination. The characteristic nitrate peak observed in Co(NO3)2·6H2O becomes significantly weaker, confirming an alternation in its coordination environment. These findings collectively confirm the successful formation of cobalt-L serine (Co-LS) metallogel network. | ||
| Fig. 4 FT-IR spectra of the xerogel form of Co-LS metallohydrogel, L-serine gelator and cobalt nitrate. | ||
3.4. PXRD analysis of Co-LS metallohydrogel
PXRD analysis of the Co-LS metallohydrogel was performed to examine its structural organization and crystalline features. The diffraction pattern, recorded over the 2θ range of 5°–80°, exhibits distinct reflections at 19.2°, 23.7°, 29.6°, 32.6°, 38.7°, 41.2°, 41.9°, and 48.3°, indicating the presence of ordered domains within the metallohydrogel network.PXRD measurements were conducted on the as-prepared gel without post-synthetic removal of KOH to preserve structural integrity. The diffraction features observed at 2θ values of 27.4°, 29.6°, 33.8°, 34.6°, and 38.7°, which are consistent with reported reflections of KOH, are attributed to residual alkaline species associated with the gel matrix.
Minor shifts in peak positions compared to pristine starting materials can be attributed to coordination-driven reorganization, hydrogen bonding interactions, and changes in lattice parameters occurring during gelation (Fig. 5).
3.5. Thin film deposition and device assembly
For assessing the electrical transport behavior and exploring the feasibility of the Co-LS metallohydrogel in semiconductor applications, forming a continuous thin film on conductive substrates such as Indium Tin Oxide (ITO) is crucial. However, due to the intrinsic viscoelastic nature of the gel, achieving a uniform coating is challenging. To mitigate this issue, Polymethylmethacrylate (PMMA) serves as a structural support, enabling effective dispersion of the gel in N,N-dimethylformamide (DMF) and improving its film-forming properties.The process begins with dissolving PMMA in DMF at 40 °C while stirring for two hours. Different concentrations of PMMA, ranging from 10 to 50 wt%, are incorporated into the gel and mixed for an additional four hours to examine the feasibility of film formation. The results indicate that a 50 wt% PMMA mixture produces the most homogeneous films, ensuring complete substrate coverage with minimal surface irregularities. In contrast, increasing the PMMA content beyond this threshold negatively impacts film integrity, leading to structural inconsistencies. Before deposition, ITO glass substrates undergo a rigorous cleaning process. The Co-LS-PMMA blend is then uniformly distributed over the substrate via spin-coating at 1000 rpm for 50 seconds, immediately followed by annealing at 60 °C for five minutes. To complete the assembly, a thin aluminum layer (100 nm) is deposited using a vacuum evaporation technique, creating a layered configuration of ITO/Co-LS-PMMA/Al. The electrical behavior of the resulting thin film device is subsequently evaluated through current–voltage (I–V) measurements.
3.6. Optical characterization
To determine the optical band gap, the optical absorbance spectrum of the synthesized metallogel was recorded. The UV-vis absorption spectrum in the wavelength range of 200–800 nm is presented in Fig. 6 (inset). The direct optical band gap energy of the metallogel was estimated using the following Tauc equation (i)| (αhν)2 = c(hν − Eg) | (i) |
3.7. Charge transport analysis and thin film device performance
The electrical characterization of the fabricated device was conducted through current–voltage (I–V) measurements usig a Keithley 2450 source meter. A bias voltage was applied within the range of 0 to ±2 V to evaluate the device's response. The resulting I–V characteristics for the device are illustrated in Fig. 7.The gel-based device demonstrates I–V characteristics comparable to those of a Schottky diode. The essential diode parameters are extracted using Cheung's method,41 and the I–V behavior is analyzed within the framework of Thermionic Emission theory. The corresponding standard equations are provided below.
 | (1) |
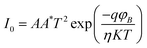 | (2) |
In this formulation, V is the applied bias voltage, k is the Boltzmann constant and q denotes the charge of an electron. The temperature is expressed in Kelvin as T, while I0 corresponds to the reverse saturation current. The effective diode area is given by A, and the Richardson constant A*, is assumed to be 32 AK−2 cm−2.
The extraction of the ideality factor (η), barrier height (φB), and series resistance (Rs) is conducted using Cheung's method, which relies on the analysis of the forward bias I–V characteristics. The method employs differential techniques to linearize the nonlinear response of the diode, facilitating accurate parameter estimation. The series resistance is determined from the slope of the modified function (d![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) V/d
V/d![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) I) versus current (Fig. 8). The barrier height is subsequently calculated using the extracted parameters in conjunction with the thermionic emission model. The governing equations for these calculations are provided below.
I) versus current (Fig. 8). The barrier height is subsequently calculated using the extracted parameters in conjunction with the thermionic emission model. The governing equations for these calculations are provided below.
 | (3) |
 | (4) |
Fig. 8 below shows the H(I) vs. I plot and as seen from the above equations, the slope and intercept of this plot combined with the ideality factor values can be used to determine the value of barrier height (φB). Fig. 9 presents the H(I) versus I plot, which serves as a crucial tool for extracting the barrier height (φB). As derived from the preceding equations, the slope and intercept of this plot, in conjunction with the ideality factor (n), facilitate the precise determination of (φB) (Table 1).
| Conductivity (Sm−1) | Ideality factor | Barrier height (eV) | Series resistance (Ω) | Mobility (cm2 V−1 s−1) | |
|---|---|---|---|---|---|
dV/d![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) I vs. I I vs. I |
H(V) vs. I | ||||
| 9.45 | 1.83 | 0.47 | 58.94 | 46.62 | 1.97 × 10−5 |
In this study, the charge carrier mobility was determined using the well-established Mott–Gurney equation,42 which is commonly employed for space-charge-limited current (SCLC) analysis in organic and semiconductor devices.
 | (5) |
In this equation, ε0 represents the permittivity of free space, while εr denotes the dielectric constant of the material blend. The parameter µ corresponds to the charge carrier mobility, and d is the thickness of the film.
The charge carrier mobility is extracted using the Mott–Gurney equation by analyzing the double logarithmic I–V plots of the devices, as shown in Fig. 10. This approach enables the identification of different conduction regimes and the determination of mobility in the space-charge-limited current (SCLC) region.
In the ohmic region (Region I), the current exhibits a direct proportionality to the applied voltage, as shown in Fig. 10. Conversely, in the space-charge-limited current (SCLC) region (Region II), the current follows a quadratic dependence on voltage.43,44 Therefore, Region II, identified in the figure, is utilized for mobility calculations. The segments of the curve exhibiting a slope of 2 (Region II) are specifically fitted to the Mott–Gurney equation (eqn (5)) to extract the charge carrier mobility values. In comparison to the traditional inorganic or organic semiconductors the mobility is (1.97 × 10−5 cm2 V−1 s−1) is low. However, the main goal of this work is to demonstrate semiconducting functionality in a soft, supramolecular metallohydrogel system rather than to compete with advanced semiconductors. Such values are typical of soft, supramolecular, and hydrogel-based semiconductors, where charge transport is governed by hopping and percolation through disordered metal–ligand networks and hydrated domains rather than band-like transport. For low-frequency and low-current applications, such as chemical and biosensors, soft rectifiers, and bio integrated electronic interfaces, comparable mobilities have been widely reported for metallogels and hydrogel-based electronic materials. In these systems, mechanical compliance, processability, and multifunctionality are often more critical than high charge carrier mobility.45,46
3.8. Inhibiting activity for pathogens
Co-LS demonstrated strong antimicrobial activity against both Gram-positive (Bacillus subtilis, Staphylococcus aureus) and Gram-negative (Escherichia coli, Pseudomonas aeruginosa) bacterial strains, as shown in Fig. 11. Streptomycin was used as a positive control at a concentration of 0.1 mg mL−1, which falls within the standard range for antibacterial assays. The Co-LS metallohydrogel was tested at 100 mg mL−1 to ensure sufficient diffusion and antimicrobial effect, as the physical gel matrix can potentially limit its dispersion in the growth medium. Both concentrations were optimized based on preliminary screening to ensure reliable detection of antimicrobial activity. Streptomycin, a broad-spectrum aminoglycoside antibiotic, inhibits bacterial protein synthesis by binding to the 30S ribosomal subunit. Due to its efficacy against a wide range of bacterial species, including uncommon strains, it serves as an effective positive control in zone of inhibition assays (Table 2).| Bacterial strain (s) | Volume of antibiotic given as positive control (µL) | Concentration of antibiotic used as positive control (mg mL−1) | Zone of inhibition (mm in diameter) |
|---|---|---|---|
| Escherichia coli | 5 | 1 | 13 ± 0.1 |
| Bacillus subtilis | 5 | 1 | 18.2 ± 0.1 |
| Pseudomonas aeruginosa | 5 | 1 | 13 ± 0.2 |
| Staphylococcus aureus | 5 | 1 | 19 ± 0.15 |
The minimum inhibitory concentration (MIC) of the metallogel Co-LS was determined against four bacterial strains. Distinct variations in antibacterial activity were observed among the tested strains. In the case of B. subtilis, strong zones of inhibition were observed starting at 80 µg mL−1, with the zone diameter increasing progressively at higher concentrations. Complete inhibition of bacterial growth was achieved at 80 µg mL−1 and above, indicating that the MIC for B. subtilis is approximately 80 µg mL−1. S. aures also showed marked susceptibility to the compound, with weak inhibition visible at 60 µg mL−1 and complete inhibition at 80 µg mL−1, suggesting an MIC value of around 80 µg mL−1.
By contrast, E. coli displayed moderate sensitivity. Inhibitory effect was noted only at 100 µg mL−1, while lower concentrations (20–80 µg mL−1) showed faint inhibition zones, indicating an MIC of approximately 100 µg mL−1. P. aeruginosa, known for its intrinsic resistance to many antimicrobial agents, exhibited the least sensitivity toward the test compound. It produced a faint zone across the concentrations, whereas no clear inhibition was observed even at 100 µg mL−1, suggesting partial resistance against Co-LS.
The control antibiotic, streptomycin, produced strong and well-defined zones of inhibition against all four bacterial species, validating the reliability of the assay and confirming the growth viability of the cultures.
Overall, the MIC values of the test compound were estimated to be 80 µg mL−1 for B. subtilis, 80 µg mL−1 for S. aureus, and 100 µg mL−1 for E. coli. These results suggest that the test compound possesses moderate to strong antibacterial potential, particularly against Gram-positive bacteria, and could serve as a promising candidate for further antimicrobial evaluation (Table 3).
| Sl no. | Bacterial strain | Zone of inhibition (mm in diameter) | |||||
|---|---|---|---|---|---|---|---|
| Positive control | Concentration of Co-LS (mg mL−1) | ||||||
| 20 mg mL−1 | 40 mg mL−1 | 60 mg mL−1 | 80 mg mL−1 | 100 mg mL−1 | |||
| 1. | Escherichia coli | 13 ± 0.2 | No zone | No zone | 10 ± 0.2 | 11 ± 0.21 | 13 ± 0.11 |
| 2. | Bacillus subtilis | 18 ± 0.22 | No zone | No zone | No zone | 10 ± 0.22 | 13 ± 0.15 |
| 3. | Pseudomonas aeruginosa | 11 ± 0.15 | No zone | 10 ± 0.12 | 12 ± 0.05 | 14 ± 0.1 | 11 ± 0.21 |
| 4. | Staphylococcus aureus | 15 ± 0.1 | No zone | 14 ± 0.15 | 15 ± 0.2 | 15 ± 0.11 | 19 ± 0.05 |
4. Conclusions
In this work, we report the successful synthesis of a supramolecular Co(II)-metallohydrogel via a rapid, room-temperature mixing of cobalt nitrate hexahydrate and L-serine in an aqueous medium. FESEM revealed a well-defined, hierarchical flake-like network, while rheological measurements confirmed the gel's mechanical robustness. FT-IR spectroscopy identified key non-covalent interactions responsible for gel formation. The metallohydrogel exhibited semiconducting behaviour, as demonstrated by optical band-gap analysis and its performance in an ITO/Co-LS-PMMA/Al thin-film diode device, where it showed distinct rectifying characteristics suited for optoelectronic applications. Notably, the gel also displayed potent antimicrobial activity, comparable to that of standard streptomycin, suggesting potential for pharmaceutical and biomedical use. This facile synthesis strategy offers a sustainable route for the development of multifunctional materials. Overall, the study presents a versatile platform for future advancements in healthcare, environmental technologies, and next-generation soft electronics through the rational design of supramolecular functional gels.Conflicts of interest
The authors declare no competing financial interests.Data availability
The authors declare that the data supporting the findings of this study are available within the paper. Should any raw data files be needed in another format they are available from the corresponding author upon reasonable request.Acknowledgements
S. S. is thankful to DST India, New Delhi for Inspire Fellowship (No. DST/inspire Fellowship/2022/IF220441). S. B. thankfully acknowledges DST Inspire Faculty Research Grant (Faculty Registration No.: IFA18-CH304; DST/INSPIRE/04/2018/000329).Notes and references
- D. Tripathy, A. S. Gadtya and S. Moharana, Polym.-Plast. Technol. Mater., 2023, 62, 306–326 CAS.
- S. Datta and S. Bhattacharya, Chem. Soc. Rev., 2015, 44, 5596–5637 RSC.
- A. Pape, M. Bastings, R. Kieltyka, H. Wyss, I. Voets, E. Meijer and P. Dankers, Int. J. Mol. Sci., 2014, 15, 1096–1111 CrossRef CAS PubMed.
- X. Ma and H. Tian, Acc. Chem. Res., 2014, 47, 1971–1981 CrossRef CAS PubMed.
- K. Karmakar, A. Dey, S. Dhibar, R. Sahu, S. Bhattacharjee, P. Karmakar, P. Chatterjee, A. Mondal and B. Saha, RSC Adv., 2023, 13, 2561–2569 RSC.
- S. Ganta and D. K. Chand, Dalton Trans., 2015, 44, 15181–15188 RSC.
- G. Yu, X. Yan, C. Han and F. Huang, Chem. Soc. Rev., 2013, 42, 6697 RSC.
- S. Dhibar, S. K. Ojha, A. Mohan, S. P. C. Prabhakaran, S. Bhattacharjee, K. Karmakar, P. Karmakar, P. Predeep, A. K. Ojha and B. Saha, New J. Chem., 2022, 46, 17189–17200 RSC.
- M. Shirakawa, N. Fujita and S. Shinkai, J. Am. Chem. Soc., 2003, 125, 9902–9903 CrossRef CAS PubMed.
- T.-A. Asoh and A. Kikuchi, Chem. Commun., 2012, 48, 10019 RSC.
- X. Yang, H. Zhang, J. Zhao, Y. Liu, Z. Zhang, Y. Liu and X. Yan, Chem. Eng. J., 2022, 450, 138135 CrossRef CAS.
- A. Rajak and A. Das, ACS Polym. Au, 2022, 2, 223–231 CrossRef CAS PubMed.
- Y. Xu, Q. Wu, Y. Sun, H. Bai and G. Shi, ACS Nano, 2010, 4, 7358–7362 CrossRef CAS PubMed.
- S. Some, P. Das, S. Pal, S. Dhibar, D. Kumari, S. Bhattacharjee, S. J. Ray, T. O. Ajiboye, S. Dam, P. P. Ray and B. Saha, RSC Adv., 2025, 15, 18392–18402 RSC.
- J. W. Steed, Chem. Soc. Rev., 2010, 39, 3686 RSC.
- K. Hanabusa, K. Hiratsuka, M. Kimura and H. Shirai, Chem. Mater., 1999, 11, 649–655 CrossRef CAS.
- A. Prathap and K. M. Sureshan, Langmuir, 2019, 35, 6005–6014 CrossRef CAS PubMed.
- A. Dawn and A. Ajayaghosh, Chem. Soc. Rev., 2010, 39, 3687–3700 Search PubMed.
- H. Sigel and R. B. Martin, Chem. Rev., 1982, 82, 385–426 CrossRef CAS.
- S. Dhibar, A. Roy, P. Das, T. Sarkar, M. Goswami, S. Some, K. Karmakar, P. Ruidas, S. Bhattacharjee, T. O. Ajiboye, A. S. Ray, K. Sarkar, S. J. Ray and B. Saha, Mater. Adv., 2025, 6, 1899–1913 RSC.
- A. Roy, S. Dhibar, K. Karmakar, S. Bhattacharjee, B. Saha and S. J. Ray, Sci. Rep., 2024, 14, 13109 CrossRef CAS PubMed.
- C. Wang, K. Xia, H. Zhang and X. Zhao, Chem. Soc. Rev., 2017, 46, 6764–6815 RSC.
- K. Karmakar, A. Roy, S. Dhibar, S. Majumder, S. Bhattacharjee, B. Mondal, S. M. Rahaman, R. Saha, S. J. Ray and B. Saha, ACS Appl. Electron. Mater., 2023, 5, 3340–3349 CrossRef CAS.
- M.-O. M. Piepenbrock, G. O. Lloyd, N. Clarke and J. W. Steed, Chem. Rev., 2010, 110, 1960–2004 CrossRef CAS.
- M. Suzuki and K. Hanabusa, Chem. Soc. Rev., 2009, 38, 967–975 RSC.
- S. Ganta and D. K. Chand, Dalton Trans., 2015, 44, 15181–15188 RSC.
- C. Po, Z. Ke, A. Y. Tam, H. Chow and V. W. Yam, Chem. – Eur. J., 2013, 19, 15735–15744 CrossRef CAS PubMed.
- B. Jiang, L.-J. Chen, G.-Q. Yin, Y.-X. Wang, W. Zheng, L. Xu and H.-B. Yang, Chem. Commun., 2017, 53, 172–175 RSC.
- S. Dhibar, A. Roy, P. Das, T. Sarkar, M. Goswami, S. Some, K. Karmakar, P. Ruidas, S. Bhattacharjee, T. O. Ajiboye, A. S. Ray, K. Sarkar, S. J. Ray and B. Saha, Mater. Adv., 2025, 6, 1899–1913 RSC.
- S. Dhibar, A. Mohan, R. Laha, S. Some, P. Kaith, A. Trivedi, S. Bhattacharjee, L. N. Nthunya, T. O. Ajiboye, A. Bera, S. K. Panja, A. K. Das, S. Dam, P. Predeep and B. Saha, RSC Adv., 2025, 15, 33494–33505 RSC.
- B. Pal, S. Dhibar, R. Mukherjee, S. Bhattacharjee, P. P. Ray and B. Saha, Mater. Adv., 2023, 4, 3628–3635 RSC.
- S. Datta and P. Dastidar, Chem. Soc. Rev., 2022, 51, 5580–5631 Search PubMed.
- S. Dhibar, A. Mohan, S. Babu, S. Bhattacharjee, S. Some, S. J. Ray, S. Roy, T. O. Ajiboye, P. Predeep and B. Saha, Discov. Mol., 2025, 2, 13 CrossRef.
- W.-L. Guan, K. M. Adam, M. Qiu, Y.-M. Zhang, H. Yao, T.-B. Wei and Q. Lin, Supramol. Chem., 2020, 32, 578–596 CrossRef CAS.
- E. M. M. Ibrahim, L. H. Abdel-Rahman, A. M. Abu-Dief, A. Elshafaie, S. K. Hamdan and A. M. Ahmed, Mater. Res. Bull., 2018, 107, 492–497 CrossRef CAS.
- E. M. M. Ahmed, L. H. Abdel-Rahman, A. M. Abu-Dief, A. Elshafaie, S. K. Hamdan and A. M. Ahmed, Phys. Scr., 2018, 93, 055801 CrossRef.
- S. Khodami, K. Kaniewska, J. Romanski, M. Karbarz and Z. Stojek, ACS Omega, 2025, 10, 12062–12075 CrossRef CAS PubMed.
- H. Sigel and R. B. Martin, Chem. Rev., 1982, 82, 385–426 CrossRef CAS.
- Metal Ions in Biological Systems, ed. H. Sigel, Vol. 2, Marcel Dekker, New York, 1973 Search PubMed.
- T. Shao, N. Falcone and H.-B. Kraatz, ACS Omega, 2020, 5, 1312–1317 CrossRef CAS PubMed.
- S. K. Cheung and N. W. Cheung, Appl. Phys. Lett., 1986, 49, 85–87 CrossRef CAS.
- P. N. Murgatroyd, J. Phys. D: Appl. Phys., 1970, 3, 151–156 CrossRef.
- D. Joung, A. Chunder, L. Zhai and S. I. Khondaker, Appl. Phys. Lett., 2010, 97, 093105 CrossRef.
- S. Dhibar, S. Some, S. Pal, R. Laha, P. Kaith, A. Trivedi, S. Bhattacharjee, L. N. Nthunya, T. O. Ajiboye, S. K. Panja, A. K. Das, A. Bera, S. Dam and B. Saha, RSC Adv., 2025, 15, 27544–27550 RSC.
- H. Sirringhaus, Adv. Mater., 2014, 26, 1319–1335 CrossRef CAS.
- V. Podzorov, Nat. Mater., 2013, 12, 443–444 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |









