Open Access Article
Open Access ArticleNovel chitosan hybrid fluorinated thiourea derivatives as dual anti-microbial and anti-biofilm agents: tailoring molecular interactions, in silico toxicity, and docking simulation
Moustafa S. Abusaif *ab,
Ahmed Ragab
*ab,
Ahmed Ragab *bc,
Mohamed A. Salemd,
Nabila A. Maziade and
Yousry A. Ammar
*bc,
Mohamed A. Salemd,
Nabila A. Maziade and
Yousry A. Ammar b
b
aDepartment for Synthesis and Characterization of Polymers, Polymer Institute of the Slovak Academy of Sciences, Dúbravská cesta 9, 845 41 Bratislava, Slovak Republic. E-mail: moustafa.samir@savba.sk
bDepartment of Chemistry, Faculty of Science (boys), Al-Azhar University, 11884 Nasr City, Cairo, Egypt. E-mail: mostafahozaifa317@azhar.edu.eg; ahmed_ragab@azhar.edu.eg
cChemistry Department, Faculty of Science, Galala University, Galala City, Suez 43511, Egypt. E-mail: ahmed.abdelwahab@gu.edu.eg
dHealth Specialties, Basic Sciences and Their Applications Unit, Applied College, Muhayl Asir, King Khalid University, Abha 62529, Saudi Arabia
ePolymer Chemistry Department, National Center for Radiation Research and Technology, Atomic Energy Authority, Cairo, Egypt
First published on 28th April 2026
Abstract
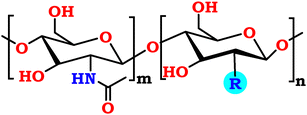
This article presents the synthesis of chitosan isothiocyanate, followed by hybridization with promising fluorinated aromatic amines, producing three chitosan fluorinated-thiourea samples (CHTUD1–3). Covalent modification of the chitosan and chitosan isothiocyanate backbones was confirmed by a notable reduction in ion exchange capacity (IEC). IEC decreased from 10.88 ± 0.23 for native chitosan and 3.21 ± 0.65 meq g−1 for CHNCS to 4.25 ± 0.35 meq g−1 for the final chitosan fluorinated-thiourea samples. This reduction strongly supports successful covalent grafting, meaning that the desired chemical modification was achieved. The modified chitosan (CHTUD1–3) was evaluated for antimicrobial activity. It showed significant effectiveness against the tested strains. These derivatives demonstrated significant antimicrobial activity, with MIC values ranging from 7.8 to 31.25 µg mL−1 against Gram-positive strains, whereas native chitosan exhibited an MIC of 62.5 µg mL−1. In contrast, the MIC values for Gram-negative strains ranged from 15.62 to 62.5 µg mL−1, compared with native chitosan, which showed an MIC of 125 µg mL−1 against the tested strains. The designed CHTUD1–3 derivatives demonstrated significant antimicrobial activity against S. aureus, with an MIC of 7.8 µg mL−1, which is significantly more promising than that of chitosan (62.5 µg mL−1), indicating that chemical modification with fluorinated derivatives enhances antibacterial potency. Furthermore, these derivatives also showed potential against C. albicans with an MIC of 15.62 µg mL−1, while native chitosan had an MIC of 62.5 µg mL−1. Regarding biofilm inhibition, the percentages against S. aureus and S. typhi showed significant dose-dependent responses. Specifically, the new derivatives achieved ≥90% inhibition at 75% MBC and 70% inhibition at 25% MBC. However, CHTUD2 exhibited 61.27% inhibition. In silico toxicity prediction revealed a favorable toxicity profile. Finally, molecular docking simulations revealed promising binding affinities. This suggests that these derivatives could inhibit biofilm formation by disrupting adhesion through sortase A and preventing biofilm formation via LpxC enzymes.
1. Introduction
The emergence of multidrug-resistant organisms poses a serious global health concern, emphasizing the urgent need for new drugs capable of treating bacterial and fungal diseases.1,2 So, creating and synthesizing new antimicrobial drugs remains a priority research goal. Out of them, novel promising aromatic compounds are being thoroughly examined as potential scaffolds to address microbial resistance to traditional treatments.3,4 In the same vein, there is a substantial movement in the field of materials science toward the replacement of synthetic polymers with environmentally friendly materials that are generated from renewable sources.5,6 Chitosan, defined as poly[(1 → 4)-β-linked 2-amino-2-deoxy-D-glucose], is a widely recognized natural polysaccharide, typically sourced from the shells of crustaceans such as crabs, shrimp, and crawfish.7,8Commercial production of chitosan, a notable semisynthetic polysaccharide, involves the partial deacetylation of chitin, the main structural component of crab shells. The result of this change is a polymer with new physicochemical properties, including increased bioactivity and enhanced solubility in acidic environments.9 The resulting biomaterial is highly suitable for sophisticated applications due to its multitude of “outstanding features.” These features include biodegradability, biocompatibility, intrinsic antibacterial activity, and minimal immunogenicity. Chitosan (CH) also has moisturizing properties and is very hygroscopic.10 Many industries, including medicine, food storage, farming, the textile industry, and environmental cleanup, utilize it because it possesses a strong set of qualities.11,12 In addition, the expanding applications of chitosan necessitate the development of versatile derivatives through innovative modification technologies to meet current industrial and biomedical requirements.13 The expanding applications of chitosan require the development of versatile derivatives through innovative modification technologies to meet current industrial and biomedical requirements.14
Chitosan's intrinsic activity, which stems from its abundance of hydroxyl and amino groups, makes it an ideal substrate for chemical modification. This flexibility is necessary for adjusting its physicochemical characteristics to particular applications.15 Significant reaction pathways involve the nucleophilic amino groups, which readily react with some electrophiles like aldehydes or ketones to form Schiff base derivatives (–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–)16,17 as well as acid chlorides or anhydrides to form amide derivatives (–CH–NH–CO–R),18 and isothiocyanates to form thiourea derivatives (–CH–NH–CS–NH–R).19 Among the distinguished functional derivatives of chitosan are the N-substituted thiourea compounds, which are generally synthesized through N-thiocarbamoylation utilizing isothiocyanate reagents. These analogs have shown great usefulness in a variety of applications. Some examples of thiourea chitosans include N-acetyl-, N-acyl-, N-fluoresceinyl-, and N-phenyl-thiourea chitosans.20,21 One study found that an N-acetyl-chitosan thiourea derivative could be used as a corrosion inhibition material.22 Another found that N-acyl-chitosans were effective antimicrobials,23 and still another found that N-phenyl-chitosans were effective macromolecular fluorophores and metal adsorbents.24,25 However, the commercial availability of the necessary isothiocyanate chemicals limits this synthesis process. A potentially more versatile strategy involves synthesizing a chitosan isothiocyanate intermediate. A polymer of this nature can subsequently undergo reactions with a diverse range of readily accessible amines, resulting in the formation of an extensive library of novel N-substituted thiourea derivatives. Notwithstanding this potential, and although the synthesis of glucosamine isothiocyanate derivatives via thiophosgene has been documented,26 there is a conspicuous absence of reports concerning analogous chitosan isothiocyanate derivatives that employ the C-2 amino group. For more clarification, one document only reported the synthesis of acyl chitosan isothiocyanate derivatives and investigated their performance of photon-to-electron conversion, indicating that they may serve as a promising material for this application.19
N–)16,17 as well as acid chlorides or anhydrides to form amide derivatives (–CH–NH–CO–R),18 and isothiocyanates to form thiourea derivatives (–CH–NH–CS–NH–R).19 Among the distinguished functional derivatives of chitosan are the N-substituted thiourea compounds, which are generally synthesized through N-thiocarbamoylation utilizing isothiocyanate reagents. These analogs have shown great usefulness in a variety of applications. Some examples of thiourea chitosans include N-acetyl-, N-acyl-, N-fluoresceinyl-, and N-phenyl-thiourea chitosans.20,21 One study found that an N-acetyl-chitosan thiourea derivative could be used as a corrosion inhibition material.22 Another found that N-acyl-chitosans were effective antimicrobials,23 and still another found that N-phenyl-chitosans were effective macromolecular fluorophores and metal adsorbents.24,25 However, the commercial availability of the necessary isothiocyanate chemicals limits this synthesis process. A potentially more versatile strategy involves synthesizing a chitosan isothiocyanate intermediate. A polymer of this nature can subsequently undergo reactions with a diverse range of readily accessible amines, resulting in the formation of an extensive library of novel N-substituted thiourea derivatives. Notwithstanding this potential, and although the synthesis of glucosamine isothiocyanate derivatives via thiophosgene has been documented,26 there is a conspicuous absence of reports concerning analogous chitosan isothiocyanate derivatives that employ the C-2 amino group. For more clarification, one document only reported the synthesis of acyl chitosan isothiocyanate derivatives and investigated their performance of photon-to-electron conversion, indicating that they may serve as a promising material for this application.19
Shibano and his colleagues developed a straightforward synthesis approach for acyl-chitosan isothiocyanates. This procedure employs a dual-phase approach: the initial N-phenylthiourea is achieved using phenyl isothiocyanate, followed by acylation through the application of either an acyl halide or an acyl anhydride. The intermediates produced consequently demonstrated improved solubility in the typically used organic solvents and were recognized as potentially useful precursors for the development of new functional chitosan derivatives.27 Qin et al. reported the synthesis of three innovative thiosemicarbazone chitosan derivatives through a condensation reaction involving thiosemicarbazide chitosan and three different aromatic aldehydes: benzaldehyde, 2-hydroxybenzaldehyde, and 4-methoxybenzaldehyde. The in vitro antimicrobial and antifungal activity of these compounds was then tested against four prevalent crop-threatening pathogenic fungi and was found to be a remarkably effective material.
On the other hand, the strategic incorporation of specific functional groups serves as a fundamental approach to bestow novel properties upon a modified compound. The thiourea subunits, in particular, are well-known as beneficial components in various areas of study, including medicinal chemistry and organic synthesis.28 Urea/thiourea compounds have gained popularity in recent years owing to their valuable chemical and biological characteristics. Both the acidity of the adjustable NH protons and their ability to function as hydrogen-bond donors make them among the most often used groups in the creation of organocatalysts and anion sensors.29 Additionally, the capacity to form H-bonds has enabled the implementation of applications in crystal engineering and as membrane transporters.30 The thiourea group serves as a crucial pharmacophore, characterized by its N–H groups and sulfur atom, which function as hydrogen bonding domains.31 This structure is linked to a wide range of biological activities, encompassing anti-HIV,32 analgesic,33 antibacterial,34 antitumor,35 antifungal,36 antiviral,37 and anti-inflammatory properties,38 among others. Moreover, the deliberate incorporation of fluorine or fluorine-containing groups, such as the trifluoromethyl (–CF3) moiety, has become a significant strategy in the advancement of biologically active compounds.39,40 The trifluoromethyl group in heterocyclic chemistry is notable for its capacity to modify the physicochemical and biological properties of the parent molecule.41 The insertion of fluorine alters important steric and electronic characteristics, impacting a drug's pharmacological and pharmacokinetic effects. The trifluoromethyl group has a distinctive profile in terms of bioisosterism, as it is sterically larger than a methyl group and smaller than an isopropyl group.42,43 The numerous US FDA-approved medications that contain fluorinated scaffolds are a testament to the potent pharmacological utility of these structures.44
Among potential modifications, the combination of modified chitosan isothiocyanate with the aromatic fluorinated moiety is of particular interest. This approach utilizes the potential for a complementary or synergistic effect, given that both moieties exhibit recognized antimicrobial properties. There is a high probability that incorporating quinoxalines, known for their potent biological activity, will enhance the natural bioactivity of chitosan. This strategy aims to develop robust derivatives that demonstrate enhanced effectiveness for applications in biomedical and environmental fields.
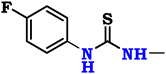
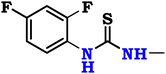
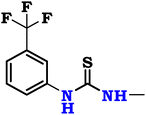
So, the article describes in depth a novel approach to synthesizing modified chitosan isothiocyanates via two different pathways. This study also examines the reactivity of these isothiocyanates with promising fluorinated amines (Scheme 1). This significant modification, not previously documented, is expected to produce a material with notably improved antimicrobial properties, making it suitable for a range of medical and environmental uses. Finally, we assessed the efficacy of these samples as antimicrobial agents through in vitro assays targeting the pathogenic bacteria B. subtilis (ATCC 6633), S. aureus (ATCC 6538), K. pneumoniae (ATCC 13883), and S. typhi (ATCC 6539), as well as the fungus C. albicans (ATCC 10221).
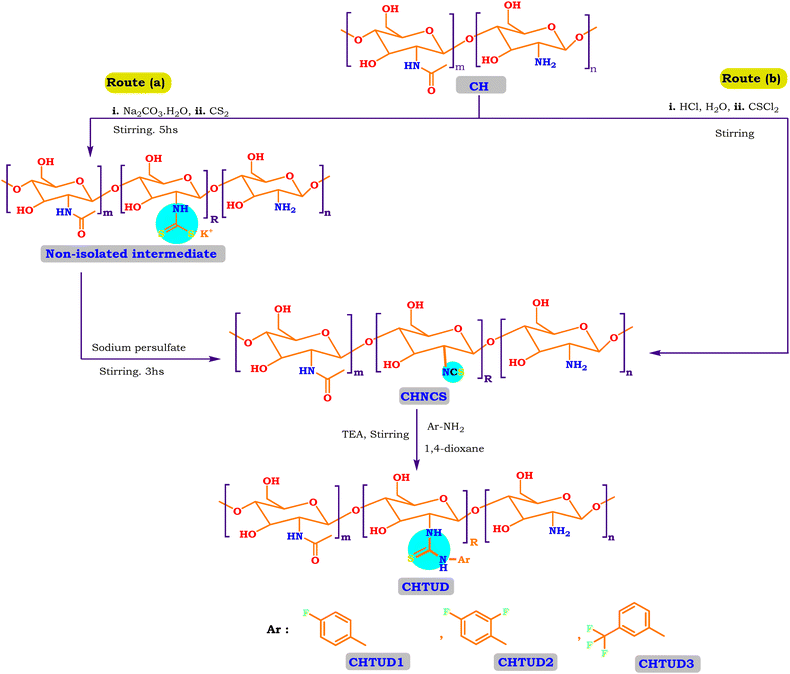 | ||
| Scheme 1 Various synthetic pathways of novel chitosan isothiocyanate (CHNCS) and chitosan thiourea derivatives (CHTUD). | ||
2. Experimental section
2.1. Materials
Chitosan, a white powdered solid, was acquired from Qingdao Yunzhou Biochemistry Co., Ltd in China, exhibiting an 85% deacetylation degree and an average molecular weight of 40 KDa g mol−1. P-Fluoroaniline (FA; purity = 98%, ID = 9349), 2,4-difluoroaniline (DFA; purity = 99%, ID = 9328), and m-trifluoromethyl aniline (TFA; purity = 96%, ID = 7097) were obtained from ChemSpider, United Kingdom. Carbon disulfide (CS2; purity ≥95%, ID = 1131) was acquired from CAMEO Chemicals, United States, while sodium persulfate (SPS; purity ≥99%, ID = 71![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 889), thiophosgene (CSCl2; purity ≥97%, ID = 79
889), thiophosgene (CSCl2; purity ≥97%, ID = 79![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 380), and triethylamine (Et3N; purity ≥99.5%, ID = 90
380), and triethylamine (Et3N; purity ≥99.5%, ID = 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 335) were obtained from Sigma Aldrich, Germany. The required solvents, like 1,4-doxane (Et3N; purity ≥99.8%, ID = 57
335) were obtained from Sigma Aldrich, Germany. The required solvents, like 1,4-doxane (Et3N; purity ≥99.8%, ID = 57![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 648
648![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 388) and acetone (C), were purchased from Sigma Aldrich, Germany. Ammonia solution (NH4OH; 26%) was acquired from El-Nasr company, Egypt.
388) and acetone (C), were purchased from Sigma Aldrich, Germany. Ammonia solution (NH4OH; 26%) was acquired from El-Nasr company, Egypt.
2.2. Assessments
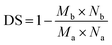 | (1) |
We used a back-titration approach to find the ion exchange capacities (IECs) of the functionalized derivatives (CHNCS, CHTUD1–3) and native chitosan (CH). Samples with known weights were submerged in a 0.1 M H2SO4 solution for 10 hours. After filtration, the residual acid in the filtrate was measured through titration using a 0.1 N NaOH standard solution. A blank titration (without a sample) was conducted as a control. The IEC was then determined based on the data using the following equation:46
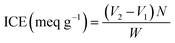 | (2) |
The proton signals of the three newly synthesized thiourea derivatives (CHTUD1–3) were meticulously characterized through the application of 1H NMR spectroscopy. Spectra were obtained using a JEOL JNM-ECA 400 II spectrometer (Japan) functioning at a frequency of 400 MHz. Samples were solubilized utilizing trifluoroacetic acid (TFA-d) and DMSO-d6 as the solvents of choice.
An X-ray diffraction (XRD) analysis was performed on the samples using Cu-Kα radiation on a PW1830 diffractometer (Japan). This allowed for the examination of the materials' crystalline structure.47 We gathered data throughout a 2θ scanning range that extended from 5° to 50°. To determine the crystalline index (CI), the following equation was employed. This equation is based on the peak height measurements of the crystalline region (110) and the amorphous region (Iam).
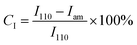 | (3) |
In the above formula, I110 indicates the highest intensity at 2θ°, and Iam corresponds to the intensity of amorphous diffraction at 15.7°.
The thermal stability of native chitosan and the synthesized derivatives was assessed utilizing a TGA (SDT Q600 V20.9 Build 20). The samples were subjected to heating from ambient temperature to 600 °C at a constant rate of 10 °C min−1 within a nitrogen atmosphere, with a flow rate of 5 mL min−1 maintained. The integrated TA-50WSI thermal analysis system was utilized for data collection and management.
Moreover, the surface morphology of the native chitosan and the modified chitosan thiourea derivatives (CHTUD1–3) was examined utilizing a JEOL JSM-6360LA scanning electron microscope (SEM) from Tokyo, Japan. Micrographs were acquired using an accelerating voltage of 10.0 kV, with initial magnifications set at ×1000 and ×2000.
2.3. Synthetic methods
Route (b): native chitosan (10 mmol, 1.6 gm) was dissolved in an aqueous hydrochloric acid solution with a pH of 2.5 while being stirred for 3 h at 0 °C to obtain a clear homogeneous solution. Then, thiophosgene (12 mmol, 0.912 mL) was added to the chitosan solution in one portion. Stirring immediately started at room temperature until the red color of thiophosgene completely disappeared, followed by a proper quantity of pure acetone being added to the mixture, resulting in the formation of the isocyanate derivative. The pale-white precipitate was filtered out, freeze-dried for 3 h, washed several times with acetone, and dried in a vacuum with N2 bubbling. Also, chitosan isothiocyanate (CHNCS) is obtained as a pale-white powder (Scheme 1).
2.4. Antimicrobial evaluation
| Biofilm inhibition (%) = [1 − (Acontrol − Ablank)(Asample − Ablank)] × 100 |
3. Results and discussion
3.1. Synthesis of the chitosan thiourea (CHTUD) derivative
Nucleophilic addition reaction involving isothiocyanate and variable amines represented a widely accepted approach for the synthesis of thiourea derivatives.56,57 But, due to the high cost and unavailability of all required isothiocyanate derivatives to react with chitosan, the authors planned to construct a modified chitosan isothiocyanate to be available to react with variable amines. Also, in the synthetic methodologies pertaining to chitosan thiourea (CHTUD1–3), novel chitosan isothiocyanate (CHNCS) emerged as a significant intermediate. In this study, the CHNCS key intermediate can be achieved through two routes: (a) through elimination reaction of non-isolable chitosan carbamodithioate using sodium persulfate; (b) through a one-step reaction of chitosan with thiophosgene in an acidic medium. For more details on route (a), chitosan isothiocyanate (CHNCS) was produced in a one-pot synthesis by combining chitosan with carbon disulfide in a basic medium, which afforded the non-isolable chitosan carbamodithioate, then subsequently eliminated using sodium persulfate (Scheme 1). This approach prevented the isolation and purification of each synthesized intermediate, thereby conserving time, solvents, and energy-intensive processes. Also, this approach is explained using the suggested mechanism as depicted in Fig. 1 under ideal circumstances. In a basic environment, an amine attacks the electrophilic carbon of carbon disulfide (CS2) to generate a carbamodithioic acid. This acid, when combined with a base, forms a stable dithiocarbamate salt. The amine attacks the central carbon of CS2, generating a zwitterionic intermediate (route a). The base subsequently deprotonates it to create the dithiocarbamate salt, a flexible intermediate for future reactions. Next, the following step involved the conversion of non-isolable dithiocarbamate salt to the corresponding isothiocyanate compound (CHNCS).58 It is widely recognized that the persulfate anion readily experiences homolytic cleavage in solutions, resulting in the formation of a sulfate radical anion. Single electron transfer (SET) from the chitosan dithiocarbamate intermediate to the sulfate radical anion generates carbamodithionyl radicals (A) and releases the sulfate anion. The process of desulfurization of carbamodithionyl radicals (A) continues, leading to the formation of carbamothionyl radicals (B), which the radical scavenger TEMPO may intercept. The sulfate radical anion abstracts hydrogen from carbamothionyl radicals (B), resulting in the formation of isothiocyanates (CHNCS) see Fig. 1. | ||
| Fig. 1 The mechanism illustrates the two different routes of preparation of chitosan isothiocyanate (CHNCS). | ||
Moreover, chitosan isothiocyanate (CHNCS) is produced using another synthetic pathway through one step, which involves the reaction of chitosan with thiophosgene in an acidic medium, and the suggested mechanism is depicted in Fig. 1 (route b). The interaction between an amine function in the chitosan skeleton and thiophosgene (CSCl2), leading to the formation of an isothiocyanate (CH–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S), occurs through a nucleophilic attack by the amine on the electrophilic carbon of thiophosgene. This process results in the generation of an intermediate, followed by the elimination of two molecules of hydrogen chloride (HCl). Firstly, the nucleophilic nitrogen atom of the principal amine hits the electrophilic carbon atom in the middle of thiophosgene (CSCl2).59 In doing so, it splits the double bond between carbon and sulfur and creates a tetrahedral intermediate. A proton is transferred from nitrogen to one of the chlorine atoms, and concurrently or in a subsequent step, one molecule of HCl is eliminated, resulting in the formation of a thiocarbamoyl chloride intermediate. The removal of the second molecule of HCl results in the synthesis of the desired isothiocyanate (CHNCS) along with a base molecule or residue.
S), occurs through a nucleophilic attack by the amine on the electrophilic carbon of thiophosgene. This process results in the generation of an intermediate, followed by the elimination of two molecules of hydrogen chloride (HCl). Firstly, the nucleophilic nitrogen atom of the principal amine hits the electrophilic carbon atom in the middle of thiophosgene (CSCl2).59 In doing so, it splits the double bond between carbon and sulfur and creates a tetrahedral intermediate. A proton is transferred from nitrogen to one of the chlorine atoms, and concurrently or in a subsequent step, one molecule of HCl is eliminated, resulting in the formation of a thiocarbamoyl chloride intermediate. The removal of the second molecule of HCl results in the synthesis of the desired isothiocyanate (CHNCS) along with a base molecule or residue.
Finally, the chitosan isothiocyanate structure (CHNCS) is used as an electrophile through its reaction with some promising selective amines, fluoroaniline, difluoroaniline and trifluoromethylaniline, which furnished the target thiourea derivatives, CHTUD1–3, respectively (Scheme 1). Amines undergo a reaction with isothiocyanates to yield thioureas, facilitated by the nucleophilic attack of the amine on the electrophilic carbon present in the isothiocyanate group. With the help of an alkaline setting and deprotonated amines, this reaction may generate a stable thiourea bond without the need for a leaving group.60
3.2. Calculations of substitution degree and ion exchange capacity assessments
Color and yield percentage of the chitosan isothiocyanate (CHNCS) and chitosan thiourea derivative (CHTUD) are assessed and depicted in Table 1. It was found that all product samples are different in color, which is more evidence for successful reactions. The CHNCS sample is obtained as a pale white color with 66.7% yield, while the corresponding thiourea derivatives (CHTUD1–3) are obtained as light yellow, yellow, and orange colors, with 73.4%, 78.5%, and 84.3% yield, respectively.| Sample | Yielda (%) | Color | Elemental analysis, calcd (found)b (%) | DSc | DSd | |||
|---|---|---|---|---|---|---|---|---|
| C | N | H | C/N | |||||
| a Yield is the ratio of CH–isothiocyanate and CH–thiourea derivative.b Theoretical elemental analysis based on the empirical formula of (C8H13NO5)m(C6H11NO4)n(C6H10NO4 + thiourea moiety)n−z, where m = 0.15 (molar ratio of chitin), y = 0.85 is the degree of deacetylation of CH and z is the experimental value of the DS of the prepared CH–thiourea derivative.c Degree of substitution of the CH–isothiocyanate and CH–thiourea derivative determined from elemental analysis.d Degree of substitution of the CH–thiourea derivatives determined from 1H NMR. | ||||||||
| CH | — | White | 45.22 (46.02) | 8.10 (7.67) | 7.27 (6.90) | 45.22 (46.02) | — | — |
| CHNCS | 66.7 | Pale white | 44.70 (44.36) | 7.98 (7.39) | 6.53 (6.03) | 44.70 (44.36) | 0.54 | — |
| CHTUD1 | 73.4 | Light yellow | 46.23 (47.51) | 8.89 (8.24) | 6.44 (6.93) | 46.23 (47.51) | 0.51 | 0.47 |
| CHTUD2 | 78.5 | Yellow | 45.87 (46.48) | 8.77 (8.03) | 6.07 (5.63) | 45.87 (46.48) | 0.52 | 0.45 |
| CHTUD3 | 84.3 | Orange | 45.21 (46.77) | 8.90 (7.52) | 4.87 (5.82) | 45.21 (46.77) | 0.50 | 0.46 |
Furthermore, an elemental analysis was conducted to ascertain the substitution degree (DS) of the modified chitosan samples using eqn (4).61
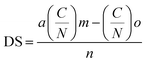 | (4) |
So, the degree of substitution of the CHNCS sample was 0.54, while the modified samples CHTUD1–3 were found to be 0.51, 0.52, and 0.50, respectively. The presented results indicate that the DS of all modified chitosan samples is around 0.50. In certain instances, the antimicrobial efficacy may be influenced directly by the extent of substitution, as the thiourea function provides a platform for improving the bioactivity of chitosan thiourea derivatives. Elemental analysis was used to calculate the M.wt. of the formula (C8H13NO5)0.15(C6H11NO4)0.85(C6H10NO4 + thiourea moiety)0.85−z, and the determined DS (z) aligned with the DS that really occurred.
At the same time, assessments of ion exchange capacity (IEC) may reveal the preparation of new thiourea functionalities through the presence of the amine group (NH2) within the chitosan framework. So, the IEC findings for the original chitosan indicated the highest value of 10.88 ± 0.23 meq g−1. Chemical modification of chitosan into CHNCS resulted in a decrease in the IEC value of 3.21 ± 0.65 meq g−1. Also, the three modified thiourea samples (CHTUD1–3) resulted in a reduction of the IEC value of 3.21 ± 0.65 meq g−1. The IEC findings indicated reduced values of 3.89 ± 0.26, 4.25 ± 0.13, and 4.13 ± 0.35 meq g−1, respectively. Moreover, the noted decrease can be ascribed to the chemical modification of chitosan into isothiocyanate or thiourea derivatives. In addition, the nitrogen atoms in the thiourea function (NH–CS–NH) did not affect the ion exchange capacity since they are not ionizable under the conditions tested. Consequently, under the experimental circumstances (employing 0.1 M H2SO4), the NH of thiourea function stays unprotonated and does not significantly affect the ion exchange capacity, owing to its acidic NH rather than fundamental nature. The changes observed in IEC can be primarily attributed to the development of the target function, rather than the protonation of the nitrogen. The measurements of ion exchange capacity (IEC) demonstrated a significant correlation with the observed degree of substitution.
3.3. Solubility test
The solubility characteristics of native chitosan (CH) and its modified derivatives (CHNCS, CHTUD1–3) were examined across a range of solvents, with the findings provided in Table 2. Native chitosan exhibited a distinct solubility profile: it was insoluble in distilled water due to extensive inter- and intramolecular hydrogen bonding, yet it dissolved readily in 1% acetic acid after the protonation of its amino groups. Chitosan demonstrated swelling in strong polar organic solvents such as DMSO, rather than achieving complete dissolution.62 Chemical changes, on the other hand, drastically changed these characteristics. There was a significant decrease in the solubility of the CHNCS intermediate and the CHTUD1–3 final thiourea derivatives in 1% acetic acid. The observed decrease in acid solubility indicates a substantial level of substitution, leading to a reduction in the availability of free amino groups for protonation. The derivatives demonstrated enhanced affinity for organic solvents; the modified thiourea samples (CHTUD1–3) exhibited high solubility in DMSO and displayed swelling behavior in DMF and CHCl3. While the CHNCS intermediate was insoluble in most organic solvents, it was observed to swell in DMSO and DMF.| Sample code | Solubility | |||||||
|---|---|---|---|---|---|---|---|---|
| Distilled H2O | 1% AcOH | DMSO | DMF | CHCl3 | MeOH | EtOH | THF | |
| Solubility parameter; + soluble; − insoluble; ± swell. | ||||||||
| CH | − | + | ± | − | − | − | − | − |
| CHNCS | − | − | ± | ± | − | − | − | − |
| CHTUD1 | − | − | + | ± | ± | − | − | − |
| CHTUD2 | − | − | + | ± | ± | − | − | − |
| CHTUD3 | − | − | + | ± | ± | − | − | − |
The inability of chitosan thiourea derivatives to dissolve in water and most organic solvents, along with their swelling capacity in polar solvents like DMSO and DMF, highlights their potential application in water purification systems. Insoluble materials serve as efficient adsorbents for pollutants, heavy metals, and pigments, as they can be readily recovered and reused without disintegrating in the treatment solution. The observed swelling behavior in polar solvents such as DMSO and DMF indicates a potential for matrix-controlled release of active agents. This may be utilized in applications including the creation of antimicrobial coatings or targeted remediation systems. Lastly, a shift in the solubility character is significant evidence for the successful synthesis of the modified thiourea samples (CHTUD1–3).
3.4. Instrumental characterization
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O axial stretching of the residual acetamide group) and a peak at 1590 cm−1 representing the N–H bending vibration of the primary amino group.63 Asymmetric stretching of the C–O–C ether linkages and skeletal vibrations of the saccharide ring were attributed to the firm peaks at 1130 cm−1, 1080 cm−1, and 1020 cm−1, respectively, in the fingerprint area, while absorptions at 1410 cm−1 and 1390 cm−1 were associated with CH3 and CH2 bending.55 In contrast, the spectrum of the CHNCS sample (Fig. 2b) showed obvious indications of effective functionalization. The most prominent new feature was the appearance of a sharp, intense absorption band at 2071 cm−1, characteristic of the asymmetric stretching vibration of the isothiocyanate (–N
O axial stretching of the residual acetamide group) and a peak at 1590 cm−1 representing the N–H bending vibration of the primary amino group.63 Asymmetric stretching of the C–O–C ether linkages and skeletal vibrations of the saccharide ring were attributed to the firm peaks at 1130 cm−1, 1080 cm−1, and 1020 cm−1, respectively, in the fingerprint area, while absorptions at 1410 cm−1 and 1390 cm−1 were associated with CH3 and CH2 bending.55 In contrast, the spectrum of the CHNCS sample (Fig. 2b) showed obvious indications of effective functionalization. The most prominent new feature was the appearance of a sharp, intense absorption band at 2071 cm−1, characteristic of the asymmetric stretching vibration of the isothiocyanate (–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S) functional group. Concurrently, there was a significant decrease in the intensity of the peak at around 1590 cm−1, which was earlier designated as 1580 cm−1 in the derivative and was related to the N–H bending of the primary amine. This weakening demonstrates that the substitution reaction devoured the principal amino groups.19 Moreover, the residual acetamide signal exhibited a shift to 1628 cm−1. The observed spectral alterations collectively validate the successful synthesis of the chitosan isothiocyanate (CHNCS) intermediate.
S) functional group. Concurrently, there was a significant decrease in the intensity of the peak at around 1590 cm−1, which was earlier designated as 1580 cm−1 in the derivative and was related to the N–H bending of the primary amine. This weakening demonstrates that the substitution reaction devoured the principal amino groups.19 Moreover, the residual acetamide signal exhibited a shift to 1628 cm−1. The observed spectral alterations collectively validate the successful synthesis of the chitosan isothiocyanate (CHNCS) intermediate.
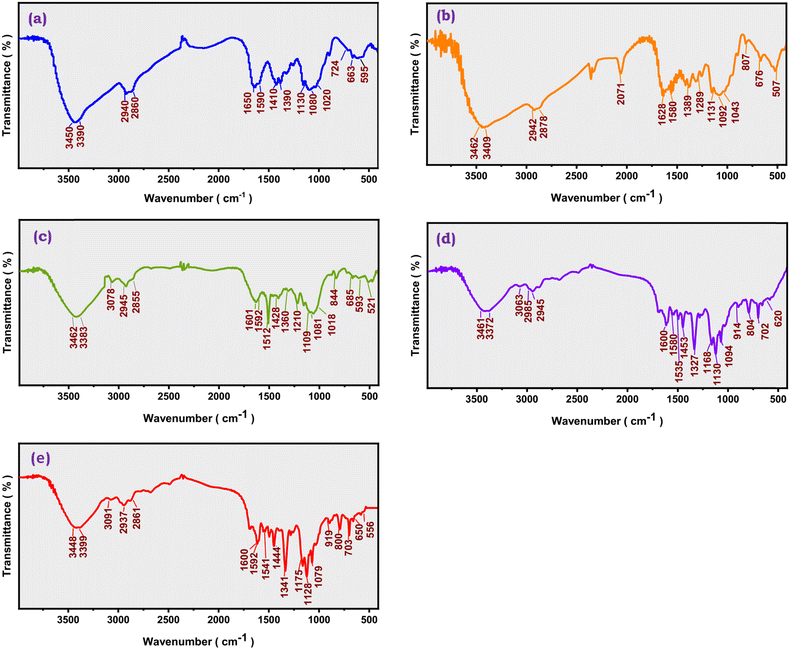 | ||
| Fig. 2 FT-IR analysis of chitosan (a) and its modified isothiocyanate CHNCS (b) and thiourea derivatives CHTUD1–3 (c–e). | ||
| Compound | FT IR (ν/cm−1) | ||||
|---|---|---|---|---|---|
| CH | CHNCS | CHTUD1 | CHTUD2 | CHTUD3 | |
| –NH2 | –NCS | –CS(NH)2–4-F–C6H4 | –CS(NH)2–2,4-(F)2–C6H3 | –CS(NH)2–3-CF3–C6H4 | |
| OH and NH2 stretching | 3450, 3390 | 3463, 3409 | 3462, 3383 | 3461, 3372 | 3448, 3399 |
| CH stretching (SP2–CH) | — | — | 3078 | 3063 | 3091 |
| CH stretching (SP3–CH) | 2940, 2860 | 2942, 2878 | 2945, 2855 | 2985, 2845 | 2937, 2861 |
| –NCS stretching | — | 2071 | — | — | — |
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching (NHCOCH3) O stretching (NHCOCH3) |
1650 | 1628 | 1601 | 1600 | 1600 |
| NH2 bending | 1590 | 1580 | 1592 | 1580 | 1592 |
| NH–CS–NH stretching | — | — | 1512 | 1535 | 1444 |
| –C–F stretching | — | — | 1210 | 1327 | 1341 |
| C–O–C stretching (glucoside linkage) | 1131, 1092 | 1131, 1092 | 1109, 1081 | 1130, 1094 | 1128, 1079 |
After modifying chitosan isothiocyanate to thiourea derivatives (CHTUD1–3), FT-IR charts of the thiourea derivatives indicate the lack of the characteristic band associated with the isothiocyanate group (–NCS) at 2070 cm−1. The appearance of several new, significant peaks characterized the spectrum of CHTUD1 (Fig. 2c). A strong band emerged at 1512 cm−1, which is ascribed to the N–H bending and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S stretching vibrations of the newly formed thiourea group (–NH–CS–NH–). Concurrently, additional bands at 3080 cm−1 (aromatic sp2 C–H stretching) and 1210 cm−1 (C–F bond stretching) emerged, which validated the presence of the fluorinated phenyl moiety (Fig. 2c). Similar evidence was observed for CHTUD2 (Fig. 2d). Its successful synthesis was indicated by the emergence of characteristic new bands at 1535 cm−1 (–NH–CS–NH– group), 3063 cm−1 (aromatic sp2 C–H), 1327 cm−1 (C–F stretching), and 702 cm−1 (phenyl ring vibration). In the case of CHTUD3 (Fig. 2e), unique absorption bands that corroborate the structure were detected at 1444 cm−1 (associated with the –NH–CS–NH– linkage), 3091 cm−1 (aromatic sp2 C–H), 1341 cm−1 (C–F3 group), and 703 cm−1 (phenyl ring). The primary absorption frequencies of the chitosan backbone, such as the wide O–H/N–H stretching (∼3462–3383 cm−1), the C–H stretching (∼2945–2855 cm−1), and the C–O–C skeletal vibrations (∼1109–1018 cm−1), were still present in all three of the derivative spectra (CHTUD1–3). This proves that the change did not damage the polysaccharide backbone.
S stretching vibrations of the newly formed thiourea group (–NH–CS–NH–). Concurrently, additional bands at 3080 cm−1 (aromatic sp2 C–H stretching) and 1210 cm−1 (C–F bond stretching) emerged, which validated the presence of the fluorinated phenyl moiety (Fig. 2c). Similar evidence was observed for CHTUD2 (Fig. 2d). Its successful synthesis was indicated by the emergence of characteristic new bands at 1535 cm−1 (–NH–CS–NH– group), 3063 cm−1 (aromatic sp2 C–H), 1327 cm−1 (C–F stretching), and 702 cm−1 (phenyl ring vibration). In the case of CHTUD3 (Fig. 2e), unique absorption bands that corroborate the structure were detected at 1444 cm−1 (associated with the –NH–CS–NH– linkage), 3091 cm−1 (aromatic sp2 C–H), 1341 cm−1 (C–F3 group), and 703 cm−1 (phenyl ring). The primary absorption frequencies of the chitosan backbone, such as the wide O–H/N–H stretching (∼3462–3383 cm−1), the C–H stretching (∼2945–2855 cm−1), and the C–O–C skeletal vibrations (∼1109–1018 cm−1), were still present in all three of the derivative spectra (CHTUD1–3). This proves that the change did not damage the polysaccharide backbone.
The complete absence of the sharp–NCS peak (at 2071 cm−1) alongside the emergence of new bands associated with the –NH–CS–NH– linkage and the distinct aromatic substituents serve as clear evidence that the phenyl thiourea derivatives have been effectively integrated into the chitosan backbone.
 | ||
| Fig. 3 1H NMR analysis of chitosan (a) and its chitosan-modified thiourea derivatives CHTUD1–3 (b–d). | ||
| Compound | 1H NMR (δ/ppm) | |||
|---|---|---|---|---|
| CH | CHTUD1 | CHTUD2 | CHTUD3 | |
| –NH2 | –CS(NH)2–4-F-C6H4 | –CS(NH)2–2,4-(F)2–C6H3 | –CS(NH)2–3-CF3–C6H4 | |
| CH3 (NHCOCH3) | 1.72 | 1.77 | 1.80 | 1.85 |
| H2 | 3.40 | 3.16 | 3.01 | 3.34 |
| H3, H4, H5, H6 (non-anomeric) | 3.49–4.48 | 3.31–4.85 | 3.08–4.42 | 3.47–4.51 |
| H1 (anomeric) | 4.70 | 4.98 | 4.60 | 4.72 |
| H-aromatic | — | 7.29 (2H, d, J = 9.6 Hz, H8), 7.75 (2H, d, J = 6.8 Hz, H7) | 6.35 (1H, d, J = 7.2 Hz, H7), 6.84 (1H, d, J = 8.0 Hz, H8), 7.54–7.64 (1H, m, H9) | 7.16 (1H, d, J = 9.6 Hz, H7), 7.99 (1H, d, J = 8.8 Hz, H9), 8.11 (1H, d, J = 7.6 Hz, H8), 8.19 (1H, s, H10) |
| –NH–CS–NH– | — | 7.47 (H9), 10.66 (H10) | 7.32 (H10), 10.59 (H11) | 7.79 (H11), 11.21 (H12) |
Moreover, Fig. 3b–d shows the spectra of the novel modified chitosan thiourea, including 4-fluoroaniline (CHTUD1), 2,4-difluoroaniline (CHTUD2), and 3-trifluoromethylaniline (CHTUD3). Crucially, the spectra revealed further signals that corroborate the effective grafting of the modified phenyl thiourea molecules. According to the chart of the CHTUD1 sample, the establishment of the new thiourea (–NH–CS–NH–) function was clearly demonstrated by the emergence of two new, deshielded singlet signals that correspond to the two N–H protons (Fig. 3b). The NH proton (H9) resonated at δ 7.47 ppm, whereas the less protected NH proton (H10) emerged at δ 10.66 ppm. Conversely, the protons of the new thiourea (–NH–CS–NH–) function in the CHTUD2 sample depicted at δ 7.32 ppm and 10.58 ppm related to the H10 and H11, respectively.65 Meanwhile, the 1H NMR spectrum of the CHTUD2 sample revealed the protons of the new thiourea at δ 7.79 and 11.21 ppm, owing to the H11 and H12, respectively (Fig. 3c). For the new aromatic protons in the CHTUD1 sample, two multiplets in the aromatic region, which are typical of a 1,4-disubstituted (para) ring, showed the 4-fluorophenyl ring. Doublet signals displayed at δ 7.29 ppm (H7, J = 9.6 Hz) and other doublet signals at δ 7.75 ppm (H8, J = 6.8 Hz) are for the two pairs of aromatic protons. The three aromatic protons in the CHTUD2 sample appeared as one doublet–doublet signal at δ 6.35 ppm, referring to the H7 proton, and multiplet signals due to the H8 and H9 protons at δ 6.81–7.74 ppm, respectively. In contrast, the aromatic protons in the CHTUD3 sample appeared as four signals, two doublet signals at δ 7.16 ppm & 7.99 ppm assigned for H7 and H9 protons, respectively, as well as a triplet signal due to the H8 proton displayed at δ 8.11 ppm (J = 7.6 Hz), besides a singlet signal at δ 8.19 ppm assigned for the H10 proton (Fig. 3d).66 Meanwhile, the 1H NMR spectra of the three novel samples CHTUD1, CHTUD2, and CHTUD3 exhibited the original signals of the chitosan backbone.
Lastly, the simultaneous detection of the native chitosan signals alongside the newly introduced aromatic and thiourea N–H signals validates the successful covalent synthesis of the target modified chitosan thiourea derivatives.
Applying scanning electron microscopy (SEM) to the surface morphology of the modified chitosan-thiourea derivatives (CHTUD1–3) allowed us to examine the structural changes that occurred as a result of chemical alteration (Fig. 4c–h). A significant change in morphology is observed when compared to the initial chitosan film (previously illustrated in Fig. 4a and b, which exhibited a smooth, dense, and non-porous surface). In stark contrast, all micrographs of the modified derivatives at 1.00k× (Fig. 4c, e and g) and 2.00k× magnification (Fig. 4d, f and h) show a very uneven, rough, and heterogeneous surface. The smooth, film-like integrity that was there in the beginning has seen total disruption. Porosity, agglomerates, and shattered, coarse particles are now visible in the changed material (for example, see Fig. 4d and f).
Additionally, a notable new characteristic is the development of unique, separate microstructures. Fig. 4g and i illustrate that the rough polymer matrix is adorned with multiple bright, well-defined, rod-like or acicular crystalline structures. This drastic shift in shape from a smooth film to a rough, porous, and crystalline-containing matrix shows that there has been a significant change in both the chemical and structural makeup of the material. The alteration of the original compact polymer architecture is ascribed to the introduction of new thiourea functional groups, which disrupt the robust and orderly intermolecular hydrogen bonding present within the chitosan chains. The effective grafting of the thiourea derivative is directly visible in the form of the new crystalline phase.69
Previous investigations on modified chitosan derivatives based on fluorinated aromatic rings have shown that, in comparison to unmodified chitosan, the modified version exhibits surface roughness that is higher and microstructures that are more noticeable, confirming the microscopic level of structural modification.70 The textured morphology has the capacity to enhance interactions and augment the surface area in applications, like antibacterial activity and drug delivery.71
 | ||
| Fig. 6 TGA/DTG curves of (a) native chitosan, (b) the CHTUD1 sample, (c) the CHTUD2 sample, and (d) the CHTUD3 sample. | ||
Conversely, fluorinated chitosan thiourea (CHTUD1–3) demonstrates superior thermal stability compared to CH, with the second degradation step occurring in CHTUD1 within the range of 210.3–327.7 °C, CHTUD2 within 229.3–342.5 °C, and CHTUD3 within 221.40–347.4 °C. At this stage, the weight loss of fluorinated chitosan thiourea is approximately ∼45%, which is lower than that of unmodified chitosan (56%). This finding provides support for the notion that fluorinated chitosan thiourea exhibits greater thermal stability compared to pristine chitosan.74 Also, the second stage of the three samples is compatible with the degree of substitution assessment, in that the order of thermal stability is CHTUD2 > CHTUD1 > CHTUD3 > CH (Fig. 6).
Continuously, thermal degradation analysis of the third stage indicates that the chitosan thiourea derivatives exhibit significant alteration from the unmodified form of chitosan. Between 273.44 and 600 °C, unmodified chitosan undergoes its third degradation phase, resulting in a weight loss of approximately 19.97% (Fig. 6a). This step, most likely involved with the breakdown of cross-linked structures from chitosan's amino groups, leaves a 10.75% residual weight at 600 °C, suggesting little incomplete decomposition. While the chitosan thiourea derivatives (CHTUD1–3) exhibit elevated temperatures compared to those of native chitosan. The third degradation phase initiates at elevated temperatures, commencing at 327 °C (CHTUD1), 342 °C (CHTUD2), and 347 °C (CHTUD3). Dehydration, thiourea bond breaking, aromatic ring elimination, and possible depolymerization are all components of this first degradation step in the derivatives.75 In addition, the T50 value quantifies this enhanced stability, representing the temperature at which a sample loses 50% of its weight.76 So, the T50 of untreated chitosan was measured at 250 °C, while the thiourea derivatives exhibited significantly elevated values: 341 °C (CHTUD1), 322 °C (CHTUD2), and 336 °C (CHTUD3). Collectively, both the delayed onset of third-stage degradation and the dramatically increased T50 values confirm that the chitosan thiourea derivatives possess significantly greater thermal stability than the original chitosan. This observation provides strong evidence for the successful formation of the target modified polymer. As a result, this superior thermal stability is a vital feature, positioning these materials for high-performance applications, such as biomedical implants or wound dressings, that must endure prolonged exposure to heat.
3.5. Antimicrobial activity
| Cpd no. | Gram-positive bacteria | Gram-negative bacteria | Fungi | ||
|---|---|---|---|---|---|
| B. subtilis (ATCC 6633) | S. aureus (ATCC 6538) | K. pneumoniae (ATCC 13883) | S. typhi (ATCC 6539) | C. albicans (ATCC 10221) | |
| AMP = ampicillin; NYS = nystatin. | |||||
| CH | 25 ± 12 | 27 ± 20 | 17 ± 18 | 20 ± 21 | 23 ± 12 |
| CHTUD1 | 28 ± 17 | 32 ± 18 | 19 ± 23 | 22 ± 19 | 28 ± 13 |
| CHTUD2 | 35 ± 11 | 32 ± 12 | 20 ± 10 | 21 ± 28 | 28 ± 16 |
| CHTUD3 | 32 ± 17 | 33 ± 16 | 21 ± 24 | 24 ± 13 | 30 ± 12 |
| AMP | 25 ± 10 | 31 ± 25 | 20 ± 18 | 22 ± 16 | — |
| NYS | — | — | — | — | 22 ± 17 |
The synthesized fluorinated-thiourea chitosan derivatives CHTUD1–3 demonstrated significant antimicrobial activity, with zones of inhibition ranging from 28 ± 17 to 35 ± 11 mm against B. subtilis (ATCC 6633) and from 32 ± 12 to 33 ± 16 mm against S. aureus. In comparison, the unmodified chitosan exhibited zones of inhibition of 25 ± 12 and 27 ± 20 mm, while ampicillin displayed zones of inhibition of 25 ± 10 and 31 ± 25 mm against the same strains. Conversely, the thiourea chitosan derivatives CHTUD1–3 exhibited moderate activity, with zones of inhibition ranging from 19 ± 23 to 21 ± 24 mm against K. pneumoniae (ATCC 13883) and from 21 ± 28 to 24 ± 13 mm against S. typhi (ATCC 6539). This was in comparison to ampicillin, which had zones of inhibition of 20 ± 18 and 22 ± 16 mm, and unmodified chitosan, which showed zones of inhibition of 17 ± 18 and 20 ± 21 mm against the same strains. Furthermore, the modified thiourea-chitosan derivatives exhibited enhanced antifungal activity, with zones of inhibition measuring 28 ± 13 mm, 28 ± 16 mm, and 30 ± 12 mm for CHTUD1, CHTUD2, and CHTUD3, respectively. This contrasted with unmodified chitosan, which had an inhibition zone of 23 ± 12 mm, and nystatin, which had an inhibition zone of 22 ± 17 mm.
Initially, the modification of chitosan with fluorinated thiourea derivatives demonstrated considerable antimicrobial activity when compared to native chitosan, suggesting that the introduction of a thiourea linker and hydrophobic substituted-phenyl groups enhances antimicrobial efficacy. Among the synthesized derivatives, the incorporation of the 2,4-difluorophenylthiourea moiety into chitosan, as represented in compound CHTUD2, revealed promising activity, with MIC values of 7.8, 7.8, 15.62, and 15.62 µg mL−1 against B. subtilis, S. aureus, K. pneumoniae, and C. albicans, respectively, comparable to ampicillin. Conversely, compound CHTUD2 demonstrated moderate activity against S. typhi with an MIC value of 62.5 µg mL−1, which is more potent than that of native chitosan (MIC = 125 µg mL−1), but still less effective than ampicillin (MIC = 15.62 µg mL−1). This enhanced activity can be attributed to the presence of two fluorinated groups at the 2 and 4 positions of the phenyl ring, which may contribute to increased lipophilicity and membrane penetration, or may enhance binding affinity to biological targets. Additionally, the presence of fluorinated groups in the ortho and para positions contributes to the stability of the aromatic ring system by repelling electron density toward the fluorinated groups due to their high electronegativity. Furthermore, the conjugation of 4-fluorophenylthiourea with chitosan to form compound CHTUD1 exhibited significant activity against S. aureus with an MIC value of 7.8 µg mL−1, equivalent to that of ampicillin, while displaying MIC values of 31.25 µg mL−1 against B. subtilis, K. pneumoniae, and S. typhi. On the other hand, compound CHTUD3, which involved the incorporation of 3-(trifluoromethyl)phenyl thiourea into chitosan, demonstrated potent antibacterial activity against S. aureus and S. typhi, with MIC values of 7.8 and 15.62 µg mL−1, respectively. Compound CHTUD3 exhibited enhanced antibacterial activity relative to native chitosan; however, it did not show a significant improvement in antibacterial efficacy when compared to ampicillin.
The synthesized chitosan thiourea derivatives, designated as CHTUD1–3, demonstrated noteworthy antifungal activity, with an MIC value of 15.62 µg mL−1. In contrast, native chitosan displayed an MIC of 62.5 µg mL−1.
The structure–activity relationship (SAR) analysis demonstrates that the introduction of fluorinated groups can enhance antimicrobial activity, particularly when these groups are positioned at the ortho and para locations on the aromatic ring. This enhancement can be attributed to the high electronegativity of the fluorine atom, which exerts an inductive (–I) effect that withdraws electron density from the aromatic phenyl ring. Consequently, an increase in the number of fluorine atoms results in a greater electron-withdrawing effect, thereby stabilizing the aromatic system and improving the overall antimicrobial efficacy of the compounds. Furthermore, the incorporation of fluorine atoms increases lipophilicity, facilitating membrane penetration and, subsequently, enhancing bioavailability. Moreover, for compound CHTUD3, which possesses a trifluoromethyl group at the meta position, its activity primarily arises from the lipophilicity of the fluorine atom, while the presence of this group in the meta position does not enhance the stability of the phenyl ring.
| Cpd no. | Anti-microbial activity expressed as MBC/MFC values represented by µg mL−1 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Gram-positive bacteria | Gram-negative bacteria | Fungi | ||||||||
| B. subtilis (ATCC 6633) | S. aureus (ATCC 6538) | K. pneumoniae (ATCC 13883) | S. typhi (ATCC 6539) | C. albicans (ATCC 10221) | ||||||
| MBC | Ratio | MBC | Ratio | MBC | Ratio | MBC | Ratio | MFC | Ratio | |
| AMP = ampicillin; NYS = nystatin. | ||||||||||
| CH | 125 | 2 | 125 | 2 | 250 | 2 | 500 | 4 | 250 | 4 |
| CHTUD1 | 62.5 | 2 | 15.62 | 2 | 62.5 | 2 | 62.5 | 2 | 15.62 | 1 |
| CHTUD2 | 15.62 | 2 | 15.62 | 2 | 31.25 | 2 | 62.5 | 1 | 15.62 | 1 |
| CHTUD3 | 15.62 | 1 | 15.62 | 2 | 62.5 | 2 | 31.25 | 2 | 31.25 | 2 |
| AMP | 7.8 | 1 | 7.8 | 1 | 31.25 | 2 | 31.25 | 2 | — | — |
| NYS | — | — | — | — | 15.62 | 2 | ||||
Regarding the efficacy of modified chitosan against B. subtilis, CHTUD2 and CHTUD3 exhibited MBC values of 15.62 µg mL−1, indicating a slightly lower bactericidal effect than ampicillin but a higher potency than CHTUD1, which had an MBC of 62.5 µg mL−1. Furthermore, the modified chitosan derivatives CHTUD1–3 displayed equipotent bactericidal activity against S. aureus with MBC values of 15.62 µg mL−1. Conversely, the modified thiourea chitosan derivatives CHTUD1–3 showed low sensitivity and exhibited bactericidal activity against Gram-negative bacterial strains, with MBC values ranging from 31.25 to 62.5 µg mL−1 for the tested strains. Moreover, CHTUD2 demonstrated an MBC value of 31.25 µg mL−1 against K. pneumoniae, which is equivalent to that of ampicillin. Additionally, CHTUD3 exhibited an identical MBC of 31.25 µg mL−1 against S. typhi, also comparable to ampicillin.
In terms of antifungal activity, CHTUD3 demonstrated an MFC value of 31.25 µg mL−1, which was two-fold less active than CHTUD1 and CHTUD2, both exhibiting MFC values of 15.62 µg mL−1, equipotent to nystatin; however, all derivatives retained fungicidal properties. It has been previously reported that the MBC/MIC ratio was approximately 4, suggesting that the synthesized derivatives exhibited bactericidal properties rather than bacteriostatic ones.79 Based on the obtained results and the determined ratio, we concluded that all derivatives demonstrated both bactericidal and fungicidal activity.
In conclusion, the synthesized fluorinated thiourea chitosan derivatives CHTUD1–3 exhibited significant bactericidal and fungicidal properties, evidenced by reduced MBC and MFC. In certain instances, these derivatives demonstrated equipotent activity when compared to established positive control drugs.
According to Table 8, all tested modified fluorinated chitosan derivatives CHTUD1–3 exhibited remarkable antibiofilm activity against S. aureus and S. typhi. The inhibition percentage increased with the concentration of the tested compounds, indicating a dose-dependent response. Notably, the modified CHTUD1 and CHTUD3 demonstrated the highest biofilm inhibitory concentrations, with their inhibitory percentages being approximately 1–2% higher at all concentrations compared to CHTUD2. However, all modified CHTUD1–3 exhibited greater biofilm activity than native chitosan. At high concentrations (75% MBC), all modified chitosans CHTUD1–3 revealed biofilm inhibitory activities exceeding 90% (IBP = 92.84 ± 0.002–96.36 ± 0.002%) against S. aureus and (IBP = 91.70 ± 0.004–93.80 ± 0.005%) against S. typhi.
| Cpd no. | Anti-biofilm activity % | |||||
|---|---|---|---|---|---|---|
| S. aureus (ATCC 6538) | S. typhi (ATCC 6539) | |||||
| 75% MBC | 50% MBC | 25% MBC | 75% MBC | 50% MBC | 25% MBC | |
| CH | 90.76 ± 0.005 | 69.37 ± 0.003 | 24.30 ± 0.003 | 90.39 ± 0.003 | 73.50 ± 0.001 | 51.24 ± 0.005 |
| CHTUD1 | 96.36 ± 0.002 | 81.84 ± 0.007 | 72.74 ± 0.003 | 93.06 ± 0.005 | 81.85 ± 0.003 | 74.31 ± 0.002 |
| CHTUD2 | 92.84 ± 0.002 | 73.15 ± 0.006 | 61.27 ± 0.005 | 91.70 ± 0.004 | 77.55 ± 0.003 | 68.49 ± 0.004 |
| CHTUD3 | 95.22 ± 0.005 | 82.04 ± 0.007 | 72.87 ± 0.003 | 93.80 ± 0.005 | 83.16 ± 0.008 | 76.27 ± 0.003 |
At low concentrations (25% MBC), the modified fluorinated chitosan derivatives CHTUD 1–3 demonstrated inhibitory percentages ranging from (IBP = 61.27 ± 0.005–72.87 ± 0.003%) against S. aureus to (IBP = 68.49 ± 0.004–76.27 ± 0.003%) against S. typhi. In contrast, native chitosan showed biofilm inhibitory activities of 24.30 ± 0.003% against S. aureus and 51.24 ± 0.005% against S. typhi (Fig. 7). This indicates that the modification of chitosan with fluorinated thiourea enhances antibiofilm formation due to increased hydrophobicity and strong electrostatic interactions between the positively charged chitosan backbone and the negatively charged bacterial cell surfaces. Additionally, the presence of fluorinated substituents may influence surface energy and membrane permeability, leading to better adhesion to bacterial biofilms and enhanced bactericidal effects.
Fig. 8 illustrates the toxicity profile of the evaluated derivatives, demonstrating that the designed fluorinated-thiourea chitosan derivatives CHTUD1–3 exhibited non-nutritional toxicity, non-ecotoxicity, and non-BBB-barrier toxicity, with commendable probability values ranging from 54% to 73%. Furthermore, these derivatives were anticipated to demonstrate no toxicity concerning cytotoxicity, mutagenicity, and carcinogenicity. However, CHTUD2 and CHTUD3 displayed immunotoxin characteristics with probability values of 90% and 60%, respectively; this observation may be attributed to the increased number of fluorinated atoms in their molecular structure. Additionally, all modified derivatives, along with native chitosan, were predicted to possess toxicity related to nephrotoxicity, cardiotoxicity, and respiratory toxicity, exhibiting moderate percentage values. Moreover, the tested derivatives were forecasted to exhibit clinical toxicity with a probability of 66%, compared to native chitosan, which had a probability of 65%. Notably, the application of these derivatives as antimicrobial agents, with potential for biofilm formation, was predicted to be safe regarding hepatotoxicity and neurotoxicity, with favorable probability values.
 | ||
| Fig. 9 3D structure of two target enzymes: (a) S. aureus sortase A (PDB: 1T2P) and (b) E. coli LpxC (PDB: 4IS9). | ||
Regarding the docking results of the designed compounds inside S. aureus sortase A (PDB: 1T2P), we found that the first derivative, the 4-fluorophenylthiourea chitosan derivative CHTUD1, exhibited a binding affinity of −17.59 kcal mol−1. The residue Lys175 interacted with CHTUD1, forming two hydrogen bonds: one with the oxygen of the carbonyl related to the acetyl group of the N-acetyl glucosamine ring (Glu-Nac) and another with the oxygen of the hydroxyl group at C3 of N-acetyl glucosamine. The bond lengths were 2.0 Å and 1.7 Å, with strengths of 16% and 99%, respectively. Furthermore, the hydroxyl group also interacted with Gln172, exhibiting a bond length of 1.8 Å and a strength of 44%. For the glucosamine ring (Glu-N), the residues Val168 and Thr180 interacted, forming hydrogen bond donors with the hydroxyl groups at C1 and C3, with bond lengths of 1.9 Å and 3.3 Å, respectively. Additionally, to confirm the stability of the complex, a new arene–cation interaction was observed between the phenyl group of the thiourea derivative and Arg157 (Fig. 10).
The second thiourea chitosan derivative, CHTUD2, exhibited a predicted binding affinity of −18.55 kcal mol−1, forming seven hydrogen bonds. These bonds are distributed throughout the compound, with four hydrogen bonds involving the N-acetyl glucosamine ring (Glu-Nac). Two of these bonds act as sidechain donors between Glu105 and the NH of the acetyl group, as well as the hydroxyl group at C3 of the Glu-Nac, with bond lengths of 1.7 and 1.8 Å and strengths of 28 and 77%, respectively. Meanwhile, the hydroxyl group at C6 of the Glu-Nac serves as a backbone donor for a hydrogen bond with Asn114, with a bond length of 2.8 Å.89 Additionally, the oxygen in the pyranose ring acts as a hydrogen bond acceptor for Ser116, with a bond length of 2.9 Å and a strength of 98%. Furthermore, the glucosamine ring (Glu-N) forms two hydrogen bond acceptors with Arg197 and the oxygen of the pyranose ring, with bond lengths of 1.7 and 2.7 Å. The hydroxyl group at C3 of the glucosamine ring (Glu-N) also creates a hydrogen bond with Asn114, measuring 1.8 Å in length and exhibiting a strength of 40%. All of these interactions are illustrated in Fig. 11a, where the conformation of CHTUD2 also indicates a hydrophobic interaction, represented as a blue shadow region over the 2,4-difluorophenyl group of the thiourea derivative and the acetyl group.
 | ||
| Fig. 11 2D and 3D docking conformation of (a) CHTUD2 and (b) CHTUD3 inside the active site of sortase A (PDB: 1T2P). | ||
Conversely, the CHTUD3 molecule was docked into the active site of S. aureus sortase A (PDB: 1T2P), revealing a binding affinity of −16.41 kcal mol−1, with four hydrogen bond donors and three hydrogen bond acceptors. To summarize and highlight the key groups in CHTUD3 that interact with the binding pocket, a visualization analysis was conducted to explore the relationship between these interactions and the observed binding affinities. The CHTUD3 demonstrated a binding affinity of −16.41 kcal mol−1. The interaction involved two pyranose rings: the glucosamine ring (Glu-N) and the N-acetylglucosamine ring (Glu-Nac). Initially, the acidic amino acid Glu105 bound to the hydroxyl groups at C3 and C4, as well as the NH group of the N-acetyl glucosamine ring (Glu-Nac), with bond lengths of 2.2, 1.8, and 2.0 Å, and strengths of 16%, 36%, and 17%, respectively, acting as three hydrogen bond donors. Similarly, the residue Ser116 formed two hydrogen bond donors with the oxygen of the pyranose ring and the hydroxyl group at C6 of the N-acetyl glucosamine ring (Glu-Nac), with bond lengths of 3.4 and 3.2 Å, resulting in bond strengths of 93 and 76%, indicating a favorable binding mode and a stable complex. Meanwhile, the glucosamine ring (Glu-N) established two hydrogen bond acceptors with the hydroxyl group at C3 and the oxygen of the pyranose ring, with bond lengths of 2.0 and 2.5 Å, and a bond strength of 30%, respectively. Additionally, hydrophobic interactions involving the trifluoromethyl, acetyl, and all hydroxyl groups within the CHTUD3 backbone were observed (Fig. 11b).
To clarify and highlight the key functional groups in the synthesized CHTUD1–3 that interact with the binding pocket of LpxC (PDB: 4IS9), a visualization analysis was conducted to explore the relationship between these interactions and the measured binding affinities. The binding affinities of the modified thiourea chitosan derivatives CHTUD1–3 ranged from −12.72 to −14.58 kcal mol−1. The interaction between the 4-fluorophenylthiourea chitosan derivative CHTUD1 and the LpxC pocket exhibited a stable complex with a binding affinity of −12.72 kcal mol−1. This interaction involved two hydrogen bond sidechain acceptors and three hydrogen bond donors, primarily interacting through the glucosamine ring (Glu-N). Meanwhile, the N-acetylglucosamine ring (Glu-Nac) displayed hydrophobic interactions with key amino acids in the pocket's active site through the fluoro atom of the aryl-thiourea derivative, the hydroxyl group attached to the pyranose ring, and the sulfur atom of thiourea of the molecule. The hydroxyl group at C3 of Glu-N formed hydrogen bonds with residues Lys239 and His265, with bond lengths of 2.3 and 3.1 Å, respectively. The hydroxyl group at C4 established a hydrogen bond as a sidechain donor with Thr191, with a bond length of 2.3 Å and a strength of 14%. Conversely, the hydroxyl group at C6 interacted with two key amino acids, Thr191 and Phe192, with bond lengths of 3.4 and 3.2 Å and strengths of 58 and 65%, respectively (Fig. 12).
In assessing the docking binding affinity of the 2,4-difluorophenylthiourea chitosan derivative, designated as CHTUD2, a value of −13.50 kcal mol−1 was recorded. This affinity was primarily attributed to the formation of two hydrogen bonds involving sidechain acceptors between Thr191 and Lys239. These interactions were facilitated by the oxygen atom of the carbonyl group in the acetyl moiety of the Glu-Nac ring and the hydroxyl group at the C3 position of Glu-Nac, exhibiting bond lengths of 2.3 Å and 1.9 Å, with corresponding binding strengths of 37 and 65%, respectively. Furthermore, the hydroxyl group at C3 of Glu-Nac also acted as a backbone donor, establishing a hydrogen bond with the residue Met61, characterized by a bond length of 3.0 Å and a binding strength of 44% (Fig. 13a). Notably, docking studies of thiourea chitosan derivatives CHTUD3 revealed significant flexibility in the binding pocket, with a binding affinity of −14.58 kcal mol−1. These results demonstrated two hydrogen bonds formed by sidechain donors between Thr191 and Glu78, involving hydroxyl groups at C3 and C6 of Glu-N, with bond lengths of 2.7 Å and 2.1 Å. Additionally, the NH of the acetyl group formed a hydrogen bond donor with the backbone of Met61, exhibiting a bond length of 1.8 Å and a strength of 27%. Furthermore, the phenyl group of the arylthiourea moiety establishes arene–cation interactions with Lys239, while CHTUD3 displayed hydrophobic interactions with the amino acid residues in the active site (Fig. 13b).
 | ||
| Fig. 13 2D and 3D docking conformation of (a) CHTUD2 and (b) CHTUD3 inside the active site of LpxC (PDB: 4IS9). | ||
Finally, the docking results indicated that thiourea-chitosan derivatives CHTUD1–3 exhibited strong binding affinities inside the active sites of sortase A (PDB: 1T2P) and LpxC (PDB: 4IS9), suggesting that these derivatives may disrupt adhesion levels and membrane formation in S. aureus and S. typhi by forming stable and favorable interactions. These results may be aligned with biofilm-inhibitory and biocidal activities, which align with experimental data on antimicrobials and antibiofilms.
4. Conclusion
This research presents the successful preparation of three novel fluorinated chitosan derivatives based on a newly synthesized chitosan isothiocyanate derivative, which is grafted with promising fluorinated amines. Consistent with studies of ion-exchange capacity, quantitative 1H NMR examination verified a high substitution degree of 0.47 with the CHTUD1 sample. Compared to the native chitosan, the chitosan thiourea derivative samples (CHTUD1–3) exhibited significantly reduced crystallinity and markedly enhanced thermal stability. Unmodified chitosan exhibited a crystallinity index of 72.12%, which systematically decreased upon modification to 53.44%, 53.13%, and 50.88% for derivatives CHTUD1–3, respectively. In addition, the chitosan-thiourea functionalization revealed superior thermal stability, with T50 values of 341 °C (CHTUD1), 322 °C (CHTUD2), and 336 °C (CHTUD3), compared to pure chitosan (250 °C). The modified chitosan sample displayed enhanced solubility in DMF and DMSO compared with chitosan. Improving the material's antimicrobial capabilities was the most important result of this chemical modification. So, the three fluorinated chitosan thiourea derivatives were evaluated for their antimicrobial activities against two Gram-positive bacteria, two Gram-negative bacteria, and C. albicans as a fungal strain. All modified samples demonstrated greater potency than chitosan itself and showed promising activity compared to ampicillin and nystatin as a benchmark for the observed in vitro antimicrobial activity through in vitro studies. All modified samples exhibited a significant increase in antimicrobial potency compared to native chitosan, particularly compounds CHTUD2 and CHTUD3, which showed potent activity against both Gram-positive and Gram-negative strains. The synthesized chitosan thiourea derivatives CHTUD1–3 also displayed notable antifungal activity, with an MIC value of 15.62 µg mL−1, compared to chitosan (MIC = 62.5 µg mL−1). The structure–activity relationship (SAR) suggests that the incorporation of fluorinated groups can improve antimicrobial activity, mainly when these groups are situated at the ortho and para positions on the aromatic ring. Moreover, these derivatives demonstrated significantly improved bactericidal and fungicidal activities compared to native chitosan. The MBC and MFC values ranged from 15.62–62.5 µg mL−1 and 15.62–31.25 µg mL−1, respectively. The MIC values obtained indicate that the modified chitosan derivatives exhibit significant in vitro antimicrobial activity, comparable to that of the reference drugs under the tested conditions. However, any direct comparison should be interpreted with caution due to the inherent physicochemical and pharmacokinetic differences between the polymeric systems and conventional small-molecule antibiotics. Fluorinated thiourea-chitosan derivatives CHTUD1–3 exhibited superior anti-biofilm properties against S. aureus and S. typhi compared to native chitosan. Enhanced biofilm integrity may result from the increased hydrophobicity and electrostatic interactions associated with the fluorine substitution, which facilitate adhesion to bacterial surfaces. In silico toxicity prediction was assessed and found to be generally non-toxic, showing no signs of cytotoxicity, mutagenicity, or carcinogenicity, except that CHTUD2 and CHTUD3 exhibited mild immunotoxic properties.4.1. Future perspectives
Further research is necessary, as the fluorinated thiourea-chitosan sample (CHTUD3) exhibited an extremely high level of biological activity. Subsequent investigations must prioritize the optimization and scaling of the synthesis process to guarantee reproducibility and cost-effectiveness. To further assess the long-term biocompatibility, toxicity, and therapeutic effectiveness of these compounds, additional in vivo investigations are necessary. The adoption of this research int clinical applications, specifically for the creation of antimicrobial coatings for medical devices, advanced wound dressings, or drug delivery systems aimed at addressing multidrug-resistant pathogens, signifies a promising direction in the domain of functionalized carbohydrate polymers.Conflicts of interest
The authors declare that they have no conflicts of interest.Data availability
Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d6ma00145a.Acknowledgements
The authors extend their appreciation to the Deanship of Research and Graduate Studies at King Khalid University for funding this work through the Large Research Project under grant number RGP2/41/46.References
- P. Dasí-Delgado, C. Andreu and M. del Olmo, Molecules, 2025, 30, 2868 CrossRef PubMed.
- R. D. Süssmuth, M. Kulike-Koczula, P. Gao and S. Kosol, Angew. Chem., Int. Ed., 2025, 64, e202414325 CrossRef PubMed.
- R. H. Abd El-Aleam, R. F. George, H. H. Georgey and H. M. Abdel-Rahman, RSC Adv., 2021, 11, 36459–36482 RSC.
- T. Qadir, A. Amin, P. K. Sharma, I. Jeelani and H. Abe, Open Med. Chem. J., 2022, 2202280 Search PubMed.
- G. Satchanska, S. Davidova and P. D. Petrov, Polymers, 2024, 16, 1159 CrossRef CAS PubMed.
- N. S. K. Gowthaman, H. N. Lim, T. R. Sreeraj, A. Amalraj and S. Gopi, Biopolymers and their Industrial Applications, Elsevier, 2021, pp. 351–372 Search PubMed.
- E. M. Elnaggar, M. S. Abusaif, Y. M. Abdel-Baky, A. Ragab, A. M. Omer, I. Ibrahim and Y. A. Ammar, Int. J. Biol. Macromol., 2024, 277, 134347 CrossRef CAS PubMed.
- M. M. Omran, M. M. Kamal, Y. A. Ammar, M. S. Abusaif, M. M. F. Ismail and H. H. Mansour, Sci. Rep., 2024, 14, 19818 CrossRef CAS PubMed.
- R. S. Alfinaikh, K. A. Alamry and M. A. Hussein, RSC Adv., 2025, 15, 4708–4767 RSC.
- M. Gao, H. Tang and H. Zhu, Compr. Rev. Food Sci. Food Saf., 2024, 70008 CrossRef PubMed.
- R. K. Pavithran, S. G. Reddy, B. Siva Kumar and S. Kugabalasooriar, ES Food Agrofor., 2025, 1–10 Search PubMed.
- S. Sigonya, B. M. Mothudi, O. J. Fakayode, T. C. Mokhena, P. Mayer, T. H. Mokhothu, T. R. Makhanya and K. Shingange, Polymers, 2025, 17, 2082 CrossRef CAS PubMed.
- D. Zhuang, R. Li, S. Wang, H. N. Ahmad and J. Zhu, Int. J. Biol. Macromol., 2024, 255, 128043 CrossRef CAS PubMed.
- R. Wang, S. Xu, M. Zhang, W. Feng, C. Wang, X. Qiu, J. Li and W. Zhao, Carbohydr. Polym., 2024, 342, 122325 CrossRef CAS PubMed.
- V. Pawariya, S. De and J. Dutta, Int. J. Biol. Macromol., 2024, 264, 130664 CrossRef CAS PubMed.
- S. S. Alharthi, T. Gomathi, J. J. Joseph, J. Rakshavi, J. A. K. Florence, P. N. Sudha, G. Rajakumar and M. Thiruvengadam, J. King Saud Univ., Sci., 2022, 34, 102177 CrossRef.
- A. A. Ali, H. Abd El-Wahab, M. S. Abusaif, A. Ragab, O. A. Abdel-Jaid, E. A. Eldeeb and Y. A. Ammar, Pigm. Resin Technol., 2024, 53, 557–568 CrossRef CAS.
- M. R. Rodrigues, J. Carbohydr. Chem., 2005, 24, 41–54 CrossRef CAS.
- M. Shibano, S. Nishida, Y. Saito, H. Kamitakahara and T. Takano, Carbohydr. Polym., 2014, 113, 279–285 CrossRef CAS PubMed.
- A. Bermúdez-Oria, G. Rodríguez-Gutiérrez, B. Vioque, F. Rubio-Senent and J. Fernández-Bolaños, Carbohydr. Polym., 2017, 178, 368–377 CrossRef PubMed.
- D. Martínez-Cisterna, L. Chen, L. Bardehle, E. Hermosilla, G. Tortella, M. Chacón-Fuentes and O. Rubilar, Int. J. Mol. Sci., 2025, 26, 4130 CrossRef PubMed.
- A. M. Fekry and R. R. Mohamed, Electrochim. Acta, 2010, 55, 1933–1939 CrossRef CAS.
- Z. Zhong, R. Xing, S. Liu, L. Wang, S. Cai and P. Li, Carbohydr. Res., 2008, 343, 566–570 CrossRef CAS PubMed.
- M. Monier and D. A. Abdel-Latif, J. Hazard. Mater., 2012, 209–210, 240–249 CrossRef CAS PubMed.
- S. Bondock, A. A. El-Zahhar, M. M. Alghamdi and S. M. A. S. Keshk, Int. J. Biol. Macromol., 2019, 137, 107–118 CrossRef CAS PubMed.
- J. Fernández-Bolaños, B. Felizón, A. Heredia, R. Guillén and A. Jiménez, Bioresour. Technol., 1999, 68, 121–132 CrossRef.
- M. Shibano, H. Kamitakahara and T. Takano, Carbohydr. Res., 2013, 382, 25–29 CrossRef CAS PubMed.
- F. Lafzi, D. Kilic, M. Yildiz and N. Saracoglu, J. Mol. Struct., 2021, 1241, 130566 CrossRef CAS.
- A. Khan, P. Dawar and S. De, Bioorg. Chem., 2025, 158, 108319 CrossRef CAS PubMed.
- A. R. Jennings and D. Y. Son, Tetrahedron, 2015, 71, 3990–3999 CrossRef CAS.
- M.-O. M. Piepenbrock, G. O. Lloyd, N. Clarke and J. W. Steed, Chem. Commun., 2008, 2644 RSC.
- T. Venkatachalam, C. Mao and F. Uckun, Bioorg. Med. Chem., 2004, 12, 4275–4284 CrossRef CAS PubMed.
- J. Lee, S.-U. Kang, H.-K. Choi, J. Lee, J.-O. Lim, M.-J. Kil, M.-K. Jin, K.-P. Kim, J.-H. Sung, S.-J. Chung, H.-J. Ha, Y.-H. Kim, L. V. Pearce, R. Tran, D. J. Lundberg, Y. Wang, A. Toth and P. M. Blumberg, Bioorg. Med. Chem. Lett., 2004, 14, 2291–2297 CrossRef CAS PubMed.
- S. A. Khan, N. Singh and K. Saleem, Eur. J. Med. Chem., 2008, 43, 2272–2277 CrossRef CAS PubMed.
- A. P. Keche, G. D. Hatnapure, R. H. Tale, A. H. Rodge, S. S. Birajdar and V. M. Kamble, Bioorg. Med. Chem. Lett., 2012, 22, 3445–3448 CrossRef CAS PubMed.
- S. Saeed, N. Rashid, P. G. Jones, M. Ali and R. Hussain, Eur. J. Med. Chem., 2010, 45, 1323–1331 CrossRef CAS PubMed.
- R. Pingaew, N. Sinthupoom, P. Mandi, V. Prachayasittikul, R. Cherdtrakulkiat, S. Prachayasittikul, S. Ruchirawat and V. Prachayasittikul, Med. Chem. Res., 2017, 26, 3136–3148 CrossRef CAS.
- K. M. Khan, S. Saeed, M. Ali, M. Gohar, J. Zahid, A. Khan, S. Perveen and M. I. Choudhary, Bioorg. Med. Chem., 2009, 17, 2447–2451 CrossRef CAS PubMed.
- A. Ragab, M. A. Salem, Y. A. Ammar, W. M. Aboulthana, M. H. Helal and M. S. Abusaif, Drug Dev. Res., 2024, 85, e22216 CrossRef CAS PubMed.
- B. E. Smart, J. Fluorine Chem., 2001, 109, 3–11 CrossRef CAS.
- H. G. Bonacorso, A. P. Wentz, R. V. Lourega, C. A. Cechinel, T. S. Moraes, H. S. Coelho, N. Zanatta, M. A. P. Martins, M. Höerner and S. H. Alves, J. Fluorine Chem., 2006, 127, 1066–1072 CrossRef CAS.
- M. Jagodzinska, F. Huguenot, G. Candiani and M. Zanda, ChemMedChem, 2009, 4, 49–51 CrossRef CAS PubMed.
- M. A. Salem, M. S. Abusaif, N. A. Gohar, Y. A. Ammar and A. Ragab, Drug Dev. Res., 2025, 86, e70085 CrossRef CAS PubMed.
- S. Shrimandilkar, P. Tryambake, S. Borgave and D. Lokhande, J. Mol. Struct., 2025, 1345, 141670 CrossRef.
- E. A. Ali, H. M. Abo-Salem, A. A. Arafa and A. A. Nada, Cellulose, 2023, 30, 3505–3522 CrossRef CAS PubMed.
- L. Yang, Y. Du, H. Wang and Y. Yang, J. Cleaner Prod., 2025, 522, 146351 CrossRef CAS.
- I. R. Fatika and F. Aligarh, in International Conference on Islamic Economics (ICIE), 2024, vol. 1, pp. 324–332.
- Z. Fu, W. Yuan, N. Chen, Z. Yang and J. Xu, Green Chem., 2018, 20, 4484–4491 RSC.
- O. A. A. Ali, A. Ragab, Y. A. Ammar and M. S. Abusaif, J. Mol. Struct., 2025, 1334, 141879 CrossRef CAS.
- A. Zieba, D. Pindjakova, M. Latocha, J. Plonka-Czerw, D. Kusmierz, A. Cizek and J. Jampilek, Molecules, 2024, 29, 4044 CrossRef CAS PubMed.
- A. Ragab, M. S. Abusaif, H. M. R. M. Selim, O. K. M. Riad, M. H. Helal, A. M. Ali, Y. A. Ammar and G. E. Ahmed, Bioorg. Chem., 2025, 165, 108977 CrossRef CAS PubMed.
- H. Khamees Thabet, A. Ragab, M. Imran, M. H. Helal, S. Ibrahim Alaqel, A. Alshehri, A. Ash Mohd, M. Rakan Alshammari, M. S. Abusaif and Y. A. Ammar, Eur. J. Med. Chem., 2024, 275, 116589 CrossRef CAS PubMed.
- H. F. Rizk, M. A. El-Borai, A. Ragab, S. A. Ibrahim and M. E. Sadek, Polycyclic Aromat. Compd., 2023, 43, 500–522 CrossRef CAS.
- M. A. Ismail, M. S. Abusaif, M. S. A. El-Gaby, Y. A. Ammar and A. Ragab, RSC Adv., 2023, 13, 12589–12608 RSC.
- Y. M. Abdel-Baky, A. M. Omer, E. M. El-Fakharany, Y. A. Ammar, M. S. Abusaif and A. Ragab, Sci. Rep., 2023, 13, 22792 CrossRef CAS PubMed.
- D. D. Babu, A. S. Pani, S. D. Joshi, P. Naik, G. K. Jayaprakash, M. Al-Ghorbani, B. Rodrigues and B. K. Momidi, Microb. Pathog., 2025, 200, 107209 CrossRef CAS PubMed.
- H. Zaman, A. Saeed, H. Ismail and M. Rashid, Arch. Biochem. Biophys., 2025, 765, 110304 CrossRef CAS PubMed.
- Z. Fu, W. Yuan, N. Chen, Z. Yang and J. Xu, Green Chem., 2018, 20, 4484–4491 RSC.
- K. Srivastava, A. Bhatt, N. Singh, R. Khare, R. Shukla, D. Chaturvedi and R. Kant, Chem. Biol., 2020, 10, 34–50 CAS.
- M. M. Ghorab, M. S. A. El-Gaby, M. S. Alsaid, Y. A. M. M. Elshaier, A. M. Soliman, F. F. El-Senduny, F. A. Badria and A. Y. A. Sherif, Adv. Anticancer Agents Med. Chem., 2017, 17, 1411–1425 CAS.
- R. Antony, S. Theodore David, K. Saravanan, K. Karuppasamy and S. Balakumar, Spectrochim. Acta, Part A, 2013, 103, 423–430 CrossRef CAS PubMed.
- G. I. Edo, W. Ndudi, A. B. M. Ali, E. Yousif, K. Zainulabdeen, P. O. Akpoghelie, E. F. Isoje, U. A. Igbuku, R. A. Opiti, A. E. Athan Essaghah, D. S. Ahmed, H. Umar and A. A. Alamiery, Carbohydr. Res., 2025, 550, 109409 CrossRef CAS PubMed.
- Y. M. Abdel-Baky, A. Ragab, M. S. Abusaif, Y. A. Ammar and A. M. Omer, Carbohydr. Polym., 2025, 124049 CrossRef CAS PubMed.
- Y. Peng, Y. Yu, Z. Su, Y. Zhong, S. Vijayakumar, Y. Chen, Y. Mao, M. Xin and M. Li, Carbohydr. Polym., 2025, 367, 124015 CrossRef CAS PubMed.
- M. M. Ahmed, N. T. I. Salama, S. Bondock, A. A. Soayed and R. M. I. Elsamra, Appl. Organomet. Chem., 2025, 70125 CrossRef.
- S. M. Tawfik, H. Jang, H. Kwon, M. Fekry, A. A. Roshdy, A. Ragab, B. Zakaria, M. Fouad, Y. A. Ammar and M. S. Abusaif, Int. J. Biol. Macromol., 2023, 150699 Search PubMed.
- A. H. dos Reis-Prado, M. Rahimnejad, R. Dal-Fabbro, P. T. A. de Toledo, C. Anselmi, P. H. C. de Oliveira, J. C. Fenno, L. T. A. Cintra, F. Benetti and M. C. Bottino, Bioact. Mater., 2025, 46, 406–422 Search PubMed.
- R. K. Cheedarala, A. M. Kurumbala and R. R. Chidambaram, Chem. Eng. J., 2025, 522, 168263 CrossRef CAS.
- T. Alizadeh and Z. Asadi, Carbohydr. Polym. Technol. Appl., 2025, 11, 100997 CAS.
- R. Malav, R. K. Sharma and S. Ray, Int. J. Biol. Macromol., 2025, 301, 140338 CrossRef CAS PubMed.
- Y. M. Abdel-Baky, A. Ragab, M. S. Abusaif, Y. A. Ammar and A. M. Omer, Carbohydr. Polym., 2025, 368, 124049 CrossRef CAS PubMed.
- N. D. Alharbi, H. H. Amer, N. A. El-Zaher and O. W. Guirguis, Polym. Polym. Compos., 2022, 26 DOI:10.1177/09673911221148826.
- F. J. Caro-León, E. Silva-Campa, R. A. Navarro-López, D. Fernández-Quiroz, V. A. Figueroa-León, M. A. Trujillo-Ramirez, L. M. López-Martínez, M. R. Aguilar and O. Álvarez-Bajo, ACS Omega, 2025, 10, 3474–3485 CrossRef PubMed.
- A. Ragab, D. M. Elsisi, E. M. Elqady, E. EL-Said, M. A. Salem, Y. A. Ammar and M. S. Abusaif, Pestic. Biochem. Physiol., 2024, 202, 105943 CrossRef CAS PubMed.
- H. R. Mardani, F. Ravari, A. Kalaki and L. Hokmabadi, J. Polym. Environ., 2020, 28, 2523–2538 CrossRef CAS.
- P. Khosravian, R. Mardani, Z. K. Boldaji, M. Javdani and M. Hashemnia, Naunyn-Schmiedeberg's Arch. Pharmacol., 2025, 1–19 Search PubMed.
- A. Ragab, R. Ayman, M. A. Salem, Y. A. Ammar and M. S. Abusaif, Eur. J. Med. Chem., 2025, 290, 117499 CrossRef CAS PubMed.
- M. S. A. El-Gaby, Y. A. Ammar, M. A. Ismail, A. Ragab and M. S. Abusaif, Heterocycl. Commun., 2023 DOI:10.1515/hc-2022-0170.
- X. Han, W. Lv, S.-Y. Guo, M. Cushman and J.-H. Liang, Bioorg. Med. Chem., 2015, 23, 6437–6453 CrossRef CAS PubMed.
- M. S. A. El-Gaby, Y. A. Ammar, M. A. Ismail, A. Ragab and M. S. Abusaif, Heterocycl. Commun., 2023, 25 DOI:10.1515/hc-2022-0170.
- Y. A. Ammar, J. A. Micky, D. S. Aboul-Magd, S. M. A. Abd El-Hafez, S. A. Hessein, A. M. Ali and A. Ragab, Chem. Biol. Drug Des., 2023, 101, 245–270 CrossRef CAS PubMed.
- R. R. Raslan, Y. A. Ammar, S. A. Fouad, S. A. Hessein, N. A. M. Shmiess and A. Ragab, RSC Adv., 2023, 13, 10440–10458 RSC.
- M. M. F. Ismail, T. Z. Shawer, R. S. Ibrahim, M. S. Abusaif, M. M. Kamal, R. M. Allam and Y. A. Ammar, RSC Adv., 2023, 13, 31908–31924 RSC.
- H. K. Thabet, A. Ragab, M. Imran, M. Hamdy Helal, S. Ibrahim Alaqel, A. Alshehri, A. Ash Mohd, S. S. Alshammari, Y. A. Ammar and M. S. Abusaif, RSC Adv., 2024, 14, 15691–15705 RSC.
- H. Khamees Thabet, Y. A. Ammar, M. Imran, M. Hamdy Helal, S. Ibrahim Alaqel, A. Alshehri, A. Ash Mohd, M. S. Abusaif and A. Ragab, Bioorg. Chem., 2024, 151, 107671 CrossRef CAS PubMed.
- A. Ragab, M. S. Abusaif, N. A. Gohar, D. S. Aboul-Magd, E. A. Fayed and Y. A. Ammar, Bioorg. Chem., 2023, 131, 106307 CrossRef CAS PubMed.
- G. A. Abd-elmaksoud, M. S. Abusaif, Y. A. Ammar, S. Al-Sharbasy and M. A. Migahed, Arabian J. Sci. Eng., 2023, 48, 16167–16185 CrossRef CAS.
- A.-M. Möller, M. Vázquez-Hernández, B. Kutscher, R. Brysch, S. Brückner, E. C. Marino, J. Kleetz, C. H. R. Senges, S. Schäkermann, J. E. Bandow and F. Narberhaus, J. Biol. Chem., 2024, 300, 107143 CrossRef PubMed.
- M. S. Abusaif, A. Ragab, E. A. Fayed, Y. A. Ammar, A. M. H. Gowifel, S. O. Hassanin, G. E. Ahmed and N. A. Gohar, Bioorg. Chem., 2025, 154, 108023 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |