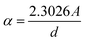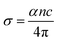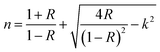Open Access Article
Open Access ArticleStudy of the structural, optical, and electronic properties of gadolinium orthophosphate: a combination of experimental and theoretical studies
I. Abdelrhafor *a,
L. H. Omari
*a,
L. H. Omari *ab,
M. Lassric,
S. Tarika,
A. El Hachmid,
H. Lassria,
E. K. Hlile and
E. Dhahri
*ab,
M. Lassric,
S. Tarika,
A. El Hachmid,
H. Lassria,
E. K. Hlile and
E. Dhahri f
f
aLPMAT, Faculté des sciences Ain Chock, Université Hassan II de Casablanca, Morocco. E-mail: imaneabdelrhafor@gmail.com; bophysiq@gmail.com
bLEST Laboratory, ESEF Berrechid, Hassan First University of Settat, Morocco
cEMINES-SAEP, UM6P, Mohammed VI Polytechnic University, Lot 660, Hay Moulay Rachid, 43150, Ben Guerir, Morocco
dPhysico-chemistry of Materials Team, Faculty of Science, Abdelmalek Essaadi University, Tetouan, Morocco
eUniv. Grenoble Alpes, CNRS, Grenoble INP, Néel Institute, 38000, Grenoble, France
fFaculté des Sciences de Sfax, Université de Sfax, 3000, BP 1171, Tunisia
First published on 28th April 2026
Abstract
We present an in-depth study combining experimental techniques with ab initio density functional theory (DFT) calculations to elucidate the electronic structure and optical properties of gadolinium orthophosphate (GdPO4). Experimentally, X-ray diffraction (XRD) and Raman spectroscopy were used to characterize the synthesized pure phase of GdPO4. Rietveld refinement analysis confirmed that GdPO4 crystallizes in the monoclinic space group P21/n with a monazite structure. UV-visible diffuse reflectance spectroscopy was performed on synthesized GdPO4 powders. The absorption coefficient was analyzed via the Kubelka–Munk transformed Tauc plot to explore the optical bandgap value of GdPO4. In addition, the absorption coefficient derived from UV-visible measurement demonstrated two peaks at 275 and 303 nm, corresponding to the 4f–4f transitions 8S7/2 → 6IJ and 8S7/2 → 6P7/2 of Gd3+, respectively. Theoretically, DFT+mBJ+SOC was employed to simulate the density of states, band structures, and complex dielectric functions. We also successfully identified both 4fn–4fn and 4fn−15d1 transitions by means of DFT. Our main findings include an accurate prediction of the energy of the possible emitted photon (3.96 eV), which aligns with the experimental measurement of luminescence. Additionally, the predicted electronic bandgap of 7.8 eV for the monazite crystal GdPO4 is consistent with previous experimental data. This large gap is typical for rare-earth orthophosphate systems. The results of our computational study are also validated by magnetic moment estimation. The Gd atom shows a saturation magnetic moment of 6.86µB, which is congruent with the experimentally measured magnetic moment µexp = 7.2 ± 0.3µB.
1. Introduction
Lanthanides, also known as rare-earth elements (REEs), belong to the f-block of the periodic table. They are typically classified into two structural groups based on their phosphate compounds. The orthophosphates of the lighter and larger lanthanides (LnPO4, where Ln = La to Gd) crystallize in a monoclinic monazite-type structure, associated with the space group P21/n. In contrast, the orthophosphates of the heavier and smaller lanthanides (Ln = Tb to Lu) have a zircon-like tetragonal structure, commonly called xenotime, with the space group I41/amd.1 Lanthanide orthophosphates (LnPO4) are attracting increasing interest due to their unique optical and structural properties, making them promising candidates for various applications, including luminescence-based temperature sensing, optical materials, and energy-related devices,2,3 and semiconductor lighting devices.4,5 Monazite-type rare earths (REEs) have been extensively researched for use in various advanced applications, including their use as geochronological microprobes,6 scintillators for X-ray and gamma-ray detection in medical imaging systems,7 and host materials in semiconductor lasers.8 Thanks to their exceptional structural stability and resistance to radiation-induced damage, they are also considered promising candidates for immobilizing minor actinides and plutonium in nuclear waste management strategies.9,10 Recently, gadolinium-based orthophosphates have attracted considerable interest due to the advantageous electronic properties of 4f elements, including their high magnetic moments and rapid electronic relaxation times, on the order of nanoseconds. These properties make them suitable for a wide range of applications, including the labelling of biological and medical samples for in vitro imaging.In particular, GdPO4 has demonstrated superior performance compared to certain commercially available contrast agents.11 Recently, GdPO4 has been identified as a promising low-temperature magnetic refrigeration material for magnetocaloric applications due to its significant magnetocaloric effect.12 It also exhibits second-order magnetic phase transition behavior.12–14 Coating cathode materials with Gd orthophosphates has proven to be a highly effective method for advanced lithium-ion battery applications. In the case of LiFePO4, the incorporation of a GdPO4 layer can significantly improve ionic and electronic conductivity while suppressing undesirable side reactions at the interface between the electrode and the electrolyte.15 In addition, Ni-rich NCA cathodes coated with GdPO4 exhibit enhanced current density performance and superior capacity retention stability.16
In this work, we report on a comparative study of GdPO4, including density functional theory (DFT) calculations and experimental optical measurements. The first part of the study presents the synthesis and structural characterization of GdPO4 using XRD and Raman spectroscopy, followed by optical characterization to identify electronic transitions. The second part will involve the study of the electronic and optical properties using DFT. To ensure reliable and accurate results, we have adopted the GGA+mBJ functional to describe the exchange–correlation potential, in addition to the spin–orbit-coupling (SOC) scheme. The electronic gap energy was predicted alongside an analysis of intra- and inter-configurational transitions. A discussion of points of agreement, similarities, and divergences then followed.
2. Experimental
2.1. Procedure of synthesis
Polycrystalline GdPO4 was prepared using a solid-state reaction method. The raw materials used were high-purity gadolinium oxide (Gd2O3, 99.99%) and ammonium dihydrogen phosphate (NH4)2HPO4, 99.9%, in stoichiometric proportions. According to the standard protocol, the gadolinium oxide precursor Gd2O3 was calcined at 1073 K for 1 hour. After that, a stoichiometric amount of (NH4)2HPO4 was then added, thoroughly ground and heated at 1623 K for 12 hours in an alumina crucible. The thermal treatment was repeated once.2.2. Characterization
Powder X-ray diffraction (PXRD) was utilized to verify the successful synthesis of GdPO4. Measurements were performed using a Bruker D8 diffractometer employing Cu-Kα radiation (λ = 0.15406 nm). The scanning range for 2θ was set from 15° to 80°, with data collected at 0.2° intervals and at a scan speed of 1 second per step. For Rietveld refinement, the established structure of GdPO4, characterized by the P21/m space group, served as the foundational model. The vibrational properties of the synthesized GdPO4 were investigated using Raman spectroscopy. Spectra were recorded in the 300–1350 cm−1 range. The UV-vis spectra were recorded via a spectrometer/Data System JASCO Corp., V-570, Rev.1.00 in the 250–850 nm wavelength range. All the spectra were recorded at room temperature (∼298 K). The measured sample was 2 millimeters thick. The spectrometer has a wavelength accuracy of ±0.3 nm. The uncertainty over absorbance is 0.13%.3. Computational methods
In order to investigate the electronic properties, a computational study was conducted using density functional theory (DFT) with the full-potential linearized augmented plane-wave method, as implemented in the Wien2k simulation package.17,18The computational approach involves resolving the Kohn–Sham equations by expanding the wave functions in terms of spherical harmonics within the atomic spheres, while plane-wave expansions are applied in the interstitial regions of the unit cell. For all calculations, the exchange–correlation potential (Exc) was treated using the generalized gradient approximation (GGA) in the Perdew–Burke–Ernzerhof (PBE) formulation.
To obtain an accurate electronic structure, the DFT+mBJ+SOC method was adopted to overcome the well-known shortcoming of GGA.19 The mBJ method was used to describe the strongly correlated electrons of the Gd3+ ion occupying the 4f orbital. Additionally, the 4f electrons of Gd were treated as valence states. The Spin–Orbit Coupling (SOC) had also been added for gadolinium. The muffin-tin radii (RMT) for the three atoms Gd, P, and O were fixed at 2.2, 1.35, and 1.48 Bohr, respectively.20
The k-point sampling of the Brillouin zone was performed using 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ×
× ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9
9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ×
× ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 k-mesh grids. The spin-polarized ferromagnetic configuration was included in the calculations self-consistently. The convergence criterion for the self-consistent field of the Kohn–Sham equations was established at 10−4 eV.
10 k-mesh grids. The spin-polarized ferromagnetic configuration was included in the calculations self-consistently. The convergence criterion for the self-consistent field of the Kohn–Sham equations was established at 10−4 eV.
4. Results and discussion
4.1. Structural properties
The crystal structure of the studied compound was investigated using the Rietveld refinement24 with the FullProf program,25 revealing that it crystallizes in the monoclinic system, with the space group P21/n (No. 14) and unit-cell parameters a = 6.6532(3) Å, b = 6.8491(3) Å, c = 6.3366(3) Å, and β = 104.001(2)°. The unit-cell volume was determined to be 280.17(2) Å3, with a calculated density of 5.981 g cm−3. The number of formula units per unit cell (Z) is 4. The XRD patterns were correctly indexed by assigning the monoclinic space group P21/n (No. 14), for which the general reflection conditions are: h0l: h + l = 2n; 0k0: k = 2n; h00: h = 2n; and 00l: l = 2n, as specified in the International Tables for Crystallography, Volume A: Space-Group Symmetry.26
Fig. 1 presents the final Rietveld refinement plot for the GdPO4 compound. Experimental X-ray diffraction data are represented by red dots, while the calculated diffractogram is represented by a solid black line. The positions of Bragg reflections are indicated by green vertical lines. The blue curve at the bottom represents the difference between the observed and calculated intensities. Table 1 presents the crystallographic data, data acquisition details, and refinement parameters, including lattice parameters, unit cell volume, crystal system, space group, and various statistical agreement indices. The reliability factors (%) – Rp = 3.89, Rwp = 4.92, Rexp = 4.07 – and the goodness-of-fit value χ2 = 1.47 indicate a high-quality fit, as these values are low. The excellent agreement between the experimental points (dots) and the calculated profile (solid line) in Fig. 1 confirms the accuracy of the refinement. The refined structure meets the criteria for chemical plausibility and statistical robustness. The fractional atomic coordinates and isotropic displacement parameters are presented in Table 2. The Gd3+, P5+, and O2− ions occupy 4-electron sites at positions (x, y, z). A P5+ cation is coordinated by four oxygen ions (4-coordination), while a Gd3+ ion is coordinated by ten oxygen ions (10-coordination). Table 3 presents the selected diatomic distances (in Å) and bond valences. The average bond lengths 〈Gd–O〉 and 〈P–O〉 determined by Rietveld refinement are congruent with the predicted bond lengths of 2.5105 Å for Gd3+–O2− and 1.5344 Å for P3+–O2−. The average crystallite size of the compound GdPO4 was estimated by X-ray diffraction line broadening analysis, using the Scherrer equation. It is important to note that X-ray diffraction peak broadening can be due to multiple factors, including crystallite size, macrostrains, instrumental contributions, temperature, and compositional inhomogeneities.
| Formula | GdPO4 |
|---|---|
| Mr | 252.2214 |
| Temperature (K) | 300 |
| dcalc (g cm−3) | 5.981 |
| Radiation | Cu-Kα |
| Wavelengths (Å) | 1.54056 (λ1) |
| 1.54439 (λ2) | |
| 2θ range (°) | 10–80 |
| 2θ step (°) | 0.020283 |
| Program | FullProf |
| Pseudo–Voigt function: [PV = ηL + (1 − η)G] | η = 0.50(6) |
| Caglioti parameters (u, v, w) | 0.022(5), −0.018(4), 0.0095(8) |
| No. of reflections | 374/2 |
| No. of fitted parameters | 35 |
| Crystal system | Monoclinic |
| Space group | P21/n (No. 14) |
| Lattice parameters | a = 6.6532(3) Å, b = 6.8491(3) Å, c = 6.3366(3) Å, β = 104.001(2) ° |
| Cell-volume (Å3) | 280.17(2) |
| Formula units per unit cell (Z) | 4 |
| Reliability factors | Rp = 3.89, Rwp = 4.92, Rexp = 4.07 |
| Goodness-of-fit (χ2) | 1.47 |
| x | y | Z | Biso (Å2) | Occ. | Site | |
|---|---|---|---|---|---|---|
| Gd3+ | 0.2798(5) | 0.1553(6) | 0.0959(6) | 0.8(1) | 1 | 4e |
| P5+ | 0.296(2) | 0.163(2) | 0.607(3) | 0.6(3) | 1 | 4e |
| O1 | 0.252(4) | −0.005(4) | 0.418(6) | 2.2(4) | 1 | 4e |
| O2 | 0.369(3) | 0.325(4) | 0.508(5) | 2.2(4) | 1 | 4e |
| O3 | 0.467(4) | 0.119(4) | 0.816(4) | 2.2(4) | 1 | 4e |
| O4 | 0.114(4) | 0.223(4) | 0.714(4) | 2.2(4) | 1 | 4e |
| Distances (Å) | Bond-valences | ||
|---|---|---|---|
| Gd–O1 | (×1) | 2.36(4) | 0.45(4) |
| (×1) | 2.34(3) | 0.48(4) | |
| Gd–O2 | (×1) | 2.79(3) | 0.14(1) |
| (×1) | 2.49(3) | 0.31(2) | |
| (×1) | 2.66(2) | 0.20(1) | |
| Gd–O3 | (×1) | 2.41(3) | 0.39(3) |
| (×1) | 2.50(3) | 0.31(2) | |
| (×1) | 3.17(3) | 0.050(4) | |
| Gd–O4 | (×1) | 2.45(2) | 0.35(2) |
| (×1) | 2.32(3) | 0.50(4) | |
| 〈Gd–O〉average, BVS | 2.549(9) | 3.19(9) | |
| P–O1 | (×1) | 1.64(4) | 0.95(9) |
| P–O2 | (×1) | 1.42(3) | 1.72(15) |
| P–O3 | (×1) | 1.55(3) | 1.19(9) |
| P–O4 | (×1) | 1.58(3) | 1.11(10) |
| 〈P–O〉average, BVS | 1.55(2) | 4.97(22) |
The Scherrer equation is defined as follows:
 | (1) |
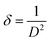 | (2) |
| Vibrational mode | GdPO4 [This work] | EuPO427,29 | TbPO431 | Assignment |
|---|---|---|---|---|
| External lattice vibrations | 398, 419 | 189–425 | 199, 383, 254 | Lattice modes |
| Symmetric O–P–O bending | 478 | 465–477 | 474 | ν2 |
| Asymmetric O–P–O bending | 628 | 618–634 | 592, 636 | ν4 |
| Symmetric P–O stretching | 985 | 965–990 | 993 | ν1 |
| Asymmetric P–O stretching | 1072, 1093 | 1055–1075 | 1043, 1106 | ν3 |
4.2. Experimental optical properties
The optical characteristics of the synthesized GdPO4 were investigated using UV-visible spectroscopy. The corresponding results are analyzed and discussed. Based on Lambert–Beer's law, the measured absorbance A is defined as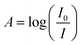 , and it is directly related to the absorption coefficient α through the following expression:
, and it is directly related to the absorption coefficient α through the following expression:where A and d are respectively the absorption and the width of the sample.
The absorption spectrum for GdPO4 is reported in Fig. 3(a). A strong absorption peak is observed at 275 nm. This corresponds to the 8S7/2 → 6IJ transition. Other peaks are observed at 307 and 313 nm, which refer to the 8S7/2 → 6P7/2 transition. Both absorption peaks were identified in previous works and reported in.32
The optical gap energy EOptg is revealed using Tauc's relation:
| αhν = C(hν − EOptg)t |
The optical conductivity, extinction coefficient, and refractive index were calculated using the following formulas:
In this study, the GdPO4 powders exhibit a direct band gap; thus, t = 1/2 was used. Fig. 3(b) represents the plot of (αhν)2 versus hν, which shows a linear behavior over a significant energy range, confirming the direct transition nature. The optical gap energy was estimated by extrapolating the linear region of the plot to intercept the photon energy axis, corresponding to (αhν)2 = 0. Which corresponds to EOptg = 4.44 eV. The obtained optical band gap does not correspond to band-to-band transitions. It deals with transitions that take place in the measuring range (250–800 nm) of the UV-vis spectrophotometer, hence only 4f–4f intraconfigurational transitions are captured. The reported value of 4.44 eV does not represent the fundamental electronic band gap. The interconfigurational 4f–5d were captured using a diffuse reflectance spectrophotometer covering a wavelength of (120–300 nm). The electronic band gap of the monazite GdPO4 was reported in a previous study to be 7.8 eV.33 Due to experimental limitations we could attain energy values greater than 5 eV, which does not allow direct observation of the fundamental band-to-band expected near 8 eV.
The dielectric and optical responses (Fig. 3(c–f)) consistently indicate that the material is a wide-band-gap insulator with localized absorption in the near-UV. The imaginary part ε2 and the optical conductivity show two peaks at 4.0 eV and 4.5 eV, which produce the corresponding dispersive structure in ε1 and a strong augmentation of the refractive index n at the same energies. The weak dispersion at energy value 4.0 eV feature is consistent with magnetic-dipole induced intra-4f transitions of Gd3+, while the relatively strong peak at 4.5 eV indicates a transition induced by the same dipole but with higher probability. The optical conductivity σ(ω) is negligible below 4 eV, confirming the insulating behavior. The crests observed at 4 and 4.5 eV are a consequence of the aforementioned transitions 8S7/2 → 6IJ and 8S7/2 → 8P7/2, considering their intensities (<800 Ω−1 cm−1) and their localized features. We conclude that at this energy range, electrons are not free to conduct.
4.3. Electronic properties
Fig. 4(a) and (b) show that the total DOS is dominated mainly by the f orbital of the Gd atom contribution taking place in both occupied states at negative energy values of spin-up of Gd and unoccupied states contained at negative energy values with major contribution from the spin-down configuration of Gd. In addition, the contribution from spin-up at negative energies resulting from spin–orbit coupling of Gd that takes place in the energy range 4–4.5 eV.
Furthermore, we observe the contribution of the d orbital at energy values around 7.95 eV. It defines the conduction band of the GdPO4. The PDOS of Gd gives evidence of the absence of the contribution from the s orbital to the total Gd PDOS, confirming that this element has lost three electrons. It is in accordance with the rare earth oxidation number 3+. Contribution from phosphorus and oxygen takes place near the Fermi level at negative energies. Partial densities of states of P and O are symmetrical with respect to the x-axis, pointing out the non-magnetic behavior of both atoms.
As expected, the GdPO4 is found to be an insulator with a band gap of 8 eV corresponding to the spin-up configuration. The electronic transition from the 4f valence band to the empty 4f states of Gadolinium is forbidden due to the electric-dipole selection rules (ΔL = ±1). Nonetheless, it is allowed by the magnetic dipole selection rules (ΔL = 0), particularly for systems having heavy elements including significant spin–orbit coupling. Basically, both initial and final bands of this transition are showing the same parity, ΔS = 0 and ΔL = 0. The 4f–4f transition corresponds to an optical band gap in the range of 4.0–4.4 eV. Electrons that absorb this energy are excited from the valence band (VB) to the 4f excited state; however, they do not contribute to electrical conductivity. Instead, these excited electrons relax back to the VB. Two possible relaxation pathways are radiative relaxation or non-radiative relaxation. We consider the relaxation to be radiative for the following reasons: first, this transition is magnetic-dipole allowed. Secondly, the empty electronic states of 4f (4–4.5 eV) are considered metastable, as can be seen in Fig. 5, 4f empty states occupy flat bands. Thirdly, the empty electronic states of 4f are well separated from the conduction band; a gap of 3.5 eV separates them. We put forward a radiative relaxation pathway of 4 eV. Experimentally, a photon emission at 313 nm (3.96 eV) is observed in the photoluminescence spectrum of GdPO4 under excitation at 254 nm, which is consistent with our analysis.32
The computed gap energy matches well the previously published experimental value of 7.8 eV.33,34 The obtained electronic energy gap using the GGA+mBJ shows better values than the theoretical band gaps obtained by the GGA+U method (5.7 eV) and the hybrid functional HSE06 (6.55 eV).35
Furthermore, the Gd atom shows a highly asymmetric DOS, which points to a magnetic behavior with 6.94µB as saturation magnetic moment carried by this atom. This finding is consistent with both the experimentally measured magnetic moment (µexp = 7.2 ± 0.3µB) and the theoretical free-ion (Gd3+) value (µtheo = 7µB).12
4.4. Numerical optical properties
In order to study the optical properties of monazite-structured GdPO4, OPTIC, JOINT, and KRAM programs, as implemented in the Wien2k simulation package, were used. Firstly, the OPTIC program was used to calculate the momentum matrix elements. Then, the JOINT program was used to compute the imaginary part of the dielectric function ε2. Finally, Kramers–Kronig analysis was carried out using the KRAM program, which is used to deduce the real part of the dielectric tensor ε1 and compute the optical conductivity and the absorption coefficient.38 All the calculations were performed corresponding to the photon energy range of 0 to 12 eV. The formulas of the real and imaginary parts of the dielectric function, respectively, are:38Additionally, with the Knowledge of the complex dielectric tensor ε = ε1 + iε2, the refractive index, extinction coefficient, and optical conductivity were calculated using the following formulas:
Fig. 6(a) and (b) illustrate the absorption coefficient α(ω) and optical conductivity σ(ω). The absorption coefficient α(ω) reflects the material's capacity to absorb incident photons having specific energies. The absorption spectra were also analyzed along the three principal crystallographic directions. As depicted in Fig. 6(a), no significant absorption phenomena are observed below 4 eV, aligning well with the computed band structure. Beyond this energy threshold, α(ω) shows a minor broad peak from 4 to 5.2 eV. This slight contribution is due to the intra-configurational 4f–4f transition. This electronic transition is reasonable and known to be very small in intensity (1–60 cm−1) in comparison to the 4f–5d transitions, owing to the nature of magnetic–dipole transitions.39 The reversed electronic transition is the origin of luminescence properties in GdPO4.2 The intra-configurational transition is observed in previous band structure calculations on lanthanide-based orthophosphate and experimental optical studies.2,40 Beyond the energy value of 8 eV, α(ω) increases drastically, showing an absorption edge in the ultraviolet region for all the crystallographic directions and reaches its peak near 11.5 eV. The calculated absorption spectrum reproduces the transparency of GdPO4 across the visible range and predicts a fundamental interband absorption edge in the deep-UV at 8 eV, where the absorption coefficient exceeds 105 cm−1. In the 4–5.2 eV window, theory yields weak features that we assigned to intra-4f transitions of Gd3+, in agreement with the experimental spectrum, which displays the 8S7/2 → 6P7/2 (4.0 eV) and a pronounced 8S7/2 → 6IJ (4.5 eV) band. The absence of an interband onset in the measured data is attributed to the limited spectral range of the utilized spectrophotometer (≤5 eV); measurements extending into the vacuum ultra-visible (VUV) would be required to observe the theoretical edge. Minor discrepancies in the intensity and sharpness of the 4f lines likely arise from the broadening parameter during calculation, where we assume that two peaks overlapped to give a bump-like feature in the energy range 4–5.2 eV. Overall, the combined results establish GdPO4 as a wide band gap insulator with localized 4f absorptions in the near-UV region and a fundamental gap located in the deep-UV region.
The optical conductivity σ(ω) represents the generation of conduction band electrons upon interaction with incident photons of specific energy. Similar to the absorption, the onset of optical conductivity occurs at the band gap energy. The small observed values (300 Ω−1 cm−1) in the energy range of 4–5.2 eV are a sign of inter-band transitions. A pronounced peak is observed around 12 eV for the zz direction, indicating strong optical transitions in the ultraviolet region of the electromagnetic spectrum. On the other hand, a delayed strong peak is observed at energy values of 10.5 and 11 eV for xx and yy directions.
Fig. 6(c and d) illustrate the refractive index n(ω) and the extinction coefficient k(ω). The simulated refractive indexes of n(xx), n(yy), and n(zz) are 1.51, 1.57, and 1.64 eV, respectively, which is closer to the experimental value 1.76 eV reported for Nd doped GdPO4.41 This is also in good agreement with a previous theoretical study 1.55–1.63.20 We notice two peaks, the first is around the 4f–4f transition peak (4 eV), where n has reached 1.8. The second strong peak depicted at 8.5 eV is due to the 4f–5d transition, which is known to have a higher probability, where n has reached a value of 2.5. The extinction coefficient k(ω) shows a bump at an energy range of 4–5 eV, then it starts to rise at the band gap energy, displaying a prominent peak within the 9.5–12 eV energy range. This peak coincides with that of the imaginary part of the dielectric function ε2(ω), indicating efficient photon absorption in this spectral region.
The simulated spectra of the real ε1(ω) and imaginary ε2(ω) parts of the complex dielectric function ε(ω) of GdPO4 are illustrated in Fig. 6(e) and (f). The dielectric function ε(ω) is calculated for the polarized incident radiations along the xx, yy, and zz directions for the purpose of examining the optical anisotropy. It is seen from Fig. 6(e) and (f) show that both the real and imaginary dielectric functions under the three directions do not coincide, demonstrating a minor optical anisotropy. From the dispersion curve of the real part, a progressive increase from the static values of ε1(ω) is shown: ε1 = 2.3 along xx, ε1 = 2.5 along yy, and ε1 = 2.6 along zz. Following the initial jump, ε1(ω) reaches a maximum value for each direction at energy values of 8.5 eV, 8.8 eV, and 8.5 eV, respectively, for xx, yy, and zz directions. Beyond this, ε1(ω) starts to decrease progressively. The imaginary part of the dielectric function ε2 designates the absorption and emission of light. It represents the transitions of electrons between occupied and unoccupied states. Every single peak describes a transition. The ε2 curve gives a clear value of the energy gap of the material, which is depicted at 8 eV; another peak is observed at 4 eV, the latter designates the emission, which is congruent with results from the electronic structure and experimental studies.
The experimentally obtained ε2 values are lower than the simulated values. This is due to the difference in the nature of the intrinsic response of an ideal, dense single crystal. Conversely, the experimental data come from a powder sample. The random orientation of the anisotropic crystallites induces an average dielectric tensor, and, more importantly, the high porosity causes significant light scattering. This scattering considerably reduces the apparent amplitude of the measured ε2, thus explaining the order-of-magnitude difference between the measured and calculated signal intensity.
5. Conclusion
Our combined DFT and spectroscopic study accurately characterizes GdPO4 as a wide-bandgap optical material, demonstrating the correctness of the fitted theoretical framework and providing a reliable reference for the bandgap estimation on lanthanide-based phosphates and luminescence property tuning for future applications in photonics, scintillation, and biomedical imaging. Rietveld analysis showed that the solid-state synthesized GdPO4 crystallizes in the monoclinic space group P21/n. Experimentally, the UV-vis absorption spectrum revealed two distinct peaks at 275 and 303 nm. These peaks correspond to the 4f–4f transitions characteristic of the Gd3+ ion. The fundamental band edge could not be depicted since it surpasses the operational spectral limits (1.5–5.4 eV) of the spectrophotometer. In the theoretical analysis, using the mBJ+SOC framework, we determined the direct electronic bandgap of the GdPO4 monazite crystal to be 7.8 eV, corresponding to the 4fn−1 5d1 transition. This value is consistent with other rare-earth orthophosphate systems and a more accurate prediction obtained with GGA+U and the HSE06 hybrid functional. Intraconfigurational 4f–4f transitions were identified by DFT and analyzed in terms of amplitude and intensity. Our study provides accurate predictions of the energy levels of GdPO4 and the possible radiative emission energy, which is equal to 3.96 eV, based on DOS and band structure calculations. This analysis presents a novelty. Finally, the magnetic moment calculated for the gadolinium (Gd) atom was 6.86µB, which is in good accordance with the experimentally measured magnetic moment of 7.2 ± 0.3µB. This combined study permits a deeper comprehension of electronic transition types in lanthanide-based phosphors and provides the scientific community with a robust basis for tuning of new luminescent materials.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
Data will be made available on request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d6ma00117c.
References
- S. N. Achary, S. Bevara and A. K. Tyagi, Recent progress on synthesis and structural aspects of rare-earth phosphates, Coord. Chem. Rev., 2017, 340, 266–297, DOI:10.1016/j.ccr.2017.03.006.
- S. K. Sharma, J. Beyer, R. Gloaguen and J. Heitmann, Non-quenching photoluminescence emission up to at least 865 K upon near-UV excitation in a single crystal of orange-red emitting SmPO4, Phys. Chem. Chem. Phys., 2019, 21, 25669–25677, 10.1039/C9CP05663G.
- N. Huittinen, Y. Arinicheva, M. Schmidt, S. Neumeier and T. Stumpf, Using Eu3+ as an atomic probe to investigate the local environment in LaPO4–GdPO4 monazite end-members, J. Colloid Interface Sci., 2016, 483, 139–145, DOI:10.1016/j.jcis.2016.08.027.
- S. K. Sharma, T. Köhler, J. Beyer, M. Fuchs, R. Gloaguen and J. Heitmann, Extending the temperature sensing range using Eu3+ luminescence up to 865 K in a single crystal of EuPO4, Phys. Chem. Chem. Phys., 2019, 21, 16329–16336, 10.1039/C9CP03501J.
- S. K. Sharma, J. Beyer, R. Gloaguen and J. Heitmann, Comparing the optical properties and thermal stability of green (TbPO4), yellow (DyPO4), and red (PrPO4) emitting single crystal samples, Phys. Chem. Chem. Phys., 2020, 22, 10247–10255, 10.1039/D0CP01561J.
- M. L. Williams, M. J. Jercinovic and C. J. Hetherington, Microprobe Monazite Geochronology: Understanding Geologic Processes by Integrating Composition and Chronology, Annu. Rev. Earth Planet. Sci., 2007, 35, 137–175, DOI:10.1146/annurev.earth.35.031306.140228.
- A. Lempicki, E. Berman, A. J. Wojtowicz, M. Balcerzyk and L. A. Boatner, Cerium-doped orthophosphates: new promising scintillators, IEEE Trans. Nucl. Sci., 1993, 40, 384–387, DOI:10.1109/23.256585.
- P. Laporta, S. Taccheo, S. Longhi, O. Svelto and C. Svelto, Erbium–ytterbium microlasers: optical properties and lasing characteristics, Opt. Mater., 1999, 11, 269–288, DOI:10.1016/S0925-3467(98)00049-4.
- S. Neumeier, Y. Arinicheva, Y. Ji, J. M. Heuser, P. M. Kowalski, P. Kegler, H. Schlenz, D. Bosbach and G. Deissmann, New insights into phosphate based materials for the immobilisation of actinides, Radiochim. Acta, 2017, 105, 961–984, DOI:10.1515/ract-2017-2819.
- R. C. Ewing and L. Wang, Phosphates as Nuclear Waste Forms, Rev. Mineral. Geochem., 2002, 48, 673–699, DOI:10.2138/rmg.2002.48.18.
- S. Rodriguez-Liviano, A. I. Becerro, D. Alcántara, V. Grazú, J. M. De La Fuente and M. Ocaña, Synthesis and Properties of Multifunctional Tetragonal Eu:GdPO4 Nanocubes for Optical and Magnetic Resonance Imaging Applications, Inorg. Chem., 2013, 52, 647–654, DOI:10.1021/ic3016996.
- M. Lassri, S. El Ouahbi, I. Abdelrhafore, L. E. Hachemi Omari, A. El Hachmi, H. Hamouda, B. Harradi, Y. Ounza, G. Viscusi, C. Minaud, C. Jama and H. Lassri, Large reversible magnetocaloric effect in GdPO4 nano-spherical particles at ultralow temperatures, J. Magn. Magn. Mater., 2025, 173291, DOI:10.1016/j.jmmm.2025.173291.
- J. Fang, B. Pan, X. Hu, N. Yu, M. Pan, H. Ge, H. Yang and Q. Wu, Giant Magnetocaloric Effect and Magnetic Critical Behavior in Polycrystalline GdPO4, J. Supercond. Nov. Magn., 2025, 38, 13, DOI:10.1007/s10948-024-06866-8.
- Y. Y. Yu, D. N. Petrov, K. C. Park, B. T. Huy and P. T. Long, Magnetic and cryogenic magnetocaloric properties of GdPO4 nanorods, J. Magn. Magn. Mater., 2021, 519, 167452, DOI:10.1016/j.jmmm.2020.167452.
- Y. Li, J. Wang, H. Huang, J. Wang, M. Zhang and Mm Liang, Co-coating effect of GdPO4 and carbon on LiFePO4 cathode surface for lithium ion batteries, Adv. Powder Technol., 2019, 30, 1442–1449, DOI:10.1016/j.apt.2019.04.017.
- Y. Li, Q. Ren, W. Xu, H. Huang, L. Chen, Y. Fu, J. Wang and Z. Wang, Surface stabilized Ni-rich LiNi0.9Co0.08Al0.02O2 cathode materials via surface modification with GdPO4 for lithium-ion batteries, Solid State Ionics, 2023, 403, 116397, DOI:10.1016/j.ssi.2023.116397.
- J. P. Perdew, K. Burke and M. Ernzerhof, Generalized Gradient Approximation Made Simple, Phys. Rev. Lett., 1996, 77, 3865–3868, DOI:10.1103/PhysRevLett.77.3865.
- K. Schwarz, P. Blaha and G. K. H. Madsen, Electronic structure calculations of solids using the WIEN2k package for material sciences, Comput. Phys. Commun., 2002, 147, 71–76, DOI:10.1016/S0010-4655(02)00206-0.
- P. Novák, F. Boucher, P. Gressier, P. Blaha and K. Schwarz, Electronic structure of the mixed valence system (YM)2 BaNiO5 (M = Ca, Sr), Phys. Rev. B: Condens. Matter Mater. Phys., 2001, 63, 235114, DOI:10.1103/PhysRevB.63.235114.
- F. Tran and P. Blaha, Accurate Band Gaps of Semiconductors and Insulators with a Semilocal Exchange-Correlation Potential, Phys. Rev. Lett., 2009, 102, 226401, DOI:10.1103/PhysRevLett.102.226401.
- S. Gates-Rector and T. Blanton, The Powder Diffraction File: a quality materials characterization database, Powder Diffr., 2019, 34, 352–360, DOI:10.1017/S0885715619000812.
- T. Degen, M. Sadki, E. Bron, U. König and G. Nénert, The HighScore suite, Powder Diffr., 2014, 29, S13–S18, DOI:10.1017/S0885715614000840.
- D. Louër and A. Boultif, Some further considerations in powder diffraction pattern indexing with the dichotomy method, Powder Diffr., 2014, 29, S7–S12, DOI:10.1017/S0885715614000906.
- H. M. Rietveld, A profile refinement method for nuclear and magnetic structures, J. Appl. Crystallogr., 1969, 2, 65–71, DOI:10.1107/S0021889869006558.
- J. Rodríguez-Carvajal, Recent advances in magnetic structure determination by neutron powder diffraction, Phys. B, 1993, 192, 55–69, DOI:10.1016/0921-4526(93)90108-I.
- T. Hahn, International tables for crystallography, 5th edn, Kluwer academic publ, Dordrecht, 2002 Search PubMed.
- E. N. Silva, A. P. Ayala, I. Guedes, C. W. A. Paschoal, R. L. Moreira, C.-K. Loong and L. A. Boatner, Vibrational spectra of monazite-type rare-earth orthophosphates, Opt. Mater., 2006, 29, 224–230, DOI:10.1016/j.optmat.2005.09.001.
- G. M. Begun, G. W. Beall, L. A. Boatner and W. J. Gregor, Raman spectra of the rare earth orthophosphates, J. Raman Spectrosc., 1981, 11, 273–278, DOI:10.1002/jrs.1250110411.
- J. Heuser, A. A. Bukaemskiy, S. Neumeier, A. Neumann and D. Bosbach, Raman and infrared spectroscopy of monazite-type ceramics used for nuclear waste conditioning, Prog. Nucl. Energy, 2014, 72, 149–155, DOI:10.1016/j.pnucene.2013.09.003.
- N. K. Sahu, R. S. Ningthoujam and D. Bahadur, Disappearance and recovery of luminescence in GdPO4:Eu3+ nanorods: Propose to water/OH• release under near infrared and gamma irradiations, J. Appl. Phys., 2012, 112, 014306, DOI:10.1063/1.4731644.
- Z. Khadraoui, K. Horchani-Naifer, M. Ferhi and M. Ferid, Electronic structure and optical properties of TbPO4: Experiment and density functional theory calculations, Opt. Mater., 2015, 47, 484–489, DOI:10.1016/j.optmat.2015.06.025.
- S. K. Sharma, Evidence of thermal ionization induced luminescence quenching in CePO4 and GdPO4, Mater. Adv., 2023, 4, 157–163, 10.1039/D2MA00951J.
- E. Nakazawa and F. Shiga, Vacuum ultraviolet luminescence-excitation spectra of RPO4: Eu3+ (R = Y, La, Gd and Lu), J. Lumin., 1977, 15, 255–259, DOI:10.1016/0022-2313(77)90024-2.
- T. Lyu and P. Dorenbos, Charge carrier trapping processes in lanthanide doped LaPO4, GdPO4, YPO4, and LuPO4, J. Mater. Chem. C, 2018, 6, 369–379, 10.1039/C7TC05221A.
- Z. Khadraoui, K. Horchani-Naifer, M. Ferhi and M. Ferid, The density functional study of electronic structure and optical properties of gadolinium monophosphate, Chin. J. Phys., 2019, 59, 333–339, DOI:10.1016/j.cjph.2019.02.025.
- J. Li, J. Liu, Y. Zhang, W. Sun, Y. Wang, H. Wu, L. Li, C. Cheng, Y. Wang, K. Tan and F. Liu, Exploring the Ln–O bonding nature and charge characteristics in monazite in relation to microwave dielectric properties, J. Am. Ceram. Soc., 2024, 107, 175–187, DOI:10.1111/jace.19402.
- I. Abdelrhafor, L. H. Omari, M. Lassri, L. Abbassi, A. El Hachmi, S. Tarik, V. Tuyikeze, H. Lassri, E. K. Hlil and M. Chafi, Structural, optical, and electronic properties of europium orthophosphate: a combination of experimental and theoretical studies, J. Mater. Chem. C, 2026, 14(11), 4598–4607, 10.1039/d5tc03857j.
- C. Ambrosch-Draxl and J. O. Sofo, Linear optical properties of solids within the full-potential linearized augmented planewave method, Comput. Phys. Commun., 2006, 175, 1–14, DOI:10.1016/j.cpc.2006.03.005.
- K. Mori, H. Nishimura, M. Nakayama and H. Ishibashi, Dynamical aspects of the core excitons formed by the 4f–4f transitions of Gd3+ in Gd2SiO5, J. Lumin., 2000, 87–89, 266–268, DOI:10.1016/S0022-2313(99)00310-5.
- Z. Khadraoui, K. Horchani-Naifer, M. Ferhi and M. Ferid, Electronic structure and optical properties of TbPO4: Experiment and density functional theory calculations, Opt. Mater., 2015, 47, 484–489, DOI:10.1016/j.optmat.2015.06.025.
- N. Ben Amar, T. Kallel, T. Koubaa, M. A. Hassairi, M. Dammak and E. Cavalli, Synthesis, characterization and optical spectroscopy of GdPO4:Er3+, Luminescence, 2020, 35, 1056–1067, DOI:10.1002/bio.3817.
| This journal is © The Royal Society of Chemistry 2026 |