 Open Access Article
Open Access ArticleSynergistic Tamra-Yasad Bhasma and cinnamaldehyde nano-shield hydrogels for diabetic wound repair
Suhela
Tyeb
 *a,
Nitesh
Kumar
b,
Vivek
Verma
*a,
Nitesh
Kumar
b,
Vivek
Verma
 c,
Ashok
Kumar
c,
Ashok
Kumar
 d and
Sanjana
Kaul
a
d and
Sanjana
Kaul
a
aSchool of Biotechnology, University of Jammu, Jammu, J&K 180006, India. E-mail: suhelat@gmail.com
bDepartment of Materials Engineering, Indian Institute of Technology Jammu, Jammu, J&K 181221, India
cDepartment of Materials Science and Engineering, Indian Institute of Technology Kanpur, UP 208016, India
dBiosciences and Bioengineering Department, Indian Institute of Technology Kanpur, UP 208016, India
First published on 21st April 2026
Abstract
Around 100 million individuals with diabetes in India are at risk of developing diabetic wounds in their lifetime. These wounds cause severe reductions in the quality of life of patients due to increased economic burden and risk of limb amputations. Indigenous wound dressings offer cost-effective solutions for the management and healing of such wounds. Loading of bioactive formulations in passive dressing material has led to successful intervention in hampered wound healing cascades. The role of traditional wound healing formulations, such as aloe vera, turmeric, and cinnamon oil extracts loaded in hydrogels, hydrocolloids, sponges and other dressing materials, has been reported earlier. Tamra Bhasma and Yasad Bhasma are two types of Ayurvedic formulations (Bhasmas) cited in the Indian traditional medicinal text “Rasa-shastra”. These Bhasmas have been studied in isolation and show beneficial attributes, such as free radical scavenging, hepatoprotective activity and anti-inflammatory effects. The present work is aimed at studying the combinatorial effect of Tamra Bhasma and Yasad Bhasma in conjunction with the potent anti-microbial “cinnamaldehyde nanoemulsion (NE)” loaded in two different forms of a polyvinyl alcohol (PVA) matrix: hydrogel and sponge. In vitro studies on three cell lines, NIH 3T3, HaCaT and HuVECs, showed sustained release of these Bhasmas and NE, leading to enhanced cell proliferation, cell migration, free radical scavenging and collagen formation. Compared with the PVA hydrogel and sponge controls, the developed bioactive wound dressing shows promise for the healing and management of infected diabetic wounds.
1. Introduction
Diabetic wounds are difficult to heal owing to hampered angiogenesis, risk of infection and impaired collagen formation. Multiple interventions have been examined in previous studies on biochemical pathways, addressing different phases of the wound-healing cascade. In this regard, hydrogels, hydrocolloids and sponges loaded with growth factors, debriding agents, antibiotics, exosomes and metallic nanoparticles have been explored.1 Hydrogels are a class of water-soluble polymers with tunable properties, such as swelling, water–vapor transmission, porosity, stiffness and non-immunogenicity, that are suitable for dermal and epidermal wound healing.2,3 Polyvinyl alcohol (PVA) is one such hydrogel polymer that has been used extensively for the healing and management of chronic diabetic wounds.4,5 Bialik-wqs et al. synthesized PVA-sodium alginate (SA) composite hydrogels for the prolonged release of aloe vera as a promising wound-healing material.6 Wang et al. developed a piezoelectric PVA-PVDF-based composite hydrogel to enhance the cellular proliferation, migration and secretion of ECM proteins for efficient healing of chronic wounds.7 Some of the earlier works studied the release of traditional medicines from PVA hydrogel matrices, such as that reported by Wang et al.8 The authors prepared a PVA-chitosan composite hydrogel for the sustained release of the Tibetan eighteen flavor dangshen pills to promote the anti-inflammatory, cell proliferative and analgesic properties of the Tibetan medicine for chronic wound healing. Ciftci et al. reported the preparation and characterization of PVA-chitosan composite mats by an electrospinning method for the sustained release of curcumin, which showed antioxidant and anti-microbial properties against E. coli and S. aureus.9 Rashid et al. studied the role of the sustained release of curcumin from PVA-gelatin for the downregulation of pro-inflammatory cytokines for full-thickness wound healing.10Tamra Bhasma and Yasad Bhasma are two nanoformulations with origins in Rasa-Shastra of the Indian Ayurvedic medicine system. Tamra Bhasma (TB) has been reported to show free-radical scavenging, hepatoprotective activity and anti-inflammatory effects.11–13 Physicochemical characterization studies and phase analysis of Tamra Bhasma show the presence of nano- to sub-micron-particles of copper sulfide.14,15Yasad Bhasma (YB) has also shown pharmaceutical benefits, such as antibacterial and anti-diabetic effects.16,17 Compositional and morphological analyses of Yasad Bhasma have shown the presence of ZnO particles in nano- to sub-micron-ranges.17,18 The biopharmaceutical benefits of these two Bhasmas are of significant interest with respect to diabetic wound healing. Cinnamon oil is known for its potent anti-microbial properties, which are useful in the management of infection in wounds.19 Ahmadi et al. studied the antioxidant and anti-bacterial properties of Cinnamon verum essential oil in mice infected-wound models.20 The study showed that topical application of cinnamon oil cream (with paraffin) to mouse wounds infected with S. aureus and P. aeruginosa bacterial strains led to a 6-fold decrease in bacterial counts on the twelfth day and granular tissue formation on the fourteenth day of treatment. However, retaining the anti-bacterial and antioxidant properties of cinnamon oil for longer durations in diabetic or chronic wounds is challenging due to loss of its chemical stability in complex micro-environments of such wound beds. Delivering cinnamon oil extract in the form of nanoemulsion particles is one strategy to ensure its sustained release in the wound bed and achieve long-term and potent antimicrobial effects. Moustafa et al. demonstrated the potent anti-bacterial effect of a cinnamon oil nanoemulsion delivered from a carboxymethyl cellulose–starch composite wound dressing on four bacterial strains: S. aureus, B. cereus, Escherichia coli and Staphylococcus typhimurium.21In the present work, the potency of these two Ayurvedic Bhasmas, in combination with cinnamon oil nanoemulsion, was explored by loading them in a PVA-based film and sponge matrix for healing and management of infected diabetic wounds. The combined bioactive system (TB + YB + NE) was assessed in vitro for its antioxidant, antibacterial, pro-proliferative, pro-migratory, collagen-forming, and angiogenic effects. The results showed that these cost-effective Ayurvedic formulations have significant promise as potent healing agents for diabetic wounds, in combination with modern drug-delivery hydrogels and sponges, possessing tunable physical and mechanical properties.
1.1. Materials
Cinnamaldehyde was purchased from HiMedia (GRM3277). Polyvinyl alcohol (PVA) was procured from Loba Chemie (mol wt. 115![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000, 99% hydrolysis), and glycerol was purchased from Fisher Scientific, Mumbai (India). Dulbecco's Modified Eagle's Medium (Thermo Fisher, 12100046), high glucose media, and trypsin-ethylenediaminetetraacetic acid (Trypsin-EDTA Thermo 25200072) were purchased from Thermo Fisher Scientific, Mumbai (India). Medium chain triglyceride oil (MCT) was procured from Novus Life Science, Mumbai, India (caprylic
000, 99% hydrolysis), and glycerol was purchased from Fisher Scientific, Mumbai (India). Dulbecco's Modified Eagle's Medium (Thermo Fisher, 12100046), high glucose media, and trypsin-ethylenediaminetetraacetic acid (Trypsin-EDTA Thermo 25200072) were purchased from Thermo Fisher Scientific, Mumbai (India). Medium chain triglyceride oil (MCT) was procured from Novus Life Science, Mumbai, India (caprylic![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) capric acid = 60
capric acid = 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40), and fetal bovine serum (FBS), of EU-approved South American origin, was procured from Gibco, Mumbai (India). NIH 3T3 cells were obtained from the National Centre for Cell Science (NCCS), Pune (India). HaCaT cells were kindly provided by Prof Kaushik Chatterjee (IISc Bangalore). HUVEC cells were kindly provided by Prof Ashok Kumar (IIT Kanpur). Alamar Blue was purchased from Himedia (India). Pseudomonas aeruginosa (MTCC-3541) and Staphylococcus aureus (MTCC-96) were procured from MTCC, Chandigarh (India). Pecam-1 (Abcam ab28364). Tamra and Yasad Bhasma were procured from a local market in India.
40), and fetal bovine serum (FBS), of EU-approved South American origin, was procured from Gibco, Mumbai (India). NIH 3T3 cells were obtained from the National Centre for Cell Science (NCCS), Pune (India). HaCaT cells were kindly provided by Prof Kaushik Chatterjee (IISc Bangalore). HUVEC cells were kindly provided by Prof Ashok Kumar (IIT Kanpur). Alamar Blue was purchased from Himedia (India). Pseudomonas aeruginosa (MTCC-3541) and Staphylococcus aureus (MTCC-96) were procured from MTCC, Chandigarh (India). Pecam-1 (Abcam ab28364). Tamra and Yasad Bhasma were procured from a local market in India.
2. Methods
2.1. Preparation of cinnamon oil NE
Cinnamaldehyde and medium-chain triglyceride were mixed in a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10. Tween 80 (3 mL) was added to the prepared CBO-MCT (cannabichromene + medium-chain triglyceride) mixture (5 mL) and stirred for 30 min at 800 rpm. To this, DI water (42 mL) was added dropwise using a 200-microliter pipette to obtain a coarse emulsion. This emulsion was further subjected to ultrasonication at 40% amplitude for 15 min under ice with a 9-s on–off cycle to obtain a nanoemulsion. The as-prepared nanoemulsion was used for further characterizations and in vitro studies.
10. Tween 80 (3 mL) was added to the prepared CBO-MCT (cannabichromene + medium-chain triglyceride) mixture (5 mL) and stirred for 30 min at 800 rpm. To this, DI water (42 mL) was added dropwise using a 200-microliter pipette to obtain a coarse emulsion. This emulsion was further subjected to ultrasonication at 40% amplitude for 15 min under ice with a 9-s on–off cycle to obtain a nanoemulsion. The as-prepared nanoemulsion was used for further characterizations and in vitro studies.
2.2. Characterization of Bhasmas and cinnamaldehyde nanoemulsion
The procured Bhasmas and synthesized nanoemulsion were subjected to morphological and physicochemical analysis. The size distribution of the Bhasma and nanoemulsion was obtained by dynamic light scattering (DLS). The hydrodynamic size distribution of the synthesized samples was determined using a Litesizer 500 (Anton Paar, Austria) instrument. Measurements were performed at a temperature of 25 °C ± 0.1 °C using quartz cuvettes in deionized water as the dispersing medium. The solvent refractive index and viscosity were set at 1.3303 and 0.8903 mPa s, respectively. Each sample was equilibrated for 2 min prior to analysis, and measurements were taken in automatic scattering mode with multiple short runs (10 s each) processed using the general cumulant analysis model. The morphological and microstructural analysis of the synthesized samples was carried out using transmission electron microscopy (TEM). A small quantity of the dried sample was ultrasonically dispersed in ethanol (99.9%) for 10 minutes to ensure uniform dispersion. A drop of the well-dispersed suspension was then placed onto a carbon-coated copper grid and air-dried at room temperature before imaging. The samples were observed under a high-resolution transmission electron microscope (JEOL JEM-2100) to capture images at various magnifications. The micrographs obtained were analyzed to determine particle morphology. Nanoemulsion particle morphology was also visualized under a Jeol high-resolution transmission electron microscope operating in the bright-field mode. The crystalline structure and phase composition of the synthesized samples were analyzed using X-ray diffraction (XRD). XRD patterns were recorded on an X-ray diffractometer (PANalytical X’Pert PRO/Bruker D8 Advance) equipped with a Cu Kα radiation source (λ = 1.5406 Å), operated at 40 kV and 30 mA. The phase composition of as-received Tamra Bhasma and Yasad Bhasma samples was studied with a PANalytical X’pert diffractometer with Bragg–Brentano geometry. The wavelength used was Cu-Kα with a line detector. The scan angle was taken between 20°–100° at a scan rate of 1° min−1. The peaks were indexed by reference to the standard Pearson's crystallographic database.2.3. Optimization of the Bhasma and NE concentrations
The biocompatibility of Tamra Bhasma, Yasad Bhasma and nanoemulsion was evaluated on NIH 3T3 fibroblast cells using the Bhasma- and nanoemulsion-spiked media. The spiked media was prepared by adding different concentrations of Bhasma and nanoemulsion in serum-free media. The spiked media was filter-sterilized (0.22 micron) and added with FBS (10%) and antibiotic (1%) to make complete media. The fibroblast cells were cultured in DMEM media, 10% FBS and 1% penicillin/streptomycin at 37 °C with 5% CO2. Around 1 × 104 cells were seeded per well and incubated for 24 h to allow attachment; thereafter, the media was replaced with complete spiked media and incubated for 48 h at 37 °C. Cells without any bioactives served as a 2D positive control. The cell viability was evaluated by Alamar Blue assay and live/dead staining.2.4. Preparation of the PVA film and sponge loaded with Tamra Bhasma, Yasad Bhasma and nanoemulsion
For the preparation of the bioactive-loaded PVA hydrogel film, the required amounts of Tamra and Yasad Bhasma were suspended in 10 mL of DI water and subjected to probe sonication at 40% power for 10 min. Separately, PVA was dissolved in 40 mL DI water at 90 °C to prepare a 7.5 wt% PVA solution. After complete dissolution of PVA, the solution was cooled to room temperature, and the required amounts of Bhasma solution and cinnamaldehyde nanoemulsion were added dropwise under stirring for another 30 min. The cooled solution was cast in a 90 mm Petri dish and dried at room temperature to prepare the bioactives-loaded PVA hydrogel films.To prepare the sponge, bioactive-loaded solution (PVA 5% w/v) was prepared as described for the PVA film, and the cooled solution was poured into a 2 mL syringe and frozen at −80 °C overnight, followed by lyophilization (Labconco 2.5 L) to obtain the cryogel sponges.
PVA films and sponges without any additives (TB, YB, and NE) were prepared as controls. The films and sponges with all additives were named PVA TYN Films and PVA TYN Sponges, respectively.
2.5. Material testing and characterization studies
The morphological features of the synthesized PVA TYN films and PVA TYN sponges were examined using scanning electron microscopy (SEM). The PVA TYN sponges were cut into cross-sectional discs (2 mm thickness, 5 mm diameter) to observe the internal porous architecture. The samples were sputter-coated with gold for 180 seconds using a sputter coater (EMLTECH 7620) and observed under SEM (Carl Zeiss EVO 50). The pore size was analyzed from SEM micrographs using the ImageJ software. For each formulation, three independent sponge samples were analyzed, and 3–4 representative images were captured per sample. The porosity of the cryogels was further quantified using the liquid displacement method with cyclohexane as a non-wetting solvent.22 Circular discs of 5 mm × 5 mm were weighed (W1), submerged in cyclohexane to measure the immersed weight (W2), and then reweighed after removal (W3). Porosity (%) was calculated using the following relation: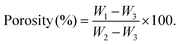 | (1) |
The mechanical properties of the PVA TYN films were evaluated using an Instron Universal Testing Machine (Model 3345). The thickness of each sample was measured using a micrometer (Mitutoyo Corporation, Japan) prior to testing. The tensile strength and elongation at break of the hydrogel films were determined in accordance with ASTM D882-12 standards. Film specimens of dimensions 70 mm × 10 mm (length × width) and thickness 200 ± 10 µm were cut with a gauge length of 40 mm. Prior to testing, the samples were immersed in phosphate-buffered saline (PBS, pH 7.4) for 24 hours at 37 °C to attain equilibrium swelling, ensuring evaluation under physiologically relevant hydrated conditions. The tensile test was performed at a crosshead speed of 10 mm min−1, and tensile strength (MPa) and elongation at break (%) were obtained from the stress–strain curves. For compressive testing, PVA TYN sponges were cut into cylindrical discs (5 mm diameter × 8 mm height) and similarly pre-soaked in PBS for 24 hours before measurement, as per a previously published protocol.22 The hydrated samples were tested using a Universal Testing Machine (BiSS India) equipped with a 100 N load cell. Compression was applied at a rate of 6 mm min−1 until the samples were 70% compressed. The compressive strength and compressive modulus were calculated from the resulting stress–strain data. The swelling behavior of the additive-loaded PVA TYN films and sponges was evaluated in phosphate-buffered saline (PBS, pH 7.4) at 37 °C under static conditions to simulate physiological environments. Pre-weighed dry samples (W0) were immersed in PBS and withdrawn at predetermined time intervals. For PVA films, swelling was monitored at 30 min and 1, 2, 4, 8, 12, 24 h. For PVA TYN sponges, swelling was measured at 30 s, 1 m, 2 m, 5 m, 10 m, 30 m, 1 h, 2 h, 4 h, 12 h and 24 h. At each interval, samples were carefully removed, blotted with filter paper and weighed (Wt). The swelling (%) was calculated using the following relation:
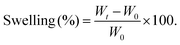 | (2) |
The water–vapor transmission rate (WVTR) of the PVA-based films was determined using the ASTM E96-96 (inverted cup method) with slight modifications. A 25 mL glass bottle was filled with reverse osmosis (RO) water, leaving approximately ½ inch headspace. Circular film specimens (18 mm diameter) were mounted over the mouth of the bottle and sealed with vacuum grease and paraffin wax to ensure airtight attachment. The open bottle served as a positive control, while a rubber-sheet-sealed bottle was used as a negative control. The initial weight (W0) was recorded at t = 0, and subsequent weights were measured every 24 hours for seven days. The WVTR (g cm−2 day−1) was calculated using the following formula:
| WVTR = G/tA | (3) |
2.6. Release kinetics study of TB, YB and NE from PVA TYN film and sponge
The release kinetics of TB, YB and NE were obtained by incubating 1 × 1 cm2 PVA hydrogel films and sponge disks (5 mm diameter × 2 mm height) in DI water for varying time intervals. Following incubation, the supernatants of the Bhasma-loaded samples were collected and digested in nitric acid. The concentration of released TB and YB in water was evaluated by ICPMS measurements after serial dilution. The concentration of these Bhasmas with time was plotted as a release rate kinetics graph. The release kinetics of NE were studied by UV-vis spectroscopy.23 The supernatant of the samples was collected as mentioned above for the Bhasma samples. The concentration of cinnamaldehyde was calculated from the standard graph. The release kinetics graph was further plotted as percentage release kinetics.2.7. Biological evaluation of additive-loaded PVATYN films and sponges
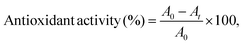 | (4) |
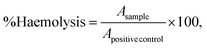 | (5) |
 | (6) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cells per well) cells were seeded in 48-well plates and allowed to attach for 24 hours. The medium was then replaced with the prepared conditioned media, and cells were incubated for 1, 3, and 5 days, with media renewal every 48 hours. Endothelial cells (HUVECs) were cultured in endothelial expansion media and seeded at a density of 5000 cells per well under the same treatment. Cell proliferation was quantified by measuring metabolic activity via Alamar Blue reduction (fluorescence at 560/590 nm). In parallel, Ki-67 staining was used to visualize actively proliferating nuclei, and phalloidin/DAPI co-staining was performed to examine cytoskeletal organization and nuclear integrity. Live/dead assays further confirmed the overall cell viability and biocompatibility.
000 cells per well) cells were seeded in 48-well plates and allowed to attach for 24 hours. The medium was then replaced with the prepared conditioned media, and cells were incubated for 1, 3, and 5 days, with media renewal every 48 hours. Endothelial cells (HUVECs) were cultured in endothelial expansion media and seeded at a density of 5000 cells per well under the same treatment. Cell proliferation was quantified by measuring metabolic activity via Alamar Blue reduction (fluorescence at 560/590 nm). In parallel, Ki-67 staining was used to visualize actively proliferating nuclei, and phalloidin/DAPI co-staining was performed to examine cytoskeletal organization and nuclear integrity. Live/dead assays further confirmed the overall cell viability and biocompatibility.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000/well) were seeded in DMEM containing 10% FBS and cultured for 16 hours to form a uniform confluent layer. The medium was replaced with 1% FBS DMEM containing Mitomycin C (5 µg mL−1) for 3 hours to inhibit proliferation. A uniform scratch was made using a sterile 200 µL pipette tip, and the detached cells were gently washed away with warm PBS. The cells were then treated with conditioned media containing 5% FBS and incubated at 37 °C. Scratch closure was monitored under an inverted microscope at 0 h and 24 h, and subsequent intervals until closure was complete. Migration efficiency was quantified as the percentage of wound closure using the ImageJ software.
000/well) were seeded in DMEM containing 10% FBS and cultured for 16 hours to form a uniform confluent layer. The medium was replaced with 1% FBS DMEM containing Mitomycin C (5 µg mL−1) for 3 hours to inhibit proliferation. A uniform scratch was made using a sterile 200 µL pipette tip, and the detached cells were gently washed away with warm PBS. The cells were then treated with conditioned media containing 5% FBS and incubated at 37 °C. Scratch closure was monitored under an inverted microscope at 0 h and 24 h, and subsequent intervals until closure was complete. Migration efficiency was quantified as the percentage of wound closure using the ImageJ software.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cells per well) were seeded in a 48-well plate and allowed to attach for 24 hours. The cells were pre-treated with conditioned media for 12 hours, followed by exposure to menadione (5.8 mM) for 2 hours to induce ROS production. After washing with PBS, the cells were incubated with 2 µM DCFH-DA for 30 minutes to detect intracellular ROS levels. Lower fluorescence intensity showed enhanced antioxidant protection mediated by the released Bhasma and NE components.
000 cells per well) were seeded in a 48-well plate and allowed to attach for 24 hours. The cells were pre-treated with conditioned media for 12 hours, followed by exposure to menadione (5.8 mM) for 2 hours to induce ROS production. After washing with PBS, the cells were incubated with 2 µM DCFH-DA for 30 minutes to detect intracellular ROS levels. Lower fluorescence intensity showed enhanced antioxidant protection mediated by the released Bhasma and NE components.
3. Results and discussions
3.1. Characterization of Bhasmas and synthesized cinnamon bark oil nanoemulsions
The particle size distributions of as-received Tamra Bhasma and Yasad Bhasma and the as-prepared nanoemulsion are shown in Fig. 1(a–c). The average particle sizes for Tamra Bhasma, Yasad Bhasma and NE were found to be 255 nm, 270 nm and 190 nm, respectively. The TEM micrographs (Fig. 1(d–f)) revealed distinct morphological differences among the three samples. The nanoemulsion showed a uniform, spherical morphology with a size distribution of 150–250 nm and an average size of 190 nm. The particles appeared well-dispersed with minimal aggregation, indicating efficient emulsification and good colloidal stability. The smooth contrast and absence of sharp crystalline boundaries suggest that the droplets were amorphous or liquid-like in nature (Fig. 1(a and d)). The Tamra sample displayed aggregated clusters of near-spherical nanoparticles, with individual particle sizes primarily in the range of 170–300 nm, with an average particle size of 250 nm (Fig. 1(b and e)). The TEM image showed irregularly shaped particle morphology with varying sizes, which might be due to particle aggregation (Fig. 1(e)). The Yasad sample exhibited a size distribution similar to that of Tamra Bhasma particles, with an average size of around 270 nm (Fig. 1(c and f)). Fig. 1(f) shows the TEM micrograph of Yasad Bhasma, which exhibits particles of various sizes arising from aggregation. The phase composition of the two Bhasmas was analyzed by X-ray diffraction. Fig. 1(g and h) shows the X-ray diffractogram of Tamra and Yasad Bhasma with CuS and ZnO being the major detectable phases, respectively. The sharpness of the peaks indicates high crystallinity, and the absence of additional impurity peaks suggests successful phase formation during the calcination process in Bhasma preparation.3.2. Optimization of the Bhasma and NE concentrations
The useful concentrations of Bhasma and NE that could be loaded on the PVA matrix for the fabrication of the film and sponge dressing were determined by viability assays with fibroblast cells. The concentrations of Bhasma from 500 µg mL−1 to 6.25 µg mL−1 were evaluated by preparing Bhasma-containing DMEM media with concentrations of 500, 250, 125, 75, 37.5, 18.75 and 9.5 µg mL−1. Similarly, nanoemulsions with concentrations of 50, 25, 12.5, 6.25, 3.12 and 0.75 µL mL−1 were evaluated. The minimum concentration that resulted in a viability of more than 80% after 48 h was selected to fabricate the dressing material. The results are presented in Fig. 2(a and b).3.3. Preparation and characterization of Bhasma- and NE-loaded PVA hydrogel films and PVA cryogel sponges
The macroscopic appearance of the fabricated PVA TYN hydrogel films and sponges revealed structural uniformity. The films exhibited smooth surfaces while the lyophilized sponges retained their shape integrity and exhibited a porous, foam-like structure (Fig. 3(a)). The SEM micrograph of the sponge (Fig. 3(b)) shows an interconnected porous architecture with pore sizes in the range of 50–150 µm. The EDS mapping (Fig. 3(b)) confirmed the homogeneous distribution of Cu, Zn, S, C, and O, indicating uniform incorporation of TB (CuS) and YB (ZnO) within the PVA matrix. The porosity measurements shown in Fig. 3(d and e) indicated a high interconnected void fraction (85%), facilitating high exudate retention and nutrient exchange. The contact angle measurement (Fig. 3(c)) demonstrated enhanced hydrophilicity of the PVATYN films compared to native PVA. The improved wettability can be attributed to the incorporation of polar metal oxide species (CuS, ZnO) and NE, increasing surface energy and water affinity. The increased hydrophilicity ensures nonadherence of the dressings, resulting in painless dressing changes and negligible damage to regenerating tissue.26 The tensile and compressive properties of the PVA TYN composites (Fig. 3(h and i)) revealed a significant enhancement compared to native PVA films and sponges. The tensile strength of the swelled PVATYN film increased to 1.6 MPa, with an elongation at break of 200%, compared to 0.5 MPa and 80%, respectively, for pure PVA. Similarly, the compressive strength of the swelled sponge improved from 0.35 MPa (control) to 0.5 MPa, and the compressive modulus increased from 1.0 to 1.3 MPa, respectively. These improvements can be attributed to the reinforcing effects of metallic nanoparticles and the plasticizing effect of the nanoemulsion between PVA hydroxyl groups. The swelling kinetics of the PVATYN films and sponges (Fig. 3(f and g)) demonstrated a rapid initial uptake within the first few hours, followed by a gradual equilibrium phase. The equilibrium swelling ratio of cryogels reached 400%, significantly higher than that of the films (180%), owing to their open-pore network and higher water-retention capacity. The slight reduction in swelling compared to pure PVA can be attributed to enhanced crosslink density resulting from interactions of the Bhasma ions and the NE with the PVA chains. The water–vapor transmission rate (WVTR) (Fig. 3(j)) of the PVA–TYN films was found to be within the optimal range of 2000–2500 g m−2 day−1, making it suitable for wound-healing applications that require balanced moisture permeability. The marginal decrease in the WVTR of PVA–TYN films compared with that of pristine PVA reflects the improved barrier properties with the incorporation of Bhasma particles.3.4. DPPH, antimicrobial and hemolysis assays
The free-radical scavenging properties of the PVA film and sponge loaded with Bhasma nanoemulsion and bioactive agents were studied with DPPH assays, as shown in Fig. 4(a). Around 80% of free-radical scavenging was observed for the YB, NE and PVA TYN films and sponges, while the TB and PVA films showed less than 50% free radical scavenging. The antimicrobial properties of the individual Bhasmas, NE and PVA films were evaluated by time kill assays performed with S. aureus and P. aeruginosa bacterial species (Fig. 4(b and d)). Iodine-incubated bacterial solutions were taken as the positive control, while untreated bacteria were taken as the negative control. All the sample groups, except for the PVA film and sponge, showed killing above 80% for S. aureus and P. aeruginosa. Hemocompatibility of the sample groups was tested on goat's blood. All the sample groups showed less than 5% hemolysis, establishing their hemocompatibility with respect to the known hemolytic material Triton-X (Fig. 4(c)).3.5. Release kinetics
The efficacy of the loaded bioactive agents, TB, YB and NE, is dependent on their release kinetics and release percentage. Release kinetics and cumulative release of the Bhasma ions (Cu and Zn) and NE were found to be faster in the sponge than in the PVA film. The Bhasma ions (Cu and Zn) showed a cumulative release of around 50% for Cu and 60% for Zn loaded in film, while for the sponge, the cumulative release was around 65% for Zn and 70% for Cu ions (Fig. 5(a)). The cumulative release percentages of NE were found to be higher than those of the Bhasma ions, with around 70% cumulative release for the film and 80% for the sponge (Fig. 5(b)). The release for all the bioactive formulations reached saturation after 24 h of incubation with DI water. To obtain the release behavior of the formulations, kinetic models were fitted to cumulative releases of Bhasma ions and NE. As per the reported literature, two widely used models for the release of nanoparticles/drugs from hydrogel and sponge matrix are the Higuchi and Korsmeyer–Peppas models.27–29 The nanoparticles and cinnamon NE release were modeled as per the two models. Based on the R-square values, all the bioactive agents, Zn ions, Cu ions, and cinnamon oil NE, were observed to follow the KP model, with an n value less than 0.45, suggesting the Fickian diffusion-controlled release of these formulations from the PVA film and sponge. | ||
| Fig. 5 Percentage cumulative release kinetics of (a) Tamra and Yasad Bhasma loaded in the PVA film and sponge and (b) NE loaded in the PVA film and sponge. | ||
The release of cinnamaldehyde nanoemulsion (NE) from the PVA matrix is primarily governed by diffusion-driven transport, facilitated by water uptake and polymer chain relaxation. Upon hydration, the porous sponge structure and swollen film matrix create interconnected pathways that allow the gradual migration of nanoemulsion droplets toward the surrounding medium. The higher release from sponges can be attributed to their increased porosity and surface area, which enhance fluid penetration and diffusion rates. The Fickian diffusion behavior (n < 0.45) suggests minimal polymer erosion, indicating controlled release. Sustained liberation of NE maintains prolonged antimicrobial activity, thereby enhancing the therapeutic potency of the developed wound dressing system. A summary of Higuchi and KP model constants and fitted R2 values is shown in Table 1. The fitted curves are shown in the SI (Fig. S3).
| Sample release | Higuchi model (KH and R2 values) | KP model (K and R2 values) |
|---|---|---|
| Zn from PVA film | K H = 16.55; R2 = 0.91 | K = 0.523; R2 = 0.98 |
| Cu from PVA film | K H = 13.38; R2 = 0.83 | K = 0.452; R2 = 0.97 |
| Zn from PVA sponge | K H = 16.83; R2 = 0.83 | K = 0.302; R2 = 0.91 |
| Cu from PVA sponge | K H = 17.67; R2 = 0.86 | K = 0.314; R2 = 0.98 |
| NE from PVA film | K H = 13.74; R2 = 0.73 | K = 0.260; R2 = 0.739 |
| NE from PVA sponge | K H = 15.64; R2 = 0.66 | K = 0.339; R2 = 0.750 |
3.6. Biofunctional assays
Overall, the in vitro studies project that Cu species primarily contribute to angiogenesis and antimicrobial action through redox cycling and ROS-mediated bacterial damage, while Zn phases regulate oxidative balance, enhance keratinocyte proliferation, and support tissue remodelling. This complementary ion activity explains the observed balance between antibacterial efficacy and cytocompatibility. NE release is governed by diffusion through the swollen PVA network and possible transient coordination with metal ions. Its anti-inflammatory and antibiofilm effects synergize with ion-mediated antimicrobial activity, while excessive phytochemical loading may modulate ion-release kinetics. Crosslinking density regulates release behavior, swelling enhances nutrient transport and cell migration, degradation governs the exposure of bioactive phases, and improved mechanical strength preserves scaffold integrity during healing.
In comparison, commercially available wound-healing hydrogels, such as DermaSyn®, Neoheal® Hydrogel, Restore Hydrogel, ActivHeal®, NU-GEL™, Purilon®, and Simpurity™ Hydrogel, primarily rely on hydrated polymeric networks (PEG, PVP, alginate, PVA, hyaluronic acid, or polyurethane) to maintain moisture balance and support autolytic healing. Most of these systems focus on hydration and exudate management rather than sustained antimicrobial delivery.59 In contrast, the developed PVA-TYN film and sponge integrate inorganic bioactives (Tamra and Yasad Bhasma) with cinnamaldehyde nanoemulsion, enabling diffusion-controlled release alongside structural support. This multifunctional design aims to combine moisture retention, antimicrobial potency, and prolonged therapeutic action, thereby addressing limitations of conventional hydrogel dressings that lack controlled bioactive release.
4. Summary and conclusions
The present work demonstrates the synergistic wound-healing capability of Tamra Bhasma and Yasad Bhasma combined with cinnamaldehyde nanoemulsion within PVA-based hydrogel films and sponges. The developed formulations showed sustained, diffusion-controlled release of the bioactive components for over 24 h, supporting prolonged therapeutic action. Compared with conventional commercial hydrogels, such as NU-GEL™, Purilon®, Neoheal® Hydrogel, and Restore Hydrogel, which mainly provide moisture retention and passive protection, the PVA-TYN system offers multifunctional performance through controlled antimicrobial and antioxidant delivery. The composites exhibited strong free-radical scavenging (>80%) and bactericidal efficacy (>90%), highlighting improved infection control potential. Incorporation of Bhasma nanoparticles and nanoemulsion also enhanced the mechanical properties, improving the tensile behavior of the films and the compressive strength of the sponges, thereby overcoming the relatively weak mechanical stability often observed in highly hydrated commercial dressings. In vitro studies with NIH 3T3 fibroblasts and HaCaT keratinocytes demonstrated enhanced cell migration, proliferation, and angiogenic response compared to neat PVA controls, indicating improved regenerative performance. Overall, the indigenous PVA-TYN film and sponge present a multifunctional wound-dressing platform that integrates sustained bioactive release, antioxidant activity, antimicrobial potency, and mechanical robustness, suggesting strong potential for further in vivo evaluation and clinical translation in advanced wound-care applications.Author contributions
Suhela Tyeb: conceptualization, methodology, and writing – original draft. Nitesh Kumar: data curation and resources. Vivek Verma: investigation and resources. Ashok Kumar: resources. Sanjana Koul: writing – reviewing and editing and supervision.Conflicts of interest
There are no conflicts of interest to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). supplementary information: includes Fig. S1 and S2 showing the FTIR spectra of Yasad Bhasma and Tamra Bhasma, respectively, and Fig. S3 showing the fitting of the Higuchi models (left column) and KP models (right column) for the release kinetics of zinc, copper and the nanoemulsion. See DOI: https://doi.org/10.1039/d6ma00091f.Acknowledgements
ST and SK would like to thank the Department of Biotechnology, Government of India, for funding the research work (DBT-RA/2023-24/call-I/RA/18). NK would like to acknowledge IIT Jammu for the characterization facilities, such as the SEM and FTIR instruments. ST would also like to thank Mr G. P Bajpai, Mr Pawan Kumar, the Department of Materials Science and Engineering and the Biosciences and Bioengineering Department, IIT Kanpur, for the laboratory facilities used for this work.References
- S. Tyeb, V. Verma and N. Kumar, Polysaccharide based transdermal patches for chronic wound healing: Recent advances and clinical perspective, Carbohydr. Polym., 2023, 316, 121038, DOI:10.1016/j.carbpol.2023.121038.
- H. Wang, Z. Xu, M. Zhao, G. Liu and J. Wu, Advances of hydrogel dressings in diabetic wounds, Biomater. Sci., 2021, 9, 1530–1546, 10.1039/d0bm01747g.
- W. Zhang, L. Liu, H. Cheng, J. Zhu, X. Li, S. Ye and X. Li, Hydrogel-based dressings designed to facilitate wound healing, Mater. Adv., 2024, 5, 1364–1394, 10.1039/D3MA00682D (accessed November 14, 2025).
- X. Liang, H. J. Zhong, H. Ding, B. Yu, X. Ma, X. Liu, C. M. Chong and J. He, Polyvinyl Alcohol (PVA)-Based Hydrogels: Recent Progress in Fabrication, Properties, and Multifunctional Applications, Polymers, 2024, 16(19), 2755, DOI:10.3390/POLYM16192755.
- W. Ji, M. Mizanur, R. Khan, M. Mahamudul and H. Rumon, Synthesis of PVA-Based Hydrogels for Biomedical Applications: Recent Trends and Advances, Gels, 2025, 11, 88, DOI:10.3390/GELS11020088.
- K. Bialik-Wąs, K. Pluta, D. Malina, M. Barczewski, K. Malarz and A. Mrozek-Wilczkiewicz, Advanced SA/PVA-based hydrogel matrices with prolonged release of Aloe vera as promising wound dressings, Mater. Sci. Eng. C, 2021, 120, 111667, DOI:10.1016/j.msec.2020.111667.
- L. Wang, Y. Yu, X. Zhao, Z. Zhang, X. Yuan, J. Cao, W. Meng, L. Ye, W. Lin and G. Wang, A Biocompatible Self-Powered Piezoelectric Poly(vinyl alcohol)-Based Hydrogel for Diabetic Wound Repair, ACS Appl. Mater. Interfaces, 2022, 14, 46273–46289, DOI:10.1021/ACSAMI.2C13026.
- Z. Wang, S. Gao, W. Zhang, H. Gong, K. Xu, C. Luo, W. Zhi, X. Chen, J. Li and J. Weng, Polyvinyl alcohol/chitosan composite hydrogels with sustained release of traditional Tibetan medicine for promoting chronic diabetic wound healing, Biomater. Sci., 2021, 9, 3821–3829, 10.1039/D1BM00346A.
- F. Ciftci, A. C. Özarslan and N. Evcimen Duygulu, Production and comprehensive characterization of PVA/chitosan transdermal composite mats loaded with bioactive curcumin; evaluation of its release kinetics, antioxidant, antimicrobial, and biocompatibility features, J. Appl. Polym. Sci., 2024, 141, e55874, DOI:10.1002/APP.55874.
- N. Rashid, S. H. Khalid, I. Ullah Khan, Z. Chauhdary, H. Mahmood, A. Saleem, M. Umair and S. Asghar, Curcumin-Loaded Bioactive Polymer Composite Film of PVA/Gelatin/Tannic Acid Downregulates the Pro-inflammatory Cytokines to Expedite Healing of Full-Thickness Wounds, ACS Omega, 2023, 8, 7575–7586, DOI:10.1021/ACSOMEGA.2C07018.
- N. Pattanaik, A. V. Singh, R. S. Pandey, B. K. Singh, M. Kumar, S. K. Dixit and Y. B. Tripathi, Toxicology and free radicals scavenging property of Tamra bhasma, Indian J. Clin. Biochem., 2003, 18, 181, DOI:10.1007/BF02867385.
- P. S. Bafna and S. D. Patil, Physicochemical characterisation and anti-inflammatory activity of ayurvedic herbo-metallic Tamra bhasma in acute and chronic models of inflammation, Mater. Technol., 2018, 33(10), 681–688, DOI:10.1080/10667857.2018.1494241.
- S. Honwad, T. S. Bairy, M. Ravi and B. Ravishankar, Hepatoprotective activity of Somanathi Tamra Bhasma in paracetamol induced liver toxicity in albino- rats, J. Phytopharmacol., 2015, 4, 143–146, DOI:10.31254/PHYTO.2015.4303.
- P. S. Bafna and S. D. Patil, Physicochemical characterisation and anti-inflammatory activity of ayurvedic herbo-metallic Tamra bhasma in acute and chronic models of inflammation, Mater. Technol., 2018, 33, 681–688, DOI:10.1080/10667857.2018.1494241.
- R. K. Singh, S. Kumar, A. K. Aman, S. M. Karim, S. Kumar and M. Kar, Study on physical properties of Ayurvedic nanocrystalline Tamra Bhasma by employing modern scientific tools, J. Ayurveda Integr. Med., 2019, 10, 88–93, DOI:10.1016/j.jaim.2017.06.012.
- D. Durgalakshmi, R. Ajay Rakkesh, T. Bhargavi Ram and S. Balakumar, Comparative studies on Indian traditional nanomedicine Yashadha Bhasma and zinc oxide nanoparticles for anti-diabetic activity, Mater. Res. Express, 2017, 4, 075016, DOI:10.1088/2053-1591/AA7A26.
- A. Pareek and N. Bhatnagar, Physico-chemical characterization of traditionally prepared Yashada bhasma, J. Ayurveda Integr. Med., 2020, 11, 228–235, DOI:10.1016/j.jaim.2018.11.004.
- A. K. Suresh and S. G. Kane, Physiochemical and Biological studies of Jasada Bhasma, a herbo-mineral Indian Traditional Medicine, 2008 Search PubMed.
- C. Shu, L. Ge, Z. Li, B. Chen, S. Liao, L. Lu, Q. Wu, X. Jiang, Y. An, Z. Wang and M. Qu, Antibacterial activity of cinnamon essential oil and its main component of cinnamaldehyde and the underlying mechanism, Front. Pharmacol., 2024, 15, 1378434, DOI:10.3389/FPHAR.2024.1378434/BIBTEX.
- S. G. Seyed Ahmadi, M. R. Farahpour and H. Hamishehkar, Topical application of Cinnamon verum essential oil accelerates infected wound healing process by increasing tissue antioxidant capacity and keratin biosynthesis, Kaohsiung J. Med. Sci., 2019, 35, 686–694, DOI:10.1002/KJM2.12120.
- H. Moustafa, H. E. Nasr and A. M. Youssef, Development of antibacterial carboxymethyl cellulose/quaternized starch bionanocomposites based on cinnamon essential oil nanoemulsion for wound healing applications, Biomass Convers. Biorefin., 2024, 14, 27477–27489, DOI:10.1007/S13399-022-03403-2/TABLES/2.
- S. Tyeb, P. A. Shiekh, V. Verma and A. Kumar, Adipose-Derived Stem Cells (ADSCs) Loaded Gelatin-Sericin-Laminin Cryogels for Tissue Regeneration in Diabetic Wounds, Biomacromolecules, 2020, 21, 294–304, DOI:10.1021/acs.biomac.9b01355.
- A. Dhillon, S. Sardana and A. R. Thakkar, Development and Validation of HPLC and UV Spectrophotometric Method for the Quantification of Cinnamaldehyde in Cinnamon Bark Extract, J. Nat. Rem., 2023, 23, 111–119, DOI:10.18311/JNR/2023/30836.
- S. Tyeb, N. Kumar, A. Kumar and V. Verma, Flexible agar-sericin hydrogel film dressing for chronic wounds, Carbohy. Polym., 2018, 200, 572–582, DOI:10.1016/j.carbpol.2018.08.030.
- J. Stephen-Haynes, Exudate management to promote healing, Nursing and Residential Care, 2015, 17, 322–326, DOI:10.12968/NREC.2015.17.6.322.
- A. Punjataewakupt and P. Aramwit, Wound dressing adherence: a review, J. Wound Care, 2022, 31, 406–423, DOI:10.12968/JOWC.2022.31.5.406.
- Y. Herdiana, N. Wathoni, S. Shamsuddin and M. Muchtaridi, Drug release study of the chitosan-based nanoparticles, Heliyon, 2022, 8, e08674, DOI:10.1016/j.heliyon.2021.e08674.
- M. Mojahedi, P. Heydari and A. Z. Kharazi, Preparation and characterization of an antibacterial CMC/PCL hydrogel films containing CIP/Cur: In vitro and in vivo evaluation of wound healing activity, Int. J. Biol. Macromol., 2024, 282, 136570, DOI:10.1016/j.ijbiomac.2024.136570.
- J. Emami, N. Mostolizadeh, M. Tabbakhian, P. Heydari, A. Z. Kharazi, M. Minaeiyan, F. Hasanzadeh, M. Mirian and A. Talebi, Development and evaluation of gelatin/hyaluronic acid nanofibrous dressing loaded with silver nanoparticles and phenytoin for enhanced wound healing: an in-vitro and in-vivo study, Res. Pharm. Sci., 2025, 20, 610, DOI:10.4103/rps.rps_60_25.
- N. K. R. Gadaime, R. B. Rabiatul, G. A. Govindasamy, Y. Bustami and S. Sreekantan, Advanced Hydrogel Dressing with Zinc Oxide-Copper Oxide Nanocomposite for Effective Wound Management: Mechanochemistry, Antibacterial Efficacy, Cytocompatibility and Wound Healing Potentials, J. Polym. Environ., 2025, 33, 1601–1614, DOI:10.1007/S10924-024-03468-2.
- Y. Yang Niu, Y. Ying Zhang, Z. Zhu, X. Qin Zhang, X. Liu, S. Ya Zhu, Y. Song, X. Jin, B. Lindholm and C. Yu, Elevated intracellular copper contributes a unique role to kidney fibrosis by lysyl oxidase mediated matrix crosslinking, Cell Death Dis., 2020, 11, 211, DOI:10.1038/S41419-020-2404-5.
- G. Borkow, Using Copper to Improve the Well-Being of the Skin, Curr. Chem. Biol., 2014, 8, 89, DOI:10.2174/2212796809666150227223857.
- Y. Qiu, Y. Gao, D. Yu, L. Zhong, W. Cai, J. Ji, F. Geng, G. Tang, H. Zhang, J. Cao, J. Zhang and S. Zhang, Genome-Wide Analysis Reveals Zinc Transporter ZIP9 Regulated by DNA Methylation Promotes Radiation-Induced Skin Fibrosis via the TGF-β Signaling Pathway, J. Invest. Dermatol., 2020, 140, 94–102.e7, DOI:10.1016/j.jid.2019.04.027.
- D. Beyersmann and H. Haase, Functions of zinc in signaling, proliferation and differentiation of mammalian cells, Biometals, 2001, 14, 331–341, DOI:10.1023/A:1012905406548.
- X. Yuan, L. Han, P. Fu, H. Zeng, C. Lv, W. Chang, R. S. Runyon, M. Ishii, L. Han, K. Liu, T. Fan, W. Zhang and R. Liu, Cinnamaldehyde accelerates wound healing by promoting angiogenesis via up-regulation of PI3K and MAPK signaling pathways, Lab. Invest., 2018, 98, 783–793, DOI:10.1038/s41374-018-0025-8.
- J. Sankaranarayanan, S. C. Lee, H. K. Kim, J. Y. Kang, S. S. Kuppa and J. K. Seon, Cinnamaldehyde-Mediated Suppression of MMP-13, COX-2, and IL-6 Through MAPK and NF-κB Signaling Inhibition in Chondrocytes and Synoviocytes Under Inflammatory Conditions, Int. J. Mol. Sci., 2024, 25, 12914, DOI:10.3390/IJMS252312914.
- W. Feng, F. Ye, W. Xue, Z. Zhou and Y. J. Kang, Copper Regulation of Hypoxia-Inducible Factor-1 Activity, Mol. Pharmacol., 2008, 75, 174, DOI:10.1124/MOL.108.051516.
- D. C. Rigiracciolo, A. Scarpelli, R. Lappano, A. Pisano, M. F. Santolla, P. De Marco, F. Cirillo, A. R. Cappello, V. Dolce, A. Belfiore, M. Maggiolini and E. M. De Francesco, Copper activates HIF-1α/GPER/VEGF signalling in cancer cells, Oncotarget, 2015, 6, 34158, DOI:10.18632/ONCOTARGET.5779.
- I. Tenaud, S. Leroy, N. Chebassier and B. Dreno, Zinc, copper and manganese enhanced keratinocyte migration through a functional modulation of keratinocyte integrins, Exp. Dermatol., 2000, 9, 407–416, DOI:10.1034/J.1600-0625.2000.009006407.x.
- A. B. G. Lansdown, U. Mirastschijski, N. Stubbs, E. Scanlon and M. S. Ågren, Zinc in wound healing: Theoretical, experimental, and clinical aspects, Wound Repair Regen., 2007, 15, 2–16, DOI:10.1111/j.1524-475x.2006.00179.x.
- R. Nosrati, S. Kheirouri, R. Ghodsi and H. Ojaghi, The effects of zinc treatment on matrix metalloproteinases: A systematic review, J. Trace Elem. Med. Biol., 2019, 56, 107–115, DOI:10.1016/j.jtemb.2019.08.001.
- R. Nosrati, S. Kheirouri, R. Ghodsi and H. Ojaghi, The effects of zinc treatment on matrix metalloproteinases: A systematic review, J. Trace Elem. Med. Biol., 2019, 56, 107–115, DOI:10.1016/j.jtemb.2019.08.001.
- H. Sharir, A. Zinger, A. Nevo, I. Sekler and M. Hershfinkel, Zinc Released from Injured Cells Is Acting via the Zn2 + -sensing Receptor, ZnR, to Trigger Signaling Leading to Epithelial Repair, J. Biol. Chem., 2010, 285, 26097, DOI:10.1074/jbc.m110.107490.
- P. H. Lin, M. Sermersheim, H. Li, P. H. U. Lee, S. M. Steinberg and J. Ma, Zinc in Wound Healing Modulation, Nutrients, 2017, 10, 16, DOI:10.3390/NU10010016.
- A. B. G. Lansdown, U. Mirastschijski, N. Stubbs, E. Scanlon and M. S. Ågren, Zinc in wound healing: Theoretical, experimental, and clinical aspects, Wound Repair Regen., 2007, 15, 2–16, DOI:10.1111/J.1524-475x.2006.00179.x.
- F. Kermani, S. Nazarnezhad, Z. Mollaei, S. Mollazadeh, A. Ebrahimzadeh-Bideskan, V. R. Askari, R. K. Oskuee, A. Moradi, S. A. Hosseini, Z. Azari, F. Baino and S. Kargozar, Zinc- and Copper-Doped Mesoporous Borate Bioactive Glasses: Promising Additives for Potential Use in Skin Wound Healing Applications, Int. J. Mol. Sci., 2023, 24, 1304, DOI:10.3390/IJMS24021304.
- B. M. Elmowafy, R. M. Ramadan, A. R. Ghazy, I. S. Fahim, T. M. Meaz, R. Ghazy and A. M. Abdelghany, Effect of copper and zinc oxide doping in 13–93B3 borate glass for enhanced wound healing, Ceram. Int., 2025, 51, 34650–34662, DOI:10.1016/j.ceramint.2025.05.188.
- D. C. Rigiracciolo, A. Scarpelli, R. Lappano, A. Pisano, M. F. Santolla, P. De Marco, F. Cirillo, A. R. Cappello, V. Dolce, A. Belfiore, M. Maggiolini and E. M. De Francesco, Copper activates HIF-1α/GPER/VEGF signalling in cancer cells, Oncotarget, 2015, 6, 34158, DOI:10.18632/oncotarget.5779.
- E. Bosch-Rué, L. Díez-Tercero, R. Rodríguez-Gonzá lez, B. M. Bosch-Canals and R. A. Perez, Assessing the potential role of copper and cobalt in stimulating angiogenesis for tissue regeneration, PLoS One, 2021, 16, e0259125, DOI:10.1371/journal.pone.0259125.
- J. Zhao, J. Du, Y. Zheng, Y. Fang, J. Huang, J. Wu, Y. Wu and T. Nie, Zinc ions coordinated carboxymethyl chitosan hydrogel doped with ellagic acid for accelerative diabetic wound healing, Int. J. Biol. Macromol., 2025, 327, 147372, DOI:10.1016/j.ijbiomac.2025.147372.
- C. Zhao, J. Yang, W. Chen, C. Lu, Z. Zeng, T. Jiang and W. Liu, Gelatin/Dopamine/Zinc-Doped Ceria/Curcumin nanocomposite hydrogels for repair of chronic refractory wounds, Int. J. Pharm., 2024, 663, 124575, DOI:10.1016/j.ijpharm.2024.124575.
- X. Yuan, L. Han, P. Fu, H. Zeng, C. Lv, W. Chang, R. S. Runyon, M. Ishii, L. Han, K. Liu, T. Fan, W. Zhang and R. Liu, Cinnamaldehyde accelerates wound healing by promoting angiogenesis via up-regulation of PI3K and MAPK signaling pathways, Lab. Invest., 2018, 98, 783–798, DOI:10.1038/S41374-018-0025-8.
- D. Liang, M. Yang, B. Guo, J. Cao, L. Yang, X. Guo, Y. Li and Z. Gao, Zinc Inhibits H2O2-Induced MC3T3-E1 Cells Apoptosis via MAPK and PI3K/AKT Pathways, Biol. Trace Elem. Res., 2012, 148(3), 420–429, DOI:10.1007/S12011-012-9387-8.
- Y. Luo, H. Zhang, Z. Wang, J. Jiao, Y. Wang, W. Jiang, T. Yu, H. Liu, L. Guan, M. Li and M. Wu, Strategic incorporation of metal ions in bone regenerative scaffolds: multifunctional platforms for advancing osteogenesis, Regen. Biomater., 2025, 12, rbaf068, DOI:10.1093/RB/RBAF068.
- E. Urso and M. Maffia, Behind the Link between Copper and Angiogenesis: Established Mechanisms and an Overview on the Role of Vascular Copper Transport Systems, J. Vasc. Res., 2016, 52, 172–196, DOI:10.1159/000438485.
- Q. Wang, Q. Li, L. Zhu, C. Lin, Q. Chen and H. Chen, Fabrication of Cu/ZnO-loaded chitosan hydrogel for an effective wound dressing material to advanced wound care and healing efficiency after caesarean section surgery, Int. Wound J, 2024, 21, e14366, DOI:10.1111/IWJ.14366.
- J. C. Palmaz, A. Benson and E. A. Sprague, Influence of surface topography on endothelialization of intravascular metallic material, J. Vasc. Interv. Radiol., 1999, 10, 439–444, DOI:10.1016/S1051-0443(99)70063-1.
- A. V. Kozlov, S. Javadov and N. Sommer, Cellular ROS and Antioxidants: Physiological and Pathological Role, Antioxidants, 2024, 13, 602, DOI:10.3390/ANTIOX13050602.
- V. Gounden and M. Singh, Hydrogels and Wound Healing: Current and Future Prospects, Gels, 2024, 10, 43, DOI:10.3390/gels10010043.
| This journal is © The Royal Society of Chemistry 2026 |









