Open Access Article
Open Access ArticleBi3+-sensitized up-conversion luminescence and non-contact optical thermometry in NaCaYV2O8:Er3+/Yb3+ phosphors
Fadwa
Ayachi
a,
Kamel
Saidi
 ab,
I.
Mediavilla-Martínez
c,
Mohamed
Dammak
ab,
I.
Mediavilla-Martínez
c,
Mohamed
Dammak
 *a and
J.
Jimenez
c
*a and
J.
Jimenez
c
aLaboratoire de Physique Appliquée, Faculté des Sciences de Sfax, Département de Physique, Université de Sfax, BP 1171, Sfax, Tunisia. E-mail: madidammak@yahoo.fr; Mohamed.dammak@fss.usf.tn
bDepartment of Physics, Sfax Preparatory Engineering Institute, University of Sfax, 1172- 3000, Sfax, Tunisia
cGdS Optronlab, Department of Condensed Matter Physics, LUCIA Building University of Valladolid, Paseo de Belen 19, 47011, Valladolid, Spain
First published on 20th February 2026
Abstract
Tri-doped NaCaYV2O8 phosphors incorporating Er3+, Yb3+, and variable Bi3+ concentrations (0–0.15 mol%) were synthesized via a sol–gel route. Phase purity and particle morphology were verified through X-ray diffraction (XRD) and Transmission electron microscopy (TEM). Optical spectroscopy revealed that Bi3+ incorporation mediates defect-state-induced band gap contraction (from 3.2 to 3.05 eV), facilitating enhanced energy transfer coupling within the Er3+/Yb3+ system. Under 980 nm excitation, pronounced green up conversion signals emerged at 525 and 552 nm, attributed to the 2H11/2 → 4I15/2 and 4S3/2 → 4I15/2 transitions of Er3+. Photoluminescence intensity scaled monotonically with Bi3+ content up to an optimum at 0.07 mol, beyond which non-radiative losses dominated. Temperature-dependent up conversion spectroscopy (298–513 K) demonstrated that fluorescence intensity ratios (FIR) derived from the thermally coupled 2H11/2 and 4S3/2 manifolds enabled reliable ratiometric thermometry independent of excitation fluctuations. The 7 mol% Bi3+-doped composition exhibited the highest absolute (Sa) and relative (Sr) thermal sensitivities, establishing this composition as optimal for simultaneous up conversion brightness and temperature-sensing performance. The findings underscore the strategic value of bismuth co-doping in vanadate hosts for synergistic enhancement of both luminescence efficiency and metrological precision.
1. Introduction
Lanthanide-doped up conversion (UC) materials have garnered significant attention in recent years due to their versatility in frontier applications ranging from photothermal therapy and bioimaging to contactless temperature sensing.1–4 The distinct optical signatures of these phosphors arise from the rich electronic structure of trivalent rare-earth (RE3+) ions, which possess abundant metastable energy levels that facilitate efficient photon absorption and emission.5,6 Among these applications, optical thermometry based on thermally coupled levels (TCLs) is scientifically paramount as it allows for precise thermal reading without physical contact. This mechanism is particularly effective in Er3+ ions, where the 2H11/2 and 4S3/2 excited states are separated by an energy gap of approximately 800 cm−1, allowing for population redistribution governed by Boltzmann statistics and mediated by host lattice phonons.7–14Despite the promise of these materials, enhancing their emission efficiency remains a critical challenge. One effective strategy involves modifying the local crystal field by introducing metal cations (e.g., Li+, Zn2+, Mg2+), which has been shown to modulate the host environment and improve UC performance.15 More recently, the incorporation of Bismuth (Bi3+) as a sensitizer has emerged as a powerful approach. Optimal Bi3+ doping can significantly amplify UC emission intensity of RE3+ systems through defect engineering and energy transfer modification.16 While the benefits of Bi3+ are known in other hosts, the specific impact of Bi3+ co-doping on the optical and thermal properties of the NaCaYV2O8:Er3+/Yb3+ system remains unexplored. This investigation is vital because the Fluorescence Intensity Ratio (FIR) technique relies heavily on optimizing host-dopant interactions to achieve the high accuracy and resolution required for practical non-contact thermometry.17,18 Consequently, maximizing the thermal sensitivity of Er3+/Yb3+ codoped phosphors continues to be a priority in contactless thermometry research.19,20
In this work, we report the synthesis of NaCaYV2O8 (NCYVO) phosphors co-doped with Er3+/Yb3+, with varying concentrations of Bi3+via a sol–gel method. Structural and morphological properties were characterized using X-ray diffraction (XRD), while the role of Bi3+ in modulating the UC emission intensity was systematically investigated. Furthermore, detailed temperature-dependent luminescence measurements were conducted to evaluate the UC mechanism and determine the suitability of these phosphors for high-sensitivity optical thermometry applications.
2. Experimental section
2.1. Synthesis
A series of Bi3+ co-doped NCYVO:0.02Er3+/0.10Yb3+ phosphors with Bi3+ concentrations (x = 0, 5, 7, 10 and 15) were prepared via a citrate-assisted sol–gel route. High-purity reagents were used as received from Sigma-Aldrich without further purification. The precursor materials comprised calcium nitrate [Ca(NO3)2, 99.9%], ammonium metavanadate [NH4VO3, 99.96%], and the rare-earth/metal nitrates: Y(NO3)3·6H2O (99.9%), Yb(NO3)3·5H2O (99.9%), Er(NO3)3·5H2O (99.9%), and Bi(NO3)3·5H2O (99.9%). Citric acid [C6H8O7, 99.0%] served as the chelating agent. In a typical synthesis, stoichiometric amounts of the metal precursors were dissolved in 120 mL of deionized water in a 250 mL beaker. The mixture was stirred continuously at ambient temperature until a transparent, homogeneous solution was achieved. Citric acid was then added in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio to the total metal ions, and the pH was adjusted to ∼7 using ammonium hydroxide under vigorous stirring to form the citrate-metal complexes. The solution was evaporated at 80 °C on a hotplate with continuous stirring until a viscous gel formed, followed by drying in an oven at 120 °C for 12 h to obtain a dry xerogel. The xerogel was ground into a fine powder and pre-calcined at 500 °C for 4 h in air to decompose organic residues. Finally, the powder was pelletized, annealed at 900 °C for 6 h in air using a muffle furnace with a heating rate of 5 °C min−1, and slowly cooled to room temperature to yield the crystalline phosphor samples.
1 molar ratio to the total metal ions, and the pH was adjusted to ∼7 using ammonium hydroxide under vigorous stirring to form the citrate-metal complexes. The solution was evaporated at 80 °C on a hotplate with continuous stirring until a viscous gel formed, followed by drying in an oven at 120 °C for 12 h to obtain a dry xerogel. The xerogel was ground into a fine powder and pre-calcined at 500 °C for 4 h in air to decompose organic residues. Finally, the powder was pelletized, annealed at 900 °C for 6 h in air using a muffle furnace with a heating rate of 5 °C min−1, and slowly cooled to room temperature to yield the crystalline phosphor samples.
2.2. Characterization
The crystal structure and phase purity of the synthesized phosphors were characterized by powder X-ray diffraction (XRD) using a Rigaku D-Max 2500 diffractometer equipped with graphite-monochromatized Cu Kα radiation (λ = 1.5406 Å). Diffraction patterns were collected over a 2θ range of 10°–80° at a scanning speed of 15° min−1. Photoluminescence (PL) spectra were recorded using a PerkinElmer spectrophotometer to evaluate the up-conversion emission properties of the phosphors.3. Results and discussions
3.1. XRD
The phase purity and structural evolution of NaCaYV2O8:0.02Er3+/0.10Yb3+/xBi3+ phosphors (x = 0, 0.05, 0.07, 0.10, 0.15 mol%) were examined using X-ray diffraction, as shown in Fig. 1(a). All diffraction patterns exhibit well-defined peaks that can be fully indexed to the standard monoclinic NaCaYV2O8 phase (JCPDS No. 96-152-7822), confirming the formation of a single-phase material across the entire Bi3+ doping range. No additional diffraction peaks corresponding to secondary phases or impurity compounds are detected, indicating that Bi3+ ions are successfully incorporated into the NaCaYV2O8 host lattice without altering its crystal structure. The mean crystallite size (DW–H) and lattice strain (ε) of all samples were determined from the Bragg peak positions and their full width at half maximum (β) by applying the Williamson–Hall method.21here, θ represents the diffraction angle corresponding to the most intense peak, K is the shape factor taken as 0.9, and λ denotes the wavelength of the Cu Kα radiation (λ = 0.154 nm). The variation observed in the Williamson–Hall plots presented in Fig. 1(b) allows the determination of the crystallite size (DW–H) and lattice strain (ε) for the synthesized samples, as summarized in Table S1. Based on the Williamson–Hall analysis, the 0.07 mol% Bi3+-doped phosphor exhibits the lowest lattice strain, indicating superior structural quality compared to the other compositions.
Rietveld refinement was performed on the representative diffraction profiles in order to quantify the structural parameters (Fig. S1). The refined crystallographic data, including lattice constants and reliability factors, are compiled in Table S1. The low residual values (Rwp, and R2) accounts for the high quality of the fit and the purity of the as-synthesized samples. The unit cell volume exhibits a consistent increase with rising Bi3+ content, as shown in Table S1, which serves as direct confirmation of the substitution of Y3+ by the larger Bi3+ ion. This expansion aligns with Vegard's law, thereby supporting the notion of Bi3+ occupancy at the Y3+ site; the ionic radius of Bi3+ (117 pm, CN = 8) is approximately 15% larger than that of Y3+ (101.9 pm, CN = 8). While Na+ (118 pm) and Ca2+ (112 pm) possess similar radii, substitution at these sites would necessitate charge compensation due to the trivalent character of Bi3+, a process that is energetically unfavorable in the absence of co-dopants or vacancies. Conversely, the isovalent substitution of Y3+ with Bi3+ circumvents these issues, thereby designating the Y-site as the most likely location for incorporation. This observation supports the effective integration of Bi3+ within the cationic sublattice of the NaCaYV2O8 host material.
3.2. TEM analysis
The microstructural evolution of the NCYVO:0.02Er3+/0.10Yb3+/xBi3+ phosphors (x = 0 and 0.07 mol) was examined by Transmission electron microscopy TEM, as shown in Fig. 2(a and b) and TEM micrographs of Bi3+-doped samples with higher dopant concentrations: (a) 0.05 Bi3+, (b) 0.1 Bi3+, and (c) 0.15 Bi3+ are provided in the Fig. S2. | ||
| Fig. 2 Transmission electron microscopy (TEM) images illustrating the surface morphology of NaCaYV2O8:0.02Er3+/0.10Yb3+ phosphors with different Bi3+concentrations: (a) 0 mol, (b) 0.07 mol%. | ||
All samples exhibit increased particle agglomeration and irregular morphologies compared to the undoped material, consistent with the trend observed for the 7% Bi3+-doped sample in the main text. The scale bar in all images is 200 nm. Fig. 2(a) shows well-defined, faceted nanoparticles corresponding to the undoped sample (0 Bi3+), where the particles are relatively uniform in size and loosely aggregated, indicating good dispersion and stable crystal growth. In contrast, Fig. 2(b) represents the sample doped with 0.07 Bi3+, which exhibits a more compact and irregular morphology with pronounced agglomeration and less distinguishable particle boundaries. The increased aggregation and morphological distortion observed in the Bi3+-doped sample suggest that higher Bi3+ incorporation affects the crystal growth process, likely by introducing lattice distortion and enhanced interparticle interactions. Overall, Bi3+ doping at 0.07 leads to reduced particle uniformity and increased agglomeration compared to the undoped material.
3.3. Optical studies
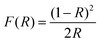 | (1) |
R represents the reflectance.
The optical band gap (Eg) of the compound was determined using the Tauc relation.
| (F(R) × hϑ)n = B(hϑ − Eg) | (2) |
The diffuse reflectance spectra (DRS) of the Bi3+ co-doped NCYVO:Er3+/Yb3+ phosphors were recorded in the 200–1100 nm range, as presented in Fig. 3. The spectra are dominated by a strong ultraviolet absorption band between 200–350 nm, corresponding to the O2− to V5+ charge transfer (CT) transition within the VO43− tetrahedral groups. Superimposed on this host absorption are sharp, discrete peaks in the visible and near infrared regions, which are assigned to the characteristic 4f–4f intra-configurational transitions of Er3+ ions.24 Notably, the broad absorption centered at 980 nm arises from the 2F7/2 to 2F5/2 transition of Yb3+, which is critical for efficient photon harvesting and subsequent energy transfer to Er3+ activators.25,26 Upon Bi3+ doping, a new absorption feature emerges at 353 nm, attributed to the 1S0 to 3P1 transition of Bi3+ ions, confirming their successful incorporation into the lattice.27 Furthermore, the absorption edge exhibits a progressive redshift with increasing Bi3+ concentration. As shown in Fig. S3, the optical band gap (Eg) narrows monotonically from 3.2 eV for the Bi-free sample to 3.05 eV for the 0.15 mol Bi3+ sample. This band gap contraction is ascribed to the hybridization of the Bi3+ 6s2 orbitals with the O–2p valence bands, which elevates the valence band maximum and introduces localized defect states within the forbidden gap.16,28 These intermediate states effectively broaden the absorption cross-section and may serve as bridges for energy transfer, thereby modulating the optical performance of the phosphor.21,22
 | ||
| Fig. 4 Up conversion (UC) emission spectra of the NCYVO:0.02Er3+/0.10 Yb3+ phosphors doped with various Bi3+ concentrations (x = 0–0.15 mol) under 980 nm excitation. | ||
As illustrated in Fig. 4, the UC emission intensity exhibits a strong dependence on Bi3+ concentration. The integrated green luminescence intensity enhances monotonically with increasing Bi3+ content, reaching a maximum at 7 mol%. This enhancement is attributed to the Bi3+-induced modification of the local crystal field symmetry around the Er3+/Yb3+ ions, which relaxes the selection rules for 4f–4f transitions, and the defect-mediated energy transfer pathways established by the narrowed band gap. However, for doping levels exceeding 0.07 mol a systematic decline in intensity is observed.
The emission spectra for each sample at different pump powers are studied without saturation at high powers. The emission intensity vs. the pump power will follow the relation:
| I ∝ (P)n | (3) |
Based on these results and the simplified energy level diagram (Fig. S3(f)), the proposed UC mechanism involves a synergistic interplay of Ground State Absorption (GSA), Energy Transfer Up conversion (ETU), and Cooperative Energy Transfer (CET). Upon 980 nm excitation, Yb3+ ions (sensitizers) efficiently absorb incident photons via the 2F7/2 → 2F5/2 transition due to their large absorption cross-section. Subsequently, excited Yb3+ transfers energy to a proximal Er3+ ion, promoting it from the ground state 4I15/2 to the intermediate 4I11/2 level. A second energy transfer event from another excited Yb3+ ion (or the absorption of a second photon by an already excited Er3+ ion via GSA/ESA) further excites the Er3+ population to the higher-lying 4F7/2 manifold. Additionally, Cooperative Energy Transfer (CET) may occur, where two excited Yb3+ ions simultaneously transfer energy to a single Er3+ ion, directly bridging the gap to the 4F7/2 state. Following rapid non-radiative relaxation to the metastable 2H11/2 and 4S3/2 levels, the ions decay radiatively to the ground state, generating the characteristic green emissions at 525 nm and 552 nm, respectively.30
 | ||
| Fig. 5 Temperature-dependent up conversion (UC) emission spectra of (a) NCYVO:Er3+/Yb3+/0Bi3+ and (b) NCYVO:Er3+/Yb3+/0.05 Bi3+ under 980 nm excitation. | ||
This redistribution follows Boltzmann statistics, allowing the fluorescence intensity ratio (FIR) to be defined as a function of absolute temperature T according to eqn (4):33,34
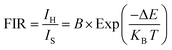 | (4) |
Fig. 6(a–e) show that the fluorescence intensity ratios (FIR) between the thermally coupled 2H11/2 and 4S3/2 levels increase non-linearly for the Er3+/Yb3+/xBi3+ tri-doped NCYVO phosphor samples (x = 0, 0.05, 0.07, 0.10, and 0.15) from 298 to 513 K. The (ΔE/KB) values obtained from the fitting results for all samples are summarized in Table 1. The temperature sensitivity of the phosphor samples is a critical factor in the development of highly responsive optical temperature sensors. Among the key indicators used to evaluate and compare sensor performance are the absolute sensitivity (Sa) and relative sensitivity (Sr). These parameters offer meaningful insights into the sensing efficiency of the material and can be calculated using the following equations eqn (5) and (6), respectively.35–37
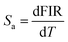 | (5) |
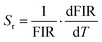 | (6) |
| Bi3+ concentration | ΔE/KB (K) |
S
a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) max max
|
S r (300 K) | σ = δFIR/FIR |
|---|---|---|---|---|
| 0 | 858.35 | 0.00540 (430 K) | 0.75% | 0.0012 |
| 0.05 | 875.01 | 0.004 (440 K) | 0.48% | 0.0013 |
| 0.07 | 980.244 | 0.005 (480 K) | 0.65% | 0.0034 |
| 0.1 | 987.02 | 0.00515 (470 K) | 0.63% | 0.0551 |
| 0.15 | 1008.818 | 0.005 (513 K) | 0.5% | 0.0065 |
Fig. 7(a–e) present the temperature-dependent evolution of the absolute sensitivity Sa for the different Bi3+ concentrations in NCYVO:Er3+/Yb3+/xBi3+. The values of Sa were obtained from the FIR–temperature relationship using eqn (5). Sa increases steadily with temperature and reaches a maximum at a characteristic temperature Tmax specific to each composition. These maximum values are listed in Table 2. A clear decline in the peak Sa is observed as the Bi3+ concentration increases, indicating that the sample without Bi3+ (x = 0) exhibits the highest absolute sensitivity, approximately 5.4 × 10−3 K−1 at 430 K.
| Sensing materials | Temperature range (K) | Excitation (nm) |
S
a![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) max (K−1) max (K−1) |
S r (%K−1) | Ref. |
|---|---|---|---|---|---|
| NaErF4@NaYF4@NaYbF4:Tm@NaYF4 NPs | 293–413 | 980 | — | 0.71 (315 K) | 43 |
| GdPVO4:2%Er3+/15%Yb3+ | 300–440 | 980 | 0.00507 (320 K) | 0.63 (300 K) | 44 |
| LaNbO4:1%Er3+/10%Yb3+ | 10–300 | 980 | — | 0.749 (300 K) | 45 |
| CaLa2ZnO5:Er3+/Yb3+ | 298–513 | 980 | — | 0.59 (513 K) | 46 |
| Tellurite glass:Yb3+, Er3+ | 298–473 | 980 | — | 0.53 (298 K) | 47 |
| K3Y(PO4)2:Yb3+/Ho3+ | 303–523 | 980 | — | 0.2 (303 K) | 48 |
| NaCaYV2O8:Er3+/Yb3+/0.07Bi3+ | 298–513 | 980 | 0.0051 (430 K) | 0.65 (300 K) | This work |
This behavior indicates that the variation of Sa is primarily governed by the pre-exponential factor B in the FIR expression, whereas the energy gap ΔE between the thermally coupled levels mainly determines the optimal operating temperature range. Within the Judd–Ofelt formalism, the parameter B can be expressed in terms of the intensity parameters Ωλ (λ = 2,4,6), which reflect the local crystal-field symmetry and the degree of covalency surrounding the Er3+ ions. In practice, these Ωλ values and thus B can be derived from room-temperature absorption or emission spectra, or extracted from literature for Er3+ in structurally similar hosts, providing a predictive framework for estimating Sa without time consuming temperature-dependent measurements.
For the FIR involving the transitions 2H11/2 → 4I15/2 and 4S3/2 → 4I15/2, B is approximated using eqn (7), which relates it to Ω2, Ω4, Ω6, and the corresponding reduced matrix elements. Among these parameters, Ω2 is particularly sensitive to local structural modifications and bonding characteristics, while Ω4 and Ω6 remain comparatively stable and are primarily dictated by the host lattice. Therefore, in the NCYVO system, Ω2 appears to be the dominant Judd–Ofelt parameter influencing the evolution of B, and consequently the absolute sensitivity Sa.38
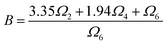 | (7) |
The variation of relative sensitivity (Sr), a critical figure of merit for comparing sensors across different temperature ranges, was calculated using eqn (6) and is presented in Fig. 7. Unlike absolute sensitivity, Sr typically follows a 1/T2 dependence. The maximum Sr values, obtained at 300 K, are listed in Table 1. Remarkably, while Bi3+ doping modulates the absolute sensitivity, the relative sensitivity remains robust, ensuring high precision for physiological temperature sensing.
To benchmark the performance of the NCYVO:Er3+/Yb3+/xBi3+ system, the obtained Sa and Sr values were compared with those of other Er3+-based optical thermometers reported in the literature (Table 2). The sensitivity values achieved in this work even for the Bi-doped samples exceed those of many established hosts (e.g., fluorides, oxides, and other vanadates). This superior performance, combined with the significant enhancement in emission intensity provided by Bi3+ sensitization, positions the 0.07 Bi3+-doped NCYVO phosphor as a highly promising candidate for practical, high-sensitivity optical thermometry applications.
Thermal resolution (δT) is another parameter used to evaluate the performance of the proposed high-temperature sensor. It quantifies the minimum temperature difference that the sensor can reliably detect, thus reflecting its ability to discern small changes in temperature with precision. A lower δT value indicates a higher sensitivity of the sensor to temperature variations. This parameter was calculated using the following equation, as presented in eqn (8).39,40
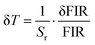 | (8) |
The parameter δFIR defines the experimental uncertainty of the thermometric signal, effectively setting the threshold for the smallest detectable change in the fluorescence ratio. While this limit is intrinsic to the sensor's performance, it is also heavily dependent on the instrumentation and environmental conditions. To minimize this uncertainty and enhance the resolution, we optimized the data acquisition protocol by increasing the signal integration time and averaging consecutive spectral scans to suppress random experimental noise.
To quantify the detection limit, a statistical analysis was performed using 50 consecutive measurements taken at room temperature for the NCYVO:Er/Yb/x%Bi samples (x = 0, 5, 7, 10, 15). The distribution of these measurements is visualized in the histograms in Fig. S4(a–e). The relative uncertainty of the signal, defined as δFIR/FIR, was derived from the standard deviation of these distributions. The calculated values for all doping concentrations are cataloged in Table 2. This metric serves as a direct indicator of measurement precision and system stability.
Furthermore, Fig. 7(f) illustrates the evolution of the thermal resolution δT as a function of temperature for the NCYVO:Er/Yb/xBi samples (x = 0, 0.05, 0.07, 0.10, 0.15). The calculated max values δTmax for all the doping concentrations are listed in Table 2. The data indicate that the thermal resolution improved with doping, yielding smaller δT as the temperature increased. This trend underscores the sensor's enhanced capability to discriminate minute temperature fluctuations in the higher temperature regimes across the investigated samples.
The estimated thermometric parameters (FIR values) were carefully measured during multiple cycles of the sample between ambient temperature (298 K) and increased temperatures (513 K), as shown in Fig. 8, in order to verify the accuracy of the used temperature sensing methods. The following formula was used to determine the repeatability(R):41,42
4. Conclusions
In summary, Bi3+-sensitized NaCaYV2O8:Er3+/Yb3+ up conversion phosphors were successfully synthesized via a sol–gel method, their structural, optical, and thermometric properties were systematically investigated. Structural analysis confirmed the formation of a single-phase solid solution, where Bi3+ incorporation induces a controlled reduction in the optical band gap (from 3.2 to 3.05 eV) via spin–orbit hybridization. This electronic structure modulation, combined with local symmetry distortion, facilitates efficient energy transfer and relaxes the selection rules for 4f–4f transitions, leading to a significant enhancement in the green up conversion emission intensity.The optimal doping concentration was identified as 0.07 mol Bi3+, which balances the emission brightness with concentration quenching effects. Temperature-dependent luminescence studies (298–513 K) demonstrated that the fluorescence intensity ratio (FIR) of the thermally coupled Er3+ levels 2H11/2 → 4S3/2 enables reliable non-contact thermometry. Although Bi3+ doping resulted in a trade-off with absolute sensitivity (Sa), the optimized phosphor retained high relative sensitivity (Sr) and excellent thermal resolution δT, superior to many existing vanadate-based sensors. These results establish the 0.02Er3+/0.10Yb3+/0.07Bi3+ tri-doped NCYVO phosphor as a robust, multifunctional material suitable for advanced applications in optical thermometry, photonic sensing, and optoelectronics.
Conflicts of interest
There are no conflicts to declare.Data availability
All data underlying the results are available as part of the article and no additional source data are required.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d6ma00035e.
References
- Z. Zhang, D. Zhu, Z. Huang, Z. Zhu, J. Liu and K. Li, Thermally boosted color-tunable up-conversion luminescence in Gd2-xLaxZr3(MoO4)9: Yb3+, Ln3+ (Ln = Er, Ho, Tm) for advanced anti-counterfeiting and wide-range-high-sensitivity optical thermometry applications, Mater. Today Chem., 2025, 48, 102971, DOI:10.1016/j.mtchem.2025.102971.
- J. Liu, W. Bu, S. Zhang, F. Chen, H. Xing, L. Pan, L. Zhou, W. Peng and J. Shi, Controlled Synthesis of Uniform and Monodisperse Upconversion Core/Mesoporous Silica Shell Nanocomposites for Bimodal Imaging, Chem. – Eur. J., 2012, 18, 2335–2341, DOI:10.1002/chem.201102599.
- C. Zheng, C. P. Teng, D.-P. Yang, M. Lin, K. Y. Win, Z. Li and E. Ye, Fabrication of luminescent TiO2:Eu3+ and ZrO2:Tb3+ encapsulated PLGA microparticles for bioimaging application with enhanced biocompatibility, Mater. Sci. Eng. C, 2018, 92, 1117–1123, DOI:10.1016/j.msec.2017.10.005.
- L.-D. Sun, Y.-F. Wang and C.-H. Yan, Paradigms and Challenges for Bioapplication of Rare Earth Upconversion Luminescent Nanoparticles: Small Size and Tunable Emission/Excitation Spectra, Acc. Chem. Res., 2014, 47, 1001–1009, DOI:10.1021/ar400218t.
- Q. Li, Z. Zhang and K. Li, Tailoring up-conversion luminescence and enhancing temperature sensitivity through Tm3+ co-doping in Yb3+, Er3+/Ho3+ activated Ca(Gd,La)SbWO8 phosphors, Ceram. Int., 2025 DOI:10.1016/j.ceramint.2025.12.503.
- A. Ćirić and S. Stojadinović, Structural and photoluminescence properties of Y2O3 and Y2O3: Ln3+ (Ln= Eu, Er, Ho) films synthesized by plasma electrolytic oxidation of yttrium substrate, J. Lumin., 2020, 217, 116762 CrossRef.
- S. Thakur, N. Dhiman, A. Sharma and A. K. Gathania, Effect of Photonic Structure on Optical Propertiesof YVO4:Eu3+ Phosphor, J. Electron. Mater., 2017, 46, 2085–2089, DOI:10.1007/s11664-016-5133-x.
- S. Kumar, R. Kumar and A. K. Gathania, Efficient and thermally stable Sm3+ -activated Na2BaCa(PO4)2 phosphors for solid-state lighting and optical thermometry, Mater. Res. Bull., 2026, 197, 113939, DOI:10.1016/j.materresbull.2025.113939.
- P. Du, L. Luo and J. S. Yu, Tunable color upconverison emissions in erbium(III)-doped BiOCl microplates for simultaneous thermometry and optical heating, Microchim. Acta, 2017, 184, 2661–2669, DOI:10.1007/s00604-017-2278-0.
- P. Du, L. Luo, H.-K. Park and J. S. Yu, Citric-assisted sol-gel based Er3+/Yb3+ -codoped Na0.5Gd0.5MoO4: A novel highly-efficient infrared-to-visible upconversion material for optical temperature sensors and optical heaters, Chem. Eng. J., 2016, 306, 840–848, DOI:10.1016/j.cej.2016.08.007.
- R. Dey and V. K. Rai, Yb3+ sensitized Er3+ doped La2O3 phosphor in temperature sensors and display devices, Dalton Trans., 2014, 43, 111–118 RSC.
- M. Mondal, V. K. Rai and C. Srivastava, Influence of silica surface coating on optical properties of Er3+ -Yb3+:YMoO4 upconverting nanoparticles, Chem. Eng. J., 2017, 327, 838–848, DOI:10.1016/j.cej.2017.06.166.
- I. Kumar, Yashwinder, A. Kumar, S. Kumar, H. Thakur and A. K. Gathania, Charge compensation-driven downconverted luminescence enhancement in Er3+ -doped SrTiO3 phosphors by co-doping with alkali ions (M +
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) =
= ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Li, Na, K) for solid-state lighting applications, Indian J. Phys., 2025, 99, 2033–2042, DOI:10.1007/s12648-024-03432-9.
Li, Na, K) for solid-state lighting applications, Indian J. Phys., 2025, 99, 2033–2042, DOI:10.1007/s12648-024-03432-9. - A. Sharma, V. Sharma and A. K. Gathania, Enhancing photoluminescence properties of reddish-orange emitting phosphor cerium fluoride doped with Eu3+ by the incorporation of N-CQDs, J. Alloys Compd., 2025, 1039, 183032, DOI:10.1016/j.jallcom.2025.183032.
- D. Li, W. Qin, T. Aidilibike, P. Zhang, S. Liu, L. Wang and S. Li, Enhanced upconversion emission and magnetization in Yb3+ -Er3+/Ho3+ codoped Gd2O3 nanocrystals by introducing Zn2+ ions, J. Alloys Compd., 2016, 675, 31–36, DOI:10.1016/j.jallcom.2016.03.110.
- R. S. Yadav, S. J. Dhoble and S. B. Rai, Improved photon upconversion photoluminescence and intrinsic optical bistability from a rare earth co-doped lanthanum oxide phosphor via Bi3+ doping, New J. Chem., 2018, 42, 7272–7282, 10.1039/C8NJ01091A.
- J. Xue, Z. Yu, H. M. Noh, B. R. Lee, B. C. Choi, S. H. Park, J. H. Jeong, P. Du and M. Song, Designing multi-mode optical thermometers via the thermochromic LaNbO4: Bi3+/Ln3+ (Ln= Eu, Tb, Dy, Sm) phosphors, Chem. Eng. J., 2021, 415, 128977 CrossRef CAS.
- P. Du, J. Tang, W. Li and L. Luo, Exploiting the diverse photoluminescence behaviors of NaLuF4:xEu3+ nanoparticles and g-C3N4 to realize versatile applications in white light-emitting diode and optical thermometer, Chem. Eng. J., 2021, 406, 127165, DOI:10.1016/j.cej.2020.127165.
- H. Lu, Y. Gao, H. Hao, G. Shi, D. Li, Y. Song, Y. Wang and X. Zhang, Judd-Ofelt analysis and temperature dependent upconversion luminescence of Er3+/Yb3+ codoped Gd2(MoO4)3 phosphor, J. Lumin., 2017, 186, 34–39 CrossRef CAS.
- K. Li and D. Zhu, Ratiometric optical temperature sensing properties based on up-conversion luminescence of novel NaLaTi2O6:Yb3+,Er3+/Ho3+ phosphors, Mater. Res. Bull., 2025, 181, 113117, DOI:10.1016/j.materresbull.2024.113117.
- V. M. Goldschmidt and D. Holmsen, Geochemische verteilungsgesetze der elemente, In Kommission bei J. Dybwad, 1927 Search PubMed.
- X. Li, P. Li, Z. Wang, S. Liu, Q. Bao, X. Meng, K. Qiu, Y. Li, Z. Li and Z. Yang, Color-Tunable Luminescence Properties of Bi3+ in Ca5(BO3)3 F via Changing Site Occupation and Energy Transfer, Chem. Mater., 2017, 29, 8792–8803, DOI:10.1021/acs.chemmater.7b03151.
- H. Li, R. Zhao, Y. Jia, W. Sun, J. Fu, L. Jiang, S. Zhang, R. Pang and C. Li, Sr1.7Zn0.3CeO4:Eu3+ Novel Red-Emitting Phosphors: Synthesis and Photoluminescence Properties, ACS Appl. Mater. Interfaces, 2014, 6, 3163–3169, DOI:10.1021/am4041493.
- P. Du, Y. Hou, W. Li and L. Luo, Ultra-high sensitivity of multicolor Sm3+ -doped LiSrVO4 phosphors for contactless optical thermometers, Dalton Trans., 2020, 49, 10224–10231 RSC.
- F. Ayachi, K. Saidi, K. Soler-Carracedo, M. Dammak and I. R. Martín, Coupled and non-coupled high sensitivity multi-mode ratiometric thermometry of Ho3+/Er3+/Yb3+ tri-doped YP0.5V0.5O4 phosphors, J. Alloys Compd., 2023, 961, 171146, DOI:10.1016/j.jallcom.2023.171146.
- F. Ayachi, K. Saidi, W. Chaabani and M. Dammak, Synthesis and luminescence properties of Er3+ doped and Er3+–Yb3+ codoped phosphovanadate YP0.5V0.5O4 phosphors, J. Lumin., 2021, 240, 118451, DOI:10.1016/j.jlumin.2021.118451.
- Y. Luo, J. Wen, J. Zhang, J. Canning and G.-D. Peng, Bismuth and erbium codoped optical fiber with ultrabroadband luminescence across O-, E-, S-, C-, and L-bands, Opt. Lett., 2012, 37, 3447–3449 CrossRef CAS PubMed.
- F. Ayachi, K. Saidi, M. Dammak, W. Chaabani, I. Mediavilla-Martínez and J. Jiménez, Dual-mode luminescence of Er3+/Yb3+ codoped LnP0.5V0.5O4 (Ln= Y, Gd, La) for highly sensitive optical nanothermometry, Mater. Today Chem., 2023, 27, 101352 Search PubMed.
- F. Ayachi, K. Saidi, W. Chaabani and M. Dammak, Synthesis and luminescence properties of Er3+ doped and Er3+–Yb3+ codoped phosphovanadate YP0.5V0.5O4 phosphors, J. Lumin., 2021, 240, 118451, DOI:10.1016/j.jlumin.2021.118451.
- A. K. Singh, Ho3+:TeO2 glass, a probe for temperature measurements, Sens. Actuators Phys., 2007, 136, 173–177, DOI:10.1016/j.sna.2006.10.045.
- L. Yu, H. Song, S. Lu, Z. Liu, L. Yang and X. Kong, Luminescent Properties of LaPO4:Eu Nanoparticles and Nanowires, J. Phys. Chem. B, 2004, 108, 16697–16702, DOI:10.1021/jp047688c.
- R. S. Yadav, D. Kumar, A. K. Singh, E. Rai and S. B. Rai, Effect of Bi3+ ion on upconversion-based induced optical heating and temperature sensing characteristics in the Er 3+/Yb3+ co-doped La2O3 nano-phosphor, RSC Adv., 2018, 8, 34699–34711, 10.1039/C8RA07438K.
- F. Ayachi, K. Saidi and M. Dammak, Exploring luminescence quenching mechanisms and temperature sensing capabilities of LiSrYW3O12: Sm3+ phosphors, Mater. Adv., 2024, 5, 6162–6169 RSC.
- K. Saidi, C. Hernández-Álvarez, M. Runowski, M. Dammak and I. R. Martín, Ultralow pressure sensing and luminescence thermometry based on the emissions of Er 3+/Yb3+ codoped Y2Mo4O15 phosphors, Dalton Trans., 2023, 52, 14904–14916 RSC.
- F. Ayachi, K. Saidi, M. Dammak, I. Mediavilla and J. Jiménez, Unlocking advanced thermometric capabilities: BiVO4: Er3+/Yb3+ nanophosphors with dual-mode up-conversion and down-shifting features, RSC Adv., 2025, 15, 655–664, 10.1039/D4RA08590F.
- N. B. Amar, K. Saidi, C. Hernández-Álvarez, M. Dammak and I. R. Martin, Ultra-high-sensitive temperature sensing based on emission Pr3+ and Yb3+ codoped Y2Mo3O12 nanostructures, Mater. Adv, 2025, 6, 827–838, 10.1039/D4MA00746H.
- F. Ayachi, K. Saidi, M. Dammak, J. J. Carvajal and M. C. Pujol, Enhancing thermometric efficiency: a wavelength excitation analysis in LiSrGdW3 O12:Tb3+ for superior single band ratiometric (SBR) thermometry, RSC Adv., 2024, 14, 13494–13504, 10.1039/D4RA00626G.
- A. Ćirić, K. Shah, M. Sekulić, B. S. Chakrabarty and M. D. Dramićanin, La2O2S: Er3+/Yb3+ nanoparticles synthesized by the optimized furnace combustion technique and their high-resolution temperature sensing, Optik, 2021, 245, 167690 CrossRef.
- M. Fhoula, K. Saidi, C. Hernández-Álvarez, K. Soler-Carracedo, M. Dammak and I. R. Martín, Unlocking the luminescent potential of Pr3+/Yb3+ Co-doped Y2Mo4O15 for advanced thermometry applications, J. Alloys Compd., 2024, 979, 173537, DOI:10.1016/j.jallcom.2024.173537.
- K. Saidi, I. Kachou, K. Soler-Carracedo, M. Dammak and I. R. Martín, Ba2YV3O11 Er3+/Yb3+ Nanostructures for Temperature Sensing in the Presence of Bismuth Ions, ACS Appl. Nano Mater., 2023, 6, 17681–17690, DOI:10.1021/acsanm.3c02911.
- Z. E. A. A. Taleb, K. Saidi and M. Dammak, Dual-mode optical ratiometric thermometry using Pr3+ -doped NaSrGd(MoO4)3 phosphors with tunable sensitivity, Dalton Trans., 2023, 52, 18069–18081, 10.1039/D3DT03242F.
- Z. Wang, M. Jia, M. Zhang, X. Jin, H. Xu and Z. Fu, Trimodal Ratiometric Luminescent Thermometer Covering Three Near-Infrared Transparency Windows, Inorg. Chem., 2021, 60, 14944–14951, DOI:10.1021/acs.inorgchem.1c02311.
- L. Lei, X. Dai, Y. Cheng, Y. Wang, Z. Xiao and S. Xu, Dual-mode color tuning based on upconversion core/triple-shell nanostructure, J. Mater. Chem. C, 2019, 7, 3342–3350, 10.1039/C8TC05467C.
- F. Ayachi, K. Saidi, M. Dammak, W. Chaabani, I. Mediavilla-Martínez and J. Jiménez, Dual-mode luminescence of Er3+/Yb3+ codoped LnP0.5V0.5O4 (Ln = Y, Gd, La) for highly sensitive optical nanothermometry, Mater. Today Chem., 2023, 27, 101352, DOI:10.1016/j.mtchem.2022.101352.
- K. Pavani, J. P. C. do Nascimento, S. K. Jakka, F. F. do Carmo, A. J. M. Sales, M. J. Soares, M. P. F. Graça, F. J. A. de Aquino, D. X. Gouveia and A. S. B. Sombra, Analogy of different optical temperature sensing techniques in LaNbO4:Er3+/Yb3+ phosphor, J. Lumin., 2021, 235, 117992, DOI:10.1016/j.jlumin.2021.117992.
- L. Li, C. Guo, S. Jiang, D. K. Agrawal and T. Li, Green up-conversion luminescence of Yb3+ -Er3+ co-doped CaLa2ZnO5 for optically temperature sensing, RSC Adv., 2014, 4, 6391, 10.1039/c3ra47264g.
- D. Manzani, J. F. da, S. Petruci, K. Nigoghossian, A. A. Cardoso and S. J. L. Ribeiro, A portable luminescent thermometer based on green up-conversion emission of Er3+/Yb3+ co-doped tellurite glass, Sci. Rep., 2017, 7, 41596, DOI:10.1038/srep41596.
- J. Zhang, Y. Zhang and X. Jiang, Investigations on upconversion luminescence of K3Y(PO4)2:Yb3+-Er3+/Ho3+/Tm3+ phosphors for optical temperature sensing, J. Alloys Compd., 2018, 748, 438–445, DOI:10.1016/j.jallcom.2018.03.127.
| This journal is © The Royal Society of Chemistry 2026 |