Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Contribution of Mg-templated porosity to activity and durability in Fe–N–C O2 reduction catalysts
Angus
Pedersen
 *abc,
Jinjie
Zhu
b,
Jesús
Barrio
*abc,
Jinjie
Zhu
b,
Jesús
Barrio
 *b,
Joseph
Parker
*b,
Joseph
Parker
 d,
Robert D.
Hunter
b,
Sarah J.
Haigh
d,
Robert D.
Hunter
b,
Sarah J.
Haigh
 d,
Tim-Patrick
Fellinger
d,
Tim-Patrick
Fellinger
 c,
Ifan E. L.
Stephens
c,
Ifan E. L.
Stephens
 a and
Maria-Magdalena
Titirici
a and
Maria-Magdalena
Titirici
 *be
*be
aDepartment of Materials, Imperial College London, London SW7 2AZ, UK. E-mail: angus-nils.pedersen@bam.de
bDepartment of Chemical Engineering, Imperial College London, London SW7 2AZ, UK. E-mail: j.barrio-hermida@imperial.ac.uk, m.titirici@imperial.ac.uk
cDivision 3.6, Electrochemical Energy Materials, Bundesanstalt für Material-forschung und -prüfung (BAM), Unter den Eichen 44-46, 12203, Berlin, Germany
dDepartment of Materials, University of Manchester, Manchester, M13 9PL, England
eAdvanced Institute for Materials Research (WPI-AIMR), Tohoku University, 2-1-1 Katahira, Aobaku, Sendai, Miyagi 980-8577, Japan
First published on 16th March 2026
Abstract
Atomically dispersed Fe in N-doped carbon (Fe–N–C) catalysts are leading platinum-group-metal-free candidates for the O2 reduction reaction in proton exchange membrane fuel cells (PEMFCs). Zeolitic imidazolate framework (ZIF-8) derived Fe–N–C present the most promising performance; however, they possess a narrow distribution of small micropores, which limits active site accessibility. Here, to induce hierarchical porosity in Fe–N–C, we report a systematic study on MgCl2·6H2O-templated ZIF-8-derived Fe–N–C catalysts for the O2 reduction reaction. MgCl2·6H2O addition induced complete Zn removal, collapse of the ZIF-8 framework, and formation of large micro- and mesopores, with graphene-like structures. N content was markedly reduced, with conversion from pyridinic to pyrrolic N species. Rotating disc electrode tests showed a progressive increase in O2 reduction activity with MgCl2·6H2O, which is strongly correlated (R2 = 0.98) to the formation of large micropores and small mesopores (1–4 nm). This introduces an indirect structure–activity design principle for Fe–N–Cs. The enhanced Fe–N–C porosity also leads to increased degradation rates under accelerated stress test conditions, which we attributed to the oxidation of disordered carbon domains and active Fe loss. This study highlights a key trade-off between porosity-driven O2 reduction activity and durability in Fe–N–C catalysts.
Introduction
Low temperature proton exchange membrane fuel cells (PEMFCs) offer an efficient and zero CO2 emission operation technology for transport and back-up power applications. One limitation of PEMFCs is the significant use of expensive precious metals, such as Pt, at the cathode, where the O2 reduction reaction occurs.1 The next-generation PEMFCs are reducing Pt content based on improved catalyst and cell designs.2 Still, techno-economic projects of Pt nanoparticle on C (Pt/C) based cathodes in PEMFC at 500![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 stacks year−1 predict the Pt catalyst would make up 26% of the stack cost, based on US DOE 2025 Pt loading targets.3 Additionally, Pt is highly susceptible to poisoning from contaminants, such as CO, H2S and SO2.4,5 As an alternative, single metal atoms in N-doped C catalysts (M–N–C), in particular Fe–N–C, show the highest non-precious metal O2 reduction activity, and exhibit high poison tolerance.6
000 stacks year−1 predict the Pt catalyst would make up 26% of the stack cost, based on US DOE 2025 Pt loading targets.3 Additionally, Pt is highly susceptible to poisoning from contaminants, such as CO, H2S and SO2.4,5 As an alternative, single metal atoms in N-doped C catalysts (M–N–C), in particular Fe–N–C, show the highest non-precious metal O2 reduction activity, and exhibit high poison tolerance.6
Among M–N–C precursors, zeolitic imidazolate framework (ZIF), such as ZIF-8, have been pivotal to PEMFC performance progress and scalability.7–10 ZIFs were first demonstrated by Yaghi and coworkers to present exceptional chemical and thermal stability compared to most metal organic frameworks.11 Additionally, many different ZIF topologies and structures are synthesizable,12,13 allowing the development of structure–activity descriptors, including the cavity size of the pristine ZIF.14 The high content of retained Zn–N4 sites15,16 and carbonaceous microporous structure of ZIF-8 following pyrolysis17 provides a suitable conductive template for O2 reduction active FeNx (x = 1–5) sites.18 Fellinger and coworkers first introduced a Zn-imprinting and ion-exchange synthesis to decouple active site formation from pyrolysis, allowing low-temperature metalation and preventing undesired pyrolysis-driven carbide formation.19–21 These transmetalation routes have achieved record FeNx active site densities and O2 reduction activity.22,23 Despite these advances, typically <10% of the FeNx sites are electrochemically utilised (based on five electron in situ nitrite stripping), limiting the accessible FeNx site density required to compete with Pt-based catalyst O2 reduction activity.23,24 This is due to the dominant microporosity of bulky ZIF-8-derived particles.16,25 Nonetheless, studies demonstrating clear correlations of porosity versus activity of M–N–Cs remain limited.14,26,27 Salt-templating with ZnCl2 or NaCl have demonstrated hierarchical porosity and opening of bottleneck pores to improve O2 reduction performance26–31 More recently, Mg-based templating has proven particularly effective.32–38 In the case of MgCl2·6H2O this was due to the generation of transient porogens39 and MgNx active site templating.32 Using this approach, our group reported MgCl2·6H2O templated Fe–N–C with record electrochemical active utilisation of 52% (based on five electron in situ nitrite stripping),37 and up to 29 ± 4% in PEMFC (based on in situ Fourier transformed alternating current voltammetry).24 Addition of MgCl2·6H2O to ZIF-8 and their subsequent pyrolysis has been explored for Na-ion anodes to generate microporosity.40 It was found that due to the salt melt, the ZIF-8 structure dissolves during the carbonisation process, creating carbon nanosheets. This led to near complete removal of Zn, from 10 wt% in directly pyrolysed ZIF-8 to <0.3 wt% in Mg-doped samples.40
Aside from activity, stability of Fe–N–C under acidic conditions is challenging and insufficient, due to a combination of Fe dissolution, reactive oxygen species and carbon corrosion.41–44 So far, high surface area Mg-templated Fe–N–C have displayed fast degradation and poor active site stability under PEMFC-relevant conditions.45,46 Recently, Wu and coworkers reported trace Mg- doping (0.5% Mg) in ZIF-derived Fe–N–C can significantly improve the catalyst stability by altering the local FeNx carbon structure, eliminating undesired N species and suppressing H2O2 formation.47
Here, we investigate how the Mg salt (MgCl2·6H2O) to ZIF-8 ratio controls the chemistry and morphology of ZIF-derived Fe–N–Cs and correlate porosity effects on activity and stability for O2 reduction in rotating disc electrode measurements.
Experimental
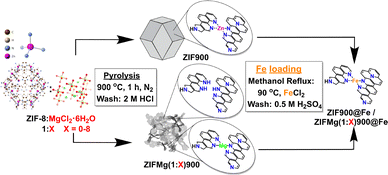
The catalysts were synthesised based on Fig. 1, with the steps detailed in the SI. Briefly, commercial ZIF-8 (Basolite® Z1200) was either directly pyrolysed or mixed with MgCl2·6H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1–8 wt. ratio) and then pyrolysed (5 °C min−1) under N2 flow to 900 °C for 1 h. The resulting powders were ground, washed with 2 M HCl and then dried at 80 °C. The catalyst was then impregnated with Fe using FeCl2 under methanol reflux for 24 h and then washed with 0.5 M H2SO4, followed by 80 °C drying. Characterisation and electrochemical testing protocols are described in the SI.
1–8 wt. ratio) and then pyrolysed (5 °C min−1) under N2 flow to 900 °C for 1 h. The resulting powders were ground, washed with 2 M HCl and then dried at 80 °C. The catalyst was then impregnated with Fe using FeCl2 under methanol reflux for 24 h and then washed with 0.5 M H2SO4, followed by 80 °C drying. Characterisation and electrochemical testing protocols are described in the SI.
Results and discussion
Catalyst characterisation
Pyrolysis of ZIF-8 at 900 °C and subsequent acid washing yielded 29% ZIF900 (Table S1), while addition of MgCl2·6H2O reducing the yield to 14–17%, indicating salt-mediated volatilisation during carbonisation. Post pyrolysis, ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)900 substantially increased in volume (Fig. S1a), whereas higher salt ratios produced denser products (Fig. S1b and c). As previously established,32,33,37 these differences arise likely due to the different templating pathways. At low Mg ratios, partial wetting of ZIF-8 produced a viscous matrix that trapped evolved gases and caused foaming, whereas at high Mg loadings, ZIF-8 dispersed in a molten salt phase that enabled gas release, yielding compact carbons. Thermal gravimetric analysis mass spectrometry (TGA–MS) confirmed distinct decomposition behaviour with increasing MgCl2·6H2O, shifting from oxidative carbon loss to chloride-driven dehydrogenation (Note S1 and Fig. S2).
1)900 substantially increased in volume (Fig. S1a), whereas higher salt ratios produced denser products (Fig. S1b and c). As previously established,32,33,37 these differences arise likely due to the different templating pathways. At low Mg ratios, partial wetting of ZIF-8 produced a viscous matrix that trapped evolved gases and caused foaming, whereas at high Mg loadings, ZIF-8 dispersed in a molten salt phase that enabled gas release, yielding compact carbons. Thermal gravimetric analysis mass spectrometry (TGA–MS) confirmed distinct decomposition behaviour with increasing MgCl2·6H2O, shifting from oxidative carbon loss to chloride-driven dehydrogenation (Note S1 and Fig. S2).
Scanning electron microscopy (SEM) reveals the ZIF-8 structure is lost after MgCl2·6H2O addition (ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)900), forming a heterogeneous mixture of smooth, bubble-like particle sizes up to 50 µm (Fig. S3b), arising from the dominant bubble templating. Increasing MgCl2·6H2O content further (ZIFMg(1
1)900), forming a heterogeneous mixture of smooth, bubble-like particle sizes up to 50 µm (Fig. S3b), arising from the dominant bubble templating. Increasing MgCl2·6H2O content further (ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2)900, Fig. S3c), the particles appear less well-defined, and a more porous structure develops as ZIF-8 is soaked by the salt. This trend continues with ZIFMg(1
2)900, Fig. S3c), the particles appear less well-defined, and a more porous structure develops as ZIF-8 is soaked by the salt. This trend continues with ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4)900 and ZIFMg(1
4)900 and ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8)900, with sheet-like open structures with lateral dimensions of up to ∼5 µm (Fig. S3d and e). Overlayed Raman spectroscopy and its deconvoluted peaks (Fig. S4) shows a distinct contribution of D3- (∼ 1495 cm−1) is present in ZIF-900 and ZIFMg(1
8)900, with sheet-like open structures with lateral dimensions of up to ∼5 µm (Fig. S3d and e). Overlayed Raman spectroscopy and its deconvoluted peaks (Fig. S4) shows a distinct contribution of D3- (∼ 1495 cm−1) is present in ZIF-900 and ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)900. This is attributed to amorphous carbon species.48 This feature largely diminishes for higher ratios of MgCl2·6H2O, as demonstrated by the reduction in ID3/IG (Table S2). Meanwhile, the ID/IG ratio does not significantly change, indicating comparable lateral size of nano-crystallites. However, the full-width half maximum of D and G bands (ca. 1585 cm−1, arising from quasi-graphitic crystallites) significantly decreases. The reduction in D3 band intensity upon adding MgCl2·6H2O, indicates less disordered sp2–sp3 carbon domains. This may be due to facilitated conversion of aliphatic intermediates due to increased reorganizational dynamics in the presence of the molten salt. X-ray diffraction (XRD) confirms collapse of ZIF-8 crystallinity with pyrolysis, and disappearance of C (002) reflection at 2-theta of ∼25° with MgCl2·6H2O addition (Fig. S5), indicating atomically thin graphene-like structure with limited long-range carbon stacking.
1)900. This is attributed to amorphous carbon species.48 This feature largely diminishes for higher ratios of MgCl2·6H2O, as demonstrated by the reduction in ID3/IG (Table S2). Meanwhile, the ID/IG ratio does not significantly change, indicating comparable lateral size of nano-crystallites. However, the full-width half maximum of D and G bands (ca. 1585 cm−1, arising from quasi-graphitic crystallites) significantly decreases. The reduction in D3 band intensity upon adding MgCl2·6H2O, indicates less disordered sp2–sp3 carbon domains. This may be due to facilitated conversion of aliphatic intermediates due to increased reorganizational dynamics in the presence of the molten salt. X-ray diffraction (XRD) confirms collapse of ZIF-8 crystallinity with pyrolysis, and disappearance of C (002) reflection at 2-theta of ∼25° with MgCl2·6H2O addition (Fig. S5), indicating atomically thin graphene-like structure with limited long-range carbon stacking.
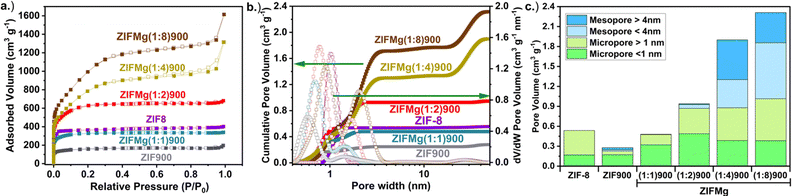
N2 sorption of as received ZIF-8 presents a Brunauer–Emmett–Teller specific surface area (SBET) of 1170 m2 g−1 (Fig. 2a and Table S3), which is comparable to previous reports.49 Upon pyrolysis, ZIF-900 shows a reduced SBET of 568 m2 g−1. Mehmood et al. showed the ZIF-8 specific surface area can largely be recovered post pyrolysis by their activation and dicyandiamide doping protocol of the Fe–N–C, reaching 1155 m2 g−1. The cumulative pore volume up to a pore width of 4 nm increased 60%, with the electrochemical active site density increasing more than fourfold.23 Here, it was found the starting SBET of ZIF-8 could be retained with ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)900, at 1138 m2 g−1. As expected, increasing the MgCl2·6H2O ratios led to increased SBET, up to 3548 m2 g−1 at ZIFMg(1
1)900, at 1138 m2 g−1. As expected, increasing the MgCl2·6H2O ratios led to increased SBET, up to 3548 m2 g−1 at ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8)900, comparable to our previously reported Mg-templated carbons.45,50
8)900, comparable to our previously reported Mg-templated carbons.45,50
The corresponding pore size distributions from the heterogeneous surface 2D non-linear density functional theory (HS 2D NLDFT) model reveal that ZIF-8 is purely microporous with pore diameters centered at 1 nm, which contracts to 0.5 nm after pyrolysis (Fig. 2b). Increasing MgCl2·6H2O progressively widens the micropores, reaching 0.9 nm at ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4)900. The introduction of additional pore diameter at 2 nm and above 40 nm becomes pronounced. The sub-defined micro- and mesopore volume values are displayed in Fig. 2c and Table S3, showcasing how micropore volumes increase up to 0.88 cm3 g−1 in ZIFMg(1
4)900. The introduction of additional pore diameter at 2 nm and above 40 nm becomes pronounced. The sub-defined micro- and mesopore volume values are displayed in Fig. 2c and Table S3, showcasing how micropore volumes increase up to 0.88 cm3 g−1 in ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2)900 and then plateau, meanwhile mesopore volumes increase up to 0.58 cm3 g−1 in ZIFMg(1
2)900 and then plateau, meanwhile mesopore volumes increase up to 0.58 cm3 g−1 in ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4)900 and then plateaus. A similar evolution was found by Pampel et al. when varying the MgCl2·6H2O ratio with adenine-derived carbons, although in their case a more tubular N–C porous structure was created due to the self-assembly of adenine and its polycondensate products with the water molecules within the Mg salt.33
4)900 and then plateaus. A similar evolution was found by Pampel et al. when varying the MgCl2·6H2O ratio with adenine-derived carbons, although in their case a more tubular N–C porous structure was created due to the self-assembly of adenine and its polycondensate products with the water molecules within the Mg salt.33
Inductively coupled plasma mass spectrometry (ICP-MS, Fig. S6a) on commercial ZIF-8, as received, contained 28.29 ± 0.70 wt% Zn, decreasing to 8.71 ± 0.65 wt% after pyrolysis (ZIF900). In ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)900 (1.12 molar ratio for MgCl2·6H2O:ZIF-8), Zn reduced to 0.46 wt% with 1.51 wt% Mg. ZIFMg(1
1)900 (1.12 molar ratio for MgCl2·6H2O:ZIF-8), Zn reduced to 0.46 wt% with 1.51 wt% Mg. ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2)900 contained only 0.1 ± 0.01 wt% Zn and 0.72 ± 0.04 wt% Mg, while only trace amounts of metals remain in ZIFMg(1
2)900 contained only 0.1 ± 0.01 wt% Zn and 0.72 ± 0.04 wt% Mg, while only trace amounts of metals remain in ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4)900, consistent with Zn volatilisation (Zn boiling point around 918 °C)15 and complete Mg template removal. This is consistent with Mehmood et al. who observed removal of the ZIF-8 structure and Zn removal upon addition of high wt. ratio of MgCl2·6H2O (1
4)900, consistent with Zn volatilisation (Zn boiling point around 918 °C)15 and complete Mg template removal. This is consistent with Mehmood et al. who observed removal of the ZIF-8 structure and Zn removal upon addition of high wt. ratio of MgCl2·6H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10).40 This is likely caused by ZIF-8 dissolution in the liquid phase and Mg salt-melt breaking the Zn coordination to 2-methyl-imidazole.
10).40 This is likely caused by ZIF-8 dissolution in the liquid phase and Mg salt-melt breaking the Zn coordination to 2-methyl-imidazole.
Fe introduced via low temperature methanol reflux showed sample-dependent incorporation. Mehmood et al. achieved up to 7.1 wt% Fe loading with only 1.0 wt% of Zn remaining by carrying out activation steps on a carbonised ZIF-8 (Fig. S6b).23 The stronger binding energy of FeN4 compared to ZnN4 allows thermodynamically favourable ion exchange, with kinetically feasible activation barriers.51 Meanwhile, directly loading Fe into ZIF900 (ZIF900@Fe) leads to 1.00 ± 0.13 wt% of Fe and reduction of Zn to 7.85 ± 0.27 wt%. In contrast, ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)900@Fe contained 0.3 wt% Fe and 0.25 wt% Zn, and Mg unchanged (1.50 wt%). The reciprocal changes in Fe and Zn content indicate preferential Fe–Zn transmetalation, rather than Fe–Mg exchange. Meanwhile, only 0.18 wt% Fe is present in ZIFMg(1
1)900@Fe contained 0.3 wt% Fe and 0.25 wt% Zn, and Mg unchanged (1.50 wt%). The reciprocal changes in Fe and Zn content indicate preferential Fe–Zn transmetalation, rather than Fe–Mg exchange. Meanwhile, only 0.18 wt% Fe is present in ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2)900@Fe and Mg dropped to 0.03 wt%, with Zn remaining equivalent (0.08 wt%). This suggests Fe may have transmetalated with Mg in MgNx sites or loaded into empty Nx sites. On the other hand, the increase in Fe content in ZIFMg(1
2)900@Fe and Mg dropped to 0.03 wt%, with Zn remaining equivalent (0.08 wt%). This suggests Fe may have transmetalated with Mg in MgNx sites or loaded into empty Nx sites. On the other hand, the increase in Fe content in ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4)900@Fe and ZIFMg(1
4)900@Fe and ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8)900@Fe likely reflects Fe metalation into empty Nx sites.
8)900@Fe likely reflects Fe metalation into empty Nx sites.
XRD patterns of Fe-loaded catalysts show identical carbon reflections to Mg-templated NC, confirming the absence of large or concentrated crystalline Fe phases (Fig. S7).
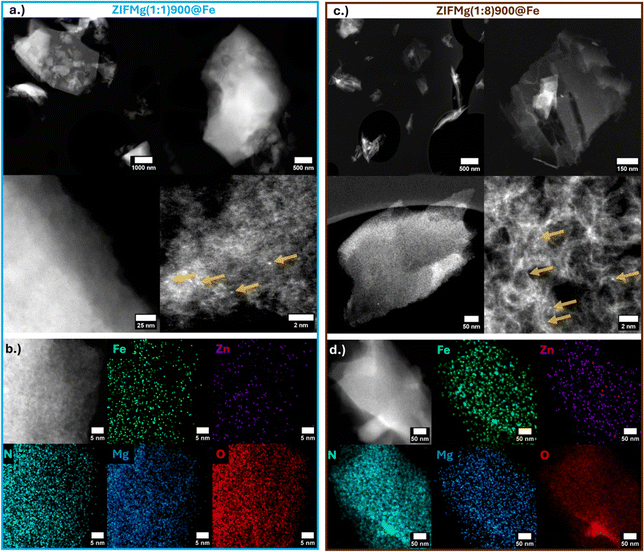
High angle annular dark field-scanning transmission electron microscopy (HAADF-STEM) imaging of ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)900@Fe reveals partially retained ZIF-8 morphology (Fig. 3a), with isolated bright atomic features attributed to Fe or residual Zn, rather than Mg, due to their higher atomic mass. Elemental mapping demonstrates a uniform dispersion of Fe, Zn and Mg, with no evidence of nanoparticle formation. (Fig. 3b and Fig. S8a, S9b, c). In ZIFMg(1
1)900@Fe reveals partially retained ZIF-8 morphology (Fig. 3a), with isolated bright atomic features attributed to Fe or residual Zn, rather than Mg, due to their higher atomic mass. Elemental mapping demonstrates a uniform dispersion of Fe, Zn and Mg, with no evidence of nanoparticle formation. (Fig. 3b and Fig. S8a, S9b, c). In ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8)900@Fe, large micro- and mesopores are clearly visible (Fig. 3c), consistent with N2 sorption data (Fig. 2b). Atomic metal dispersion is also evident in the largely exfoliated graphene-like layers (Fig. 3c and d), showing nm-sized graphitic carbon domains, analogous to previous Mg-templated N–C.24 Elemental mapping show the even dispersion of elements, with energy dispersive X-ray spectroscopy showing a weak but detectable Fe signal, with little to no Zn or Mg remaining (Fig. S8d). Based on ICP-MS (Fig. S7) and STEM-EDXS, the identified single atoms are therefore expected to be predominantly Fe. Due to the low Fe loading in Mg-templated Fe–N–Cs here (≤0.3 wt% Fe), clear characterisation on the FeNx active sites beyond HAADF-STEM and STEM-EDXS remains challenging. For insights into how FeNx changes with Mg-templating we refer the reader to our recent works.37,38,52 Here, focus is made on the clearly distinguishable catalyst properties on performance, such as porosity and N type.
8)900@Fe, large micro- and mesopores are clearly visible (Fig. 3c), consistent with N2 sorption data (Fig. 2b). Atomic metal dispersion is also evident in the largely exfoliated graphene-like layers (Fig. 3c and d), showing nm-sized graphitic carbon domains, analogous to previous Mg-templated N–C.24 Elemental mapping show the even dispersion of elements, with energy dispersive X-ray spectroscopy showing a weak but detectable Fe signal, with little to no Zn or Mg remaining (Fig. S8d). Based on ICP-MS (Fig. S7) and STEM-EDXS, the identified single atoms are therefore expected to be predominantly Fe. Due to the low Fe loading in Mg-templated Fe–N–Cs here (≤0.3 wt% Fe), clear characterisation on the FeNx active sites beyond HAADF-STEM and STEM-EDXS remains challenging. For insights into how FeNx changes with Mg-templating we refer the reader to our recent works.37,38,52 Here, focus is made on the clearly distinguishable catalyst properties on performance, such as porosity and N type.
Overlaid XPS spectra of C1s, O1s, N1s, Zn2p and Mg1s of ZIF900 and ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) X)900 catalysts are shown in Fig. S9 with the corresponding at% element composition in Fig. 4a. The increased O content in ZIFMg(1
X)900 catalysts are shown in Fig. S9 with the corresponding at% element composition in Fig. 4a. The increased O content in ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)900 may correspond to MgO remaining after acid washing due to limited accessibility. With increasing Mg precursor ratio, the C content increases while O and Mg content decreases. The 4.0 at% of Zn in ZIF900 is almost entirely removed for ZIFMg(1
1)900 may correspond to MgO remaining after acid washing due to limited accessibility. With increasing Mg precursor ratio, the C content increases while O and Mg content decreases. The 4.0 at% of Zn in ZIF900 is almost entirely removed for ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)900, demonstrating the dissolution of the ZIF-8 structure.
1)900, demonstrating the dissolution of the ZIF-8 structure.
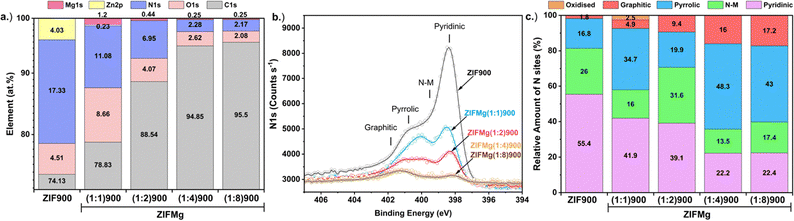 | ||
| Fig. 4 XPS of ZIF-8 derived and Mg-doped catalysts. (a) Elemental composition. (b) Overlays of N1s and (c) corresponding fitted N species composition. | ||
The overlaid N1s spectra demonstrates the reduction in N species upon increasing MgCl2·6H2O ratios. Overlaid N1s spectra are shown in Fig. 4b with their quantities plotted in Fig. 4c and fittings displayed in Fig. S10. N content falls from 17.3 at% in ZIF900 to 2.2 at% in ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8)900. Meanwhile, increasing MgCl2·6H2O results in a relative increase in graphitic N, from 1.8 to 17.2% between ZIF900 and ZIFMg(1
8)900. Meanwhile, increasing MgCl2·6H2O results in a relative increase in graphitic N, from 1.8 to 17.2% between ZIF900 and ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8)900, respectively. Additionally, a clear trend of relative decreasing pyridinic N content and increasing pyrrolic N is found with increasing MgCl2·6H2O. According to density functional theory studies and recent X-ray absorption spectroscopy observations, this could be due to Mg having a higher affinity towards pyrrolic motifs than Zn, which could lead to greater pyrrolic retainment.52,53 Elemental analysis also confirms a sharp decrease in N content from 15.2 to 2.3 wt% with increasing MgCl2·6H2O precursor, indicating comparable bulk and surface composition (Fig. S11).
8)900, respectively. Additionally, a clear trend of relative decreasing pyridinic N content and increasing pyrrolic N is found with increasing MgCl2·6H2O. According to density functional theory studies and recent X-ray absorption spectroscopy observations, this could be due to Mg having a higher affinity towards pyrrolic motifs than Zn, which could lead to greater pyrrolic retainment.52,53 Elemental analysis also confirms a sharp decrease in N content from 15.2 to 2.3 wt% with increasing MgCl2·6H2O precursor, indicating comparable bulk and surface composition (Fig. S11).
Cryogenic (5 K) X-band electron paramagnetic resonance (EPR) reveals suppression of carbon-centred radicals upon Mg addition (g ∼ 2) and predominantly EPR-inactive Fe2+ species after Fe loading (Fig. S12), which is regarded as the less active but more durable FeNx site for O2 reduction.54
Correlating with O2 reduction performance
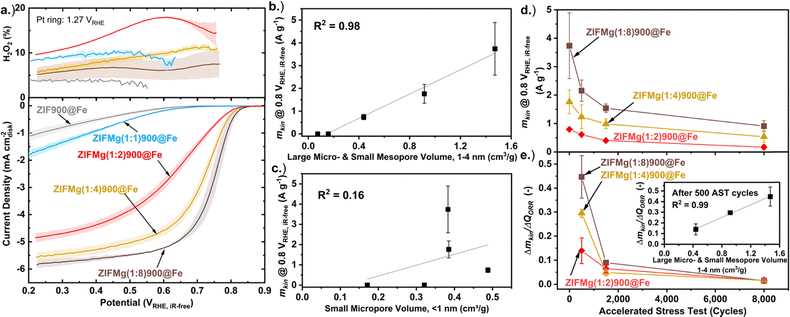
The O2 reduction performance of the catalysts was tested in O2-saturated 0.1 M HClO4 three-electrode rotating ring disc electrode (RRDE) setup under 1600 rpm (0.26 mgM–N–C cm−2 loading). Prior to Fe loading, all ZIF-derived catalyst displayed negligible O2 reduction activity, only beginning to show current onset (>0.1 mA cm−2) below 0.6 VRHE (Fig. S13). Following Fe incorporation, ZIF900@Fe displayed H2O2 production <5% across the potential range; however, the disk current density did not exceed 1.25 mA cm−2, despite high Fe content (1.0 wt%, Fig. S6b), suggesting limited FeNx accessibility. The H2O2% decreases from ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2)900@Fe to ZIFMg(1
2)900@Fe to ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8)900@Fe (Fig. 5a and eqn (S4)). This improvement likely stems from enhanced porosity and surface area, enabling secondary reduction of H2O2 within the catalyst layer,55 and/or increased electrochemical accessibility of FeNx active sites.37
8)900@Fe (Fig. 5a and eqn (S4)). This improvement likely stems from enhanced porosity and surface area, enabling secondary reduction of H2O2 within the catalyst layer,55 and/or increased electrochemical accessibility of FeNx active sites.37
Increasing the MgCl2·6H2O precursor ratio leads to improved O2 reduction activity, particularly for Mg-templated samples with higher Mg precursor ratios (X ≥ 2, Fig. 5), despite comparable Fe loadings (0.18–0.30 wt%, Fig. S7). This correlation is strongest (R2 = 0.98) when considering the increasing volume of large micropores and small mesopores (1–4 nm, Fig. 2c) with the kinetic mass activity (mkin, eqn (S2)) at 0.8 VRHE,iR-free (Fig. 5b). This follows observations by Wang et al. where pores between 0.8–2.0 nm contributed the majority of Fe–N–C O2 reduction activity.27 Additionally, Wan et al. reported mesopore incorporation into ZIF-8-derived Fe–N–C increased FeNx electrochemical utilisation and site density, leading to improved PEMFC performance.30 We relate these combined findings to FeNx active sites becoming electrochemically accessible above specific pore diameters. We note that porosity has an indirect effect on activity, rather than direct changes in the intrinsic activity of FeNx sites. Consequently, porosity can be considered as a secondary (indirect), rather than primary, structure–activity descriptor. No correlation is observed when considering small micropores <1 nm (R2 = 0.16, Fig. 5c), indicating small micropores do not contribute to O2 reduction activity. The correlation of other pore volumes with activity is shown in Fig. S14.
We note that our previously Mg-templated Fe–N–C presented a low intrinsic O2 reduction activity (turnover frequency) compared to the state-of-the-art.37 This also likely restricts the measured initial kinetic mass activity here (Fig. 5d), in addition to the limited site density from the low Fe loading (Fig. S6b).
Accelerated stress testing (AST; 0.80-0.40 VRHE, 100 mV s−1 O2-saturated, 1600 rpm; Fig. 5d) shows that while ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8)900@Fe exhibits the highest initial kinetic mass activity (3.74 ± 1.14 A gFe–N–C−1), it undergoes the fastest degradation. After 8000 AST cycles all electrocatalysts displayed H2O2 yield >20% across the potential range (Fig. S15). When normalised to the total O2 reduction charge (QORR, eqn (S5)), degradation scales with the volume of 1–4 nm pores (inset, Fig. 5e). This shows the most active regions of the Fe–N–Cs are also the most unstable. After 8000 AST cycles the rate of degradation converge and become comparable. This may due to a shift of degradation mechanism from initially primarily Fe dissolution to reactive oxygen species attack and carbon corrosion,41,42,44 as we previously resolved for another Mg-templated Fe–N–C.45,46 It is also noted there is no observable stabilisation effect from Mg doping, unlike in the work by Wu and coworkers, where Mg acetylacetonate was added during ZIF-8 synthesis which led to elimination of undesired N-species in their Fe–N–C.
8)900@Fe exhibits the highest initial kinetic mass activity (3.74 ± 1.14 A gFe–N–C−1), it undergoes the fastest degradation. After 8000 AST cycles all electrocatalysts displayed H2O2 yield >20% across the potential range (Fig. S15). When normalised to the total O2 reduction charge (QORR, eqn (S5)), degradation scales with the volume of 1–4 nm pores (inset, Fig. 5e). This shows the most active regions of the Fe–N–Cs are also the most unstable. After 8000 AST cycles the rate of degradation converge and become comparable. This may due to a shift of degradation mechanism from initially primarily Fe dissolution to reactive oxygen species attack and carbon corrosion,41,42,44 as we previously resolved for another Mg-templated Fe–N–C.45,46 It is also noted there is no observable stabilisation effect from Mg doping, unlike in the work by Wu and coworkers, where Mg acetylacetonate was added during ZIF-8 synthesis which led to elimination of undesired N-species in their Fe–N–C.
Post-AST HAADF-STEM of ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8)900@Fe shows retained morphology and some single atoms remaining (Fig. S16), with EDXS demonstrating reduced Fe signal (Fig. S17 versus Fig. S8), indicating partial removal of active Fe single atoms. The rapid Fe and activity loss in highly porous Mg-templated samples aligns with our prior reports of rapid dissolution from exposed FeNx sites, as confirmed by online ICP-MS in both flow cell and gas diffusion electrode setups.46 Moreover, Fig. S9a demonstrates an increase in C–O, C
8)900@Fe shows retained morphology and some single atoms remaining (Fig. S16), with EDXS demonstrating reduced Fe signal (Fig. S17 versus Fig. S8), indicating partial removal of active Fe single atoms. The rapid Fe and activity loss in highly porous Mg-templated samples aligns with our prior reports of rapid dissolution from exposed FeNx sites, as confirmed by online ICP-MS in both flow cell and gas diffusion electrode setups.46 Moreover, Fig. S9a demonstrates an increase in C–O, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and CxZyOw species with increasing Mg precursor. Based on previous post-mortem Raman and temperature programmed desorption on equivalently Mg-templated N–Cs,45,56 carbon oxidation likely further contributes to instability and reduced turnover frequency,42 as amorphous and defective graphitic domains (Fig. S4) are preferentially corroded within the 0.8–0.4 VRHE.57 N2-saturated cyclic voltammograms confirms this: only ZIFMg(1
O and CxZyOw species with increasing Mg precursor. Based on previous post-mortem Raman and temperature programmed desorption on equivalently Mg-templated N–Cs,45,56 carbon oxidation likely further contributes to instability and reduced turnover frequency,42 as amorphous and defective graphitic domains (Fig. S4) are preferentially corroded within the 0.8–0.4 VRHE.57 N2-saturated cyclic voltammograms confirms this: only ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8)900@Fe displays the clear emergence of ill-defined redox, indicative of quinone/hydroquinone species resulting from carbon oxidation (Fig. S18).58 In summary, this is indicative that the Mg-templating process, which generates porosity under these conditions, yields N–C (and subsequent Fe–N–C) that are oxidised and prone to oxidation and corrosion within the applied potential range used for the AST. Consequently, unlike Mg acetylacetonate doping during ZIF synthesis reported by Wu et al.,47 no Mg stabilisation effect is observed here. This underscores the timing, type and mechanism of Mg introduction critically determine the balance between activity and durability in Fe–N–C catalysts.
8)900@Fe displays the clear emergence of ill-defined redox, indicative of quinone/hydroquinone species resulting from carbon oxidation (Fig. S18).58 In summary, this is indicative that the Mg-templating process, which generates porosity under these conditions, yields N–C (and subsequent Fe–N–C) that are oxidised and prone to oxidation and corrosion within the applied potential range used for the AST. Consequently, unlike Mg acetylacetonate doping during ZIF synthesis reported by Wu et al.,47 no Mg stabilisation effect is observed here. This underscores the timing, type and mechanism of Mg introduction critically determine the balance between activity and durability in Fe–N–C catalysts.
Conclusions
Increasing MgCl2·6H2O precursor with ZIF-8 led to atomically thin carbon sheets, and reduced N content, shifting from predominantly pyridinic to pyrrolic N species. Clear increases in large micropores and mesopores were observed, which correlated strongly with improved electrocatalytic activity, culminating in a peak kinetic mass activity of 3.74 ± 1.14 A g−1 at 0.80 VRHE,iR-free for ZIFMg(1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8)900@Fe. Despite the high activity and low peroxide yield, this catalyst displayed the most significant degradation during the AST. This is related to porosity via increased (1) risk of Fe dissolution from increased exposure and (2) amounts of oxidised and oxidisable carbon species from Mg-templating. This reveals the multifaceted role of porosity on electrocatalyst performance.
8)900@Fe. Despite the high activity and low peroxide yield, this catalyst displayed the most significant degradation during the AST. This is related to porosity via increased (1) risk of Fe dissolution from increased exposure and (2) amounts of oxidised and oxidisable carbon species from Mg-templating. This reveals the multifaceted role of porosity on electrocatalyst performance.
Author contributions
A. P. conceptualised the work and wrote the initial draft and led the investigation. A. P. synthesised the materials and performed XRD, Raman, N2 sorption, EPR, ICP-MS, and electrochemical testing and their analysis. J. Z. performed SEM measurements. R. H. provided TGA-MS measurements and Raman analysis. J. P. performed HAADF-STEM and EDXS. J. B. carried out XPS measurements and analysis. J. B., S. J. H., M.-M. T., and I. E. L. S. provided project supervision, and funded the project and resources. All authors contributed to reviewing and editing the manuscript.Conflicts of interest
There are no conflicts of interest to declare.Data availability
Experimental data files from the manuscript are available at BAM Publica: https://doi.org/10.26272/opus4-65228.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5ma01488c.
Acknowledgements
The authors acknowledge financial support from the Engineering and Physical Sciences Research Council (EPSRC) (EP/W031019/1, EP/S023259/1 EP/W03395X/1 and EP/Y024303/1), the European Research Council (ERC) under the European Union's Horizon 2020 research and innovation programme (grant agreement no. 866402 and no. 715502, EvoluTEM). A. P. thanks the EPSRC Centre for Doctoral Training in the Advanced Characterisation of Materials (grant number EP/L015277/1) and EPSRC Doctoral Prize Fellowship (EP/W524323/1). J. B. acknowledges financial support from Imperial College London through the Imperial College Research Fellowship. Electron microscopy access was supported by the Henry Royce Institute for Advanced Materials, funded through EPSRC grants EP/R00661X/1, EP/S019367/1, EP/P025021/1 and EP/P025498/1.References
- H. A. Gasteiger, S. S. Kocha, B. Sompalli and F. T. Wagner, Appl. Catal., B, 2005, 56, 9–35 CrossRef CAS.
- K. Jiao, J. Xuan, Q. Du, Z. Bao, B. Xie, B. Wang, Y. Zhao, L. Fan, H. Wang, Z. Hou, S. Huo, N. P. Brandon, Y. Yin and M. D. Guiver, Nature, 2021, 595, 361–369 CrossRef CAS PubMed.
- A. Pedersen, J. Pandya, G. Leonzio, A. Serov, A. Bernardi, I. Stephens, M.-M. Titirici, C. Petit and B. Chachuat, Green Chem., 2023, 25, 10458–10471 RSC.
- J. J. Baschuk and X. Li, Int. J. Energy Res., 2001, 25, 695–713 CrossRef CAS.
- R. Mohtadi, W.-K. Lee and J. W. Van Zee, J. Power Sources, 2004, 138, 216–225 CrossRef CAS.
- D. Malko, T. Lopes, E. Symianakis and A. R. Kucernak, J. Mater. Chem. A, 2015, 4, 142–152 Search PubMed.
- W. Chaikittisilp, M. Hu, H. Wang, H.-S. Huang, T. Fujita, K. C.-W. Wu, L.-C. Chen, Y. Yamauchi and K. Ariga, Chem. Commun., 2012, 48, 7259–7261 RSC.
- N. Y. Gugin, J. A. Villajos, O. Dautain, M. Maiwald and F. Emmerling, ACS Sustainable Chem. Eng., 2023, 11, 5175–5183 CrossRef CAS.
- A. Deacon, L. Briquet, M. Malankowska, F. Massingberd-Mundy, S. Rudić, T. I. Hyde, H. Cavaye, J. Coronas, S. Poulston and T. Johnson, Commun. Chem., 2022, 5, 1–10 Search PubMed.
- A. Pedersen, A. Bagger, J. Barrio, F. Maillard, I. Stephens and M.-M. Titirici, J. Mater. Chem. A, 2023, 11, 23211–23222 RSC.
- K. S. Park, Z. Ni, A. P. Côté, J. Y. Choi, R. Huang, F. J. Uribe-Romo, H. K. Chae, M. O’Keeffe and O. M. Yaghi, Proc. Natl. Acad. Sci. U. S. A., 2006, 103, 10186–10191 CrossRef CAS PubMed.
- Z. Zheng, Z. Rong, H. L. Nguyen and O. M. Yaghi, Inorg. Chem., 2023, 62, 20861–20873 CrossRef CAS PubMed.
- J. Castells-Gil, J. Zhu, I. Itskou, E. H. Wolpert, R. D. Hunter, J. P. Tidey, A. Pedersen, E. Solvay, H. Tyrrell, C. Petit and J. Barrio, J. Mater. Chem. A, 2025, 13, 28006–28018 RSC.
- V. Armel, S. Hindocha, F. Salles, S. Bennett, D. Jones and F. Jaouen, J. Am. Chem. Soc., 2017, 139, 453–464 CrossRef CAS PubMed.
- G. W. C. Kaye, D. Ewen and R. T. Glazebrook, Proc. R. Soc. London, Ser. A, 1997, 89, 58–67 Search PubMed.
- Q. Wang, T. Ina, W.-T. Chen, L. Shang, F. Sun, S. Wei, D. Sun-Waterhouse, S. G. Telfer, T. Zhang and G. I. N. Waterhouse, Sci. Bull., 2020, 65, 1743–1751 CrossRef CAS PubMed.
- F. Jaouen, E. Proietti, M. Lefèvre, R. Chenitz, J.-P. Dodelet, G. Wu, H. Taek Chung, C. Marie Johnston and P. Zelenay, Energy Environ. Sci., 2011, 4, 114–130 RSC.
- Z. Yang, Y. Chen, S. Zhang and J. Zhang, Adv. Funct. Mater., 2023, 33, 2215185 CrossRef CAS.
- D. Menga, F. Ruiz-Zepeda, L. Moriau, M. Šala, F. Wagner, B. Koyutürk, M. Bele, U. Petek, N. Hodnik, M. Gaberšček and T. Fellinger, Adv. Energy Mater., 2019, 9, 1902412 CrossRef CAS.
- H. J. T. Ellingham, J. Soc. Chem. Ind., 1944, 63, 125–160 CrossRef CAS.
- R. D. Hunter, J. Ramírez-Rico and Z. Schnepp, J. Mater. Chem. A, 2022, 10, 4489–4516 RSC.
- L. Jiao, J. Li, L. L. Richard, Q. Sun, T. Stracensky, E. Liu, M. T. Sougrati, Z. Zhao, F. Yang, S. Zhong, H. Xu, S. Mukerjee, Y. Huang, D. A. Cullen, J. H. Park, M. Ferrandon, D. J. Myers, F. Jaouen and Q. Jia, Nat. Mater., 2021, 20, 1385–1391 CrossRef CAS PubMed.
- A. Mehmood, M. Gong, F. Jaouen, A. Roy, A. Zitolo, A. Khan, M. Sougrati, M. Primbs, A. M. Bonastre, D. Fongalland, G. Drazic, P. Strasser and A. Kucernak, Nat. Catal., 2022, 5, 311–323 CrossRef CAS.
- A. Pedersen, R. Z. Snitkoff-Sol, Y. Presman, J. Barrio, R. Cai, T. Suter, G. Yang, S. J. Haigh, D. Brett, R. Jervis, M.-M. Titirici, I. E. L. Stephens and L. Elbaz, J. Power Sources, 2024, 609, 234683 CrossRef CAS.
- Y. Huang, Y. Chen, M. Xu, A. Ly, A. Gili, E. Murphy, T. Asset, Y. Liu, V. De Andrade, C. U. Segre, A. L. Deriy, F. De Carlo, M. Kunz, A. Gurlo, X. Pan, P. Atanassov and I. V. Zenyuk, Mater. Today, 2023, 69, 66–78 CrossRef CAS.
- F. Jaouen, M. Lefèvre, J.-P. Dodelet and M. Cai, J. Phys. Chem. B, 2006, 110, 5553–5558 CrossRef CAS PubMed.
- Y.-C. Wang, W. Huang, L.-Y. Wan, J. Yang, R.-J. Xie, Y.-P. Zheng, Y.-Z. Tan, Y.-S. Wang, K. Zaghib, L.-R. Zheng, S.-H. Sun, Z.-Y. Zhou and S.-G. Sun, Sci. Adv., 2022, 8, eadd8873 CrossRef CAS PubMed.
- M. Mazzucato, G. Daniel, A. Mehmood, T. Kosmala, G. Granozzi, A. Kucernak and C. Durante, Appl. Catal., B, 2021, 291, 120068 CrossRef CAS.
- L. Mazzoli, A. Pedersen, S. Kellner, R. D. Hunter, R. Cai, M. Wang, K. Sivula, S. J. Haigh and J. Barrio, Green Chem., 2024, 26, 3271–3280 RSC.
- X. Wan, X. Liu, Y. Li, R. Yu, L. Zheng, W. Yan, H. Wang, M. Xu and J. Shui, Nat. Catal., 2019, 2, 259–268 CrossRef CAS.
- J. Pampel and T.-P. Fellinger, Adv. Energy Mater., 2016, 6, 1502389 CrossRef.
- A. Mehmood, J. Pampel, G. Ali, H. Y. Ha, F. Ruiz-Zepeda and T.-P. Fellinger, Adv. Energy Mater., 2018, 8, 1701771 CrossRef.
- J. Pampel, A. Mehmood, M. Antonietti and T.-P. Fellinger, Mater. Horiz., 2017, 4, 493–501 RSC.
- K. Kisand, A. Sarapuu, J. C. Douglin, A. Kikas, M. Käärik, J. Kozlova, J. Aruväli, A. Treshchalov, J. Leis, V. Kisand, K. Kukli, D. R. Dekel and K. Tammeveski, ChemSusChem, 2025, 18, e202401332 CrossRef CAS PubMed.
- U. Sajjad, A. Sarapuu, J. C. Douglin, A. Kikas, A. Treshchalov, M. Käärik, J. Kozlova, J. Aruväli, J. Leis, V. Kisand, K. Kukli, D. R. Dekel and K. Tammeveski, ACS Catal., 2024, 14, 9224–9234 CrossRef CAS.
- F. O. Ibrahim, K. Kisand, J. C. Douglin, A. Sarapuu, A. Kikas, M. Käärik, J. Kozlova, J. Aruväli, A. Treshchalov, J. Leis, V. Kisand, K. Kukli, K. Yassin, D. R. Dekel and K. Tammeveski, Chem. Eng. J., 2025, 510, 161560 CrossRef CAS.
- J. Barrio, A. Pedersen, S. Ch Sarma, A. Bagger, M. Gong, S. Favero, C. Zhao, R. Garcia-Serres, A. Y. Li, Q. Zhang, F. Jaouen, F. Maillard, A. Kucernak, I. E. L. Stephens and M. Titirici, Adv. Mater., 2023, 35, 2211022 CrossRef CAS PubMed.
- E. Petitdemange, J. Zhu, A. Pedersen, J. Parker, S. E. Balaghi, S. Li, S. Favero, J. I. Martínez, S. Haigh, M.-M. Titirici, A. Fischer and J. Barrio, Adv. Funct. Mater., 2025, e18944 Search PubMed.
- K. Sugimoto, R. E. Dinnebier and J. C. Hanson, Acta Crystallogr., Sect. B: Struct. Sci., 2007, 63, 235–242 CrossRef CAS PubMed.
- A. Mehmood, G. Ali, B. Koyutürk, J. Pampel, K. Y. Chung and T.-P. Fellinger, Energy Storage Mater., 2020, 28, 101–111 CrossRef.
- C. H. Choi, H. K. Lim, M. W. Chung, G. Chon, N. Ranjbar Sahraie, A. Altin, M. T. Sougrati, L. Stievano, H. S. Oh, E. S. Park, F. Luo, P. Strasser, G. Dražić, K. J. J. Mayrhofer, H. Kim and F. Jaouen, Energy Environ. Sci., 2018, 11, 3176–3182 RSC.
- G. Bae, M. M. Kim, M. H. Han, J. Cho, D. H. Kim, M.-T. Sougrati, J. Kim, K.-S. Lee, S. H. Joo, W. A. Goddard, H.-S. Oh, H. Kim, F. Jaouen and C. H. Choi, Nat. Catal., 2023, 6, 1140–1150 CrossRef CAS.
- L. Liu, X. Rao, S. Zhang and J. Zhang, Chem, 2024, 10, 1994–2030 CAS.
- S. Liu, Q. Meyer, C. Jia, S. Wang, C. Rong, Y. Nie and C. Zhao, Energy Environ. Sci., 2023, 16, 3792–3802 RSC.
- A. Pedersen, R. Z. Snitkoff-Sol, Y. Presman, L. Dubau, R. Cai, J. Barrio, S. J. Haigh, F. Maillard, I. E. L. Stephens, M.-M. Titirici and L. Elbaz, Adv. Energy Mater., 2025, 15, 2403920 CrossRef CAS.
- A. Pedersen, K. Kumar, Y.-P. Ku, V. Martin, L. Dubau, K. T. Santos, J. Barrio, V. A. Saveleva, P. Glatzel, V. K. Paidi, X. Li, A. Hutzler, M.-M. Titirici, A. Bonnefont, S. Cherevko, I. E. L. Stephens and F. Maillard, Energy Environ. Sci., 2024, 17, 6323–6337 RSC.
- X. Li, J. Li, G. Wu and S. Litster, presented in part at the Annual Merit Review, 2023.
- T. Jawhari, A. Roid and J. Casado, Carbon, 1995, 33, 1561–1565 CrossRef CAS.
- N. A. H. M. Nordin, A. F. Ismail, A. Mustafa, R. S. Murali and T. Matsuura, RSC Adv., 2014, 4, 52530–52541 RSC.
- J. Zhu, A. Pedersen, S. Kellner, R. D. Hunter and J. Barrio, Commun. Chem., 2025, 8, 1–8 Search PubMed.
- D. Menga, J. L. Low, Y.-S. Li, I. Arčon, B. Koyutürk, F. Wagner, F. Ruiz-Zepeda, M. Gaberšček, B. Paulus and T.-P. Fellinger, J. Am. Chem. Soc., 2021, 143, 18010–18019 CrossRef CAS PubMed.
- A. Rieck, J. L. Low, S. Dietzmann, J. Radnik, Z. Teimouri, D. Higgins, D.-V. Hodoroaba, A. Mehmood and T.-P. Fellinger, ChemRxiv, 2025, preprint, chemrxiv-2025-s59s5 DOI:10.26434/chemrxiv-2025-s59s5.
- J. L. Low and B. Paulus, Catalysts, 2023, 13, 566 CrossRef CAS.
- J. Li, M. T. Sougrati, A. Zitolo, J. M. Ablett, I. C. Oğuz, T. Mineva, I. Matanovic, P. Atanassov, Y. Huang, I. Zenyuk, A. Di Cicco, K. Kumar, L. Dubau, F. Maillard, G. Dražić and F. Jaouen, Nat. Catal., 2021, 4, 10–19 CrossRef CAS.
- G. Bae, H. C. Kwon, M. H. Han, H.-S. Oh, F. Jaouen and C. H. Choi, ACS Catal., 2024, 8184–8192 CrossRef CAS.
- Z. Xu, A. Pedersen, S. Shimizu, T. Yoshii, H. Nishihara, M.-M. Titirici, J. Barrio and A. C. Forse, ChemRxiv, 2026, chemrxiv.10001886/v1 DOI:10.26434/chemrxiv.10001886/v1.
- L. Castanheira, L. Dubau, M. Mermoux, G. Berthomé, N. Caqué, E. Rossinot, M. Chatenet and F. Maillard, ACS Catal., 2014, 4, 2258–2267 CrossRef CAS.
- K. Kinoshita and J. A. S. Bett, Carbon, 1973, 11, 403–411 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |