 Open Access Article
Open Access ArticleTunable drug delivery via functionalized C18 nanorings: a DFT-MD investigation of 5-fluorouracil adsorption and release
Alaa M.
Khudhair
ab and
Ali
Ben Ahmed
 *bc
*bc
aDepartment of Physics, College of Science, University of Sumer, Iraq, Thi-Qar 64000, Iraq
bDepartment of Physics, College of Science, University of Sfax, Sfax 3000, Tunisia
cDepartment of Biomedical, Higher Institute of Biotechnology of Sfax, University of Sfax, Sfax 3000, Tunisia. E-mail: ali.benahmed@isbs.usf.tn
First published on 6th February 2026
Abstract
Nanomaterials have transformed the field of targeted drug delivery by providing exceptional surface-to-volume ratios, tunable electronic characteristics, and the potential for controlled release mechanisms. They can enhance treatment efficiency, reduce adverse effects, and increase bioavailability by precisely interacting with therapeutic molecules. This investigation investigates the electronic properties and adsorption behavior of 5-fluorouracil (FU) on pristine and doped C18 nanorings (B–C18, N–C18, and Si–C18) using density functional theory (DFT) considering these advantages. Stable and thermodynamically advantageous physisorption is confirmed by the calculated adsorption energies (Eads) of FU@B–C18 and FU@N–C18, which range from −0.772 eV to −0.930 eV. The band structure is effectively customized by doping, as evidenced by the narrowest band gap (0.463 eV) in FU@Si–C18 compared to 1.861 eV in FU@C18. This suggests that the electronic sensitivity has been enhanced. The Mulliken charge transfer (CT) analysis suggests that FU functions as an electron donor, with the highest charge transfer observed for FU@B–C18 (−0.060e). FU@B–C18 exhibits rapid desorption (9.3 s), while FU@N–C18 exhibits robust retention (4.2 × 103 s), indicating a balance between release efficiency and stability. Recovery time (τ) varies accordingly. The thermodynamic stability and structural integrity of the FU–C18 complexes are further confirmed by complementary molecular dynamics (MD) simulations, which guarantee dependable performance under physiological conditions. The potential of doped and functionalized C18 nanorings as advanced nanocarriers for FU cancer therapy is underscored by these findings.
1. Introduction
Cancer is a significant global health concern in the 21st century, being the second highest cause of mortality globally. Recent estimates from the World Health Organization indicate that cancer was responsible for almost 10 million deaths in 2020, with forecasts suggesting a persistent increase in incidence due to aging populations, environmental exposures, and changing lifestyle factors.1 The many characteristics of cancer, its ability to evade the immune system, and the development of treatment resistance all impede sustained therapeutic efficacy. Traditional chemotherapeutic techniques, while fundamental to clinical practice, often exhibit limited therapeutic windows and systemic toxicity, highlighting the need for more targeted and effective delivery methods.2Nanotechnology has emerged as a breakthrough platform in oncology, allowing the strategic design of nanocarriers capable of encapsulating or adsorbing anticancer drugs, traversing biological barriers, and delivering their payload in a regulated and site-specific manner.3 Ring-shaped or cyclic nanostructures, including cyclodextrins and other macrocyclic compounds, are crucial for drug delivery because they can form inclusion complexes with drug molecules. Improved solubility, stability, and bioavailability are achieved by encapsulating medications that are inadequately water-soluble in their distinctive cavities. These structures can also facilitate controlled release and safeguard pharmaceuticals from enzymatic degradation. Macrocyclic carriers that are functionalized can be customized to accomplish site-specific delivery, which can reduce adverse effects and improve therapeutic efficacy. These characteristics render them valuable platforms for advanced pharmaceutical applications, particularly in the fields of cancer therapy and targeted drug release systems.4,5
Carbon-based nanomaterials, including carbon nanotubes, graphene derivatives, fullerenes, and lately carbon nanorings, have garnered significant attention owing to their distinctive physicochemical features, elevated surface area-to-volume ratios, and adjustable surface functions.6 Carbon nanorings, which are distinguished by their cyclic π-conjugated architecture of sp2-hybridized carbon atoms, provide a symmetrical substrate with a well-defined structure and strong π–π interaction potential for aromatic medicinal molecules, including 5-FU.7 In comparison to planar graphene fragments, their curvature and topological characteristics also confer unique electronic properties.
Heteroatom doping and functionalization of C18 nanorings constitute an advanced atomic-scale engineering approach, facilitating precision modulation of structural, electronic, and interfacial characteristics. The introduction of dopants, including boron (B), nitrogen (N), and silicon (Si), enables the modification of the material's band gap, charge distribution, and adsorption sites, thereby directly affecting the adsorption strength of 5-fluorouracil (5-FU), charge transfer (CT) processes, and drug-release characteristics. This modification improves the capacity of C18 nanorings to enable targeted drug delivery, thereby optimizing the system's stability and responsiveness. Furthermore, these alterations in electronic characteristics facilitate the effective modulation of interactions with therapeutic agents, thereby enhancing the material's efficacy in the treatment of cancer. The influence of heteroatom doping on these characteristics has been extensively reported within the existing literature concerning dopant-based atomic-scale functionalization and electronic structure modification.8–10
5-Fluorouracil (5-FU) is a widely used chemotherapeutic agent for various solid tumors, yet its clinical efficacy is limited by poor selectivity, systemic toxicity, and rapid metabolic clearance. These limitations necessitate the development of nanocarriers capable of enhancing 5-FU's bioavailability and enabling controlled release.11–13 Doping carbon-based nanomaterials with heteroatoms (e.g., B, N, Si) is a well-established approach to modulate surface reactivity, improve adsorption behavior, and facilitate selective interactions with drug molecules.
Several theoretical and experimental investigations have already analyzed the impact of heteroatom substitutions on drug delivery efficacy.14 Density functional theory (DFT) simulations have shown increased adsorption energies for anticancer medicines on nitrogen- or boron-doped graphene and carbon nanotube systems.15 Likewise, silicon-doped carbon frameworks have shown potential in modulating electrostatic interactions and enhancing drug retention in adverse biological conditions.16 Thus far, cyclic carbon nanorings have received little attention as a delivery platform, particularly regarding heteroatom doping and their relative efficacy as a 5-FU carrier.
Prior DFT-based investigations of drug carriers have mostly concentrated on expansive 2D sheets and inorganic nanocages (e.g., graphene oxide, Ca12O12) or heteroatomic rings/cages (e.g., C3N3/B3O3 and Al8N8/B8N8). This work investigates cyclocarbon (C18) nanorings, a relatively unexplored cyclic carbon structure, and quantifies the modulation of interaction with 5-fluorouracil (FU) by heteroatom doping (B, N, and Si). We provide a comprehensive DFT–MD study that correlates adsorption energetics and charge transfer with electrical response, including band structure/DOS trends and work-function (Φ) modulation. Additionally, we evaluate the thermal stability of FU@C18 complexes by NVT molecular dynamics at 310 K. This framework delineates structure–property principles for doping-modulated FU binding and electrical behavior in cyclic carbon nanorings.
In this study, density functional theory (DFT) calculations are employed to investigate the adsorption properties, electronic characteristics, and reactivity profiles of 5-FU on pristine C18 and its doped derivatives (B–C18, N–C18, Si–C18). Adsorption energies, band structures, density of states (DOS), global reactivity indices, Mulliken charge transfer, and molecular dynamics analyses were systematically performed to assess the influence of doping on FU binding affinity and carrier potential.
2. Computational methodology
We used ab initio calculations for computational design, structural improvement, and to check the main features of the structures we were looking at. These simulations used basis sets from a numerical linear combination of atomic orbitals (LCAO), supported by Quantum ATK software.17–20To accurately model how adsorbed molecules and the substrate interact, we used the Perdew–Burke–Ernzerhof (PBE) generalized gradient approximation (GGA), along with van der Waals corrections (Grimme DFT-D3 parameters) for iterative calculations.21–23 Computational methodology and ab initio molecular dynamics protocol. All density functional theory (DFT) calculations were conducted utilizing QuantumATK within the linear combination of atomic orbitals (LCAO) formalism. The simulation cell incorporated a vacuum region of 20 Å to mitigate artefactual interactions between periodic replicas. Brillouin zone sampling was performed at the Γ-point (1 × 1 × 1), and the real-space mesh cutoff was established at 150 Hartree. Convergence of the geometry optimizations was established when the total energy variation was less than 1 × 10−5 eV and the maximal residual force acting on any particle was below 0.01 eV Å−1. Furthermore, ab initio molecular dynamics (AIMD) simulations were conducted to evaluate the finite-temperature stability of the FU–C18 complexes within the NVT ensemble at 310 K, employing a Nosé–Hoover thermostat (coupling time of 100 fs), a time step of 1.0 fs, and a total trajectory duration of 10 ps (following a 1 ps equilibration period). The 10 ps AIMD simulation window predominantly assesses the short-term thermal stability and endurance of FU-C18 contacts; however, uncommon desorption events and long-term release kinetics, which occur on nanosecond to microsecond timescales, are not comprehensively characterized within this timeframe. Consequently, ab initio molecular dynamics (AIMD) simulations serve as a qualitative assessment of stability, thereby augmenting the static density functional theory (DFT) analysis of adsorption energetics and electronic response. We also added PseudoDojo potentials24 using the GGA for exchange and correlation.25 The calculations were done using molecular dynamics simulation methods.
The following formulas are used to determine the Fermi level energy (EFL) and electronic band gap (Egap):26–28
| Eg = ELUMO − EHOMO | (1) |
The adsorption energy (Eads) was determined by the following equation:29,30
| Eads = EFU–substrate − Esubstrate − EFU | (2) |
| IP = −EHOMO | (3) |
| EA = −ELUMO | (4) |
 | (5) |
 | (6) |
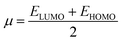 | (7) |
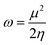 | (8) |
Consequently, Φ was determined from the electrostatic potential within the vacuum region and the Fermi level, as delineated in eqn (9).34 To quantify the modulation induced by adsorption, the percentage variation in the work function was determined using eqn (10), wherein Φb and Φa represent the work function of the substrate prior to and following FU adsorption, respectively:
| Φ = Vel(+∞) − EFL | (9) |
We quantified the effect of FU adsorption on the work function utilizing eqn (10).
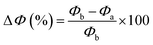 | (10) |
The discussion surrounding recovery time (τ) underscores its significance in both drug delivery systems. By illuminating the relationship between recovery time and adsorption energy through traditional transition state theory, this study lays the groundwork for future technological advancements. Understanding these dynamics can lead to the creation of more responsive and efficient drug delivery methodologies. The insights gained from this exploration can drive innovations that significantly impact medical solutions:35,36
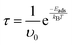 | (11) |
3. Results and discussion
3.1 Optimized geometries and structural features
The QuantumATK package was employed to conduct density functional theory (DFT) simulations to investigate the interactions between the FU anticancer drug and both pristine C18 and B-, N-, and Si-doped C18 substrates. The optimized geometries of the pristine and doped C18 structures are illustrated in Fig. 1 and 2, respectively, in both the absence and presence of the FU anticancer drug.The overall ring structure of C18 maintains stability upon doping with B, N, or Si atoms, but some local distortions occur at the dopant site. The impact is more significant in the Si-doped structure owing to the greater atomic radius of silicon relative to carbon, as shown in Fig. 1.
The substrate comprises 18 carbon atoms (C18) and is doped with diverse elements, such as boron, nitrogen, and silicon (B, N, Si). The optimized configurations, as illustrated in Fig. 2, indicate that the FU drug interacts with the pristine C18 substrate at distances of 2.89 Å (between the oxygen atom of the drug and a carbon atom of the substrate) and 2.80 Å (between the hydrogen atom of the drug and a carbon atom of the substrate). The O–B and H–C distances for the B-doped C18 system are 2.93 Å and 3.03 Å, respectively. In contrast, the O–N and H–C distances for the N-doped substrate are both 2.97 Å. In contrast, the Si-doped substrate exhibits interaction distances of 2.82 Å (H–C) and 3.13 Å (O–Si).
In addition, for the lowest energy structures, vibrational calculations were performed inside the harmonic approximation to confirm the identification of local minima and as a measure of structural stability; the results showed positive values, as shown in Fig. 3 and 4.
3.2 The electronic properties
The band structure diagrams in Fig. 5 and 6(a)–(d) demonstrate significant changes in the electronic characteristics of the various functionalized and doped C18-based structures.The total energy (E) values in Table 1 provide essential insights into the thermodynamic stability of the virgin, doped, and FU-functionalized C18 structures. Pristine C18 demonstrates the lowest total energy among the analyzed systems, indicating its fundamentally stable form. Doping with B, N, or Si results in a little elevation in total energy, attributable to lattice deformation and the incorporation of impurity atoms that alter the local electronic environment. Among the doped systems, Si–C18 exhibits the most significant rise in E, indicative of Si's larger atomic radius relative to C and the enhanced structural relaxation necessary for stability. For FU-functionalized C18 (FU@C18), the total energy is marginally less negative than that of pristine C18, indicating that the adsorption of FU groups induces more structural strain and electron redistribution. The combination of FU functionalization with doping (FU@B–C18, FU@N–C18, FU@Si–C18) results in an increased total energy, indicating that the synergistic effects of functionalization and doping need more structural modifications to attain equilibrium. Nonetheless, the total energy remains negative across all configurations, affirming their thermodynamic viability and promise for experimental realization. These patterns suggest that whereas doping and FU functionalization somewhat diminish the thermodynamic stability compared to pure C18, they provide considerable electrical tunability, as shown by the band structure, HOMO–LUMO levels, and DOS profiles.
| Structure | E | E FL | Φ | ΔΦ (%) | E HOMO | E LUMO | E g | ΔEg (%) |
|---|---|---|---|---|---|---|---|---|
| C18 | −2799.183 | −7.766 | 7.766 | — | −8.460 | −6.991 | 1.470 | — |
| B–C18 | −2719.441 | −7.713 | 7.713 | −0.68 | −7.714 | −6.827 | 0.887 | −39.66 |
| N–C18 | −2915.523 | −7.838 | 7.838 | 0.92 | −8.753 | −7.839 | 0.914 | −37.82 |
| Si–C18 | −2815.761 | −7.539 | 7.539 | −2.92 | −7.910 | −7.174 | 0.736 | −92.66 |
| FU@C18 | −5533.383 | −7.724 | 7.724 | −0.54 | −8.598 | −6.737 | 1.861 | 26.60 |
| FU@B–C18 | −5453.679 | −7.853 | 7.853 | 1.12 | −7.880 | −6.519 | 1.361 | −7.41 |
| FU@N–C18 | −5649.895 | −7.699 | 7.699 | −0.86 | −8.596 | −7.700 | 0.896 | −39.04 |
| FU@Si–C18 | −5550.038 | −7.621 | 7.621 | −1.86 | −7.872 | −7.409 | 0.463 | −68.50 |
The band structure of pure C18 has a significant energy gap of 1.47 eV, as shown in Table 1, signifying semiconducting characteristics with distinctly differentiated valence and conduction bands. Doping with B, N, or Si (B–C18, N–C18, and Si–C18) results in a significant reduction in the energy gap, with the minimum gap of 0.736 eV recorded for Si–C18. These substantial decreases, especially with Si doping (ΔEg = −92.66%), indicate that these dopants create states proximate to the Fermi level, hence promoting electronic transitions and improving conductivity.
Functionalization with FU groups (FU@C18), seen in Fig. 5a, results in an expansion of the energy gap (1.861 eV), suggesting that FU functionalization enhances the semiconducting properties of the structure. When FU groups are amalgamated with dopants (refer to Fig. 5b–d for FU@B–C18, FU@N–C18, and FU@Si–C18), the band gaps are significantly reduced (1.361 eV, 0.896 eV, and 0.463 eV, respectively), hence affirming the substantial impact of dopant-induced states that prevail over the influence of FU. The HOMO and LUMO level placements shown in Table 1 align with these patterns.
The HOMO and LUMO levels of N–C18 are nearer to the Fermi level than those of pure C18, indicating a decreased gap of 0.914 eV. In contrast, FU@C18 exhibits a more significant distinction between the HOMO and LUMO levels, aligning with its greater energy gap. These findings demonstrate that regulated doping (B, N, Si) and FU functionalization may effectively modify the electrical characteristics of the C18 structures. Doping lowers the band gap and improves conductivity, while FU functionalization may either restore or augment semiconducting properties, making these materials viable candidates for electrical and optoelectronic applications.
The density of states (DOS) profiles shown in Fig. 7 provide enhanced understanding of the electrical structure, supplementing the band structure diagrams presented in Fig. 5 and 6(a)–(d). The density of states (DOS) for pure C18 reveals a significant separation between the valence and conduction bands, exhibiting few states at the Fermi level, hence affirming its semiconducting characteristics and a band gap of 1.47 eV (Table 1). This characteristic aligns with other carbon-based nanostructures, whereby a substantial HOMO–LUMO gap enhances electrical stability and diminishes intrinsic conductivity.
 | ||
| Fig. 7 Density of states (DOS) of pristine and doped graphene surfaces before and after FU adsorption. | ||
Doping with heteroatoms like B, N, or Si significantly modifies the density of states, creating localized states near the Fermi energy. This leads to a significant decrease in the band gap, particularly for Si–C18 (0.736 eV), where distinct peaks at EF signify improved conductivity owing to the heightened density of available states. Comparable phenomena have been found in doped graphene systems.
In FU-functionalized C18 (FU@C18), the band gap rises to 1.861 eV, accompanied by a reduction in states at the Fermi level, hence enhancing its semiconducting characteristics. The synergistic impact of FU groups and doping (FU@B–C18), FU@N–C18 FU@Si–C18 reintroduces states next to the Fermi level, reducing the energy gap and altering the distribution of the density of states. This illustrates how doping and functionalization may collaboratively regulate the band structure and density of states (DOS).
3.3 Adsorption energy and charge transfer
The adsorption energy (Eads) and charge transfer (CT) are essential metrics for comprehending the interaction between a molecule and a substrate, as they directly influence the stability, electronic characteristics, and possible sensing efficacy of the system. Eads assesses the thermodynamic favorability and binding strength of adsorption, whereas CT measures the degree of electronic exchange, offering critical insights into reactivity and surface activity.The adsorption energies shown in Table 2 are uniformly negative, indicating that FU adsorption on both pure and doped C18 surfaces is exothermic and thermodynamically advantageous. FU@N–C18 demonstrates the most robust interaction (−0.930 eV), ascribed to the significant electronic coupling resulting from nitrogen doping. Conversely, FU@B–C18 has the least negative Eads (−0.772 eV), signifying weaker binding, whilst FU@C18 and FU@Si–C18 have intermediate values (−0.824 eV and −0.812 eV), illustrating that doping efficiently modulates adsorption strength.
| Structures | E ads (eV) | BSSE correction | CT | τ (s) | σ (A m−2) |
|---|---|---|---|---|---|
| FU@C18 | −0.824 | 0.525 | −0.011 | 7 × 101 | 1.170 × 10−6 |
| FU@B–C18 | −0.772 | 0.630 | −0.060 | 9.3 × 100 | 1.964 × 10−2 |
| FU@N–C18 | −0.930 | 0.578 | −0.036 | 4.2 × 103 | 1.66 × 102 |
| FU@Si–C18 | −0.812 | 0.604 | −0.053 | 4.4 × 101 | 7.60 × 105 |
The Mulliken study reveals that charge transfer occurs from FU to the C18-based surfaces, as shown by the negative CT values. A negative CT indicates that the FU molecule functions as an electron donor, while the C18 surface operates as an electron acceptor, aligning with observations documented for analogous functionalized carbon systems.37 The maximum charge transfer (CT) is recorded for FU@B–C18 (−0.060e), followed by FU@Si–C18 (−0.053e), whilst FU@C18 and FU@N–C18 demonstrate lower charge transfers of −0.011e and −0.036e, respectively. This pattern indicates that dopants such as B and Si augment the charge exchange process, thus affecting the electrical reactivity of the surface. These results align with publications on doped carbon nanostructures, whereby charge transfer (CT) is crucial for improving sensing and catalytic efficacy. The sign of the charge transfer (CT) values indicates the donor–acceptor characteristics of the system. A negative CT value indicates that electrons are transported from the adsorbed molecule (FU) to the C18-based substrate, indicating that FU functions as the electron donor, whilst the C18 surface serves as the electron acceptor. If the CT value were positive, the substrate would function as the donor and the molecule as the acceptor. The current work indicates that all CT values are negative (Table 2), thus demonstrating that electron transport transpires from FU to both pure and doped C18 surfaces, aligning with donor–acceptor interactions documented in analogous carbon nanostructures.38,39
Electrical conductivity (σ) is a fundamental feature associated with adsorption behavior, charge transfer (CT), and band gap, indicating a material's capacity to conduct electricity. Table 2 illustrates that the conductivity (σ) of FU@C18 systems exhibits considerable variation, with FU@C18 measuring 1.170 × 10−6 A m−2, while FU@Si–C18 attains 7.60 × 105 A m−2. The reduced conductivity of FU@C18 aligns with its comparatively broad band gap (1.861 eV), limiting charge carrier movement. Conversely, the elevated conductivity of FU@Si–C18 is ascribed to the pronounced electron delocalization resulting from Si doping and a markedly decreased band gap of 0.463 eV. FU@N–C18 exhibits significant conductivity (1.66 × 102 A m−2), consistent with its intermediate band gap (0.896 eV) and elevated electron density. Boron doping in FU@B–C18 (σ = 1.964 × 10−2 A m−2) improves conductivity relative to FU@C18, however its impact is less significant than that of silicon or nitrogen doping. The observations demonstrate a distinct correlation among σ, CT, and band gap: reduced band gaps and enhanced electron transport (more negative CT) result in increased conductivity. The work function (ΔΦ) indirectly influences σ by affecting electron emission and carrier availability. The results indicate that Si and N doping significantly improve conductivity, essential for nanoelectronics, sensors, and catalytic systems. The tunability underscores the promise of FU-functionalized C18 derivatives as multifunctional materials, tailored for adsorption stability and electrical responsiveness.
3.4 Work function (Φ) and its modification
Recent DFT/MD investigations into nanoscale drug-carrier systems have underscored that adsorption affinity, and charge-transfer characteristics serve as valuable design parameters for modulating nanocarrier performance, including in the case of 5-fluorouracil. Building upon these recent advancements, we analyze the current FU–C1T findings by correlating adsorption energetics with charge transfer (CT) and the related electronic perturbation, whereas the work-function response is addressed independently in the dedicated Φ section.40–42The work function (Φ), which is the minimal energy required to extract an electron from the Fermi level to the vacuum, is a crucial parameter influencing the surface electrical behaviour, field emission characteristics, and sensing capacities of carbon-based nanomaterials.43 In these systems, little variations in Φ may substantially modify the electrical response, becoming a crucial element in the development of chemical sensors, nanoelectronics, and catalytic devices.44 Chemical perturbations, such as doping or molecule adsorption, cause charge redistribution and surface dipole production, leading to a shift in the Fermi level and observable alterations in Φ.45
The data presented in Table 1 indicate that pure C18 has a work function of 7.766 eV. Boron doping marginally decreases Φ to 7.713 eV (ΔΦ = −0.68%), signifying modest electron donation, whereas nitrogen doping elevates it to 7.838 eV (ΔΦ = +0.92%) owing to its electron-withdrawing properties.46 Silicon doping results in the most significant reduction, producing a Φ of 7.539 eV (ΔΦ = −2.92%), which correlates with a rise in surface electron density. The functionalization with FU groups further adjusts Φ, with FU@C18 demonstrating a marginally reduced value of 7.724 eV (ΔΦ = −0.54%). The variance becomes more pronounced when dopants are included, with FU@B–C18 attaining 7.853 eV (+1.12%), but FU@N–C18 and FU@Si–C18 decrease to 7.699 eV (−0.86%) and 7.621 eV (−1.86%), respectively47 (see Fig. 8).
 | ||
| Fig. 8 Percentage variation in work function (ΔΦ) and energy gap (ΔEg) for pristine, doped, and FU-functionalized C18 structures. | ||
An important discovery is that the sign and magnitude of ΔΦ are correlated with the charge transfer (CT) values. Systems exhibiting negative CT (electron transport from FU to the surface) often show a decrease in Φ because of the heightened surface electron density. Conversely, when ΔΦ is positive (e.g., FU@B–C18), the surface exhibits characteristics akin to an electron acceptor, indicating enhanced electron-withdrawing effects. A high ΔΦ promotes sensor sensitivity by elevating the energy barrier for electron emission, whereas a low ΔΦ boosts field emission and catalytic activity by augmenting electron availability.48
The electronic behavior of pristine and doped C18 systems is comprehensively understood through the interplay between adsorption energy (Eads), charge transfer (CT), and work function variation (ΔΦ). Stronger molecule–surface interactions are typically associated with a more negative Eads, which is frequently accompanied by higher CT values because of increased electronic coupling. Electron transfer from the FU molecule to the surface is indicated by negative CT values, and typically reduces Φ (negative ΔΦ) by increasing the density of surface electrons, as demonstrated in FU@Si–C18 and FU@N–C18. In contrast, systems with positive ΔΦ, such as FU@B–C18, demonstrate electron-withdrawing behavior, resulting in a slight increase in Φ and an enhanced sensing potential. The simultaneous tailoring of Eads, CT, and Φ can be strategically employed to optimize both adsorption stability and electronic response for targeted applications in sensing, catalysis, and nanoelectronics, as these correlations highlight.
3.5 Recovery time
The recovery time (τ) is a critical measure in assessing material performance for applications like medication administration and gas sensing, since it denotes the duration needed for the adsorbed FU molecule to disengage from the surface and restore the substrate to its original condition. The values of τ are directly affected by the adsorption energy (Eads) (see Fig. 9), with stronger adsorption generally leading to an extended recovery time owing to the enhanced stability of the adsorbate–substrate combination.Table 2 indicates notable discrepancies in recovery time between the pristine and doped C18 structures. FU@N–C18 has the greatest recovery duration (4.2 × 103 s), correlating with the highest adsorption energy (−0.930 eV). The robust interaction indicates that the desorption of FU from N-doped C18 may need a prolonged duration, thus limiting its use in scenarios requiring rapid reaction and regeneration. Conversely, FU@B–C18 exhibits the briefest recovery time (9.3 × 100 s) owing to its comparatively lower binding energy (−0.772 eV), rendering it beneficial for dynamic applications that need rapid adsorption and desorption cycles.
The intermediate values of τ for FU@C18 (7 × 101 s) and FU@Si–C18 (4.4 × 101 s) indicate moderate binding energies (−0.824 eV and −0.812 eV, respectively), suggesting a balanced compromise between stability and reversibility. These patterns underscore the need of customizing adsorption strength to get optimum recovery durations. Although elevated adsorption energies improve the stability of the drug-surface interface, they may impede desorption, thereby diminishing system efficiency, particularly in sensing and drug delivery applications. This behavior aligns with previous research on functionalized and doped carbon nanostructures, which has highlighted the regulation of adsorption strength as a crucial element for enhancing recovery dynamics.49
3.6 Global reactivity indices
The global reactivity indices (shown in Table 3) provide essential insights into the electrical reactivity and stability of both pure and modified C18 structures. These factors are often used to forecast chemical behavior, donor–acceptor proclivities, and the suitability of materials for catalytic and sensing applications. They originate from frontier molecular orbital theory.50,51| Structure | IP | EA | S | H | μ | ω |
|---|---|---|---|---|---|---|
| C18 | 8.460 | 6.991 | 0.680 | 0.735 | 7.725 | 40.609 |
| B–C18 | 7.714 | 6.827 | 1.128 | 0.443 | 7.270 | 59.598 |
| N–C18 | 8.753 | 7.839 | 1.094 | 0.457 | 8.296 | 75.265 |
| Si–C18 | 7.910 | 7.174 | 1.358 | 0.368 | 7.542 | 77.262 |
| FU@C18 | 8.598 | 6.737 | 0.537 | 0.930 | 7.668 | 31.594 |
| FU@B–C18 | 7.880 | 6.519 | 0.735 | 0.680 | 7.200 | 38.093 |
| FU@N–C18 | 8.596 | 7.700 | 1.116 | 0.448 | 8.148 | 74.096 |
| FU@Si–C18 | 7.872 | 7.409 | 2.161 | 0.231 | 7.641 | 126.142 |
The ionization potential (IP) and electron affinity (EA), denoting the energy necessary to remove an electron and the energy acquired by adding an electron, respectively, reflect electronic stability. The immaculate C18 structure has an ionization potential of 8.460 eV and an electron affinity of 6.991 eV, indicating considerable electronic stability. Doping with boron or silicon decreases the ionization potential (7.714 eV for B–C18 and 7.910 eV for Si–C18), signifying enhanced propensity for electron donation, advantageous for charge-transfer applications. Conversely, N–C18 has the greatest ionization potential (8.753 eV) and electron affinity (7.839 eV), indicating enhanced electron-accepting characteristics.
The oxidative stability of FU@C18 is enhanced by the slight elevation in ionization potential (8.598 eV) resulting from the functionalization with FU groups. The softness (S) and hardness (H) serve as inverse indicators of chemical reactivity, with higher S values signifying enhanced reactivity. Si–C18 demonstrates peak softness (1.358 eV−1) and minimal hardness (0.368 eV), indicating increased chemical reactivity. FU@C18 has the lowest softness (0.537 eV−1), indicating enhanced stability and less reactivity after FU functionalization. The electron-accepting characteristics of N–C18 (8.296 eV) and FU@N–C18 (8.148 eV) are shown by their elevated chemical potential (μ), indicating a propensity for electron ejection from the structure. In contrast, B–C18 and FU@B–C18 have reduced μ values (7.270 eV and 7.200 eV, respectively), corroborating their electron-donating characteristics.
The electrophilicity index (ω), which measures stability with the acquisition of an extra charge, differs across the systems. Si–C18 and FU@Si–C18 have the highest values (77.262 and 126.142, respectively), indicating robust electron uptake. The lowest ω (31.594) is recorded in FU@C18, indicating a reduction in electrophilicity attributed to FU passivation.
In general, these global reactivity indices indicate that doping, especially with Si or N, increases the electrical reactivity and electron-accepting capacity of C18, whereas FU functionalization stabilizes the structure and diminishes its electrophilicity. These characteristics render the systems exceptionally appropriate for electrical device engineering, catalysis, and sensing applications.52
3.7 Molecular dynamics
Molecular dynamics simulations were utilized to investigate the thermal stability of the FU@C18 complexes. Specifically, four independent NVT ab initio molecular dynamics (AIMD) trajectories were conducted at 310 K to assess whether the optimized adsorption configurations maintain their structural integrity under thermal fluctuations. The comprehensive AIMD protocol and all computational parameters, including the thermostat scheme, time step, and trajectory duration, are documented in Section 2 (Computational methodology) to consolidate the simulation details in a single section. The discussion below, therefore, concentrates on the observed trajectory behaviour and stability characteristics during the simulations.The four different molecular dynamics simulations (Fig. 10 and 11) consistently show that the FU delivery system undergoes rapid thermal and energetic equilibration after initialization. Randomized initial velocity distributions are associated with the abrupt temperature increases that are observed at the commencement of each cycle. According to thermodynamic predictions for solvated molecular systems53, the system stabilizes within sub-picosecond timescales, achieving equilibrium with the reservoir temperature within approximately 0.3–0.6 ps. A local energy minimum indicative of configurational relaxation is suggested by the stabilization of potential energy, which also occurs within similar timescales.
 | ||
| Fig. 10 Molecular dynamics trajectories depicting conformational transitions: potential energy stabilization. | ||
 | ||
| Fig. 11 Molecular dynamics trajectories depicting conformational transitions: temperature convergence. | ||
The consistent equilibration behavior seen in all four tests reinforces the reliability of the FU delivery arrangement. The efficacy of the Nosé–Hoover thermostat in properly modeling canonical ensemble dynamics has been substantiated in prior investigations,54 and a thermostat relaxation duration of 100 fs has shown effectiveness for thermal coupling without the introduction of artifacts. The observed thermal behavior corresponds with results from analogous research on drug encapsulation and release utilizing nanoscale carriers.55 Therefore, our simulations demonstrate that the FU delivery system maintains stable dynamic behavior during physiologically relevant heat circumstances, and the used methodological framework may be dependably utilized for future investigation of transport, diffusion, and absorption processes.
We observe that the 10 ps AIMD trajectories are predominantly designed to serve as a brief assessment of the thermal stability of the FU@C18 complexes. Extended stability, infrequent desorption events, and release kinetics may manifest over prolonged (nanoseconds to microseconds) time scales and are consequently outside the scope of the current simulation window.
3.8 Biological feasibility and environmental stability considerations
The biocompatibility of C18 nanorings has been investigated in numerous studies, demonstrating minimal cytotoxicity in human cells, thereby suggesting their potential utility in biomedical applications. The incorporation of heteroatoms, including boron (B), nitrogen (N), and silicon (Si), improves biocompatibility. These modifications subtly alter the electronic characteristics of the nanorings, thereby optimizing their interactions within biological systems. Furthermore, multiple investigations have indicated that C18 nanorings demonstrate robust stability within aqueous environments, exhibiting negligible degradation across brief to intermediate durations, thereby rendering them appropriate for drug delivery and sensing applications.56,57Nevertheless, the toxicity of C18 nanorings necessitates additional research, especially regarding prolonged exposure and potential bioaccumulation. Recent investigations indicate that surface functionalization employing specific moieties, including FU groups, may attenuate the toxicity of nanostructures by diminishing cellular harm and enhancing their selective absorption within target tissues.58,59 The effect of these alterations on the comprehensive toxicity profile necessitates assessment within more sophisticated in vitro and in vivo models.
To evaluate the stability under physiological conditions, molecular dynamics (MD) simulations were conducted to examine the behavior of C18 within simulated biological fluids. The simulations indicate that C18 nanorings exhibit considerable stability within biological environments over a brief duration. However, additional long-term simulations, extending from nanoseconds to microseconds, are required to comprehensively assess their behavior and degradation characteristics under authentic physiological conditions.59–61
4. Conclusion
In this study, DFT was employed to conduct a comprehensive theoretical analysis of the electronic properties and adsorption of 5-fluorouracil (FU) on pristine and doped C18 nanorings (B–C18, N–C18, and Si–C18). The results indicate that the electronic structure and adsorption characteristics of C18 nanorings can be precisely tailored through the use of doping and FU functionalization. The adsorption energies (Eads) that were calculated, which range from −0.772 eV for FU@B–C18 to −0.930 eV for FU@N–C18, confirm the thermodynamic stability and moderate binding strength of the compounds. These properties are perfect for reversible drug loading and release.The band gap has been significantly reduced, particularly for FU@Si–C18 (0.463 eV), which suggests that the electronic responsiveness has been improved. The recovery time (τ) analysis has revealed a complex equilibrium between rapid desorption (τ = 9.3 s for FU@B–C18) and strong adsorption (τ = 4.2 × 103 s for FU@N–C18). This allows for the tunable release of the drug. The Mulliken charge transfer (CT) analysis further demonstrates that FU functions as an electron donor, thereby modulating the surface reactivity of the nanorings. In addition, the structural stability of FU–C18 complexes under thermal fluctuations is further validated by complementary molecular dynamics (MD) simulations, which further supports their reliability for biomedical applications. The potential of doped C18 nanorings as versatile substrates for drug delivery and nanosensing is underscored by this work, which also offers fundamental insights into the adsorption mechanism of anticancer medications on carbon-based nanomaterials. The development of next-generation nanocarriers is facilitated by the combination of controllable electronic tuning and stable and reversible adsorption in these nanostructures. This approach allows for the optimization of efficiency, selectivity, and adaptability for advanced biomedical applications.
Author contributions
All authors contributed to the conception and design of the study. Alaa M. Khudhair performed material preparation, computations, data collection and analysis and Ali Ben Ahmed supervised this study. All authors discussed the results and contributed to reading and approving the final manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information (SI) is available. The optimized structures FU@C18, FU@B-C18, FU@N-C18 and FU@Si-C18. See DOI: https://doi.org/10.1039/d5ma01445j.References
- H. Sung, et al., Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries, CA-Cancer J. Clin., 2021, 71(3), 209–249 Search PubMed.
- S. K. Chakrabarti and D. Chattopadhyay, Cancer Immunotherapy: Bridging Concept with Practice–A Broad-Brush Stroke, Health, 2024, 5(3), 1108 Search PubMed.
- R. Singh and J. W. Lillard Jr, Nanoparticle-based targeted drug delivery, Exp. Mol. Pathol., 2009, 86(3), 215–223 CrossRef CAS PubMed.
- S. V. Kurkov and T. Loftsson, Cyclodextrins, Int. J. Pharm., 2013, 453(1), 167–180 CrossRef CAS PubMed.
- T. Loftsson and D. Duchene, Cyclodextrins and their pharmaceutical applications, Int. J. Pharm., 2007, 329(1–2), 1–11 Search PubMed.
- S. Iijima, Helical microtubules of graphite carbon, Nature, 1999, 56, 354 Search PubMed.
- S. Yamago, E. Kayahara and T. Iwamoto, Organoplatinum-Mediated Synthesis of Cyclic π-Conjugated Molecules: Towards a New Era of Three-Dimensional Aromatic Compounds, Chem. Rec., 2014, 14(1), 84–100 CrossRef CAS PubMed.
- H. Kurban, S. Alaei and M. Kurban, Effect of Mg content on electronic structure, optical and structural properties of amorphous ZnO nanoparticles: A DFTB study, J. Non-Cryst. Solids, 2021, 560, 120726–120732, DOI:10.1016/j.jnoncrysol.2021.120726.
- H. Kurban, P. Sharma, M. M. Dalkilic and M. Kurban, Accelerating density of states prediction in Zn-doped MgO nanoparticles via kernel-optimized weighted k-NN, Sci. Rep., 2025, 15(1), 30524, DOI:10.1038/s41598-025-07887-6.
- Z. Shariatinia and M. Kurban, Sustainable design of green-functionalized-conjugated BTBF-DPA derivatives for high-mobility hole transport, Tetrahedron, 2025, 188, 134984–134994, DOI:10.1016/j.tet.2025.134984.
- G. J. Ogunwale, et al., Interaction of 5-fluorouracil on the surfaces of pristine and functionalized Ca12O12 nanocages: an intuition from DFT, ACS Omega, 2023, 8(15), 13551–13568 CrossRef CAS PubMed.
- F. Safdari, H. Raissi, M. Shahabi and M. Zaboli, DFT calculations and molecular dynamics simulation study on the adsorption of 5-fluorouracil anticancer drug on graphene oxide nanosheet as a drug delivery vehicle, J. Inorg. Organomet. Polym. Mater., 2017, 27(3), 805–817 CrossRef CAS.
- Z. Yulin, O. Shuosi and J. Zhao, Potential application of pristine and Al-doped graphyne-like BN nanosheet for detection of anticancer fluorouracil drug, J. Mol. Model., 2020, 26(7), 169 Search PubMed.
- H. O. Edet, et al., Heteroatoms (B, N, S) doped quantum dots as potential drug delivery system for isoniazid: insight from DFT, NCI, and QTAIM, Heliyon, 2023, 9(1), e12599–e12614, DOI:10.1016/j.heliyon.2022.e12599.
- M. A. Ibrahim, et al., A comparative DFT investigation of the adsorption of temozolomide anticancer drug over beryllium oxide and boron nitride nanocarriers, ACS Omega, 2024, 9(23), 25203–25214 CrossRef CAS PubMed.
- Z. H. Al-Sawaff, S. S. Dalgic and F. Kandemirli, A DENSITY FUNCTIONAL THEORY (DFT) STUDY ON SILICON DOPED CARBON NANOTUBE SI–CNT AS A CARRIER FOR BMSF-BENZ DRUG USED FOR OSTEOPOROSIS DISEASE, Momento, 2022, 65, 1–24 Search PubMed.
- M. B. Kanoun and S. Goumri-Said, Insights into the impact of Mn-doped inorganic CsPbBr3 perovskite on electronic structures and magnetism for photovoltaic application, Mater. Today Energy, 2021, 21, 100796 CrossRef CAS.
- A. Shah, A. H. Munshi, A. P. Nicholson, A. Thiyagarajan, U. M. Pozzoni and W. S. Sampath, Atomistic modeling of energy band alignment in CdSeTe surfaces, Appl. Surf. Sci., 2021, 544, 148762 Search PubMed.
- A. M. Khudhair and A. Ben Ahmed, Adsorption Characteristics of the Anticancer Drug Hydroxyurea with Armchair BN Graphene Nanoribbons Containing and Lacking Vacancy Defects: Insight via DFT Calculations, J. Supercond. Novel Magn., 2024, 37(8), 1509–1518 CrossRef CAS.
- B. Chettri, P. K. Patra, Z. Renthlei, A. Laref and D. P. Rai, Bilayer heterostructure of boron nitride and graphene for hydrogen storage: a first-principles study, Energy Fuels, 2022, 36(21), 13307–13316 CrossRef CAS.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu, J. Chem. Phys., 2010, 132(15), 154104, DOI:10.1063/1.3382344.
- A. M. Khudhair, A. B. Ahmed, F. N. Ajeel and M. H. Mohammed, Theoretical investigation on the therapeutic applications of C2B and C2O as targeted drug delivery systems for hydroxyurea and 6-thioguanine in cancer treatment, Nano-Struct. Nano-Objects, 2024, 38, 101135 CrossRef CAS.
- J. Hostas and J. Rezac, Accurate DFT-D3 calculations in a small basis set, J. Chem. Theory Comput., 2017, 13(8), 3575–3585 CrossRef CAS PubMed.
- M. J. van Setten, et al., The PseudoDojo: Training and grading a 85 element optimized norm-conserving pseudopotential table, Comput. Phys. Commun., 2018, 226, 39–54 CrossRef CAS.
- J. P. Perdew, K. Burke and M. Ernzerhof, Generalized gradient approximation made simple, Phys. Rev. Lett., 1996, 77(18), 3865 CrossRef CAS PubMed.
- F. N. Ajeel, K. H. Bardan, S. H. Kareem and A. M. Khudhair, Pd doped carbon nanotubes as a drug carrier for Gemcitabine anticancer drug: DFT studies, Chem. Phys. Impact, 2023, 7, 100298 CrossRef.
- F. N. Ajeel, M. N. Mutier, K. H. Mohsin, S. K. Khamees, A. M. Khudhair and A. B. Ahmed, Theoretical Study on Electronic Properties of BN Dimers Doped Graphene Quantum dots, Bionanoscience, 2024, 1–9 Search PubMed.
- A. M. Khudhair, F. N. Ajeel and M. H. Mohammed, Theoretical study of the electronic and optical properties to design dye-sensitivity for using in solar cell device, Russ. J. Phys. Chem. B, 2018, 12, 645–650 CrossRef CAS.
- F. N. Ajeel, M. H. Mohammed and A. M. Khudhair, SWCNT as a model nanosensor for associated petroleum gas molecules: via DFT/B3LYP investigations, Russ. J. Phys. Chem. B, 2019, 13, 196–204 CrossRef CAS.
- F. N. Ajeel, Y. W. Ouda and A. M. Khudhair, Graphene Nanoflakes as a New Nanobiosensor for Histidine Amino Acid in Fish with Gas, Ethanol, DMSO and Water Environment, J. Basrah Res., 2019, 45(2A), 1–6 Search PubMed.
- A. M. Khudhair and A. Ben Ahmed, Pure and stone-wales defect armchair boron nitride graphene nanoribbons as anticancer drug delivery vehicles: a theoretical investigation, J. Cluster Sci., 2024, 35(2), 451–460 CrossRef CAS.
- F. Mollania, N. L. Hadipour and N. Mollania, CNT-based nanocarrier loaded with pyrimethamine for adipose mesenchymal stem cells differentiation and cancer treatment: The computational and experimental methods, J. Biotechnol., 2020, 308, 40–55 CrossRef CAS PubMed.
- S. Keshtkar, et al., A novel highly sensitive and selective H2S gas sensor at low temperatures based on SnO2 quantum dots-C60 nanohybrid: experimental and theory study, Talanta, 2018, 188, 531–539 CrossRef CAS PubMed.
- C. Xiao, K. Ma, G. Cai, X. Zhang and E. Vessally, Borophene as an electronic sensor for metronidazole drug: a computational study, J. Mol. Graphics Modell., 2020, 96, 107539 Search PubMed.
- N. L. Hadipour, A. Ahmadi Peyghan and H. Soleymanabadi, Theoretical study on the Al-doped ZnO nanoclusters for CO chemical sensors, J. Phys. Chem. C, 2015, 119(11), 6398–6404 Search PubMed.
- K. H. Bardan, F. N. Ajeel, M. H. Mohammed, A. M. Khudhair and A. B. Ahmed, DFT study of adsorption properties of the ammonia on both pristine and Si-doped graphene nanoflakes, Chem. Phys. Impact, 2024, 8, 100561 Search PubMed.
- M. Liedke, K. Potzger, N. Srinivasan and A. Wagner, Publications Repository-Helmholtz-Zentrum Dresden-Rossendorf.
- A. M. Khudhair and A. B. Ahmed, First investigation survey on the pristine C3N3 and B3O3 monolayer as a promising vehicle for delivery of 5-fluorouracil and cisplatin anticancer drugs, Chin. J. Phys., 2025, 95, 674–684 CrossRef CAS.
- A. M. Khudhair, I. Dhouib, F. N. Ajeel, A. B. Ahmed and B. Khemakhem, A Novel Al8N8 and B8N8 Nanoring Monolayer for Sensing and Drug Delivery of Cisplatin and Nitrosourea Anticancer Drugs: A DFT Insight, J. Inorg. Organomet. Polym. Mater., 2025, 1–14 Search PubMed.
- L. L. Bechohra, M. Kurban, N. E. H. Medigue and S. Kellou-Taïri, Drug delivery potential of carbon and boron nitride nanotubes: A DFT-D3 analysis of curcumin binding interactions, Diamond Relat. Mater., 2024, 149, 111626–111643, DOI:10.1016/j.diamond.2024.111626.
- A. Maurya, et al., Drug delivery potential of γ-graphyne, 6,6,12-graphyne and γ-graphdiyne for 5-Fluorouracil: insights from DFT calculations, Compos. Interfaces, 2025, 32(8), 1193–1213, DOI:10.1080/09276440.2025.2460350.
- A. Maurya, et al., A Novel Pyridopyrimidine Derivative as a Potential Breast Cancer Agent: DFT, Docking, MD Simulation, and Cytotoxic Studies, Chem. Biodiversity, 2025, 22(12), e01955, DOI:10.1002/cbdv.202501955.
- K.-i Murata and H. Tanaka, Liquid–liquid transition without macroscopic phase separation in a water–glycerol mixture, Nat. Mater., 2012, 11(5), 436–443 CrossRef CAS PubMed.
- T. Sahoo and P. Kale, Work function-based metal–oxide–semiconductor hydrogen sensor and its functionality: a review, Adv. Mater. Interfaces, 2021, 8(23), 2100649 CrossRef CAS.
- J.-X. Zhang and G.-H. Yang, Low-computation adaptive fuzzy tracking control of unknown nonlinear systems with unmatched disturbances, IEEE Trans. Fuzzy Syst., 2019, 28(2), 321–332 Search PubMed.
- S. Ajmal, et al., Electron deficient boron-doped amorphous carbon nitride to uphill N2 photo-fixation through π back donation, Appl. Catal., B, 2023, 321, 122070 CrossRef CAS.
- E. Caillosse, et al., Photo-induced self-assembly of silver nanoparticles for rapid generation of first and second surface mirrors, ACS Appl. Nano Mater., 2020, 3(7), 6531–6540 CrossRef CAS.
- L. Chen, J. T. Ren and Z. Y. Yuan, Enabling internal electric fields to enhance energy and environmental catalysis, Adv. Energy Mater., 2023, 13(11), 2203720 CrossRef CAS.
- G. Speranza, Carbon nanomaterials: Synthesis, functionalization and sensing applications, Nanomaterials, 2021, 11(4), 967 CrossRef CAS PubMed.
- P. Verma, A. Srivastava, P. Prajapati, P. Tandon and M. R. Shimpi, Molecular structure, hydrogen bonding interactions and docking simulations of nicotinamide (monomeric and trimeric models) by using spectroscopy and theoretical approach, Polycyclic Aromat. Compd., 2024, 44(3), 1537–1555 CrossRef CAS.
- O. C. Godfrey, et al., Impact of polar (DMSO, ethanol, water) solvation on geometry, spectroscopy (FT-IR, UV, NMR), quantum chemical parameters, and the antifungal activities of benzothiazole derivative by molecular docking approach, Chem. Phys. Impact, 2023, 7, 100349 CrossRef.
- R. Meenakshi, Spectral investigations, DFT based global reactivity descriptors, Inhibition efficiency and analysis of 5-chloro-2-nitroanisole as π-spacer with donor-acceptor variations effect for DSSCs performance, J. Mol. Struct., 2017, 1127, 694–707 Search PubMed.
- A. I. Väkeväinen, A. J. Moilanen, M. Nečada, T. K. Hakala, K. S. Daskalakis and P. Törmä, Sub-picosecond thermalization dynamics in condensation of strongly coupled lattice plasmons, Nat. Commun., 2020, 11(1), 3139 CrossRef PubMed.
- G. J. Martyna, M. L. Klein and M. Tuckerman, Nosé–Hoover chains: The canonical ensemble via continuous dynamics, The, J. Chem. Phys., 1992, 97(4), 2635–2643 CrossRef.
- D. M. Long, J. A. Greathouse, G. Xu and K. L. Jungjohann, Molecular dynamics simulation and cryo-electron microscopy investigation of AOT surfactant structure at the hydrated mica surface, Minerals, 2022, 12(4), 479 CrossRef CAS.
- S. K. Jana, N. N. Som and P. K. Jha, Size-dependent fullerenes for enhanced interaction of l-leucine: a combined DFT and MD simulations approach, Langmuir, 2024, 40(27), 13844–13859 CrossRef CAS PubMed.
- Z. Shariatinia and M. Kurban, Sustainable Design of Green-Functionalized π-Conjugated BTBF-DPA Derivatives for High-Mobility Hole Transport, Tetrahedron, 2025, 134984 CrossRef CAS.
- H. Kurban, P. Sharma, M. M. Dalkilic and M. Kurban, Accelerating density of states prediction in Zn-doped MgO nanoparticles via kernel-optimized weighted k-NN, Sci. Rep., 2025, 15(1), 30524 Search PubMed.
- R. Sainda, S. K. Jana and P. K. Jha, Multiscale simulation of pesticides adsorption on passivated carbon nanotubes, Diamond Relat. Mater., 2025, 113279 Search PubMed.
- A. Bunker and T. Róg, Mechanistic understanding from molecular dynamics simulation in pharmaceutical research 1: drug delivery, Front. Mol. Biosci., 2020, 7, 604770 CrossRef CAS PubMed.
- X. Xu, et al., Application of molecular dynamics simulation in self-assembled cancer nanomedicine, Biomater. Res., 2023, 27(1), 39 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |







