Open Access Article
Open Access ArticleBiocompatible melanin-functionalized CaCO3 nanoparticles for cell protection against photoinduced oxidative stress
Giuseppe Vitiello *ab,
Francesca Baldassarrec,
Chiara Boncristiani
*ab,
Francesca Baldassarrec,
Chiara Boncristiani de,
Alessandro Pezzella
de,
Alessandro Pezzella fg,
Giuseppina Luciania,
Giuseppe Ciccarella
fg,
Giuseppina Luciania,
Giuseppe Ciccarella cd and
Viviana Vergaro
cd and
Viviana Vergaro *e
*e
aDepartment of Chemical, Materials and Production Engineering, University of Naples Federico II, p.le V. Tecchio 80, 80125 Naples, Italy. E-mail: giuseppe.vitiello@unina.it
bCenter for Colloid and Surface Science, CSGI, Via della Lastruccia 3, Florence, 50019, Italy
cBiological and Environmental Sciences Department, UdR INSTM of Lecce University of Salento, Via Monteroni, 73100 Lecce, Italy
dInstitute of Nanotechnology CNR NANOTEC, Consiglio Nazionale delle Ricerche, Via Monteroni, 73100 Lecce, Italy
eDepartment of Experimental Medicine, University of Salento, Via Monteroni, 73100 Lecce, Italy. E-mail: viviana.vergaro@unisalento.it
fDepartment of Physics “Ettore Pancini”, University of Naples Federico II, Via Cintia 4, 80126 Naples, Italy
gInstitute of Polymers, Composites and Biomaterials, National Research Council, 80125 Naples, Italy
First published on 14th May 2026
Abstract
Melanin is a natural biopolymer with intrinsic antioxidant, photoprotective, and anti-inflammatory properties, making it a promising component for bioinspired functional materials. In this study, we report a green, one-pot synthesis of hybrid melanin–calcium carbonate nanoparticles (melanin–CaCO3 NPs) via in situ polymerization of 5,6-dihydroxyindole-2-carboxylic acid (DHICA) on amino-functionalized CaCO3 templates. The resulting nanoparticles combine the biocompatibility of the inorganic scaffold with the multifunctionality of melanin. Detailed physicochemical characterization confirmed the successful formation of the hybrid system, while in vitro studies on two human cell lines demonstrated excellent cytocompatibility, promotion of cell proliferation, and marked reactive oxygen species (ROS)-scavenging ability, achieving approximately 70% ROS reduction at a dose of 10 µg mL−1. Notably, even with a low melanin content (1%), the NPs exhibited significant intracellular photoprotective activity against photoinduced oxidative stress. These findings suggest that melanin–CaCO3 NPs hold strong potential as green-engineered, multifunctional nanomaterials for biomedical applications, particularly in oxidative stress-related therapies and photoprotection.
1. Introduction
Oxidative stress (OS) is a critical contributor to the onset and progression of numerous pathological states, as it is closely linked to a wide range of cellular and tissue injuries that adversely affect various organs and biological systems.1–3 OS is caused by the imbalance between the generation and clearance of oxidant species, which are mainly composed of reactive oxygen species (ROS).4–6 These species are generated through a combination of endogenous mechanisms – such as the activity of fibroblasts, epithelial and endothelial cells, immune cells, mitochondrial respiration, and NADPH oxidase – and exogenous influences, including exposure to environmental pollutants, radiation, toxic compounds, and certain pharmaceuticals. Prolonged exposure to UVA radiation is especially detrimental, as it enhances the intracellular buildup of ROS, exacerbates oxidative stress, and triggers molecular damage—manifesting in lipid peroxidation, alterations of the protein structure, and genomic instability within skin cells. These can significantly contribute to accelerated photoaging, weakened immune response to UV exposure, and the development of UV-related skin tumors.7,8 Under normal physiological conditions, human cells exploit their enzymatic and non-enzymatic antioxidant defence systems for maintaining the cellular redox balance.9 In the presence of inefficient and insufficient defence systems, the oxidant species can accumulate favouring serious pathological conditions such as diseases, disorders and aging with a progressive loss of function in tissues and organs.1In recent years, considerable research has focused on the creation of biocompatible nanomaterials that emulate natural systems and exhibit strong radical-scavenging capabilities.10–12 These materials play a key role in modulating the redox balance by neutralizing oxidant species and sustaining antioxidant defences, thereby contributing to cellular protection under physiological conditions. Current strategies have further evolved from simple radical scavenging to the engineering of intelligent 'synthetic organelles' capable of autoregulatory protection. For instance, recent work has demonstrated the potential of enzyme-loaded nanoreactors that not only scavenge ROS directly but also trigger endogenous glutathione biosynthesis in response to oxidative stress, ensuring durable maintenance of intracellular redox homeostasis.13 Consequently, there is increasing interest in the design and development of bio- and eco-compatible nanotechnological formulations aimed at protecting the skin and other tissues from the harmful effects of UV radiation. This includes the advancement of sunscreens and antioxidant-enriched skincare products with improved stability, effectiveness, and targeted delivery.14,15 In this context, melanin-like materials are considered highly promising candidates for these aims. They are polyphenolic-like pigments found in many different natural sources, from bacteria, fungi and plants to fish, mollusks, mammals, and humans, which are involved in a variety of biological functions and shield organisms from harmful environmental factors.16 Notably, melanins contribute significantly to physiological balance, especially by regulating functions in the skin, hair, and eyes and by offering protection against stress-related genetic damage.17,18 Due to their notable physicochemical properties—such as biocompatibility, ability to scavenge reactive oxygen and nitrogen species (RONS), broad-spectrum light absorption spanning UV and visible ranges, high photostability, heat conversion efficiency, electrical conductivity, and paramagnetism19—melanins are promising macromolecules for developing functional biomaterials aimed at skin protection,20 wound repair,21,22 and eco-friendly antioxidant, antibacterial,23,24 or photoresponsive biomedical applications.25 Interestingly, melanin precursors, such as 5,6-dihydroxyindole (DHI), 5,6-dihydroxyindole-2-carboxylic acid (DHICA), and dopamine (DA), have been successfully integrated at the molecular level with ceramic components – including titanium,26 silica,27 and cerium28 oxides, as well as doped hydroxyapatite29 – resulting in hybrid nanoplatforms that exhibit strong antimicrobial and antioxidant activities, along with notable bioactive and diagnostic properties. Recent advances in the design and tuning of such hybrid nanostructures, including core–shell and cloud-type morphologies, are discussed in detail in the work by Pota et al.,30 which explores how structural modifications can optimize functional properties for biomedical applications. Despite the significant progress in melanin–hybrid nanostructures, many existing systems remain constrained by the use of persistent, non-biodegradable inorganic cores, such as silica or metal oxides, which pose long-term biosafety concerns regarding tissue accumulation. Furthermore, most reported melanin–CaCO3 architectures rely on the physical adsorption of the pre-formed pigment or multi-step coating procedures that often result in limited interfacial stability and low radical-scavenging efficiency. There is a clear need for simpler, ‘green’ synthetic routes that can achieve a high degree of integration between the biopolymer and the inorganic scaffold. By leveraging the in situ oxidative polymerization of DHICA directly on CaCO3 templates, we overcome these hurdles, producing a highly stable, biomimetic hybrid that ensures maximum synergy between the mineral phase and the melanin network while maintaining full biodegradability. Beyond traditional ceramic hybrids, a variety of other nanostructures have been extensively explored for photoinduced ROS scavenging and photoprotective applications, demonstrating the diversity of the field. For instance, recent studies have highlighted the efficacy of specific polymer-based and metal-doped nanoparticles in mitigating oxidative stress under irradiation.31–33 However, despite the progress in these systems, CaCO3 stands out as a particularly compelling platform due to its unique combination of extreme biocompatibility, pH-dependent biodegradability, and high loading capacity for bioactive macromolecules. Naturally abundant and found in structures like mollusk shells, CaCO3 offers a safe and sustainable ‘green chemistry’ route that avoids the potential long-term accumulation or toxicity issues sometimes associated with synthetic metal-oxide or polymeric scaffolds.
Calcium carbonate (CaCO3), a naturally occurring inorganic material, is valued in biomedical contexts for its biocompatibility and ability to biodegrade safely. In diverse biomedical applications, CaCO3 shows significant potential, especially in drug delivery, tissue regeneration, and imaging techniques. CaCO3 nanoparticles can serve as adjuvants in vaccine formulations, helping to boost the immune response. Current studies aim to enhance the properties of these materials and broaden their applications to achieve better therapeutic results. In recent years, CaCO3 nanoparticles have been widely utilized in combination with imaging contrast agents and therapeutic compounds for diverse diagnostic and therapeutic applications.34 Naturally abundant, calcium carbonate is a major component of materials, such as mollusk shells, limestone, or eggshells, and is highly intriguing for driving advancements in novel biomedical systems in accordance with the safe and sustainable nanotechnology design principle. Further advantages include ease of preparation using gentle methods such as co-precipitation and their ability to biodegrade into harmless by-products at neutral pH, preventing the accumulation of these materials in the body over time.35 Furthermore, due to CaCO3's strong ability to capture bioactive molecules, it has been extensively utilized in biomedical research as a sacrificial template for creating functional materials to explore cellular vesicle acidification or enzyme compartmentalization effects in catalytic cascades.36,37 Through the use of CaCO3 nanoparticles combined with drugs, a variety of treatments such as chemical therapy, gene therapy, photothermal therapy (PTT)/photodynamic therapy (PDT), and immunotherapy can be accomplished.38 One of the unique features of these inorganic nanomaterials is that their intrinsic physical, chemical and biological properties can be easily tuned and enhanced by surface functionalization. By exploiting their surface functionalization, multifunctional nanosystems can easily achieve (i) modification of their original surface chemical termination, (ii) incorporation of new functional molecules onto the surface, or (iii) coating of the surface with an additional layer.34 This study focused on designing and synthesizing melanin-functionalized calcium carbonate nanoparticles (melanin–CaCO3 NPs) endowed with enhanced protective capabilities against oxidative stress, including that induced by light exposure. A feasible synthetic strategy was proposed to realize bio-functional nanostructures, in which crystalline nano-calcium carbonate platforms were combined with DHICA monomers to induce the oxidative polymerization to melanin. The formation, chemical composition, and structural properties of the hybrid nanoparticles were thoroughly characterized, along with evaluations of their biocompatibility, antioxidant activity, and reactive oxygen species (ROS) scavenging under light exposure, indicating strong prospects for biomedical uses.
2. Materials and methods
2.1 Materials
EDC (protein seq grade), NHS (98%), APTES (99.999%), ethanol (absolute, ≥99.8%), fluorescein isothiocyanate (FITC), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT), Hoechst 33342 (trihydrochloride and trihydrate), phosphate buffer saline (PBS) and Annexin V were purchased from Sigma-Aldrich and used as received. H2DCFDA was purchased from Thermo Fisher Scientific and used as received. The DHICA monomer and DHICA–melanin were prepared as described elsewhere.392.2 Synthesis of melanin–CaCO3 nanoparticles
CaCO3 nanocrystals (CaCO3-NCs) were prepared through a spray drying process as described in our previous work.35 Briefly, in spray drying, CaCl2 solution and NaHCO3 are mixed under controlled conditions, because the concentration of the precursor solutions ensures the optimal precipitation and nanocrystal formation. The pH of the solution is also crucial, as it can affect the size and morphology of the obtained CaCO3 crystals. Thanks to the high temperature, the liquid feed is atomized into fine droplets, which are then rapidly dried in a hot air stream. This process results in the formation of solid particles with a desired size distribution and morphology.Then, the surface of the obtained CaCO3 nanocrystals was functionalized with 3-aminopropyl-triethoxysilane (APTES), as previously described in the literature.40,41 In particular, a mixture of APTES (1.0 M) and CaCO3-NCs (0.150 g) in toluene was prepared and the reaction was allowed to proceed at 90 °C for 18 h in the dark under continuous stirring. Then, the amino-functionalized CaCO3 nanocrystals (named NH2-CaCO3-NCs) were recovered by centrifugation at 3500 rpm for 5 minutes, washed three times with distilled water and dried at 100 °C for 24 h. The NH2-CaCO3-NC weight was then calculated to obtain the NH2-CaCO3-NC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DHICA molar ratio of 2
DHICA molar ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 in the reaction mixture. The DHICA/NH2-CaCO3-NC precursor was synthesized by coupling DHICA carboxyl (–COOH) groups to amino groups of APTES molecules through EDC/NHS chemistry. EDC (0.021 mM), NHS (0.25 mM), and NH2-CaCO3-NCs (6.0 mg) were added to a DHICA deaerated solution (0.4 mM) in ethanol (43.2 mL) and water (9.00 mL). The reaction was allowed to proceed under stirring for 15 min at 4 °C in an ice bath. The pH of solution was measured (pH 10.5) to verify the alkalinity of experimental conditions. Alkaline experimental conditions facilitate the polymerization of melanin by promoting the oxidation of DHICA precursors, improving the interaction between melanin and inorganic nanoparticles, enhancing melanin's stability and structure, and enabling better control over the functionalization of nanoparticles. The NPs were recovered by centrifugation and repeatedly washed (5 times with distilled water).
1 in the reaction mixture. The DHICA/NH2-CaCO3-NC precursor was synthesized by coupling DHICA carboxyl (–COOH) groups to amino groups of APTES molecules through EDC/NHS chemistry. EDC (0.021 mM), NHS (0.25 mM), and NH2-CaCO3-NCs (6.0 mg) were added to a DHICA deaerated solution (0.4 mM) in ethanol (43.2 mL) and water (9.00 mL). The reaction was allowed to proceed under stirring for 15 min at 4 °C in an ice bath. The pH of solution was measured (pH 10.5) to verify the alkalinity of experimental conditions. Alkaline experimental conditions facilitate the polymerization of melanin by promoting the oxidation of DHICA precursors, improving the interaction between melanin and inorganic nanoparticles, enhancing melanin's stability and structure, and enabling better control over the functionalization of nanoparticles. The NPs were recovered by centrifugation and repeatedly washed (5 times with distilled water).
Fluorescent melanin–CaCO3 nanoparticles (melanin–CaCO3 NPs) were prepared by incubating NH2-CaCO3 nanocrystals (20 mg) with fluorescent isothiocyanate (0.4 mM) in an ethanol/water solution. The reaction was allowed to proceed for 6 h in the dark under continuous stirring. The chemical conjugation between CaCO3-NH2 and fluorescein isothiocyanate (FITC) typically involves a reaction between the amino group on the CaCO3-NH2 surface and the isothiocyanate group (–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S) of the FITC molecule. FITC is an isothiocyanate derivative, which contains a reactive –N
S) of the FITC molecule. FITC is an isothiocyanate derivative, which contains a reactive –N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S group. The primary amino group (NH2) on the surface of the CaCO3 particles can undergo a nucleophilic attack on the carbon atom of the isothiocyanate group of FITC. This reaction leads to the formation of a stable thiourea bond (–NH–C(S)–N–) between the CaCO3-NH2 surface and FITC. The reaction is often carried out under mildly alkaline conditions (pH 8–9), which promotes the deprotonation of the amino group (NH2), enhancing its nucleophilicity and thus improving the conjugation efficiency.42
S group. The primary amino group (NH2) on the surface of the CaCO3 particles can undergo a nucleophilic attack on the carbon atom of the isothiocyanate group of FITC. This reaction leads to the formation of a stable thiourea bond (–NH–C(S)–N–) between the CaCO3-NH2 surface and FITC. The reaction is often carried out under mildly alkaline conditions (pH 8–9), which promotes the deprotonation of the amino group (NH2), enhancing its nucleophilicity and thus improving the conjugation efficiency.42
2.3 Physicochemical characterization
2.4 Biological investigation
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 dilution. Prior to passaging, the culture medium was aspirated, the cells were rinsed with phosphate-buffered saline (PBS) and then detached with trypsin–EDTA. Following centrifugation at 1200 rpm for five minutes, the resulting cell pellet was resuspended in fresh culture medium.
5 dilution. Prior to passaging, the culture medium was aspirated, the cells were rinsed with phosphate-buffered saline (PBS) and then detached with trypsin–EDTA. Following centrifugation at 1200 rpm for five minutes, the resulting cell pellet was resuspended in fresh culture medium.2.5 Cell proliferation
As has been previously demonstrated in ref. 45, cell proliferation was evaluated in human fibroblasts (WS1) and human keratinocytes (HaCaT) using the Hoechst assay, which enables quantification of cellular DNA.46 Briefly, cells were incubated for 24 hours with 100 µg mL−1 of CaCO3 nanocrystals (CaCO3-NCs), as well as 100 µg mL−1 of NH2-functionalized CaCO3 nanocrystals (NH2-CaCO3-NCs) and melanin-coated CaCO3 nanoparticles (melanin–CaCO3 NPs). Following incubation, the cells were harvested from 12-well plates and suspended in 100 µL of Hoechst buffer (composed of 0.1 M NaCl, 1 mM EDTA, and 10 mM Tris, pH 7.4). To facilitate cell lysis and DNA release, the suspensions were subjected to five cycles of freezing and thawing. The assay was performed in black, fat-bottom plates with low background fluorescence to enhance detection sensitivity. Fluorescence measurements were obtained using a PerkinElmer LS50 spectrofluorometer (excitation at 360 nm and emission at 460 nm). A calibration curve was prepared using calf thymus DNA standards to quantify the DNA concentration. Each sample's DNA content, expressed in micrograms per milliliter (µg mL−1), was determined by comparing its fluorescence intensity to the standard curve, following the addition of 2 µL of Hoechst dye to each sample.2.6 Protective effect of melanin–CaCO3 NPs by flow cytometry analysis
As reported in our previous work48 to analyze the cytocompatibility and the protective effects of melanin–CaCO3 NPs against the cytotoxic effect of hydrogen peroxide in apoptosis induction, both HaCaT and WS1 human cells were incubated for 24 h with the melanin–CaCO3 NPs and, after washing out of the particles, were treated with 1 mM H2O2 for 30 min to induce cell death. After 24 h of stress induction, cells are extensively washed with PBS, gently collected using 1× PBS/2 mM EDTA and incubated with Annexin V (BD Biosciences) for 20 min at room temperature in the dark according to the manufacturer's instructions. Annexin V marks phosphatidylserine exposed on the outer leaflet of the plasma membrane of dying cells. CaCO3-NCs and NH2-CaCO3-NCs were also tested as control. Apoptosis (percentage of cells positive for annexin V) was measured with a FACS flow cytometer Accuri C6 (Becton Dickinson).3. Results and discussion
3.1 Chemical conjugation and physicochemical properties of melanin–CaCO3 NPs
The experimental approach developed to obtain melanin–CaCO3NPs is illustrated in Fig. 1. Melanin–CaCO3 NPs were obtained through the oxidative polymerization of DHICA (0.4 mM) on CaCO3 NPs functionalized with 3-aminopropyltrimethoxysilane (APTES). DHICA was selected as the melanin precursor due to its structural and electronic features. The presence of the carboxyl group affects both the polymerization pattern and the supramolecular organization, leading to eumelanin-like materials with distinct redox properties compared to DHI- or dopamine-derived melanins. This is particularly relevant for applications where controlled radical behavior and redox buffering capacity are required.More in detail, the synthetic approach relied at first on the formation of amino-functionalized CaCO3 NPs (6.0 mg)36 and then, the conjugation of APTES–DHICA was obtained through the coupling reaction between the amino group of APTES and the carboxyl group of DHICA using the N-(3-(dimethylamino)propyl)-N′-ethyl-carbodiimide hydrochloride (EDC)/N-hydroxy succinimide (NHS) reaction. The reaction parameters were adapted from existing studies,37 with a controlled low temperature to prevent unwanted side reactions.38 The reaction yield was 90% based on weight measurement. The alkaline experimental condition is guaranteed by the presence of CaCO3 alone. The pH plays a crucial role in the polymerization of DHICA to melanin on the inorganic nanoparticles: the functional groups on both the CaCO3 nanocrystals and melanin molecules can interact more efficiently under alkaline conditions. The pH can influence the redox reactions occurring in the precursor molecule; in fact, DHICA undergoes oxidation and polymerization under alkaline conditions. The hydroxyl groups on the precursor molecules are deprotonated at high pH, promoting the nucleophilic attack on the substrates and aiding the formation of reactive intermediates that drive melanin formation. Alkaline conditions stabilize certain intermediates and may facilitate better polymerization of melanin by enabling the aggregation of the oxidative products that eventually form the melanin structure.
To produce FITC-labeled melanin–CaCO3 nanoparticles, the amino-functionalized CaCO3 particles were allowed to react with fluorescein isothiocyanate (FITC). NP characterization was assessed using both dynamic light scattering (DLS) measurements and TEM images. DLS measurements were assessed for each step of synthesis procedure as summarized in Table 1. Naked CaCO3 nanocrystals in water tend to aggregate, as demonstrated by the higher hydrodynamic diameter and polydispersity index. The amino functionalization improves colloidal stability, as suggested by a lower PDI, and increases the surface charge to a positive value (+14.8 mV). Indeed, the oxidative polymerization of DHICA on amino-functionalized CaCO3 nanoparticles led to a significant modification of the ζ-potential value (−17.4 ± 3.8 mV), and the size seems to be smaller (323 ± 5.0 nm) probably due to the minor aggregation phenomena that occur in water, as confirmed by both the single broad population (Fig. S1) and the low PDI (0.15). The morphological characterization of melanin–CaCO3 NPs was conducted by TEM analysis, which revealed good sample preparation and dispersion of nanoparticles with an almost round-shaped nanostructures (Fig. S1 and Fig. 2a). According to the nanoparticle size distribution (Fig. 2b), almost 50% of analyzed nanoparticles show a dimension of about 104 ± 1 nm in diameter. This result compared to the DLS one confirmed the tendency of nanoparticles to self-aggregate in aqueous medium, leading to the formation of clusters of melanin–CaCO3 NPs with a mean size greater than that of single one as observed by TEM. Furthermore, the DLS measurements were performed for 21 days to verify the stability of melanin–CaCO3 synthetized NPs (Fig. S2). Fig. S2 indicates that DHICA functionalization enhances colloidal stability and reduces the tendency to aggregate, which are critical features for biomedical applications such as biodistribution and cellular uptake.
| Sample | Size (nm) | PDI (polydispersity index) | ζ-Potential (MV) |
|---|---|---|---|
| CaCO3-NC | 760.1 ± 1.6 | 0.426 | −4.5 ± 1.23 |
| NH2-CaCO3-NC | 427.2 ± 3.5 | 0.313 | +14.8 ± 0.70 |
| Melanin–CaCO3 NPs | 323 ± 5.0 | 0.15 | −17.4 ± 3.8 |
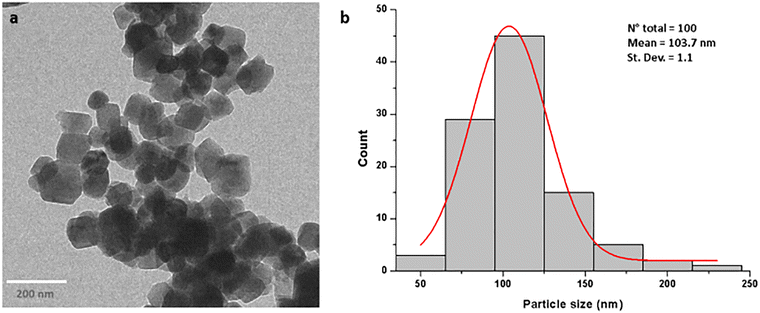 | ||
| Fig. 2 TEM image of melanin–CaCO3 NPs (a) and particle size distribution (b) obtained from the statistical analysis carried out after a random sampling of the nanoparticles. | ||
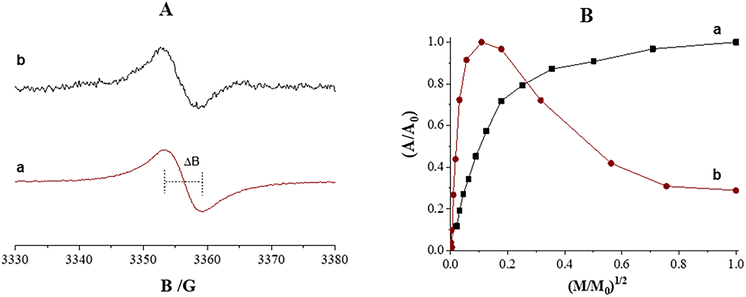
EPR analysis was performed to evaluate the polymerization of DHICA into melanin and to explore the chemical and structural features of the pigment component within the melanin–CaCO3 nanoparticles. As shown in Fig. 3A, the EPR spectrum of melanin–CaCO3 NPs exhibited a single symmetric signal centered at a g-factor of 2.0037 ± 0.0003, with a linewidth (ΔB) of 6.8 ± 0.2. These parameters, derived from the experimental spectrum described in Fig. 3A, provided indirect insight into the chemical environment and spatial distribution of paramagnetic centers within the pigment matrix. This signal is characteristic of carbon-centered radicals typical of melanin and melanin-like pigments, as previously reported.34,49,50 A similar EPR profile was observed for pure DHICA-derived melanin, showing a g-factor of 2.0035 ± 0.0003 and ΔB = 5.7 ± 0.2. The difference observed in the ΔB values confirms that the supramolecular organization of the melanin coating onto CaCO3 nanoparticles is directly influenced by its interaction with the inorganic matrix, in contrast to pure melanin. This interpretation is further supported by the distinct trends observed in the power saturation curves, as illustrated in Fig. 3B, indicating that melanin supported on CaCO3 exhibits a different microstructural organization, which may favour the exposure of redox-active sites, in agreement with previous evidence.37 Complementary to the paramagnetic characterization, the optical properties were investigated by UV-vis spectroscopy. The absorption spectrum (Fig. S4) exhibits a characteristic broadband profile with a monotonic increase towards the UV region, culminating in a maximum intensity around 300 nm. This profile is typical of melanin-like pigments and confirms the successful functionalization of the CaCO3 matrix. Importantly, this strong absorption in the UV range is a critical functional feature for the proposed application: it enables the nanoparticles to effectively act as ‘optical shields’ that filter high-energy radiation, thereby reducing the photo-oxidative burden on cells during the irradiation experiments described in Section 3.2.1. The close agreement of both the g-factor and ΔB values strongly supports the successful formation of melanin on the CaCO3 NPs. Moreover, the spin density was estimated to be equal to 1.87 × 1017 spins g−1, confirming the presence of melanin's radical centers and reflecting the total radical concentration per gram of nanostructured material, consistent with previously reported systems.28,50,51 Finally, thermogravimetric analysis (TGA) provided a quantitative estimate of the melanin associated with CaCO3 NPs, revealing a content of approximately 1%, as indicated by the weight loss (Fig. S5), thus confirming the low pigment loading in the hybrid melanin–CaCO3 NPs. On the other hand, FTIR/ATR indicates similarities between the spectra of uncoated and melanin-coated CaCO3 NPs (Fig. S6), which might be due to the fact that the main functional group of melanin is exposed in the same region of interest as CaCO3's ones, indirectly confirming that the coated sample is composed of a very low amount of pigment. All these findings clearly indicate that the inorganic CaCO3 matrix facilitated the oxidative polymerization of the DHICA precursor into melanin.
3.2 Biological assays
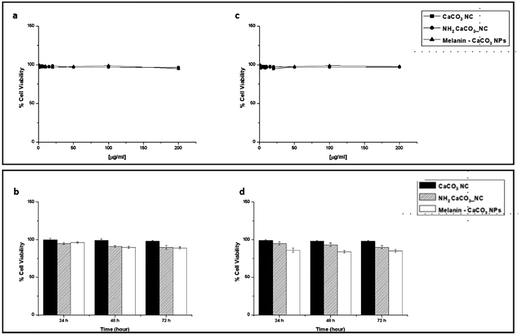
In this work, before investigating the antioxidant and the cytoprotective effect of melanin–CaCO3 NPs, at first, we evaluated the effect of exposure of HaCaT (human epidermal keratinocyte) and WS1 (fibroblast cell isolated from the skin) cell lines, to naked CaCO3-NCs, NH2-CaCO3-NCs and melanin–CaCO3 NPs. The use of HaCaT and WS1 is appropriate because these are the primary cell types found in human skin, the organ most directly affected by UV radiation and photo-oxidative stress. A very important aspect is the identification of the most relevant dose for particle toxicity, so in this context, HaCaT (Fig. 4a) and WS1 (Fig. 4c) cells were exposed for 24 hours to different concentrations of NPs (from 1 up to 200 µg mL−1). No cytotoxic effect was observed; then, having fixed the concentration of 100 µg mL−1, a time-course was carried out up to 72 h, for both cell lines (Fig. 4b and d). At 72 hours, there is a slight decrease in cell viability, but it does not appear to be statistically significant.
Furthermore, cell proliferation was examined by measuring the DNA concentration where an increase indicates cell growth. While melanin–CaCO3 NPs did not seem to influence the metabolic activity of HaCaT and WS1 cells (Fig. 4), it significantly increased cell proliferation (Fig. 5). The evaluation of cytotoxicity and proliferation demonstrated the harmlessness of CaCO3-NCs and NH2-CaCO3-NCs in both selected cell lines in vitro at the selected concentrations. In contrast, although melanin–CaCO3 NPs do not affect cell viability by reducing metabolic activity, the proliferation is considerably increased. Bare CaCO3-NCs were employed as direct control because free DHICA-melanin was not readily obtainable in a form directly comparable to that produced on CaCO3 nanoparticles. Indeed, DHICA-melanin functions as an organic dopant within the carbonate scaffold, modulating surface chemistry and redox behavior, thereby enabling a meaningful assessment of the hybrid material's biological effects.52 To carry out the tests on photoprotective ability, the next culture conditions were chosen for being innocuous to both cell lines and, at the same time, containing the highest amount of melanin–CaCO3 NPs.
As validated in our prior work,35 CaCO3 nanocrystals ensure safe long-term fate by dissolving in lysosomal acidity (∼pH 5), preventing accumulation. In this study, cellular internalization, at different time points (3 h and 24 h), was evaluated by also confocal microscopy using melanin–CaCO3 NPs conjugated with FITC. HaCaT and WS1 cells were incubated with 100 µg mL−1 of fluorescent melanin–CaCO3 NPs, and the micrographs reported in Fig. 6 show how these NPs were internalized in the cells already only after 3 hours of exposure, as suggested by the green fluorescence reported in the central panel of the figure. The morphology of the nuclei shows that no alterations are observed even after long periods of incubation, confirming that there are no cytotoxic effects from the exposure of the cells to these NPs. To confirm internalization into cells, confocal Z-stacks were acquired (Fig. S3). To quantify the amount of internalized melanin–CaCO3 NPs by cells, HaCaT and WS1 cells were subjected to incubation with melanin CaCO3 NPs at a concentration of 10 µg mL−1 for a duration of 60 min. Subsequently, the complete cell lysates were carefully prepared. The ultraviolet-visible (UV-vis) spectra of lysates derived from both untreated and treated cells were recorded. Through this analysis, the extent of melanin internalized by the cells was estimated to be approximately 40% in HaCaT cells and 32% in WS1 cells. This estimation was based upon the absorbance measured at 330 nm in the lysates of the treated cells, with reference to the calibration curve established using pure melanin–CaCO3 NPs (Fig. S4). The rate and the mechanism of internalization of CaCO3 and NH2-CaCO3-NCs have been extensively investigated in different cancer cell line models, in our previous studies.35,36
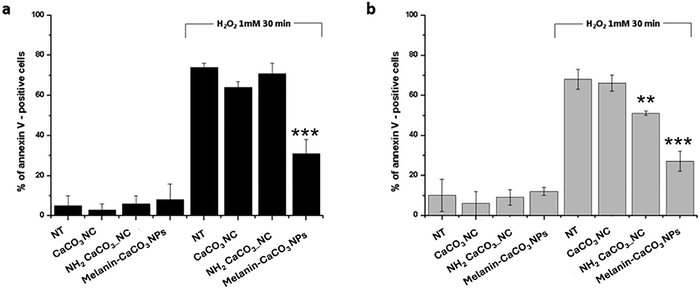
The protective effect of melanin–CaCO3 NPs against hydrogen peroxide-induced apoptosis was verified incubating HaCaT and WS1 cells for 24 h with NPs; then, the cells were treated with 1 mM H2O2 for 30 min. The apoptosis was assessed through FACS analysis staining the cells with annexin V. Fig. 7 demonstrates that all samples tested (naked CaCO3 nanocrystals, amino-functionalized CaCO3 nanocrystals, and melanin CaCO3 NPs) were biocompatible both in HaCaT (Fig. 7a) and WS1 (Fig. 7b) cell lines. Hydrogen peroxide treatment induces a strong cell death and over 80% of cells were positive for annexin V in both cell lines. Instead, the preventive incubation of cells with melanin CaCO3 NPs revealed a significative protective effect; indeed, the apoptotic cells were at most 32–35% in HaCaT and WS1 cells, respectively. This effect was due to the redox properties of dark eumelanin which plays a role in the protection from free radicals and oxidative insults.51,53–55 This mechanistic interpretation is further supported by EPR measurements, which revealed the intrinsic radical character of melanin within the nanoparticles. Importantly, the paramagnetic character evidenced by EPR is directly linked to melanin's radical scavenging activity, as the presence of stable unpaired electrons provides redox-active centers capable of interacting with and neutralizing reactive species. These stable paramagnetic centers underpin its redox capacity, providing a direct link between the radical nature of melanin and the observed reduction of oxidative stress and apoptosis in treated cells. Furthermore, the protective role of melanin CaCO3 NPs in the photoinduced oxidative stress against HaCaT and WS1 cell lines was assessed under UVA irradiation, chosen as a source of stress.56 Ultraviolet irradiation may cause serious cellular and molecular tissue damage when living organisms are directly exposed without filtering.57 This assay focused on UV blocking ability of our melanin CaCO3 NPs against the damaging effects of UVA irradiation. A dose–response experiment was first performed incubating HaCaT and WS1 cells with increasing concentrations of naked CaCO3-NCs, NH2-CaCO3-NCs, and melanin–CaCO3 NPs (0.1–10 µg mL−1) for 2 h prior to UVA irradiation treatment, and immediately after irradiation, ROS production was evaluated by the H2DCFDA assay.
As shown in Fig. 8, at first, the HaCaT (a) and WS1 (b) cells were incubated at the maximum chosen concentration (10 µg mL−1) of each sample to evaluate a possible ROS production related to nanomaterial exposure without UVA irradiation. In this case, DCF fluorescence did not show a significant increase, also in the presence of melanin–CaCO3 NPs. Instead, ROS levels increased considerably on irradiated cells (Fig. 8c and d) under all tested conditions, but interestingly, only when cells were preincubated with melanin–CaCO3 NPs prior to UVA exposure, ROS production was decreased in a dose dependent manner, reaching the levels observed in non-irradiated cells when the melanin–CaCO3 NPs were tested already at 5 µg mL−1. This evidence confirmed that the cytoprotective activity was exerted by the internalized melanin–CaCO3 NPs, which acted inside the cells to preserve their integrity and not simply through a UV-shielding mechanism. Together with this peculiar behavior, these results appeared to be much more valuable considering the low content of melanin included in the final NPs which has already proven to be useful to confer a significant photoprotection ability to CaCO3.58–60 Several studies have shown that melanin nanoparticles, including those combined with CaCO3, can exert protective effects within cells by scavenging ROS and reducing oxidative stress. This is especially significant because melanin's antioxidant properties are not restricted to UV protection but also include direct neutralization of free radicals and ROS in the cytoplasm, mitochondria, and other cellular compartments. The choice of CaCO3 as an inorganic support offers distinct advantages over traditional persistent materials like SiO2, TiO2, and CeO2, particularly regarding intracellular fate and antioxidant safety. A comprehensive summary of the biological performance of the different nanoparticle systems, highlighting the superior properties of the melanin–hybrid design, is provided in Table 2. While metal-oxide supports such as TiO2 can exhibit unintended pro-oxidant behaviour under UV exposure or cause ‘dark toxicity’ through mitochondrial interference, CaCO3 is metabolically inert and undergoes pH-dependent biodegradation within acidic endo-lysosomal compartments. This ensures that the nanoparticles do not accumulate permanently within the cell, instead decompose into essential calcium ions that are safely integrated into cellular homeostasis. From a mechanistic standpoint, the CaCO3 matrix facilitates a superior antioxidant performance by providing a high-surface-area, biocompatible scaffold that maximizes the accessibility of melanin's paramagnetic centers for ROS scavenging, avoiding the environmental fouling or surface-masking effects often observed with more chemically rigid inorganic cores. Furthermore, as reported in the literature, the size of our nanoparticles plays a crucial role in their antioxidant activity.61,62 Recently, Carmignani et al. synthesized a series of melanin-mimic nanoparticles with sizes ranging from 150 to 960 nm and found that the scavenging efficiency was highest for the smallest nanoparticles.63 Gianneschi and coworkers synthesized melanin-like NPs by simple polymerization of dopamine in an alkaline environment. They reported the ROS-scavenging properties of these nanoparticles in human epidermal keratinocytes (HEKa).64 All these studies underscored the critical relationship between the size and structural organization of melanin-like nanoparticles and their antioxidant efficacy, which could be presumably driven by the differential exposure and accessibility of specific chemical functional groups integral to radical scavenging mechanisms.
| Sample | Cell viability (up to 200 µg mL−1) | Cell proliferation | ROS scavenging efficiency | Cytoprotection (H2O2-induced apoptosis) |
|---|---|---|---|---|
| CaCO3 NC | High (>95% – biocompatible) | Baseline | Negligible | None |
| NH2-CaCO3NC | High (>95% – biocompatible) | Baseline | Negligible | None |
| Melanin–CaCO3 NPs | High (>95% – biocompatible) | Significantly increased | Very high (∼50% reduction at 5 µg mL−1) | Significant (apoptosis reduced by ∼60%) |
4. Conclusions
Oxidative stress occurs when there is an imbalance between the generation and accumulation of ROS in cells and tissues and the ability of the biological system to eliminate these reactive substances. The accumulation of oxidative damage can impair cellular function leading to DNA mutations or can trigger inflammatory responses exacerbating tissue damage and increasing cancer risk. Furthermore, chronic oxidative stress is associated with aging and the development of age-related diseases. The body employs antioxidants (e.g., vitamin C, vitamin E, and glutathione) to neutralize ROS and minimize damage. Melanin nanoparticles have gained attention for their potential cytoprotective action, particularly in counteracting oxidative stress by absorbing UV radiation and scavenging ROS.51,65,66This paper presents the synthesis of melanin–CaCO3 NPs according to the in situ polymerization of DHICA on amino-functionalized CaCO3 nanoparticles. The proposed strategy of using APTES-functionalized CaCO3 nanoparticles ensures the colloidal stability of melanin-based nanosystems and the success of the polymerization process also favored by the alkaline conditions. The basic pH, promoting the oxidative polymerization of DHICA onto CaCO3 nanoparticles, plays a crucial role in facilitating the conversion of DHICA into a stable polymeric structure, which is essential for the formation of melanin-like nanoparticles with enhanced stability and ROS-scavenging ability.
The produced NPs show a round shape morphology with a good biocompatibility and a high rate of cell internalization, important features for dermo-cosmetic applications.67 The findings demonstrate that the melanin–CaCO3 NPs are biocompatible, internalized by skin cells, and effectively protect them from oxidative stress induced by H2O2 and UVA irradiation. Indeed, these NPs protect immortalized human keratinocytes (HaCat) and fibroblasts (WS1) from damage caused by oxidative stress, as proved by the significantly reduction of stress parameters including ROS. Therefore, the protective action of melanin–CaCO3 nanoparticles extends to intracellular mechanisms of oxidative stress mitigation, not just UV protection. The mechanism of antioxidant activity relies on the melanin's chemical structure, because it contains multiple phenolic groups and heteroatoms (such as nitrogen), which allow it to interact with and neutralize reactive molecules by scavenging free radicals and stabilizing cellular membranes. Also, these processes can help to protect cells from oxidative damage to lipids, proteins, and DNA.
The findings presented here provide new opportunities for the design of innovative hybrid nanoparticles with cytoprotective activity for nanomedicine applications. These nanoparticles offer a promising avenue for combating oxidative stress through their potent antioxidant activity and ability to protect cellular structures and modulate inflammatory responses. Their potential applications in therapeutic settings—particularly for neuroprotection, drug delivery, and overall cytoprotection—make them a subject of active further research in the fields of nanomedicine and biochemistry. However, further studies will need to include in vivo experiments, assess inflammatory markers, and evaluate long-term toxicity. These will be critical to fully determine the translational potential before any clinical application can be considered. On the other hand, advances in the precise tuning of nanostructures—from core–shell to cloud-type architectures—can further enhance their functional performance, as highlighted in the study.30
Author contributions
Vitiello G. and Vergaro V. wrote the manuscript, realized data analysis and prepared figures; Baldassarre F. contributed to physical–chemical and morphological characterization of nanoparticles. Pezzella A., Luciani G., Ciccarella G. and Boncristiani C. contributed to the revision of the manuscript; Vergaro V. coordinated the study and revised the manuscript. All authors read and approved the final manuscript. The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The authors confirm that all the supporting data and the findings of this study are included in the article and the attached supplementary information (SI). The supplementary materials include the following additional data concerning the (1) CaCO3 nanoparticles characterization obtained by Transmission Electron Microscopy; (2) the stability characterization of nanoparticles performed by zeta potential measurements; (3) confocal micrographs: orthogonal projection image to confirm the internalization of nanoparticles; and (4) the quantification of internalized melanin–CaCO3 nanoparticles performed by recording the UV-vis spectra; (5) TG and DTA curves of bare CaCO3 NC and melanin–CaCO3 NPs; (6) FTIR spectra of bare CaCO3 and melanin–CaCO3 NPs. See DOI: https://doi.org/10.1039/d5ma01417d.Acknowledgements
We acknowledge financial support under the National Recovery and Resilience Plan (NRRP), Mission 4, Component 2, Investment 1.1, Call for tender no. 104 published on 2.2.2022 by the Italian Ministry of University and Research (MUR), funded by the European Union – NextGenerationEU – Project Title MulticEllular spheRoids model for muLtiparametric blood–brain barrier Injury detectioN in microfluidics (MERLIN) – CUP B53D23018380006 by the Italian Ministry of Ministry of University and Research (MUR). The authors also thank the Department of Chemical Science of University of Naples Federico II for awarding time for EPR measurements. Regione Puglia within “Tecnopolo per la medicina di precisione” (TecnoMed Puglia): DGR n.2117 del 21/11/2018, CUP: B84I180 0 0540 0 02.References
- Y. A. Hajam, R. Rani, S. Y. Ganie, T. A. Sheikh, D. Javaid, S. S. Qadri, S. Pramodh, A. Alsulimani, M. F. Alkhanani, S. Harakeh, A. Hussain, S. Haque and M. S. Reshi, Cells, 2022, 11, 552 CrossRef CAS PubMed.
- D. Rotariu, E. E. Babes, D. M. Tit, M. Moisi, C. Bustea, M. Stoicescu, A. F. Radu, C. M. Vesa, T. Behl, A. F. Bungau and S. G. Bungau, Biomed. Pharmacother., 2022, 152, 113238 CrossRef CAS PubMed.
- A. Laurent, C. Nicco, C. Chéreau, C. Goulvestre, J. Alexandre, A. Alves, E. Lévy, F. Goldwasser, Y. Panis, O. Soubrane, B. Weill and F. Batteux, Controlling Tumor Growth by Modulating Endogenous Production of Reactive Oxygen Species, Cancer Res., 2005, 65, 948–956 CrossRef CAS PubMed.
- B. Kalyanaraman, G. Cheng and M. Hardy, Redox Biol., 2024, 71, 103092 CrossRef CAS PubMed.
- S. Di Meo, T. T. Reed, P. Venditti and V. M. Victor, Oxid. Med. Cell. Longev., 2016, 2016, 1245049 CrossRef PubMed.
- N. Zarkovic, Cells, 2020, 9, 767 CrossRef CAS PubMed.
- K. Jomova, R. Raptova, S. Y. Alomar, S. H. Alwasel, E. Nepovimova, K. Kuca and M. Valko, Arch. Toxicol., 2023, 97, 2499–2574 CrossRef CAS PubMed.
- M. Lodovici and E. Bigagli, J. Toxicol., 2011, 2011, 487074 Search PubMed.
- P. Irato and G. Santovito, Antioxidants, 2021, 10, 579 CrossRef CAS PubMed.
- Y. Dai, Y. Guo, W. Tang, D. Chen, L. Xue, Y. Chen, Y. Guo, S. Wei, M. Wu, J. Dai and S. Wang, J. Nanobiotechnol., 2024, 22, 252 CrossRef PubMed.
- F. Wei, W. Yang, H. Wang, S. Song, Y. Ji, Z. Chen, Y. Zhuang, J. Dai and H. Shen, Biomater. Sci., 2025, 13, 343–363 RSC.
- F. Fragou, A. Theofanous, Y. Deligiannakis and M. Louloudi, Micromachines, 2023, 14, 383 CrossRef PubMed.
- P. Wen, A. Dirisala, H. Guo, X. Liu, S. Kobayashi, H. Kinoh, T. Anada, M. Tanaka, K. Kataoka and J. Li, J. Controlled Release, 2025, 382, 113683 CrossRef CAS PubMed.
- A. R. Hegde, M. U. Kunder, M. Narayanaswamy, S. Murugesan, S. C. Furtado, B. B. Veerabhadraiah and B. Srinivasan, Environ. Sci. Pollut. Res. Int., 2024, 31, 38061–38082 CrossRef CAS PubMed.
- M. Burkard, A. Betz, K. Schirmer and A. Zupanic, Environ. Sci. Technol., 2020, 54, 335–344 CrossRef CAS PubMed.
- L. Guo, W. Li, Z. Gu, L. Wang, L. Guo, S. Ma, C. Li, J. Sun, B. Han and J. Chang, Int. J. Mol. Sci., 2023, 24, 4360 CrossRef CAS PubMed.
- J. Y. Lin and D. E. Fisher, Nature, 2007, 445, 843–850 CrossRef CAS PubMed.
- M. Brenner and V. J. Hearing, Photochem. Photobiol., 2008, 84, 539–549 CrossRef CAS PubMed.
- M. Jinek, K. Chylinski, I. Fonfara, M. Hauer, J. A. Doudna and E. Charpentier, A programmable dual RNA-guided DNA endonuclease in adaptive bacterial immunity One-Sentence Summary, Science, 2012, 337, 816–821 CrossRef CAS PubMed.
- S. D. Seelam, D. Agsar, S. K. Halmuthur M., P. Reddy Shetty, S. Vemireddy, K. M. Reddy, M. K. Umesh and C. H. Rajitha, J. Photochem. Photobiol., B, 2021, 216, 112126 CrossRef CAS PubMed.
- Z. Lin, L. Liu, W. Wang, L. Jia, Y. Shen, X. Zhang, D. Ge, W. Shi and Y. Sun, Biomater. Sci., 2021, 9, 5951–5964 RSC.
- S. Zhang, Q. Ou, P. Xin, Q. Yuan, Y. Wang and J. Wu, Biomater. Sci., 2019, 7, 4230–4236 RSC.
- T. Rahmani Eliato, J. T. Smith, Z. Tian, E. S. Kim, W. Hwang, C. P. Andam and Y. J. Kim, J. Mater. Chem. B, 2021, 9, 1536–1545 RSC.
- J. J. Oh, J. Y. Kim, S. H. Son, W. J. Jung, D. H. Kim, J. W. Seo and G. H. Kim, RSC Adv., 2021, 11, 19682–19689 RSC.
- W. Cheng, X. Zeng, H. Chen, Z. Li, W. Zeng, L. Mei and Y. Zhao, ACS Nano, 2019, 13, 8537–8565 CrossRef CAS PubMed.
- G. Pota, A. Zanfardino, M. Di Napoli, D. Cavasso, M. Varcamonti, G. D’Errico, A. Pezzella, G. Luciani and G. Vitiello, Colloids Surf., B, 2021, 202, 111671 CrossRef CAS PubMed.
- B. Silvestri, G. Vitiello, G. Luciani, V. Calcagno, A. Costantini, M. Gallo, S. Parisi, S. Paladino, M. Iacomino, G. D’Errico, M. F. Caso, A. Pezzella and M. D’Ischia, ACS Appl. Mater. Interfaces, 2017, 9, 37615–37622 CrossRef CAS PubMed.
- G. Pota, B. Silvestri, G. Vitiello, N. Gallucci, R. Di Girolamo, S. Scialla, M. G. Raucci, L. Ambrosio, M. Di Napoli, A. Zanfardino, M. Varcamonti, A. Pezzella and G. Luciani, Biomater. Adv., 2023, 153, 213558 CrossRef CAS PubMed.
- F. Furlani, G. Pota, A. Rossi, G. Luciani, E. Campodoni, F. Mocerino, G. D’Errico, A. Pezzella, S. Panseri, G. Vitiello and M. Sandri, Colloids Surf., B, 2024, 235, 113756 CrossRef CAS PubMed.
- G. Pota, G. Vitiello, V. Venezia, F. Della Sala, A. Borzacchiello, A. Costantini, L. Paduano, L. P. Cavalcanti, F. Tescione, B. Silvestri and G. Luciani, Polymers, 2022, 14, 3568 CrossRef CAS PubMed.
- P. Durgadevi, K. Girigoswami and A. Girigoswami, Physics, 2025, 7, 28 CrossRef CAS.
- P. Pallavi, K. Girigoswami, K. Harini, P. Gowtham, A. Thirumalai and A. Girigoswami, Naunyn. Schmiedebergs. Arch. Pharmacol., 2025, 398, 867–880 CrossRef CAS PubMed.
- P. Pallavi, K. Harini, S. Crowder, D. Ghosh, P. Gowtham, K. Girigoswami and A. Girigoswami, Appl. Biochem. Biotechnol., 2023, 195, 6979–6993 CrossRef CAS PubMed.
- Y. Zhao, Y. Bian, X. Xiao, B. Liu, B. Ding, Z. Cheng, P. Ma and J. Lin, Small, 2022, 18, 2204047 CrossRef CAS PubMed.
- V. Vergaro, E. Carata, F. Baldassarre, E. Panzarini, L. Dini, C. Carlucci, S. Leporatti, B. F. Scremin, D. Altamura, C. Giannini, F. P. Fanizzi and G. Ciccarella, Adv. Powder Technol., 2017, 28, 2445–2455 CrossRef CAS.
- V. Vergaro, I. Pisano, R. Grisorio, F. Baldassarre, R. Mallamaci, A. Santoro, G. P. Suranna, P. Papadia, F. P. Fanizzi and G. Ciccarella, Materials, 2019, 12, 1481 CrossRef CAS PubMed.
- D. Vecchione, A. M. Grimaldi, E. Forte, P. Bevilacqua, P. A. Netti and E. Torino, Sci. Rep., 2017, 7, 45121 CrossRef CAS PubMed.
- U. Freudenberg, Y. Liang, K. L. Kiick and C. Werner, Adv. Mater., 2016, 28, 8861–8891 CrossRef CAS PubMed.
- M. D’Ischia, K. Wakamatsu, A. Napolitano, S. Briganti, J. C. Garcia-Borron, D. Kovacs, P. Meredith, A. Pezzella, M. Picardo, T. Sarna, J. D. Simon and S. Ito, Pigm. Cell Melanoma Res., 2013, 26, 616–633 CrossRef PubMed.
- V. Vergaro, P. Papadia, S. Leporatti, S. A. De Pascali, F. P. Fanizzi and G. Ciccarella, J. Inorg. Biochem., 2015, 153, 284–292 CrossRef CAS PubMed.
- E. Wondu, Z. C. Lule and J. Kim, Polymers, 2020, 12, 747 CrossRef CAS PubMed.
- N. Chekina, D. Horák, P. Jendelová, M. Trchová, M. J. Beneš, M. Hrubý, V. Herynek, K. Turnovcová and E. Syková, J. Mater. Chem., 2011, 21, 7630–7639 RSC.
- G. Vitiello, A. Pezzella, V. Calcagno, B. Silvestri, L. Raiola, G. D’Errico, A. Costantini, F. Branda and G. Luciani, J. Phys. Chem. C, 2016, 120, 6262–6268 CrossRef CAS.
- D. Liberti, M. L. Alfieri, D. M. Monti, L. Panzella and A. Napolitano, Antioxidants, 2020, 9, 270 CrossRef CAS PubMed.
- É. Velot, F. Ducrocq, L. Girardeau, A. Hehn, S. Piutti, C. Kahn, M. Linder, A. Bianchi and E. Arab-Tehrany, Int. J. Mol. Sci., 2022, 23, 12423 CrossRef PubMed.
- A. Bianchi, É. Velot, H. Kempf, K. Elkhoury, L. Sanchez-Gonzalez, M. Linder, C. Kahn and E. Arab-Tehrany, Int. J. Mol. Sci., 2020, 21, 3436 CrossRef CAS PubMed.
- V. Vergaro, F. Baldassarre, F. De Castro, D. Migoni, M. M. Dell’Anna, P. Mastrorilli, F. P. Fanizzi and G. Ciccarella, Bioinorg. Chem. Appl., 2022, 2022, 9571217 CrossRef PubMed.
- B. Silvestri, G. Vitiello, G. Luciani, V. Calcagno, A. Costantini, M. Gallo, S. Parisi, S. Paladino, M. Iacomino, G. D’Errico, M. F. Caso, A. Pezzella and M. d’Ischia, ACS Appl. Mater. Interfaces, 2017, 9, 37615–37622 CrossRef CAS PubMed.
- V. Vergaro, M. Civallero, C. Citti, M. Cosenza, F. Baldassarre, G. Cannazza, S. Pozzi, S. Sacchi, F. P. Fanizzi and G. Ciccarella, Cancers, 2018, 10, 31 CrossRef PubMed.
- G. Pota, A. Puzone, E. Carrella, A. Pezzella, G. Luciani, G. D’Errico and G. Vitiello, Appl. Surf. Sci., 2025, 686, 162134 CrossRef CAS.
- L. Panzella, G. Gentile, G. D’Errico, N. F. Della Vecchia, M. E. Errico, A. Napolitano, C. Carfagna and M. D’Ischia, Angew. Chem., Int. Ed., 2013, 52, 12684–12687 CrossRef CAS PubMed.
- K. Urabe, P. Aroca, K. Tsukamoto, D. Mascagna, A. Palumbo, G. Prota and V. J. Hearing, Biochim. Biophys. Acta, 1994, 1221, 272–278 CrossRef CAS PubMed.
- B.-L. L. Seagle, K. A. Rezai, Y. Kobori, E. M. Gasyna, K. A. Rezaei and J. R. Norris, Proc. Natl. Acad. Sci. U. S. A., 2005, 102, 8978–8983 CrossRef CAS PubMed.
- K. Y. Ju, Y. Lee, S. Lee, S. B. Park and J. K. Lee, Biomacromolecules, 2011, 12, 625–632 CrossRef CAS PubMed.
- T. Sarna, Photochem. Photobiol., 2023, 99, 866–868 CrossRef CAS PubMed.
- R. M. Haywood, M. Lee and C. Linge, J. Photochem. Photobiol., B, 2006, 82, 224–235 CrossRef CAS PubMed.
- R. Brem, M. Guven and P. Karran, Free Radical Biol. Med., 2017, 107, 101–109 CrossRef CAS PubMed.
- L. Guo, T. Wang, Z. Li, S. Wu, Y. Xu, Z. Yang, Y. Li, Z. Gu and X. Jiang, Chem. Mater., 2023, 35, 5420–5432 CrossRef CAS.
- Z. Dong, L. Feng, Y. Hao, M. Chen, M. Gao, Y. Chao, H. Zhao, W. Zhu, J. Liu, C. Liang, Q. Zhang and Z. Liu, J. Am. Chem. Soc., 2018, 140, 2165–2178 CrossRef CAS PubMed.
- X. Ru, Y. Guo, Z. Bai, X. Xie, X. Ma, L. Zhu, K. Wang, F. Wang, L. Yang and J. Lu, Commun. Chem., 2019, 2, 105 CrossRef.
- A. Mavridi-Printezi, A. Menichetti, D. Mordini, R. Amorati and M. Montalti, Antioxidants, 2023, 12, 863 CrossRef CAS PubMed.
- G. Zhong, X. Yang, X. Jiang, A. Kumar, H. Long, J. Xie, L. Zheng and J. Zhao, Nanoscale, 2019, 11, 11605–11616 RSC.
- A. Carmignani, M. Battaglini, E. Sinibaldi, A. Marino, V. Vighetto, V. Cauda and G. Ciofani, ACS Appl. Nano Mater., 2022, 5, 1702–1713 CrossRef CAS.
- Y. Huang, Y. Li, Z. Hu, X. Yue, M. T. Proetto, Y. Jones and N. C. Gianneschi, ACS Cent. Sci., 2017, 3, 564–569 CrossRef CAS PubMed.
- S. Jiang, X. M. Liu, X. Dai, Q. Zhou, T. C. Lei, F. Beermann, K. Wakamatsu and S. Z. Xu, Free Radical Biol. Med., 2010, 48, 1144–1151 CrossRef CAS PubMed.
- S. Ito, M. Kikuta, S. Koike, G. Szewczyk, M. Sarna, A. Zadlo, T. Sarna and K. Wakamatsu, Pigm. Cell Melanoma Res., 2016, 29, 340–351 CrossRef CAS PubMed.
- R. Micillo, M. Iacomino, M. Perfetti, L. Panzella, K. Koike, G. D’Errico, M. d’Ischia and A. Napolitano, Pigm. Cell Melanoma Res., 2018, 31, 475–483 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |