Open Access Article
Open Access ArticleEnhanced photodegradation of antibiotics using a novel BiVO4/P-doped TiO2 heterostructure: performance evaluation, property and kinetic modelling
Tanzim Ur
Rahman
a,
Musfekur Rahman
Dihan
 a,
Abdul
Hai
b and
Md. Shahinoor
Islam
a,
Abdul
Hai
b and
Md. Shahinoor
Islam
 *a
*a
aDepartment of Chemical Engineering, Bangladesh University of Engineering and Technology, Dhaka-1000, Bangladesh. E-mail: shahinoorislam@che.buet.ac.bd; Tel: +8801732782120
bDepartment of Chemical & Petroleum Engineering, Khalifa University, 127788, Abu Dhabi, United Arab Emirates
First published on 26th January 2026
Abstract
This study presents the performance evaluation of a novel P-doped TiO2–BiVO4 heterostructured photocatalyst against two model antibiotics, metronidazole (MNZ) and ciprofloxacin (CIP), in both single and binary solution system. The morphological analysis confirms the successful incorporation of anatase P-TiO2 into monoclinic BiVO4 and the reduction in BiVO4 particle size with increasing surface roughness. Two multivariable linear regression models were developed to predict the crystallite sizes and bandgap of the photocatalyst. Among the seven photocatalysts, the composite with 80 wt% P-TiO2 in BiVO4 exhibited the highest degradation for CIP and MNZ (94.48% and 96.07%, respectively), with a crystallite size of 14 nm and an optimized bandgap of 2.59 eV. Kinetic studies revealed that the degradation followed a first-order model, with P-TiO2/BiVO4-80 exhibiting rate constants 4.92 and 9.50 times those of pristine TiO2 and BiVO4, respectively. Additionally, the estimated catalytic potential and the synergy index showed the effectiveness of coupling P-TiO2 with BiVO4 for potential use in AOPs. The effects of operational parameters were evaluated to identify the optimized conditions (10 ppm antibiotics, 1.2 g L−1 catalyst loading, natural pH, and 20 mM H2O2), under which nearly complete degradation of both MNZ and CIP was achieved. Similar degradation efficiency in the binary antibiotic mixture, and while retaining 78% efficiency after five cycles, demonstrated the robustness, recyclability, and stability of the photocatalytic activity of the synthesized photocatalyst. Hence, we believe that the demonstrated insights of the proposed highly efficient novel P-TiO2/BiVO4 can further facilitate the transition towards sustainable wastewater treatment, providing clean and safer water for everyone.
1. Introduction
The demand for antibiotics to treat infectious diseases in humans and animals, including livestock and aquaculture, has increased significantly; however, a substantial portion of these antibiotics is released into the environment because only a small fraction is metabolized.1,2 Browne et al. estimated that global antibiotic consumption increased by approximately 46% between 2000 and 2018, with South Asian countries, including Bangladesh, India, Pakistan, and Nepal, accounting for about 25% of total consumption.3 This trend of rising consumption and environmental release is also linked to antimicrobial resistance (AMR), which can be detrimental to ecosystems and public health. The threat of antibiotic pollution and the emergence of AMR is particularly high in low- to middle-income countries (LMICs) like Bangladesh, mainly due to a lack of awareness and the unregulated, over-the-counter availability of antibiotics without prescriptions. In addition to contamination from excessive use, antibiotics enter water bodies from agricultural runoff, the pharmaceutical industry, and hospital wastewater. They are also released into water bodies via municipal and industrial wastewater treatment plants (WWTPs), as current treatment technologies are insufficient to remove and effectively mineralize such pollutants.1,2,4 In Bangladesh, the most commonly used antibiotics include ciprofloxacin (CIP) and metronidazole (MNZ). In a study, Akhter et al. estimated that approximately 273 kg of CIP is released into the environment each year in the Chittagong Metropolitan area. They also reported the prevalence of CIP-resistant E. coli bacteria.5 On the other hand, MNZ has been detected at high concentrations across multiple sampling sites in Barisal.6 Antibiotics in water bodies, like other persistent organic pollutants (POPs), are challenging to mineralize using current wastewater treatment technologies. Thus, it is imperative to place greater emphasis on research into the effective elimination of POPs, such as antibiotics, from water reclamation pathways.7,8Heterogeneous photocatalysis has emerged as a sustainable option to eliminate POPs from water bodies.9 Photocatalytic treatment of pollutants is a type of advanced oxidation process (AOP) that degrades pollutants by generating highly reactive free radicals.2 Among various semiconductor photocatalysts, TiO2 is most widely utilized because of its low toxicity, stability, and availability.10 However, the main issue of TiO2 is its large band gap of 3.2 eV, which limits its photoactivity under irradiation with longer wavelengths.11 Therefore, doping it with metal/non-metals and the formation of a heterojunction with another photocatalyst can be utilized for effective electron–hole pair separation and inhibition of their recombination.12–14
Doping of TiO2 with non-metals (e.g., P, N, and C) has become increasingly desirable because it can reduce the band gap by modifying the band structure, thereby improving photodegradation.15,16 Studies have shown that dopant P in TiO2, resulted in enhanced photocatalytic degradation of organic pollutants. P-doped TiO2 exhibits a narrower band gap, diverse surface and bulk properties, improved thermal stability, and a smaller particle size.17 Feng et al. synthesized P-doped TiO2 with surface oxygen vacancies and utilized it for the degradation of CIP. The results revealed a 16.2-fold increase in the degradation rate relative to pristine TiO2, and a reduction in the band gap to 3.02 eV.18 In another study, Gopal et al. synthesized P-doped TiO2 by the sol–gel method using H3PO4 as the precursor. They observed the presence of a Ti–O–P bond, which is representative of the substitution of Ti4+ by P5+. Moreover, doping has resulted in a red shift of the absorption edge, a reduction in particle size, an increase in thermal stability, and inhibition of the phase transition of anatase TiO2.19 Photocatalytic performance can be further improved by forming heterojunctions with other semiconductors, such as ZnO, BiVO4, and SnO2, whose suitable band structures can suppress electron–hole recombination.11,20,21 For instance, the coupling of TiO2 with BiVO4 has been studied extensively for enhanced photocatalytic performance owing to heterojunction formation, improved charge-carrier separation, and reduced recombination. Monoclinic BiVO4, with a low band gap of approximately 2.4 eV, is prone to high electron–hole recombination, resulting in poor photocatalytic performance.22 However, this limitation can be overcome by coupling BiVO4 with pristine or doped TiO2. For instance, Liaqat et al. synthesized TiO2/BiVO4via a hydrothermal method and used it to degrade tetracycline (TC) and methylene blue (MB). A maximum of 72% TC degradation was observed with TiO2/BiVO4, whereas degradation was 35% and 40% for pristine BiVO4 and TiO2, respectively.23 On the other hand, MB degradation was 38% and 43% for pristine BiVO4 and TiO2, respectively, whereas it increased to 87% for TiO2/BiVO4. Moreover, studies have shown that non-metal (nitrogen and boron) doped TiO2 coupled with BiVO4 exhibits improved photocatalytic performance.24,25 However, to the best of our knowledge, no studies have reported the photocatalytic performance of P-doped TiO2 coupled with BiVO4; therefore, it remains unclear whether this combination can further improve photocatalytic performance. Moreover, industrial wastewater contains multiple contaminants, and their presence can both contribute synergistically or act competitively during the photocatalytic degradation. For instance, the wastewater matrix may contain both CIP and MNZ, which differ in molecular structure and adsorption behavior and follow separate AOP-based degradation pathways.26 Most studies report the photocatalytic efficiencies of the synthesized heterojunctions for single contaminants; thus, it is imperative to determine whether the material can simultaneously remove multiple pollutants, benefiting real wastewater treatment applications.
Thus, in this study, we synthesized and evaluated the photocatalytic performance of P-TiO2/BiVO4. Seven types of photocatalysts were synthesized, with varying mass ratios of P-TiO2 in BiVO4, to identify the optimal combination, and different characterization techniques, XRD, FESEM-EDX, FTIR, and UV-vis spectroscopy, were utilized for examining the structural and functional properties of the synthesized catalysts. Two multivariable linear regression (MLR) models were also proposed to predict the photocatalyst's crystallite size and bandgap from elemental composition. The photocatalytic performance evaluations were conducted against CIP and MNZ, both in single and binary mixtures, along with catalytic potential, synergy index, and kinetic studies to assess the effectiveness of the synthesized photocatalysts. The effects of various operational parameters, including solution pH, catalyst dosage, initial pollutant concentration, and hydrogen peroxide concentration, were assessed to determine the optimal operational conditions. Thus, we believe this study will provide fresh insights into the properties, effectiveness, and behavior of the novel P-TiO2/BiVO4 heterojunction photocatalyst developed for the degradation of antibiotics in wastewater.
2. Materials and methodology
2.1. Materials
Absolute ethanol (C2H5OH) (Tedia, USA), ortho-phosphoric acid (H3PO4) (Merck, Germany, 85%), titanium(IV) isopropoxide (C14H28O4Ti) (Sigma-Aldrich, Germany 98%), bismuth nitrate pentahydrate (Bi(NO3)3·5H2O) (Sigma-Aldrich, Germany, 98%), nitric acid (HNO3), citric acid (C6H8O7), ammonium metavanadate (NH4VO3) (Sigma-Aldrich, Germany, 99%), ammonium hydroxide (NH4OH), metronidazole (C6H9N3O3), ciprofloxacin (C17H18FN3O3), and hydrogen peroxide (H2O2) (Sigma-Aldrich, 30% (w/w)) were used. All chemicals used for experimentation were of analytical grade.2.2. Methodology
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Solution II was stirred at 80 °C until a brown-colored solution was obtained. Subsequently, solution I was added dropwise to solution II, resulting in a blue-colored solution. Stoichiometric amounts of P-doped TiO2 were added to the constant concentration of BiVO4 to obtain composites of three different mass ratios of P-doped TiO2 to BiVO4 (1
1. Solution II was stirred at 80 °C until a brown-colored solution was obtained. Subsequently, solution I was added dropwise to solution II, resulting in a blue-colored solution. Stoichiometric amounts of P-doped TiO2 were added to the constant concentration of BiVO4 to obtain composites of three different mass ratios of P-doped TiO2 to BiVO4 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.25, 1
0.25, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, and 1
1, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4). The mixture was adjusted to pH 6.5 and was sonicated for 30 minutes to ensure better mixing and homogenous dispersion of P-doped TiO2 in P-doped TiO2/BiVO4.25,30 The resulting solution was dried overnight at 80 °C, calcined at 500 °C for 4 hours, and then ground to obtain a fine powder. Depending on the compositions of P-TiO2 and BiVO4, the composites are labeled as P-TiO2/BiVO4-x, where x is the theoretical mass percentage of P-TiO2. The synthesis route is depicted in Fig. 1.
4). The mixture was adjusted to pH 6.5 and was sonicated for 30 minutes to ensure better mixing and homogenous dispersion of P-doped TiO2 in P-doped TiO2/BiVO4.25,30 The resulting solution was dried overnight at 80 °C, calcined at 500 °C for 4 hours, and then ground to obtain a fine powder. Depending on the compositions of P-TiO2 and BiVO4, the composites are labeled as P-TiO2/BiVO4-x, where x is the theoretical mass percentage of P-TiO2. The synthesis route is depicted in Fig. 1.
2.3. Photocatalytic activity and kinetic modelling
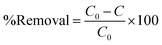 | (i) |
Zero-order kinetic model:
| Ct − C0 = k0t | (ii) |
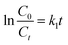 | (iii) |
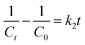 | (iv) |
Furthermore, photocatalytic antibiotic degradation involves surface adsorption, the photocatalyst's oxidation under UV irradiation, and photolysis under UV irradiation alone. Thus, for this hybrid system, it is imperative to determine whether the combined processes increase overall degradation or exhibit synergistic effects. To assess this, the catalytic potential (CP), the synergy index (SI), and the relative contributions (RC) are evaluated for the synthesized photocatalyst, and the catalytic performance is compared using the equations presented.
The following equation defines the catalytic potential of the composites.
| %Catalytic potential = R(catalyst+UV) − (Radsorption + RUV) | (v) |
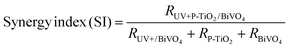 | (vi) |
The relative contribution (RC) of each process in a hybrid system is defined by
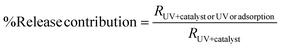 | (vii) |
3. Results and discussion
3.1. Structural and functional properties of the synthesized photocatalysts
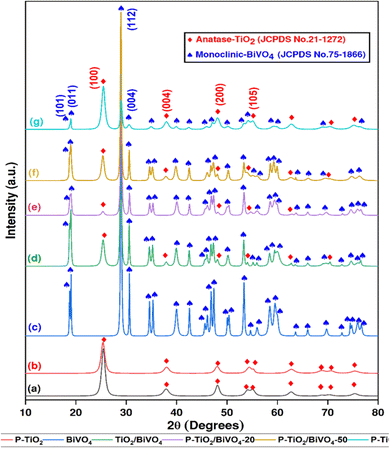
The major diffraction peaks at 2θ = 18.68°, 19°, 28.3°, and 30.61°, corresponding to crystallite phases (101), (011), (114), and (004), indicate the presence of a monoclinic crystallite of BiVO4.20 These major peaks and other diffraction peaks comply with the JCPDS Card No. 75-1866 of monoclinic BiVO4. The absence of any peaks other than those corresponding to the crystalline phases of TiO2 and BiVO4 in the XRD pattern suggests that there are no detectable impurities present. The peak intensity of anatase-TiO2 at phase (100) is also observed to be reduced with an increasing amount of BiVO4 content in the sample, as evident in Fig. 2.
The estimated average crystallite size of the synthesized photocatalysts, as well as the obtained lattice parameters (a and c), are tabulated in Table 1, where the average crystallite size was calculated using the Debye–Scherrer equation. Notably, the values of lattice parameters a and c are pretty close to the theoretical values of the JCPDS cards of the components. The crystallite size of TiO2 was found to be 9.02 nm, which was reduced to 6.04 nm upon P doping, and the average crystallite size increased after coupling with BiVO4. The crystallite sizes of P-TiO2/BiVO4-20, P-TiO2/BiVO4-50, and P-TiO2/BiVO4-80 were 24.67 nm, 19.65 nm, and 13.99 nm, respectively. These larger crystallite sizes are attributable to the higher crystallinity of BiVO4 (44.89 nm) and its larger atomic radius.
| Catalysts | Average crystallite size from XRD (nm) | Lattice parameters, A° | Band gap (eV) | Average particle size from ImageJ (nm) | Elemental composition (wt%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| a | c | Ti | O | P | Bi | V | ||||
| TiO2 | 9.02 | 3.5124 | 9.7827 | 3.15 | 59.94 ± 22.71 | 62.75 | 37.25 | — | — | — |
| P-TiO2 | 6.04 | 3.4977 | 9.4543 | 2.88 | 56.12 ± 41.87 | 53.53 | 45.53 | 0.94 | — | — |
| BiVO4 | 44.89 | 5.1841 | 11.678 | 2.31 | 292.60 ± 143.11 | — | 9.13 | — | 73.38 | 17.49 |
| TiO2/BiVO4 | 24.89 | 3.506 | 11.688 | 2.63 | 115.95 ± 74.19 | 15.06 | 18.12 | — | 52.34 | 14.48 |
| P-TiO2/BiVO4-20 | 24.67 | 3.513 | 11.671 | 2.38 | 152.01 ± 131.32 | 5.03 | 10.32 | 0.11 | 64.96 | 17.16 |
| P-TiO2/BiVO4-50 | 19.65 | 3.506 | 11.673 | 2.45 | 105.7 ± 53.78 | 22.71 | 50.53 | 0.36 | 48.53 | 12.63 |
| P-TiO2/BiVO4-80 | 13.99 | 3.499 | 11.650 | 2.59 | 61.43± 19.55 | 44.26 | 29.61 | 0.92 | 17.03 | 4.76 |
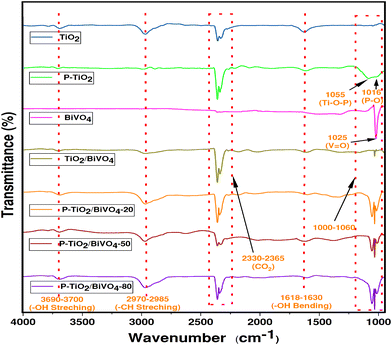
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond,34 which is present in all samples containing BiVO4. For the samples having P as the dopant, two common peaks at 1016 cm−1 and 1055 cm−1 mark the presence of Ti–O–P stretching and P–O bonds, respectively.31 Thus, the presence of a Ti–O–P bond indicates the successful incorporation of P in the lattice of TiO2, substituting Ti4+, and the presence of P–O vibration indicates the coordination of the phosphate ion with TiO2.31,35 Along with these, two weak absorbance peaks are observed at 1618–1630 cm−1 and 3690–3700 cm−1, and the twin peaks in the range of 2330–2965 cm−1 are also marked. These indicate the bending and stretching vibrations of O–H molecules present due to the absorbed water molecule on the surface of the photocatalysts and the presence of adsorbed CO2 molecules.36
O bond,34 which is present in all samples containing BiVO4. For the samples having P as the dopant, two common peaks at 1016 cm−1 and 1055 cm−1 mark the presence of Ti–O–P stretching and P–O bonds, respectively.31 Thus, the presence of a Ti–O–P bond indicates the successful incorporation of P in the lattice of TiO2, substituting Ti4+, and the presence of P–O vibration indicates the coordination of the phosphate ion with TiO2.31,35 Along with these, two weak absorbance peaks are observed at 1618–1630 cm−1 and 3690–3700 cm−1, and the twin peaks in the range of 2330–2965 cm−1 are also marked. These indicate the bending and stretching vibrations of O–H molecules present due to the absorbed water molecule on the surface of the photocatalysts and the presence of adsorbed CO2 molecules.36
The EDX elemental mappings and the spectra (Fig. 4 and Fig. S4–S9) of the photocatalysts further confirm the successful doping of P in TiO2 and the incorporation of P-TiO2 in the final P-TiO2/BiVO4 photo-composites. For instance, in Fig. S5, the color map and spectra confirm the presence of P in P-TiO2, with an estimated amount of approximately 1%. Additionally, EDX elemental mapping of the P-TiO2/BiVO4-80 sample shows a homogeneous elemental distribution (Fig. 4), and the spectra confirm the presence of Ti, O, Bi, V, and P contents in the composite. Moreover, the absence of additional peaks aside from these confirms the absence of probable impurities, as indicated by the XRD analysis. The elemental compositions obtained by EDX mapping for all samples are provided in Table 1, and it is clear that the Ti and P contents increase, whereas the Bi content decreases, as the TiO2 content in the P-TiO2/BiVO4 photo-composite increases. Also, a comparison of elemental mappings in Fig. S8 and S9, and Fig. 4 shows an increase in P content in the synthesized P-TiO2/BiVO4 composites as the amount of P–TiO2 in the matrix increased. The resulting elemental composition was close to the expected values. It is also noteworthy that the P content in the P-TiO2/BiVO4-80 sample is found to be close to that in P-TiO2. In P-TiO2/BiVO4-80, P-TiO2 is expected to constitute 80% of the total mass, and thus the P content should be lower than 0.90%. However, Fig. 4 shows higher P content in the elemental mapping of the P-TiO2/BiVO4-80 sample than that in P-TiO2 shown in Fig. S5. EDX is a standardless semi-quantitative analysis, and due to the selection of different surfaces with non-homogeneous elemental distribution, the obtained elemental composition may differ from the bulk composition. However, our target of successful incorporation and availability of P, TiO2, and BiVO4 in the final composites has been achieved, as further supported by the XRD, FTIR, and UV-vis analyses discussed.
3.2. Regression models for predicting the crystallite size and bandgap
Furthermore, a multivariable linear regression (MLR) model for the synthesized photocatalyst was developed to predict the average crystallite size and bandgap as functions of the photocatalyst's atomic composition. MLR models provide a lower-dimensional estimate of a single dependent variable using multiple independent variables. Here, we set the average crystallite size and the band gap as the main predictive variables. The individual atoms (P, Ti, O, Bi, etc.) present in the synthesized photocatalyst play a significant role in determining the crystallite size and band gap, as discussed in Section 3.1. To reaffirm the collinearity between the crystallite size and the bandgap and the individual atomic compositions of the seven prepared photocatalysts (Table 1), Pearson correlation matrices are constructed (Table 2) for both the crystallite size and the bandgap. In general, the parametric Pearson correlation coefficient, ρ ≥ ±0.8, indicates a stronger linear relationship between two variables, either by positive or negative means, whereas ρ ≥ ±0.6 and ρ ≥ 0.3 are considered moderately firm and fair relationships. Table S1 presents the parametric two-tailed Pearson correlation matrices between the crystallite size and the bandgap for the five atomic component compositions (Ti, P, Bi, V, and O). From there, it is clear that the average crystallite size is strongly statistically correlated with Bi and V and negatively with Ti, that is, the higher the Ti concentration, the smaller the average crystallite size; conversely, the higher the Bi concentration, the larger the average crystallite size. This is also stated in Section 3.1, in accordance with the atomic size. For the bandgap, strong statistical correlations are observed across the same Ti, Bi, and V compositions, with Ti positively correlated and Bi and V negatively correlated. As Ti composition in the catalyst increases, the bandgap increases. The developed MLR models are presented in Table 2, along with the fitting performance indicators (R2, RMSE, and MSE). The predicted bandgap and crystallite sizes from the MLR models, compared with the actual values, are shown in Fig. 5(a) and (b). The R2 values for both models are excellent: 0.958 and 0.999 for the crystallite size and bandgap, respectively. Thus, it is evident that these MLR models are effective for predicting the bandgap and crystallite size from the elemental compositions of the photocatalysts, which may further support property optimization.| Predictive variable | Predictor variable | MLR models | Performance statistics |
|---|---|---|---|
| Crystallite size (nm) | Atomic compositions of Ti, O, P, Bi, and V | CS = 8.30 + 0.134Ti–0.183O–0.438P + 1.933Bi–6.005V | R 2: 0.958; RMSE: 6.502; MSE: 42.27 |
| Band gap (eV) | BG = 4.269-0.0186Ti + 0.00168O–0.485P–0.0284Bi + 0.0066V | R 2: 0.999; RMSE: 0.022.; MSE: 0.0005 |
 | ||
| Fig. 5 The predicted bandgap (a) and crystallite size (b) values of the photocatalysts from the developed MLR models. | ||
3.3. Photocatalytic activity assessment and modelling
As noted, P-TiO2 can increase the number of active sites, enhance electron transfer (due to an altered crystalline structure), and increase photoelectron generation (by lowering the bandgap); its coupling with BiVO4 is expected to further improve the degradation of CIP and MNZ. This is observed for the P-TiO2/BiVO4-x composites, which exhibited higher degradation capacity for both CIP and MNZ than the constituents, P-TiO2, BiVO4, or TiO2/BiVO4. For instance, P-TiO2/BiVO4-50 showed 50.6% and 30.6% higher CIP and MNZ degradation than BiVO4 and TiO2, respectively. A similar trend is found for 20% and 80% composites. In the P-TiO2/BiVO4 composite, the shortcomings of both TiO2 and BiVO4 are offset by each other. For instance, the bandgap of TiO2 is reduced by BiVO4, thereby increasing the utilization of incident light and thereby increasing the number of photogenerated electron–hole pairs, whereas P-TiO2 improves the mobility of photogenerated electrons and enhances adsorption through increased surface roughness. This trend is also reported in ref. 2, where the lowered bandgap of NiSe2/WO3 (2.66 eV) and the suppression of rapid electron–hole recombination by incorporating NiSe2 resulted in 89% degradation of CIP within 120 minutes of treatment, whereas WO3 removed 48%.
Among the three composites, the highest degradation efficiencies were observed for P-TiO2/BiVO4-80, which effectively degraded 94.48% of CIP and 96.07% of MNZ. This composite constitutes 80% of P-TiO2 by mass; the properties of the P-TiO2 are dominant here. Notably, as the proportion of BiVO4 increased in P-TiO2/BiVO4 composites, the photodegradation of CIP and MNZ was reduced. This can be attributed to the inherent shortcomings of BiVO4, including poor photogenerated electron mobility due to its lower bandgap, and reduced adsorption capacity and active sites associated with larger crystallite sizes.23 Moreover, excess BiVO4 can impede UV absorption by blocking the active sites of P-TiO2, thereby hindering electron–hole separation at the heterojunction.25 Thus, the dominant effect of BiVO4 resulted in poor performances among the heterostructures. While P-TiO2/BiVO4-80 had an optimal bandgap (2.59 eV), it exhibited the smallest crystallite size (14 nm) among the heterostructures, enabling higher adsorption capacities and enhanced effective photogenerated electron–hole mobility. Thus, we report the enhanced photocatalytic activity of the synthesized heterostructured photocatalysts, with an optimal preparation ratio of 80% P-TiO2 and 20% BiVO4.
For 180 minutes of CIP treatment, 5.51 ± 2.2% removal was due to UV photolysis, and 25.50 ± 0.77% was from surface adsorption (Fig. 6a). The estimated CP of P-TiO2-BiVO4-80 was 47.48 ± 10.2%, indicating that the photocatalyst addition resulted in, on average, a 47.5% increase in degradation efficiency over that achievable by sole adsorption and photolysis. Hence, the coupled photocatalysts exhibit a synergistic effect across these hybrid processes. For MNZ, the trend is similar, with an estimated CP of 46.50 ± 12.45%, reaffirming the superior activity of the P-TiO2/BiVO4-80 photocatalyst coupled with photolysis.
Now, the contribution of each process to the total degradation can be further visualized using relative contribution (RC) analysis. Among the total CIP degradation, UV photolysis and surface adsorption accounted for 9% and 28%, respectively, and the remaining 63% was attributed to the photocatalyst's oxidative activity. For MNZ degradation, the pattern is similar: 9.90% and 28.22% are attributed to photolysis and adsorption, whereas the majority of degradation (61.88%) is due to photocatalytic activity. Although photogenerated radicals primarily contribute to antibiotic degradation, surface adsorption plays a crucial role in the overall degradation process by bringing pollutants to active sites, after which subsequent degradation occurs. Thus, from the CP and RC analyses, the synergistic adsorption–degradation process and its benefits in achieving high degradation performance are evident.
Now, to understand whether it is beneficial or to what extent the coupling of P-TiO2 with BiVO4 helped to obtain higher photocatalytic performance, the synergy index (SI) values of the TiO2/BiVO4-80 heterostructure are calculated for the CIP and MNZ degradation. Noting the positive, SI ≥ 1 indicates the positive synergy of coupling P-TiO2 with BiVO4 and with the UV photolysis, and SI ≤ 1 indicates ineffective coupling. For 180 minutes of CIP and MNZ treatment, the SI values of P-TiO2-BiVO4-80 are estimated to be 1.09 ± 0.03 and 1.10 ± 0.06, respectively. This confirms the synergistic nature of coupling P-TiO2 with BiVO4 in the UV system for the efficient removal of the antibiotics. This also ratifies that P-TiO2-BiVO4-80 can successfully harness the benefits of both P-TiO2 and BiVO4, as discussed in Section 3.3.1, as well as those of UV-mediated photocatalysis.
3.3.4.1. Effect of pH. The impact of the change of the solution pH on the photodegradation (both for CIP and MNZ) was studied by varying the solution pH at 3, 10, and the natural pH of the antibiotics (6.5 for CIP and 6.3 for MNZ), while the photocatalyst loading was kept at 1 g L−1 and the antibiotic concentration at 20 ppm. The degradation profiles of CIP and MNZ across these pH ranges are shown in Fig. 7, along with the corresponding kinetic models. For CIP, the highest degradation was observed under natural pH conditions (94.5%), approximately 15% and 6.4% higher than that at pH 3 and 10, respectively. CIP exists primarily as a zwitterionic structure with both carboxylic acid and piperazine groups, giving two pKa values at pKa1 6.1 and pKa2 = 8.7. Therefore, the CIP molecules are positively charged at pH below 6.1 and negatively charged at pH above 8.7. Since the calculated point of zero charge (PZC) of the catalyst is 6.31 (Fig. S10), electrostatic repulsion between CIP and the catalyst surface at both acidic and basic pH, hinders the adsorption.37 This is why photodegradation is faster at neutral pH (1.68 and 1.50 times higher than at pH 3 and pH 10), as shown in Fig. 7(a and b). Moreover, photodegradation at basic pH (pH 10) was higher than that at acidic pH, owing to the readily available hydroxide ion, which facilitates the formation of OH˙ as a key component of the AOP.
On the other hand, although MNZ is zwitterionic, its structure, with a highly negative nitro group, makes it a polar compound with a pKa of 2.87. Hence, it is cationic at pH < 2.87 and anionic at pH > 2.87. The MNZ degradation is also maximal at the antibiotic's natural pH (pH 6.3), degrading 96% of MNZ within 180 minutes, whereas the degradation was 84% and 90.2% at pH 3 and pH 10, respectively. The rate of degradation at natural pH was 1.82 and 1.52 times higher than that at pH 3 and pH 10 (Fig. 7c and d). The low degradation at pH 3 is attributable to the scarcity of hydroxide ions. Moreover, particles tend to agglomerate at acidic pH, whereas at basic pH, electrostatic repulsion between the photocatalyst surface and MNZ reduces adsorption.38
3.3.4.2. Effect of initial antibiotic concentration. Additionally, the effects of initial concentrations on the degradation of MNZ and CIP, as well as on the corresponding rate constants, are illustrated in Fig. 8. The antibiotic concentrations were varied from 10 to 30 mg mL−1 for CIP and MNZ, while the catalyst loading was maintained at 1 g L−1 and at the natural pH of the antibiotics. The maximum degradation of both CIP and MNZ occurred at 10 mg L−1, and the corresponding rate constants were also highest. At this initial concentration, the CIP degradation efficiency was 96%, and the rate constant was 1.67 times that at 30 mg L−1. The same trend was observed for MNZ degradation. The degradation efficiency of MNZ at an initial concentration of 10 mg L−1 was 98.24%. The lower degradation at higher concentrations is mainly due to the scarcity of ˙OH, O2˙−, and other oxidative radicals, since the catalyst amount remains constant.39 Moreover, higher pollutant concentrations saturate the active sites on the catalyst surface, thereby hindering adsorption.40,41 This is also evident from the total obtained adsorption of CIP and MNZ after exposure to the dark conditions. As pollutant concentration increased, adsorption capacity decreased for both CIP and MNZ.
3.3.4.3. Effect of catalyst dose. Fig. 9 demonstrates how the photodegradation of CIP and MNZ varies with changes in photocatalyst dosages (0.8, 1, 1.2, and 1.4 g L−1), while keeping the other treatment parameters constant. For both CIP and MNZ, degradation increased as the catalyst concentration rose from 0.8 g L−1 to 1.2 g L−1 and then decreased when the catalyst dosage was further increased to 1.4 g L−1. The maximum degradation for CIP and MNZ was 96.4% and 98.3%, respectively, for 1.2 g L−1; therefore, the optimum catalyst dosage is 1.2 g L−1. The degradation efficiency increases with catalyst dose up to a certain extent because of a greater number of available active sites for adsorption and the availability of oxidative radicals for degradation. However, as the dosage increases further, degradation decreases because the particles impede radiation penetration, thereby reducing the exposure of active sites to UV light.42 This turbidity of the suspension affects charge-carrier separation by reducing UV penetration to active sites. Therefore, the degradation rate decreases with increasing catalyst dosage.43,44
3.3.4.4. Effect of peroxide dose. External oxidizing agents can substantially alter the photocatalyst's degradation rate. In many heterogeneous catalytic AOPs, peroxides (H2O2), peroxymonosulfate (PMS, HSO5−), ozone (O3), and persulfate (S2O82−) are used to mediate the oxidation process. These, upon UV irradiation, greatly facilitate the formation of highly reactive hydroxyl and sulfate radicals and enhance the degradation of organic pollutants.26,45 Thus, we employed H2O2 to assess the impact of external oxidizing agents on the overall degradation of CIP and MNZ, by varying H2O2 doses (10 mM, 20 mM, and 30 mM) while maintaining other operational parameters constant (Fig. S11).
The degradation increases with H2O2 concentration up to 20 mM for both MNZ and CIP and then decreases at 30 mM. Nearly complete degradation of both MNZ and CIP was achieved at a H2O2 concentration of 20 mM. For CIP, the maximum rate constant of 0.02691 min−1 was observed at 20 mM H2O2, decreasing to 0.01192 min−1 at 30 mM. On the other hand, the highest rate constant, 0.02511 min−1, was observed at 20 mM MNZ, decreasing to 0.01135 min−1 as the H2O2 concentration increased to 30 mM. Initially, the degradation rate increased because H2O2 acted as an electron scavenger, thereby reducing electron–hole recombination and hydroxyl radical formation, as shown in eqn (VIII).46 In addition, H2O2 present in the system can aid in the generation of hydroxyl radicals by reacting with superoxide radicals as per eqn (ix).47 Alongside, H2O2 itself can split to form OH˙ upon excitation by UV radiation (eqn (x)).48
| e−(CB) + H2O2 → OH− + OH˙ | (viii) |
| H2O2 + O2−˙ → OH− + OH˙ + O2 | (ix) |
| H2O2 + hν → 2OH˙ | (x) |
| H2O2 + OH˙ → HO2˙ + H2O˙ | (xi) |
| HO2˙ + OH˙ → H2O + O2 | (xii) |
The presence of the nitro (–NO2) group in the structure of MNZ makes it more susceptible to photocatalytic degradation.51 On the other hand, CIP has a complex fluoroquinolone structure, which makes it comparatively more stable than MNZ.52 Here, the degradation in the binary mixture differs little from that in the single solution. Thus, there is no synergistic effect or suppression of photodegradation in the binary solution of CIP and MNZ. This behavior was also reported for the homogeneous AOP, using persulfate-peroxide for the photodegradation of CIP and MNZ.26 Thus, the synthesized TiO2/BiVO4-80 can be used for the simultaneous removal of multiple antibiotics from wastewater.
3.4. Photocatalytic degradation mechanism
The photocatalytic degradation mechanism of MNZ and CIP using the synthesized P-TiO2/BiVO4 composite is depicted in Fig. 12. Under UV irradiation and upon adsorption on the composite, electron–hole pair separation occurs in P-TiO2 and BiVO4. For P-doped TiO2, the substitution of Ti4+ by P5+ results in shallow donor states (P 3p) below the conduction band of TiO2.54 The transfer is mainly due to the relative positions of the CBs. The holes are transferred from the VB of BiVO4 to the more negatively positioned VB of P-TiO2.55 This separation of electron–hole pairs reduces recombination. Moreover, the effective band gap of the heterojunction is also reduced as P-TiO2/BiVO4-80 has a band gap of 2.59 eV. The photogenerated holes react with adsorbed water and hydroxide molecules to generate OH˙ radicals, and the electrons interact with electron scavengers, such as dissolved O2, to generate O2˙− radicals. These O2−˙ radicals act as strong oxidizing agents and undergo further reactions to form OH˙ radicals eventually. Thus, adsorbed CIP and MNZ molecules react with the formed OH˙ and O2˙−, resulting in the formation of degradation products such as CO2 and H2O. The transfer of electron–hole pairs at a heterojunction inhibits electron–hole separation and reduces the band gap, thereby facilitating the degradation of adsorbed antibiotic molecules.4. Concluding remarks, study shortfalls, and way forward
This study systematically investigated the photocatalytic performance of a novel P-doped TiO2/BiVO4 heterostructure photocatalyst, both for individual and binary mixtures of CIP and MNZ antibiotics.The characterization revealed that the incorporation of anatase TiO2 and monoclinic BiVO4 substantially benefited from the favorable features of both, with reduced crystal size and improved surface roughness, leading to higher adsorption capacity, an optimized bandgap, and excellent photogenerated electron–hole transfer. Among the synthesized composites, P-TiO2/BiVO4-80 exhibited superior photocatalytic performance, with an optimal crystal size of 14 nm and a band gap of 2.59 eV, achieving degradation efficiencies exceeding 94% for CIP and 96% for MNZ under UV-A irradiation. In addition, two multivariable linear regression models were developed to predict the crystal structure size and the bandgap from elemental composition. The estimated catalytic potential and the synergy index showed the effectiveness of coupling P-TiO2 with BiVO4 for potential use in AOPs. The photocatalyst exhibited excellent performance in a binary mixture of CIP and MNZ, with a degradation rate comparable to that observed for the individual antibiotics. Moreover, the photocatalyst demonstrated excellent reusability, maintaining over 78% efficiency after five cycles.
This is the first attempt to investigate this novel photocatalyst as a potential solution for the degradation of recalcitrant pollutants in surface water; however, the study has some shortcomings that can guide future improvements to this material for large-scale implementation. Here, we discussed some limitations and suggested future directions for detailed investigation.
• In this study, the material characterization was done using FESEM-EDX, XRD, FTIR, and UV-vis, which couldn’t provide more detailed information on the structural and functional properties of the synthesized catalyst. Advanced spectroscopic and electrochemical techniques, such as transient photocurrent responses and electrochemical impedance spectroscopy, could further elucidate charge-transfer dynamics under different illumination regimes. TEM and TGA can provide essential insights into the successful doping and the thermal stability of the materials. Thus, we suggest further exploration of these.
• Photocatalytic experiments were conducted under controlled UV-A irradiation. While this approach allowed isolation of intrinsic heterojunction effects and reliable benchmarking against TiO2-based systems, it did not fully represent solar-driven operation. Although the composite exhibited a reduced band-gap energy within the visible-light utilization range, the contribution of solar irradiation to long-term degradation efficiency was not analyzed in this initial study. Thus, further investigation of P-TiO2/BiVO4 under visible-light and simulated solar irradiation is required to assess its applicability to real effluents containing natural organic matter, inorganic ions, and competing radical scavengers.
• This study primarily focused on parent compound removal to investigate the catalytic potential of this synthesized novel composite; mineralization extent and the toxicity of transformation intermediates were not comprehensively evaluated. Additionally, UV-vis-based concentration determination for degradation has a well-known limitation: it can overestimate the parent compound if the intermediates have the same absorption wavelength, and this method doesn’t provide information about partially degraded intermediates. Moreover, partial degradation may still yield biologically active or toxic by-products, thereby limiting direct environmental risk assessment. Thus, comprehensive mineralization studies, including total organic carbon (TOC) analysis, identification of degradation intermediates via HPLC or LC-MS analysis, and toxicity assessment of degradation by-products, can help ensure environmental safety and elucidate degradation mechanisms.
• From the practical viewpoint, the large-scale implementation of powder photocatalysts in a wastewater treatment plant is associated with post-processing difficulties, mass loss, turbidity, separation, and recycling issues. The immobilization, specifically biopolymer-based immobilization, provides a solution to these problems, with an additional enhancement in pollutant uptake capacity through a synergistic adsorption–degradation mechanism.33 Further studies of our synthesized photocatalyst immobilized in biopolymer hydrogel matrices (chitosan, alginate, carboxymethyl cellulose, PVA, PANI, etc.) can provide an efficient means for efficient absorber and photocatalytic reactor design. In addition, the benefits, sustainability, and large-scale feasibility of the synthesized P-TiO2/BiVO4 photocatalyst can be evaluated through techno-economic analysis (TEA) integrated with life cycle analysis (LCA). Sometimes, nanomaterials show significant improvements in energy requirements in smaller-scale processes but exhibit substantial energy consumption during synthesis and preparation and throughout the life cycle.12,56 Thus, an integrated TEA-LCA with hotspot analysis can be conducted to identify energy-intensive routes, improve energy efficiency, and reduce the environmental footprint.
Overall, this study offers significant insights into the development of a highly efficient P-TiO2/BiVO4 photocatalyst, which holds great promise for removing antibiotics and other persistent organic pollutants from water. We believe that the proposed future work in these directions would benefit the transition toward sustainable wastewater treatment, reduce the environmental footprint, and help ensure clean water availability for everyone.
Author contributions
Tanzim Ur Rahman: writing – methodology, investigation, data collection, data analysis, and original draft; Musfekur Rahman Dihan: writing – methodology, data analysis, and original draft, review and editing; Abdul Hai: review and editing; Md. Shahinoor Islam: review and editing, supervision.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
Data obtained from the laboratory based experiments were used to produce figures, and analysis and the remaining data are included in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5ma01398d.Further availability of the data will be provided upon request to the corresponding author.
Acknowledgements
This research project was funded by the CASR Postgraduate Research Fund, Bangladesh University of Engineering and Technology (BUET), and BUET Chemical Engineering Forum (BCEF). The authors would like to acknowledge the laboratory support from the Department of Chemical Engineering, Bangladesh University of Engineering and Technology (BUET), Dhaka, Bangladesh.References
- F. Mahmud, T. S. Banhi, H. Roy, M. R. Dihan, Md. S. Islam and Y. Cai, et al., Antibiotic-contaminated wastewater treatment and remediation by electrochemical advanced oxidation processes (EAOPs), Groundw. Sustain. Dev., 2024, 25, 101181 CrossRef.
- T. L. Yusuf, B. O. Ojo, T. Mushiana, N. Mabuba, O. A. Arotiba and S. Makgato, Synergistically enhanced photoelectrocatalytic degradation of ciprofloxacin via oxygen vacancies and internal electric field on a NiSe2/WO3 photoanode, Catal. Sci. Technol., 2024, 14(20), 6015–6026 RSC.
- A. J. Browne, M. G. Chipeta, G. Haines-Woodhouse, E. P. A. Kumaran, B. H. K. Hamadani and S. Zaraa, et al., Global antibiotic consumption and usage in humans, 2000–18: a spatial modelling study, Lancet Planet. Health, 2021, 5(12), e893–e904 CrossRef PubMed.
- A. Guha Roy, Antibiotics in water, Nat. Sustainability, 2019, 2(5), 356 Search PubMed.
- F. Akter, M. R. Amin, K. T. Osman, M. N. Anwar, M. M. Karim and M. A. Hossain, Ciprofloxacin-resistant Escherichia coli in hospital wastewater of Bangladesh and prediction of its mechanism of resistance, World J. Microbiol. Biotechnol., 2012, 28(3), 827–834 CrossRef CAS PubMed.
- J. L. Wilkinson, A. B. A. Boxall, D. W. Kolpin, K. M. Y. Leung, R. W. S. Lai and C. Galbán-Malagón, et al., Pharmaceutical pollution of the world's rivers, Proc. Natl. Acad. Sci. U. S. A., 2022, 119, 8 CrossRef PubMed.
- S. Akter, M. S. Islam, M. H. Kabir, M. A. A. Shaikh and M. A. Gafur, UV/TiO2 photodegradation of metronidazole, ciprofloxacin and sulfamethoxazole in aqueous solution: An optimization and kinetic study, Arabian J. Chem., 2022, 15(7), 103900 CrossRef CAS.
- T. U. Rahman, H. Roy, A. Fariha and A. Z. Shoronika, Al-Mamun MdR, Islam SZ, et al. Progress in plasma doping semiconductor photocatalysts for efficient pollutant remediation and hydrogen generation, Sep. Purif. Technol., 2023, 320, 124141 Search PubMed.
- M. Sharma, D. Rajput, V. Kumar, I. Jatain, T. M. Aminabhavi and G. Mohanakrishna, et al., Photocatalytic degradation of four emerging antibiotic contaminants and toxicity assessment in wastewater: A comprehensive study, Environ. Res., 2023, 231, 116132 Search PubMed.
- M. Rashid Al-Mamun, M. Shofikul Islam, M. Rasel Hossain, S. Kader, M. Shahinoor Islam and M. Zaved Hossain Khan, A novel and highly efficient Ag and GO co-synthesized ZnO nano photocatalyst for methylene blue dye degradation under UV irradiation, Environ. Nanotechnol., Monit. Manage., 2021, 16, 100495 CAS.
- T. U. Rahman, H. Roy, A. Z. Shoronika, A. Fariha, M. Hasan and Md. S. Islam, et al., Sustainable toxic dye removal and degradation from wastewater using novel chitosan-modified TiO2 and ZnO nanocomposites, J. Mol. Liq., 2023, 388, 122764 Search PubMed.
- M. R. Dihan, T. U. Rahman, M. A. J. R. Khan, T. S. Banhi, S. Sadaf and M. S. Reza, et al., Photocatalytic and photoelectrochemical H2 generation for sustainable future: Performance improvement, techno-economic analysis, and life cycle assessment for shaping the reality, Fuel, 2025, 392, 134356 CrossRef CAS.
- Q. Guo, C. Zhou, Z. Ma and X. Yang, Fundamentals of TiO2 Photocatalysis: Concepts, Mechanisms, and Challenges, Adv. Mater., 2019, 31, 50 Search PubMed.
- T. L. Yusuf, O. C. Olatunde, D. Masekela, K. D. Modibane, D. C. Onwudiwe and S. Makgato, Charge Redistribution in NiSe2/MoS2 n–n Heterojunction towards the Photoelectrocatalytic Degradation of Ciprofloxacin, ChemElectroChem, 2024, 11(16), e202400309 CrossRef CAS.
- X. Wang, L. L. Wang, D. Guo, L. L. Ma, B. L. Zhu and P. Wang, et al., Fabrication and photocatalytic performance of C, N, F-tridoped TiO 2 nanotubes, Catal. Today, 2019, 327(2010), 182–189 CrossRef CAS.
- M. R. Al-Mamun, S. Kader, M. S. Islam and M. Z. H. Khan, Photocatalytic activity improvement and application of UV-TiO2 photocatalysis in textile wastewater treatment: A review, J. Environ. Chem. Eng., 2019, 7, 5 Search PubMed.
- L. G. Devi and R. Kavitha, A review on non metal ion doped titania for the photocatalytic degradation of organic pollutants under UV/solar light: Role of photogenerated charge carrier dynamics in enhancing the activity, Appl. Catal., B, 2013, 140–141, 559–587 Search PubMed.
- X. Feng, P. Wang, J. Hou, J. Qian, Y. Ao and C. Wang, Significantly enhanced visible light photocatalytic efficiency of phosphorus doped TiO2 with surface oxygen vacancies for ciprofloxacin degradation: Synergistic effect and intermediates analysis, J. Hazard. Mater., 2018, 351, 196–205 CrossRef CAS PubMed.
- N. O. Gopal, H. H. Lo, T. F. Ke, C. H. Lee, C. C. Chou and J. D. Wu, et al., Visible Light Active Phosphorus-Doped TiO2 Nanoparticles: An EPR Evidence for the Enhanced Charge Separation, J. Phys. Chem. C, 2012, 116(30), 16191–16197 CrossRef CAS.
- V. Liapun, M. B. Hanif, M. Sihor, X. Vislocka, S. Pandiaraj, U. V. K., G. K. Thirunavukkarasu, M. F. Edelmannová, M. Reli, O. Monfort, K. Kočí and M. Motola, et al., Versatile application of BiVO4/TiO2 S-scheme photocatalyst: Photocatalytic CO2 and Cr(VI) reduction, Chemosphere, 2023, 337, 139397 CrossRef CAS PubMed.
- H. Roy, T. U. Rahman, Md. A. J. R. Khan, Md. R. Al-Mamun, S. Z. Islam and Md. A. Khaleque, et al., Toxic dye removal, remediation, and mechanism with doped SnO2-based nanocomposite photocatalysts: A critical review, J. Water Proc. Eng., 2023, 54, 104069 CrossRef.
- B. Y. Cheng, J. S. Yang, H. W. Cho and J. J. Wu, Fabrication of an Efficient BiVO4 –TiO2 Heterojunction Photoanode for Photoelectrochemical Water Oxidation, ACS Appl. Mater. Interfaces, 2016, 8(31), 20032–20039 CrossRef CAS PubMed.
- M. Liaqat, S. Kausar, T. Iqbal, S. Afsheen, A. Younas and M. Zubair, et al., Synergistic Photocatalytic Activity of TiO2/BiVO4 Nanocomposites: Optimization, Characterization, and Recyclability for Dye and Antibiotic Degradation, J. Inorg. Organomet. Polym. Mater., 2024, 34(7), 3246–3257 CrossRef CAS.
- S. Cipagauta-Díaz, A. Estrella-González, M. Navarrete-Magaña and R. Gómez, N doped -TiO2 coupled to BiVO4 with high performance in photodegradation of Ofloxacin antibiotic and Rhodamine B dye under visible light, Catal. Today, 2022, 394–396, 445–457 Search PubMed.
- C. Wu, J. Dai, J. Ma, T. Zhang, L. Qiang and J. Xue, Mechanistic study of B-TiO2/BiVO4 S-scheme heterojunction photocatalyst for tetracycline hydrochloride removal and H2 production, Sep. Purif. Technol., 2023, 312, 123398 CrossRef CAS.
- M. R. Dihan, Md. A. Alam, S. Akter, Md. A. Gafur and Md. S. Islam, Statistical modeling and application of machine learning for antibiotic degradation using UV/persulfate-peroxide based advanced oxidation process, Heliyon., 2025, 11(13), e43664 CrossRef CAS.
- M. R. Al-Mamun, S. Kader and M. S. Islam, Solar-TiO2 immobilized photocatalytic reactors performance assessment in the degradation of methyl orange dye in aqueous solution, Environ. Nanotechnol., Monit. Manage., 2021, 16, 100514 Search PubMed.
- R. Rescigno, O. Sacco, V. Venditto, A. Fusco, G. Donnarumma and M. Lettieri, et al., Photocatalytic activity of P-doped TiO2 photocatalyst, Photochem. Photobiol. Sci., 2023, 22(6), 1223–1231 Search PubMed.
- B. Xie, C. He, P. Cai and Y. Xiong, Preparation of monoclinic BiVO4 thin film by citrate route for photocatalytic application under visible light, Thin Solid Films, 2010, 518(8), 1958–1961 CrossRef CAS.
- K. T. Drisya, M. Solís-López, J. J. Ríos-Ramírez, J. C. Durán-Álvarez, A. Rousseau and S. Velumani, et al., Electronic and optical competence of TiO2/BiVO4 nanocomposites in the photocatalytic processes, Sci. Rep., 2020, 10(1), 13507 CrossRef CAS PubMed.
- S. Y. Mendiola-Alvarez, Ma. A. Hernández-Ramírez, J. L. Guzmán-Mar, L. L. Garza-Tovar and L. Hinojosa-Reyes, Phosphorous-doped TiO2 nanoparticles: synthesis, characterization, and visible photocatalytic evaluation on sulfamethazine degradation, Environ. Sci. Pollut. Res., 2019, 26(5), 4180–4191 CrossRef CAS PubMed.
- S. Y. Mendiola-Alvarez, M. A. Hernández-Ramírez, J. L. Guzmán-Mar, L. L. Garza-Tovar and L. Hinojosa-Reyes, Phosphorous-doped TiO 2 nanoparticles: synthesis, characterization, and visible photocatalytic evaluation on sulfamethazine degradation, Environ. Sci. Pollut. Res., 2019, 26(5), 4180–4191 CrossRef CAS PubMed.
- M. R. Dihan, S. Himel, Md. F. Abedin, S. M. A. Nayeem, A. A. Promy and Md. S. Islam, A Partial Air-Freeze Drying Strategy for TiO2-Immobilized Chitosan Beads: Balancing Swelling and Stability for Improved Photocatalysis, J. Mater. Res. Technol., 2025, 38, 4115–4132 CrossRef CAS.
- V. Sivakumar, R. Suresh, K. Giribabu and V. Narayanan, BiVO4 nanoparticles: Preparation, characterization and photocatalytic activity, ed A. M. Z. Slawin, Cogent Chemistry. 2015;vol. 1(1):1074647 Search PubMed.
- Y. Xia, Y. Jiang, F. Li, M. Xia, B. Xue and Y. Li, Effect of calcined atmosphere on the photocatalytic activity of P-doped TiO2, Appl. Surf. Sci., 2014, 289, 306–315 CrossRef CAS.
- Md. R. Al-Mamun, Md. Z. Iqbal Rokon, Md. A. Rahim, Md. I. Hossain, Md. S. Islam and Md. R. Ali, et al., Enhanced photocatalytic activity of Cu and Ni-doped ZnO nanostructures: A comparative study of methyl orange dye degradation in aqueous solution, Heliyon., 2023, e16506 CrossRef CAS PubMed.
- A. Salma, S. Thoröe-Boveleth, T. C. Schmidt and J. Tuerk, Dependence of transformation product formation on pH during photolytic and photocatalytic degradation of ciprofloxacin, J. Hazard. Mater., 2016, 313, 49–59 CrossRef CAS PubMed.
- F. Bashiri, S. M. Khezri, R. R. Kalantary and B. Kakavandi, Enhanced photocatalytic degradation of metronidazole by TiO2 decorated on magnetic reduced graphene oxide: Characterization, optimization and reaction mechanism studies, J. Mol. Liq., 2020, 314, 113608 Search PubMed.
- P. A. Mangrulkar, S. P. Kamble, M. M. Joshi, J. S. Meshram, N. K. Labhsetwar and S. S. Rayalu, Photocatalytic Degradation of Phenolics by N-Doped Mesoporous Titania under Solar Radiation, Int. J. Photoenergy, 2012, 2012, 1–10 CrossRef.
- B. Gupta, A. K. Gupta, C. S. Tiwary and P. S. Ghosal, A multivariate modeling and experimental realization of photocatalytic system of engineered S–C3N4/ZnO hybrid for ciprofloxacin removal: Influencing factors and degradation pathways, Environ. Res., 2021, 196, 110390 CrossRef CAS PubMed.
- A. K. Jha and S. Chakraborty, Photocatalytic degradation of tetracycline and ciprofloxacin antibiotic residues in aqueous phase by biosynthesized nZVI using Sal (Shorea robusta) leaf extract, Aqua, 2023, 72(3), 230–245 Search PubMed.
- L. Zang, Photocatalytic Degradation of Water Pollutants Using Nano-TiO2, Green Energy and Technology, Vol. 33, 2011 Search PubMed.
- M. A. Rauf and S. S. Ashraf, Fundamental principles and application of heterogeneous photocatalytic degradation of dyes in solution, Chem. Eng. J., 2009, 151(1–3), 10–18 CrossRef CAS.
- D. Zhao, J. Wang, Z. Zhang and J. Zhang, Photocatalytic degradation of omethoate using NaY zeolite-supported TiO 2, Front. Chem. Eng. China, 2009, 3(2), 206–210 Search PubMed.
- M. H. Moslehi, M. S. Zadeh, K. Nateq, Y. D. Shahamat, N. A. Khan and N. Nasseh, Statistical computational optimization approach for photocatalytic-ozonation decontamination of metronidazole in aqueous media using CuFe2O4/SiO2/ZnO nanocomposite, Environ. Res., 2024, 242, 117747 CrossRef CAS PubMed.
- Y. Zang and R. Farnood, Effect of Hydrogen Peroxide on the Photocatalytic Degradation of Methyl tert-butyl Ether, Top. Catal., 2006, 37(2–4), 91–96 CrossRef CAS.
- A. Touati, T. Hammedi, W. Najjar, Z. Ksibi and S. Sayadi, Photocatalytic degradation of textile wastewater in presence of hydrogen peroxide: Effect of cerium doping titania, J. Ind. Eng. Chem., 2016, 35, 36–44 Search PubMed.
- M. R. Al-Mamun, M. N. Karim, N. A. Nitun, S. Kader, M. S. Islam and M. Z. H. Khan, Photocatalytic performance assessment of GO and Ag co-synthesized TiO2 nanocomposite for the removal of methyl orange dye under solar irradiation, Environ. Technol. Innovation, 2021, 22 Search PubMed.
- N. Bouanimba, R. Zouaghi, N. Laid and T. Sehili, Factors influencing the photocatalytic decolorization of Bromophenol blue in aqueous solution with different types of TiO2 as photocatalysts, Desalination, 2011, 275(1–3), 224–230 CrossRef CAS.
- G. S. Wang, C. H. Liao, H. W. Chen and H. C. Yang, Characteristics of natural organic matter degradation in water by UV/H2O2 treatment, Environ. Technol., 2006, 27(3), 277–287 CrossRef CAS PubMed.
- J. Sun, R. Chu and Z. U. H. Khan, A Theoretical Study on the Degradation Mechanism, Kinetics, and Ecotoxicity of Metronidazole (MNZ) in ˙OH- and SO4˙−-Assisted Advanced Oxidation Processes, Toxics., 2023, 11, 9 Search PubMed.
- P. P. Singh, G. Pandey, Y. Murti, J. Gairola, S. Mahajan and H. Kandhari, et al., Light-driven photocatalysis as an effective tool for degradation of antibiotics, RSC Adv., 2024, 14(29), 20492–20515 RSC.
- N. P. Rini, N. I. Istiqomah and E. Suharyadi, Enhancing photodegradation of methylene blue and reusability using CoO/ZnO composite nanoparticles, Case Stud. Chem. Environ. Eng., 2023, 7, 100301 CrossRef CAS.
- C. Sotelo-Vazquez, N. Noor, A. Kafizas, R. Quesada-Cabrera, D. O. Scanlon and A. Taylor, et al., Multifunctional P-Doped TiO 2 Films: A New Approach to Self-Cleaning, Transparent Conducting Oxide Materials, Chem. Mater., 2015, 27(9), 3234–3242 CrossRef CAS.
- Y. R. Lv, C. J. Liu, R. K. He, X. Li and Y. H. Xu, BiVO4/TiO2 heterojunction with enhanced photocatalytic activities and photoelectochemistry performances under visible light illumination, Mater. Res. Bull., 2019, 117, 35–40 CrossRef CAS.
- M. R. Dihan, T. Ahmed, T. U. Rahman, K. Bahar, N. M. Likhon, S. S. Sayem, et al., in Sustainable nano-innovations at the heart of the water–energy–food nexus, Climate Change Impacts on the Water-Energy-Food Nexus, Elsevier, 2026, p. 539–56, Available from: https://www.sciencedirect.com/science/chapter/edited-volume/abs/pii/B978044322320400027X Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |