Open Access Article
Open Access ArticleDFT-driven insights into the electronic, magnetic, and transport properties of a 2D Nb3C2 MXene for high-performance Li-ion batteries
Samia
Tahira
a,
Safwara
Siddiqua
a,
Fouzia Perveen
Malik
 *a and
Hummera
Rafique
b
*a and
Hummera
Rafique
b
aMaterials Modelling Lab, School of Interdisciplinary Engineering & Science (SINES), National University of Sciences and Technology (NUST), Sector H-12, Islamabad 44000, Pakistan. E-mail: fouzia@sines.nust.edu.pk
bDepartment of Chemistry, University of Gujrat, Gujrat-50700, Pakistan
First published on 7th April 2026
Abstract
‘MXenes’, two-dimensional materials, have attained significant attention for their outstanding characteristics inherent to their nanostructures. However, to date, the Nb3C2 MXene, particularly in Cr-doped form, has not been theoretically explored for Li-ion battery applications. In this work, first-principles calculations were performed to explore the structural, electronic, magnetic, and transport properties of newly designed pristine Nb3C2 and Cr-doped Nb3C2, along with their energy storage potential, using the FP-LAPW approach. Both structures are dynamically and thermally stable, as confirmed from phonon dispersion and AIMD simulations. Electronic properties, including the band structure and density of states, indicate metallic behavior in both structures with an indirect band gap, fulfilling a key requirement for electrode materials in energy storage systems. Pristine Nb3C2 exhibits an essentially non-magnetic ground state, while Cr-doped Nb3C2 exhibits ferromagnetic behavior. Theoretical capacities of 169 mAh g−1 for pristine Nb3C2 and 280 mAh g−1 for Cr-doped Nb3C2 were obtained, indicating a substantial enhancement upon Cr doping and exceeding that of pristine Nb2C (170 mAh g−1) reported in the literature. The predicted electrochemical properties unveil that both pristine and Cr-doped Nb3C2 possess favorable open-circuit voltages within the desirable range for anode materials, along with high electronic conductivity and improved gravimetric capacity. Furthermore, transport property analysis based on semi-classical Boltzmann theory highlights their promising thermoelectric behavior, complementing their electrochemical performance and providing a comprehensive evaluation of MXenes in energy storage devices.
1. Introduction
The successful synthesis and research on graphene have compelled scientists to explore more graphene-like 2D materials, both experimentally and theoretically, made of elements beyond carbon, such as phosphorene, hexagonal boron nitride (h-BN), and transition metal dichalcogenides. These materials possess unique properties, such as a high surface area-to-volume ratio and excellent electrical and thermal conductivity, making them promising candidates for use in supercapacitors and batteries.1However, the performance of current batteries is insufficient to adequately satisfy the escalating demand for extensive energy storage. In order to address and comprehend this challenge, an immediate imperative arises to actively pursue novel materials that exhibit enhanced performance attributes within energy storage systems.2 Recently, a new class of 2D materials known as MXenes have been drawing much interest because of their exceptional geometries and electronic topologies, i.e., excellent conductivity, fast ion diffusion, and hydrophilic nature, which make them competent materials for storage systems.3 MXenes are graphene-like layered 2D early transition metal carbides, nitrides, or carbonitrides that have been prepared by selectively etching the A layers from 3D MAX phases (Mn+1AXn, where n ranges from 1 to 3), for example, M2AX, M3AX2, and M4AX3, where M denotes a transition metal (e.g., Ti, Nb, Mo, Ta, Sc, V, Zr, etc.); A is an element from group III-A or IV-A (Al, Si, or Ga); and X corresponds to carbides, nitrides, or carbonitrides.4 Because the M–X bond is stronger than the M–A bond, the A atom can be selectively etched to yield 2D MXenes.5
A number of studies have been carried out on Nb2C and Nb4C3 MXenes because of their potential use in energy storage devices, catalysis, and biomedical applications. Ghidiu et al. reported the synthesis and characterization of the Nb4C3 MXene obtained by selectively etching Nb4AlC3.6 Shen et al. demonstrated the synthesis of the Nb2C MXene and explored its electrochemical performance for supercapacitors.7 Nishat et al. described the electrochemical performance of Nb2C and Nb2CO2 for Na- and Li-ion batteries using an ab initio approach.8 Although several studies have been conducted on Nb-based MXenes, such as Nb2C and Nb4C3, studies on Nb3C2 remain limited. Furthermore, Cr doping in Nb2C and Nb4C3 MXenes has not yet been explored.6,7 In particular, a systematic investigation of their structural stability and electronic, magnetic, and electrochemical properties upon transition metal doping, especially with Cr, has not been explored. Hence, a detailed first-principles study is required to evaluate the potential of Cr-doped Nb3C2 MXenes for high-performance Li-ion battery applications.
Furthermore, niobium-based MXenes show superior performance as anode materials in Li-ion batteries compared to Ti-based MXenes, as reported in the literature.9 Nb3C2 holds a different M/X ratio compared to other members of the MXene family, making it unique and influencing its layer thickness, electron density, surface chemistry, and mechanical strength, thereby positioning it as a promising and versatile candidate for future energy devices.10 Due to its layered configuration, the Nb3C2 MXene possesses a relatively small interlayer spacing, which hinders the intercalation and deintercalation of Li+ ions during battery operation.11 To overcome this limitation, various dopant atoms have been introduced to expand the interlayer distance and investigate their influence on the electrocatalytic behavior of MXenes.12 Doping not only increases the lattice parameters but also tunes the band gap and enhances the electrical conductivity, thereby improving the overall electrochemical performance of the material.13
In recent years, several studies have demonstrated that doping can effectively modify the interlayer spacing and surface chemistry of MXenes, thereby improving their electrochemical performance.14 Among potential dopants, Cr is notable for its good electrical conductivity and ionic radius (0.62 Å), closely matching that of niobium (0.64 Å), making it a promising candidate for Nb3C2 MXene doping.15
Herein, a systematic first-principles investigation of pristine Nb3C2 and Cr-doped Nb3C2 MXenes is performed to explore their structural stability, electronic structure, magnetic behavior, transport properties, and electrochemical performance as potential anode materials for Li-ion batteries. Furthermore, the effect of Cr doping on the structural, electronic, and electrochemical properties of Nb3C2 is comprehensively examined.
The findings of this work provide valuable theoretical insights into the properties of the Nb3C2 MXene and highlight the potential of Cr doping to improve its electrochemical performance, thereby supporting future experimental investigations and the development of advanced MXene-based energy storage devices.
2. Computational method
All computational calculations were executed within the density functional theory (DFT) framework, employing the full-potential augmented plane waves plus local orbital (FP-LAPW+lo) method, as implemented through the WIEN2k code.16 The exchange–correlation potential energies of the electrons were calculated in the generalized gradient approximation (GGA) via the Perdew–Burke–Ernzerhof (PBE) functional.17The two-dimensional Nb3C2 MXene structure was constructed by selectively etching the A-group element from its corresponding MAX phase precursor to obtain a Nb3C2 monolayer. A vacuum layer of 15–20 Å was introduced along the c-direction to avoid the interactions between periodic layers. To evaluate the effect of substitutional Cr doping, one Nb atom in the supercell was substituted with a Cr atom. All structures were fully optimized prior to the electronic, magnetic and transport property calculations.2,6
Firstly, the unit cell and supercell structure of Nb3C2 and Cr-doped Nb3C2 were generated and optimized at 1000 (19 × 19 × 2) and 200 k-points within a 2 × 12 × 7 k-mesh in the irreducible Brillouin zone (IBZ), respectively, using GGA-PBE. The expansion of valence wave functions within the Nb spheres was conducted up to lmax = 10. Additionally, the Fourier expansion of the charge density was pursued up to Gmax = 14. For structural and electronic calculations, the potential was expanded within the Nb spheres, employing a combination of spherical harmonic functions and the Brillouin zone. The cut-off energy for the separation of valence and core electrons was chosen as −6 Ry, which corresponds to RMT Kmax = 7, where RMT is the smallest of all atomic sphere radii and Kmax determines the truncation of the reciprocal lattice expansion of the wave functions in the interstitial region.18,19 Convergence of the total energy was set to 10−5 eV between successive iterations, with a Gaussian smearing factor of 0.05 eV. Atomic positions were fully relaxed until the maximum force on each atom was below 10−3 eV Å−1. The plane-wave basis set was taken with an energy cut-off of 600 eV. For the electronic structure analysis, the Fermi level (EF), defined as the highest occupied electronic state at 0 K, was set to 0 eV as the reference energy.20,21
Phonon dispersion calculations were performed using the PHONOPY package interfaced with WIEN2k to evaluate the dynamical stability of pristine and Cr-doped Nb3C2 structures. In addition, ab initio molecular dynamics (AIMD) simulations were carried out to examine thermal stability. The AIMD simulations were conducted in the canonical ensemble (NVT) using a Nosé–Hoover thermostat at 500 K. A time step of 1 fs was employed, and the total simulation time was 100 ps, corresponding to 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 MD steps.22
000 MD steps.22
Spin-polarization calculations were carried out to determine the magnetic ordering of the 2D MXenes. Pristine Nb3C2 exhibited negligible magnetism (very small magnetic moments within numerical error), while Cr-doping stabilized a ferromagnetic state. The magnetic behavior was further evaluated using both GGA-PBE and GGA-PBE+U, where U represents the Hubbard potential. The U values of Nb (0.22 Ry = 3 eV)20 and Cr (U = 0.15 Ry = 2 eV) were adopted from the literature, where similar parameters successfully captured the localized d-electron correlations.23
Although applying the U slightly increased the bandgap, the valence and conduction band levels near the Fermi level remained consistent, ensuring reliable results for electronic and optical properties.24 The simplicity of this approach, along with the availability of all required inputs from first principles, makes it highly suitable for high-throughput computations, where it has gained wide acceptance.25 The electronic transport properties were calculated using the BoltzTraP2 code interfaced with WIEN2k under the constant relaxation time approximation (RTA), yielding the thermal and electrical conductivities.26 To evaluate the Li storage capability of pristine and Cr-doped Nb3C2 for Li-ion batteries, we computed the adsorption energy,27 the open circuit voltage (OCV),28 and the theoretical capacity (Q).29
The adsorption energy (Eads) was determined using:
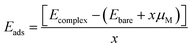 | (1) |
| V = [Etotal(Nb3C2) + xEtotal(Li) − Etotal(LixNb3C2)]/x | (2) |
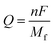 | (3) |
3. Results and discussion
3.1. Structure prediction of novel MXenes
According to results obtained via first-principles DFT calculations, the newly designed Nb3C2 MXene structure was found to belong to the hexagonal geometry with the space group number 194 (P63/mmc) and lattice parameters a = b = 3.131 Å, c = 19.24 Å with α = β = 90° and γ = 120°. Two inequivalent niobium atoms were identified as Nb(1) and Nb(2), along with one carbon atom, having fractional coordinates Nb(1) (2/3, 1/3, 0.875), Nb(2) (0, 0, 0.5) and C: (1/3, 2/3, 0.924). The obtained lattice constant values are very close to the reported literature value of 3.3008 Å.30To elucidate the effect of Cr doping in the Nb3C2 structure, a 2 × 1 × 1 supercell was generated. This supercell contains six Nb atoms and two C atoms. A 25% Cr-doped Nb3C2 structure was then optimized using 1000 k-points within a 2 × 12 × 7 k-mesh in the irreducible Brillouin zone (IBZ). The schematic views of pristine and Cr-doped Nb3C2 are shown in Fig. 1.
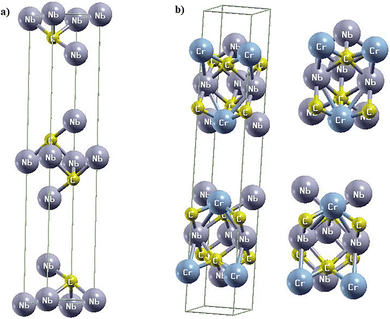 | ||
| Fig. 1 (a) Crystal structure of pristine Nb3C2. (b) Schematic view of the 2 × 1 × 1 supercell of Cr-doped Nb3C2. | ||
3.2. Structural and thermal stability
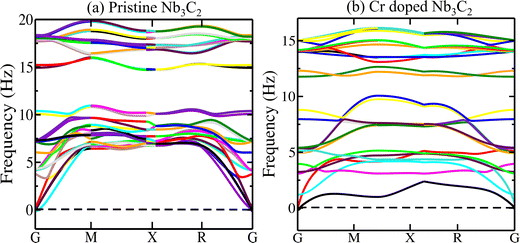
The phonon dispersion curves for both pristine and Cr-doped Nb3C2 are presented in Fig. 2. It is evident that no imaginary (negative) frequencies are present throughout the Brillouin zone, confirming the dynamical stability of both structures. A slight shift in phonon band separation is observed in the frequency range of approximately 10–15 THz between pristine and doped Nb3C2 (Fig. 2a and b), indicating modifications in interatomic interactions and bonding strength due to doping. Both systems exhibit three acoustic and several optical branches, as typically observed in two-dimensional materials.The slope of the acoustic branches is slightly higher in pristine Nb3C2 as shown in Fig. 2a, compared to the doped structure in Fig. 2(b), suggesting a relatively higher phonon group velocity. Furthermore, the absence of a distinct gap between acoustic and optical modes may enhance acoustic–optical phonon scattering, which can influence the thermal conductivity of both pristine and doped Nb3C2 MXene layers.31
The energy evolution of pristine and Cr-doped Nb3C2 MXene systems during the AIMD simulations is presented in Fig. 3, with pristine Nb3C2 shown in Fig. 3a and Cr-doped Nb3C2 in Fig. 3b. The total energy of both structures remains nearly constant throughout the simulation, indicating good numerical stability. The potential, kinetic, and non-bonding energies exhibited moderate fluctuations around their average values, which are expected due to the thermal motion of atoms. For pristine Nb3C2 (Fig. 3a), energy variations remain confined within a narrow range over the 10 ps simulation, suggesting that the structural framework remains intact without significant distortion. A similar trend is observed for the Cr-doped Nb3C2 system (Fig. 3b), where the energy components fluctuate around equilibrium values without any noticeable drift in total energy. These stable energy oscillations confirm that Cr incorporation does not compromise the structural integrity of the MXene lattice, demonstrating that both systems maintain thermal stability under the simulated conditions.22
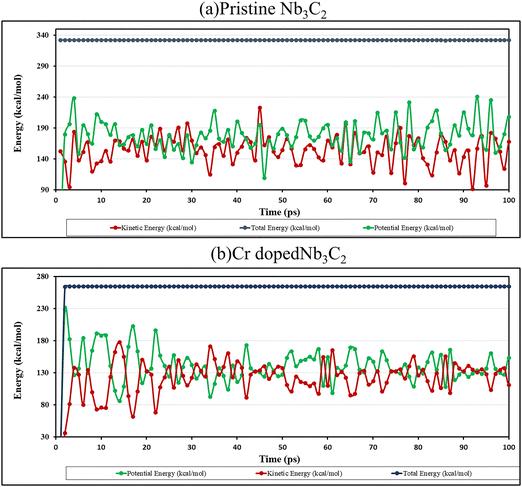 | ||
| Fig. 3 AIMD simulation of (a) pristine Nb3C2 and (b) Cr-doped Nb3C2 at 500 K, showing energy evolution with atoms in their most stable position. | ||
3.3. Electronic properties of pristine and Cr-doped Nb3C2
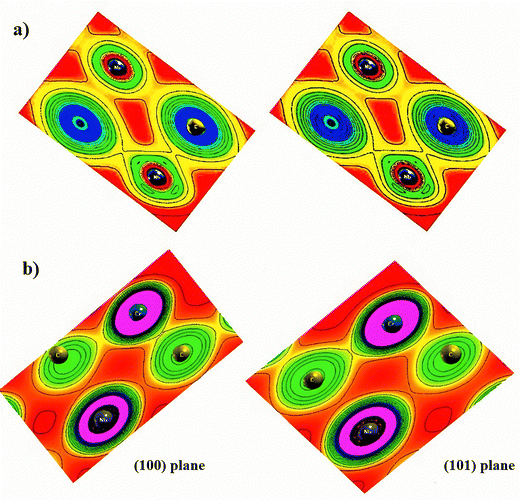
Exploring the electronic properties of a potential electrode requires determining whether the material exhibits metallic, semiconducting, or insulating behavior.32 In this study, Cr atoms were employed as dopants to investigate the electronic characteristics, including charge density distribution, band structure, and density of states (DOS). The energy gaps between valence and conduction bands were identified from the calculated density of states (DOS) and band structure.33Charge density plots provide direct insight into the bonding characteristics of the system.34Fig. 4(a) and (b) shows the electron density of pristine and Cr-doped Nb3C2 along the (100) and (110) planes. It is revealed that Cr-doping modifies the bonding nature between Nb and C atoms. As shown in Fig. 4(a), the charge density contour of pristine Nb3C2 indicates a covalent bond between C and Nb atoms, while in Fig. 4(b), Cr-doped Nb3C2 exhibits a covalent bond between Cr and C atoms due to the overlap of their valence electrons. In addition, the spherical charge distribution around Nb atoms in Nb3C2 indicates ionic bonding between the electronegative C atoms and electropositive Nb atoms. In the Cr-doped Nb3C2, the weak covalent interaction between Nb and C atoms suggests a weaker hybridization tendency of Nb towards C, as compared with pristine Nb3C2.35 These features confirm that Cr doping significantly alters the bonding environment and electronic interactions in Nb3C2.
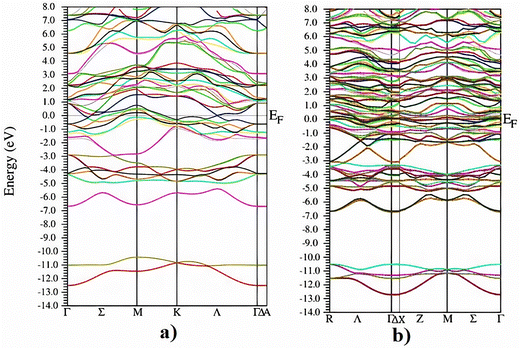
The interpretation of the band structure provides detailed insights into the electronic behaviour of materials, serving as a fundamental indicator of their conductivity and their responsiveness to doping-induced modifications. The band structure of pristine Nb3C2 (Fig. 5a) indicated metallic behavior, as the band gap was found to be zero.36
In contrast, Cr-doping (Fig. 3b) increased the density of electronic states around the Fermi level; however, the band gap for the Cr doped Nb3C2 structure also remained zero. This confirms that Nb3C2 retains its metallic character even after Cr doping. Moreover, Cr doping increases the electronic states above the Fermi level due to the additional energy levels introduced by Cr atoms within the crystal lattice, as evident from Fig. 5b.37
This expansion is shown to be due to the n-type doping effect of Cr atoms, bringing about distinct modifications in the pristine Nb3C2 band structure, which in turn influences the electrical conductivity of the material.38 Band gap analysis therefore suggests that both pristine and doped Nb3C2 exhibit effective electrical conductivity, as this enables efficient electron transport during electrochemical processes.39
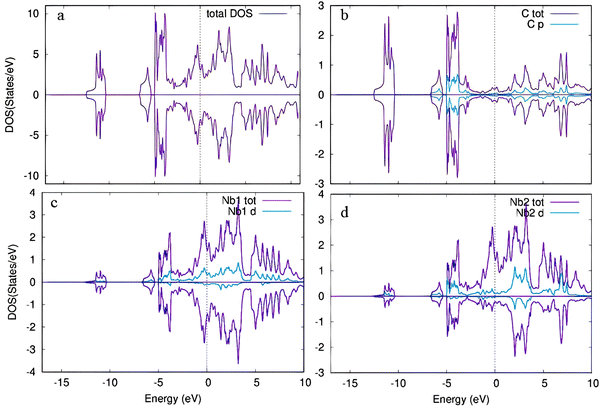
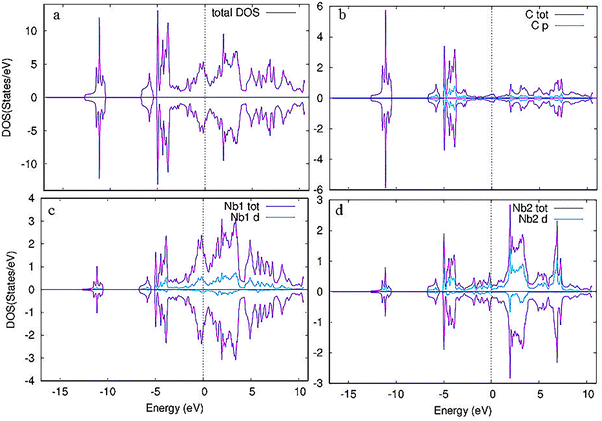
To estimate the number of states at different energy levels occupied by electrons, the partial and total density of states (PDOS and TDOS) for pristine and Cr-doped Nb3C2 were calculated, as shown in Fig. 6 and 7, respectively. For the TDOS, Fig. 6(a) shows a maximum peak in the density of states around −6.5 eV, indicating a high concentration of electrons at this energy level.40
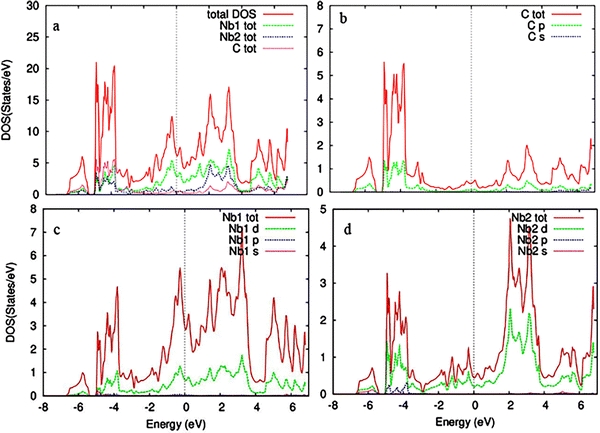 | ||
| Fig. 6 (a) Total density of states (TDOS) of pristine Nb3C2 and (b)–(d) partial density of states (PDOS) of C, Nb1, and Nb2 atoms, respectively. | ||
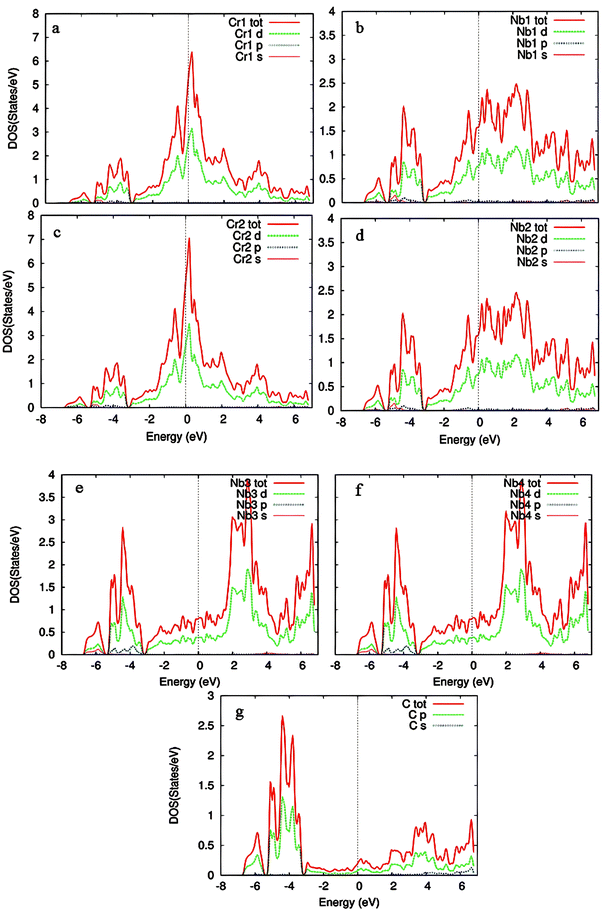 | ||
| Fig. 7 Partial density of states (PDOS) of Cr-doped Nb3C2 in a 2 × 1 × 1 supercell: (a) Cr1, (b) Nb1, (c) Cr2, (d) Nb2, (e) Nb3, (f) Nb4, and (g) C atoms. | ||
For the PDOS of pristine Nb3C2 (Fig. 6a–d), the maximum electronic states at the Fermi level (EF) mainly originate from the Nb1-4d orbital states.41 The valence band is dominated by C-2p states, with an admixture of Nb d-orbital contributions, confirming covalent Nb–C bonding.42 Above the Fermi level, the conduction band is primarily composed of Nb1-4d orbitals, with additional contributions from Nb2-4d states, suggesting anisotropic electronic transport.43 The dominance of Nb-4d orbitals near the Fermi level confirms its metallic conductivity, which is beneficial for electrochemical performance when used as an electrode material.44
Cr doping significantly increases the density of states per electron volt. At the Fermi level, the dominant contributions arise from Cr1-3d and Cr2-3d orbitals, with smaller contributions from Nb1-4d and Nb2-4d states. The upper part of the conduction band consists of states from Cr1, Cr2, Nb1, Nb2, Nb3, and Nb4 d orbitals. In the energy range of 2–4 eV above EF, the conduction band is mainly dominated by Nb3-4d and Nb4-4d orbitals, with minor contributions from Cr1-3d, Cr2-3d, and Nb1-4d and Nb2-4d states. Meanwhile, C-2p orbitals contribute negligibly to the conduction band but play a major role in the valence band through hybridization with Nb3-4d and Nb4-4d orbitals.
As evident from Fig. 7, the density of states in both the valence and conduction bands cross the Fermi level without opening a gap, thereby confirming the metallic character of Cr-doped Nb3C2.45 The PDOS plots show the orbital contributions of Cr, Nb, and C atoms in Cr-doped Nb3C2. Cr atoms (a) and (c) mainly contribute through d-states near the Fermi level, indicating strong hybridization. Nb atoms (b)–(f) also show dominant d-orbital contributions, crucial for electronic conductivity. C atoms (g) contribute primarily through p-states, supporting the overall electronic interaction in the heterostructure.
3.4. Magnetic properties
Magnetism in MXenes has not been extensively explored for the M3C2 phase, either theoretically or experimentally. For comparison, Ti3C2 magnetism has been briefly studied by Shien et al., who reported ferromagnetic behavior.46,47Herein, pristine Nb3C2 exhibited an essentially non-magnetic ground state, as the calculated magnetic moments for Nb2 were −0.00002µB (GGA) and −0.00007µB (GGA+U), which were negligibly small and within the margin of computational error.48 Although the PDOS of Nb2 shows a slight asymmetry between spin-up and spin-down states (Fig. 8a), the imbalance is too weak to generate a measurable magnetic response in the whole compound. Consequently, the net magnetic moment is effectively zero, confirming the non-magnetic nature of pristine Nb3C2.
Fig. 8(b)–(d) further illustrates the DOS and PDOS of C-2p, Nb1-4d, and Nb2-4d states. By applying the Hubbard potential (GGA+U), the magnetic moment of the carbon atom became aligned with that of Nb2 and anti-parallel to Nb1, which enhanced the overall magnetic moment. This increase arises because the GGA+U correction shifts the partial density of states of Nb1 and Nb2, accounting for stronger electron–electron correlation effects49 as shown in Fig. 9. Cr doping in Nb3C2 enhanced the magnetism and stabilized a ferromagnetic ground state.
Table 1 shows that pristine Nb3C2 is nearly non-magnetic with negligible total magnetic moment using both PBE-GGA and PBE-GGA+U calculations. Upon Cr doping, the system exhibits a substantial increase in magnetism (the total moment rising from ∼7.5 to ∼12.0µB), primarily originating from the strong spin polarization of Cr-3d states and significant interstitial contributions, while Nb and C atoms provide smaller site-dependent moments. Fig. 8 further supports this, as the spin-polarized PDOS confirms the ferromagnetic nature through asymmetry of electronic states below and above the Fermi level for majority and minority spin channels.50 The Cr-d orbitals (Fig. 10c and d) dominate the magnetism with strong spin polarization, whereas C-p orbitals (Fig. 10b) remain nearly symmetric. Nb-d states (Fig. 10e–h) display moderate asymmetry, indicating induced contributions through hybridization with Cr-d states. Overall, the net magnetic moment mainly arises from unpaired Cr-d electrons, with Nb atoms providing secondary polarization, thereby stabilizing robust ferromagnetism in Cr-doped Nb3C2, which is beneficial for charge storage and transport in lithium-ion batteries.
| Compound | Atoms | Magnetic moment (PBE-GGA) | Magnetic moment (PBE-GGA+U) |
|---|---|---|---|
| Pristine Nb3C2 | Nb1 (µB) | 0.00003 | 0.00026 |
| Nb2 (µB) | −0.00002 | −0.00007 | |
| C (µB) | 0.00001 | −0.000001 | |
| Interstitial (µB) | 0.00013 | 0.00059 | |
| Total (µB) | 0.00024 | 0.00146 | |
| Cr-doped Nb3C2 | Cr1 (µB) | 1.76157 | 2.51618 |
| Cr2 (µB) | 1.76010 | 2.55740 | |
| Nb1–Nb4 (µB) | −0.03548 | 0.11141 | |
| −0.03698 | 0.16468 | ||
| −0.02989 | −0.07683 | ||
| −0.02989 | −0.07441 | ||
| C (µB) | −0.04485 | −0.07782 | |
| −0.04473 | −0.08433 | ||
| −0.01336 | −0.02645 | ||
| −0.01323 | −0.03146 | ||
| Interstitial (µB) | 0.94320 | 2.08746 | |
| Total (µB) | 7.48972 | 12.04418 | |
The calculated magnetic moment values for pristine Nb3C2 and Cr-doped Nb3C2 are summarized in Table 1.
3.5. Transport properties
The transport properties, including electrical conductivity, Seebeck coefficient, and thermal conductivity, were evaluated as a function of chemical potential at temperatures of 300 K, 500 K, and 700 K. These parameters provide insight into the charge-carrier behavior and electron transport efficiency in Nb3C2, which is essential for assessing its potential as an electrode material for energy storage applications.51 Transport properties were also estimated for the lithiated systems of both pristine and Cr-doped Nb3C2, since lithiation and delithiation during Li-ion battery charge/discharge significantly influence charge-carrier mobility and the density of states near the Fermi level.52The Seebeck coefficient is a key parameter for understanding charge-carrier behavior and transport efficiency.53Fig. 11 depicts the Seebeck coefficient as a function of chemical potential for various Nb3C2-based compounds. For pristine Nb3C2, Fig. 11(a) shows four distinct peaks at chemical potentials of −5.9 eV, −5.2 eV, −3.2 eV, and −3.0 eV. Outside this range, the Seebeck coefficient rapidly approaches zero, which indicates good transport performance. In contrast, lithiated Nb3C2, Cr-doped Nb3C2, and lithiated Cr-doped Nb3C2 (Fig. 11(b)–(d)) show smaller Seebeck values over the entire chemical potential range, consistent with strong metallic behavior. In Li-ion battery electrodes, metallic conductivity is essential for efficient charge transport.54 These results suggest that the lithiated phases, particularly Li2Nb3C2 and Li4Cr-doped Nb3C2, exhibit high metallic conductivity and may provide more efficient electronic transport during Li-ion battery operation compared to pristine Nb3C2.
 | ||
| Fig. 11 Seebeck coefficient as a function of chemical potential at three different temperatures for the compounds: (a) pristine Nb3C2, (b) Li2Nb3C2, (c) Cr-doped Nb3C2, and (d) Li4–Cr-doped Nb3C2. | ||
The maximum values of the Seebeck coefficient for the different compounds at various temperatures are summarized in Table 2. The results show that, with increasing temperature, the carrier concentration increases, leading to a reduction in the Seebeck coefficient, as expected for metallic systems. This behavior is consistent with the metallic nature of all compounds.
| Temperature (K) | Nb3C2 | Li2Nb3C2 | Cr-doped Nb3C2 | Li4CrNb3C2 |
|---|---|---|---|---|
| 300 | 4.2 × 10−4 | 1.5 × 10−4 | 2.9 × 10−4 | 8.0 × 10−5 |
| 500 | 2.0 × 10−4 | 1.3 × 10−4 | 1.8 × 10−4 | 6.5 × 10−5 |
| 700 | 1.0 × 10−4 | 1.2 × 10−4 | 1.2 × 10−4 | 6.0 × 10−5 |
Electrical conductivity reflects the ease of electron flow in a material, and materials with efficient transport typically exhibit high conductivity. In metallic systems, conductivity generally decreases with increasing temperature but increases with chemical potential, due to higher carrier concentration and improved mobility.55 As shown in Fig. 12a and c, the electrical conductivity approaches zero near −5.5 eV at 300 K for both delithiated Nb3C2 and delithiated Cr-doped Nb3C2. In contrast, the lithiated phases shown in Fig. 12(b) and (d) exhibited significantly higher conductivity than their delithiated counterparts, indicating enhanced electron conduction during Li-ion battery operation. The maximum values of electrical conductivity are summarized in Table 3.
 | ||
| Fig. 12 Electronic conductivity as a function of chemical potential at three different temperatures for: (a) pristine Nb3C2, (b) Li2Nb3C2, (c) Cr-doped Nb3C2, and (d) Li4–Cr-doped Nb3C2. | ||
| Compound | Electrical conductivity (S m−1) |
|---|---|
| Li2Nb3C2 | 4.7 × 1020 |
| Nb3C2 | 4.4 × 1020 |
| LiCr–Nb3C2 | 1.9 × 1020 |
| Cr–Nb3C2 | 1.7 × 1020 |
Fig. 13 depicts the thermal conductivity of Nb3C2-based systems as a function of chemical potential at three different temperatures. For all compounds, κ increases with temperature, consistent with their metallic nature.56 Pristine Nb3C2 shows a smoother variation in thermal conductivity with chemical potential, whereas the other systems exhibit stronger variations, maintaining higher conductivity over a broader chemical potential (μ) range. Combined with the Seebeck coefficient and electrical conductivity results, these trends indicate that lithiation significantly enhances both charge and heat transport.57 In particular, Li2Nb3C2 and Li4–Cr-doped Nb3C2 demonstrate excellent transport properties, making them promising candidates for efficient Li-ion battery electrodes.
 | ||
| Fig. 13 Thermal conductivity as a function of chemical potential at three different temperatures for the compounds: (a) pristine Nb3C2, (b) Li2Nb3C2, (c) Cr-doped Nb3C2, and (d) Li4–Cr-doped Nb3C2. | ||
3.6. Electrochemical performance of MXenes as electrode materials
Metallic behavior is a desirable characteristic for electrode materials.58 The electronic properties confirm that both pristine Nb3C2 and Cr-doped Nb3C2 are metallic, thereby fulfilling this requirement. In electrochemical applications, intercalating species play a crucial role, with Li+ ions being the most widely used in Li-ion batteries.59 In this study, Li+ intercalation into pristine Nb3C2 and Cr-doped Nb3C2 MXenes was investigated to evaluate their storage capability as electrode materials. The electrochemical suitability of these MXenes was examined through adsorption energy analysis.| V = [Etotal(Nb3C2) + xEtotal(Li) − Etotal(Li2Nb3C2)]/x | (4) |
| V = [Etotal(CrNb3C2) + xEtotal(Li) − Etotal(Li4CrNb3C2)]/x | (5) |
The corresponding intercalation reactions can be expressed as:
| Nb3C2 + 2Li → Li2Nb3C2 | (6) |
| CrNb3C2 + 4Li → Li4CrNb3C2 | (7) |
The theoretical capacity of pristine Nb3C2 for Li-ion batteries was found to be 169 mAh g−1, which is comparable to the reported value of Nb2C (170 mAh g−1).69 In contrast, Cr-doped Nb3C2 exhibits a significantly higher storage capacity of 280 mAh g−1, indicating that Cr doping substantially enhances the Li-ion storage performance of the MXene.
4. Conclusions
Using first-principles calculations, we systematically investigated the structural, electronic, magnetic, and transport properties of pristine Nb3C2 and Cr-doped Nb3C2, as well as their potential as electrode materials for Li-ion batteries. Structural analysis confirmed that Nb3C2 and Cr-doped Nb3C2 adopt hexagonal (P63/mmc) and orthorhombic (Pmm2) crystal structures, respectively. Phonon and AIMD results confirm that both pristine Nb3C2 and Cr-doped Nb3C2 are dynamically and thermally stable, with no significant structural distortions observed. These findings validate the reliability of the subsequent electronic and magnetic property calculations.Charge density contours revealed that Cr doping substantially modifies the Nb–C bonding nature, while the band structures and density of states (DOS) confirm metallic behavior (indirect zero-band gap) in both systems, primarily originating from Nb 4d states. Moreover, substitutional Cr doping transforms non-magnetic pristine Nb3C2 into a ferromagnetic material with a finite magnetic moment, making it a promising candidate for 2D spintronic applications.
Transport property evaluations based on Boltzmann transport theory demonstrate that lithiation significantly enhances charge and heat transport. In particular, Li2Nb3C2 and Li4Cr-doped Nb3C2 exhibit high electrical and thermal conductivities with low Seebeck coefficients, confirming their metallic character and suitability as Li-ion battery electrodes. Furthermore, Cr-doped Nb3C2 outperforms pristine Nb3C2, suggesting superior transport performance for high-efficiency energy storage applications.
Electrochemical analysis further supports Cr-doped Nb3C2 as a promising electrode material, owing to its higher electronic conductivity, higher open-circuit voltage, and improved theoretical capacity compared to pristine Nb3C2. These improved electrochemical and transport properties make Cr-doped Nb3C2 a potential candidate for high performance lithium-ion battery electrodes, particularly in applications requiring fast charge transport and high energy density, such as portable electronic devices and electric vehicles. Overall, these findings demonstrate that Cr doping is an effective strategy for tailoring the properties of Nb3C2 MXenes, underscoring their strong potential for next-generation spintronic and energy storage applications.
Conflicts of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.Data availability
The computational data that support the findings of this study are available upon request. All the related data are presented in the form of figures and tables in the manuscript.Acknowledgements
The authors gratefully acknowledge the School of Interdisciplinary Engineering & Science (SINES) Islamabad, National University of Science and Technology, for providing computational resources and facilities to support this work.References
- B. Zhang, Y. Huang, W. Bao, B. Wang, Q. Meng, L. Fan and Q. Zhang, Two-Dimensional Stable Transition Metal Carbides MnC and NbC with Prediction and Novel Functionalizations, RSC Adv., 2018, 8, 12345–12356, 10.1039/C8RA12345A.
- X. Zhang, Z. Zhang and Z. Zhou, MXene-based materials for electrochemical energy storage, J. Energy Chem., 2018, 27, 73–85, DOI:10.1016/j.jechem.2017.08.004.
- Y. I. Jhon, I. K. Han, J. H. Lee and Y. M. Jhon, Microscopic understanding of exceptional orientation-dependent tensile and fracture responses of two-dimensional transition-metal carbides, Appl. Surf. Sci., 2022, 585, 152557, DOI:10.1016/j.apsusc.2021.152557.
- J. Pang, R. G. Mendes, A. Bachmatiuk, L. Zhao, H. Q. Ta, T. Gemming, H. Liu, Z. Liu and M. H. Rümmeli, Applications of 2D MXenes in energy conversion and storage systems, Chem. Soc. Rev., 2019, 48, 72–133, 10.1039/C8CS00324F.
- A. Zaheer, S. A. Zahra, M. Z. Iqbal, A. Mahmood, S. A. Khan and S. Rizwa, Nickel-adsorbed two-dimensional Nb2C MXene for enhanced energy storage applications, RSC Adv., 2022, 12, 4624–4633, 10.1039/D2RA00014H.
- M. Ghidiu, M. Naguib, C. Shi, O. Mashtalir, L. M. Pan, B. Zhang, J. Yang, Y. Gogotsi, S. J. L. Billinge and M. W. Barsoum, Synthesis and characterization of two-dimensional Nb4C3 (MXene), Chem. Commun., 2014, 50, 9517–9520, 10.1039/C4CC03366C.
- B. Shen, X. Liao, X. Zhang, H.-T. Ren, J.-H. Lin, C.-W. Lou and T.-T. Li, Synthesis of Nb2C MXene-based 2D layered structure electrode material for high-performance battery-type supercapacitors, Phys. Chem. Chem. Phys., 2020, 22, 23900–23908, 10.1039/D0CP03726A.
- N. Sultana, A. A. Amin, E. J. Payton and W. K. Kim, Insights into Nb2C and Nb2CO2 as high-performance anodes for sodium- and lithium-ion batteries: An ab initio investigation, arXiv, 2025, preprint, arXiv:2504.08953 DOI:10.48550/arXiv.2504.08953.
- U. Kalsoom, S. Khan, M. Kashif, H. S. Yaseen, S. A. Hussain, S. Azizi and M. Maaza, MXene-based hybrid composites for lithium-ion batteries: advances in synthesis strategies and electrochemical performance, Ionics, 2025, 1–21 Search PubMed.
- R. Akhter and S. S. Maktedar, MXenes: a comprehensive review of synthesis, properties, and progress in supercapacitor applications, J. Materiomics, 2023, 9(6), 1196–1241, DOI:10.1016/j.jmat.2023.08.011.
- J. Jiang, Y. Zou, A. Arramel, F. Li, J. Wang, J. Zou and N. Li, Intercalation engineering of MXenes towards highly efficient photo(electrocatalytic) hydrogen evolution reactions, J. Mater. Chem. A, 2021, 9, 24195–24214, 10.1039/D1TA07332J.
- Z. Wang, Z. Dong, B. Wu, Z. Wang, Z. Qiu, D. Wang, Q. Zeng, X. Liu, K. N. Hui, Z. Liu and Y. Zhang, Unlocking the critical roles of N, P co-doping in MXene for lithium–oxygen batteries: elevated d-band center and expanded interlayer spacing, J. Colloid Interface Sci., 2024, 676, 368–377, DOI:10.1016/j.jcis.2024.07.138.
- K. Arole, S. E. Pas, R. M. Thakur, L. A. Amiouny, M. H. Kabir, M. Dujovic, M. Radovic, J. L. Lutkenhaus, M. J. Green and H. Liang, Effects of intercalation on ML-Ti3C2Tz MXene properties and friction performance, ACS Appl. Mater. Interfaces, 2024, 16(46), 60538–60548, DOI:10.1021/acsami.4c12157.
- H. Qian, H. Ren, Y. Zhang, X. He, W. Li, J. Wang, J. Hu, H. Yang, H. M. K. Sari, Y. Chen and X. Li, Surface doping vs. bulk doping of cathode materials for lithium-ion batteries: a review, Electrochem. Energy Rev., 2022, 5(4), 2 CrossRef CAS.
- R. D. Shannon, Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides, Acta Crystallogr., Sect. A: Cryst. Phys., Diffr., Theor. Gen. Crystallogr., 1976, 32, 751–767, DOI:10.1107/S0567739476001551.
- G. R. Berdiyorov, Optical properties of functionalized Ti3C2T2 (T = F, O, OH) MXene: first-principles calculations, AIP Adv., 2016, 6(5), 055105, DOI:10.1063/1.4943752.
- P. Blaha, K. Schwarz, G. K. H. Madsen, D. Kvasnicka and J. Luitz, WIEN2k, An Augmented Plane Wave + Local Orbitals Program for Calculating Crystal Properties, Technical University of Vienna, Austria, 2001, pp. 1–302 Search PubMed.
- T. W. Keal and D. J. Tozer, A semiempirical generalized gradient approximation exchange–correlation functional, J. Chem. Phys., 2004, 121(12), 5654–5660, DOI:10.1063/1.1778374.
- W. Chen, J. H. Pöhls, G. Hautier, D. Broberg, S. Bajaj, U. Aydemir, Z. M. Gibbs, H. Zhu, M. Asta, G. J. Snyder and B. Meredig, Understanding thermoelectric properties from high-throughput calculations: trends, insights, and comparisons with experiment, J. Mater. Chem. C, 2016, 4(20), 4414–4426, 10.1039/C5TC04339E.
- J. Carrete; N. Mingo; S. Wang and S. CurtaroloNanograined half-Heusler semiconductors as advanced thermoelectrics: an ab initio high-throughput statistical study, arXiv, 2014, preprint, arXiv:1408.5859 DOI:10.48550/arXiv.1408.5859.
- M. Miyata, T. Ozaki, T. Takeuchi, S. Nishino, M. Inukai and M. Koyano, High-throughput screening of sulfide thermoelectric materials using electron transport calculations with OpenMX and BoltzTraP, J. Electron. Mater., 2018, 47, 3254–3259, DOI:10.1007/s11664-017-6020-9.
- A. Shamsieva, A. Evseev, S. Kaviani, O. V. Nedopekin, H. Zakaryan and I. Piyanzina, DFT analysis of furan-based covalent organic framework as electrode materials for lithium and calcium ion batteries, Comput. Theor. Chem., 2025, 125, 115445, DOI:10.1016/j.comptc.2025.115445.
- J. Fatheema, M. Fatima, N. B. Monir, S. A. Khan and S. Rizwan, A comprehensive computational and experimental analysis of stable ferromagnetism in layered 2D Nb-doped Ti3C2 MXene, Phys. E, 2020, 124, 114253, DOI:10.1016/j.physe.2020.114253.
- Z. Tong, Y. Suo, S. Zhang and J. Yang, Spin-polarized DFT+U study of surface-functionalized Cr3C2 MXenes: tunable electronic and magnetic behavior for spintronics, Materials, 2025, 18(15), 3709, DOI:10.3390/ma18153709.
- F. Aguilera-Granja, R. H. Aguilera-del-Toro and J. L. Morán-López, A first-principles systematic study of the structural, electronic, and magnetic properties of Heusler X2MnZ with X = Fe, Co, Ni, Cu, Ru, Rh, Pd, Ag, Pt, Au and Z = Al, Si, Ga, Ge, In and Sn, Mater. Res. Express, 2019, 6(10), 106118, DOI:10.1088/2053-1591/ab243c.
- L. Bassman Oftelie, P. Rajak and R. K. Kalia, et al., Author Correction: Active learning for accelerated design of layered materials, npj Comput. Mater., 2022, 8, 215, DOI:10.1038/s41524-022-00902-8.
- F. Ricci, W. Chen, U. Aydemir, G. J. Snyder, G. M. Rignanese, A. Jain and G. Hautier, An ab initio electronic transport database for inorganic materials, Sci. Data, 2017, 4, 170085, DOI:10.1038/sdata.2017.85.
- L. Wang, W. Liu, F. Bai, X. Zheng, C. Yin, J. Wei, J. Ma, H. Bai and B. Dong, Theoretical study on lithium storage performance of V-doped Ti2CO2 MXene, RSC Adv., 2024, 14, 19945, 10.1039/d4ra03618b.
- J. W. Gonzalez; S. Vizcaya and E. Suarez MorellV2C-based lithium batteries: the influence of magnetic phase and Hubbard interaction, arXiv, 2022, preprint, arXiv:2204.04764 DOI:10.48550/arXiv.2204.04764.
- R.-W. Yan, X.-H. Li, R.-Z. Zhang and H.-L. Cui, Computational investigation on Cr-doped Sc2CO2 MXene under strain for electronic properties, quantum capacitance, and photocatalytic activity, Langmuir, 2024, 40(37), 19619–19630, DOI:10.1021/acs.langmuir.4c02282.
- X. Yan, W. Cao and H. Li, Thermoelectric properties of X3N2O2 (X = Hf, Zr) MXene monolayers: a first-principles study, RSC Adv., 2023, 13, 18323–18327, 10.1039/D3RA02835F.
- F. B. Oliveira, E. N. Lima, M. C. da Silva, A. L. da Rosa and T. Frauenheim, Exploring charge density distribution and electronic properties of hybrid organic-germanium layers, Phys. Chem. Chem. Phys., 2020, 22, 22055–22065, 10.1039/D0CP03024D.
- M. Fatima, J. Fatheema, N. B. Monir, A. H. Siddique, B. Khan, A. Islam, D. Akinwande and S. Rizwan, Nb-doped MXene with enhanced energy storage capacity and stability, Front. Chem., 2020, 8, 168, DOI:10.3389/fchem.2020.00168.
- Q. Sun, Z. Fu and Z. Yang, Tunable magnetic and electronic properties of the Cr-based MXene (Cr2C) with functional groups and doping, J. Magn. Magn. Mater., 2020, 514, 167141, DOI:10.1016/j.jmmm.2020.167141.
- M. Khazaei, M. Arai, T. Sasaki, C.-Y. Chung, N. S. Venkataramanan, M. Estili, Y. Sakka and Y. Kawazoe, Novel electronic and magnetic properties of two-dimensional transition metal carbides and nitrides, Adv. Funct. Mater., 2013, 23, 2185–2192, DOI:10.1002/adfm.201202502.
- B. Soundiraraju and B. K. George, Two-dimensional titanium nitride (Ti2N) MXene: synthesis, characterization, and potential application as surface-enhanced Raman scattering substrate, ACS Nano, 2017, 11, 8892–8900, DOI:10.1021/acsnano.7b03129.
- B. Anasori, M. R. Lukatskaya and Y. Gogotsi, 2D metal carbides and nitrides (MXenes) for energy storage, Nat. Rev. Mater., 2017, 2, 16098, DOI:10.1038/natrevmats.2016.98.
- F. Ebisawa, T. Kurokawa and S. Nara, Electrical properties of polyacetylene/polysiloxane interface, J. Appl. Phys., 1983, 54, 3255–3259, DOI:10.1063/1.332488.
- I. R. Shein and A. L. Ivanovskii, Planar nano-block structures Tin+1Al0.5Cn and Tin+1Cn (n = 1, 2) from MAX phases: structural, electronic properties and relative stability from first-principles calculations, Superlattices Microstruct., 2012, 52(2), 147–157, DOI:10.1016/j.spmi.2012.04.014.
- A. N. Yaresko, Electronic band structure and exchange interactions in ferromagnetic CrO2, Phys. Rev. B: Condens. Matter Mater. Phys., 2008, 77, 115106, DOI:10.1103/PhysRevB.77.115106.
- S. L. Dudarev, G. A. Botton, S. Y. Savrasov, C. J. Humphreys and A. P. Sutton, Electron-energy-loss spectra and the structural stability of nickel oxide: an LSDA + U study, Phys. Rev. B:Condens. Matter Mater. Phys., 1998, 57, 1505–1509, DOI:10.1103/PhysRevB.57.1505.
- Y. Chen, X. Li, Z. Wang, J. Zhou and Z. Sun, Magnetic properties and tunable electronic structures of transition-metal-doped MXenes: a first-principles study, J. Phys. Chem. C, 2019, 123, 17987–17994, DOI:10.1021/acs.jpcc.9b03654.
- H. Wang, H. Feng and J. Li, et al., Graphene and graphene-like anode materials for lithium-ion batteries, Adv. Mater., 2014, 26, 4880–4900, DOI:10.1002/adma.201306031.
- G. J. Snyder and E. S. Toberer, Complex thermoelectric materials, Nat. Mater., 2008, 7, 105–114, DOI:10.1038/nmat2090.
- B. Xu, Y. Qi and C. Liu, et al., Recent progress in MXene-based materials for electrochemical energy storage, Adv. Funct. Mater., 2020, 30, 1905812, DOI:10.1002/adfm.201905812.
- D. Beretta, N. Neophytou, J. M. Hodges, M. G. Kanatzidis, D. Narducci, M. Martin-Gonzalez, M. Beekman, B. Balke, G. Cerretti, W. Tremel, A. Zevalkink, A. I. Hofmann, C. Müller, B. Dörling, M. Campoy-Quiles and M. Caironi, Thermoelectrics: from history, a window to the future, Mater. Sci. Eng., R, 2019, 138, 100501, DOI:10.1016/j.mser.2018.09.001.
- G. Berdiyorov, Effect of lithium and sodium ion adsorption on the electronic transport properties of Ti3C2 MXene, Appl. Surf. Sci., 2015, 359, 161–165, DOI:10.1016/j.apsusc.2015.10.050.
- J.-M. Tarascon and M. Armand, Issues and challenges facing rechargeable lithium batteries, Nature, 2001, 414, 359–367, DOI:10.1038/35104644.
- B. Hammer and J. K. Nørskov, Theoretical surface science and catalysis—calculations and concepts, Adv. Catal., 2000, 45, 71–129, DOI:10.1016/S0360-0564(02)45013-4.
- M. Xia, T. Zhang, J. Wu, W. Lu, Z. Pan and J. Qiao, Transition metal layer substitution in Mo2CS2 MXene: enhanced Li adsorption and electronic structure modulation, ACS Omega, 2023, 8, 23412–23420, DOI:10.1021/acsomega.3c02080.
- Q. Tang, Z. Zhou and P. Shen, Are MXenes promising anode materials for Li ion batteries? Computational studies on electronic properties and Li storage capability of Ti3C2 and Ti3C2X2 (X = F, OH) monolayer, J. Am. Chem. Soc., 2012, 134, 16909–16916, DOI:10.1021/ja308463r.
- C. Eames and M. S. Islam, Ion intercalation into two-dimensional transition-metal carbides: global screening for new high-capacity battery materials, J. Am. Chem. Soc., 2014, 136, 16270–16276, DOI:10.1021/ja508154e.
- N. Lu, L. Li and M. Liu, A review of carrier thermoelectric-transport theory in organic semiconductors, Phys. Chem. Chem. Phys., 2016, 18(29), 19503–19525 RSC.
- P. Sun, B. Wei, J. Zhang, J. M. Tomczak, A. M. Strydom, M. Søndergaard, B. B. Iversen and F. Steglich, Large Seebeck effect by charge-mobility engineering, Nat. Commun., 2015, 6(1), 7475 CrossRef CAS PubMed.
- S. Poncé, W. Li, S. Reichardt and F. Giustino, First-principles calculations of charge carrier mobility and conductivity in bulk semiconductors and two-dimensional materials, Rep. Prog. Phys., 2020, 83(3), 036501 CrossRef PubMed.
- G. Frenking and N. Fröhlich, The nature of the bonding in transition-metal compounds, Chem. Rev., 2000, 100(2), 717–774 CrossRef CAS PubMed.
- M. M. Mallick and S. Vitta, Giant Enhancement in High-Temperature Thermoelectric Figure-of-Merit of Layered Cobalt Oxide, LiCoO2, Due to a Dual Strategy—Co-Substitution and Lithiation, Inorg. Chem., 2017, 56(10), 5827–5838 CrossRef CAS PubMed.
- Y. Ohno, D. K. Young, B. A. Beschoten, F. Matsukura, H. Ohno and D. D. Awschalom, Electrical spin injection in a ferromagnetic semiconductor heterostructure, Nature, 1999, 402(6763), 790–792 CrossRef CAS.
- Y. Zhu, T. Gao, X. Fan, F. Han and C. Wang, Electrochemical techniques for intercalation electrode materials in rechargeable batteries, Acc. Chem. Res., 2017, 50(4), 1022–1031 CrossRef CAS PubMed.
- C. Eames and M. S. Islam, Ion intercalation into two-dimensional transition-metal carbides: global screening for new high-capacity battery materials, J. Am. Chem. Soc., 2014, 136(46), 16270–16276 CrossRef CAS PubMed.
- W. Ju, T. Li, X. Su, H. Li, X. Li and D. Ma, Au cluster adsorption on perfect and defective MoS 2 monolayers: structural and electronic properties, Phys. Chem. Chem. Phys., 2017, 19(31), 20735–20748 RSC.
- H. Naaz, F. P. Malik, A. Mahmood and A. Irfan, Investigation of 2D Nb3C2-Based MXenes as the Anode Material for LIBs: A Theoretical Study, ACS Omega, 2025, 10, 5283–5295 CrossRef CAS PubMed.
- L. Wang, W. Liu, F. Bai, X. Zheng, C. Yin, J. Wei, J. Ma, H. Bai and B. Dong, Theoretical study on lithium storage performance of V-doped Ti 2 CO 2 MXene, RSC Adv., 2024, 14(28), 19945–19952 RSC.
- J. Wang and W. Q. Han, A review of heteroatom doped materials for advanced lithium–sulfur batteries, Adv. Funct. Mater., 2022, 32(2), 2107166 CrossRef CAS.
- T. Hiemstra and W. H. Van Riemsdijk, A surface structural approach to ion adsorption: the charge distribution (CD) model, J. Colloid Interface Sci., 1996, 179(2), 488–508 CrossRef CAS.
- M. P. Mercer, C. Peng, C. Soares, H. E. Hoster and D. Kramer, Voltage hysteresis during lithiation/delithiation of graphite associated with meta-stable carbon stackings, J. Mater. Chem. A, 2021, 9(1), 492–504 RSC.
- C. Yang, S. Yu, Y. Ma, C. Lin, Z. Xu, H. Zhao, S. Wu, P. Zheng, Z. Z. Zhu, J. Li and N. Wang, Cr3+ and Nb5+ co-doped Ti2Nb10O29 materials for high-performance lithium-ion storage, J. Power Sources, 2017, 360, 470–479 CrossRef CAS.
- K. E. Prenger, MXenes and Their Heterostructures with Oxides as Electrodes for Supercapacitors, Doctoral dissertation, Tulane University, 2022 Search PubMed.
- R. Santoy-Flores, H. N. Fernández-Escamilla, J. I. Páez-Ornelas, E. G. Perez-Tijerina, J. Guerrero-Sánchez, R. Ponce-Pérez, N. Takeuchi and M. G. Moreno-Armenta, Nb2C and Nb2CO2 MXenes as anodes in Li-ion batteries: a comparative study by first-principles calculations, ACS Omega, 2024, 9(26), 28903–28911 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |