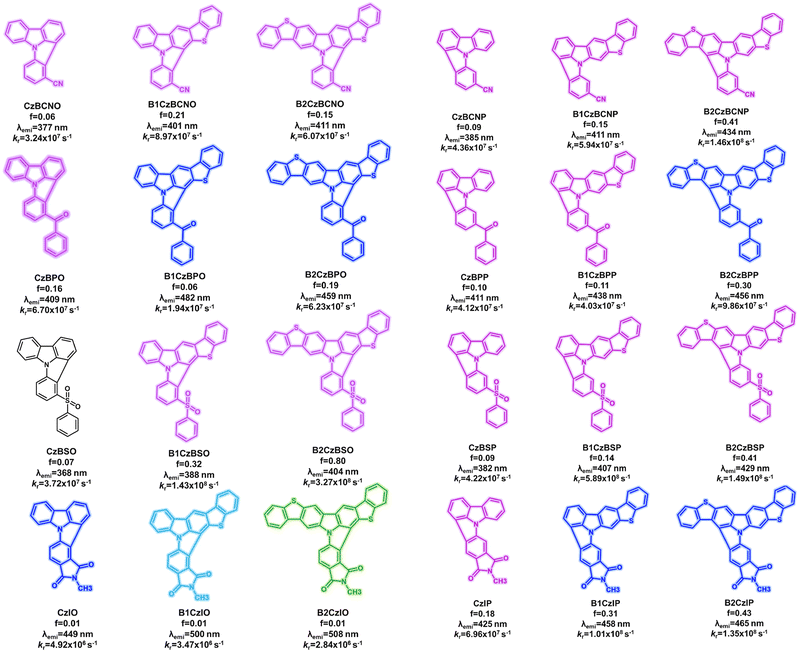
Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Rigidifying donor–acceptor frameworks via C–C interlocking to modulate excited-state dynamics in HLCT emitters
L. N. S. Laya
Pagadala
 a,
Sivakumar
Vaidyanathan
a,
Sivakumar
Vaidyanathan
 b and
Mahesh Kumar
Ravva
b and
Mahesh Kumar
Ravva
 *ac
*ac
aDepartment of Chemistry, SRM University-AP, Amaravati, 522240, India. E-mail: mahesh.r@srmap.edu.in
bDepartment of Chemistry, Indian Institute of Technology Hyderabad, Kandi, Sangareddy, 502285, India
cCentre for Computational and Integrative Sciences, SRM University-AP, Amaravati, 522240, India
First published on 14th March 2026
Abstract
Thermally activated delayed fluorescence (TADF) exploits intramolecular charge-transfer states to harvest triplet excitons in organic light-emitting diodes (OLEDs) through the twisted configuration of the donor and acceptor units. However, the highly twisted conformation of TADF molecules results in limited fluorescence yield and leads to energy loss. In this study, we designed rigid and planar fused HLCT emitters by interlocking the C–C bond between the donor and the acceptor. Carbazole and π-extended carbazole derivatives were used as donors, and cyano benzene, benzophenone, sulfonyldibenzene, and N-methyl phthalimide were employed as acceptors. Density functional theory (DFT) and time-dependent density functional theory (TD-DFT) were used to evaluate the electronic, excited-state, and photophysical properties of the newly designed molecules. The radiative and non-radiative decay rates were estimated using the thermal vibrational correlated formalism (TVCF). Furthermore, the light emitting properties of organic emitters with and without interlocked structures were systematically evaluated. These results reveal the crucial role of molecular rigidity in enhancing radiative and non-radiative properties of organic emitters. Overall, our findings establish intricate relationships between the molecular structure and key photochemical properties, including intersystem crossing rates, reverse intersystem crossing rates, radiative decay rates, and charge transfer characteristics.
1. Introduction
Thermally activated delayed fluorescence (TADF) is a mechanism by which triplet electrons can be harvested to generate fluorescence, and is used in organic light-emitting diodes (OLEDs), which enables high efficiency without the involvement of noble metals.1,2 In optoelectronic devices, these TADF emitters play a critical role in ultrapure luminescence, providing a wide colour gamut. The advantage of TADF emitters is their capability to utilize both singlet and triplet excitons, enabling 100% internal quantum efficiency (IQE) in OLEDs.3,4 Compared with traditional fluorescence (first-generation OLEDs) and phosphorescence (second-generation OLEDs), researchers have focused on enhancing the device efficiency and colour purity by establishing TADF emitters (third-generation OLEDs).5,6The main factor in the TADF mechanism is the maintenance of a smaller energy gap between the singlet and triplet excited states (ΔEST) and spin–orbit coupling (SOC), which facilitates faster reverse intersystem crossing (RISC).7,8 These factors depend on the structure of the molecule (twisted donor and acceptor), which can promote spatial separation between the highest occupied molecular orbital (HOMO) and the lower unoccupied molecular orbital (LUMO). The separation between the HOMO and LUMO wavefunctions depends on the dihedral angle between the donor and acceptor units.9 The larger dihedral angles between the donor and acceptor units effectively separated the HOMO and LUMO distributions, resulting in smaller ΔEST values. These reduced energy gaps enable efficient harvesting of triplet excitons through the RISC process.10 Three mechanisms have been proposed for conversion of triplet excitons to singlet excitons to realize a theoretical internal quantum efficiency (IQE) of 100%: triplet–triplet annihilation (TTA),11,12 TADF and hybrid local and charge transfer (HLCT) emission.13,14
To enhance the TADF properties, researchers have explored replacing planar acceptors with non-planar acceptors to increase the twist angle.15 Various molecular architectures have been developed to achieve smaller singlet–triplet energy gaps, including donor–acceptor (D–A) systems with spiro-linkages16 and through-space charge transfer (TSCT)17 configurations that optimize the spatial arrangement of the donor and acceptor units. However, the lack of interlocking between the donor and acceptor units induces structural relaxation in TADF emitters and leads to an increased Stokes shift and broadened emission spectra.18,19 Typical TADF emitters exhibit the emission spectra with a full-width at half-maximum (FWHM) of approximately 100 nm.20–23 OLED displays incorporate color filters or optical microcavities to meet the broadcasting standards of color purity by trimming the broad emission tail.24 However, this approach leads to efficiency loss, which makes it challenging to realize high efficiency OLED displays with the existing TADF emitters.25–27
It has been shown that RISC exists from high-lying triplet states (hRISC, Tn, n ≥ 2) in organic emitters.13,14 The molecules exhibiting hRISC up-conversion are referred to as HLCT molecules. In order to achieve high color purity, it is important to develop molecular design strategies that can effectively narrow the FWHM of emission in HLCT emitters with D–A structures.28 The traditional HLCT emitters contain large twist between the donor and acceptor units, which facilitates spatially separated HOMO–LUMO. During the excitation process, such separation promotes the charge-transfer (CT) character but also enables facile torsional motion between the donor and acceptor units. It is important to understand whether broad emission originates from the CT process, structural relaxations, or their combined effects.29 It has been shown that the large dihedral angles introduced in typical D–A configurations facilitate twisted intramolecular charge transfer (TICT).28
Through bond charge transfer (TBCT) and TICT are emerging as common design strategies to enhance the TADF properties. However, TBCT has gained much attention compared to TICT to regulate excited-state properties in D–A systems. In TBCT systems, donor and acceptor units are electronically coupled through a π-conjugated backbone rather than a twisted configuration. Such bonding interactions enable controlled delocalization of Frontier molecular orbitals while maintaining structural rigidity. Compared to TICT-based emitters, C–C interlocking along with TBCT systems can suppress excessive structural relaxation and reduce emission broadening while still maintaining a small ΔEST.30–32 The balance between orbital overlap and spatial separation in TBCT architectures provides an effective pathway to achieve fast RISC with improved colour purity. Therefore, incorporating through-bond interactions in rigid D–A frameworks represents a promising strategy to overcome the limitations of conventional twisted architectures.33,34
Recently, multiple resonance MR-TADF emitters have been developed with extremely narrow FWHM.35 Typically, MR-TADF emitters exhibit a slower rate of reverse intersystem crossing. Various strategies such as integration of heavy atoms into the core, π-conjugation extension, and peripheral substitution of various donor and acceptor units are used to improve the rate of reverse intersystem crossing.36 In general, these strategies are limited to MR-TADF materials and aren’t applicable to traditional D–A architecture HLCT emitters. Therefore, it is important to develop design strategies to achieve faster RISC and decrease the emission FWHM of HLCT emitters with D–A architectures. Recently, Chan et al. proposed a D–A-based deep-blue emitter which contains a 3,6-substituted carbazole as the donor and benzonitrile as the acceptor.37 These authors have shown that a reduced FWHM from 79 nm to 55 nm in obtained through functional group modification. However these emitters suffer from a low external quantum efficiency.37 To address these challenges, emitters with a lower FWHM and higher external quantum efficiency are essential.
Recently, Patil et al. proposed a new design strategy involving the interlocking of carbazole and benzene in N-phenyl carbazole (indolo[3,2,1-jk] carbazole (ICz)). The ICz motif has a more planar structure than triphenyl amine and N-phenyl carbazole.38 Compared with state-of-the-art narrow-emitting boron–nitrogen-type materials, ICz-based TADF emitters exhibit an exceptionally narrow FWHM (below 20 nm) along with high color purity and an external quantum efficiency above 30%. The planar and rigid nitrogen-centered tripod structure of ICz has extended π-electron conjugation and delocalization of non-bonding electrons over three benzene rings.39 As reported previously, this building block possesses unique characteristics, such as charge transport and emission properties due to its deeper HOMO and LUMO compared to the other indolocarbazole derivatives.38,40
Inspired by this design principle, our research work explores an unconventional approach to develop a rigid, planar D–A-type emitter through C–C interlocking at the ortho- and para positions between the donor and acceptor fragments (Scheme 1). By systematically modifying the strength of the donor and acceptor units, we investigated how structural rigidity influences intersystem crossing, and reverse intersystem crossing, radiative emission, and non-radiative emission using density functional theory methods. Furthermore, comparative analysis between rigid and non-rigid systems, an aspect not reported previously, reveals that the interlocking between the donor and acceptor fragments effectively suppresses the non-radiative decay. The non-rigid analogues show higher non-radiative decay through structural relaxation leading to poor quantum yield.
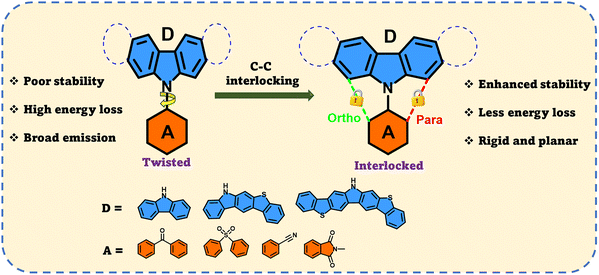 | ||
| Scheme 1 Schematic representation of the molecular design of the ortho- and para-configured donor–acceptor HLCT emitters considered in this study. | ||
2. Molecular design strategy
In this study, we developed a series of novel rigid π-conjugated and planar donor (D)–acceptor (A)-type molecules by interlinking the carbon-3 (C3) and carbon-12 (C12) positions of the carbazole (CZ) unit, referred to as the ortho (O) and para (P) configurations (Scheme 1). Our analysis revealed that the electronic properties of these newly designed ortho- and para-substituted molecules can aid in the development of OLED materials using the “rigid π-conjugated and planar” approach. Carbazole (CZ), carbazole fused with one benzothiophene group (B1Cz), and carbazole fused with two benzothiophene groups (B2Cz) were selected as the donor units because of their electron-donating ability and electron-withdrawing groups such as cyano benzene, benzophenone, diphenyl sulfide, and n-methyl phthalimide. A total of 24 π-conjugated rigid donor (D)–acceptor (A) substructure ortho- and para-substituted molecules were designed by combining the aforementioned ortho and para donor–acceptor fragments, as shown in Fig. S1.3. Computational methodology
The ground-state (S0) geometries were optimized using density functional theory (DFT) with the PBE041 functional and the 6-31G(d,p)42 basis set. The five lowest excited singlet states and the triplet states were computed using TD-DFT43 with the same functional and basis set. Natural transition orbital (NTO)44 analysis was performed to characterize the nature of the excited states. Benchmark calculations were carried out to assess the performance of different hybrid and range-separated functionals (B3LYP, PBE0, BMK, M06-2X, ωB97XD, and CAM-B3LYP). The calculated values are listed in Table S1. Among the functionals considered, the values obtained using PBE0 are in good agreement with the experimental values. The PBE0 hybrid density functional was found to be particularly effective for modelling charge-transfer excitations.45 All calculations for both the ground and excited states were performed using the Gaussian 16 software.46 In addition, the charge transfer values were determined using the Multiwfn code. The spin–orbit coupling (SOC) values were computed via TD-DFT calculations using the PBE0 functional and DEF2-TZVP as the basis set in the ORCA47 software package. The RISC rates were derived from the classical Marcus rate equation,48 and both the RISC and ISC rates were estimated using the Fortran code.4. Results and discussion
4.1. Ground-state geometries
Fig. 1 shows the chemical structures of the designed ortho- and para-interlocked HLCT emitters. In these systems, the covalent bond between the C3/C12 positions of carbazole and the ortho/para positions of the benzene ring results in a planar conformation of the N-substituted benzo carbazole unit. Such planarity can enhance π-electron delocalization between the carbazole and benzene rings. However, benzophenone and diphenyl sulphide-substituted molecules (CzBPO, B1CzBPO, B2CzBPO, CzBSO, B1CzBSO, B2CzBSO, CzBPP, B1CzBPP, B2CzBPP, CzBSP, B1CzBSP, and B2CzBSP) exhibit a slight twist in their geometry due to steric hindrance between hydrogen atoms of the phenyl rings. The calculated dihedral angles between the two benzene units for both the ortho- and para-substituted molecules are listed in Table S2. It is interesting note that in the cases of CzBPO, B1CzBPO, and B2CzBPO, the calculated twist angle values are 29°, 22°, and 22°, respectively. In the case of B1CzBPO and B2CzBPO, the interaction between the oxygen atom of the ketone group and the sulphur atom of the thiophene unit alters the twist angle between the phenyl rings. A similar phenomenon is observed for B1CzBSO and B2CzBSO. However, no change in the twist angle is observed for para-interlocked molecules, such as B1CzBPP and B2CzBPP, owing to the absence of S⋯O interactions. All ground state optimized geometries are depicted in Fig. 2.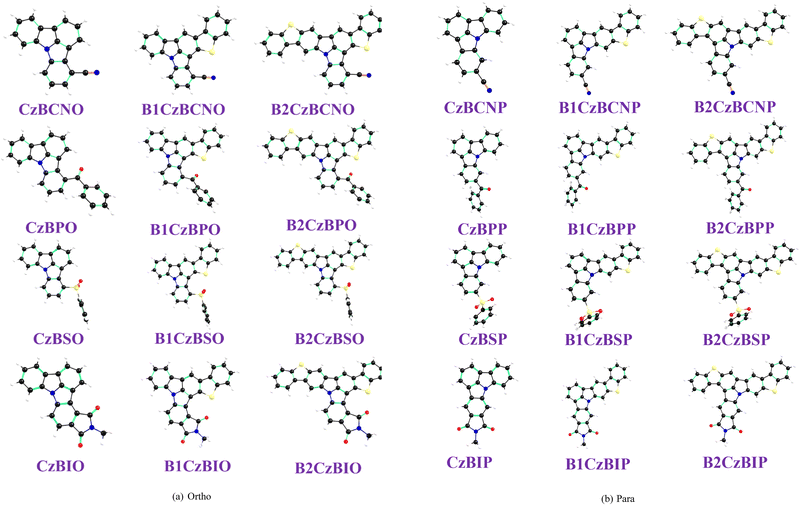 | ||
| Fig. 2 Optimized π-conjugated rigid (a) ortho- and (b) para-configured molecules obtained at the PBE0/6-31G(d,p) level of theory. | ||
4.2. Frontier molecular orbital (FMO) analysis
The electronic structures of the designed molecules were analysed using the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO). Fig. S1 shows the electron density distributions of the HOMO and the LUMO across the donor and acceptor units in the ortho- and para-substituted molecules. The HOMO was primarily localized on the electron-donating CZ, B1Cz, and B2Cz units in the CZ derivatives and extended to the adjacent benzene ring. The LUMO is mainly localized in the electron-acceptor region, with a slight extension to the benzene ring due to π-conjugation between the donor and acceptor moieties. The HOMO energy values became less negative with an increasing number of donor units from Cz to B2Cz with all different acceptors in both ortho- and para-interlocked molecules (Fig. 3(a) and (b) and Table S3). The LUMO energy values varied in the following order: N-methyl phthalimide > benzophenone > cyanobenzene > diphenyl sulfide. As described previously, the presence of S⋯O interactions alters the twist angle between the phenyl rings in the acceptor group in the B1Cz and B2Cz substituted molecules. Consequently, different trends were observed in the LUMO energies. Notably, this trend shifted in the presence of stronger donor units, such as B1Cz and B2Cz, with the LUMO stabilization sequence: N-methyl phthalimide > cyano benzene > benzophenone > sulfonyl dibenzene (Fig. 3(b)).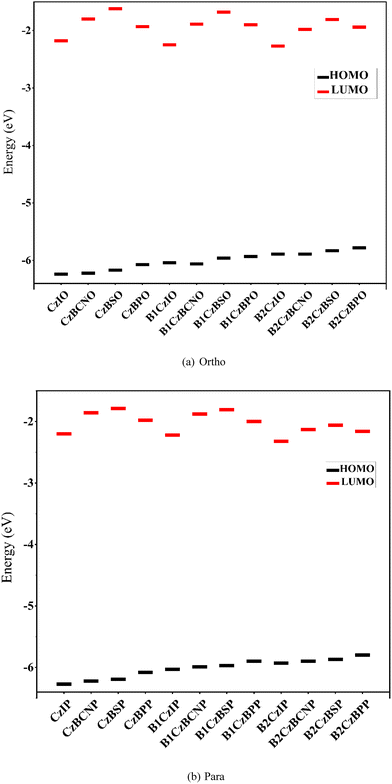 | ||
| Fig. 3 FMO analysis for the designed (a) ortho and (b) para interlocked molecules obtained at the PBE0/6-31G(d,p) level of theory. | ||
Additionally, a comparison between ortho- and para-interlocked systems revealed that para-interlocked molecules generally exhibited slightly higher LUMO energy values. From these observations, it can be concluded that N-methyl phthalimide acts as a strong electron acceptor, whereas dibenzothiophene–carbazole (B2Cz) serves as a strong donor among the studied systems. The donor–acceptor pairing follows the order of effectiveness: B2CzIO > B1CzIO > CzIO in both the ortho and para configurations. This trend is rationalized by the capacity of the strong electron-donating and electron-withdrawing groups to induce a favourable spatial overlap between the HOMO and the LUMO, facilitating intramolecular charge transfer, although it has a rigid and planar structure.
4.3. Excited state characteristics
The calculated absorption energies for CzIO and CzIP are 380 and 370 nm, respectively (Table 1). These values are in good agreement with the experimental values;49 however, systems incorporating strong electron-withdrawing groups (e.g., cyanobenzene and phthalimide) in conjunction with strong electron-donating units (e.g., bis-benzothiophene) show distinct absorption properties extending into the visible region (listed in Table 1). This effect is attributed to enhanced π-electron delocalization between the donor and acceptor segments, which promotes a more rigid and planar molecular conformation, thereby facilitating lower-energy electronic transitions.| Molecules | S1 (eV) | T1 (eV) | T2 (eV) | T3 (eV) | T4 (eV) | T5 (eV) |
|---|---|---|---|---|---|---|
| CzBCNO | 3.61 | 2.80 | 2.94 | 3.24 | 3.48 | 3.83 |
| CzBPO | 3.41 | 2.76 | 2.88 | 3.05 | 3.18 | 3.38 |
| CzBSO | 3.71 | 2.86 | 3.08 | 3.27 | 3.58 | 3.65 |
| CzIO | 3.27 | 2.66 | 2.85 | 3.20 | 3.25 | 3.48 |
| B1CzBCNO | 3.42 | 2.58 | 2.83 | 3.21 | 3.23 | 3.39 |
| B1CzBPO | 3.33 | 2.63 | 2.87 | 3.08 | 3.20 | 3.24 |
| B1CzBSO | 3.54 | 2.66 | 2.95 | 3.26 | 3.29 | 3.59 |
| B1CzIO | 3.07 | 2.50 | 2.67 | 3.03 | 3.08 | 3.23 |
| B2CzBCNO | 3.22 | 2.51 | 2.69 | 2.97 | 3.19 | 3.27 |
| B2CzBPO | 3.18 | 2.54 | 2.72 | 2.96 | 3.07 | 3.18 |
| B2CzBSO | 3.31 | 2.54 | 2.81 | 3.00 | 3.27 | 3.33 |
| B2CzIO | 2.95 | 2.46 | 2.53 | 2.92 | 2.94 | 3.11 |
| CzBCNP | 3.56 | 2.78 | 3.01 | 3.06 | 3.77 | 3.85 |
| CzBPP | 3.36 | 2.72 | 2.90 | 3.01 | 3.14 | 3.41 |
| CzBSP | 3.61 | 2.85 | 3.08 | 3.13 | 3.57 | 3.78 |
| CzIP | 3.35 | 2.68 | 2.87 | 2.98 | 3.54 | 3.62 |
| B1zBCNP | 3.40 | 2.71 | 2.88 | 2.97 | 3.24 | 3.49 |
| B1CzBPP | 3.23 | 2.69 | 2.80 | 2.89 | 3.11 | 3.22 |
| B1CzBSP | 3.45 | 2.74 | 2.96 | 3.03 | 3.29 | 3.50 |
| B1CzIP | 3.18 | 2.64 | 2.73 | 2.90 | 3.19 | 3.36 |
| B2CzBCNP | 2.90 | 2.28 | 2.36 | 2.79 | 3.03 | 3.22 |
| B2CzBPP | 3.10 | 2.44 | 2.52 | 2.84 | 3.05 | 3.35 |
| B2CzBSP | 3.14 | 2.47 | 2.63 | 2.91 | 3.15 | 3.39 |
| B2CzIP | 3.00 | 2.42 | 2.56 | 2.70 | 3.11 | 3.24 |
Among all ortho-substituted molecules, B2CzIO showed the lowest absorption energy value (2.95 eV). The strong electron-donating (benzothiophene) and electron-withdrawing (phthalimide) natures led to the lowest absorption energies compared to the other substituents. For para-interlocked molecules, B2CzBCNP exhibited the lowest absorption energy (S1) of 2.90 eV. Overall, all the designed molecules exhibited absorption energies in the range of 2.90 to 3.71 eV. The marginal difference between the absorption energies of the ortho- and para-interlocked molecules reveals that both have similar π-electron delocalization. From Cz to B2Cz, the strength of the donor increases and the value of the excitation energy decreases.
To gain deeper insights into the “π-conjugated rigid” molecules, we need to explore the characteristics of the excited states, energy level alignments, and transition properties. A natural transition orbital (NTO) analysis was performed to understand and characterize the nature of the excited state. NTOs offer a clear representation of electronic excitations, characterizing the relevant excited states as a pair of orbitals that represent the hole and electron wave functions. The NTOs of the designed molecules are shown in Fig. S2. Based on the delocalization of the hole and electron wavefunctions, the excitation can be classified into local excitation (LE), charge transfer (CT), and hybridized local and charge transfer (HLCT). LE occurs when holes and electrons overlap within the same spatial region. CT occurs when there is a significant spatial separation between the hole and electron, leading to a noticeable shift in the charge density. HLCT refers to an excited state in which both LE and CT characteristics co-exist.50,51 In addition, the nature of an excited state can be qualitatively described by hole and electron wavefunctions (as represented by NTOs). In addition, the Δr index, which represents the distance between the hole and the electron, can quantitatively characterize the nature of the excited states. The calculated Δr indices of the designed ortho- and para-substituted molecules are listed in Table S4. By examining the delocalization of the hole and electron wavefunctions (Fig. S2) and Δr index values (Table S4), one can determine whether the excitation was LE, CT, or HLCT. In the LE state, the hole and electron wave functions overlap significantly and the Δr value is small. In the CT state, holes and electrons are spatially separated in different regions of the molecule, resulting in larger Δr values. As mentioned earlier, HLCT is a hybrid state with the features of both CT and LE. Unlike the LE and CT states, the hole and electron wavefunctions in HLCT exhibit partial overlap rather than complete separation (as in CT) or strong overlap (as in LE). From the above information, we observed that both benzophenone and phthalimide acceptor units have higher Δr values (>3 Å) with Cz, B1Cz, and B2Cz donors. This clearly reveals that the centroids of both the hole and electron wavefunctions are separated in the aforementioned molecules. In contrast, sulfonyldibenzene and cyanobenzene acceptor units show lower Δr values (<3 Å) with three donor units in the ortho-substituted molecules. We found that the Δr values depend on the strength of the acceptor units. Molecules with stronger acceptor units, such as I and BP units, exhibit a large distance between the hole and electron wavefunctions. Visual inspection of the hole and electron wavefunction delocalization and moderate values of the Δr index reveal that the nature of the excited state in these molecules is HLCT, as overlap in the hole and electron wavefunctions is observed. Overall, all designed molecules showed either HLCT- or LE-dominated HLCT properties in the lowest S1 state.
4.4. Photophysical properties
Thus, a rapid reverse intersystem crossing (RISC) from the lower triplet excited states (Tm) to the lowest singlet excited states (S1) can be obtained according to the classical Marcus eqn (1)48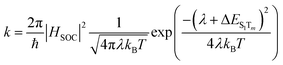 | (1) |
We observed that the calculated ΔES1Tm values for all designed molecules are less than 0.2 eV, except B1CzBSO, CzBCNP, CzIP, B1CzIP, B2CzBSP, and B2CzIP. For efficient exciton utilization efficiency (EUE), the gap between the lowest singlet state (S1) and the nearest triplet state (Tm) should be less than 0.2 eV.48,52 It is important to note that, in traditional HLCT emitters, the donor and acceptor units are separated by twist angle to spatially separate the HOMO and the LUMO. As discussed previously, such a spatial separation leads to a smaller energy gap between the singlet and triplet states. In our designed molecules, even though the donor and acceptor molecules exhibit a planar conformation, they exhibit smaller ΔES1Tm values. This enables an efficient RISC process, even for the planar confirmation of the HLCT emitters.
Another important factor that plays a crucial role in the EUE process is spin–orbit coupling (SOC). We have calculated the SOC between S1 and the lowest five triplet states (T1–T5) and the same values are depicted in Fig. 4. Among all the designed molecules, the CzBPO molecule showed a higher SOC value (3.75 cm−1). The SOC value depends on the presence of heteroatoms, such as N, O, and S. According to El-Sayed's rule, stronger SOC for transitions involves a change in orbital type. Thus, the S1 state with CT character and higher-lying triplet state (Tm) with LE character exhibit stronger SOC values. In the cases of CzBPO, CzIO, B1CzBPO, and B2CzBPP, we have observed stronger SOC values (Table 2 and Fig. S3).
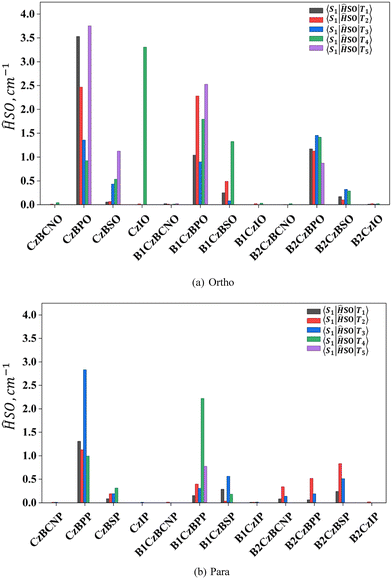 | ||
| Fig. 4 Calculated SOC between S1 and the lowest five triplet states (T1–T5) for designed (a) ortho and (b) para interlocked molecules obtained at the PBE0/DEF2-TZVP level of theory. | ||
| Molecules | ΔES1Tm (eV) | SOC (cm−1) | k ISC (s−1) | k RISC (s−1) |
|---|---|---|---|---|
| CzBCNO | 0.13 | 0.04 | 1.10 × 106 | 7.50 × 103 |
| CzBPO | 0.03 | 3.75 | 7.10 × 109 | 2.20 × 109 |
| CzBSO | 0.06 | 1.12 | 8.70 × 108 | 8.40 × 107 |
| CzIO | 0.02 | 3.31 | 4.70 × 109 | 2.20 × 109 |
| B1CzBCNO | 0.03 | 0.02 | 2.00 × 105 | 6.30 × 104 |
| B1CzBPO | 0.09 | 2.52 | 5.10 × 109 | 1.50 × 108 |
| B1CzBSO | 0.25 | 1.33 | 3.60 × 104 | 9.60 × 103 |
| B1CzIO | 0.04 | 0.00 | 0 | 0 |
| B2CZBCNO | 0.02 | 0.02 | 1.70 × 105 | 1.70 × 105 |
| B2CzBPO | 0.00 | 0.86 | 2.20 × 108 | 2.20 × 108 |
| B2CzBSO | 0.04 | 0.29 | 4.80 × 107 | 1.00 × 107 |
| B2CzIO | 0.01 | 0.02 | 1.40 × 105 | 1.00 × 105 |
| CzBCNP | 0.50 | 0.01 | 1.40 × 10−2 | 5.00 × 10−11 |
| CzBPP | 0.21 | 0.99 | 2.40 × 108 | 6.90 × 104 |
| CzBSP | 0.05 | 0.31 | 6.10 × 107 | 8.70 × 106 |
| CzIP | 0.37 | 0.01 | 6.70 × 101 | 3.70 × 10−5 |
| B1zBCNP | 0.15 | 0.00 | 0 | 0 |
| B1CzBPP | 0.01 | 0.77 | 2.10 × 108 | 1.40 × 108 |
| B1CzBSP | 0.16 | 0.18 | 5.70 × 104 | 3.60 × 104 |
| B1CzIP | 0.28 | 0.01 | 3.40 × 103 | 6.40 × 10−4 |
| B2CZBCNP | 0.11 | 0.14 | 7.20 × 106 | 2.10 × 105 |
| B2CzBPP | 0.05 | 2.09 | 2.70 × 109 | 3.90 × 108 |
| B2CzBSP | 0.23 | 0.51 | 4.00 × 107 | 5.30 × 103 |
| B2CzIP | 0.30 | 0.17 | 4.80 × 105 | 4.4 |
The calculated ISC and RISC rates for all molecules are listed in Table 2. Among all designed molecules, CzBPO, CzIO, B1CzBPO, B2CzBPO and B1CzBPP have shown higher RISC values (>107 S−1).
5. Fluorescence efficiency
5.1. Rate of radiative emission (kr)
Traditional HLCT emitters have twisted dihedral angles between the donor and acceptor units. As a result, the excited state in the lowest S1 state is generally CT in nature. The HLCT emitters, which have CT dominated character in the S1 state, exhibit a reduced rate of radiative emission (fluorescence) due to the spatial separation between the HOMO and LUMO distributions on the donor and acceptor.In the case of our newly designed molecules, as described earlier, most of the molecules exhibited LE dominated HLCT characteristics in the S1 state. Consequently, the oscillator strength of the designed molecules is higher, which leads to an increase in the radiative emission rate (fluorescence) because of the improved HOMO and LUMO overlap between the donor and acceptor. According to Einstein's spontaneous eqn (2), the radiative emission is directly proportional to the oscillator strength and vertical emission energy:
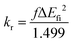 | (2) |
The combined analysis of radiative and RISC rates provides insights into potential molecules as TADF emitters. CzBPO and CzIO show much faster kRISC due to strong SOC and smaller ΔEST, but weaker kr (6.70 × 107 and 4.63 × 106 s−1) limits the emission efficiency. Molecules such as B1CzBSO, B2CzBSO, B1CzIP, B2CzBCNP, B2CzBSP and B2CzIP exhibited high kr rates. In particular, B2CzBSO combines with a higher kRISC (1.00 × 107 s−1), indicating that this molecule can efficiently up-convert triplet excitons into emissive singlets, making it a potential HLCT emitter compared to experimental molecules CzIO and CzIP.
5.2. Rate of non-radiative emission (knr)
The rate of non-radiative decay depends on two factors: the Huang–Rhys (HR) factors and the reorganization energy (λ). To more clearly define the nonradiative energy dissipation pathways of the S1 state, we performed a detailed analysis of the relationship between the HR factor and λ with normal mode frequencies. This analysis provides quantitative data on the nonradiative decay rates in solvents. The HR factor describes the change in vibrational quanta that occurs when the electronic state changes. The following equation was used to calculate the HR factor: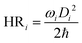 | (3) |
In the above equation, ωi is the vibrational frequency of the ith normal mode, ħ is the reduced Planck constant and Di is the displacement for mode i between the equilibrium geometry of two electronic states (S0 and S1). A greater HR factor (or stronger vibronic coupling) can lead to higher nonradiative decay rates by enabling nonradiative decay pathways through increased interactions with vibrational modes.
λ es can be calculated using the harmonic oscillator approximation as the sum of the contributions from the normal-mode analysis equation:
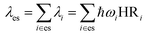 | (4) |
In order to highlight the importance of C–C bond interlocking, we considered two different configurations, viz., (i) B2CzBSO-R, which contains the C–C interlocking and represents the rigid structure, and (ii) B2CzBSO-NR, a non-interlocked analogue designed to evaluate the effect of structural flexibility. We analysed the Huang–Rhys (HR) factors and reorganization energy for the B2CzBSO-R and B2CzBSO-NR emitters as a function of the normal mode frequency to gain deeper insight into the vibrational contributions to non-radiative decay pathways (Fig. 6). The results reveal that the rigid B2CzBSO (B2CzBSO-R) molecule exhibits notably lower HR factors, particularly in the low-frequency region (<500 cm−1). Because the non-radiative decay rate (knr) is directly proportional to the HR factor, the reduced HR factor in B2CzBSO-R correlates with a suppressed knr. The reduction in knr enhances the radiative efficiency and maintains high photoluminescence quantum yields (Fig. 6(c)).
To understand the impact of structural rigidity, we considered a nonrigid analog of B2CzBSO (B2CzBSO-NR, shown in Fig. 6(b)). A comparison of the knr, HR factors, and reorganization energies will provide insights into the impact of structural rigidity. The non-rigid analogue of B2CzBSO-NR displayed a higher HR factor of 0.30 within the same low-frequency range (<500 cm−1), as shown in Fig. 6(d). This increase was attributed to the enhanced structural flexibility during the electronic excitation. These findings reinforce the importance of molecular rigidity in designing efficient HLCT emitters, as lower HR factors help reduce energy loss through nonradiative channels and support narrowband emission profiles critical for high-performance optoelectronic applications.
5.3. Distribution of S1 energy
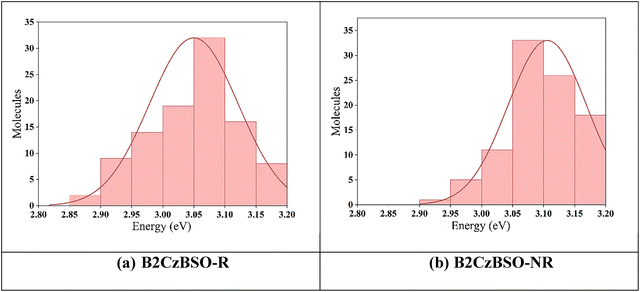
A detailed description of the atomistic molecular dynamic simulation is provided in the SI. The resulting distributions of S1 energies are depicted in Fig. 7(a) and (b) for the rigid and non-rigid systems of B2CzBSO, respectively.In the case of the non-rigid B2CzBSO-NR, the S1 energies are broadly distributed, with a mean value of 3.04 eV and a standard deviation of ±0.06 eV. Notably, approximately 32% of the sampled conformers exhibited excitation energies in the range of 2.9–3.2 eV, indicating significant conformational flexibility and energetic dispersion.
Conversely, the rigid B2CzBSO-R molecules exhibited a narrower distribution centred around 2.94 eV with a standard deviation of ±0.122 eV. The majority of conformers (nearly all) were clustered within a more confined energy window of 2.9–3.1 eV, reflecting a more homogeneous conformational landscape and reduced structural dynamics. These results highlight the influence of molecular rigidity on the photophysical behaviour of B2CzBSO, where structural constraints in the rigid derivative led to narrower S1 energy distributions, potentially favouring more consistent optical and electronic properties that are desirable for device applications.
6. Conclusion
In this study, we systematically designed and computationally analysed a series of 24 hybrid local and charge transfer (HLCT) emitters by systematically varying the strengths of the donor and acceptor units and interlocking patterns. Geometrical, electronic, excited-state properties are evaluated on these molecules to elucidate the role of backbone rigidification in modulating the excited state dynamics. The designed molecules consisted of ortho- and para-interlocked configurations constructed using three donor moieties (carbazole (Cz), mono benzo thiophene carbazole (B1Cz), and dibenzo thiophene carbazole (B2Cz)) in combination with four distinct acceptors: cyano benzene (BCN), benzophenone (BP), sulfonyl dibenzene (BS), and n-methyl phthalimide (I). We established a structure–property relationship linking interlocking geometry, excited-state character, radiative, and non-radiative decay properties.We found that the ortho-interlocked structures exhibited smaller ΔEST values owing to the increased charge-transfer character and spatial separation of the Frontier molecular orbitals. In addition to ΔEST, spin–orbit coupling (SOC) values were analysed, as they play a crucial role in promoting RISC from higher-lying triplet states. Furthermore, the interlocking strategy improved the overlap between the HOMO and the LUMO. As a result, high oscillator strength (f) values were observed for the newly designed molecules. Among all the designed molecules, the B2CzBSO emitter exhibited a favourable balance of small ΔEST, enhanced SOC, efficient RISC, and strong oscillator strength, identifying it as a promising candidate.
Comparison between rigid B2CzBSO (B2CzBSO-R) and non-rigid analogues of B2CzBSO (B2CzBSO-NR) reveals that C–C interlocking reduces the low-frequency vibrational contributions and HR factors, thereby suppressing the non-radiative decay pathways. Although most of the properties are similar in the rigid and non-rigid structures, we noted that backbone interlocking can simultaneously maintain radiative efficiency and suppress the non-radiative pathways in this structural class. Overall, this study establishes structural interlocking as an effective molecular-design strategy for achieving balanced radiative and non-radiative decay processes.
Conflicts of interest
The authors declare no competing financial interests.Data availability
The data supporting this article have been included as part of the supplementary information (SI). The supplementary information includes additional details on comparison of performance of various DFT functionals, HOMO and LUMO wavefunctions, NTOs. See DOI: https://doi.org/10.1039/d5ma01318f.Acknowledgements
L. N. S. L. P. thanks SRM University-AP for the junior research fellowship. M. K. R. also thanks SRM University-AP, Andhra Pradesh, for providing the central computational facility.References
- C. W. Tang and S. A. VanSlyke, Organic electroluminescent diodes, Appl. Phys. Lett., 1987, 51, 913–915 CrossRef CAS.
- M. A. Baldo, D. F. O’Brien, M. E. Thompson and S. R. Forrest, Excitonic singlet-triplet ratio in a semiconducting organic thin film, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 60, 14422–14428 CrossRef CAS.
- M. A. Baldo, D. F. O’Brien, Y. You, A. Shoustikov, S. Sibley, M. E. Thompson and S. R. Forrest, Highly efficient phosphorescent emission from organic electroluminescent devices, Nature, 1998, 395, 151–154 CrossRef CAS.
- D. Zhang, T. Huang and L. Duan, Emerging Self-Emissive Technologies for Flexible Displays, Adv. Mater., 2020, 32, 1902391 CrossRef CAS.
- R. H. Friend, R. W. Gymer, A. B. Holmes, J. H. Burroughes, R. N. Marks, C. Taliani, D. D. C. Bradley, D. A. D. Santos, J. L. Brédas, M. Lögdlund and W. R. Salaneck, Electroluminescence in conjugated polymers, Nature, 1999, 397, 121–128 CrossRef CAS.
- J. Huang, J.-H. Su and H. Tian, The development of anthracene derivatives for organic light-emitting diodes, J. Mater. Chem., 2012, 22, 10977 RSC.
- J. Eng and T. J. Penfold, Understanding and Designing Thermally Activated Delayed Fluorescence Emitters: Beyond the Energy Gap Approximation, Chem. Rec., 2020, 20, 831–856 CrossRef CAS PubMed.
- T. Chen, L. Zheng, J. Yuan, Z. An, R. Chen, Y. Tao, H. Li, X. Xie and W. Huang, Understanding the Control of Singlet-Triplet Splitting for Organic Exciton Manipulating: A Combined Theoretical and Experimental Approach, Sci. Rep., 2015, 5, 10923 CrossRef PubMed.
- Q. Zhang, H. Kuwabara, W. J. Potscavage, S. Huang, Y. Hatae, T. Shibata and C. Adachi, Anthraquinone-Based Intramolecular Charge-Transfer Compounds: Computational Molecular Design, Thermally Activated Delayed Fluorescence, and Highly Efficient Red Electroluminescence, J. Am. Chem. Soc., 2014, 136, 18070–18081 CrossRef CAS PubMed.
- A. Monkman, Why Do We Still Need a Stable Long Lifetime Deep Blue OLED Emitter?, ACS Appl. Mater. Interfaces, 2022, 14, 20463–20467 CrossRef CAS PubMed.
- C. Rothe and A. P. Monkman, Triplet exciton migration in a conjugated polyfluorene, Phys. Rev. B: Condens. Matter Mater. Phys., 2003, 68, 075208 CrossRef.
- C.-J. Chiang, A. Kimyonok, M. K. Etherington, G. C. Griffiths, V. Jankus, F. Turksoy and A. P. Monkman, Ultrahigh Efficiency Fluorescent Single and Bi-Layer Organic Light Emitting Diodes: The Key Role of Triplet Fusion, Adv. Funct. Mater., 2013, 23, 739–746 CrossRef CAS.
- S. Xiao, Y. Gao, R. Wang, H. Liu, W. Li, C. Zhou, S. Xue, S.-T. Zhang, B. Yang and Y. Ma, Highly efficient hybridized local and Charge-transfer (HLCT) Deep-blue electroluminescence with excellent molecular horizontal orientation, Chem. Eng. J., 2022, 440, 135911 CrossRef CAS.
- D. Cui, L. Zhang, J. Zhang, W. Li, J. Chen, Z. Guo, C. Sun, Y. Wang, W. Wang, S. Li, W. Huang, C. Zheng and R. Chen, Hybrid Local and Charge-Transfer Material with Ultralong Room Temperature Phosphorescence for Efficient Organic Afterglow Light-Emitting Diodes, Angew. Chem., Int. Ed., 2024, 63, e202411588 CrossRef CAS PubMed.
- X. Hong, D. Zhang, C. Yin, Q. Wang, Y. Zhang, T. Huang, J. Wei, X. Zeng, G. Meng, X. Wang, G. Li, D. Yang, D. Ma and L. Duan, TADF molecules with π-extended acceptors for simplified high-efficiency blue and white organic light-emitting diodes, Chem, 2022, 8, 1705–1719 CAS.
- T. Nakagawa, S.-Y. Ku, K.-T. Wong and C. Adachi, Electroluminescence based on thermally activated delayed fluorescence generated by a spirobifluorene donor–acceptor structure, Chem. Commun., 2012, 48, 9580 RSC.
- Y. Zhang, X. Liang, X. Luo, S. Song, S. Li, Y. Wang, Z. Mao, W. Xu, Y. Zheng, J. Zuo and Y. Pan, Chiral Spiro-Axis Induced Blue Thermally Activated Delayed Fluorescence Material for Efficient Circularly Polarized OLEDs with Low Efficiency Roll-Off, Angew. Chem. Int. Ed., 2021, 60, 8435–8440 CrossRef CAS PubMed.
- H. Bässler and B. Schweitzer, Site-Selective Fluorescence Spectroscopy of Conjugated Polymers and Oligomers, Acc. Chem. Res., 1999, 32, 173–182 CrossRef.
- D. J. Stewart, M. J. Dalton, S. L. Long, R. Kannan, Z. Yu, T. M. Cooper, J. E. Haley and L.-S. Tan, Steric hindrance inhibits excited-state relaxation and lowers the extent of intramolecular charge transfer in two-photon absorbing dyes, Phys. Chem. Chem. Phys., 2016, 18, 5587–5596 RSC.
- X. Liu, Z. Chen, C. Zheng, M. Chen, W. Liu, X. Zhang and C. Lee, Nearly 100% Triplet Harvesting in Conventional Fluorescent Dopant-Based Organic Light-Emitting Devices Through Energy Transfer from Exciplex, Adv. Mater., 2015, 27, 2025–2030 CrossRef CAS PubMed.
- S. Youn Lee, T. Yasuda, H. Nomura and C. Adachi, High-efficiency organic light-emitting diodes utilizing thermally activated delayed fluorescence from triazine-based donor–acceptor hybrid molecules, Appl. Phys. Lett., 2012, 101, 093306 CrossRef.
- Q. Zhang, B. Li, S. Huang, H. Nomura, H. Tanaka and C. Adachi, Efficient blue organic light-emitting diodes employing thermally activated delayed fluorescence, Nat. Photon., 2014, 8, 326–332 CrossRef CAS.
- J. Lee, K. Shizu, H. Tanaka, H. Nomura, T. Yasuda and C. Adachi, Oxadiazole- and triazole-based highly-efficient thermally activated delayed fluorescence emitters for organic light-emitting diodes, J. Mater. Chem. C, 2013, 1, 4599 RSC.
- T. Hatakeyama, K. Shiren, K. Nakajima, S. Nomura, S. Nakatsuka, K. Kinoshita, J. Ni, Y. Ono and T. Ikuta, Ultrapure Blue Thermally Activated Delayed Fluorescence Molecules: Efficient HOMO–LUMO Separation by the Multiple Resonance Effect, Adv. Mater., 2016, 28, 2777–2781 CrossRef CAS PubMed.
- M. Liu, X. Li, D. C. Chen, Z. Xie, X. Cai, G. Xie, K. Liu, J. Tang, S. Su and Y. Cao, Study of Configuration Differentia and Highly Efficient, Deep-Blue, Organic Light-Emitting Diodes Based on Novel Naphtho[1,2-d]imidazole Derivatives, Adv. Funct. Mater., 2015, 25, 5190–5198 CrossRef CAS.
- J. W. Sun, J. Y. Baek, K.-H. Kim, C.-K. Moon, J.-H. Lee, S.-K. Kwon, Y.-H. Kim and J.-J. Kim, Thermally Activated Delayed Fluorescence from Azasiline Based Intramolecular Charge-Transfer Emitter (DTPDDA) and a Highly Efficient Blue Light Emitting Diode, Chem. Mater., 2015, 27, 6675–6681 CrossRef CAS.
- J. Zhang, D. Ding, Y. Wei, F. Han, H. Xu and W. Huang, Multiphosphine-Oxide Hosts for Ultralow-Voltage-Driven True-Blue Thermally Activated Delayed Fluorescence Diodes with External Quantum Efficiency beyond 20%, Adv. Mater., 2016, 28, 479–485 CrossRef CAS PubMed.
- D.-Y. Chen, W. Liu, C.-J. Zheng, K. Wang, F. Li, S. Tao, X.-M. Ou and X.-H. Zhang, Isomeric Thermally Activated Delayed Fluorescence Emitters for Color Purity-Improved Emission in Organic Light-Emitting Devices, ACS Appl. Mater. Interfaces, 2016, 8, 16791–16798 CrossRef CAS PubMed.
- R. Ansari, W. Shao, S.-J. Yoon, J. Kim and J. Kieffer, Charge Transfer as the Key Parameter Affecting the Color Purity of Thermally Activated Delayed Fluorescence Emitters, ACS Appl. Mater. Interfaces, 2021, 13, 28529–28537 CrossRef CAS PubMed.
- A. Kumar, P. Keerthika, S. W. Han, J. Y. Lee and R. K. Konidena, One Shift, Huge Impact: Isomeric Engineering Unlocks Pure Organic Ultra-Narrowband Dual Functional Ultraviolet Emitters for High-Performance OLEDs, ACS Mater. Lett., 2026, 8, 544–550 CrossRef CAS.
- S. Kothavale, R. K. Konidena, H. Lee and J. Y. Lee, Stepwise Emission Tuning of Thermally Activated Delayed Fluorescence Emitters from the Orange to Near-Infrared Region by Rational Molecular Design, Org. Lett., 2025, 27, 12933–12938 CrossRef CAS PubMed.
- A. Kumar, S. Han, D. Kumar Sharma, J. Yeob Lee and R. Kumar Konidena, A streamlined steric-shielding approach toward efficient narrowband (FWHM ∼ 18 nm) ultra-violet emitters for OLEDs, Chem. Commun., 2025, 61, 18116–18119 RSC.
- H. Uoyama, K. Goushi, K. Shizu, H. Nomura and C. Adachi, Highly efficient organic light-emitting diodes from delayed fluorescence, Nature, 2012, 492, 234–238 CrossRef CAS PubMed.
- F. B. Dias, J. Santos, D. R. Graves, P. Data, R. S. Nobuyasu, M. A. Fox, A. S. Batsanov, T. Palmeira, M. N. Berberan-Santos, M. R. Bryce and A. P. Monkman, The Role of Local Triplet Excited States and D-A Relative Orientation in Thermally Activated Delayed Fluorescence: Photophysics and Devices, Adv. Sci., 2016, 1600080 CrossRef.
- T. Hatakeyama, K. Shiren, K. Nakajima, S. Nomura, S. Nakatsuka, K. Kinoshita, J. Ni, Y. Ono and T. Ikuta, Ultrapure Blue Thermally Activated Delayed Fluorescence Molecules: Efficient HOMO–LUMO Separation by the Multiple Resonance Effect, Adv. Mater., 2016, 28, 2777–2781 CrossRef CAS PubMed.
- X.-F. Luo, X. Xiao and Y.-X. Zheng, Recent progress in multi-resonance thermally activated delayed fluorescence emitters with an efficient reverse intersystem crossing process, Chem. Commun., 2024, 60, 1089–1099 RSC.
- C.-Y. Chan, L.-S. Cui, J. U. Kim, H. Nakanotani and C. Adachi, Rational Molecular Design for Deep-Blue Thermally Activated Delayed Fluorescence Emitters, Adv. Funct. Mater., 2018, 28, 1706023 CrossRef.
- V. V. Patil, W. P. Hong and J. Y. Lee, Indolocarbazole Derivatives for Highly Efficient Organic Light-Emitting Diodes, Adv. Energy Mater., 2025, 15, 2400258 CrossRef CAS.
- X. Wang, L. Duan and D. Zhang, Indolocarbazole-Based Multiple-Resonance Molecules: an Emerging Class of Full-Color, Narrowband Emitters for Organic Light-Emitting Diodes, Chem. – Eur. J., 2023, 29, e202300701 CrossRef CAS PubMed.
- V. V. Patil, J. Lim and J. Y. Lee, Strategic Synchronization of 7,7-Dimethyl-5,7-dihydroindeno[2,1-b]carbazole for Narrow-Band, Pure Violet Organic Light-Emitting Diodes with an Efficiency of > 5% and a CIE y Coordinate of < 0.03, ACS Appl. Mater. Interfaces, 2021, 13, 14440–14446 CrossRef CAS PubMed.
- H. Sun, C. Zhong and J.-L. Brédas, Reliable Prediction with Tuned Range-Separated Functionals of the Singlet–Triplet Gap in Organic Emitters for Thermally Activated Delayed Fluorescence, J. Chem. Theory Comput., 2015, 11, 3851–3858 CrossRef CAS PubMed.
- W. J. Hehre, R. Ditchfield and J. A. Pople, Self—Consistent Molecular Orbital Methods. XII. Further Extensions of Gaussian—Type Basis Sets for Use in Molecular Orbital Studies of Organic Molecules, J. Chem. Phys., 1972, 56, 2257–2261 CrossRef CAS.
- J. Schirmer, Review of the foundations of time-dependent density-functional theory (TDDFT), Phys. Chem. Chem. Phys., 2025, 27, 4992–5005 RSC.
- R. L. Martin, Natural transition orbitals, J. Chem. Phys., 2003, 118, 4775–4777 CrossRef CAS.
- S. Kumar, L. G. Franca, K. Stavrou, E. Crovini, D. B. Cordes, A. M. Z. Slawin, A. P. Monkman and E. Zysman-Colman, Investigation of Intramolecular Through-Space Charge-Transfer States in Donor–Acceptor Charge-Transfer Systems, J. Phys. Chem. Lett., 2021, 12, 2820–2830 CrossRef CAS PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery Jr., J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman and D. J. Fox, 2016.
- F. Neese, F. Wennmohs, U. Becker and C. Riplinger, The ORCA quantum chemistry program package, J. Chem. Phys., 2020, 152, 224108 CrossRef CAS PubMed.
- P. K. Samanta, D. Kim, V. Coropceanu and J.-L. Brédas, Up-Conversion Intersystem Crossing Rates in Organic Emitters for Thermally Activated Delayed Fluorescence: Impact of the Nature of Singlet vs. Triplet Excited States, J. Am. Chem. Soc., 2017, 139, 4042–4051 CrossRef CAS PubMed.
- S. Kuila, H. Miranda-Salinas, J. Eng, C. Li, M. R. Bryce, T. J. Penfold and A. P. Monkman, Rigid and planar π-conjugated molecules leading to long-lived intramolecular charge-transfer states exhibiting thermally activated delayed fluorescence, Nat. Commun., 2024, 15, 9611 CrossRef CAS PubMed.
- T. Liu, X. Chen, J. Zhao, W. Wei, Z. Mao, W. Wu, S. Jiao, Y. Liu, Z. Yang and Z. Chi, Hybridized local and charge-transfer excited state fluorophores enabling organic light-emitting diodes with record high efficiencies close to 20%, Chem. Sci., 2021, 12, 5171–5176 RSC.
- C. Zhou, S. Xiao, M. Wang, W. Jiang, H. Liu, S. Zhang and B. Yang, Modulation of Excited State Property Based on Benzo[a, c]phenazine Acceptor: Three Typical Excited States and Electroluminescence Performance, Front Chem., 2019, 7, 141 CrossRef CAS PubMed.
- X.-K. Chen, D. Kim and J.-L. Brédas, Thermally Activated Delayed Fluorescence (TADF) Path toward Efficient Electroluminescence in Purely Organic Materials: Molecular Level Insight, Acc. Chem. Res., 2018, 51, 2215–2224 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |