Open Access Article
Open Access ArticleIn situ synchrotron X-ray diffraction studies of the high-temperature hydrogen reduction of New Zealand titanomagnetite ironsand
Morgan
Lowther
 *a,
Bridget
Ingham
*a,
Bridget
Ingham
 b,
Martin J.
Ryan†
b,
Martin J.
Ryan†
 b,
Sigit
Prabowo
b,
Sigit
Prabowo
 a,
Mohammad
Nusheh
a,
Mohammad
Nusheh
 a,
Raymond J.
Longbottom
a,
Raymond J.
Longbottom
 c,
Bavinesh
Maisuria
c,
Bavinesh
Maisuria
 a,
Sarah
Spencer
a,
Brian J.
Monaghan
a,
Sarah
Spencer
a,
Brian J.
Monaghan
 c,
Mark H.
Reid
c,
Mark H.
Reid
 d and
Chris W.
Bumby
d and
Chris W.
Bumby
 a
a
aRobinson Research Institute, Victoria University of Wellington, P. O. Box 33436, Lower Hutt 5046, New Zealand
bCallaghan Innovation, P. O. Box 31310, Lower Hutt 5040, New Zealand
cPyrometallurgical Research Group, Faculty of Engineering and Information Sciences, University of Wollongong, Wollongong, NSW 2522, Australia
dAustralian Centre for Neutron Scattering, Australian Nuclear Science and Technology Organisation, Sydney, NSW, Australia
First published on 11th March 2026
Abstract
To characterise the hydrogen fluidised bed reduction of New Zealand titanomagnetite ironsand, this study reports the hydrogen reduction behaviour in situ via synchrotron X-ray diffraction at 938 °C. Titanomagnetite is directly compared with a synthetic magnetite. The crystalline phase evolution and changes in lattice parameter confirm that Ti significantly alters the reduction pathway at high temperature, following a 5-stage reduction process. Stabilisation of Fe(3−x)TixO4 (inverse spinel) by Ti results in two distinct reduction pathways that favour reduction of low Ti content crystallites. First, reduction of inverse spinel via wüstite results in the exsolution of an amorphous Ti rich oxide phase. This reaction halts as FeO is metallised, causing a plateau in the net lattice parameter of Fe(3−x)TixO4. A transition to the accumulation of Ti during direct reduction of the remaining inverse spinel is followed by conversion to ilmenite and pseudobrookite phases. Lower Ti content inverse spinel reduces first throughout the entire reduction process, emphasising the significant stabilising effect of Ti content.
1. Introduction
Decarbonisation of steelmaking is a key requirement for reducing anthropogenic CO2 output, accounting for 8% of global energy system emissions in 2022 and the largest source of greenhouse emissions for a single industrial process.1,2 Hydrogen direct reduction represents the primary alternative to traditional carbothermic ironmaking processes, with significant potential decrease in emissions as the renewable production of hydrogen increases.3,4Parallel to the need for low emission technology, the decreasing availability of high-grade binary iron oxide ores makes the reduction behaviour of ternary oxides crucial for long-term sustainability.5 Significant quantities of titanium-iron oxide ores are available globally, with large exemplar deposits in China, Indonesia, India, and New Zealand.6–10 The concentration and quality of such ore varies by location, with extensive deposits of very high grade mineral sands occurring on the coast of North Island Aotearoa, New Zealand (NZ).11,12 The combination of high grade ore, abundant local renewable energy sources, and the global need to decarbonise steelmaking, make the hydrogen reduction of NZ ironsands of particular interest.13
These ores consist primarily of two solid solution series. First, the titanomagnetite inverse spinel Fe(3−x)TixO4 solid solution, ranging from magnetite (x = 0) to ulvospinel (x = 1). Second, the Fe(2−x)TixO3 series, between hematite (x = 0) and ilmenite (x = 1). Alongside acting as a significant source of metallic iron, reduction of titanomagnetite is a necessary first step in the co-production of high value companion metals such as Ti and V.14 For Fe production, a high Fe content titanomagnetite is preferable, with ilmenite primarily exploited for Ti production.
Unlike the sequential reduction of iron oxides as described by the Bauer–Glaessner diagram, the hydrogen reduction of titanomagnetite ores follows a complex multi-phase pathway that is highly temperature and composition dependent. Previous studies have established the importance of Ti content in determining this behaviour. High Ti ores (Ti/(Ti + Fe) > 20%) progress directly from inverse spinel to ilmenite formation across a wide temperature range.15 At lower Ti content, however, varying reduction pathways have been observed in the 700–1000 °C range.16 Understanding the order in which phases form and the distribution of Ti is crucial for optimisation of the reduction process. This is particularly the case when reducing particulate ores in a fluidised bed, in which the migration of Ti plays a major role in regulating sticking.17
Alongside Ti and Fe, titanomagnetite ores often contain other contaminant elements such as Al, Mg, and lower concentration substituents such as V and Mn. Both Mg and Al are able to substitute for Fe throughout the matrices of magnetite and hematite, forming equivalent solid solutions to the Fe(2−x)TixO3 and Fe(3−x)TixO4 phases.18–21 Microstructural studies of ironsand reduction indicate that Al is strongly co-located with Ti throughout the reduction process, whilst Mg is more evenly dispersed within Fe bearing phases.16,22,23
Temperature dependent miscibility gaps and phase instabilities within the Fe–Ti–O ternary further complicates predicting the reduction pathway at elevated temperature. The ferropseudobrookite solid solution (FexTi(3−x)O5, 1 ≤ x ≤ 2) is stabilised at high temperature, but has unstable regions in the temperature range of 600–1150 °C.24 Further, miscibility gaps in the inverse spinel Fe(3−x)TixO4 and hematite-ilmenite Fe(2−x)TixO3 series occur below 550 °C and 800 °C respectively.25,26
Quantitative in situ monitoring of crystallographic phases is required to understand reaction pathways. Here we report in situ powder diffraction measurements of the high temperature hydrogen reduction of both mineral titanomagnetite and synthetic magnetite using high-resolution synchrotron X-rays. This technique allows us to not only track the major phases which evolve during the reaction, but also offers additional insight into materials parameters such as crystallite size, strain, and other structural variations. Coupled with sampling of partially reduced material from a fluidised bed reactor under equivalent conditions, microstructural changes can be correlated with the in-depth in situ crystallographic data.
Results show that the presence of Ti within the magnetite lattice has a significant effect on the reduction pathway, increasing the number of intermediate phases to achieve complete metallisation. As the titanium content of inverse spinel increases, production of wüstite becomes unfavourable. This results in a switch in the reduction pathway to direct metallisation of the inverse spinel, and the exsolution of Ti.
2. Experimental method
2.1. Materials
Titanomagnetite ironsand was sourced as an unmilled mineral concentrate (Waikato North Head, New Zealand), with a synthetic magnetite (Höganäs) used as a comparator. These samples are referred to as ‘TTM’ and ‘MAG’ respectively. Elemental analysis was performed by X-ray fluorescence (XRF) (Shimadzu EDX-7200), with results given in Table 1.| Mass % | Cation ratio | ||||
|---|---|---|---|---|---|
| TTM | MAG | TTM | MAG | ||
| Total iron (FeT) | 60.5 | 69 | Fe | 80 | 98.6 |
| TiO2 | 8.1 | 0.3 | Ti | 7.5 | 0.3 |
| Al2O3 | 3.5 | 0.2 | Al | 5.1 | 0.3 |
| MgO | 2.6 | 0.2 | Mg | 4.8 | 0.4 |
| V2O5 | 0.6 | 0.2 | V | 0.5 | 0.2 |
| MnO | 0.6 | 0.05 | Mn | 0.6 | 0.1 |
| CaO | 0.2 | 0.03 | Ca | 0.3 | 0.04 |
| P2O5 | 0.1 | 0.02 | P | 0.1 | 0.02 |
| SiO2 | 1 | 0.1 | Si | 1.2 | 0.1 |
2.2. Fluidised bed experiments
The hydrogen reduction behaviour of TTM was investigated using isothermal fluidised bed experiments. A simplified schematic of the fluidised bed reactor and sampling system is shown in Fig. 1. Further details of the experimental apparatus can be found in previously published literature.16,17,27Unmilled TTM was sieved to a 90–150 µm size fraction. A bed mass of 100 g was used for each experiment. The reactor was purged with nitrogen gas at an input flow rate of 50 cm3 s−1 (3 standard litres per minute) during heating to a bed temperature of 938 °C. Gas flow was then switched to hydrogen at an input flow rate of 83 cm3 s−1 (5 standard litres per min) for the duration of the experiment. Samples of approximately 2 g were drawn into a water quenched sample pot at predetermined timepoints during the reduction. In order to acquire samples at timepoints of 1 min and 3 min, the entire bed was quenched with nitrogen.
Additional samples were mounted and polished to cross-section particles for scanning electron microscopy. Backscattered electron (BSE) images were taken (Hitachi SU7000) of representative particles. Electron dispersive X-ray spectroscopy (EDS) mapping of selected regions was performed (Oxford EDS Ultimax 100, AZtec Version 6.1) to assess the distribution of key elements. Maps are presented as apparent concentration.
2.3. Synchrotron experiments
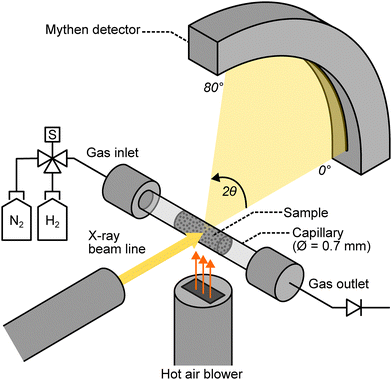
Samples of MAG and TTM were ground to a particle size of 5–10 µm for synchrotron measurements. The ground samples were packed in 0.7 mm diameter quartz capillaries (Charles Supper) and mounted in a gas flow cell on the Powder Diffraction beamline at the Australian Synchrotron, shown in schematic form in Fig. 2. A Cyberstar hot air blower was mounted beneath the capillary and heated to its maximum setpoint temperature of 1000 °C, with a target experimental temperature of approximately 950 °C. The actual temperature at the sample position was measured as 938 °C using a thermocouple and calibrated using the lattice parameter of an in situ platinum wire. The hot zone was approximately 2 mm wide; the X-ray beam size at the sample was 1 mm and centred in the middle of the hot zone. The X-ray wavelength was calibrated using a LaB6 standard (NIST 660b), and established as 0.5889 Å (21.00 keV). Data were collected in a continuous fashion using a Mythen detector spanning 80° of arc in 2θ. To cover gaps between the detector modules, the detector was rotated back and forth by 0.5° in 2θ from one measurement to the next. The exposure time for each measurement was 30 s. Data were normalised to the incident beam intensity, merged in time and averaged to a step size of 0.005° in 2θ according to the method described in Materić et al.,28 which results in no loss of time resolution.Experiments were performed under isothermal conditions, with an input gas flow rate of 2.08 cm3 s−1 (125 standard cubic centimetres per min), equivalent to approximately 8.3 cm3 s−1 at 938 °C. The hot air blower was heated to temperature and moved into place beneath the sample with nitrogen gas flowing through the capillary. Continuous diffraction measurements were begun, and after 10 measurements the gas was remotely switched to hydrogen. The experiment was stopped after 90 minutes or once the sample had fully reacted and reached a steady state, whichever came first. At the conclusion of each experiment, the gas flow was switched back to nitrogen, measurements were stopped, the hot air blower was moved out of position, the inlet and outlet gas valves were closed, and the capillary was removed from the flow cell.
The estimated reduction degree (RD) was calculated on a per-cation site basis, calculating the molar ratio of phases present assuming ideal stoichiometry and the resulting weighted average oxidation state of iron. Note that this neglects any compositional changes within phases, and is provided as an indicator of reaction progress and not as a precise instantaneous measure of the degree of reduction.
3. Results
3.1. Fluidised bed reduction behaviour
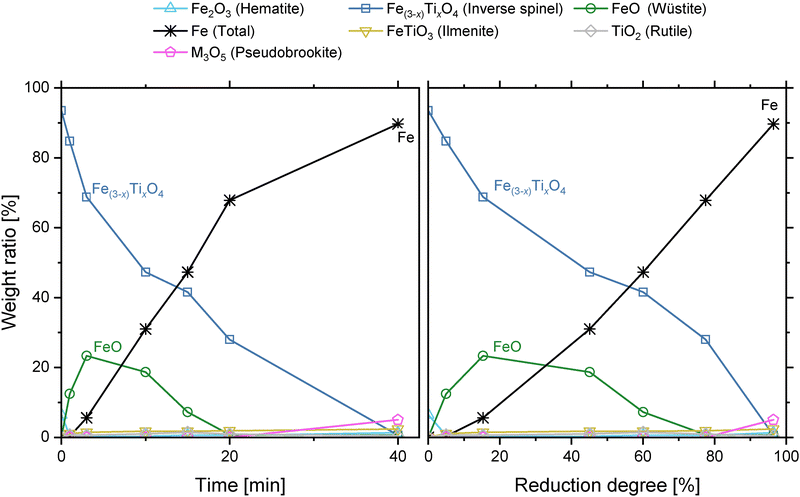
Both inverse spinel and wüstite reduce during the remaining reaction, with formation of pseudobrookite observed in the final timepoint (tf = 40 min). A metallisation degree of ∼90% and calculated reduction degree of 96% is achieved.
Wüstite is shown to have a lower solubility for Ti than the surrounding inverse spinel matrix with similar behaviour observed for Al (Fig. 5a). As wüstite reduces, the formation of voids and metallised Fe follows the microstructure of prior wüstite channels.
The outer shell consists of a 0.5 µm thick outermost oxide layer enriched in Mg, and an Fe denuded oxide region enriched in Ti and Al (Fig. 5b). A visible correlation of Ti, Al, and O distribution is present within the particle as reduction progresses (Supplementary C). An increase in the volume fraction of Fe within regions where wüstite has already been reduced indicates continued reduction of the remaining inverse spinel. This behaviour correlates with the observed crystalline phase behaviour in synchrotron and XRD analyses. No other segregated Ti enriched phase can be directly observed in these images, excluding the Fe-denuded outer shell.
3.2. Synchrotron reduction behaviour
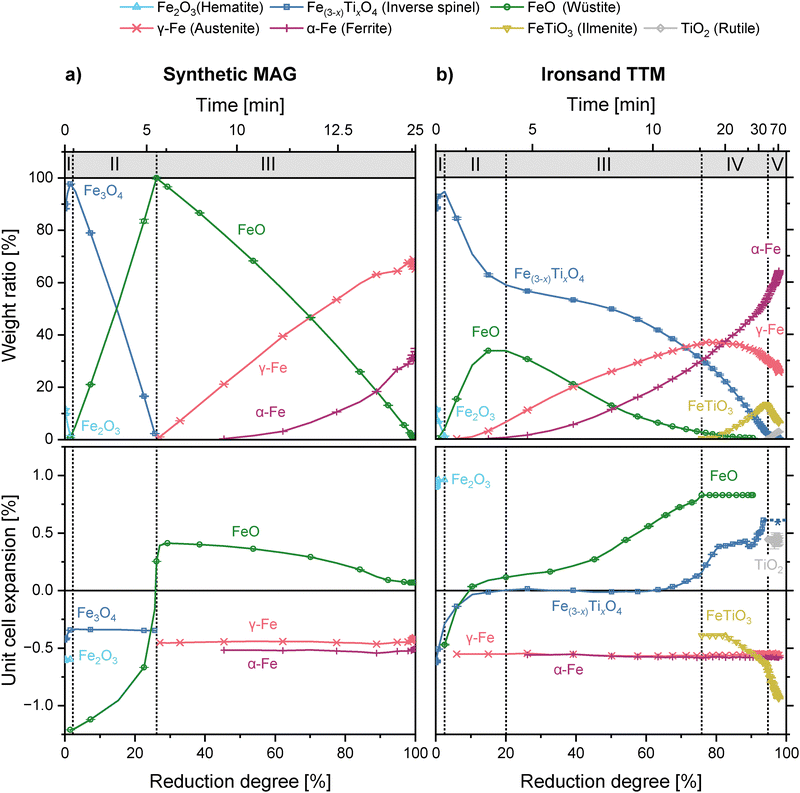
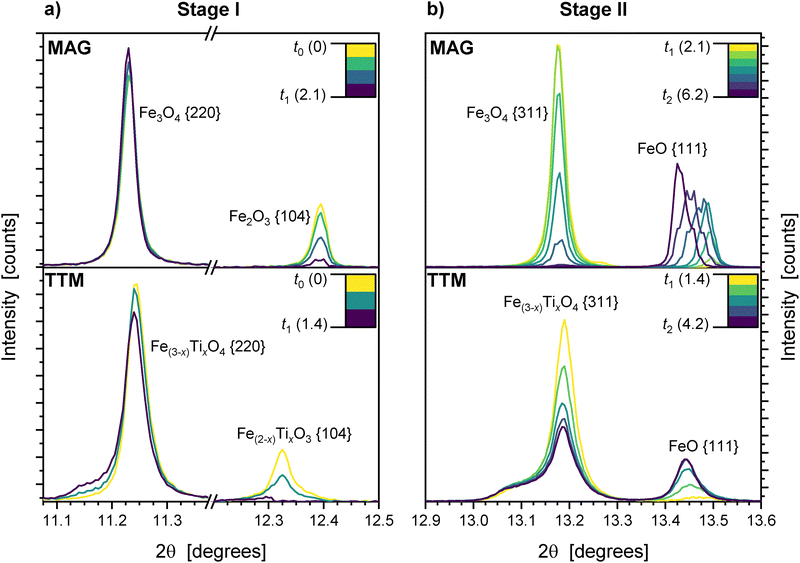
The rapid onset of hematite reduction to inverse spinel indicates the start of reaction of hydrogen gas with the sample at the measurement position (t0 = 0). The MAG sample follows a well-defined stepwise reduction with each reaction reaching the endpoint concurrent with the start of the next reaction:
| Stage I: 3Fe2O3 + H2 → 2Fe3O4 + H2O | (1) |
| Stage II: Fe3O4 + H2 → 3FeO + H2O | (2) |
| Stage III: FeO + H2 → Fe + H2O | (3) |
This three-stage hydrogen reduction pathway is consistent with existing literature.29,30 The small 5–10 µm particle size will encourage completion of sequential reactions compared to coarser material.
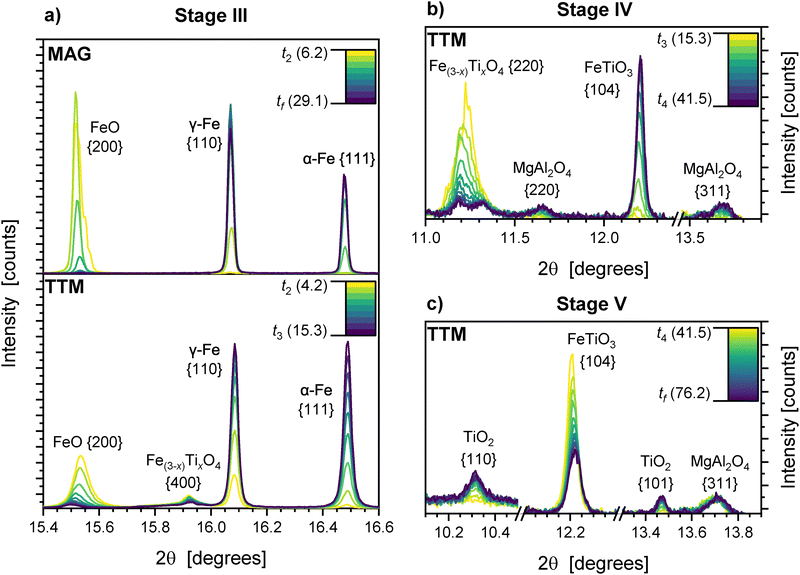
The reduction process observed in TTM is significantly more complex than MAG, with five distinct reduction stages. The approximate boundaries of these stages are demarcated in Fig. 6b. Theoretical reduction equations are as follows, with the x representing varying degrees of Ti enrichment and Δ representing arbitrary hydrogen quantities for a given reaction:
| Stage I: 3Fe(2-x)TixO3 + H2 → 2Fe(3-1.5x)Ti1.5xO4 + H2O | (4) |
| Stage II: (x + Δ)·Fe(3−x)TixO4 + Δ·H2 → 3Δ·FeO + x·Fe(3−x−Δ)Ti(x+Δ)O4 + Δ·H2O | (5) |
| Stage III: FeO + H2 → Fe + H2O | (6) |
| Stage IV: Fe(3−x)TixO4 + (4 − 3x)·H2 → xFeTiO3 + (3 − 2x)·Fe + (4 − 3x)·H2O | (7) |
| Stage V: FeTiO3 + H2 → Fe + TiO2 + H2O | (8) |
The formation of ilmenite in Stage IV (eqn (7)) has been simplified to assume strict stoichiometry of the product phase. Unlike in MAG, these reactions are not strictly confined to the boundaries of each stage as shown, but are instead the predominant or characteristic reaction during that stage. For example, reduction of wüstite to metallic iron begins prior to the demarcated start of Stage III.
Comparing behaviour to that observed in fluidised bed samples, a good correlation is shown by these data and comparison of synchrotron and ex situ XRD patterns (Supplementary E). Synchrotron data is representative of the fluidised bed reduction behaviour.
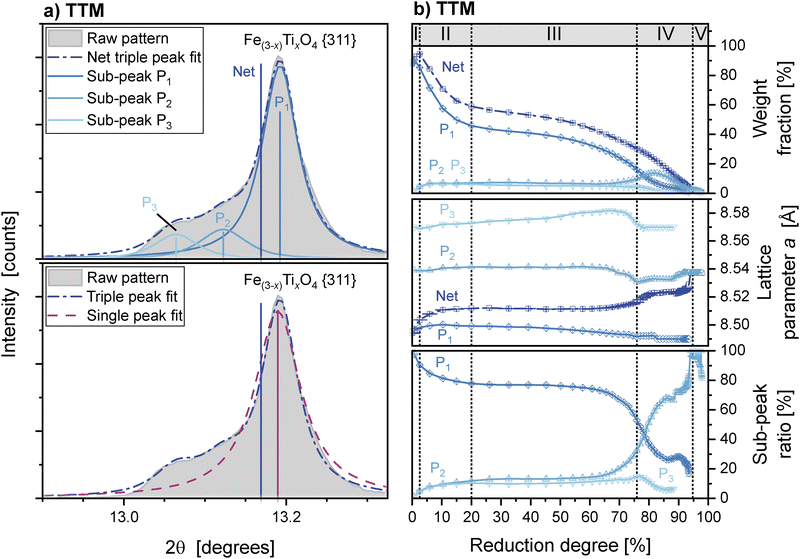
The diffraction peaks of the inverse spinel phase in TTM show significant asymmetry once reduction begins. Asymmetric peaks were fitted using the sum of three separate inverse spinel structural models (Fig. 7a), each having different lattice parameters but common peak shape parameters (i.e. crystallite size and strain parameters). These peaks represent three populations of inverse spinel crystallites in the sample: (P1) raw TTM, (P2) an intermediate Ti enriched population, and (P3) Ti enriched crystallites at the highest Ti content.
The inclusion of three such sub-peaks was sufficient to fit the inverse spinel peaks. The inclusion of additional phases did not statistically improve the fit, whilst using only two phases yielded poor agreement with the experimental data. The lattice parameters fitted for each structural model were stable throughout the reduction (Fig. 7b), and the peak shape parameters were fixed.
Fig. 7b shows that during the reduction, the three sub-peak populations show fundamentally different behaviours. The initially dominant phase with the lowest Ti content, sub-peak P1, is the only population to decrease in intensity during Stages II and III. This decrease continues until its intensity decreases to a similar magnitude as the other inverse spinel phases.
The detailed progression of each reduction stage is presented in further detail in the following sections.
In MAG, the direct reduction of hematite to magnetite occurs with no significant changes in peak shape or position (Fig. 8a).
For TTM, an increase in the titanohematite lattice parameter is observed during Stage I (t0 to t1). The inverse spinel unit cell simultaneously expands by 0.3% (Fig. 6b). This correlates with an increased Ti content in the phase as titanohematite reduces, producing the P2 and P3 crystallite populations (see Fig. 7b). Significant asymmetry of the diffraction peaks for inverse spinel in TTM occurs as a result (Fig. 8a). An increase in the low-angle side of the diffraction peaks (i.e., larger unit cell) is particularly prominent. In addition, the fitted strain parameter of the inverse spinel phase shows a rapid increase by 40% during the hematite reduction.46
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) O ratio is shown by the increasing lattice parameter as reduction progresses (Fig. 8b). Wüstite is sub-stoichiometric with composition dependent on local oxygen fugacity, with increasing lattice parameter associated with an Fe
O ratio is shown by the increasing lattice parameter as reduction progresses (Fig. 8b). Wüstite is sub-stoichiometric with composition dependent on local oxygen fugacity, with increasing lattice parameter associated with an Fe![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) O ratio increasing towards 1.47,48 This indicates that the first wüstite formed has a high oxygen content, with the average iron content increasing as the samples reduce.
O ratio increasing towards 1.47,48 This indicates that the first wüstite formed has a high oxygen content, with the average iron content increasing as the samples reduce.
Production of wüstite is the only reaction pathway in MAG during stage II (eqn (2)), reaching completion 4 minutes after the start of Stage II (Fig. 6a), with no Fe3O4 present after this point (t2 = 6.2 min). The apparent rate of reduction slows at the end of Stage II, correlating with the increased stability and widening of the wüstite phase field at elevated temperature allowing stoichiometric deviation not captured in the estimated reduction degree.49 There is no change in inverse spinel unit cell volume throughout this stage.
Total conversion to wüstite does not occur in TTM, with 59 wt% inverse spinel remaining at the peak wüstite concentration of 34 wt% (t2 = 4.2 min). Simultaneous reduction of wüstite is indicated by the formation of face-centred cubic austenite (γ-Fe), which increases in rate as the stage progresses and the reduction of Fe(3−x)TixO4 slows.
Increasing net concentration of Ti in the inverse spinel lattice is shown by a 0.3% expansion in the net unit cell volume (Fig. 6b). This is caused by reduction of low-Ti content crystallites, as represented by sub-peak P1 (Fig. 7b), and is visible as a decrease in the high-angle sub-peak of the inverse spinel diffraction peaks (Fig. 8b). No reduction of P2 or P3 occurs during this stage.
Wüstite in MAG is directly reduced to iron during Stage III, forming austenite and ferrite. This reaches completion after 25 minutes. No additional phases are formed during this process.
Metallic iron product phases had stable lattice parameters and peak shapes throughout.
The majority of wüstite present in TTM is reduced to iron during Stage III. The wüstite unit cell size continues to increase, reaching a stable maximum of 4.365 Å. The reduction of inverse spinel also continues during Stage III at a decreased rate, with the continued reduction of the P1 sub-population. No accumulation of Ti occurs, shown by a plateau in unit cell size and the absence of P2 or P3 crystallite generation (Fig. 7b). This behaviour is the continued production of wüstite as channels coarsen at the core of the reducing particles.
At a reduction degree of approximately 60%, a change in the reduction mode of inverse spinel is observed. The reduction of P1 crystallites increases, with the generation of additional P2 crystallites. This indicates the direct reduction of inverse spinel, with the accumulation of Ti increasing the lattice parameter of the remaining inverse spinel. No new intermediate or Ti bearing phases are observed during this stage.
This correlates well with the intermediate regime established in fluidised bed reduction by Prabowo et al. between 850 and 950 °C,16 above which the reduction of Fe(3−x)TixO4 halts, and below which no intermediate wüstite is formed.
The unit cell volume of the ilmenite phase reduces by 0.3% throughout Stage IV, indicating a change in stoichiometry as more ilmenite is formed. Ilmenite peaks are symmetric throughout this stage. The crystallisation of a magnesium aluminate spinel phase, labelled as MgAl2O4, also occurs during Stage IV (Fig. 9b).
The rutile content increases as Stage V progresses towards completion, with 2.9 wt% present at the final reduction degree of 91% after 75 minutes. No significant changes in lattice parameters for TiO2 are observed. The lattice cell volume of ilmenite continues to decrease as observed in Stage IV, indicating further changes in composition, with the resulting diffraction peaks shifting to higher angle.
4. Discussion
Combining measurement in situ with faster data sampling and increased angular resolution, the synchrotron diffraction results reveal significant new details of Ti migration during the reduction of TTM. Previous studies have lacked the time-resolution to observe the reduction of Ti enriched Fe(2−x)TixO3 during Stage I.16,17,27 In this work, a significant change in the lattice parameter of inverse spinel is caused by this reduction (a). This is directly correlated with an increased and non-uniform Ti content in the inverse spinel matrix that forms the bulk of TTM particles. This contrasts with the MAG reference sample, where the reduction of hematite has no significant effect on the inverse spinel lattice parameter. No significant changes in the inverse spinel lattice parameter will be caused by oxygen content at this temperature, as the phase field remains narrow and approximately stoichiometric.The higher solubility of Ti in the hematite-ilmenite solid solution compared to the magnetite-ulvospinel solution under equal oxygen partial pressures results in a higher total Ti content.50 Titanohematite is not uniformly distributed in TTM, with approx. 8% of particles showing a lamellar microstructure consisting of inverse spinel and titanium enriched titanohematite, visible in the BSE micrographs shown in Supplementary C.17,51 The Stage I reduction of hematite therefore produces a locally enriched inverse spinel, represented by sub-peak P3 (Fig. 7). Resulting in a larger unit cell and a multimodal distribution of lattice parameter within the sample. A schema presenting the changes in sub-peaks and the attributed causes is presented below (Fig. 10).
 | ||
| Fig. 10 Schema summarising the changes in the inverse spinel crystallite sub-populations during each reaction stage, and the causes of each change. | ||
From the changing ratio of sub-peaks during reduction, we posit that a Ti rich oxide is exsolved from the inverse spinel phase as it reduces. During Stage II, the net lattice parameter of the inverse spinel increases. This is caused predominantly by reduction of low Ti crystallites, represented by sub-peak P1 (Fig. 7b and 8b). There is no concurrent increase in sub-peaks P2 or P3. The Ti contained in the reducing inverse spinel is therefore not retained in the remaining inverse spinel as posited in previous works.16,17
The crystalline product phase in Stage II is wüstite. Ti exhibits poor solubility in wüstite compared to the inverse spinel phase (Fig. 5a). Experimental reports indicate a solubility limit around 5at% at the liquidus of 1314 °C, with more recent thermodynamic analyses suggesting a maximum of <1 at% at 938 °C.52–54 Tetravalent titanium must therefore be partitioned away from wüstite as reduction proceeds.
Given that no other crystalline phases are produced, this is direct evidence for the formation of an amorphous Ti rich oxide (a-TiOn) phase starting in Stage II. This correlates with the Ti-rich oxide shell formation observed in fluidised bed samples, that precipitates during Stage II and thickens throughout reduction (Fig. 5 and Supplementary C). The thickening of the shell correlates with wüstite formation, with the coarsening of wüstite channels at the core of the particle and the thickening of the shell terminating mid Stage III (Supplementary C). This behaviour disagrees with the theoretical reaction described in eqn (5), and can instead be better approximated as below:
| Fe(3−x)TixO4 + (4 − 2x)·H2 → (3 − x)·Fe + xTiO2 + (4 − 2x)·H2O | (9) |
This hypothesis is validated by considering the required diffusion of Ti in the inverse spinel phase to form the shell. Based on the 5 µm thick reacted layer of wüstite channels at t = 60 s, the estimated diffusion constant in inverse spinel is DTi ≈ 10−13 m2 s−1. This lies well within the predicted diffusion rate of Fe in spinel structures at 950 °C and estimated oxygen partial pressure of pO2 = 10−16 (pH2O = 0.3 atm).55 Accurate data for the diffusion rate of Ti in inverse spinel is, however, limited to temperatures above 1100 °C and oxygen fugacity above 10−11.56 This comparison neglects accelerated diffusion of grain boundaries and high vacancy content during reduction, which would further encourage the likely shell-exsolution mechanism.
A significant transition in reduction mechanism occurs in Stage III. As the available wüstite approaches zero, the reduction of remaining P1 crystallites accelerates. An increase in P2 crystallites indicates a transition in reaction pathway, with Ti being accumulated rather than primarily exsolved. This represents the direct reduction of inverse spinel to metallic iron, described by eqn (10):
| (x + Δ)·Fe(3−x)TixO4 + 4Δ·H2 → 3Δ·Fe + x·Fe(3−x−Δ)Ti(x+Δ)O4 + 4Δ·H2O | (10) |
A temperature dependent transition in the reduction of magnetite directly to Fe is well established, occurring below 570 °C.57 The stabilising effect of Ti plays a similar role, lowering the favourability of the wüstite reduction route. The continued reduction of the inverse spinel during Stage III is visible as increasing metallisation within regions where wüstite reduction is already complete (Fig. 5b and Supplementary C).
The final key transition in behaviour is the direct conversion of inverse spinel to ilmenite in Stage IV. Each of these reduction pathways – described here as exsolution, accumulation, and conversion – demonstrate the preferential reduction of low-Ti content inverse spinel Fe(3−x)TixO4, i.e., the stabilisation of the phase by Ti. It is exclusively the low Ti P1 crystallites that are reduced throughout Stages II–III (Fig. 10), with the P2 crystallites dominant at the end of reduction.
Whilst there is an inhomogeneous distribution of inverse spinel across these three populations, considered as a collective body, the crystalline system follows the anticipated phase diagram well. An approximate ternary phase diagram for the Fe–Fe2O3–TiO2 system at 950 °C is presented in Fig. 11a.49,50,58–60 Of note is the instability of pseudobrookite (FeTi2O5), with the limit of stability for the M3O5 solid solution lying at approximately Fe1.3Ti1.7O5.24
 | ||
| Fig. 11 (a) Schematic molar fraction ternary Fe–Fe2O3–TiO2 phase diagram for 950 °C, with the inclusion of established ferro-pseudobrookite stability limit at Fe1.3Ti1.7O5.24,49,50,58–60 The theoretical reduction pathway for titanomagnetite with a Ti/(Ti + Fe) ratio of 8.6% is shown. (b) Summary of phase assemblages A–J for the ternary diagram, where M represents metal cation; (ss) = solid solution. (c) Conceptual ternary phase diagram overlaid with the phase assemblages C*–F* for lower Ti content invariant points in inverse spinel. (d) Phase fields H*–J* if pseudobrookite is stabilised. | ||
Cross-referencing the reduction stages described for TTM with the equilibrium phase assemblages A–J results in the following:
(1) Stage I: conversion of titanohematite to a fully inverse spinel sample (A).
(2) Stage II: net Ti content in inverse spinel increases as wüstite is formed (B).
(3) Stage III: transition to the reduction of wüstite to metallic Fe, with no change in the Ti content of inverse spinel during this period (C). At the end of Stage III, see the direct reduction of inverse spinel, resulting in an increasing lattice parameter (E).
(4) Stage IV: remaining inverse spinel is converted to ilmenite and metallic Fe (F and G).
(5) Stage V: ilmenite is converted to rutile and metallic Fe (J).
Minor deviations can be considered in terms of the position of specific vertices. Given that the net Ti content of inverse spinel never reaches that of P3, it is clear that the ulvospinel point is never reached. Combined with the transition to Stage III, a conceptual modification is shown in Fig. 11c. Instability of the ulvospinel has previously been reported, no longer co-occurring with metallic Fe below 680 °C.50 It is important to note that this neglects the exsolved Ti rich amorphous oxide, which is not observed in previous reports of the Fe–Ti–O ternary at 950 °C and is incompatible with the thermodynamic predictions of the equilibrium ternary.58,61
This ternary phase diagram is also broadly consistent with the reported behaviour of high Ti titanomagnetite ores. At a Ti/(Ti + Fe) ratio of 22%, material sourced from Chaoyang is reported to partially metallise before directly reducing to ilmenite (transiting phase assemblages A–E–F–G), with no wüstite formation reported.15 This contrasting behaviour emphasises the significance of the Ti concentration in the TTM source material in determining the reduction pathway for titanomagnetites.
The formation of pseudobrookite or rutile during Stage V is the only significant difference between fluidised bed and synchrotron reduction experiments. The Ti rich end-member of Fe(2−x)Ti(1+x)O5 is typically unstable below 1150 °C, and less stable than rutile at high oxygen fugacity.24,50 Magnesium cations are the likely cause of pseudobrookite formation as a stabiliser of M3O5 phases.62 The observed pseudobrookite has a high Mg content, shown by a contracted c axis and elongated b axis (a = 9.742 Å, b = 10.09 Å, c = 3.738 Å).42,63 As shown schematically in Fig. 11d, stabilisation of a pseudobrookite phase will encourage its formation at high reduction degrees. Considering the migration of Mg to the shell (Fig. 5b and Supplementary C), the smaller particle size and difference in gas flow behaviour between experiments may play a role in this variation.
Magnesium cations also play a role in the formation of a spinel phase during Stage IV. A shift in lattice parameter compared to the titanomagnetite inverse spinel (8.18 Å vs. 8.54 Å) correlates well with a primarily magnesium aluminate spinel, with low Ti or Fe content.64,65 This phase however is present in low concentrations and only apparent during Stage IV–V. The remaining Al and Mg cations must instead be substituting for Fe or Ti in the inverse spinel or ilmenite lattices or exsolving as amorphous oxides during Stages I–III.
This same cationic substitution prevents direct extrapolation of Ti content from lattice parameters, and may explain the anomalously low inverse spinel unit cell size in TTM compared to MAG. Al and Mg account for 5.1 and 4.8 at% of cations respectively (Table 1). Microstructural analyses indicate co-location of Ti and Al during reduction (Section 3.1.2. and Supplementary C), both of which preferentially occupy the octahedral lattice site in the inverse spinel phase.66–68 Titanium in particular is well-established to occupy the octahedral vacancy site, even at high Ti:Fe ratios approaching the ulvospinel composition.69 The combination of their impact on lattice expansion is, however, unlikely to be linear given the significance of cation ordering in determining lattice parameters.38,70 Local oxygen fugacity, interstitial hydrogen and the established discontinuity in thermal expansion above the Curie temperature will further confound lattice expansion.38,39,71 This provides a likely explanation for the lower initial lattice parameter observed in TTM than MAG at temperature, despite the anticipated lattice expansion caused by Ti.
Similar confounding effects may be anticipated in ilmenite. Oxygen vacancies, interstitial hydrogen, decreasing Ti or increasing Al content would all contribute to an observed decrease in lattice parameter.20,31,72 This further reinforces the challenges of accurately measuring the movement of a single solute species in mineral systems, where the migration of other solutes present alters lattice parameters. Further work with high purity synthetic titanomagnetite and magnetite would be needed to decouple these effects.
Given the established γ–α transition temperature of 912 °C in pure Fe, the presence of ferrite in both MAG and TTM at 938 °C is unexpected, and can only be explained as a compositional effect. First, the low illuminated volume (>0.4 mm3) eliminates the possibility of a large thermal gradient across the sample. Second, the α![[thin space (1/6-em)]](https://www.rsc.org/images/entities/i_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) γ ratio increases in both MAG and TTM with time, ruling out local endothermic cooling during the rapid early reaction, which would favour early ferrite formation. Finally, in situ platinum wire calibration indicates a constant temperature within the hot zone during the experiment, ruling out a thermal drift of the whole system. There remains considerable ferrite present at the endpoint for both MAG and TTM at 938 °C. Nitridation by nitrogen carrier gas has been observed via synchrotron to widen the γ–α transition temperature,73 but the use of pure hydrogen in this work excludes this factor. Both samples contain several strongly α-stabilising elements (Ti, Al, V, Si), with approximately 0.4 at% Ti in solution sufficient to completely eliminate austenite at 938 °C.49 None of these elements are directly reducible to a metallic state from their oxides under the reaction conditions investigated in this work. However, the presence of a large sink of metallic Fe will alter the activity of these elements.
γ ratio increases in both MAG and TTM with time, ruling out local endothermic cooling during the rapid early reaction, which would favour early ferrite formation. Finally, in situ platinum wire calibration indicates a constant temperature within the hot zone during the experiment, ruling out a thermal drift of the whole system. There remains considerable ferrite present at the endpoint for both MAG and TTM at 938 °C. Nitridation by nitrogen carrier gas has been observed via synchrotron to widen the γ–α transition temperature,73 but the use of pure hydrogen in this work excludes this factor. Both samples contain several strongly α-stabilising elements (Ti, Al, V, Si), with approximately 0.4 at% Ti in solution sufficient to completely eliminate austenite at 938 °C.49 None of these elements are directly reducible to a metallic state from their oxides under the reaction conditions investigated in this work. However, the presence of a large sink of metallic Fe will alter the activity of these elements.
Calculation of the equilibrium phase distribution and activities of elements in each phase (FactSage v8.2, FactPS/FactOxid/FactStel) does not indicate body-centred cubic ferrite as a stable end-phase for either MAG or TTM (Supplementary F). Austenite and spinel phases are indicated as the predominant stable phases. The microstructural partitioning of solutes during the reduction process clearly plays a key role in determining the stable end phases present.
This work demonstrates the significant effect of Ti on direct reduction behaviour of titanomagnetite ore. The exsolution of amorphous oxides has an established effect on particle sticking during fluidised bed reduction,16,17 and will influence downstream processing of reduced material. Further study is required to confirm the mechanistic cause of this exsolution behaviour, and the role of other cation substituents in the inverse spinel matrix.
Conclusions
In this study, in situ synchrotron X-ray diffraction was used to analyse the hydrogen reduction of New Zealand titanomagnetite ironsand at 938 °C, to better characterise the fluidised bed reduction behaviour. The importance of Ti content in determining the reduction pathway was confirmed. The key findings are:(1) The behaviour observed in situ is representative of fluidised bed reduction behaviour, with only minor deviations in reduction pathway at the end stage of reduction.
(2) A 5-stage reduction pathway is observed compared to the typical 3-stage pathway of a synthetic magnetite sample.
(3) The presence of titanium enriched titanohematite leads to a trimodal Ti content in the inverse spinel phase and asymmetric diffraction peaks during reduction.
(4) Ti stabilises the inverse spinel phase, preventing total reduction via wüstite at this temperature, and favouring the reduction of low Ti content crystallites first.
(5) Titanium is exsolved from the inverse spinel during reduction to wüstite, resulting in an amorphous Ti rich oxide shell.
(6) A transition to direct reduction of inverse spinel occurs late in Stage III, resulting in the accumulation of Ti in the remaining inverse spinel.
Author contributions
The contributions of the authors to the current work are as follows: conceptualization: B. I., M. J. R., S. P., M. N., M. H. R. and C. W. B.; data curation: M. L., B. I., B. M., S. S. and M. H. R.; formal analysis: M. L., B. I. and R. J. L.; funding acquisition: R. J. L., B. J. M., M. H. R. and C. W. B.; investigation: B. I., M. J. R., B. M., S. S. and M. H. R.; methodology: B. I., M. J. R., B. M. and M. H. R.; supervision: B. J. M., M. H. R. and C. W. B.; visualization: M. L.; writing – original draft: M. L. and B. I.; writing – review and editing: M. L., S. P., M. N., R. J. L., B. J. M. and C. W. B.Conflicts of interest
There are no conflicts to declare.Data availability
Data for this article are available at https://doi.org/10.17605/OSF.IO/WQT58. This includes data collected from the ANSTO synchrotron diffraction facility including raw and intensity corrected files.Supplementary information (SI): (A) photographs of experimental apparatus; (B) parameters for TOPAS fitting; (C) SEM and EDS maps of fluidised bed reduced particles; (D) reported data for compositional and thermal dependence of lattice parameters; (E) comparison of in situ synchrotron and fluidised bed XRD; (F) calculation of theoretical equilibrium phases. See DOI: https://doi.org/10.1039/d5ma01165e.
Acknowledgements
This research was undertaken on the Powder Diffraction beamline at the Australian Synchrotron, part of ANSTO [reference number AS171/PD/11627]. This work was supported by New Zealand Ministry of Business, Innovation and Employment [grant numbers RTVU1404 and RTVU1907]. Martin J. Ryan is acknowledged posthumously as a significant contributor to this work.References
- J. Kim, B. K. Sovacool, M. Bazilian, S. Griffiths, J. Lee, M. Yang and J. Lee, Energy Res. Soc. Sci., 2022, 89, 102565 CrossRef.
- International Energy Agency, Emissions Measurement and Data Collection for a Net Zero Steel Industry, OECD, Paris, 2023 Search PubMed.
- W. Choi and S. Kang, J. Environ. Manage., 2023, 335, 117569 CrossRef CAS PubMed.
- M. Sun, K. Pang, M. Barati and X. Meng, J. Sustainable Metallurgy, 2024, 10, 10–25 CrossRef.
- R. J. Holmes, Y. Lu and L. Lu, Iron Ore, Elsevier, 2022, pp. 1–56 Search PubMed.
- J. H. Luo, K. H. Qiu, Y. C. Qiu and P. C. Zhang, Adv. Mater. Res., 2013, 813, 292–297 CAS.
- L. M. L. Harahap, A. Idrus, Ernowo, I. G. Sukadana, Suwahyadi and T. Handayani, IOP Conf. Ser.: Earth Environ. Sci., 2023, 1233, 012022 CrossRef.
- S. Samanta, M. C. Goswami, T. K. Baidya, S. Mukherjee and R. Dey, Int. J. Miner., Metall. Mater., 2013, 20, 917–924 CrossRef CAS.
- R. L. Brathwaite, M. F. Gazley and A. B. Christie, J. Geochem. Explor., 2017, 178, 23–34 CrossRef CAS.
- Z. Yan, S. Zheng, Y. Zhang and Y. Zhang, JOM, 2025, 77, 3354–3367 CrossRef.
- A. Orpin, H. Bostock, S. Nodder, P. Barnes and G. Lamarche, Coasts and Ports 2009: In a Dynamic Environment, 2009, pp. 90–96 DOI:10.3316/INFORMIT.866385506331874.
- R. L. Brathwaite, A. B. Christie and M. F. Gazley, Miner. Depos., 2021, 56, 343–362 CrossRef CAS.
- C. van Vuuren, A. Zhang, J. T. Hinkley, C. W. Bumby and M. J. Watson, Cleaner Chem. Eng., 2022, 4, 100075 CrossRef.
- M. Girdwood, A. C. Pearcy, T. Arif, K. Dahm, A. T. Marshall and C. W. Bumby, ACS Sustainable Chem. Eng., 2025, 13, 4376–4385 CrossRef CAS.
- J. Yu, N. Hu, H. Xiao, P. Gao and Y. Sun, Powder Technol., 2021, 385, 83–91 Search PubMed.
- S. W. Prabowo, R. J. Longbottom, B. J. Monaghan, D. del Puerto, M. J. Ryan and C. W. Bumby, Powder Technol., 2022, 398, 117032 CrossRef CAS.
- S. W. Prabowo, R. J. Longbottom, B. J. Monaghan, D. del Puerto, M. J. Ryan and C. W. Bumby, Metall. Mater. Trans. B, 2019, 50, 1729–1744 CrossRef CAS.
- F. J. Berry, C. Greaves, Ö. Helgason, J. McManus, H. M. Palmer and R. T. Williams, J. Solid State Chem., 2000, 151, 157–162 CrossRef CAS.
- C. Melai, T. Boffa Ballaran, L. Uenver-Thiele, A. Kurnosov, A. I. Chumakov, D. Bessas and D. J. Frost, Phys. Chem. Miner., 2023, 50 DOI:10.1007/s00269-022-01217-2.
- J. Majzlan, A. Navrotsky and B. J. Evans, Phys. Chem. Miner., 2002, 29, 515–526 CrossRef CAS.
- Y. Kapelyushin, Y. Sasaki, J. Zhang, S. Jeong and O. Ostrovski, Metallu. Mater. Trans. B, 2015, 46, 2564–2572 CrossRef CAS.
- Z. Wang, J. Zhang, Z. Liu, K. Jiao and X. Xing, JOM, 2019, 71, 1776–1784 CrossRef CAS.
- J. Yu, Y. Ou, Y. Sun, Y. Li and Y. Han, Powder Technol., 2022, 402, 117340 CrossRef CAS.
- I. E. Grey and R. R. Merritt, J. Solid State Chem., 1981, 37, 284–293 CrossRef CAS.
- K. I. Lilova, C. I. Pearce, C. Gorski, K. M. Rosso and A. Navrotsky, Am. Mineral., 2012, 97, 1330–1338 CrossRef CAS.
- R. J. Harrison, U. Becker and S. A. T. Redfern, Am. Mineral., 2000, 85, 1694–1705 CrossRef CAS.
- S. W. Prabowo, R. J. Longbottom, B. J. Monaghan, D. del Puerto, M. J. Ryan and C. W. Bumby, JOM, 2022, 74, 885–898 CrossRef CAS.
- V. Materić, B. Ingham and R. Holt, CrystEngComm, 2015, 17, 7306–7315 RSC.
- S. K. Kuila, R. Chatterjee and D. Ghosh, Int. J. Hydrogen Energy, 2016, 41, 9256–9266 CrossRef CAS.
- A. Heidari, M. K. Ghosalya, M. Alaoui Mansouri, A. Heikkilä, M. Iljana, E. Kokkonen, M. Huttula, T. Fabritius and S. Urpelainen, Int. J. Hydrogen Energy, 2024, 83, 148–161 CrossRef CAS.
- N. E. Brown, A. Navrotsky, G. L. Nord and S. K. Banerjee, Am. Mineral., 1993, 78, 941–951 CAS.
- P. Fabrykiewicz, M. Stękiel, I. Sosnowska and R. Przeniosło, Acta Crystallogr., B: Struct. Sci. Cryst. Eng. Mater., 2017, 73, 27–32 CrossRef CAS.
- J. Song, D. Fan, S. Huang, S. Zhang, M. Wu, W. Chen and W. Zhou, Acta Geochim., 2023, 42, 988–997 CrossRef CAS.
- R. J. Harrison and S. A. T. Redfern, Phys. Chem. Miner., 2001, 28, 399–412 CrossRef CAS.
- B. A. Wechsler and C. T. Prewitt, Am. Mineral., 1984, 69, 176–185 CAS.
- C. I. Pearce, C. M. B. Henderson, N. D. Telling, R. A. D. Pattrick, J. M. Charnock, V. S. Coker, E. Arenholz, F. Tuna and G. van der Laan, Am. Mineral., 2010, 95, 425–439 CrossRef CAS.
- G. R. Holcomb, Mater. High Temp., 2019, 36, 232–239 CrossRef CAS.
- D. Levy, R. Giustetto and A. Hoser, Phys. Chem. Miner., 2012, 39, 169–176 CrossRef CAS.
- D. Levy, G. Artioli and M. Dapiaggi, J. Solid State Chem., 2004, 177, 1713–1716 CrossRef CAS.
- C. A. McCammon and L. Liu, Phys. Chem. Miner., 1984, 10, 106–113 CrossRef CAS.
- I. Seki and K. Nagata, ISIJ Int., 2005, 45, 1789–1794 CrossRef CAS.
- W. Q. Guo, S. Malus, D. H. Ryan and Z. Altounian, J. Phys.: Condens. Matter, 1999, 11, 6337–6346 CrossRef CAS.
- D. Y. Chung, P. J. Heaney, J. E. Post, J. E. Stubbs and P. J. Eng, J. Phys. Chem. Solids, 2023, 177, 111284 CrossRef CAS.
- Y. Hamano, J. Geomagnet. Geoelectricity, 1989, 41, 65–75 CrossRef CAS.
- R. A. Fischer, A. J. Campbell, G. A. Shofner, O. T. Lord, P. Dera and V. B. Prakapenka, Earth Planet. Sci. Lett., 2011, 304, 496–502 CrossRef CAS.
- Y. Waseda, E. Matsubara and K. Shinoda, X-Ray Diffraction Crystallography, Springer Berlin Heidelberg, Berlin, Heidelberg, 2011, pp. 107–167 Search PubMed.
- R. M. Hazen and R. Jeanloz, Rev. Geophys., 1984, 22, 37–46 CrossRef CAS.
- M. Hamada, S. Ganschow, D. Klimm, G. Serghiou, H.-J. Reichmann and M. Bickermann, Z. Naturforsch. B, 2022, 77, 463–468 CrossRef CAS.
- A. Walnsch, A. Leineweber and M. J. Kriegel, Calphad, 2023, 81, 102555 CrossRef CAS.
- K. Borowiec and T. Rosenqvist, Scand. J. Metall., 1981, 10, 217–224 CAS.
- E. Park and O. Ostrovski, ISIJ Int., 2003, 43, 1316–1325 CrossRef CAS.
- C. Gleitzer, Diversity and complexity of the wustite solid solutions i-tentative rationalization of the miscibility data and classification of the wustite ternary fields and of the postsaturation reactions, 1980, vol. 15 Search PubMed.
- J. B. MacChesney and A. Muan, Am. Mineral.: J. Earth Planetary Mater., 1961, 46, 572–582 CAS.
- T. Jantzen, K. Hack, E. Yazhenskikh and M. Müller, Calphad, 2018, 62, 187–200 CrossRef CAS.
- J. A. Van Orman and K. L. Crispin, Rev. Mineral. Geochem., 2010, 72, 757–825 CrossRef CAS.
- S. Aggarwal and R. Dieckmann, Phys. Chem. Miner., 2002, 29, 707–718 CrossRef CAS.
- P. Li, Y. Li, J. Yu, P. Gao and Y. Han, Int. J. Hydrogen Energy, 2022, 47, 31140–31151 CrossRef CAS.
- S. K. Gupta, V. Rajakumar and P. Grieveson, Canadian Metall. Quarterly, 1989, 28, 331–335 CrossRef CAS.
- P. Den Hoed and A. Luckos, Oil Gas Sci. Technol.–Revue d’IFP Energies nouvelles, 2011, 66, 249–263 CrossRef CAS.
- N. Kumagai, T. Hiraki, U. B. Pal, E. Kasai and T. Nagasaka, Metall. Mater. Trans. B, 2018, 49, 2278–2284 CrossRef CAS.
- N. Kumagai, T. Hiraki, U. B. Pal, E. Kasai and T. Nagasaka, Metall. Mater. Trans. B, 2018, 49, 2278–2284 CrossRef CAS.
- O. M. Borysenko, S. M. Logvinkov, G. M. Shabanova and I. A. Ostapenko, Voprosy Khimii i Khimicheskoi Tekhnologii, 2021, 12–15 CrossRef CAS.
- D. Xirouchakis, A. Smirnov, K. Woody, D. H. Lindsley and D. J. Andersen, Am. Mineral., 2002, 87, 658–667 CrossRef CAS.
- S. A. T. Redfern, R. J. Harrison, H. S. C. O’Neill and D. R. R. Wood, Am. Mineral., 1999, 84, 299–310 CrossRef CAS.
- W. Sugimoto, N. Kaneko, Y. Sugahara and K. Kuroda, J. Ceram. Soc. Jpn., 1997, 105, 101–105 CrossRef CAS.
- F. Bosi, U. Halenius and H. Skogby, Am. Mineral., 2009, 94, 181–189 CrossRef CAS.
- A. V. Kovalevsky, E. N. Naumovich, A. A. Yaremchenko and J. R. Frade, J. Eur. Ceram. Soc., 2012, 32, 3255–3263 CrossRef CAS.
- A. V. Kovalevsky, A. A. Yaremchenko, E. N. Naumovich, N. M. Ferreira, S. M. Mikhalev, F. M. Costa and J. R. Frade, J. Eur. Ceram. Soc., 2013, 33, 2751–2760 CrossRef CAS.
- M. Okube, T. Oshiumi, T. Nagase, R. Miyawaki, A. Yoshiasa, S. Sasaki and K. Sugiyama, J. Synchrotron Radiat., 2018, 25, 1694–1702 CrossRef CAS PubMed.
- S. M. Antao, I. Hassan and J. B. Parise, Am. Mineral., 2005, 90, 219–228 CrossRef CAS.
- I. Kinebuchi and A. Kyono, J. Mineral. Petrol. Sci., 2021, 116, 211–219 CrossRef CAS.
- C. E. Cava, L. S. Roman and C. Persson, Phys. Rev. B:Condens. Matter Mater. Phys., 2013, 88, 045136 CrossRef.
- Y. Wang, A. Heidari, H. Singh, G. King, S. Wang, R. F. Chuproski, M. Huttula, T. Fabritius and S. Urpelainen, Metall. Mater. Trans. B, 2025, 56, 5759–5772 CrossRef CAS.
Footnote |
| † Martin Ryan is acknowledged posthumously as a significant contributor to this work. |
| This journal is © The Royal Society of Chemistry 2026 |