Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Theoretical insights into lattice dynamics and thermal transport properties of lead-free quadruple halide perovskite Cs4CuSb2Cl12
Un-Sok
Pak
 *,
Dok-Song Che
Gal
,
Song-Myong
Jang
and
Un-Gi
Jong
*,
Dok-Song Che
Gal
,
Song-Myong
Jang
and
Un-Gi
Jong
 *
*
Metallic Material Institute, Faculty of Materials Science and Technology, Kim Chaek University of Technology, PO Box 76, Pyongyang, Democratic People's Republic of Korea. E-mail: pus93916@star-co.net.kp; ug.jong@ryongnamsan.edu.kp
First published on 20th November 2025
Abstract
In spite of extensive studies of halide perovskites, little attention has been paid to lattice dynamics and thermal transport properties of halide perovskite Cs4CuSb2Cl12 so far. In this work, we systematically investigate the structural, mechanical, and lattice dynamics and thermal transport properties for lead-free quadruple halide perovskite Cs4CuSb2Cl12 in the monoclinic phase using state-of-the-art first-principles methods. Scrutiny of the crystal structure reveals that Cs atoms are located inside the over-sized cage-like structure CsCl12, while Cu (Sb) atoms are tightly filled into the octahedral Cu(Sb)Cl6, expecting a role of atomic rattler for the Cs atoms. We carry out anharmonic lattice dynamics calculations at finite temperature based on self-consistent phonon theory, finding that monoclinic Cs4CuSb2Cl12 is dynamically stable at elevated temperature. Moreover, we find that the Cs atomic rattlers result in lattice anharmonicity and severely scatter heat-carrying acoustic and low-energy optical phonons, consequently leading to a low phonon group velocity and extremely short phonon lifetime. We then calculate the temperature-dependent lattice thermal conductivity κl of monoclinic Cs4CuSb2Cl12 using a unified theory of thermal transport for both crystals and glasses, demonstrating an extremely low κl of 0.27, 0.36 and 0.14 W m−1 K−1 at 300 K along x-, y- and z-axes. Moreover, it is found that nano-structuring can further suppress the κl by half. Strong lattice anharmonicity is again confirmed from the Grüneisen parameter calculation and temperature dependence of κl ∝ T−0.72. In addition, the elastic constant calculation demonstrates that monoclinic Cs4CuSb2Cl12 is mechanically stable with a brittle nature. Our work highlights theoretical insights into the lattice dynamics and thermal transport properties of monoclinic Cs4CuSb2Cl12.
1 Introduction
Lead-based simple perovskite halides APbX3 (A = Cs, CH3NH3; X = I, Br, Cl) have been studied extensively for light-absorber applications in solar cells,1 X-ray detectors2 and light-emitting diodes.3,4 Most prominently, perovskite solar cells have reached power conversion efficiencies of up to 25.2%5 in only ten years. Computational investigations6–8 have demonstrated that the superior optoelectronic properties of APbX3 are mostly ascribed to the unique electronic configuration of Pb 6s26p0, high crystalline symmetry and high dimensionality of electronic properties. However, the toxicity of lead and poor stability under ambient environmental conditions are the major drawbacks which hinders them from large-scale applications. This leads to great interest in lead-free perovskite exploration.9–11In this regard, the substitution of Pb with the same group elements of Sn or Ge can maintain the perovskite structure and advanced optoelectronic properties,12–15 but unfortunately, this causes more serious stability issues16–19 owing to the easy oxidation of Sn2+ (Ge2+) to Sn4+ (Ge4+), which destroys both the high structural symmetry and electronic configuration of Sn 5s25p0 (Ge 4s24p0). Replacing Pb with heterovalent elements such as Bi and Sb results in A3B2X9 compounds with improved phase stability. However, their optoelectronic properties become worse compared to those of APbX3 due to the unfavorable low dimensionality of their crystal and electronic structures.20–23 In order to maintain the high dimensionality of the crystal structure, the double halide perovskite A2BIBIIIX6 was derived by substituting two Pb2+ cations in APbX3 with BI and BIII cations.24–27 Although great freedom of combination between BI and BIII cations provides over 200 double perovskite halides with various chemical and physical properties, they cannot support efficient solar cells because of their unfavorable bandgap and likely formation of cation–cation antisites.28–31 A new 3D perovskite-type compound A4○CIIBIIIX1232–39 (○ = vacancy) was designed by substituting two BI cations in A2BIBIIIX6 by ○ and CII cations to provide an alternative platform for designing solar cell materials. Lin et al.32 synthesized quadruple perovskites Cs4CdSb2Cl12 and Cs4CdBi2Cl12 by a solvothermal method and observed steady-state photoluminescence showing warm orange emission. The quadruple perovskite Cs4CuSb2Cl1236 was fabricated by using a top-down ultrasonic exfoliation technique with a direct bandgap of 1.6 eV and superior optoelectronic properties due to a reduced electron effective mass. Cai et al.39 reported the first colloidal fabrication of Cs4CuSb2Cl12 perovskite-type nanocrystals with a direct bandgap of 1.79 eV, an ultrafast photo-response and narrow bandwidth. On the other hand, Nag's group37 mechanochemically synthesized Cs4CuSb2Cl12, demonstrating the magnetic response of the Cu2+ cation which controls the electronic properties, its thermal and chemical stabilities. Moreover, Karmaker and co-workers40 recently predicted a high power conversion efficiency of 23.1% and an open-circuit voltage of 1.2 V in the Cs4CuSb2Cl12-based solar cell by computationally optimizing perovskite layer thickness, doping level, defect density and so on.
To the best of our knowledge following a literature search, most investigations mainly concentrated on synthesis methods and optoelectronic properties for the quadruple perovskite halide Cs4CuSb2Cl12, and thus its mechanical, lattice dynamics and transport properties have never been explored so far. Recently, Delaire et al. investigated the structural instability, optical and thermal properties of the lead halide perovskite CsPbBr3, linking soft anharmonic lattice dynamics and optoelectronic properties.41 Moreover, Gupta and co-workers42 performed ab initio molecular dynamics simulations combined with neutron scattering measurements in the superionic argyrodite Cu7PSe6, highlighting the importance of strongly anharmonic effects on the ionic and thermal transport properties of the superionic systems. In this work, we report a systematic investigation of the lattice dynamics and transport properties including phonon dispersion curves, phonon density of states and lattice thermal conductivity for the all-inorganic lead-free quadruple halide perovskite Cs4CuSb2Cl12 using state-of-the-art first-principles approaches and methods.
2 Computational methods
All the density functional theory (DFT) calculations were performed using the Vienna ab initio simulation package (VASP),43,44 employing the projector augmented wave (PAW) potentials45,46 with the valence electron configurations of Cs-5s25p66s1, Cu-3d104p1, Sb-5s25p3 and Cl-3s23p5. We utilized the PBEsol functional47 known to accurately predict the lattice constants of all-inorganic halide perovskites48–50 which is crucial for a reliable description of lattice dynamics properties. We used a kinetic energy cutoff of 600 eV for a plane-wave basis set and a k-point mesh of 10 × 10 × 6 for Brillouin zone (BZ) integrations, which were enough to guarantee a total energy accuracy as 3.5 meV per atom. Lattice dynamics and thermal conductivity calculations were performed using the ALAMODE code.51,52 The atomic positions were relaxed until the atomic forces are smaller than 5 × 10−3 eV Å−1, while the energy convergence criterion was set to be 10−7 eV. The interatomic force constants (IFCs) were calculated using a 2 × 2 × 2 supercell expansion, and for supercell calculations, we used the same convergence thresholds with a reduced kinetic energy cutoff of 400 eV and 5 × 5 × 3 k-point mesh. In this work, we mainly focused on the lattice dynamics and thermal transport properties of Cs4CuSb2Cl12, and therefore, the spin-polarization of Cu2+ atoms was not considered.In order to calculate the harmonic phonon dispersion curves and phonon density of states (DOS), we used the second-order (harmonic) IFCs by utilizing the finite displacement method with 2 × 2 × 2 supercell, as implemented in the ALAMODE code.51,52 We prepared 20 different supercells where all the atoms were randomly displaced by 0.01 Å from their equilibrium positions, and then, calculated the atomic forces for each displaced configuration, extracting harmonic IFCs by using the ALAMODE code. We performed the convergence tests of the phonon eigenvalues with respect to the supercell size, revealing that the results of the 2 × 2 × 2 supercell agree well with the ones of 3 × 3 × 3 the supercell for Cs4CuSb2Cl12 as shown in Fig. S1, SI. We performed self-consistent phonon (SCP) calculations in order to include the anharmonic effects at finite temperatures.51–53 Within the SCP theory, the anharmonic phonon eigenvalues were computed as functions of temperature from the pole of the Green's function beyond perturbation theory, as implemented in the ALAMODE code. In the SCP equation, the phonon self-energy becomes frequency-dependent only treating the loop diagram related to the quartic IFCs. Note that the off-diagonal elements of the self-energy should be accounted for to allow the phonon eigenvectors to change through anharmonic effects.52 The current SCP approach applied to this work was confirmed to be efficient and accurate in accounting for lattice dynamics and thermal transport properties of systems with severe anharmonicity. For estimating anharmonic IFCs, we employed the compressive sensing lattice dynamics (CSLD) approach,54 as implemented in the ALAMODE code. To do this, we prepared over 60 configurations where all of the atoms are displaced randomly with large displacements54 and computed atomic forces for each configuration using precise DFT calculations. For cubic and quartic IFCs, we included up to 5th- and 3rd-nearest neighbor shells, whereas fifth- and sixth-order IFCs were considered for nearest-neighbor pairs. In order to estimate the accuracy of the IFCs estimated by the CSLD approach, we show the comparison of the atomic forces obtained from DFT and CSLD calculations in Fig. S2, SI. By using the higher-order IFCs, we guaranteed a relative error below 2.6% for the fitting of the atomic forces compared with the DFT-calculated forces.
With the calculated 3rd-order IFCs, we solved the Boltzmann transport equation (BTE) for phonons within the relaxation time approximation (RTA) and estimated the lattice thermal conductivity, as implemented in the ALAMODE code. We performed convergence tests for the calculations of lattice thermal conductivity κl according to the size of q-point mesh in the phonon BZ, demonstrating that a 20 × 20 × 15 q-point mesh is sufficient to determine κl with an error less than 0.01 W m−1 K−1 (see Fig. S3, SI). On the other hand, it was recently suggested that both diagonal and off-diagonal contributions to κl can be calculated from the phonon velocity operator based on the unified theory of thermal transport for both crystals and glasses.55 Furthermore, it was demonstrated that the off-diagonal contributions were found to be essential even after the anharmonic phonon renormalization was taken into account in recent studies56–58 on the lattice thermal conductivity. By solving the Boltzmann transport equation (BTE) on top of the SCP phonon dispersions, the Peierls's contribution κlP to the lattice thermal conductivity κl was estimated from the harmonic and cubic IFCs, as implemented in the ALAMODE code. Meanwhile, the coherent contribution κlC was calculated using the phonon velocity operator within the framework of unified theory for the lattice transport properties,55 as implemented in the ALAMODE code.
The mechanical stability of materials with polycrystalline structure can be determined by elastic constants including bulk (B), shear (G) and Young's (E) moduli. Such elastic moduli were estimated from the elastic stiffness (Cij) and compliance (Sij) constants for the single crystal material, which were obtained from the density functional perturbation theory (DFPT) calculations.59 For a monoclinic structure, the mechanical stability criterion can be expressed as follows.
| C11 > 0, C22 > 0, C33 > 0, C44 > 0, C55 > 0, C66 > 0 | (1) |
| C11 + C22 + C33 + 2(C12 + C13 + C23) > 0 | (2) |
| C33C55 − C352 > 0, C44C66 − C462 > 0, C22C33 − 2C23 > 0 | (3) |
| C22(C33C55 − C352) + 2C23C25C35 − C232C55 − C252C33 > 0 | (4) |
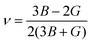 smaller (larger) than 1.75 and 0.26, respectively, are regarded as brittle (ductile) materials.
smaller (larger) than 1.75 and 0.26, respectively, are regarded as brittle (ductile) materials.
3 Results and discussion
3.1 Structural properties
We begin with scrutinizing the crystalline structure of the quadruple halide perovskite Cs4CuSb2Cl12. According to previous experiment,39 Cs4CuSb2Cl12 crystallizes in the monoclinic phase with the C2/m space group. By performing the variable-cell structural optimization, we obtained the PBEsol-optimized lattice constants of a = 13.12 Å, b = 7.36 Å, c = 13.02 Å and β = 112.59°, in good agreement with the previous experimental results39 of a = 13.04 Å, b = 7.30 Å, c = 12.97 Å and β = 111.73°. Fig. 1 displays the polyhedral view of the PBEsol-optimized crystal structure for the quadruple halide perovskite Cs4CuSb2Cl12 with the monoclinic C2/m space group. A particular structural feature is that its crystalline structure consists of corner- and face-sharing cage-like structures composed of Cl− anions, which accommodate one metal cation from among the Cs+, Cu2+ and Sb3+. That is, the Cu2+ (Sb3+) cations are at the center of the CuCl6 (SbCl6) octahedral cages, while the Cs+ cations are located inside the cub-octahedral cages. Moreover, the average value of the Cu–Cl (Sb–Cl) bond length was measured to be 2.47 (2.61) Å which is slightly larger (smaller) than the sum of ionic radii of 2.54 (2.57) Å for Cu2+ (Sb3+) and Cl− ions. However, the average distance 3.69 Å between the Cs+ cation and the Cl− anion is much greater than the sum of ionic radii of 3.48 Å for the corresponding ions. This obviously indicates that the Cs atoms are located inside the over-sized cub-octahedral CsCl12 cages, whereas the Cu and Sb atoms are tightly filled into the octahedral CuCl6 and SbCl6 cages, respectively. From such structural features of Cs4CuSb2Cl12, it can be expected that the Cs atoms play roles as the vibrational rattlers because of the fairly large interstitial space in the CsCl12 cub-octahedral cage, driving the lattice anharmonicity like A atoms in the halide perovskites ABX3.61,623.2 Mechanical and elastic properties
Solar cell materials are utilized to directly generate electricity by absorbing sunlight for the application of the solar cell system. For that reason, their mechanical property is an important factor as it determines the solar cell system's safety and durability under operation. Within the DFPT approach, we estimated the independent elastic stiffness constants of C11, C22, C33, C44, C55, C66, C12, C13, C23, C25, C35 and C46 to be 16.49, 20.97, 18.39, 5.62, 5.60, 6.50, 6.65, 5.94, 6.47, 0.99, 2.02 and 0.22 GPa, respectively for the monoclinic Cs4CuSb2Cl12. We found that these elastic stiffness constants satisfy the Born's stability criteria for the monoclinic solid crystals, expressed by eqn (1)–(4), thus indicating mechanical stability at ambient conditions. It should be noted that the deformation resistance and mechanical stability along the x-, y- and z-axes are superior to the shear deformation resistance and stability because the Cii (i = 1, 2, 3) is larger by one order in magnitude than the Cij (i ≠ j). Meanwhile, we obtained B = 10.44 GPa, E = 15.10 GPa and G = 6.00 GPa for the polycrystalline Cs4CuSb2Cl12, implying that uniform pressure and uniaxial stress are larger than the shear stress owing to the larger value of B and E than the value of Y. It was found that the monoclinic Cs4CuSb2Cl12 is considered as a brittle material because of its Poisson's ratio ν of 0.26 and Pugh's ratio B/G of 1.74 being smaller than the threshold values of 0.26 and 1.75. For describing the elastic anisotropy, we obtained the universal anisotropic index AU = 0.29, finding much deviation from zero, i.e., from the isotropic nature. Furthermore, we determined the relatively low average sound velocity and Debye temperature of 1469.98 m s−1 and 136.60 K by using the calculated elastic moduli.3.3 Lattice dynamics properties
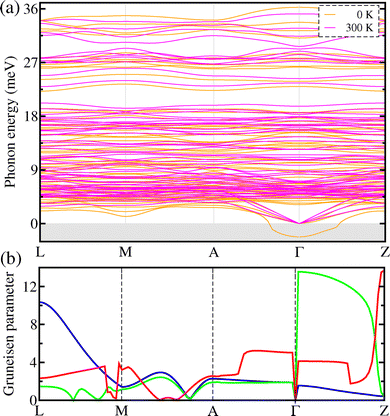
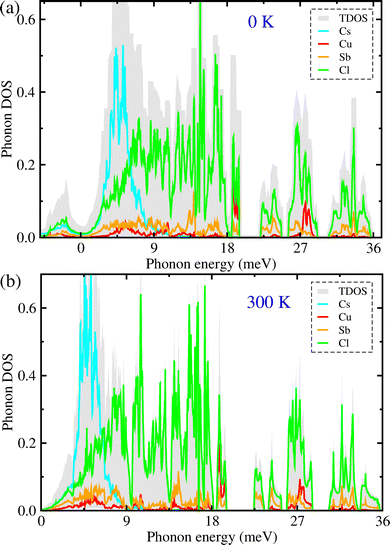
In the next step, we carried out lattice dynamics calculations for the monoclinic Cs4CuSb2Cl12 using 2 × 2 × 2 supercells in order to investigate its dynamical stability, phonon energies and density of state (DOS). The dynamic stability and thermal vibration of constituent atoms at a given temperature are determined by lattice dynamics for crystalline solids. Fig. 2(a) shows the harmonic phonon dispersion curves (brown) calculated at 0 K along the L–M–A–Γ–Z line of the phonon Brillouin zone (BZ). As can be seen, the soft phonon modes with imaginary phonon energies were identified at the Γ point for the monoclinic Cs4CuSb2Cl12. This fact seems to be at odds with the experimental finding that Cs4CuSb2Cl12 adopts the monoclinic phase at ambient conditions.38,39 However, Xie and co-workers62 already found that the anharmonic soft modes could be shown even for the dynamically stable cubic CsSnI3, which was associated with the strong lattice vibrations of atomic rattlers, resulting in the octahedral distortions of SnI6. Actually, we observed the Cu(Sb)Cl6 octahedral and CsCl12 cub-octahedral distortions with elongation along the z direction and shortening in the x−y plane for the metal–chlorine bond lengths being ascribed to the Jahn–Teller effect owing to the presence of metal cations with the unpaired electrons as confirmed in the previous experiment.39 That is to say, it is expected that the anharmonic phonon modes are just induced by the rattling vibrations of the metal cations, especially by the Cs-atomic rattling vibrations.In order to directly measure the lattice anharmonicity, we also computed the Grüneisen parameter associated with the acoustic phonon modes for the monoclinic Cs4CuSb2Cl12 as implemented in the ALAMODE code.51,52 In general, the Grüneisen parameters are determined by the change of phonon energies according to the volume of the unit cell.63 As can be seen in Fig. 2(b), the Grüneisen parameters reached the maximum value over 12 in the Γ−Z line of the phonon BZ with an average value of 3.9. Such a large Grüneisen parameter is comparable to the one of 4.1 for the strongly anharmonic SnSe,64 suggesting strong lattice anharmonicity and thus low lattice thermal conductivity for Cs4CuSb2Cl12. On the other hand, we calculated the atom-resolved phonon density of states (DOS) of the monoclinic Cs4CuSb2Cl12 at 0 K. As shown in Fig. 3(a), the phonon DOS calculation revealed that the soft phonon modes predominantly originate from the Cs and Cl atoms with a small contribution from the Cu and Sb atoms. The Cs atoms make a major contribution to the acoustic and low-energy optical phonon modes, whereas the Cl contributes to the mid- and high-energy optical modes. From the presence of such soft modes, it can be assumed that at 0 K, the quadruple halide perovskite Cs4CuSb2Cl12 is dynamically unstable in the monoclinic phase. Moreover, such dynamical instability implies that the harmonic lattice dynamics at 0 K cannot accurately capture the lattice vibrational properties of the constituent atoms at an elevated temperature, and consequently, anharmonic lattice dynamics is necessary to reasonably describe the lattice dynamic stability and vibrational properties at finite temperature for the monoclinic Cs4CuSb2Cl12.
To this end, we then performed self-consistent phonon (SCP) calculations to compute the phonon eigenvalues by considering the anharmonic effects at finite temperatures. Fig. 2(a) also shows the anharmonic phonon dispersion curves (magenta) calculated at 300 K for the monoclinic Cs4CuSb2Cl12. In comparison with the harmonic phonon dispersion curves (brown) at 0 K, the soft phonon modes with the imaginary eigenvalues were renormalized to become real energies at 300 K. This clearly demonstrates that the monoclinic phase of the quadruple halide perovskite Cs4CuSb2Cl12 becomes dynamically stable at finite temperatures. When the temperature increases from 0 K to 300 K, the acoustic as well as optical phonon modes are noticeably renormalized across the whole range of the phonon BZ, demonstrating the strong lattice anharmonicity. Importantly, the lowest optical phonon modes are present at about 4.5 meV in the acoustic region at the phonon BZ center, indicating the strong coupling between the heat-carrying acoustic and low-energy optical modes. In the previous studies,65,66 it was confirmed that the low-energy optical phonons coupled with the acoustic modes play roles as strong vibrational rattlers, severely scattering heat-carrying acoustic phonons. Moreover, the phonon DOS at 300 K (Fig. 3(b)) was found to have the Cs-related phonon modes peaked around 5 meV in the acoustic phonon and low-energy optical phonon region, which are mostly attributed to the rattling vibrations of Cs atoms. In Fig. 5(c), we intuitively show the atomic vibrations corresponding to the lowest-energy optical phonon mode at the Γ point. As can be seen, the Cs atoms have the largest amplitude among the constituent atoms, indicating a strong atomic rattler. It can be inferred that the rattling vibrations of the Cs atoms strongly scatter the heat-carrying acoustic and low-energy optical phonons, accordingly decreasing the lattice thermal conductivity in the monoclinic Cs4CuSb2Cl12. On the other hand, upon increasing temperature, the SCP dispersions broaden towards the higher phonon energy region due to the stronger thermal vibrations of the constituent atoms (see Fig. S6 in the SI).
3.4 Lattice transport properties
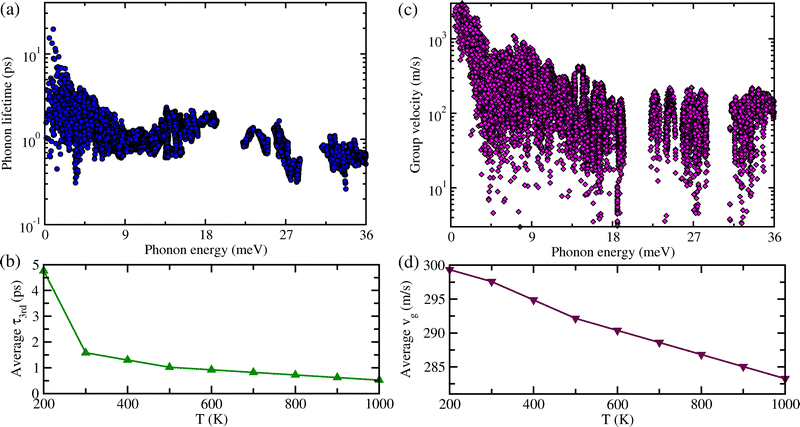
In the final stage, with the use of the SCP dispersions and DOS, and third-order interatomic force constants, we estimated the lattice transport properties such as phonon lifetime τ3rd, phonon group velocity vg and lattice thermal conductivity κl for the monoclinic Cs4CuSb2Cl12. Fig. 4(a) and (c) respectively display the calculated τ3rd and vg at 300 K as functions of phonon energy. It was found that the values of τ3rd are extremely low (<20 ps) which is one order of magnitude smaller than the τ3rd of the conventional thermoelectric materials, whereas the vg is lower than ∼ 2000 m s−1 in the whole range of phonon energy. Such low vg at the Γ point is in reasonable agreement with the average sound velocity of 1469.98 m s−1 assessed by the elastic constant calculations. In particular, we note that the rapid decrement of τ3rd and vg according to the increment of phonon energy from 0 meV to 9 meV is attributed to the strong phonon scattering caused by the rattling vibrations of the Cs atoms, as confirmed above. Such small vg and suppressed τ3rd suggest that the monoclinic Cs4CuSb2Cl12 can have extremely low lattice thermal conductivity. On the other hand, the average τ3rd and vg slowly decrease with increasing temperature because the phonon scattering intensity becomes strong according to the increment of temperature as shown in Fig. 4(b) and (d).By using the calculated τ3rd and vg, we eventually assessed the Peierls's contribution κlP to the lattice thermal conductivity κl following the formula 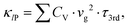 where CV, τ3rd and vg are the heat capacity, phonon lifetime and group velocity, respectively. As shown in Fig. S4 in the SI, the calculated CV increases rapidly when the temperature goes from 0 K to 400 K and after that reaches a plateau, following the general tendency of a solid crystal. Fig. 5(a) shows the Peierls's contribution κlP (dashed), the coherent contribution κlC (dotted) and total κl (solid) calculated along the x-, y- and z-axes for Cs4CuSb2Cl12. It was observed that the κl is strongly anisotropic (κl,yy > κl,xx > κl,zz), i.e., it has different values along the different axes. In the x- and y-axis, the κlP is dominant in comparison with the κlC over the whole range of temperature, while in the z-axis, the two contributions to the κl are comparable with each other. With reference to the magnitude of lattice thermal conductivity, the κl was estimated to be 0.27, 0.36 and 0.14 W m−1 K−1 at 300 K along the x-, y- and z-axes, respectively, values which are comparable to 0.48 W m−1 K−1 of conventional thermoelectric material SnSe64 and even one order of magnitude smaller than the 6.96 W m−1 K−1 of GeTe.67 Meanwhile, it should be noted that the κl computed by iteratively solving the BTE is slightly overestimated in comparison with the none-iteratively calculated one (see Fig. S7, SI). Such extremely low κl makes suggestion that the monoclinic Cs4CuSb2Cl12 can be very promising for applications as a thermoelectric material.
where CV, τ3rd and vg are the heat capacity, phonon lifetime and group velocity, respectively. As shown in Fig. S4 in the SI, the calculated CV increases rapidly when the temperature goes from 0 K to 400 K and after that reaches a plateau, following the general tendency of a solid crystal. Fig. 5(a) shows the Peierls's contribution κlP (dashed), the coherent contribution κlC (dotted) and total κl (solid) calculated along the x-, y- and z-axes for Cs4CuSb2Cl12. It was observed that the κl is strongly anisotropic (κl,yy > κl,xx > κl,zz), i.e., it has different values along the different axes. In the x- and y-axis, the κlP is dominant in comparison with the κlC over the whole range of temperature, while in the z-axis, the two contributions to the κl are comparable with each other. With reference to the magnitude of lattice thermal conductivity, the κl was estimated to be 0.27, 0.36 and 0.14 W m−1 K−1 at 300 K along the x-, y- and z-axes, respectively, values which are comparable to 0.48 W m−1 K−1 of conventional thermoelectric material SnSe64 and even one order of magnitude smaller than the 6.96 W m−1 K−1 of GeTe.67 Meanwhile, it should be noted that the κl computed by iteratively solving the BTE is slightly overestimated in comparison with the none-iteratively calculated one (see Fig. S7, SI). Such extremely low κl makes suggestion that the monoclinic Cs4CuSb2Cl12 can be very promising for applications as a thermoelectric material.
We then considered the temperature dependence of the κl averaged in the x-, y- and z-axes, finding the relation of κl ∝ T−0.72 for the monoclinic Cs4CuSb2Cl12 (see Fig. S5 in the SI). Such a large deviation from the κl ∝ T−1 tendency again confirms the strong lattice anharmonicity of Cs4CuSb2Cl12. To establish deep insight into the lattice transport properties, we computed the lattice thermal conductivity spectra κl(hω) as a function of phonon energy and cumulative thermal conductivity as a function of mean free path (MFP) at 300 K within the SCP approach. As shown in Fig. 5(b) (solid), the spectra κl(hω) is dominant in the phonon energies ranging from 0 meV to 5 meV, while is negligible in the energies ranging from 5 meV to 9 meV and in the high-energy region. This indicates that the acoustic phonon modes coupled with the low-energy optical modes below 5 meV have a major contribution to the κl, while the phonons corresponding to the optical modes over 5 meV are strongly scattered by the Cs atomic rattling vibrations, severely suppressing the κl for the monoclinic Cs4CuSb2Cl12. Furthermore, we compared the κl of Cs4CuSb2Cl12 with Cu2+ atoms and CaCuP with Cu1+ atoms to gain a deeper understanding of the lattice thermal transport properties. The Cs4CuSb2Cl12 exhibits one order of magnitude smaller κl than the CaCuP68 due to the strong rattling vibrations of the Cs atoms. In addition, the cumulative κl (dashed) in Fig. 5(b) is saturated at the MFP of about 40 nm, showing that the dominant heat-carrying phonons have MFPs ranging from 2 nm to 20 nm. The MFPs are significantly suppressed and much less than the phonon wavelength or inter-atomic spacing, violating the Ioffe–Regel limit.69 This violation supports the strong lattice anharmonicity and ultralow lattice thermal conductivity. Moreover, we note that the κl can be decreased by half for the nano-structure Cs4CuSb2Cl12 with a size less than 10 nm.
4 Conclusions
In this work, we have systemically investigated structural, mechanical, lattice dynamics and thermal transport properties of quadruple halide perovskite Cs4CuSb2Cl12 in monoclinic phase using state-of-the-art first-principles methods. Structural optimization calculations revealed that PBEsol-optimized lattice constants and bond lengths are in good agreement with available experimental data. In particular, scrutiny of crystalline structure turned out that Cs atoms are located inside over-sized cub-octahedral cage-like structure CsCl12, while Cu (Sb) atoms are tightly filled into octahedral Cu(Sb)Cl6 cages, expecting a role of atomic rattler for the Cs atoms. We then performed anharmonic lattice dynamics calculations at finite temperature based on self-consistent phonon theory, demonstrating that the monoclinic phase of Cs4CuSb2Cl12 is dynamically stable at elevated temperature. Moreover, we found that the Cs atomic rattlers stimulate lattice anharmonicity and severely scatter heat-carrying acoustic and low-energy optical phonons, consequently leading to small phonon group velocity and extremely short phonon lifetime. At the end, we calculated temperature-dependent lattice thermal conductivity of the monoclinic Cs4CuSb2Cl12 using unified theory of thermal transport for both crystals and glasses, demonstrating an extremely low lattice thermal conductivity of 0.27, 0.36 and 0.14 W m−1 K−1 at 300 K along the x-, y- and z-axis. Moreover, it was found that nano-structuring can further suppress the κl by half for Cs4CuSb2Cl12. Strong lattice anharmonicity was again confirmed from Grüneisen parameter calculation and temperature dependence of lattice thermal conductivity, i.e., κl ∝ T−0.72. In addition, elastic constant calculation illustrated that the monoclinic Cs4CuSb2Cl12 is mechanically stable with a brittle nature. Our work highlights atomic insights into lattice dynamics and thermal transport properties of the quadruple halide perovskite Cs4CuSb2Cl12 with monoclinic phase.Author contributions
Un-Sok Pak developed the original project, performed the calculations and drafted the first manuscript. Dok-Song Che Gal and Song-Myong Jang assisted with the DFT calculations and the post-processing of calculation results, and contributed to useful discussions. Un-Sok Pak and Un-Gi Jong supervised the work. All authors reviewed the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.Supplementary information (SI): convergence tests of phonon dispersion curve according to the supercell size, comparison of the atomic forces obtained from the density functional theory and compressive sensing lattice dynamics calculations, convergence tests of lattice thermal conductivity according to the size of q-point mesh, heat capacity as a function of temperature, average phonon lifetime and group velocity and self-consistent phonon dispersion curves at different temperatures for the monoclinic Cs4CuSb2Cl12. See DOI: https://doi.org/10.1039/d5ma01099c.
Acknowledgements
This work made use of the High-Performance Computing Service via our membership of the Computing Center in Kim Chaek University of Technology. The authors are extremely grateful to Prof. S.-I. Jo, Song-Gun Kim and Gyong-Sok Kim for the useful discussion and provision of scripts allowing the calculation.References
- A. Kojima, K. Teshima, Y. Shirai and T. Miyasaka, J. Am. Chem. Soc., 2009, 131, 6050–6051 CrossRef CAS PubMed.
- G. Niu, W. Li, J. Li, X. Liang and L. Wang, RSC Adv., 2017, 7, 17473–17479 RSC.
- W. Xu, Q. Hu, S. Bai, C. Bao, Y. Miao, Z. Yuan, T. Borzda, A. J. Barker and E. Tyukalova, Nat. Photonics, 2019, 13, 418–424 CrossRef CAS.
- K. Lin, J. Xing, L. N. Quan, F. P. G. de Arquer, X. Gong, J. Lu, L. Xie, W. Zhao and D. Zhang, Nature, 2018, 562, 245–248 CrossRef CAS PubMed.
- National Renewable Laboratory, Best Research-Cell Efficiency Chart, Accessed 2019-07-12, https://www.nrel.gov/pv/cell-efficiency.html.
- W. J. Yin, T. Shi and Y. Yan, Adv. Mater., 2014, 26, 4653–4658 CrossRef CAS PubMed.
- Z. Xiao, W. Meng, J. Wang, D. B. Mitzi and Y. Yan, Mater. Horiz., 2017, 4, 206–216 RSC.
- Z. Xiao and Y. Yan, Adv. Energy Mater., 2017, 7, 1701136 CrossRef.
- R. A. Evarestov, E. A. Kotomin, A. Senocrate, R. K. Kremer and J. Maier, Phys. Chem. Chem. Phys., 2020, 22, 3914–3920 RSC.
- Y.-X. Zhang, H.-Y. Wang, Z.-Y. Zhang, Y. Zhang, C. Sun, Y.-Y. Yue, L. Wang, Q.-D. Chen and H.-B. Sun, Phys. Chem. Chem. Phys., 2017, 19, 1920–1926 RSC.
- F. Dybala, R. Kudrawiec, M. P. Polak, A. P. Herman, A. Sieradzki and M. Maczka, Mater. Adv., 2025, 6, 569–579 RSC.
- T. Krishnamoorthy, H. Ding, C. Yan, W. L. Leong, T. Baikie, Z. Zhang, M. Sherburne, S. Li, M. Asta, N. Mathews and S. G. Mhaisalkar, J. Mater. Chem. A, 2015, 3, 23829–23832 Search PubMed.
- X. Zhang, Y. Ma, X. Chen, X. Li, W. Zhou, N. A. N. Ouedraogo, Y. Shirai, Y. Zhang and H. Yan, RSC Adv., 2021, 11, 33637–33645 Search PubMed.
- W. Chen, X. Xin, Z. Zang, X. Tang, C. Li, W. Hu, M. Zhou and J. Du, Phys. Chem. Chem. Phys., 2017, 19, 115–120 Search PubMed.
- Y. Takahashi, R. Obara, Z. Z. Lin, T. Naito, T. Inabe, S. Ishibashi and K. Terakura, Dalton Trans., 2011, 40, 5563–5568 RSC.
- Y. Han, X. Cheng and B.-B. Cui, Mater. Adv., 2023, 4, 355–373 RSC.
- N. K. Noel, S. D. Stranks, A. Abate, C. Wehrenfennig, S. Guarnera, A.-A. Haghighirad, A. Sadhanala, G. E. Eperon, S. K. Pathak and M. B. Johnston, Energy Environ. Sci., 2014, 7, 3061–3068 RSC.
- A. E. Shalan, Mater. Adv., 2021, 1, 292–309 RSC.
- X. Jiang, Z. Zang, Y. Zhou, H. Li, Q. Wei and Z. Ning, Acc. Mater. Res., 2021, 2, 210–219 CrossRef CAS.
- T. L. Hodgkins, C. N. Savory, K. K. Bass, B. L. Seckman, D. O. Scanlon, P. I. Djurovich, M. E. Thompson and B. C. Melot, Chem. Commun., 2019, 55, 3164–3167 RSC.
- S. Chander and S. K. Tripathi, Mater. Adv., 2022, 3, 7198–7211 RSC.
- K. Mahmood, S. Sarwar and M. T. Mehran, RSC Adv., 2017, 7, 17044–17062 RSC.
- G. Volonakis, M. R. Filip, A. A. Haghighirad, N. Sakai, B. Wenger, H. J. Snaith and F. Giustino, J. Phys. Chem. Lett., 2016, 7, 1254–1259 CrossRef CAS PubMed.
- M. R. Filip, S. Hillman, A. A. Haghighirad, H. J. Snaith and F. Giustino, J. Phys. Chem. Lett., 2016, 7, 2579–2585 CrossRef CAS PubMed.
- X.-G. Zhao, D. Yang, J.-C. Ren, Y. Sun, Z. Xiao and L. Zhang, Joule, 2018, 2, 1662–1673 CrossRef CAS.
- T. T. Tran, J. R. Panella, J. R. Chamorro, J. R. Morey and T. M. McQueen, Mater. Horiz., 2017, 4, 688–693 RSC.
- E. T. McClure, M. R. Ball, W. Windl and P. M. Woodward, Chem. Mater., 2016, 28, 1348–1354 CrossRef CAS.
- F. Giustino and H. J. Snaith, ACS Energy Lett., 2016, 1, 1233–1240 Search PubMed.
- M. K. Jana, S. M. Janke, D. J. Dirkes, S. Dovletgeldi, C. Liu, X. Qin, K. Gundogdu, W. You and V. Blum, J. Am. Chem. Soc., 2019, 141, 7955–7964 Search PubMed.
- B. A. Connor, F. Leppert, M. D. Smith, J. B. Neaton and H. I. Karunadasa, J. Am. Chem. Soc., 2018, 140, 5235–5240 CrossRef CAS PubMed.
- E. Greul, M. L. Petrus, A. Binek, P. Docampo and T. Bein, J. Mater. Chem. A, 2017, 5, 19972–19981 RSC.
- Y. P. Lin, S. Hu, B. Xia, K. Q. Fan, L. K. Gong, J. T. Kong, X. Y. Huang, Z. Xiao and K. Z. Du, J. Phys. Chem. Lett., 2019, 10, 5219–5225 CrossRef CAS PubMed.
- B. Vargas, R. Torres-Cadena, J. Rodriguez-Hernandez, M. Gembicky, H. Xie, J. Jimenez-Mier, Y.-S. Liu, E. Menendez-Proupin, K. R. Dunbar, N. Lopez, P. Olalde-Velasco and D. Solis-Ibarra, Chem. Mater., 2018, 30, 5315–5321 CrossRef CAS.
- B. Vargas, R. Torres-Cadena, D. T. Reyes-Castillo, J. Rodriguez-Hernandez, M. Gembicky, E. Menendez-Proupin and D. Solis-Ibarra, Chem. Mater., 2019, 32, 424–429 CrossRef.
- J. Xu, J.-B. Liu, J. Wang, B.-X. Liu and B. Huang, Adv. Funct. Mater., 2018, 28, 1800332 CrossRef.
- X. D. Wang, N. H. Miao, J. F. Liao, W. Q. Li, Y. Xie, J. Chen, Z. M. Sun, H. Y. Chen and D. B. Kuang, Nanoscale, 2019, 11, 5180–5187 RSC.
- N. Singhal, R. Chakraborty, P. Ghosh and A. Nag, Chem. – Asian J., 2018, 13, 2085–2092 CrossRef CAS PubMed.
- B. Vargas, E. Ramos, E. Perez-Gutierrez, J. C. Alonso and D. Solis-Ibarra, J. Am. Chem. Soc., 2017, 139, 9116–9119 CrossRef CAS PubMed.
- T. Cai, W. Shi, S. Hwang, K. Kobbekaduwa, Y. Nagaoka, H. Yang, K. Hills-Kimball, H. Zhu, J. Wang, Z. Wang, Y. Liu, D. Su, J. Gao and O. Chen, J. Am. Chem. Soc., 2020, 142, 11927–11936 CrossRef CAS PubMed.
- H. Karmaker, A. Siddique, B. K. Das and M. N. Islam, Results Eng., 2024, 22, 102106 CrossRef CAS.
- O. Delaire, R. Osborn, S. Rosenkranz, M. G. Kanatzidis, D.-Y. Chung, Z. Xu, G. Xu, D. L. Abernathy, D. M. Pajerowski, M. J. Krogstad, X. He and T. Lanigan-Atkins, Nat. Mater., 2021, 20, 977–983 CrossRef PubMed.
- O. Z. Delaire, G. Wolfgang, C. O. Naresh, G. Ehlers, D. L. Abernathy, D. Bansal, J. Ding and M. K. Gupta, Adv. Energy Mater., 2022, 12, 00596–00604 Search PubMed.
- G. Kresse and J. Furthmüller, Comput. Mater. Sci., 1996, 6, 15–50 CrossRef CAS.
- G. Kresse and J. Furthmüller, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 54, 11169–11186 CrossRef CAS PubMed.
- G. Kresse and D. Joubert, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 59, 1758–1775 CrossRef CAS.
- P. E. Blöchl, Phys. Rev. B: Condens. Matter Mater. Phys., 1994, 50, 17953–17979 CrossRef PubMed.
- J. P. Perdew, A. Ruzsinszky, G. I. Csonka, O. A. Vydrov, G. E. Scuseria, L. A. Constantin, X. Zhou and K. Burke, Phys. Rev. Lett., 2008, 100, 136406–136412 CrossRef PubMed.
- U.-G. Jong, C.-J. Yu, Y.-H. Kye, Y.-S. Kim, C.-H. Kim and S.-G. Ri, J. Mater. Chem. A, 2018, 6, 17994 RSC.
- U.-G. Jong, C.-J. Yu and Y.-H. Kye, RSC Adv., 2020, 10, 201–209 RSC.
- C.-J. Yu, U.-S. Hwang, Y.-C. Pak, K. Rim, C. Ryu, C.-R. Mun and U.-G. Jong, New J. Chem., 2020, 44, 21218–21227 RSC.
- T. Tadano, Y. Gohda and S. Tsuneyuki, J. Phys.: Condens. Matter, 2014, 26, 225402 CrossRef CAS PubMed.
- T. Tadano and S. Tsuneyuki, Phys. Rev. B: Condens. Matter Mater. Phys., 2015, 92, 054301 CrossRef.
- C.-J. Pak, U.-G. Jong, C.-J. Kang, Y.-S. Kim, Y.-H. Kye and C.-J. Yu, Mater. Adv., 2023, 4, 4528–4536 RSC.
- F. Zhou, W. Nielson, Y. Xia and V. Ozolins, Phys. Rev. Lett., 2014, 113, 185501 CrossRef PubMed.
- M. Simoncelli, N. Marzari and F. Mauri, Nat. Phys., 2019, 15, 809–816 Search PubMed.
- U.-G. Jong, C.-H. Ri, C.-J. Pak, C.-H. K. S. Cottenier and C.-J. Yu, New J. Chem., 2021, 45, 21569–21576 RSC.
- U.-G. Jong, C. Ryu, J.-M. Hwang, S.-H. Kim, I.-G. Ju and C.-J. Yu, Chem. Commun., 2024, 60, 13400–13403 RSC.
- U.-G. Jong, S.-H. Kim, R.-W. Ham, S. Ri, R.-J. Ri and C.-J. Yu, Appl. Phys. Lett., 2024, 125, 042203 CrossRef CAS.
- S. Sharma, J. K. Dewhurst and C. Ambrosch-Draxl, Phys. Rev. B: Condens. Matter Mater. Phys., 2003, 67, 165332 CrossRef.
- S. F. Pugh, Philos. Mag., 1954, 45, 823–843 Search PubMed.
- U.-G. Jong, Y.-S. Kim, C.-H. Ri, Y.-H. Kye, C.-J. Pak, S. Cottenier and C.-J. Yu, Chem. Commun., 2022, 58, 4223–4226 RSC.
- H. Xie, S. Hao, J. Bao, T. J. Slade, G. J. Snyder, C. Wolverton and M. G. Kanatzidis, J. Am. Chem. Soc., 2020, 142, 9553–9563 CrossRef CAS PubMed.
- E. J. Skoug, J. D. Cain and D. T. Morelli, Appl. Phys. Lett., 2010, 96, 181905 CrossRef.
- L. D. Zhao, S. H. Lo, Y. Zhang, H. Sun, G. Tan, C. Uher, C. Wolverton, V. P. Dravid and M. G. Kanatzidis, Nature, 2014, 508, 373–377 CrossRef CAS PubMed.
- M. Christensen, A. B. Abrahamsen, N. B. Christensen, F. Juranyi, N. H. Andersen, K. Lefmann, J. Andreasson, C. R. Bahl and B. B. Iversen, Nat. Mater., 2008, 7, 811–815 CrossRef CAS PubMed.
- U.-G. Jong, C.-J. Kang, S.-Y. Kim, H.-C. Kim and C.-J. Yu, Phys. Chem. Chem. Phys., 2022, 24, 5729–5737 Search PubMed.
- Y. Pei, J. Lensch-Falk, E. S. Toberer, D. L. Medlin and G. J. Snyder, Adv. Funct. Mater., 2011, 21, 241–249 Search PubMed.
- U.-G. Jong, C. Ryu, J.-M. Hwang, S.-H. Kim, I.-G. Ju and C.-J. Yu, Chem. Commun., 2024, 60, 13400–13403 RSC.
- A. F. Ioffe and A. R. Regel, Progress in Semiconductors, Heywood, London, 1960, vol. 4, pp. 237–291 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |