 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Studies on the structural, microstructure, dielectric, electrical and optical properties of Gd-doped BiFeO3 ceramic and its NTC thermistor application
Swati
Panda
and
S. K.
Parida
 *
*
Department of Physics, ITER, Siksha ‘O’ Anusandhan Deemed to be University, Bhubaneswar-751030, India. E-mail: santoshparida@soa.ac.in
First published on 4th February 2026
Abstract
This study presents a comprehensive investigation of Gd-doped BiFeO3 [Bi0.9Gd0.1FeO3, BGFO] ceramics synthesized via a conventional solid-state reaction method. The preliminary structural Rietveld analysis confirms the formation of a rhombohedral structure (#R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) c) with an average crystallite size of 65.5 nm, a micro-lattice strain of 0.00106, and a dislocation density of 2.33 × 1014 m−2. The analysis of the FTIR spectrum confirms the presence of all atomic vibration bands in the studied sample. The analysis of UV visible data shows a wide bandgap energy of 2.9 eV, which may be suitable for applications in photodetectors, LEDs, etc. The study of dielectric properties versus frequency and temperature reveals that the studied sample has a high dielectric constant and low loss, making the material a better candidate for energy storage devices. The study of impedance plots versus frequency supports the semiconducting nature of the sample. The study of modulus plots versus frequency confirms the non-Debye type of relaxation mechanism. The study of the ac conductivity versus both frequency and temperature confirms a thermally activated hopping mechanism. Both the Nyquist and Cole–Cole plots support the semiconducting nature of the sample. Additionally, the material exhibits negative temperature coefficient (NTC) thermistor behavior, confirming its potential for temperature sensor applications.
c) with an average crystallite size of 65.5 nm, a micro-lattice strain of 0.00106, and a dislocation density of 2.33 × 1014 m−2. The analysis of the FTIR spectrum confirms the presence of all atomic vibration bands in the studied sample. The analysis of UV visible data shows a wide bandgap energy of 2.9 eV, which may be suitable for applications in photodetectors, LEDs, etc. The study of dielectric properties versus frequency and temperature reveals that the studied sample has a high dielectric constant and low loss, making the material a better candidate for energy storage devices. The study of impedance plots versus frequency supports the semiconducting nature of the sample. The study of modulus plots versus frequency confirms the non-Debye type of relaxation mechanism. The study of the ac conductivity versus both frequency and temperature confirms a thermally activated hopping mechanism. Both the Nyquist and Cole–Cole plots support the semiconducting nature of the sample. Additionally, the material exhibits negative temperature coefficient (NTC) thermistor behavior, confirming its potential for temperature sensor applications.
1. Introduction
The performance of single perovskite materials in optoelectronic applications heavily depends on their structural stability and phase purity, which are critical for achieving consistent electronic and optical properties.1 Single perovskite materials offer tunable band gaps through compositional engineering, making them versatile for applications ranging from photovoltaics to light-emitting diodes.2 A single perovskite is a type of material that follows a specific crystal structure, originally found in the mineral calcium titanium oxide (CaTiO3). This structure is represented by the formula ABX3, where “A” is a larger positive ion, “B” is a smaller metal ion, and “X” is a negatively charged oxygen or halide like iodide, bromide, or chloride. Single perovskite materials are widely applied in optoelectronic devices such as solar cells, light-emitting diodes (LEDs), and photodetectors due to their tunable bandgap, and in energy storage devices due to their high dielectric constant and low loss, high absorption coefficients, and excellent charge transport properties.3–7Doping in single perovskite materials is important because it enables precise tuning of their electrical, magnetic, and structural properties to meet specific functional requirements. By introducing controlled impurities, researchers can enhance performance, stabilize phases, or induce new properties that are essential for advanced electronic and energy applications.8–13 BiFeO3 is one of the few single-phase multiferroic materials that exhibits both ferroelectricity and antiferromagnetism at room temperature, making it highly attractive for multifunctional devices.14,15 Pure BFO ceramics often suffer from high leakage current due to oxygen vacancies and volatile bismuth loss during sintering, which limits their practical ferroelectric applications.16,17 Doping BiFeO3 is essential because it enables tuning of its multiferroic behavior, which is vital for oxide memory technologies and other multifunctional devices. By introducing dopants, the magnetic and ferroelectric characteristics of the material can be adjusted to achieve targeted functionalities. Doping with rare-earth or transition metal ions and reducing grain size through nanostructuring significantly improves the electrical, magnetic, and structural stability of BiFeO3 ceramics.18,19
BiFeO3 is a room-temperature multiferroic material, but its practical use is limited due to high leakage current, secondary phases, and weak magnetism. Doping with rare-earth ions like Gd3+ (gadolinium) at the Bi-site has proven effective in enhancing its magnetoelectric performance and structural stability.20–24 Gd3+ (ionic radius ∼0.938 Å) has a smaller ionic size than Bi3+ (∼1.03 Å), introducing internal chemical pressure, reducing distortion, and enhancing magnetism and dielectric properties. Taking the problem of doping Gd in the bismuth site in the BiFeO3 ceramic host, the authors decided to explore the structural, dielectric, electric, and optical properties, along with searching for NTC thermistor applications.
2. Experimental details
2.1 Materials required
Bi0.9Gd0.1FeO3 nanoparticles were synthesized using a conventional solid-state reaction method. For the synthesis of this polycrystalline compound, raw materials Bi2O3, Fe2O3, and Gd2O3 of AR grade with 99% purity were procured from LOBA Chemie Pvt. Ltd. The required metal oxide powders were measured stoichiometrically using an electronic digital balance [MODEL: ML204/A01] with an accuracy of 0.0001 g. The required chemical equation for the synthesis of the final product through a stoichiometric ratio is given as: 0.45Bi2O3 + 0.05Gd2O3 + 0.5Fe2O3 → Bi0.9Gd0.1FeO3. This equation decides the quantity of the procured metal oxide required for the synthesis of the final product.2.2 Sample synthesis
The metal powders were thoroughly mixed using an agate mortar and pestle in a dry medium for 3 h, followed by wet mixing for another 3 h for better homogeneity. The mixture was then calcinated at a rate of 3 °C min−1 for 6 h at 850 °C. The XRD pattern of the calcinated sample was recorded to confirm the formation of a stable crystal structure. Subsequently, polyvinyl alcohol (PVA) was added to the calcinated powder to prepare cylindrical pellets with a diameter of 12 mm and a thickness of 2 mm using a KBr hydraulic press under a pressure of 4.0 × 106 N m−2. The pellets were sintered at 900 °C for 5 h in a muffle furnace to remove impurities of carbon and oxygen, if any, and also to make a dense material with better electrical properties.2.3 Characterization
Structural analysis was performed at room temperature using an X-ray diffractometer [MODEL: Rigaku Ultima IV, source = Cu Kα, step size = 0.02°] over a wide range of Bragg angles (20° ≤ θ ≤ 80°). Fourier transform infrared (PerkinElmer Model no. Spectrum Two Serial no. 105627 FT-IR) spectroscopy was used to record the FTIR spectrum of the prepared samples. To measure the bandgap energy and hence to investigate the semiconducting behavior of the proposed sample, the UV-vis spectroscopy analysis has been tailored. The UV-vis data were recorded using the MODEL: V670, being customized for source D2/WI and a bandwidth equal to 2 nm with a scan speed limit of 400 nm min−1. High-temperature dielectric data were recorded using an LCR analyzer [MODEL: N4L PSM 1735] over a frequency range of 1 kHz to 1 MHz and a temperature range of 25 °C to 500 °C.3. Results and discussion
3.1 Structure stability
To understand the stability of the Gd-doped BiFeO3 ceramics, the Goldschmidt tolerance factor (t) must be evaluated. The mathematical relation for the tolerance factor can be written as: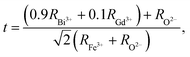 where all symbols have their usual meaning.25,26 The permitted value of t for a stable perovskite material should fall in the range 0.75 ≤ t ≤ 1.02.27 In the present study, the value of the tolerance factor of the BGFO ceramic is found to be 0.83, which supports the formation of a rhombohedral structure.
where all symbols have their usual meaning.25,26 The permitted value of t for a stable perovskite material should fall in the range 0.75 ≤ t ≤ 1.02.27 In the present study, the value of the tolerance factor of the BGFO ceramic is found to be 0.83, which supports the formation of a rhombohedral structure.
3.2 XRD analysis
XRD is essential for ceramic structural analysis as it identifies crystalline phases, determines the crystal structure, and assesses phase purity and lattice parameters.Here, Fig. 1(a) shows the XRD pattern of the BGFO sample at room temperature. The preliminary analysis of the title compound was performed by using a computer-generated analysis software called X'Pert High Score Plus.28 The XRD analysis shows that the sample has a rhombohedral (#R3c) crystal symmetry with cell parameters: a = b = 5.5876 Å, c = 13.8670 Å, α = β = 90°, γ = 120°, volume (V) = 374.94 Å3, and density = 8.31 g cm−3, respectively (#BiFeO3: JCPDS file no: 01-071-2494). Two weak secondary peaks of Gd2O3 are observed as shown in the XRD pattern [JCPDS file no: 00-012-0797 & 00-024-0430]. To understand the structural analysis better, Rietveld analysis was done, taking the BiFeO3 CIF file: 2101912 as the input file. Fig. 1(b) shows the Rietveld fitting plot of the BGFO ceramic. It is observed that all peaks are fitted well, except for two Gd2O3 peaks. The recorded fitting parameters are Rwp (%) = 12.3621, Rb (%) = 9.6011, Rexp (%) = 7.4035 and σ = 1.67. Therefore, the presence of the Gd signal in the fitting of the pure BeFeO3 ceramic confirms the formation of a solid solution of the Bi0.9Gd0.1FeO3 ceramic and the obtained rhombohedral (#R3c) crystal symmetry.22
 | ||
| Fig. 1 (a) XRD pattern and (b) Rietveld fitting plot of the BGFO ceramic. (c) W–H plots of the BGFO ceramic. | ||
To calculate the average crystallite size and micro-lattice strain, we have usedthe Williamson–Hall (W–H) method. The mathematical relation for the W–H method can be written as: 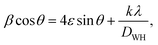 where all the symbols have their usual meaning.29–31Fig. 1(c) shows a 4ε
where all the symbols have their usual meaning.29–31Fig. 1(c) shows a 4ε![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sin
sin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ versus β
θ versus β![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos
cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ plot of the BGFO ceramic. We can evaluate the slope and y-intercept for the final value of the lattice strain and average crystallite size. In the present study, the micro-lattice strain is found to be 1.06 × 10−3 [error bar = 8.94 × 10−4], whereas the value of the average crystallite size (D) is found to be 65.5 nm [error bar = 1.17 × 10−3]. We can calculate the average dislocation density (δ) using the relation δ = 1/D2, which is found to be 2.33 × 1014 m−2.
θ plot of the BGFO ceramic. We can evaluate the slope and y-intercept for the final value of the lattice strain and average crystallite size. In the present study, the micro-lattice strain is found to be 1.06 × 10−3 [error bar = 8.94 × 10−4], whereas the value of the average crystallite size (D) is found to be 65.5 nm [error bar = 1.17 × 10−3]. We can calculate the average dislocation density (δ) using the relation δ = 1/D2, which is found to be 2.33 × 1014 m−2.
3.3 FTIR study
Fig. 2 shows the FTIR spectrum of the BGFO ceramic at room temperature. The fingerprint region of the metal oxygen bond shows the characteristic absorption bands in the wavenumber region 400 to 600 cm−1.32 Hussain et al. (2022) show that metal–oxide stretching modes (Bi–O and Fe–O) in pure BiFeO3 ceramics occur between 492 and 538 cm−1.33 The FTIR spectrum of pure BiFeO3 displays two prominent absorption bands in the 400–1000 cm−1 region: ∼527 cm−1, corresponding to Fe–O stretching vibrations in FeO6 octahedral units and ∼821 cm−1, attributed to higher-energy metal–oxygen vibrational modes associated with the perovskite R3c phase.In the present study, upon Gd3+ doping (e.g., x = 0.1), these bands shift slightly to ∼535 cm−1 and ∼827 cm−1, respectively, confirming successful lattice incorporation.34 Bi–O stretching vibrations typically appear in the low-frequency region near 440–460 cm−1. In the present study, Fe–O bands are broad or intense, and they overlap and mask the weak Bi–O peaks.34 Good doping levels are small; bands may be too weak to resolve. Gd–O (∼440–460 cm−1) coincides with strong Fe–O absorptions. Therefore, it can be concluded that the studied sample has all of the constituent metal-oxide vibration bands and complements the XRD analysis.
3.4 Optical property analysis
UV-Visible spectroscopy is essential for evaluating the optical absorption and bandgap energy of single perovskite materials, which directly influences their photovoltaic and optoelectronic performance. This technique helps identify transitions and defect states critical to optimizing device efficiency.35,36 The absorption peak originates from an electronic transition in the molecule: when the photon energy matches the energy separation between the ground and excited states, efficient absorption occurs, producing a peak in the spectrum. The position and intensity of the peak reflect the molecular structure and its environment.Fig. 3(a) illustrates the UV-visible absorption spectrum plotted as wavelength versus absorbance. The obtained optical data is analyzed using the Tauc relation, (αhν)n = A(hν − Eg), where α is the absorption coefficient, hν is the photon energy (eV), Eg is the optical bandgap energy, A is a constant, and the exponent n = 2 indicates a direct allowed transition.37 The development of energy bands arises due to interactions between the charged ions of half-filled d-block cations, facilitating electronic transitions from the filled oxygen orbitals to the partially filled d-orbitals of transition metal ions. The bandgap energy of pure BiFeO3 ceramic typically lies in the range of 2.1 to 2.8 eV, depending on factors such as synthesis method, grain size, and crystallinity.38–40Fig. 3(b) shows energy versus (αhν)2 of the BGFO ceramic. Extrapolation of the linear fit to the curve obtained between hν versus (αhν)2 provides a bandgap of 2.9 eV, which is well-matched with the results from Fig. 3(a) and (b) as shown. Gadolinium (Gd3+) doping in BiFeO3 can cause an increase in the bandgap energy (e.g., from ∼2.3 eV to ∼2.9 eV) due to two reasons: (i) Gd3+ ions (ionic radius ≈ 0.938 Å) substitute Bi3+ ions (≈ 1.03 Å), leading to lattice contraction and internal strain so that the Fe–O–Fe bond angles are reduced and the orbital overlap between Fe 3d and O 2p is modified, which affects the electronic band structure,41,42 and (ii) Gd3+ substitution may alter local electric fields and the Fe3+–O2− bonding environment, shifting energy levels upward or downward, which modifies the conduction/valence band positions, possibly increasing the energy gap.43
3.5 Dielectric properties
Dielectric studies are essential for ceramic materials because they provide critical insights into polarization mechanisms, electrical insulation behavior, and energy storage capabilities, which are vital for applications in capacitors, sensors, memory devices, and high-frequency electronics.44–47 To evaluate the dielectric constant (εr) and dielectric loss (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) of ceramic compounds, a mathematical relation is used: εr = Cdε0/A and tan
δ) of ceramic compounds, a mathematical relation is used: εr = Cdε0/A and tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ = ε″/ε′, where all symbols have their usual meaning.48Fig. 4(a) shows the variation of dielectric constant versus frequency, whereas Fig. 4(b) shows the variation of tan
δ = ε″/ε′, where all symbols have their usual meaning.48Fig. 4(a) shows the variation of dielectric constant versus frequency, whereas Fig. 4(b) shows the variation of tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ versus frequency of the BGFO ceramic at some selected temperatures. Polarization plays a key role in determining the dielectric properties of doped BiFeO3, as doping can modify the strength and orientation of spontaneous polarization within the crystal. When polarization is enhanced or better aligned due to dopant-induced structural changes, the material exhibits higher dielectric permittivity and improved charge-storage capability. At the same time, doping can suppress unwanted polarization-related defects such as oxygen vacancies and domain wall pinning, which helps reduce dielectric loss and stabilizes the dielectric response across different frequencies. Overall, the interaction between polarization and dopant-induced lattice distortions is crucial for optimizing the dielectric performance of BiFeO3-based ceramics.49–51 In Gd-doped BiFeO3 ceramics, the dielectric constant (ε′) typically decreases with increasing frequency, which is attributed to the Maxwell–Wagner interfacial polarization and dipolar relaxation. At lower frequencies, space charge and grain boundary effects contribute to high dielectric values, while at higher frequencies, dipoles fail to follow the rapidly changing electric field, leading to reduced permittivity.52
δ versus frequency of the BGFO ceramic at some selected temperatures. Polarization plays a key role in determining the dielectric properties of doped BiFeO3, as doping can modify the strength and orientation of spontaneous polarization within the crystal. When polarization is enhanced or better aligned due to dopant-induced structural changes, the material exhibits higher dielectric permittivity and improved charge-storage capability. At the same time, doping can suppress unwanted polarization-related defects such as oxygen vacancies and domain wall pinning, which helps reduce dielectric loss and stabilizes the dielectric response across different frequencies. Overall, the interaction between polarization and dopant-induced lattice distortions is crucial for optimizing the dielectric performance of BiFeO3-based ceramics.49–51 In Gd-doped BiFeO3 ceramics, the dielectric constant (ε′) typically decreases with increasing frequency, which is attributed to the Maxwell–Wagner interfacial polarization and dipolar relaxation. At lower frequencies, space charge and grain boundary effects contribute to high dielectric values, while at higher frequencies, dipoles fail to follow the rapidly changing electric field, leading to reduced permittivity.52
 | ||
Fig. 4 (a) The dielectric constant versus frequency, (b) tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ versus frequency, (c) dielectric constant versus temperature, and (d) tan δ versus frequency, (c) dielectric constant versus temperature, and (d) tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ versus temperature of the BGFO ceramic. δ versus temperature of the BGFO ceramic. | ||
Similarly, the tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ (dielectric loss) also decreases with frequency, indicating a reduction in energy dissipation due to delayed polarization processes. The high tan
δ (dielectric loss) also decreases with frequency, indicating a reduction in energy dissipation due to delayed polarization processes. The high tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ at low frequencies is generally linked to defect-induced hopping conduction (e.g., Fe3+/Fe2+), while its reduction at higher frequencies shows that the dielectric loss becomes minimal.53Fig. 4(c) shows the variation of dielectric constant versus temperature, whereas Fig. 4(d) shows the variation of tan
δ at low frequencies is generally linked to defect-induced hopping conduction (e.g., Fe3+/Fe2+), while its reduction at higher frequencies shows that the dielectric loss becomes minimal.53Fig. 4(c) shows the variation of dielectric constant versus temperature, whereas Fig. 4(d) shows the variation of tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ versus temperature of the BGFO ceramic at some selected frequencies. In Gd-doped BiFeO3 ceramics, the dielectric constant (ε′) increases with rising temperature, especially near the ferroelectric–paraelectric transition temperature of 390 °C, due to enhanced thermal activation of dipoles and increased space charge polarization. This behavior is typical for ferroelectric materials, where higher thermal energy allows dipoles to reorient more easily, leading to an increase in permittivity.54,55 The tan
δ versus temperature of the BGFO ceramic at some selected frequencies. In Gd-doped BiFeO3 ceramics, the dielectric constant (ε′) increases with rising temperature, especially near the ferroelectric–paraelectric transition temperature of 390 °C, due to enhanced thermal activation of dipoles and increased space charge polarization. This behavior is typical for ferroelectric materials, where higher thermal energy allows dipoles to reorient more easily, leading to an increase in permittivity.54,55 The tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ also increases with temperature, mainly because of the enhanced mobility of charge carriers and leakage current at higher temperatures. A distinct peak in tan
δ also increases with temperature, mainly because of the enhanced mobility of charge carriers and leakage current at higher temperatures. A distinct peak in tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ may appear near 290 °C, indicating a relaxation process.56,57 The shift of the relaxation peak in tan
δ may appear near 290 °C, indicating a relaxation process.56,57 The shift of the relaxation peak in tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ with increasing temperature indicates a thermally activated relaxation process, where dipoles or charge carriers require less energy to reorient at higher temperatures, causing the loss peak to move toward lower frequencies or higher temperatures, depending on the relaxation mechanism.
δ with increasing temperature indicates a thermally activated relaxation process, where dipoles or charge carriers require less energy to reorient at higher temperatures, causing the loss peak to move toward lower frequencies or higher temperatures, depending on the relaxation mechanism.
3.6 Impedance analysis
Impedance spectroscopy is essential for BiFeO3 ceramics as it provides critical insights into their electrical conduction mechanisms, grain and grain boundary contributions, and dielectric relaxation behavior, which are key for optimizing their multiferroic and functional properties. Fig. 5(a) and (b) show the variation of real (Z′) and imaginary (Z″) components of impedance with frequency. Z′ shows a decreasing trend with frequency, indicating enhanced conductivity due to reduced grain boundary resistance at higher frequencies. It supports the semiconducting nature of the sample under study.58 | ||
| Fig. 5 (a) The Z′ versus frequency, (b) Z″ versus frequency, and (c) Z′ versus Z″ of the BGFO ceramic. | ||
The Z″ versus frequency plots for BGFO exhibit one or more characteristic peaks, whose positions shift toward higher frequencies as temperature increases, indicating thermally activated relaxation processes.59Fig. 5(c) shows the graph of Z′ versus Z″ of the BGFO ceramic at some selected temperatures. The radius of the semicircular arcs decreases with the rise of temperature, supporting the semiconducting maturity of the sample.60
3.7 Modulus analysis
Modulus study is required for ceramics to analyze dielectric relaxation and distinguish between electrode effects and bulk material responses, offering deeper insight into charge carrier dynamics and localized conduction processes. The complex electric modulus M* is related to the complex permittivity ε* by: M* = 1/ε* = M′ + iM″, where ε* = ε′ − iε‴, M′ = ε′/[(ε′)2 + (ε″)2], M″ = ε″/[(ε′)2 + (ε″)2], M′ = real part of the modulus (storage modulus) and M″ = imaginary part (loss modulus), indicating relaxation processes.61,62Fig. 6(a) and (b) show the variation of M′ with frequency and M″ versus frequency at some selected temperatures. At lower frequencies, the M′ values are nearly zero and coincide across compositions, indicating negligible electrode polarization; as frequency increases, M′ gradually rises and plateaus at high frequency, reflecting the transition from long-range conduction to localized charge relaxation and confirming non-Debye–type behavior in the material.63 In the present study, the imaginary modulus M″ exhibits broad peaks whose relaxation frequency (fn) shifts to higher frequencies with both increasing temperature and 10% Gd content. These peaks are asymmetric and broadened, indicating a distribution of relaxation times and non-Debye-type relaxation behavior, typically associated with localized hopping of charge carriers within grains or across grain boundaries.64Cole–Cole plots help identify and distinguish grain and grain boundary responses through separate semicircular arcs, revealing resistive and capacitive behavior of different microstructural regions in polycrystalline BGFO ceramics. Fig. 6(c) shows the Cole–Cole plots of the BGFO ceramic at some selected temperatures. The presence of the semicircular arcs in the Cole–Cole plots confirms the semiconducting nature.65
3.8 AC conductivity study
The study of ac conductivity in Gd-doped BiFeO3 ceramics is essential to understand the frequency-dependent charge transport mechanisms and assess the influence of Gd doping on electrical conduction behavior.The ac conductivity can be calculated from the dielectric data as: σac(ω) = ε0ω![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) tan
tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ, where ε0 = permittivity of free space (8.854 × 10−12 F m−1), tan
δ, where ε0 = permittivity of free space (8.854 × 10−12 F m−1), tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ = dielectric loss, and ω = 2πf = angular frequency.66Fig. 7(a) shows the ac conductivity versus frequency at some selected temperatures. At low frequencies, σac remains nearly frequency-independent, forming a plateau corresponding to σdc (dc conductivity), a regime dominated by long-range charge transport across grains. As frequency increases, σac begins to rise following Jonscher's universal power law, i.e., σac = σdc + Aωn, where exponent n (0 < n < 1) and pre-exponential factor A reveal a hopping conduction mechanism involving small polarons or localized charge carriers.67 As the temperature rises or when Gd concentration reaches 10%, the dispersion begins at lower frequencies and the slope parameter (n) decreases, suggesting improved charge carrier mobility and a lower activation energy for hopping conduction. Fig. 7(b) shows ac conductivity versus 1000/T at some selected frequencies. Temperature-dependent ac conductivity obeys the Arrhenius relationship:
δ = dielectric loss, and ω = 2πf = angular frequency.66Fig. 7(a) shows the ac conductivity versus frequency at some selected temperatures. At low frequencies, σac remains nearly frequency-independent, forming a plateau corresponding to σdc (dc conductivity), a regime dominated by long-range charge transport across grains. As frequency increases, σac begins to rise following Jonscher's universal power law, i.e., σac = σdc + Aωn, where exponent n (0 < n < 1) and pre-exponential factor A reveal a hopping conduction mechanism involving small polarons or localized charge carriers.67 As the temperature rises or when Gd concentration reaches 10%, the dispersion begins at lower frequencies and the slope parameter (n) decreases, suggesting improved charge carrier mobility and a lower activation energy for hopping conduction. Fig. 7(b) shows ac conductivity versus 1000/T at some selected frequencies. Temperature-dependent ac conductivity obeys the Arrhenius relationship: 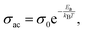 where Ea = activation energy, and kB = Boltzmann's constant = 1.38 × 1023 J K−1.68 The activation energy values are calculated from the slope of ln(σ) vs. 1/T plots. The calculated values of the activation energy are 0.511 eV, 0.384 eV, 0.257 eV, 0.186 eV, 0.167 eV, 0.158 eV, and 0.151 eV at 1 kHz, 10 kHz, 100 kHz, 300 kHz, 500 kHz, 700 kHz, and 1 MHz, respectively. The gradual decrease in activation energy with increasing frequency suggests that charge carriers require less energy to hop at higher frequencies, supporting a thermally activated conduction process.69
where Ea = activation energy, and kB = Boltzmann's constant = 1.38 × 1023 J K−1.68 The activation energy values are calculated from the slope of ln(σ) vs. 1/T plots. The calculated values of the activation energy are 0.511 eV, 0.384 eV, 0.257 eV, 0.186 eV, 0.167 eV, 0.158 eV, and 0.151 eV at 1 kHz, 10 kHz, 100 kHz, 300 kHz, 500 kHz, 700 kHz, and 1 MHz, respectively. The gradual decrease in activation energy with increasing frequency suggests that charge carriers require less energy to hop at higher frequencies, supporting a thermally activated conduction process.69
 | ||
| Fig. 7 (a) and (b) The ac conductivity versus frequency and the ac conductivity versus 1000/T of the BGFO ceramic. | ||
3.9 Thermistor behaviour
Thermistor studies in ceramics are essential to evaluate their temperature-dependent resistive behavior, enabling their application in temperature sensing and thermal protection devices. Fig. 8(a) shows the variation of resistance versus temperature, while Fig. 8(b) shows the variation of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) R versus 1/T of the BGFO ceramic. Thermistive materials fall into two primary categories: NTCR, where resistance decreases with rising temperature, and PTCR, where the opposite occurs. In the present study, the nature of NTCR and PTCR of BGFO was analyzed in depth. Resistance data were extracted from Nyquist plots generated via complex impedance spectroscopy, across a temperature range from 300 K to 800 K in steps of 25 K. Plotting these resistance values against temperature revealed a consistent downward trend from 300 to 500 °C, affirming the NTC thermistor character of the sample.70 Additionally, a graph of ln(R) versus 1/T further confirmed this thermally activated semiconducting behavior, an indication that increased thermal energy facilitates carrier excitation and movement, thereby reducing resistance.71Fig. 8(c) shows the variation of the thermistor constant versus temperature. To quantify this thermistor behavior, the thermistor constant (β) was calculated using resistance values at two adjacent temperatures, based on the following empirical relation:
R versus 1/T of the BGFO ceramic. Thermistive materials fall into two primary categories: NTCR, where resistance decreases with rising temperature, and PTCR, where the opposite occurs. In the present study, the nature of NTCR and PTCR of BGFO was analyzed in depth. Resistance data were extracted from Nyquist plots generated via complex impedance spectroscopy, across a temperature range from 300 K to 800 K in steps of 25 K. Plotting these resistance values against temperature revealed a consistent downward trend from 300 to 500 °C, affirming the NTC thermistor character of the sample.70 Additionally, a graph of ln(R) versus 1/T further confirmed this thermally activated semiconducting behavior, an indication that increased thermal energy facilitates carrier excitation and movement, thereby reducing resistance.71Fig. 8(c) shows the variation of the thermistor constant versus temperature. To quantify this thermistor behavior, the thermistor constant (β) was calculated using resistance values at two adjacent temperatures, based on the following empirical relation: 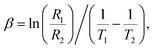 where R1 and R2 are the resistances at absolute temperatures T1 and T2, respectively.72
where R1 and R2 are the resistances at absolute temperatures T1 and T2, respectively.72
This parameter provides insight into how sharply resistance changes with temperature. The calculated β values ranged from 2334 K to 4406 K, depending on the specific temperature interval. According to established criteria, β values between 2000 K and 5000 K are typically associated with low-temperature thermistors, while much higher values are characteristic of high-temperature sensing materials. Therefore, the β values observed for BGFO suggest strong potential for high-temperature thermistor applications.
Another significant performance metric is the sensitivity factor (α), which measures the relative rate of resistance change with temperature. It is calculated using: 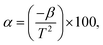 where α is the sensitivity in percentage per Kelvin, β is the thermistor constant, and T is the absolute temperature in Kelvin.73 This relationship shows that sensitivity increases with higher β and decreases with rising temperature. The plot in Fig. 8(d) shows an inverse nonlinear relationship, indicating that sensitivity gradually declines as temperature increases, a hallmark of NTC thermistor behavior. This trend reflects reduced responsiveness at elevated temperatures, which aligns with theoretical expectations.
where α is the sensitivity in percentage per Kelvin, β is the thermistor constant, and T is the absolute temperature in Kelvin.73 This relationship shows that sensitivity increases with higher β and decreases with rising temperature. The plot in Fig. 8(d) shows an inverse nonlinear relationship, indicating that sensitivity gradually declines as temperature increases, a hallmark of NTC thermistor behavior. This trend reflects reduced responsiveness at elevated temperatures, which aligns with theoretical expectations.
The observed thermal behavior can be attributed to the crystal structure of BGFO, particularly the octahedral coordination of transition metal cations at the B-site with surrounding oxygen anions. These structural features facilitate charge transfer through partially filled d-orbitals, enhancing mobility and improving thermistor efficiency. Finally, the activation energy (Ea) corresponding to the thermally driven conduction was calculated from the thermistor constant using: Ea = kB × β, where Ea is the activation energy in eV, β is the thermistor constant, and kB is Boltzmann's constant (8.617 × 10−5 eV K−1).74,75 The activation energy values, plotted as a function of temperature, mirrored the trend of β, consistent with their direct proportional relationship as shown in Fig. 8(e). These moderate activation energies support the presence of easily excitable charge carriers, further confirming the semiconducting and NTC thermistor characteristics in the high temperature range.
4. Conclusion
In this communication, a comprehensive study on the structural, dielectric, electrical, and optical properties of the Gd-doped BiFeO3 ceramic is presented. The preliminary structural analysis by MADU Rietveld refinement confirms that the BGFO ceramic has a rhombohedral structure with an average crystallite size of 65.5 nm, micro-lattice strain of 0.00106, and dislocation density of 2.33 × 1014 m−2. The analysis of the FTIR spectrum confirms the presence of a wide and strongly intense vibration band of Fe–O at 535 cm−1, and two other weak bands of Bi–O and Gd–O are overlapped with the Fe–O stretching band. So, our prepared sample has all constituent elements and this is supported by the results of the XRD analysis. The analysis of the UV-visible data provides the bandgap energy of 2.9 eV, supporting the application for photodetectors and light-detecting devices. A wide range of dispersed values of dielectric constant and loss are observed at low frequency due to the strong effect of dipolar and space charge polarization. Interestingly, with the rise of frequency, both the dielectric constant and loss decrease due to the reduction of space charge polarization. The study of impedance plots versus frequency confirms the presence of semiconducting and relaxation behaviour. Again, the study of modulus plots with frequency confirms a non-Debye type of relaxation mechanism in the studied sample. AC conductivity studies suggest a thermally activated hopping process that controls the whole conductivity mechanism in the sample. The study of resistance versus temperature plots confirms the semiconducting nature, and analysis of the thermistor coefficient versus temperature confirms the NTC thermistor character at high temperature.Author contributions
Swati Panda: data curation, writing – original draft, conceptualization, and methodology, and S. K. Parida: validation, software, writing – reviewing and editing, visualization, and supervision.Conflicts of interest
There are no conflicts of interest to declare.Data availability
Data will be made available upon reasonable request.Acknowledgements
There is no funding for the preparation of the presented work. The authors would like to thank the host Institute for the XRD characterization.References
- J. H. Noh, S. H. Im, J. H. Heo, T. N. Mandal and S. I. Seok, Chemical management for colorful, efficient, and stable inorganic–organic hybrid nanostructured solar cells, Nano Lett., 2013, 13, 1764–1769 CrossRef CAS PubMed.
- M. Saliba, T. Matsui, J. Y. Seo, K. Domanski, J. P. Correa-Baena, M. K. Nazeeruddin and M. Grätzel, Cesium-containing triple cation perovskite solar cells: improved stability, reproducibility, and high efficiency, Energy Environ. Sci., 2016, 9, 1989–1997 RSC.
- A. Kojima, K. Teshima, Y. Shirai and T. Miyasaka, Organometal halide perovskites as visible-light sensitizers for photovoltaic cells, J. Am. Chem. Soc., 2009, 131, 6050–6051 CrossRef CAS PubMed.
- S. D. Stranks and H. J. Snaith, Metal–halide perovskites for photovoltaic and light-emitting devices, Nat. Nanotechnol., 2015, 10, 391–402 Search PubMed.
- Z. K. Tan, R. S. Moghaddam, M. L. Lai, P. Higler, R. Docampo, F. Deschler and R. H. Friend, Bright light-emitting diodes based on organometal halide perovskite, Nat. Nanotechnol., 2014, 9, 687–692 CrossRef CAS PubMed.
- L. Dou, Y. M. Yang, J. You, Z. Hong, W. H. Chang, G. Li and Y. Yang, Solution-processed hybrid perovskite photodetectors with high detectivity, Nat. Commun., 2014, 5, 5404 CrossRef CAS PubMed.
- M. Saliba, T. Matsui, J. Y. Seo, K. Domanski, J. P. Correa-Baena, M. K. Nazeeruddin and M. Grätzel, Cesium-containing triple cation perovskite solar cells: improved stability, reproducibility, and high efficiency, Energy Environ. Sci., 2016, 9, 1989–1997 RSC.
- S. Supriya, Recent trends and morphology mechanisms of rare-earth based BiFeO3 nano perovskites with excellent photocatalytic performances, J. Rare Earths, 2023, 41, 331–341 Search PubMed.
- D. Dash, A. Palai and D. Sahu, Nanocrystalline gadolinium doped ZnO: an excellent photoluminescent material and efficient photocatalyst towards optoelectronic and environment remedial applications, Ceram. Int., 2022, 48, 28835–28842 CrossRef CAS.
- R. Balakrishnan, A. Dixit, R. Naik and M. S. R. Rao, Enhancement in electrical and magnetodielectric properties of Ca- and Ba-doped BiFeO3 polycrystalline ceramics, J. Am. Ceram. Soc., 2018, 101, 782–788 CrossRef CAS.
- A. Palai, N. Ranjan Panda, S. Chhotaray and D. Sahu, Sm doped ZnO-SnO2 nanocomposites with improved photo-degradation strength and antibacterial properties, Surf. Interfaces, 2023, 41, 103217 CrossRef CAS.
- D. Sahu, N. R. Panda and B. S. Acharya, Effect of Gd doping on structure and photoluminescence properties of ZnO nanocrystals, Mater. Res. Express, 2017, 4, 114001 Search PubMed.
- P. Banerjee and A. Franco Júnior, Rare earth and transition metal doped BiFeO3 ceramics: structural, magnetic and dielectric characterization, J. Mater. Sci.: Mater. Electron., 2016, 27, 6053–6059 CrossRef CAS.
- J. Wang, J. B. Neaton, H. Zheng, V. Nagarajan, S. B. Ogale, B. Liu and R. Ramesh, Epitaxial BiFeO3 multiferroic thin film heterostructures, Science, 2003, 299, 1719–1722 CrossRef CAS PubMed.
- Q. Wu, Y. Song, C. Jia, Z. Gao and W. Zhang, Effect of Nd and Mn Co-Doping on Dielectric, Ferroelectric and Photovoltaic Properties of BiFeO3, Crystals, 2022, 12, 500 CrossRef CAS.
- T. J. Park, G. C. Papaefthymiou, A. J. Viescas, A. R. Moodenbaugh and S. S. Wong, Size-dependent magnetic properties of single-crystalline multiferroic BiFeO3 nanoparticles, Nano Lett., 2007, 7, 766–772 CrossRef CAS.
- M. Kumar and K. L. Yadav, Large dielectric and weak ferromagnetic behavior of BiFeO3 ceramics synthesized using Fe2O3 nanopowder, J. Phys. D: Appl. Phys., 2007, 40, 5317–5320 Search PubMed.
- A. Singh, B. C. Yadav and R. S. Tiwari, Enhancement of multiferroic properties in rare-earth substituted BiFeO3, J. Alloys Compd., 2014, 615, 877–884 CrossRef.
- S. K. Singh, R. S. Katiyar and J. F. Scott, Optical properties and Raman scattering of multiferroic BiFeO3 thin films, J. Appl. Phys., 2008, 103, 074103 CrossRef.
- A. Singh, B. C. Yadav and R. S. Tiwari, Structural, dielectric, and magnetic properties of Gd-doped BiFeO3 multiferroic ceramics, J. Alloys Compd., 2014, 615, 877–884 CrossRef.
- V. A. Khomchenko, D. A. Kiselev, J. M. Vieira, A. L. Kholkin and P. M. Vilarinho, Effect of rare-earth substitution on the structure and multiferroic properties of BiFeO3 ceramics, J. Appl. Phys., 2008, 103, 024105 CrossRef.
- D. K. Pradhan, P. R. Das, R. N. P. Choudhary and S. Das, Dielectric and magnetic properties of Gd-modified BiFeO3 multiferroic ceramics, Mater. Chem. Phys., 2013, 139, 609–615 CrossRef.
- D. Kothari, G. Sharma and R. K. Kotnala, Enhanced magnetic and dielectric properties of Gd-doped BiFeO3 nanoparticles, Mater. Res. Bull., 2011, 46, 1837–1842 CrossRef.
- S. K. Mendiratta, R. Kumar and R. K. Dwivedi, Improved dielectric and ferroelectric behavior of Gd-doped BiFeO3 ceramics, J. Mater. Sci.: Mater. Electron., 2016, 27, 7392–7400 Search PubMed.
- V. M. Goldschmidt, Die gesetze der krystallochemie, Naturwissenschaften, 1926, 14, 477–485 CrossRef CAS.
- R. D. Shannon, Revised Effective Ionic Radii and Systematic Studies of Interatomic Distances in Halides and Chalcogenides, Acta Crystallogr., Sect. A, 1976, 32, 751–767 CrossRef.
- C. J. Bartel, C. Sutton, B. R. Goldsmith, R. Ouyang, C. B. Musgrave, L. M. Ghiringhelli and M. Scheffler, New tolerance factor to predict the stability of perovskite oxides and halides, Sci. Adv., 2019, 5, eaav0693 CrossRef CAS PubMed.
- M. Padhi, S. Mishra, A. Prasad Sahu and S. K. Parida, A comprehensive study on the structure, microstructure, dielectric, electrical, and infrared properties of the BiNaCoMoO6 ceramic for NTC thermistor applications, J. Mater. Sci.: Mater. Electron., 2025, 36, 1966 CrossRef CAS.
- G. K. Williamson and W. H. Hall, X-ray line broadening from fielded aluminum and wolfram, Acta Metall., 1953, 1, 22–31 CrossRef CAS.
- S. Mishra and S. K. Parida, Electrical and optical properties of a lead-free complex double perovskite BaNaFeMoO6: photovoltaic and thermistor applications, Mater. Sci. Eng., B, 2023, 296, 116629–116645 CrossRef CAS.
- M. G. Hajiabadi, M. Zamanian and D. Souri, Williamson-Hall analysis in evaluation of lattice strain and the density of lattice dislocation for nanometer-scaled ZnSe and ZnSe:Cu particles, Ceram. Int., 2019, 45, 14084–14089 CrossRef.
- A. Hezam, K. Namratha, Q. A. Drmosh, Z. H. Yamani and K. Byrappa, Synthesis of Heterostructured Bi2O3–CeO2–ZnO Photocatalyst with Enhanced Sunlight Photocatalytic Activity, Ceram. Int., 2017, 43, 5292–5301 CrossRef CAS.
- G. Hussain, I. Ahmed, A. U. Rehman, M. U. Subhani, N. Morley and M. Akhtar, et al., Study of the role of dysprosium substitution in tuning structural, optical, electrical, dielectric, ferroelectric, and magnetic properties of bismuth ferrite multiferroic, J. Alloys Compd., 2022, 919, 165743 CrossRef CAS.
- S. R. Dhanya, S. G. Nair, J. Satapathy and N. Pavan Kumar, Structural and spectroscopic characterization of bismuth-ferrites, AIP Conf. Proc., 2019, 2166, 020017 CrossRef CAS.
- J. H. Heo, et al., Efficient inorganic–organic hybrid heterojunction solar cells containing perovskite compound and polymeric hole conductors, Nat. Photonics, 2013, 7, 486–491 CrossRef CAS.
- G. Xing, et al., Long-range balanced electron- and hole-transport lengths in organic-inorganic CH3NH3PbI3, Science, 2013, 342, 344–347 CrossRef CAS PubMed.
- J. Tauc, R. Grigorovici and A. Vancu, Optical properties and electronic structure of amorphous germanium, Phys. Status Solidi B, 1966, 15, 627–637 CrossRef CAS.
- G. Catalan and J. F. Scott, Physics and applications of bismuth ferrite, Adv. Mater., 2009, 21, 2463–2485 CrossRef CAS.
- S. M. Selbach, et al., Band gap and electronic structure of BiFeO3 from hybrid functional and O 1s X-ray absorption spectroscopy, J. Solid State Chem., 2012, 196, 249–254 CrossRef CAS.
- R. Mazumder, et al., Structural, optical and magnetic properties of sol–gel synthesized BiFeO3 ceramics, Ceram. Int., 2014, 40, 10459–10466 Search PubMed.
- M. Selvakumar, et al., Influence of rare earth doping on structural, optical and magnetic properties of BiFeO3 nanoparticles, J. Alloys Compd., 2011, 509, 9800–9806 Search PubMed.
- A. Sharma, et al., Influence of Gd doping on structure, bandgap, and magnetic properties of BiFeO3 ceramics, Ceram. Int., 2016, 42, 13847–13854 Search PubMed.
- Q. Liu, et al., Enhanced multiferroic properties and bandgap tuning in Gd-doped BiFeO3 ceramics, J. Mater. Sci.: Mater. Electron., 2016, 27, 10758–10765 Search PubMed.
- M. M. Kumar, A. Srinivas and S. V. Suryanarayana, Structure–property relation in Nd-substituted BiFeO3, J. Appl. Phys., 2000, 87, 855–862 Search PubMed.
- S. Ghosh and R. N. P. Choudhary, Impedance and dielectric studies of BiFeO3–PbTiO3 ceramics, Mater. Chem. Phys., 2009, 114, 44–49 Search PubMed.
- P. Lunkenheimer, et al., Origin of apparent colossal dielectric constants, Phys. Rev. B: Condens. Matter Mater. Phys., 2002, 66, 052105 Search PubMed.
- S. Suthar, et al., Dielectric and impedance analysis of rare earth doped BiFeO3 ceramics, Ceram. Int., 2016, 42, 12529–12536 Search PubMed.
- B. Kumar Vashisth, J. S. Bangruwa, S. P. Gairolaa and V. Verma, Structural, dielectric, ferroelectric, and magnetic properties of doped BiFeO3, Integr. Ferroelectr., 2018, 194, 21–27 Search PubMed.
- N. Ranjan Panda, S. K. Sahu, A. Palai, T. Yadav, D. Behera and D. Sahu, Unraveling the synergistic effects in ZnO–MoS2 nanocomposite leading to enhanced photocatalytic, antibacterial, and dielectric characteristics, Chem. Phys. Impact, 2024, 8, 100550 Search PubMed.
- D. Sahu and N. Ranjan Panda, Synthesis of novel nanocomposite of g-C3N4 coated ZnO–MoS2 for energy storage and photocatalytic applications, Chemosphere, 2024, 350, 141014 CrossRef CAS PubMed.
- Y. Saad, I. Álvarez-Serrano, M. L. López and M. Hidouri, Dielectric response and thermistor behavior of lead-free xNaNbO3 − (1 − x)BiFeO3 electroceramics, Ceram. Int., 2018, 44, 18560–18570 CrossRef CAS.
- S. Ghosh, et al., Dielectric relaxation and AC conductivity in Gd-modified BiFeO3 ceramics, J. Alloys Compd., 2011, 509, 7584–7592 Search PubMed.
- A. K. Jonscher, The ‘universal’ dielectric response, Nature, 1977, 267, 673–679 CrossRef CAS.
- S. Suthar, Y. Sharma and N. Ahlawat, Dielectric and impedance analysis of rare earth doped BiFeO3 ceramics, Ceram. Int., 2016, 42, 12529–12536 Search PubMed.
- M. K. Singh, R. S. Katiyar and J. F. Scott, New magnetic phase transitions in BiFeO3, J. Phys.: Condens. Matter, 2008, 20, 252203 Search PubMed.
- M. M. Kumar, A. Srinivas and S. V. Suryanarayana, Structure–property relation in Nd-substituted BiFeO3, J. Appl. Phys., 2000, 87, 855–862 CrossRef CAS.
- K. L. Yadav, Dielectric and magnetic behavior of multiferroic BiFeO3 ceramics, J. Phys. D: Appl. Phys., 2008, 41, 135005 CrossRef.
- A. Khanna, et al., Structural, dielectric and magnetic properties of Gd-doped BiFeO3 ceramics, Mater. Res. Bull., 2013, 48, 3907–3913 Search PubMed.
- S. K. Parida, P. K. Das and R. N. P. Choudhary, Structural and Electrical Characterization of SrMn0.97Ce0.03O3 Ceramics, Integr. Ferroelectr., 2021, 221, 215–230 CrossRef CAS.
- S. Mishra and S. K. Parida, Lead-free complex double perovskite SrLiFeWO6: structural, microstructure, electrical and optical study, Phys. B, 2023, 668, 415246 CrossRef CAS.
- T. Md Rahman, M. Vargas and C. V. Ramana, structural characteristics, electrical conduction, and dielectric properties of gadolinium-substituted cobalt ferrite, J. Alloys Compd., 2014, 617, 547–562 CrossRef.
- Y. D. Kolekar, et al., Impedance spectroscopy and modulus studies of BiFeO3 ceramics, J. Alloys Compd., 2011, 509, 942–948 CrossRef.
- S. Kumar, et al., Effect of Nd Doping on Structural, Electrical and Optical Properties of BiFeO3 Ceramics, Ceram. Int., 2017, 43, 14859–14867 Search PubMed.
- S. Pattanayak, B. N. Parida, P. R. Das and R. N. P. Choudhary, Impedance spectroscopy of Gd-doped BiFeO3 multiferroics, Appl. Phys. A, 2013, 112, 387–395 CrossRef CAS.
- S. Mishra, R. N. P. Choudhary and S. K. Parida, Structural, dielectric, electrical, and optical properties of a double perovskite: BaNaFeWO6 for some device applications, J. Mol. Struct., 2022, 1265, 133353 CrossRef CAS.
- S. Mishra and S. K. Parida, Investigation of structure, dielectric, thermistor, and optical properties study of an eco-friendly double perovskite for photovoltaic and optoelectronic applications: CaLiFeWO6, Curr. Appl. Phys., 2024, 65, 75–90 CrossRef.
- R. Das and R. N. P. Choudhary, Studies of structural, dielectric relaxation and impedance spectroscopy of lead-free double perovskite: Dy2NiMnO6, J. Mater. Sci.: Mater. Electron., 2018, 29, 19099–19110 CrossRef CAS.
- A. Goswami and P. K. Mahapatra, Impedance spectroscopy and Cole–Cole analysis of dielectric properties in lead-based ceramic materials, Ceram. Int., 2014, 40, 7291–7300 CrossRef.
- R. Kumar and S. B. Rai, Electrical and Dielectric Properties of Ferrite Ceramics Through Nyquist Plot Interpretation, J. Mater. Sci.: Mater. Electron., 2017, 28, 7893–7900 Search PubMed.
- S. Mishra and S. K. Parida, Lead-free complex double perovskite SrLiFeWO6: structural, microstructure, electrical and optical study, Phys. B, 2023, 668, 415246 Search PubMed.
- S. Mishra and S. K. Parida, Electrical and optical properties of a lead-free complex double perovskite BaNaFeMoO6: photovoltaic and thermistor applications, Mater. Sci. Eng., B, 2023, 296, 116629–116645 CrossRef CAS.
- S. Sahoo, Enhanced time response and temperature sensing behavior of thermistor using Zn-doped CaTiO3 nanoparticles, J. Adv. Ceram., 2018, 7, 99–108 CrossRef CAS.
- D. Saha, A. Das Sharma, A. Sen and H. S. Maiti, Preparation of bixbyite phase (MnxFe1−x)2O3 for NTC thermistor applications, Mater. Lett., 2002, 55, 403–406 CrossRef CAS.
- F. C. S. Luz, S. A. Pianaro, C. E. Yurk, G. Capobianco, A. J. Zara and S. M. Tebcherani, Construction and testing of a system for the electrical characterization of ceramic thermistors at low temperatures, Ceramica, 2014, 60, 96–101 CrossRef CAS.
- A. Feteira, Negative Temperature Coefficient Resistance (NTCR) Ceramic Thermistors: An Industrial Perspective, J. Am. Ceram. Soc., 2009, 92, 967–983 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |




