Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Toward sustainable SLA 3D printing: glycerol-based 1,3-diether-2-methacrylates with solvent capability for in situ polymer blend formation
Zahra
Sekhavat Pour
 ,
Pravin S.
Shinde
,
Pravin S.
Shinde
 ,
Jun
Wang
,
Mustapha
Iddrisu
,
Tibor
Szilvási
,
Jun
Wang
,
Mustapha
Iddrisu
,
Tibor
Szilvási
 and
Jason E.
Bara
and
Jason E.
Bara
 *
*
Department of Chemical & Biological Engineering, The University of Alabama, Tuscaloosa, AL, 35487-0203 USA. E-mail: jbara@eng.ua.edu
First published on 20th February 2026
Abstract
Development of sustainable and environmentally friendly stereolithography (SLA) 3D printing resins has gained considerable attention in recent years. Towards this goal, we have successfully employed glycerol-based 1,3-diether-2-methacrylate monomers that offer tunable mechanical and thermal properties. These monomers also act as solvents for thermoplastics such as polystyrene (PS) and poly(methyl-methacrylate) PMMA. This dual functionality enables the direct fabrication of polymer blends using only a consumer-level SLA 3D printer. The resulting blends exhibit composition-dependent phase morphologies that significantly influence mechanical properties. For instance, incorporating 5 wt% waste PS (wPS) results in complete phase separation with a stratified morphology in each layer and substantially diminished elongation at break, whereas 10 wt% wPS led to a more uniform dispersion and improved mechanical behavior. In contrast, PMMA-containing systems provide more transparent blends and continuous matrix morphology without compromising elasticity. Thermal analysis revealed the presence of two distinct glass transition temperatures (Tg) corresponding to the matrix formed via monomer curing and the thermoplastic phase, confirming the immiscibility of the blends. However, shifts and broadening of Tg values indicate improved interfacial interactions across the studied composition range. These results highlight the potential of glycerol-based methacrylate monomers as multifunctional platforms for tunable resin formulations and sustainable SLA 3D printing.
1. Introduction
Glycerol (also known as glycerin, 1,2,3-propanetriol, etc.) has transitioned from being a critical industrial chemical in limited supply to an abundant by-product of biodiesel production over the past seven decades. Global biodiesel production was projected to reach 41.4 × 109 L by 2025, a 33% increase from 2015.1,2 Since ∼10% of the weight of biodiesel is generated as glycerol, a significant amount of waste glycerol has been produced. Therefore, the reuse and valorization of glycerol have become urgent priorities.3 Numerous catalytic strategies, including oxidation, etherification, esterification, and acetalization, have been explored to convert glycerol into value-added chemicals, such as organic acids, diols, and fuels.4–10The three hydroxyl (-OH) groups of glycerol make it an excellent precursor for multifunctional monomers and bio-based polymers. Its non-flammability, low volatility, and low toxicity further enhance its suitability as a sustainable feedstock.11,12 Glycerol and its derivatives have been used in polycondensation polymerization to produce materials such as polyglycerols, bio-polyesters, and multifunctional polyurethanes with properties like self-healing and shape recovery.13,14 However, most reported glycerol polycondensates remain limited to oligomeric or crosslinked structures and often require complex synthesis steps, which hinder broader commercial adoption.15
Radical polymerization has also been utilized in the synthesis of glycerol-derived polymers.16–18 Methods such as (trans)esterification19 or transvinylation20 are typically employed to functionalize glycerol with (meth)acrylic, vinylic, or allylic groups. Such biobased monomers are compatible with various radical polymerization methods and have enabled the synthesis of high-performance thermoplastics and adhesives. For instance, esterifying crude glycerol with acrylic acid, followed by RAFT polymerization to mitigate gelation, has resulted in the production of glycerol-based thermoplastics with high molecular weights and performance comparable to conventional wood adhesives.15 Glycerol ketal(meth)acrylate monomers are synthesized by the reaction of various glycerol ketals and methyl(meth)acrylate via enzymatic transesterification and used to develop novel biopolymers with potential application in hard block polymers and adhesives.21 The results of this study demonstrate how the choice of (meth)acrylate backbone and pendant side chain groups can be used to influence the thermal and rheological properties of the resulting polymers.
3D printing, also known as additive manufacturing (AM), is a transformative technology that builds complex three-dimensional objects layer by layer from digital models, enabling geometries that are difficult or impossible to achieve with traditional methods.22 The global market for 3D printing materials has experienced exponential growth over the past decade, with photopolymers now accounting for nearly half of global material sales.23 The global revenues for AM materials are expected to reach $30 B by 2032, with the largest market shares for the aerospace, defense, medical, and dental industries.24 Stereolithography (SLA) is a well-established AM technique that enables the layer-by-layer photopolymerization of liquid resins into high-resolution parts with fast production times. Although it is an efficient and low-waste production method, the lack of sustainable resins for vat photopolymerization remains a major limitation, underscoring the need for environmentally friendly alternatives to support broader adoption.25
In recent years, there has been interest in the formulation and application of resins derived from renewable feedstocks such as lignin and vegetable oils.26–28 For example, soybean oil methacrylates have been formulated into printable resins that showed full layer fusion, high print quality, and tunable mechanical properties, including stiffness and toughness, by varying their chemical composition and functionality.29 Another renewable soybean oil-based epoxidized acrylate has also been processed by 3D laser printing to produce porous, biocompatible scaffolds capable of supporting human bone marrow stem cell growth.30 Five 3D-printable, self-healing, reprocessable, and chemically degradable thermoset polymers were fabricated by the functionalization of different Jeffamines with vanillin methacrylate and formulated with vanillin acrylate to allow for printability. The resulting thermosets exhibited a broad range of mechanical properties (Young's modulus: 2.05–332 MPa), enabling diverse applications.31 However, considerable efforts are still needed to develop environmentally friendly materials for SLA 3D printing that can replace commercial fossil fuel-based resins.
We previously reported the controlled synthesis of symmetric and non-symmetric 1,3-diether-2-propanol and 1,2,3-triether derivatives, starting from epichlorohydrin (ECH), a key molecule that can be obtained from glycerol via reaction with two equivalents of HCl.8,32–34 While 1,2,3-triether compounds show excellent promise as tunable solvents, particularly for CO2 absorption,33 1,3-diether-2-propanol serves as a versatile intermediate for synthesizing various functional molecules, including esters, ketones, and amines.32,34–37 We recently utilized 1,3-diether-2-propanol compounds as precursors to synthesize UV-curable glycerol-based methacrylate monomers designed to serve as critical components in SLA 3D printing resins. Comprehensive evaluations of the thermal and mechanical properties of the 3D-printed objects were performed and reported in our recently published paper,38 revealing that these straightforward monomers can create parts indistinguishable in appearance from highly optimized commercial resins. The versatility of these methacrylate monomers extends beyond their role in 3D printing resins. They can also function as solvents for certain commodity thermoplastics, effectively dissolving polystyrene (PS) and poly(methyl methacrylate) PMMA. This dissolved plastic is integrated into the printed structures, enabling the customization of their material properties, including mechanical properties. Therefore, these glycerol-based methacrylate monomers open new opportunities for creating polymer blends through SLA 3D printing. While polymer blends are commonly achieved through melt blending using equipment like extruders, the dual functionality of MAA-DEP as both a monomer and a solvent offers an innovative alternative for preparing polymer blends through a fundamentally distinct method.
Incorporation of non-reactive polymers or prepolymers into photopolymerizable monomers is a strategy to generate phase-separated structures through photopolymerization-induced phase separation.39–41 In these systems, an initially homogeneous mixture becomes thermodynamically unstable as polymerization proceeds, owing to increasing molecular weight, reduced entropy of mixing, and rising viscosity of the forming network. As a result, the dissolved polymer or prepolymer demixes from the growing photopolymer matrix, producing composition-dependent phase morphologies. This behavior has been widely reported in UV-curable coatings, films, and bulk photopolymer systems containing dissolved polymers or oligomeric modifiers, where phase separation and domain size are governed by additive loading, molecular weight, and curing kinetics, and exploited to tailor optical, mechanical, and thermal properties.42,43 This phenomenon has been rarely reported in the context of vat photopolymerization and SLA resin design; however, extending this concept to SLA printing enables composition-dependent control of microstructure and properties within printed objects.
In this work, we demonstrate how dissolving PS and PMMA into glycerol-based diether-methacrylate monomers significantly impacts the microstructure of the resulting polymer blends, thereby influencing their mechanical and thermal properties. Moreover, PS is a major contributor to plastic waste streams and is notoriously difficult to recycle by conventional means44 such that incorporating materials like waste PS (wPS), PMMA, and potentially other thermoplastics into 3D printing resins provides a promising upcycling route that transforms discarded plastics into higher-value materials and proportionally reduces the volume of SLA resin required. By blending renewable glycerol-derived monomers with wPS and PMMA, this work demonstrates a circular materials design strategy in which bio-based feedstocks and waste plastics are combined to create sustainable resins for AM applications.
2. Experimental
2.1. Materials
(±)-ECH (99%) and 2-methoxyethanol (99%) were purchased from Beantown Chemical. ACS-grade ethanol (EtOH), methanol (MeOH), chloroform (CHCl3), and sodium hydroxide (NaOH, 97%) were received from VWR, while methacrylic anhydride (MAA, 94%) was purchased from Sigma-Aldrich. 4-(Dimethylamino)pyridine (DMAP) was acquired from Chem-Impex International, and phenylbis(2,4,6-trimethylbenzoyl)phosphine oxide (BAPO), used as the initiator, was obtained from TCI America. Triethylamine (Et3N) was purchased from Macron Fine Chemicals. wPS was obtained from food containers marked with resin identification code (RIC) 6. These containers were manually cut into small flakes (0.5–1.0 cm), washed with deionized water and EtOH, and dried in a vacuum oven at 60 °C overnight before use. The weight-average molecular weight (Mw) of wPS was determined to be 174.7 kDa using gel permeation chromatography (GPC).45 Virgin PS (PS(45k): Mw = 45 kDa) and PMMA (Mw = 15 kDa) were purchased from Scientific Polymer Products (Ontario, NY, USA).2.2. Synthesis of symmetric 1,3-diethers
Symmetric 1,3-diether-2-propanol compounds, including 1,3-diethoxypropan-2-ol (DEP), 1,3-dibutoxypropan-2-ol (DBP), and 1,3-bis(2-methoxyethoxy)propan-2-ol (DMEP), were synthesized from ECH and the corresponding alcohols following the method outlined in our previous studies.2,342.3. Synthesis of symmetric 1,3-diether-2-methacrylates
Three glycerol-based monomers, 1,3-diethoxypropan-2-yl methacrylate (MAA-DEP), 1,3-dibutoxypropan-2-yl methacrylate (MAA-DBP), and 2,5,9,12-tetraoxatridecan-7-yl methacrylate (MAA-DMEP), were synthesized through the reaction of the corresponding symmetric 1,3-diether-2-propanol compounds with MAA, using Et3N and DMAP as catalysts (Scheme S1). The synthetic procedure followed the strategy previously reported in our earlier publication,38 and is briefly summarized in the SI. Fig. 1 illustrates the chemical structures of the 1,3-diether-2-methacrylate monomers.2.4. Resin formulation for 3D printing
To prepare the PS or PMMA solutions with symmetric 1,3-diether-2-methacrylate monomers, a specific amount of wPS (5 and 10 wt%), PS(45k) (10 wt%) or PMMA (5 and 10 wt%) was weighed and added to the monomer. The mixture was heated at 45 °C while stirring for a minimum of 5 h to obtain a clear solution. SLA 3D printing resins were formulated by incorporating 2.5 wt% BAPO photoinitiator into each PS or PMMA monomer solution. The mixtures were stirred for 10 min until a clear solution was achieved.2.5. SLA 3D printing
3D printing was performed using an Elegoo Mars 4 MSLA printer (China) equipped with a 405 nm light source. Each layer with a thickness of 50 μm was exposed for 40 s for pure monomers and 50 s for PS or PMMA/monomer solutions. After printing, the 3D-printed parts were cleaned to remove any residual monomer, and finally, the parts were post-cured in a UV oven (Elegoo Mercury Plus) for an additional 10 min.2.6. Characterization
1H NMR spectra were acquired using a 500 MHz Bruker Avance instrument (Billerica, MA, USA). The viscosity of PS or PMMA solution was measured at 25 °C using an Anton Paar ViscoQC 300-L rotational viscometer equipped with a CP40 cone plate spindle. Glass transition temperature (Tg) values of the polymers were determined by differential scanning calorimetry (DSC) using a TA Instruments DSC Q20 (New Castle, DE, USA). 3D-printed samples were heated and cooled from −40 to 150 °C at a scan rate of 10 °C min−1 under a nitrogen atmosphere. Multiple heating and cooling cycles were performed to ensure reliable results.Thermogravimetric Analysis (TGA) measurements were conducted using a SETARAM Labsys Evo thermal analyzer (KEP Technologies, TX, USA). An appropriate amount of 3D-printed polymer sample was placed in alumina (Al2O3) pans and heated from 25 to 700 °C at a heating rate of 10 °C min−1 under a constant flow of ultra-high purity argon gas (AR UHP300, Airgas). Tensile tests were performed using a compact table-top electromechanical-driven single-column load-frame universal testing machine (Test Resources Inc, Shakopee, MN, USA) equipped with a 1.1 kN load cell. Tests were conducted according to ASTM D 638 standards on specimens with dimensions of 82 mm × 12 mm × 2 mm. Degree of Conversion (DC) was determined using Fourier Transform Infrared Spectroscopy (FTIR) with a PerkinElmer Spectrum Two ATR-FTIR instrument. DC was calculated based on the change in the absorption intensity of the methacrylate C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibration at 1638 cm−1 before and after 3D printing, using the C
C stretching vibration at 1638 cm−1 before and after 3D printing, using the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration (1715–1725 cm−1) as an internal standard. Eqn (1) was used to calculate DC:
O stretching vibration (1715–1725 cm−1) as an internal standard. Eqn (1) was used to calculate DC:
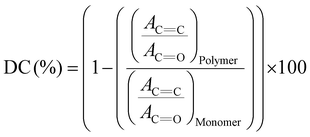 | (1) |
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C represents the absorbance at 1638 cm−1 before and after 3D printing, while AC
C represents the absorbance at 1638 cm−1 before and after 3D printing, while AC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O refers to the absorbance peak around 1715–1725 cm−1 in the monomer and polymer.46,47
O refers to the absorbance peak around 1715–1725 cm−1 in the monomer and polymer.46,47
The morphology of 3D-printed samples prepared from solutions of PS or PMMA in 1,3-diether-2-methacrylates was analyzed using a Thermo Fisher Scientific Apreo 2 SEM. Samples were cryogenically fractured in liquid nitrogen, coated with silver, and analyzed at an accelerating voltage of 5 kV. XRD measurements were performed using a Panalytical MPD X'pert Pro diffractometer (model: PW3050/60).
3. Results and discussion
3.1. Polymer blend formation through SLA 3D printing
This study explores a novel approach to polymer blend formation using SLA 3D printing. We previously reported the efficient synthesis of 1,3-diether-2-methacrylate monomers via Steglich esterification of glycerol-derived 1,3-diether-2-propanol with MAA, catalyzed by DMAP.38 The resulting pure monomers (Fig. 1) demonstrate a capability to dissolve some thermoplastics, such as PS and PMMA. 3D printing of 1,3-diether-2-methacrylate monomers containing varying fractions of dissolved PS and PMMA, in the presence of BAPO as a photoinitiator, resulted in the formation of polymer blends via a process distinct from conventional melt or solution blending, which typically requires heating and mechanical mixing in an extruder or solvent medium. This promising approach significantly influences both the mechanical properties and the microstructure of the resulting blend, which will be discussed in the following sections. In this study, wPS (Mw = 174.7 kDa), low molecular weight PS (Mw = 45 kDa), and PMMA (Mw = 15 kDa) were dissolved in the three different 1,3-diether-2-methacrylate monomers at 5 and 10 wt% to evaluate the influence of PS and PMMA on the properties of the resulting 3D printed polymer blends. The thermoplastic loadings were selected based on polymer solubility, resin viscosity, and SLA printability. Although MAA-DEP could dissolve up to 15 wt% wPS, formulations at this loading exhibited excessively high viscosity and could not be printed reliably, resulting in part adhesion to the resin tank even at extended exposure times. In contrast, PMMA loadings above 10 wt% were not fully soluble, leading to heterogeneous resins. Therefore, 5 and 10 wt% loadings were chosen as the highest compositions that ensured complete dissolution and reproducible printing while enabling evaluation of the effect of thermoplastic content on phase morphology and properties. The physical appearance of pure polymer and 3D-printed poly(1,3-diether-2-methacrylate)/PS or PMMA is shown in Fig. 2. The incorporation of PS notably altered the color and transparency of the resulting 3D-printed specimens, leading to yellowish blends. In contrast, PMMA-based blends retained some degree of transparency, particularly at lower concentrations, highlighting their better compatibility and optical clarity with the poly(MAA-DEP) matrix.3.2. Hansen solubility parameters
To further assess the miscibility of glycerol-derived methacrylate monomers with PS and PMMA, Hansen Solubility Parameters (HSPs) and relative energy differences (RED) were calculated (Table 1 and Table S1). HSP provides a useful framework for understanding polymer solubility in terms of intermolecular interactions relevant to solvation; however, they are not a perfect model, with reported accuracies of only ∼67% for “good” solvents and even lower for polar polymers.48 PS and PMMA were used as reference polymers with δD (dispersion), δP (polar), and δH (H-bonding) values of 18.5, 4.5, 2.9, and 18.6, 10.5, 5.1 (MPa)1/2, respectively, and R0 (interaction radius) of 8 (MPa)1/2 (Table S1). The calculated RED factors for the methacrylate monomers with PS were typically <1 (0.6–0.7), indicating favorable compatibility and consistent with the observed dissolution of PS in 1,3-diether-2-methacrylate monomers. In contrast, the RED values for the same monomers with PMMA were closer to unity (0.9–1.1), suggesting marginal miscibility. Interestingly, despite these predictions, PMMA is soluble in MAA-DEP, indicating that molecular weight, chain mobility, and processing conditions, factors not captured in HSP/RED calculations, can significantly influence experimental solubility.| Monomers | δ d ((MPa)1/2) | δ p ((MPa)1/2) | δ h ((MPa)1/2) | R a ((MPa)1/2) | RED | ||
|---|---|---|---|---|---|---|---|
| PS | PMMA | PS | PMMA | ||||
| δ D: dispersion forces. δP: polar interactions. δH: hydrogen-bonding interactions. RED = Ra/R0. | |||||||
| MAA-DEP | 16.0 | 4.7 | 5.0 | 5.4 | 7.8 | 0.7 | 1.0 |
| MAA-DBP | 16.1 | 3.7 | 4.2 | 5.0 | 8.5 | 0.6 | 1.1 |
| MAA-DMEP | 16.2 | 5.3 | 6.3 | 5.8 | 7.2 | 0.7 | 0.9 |
3.3. Viscosity
Resin viscosity plays a crucial role in SLA 3D printing, significantly influencing print quality, mechanical properties, and the overall success of the printing process.49 In general, relatively low viscosity is preferred to ensure proper recoating of the liquid resin between the previously cured layer and the resin tank surface.50 Formulations with viscosities <5000 mPa s (5000 cP) are within the capabilities of SLA printers, ensuring proper layer recoating and adhesion during the printing process.51Table 2 summarizes the viscosities of the pure monomers and their corresponding PS and PMMA solutions at ∼25 °C and atmospheric pressure. The pure 1,3-diether-2-methacrylate monomers exhibit low viscosities at 25 °C, ranging from 2.5 to 5.5 mPa s. The dissolution of PS and PMMA into the monomers significantly increases resin viscosity. For example, incorporating 5 and 10 wt% of wPS into MAA-DEP raises the viscosity to 44.1 and 166.2 mPa s, respectively. Higher monomer viscosity generally results in higher PS solution viscosity, with the MAA-DMEP/PS (10 wt%) solution exhibiting the highest value of 428.6 mPa s among the systems studied. In contrast, dissolving lower molecular weight PS, PS(45k), at 10 wt% in MAA-DEP resulted in a lower viscosity. The lowest viscosities among the polymer solutions, 5.2 and 10.7 mPa s, were observed for 5 and 10 wt% low molecular weight PMMA solutions in MAA-DEP. All formulations developed in this study demonstrated viscosities suitable for use in vat photopolymerization-based printing. However, since PS or PMMA lack reactive functional groups for photopolymerization, it is likely to interfere with the curing of active monomers, potentially influencing the final material properties.| Sample | μ (mPa s) |
|---|---|
| MAA-DEP | 2.80 |
| MAA-DEP/wPS (5 wt%) | 44.1 |
| MAA-DEP/wPS (10 wt%) | 166.2 |
| MAA-DEP/PS(45k) (10 wt%) | 17.7 |
| MAA-DEP/PMMA (5 wt%) | 5.2 |
| MAA-DEP/PMMA (10 wt%) | 10.7 |
| MAA-DBP | 5.53 |
| MAA-DBP/wPS (10 wt%) | 284.1 |
| MAA-DMEP | 4.95 |
| MAA-DMEP/wPS (10 wt%) | 428.6 |
3.4. Degree of conversion (DC, %)
The degree of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond conversion (DC%) in SLA 3D printing refers to the proportion of monomers that have polymerized into the solid structure during the curing process. A higher DC indicates a more complete polymerization, which significantly influences the mechanical properties and overall performance of the printed material. Fig. S1 presents the FTIR spectra of the 1,3-diether-2-methacrylate monomer blended with dissolved PS or PMMA, both before and after 3D printing. In most of the studied systems, the characteristic C
C double bond conversion (DC%) in SLA 3D printing refers to the proportion of monomers that have polymerized into the solid structure during the curing process. A higher DC indicates a more complete polymerization, which significantly influences the mechanical properties and overall performance of the printed material. Fig. S1 presents the FTIR spectra of the 1,3-diether-2-methacrylate monomer blended with dissolved PS or PMMA, both before and after 3D printing. In most of the studied systems, the characteristic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching peak at 1638 cm−1 was almost eliminated after 3D printing, indicating efficient polymerization. However, in certain polymer blends—such as poly(MAA-DEP)/PS(45k) (10 wt%) and poly(MAA-DBP)/wPS (10 wt%)—this peak persisted with reduced intensity. Table 3 summarizes the DC calculated from eqn (1) for the 3D-printed 1,3-diether-2-methacrylate polymers containing 5 and 10 wt% of PS and PMMA. Blending poly(MMA-DEP) with wPS preserved high polymerization efficiency, achieving DC values >80%. This suggests that the increased viscosity of the 10 wt% wPS solution did not hinder C
C stretching peak at 1638 cm−1 was almost eliminated after 3D printing, indicating efficient polymerization. However, in certain polymer blends—such as poly(MAA-DEP)/PS(45k) (10 wt%) and poly(MAA-DBP)/wPS (10 wt%)—this peak persisted with reduced intensity. Table 3 summarizes the DC calculated from eqn (1) for the 3D-printed 1,3-diether-2-methacrylate polymers containing 5 and 10 wt% of PS and PMMA. Blending poly(MMA-DEP) with wPS preserved high polymerization efficiency, achieving DC values >80%. This suggests that the increased viscosity of the 10 wt% wPS solution did not hinder C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond conversion. The incorporation of 10 wt% PS (45 kDa) into MAA-DEP reduced the DC to 68.9%. This reduction is likely due to the PS chains interfering with the formation of a continuous polymer matrix, possibly through the formation of microdomains that disrupt network uniformity. A notably lower DC (44.8%) was observed for the poly(MAA-DBP)/wPS (10 wt%) blend. The significant reduction may stem from the greater interference of wPS with the curing of MAA-DBP, whose longer alkyl side chains (compared to MAA-DEP) introduce additional steric hindrance and reduce compatibility during network formation. As will be shown later, the SEM images clearly illustrate the resulting differences in phase structure. However, the poly(MAA-DMEP)/wPS (10 wt%) blend exhibited a relatively high DC of 77.1%, despite having the highest viscosity among the systems studied. This suggests that the more flexible chemical structure of the DMEP monomer may facilitate better compatibility or interaction with wPS, enabling more efficient polymerization under high-viscosity conditions. These findings highlight the significant impact of PS molecular weight and chemical structure of the monomer on the polymerization efficiency of SLA-printed blends.
C double bond conversion. The incorporation of 10 wt% PS (45 kDa) into MAA-DEP reduced the DC to 68.9%. This reduction is likely due to the PS chains interfering with the formation of a continuous polymer matrix, possibly through the formation of microdomains that disrupt network uniformity. A notably lower DC (44.8%) was observed for the poly(MAA-DBP)/wPS (10 wt%) blend. The significant reduction may stem from the greater interference of wPS with the curing of MAA-DBP, whose longer alkyl side chains (compared to MAA-DEP) introduce additional steric hindrance and reduce compatibility during network formation. As will be shown later, the SEM images clearly illustrate the resulting differences in phase structure. However, the poly(MAA-DMEP)/wPS (10 wt%) blend exhibited a relatively high DC of 77.1%, despite having the highest viscosity among the systems studied. This suggests that the more flexible chemical structure of the DMEP monomer may facilitate better compatibility or interaction with wPS, enabling more efficient polymerization under high-viscosity conditions. These findings highlight the significant impact of PS molecular weight and chemical structure of the monomer on the polymerization efficiency of SLA-printed blends.
| Printed blend | DC (%) |
|---|---|
| Poly(MAA-DEP) | 78.3 |
| Poly(MAA-DEP)/wPS (5 wt%) | 84.7 |
| Poly(MAA-DEP)/wPS (10 wt%) | 80.6 |
| Poly(MAA-DEP)/PS(45k) (10 wt%) | 68.9 |
| Poly(MAA-DBP) | 82.1 |
| Poly(MAA-DBP)/wPS (10 wt%) | 44.8 |
| Poly(MAA-DMEP) | 80.7 |
| Poly(MAA-DMEP)/wPS (10 wt%) | 77.1 |
| MAA-DEP/PMMA (5 wt%) | 85.8 |
| MAA-DEP/PMMA (10 wt%) | 84.6 |
Dissolving PMMA into MAA-DEP at both 5 and 10 wt% resulted in high DC values of 85.8% and 84.6%, respectively. Unlike the PS blends, the PMMA blends remained optically transparent (Fig. 2f and g) to some extent and highly flexible, likely due to excellent molecular-level dispersion of PMMA within the matrix. The structural similarity between PMMA and the poly(methacrylate) matrix likely enhances miscibility and interfacial compatibility, reducing phase separation and optical scattering that could otherwise impede light penetration and hinder efficient photopolymerization.
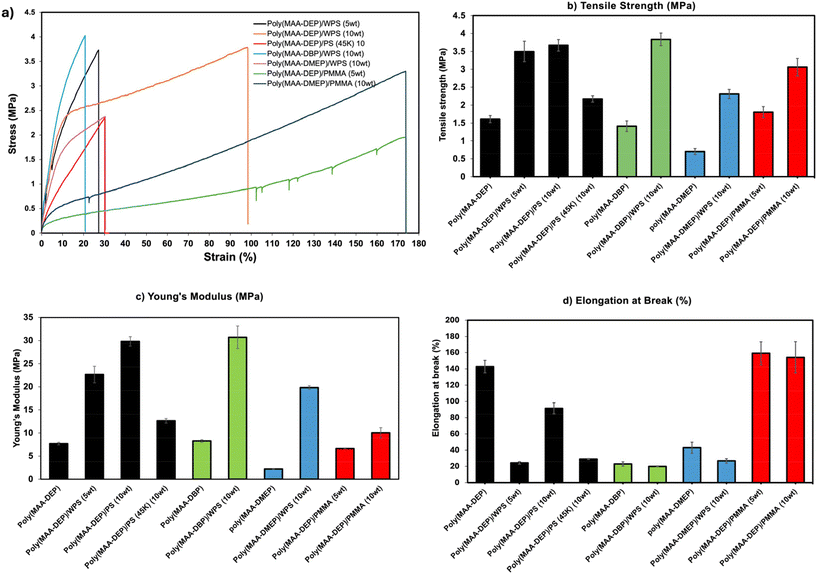
3.5. Mechanical properties
The mechanical performance of the 3D-printed polymer blends was evaluated via tensile testing, with results reported in terms of tensile strength, Young's modulus, and elongation at break (Fig. 3 and Table S2). The mechanical behavior of the pristine 3D-printed monomers has been previously reported; among them, poly(MAA-DEP) exhibited the best performance, displaying a characteristic elastic response with a tensile strength of 1.61 MPa, Young's modulus of 7.67 MPa, and elongation at break of 142.94%.38The incorporation of PS into 1,3-diether-2-methacrylate polymers generally enhanced the tensile strength and stiffness of the resulting blends while reducing their elongation at break. This trend is attributed to the reinforcing effect of PS, which inherently possesses high tensile strength and rigidity, but also introduces brittleness into the system. For example, blending 5 wt% wPS with poly(MAA-DEP) significantly increased the tensile strength (3.5 MPa) and modulus (22.6 MPa) compared to the neat resin; however, the elongation at break dropped sharply to 24.2% (Fig. 3). Interestingly, increasing the wPS loading to 10 wt% in poly(MAA-DEP) further enhanced strength and stiffness, while the elongation at break recovered to ∼90%. This behavior suggests that the dispersion of the PS domain and phase morphology in the resulting blend are influenced by composition, as will be further examined in the following microstructure analysis section. The incorporation of 10 wt% PS(45k) into poly(MAA-DEP) yielded lower tensile strength (2.2 MPa) and modulus (12.6 MPa), along with limited elongation (∼29%) (Fig. 3d and Table S2). The inferior performance is likely due to lower molecular weight, reduced chain entanglement, and weaker phase interaction between the short-chain PS and the matrix. The poly(MAA-DBP)/wPS (10 wt%) formulation exhibited a relatively high tensile strength of 3.9 MPa and the highest Young's modulus of 30.7 MPa, primarily due to the intrinsically rigid nature of the poly(MAA-DBP) matrix and the reinforcing effect of the incorporated PS. However, the elongation at break decreased to ∼20%, indicating limited elasticity (Fig. 4d). The poly(MAA-DMEP)/wPS (10 wt%) blend exhibited significantly enhanced mechanical strength and stiffness, over threefold and ninefold increases, respectively, compared to its neat counterpart, likely due to improved interfacial interaction between the two polymers. However, its elongation at break was reduced to ∼27%.
 | ||
| Fig. 4 SEM images of cryofracture surface of 3D printed (a) pure poly(MAA-DEP), b and (c) poly(MAA-DEP)/wPS (5 wt%), d and (e) poly(MAA-DEP)/wPS (10 wt%) and f and (g) poly(MAA-DEP)/PS(45k) (10 wt%). | ||
The incorporation of PMMA into poly(MAA-DEP) led to noticeable changes in mechanical performance. At 5 wt%, PMMA improved the elongation at break to 159.2%, surpassing even the pristine polymer and all PS-containing blends. This remarkable elasticity is likely due to better dispersion of PMMA domains and favorable interaction between low molecular weight PMMA and the poly(MAA-DEP) matrix, enabling more energy dissipation during deformation. While the tensile strength remained comparable to the neat polymer (1.80 MPa vs. 1.6 MPa for pure polymer), the Young's modulus decreased slightly to 6.6 MPa at low PMMA loading. At 10 wt%, the increased content of PMMA led to significant improvements in tensile strength (3.1 MPa) and Young's modulus (10.0 MPa) without a significant change in elongation at break. This behavior contrasts with the trends observed in PS blends, which exhibited low elongation at break, underscoring the favorable nature of PMMA-matrix interactions despite its low molecular weight.
3.6. Thermal properties
| Sample |
T
g1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a (°C) a (°C) |
T
g2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) b (°C) b (°C) |
T
d,max1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c (°C) c (°C) |
T
d,max2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) d (°C) d (°C) |
|---|---|---|---|---|
| a T g of main polymer matrix. b T g of PS or PMMA phase. c Primary maximum decomposition temperature of polymer matrix as measured by TGA. d Secondary maximum decomposition temperature of PS or PMMA component. | ||||
| Poly(MAA-DEP) | 0.29 | — | 291 | — |
| wPS | — | 103.73 | — | 410 |
| Poly(MAA-DEP)/wPS (5 wt%) | −2.28 | 80.6 | 331 | 431 |
| Poly(MAA-DEP)/wPS (10 wt%) | 0.72 | 95.82 | 309 | 395 |
| PS (45k) | — | 60.72 | — | 405 |
| Poly(MAA-DEP)/PS45 (10 wt%) | −0.92 | 59.04 | 338 | 416 |
| Poly(MAA-DBP) | 27.17 | — | 326 | — |
| Poly(MAA-DBP)/wPS (10 wt%) | 27.11 | 89.49 | 301 | 419 |
| Poly(MAA-DMEP) | 26.17 | — | 309 | — |
| Poly(MAA-DMEP)/wPS (10 wt%) | 22.77 | 71.88 | 351 | 420 |
| PMMA | — | 80.50 | — | 361 |
| Poly(MAA-DEP)/PMMA (5 wt%) | −6.39 | 77.11 | 326 | — |
| Poly(MAA-DEP)/PMMA (10 wt%) | −0.80 | 74.31 | 324 | 410 |
DSC thermograms of pure 1,3-diether-2-methacrylate polymers, wPS, PS (45k) and PMMA are shown in Fig. S2. Among the pure polymers, poly(MAA-DEP) exhibited the lowest Tg, measured at 0.29 °C. The glass transition behavior of a polymer blend is a key indicator of its phase miscibility. While fully miscible blends exhibit a single Tg somewhere between the Tgs of the two pure components, immiscible blends typically exhibit two distinct Tg values corresponding to those of the pure components, indicating the presence of two separate phases.52 DSC analysis of the 3D-printed PS blends revealed two distinct Tgs in all samples: one corresponding to the main matrix, 1,3-diether-2-methacrylate polymers (Tg1), and the other to the PS or PMMA phase (Tg2), indicating that the two polymers are immiscible. However, the Tg peaks of the resulting polymer blends are generally broadened and slightly shifted compared to those of the corresponding pure polymers. For instance, the Tg of poly(MAA-DEP) containing 10 wt% wPS increased slightly to 0.72 °C with a broadened peak. In contrast, the blend containing 5 wt% wPS exhibited a decreased Tg of −2.28 °C, suggesting composition-dependent variations in segmental mobility and possible phase interactions between PS and the poly(MAA-DEP) matrix. Such Tg shifts and broadening are commonly attributed to partial miscibility between the polymer phases, particularly at interfacial regions.53,54 The Tg associated with the PS phase was consistently reduced across all blends. Residual unreacted monomers likely act as plasticizers, diffusing into the PS domains and reducing their Tg through enhanced segmental mobility and interfacial molecular interpenetration.
The poly(MAA-DEP)/PMMA blends also exhibited two Tg values. For the 5 wt% PMMA blend, the Tg of the matrix (Tg1) and the PMMA phase (Tg2) decreased to −6.39 °C and 77.11 °C, respectively. The reduction in Tg1 at low PMMA content implies enhanced segmental mobility in the matrix, likely due to the good dispersion of low molecular weight PMMA and partial intermixing at the molecular level, which increases free volume and reduces Tg. The structural similarity between the two polymers may promote this compatibility, facilitating better dispersion and interfacial adhesion. At a higher PMMA content (10 wt%), the diminished Tg1 depression suggests more significant phase separation, reducing the extent of interfacial mixing and its influence on matrix mobility. The decrease in Tg2 associated with the PMMA phase was also observed for these blends, attributed to enhanced chain mobility, likely due to interfacial mixing with the surrounding matrix and the presence of unreacted monomers. This interaction may locally disrupt PMMA's chain packing and reduce its Tg.
The more pronounced Tg2 shifts observed for the wPS-containing blends might be attributed to the polydisperse nature of the waste polystyrene. Although the Mw of wPS is relatively high (174.7 kDa), its broad molecular weight distribution (PDI = 2.31)45 implies the presence of a significant fraction of lower-molecular-weight chains. These shorter PS chains are likely to be more susceptible to partial interpenetration into the poly(methacrylate) matrix and to plasticization by residual monomers, leading to greater Tg depression.
3.7. Morphology analysis by SEM
Microstructural analysis of the 3D-printed polymer blends was conducted using SEM to investigate phase morphology and domain dispersion (Fig. 4 and 5, and S11). Most polymer blends are thermodynamically immiscible because the mixing of high-molecular-weight chains results in minimal entropy gain. In contrast, the enthalpy of mixing is typically positive, making the overall process energetically unfavorable. Additionally, the high viscosities of the individual polymers hinder phase mobility and coalescence, resulting in kinetically trapped, partially mixed systems. Consequently, conventional blending techniques such as extrusion or internal mixing often yield phase-separated morphologies, with domain sizes typically ranging from 0.1 to 50 μm. PS, a non-polar polymer with a rigid backbone, tends to be immiscible with many polar or flexible polymers due to unfavorable enthalpic interactions, often leading to phase separation in the resulting blends.58 The remarkable ability of the synthesized 1,3-diether-2-methacrylates in dissolving PS with different molecular weights and their application in SLA 3D printing leads to polymer blends with interesting and distinct morphology depending on the PS content and molecular weight. The cryofracture surface of 3D-printed poly(MAA-DEP) (Fig. 4a) displayed a smooth, homogeneous morphology without visible layer lines, suggesting uniform curing and strong interlayer adhesion. Fig. 4b and c show the fracture surface of the 3D-printed blend containing 5 wt% wPS in MAA-DEP with different magnifications. Unlike the smooth morphology observed in the pure poly(MAA-DEP), these images reveal layer-by-layer curing patterns characteristic of the SLA 3D printing process. At low PS concentrations, SEM analysis reveals that during 3D printing, the dissolved PS tends to migrate toward the top of each printed layer, forming PS-rich regions that are phase-separated from the main poly(MAA-DEP) matrix. The PS-rich regions themselves are characterized by spherical PS domains dispersed within the remaining poly(MAA-DEP) matrix. This creates a stratified microstructure, with each layer exhibiting a distinct boundary between the flexible matrix and the rigid PS domains. Such morphology disrupts the uniform stress distribution during deformation and introduces stress concentration points at the matrix–domain interfaces, resulting in a substantial loss of elasticity (Fig. 3d). | ||
| Fig. 5 SEM images of cryofractured surface of (a) and (b) poly(MAA-DEP)/PMMA (5 wt%), c and (d) poly(MAA-DEP)/PMMA (10 wt%). | ||
SEM analysis of the 3D-printed poly(MAA-DEP) blend containing 10 wt% wPS (Fig. 4d and e) reveals a markedly different microstructure compared to the 5 wt% sample. At the higher loading, PS appears more uniformly dispersed as discrete domains within the polymer matrix, suggesting improved trapping during photopolymerization. This behavior might be attributed in part to the higher resin viscosity at increased wPS content, which restricts polymer chain mobility and suppresses long-range phase migration during the layer-by-layer curing process. Although some heterogeneity persists, evident from the slightly higher PS domain concentration with smaller size near the top of each printed layer, the overall morphology shows the wPS phase is well dispersed compared to the blend with 5 wt%. By limiting PS mobility prior to gelation, the increased viscosity promotes kinetic stabilization of the dispersed phase, resulting in reduced stratification. This improved dispersion of PS domains contributes directly to the enhanced mechanical performance of the blend, leading to a significant recovery in elongation at break. These observations highlight resin viscosity as a key formulation parameter governing microstructure development and mechanical performance in SLA-printed polymer blends. However, SEM analysis of the poly(MAA-DEP) blend containing 10 wt% low molecular weight PS, PS(45k), reveals that spherical PS domains with different sizes from <1 µm to >10 µm are dispersed within the continuous poly(MAA-DEP) matrix. Additionally, larger PS domains consisting of aggregated smaller ones suggest incomplete coalescence or secondary phase separation during printing and curing. This morphology contrasts with the more uniformly dispersed structure observed for blends containing higher-molecular-weight PS.
The 3D-printed poly(MAA-DBP)/wPS (10 wt%) blend exhibited a distinctly different morphology, resembling a honeycomb-like droplet structure (Fig. S11a and b), which highlights the sensitivity of microstructure development to slight variations in monomer chemical structure. The larger droplets of the main matrix are observed at the bottom of each printed layer, encapsulated or surrounded by small PS domains (∼1 μm), while smaller matrix droplets dominate the top. This gradient structure indicates upward migration of the PS phase during curing, with the domains exhibiting relatively well-defined boundaries, indicative of poor miscibility and limited interfacial mixing between PS and the main matrix. The cryo-fractured surfaces of 3D-printed poly(MAA-DMEP) containing 10 wt% wPS exhibit a uniform dispersion of PS domains throughout the matrix (Fig. S11c and d). However, the layer boundaries appear more distinct compared to other systems, indicating less effective interlayer fusion during the SLA printing process.
SEM images of 3D-printed poly(MAA-DEP) blends containing 5 wt% and 10 wt% PMMA are presented in Fig. 5. At a lower magnification, Fig. 5a, the 5 wt% PMMA blend exhibits a homogeneous appearance, without characteristic layer-by-layer deposition patterns typically observed in 3D-printed PS-based blends. However, higher magnification (Fig. 5b) reveals a dilute droplet-matrix morphology, characterized by spherical PMMA domains approximately 1 μm in diameter dispersed within the continuous poly(MAA-DEP) matrix. Such morphology is common in blends with low concentrations of the dispersed phase.59 The interfacial adhesion between PMMA droplets and the poly(MAA-DEP) matrix, arising from favorable interactions between their ester functionalities, can enhance interfacial compatibility and reduce phase separation. These interactions facilitate more uniform stress distribution during deformation, promoting energy dissipation and contributing to improved elasticity and toughness60 (Fig. 3d). In contrast, the 10 wt% PMMA blend (Fig. 5c) exhibits larger, oval-shaped PMMA domains with less uniform dispersion, predominantly concentrated near the top of each printed layer with no distinct interlayer boundaries. Despite the significantly increased domain size, these PMMA regions maintain strong interfacial adhesion with the poly(MAA-DEP) matrix, as evidenced in Fig. 5d. The presence of larger domains may introduce stress concentration points; however, the overall matrix continuity is preserved, with the polymer matrix connected at several locations across each layer. This interconnected structure contributes to a continuous matrix, which is essential for maintaining mechanical integrity.
4. Conclusions
In this study, glycerol-derived 1,3-diether-2-methacrylate monomers were synthesized and employed as dual-function molecules, serving as both photopolymerizable monomers and solvents to dissolve thermoplastics such as PS and PMMA. These formulations were used to fabricate polymer blends via SLA 3D printing to investigate the effects of thermoplastic incorporation on morphology, mechanical, and thermal properties. The blends exhibited distinct phase separation depending on monomer structure, thermoplastic structure, and loading. All polymer blends displayed two Tg values, indicating the formation of immiscible systems; however, peak broadening and slight shifts suggest partial miscibility at the interface. The poly(MAA-DEP)/wPS system with low concentrations of wPS led to pronounced phase separation and a loss of elasticity, while increasing the wPS content to 10 wt% improved PS domain dispersion, enabling recovery of elongation at break, and more favorable mechanical properties. Incorporation of low-molecular-weight PMMA, with a structure closer to that of glycerol-derived 1,3-diether-2-methacrylate polymers, particularly at lower concentrations, yields a continuous polymer matrix with indistinguishable printed layer boundaries and a slight increase in elongation at break.This method provides an alternative to conventional blend formation, which typically relies on melt-blending polymers in an extruder. In contrast, the approach demonstrated here enables blend formation through photopolymerization in the presence of dissolved thermoplastics, thereby reducing energy input and processing complexity. Importantly, the potential use of waste PS and PMMA in these systems offers a promising route to reuse two of the most widely produced thermoplastics, aligning with circular economy principles. Further optimization through a more sophisticated monomer design with different types of functional groups (e.g., aromatics) and/or non-symmetric functional groups (e.g., one alkyl chain, one aromatic group) and/or through the inclusion of crosslinkers or compatibilizers could enhance material performance and expand the range of potential applications and types of polymers that could be dissolved. Nonetheless, it is clear that the use of glycerol-derived 1,3-diether-2-methacrylates offer a very wide range of design possibilities for sustainable and advanced SLA 3D printing resin formulations.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: detailed synthesis of symmetric 1,3-diether-2-methacrylates. Scheme S1. General synthesis of 1,3-diether-2-methacrylates. Table S1. Hansen solubility parameters (HSP) of PS and PMMA polymers. Table S2. Tensile properties of the 3D printed pure polymers and their blends. Fig. S1. FTIR spectra of poly(1,3-diether-2-methacrylate)/thermoplastic blends. Fig. S2. DSC thermograms of pure polymers. Fig. S3. DSC thermograms of poly(MAA-DEP)/WPS (5 wt%). Fig. S4. DSC thermograms of poly(MAA-DEP)/WPS (10 wt%). Fig. S5. DSC thermograms of poly(MAA-DEP)/PS(45k) (10 wt%). Fig. S6. DSC thermograms of poly(MAA-DBP)/WPS (10 wt%). Fig. S7. DSC thermograms of poly(MAA-DMEP)/WPS (10 wt%). Fig. S8. DSC thermograms of poly(MAA-DEP)/PMMA (5 wt%). Fig. S9. DSC thermograms of poly(MAA-DEP)/PMMA (10 wt%). Fig. S10. TGA and DTG thermograms of poly(1,3-diether-2-methacrylate)/thermoplastic blends. Fig. S11. SEM images of cryofracture surface of a and (b) poly(MAA-DBP)/WPS (10 wt%), c and (d) poly(MAA-DMEP)/WPS (10 wt%). See DOI: https://doi.org/10.1039/d6lp00005c.Acknowledgements
Support for this work provided by the US National Science Foundation (EFMA-2029387) is gratefully acknowledged. During the preparation of this work the author(s) used Adobe Firefly to generate portions of the TOC graphic. After using this tool/service, the authors reviewed and edited the content as needed and take full responsibility for the content of the published article. The authors’ university has a license for Adobe Firefly and terms of use associated with the software are being followed. All other images/photographs were fully created by the authors.References
- H. I. Mahdi, N. N. Ramlee, J. L. da Silva Duarte, Y.-S. Cheng, R. Selvasembian, F. Amir, L. H. de Oliveira, N. I. W. Azelee, L. Meili and G. Rangasamy, A comprehensive review on nanocatalysts and nanobiocatalysts for biodiesel production in Indonesia, Malaysia, Brazil and USA, Chemosphere, 2023, 319, 138003 CrossRef CAS PubMed.
- S. Qian, X. Liu, G. P. Dennis, C. H. Turner and J. E. Bara, Properties of symmetric 1, 3-diethers based on glycerol skeletons for CO2 absorption, Fluid Phase Equilib., 2020, 521, 112718 CrossRef CAS.
- A. Zanoni, G. Gardoni, M. Sponchioni and D. Moscatelli, Valorisation of glycerol and CO2 to produce biodegradable polymer nanoparticles with a high percentage of bio-based components, J. CO2 Util., 2020, 40, 101192 CrossRef CAS.
- G. Dodekatos, S. Schünemann and H. Tüysüz, Recent advances in thermo-, photo-, and electrocatalytic glycerol oxidation, ACS Catal., 2018, 8(7), 6301–6333 CrossRef CAS.
- S. Basu and A. K. Sen, A review on catalytic dehydration of glycerol to acetol, ChemBioEng Rev., 2021, 8(6), 633–653 CrossRef CAS.
- D. Spataru, A. P. S. Dias and L. F. V. Ferreira, Acetylation of biodiesel glycerin using glycerin and glucose derived catalysts, J. Cleaner Prod., 2021, 297, 126686 CrossRef CAS.
- S. Qian, X. Liu, V. N. Emel'yanenko, P. Sikorski, I. Kammakakam, B. S. Flowers, T. A. Jones, C. H. Turner, S. P. Verevkin and J. E. Bara, Synthesis and properties of 1, 2, 3-Triethoxypropane: a glycerol-derived green solvent candidate, Ind. Eng. Chem. Res., 2020, 59(45), 20190–20200 CrossRef CAS.
- I. V. Andreeva, D. H. Zaitsau, S. Qian, V. V. Turovtzev, A. A. Pimerzin, J. E. Bara and S. P. Verevkin, Glycerol valorisation towards biofuel additivities: Thermodynamic studies of glycerol ethers, Chem. Eng. Sci., 2022, 247, 117032 CrossRef CAS.
- K. Pandit, C. Jeffrey, J. Keogh, M. S. Tiwari, N. Artioli and H. G. Manyar, Techno-economic assessment and sensitivity analysis of glycerol valorization to biofuel additives via esterification, Ind. Eng. Chem. Res., 2023, 62(23), 9201–9210 CrossRef CAS PubMed.
- L. D. da Silva, R. C. Santos, J. G. A. B. Silva, E. de Paiva Alves, R. T. F. Fréty and L. A. M. Pontes, Direct ammoxidation of glycerol to nitriles using Mo/alumina catalysts, React. Kinet., Mech. Catal., 2022, 1–15 CAS.
- N. Ebadipour, S. Paul, B. Katryniok and F. Dumeignil, Alkaline-based catalysts for glycerol polymerization reaction: A review, Catalysts, 2020, 10(9), 1021 CrossRef CAS.
- M. Ionescu and Z. S. Petrović, On the Mechanism of Base–Catalyzed Glycerol Polymerization and Copolymerization, Eur. J. Lipid Sci. Technol., 2018, 120(6), 1800004 CrossRef.
- F. Zeng, X. Yang, D. Li, L. Dai, X. Zhang, Y. Lv and Z. Wei, Functionalized polyesters derived from glycerol: Selective polycondensation methods toward glycerol–based polyesters by different catalysts, J. Appl. Polym. Sci., 2020, 137(16), 48574 CrossRef CAS.
- T. Ghosh and N. Karak, Biobased multifunctional macroglycol containing smart thermoplastic hyperbranched polyurethane elastomer with intrinsic self-healing attribute, ACS Sustainable Chem. Eng., 2018, 6(3), 4370–4381 CrossRef CAS.
- M. Forrester, A. Becker, A. Hohmann, N. Hernandez, F.-Y. Lin, N. Bloome, G. Johnson, H. Dietrich, J. Marcinko and R. C. Williams, RAFT thermoplastics from glycerol: a biopolymer for development of sustainable wood adhesives, Green Chem., 2020, 22(18), 6148–6156 RSC.
- P. D. Pham, S. Monge, V. Lapinte, Y. Raoul and J. J. Robin, Various radical polymerizations of glycerol–based monomers, Eur. J. Lipid Sci. Technol., 2013, 115(1), 28–40 CrossRef CAS.
- A. Mhanna, F. Sadaka, G. Boni, C.-H. Brachais, L. Brachais, J.-P. Couvercelle, L. Plasseraud and L. Lecamp, Photopolymerizable synthons from glycerol derivatives, J. Am. Oil Chem. Soc., 2014, 91, 337–348 CrossRef CAS.
- X. Sun, J.-P. Lindner, B. Bruchmann and A. D. Schlüter, Synthesis of Neutral, Water-Soluble Oligo–Ethylene Glycol-Containing Dendronized Homo-and Copolymers of Generations 1, 1.5, 2, and 3, Macromolecules, 2014, 47(21), 7337–7346 CrossRef CAS.
- F.-Y. Lin, B. Claypool, M. J. Forrester, N. Hernández, A. Buss, B. Kuehl and E. W. Cochran, Glycerol-Based Pressure Sensitive Adhesives: Synthesis and Applications, ACS Sustainable Chem. Eng., 2024, 12(32), 12042–12051 CrossRef CAS.
- L. Pichavant, C. Guillermain and X. Coqueret, Reactivity of vinyl ethers and vinyl ribosides in UV-initiated free radical copolymerization with acceptor monomers, Biomacromolecules, 2010, 11(9), 2415–2421 CrossRef CAS PubMed.
- S. Goyal, F.-Y. Lin, M. Forrester, W. Henrichsen, G. Murphy, L. Shen, T.-P. Wang and E. W. Cochran, Glycerol ketals as building blocks for a new class of biobased (meth) acrylate polymers, ACS Sustainable Chem. Eng., 2021, 9(31), 10620–10629 CrossRef CAS.
- S. C. Ligon, R. Liska, J. Stampfl, M. Gurr and R. Mülhaupt, Polymers for 3D printing and customized additive manufacturing, Chem. Rev., 2017, 117(15), 10212–10290 CrossRef CAS PubMed.
- V. S. Voet, J. Guit and K. Loos, Sustainable photopolymers in 3D printing: a review on biobased, biodegradable, and recyclable alternatives, Macromol. Rapid Commun., 2021, 42(3), 2000475 CrossRef CAS PubMed.
- V. S. Voet, Closed-loop additive manufacturing: dynamic covalent networks in vat photopolymerization, ACS Mater. Au, 2022, 3(1), 18–23 Search PubMed.
- E. M. Maines, M. K. Porwal, C. J. Ellison and T. M. Reineke, Sustainable advances in SLA/DLP 3D printing materials and processes, Green Chem., 2021, 23(18), 6863–6897 RSC.
- E. Skliutas, M. Lebedevaite, S. Kasetaite, S. Rekštytė, S. Lileikis, J. Ostrauskaite and M. Malinauskas, A bio-based resin for a multi-scale optical 3D printing, Sci. Rep., 2020, 10(1), 9758 CrossRef CAS PubMed.
- J. Stouten, G. H. Schnelting, J. Hul, N. Sijstermans, K. Janssen, T. Darikwa, C. Ye, K. Loos, V. S. Voet and K. V. Bernaerts, Biobased photopolymer Resin for 3D printing containing dynamic imine bonds for fast reprocessability, ACS Appl. Mater. Interfaces, 2023, 15(22), 27110–27119 Search PubMed.
- L. Pezzana, R. Wolff, J. Stampfl, R. Liska and M. Sangermano, High temperature vat photopolymerization 3D printing of fully bio-based composites: Green vegetable oil epoxy matrix & bio-derived filler powder, Addit. Manuf., 2024, 79, 103929 CAS.
- J. Guit, M. B. Tavares, J. Hul, C. Ye, K. Loos, J. Jager, R. Folkersma and V. S. Voet, Photopolymer resins with biobased methacrylates based on soybean oil for stereolithography, ACS Appl. Polym. Mater., 2020, 2(2), 949–957 Search PubMed.
- S. Miao, W. Zhu, N. J. Castro, M. Nowicki, X. Zhou, H. Cui, J. P. Fisher and L. G. Zhang, 4D printing smart biomedical scaffolds with novel soybean oil epoxidized acrylate, Sci. Rep., 2016, 6(1), 27226 Search PubMed.
- K. P. Cortés-Guzmán, A. R. Parikh, M. L. Sparacin, A. K. Remy, L. Adegoke, C. Chitrakar, M. Ecker, W. E. Voit and R. A. Smaldone, Recyclable, biobased photoresins for 3d printing through dynamic imine exchange, ACS Sustainable Chem. Eng., 2022, 10(39), 13091–13099 CrossRef.
- S. Chatterjee, S. Qian, A. Soyemi, T. Szilvási and J. E. Bara, Synthesis and Properties of 2-Halo-1, 3-diether-propanes: Diversifying the Range of Functionality in Glycerol-Derived Compounds, Ind. Eng. Chem. Res., 2023, 62(6), 2959–2967 Search PubMed.
- S. Qian, X. Liu, C. H. Turner and J. E. Bara, Synthesis and properties of symmetric glycerol-derived 1, 2, 3-triethers and 1, 3-diether-2-ketones for CO2 absorption, Chem. Eng. Sci., 2022, 248, 117150 Search PubMed.
- S. Qian, J. D. Leah, S. Chatterjee, A. Soyemi, T. Szilvási and J. E. Bara, Properties of Imidazolium Ionic Liquids with Glycerol-Derived Functional Groups, J. Chem. Eng. Data, 2022, 67(8), 1905–1914 CrossRef CAS.
- J. Wang, S. Qian, G. Olajide, S. Chatterjee, T. Szilvasi and J. E. Bara, Synthesis and properties of symmetric glycerol-derived (E/Z)-1, 3-diether-2-alkenes, Ind. Chem. Mater., 2026, 4, 131–141 RSC.
- G. Lingua, G. Depraetère, J. Wang, J. E. Bara, M. Forsyth and D. Mecerreyes, Solvate Ionic Liquids based on branched glymes enabling high performance lithium metal batteries, J. Power Sources, 2024, 624, 235535 CrossRef CAS.
- J. Wang, M. Iddrisu, Z. S. Pour, S. Ravula, T. Szilvási and J. E. Bara, Synthesis and Properties of Novel Glycerol–Derived Liquids with Dual Functional Groups: Nonsymmetric (E/Z)–Isomeric Mixtures of 1, 3-Diether-2-Alkenes, Chem. – Eur. J., 2025, e01406 CrossRef CAS PubMed.
- Z. Sekhavat Pour, P. S. Shinde, J. Wang, C. Woods, S. Taylor, S. Chatterjee and J. E. Bara, 1, 3-Diether-2-Methacrylates with Glycerol Skeletons: Tunable Resins for Stereolithography 3D Printing, Polym. Chem., 2025, 16, 2840–2850 RSC.
- L. Zakrzewski, Y. Kim, Y. Song, C. Y. Ryu, C. Bae and C. R. Picu, Interplay of photopolymerization and phase separation kinetics and the resulting structure-property relationship of photocurable resins, Polymer, 2023, 280, 126032 CrossRef CAS.
- Y. Kawagoe, G. Kikugawa, K. Shirasu, Y. Kinugawa and T. Okabe, Dissipative particle dynamics simulation for reaction-induced phase separation of thermoset/thermoplastic blends, J. Phys. Chem. B, 2024, 128(8), 2018–2027 CrossRef CAS PubMed.
- S. J. Curley and C. R. Szczepanski, Applying Hansen Solubility Parameters to Dynamically Reacting Systems– A Case Study of Photopolymerization Induced Phase Separation, ACS Mater. Lett., 2025, 7(9), 3206–3212 CrossRef CAS.
- E. Hasa, T. Y. Lee and C. A. Guymon, Controlling phase separated domains in UV-curable formulations with OH-functionalized prepolymers, Polym. Chem., 2022, 13(21), 3102–3115 RSC.
- J. Joy, K. Winkler, K. Joseph, S. Anas and S. Thomas, Epoxy/methyl methacrylate acrylonitrile butadiene styrene (MABS) copolymer blends: Reaction-induced viscoelastic phase separation, morphology development and mechanical properties, New J. Chem., 2019, 43(23), 9216–9225 RSC.
- Z. Xu, D. Sun, J. Xu, R. Yang, J. D. Russell and G. Liu, Progress and challenges in polystyrene recycling and upcycling, ChemSusChem, 2024, 17(17), e202400474 CrossRef CAS PubMed.
- Z. Sekhavat Pour, P. S. Shinde and J. E. Bara, Upcycling of Polystyrene Waste to Poly (ionic liquid) Materials, ACS Appl. Polym. Mater., 2024, 6(23), 14309–14319 CrossRef.
- J.-J. Tzeng, T.-S. Yang, W.-F. Lee, H. Chen and H.-M. Chang, Mechanical properties and biocompatibility of urethane acrylate-based 3D-printed denture base resin, Polymers, 2021, 13(5), 822 CrossRef CAS PubMed.
- M. Diamantopoulou, N. Karathanasopoulos and D. Mohr, Stress-strain response of polymers made through two-photon lithography: Micro-scale experiments and neural network modeling, Addit. Manuf., 2021, 47, 102266 CAS.
- S. Venkatram, C. Kim, A. Chandrasekaran and R. Ramprasad, Critical assessment of the Hildebrand and Hansen solubility parameters for polymers, J. Chem. Inf. Model., 2019, 59(10), 4188–4194 CrossRef CAS PubMed.
- V. S. Voet, T. Strating, G. H. Schnelting, P. Dijkstra, M. Tietema, J. Xu, A. J. Woortman, K. Loos, J. Jager and R. Folkersma, Biobased acrylate photocurable resin formulation for stereolithography 3D printing, ACS Omega, 2018, 3(2), 1403–1408 CrossRef CAS PubMed.
- Z. Weng, Y. Zhou, W. Lin, T. Senthil and L. Wu, Structure-property relationship of nano enhanced stereolithography resin for desktop SLA 3D printer, Composites, Part A, 2016, 88, 234–242 CrossRef CAS.
- X. Kong, M. Dong, M. Du, J. Qian, J. Yin, Q. Zheng and Z. L. Wu, Recent Progress in 3D Printing of Polymer Materials as Soft Actuators and Robots, Chem Bio Eng., 2024, 1(4), 312–329 CrossRef CAS PubMed.
- S.-A. Xu and C.-M. Chan, Polystyrene/high density polyethylene blends compatibilized by a tri-block copolymer I. Properties and morphology, Polym. J., 1998, 30(7), 552–558 CrossRef CAS.
- C. Chuai, K. Almdal, I. Johannsen and J. Lyngaae-Jørgensen, Miscibility evolution of polycarbonate/polystyrene blends during compounding, Polym. Eng. Sci., 2002, 42(5), 961–968 CrossRef CAS.
- Y. Komori, A. Taniguchi, H. Shibata, S. Goto and H. Saito, Partial miscibility and concentration distribution of two-phase blends of crosslinked NBR and PVC, Polymers, 2023, 15(6), 1383 CrossRef CAS PubMed.
- F. P. La Mantia, M. Morreale, L. Botta, M. C. Mistretta, M. Ceraulo and R. Scaffaro, Degradation of polymer blends: A brief review, Polym. Degrad. Stab., 2017, 145, 79–92 CrossRef CAS.
- N. Netsch, L. Schröder, M. Zeller, I. Neugber, D. Merz, C. O. Klein, S. Tavakkol and D. Stapf, Thermogravimetric study on thermal degradation kinetics and polymer interactions in mixed thermoplastics, J. Therm. Anal. Calorim., 2025, 150(1), 211–229 CrossRef CAS.
- R. Tuffi, S. D’abramo, L. Cafiero, E. Trinca and S. V. Ciprioti, Thermal behavior and pyrolytic degradation kinetics of polymeric mixtures from waste packaging plastics, eXPRESS Polym. Lett., 2018, 12(1), 82–99 CrossRef CAS.
- U. V. Nikulova and A. E. Chalykh, Phase Equilibrium and Interdiffusion in Blends of Polystyrene with Polyacrylates, Polymers, 2021, 13(14), 2283 CrossRef CAS PubMed.
- M. Grosso and P. L. Maffettone, Fourier transform rheology: A new tool to characterize material properties, Fourier Transforms: New Anal. Approaches FTIR Strategies, 2011, 285, 1–20 Search PubMed.
- A. Thirugnanasamabandam, R. Nallamuthu, M. Renjit and C. L. Gnanasagaran, 3D–printed PLA/PMMA polymer composites: A consolidated feasible characteristic investigation for dental applications, Polym. Eng. Sci., 2024, 64(9), 4019–4031 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |