Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Bio-based vitrimers: chemistry, performance and applications
Edoardo
Albertini
 *,
Sara
Dalle Vacche
*,
Sara
Dalle Vacche
 and
Alessandra
Vitale
and
Alessandra
Vitale
 *
*
Politecnico di Torino, Corso Duca degli Abruzzi 24, 10129, Turin, Italy. E-mail: edoardo.albertini@polito.it; alessandra.vitale@polito.it
First published on 13th March 2026
Abstract
Despite being widely used in industrial applications, thermoset polymers arguably present a major challenge to modern society due to the near impossibility of their end-of-life recycling and reprocessing. Furthermore, there are difficulties in finding bio-based alternatives to the fossil-based building blocks used for their production that can deliver comparable mechanical performance. These limitations have driven growing interest in vitrimers, associative covalent adaptable networks capable of being recycled, welded and reprocessed without changes in cross-linking density through dynamic covalent exchange. Vitrimers provide a promising strategy to address both the recyclability of thermosets and the need for more sustainable materials. The use of bio-based chemicals in vitrimer production can further help reduce the environmental impact and toxicity, supporting the transition to circular polymer systems. This review summarizes recent advances in bio-based vitrimer systems, with particular emphasis on renewable feedstocks such as carbohydrates, lignin-derived aromatics, and vegetable oils, and their translation into functional materials. Rather than focusing solely on exchange chemistry concepts, the manuscript highlights the emerging applications of newly developed bio-based vitrimers in composites, packaging, additive manufacturing, adhesives, electronics, and foams. By organizing recent literature according to feedstock families and application domains, this work provides an updated, application-oriented perspective on the development of sustainable vitrimer technologies. Overall, bio-based vitrimers represent a promising platform for the development of next-generation recyclable thermosets within a circular and sustainable polymer framework.
1. Introduction
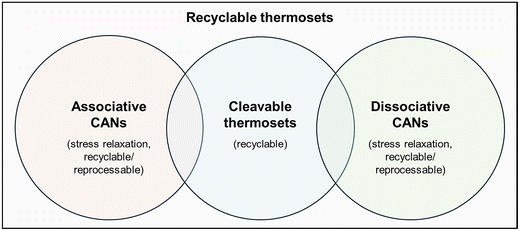
The persistent accumulation of plastic in soils, oceans, and deep-sea environments has become a critical global concern. Estimates of environmental half-lives remain uncertain, as degradation depends on polymer chemistry, additives, and local conditions.1 As plastics fragment into micro- and nanoparticles, they integrate into ecosystems, with documented impacts across environmental compartments and implications for climate processes.2 Forecasts indicate that global plastic consumption may rise from 464 Mt in 2020 to nearly 884 Mt by 2050, further intensifying waste accumulation.3 In response, advanced recycling strategies have been developed to valorize plastic waste by converting it into new monomers, polymers, or higher-value chemicals, thereby extending material lifetimes within a circular economy framework.4 From a materials perspective, plastics are classified into two major categories: thermoplastics and thermosets. Thermoplastics are characterized by two transition temperatures: the glass transition temperature (Tg) and the melting temperature (Tm, in the case of semi-crystalline polymers). Thermosets (primarily epoxies, polyesters and polyurethanes), on the other hand, are formed when monomers (usually referred to as resins) are covalently bonded through cross-linking agents to form amorphous cross-linked networks characterized by a single transition temperature, Tg. While this permanent cross-linked structure imparts superior thermal, mechanical, and chemical resistance, it also renders conventional thermosets intrinsically difficult to reprocess or recycle.5 Thermosets account for ca. 18% of the polymers currently in global circulation, are predominantly derived from fossil resources, and are widely used in many industrial applications such as packaging, automotive components, composite manufacturing, adhesives, lightweight construction, and aviation.6 To enhance the sustainability of this important class of materials, the development of thermosets that are both readily recyclable and derived from renewable resources is essential.7,8 In this context, bio-based feedstocks (e.g., lignocellulosic biomass, lignin-derived aromatics, and vegetable oils) have recently been studied as substitutes for fossil-based chemicals, with some already finding industrial applications.9–11 However, despite these efforts, difficulties in matching the mechanical properties and production costs of fossil-based raw chemicals used in thermoset production have been faced so far. In fact, only 0.7% of thermoset and thermoplastic polymers are produced from bio-based sources, as stated by the 2024 report by Plastics Europe.12 Biodegradation is another way for reducing the environmental impact of thermosets. Despite being mainly used for polyester-based thermosets, recent studies on the biodegradation of epoxy thermosets have yielded encouraging results as well.13–15 In this direction, the use of bio-based feedstocks can help biodegradation using more degradable building blocks.16,17 Typical bio-based platform compounds used for resin synthesis are reported in Fig. 1. The high abundance of hydroxyl groups in bio-derived molecules makes them particularly attractive for chemical functionalization and the synthesis of bio-based epoxy resins and cross-linkers.11An alternative and increasingly powerful strategy for reducing the environmental footprint of thermoset materials involves the deliberate modification of network chemistry to enable reprocessing/recyclability. Two main approaches can be identified: (a) the introduction of labile bonds or linkages that can be cleaved during recycling through chemical or thermal decross-linking, often accompanied by partial degradation of the polymer structure; and (b) the development of covalent adaptable networks (CANs). CANs are materials designed using building blocks that enable recyclable and reformable cross-linked structures based on dynamic covalent bonds while maintaining the advantages of conventional thermosets, such as high chemical and thermal resistance and, in some cases, depending on the specific chemistry, good mechanical performance. A classification of recyclable thermoset materials is shown in Fig. 2.
Within the broad family of CANs, vitrimers constitute a distinctive class of associative dynamic networks in which bond exchange reactions occur without a change in cross-link density. This unique behavior enables thermoset-like performance under service conditions combined with thermoplastic-like reprocessability at elevated temperatures.
In this review, particular emphasis is placed on bio-based vitrimers derived from renewable resources (including carbohydrates, lignin and its derivatives, and vegetable oils), with specific attention to their emerging applications reported in recent years. While several comprehensive reviews have examined bio-based vitrimers from the perspectives of dynamic covalent chemistries,18–20 sustainable feedstocks and synthetic strategies,19,20 or general progress in the field,21 a structured overview specifically centered on the application landscape of newly developed bio-based vitrimer systems remains limited.
The present work therefore aims to summarize and critically discuss recent advances in bio-based vitrimer formulations with demonstrated application potential, including composites, packaging, additive manufacturing, adhesives, electronics, and foams. Rather than reiterating well-established mechanistic aspects in detail, this review highlights how different renewable building blocks and exchange chemistries have been translated into functional materials tailored to specific industrial sectors. Following a brief introduction and a general description of vitrimer materials, the recent literature is organized according to feedstock families and application domains. In doing so, we aim to provide an updated, application-driven perspective that complements existing chemistry-focused reviews and offers a rational framework for polymeric material design, as well as guidance for the future development and implementation of bio-based vitrimer technologies within circular polymer systems.
2. Dynamic polymer materials
2.1. Covalent adaptable networks (CANs)
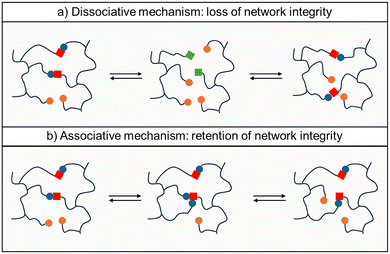
CANs owe their name to the ability of their covalently cross-linked structures to reorganize their topology in response to an external stimulus (e.g., temperature, light, or pressure) while maintaining overall connectivity.22 CANs can be divided into two main categories, depending on the bond-exchange reaction mechanism: dissociative and associative systems. In dissociative CANs, dynamic bonds first cleave to generate reactive intermediates, followed by bond reformation. During this intermediate stage, the cross-link density temporarily decreases, leading to a transient loss of network integrity. As a consequence, dissociative systems often exhibit a viscosity drop near the bond dissociation temperature and may behave similarly to thermoplastics above this transition. In contrast, associative CANs undergo bond exchange through a concerted mechanism in which a new covalent bond is formed at a different site before the original bond is broken.23,24 Because bond formation precedes bond cleavage, the cross-link density remains constant throughout the exchange process. This preservation of network connectivity prevents macroscopic depolymerization and enables gradual stress relaxation without a distinct gel–sol transition. Vitrimers belong to this associative category.The fundamental distinction between these two mechanisms is illustrated schematically in Fig. 3: in dissociative systems, all dynamic groups may temporarily exist in an unbound state, whereas in associative systems, the network connectivity is continuously maintained. This mechanistic difference has important consequences for thermal behavior, viscoelastic response, and reprocessability, as discussed in the following sections.
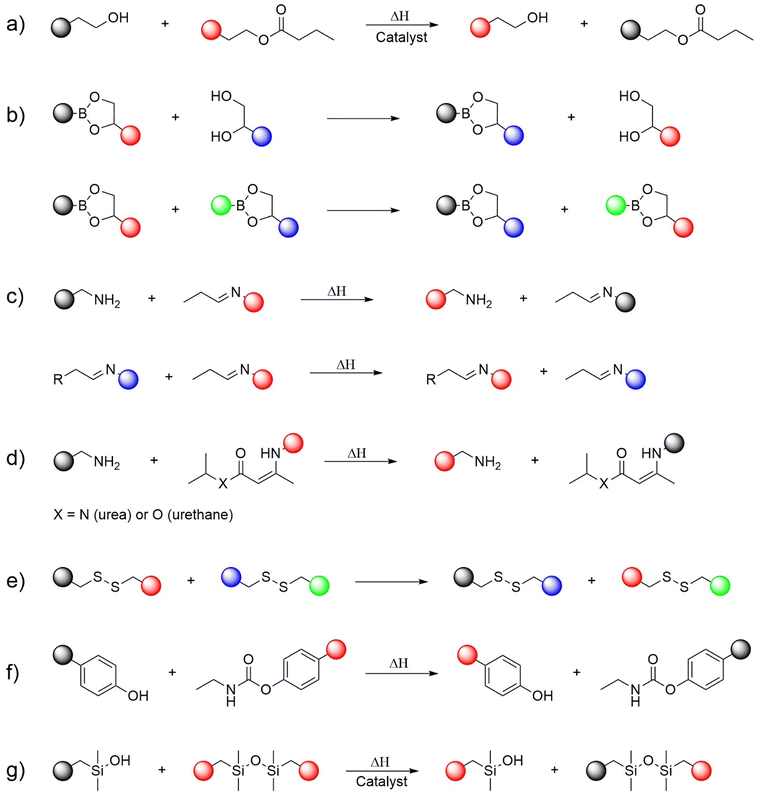
Typical reactions involved in associative CAN systems include the following,20,26 as illustrated in Fig. 4:
(a) Transesterification reaction: an exchange reaction between an alcohol group and an ester, usually occurring at high temperatures involving the use of a catalyst.27
(b) Boronic-ester-based exchange: transesterification of boronic esters or metathesis of boronic esters.28,29
(c) Amino–imine and imine–imine exchange reactions (Schiff base systems): exchange reactions between a free amine and an imine or between an imine and an imine.30,31
(d) Vinylogous urethane/urea exchanges: Michael addition of free primary amines occurring in the absence of a catalyst.32
(e) Disulfide bond exchange: dynamic reshuffling of disulfide bonds via radical-mediated or thiolate-mediated mechanisms, typically occurring at moderate temperatures and without a catalyst.33,34
(f) Transcarbamoylation: an exchange reaction involving reversible urethane (carbamate) linkages, typically occurring between sterically hindered phenolic hydroxyl groups and carbamate moieties without a catalyst.35
(g) Siloxane exchange: relatively fast base-catalyzed siloxane exchange reactions involving siloxane moieties and hydroxyl groups.36,37
Vitrimers rely on associative exchange reactions that preserve cross-link density and allow the network to change topology under heat without loss of structural integrity. At high temperatures, these materials undergo stress relaxation and viscoelastic flow because the rate of exchange reactions controls their viscosity, which follows Arrhenius behavior.25 This enables vitrimers to evolve from a viscoelastic solid to a viscoelastic liquid state in an abrupt way, allowing reprocessability/recyclability of the polymeric system while retaining thermoset-like mechanical performance under service conditions.38 Additionally, as seen in the classification matrix in Fig. 2, vitrimers based on certain bond chemistries, such as imine/amine–imine or disulfide bond exchange, can also be designed to yield cleavable thermoset networks. These systems combine the vitrimers’ thermal reprocessability with the possibility of chemical degradation under specific stimuli, thereby offering complementary end-of-life options and further enhancing the sustainability potential of vitrimeric materials.39
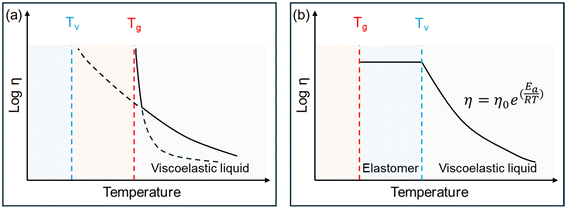
The viscoelastic behavior of vitrimers is governed by two characteristic transition temperatures: (i) the glass transition temperature (Tg), which marks the onset of long-range coordinated segmental motion and separates the glassy and rubbery states of the polymer network; and (ii) the topological freezing transition temperature (Tv), conventionally defined as the temperature at which the viscosity reaches 1012 Pa s, as introduced by Leibler and co-workers.40,41Tv originates from thermally activated dynamic bond exchange reactions within the cross-linked network and corresponds to the transition from a viscoelastic solid to a viscoelastic liquid regime.
As illustrated in Fig. 5, the macroscopic behavior of vitrimers depends on the relative positions of Tg and Tv. When Tg > Tv, the material remains rigid up to Tg and flows as a viscoelastic liquid above this temperature. Conversely, when Tg < Tv, the vitrimer behaves like an elastomer in the range Tg < T < Tv and flows like a viscoelastic liquid for T > Tv, following Arrhenius-type kinetics dictated by the exchange reactions.
Stress relaxation measurements are widely used to characterize vitrimer dynamics. In these measurements, the decay of stress under constant strain provides the characteristic relaxation time (τ), which reflects the rate of network rearrangement. An ideal vitrimer system is capable of complete stress relaxation, reflecting efficient and continuous bond exchange within the network. The activation energy (Ea) of the dynamic exchange process can be determined by fitting temperature-dependent relaxation times to the Arrhenius equation. In some systems, dual relaxation mechanisms have been reported, arising from the coexistence of multiple dynamic bond chemistries within the network structure.42,43 Despite their dynamic nature, vitrimers retain the characteristic solvent resistance of conventional thermosets: they typically swell but do not dissolve in common organic solvents due to their permanent cross-linked structure. For example, Albertini et al. proved the resistance to common solvents like acetone, toluene, hexane and iso-propanol of a bio-based vitrimer system relying on transesterification exchange reactions.44
2.2. Application pathways toward sustainable polymer systems
Since vitrimers can be easily molded upon heating due to the gradual reduction of viscosity with increasing temperature beyond the topological freezing transition temperature, there are several chances for thermal recycling, offering closed-loop circularity in plastics.44–46 Additionally, this property creates new processing opportunities, like extrusion, which is usually applied in standard thermoplastic processing.47 For instance, Taplan et al. developed covalently cross-linked permanent networks producing densely cross-linked vitrimers with pronounced viscoelastic-liquid behavior, and they used these networks for extrusion processing.48 Furthermore, chances for chemical recycling have been created, thanks to the covalent bonds’ dynamic nature: depolymerization and monomer recovery can be accomplished by using a reactive solvent.42 Because of this specific recyclability, these features may contribute to extended service life and improved end-of-life management. Thermally activated self-healing behavior can also originate from the capacity of vitrimers to rearrange their network topology upon heating.49,50 Through the reformation of covalent bonds between the two pieces of material that come into contact, welding can also be readily achieved: the ability to be repeatedly welded thanks to the exchange reactions opens the way towards joining composite parts without adhesives or mechanical fasteners.51 It is interesting to note that covalent bonds can form at the interface, allowing for the welding of dissimilar and even incompatible polymers if they share the same dynamic linkages.52 Eventually, dynamic covalent bonds can be inserted into thermoplastic materials via different approaches. For instance, Van Zee et al. and Röttger et al. inserted dynamic covalent functionalities through post-polymerization reactive extrusion, typically relying on radical-initiated reactions.38,52 Additionally, Lepage et al. designed a bis-diazirine cross-linker capable of generating carbenes under controlled conditions, enabling double C–H insertion and covalent network formation in otherwise inert polymers.533. Approaches to sustainable bio-based epoxy vitrimers
Conventional thermoset polymers are often synthesized from fossil-based precursors that raise significant concerns from both regulatory and life-cycle perspectives, particularly regarding toxicity, greenhouse gas emissions, and end-of-life management.54 For example, diglycidyl ether of bisphenol A (DGEBA), the most common epoxy resin used in high-value applications, is synthesized from bisphenol A and epichlorohydrin in the presence of sodium hydroxide. Bisphenol A is a well-documented endocrine disruptor associated with adverse effects on immune and reproductive functions, as well as alterations in neurological development.55,56 As a result, bisphenol A and related substances have become targets of increasing regulatory scrutiny worldwide. Substituting bisphenol A-derived epoxy resins therefore represents not only a materials innovation challenge but also a strategic pathway to reduce hazard profiles and improve regulatory compliance across product life cycles. Similar issues arise with conventional epoxy hardeners, which are mainly fossil-based and often hazardous.From a sustainability and life-cycle assessment (LCA) standpoint, biomass-derived resources, including lignin, cellulose, carbohydrates, vegetable oils, terpenes, and polyphenols, offer attractive alternatives as renewable feedstocks for thermoset synthesis. These resources can reduce dependence on non-renewable carbon sources and, when responsibly sourced, may lower cradle-to-gate greenhouse gas emissions. Table 1 summarizes key natural resources, representative platform chemicals, and functional groups relevant to bio-based thermoset development. Among these, lignin, carbohydrates and vegetable oils are particularly important platforms for bio-based epoxy resins due to their availability, functionality, and compatibility with existing polymer processing technologies.57
| Resources | Related chemicals | Functional groups |
|---|---|---|
| Carbohydrates | Itaconic acid, furfuryl amine, isosorbide | Carboxyl, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond, furan group, alcohols, diheterocycles C double bond, furan group, alcohols, diheterocycles |
| Lignin | Vanillin, eugenol, guaiacol/bisguaiacol | Aldehyde, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond C double bond |
| Vegetable oils | Glyceride, cardanol, soybean oil, linseed oil | Ester group, unsaturated aliphatic chain |
As stated above, integrating bio-based platform chemicals into vitrimer design also advances sustainability at the molecular scale. Using properly functionalized bio-based chemicals (or, in some cases, minimally modified bio-derived compounds) with chemical groups capable of forming dynamic covalent bonds helps decrease the environmental impact of thermosets by both reducing the potential impact of raw materials and improving end-of-life options. From an LCA perspective, these features can substantially extend service life, reduce material losses, and decrease the demand for virgin polymer production, thereby lowering cumulative environmental impacts. Ultimately, such strategies align with circular-economy principles by minimizing greenhouse gas emissions, reducing waste generation, and mitigating biodiversity loss through prolonged resource circulation.58
In the following subsections, representative examples of bio-based vitrimer systems synthesized from the natural feedstocks listed in Table 1 are presented. These case studies highlight how renewable resources, when combined with dynamic covalent chemistry, can contribute to the development of reprocessable thermoset networks with improved environmental profiles and demonstrated application potential, offering concrete pathways toward scalable and policy-relevant sustainable polymer technologies. It should be noted that Table 1 does not aim to provide an exhaustive overview of all renewable resources suitable for vitrimer synthesis. Instead, it highlights the most widely investigated and industrially relevant biomass platforms, such as carbohydrates, lignin derivatives, and vegetable oils, that have recently led to vitrimer systems with demonstrated functional applications. Other renewable building blocks and niche bio-derived monomers have been reported in the literature but fall outside the scope of the present application-oriented discussion. Chemicals primarily used for thermoplastic production are likewise not included, as they are beyond the focus of this review.
3.1. Carbohydrates
Carbohydrates are naturally occurring organic compounds derived from plant and animal sources and are broadly classified as simple sugars or complex polysaccharides. Owing to their high functionality and structural diversity, carbohydrate-derived molecules have attracted significant attention as renewable building blocks for thermoset and vitrimer synthesis. Representative examples of carbohydrate-derived platform chemicals employed in cross-linked thermoset vitrimer systems are depicted in Fig. 6. It should be noted that this section focuses on carbohydrate-derived molecules that have been utilized in permanent, cross-linked vitrimer networks. Polyether- or polyethylene glycol (PEG)-based motifs, which are more commonly associated with thermoplastic or elastomeric architectures, are not discussed in detail unless incorporated into covalently cross-linked vitrimer systems, as they fall outside the primary thermoset-oriented scope of this review. | ||
| Fig. 6 Representative carbohydrate-derived platform chemicals used in thermoset vitrimer systems: itaconic acid, furfuryl amine, and isosorbide. | ||
Itaconic acid is a bio-based monounsaturated organic acid obtained through the fermentation of sugars by the filamentous fungus Aspergillus terreus.59 It is a potential alternative to acrylic and methacrylic acids and was included by the U.S. Department of Energy in its list of the most promising chemical platforms derived from sugars.60 Its dual functionality (i.e., two carboxylic acid groups and an aliphatic double bond) makes it particularly suitable for dynamic covalent networks. In vitrimer design, the transesterification pathway is frequently exploited. For instance, Albertini et al. used itaconic acid to prepare a vitrimer based on transesterification through the direct reaction between itaconic acid and a bio-based epoxy resin mediated by a zinc catalyst.44 Pan et al. produced natural peach gum-based epoxy vitrimers with superior mechanical properties and thermal stability by incorporating peach gum and itaconic acid and reacting them with a bio-based epoxy resin.61 Itaconic acid has also been used as a structural element of the epoxy resin itself. Yang et al. synthesized an itaconic acid-derived epoxy resin and cross-linked it by using a boron-containing cross-linker. The cured vitrimer exhibited rapid stress relaxation (54 s at 150 °C) with low activation energy (Ea = 31.6 kJ mol−1) and excellent repairing, welding, reprocessing, and recycling capabilities.62 Collectively, these examples highlight the chemical versatility of itaconic acid for enabling vitrimer functionality through different molecular architectures.
Another relevant carbohydrate-derived compound is furfuryl amine, although its application in vitrimer systems remains almost unexplored. However, few examples of furfuryl amine used to produce dissociative CANs exist in the literature.63,64 These studies demonstrate how the reactivity of the furan ring can be potentially leveraged to introduce reversible linkages into cross-linked networks.
Isosorbide, a renewable sugar-based chemical, is another key carbohydrate-based molecule and a well-known building block for bio-based thermoset production.65 Although its application in vitrimers is less common, recent work has shown promising potential. A high Tg and degradable isosorbide-based poly-benzoxazine vitrimer was obtained by Adjaoud et al. A two-step synthesis was investigated (see Fig. 7): first, isosorbide was esterified with phloretic acid and afterwards reacted with mono-ethanolamine and/or furfuryl amine and paraformaldehyde. In this way, a chemical structure including a tertiary amine capable of catalyzing the transesterification reaction was obtained, leading to fast dynamic bond exchange. Furthermore, the presence of isosorbide, furan and benzoxazine moieties resulted in high Tg values (from 143 to 193 °C) for the final vitrimer, combined with degradability under relatively mild conditions.66 In another approach, Yang et al. used a post-cross-linking technique to produce bio-based vinylogous urethane epoxy thermosets from isosorbide. Direct functionalization of isosorbide resulted in isosorbide-bis-acetoacetate, which could then react with primary amines present in diamine and triethylenetetramine chemicals to produce linear prepolymers with vinylogous urethane linkages. After that, the secondary amines in the linear prepolymer were used to create covalent cross-links by ring-opening the epoxy groups of epoxidized vanillin. The bio-based vitrimers produced by combining various diamine types had elongation at break between 9.5 and 150% and tensile strength between 31.5 and 114.6 MPa, demonstrating the excellent versatility of the system. The incorporation of vinylogous urethane bonds conferred recyclability: the thermoset was immersed in ethanol with benzylamine and heated for 12 h at 50 °C, leading to complete dissolution (see Fig. 8).67
 | ||
| Fig. 7 Synthesis reactions of a high Tg and degradable isosorbide-based poly-benzoxazine vitrimer. Reproduced from ref. 66 with permission from American Chemical Society, Copyright © 2021. | ||
 | ||
| Fig. 8 Chemical degradation of bio-based vinylogous urethane epoxy thermosets derived from isosorbide by immersion in ethanol for 24 h at room temperature with benzylamine. Reproduced from ref. 67 with permission from American Chemical Society, Copyright © 2023. | ||
Overall, these studies demonstrate that carbohydrate-derived molecules, from fermentation products like itaconic acid to rigid sugar-based monomers such as isosorbide, provide versatile platforms for creating dynamic, reprocessable vitrimer networks. Their diverse functionalities support multiple exchange chemistries, highlighting carbohydrates as a promising feedstock class for sustainable vitrimer development.
3.2. Lignin and its derivatives
Lignin, whose representative structure is shown in Fig. 9, is the second main polymer found in nature, accounting for approximately 25–35% of the organic matrix of wood and lignocellulosic biomass. It is an abundant byproduct of the paper and pulp industry, where it is still predominantly combusted for energy recovery rather than valorized into higher-value materials.68 Structurally, lignin is a complex, three-dimensional aromatic biopolymer composed primarily of guaiacyl, syringyl, and p-hydroxyphenyl units interconnected through various ether (e.g., β-O-4) and carbon–carbon linkages. This structural diversity results in a high density of phenolic and aliphatic hydroxyl groups, methoxy substituents, and aromatic functionalities, offering multiple reactive sites for chemical modification. Its high carbon content and aromatic backbone make lignin an ideal candidate as a platform for the development of thermoset materials with high bio-based carbon content, thermal stability, and mechanical robustness. However, the lignin structure varies greatly with the source and extraction conditions, leading to variability in molecular weight, functional group distribution, and reactivity.69,70 To overcome issues related to heterogeneity and limited processability, refining, depolymerization, and targeted functionalization strategies have been extensively investigated.71 | ||
| Fig. 9 Chemical structures of a representative lignin fragment and selected lignin-derived platform chemicals: vanillin, eugenol and guaiacol. | ||
Beyond serving as a source of aldehyde or alkene functionalities, lignin can be directly incorporated into polymer networks or selectively depolymerized into well-defined aromatic platform chemicals such as vanillin, eugenol, and guaiacol. These derivatives constitute versatile building blocks for epoxy resins, phenolic resins, polyurethanes, and vitrimer systems. Comprehensive reviews have examined lignin-derived thermosets and vitrimers from a chemistry-centered perspective.71,72 In the present work, emphasis is placed on lignin-derived molecules that have recently enabled bio-based vitrimer systems with demonstrated application potential. Accordingly, vanillin-, eugenol-, and guaiacol-based vitrimers, whose structures are depicted in Fig. 9, are discussed below.
Vanillin (4-hydroxy-3-methoxybenzaldehyde) is the most well-known product of lignin depolymerization. It is produced by the controlled oxidation of lignin at strongly alkaline pH and high temperatures with air, oxygen or metallic oxides used as oxidants.73,74 In this way, degradation of lignin occurs, producing vanillin and other by-products. The resulting vanillin can be used in the pharmaceutical sector, but it can also be used as a raw chemical for the synthesis of bio-based epoxy resins or cross-linkers with excellent mechanical and thermal properties, thanks to the high concentration of aromatic rings in the products’ cross-linked structure.75,76 Derivatives of vanillin have been used successfully as both polymer resins and cross-linkers in the production of bio-based vitrimers.
When used as cross-linkers, vanillin-based imine hardeners have demonstrated the ability to deliver thermomechanical properties comparable to those of conventional fossil-derived counterparts while introducing dynamic covalent functionality. For example, Memon et al. synthesized an imine-containing hardener from vanillin and compared it to an imine-containing hardener derived from petroleum-based p-hydroxy benzaldehyde for curing DGEBA. Comparable Tg (>120 °C), tensile strength and modulus (>60 MPa and >2500 MPa, respectively) and good solvent resistance in organic solvents, aqueous acids, and alkaline solutions were obtained with the two hardeners; the resulting cured epoxy resins showed good reprocessability and degradability.77 In a related study, a vanillin-imine-containing hardener was synthesized by using a simple one-step procedure combining bio-based vanillin and xylylene diamine. The flexural strength (125 MPa) of the resulting vitrimer was similar to that of commercial counterparts, and the storage modulus (up to 3448 MPa) was noticeably higher. The cross-linked vitrimer was able to relax stresses (relaxation time of 35 s at 130 °C and Ea = 119 kJ mol−1) and was recycled repeatedly with recovery of flexural characteristics even after three reprocessing cycles. Additionally, despite its resistance to common organic solvents, within 12 h at 50 °C, the epoxy vitrimer degraded rapidly under acidic conditions, allowing chemical recycling.78 Representative imine–imine and amine–imine exchange reactions underpinning vanillin-based vitrimer networks are illustrated in Fig. 11.79 Additionally, Monteserin et al. developed a series of sustainable vitrimers through the reaction between a vanillin-derived Schiff base hardener and epoxidized linseed oil. The scheme of the condensation reaction forming the imine group is reported in Fig. 10. Different epoxy-to-hardener ratios were tested, with the stoichiometric ratio yielding the highest glass transition temperature (Tg = 125 °C) and thermal degradation resistance (Tdeg = 329.5 °C). All vitrimers showed good reprocessability via dynamic imine bond exchange, maintaining or slightly improving their mechanical and thermal properties after 10 cycles. Additionally, the materials demonstrated acid solubility, supporting their recyclability. Flax fiber-reinforced vitrimer composites were also prepared, showing strong fiber-matrix adhesion without surface treatment, along with suitable thermal and mechanical performance for lightweight engineering applications.80
 | ||
| Fig. 10 Scheme of the condensation reaction forming the imine group in a linseed oil-based vitrimer. Adapted from ref. 80 with permission from Elsevier Ltd, Copyright © 2023. | ||
 | ||
| Fig. 11 Reactions involved in vanillin-based vitrimers based on imine–imine and amine–imine exchange reactions. Adapted from ref. 79 with permission from American Chemical Society, Copyright © 2024. | ||
Vanillin can also be epoxidized and used as a polymer matrix for vitrimer production. For example, Guggari et al. investigated the reaction between a bio-based epoxy resin derived from vanillin and an aliphatic disulfide (cystamine) used as a cross-linker, demonstrating high reactivity and low relaxation times (22 s at 170 °C with an activation energy of 53 kJ mol−1) with possible suitability for a wide range of applications.45 Eventually, Yu et al. used a mono-glycidyl derivative of vanillin to prepare vanillin-based epoxy vitrimers through reaction with isophorone diamine. The resulting vitrimers exhibited mechanical properties and thermal stability comparable with those of bisphenol-A epoxy cured with amine hardeners, with a Tg of 121 °C. Furthermore, the high content of dynamic imine bonds in the networks resulted in fast stress relaxation (50 s) at a temperature 30 °C higher than Tg. The properties were maintained after each reprocessing cycle, and thanks to the Schiff structure, the studied vitrimer was also degradable under acidic conditions; this result is illustrated in Fig. 12.81
 | ||
| Fig. 12 Epoxidized vanillin–imine exchange reaction in dynamic epoxy networks. Reproduced from ref. 81 with permission from Elsevier Ltd, Copyright © 2019. | ||
Eugenol, which constitutes around 80% of clove oil, is an example of a non-toxic renewable phenol and can be synthesized through lignin depolymerization. Its chemical structure allows easy modification, making it suitable for obtaining a wide and versatile range of bio-based monomers.82,83 The terminal olefin group serves as a synthetic handle for polymerization and chemical modification, while the aliphatic chain and methoxy group have a significant impact on the physical properties of polymers generated from eugenol. For example, it has been extensively used to design polymer networks exploiting self-metathesis and olefin metathesis coupling reactions.84 Furthermore, its hydroxyl and alkene functionalities can be used to prepare bio-based epoxy thermosets with high performance, heat resistance and flame-retardant properties thanks to the aromatic ring in the molecular structure.85–88 Eugenol has also been used as a natural starting material to prepare bifunctional unsaturated monomers (polyethers and polycarbonates). The resulting diene-terminated ethers and carbonates can then be subjected to step-growth acyclic diene metathesis (ADMET) polymerization obtaining unsaturated polymers.89 These features make eugenol an interesting building block for vitrimeric structures. For instance, Niu et al. synthesized eugenol-based epoxy monomers for the preparation of transparent UV- and thermally curable covalent adaptable thermosets with UV-shielding properties. By adjusting the proportion of rigid to flexible structural units, the thermomechanical properties of the eugenol-based thermosets were controlled. The resulting vitrimer exhibited efficient stress relaxation (Ea = 119–127 kJ mol−1) and showed thermally activated repair capability and shape memory behavior arising from dynamic bond exchange. Furthermore, the material could be reprocessed by hot pressing, maintaining 85% of the initial mechanical properties.90 Similarly, Ocando et al. used epoxidized eugenol for producing CANs based on disulfide bonds. After thermal cross-linking with a disulfide bond-containing amine, the thermoset was successfully weldable, healable and recyclable in the absence of catalysts under thermal stimulus, as shown in Fig. 13. Notably, the material also displayed a higher modulus and improved thermal properties compared to its fossil-based counterpart.91 In a similar way, a disulfide vitrimeric material based on a bio-based cross-linker with disulfide bonds and epoxidized eugenol was obtained by Roig et al. The authors studied the effect of the addition of various fractions of tris(2-aminoethyl) amine to enhance the cross-linking density and increase the proportion of tertiary amines, which may act as catalysts in disulfide bond exchange. The resulting material was able to rapidly relax 63% of the initial stress in less than 8.5 min, and the authors demonstrated the catalytic activity of the tertiary amine in the exchange reaction.92 Finally, Tannert et al. investigated the influence of epoxy impurities, such as mono- and di-epoxidized eugenol derivatives, on vitrimer performance and showed that variations in epoxy purity did not significantly affect the dynamic exchange reactions.93
 | ||
| Fig. 13 Cutting and welding by hot pressing at 230 °C of an epoxidized eugenol-based vitrimer. The sample was manually bent to show cohesion between the welded pieces. Reproduced from ref. 91 with permission from Elsevier Ltd, Copyright © 2020. | ||
Lastly, guaiacol is a non-toxic monosubstituted bio-based phenol, suitable as an alternative to more commonly used fossil-based phenols. Its condensation with vanillyl alcohol under acidic conditions yields a bisphenolic compound (bisguaiacol), which can be used as a substitute for fossil-based bisphenols in thermoset and epoxy resin synthesis.94 Several examples of guaiacol-derived vitrimers have been reported. For instance, thermally healable thiol-acrylate vitrimers, reprocessable via transesterification reactions, were synthesized by Wong et al. starting from guaiacol and bisguaiacol F with thiol and phosphorus cross-linkers. The authors demonstrated how the additional methoxy groups in the lignin-derived vitrimers promoted inter-molecular interactions during UV curing by acting as additional hydrogen-bond acceptors, enhancing the resulting cross-linking density compared to their fossil-based counterparts (phenol and bisphenol F), improving robustness and toughness. The higher cross-linking density helped lignin-derived vitrimers in the thermal healing process probably due to the denser network facilitating bond exchange (Ea = 55 kJ mol−1, relaxation time at 110 °C equal to ca. 500 s and Tv = −7 °C), demonstrating good recovery of tensile properties.95 Another interesting example was reported by Liu et al., who developed a self-healable bio-based epoxy vitrimer with a high Tg by reacting a tri-epoxy monomer synthesized from vanillin and guaiacol, as illustrated in Fig. 14. The resulting resin was thermally cured using an anhydride cross-linker in the presence of zinc as a catalyst. The cured epoxy thermoset showed similar mechanical properties and thermal stability comparable to those of the cured epoxy resin from bisphenol A, while also demonstrating good reprocessability.96
 | ||
| Fig. 14 Synthetic route for a tri-epoxy resin using vanillin and guaiacol as raw chemicals. Adapted from ref. 96 with permission from American Chemical Society, Copyright © 2018. | ||
From a structure–property perspective, vanillin-, eugenol-, and guaiacol-derived building blocks impart distinct effects on vitrimer dynamics due to differences in aromatic substitution and functional group chemistry. Vanillin-based structures, featuring aldehyde and phenolic groups on a rigid aromatic ring, readily form imine linkages that combine high Tg with fast associative exchange kinetics, enabling efficient stress relaxation without sacrificing stiffness. In contrast, eugenol-derived systems incorporate a flexible aliphatic side chain and an alkene moiety, which reduce network rigidity and Tg while enhancing molecular mobility, thereby facilitating dynamic bond exchange at lower temperatures and improving toughness and shape-memory behavior. Guaiacol-based building blocks offer a balance between rigidity and reactivity: their methoxy and hydroxyl groups promote strong intermolecular interactions and high cross-link density while still enabling dynamic exchange through transesterification or disulfide mechanisms. Overall, these lignin-derived aromatics provide a versatile toolbox for tuning vitrimer networks, allowing controlled adjustment of mechanical performance, relaxation kinetics, and processing windows through molecular design.
3.3. Vegetable oils
Various vegetable oils derived from seeds, plants, and nuts have been widely explored as renewable feedstocks for the synthesis of bio-based thermosets, offering sustainable alternatives to conventional, often toxic, fossil-derived chemicals. Well-known examples include soybean oil, linseed oil, castor oil, palm oil, tung oil, and cardanol oil. Owing to their inherent functionality and availability, these oils have also found extensive application in the vitrimer field, where they serve as precursors for both resin matrices and curing agents. The chemical structures of representative vegetable oils are depicted in Fig. 15. | ||
| Fig. 15 Main vegetable oil platforms used for vitrimers: soybean oil, linseed oil, castor oil, tung oil and cardanol. | ||
Soybean oil is probably the most used vegetable oil for vitrimer production. The presence of double bonds along the aliphatic chains makes it suitable for epoxidation reactions. Once epoxidized, it can be used in combination with other bio-based hardeners to make CANs based, for example, on transesterification, disulfide bond and imine–imine/amine bond exchanges.97 For instance, Di Mauro et al. used epoxidized soybean oil in combination with epoxidized linseed oil and itaconic acid as a hardener to obtain fully bio-based materials with a high bio-based carbon content. The materials demonstrated good recyclability via both mechanical and chemical methods thanks to transesterification reactions. The fully reprocessed bio-based thermosets retained nearly identical thermomechanical properties and chemical structures.98 Nepomuceno et al. prepared a fully bio-based epoxy vitrimer starting from epoxidized soybean oil with a eutectic hardener made of L-tartaric acid and ethyl lactate in aqueous solution, resulting in a fully bio-based adaptable covalent network that can be reprocessed via transesterification reactions without requiring an external catalyst. Furthermore, the presence of free hydroxyl groups in tartaric acid promoted the ring-opening of epoxides and transesterification reactions, thereby improving both the curing process and the kinetics of covalent bond exchange.99 Li et al. synthesized catalyst-free fully bio-based hydroxyester vitrimers using epoxidized soybean oil and acetylated starch succinate monoesters as raw materials. The epoxidized soybean oil was cross-linked with acetylated starch succinate monoesters (ratio of epoxy groups![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) carboxyl groups = 2
carboxyl groups = 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) to form a dynamically cross-linked network where the long segments with unreacted epoxy groups from epoxidized soybean oil acted as plasticizers. Consequently, a maximum strain of 230% was achieved, helping to avoid the retrogradation and brittleness commonly observed in starch-based materials. Moreover, the mechanical properties remained stable even after three reprocessing cycles.100 Fully bio-based vitrimers with high biomass content (up to 77.1%) were also developed by Jeon et al., who synthesized bio-based di-epoxy, tri-epoxy, and soybean oil epoxy monomers derived from natural resources including 2,5-furandicarboxylic acid, protocatechuic acid and soybean oil, respectively, and cured them with a tetra-functional thiol cross-linker derived from α-lipoic acid. The resulting thermosets demonstrated impressive mechanical properties, with tensile strengths of up to 55.76 MPa and stress relaxation behavior (with a minimum activation energy for dynamic bond exchange equal to 123.05 kJ mol−1). They reprocessed the vitrimers through thiol-epoxy reactions and transesterification, representing a major step forward in developing sustainable alternatives to conventional petroleum-based thermosets.101 An interesting fully bio-based vitrimer was also prepared by Altuna et al. by cross-linking epoxidized soybean oil with an aqueous citric acid solution in the absence of any catalysts. The cross-linked polymer was characterized by a network of β-hydroxyester groups and a fraction of vicinal diol groups. Stress relaxation and lap shear tests evidenced the vitrimeric properties of the system with an Ea = 106 kJ mol−1 and disappearance of fracture lines after mending the samples at 160 °C by compression molding.97,102 Furthermore, Veloso-Fernández et al. synthesized a sustainable epoxy vitrimer by cross-linking epoxidized soybean oil with a vanillin-derived Schiff base aliphatic diamine as a dynamic hardener with 1,2-dimethylimidazole as an accelerator. The resulting vitrimer was compared with an equivalent commercial epoxy resin (poly(bisphenol A-co-epichlorohydrin) glycidyl end-capped). A mixture of the two epoxies (70
1) to form a dynamically cross-linked network where the long segments with unreacted epoxy groups from epoxidized soybean oil acted as plasticizers. Consequently, a maximum strain of 230% was achieved, helping to avoid the retrogradation and brittleness commonly observed in starch-based materials. Moreover, the mechanical properties remained stable even after three reprocessing cycles.100 Fully bio-based vitrimers with high biomass content (up to 77.1%) were also developed by Jeon et al., who synthesized bio-based di-epoxy, tri-epoxy, and soybean oil epoxy monomers derived from natural resources including 2,5-furandicarboxylic acid, protocatechuic acid and soybean oil, respectively, and cured them with a tetra-functional thiol cross-linker derived from α-lipoic acid. The resulting thermosets demonstrated impressive mechanical properties, with tensile strengths of up to 55.76 MPa and stress relaxation behavior (with a minimum activation energy for dynamic bond exchange equal to 123.05 kJ mol−1). They reprocessed the vitrimers through thiol-epoxy reactions and transesterification, representing a major step forward in developing sustainable alternatives to conventional petroleum-based thermosets.101 An interesting fully bio-based vitrimer was also prepared by Altuna et al. by cross-linking epoxidized soybean oil with an aqueous citric acid solution in the absence of any catalysts. The cross-linked polymer was characterized by a network of β-hydroxyester groups and a fraction of vicinal diol groups. Stress relaxation and lap shear tests evidenced the vitrimeric properties of the system with an Ea = 106 kJ mol−1 and disappearance of fracture lines after mending the samples at 160 °C by compression molding.97,102 Furthermore, Veloso-Fernández et al. synthesized a sustainable epoxy vitrimer by cross-linking epoxidized soybean oil with a vanillin-derived Schiff base aliphatic diamine as a dynamic hardener with 1,2-dimethylimidazole as an accelerator. The resulting vitrimer was compared with an equivalent commercial epoxy resin (poly(bisphenol A-co-epichlorohydrin) glycidyl end-capped). A mixture of the two epoxies (70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 = synthesized
30 = synthesized![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) commercial) provided the best results with high thermo-mechanical properties while retaining the material's inherent reprocessability and recyclability (hot pressing at 150 °C for 60 min at 10 bar), despite the decrease in Tg values with increasing bio-based epoxy resin content in the formulations.103 An equivalent study was conducted by Zhao et al., who studied a sustainable vitrimer based on epoxidized soybean oil and a vanillin-based hardener with 1,2-dimethylimidazole as an accelerator. They also proved excellent stress relaxation, resulting in Ea = 39.67 kJ mol−1, and the reprocessability and weldability of the epoxy vitrimer, as shown in Fig. 16.104 Eventually, Thomas et al. developed vitrimers using epoxidized norbornylized seed oils, specifically derived from soybean and linseed oils for composite applications. To enhance the sustainability of the system, carbonized biomass derived from sorghum was incorporated as a filler, contributing to increased bio-based content, reduced cost, and lower weight. These sustainable materials were cured with an acid hardener containing a dynamic disulfide bond (dithiodibenzoic acid). Additionally, glass-fiber-reinforced composites were prepared with the studied vitrimer resin. The thermosets maintained their cross-linking degree and strong tensile properties after reprocessing.105
commercial) provided the best results with high thermo-mechanical properties while retaining the material's inherent reprocessability and recyclability (hot pressing at 150 °C for 60 min at 10 bar), despite the decrease in Tg values with increasing bio-based epoxy resin content in the formulations.103 An equivalent study was conducted by Zhao et al., who studied a sustainable vitrimer based on epoxidized soybean oil and a vanillin-based hardener with 1,2-dimethylimidazole as an accelerator. They also proved excellent stress relaxation, resulting in Ea = 39.67 kJ mol−1, and the reprocessability and weldability of the epoxy vitrimer, as shown in Fig. 16.104 Eventually, Thomas et al. developed vitrimers using epoxidized norbornylized seed oils, specifically derived from soybean and linseed oils for composite applications. To enhance the sustainability of the system, carbonized biomass derived from sorghum was incorporated as a filler, contributing to increased bio-based content, reduced cost, and lower weight. These sustainable materials were cured with an acid hardener containing a dynamic disulfide bond (dithiodibenzoic acid). Additionally, glass-fiber-reinforced composites were prepared with the studied vitrimer resin. The thermosets maintained their cross-linking degree and strong tensile properties after reprocessing.105
 | ||
| Fig. 16 Sustainable epoxy vitrimer synthesis with reprocessability and weldability properties. Reproduced from ref. 104 with permission from American Chemical Society, Copyright © 2020. | ||
Linseed oil has been standing out as a promising alternative to petroleum-based resources due to its distinctive composition, particularly its high linolenic content (56%). Its significant degree of unsaturation, averaging 6.6 double bonds per triglyceride, makes it especially valuable as a biomass-derived building block for synthesizing prepolymers and polymers.106 As for the other vegetable oils, linseed oil has found some applications in epoxy vitrimer synthesis too. Di Mauro et al. synthesized reprocessable thermosets combining disulfide metathesis and transesterification using epoxidized linseed oil. The synthesis of epoxidized linseed cross-linked structures and their network rearrangement by simultaneous exchange reactions is depicted in Fig. 17. Interestingly, the authors tested a series of 10 different initiators capable of initiating the cross-linking reaction while also influencing the reprocessing step. Thermosets synthesized using imidazole as the initiator demonstrated excellent reprocessability. Furthermore, they exhibited complete chemical recyclability under basic conditions at 80 °C within three days. The reprocessed materials retained performance comparable to that of the original ones, even after ten reprocessing cycles.107 The same authors studied a series of novel bio-based epoxy thermosets obtained by polymerizing epoxidized vegetable oils (including linseed oil) with a disulfide aromatic dicarboxylic acid. The obtained thermosets were able to be chemically dissolved in a solution of 2-mercaptoethanol in DMF. Notably, the authors also observed that increasing the functionality of the epoxidized vegetable oils led to enhanced reactivity, greater cross-linking density, and improved overall performance, with Tg ranging from 34 °C to 111 °C.108 Nardi et al. explored boronic ester vitrimers containing lignin microparticles for adhesive applications. The adhesive was prepared from epoxidized linseed oil and a boronic ester dithiol cross-linker. Lignin microparticles (20–50 wt%) were used as reinforcing fillers to enhance the thermal and mechanical properties of the adhesive. The process was solvent- and catalyst-free, which was advantageous from environmental and industrial perspectives. The obtained vitrimers exhibited lap shear strengths of 9–17 MPa when tested on aluminum, stainless steel, and wood and demonstrated good to excellent rewelding potential, even after multiple bonding cycles. Furthermore, the vitrimer maintained 83% of its initial adhesion strength after 24 hours in water, indicating water resistance, although it demonstrated enhanced biodegradability in soil and seawater due to the presence of lignin.109
 | ||
| Fig. 17 Synthesis of epoxidized linseed oil/2,2′-dithiodibenzoic acid cross-linked structures and network rearrangement by simultaneous exchange reactions: disulfide metathesis and transesterification. Reproduced from ref. 107 with permission from American Chemical Society, Copyright © 2020. | ||
Castor oil is a vegetable oil extracted from castor seeds, which contain 45% to 60% oil content.110 Its use in polymer production is well documented, and it has recently attracted attention for vitrimer production too.106,111 Few examples are discussed in this section. Zhang et al. recently developed repairable and reprocessable bio-based polyurethanes with robust mechanical properties using hindered urea bonds derived from castor oil. The relaxation times of the prepared vitrimers were found to be short, ranging from 12.3 s to 221 s at 100 °C, and the vitrimers showed good healing efficiency of 88.9–100% at 100 °C for 10 min and recyclability too. The shape recovery was measured to be 81.3%. Interestingly, the polymers could undergo rapid and reversible solid/liquid transformation under cooling and heating treatments.112 A castor oil-based epoxy vitrimer based on dual dynamic bonds (disulfide and ester bonds) was also prepared by Zhang et al. Methacrylated castor oil was used as the epoxy prepolymer, glycerol methacrylate as a reactive diluent, and itaconic acid and 4,4′-dithiodiphenylamine as curing agents. A 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 molar ratio between the two cross-linkers was found to provide the most balanced comprehensive performance with a storage modulus of 1715 MPa, a tensile strength of 16.8 MPa and a Tg of 44.6 °C. The activation energy of the reversible exchange reactions was found to be 89.3 kJ mol−1. The vitrimer demonstrated high thermally activated repair efficiency (94.6%) and efficient reprocessability under heat and pressure, together with confirmed chemical degradability.113 Additionally, vitrimers derived from castor oil were synthesized by Zhu et al., who prepared fully bio-based catalyst-free vinylogous urethane vitrimers using aminated DL-limonene as a cross-linker, demonstrating the possibility of generalizing the used synthetic procedure to other bio-based raw materials rich in double bonds (Fig. 18). A polymer film was obtained after cross-linking at 25 °C for 48 h, and it was able to be thermally reprocessed (130 °C and 6 MPa for 30 min), exhibited good shape-memory properties, and showed complete dissolution after immersion in benzylamine at 120 °C for 30 min. Furthermore, a 95% gel fraction in THF/acetone (v/v = 1
5 molar ratio between the two cross-linkers was found to provide the most balanced comprehensive performance with a storage modulus of 1715 MPa, a tensile strength of 16.8 MPa and a Tg of 44.6 °C. The activation energy of the reversible exchange reactions was found to be 89.3 kJ mol−1. The vitrimer demonstrated high thermally activated repair efficiency (94.6%) and efficient reprocessability under heat and pressure, together with confirmed chemical degradability.113 Additionally, vitrimers derived from castor oil were synthesized by Zhu et al., who prepared fully bio-based catalyst-free vinylogous urethane vitrimers using aminated DL-limonene as a cross-linker, demonstrating the possibility of generalizing the used synthetic procedure to other bio-based raw materials rich in double bonds (Fig. 18). A polymer film was obtained after cross-linking at 25 °C for 48 h, and it was able to be thermally reprocessed (130 °C and 6 MPa for 30 min), exhibited good shape-memory properties, and showed complete dissolution after immersion in benzylamine at 120 °C for 30 min. Furthermore, a 95% gel fraction in THF/acetone (v/v = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) was determined. Relaxation times ranging from 179 to 595 s were recorded as the temperature increased from 70 to 130 °C, validating the temperature dependence of the exchange reactions in the vitrimer network. An activation energy of 26 kJ mol−1 was determined, which is generally lower than those reported for previously described vinylogous urethane vitrimers.114 Eventually, Shao et al. synthesized a castor oil-derived vitrimer incorporating dual dynamic networks through carefully designing a molecule based on ester–imine bonds and an aromatic Schiff base-conjugated cross-linker. These vitrimers enabled controlled self-healing triggered by photothermal conversion with an efficiency of 100%. The activation energy of the dynamic bond exchange was found to be 37.25 kJ mol−1, and the vitrimer exhibited a Tv of 123.13 °C. The authors found favorable dielectric properties, presenting a novel approach for the design of biomass-based epoxy vitrimers for electronic applications.115
1) was determined. Relaxation times ranging from 179 to 595 s were recorded as the temperature increased from 70 to 130 °C, validating the temperature dependence of the exchange reactions in the vitrimer network. An activation energy of 26 kJ mol−1 was determined, which is generally lower than those reported for previously described vinylogous urethane vitrimers.114 Eventually, Shao et al. synthesized a castor oil-derived vitrimer incorporating dual dynamic networks through carefully designing a molecule based on ester–imine bonds and an aromatic Schiff base-conjugated cross-linker. These vitrimers enabled controlled self-healing triggered by photothermal conversion with an efficiency of 100%. The activation energy of the dynamic bond exchange was found to be 37.25 kJ mol−1, and the vitrimer exhibited a Tv of 123.13 °C. The authors found favorable dielectric properties, presenting a novel approach for the design of biomass-based epoxy vitrimers for electronic applications.115
 | ||
| Fig. 18 Vitrimer derived from castor oil and DL-limonene: (a) synthetic route of the vinylogous urethane vitrimer, (b) synthesis of the aminated DL-limonene cross-linker, and (c) dissolution reaction in benzylamine at 120 °C. Adapted from ref. 114 with permission from Elsevier Ltd, Copyright © 2020. | ||
Tung oil is a nonedible oil derived from the seeds of the tung tree, and, like other vegetable oils, it has been used for the preparation of vitrimers. Chen et al. used tung maleic anhydride and epoxidized tung oil ethanolamides synthesized from tung oil to prepare a vitrimer based on dynamic hydroxyl–ester exchange reactions, as depicted in Fig. 19. The so-obtained vitrimer showed fast and excellent healing performance under thermal stimulus and good reprocessing efficiency (94.7%) at 160 °C for 10 min in the absence of a catalyst. Furthermore, degradation experiments indicated that the vitrimer could be hydrolyzed in alkaline solutions.116 An adduct of tung oil-based diacid and maleic anhydride with different contents of carboxylic acid and anhydride groups was used by Xiao et al. to cross-link DGEBA to prepare vitrimers. The resulting tung oil-based vitrimers demonstrated outstanding mechanical strength and toughness, with a tensile strength of 63.17 MPa, an elongation at break of 5.34% and bending strength equal to 92.96 MPa. Interestingly, the presence of secondary hydroxyl groups enhanced the transesterification reaction during the recycling step. Remolding was performed without any catalyst, and the recycled materials retained nearly 100% of their original tensile strength, indicating excellent recyclability and malleability. The activation energies of the vitrimers ranged from 110 to 126.7 kJ mol−1, and the relaxation times were less than 1000 s.117 More recently, Chang et al. synthesized a tung oil-based tetrafunctional carboxylic acid curing agent through a one-pot method and used it to cross-link DGEBA for preparing carbon fiber-reinforced composites. The tensile strength, tensile modulus and elongation at break of the bare vitrimer were 64.62 MPa, 2.55 GPa, and 4.31%, respectively. Additionally, the vitrimer exhibited excellent thermally activated repair, shape memory behavior, and recyclability: it could undergo reprocessing in the absence of a catalyst with a recovery efficiency of 91.3% even after the third cycle. Relaxation times ranging from 7166 to 255 s at 180 and 240 °C, respectively, were determined, and an activation energy of 106.4 kJ mol−1 was calculated.118
 | ||
| Fig. 19 Synthesis reactions of a tung oil-based vitrimer. Adapted from ref. 116 with permission from American Chemical Society, Copyright © 2023. | ||
Cardanol is an unsaturated alkylphenol derived from cashew nutshell liquid (CNSL), which accounts for 25% of the weight of the nuts and is an important industrial byproduct. CNSL contains four different meta-alkylphenols that vary in the degree of unsaturation of their side chains.10 Through thermal treatment and subsequent purification, CNSL is processed into industrial-grade cardanol. Cardanol serves as a key starting material for synthesizing chemical building blocks for thermosetting polymers, such as epoxy resins and cross-linkers and cardanol derivatives have also been explored for use in vitrimers. Once epoxidized, the presence of epoxy groups provides excellent reactivity, while the aliphatic chains contribute to flexibility. Additionally, the aromatic core of the molecule ensures high resistance to flame and organic solvents.44 Usually, due to the long aliphatic chains, elastomeric materials with low Tg are obtained.
Ferretti et al. developed a vitrimer combining epoxidized cardanol and a di-boronic ester dithiol as a cross-linking agent. The reaction was carried out in the absence of a catalyst through a thiol-epoxy “click” mechanism. The resulting vitrimer was characterized by short relaxation times, around 4 s at 70 °C and 0.3 s at 110 °C, and a dynamic bond-exchange activation energy of ca. 48 kJ mol−1. Furthermore, the material exhibited ready biodegradability in seawater and demonstrated favorable characteristics under flame exposure.119 Liu et al. studied a cardanol-based vitrimer with self-healing, shape memory, and recyclability properties using citric acid as a cross-linking agent without a catalyst. They mechanically characterized the vitrimers for adhesion purposes; excellent thermal stability was also demonstrated. Stress relaxation analysis was conducted, confirming the autocatalysis of hydroxyl groups at elevated temperatures, with an Ea = 34.03 kJ mol−1. The studied vitrimers were chemically recyclable and showed good thermally activated repairability, shape-memory behavior and recovery of bonding strength after reprocessing, supporting their application as curable and repairable adhesives.120 Trejo-Machin et al. synthesized a cardanol-based polybenzoxazine vitrimer that could be recycled up to 5 times, reshaped and used for reversible adhesion (adhesion strength up to ca. 2.8 MPa on aluminum) thanks to a fast stress relaxation process (18 s at 120 °C) and a low activation energy (64.5 kJ mol−1). The associative nature of the CAN formed was confirmed by frequency sweep measurements at different temperatures. At 140 °C, the crossover of G′ and G″ occurred around 0.19 rad s−1, which corresponds to a relaxation time of 33 s. The shift in the crossover frequency of G′ and G″ with increasing temperature indicated enhanced molecular mobility, characteristic of dynamic bond exchange, while the stable storage modulus at higher frequencies suggested preservation of the network cross-linking.121 Chen et al. reported a cardanol-based vinylogous urethane vitrimer with Tg = 42 °C and Tv = −42 °C. Being Tv < Tg, the chain relaxation at T < 42 °C was limited by segmental mobility. However, at higher T, chain relaxation is then governed by the exchange reactions. The vitrimers were characterized by high reprocessability due to low Ea values (inferred from stress relaxation and creep experiments) of 33 kJ mol−1 and 25 kJ mol−1, respectively. Investigation of shape-memory behavior demonstrated a correlation between the shape programming time and the extent of shape recovery. This behavior was attributed to the low activation energy, which allowed the vitrimer network to reorganize quickly at the programming temperature, thereby facilitating the dissipation of stored elastic energy resulting from the shape deformation.122 Eventually, Bo et al. investigated a renewable benzoxazine-based vitrimer derived from cardanol. The vitrimer showed excellent reprocessability, self-healing properties, shape-memory behavior, and antibacterial activity, with an elongation at break of over 587% and a tensile strength equal to 5.88 MPa. It was recycled many times and was also healed by thermal treatment at 100 °C for 5 minutes. Images of the mechanical and chemical recycling are shown in Fig. 20.123
 | ||
| Fig. 20 (A) Reprocessing process of a benzoxazine-based vitrimer derived from cardanol by compression to reform a film, and the proposed mechanism of cross-linking network remodeling; (B) recycling process of the vitrimer (called PCB1). Reproduced from ref. 123 with permission from Elsevier Ltd, Copyright © 2022. | ||
From a structure–property standpoint, vegetable oil-derived building blocks impart markedly different vitrimer behaviors depending on chain architecture, degree of unsaturation, and functional group density. Oils rich in multiple unsaturated fatty acid chains, such as soybean and linseed oils, enable high epoxy or ester functionality after chemical modification, favoring densely cross-linked networks with good mechanical strength and thermally activated exchange dynamics governed by transesterification or disulfide reactions. Castor oil, distinguished by the presence of intrinsic hydroxyl groups along the fatty acid backbone, facilitates network formation at lower temperatures and promotes efficient dynamic exchange, often resulting in softer, more elastomeric vitrimers with enhanced healing efficiency. Tung oil, characterized by conjugated unsaturation, provides higher reactivity and cross-link density, yielding vitrimers with increased stiffness and thermal resistance while retaining reprocessability. In contrast, cardanol-based structures combine a rigid aromatic core with a long flexible aliphatic side chain, leading to vitrimers with low Tg, high toughness, and rapid stress relaxation. Collectively, these structural variations offer a versatile molecular toolbox for tailoring vitrimer mechanical performance, relaxation kinetics, and processing windows through the judicious selection of vegetable oil feedstocks.
4. Application examples of bio-based vitrimers
Vitrimers have demonstrated broad applicability across key sectors including thermoset composites, packaging, additive manufacturing, adhesives, electronics, and foams, owing to their unique ability to combine thermoset-like mechanical and thermal performance with dynamic covalent bond exchange. In fiber-reinforced composites, vitrimers enable damage repair, reshaping, and recovery of reinforcement fibers, addressing critical recyclability challenges. In packaging, vitrimer chemistries, particularly when derived from bio-based feedstocks, offer a balance of processability, barrier performance, and controlled recyclability. Additive manufacturing benefits from vitrimer networks that allow printed parts to be welded, reshaped, or reprocessed after fabrication, overcoming the irreversibility of conventional thermosets. Adhesive applications exploit vitrimer-enabled debonding, rebonding, and repair under thermal or chemical stimuli, while in electronics, reprocessable vitrimer matrices support repairable circuit boards, conductive composites, and improved electronic waste management. Vitrimer foams further extend these advantages to lightweight, repairable, and potentially degradable cellular materials. Collectively, these application-specific benefits stem from the ability of vitrimers to undergo network rearrangement without loss of cross-link density, enabling extended service lifetimes, repair, reuse, and reduced plastic waste generation within a circular economy framework.124 For instance, the diverse application landscape of vegetable oil-based vitrimers has been comprehensively summarized by Chong et al. as depicted in Fig. 21.125 The following sections review representative vitrimer systems with emphasis on application-driven material design, processing strategies, and functional performance. | ||
| Fig. 21 Potential applications of epoxidized vegetable oil-based epoxy vitrimers. Reproduced from ref. 125 with permission from Elsevier Ltd, Copyright © 2022. | ||
4.1. Composites
Thermosets are widely used in composite production, and it is therefore fundamental to explore bio-based alternatives with lower environmental and toxicological impacts. The use of bio-based vitrimer thermosets can help reduce thermoset composite waste by facilitating recycling and repairability. Numerous applications of vitrimer thermosets in the composite sector can be found in the literature, particularly in carbon fiber-reinforced systems.26,126–128 Recently, Arano et al. developed a self-healing carbon fiber-reinforced epoxy vitrimer using bio-based carboxylic acids to cross-link DGEBA. The cross-linked vitrimer exhibited a Tg of 94 °C and a stress relaxation time of 1.1 h at 200 °C, while the activation energy of the transesterification reaction was found to be in the range of 117–133 kJ mol−1. Furthermore, this study demonstrated that vitrimer-based resins can significantly extend the service life of carbon fiber composites. When a damaged composite containing 50 vol% carbon fibers was heated to an elevated temperature, the transesterification reaction enabled the healing of microcracks and scratches, as depicted in Fig. 22.129 | ||
| Fig. 22 Thermal healing of a carbon fiber-reinforced epoxy vitrimer composite obtained from bio-based carboxylic acids and DGEBA. Reproduced from ref. 129 with permission from American Chemical Society, Copyright © 2024. | ||
Xu et al. developed a self-healing, bio-based vitrimer by combining a tung oil-based triglycidyl ester with a disulfide bond-containing molecule. The resulting material was reinforced with carbon fibers to yield a composite exhibiting dual dynamic network behavior involving both transesterification and disulfide metathesis mechanisms. A relatively low activation energy for dynamic bond exchange was observed (66.51 kJ mol−1), and complete stress relaxation was achieved within 50 s at 160 °C. The composite also demonstrated thermally triggered healing behavior and recycling capabilities, and chemical degradation was achieved using mercaptoethanol, ethanolamine, and ethylene glycol.130 Zhang et al. prepared a carbon fiber-reinforced bio-based epoxy vitrimer composite from camphoric acid and epoxidized soybean oil. The cross-linked network underwent transesterification reactions, demonstrating good reprocessability, repairability, and recyclability. Stress relaxation measurements at elevated temperatures revealed a bond-exchange activation energy of 84 kJ mol−1, with a relaxation time of 7245 s at 200 °C under an excess of hardener. Complete recovery of the carbon fibers was achieved by dissolving the vitrimer matrix in ethylene glycol.131 Liu et al. developed a high-performance bio-based epoxy vitrimer and applied it in carbon fiber-reinforced composites. The vitrimer was synthesized using glycerol triglycidyl ether as the epoxy resin and an imine-containing vanillin derivative as the hardener. Excellent reprocessability and recyclability were demonstrated, and the mechanical properties were comparable to those of amine-cured DGEBA systems. After curing, the carbon fiber composite was recycled by dissolving the resin in an amine solution, enabling complete recovery of the composite constituents.132 Hong et al. reported the use of a ternary aqueous solution of tannic acid, boric acid, and poly(vinyl alcohol) to produce a robust bio-based vitrimer combined with carbon fibers. The resulting composite exhibited high mechanical performance, with a tensile strength of 570 MPa and a tensile modulus of 21 GPa, as well as recyclability and reprocessability above Tv (78 °C) due to dynamic covalent bonding, with an activation energy of 51.83 kJ mol−1.133 Bio-based boronic ester vitrimers were also prepared by Sangaletti et al., starting from epoxidized linseed oil. The resulting vitrimer was used to prepare carbon fiber composites, showing good mechanical properties and recyclability in ethanol via reversible hydrolysis of the boronic ester cross-links.134 Dong et al. studied a guaiacol-derived epoxy vitrimer based on disulfide bond exchange and applied it in carbon fiber-reinforced composites. The vitrimer exhibited fast stress relaxation (20 min at 140 °C and 15.8 s at 200 °C), could be reprocessed at 180 °C and 10 MPa for 30 min, and showed effective thermal repair capability. The activation energy of the dynamic bond-exchange reaction was calculated to be 95.42 kJ mol−1, and a Tv of 64 °C was determined.135 Finally, Bourdon et al. investigated bio-based benzoxazine vitrimer composites reinforced with flax and carbon fibers, demonstrating the importance of stacking sequence and fiber selection in optimizing the healing performance of vitrimer composites.136
4.2. Packaging
Packaging represents a major share of global plastic production. Although this sector is currently dominated by thermoplastic polymers, further research into bio-based materials with high recyclability and biodegradability is required. In this context, the incorporation of functional groups capable of forming dynamic covalent networks can improve both the mechanical performance and recyclability of packaging materials. Several examples of vitrimer-based systems for packaging applications have been reported in the literature. For instance, Safarpour et al. synthesized a bio-based and biodegradable vitrimer using soybean oil as a building block for packaging applications. A combination of vanillin and a bio-based diamine was used as the cross-linker, while oleic acid was incorporated to tune the mechanical properties and control the cross-linking density. Increasing the oleic acid content up to 23 wt% reduced the Tg from 0 °C to −30 °C and the tensile modulus from 2.41 to 0.66 MPa. At the same time, the oleic acid addition shortened the stress relaxation times and reduced the activation energy of dynamic bond exchange due to its plasticizing effect. Without oleic acid, Ea and Tv were 33.6 kJ mol−1 and −152.64 °C, respectively, whereas values of 27.89 kJ mol−1 and −175.5 °C were obtained in its presence. The vitrimers exhibited excellent reprocessability and recyclability, as well as good moldability and adhesion to surfaces. Oxygen and water vapor barrier properties were comparable to those of low-density polyethylene, with very low migration into food simulants. These combined properties make this system a promising alternative for flexible food and general packaging applications.14 Yiga et al. developed recyclable and biodegradable epoxy thermosets using modified rice husk as a component. Rice husks were treated with an alkaline solution to increase the cellulose content, followed by succinylation and curing with trimethylolpropane triglycidyl ether to produce thermoset films. These films could be thermally reprocessed by hot pressing through transesterification reactions, achieving mechanical property recovery efficiencies between 92% and 96%. Additionally, biodegradation levels of 80–84% were achieved after 150 days under composting conditions.137 Tang et al. developed a fully bio-based vitrimer containing 70% lignin using epoxidized fractionated lignin and sebacic acid, without additional chemical modification, for advanced packaging applications involving UV-sensitive products. As discussed in Section 3.2, the phenolic hydroxyl content and the ratio of flexible to rigid linkages in lignin strongly influence tensile strength, toughness, self-healing behavior, and reprocessability. The authors also reported a strong correlation between lignin molecular weight, the syringyl/guaiacyl ratio, and the thermal properties of the resulting materials.1384.3. 3D printing
3D printing has emerged as a high-value sector for polymer applications. Various 3D printing techniques exist, and some of them, such as Digital Light Projection (DLP), Liquid Deposition Modeling (LDM), and Direct Ink Writing (DIW), can be applied to vitrimer materials.139,140Cortés-Guzmán et al. studied a recyclable bio-based vitrimer for DLP 3D printing based on dynamic imine exchange. The obtained vitrimer could be reprocessed in the absence of any catalysts. Five bio-based resin formulations derived from vanillin, comprising vanillin acrylate and vanillin methacrylate-functionalized amines, were synthesized. Complex structures were 3D printed and simultaneously cured for each formulation. The cured vitrimers exhibited tensile moduli ranging from 2.05 MPa to 332 MPa. These materials also demonstrated thermally activated repair and reprocessing capabilities when heated above their glass transition temperatures under high pressure (1500 psi at 140 °C for 3 h).30 Fei et al. also synthesized bio-based UV-curable dimethacrylate vitrimers for DLP 3D printing via the reaction of vegetable oil-derived dimer acid with glycidyl methacrylate. A resin formulation containing a photoinitiator and a transesterification catalyst was used for 3D printing, while ethyl acetate served as a diluent. The authors tuned the mechanical properties of the cross-linked vitrimers through molecular design, achieving a maximum tensile strength of 9.2 MPa and an elongation at break of 66.4%. The vitrimer showed good reprocessability and repairability due to transesterification reactions between hydroxyl and ester groups. Furthermore, the printed structures exhibited notable welding behavior enabled by the dynamic covalent bonds.141 Additionally, a bio-based epoxy vitrimer/cellulose composite was 3D printed via LDM by Capannelli et al. The vitrimer was prepared by mixing epoxidized cardanol with a bio-based polycarboxylic acid in the presence of a zinc catalyst for transesterification. Cellulose was used as a rheology modifier to impart suitable flow properties to the polymer. The formulation was successfully 3D printed and cured at 200 °C for 6 h. Chemical recycling of the vitrimer was achieved by immersion in ethylene glycol at 180 °C for 6 h, and an activation energy of 85 kJ mol−1 and a Tv of 71 °C were determined.142 Finally, Shi et al. developed an epoxy vitrimer for DIW 3D printing that could be recycled and reprinted via transesterification reactions. The vitrimer was based on DGEBA and fatty acids with a zinc catalyst, and nanoclay was incorporated to impart rheological properties suitable for DIW processing. The vitrimer was recycled by soaking in ethylene glycol at 180 °C for 6 h, followed by solvent evaporation and partial repolymerization under vacuum at 180 °C for 8 h to reform an ink suitable for 3D printing. The cross-linking/dissolution/re-cross-linking cycle of the nanoclay-reinforced vitrimer epoxy is illustrated in Fig. 23.143
 | ||
| Fig. 23 Illustration of the cross-linking/dissolution/re-cross-linking cycle for recyclable 3D printing of a nanoclay-reinforced vitrimer epoxy. Reproduced from ref. 143 with permission from The Royal Society of Chemistry, Copyright © 2017. | ||
4.4. Adhesives
The adhesives sector is highly dominated by thermoset polymers. In this context, the introduction of CANs for adhesive production can have a significant impact, and several examples of vitrimer-based adhesives have already been reported.144–146 Notably, Zhang et al. studied a bio-based vitrimer with excellent adhesion properties by modifying wheat straw-derived lignin via a catalyst-free reaction with tert-butyl acetoacetate and cross-linking the resulting product with a fatty acid–based diamine. This approach yielded a fully bio-based vitrimer containing 45–50% lignin. Recycling and reprocessing were enabled by dynamic vinylogous urethane bonds activated at temperatures above 100 °C. Relaxation times ranging from 104 s at 150 °C to 9 s at 180 °C were recorded in the absence of a catalyst, and an activation energy of 133.5 kJ mol−1 was calculated. Mechanical testing revealed a dry shear strength of 12.7 MPa and a wet shear strength of 3.5 MPa. Importantly, adhesive removal by heat or solvent was demonstrated, and the recovered adhesive was fully recycled.147 Lignin-based vitrimer adhesives were also investigated by Yang et al., who produced a polyphenol lignin-based vitrimer adhesive with photothermal conversion capability and a wood lap shear strength of 12.87 MPa. The lap shear strength was maintained even under extreme environmental conditions. Furthermore, owing to lignin's efficient light-to-heat conversion, phenol–lignin-based vitrimers could be repaired using infrared laser irradiation, with the repaired adhesive retaining approximately 93% of its original strength after reprocessing.148 In related work, Du et al. developed a vitrimer adhesive using epoxidized lignin and a lignin-based carboxylic acid curing agent obtained via reaction with maleic anhydride. The rigidity of lignin's aromatic structure increased the mechanical strength up to 68 MPa, while the incorporation of polyethylene glycol significantly improved the elongation at break and eliminated the need for metal catalysts during recycling. The vitrimer exhibited excellent solvent resistance and recyclability through multiple methods, including hot pressing at 160 °C and 4 MPa for 15 min and chemical recycling in ethylene glycol. The activation energy of the bond exchange reaction was calculated to be 18.12 kJ mol−1.149Adhesive materials were also developed by Ma et al., starting from soybean oil. Soybean oil-based polymers with hydrogen-bonded cross-linked networks were formed under UV irradiation via random polymerization. Adhesives were obtained through ammonolysis of soybean oil with ethanolamine, esterification of N-hydroxyalkyl fatty amides with methacrylic anhydride, and free-radical polymerization with allyl thiourea. The resulting networks demonstrated effective adhesion to various substrates, including wood, metal, rubber, glass, and PTFE, with dry shear strengths of 2.21 MPa on wood and 2.06 MPa on steel. These materials exhibited thermal repair capability, reprocessability, shape memory behavior, and tunable adhesion. Solubility tests showed complete dissolution in tetrahydrofuran after 12 h, confirming chemical recyclability.150 Finally, Tratnik et al. studied starch-based reversible adhesives relying on transesterification reactions. By varying the epoxy-to-cross-linker molar ratio, the authors evaluated its effect on vitrimer properties. An excess of cross-linker reduced Tg, cross-link density, Tv, and activation energy. Notably, increasing the ratio from 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 1
1 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.5 lowered the activation energy from 90 to 31 kJ mol−1. Adhesion tests on wood revealed that a 1
1.5 lowered the activation energy from 90 to 31 kJ mol−1. Adhesion tests on wood revealed that a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.5 ratio yielded the highest adhesion strength (3.02 MPa), with adhesion recovery up to 91%. These results highlight the critical role of dynamic bond density and functional group concentration in designing bio-based vitrimer adhesives.151
1.5 ratio yielded the highest adhesion strength (3.02 MPa), with adhesion recovery up to 91%. These results highlight the critical role of dynamic bond density and functional group concentration in designing bio-based vitrimer adhesives.151
4.5. Electronics
Vitrimers can help reduce the environmental impact of electronic waste owing to their reprocessability and repairability. While many vitrimer applications in electronics have been reported, examples of fully bio-based vitrimers in this field remain limited. Reprocessable vitrimers for electronic applications were studied by Biswal et al. Starting from adipic acid and glycerol, a cross-linker was synthesized and combined with DGEBA to form a CAN exploiting transesterification reactions. These vitrimers exhibited high elongation at break (up to 250%) and toughness (466 J m−1), with Tv values between 185 °C and 248 °C depending on catalyst concentration and chain length. The authors demonstrated a functional USB cable for power and data transfer (Fig. 24), highlighting both repairability and recyclability via a solvent-based process.152 Li et al. developed a printed circuit board using a transesterification-based vitrimer and conducted a life cycle assessment showing a significant reduction in environmental impact compared to conventional boards. Fractures and holes were effectively repaired while maintaining consistent performance over multiple cycles. A non-destructive recycling method based on polymer swelling with small-molecule solvents was also introduced, preserving material integrity and enabling the fabrication of new printed circuit boards without performance loss.153 Ho et al. investigated recyclable and healable electronic materials based on vitrimer–liquid metal microdroplet composites, combining the electrical conductivity of liquid metals with the mechanical robustness of thermosets. The composites exhibited a glass transition temperature of 130 °C and a topology freezing temperature of 165 °C. Tv was determined from strain response under constant stress via dynamic mechanical analysis. The materials showed strong solvent resistance, high electrical conductivity, and reprocessability (170 °C, 1.5 metric tons, 30 min, up to four cycles). The activation energy derived from stress relaxation was 45.3 kcal mol−1. With only 5 vol% liquid metal loading, conductive pathways enabled the fabrication of vitrimer-based circuit boards incorporating sensors and LEDs.154 Notably, Wang et al. used carbon nanotube sheets to prepare electricity-triggered healing conductive vitrimers. The sheets were attached to liquid crystal elastomer composites, enabling healing under an applied voltage of 1.18 V mm−1. Electrical healing arose from nanotube reconnection, while mechanical healing was driven by transesterification-induced network rearrangement.155 Yang et al. employed poly(lipoic acid)-modified liquid metal as a cross-linker to prepare sustainable epoxy composites with high thermal conductivity (0.82 W m−1 K−1). Dynamic disulfide bonds anchored the liquid metal to the polymer matrix, enabling reprocessing while maintaining thermal conductivity and allowing degradation under mild reducing conditions.156 Finally, Xu et al. engineered high-Tg vitrimers for electronic packaging through molecular design of thermoset precursors, incorporating rigid moieties and CAN functionalities. The resulting materials exhibited Tg values above 125 °C and Tv around 230 °C, with slow and fast stress relaxation below and above Tv, respectively. Thermally activated repair efficiencies exceeded 80%, demonstrating a promising strategy for advanced electronic packaging materials.157 | ||
| Fig. 24 Example of a vitrimer application in electronics. Measured dielectric constant (a) and volume resistivity (b) of the flexible vitrimers; cable fabricated with the flexible vitrimer (c) and its thermal healing (d); use of the cable to charge a mobile phone (e) and to transfer data (f). Reproduced from ref. 152 with permission from American Chemical Society, Copyright © 2025. | ||
4.6. Foams
Thermoset foams are extensively used in industrial sectors such as automotive, aviation, and electronics. However, they are predominantly fossil-based and difficult to recycle. For these reasons, vitrimers have been investigated as potential alternatives. Tian et al. reported the production of a fully bio-based degradable vitrimer foam using epoxidized malepimaric anhydride, 1,5-diaminopentane, and 1,5-diaminopentane carbamate as a latent curing–blowing agent. The foam was cured at 50 °C and exhibited excellent mechanical performance, with a compressive strength of 2.86 MPa. The vitrimer showed catalyst-free thermally activated repair via dynamic β-hydroxy ester bonds and tertiary amine autocatalysis, as well as shape-memory behavior and chemical degradability in ethanolamine or NaOH under mild conditions. The activation energy was 73.82 kJ mol−1, with a relaxation time of 285 s at 180 °C.158 Tian et al. also developed a degradable tung oil-based vitrimer foam using a latent foaming–curing agent combined with triglycidyl ester of eleostearic acid, maleic anhydride and isophorone diamine. The cured foam exhibited good compressive strength (3.55 MPa) and thermal stability. Tertiary amine groups catalyzed dynamic hydroxyl–ester exchange, enabling thermally triggered healing behavior, degradability in ethanolamine, and shape-memory behavior. Complete stress relaxation occurred between 140 °C and 180 °C, with relaxation times ranging from 1045 to 105 s and an activation energy of 89.29 kJ mol−1.159 Liu et al. studied polyurethane foams with vitrimeric properties by replacing up to 50% of fossil-based polyols with soy polyols. Dibutyltin dilaurate catalyzed bond-exchange reactions, allowing mild reprocessing at 160 °C. The resulting foams exhibited improved stress relaxation and satisfactory reprocessability.160 Finally, Han et al. developed a bio-based, catalyst-free poly(ethylene 2,5-furandicarboxylate)/poly(trimethylene carbonate) blend vitrimer with good foamability, high mechanical performance, thermal stability, and excellent reprocessability.1615. Conclusions and outlook
Bio-based vitrimers have emerged as a highly promising class of materials capable of addressing two of the most critical limitations of conventional thermosets: their reliance on fossil-derived feedstocks and their near-impossibility of reprocessing and recycling. By combining renewable building blocks with associative CAN structures, vitrimers enable thermoset-like mechanical and thermal performance together with reprocessability, repairability, and, in some cases, controlled chemical degradation. As highlighted throughout this review, a wide range of bio-based feedstocks, including carbohydrates, lignin-derived aromatics, and vegetable oils, can be successfully integrated into vitrimer architectures, enabling application-driven tuning of properties for sectors such as composites, packaging, additive manufacturing, adhesives, electronics, and foams. These advances position bio-based vitrimers as strong candidates for next-generation materials aligned with circular economy principles.Despite this significant progress, several scientific and technological challenges must still be addressed before bio-based vitrimers can be implemented at an industrial scale. Achieving mechanical, thermal, and long-term durability comparable to or exceeding that of benchmark fossil-based thermosets under demanding service conditions (e.g., elevated temperatures, aggressive chemical environments, and cyclic loading) remains non-trivial. In this context, the intrinsic variability of biomass-derived feedstocks, including differences in lignin origin and extraction methods or variations in vegetable oil composition, introduces additional complexity in ensuring reproducibility, consistency, and quality control.
Equally critical is the precise control of vitrimer dynamic behavior. The density and kinetics of dynamic covalent bonds must be carefully balanced: while fast exchange reactions enhance reprocessability, healing, and welding, they may also promote creep or dimensional instability during service; conversely, overly sluggish exchange kinetics can limit recyclability and repair. Designing vitrimers with an optimal separation and positioning of Tg and Tv for specific applications therefore remains a central challenge in molecular and network design.
Formulation strategies also require further refinement. Many current vitrimer systems rely on metal-based catalysts or non-benign additives to activate bond exchange reactions, which may restrict their use in sensitive applications, such as food packaging, biomedical devices, or electronics, and complicate end-of-life management. The development of catalyst-free systems, self-catalyzing networks, or vitrimers based on inherently benign components is thus a key research direction for improving safety, regulatory acceptance, and sustainability.
From a processing perspective, translating bio-based vitrimer chemistries to industrially relevant manufacturing routes (e.g., reactive extrusion, resin transfer molding, prepregging, additive manufacturing, and foaming) requires formulations that tolerate realistic processing windows, temperature profiles, and residence times. In parallel, compatibilization with existing polymer and composite waste streams will be essential to fully exploit vitrimer recyclability within current industrial infrastructures.
Finally, robust sustainability validation is indispensable. Experimental advances must be increasingly coupled with LCA and techno-economic analysis to quantify the true environmental benefits of bio-based vitrimer technologies, identify trade-offs, and guide the rational selection of feedstocks, chemistries, and processing routes. Such integrated assessments will be crucial to avoid burden shifting and to ensure that vitrimer solutions deliver measurable improvements over incumbent materials.
Looking ahead, the convergence of molecular design, processing science, and sustainability assessment will be decisive for the future of bio-based vitrimers. The combination of renewable feedstocks, advanced dynamic covalent chemistries, including dual-exchange and multi-network systems, and scalable manufacturing strategies has the potential to move these materials beyond laboratory demonstrations toward real-world, high-value applications. Successfully addressing the remaining challenges would establish bio-based vitrimers as key enablers of circular thermoset technologies and as an important pillar of sustainable polymer materials development.
Author contributions
EA: conceptualization, writing – original draft, and visualization; SDV: writing – review & editing; and AV: writing – review & editing and funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included, and no new data were generated or analyzed as part of this review.Acknowledgements
This study was carried out within the MICS (Made in Italy–Circular and Sustainable) Extended Partnership and received funding from the European Union NextGenerationEU (PIANO NAZIONALE DI RIPRESA E RESILIENZA (PNRR)−MISSIONE 4 COMPONENTE 2, INVESTIMENTO 1.3−D.D. 1551.11-10-2022, PE00000004). This manuscript reflects only the authors’ views and opinions, and neither the European Union nor the European Commission can be considered responsible for them. The graphical abstract was created using BioRender.References
- J. N. Hahladakis, C. A. Velis, R. Weber, E. Iacovidou and P. Purnell, An overview of chemical additives present in plastics: Migration, release, fate and environmental impact during their use, disposal and recycling, J. Hazard. Mater., 2018, 344, 179–199, DOI:10.1016/j.jhazmat.2017.10.014.
- M. Macleod, H. Peter, H. Arp, M. B. Tekman and A. Jahnke, The global threat from plastic pollution, Science, 1979, 373(2021), 61–65, DOI:10.1126/science.abg5433.
- M. Dokl, A. Copot, D. Krajnc, Y. Van Fan, A. Vujanović, K. B. Aviso, R. R. Tan, Z. Kravanja and L. Čuček, Global projections of plastic use, end-of-life fate and potential changes in consumption, reduction, recycling and replacement with bioplastics to 2050, Sustainable Prod. Consumption, 2024, 51, 498–518, DOI:10.1016/j.spc.2024.09.025.
- K. D. Nixon, Z. O. G. Schyns, Y. Luo, M. G. Ierapetritou, D. G. Vlachos, L. T. J. Korley and T. H. Epps, Analyses of Circular Solutions for Advanced Plastics Waste Recycling, Nat. Chem. Eng., 2024, 1, 615–626, DOI:10.1038/s44286-024-00121-6.
- G. Rajeshkumar, S. A. Seshadri and T. K. G. Keerthi, Recycling and Biodegradation Studies of Epoxy/Natural Fiber Composites, in Handbook of Epoxy/Fiber Composites, ed. S. Mavinkere Rangappa, J. Parameswaranpillai, S. Siengchin and S. Thomas, Springer Nature Singapore, Singapore, 2022, pp. 837–850. DOI:10.1007/978-981-19-3603-6_49.
- R. Morales Ibarra, Recycling of thermosets and their composites, in: Thermosets: Structure, Properties, and Applications (Second Edition), 2nd ed, Elsevier, 2018, pp. 639–666. DOI:10.1016/B978-0-08-101021-1.00020-4.
- B. P. F. Bryce and T. Nicholls, Closing the loop on thermoset plastic recycling, Science, 1979, 384(2024), 156–157, DOI:10.1126/science.ado8562.
- X. Wu, P. Hartmann, D. Berne, M. De Bruyn, F. Cuminet, Z. Wang, J. M. Zechner, A. D. Boese, V. Placet, S. Caillol and K. Barta, Closed-loop recyclability of a biomass-derived epoxy-amine thermoset by methanolysis, Science, 1979, 384(2024) DOI:10.1126/science.adj9989.
- R. Dinu, S. Montes, F. Orange and A. Mija, Reprocessable humins thermosets and composites, Compos. Sci. Technol., 2021, 207, 108655, DOI:10.1016/j.compscitech.2021.108655.
- F. Jaillet, E. Darroman, A. Ratsimihety, R. Auvergne, B. Boutevin and S. Caillol, New biobased epoxy materials from cardanol, Eur. J. Lipid Sci. Technol., 2014, 116, 63–73, DOI:10.1002/ejlt.201300193.
- Y. Zhang, X. Liu, M. Wan, Y. Zhu and K. Zhang, Recent Development of Functional Bio-Based Epoxy Resins, Molecules, 2024, 29, 4428, DOI:10.3390/molecules29184428.
- Plastics Europe, Plastics - the fast Facts 2024, (2024). https://plasticseurope.org/knowledge-hub/plastics-the-fast-facts-2024/(accessed September 26, 2025).
- A. Zych, J. Tellers, L. Bertolacci, L. Ceseracciu, L. Marini, G. Mancini and A. Athanassiou, Biobased, Biodegradable, Self-Healing Boronic Ester Vitrimers from Epoxidized Soybean Oil Acrylate, ACS Appl. Polym. Mater., 2021, 3, 1135–1144, DOI:10.1021/acsapm.0c01335.
- M. Safarpour, A. Zych, M. Najafi, G. Tedeschi, L. Ceseracciu, L. Marini, G. Perotto and A. Athanassiou, Biobased and biodegradable imine vitrimers from epoxidized soybean oil as packaging, J. Appl. Polym. Sci., 2024, 141, e55527, DOI:10.1002/app.55527.
- L. Klose, N. Meyer-Heydecke, S. Wongwattanarat, J. Chow, P. Pérez García, C. Carré, W. Streit, G. Antranikian, A. M. Romero and A. Liese, Towards Sustainable Recycling of Epoxy-Based Polymers: Approaches and Challenges of Epoxy Biodegradation, Polymers, 2023, 15, 2653, DOI:10.3390/polym15122653.
- M. Fache, E. Darroman, V. Besse, R. Auvergne, S. Caillol and B. Boutevin, Vanillin, a promising biobased building-block for monomer synthesis, Green Chem., 2014, 16, 1987–1998, 10.1039/c3gc42613k.
- T. N. M. T. Ismail, H. A. Hassan, S. Hirose, Y. Taguchi, T. Hatakeyama and H. Hatakeyama, Synthesis and thermal properties of ester-type crosslinked epoxy resins derived from lignosulfonate and glycerol, Polym. Int., 2010, 59, 181–186, DOI:10.1002/pi.2705.
- V. R. Madduluri, A. Bendi, M. Chinmay, G. P. Maniam, R. Roslan and M. H. Ab Rahim, Recent Advances in Vitrimers: A Detailed Study on the Synthesis, Properties and Applications of Bio-Vitrimers, J. Polym. Environ., 2025, 33, 301–322, DOI:10.1007/s10924-024-03416-0.
- D. Bulutcu and S. Eschig, Bio-based vitrimers and covalent adaptable networks – a short review, Next Sustainability, 2026, 7, 100235, DOI:10.1016/j.nxsust.2025.100235.
- M. A. Lucherelli, A. Duval and L. Avérous, Biobased vitrimers: Towards sustainable and adaptable performing polymer materials, Prog. Polym. Sci., 2022, 127, 101515, DOI:10.1016/j.progpolymsci.2022.101515.
- B. Zhao, T. Liu, M. Fei, C. Hao, B. Bliss, Y. Cao and J. Zhang, Biobased Vitrimers: A Sustainable Future, Sustainable Polym. Energy, 2024, 2, 10007, DOI:10.35534/spe.2024.10007.
- C. N. Bowman and C. J. Kloxin, Covalent adaptable networks: Reversible bond structures incorporated in polymer networks, Angew. Chem., Int. Ed., 2012, 51, 4272–4274, DOI:10.1002/anie.201200708.
- C. J. Kloxin and C. N. Bowman, Covalent adaptable networks: Smart, reconfigurable and responsive network systems, Chem. Soc. Rev., 2013, 42, 7161–7173, 10.1039/c3cs60046g.
- C. J. Kloxin, T. F. Scott, B. J. Adzima and C. N. Bowman, Covalent adaptable networks (CANs): A unique paradigm in cross-linked polymers, Macromolecules, 2010, 43, 2643–2653, DOI:10.1021/ma902596s.
- W. Denissen, J. M. Winne and F. E. Du Prez, Vitrimers: Permanent organic networks with glass-like fluidity, Chem. Sci., 2016, 7, 30–38, 10.1039/c5sc02223a.
- V. Schenk, K. Labastie, M. Destarac, P. Olivier and M. Guerre, Vitrimer composites: current status and future challenges, Mater. Adv., 2022, 3, 8012–8029, 10.1039/d2ma00654e.
- A. Kumar and L. A. Connal, Biobased Transesterification Vitrimers, Macromol. Rapid Commun., 2023, 44, 2200892, DOI:10.1002/marc.202200892.
- O. R. Cromwell, J. Chung and Z. Guan, Malleable and Self-Healing Covalent Polymer Networks through Tunable Dynamic Boronic Ester Bonds, J. Am. Chem. Soc., 2015, 137, 6492–6495, DOI:10.1021/jacs.5b03551.
- Q. Li, D. Wang, T. Wang, Y. Zhang, S. Liu, S. Zhang, Z. Hu, L. Li, G. Wang and Y. Zhao, Reversible B-O bond-based epoxy vitrimers with high thermomechanical and dynamic properties enhanced by intermolecular B-N coordination, RSC Appl. Polym., 2025, 3, 1183–1192, 10.1039/d5lp00144g.
- K. P. Cortés-Guzmán, A. R. Parikh, M. L. Sparacin, A. K. Remy, L. Adegoke, C. Chitrakar, M. Ecker, W. E. Voit and R. A. Smaldone, Recyclable, Biobased Photoresins for 3D Printing Through Dynamic Imine Exchange, ACS Sustainable Chem. Eng., 2022, 10, 13091–13099, DOI:10.1021/acssuschemeng.2c03541.
- X. Liu, L. Liang, M. Lu, X. Song, H. Liu and G. Chen, Water-resistant bio-based vitrimers based on dynamic imine bonds: Self-healability, remodelability and ecofriendly recyclability, Polymer, 2020, 210, 123030, DOI:10.1016/j.polymer.2020.123030.
- W. Denissen, G. Rivero, R. Nicolaÿ, L. Leibler, J. M. Winne and F. E. Du Prez, Vinylogous urethane vitrimers, Adv. Funct. Mater., 2015, 25, 2451–2457, DOI:10.1002/adfm.201404553.
- I. Azcune, A. Huegun, A. Ruiz de Luzuriaga, E. Saiz and A. Rekondo, The effect of matrix on shape properties of aromatic disulfide based epoxy vitrimers, Eur. Polym. J., 2021, 148, 110362, DOI:10.1016/j.eurpolymj.2021.110362.
- N. Tratnik, N. R. Tanguy and N. Yan, Recyclable, self-strengthening starch-based epoxy vitrimer facilitated by exchangeable disulfide bonds, Chem. Eng. J., 2023, 451, 138610, DOI:10.1016/j.cej.2022.138610.
- N. Zheng, Z. Fang, W. Zou, Q. Zhao and T. Xie, Thermoset Shape–Memory Polyurethane with Intrinsic Plasticity Enabled by Transcarbamoylation, Angew. Chem., 2016, 128, 11593–11597, DOI:10.1002/ange.201602847.
- F. Lossada, J. Guo, D. Jiao, S. Groeer, E. Bourgeat-Lami, D. Montarnal and A. Walther, Vitrimer Chemistry Meets Cellulose Nanofibrils: Bioinspired Nanopapers with High Water Resistance and Strong Adhesion, Biomacromolecules, 2019, 20, 1045–1055, DOI:10.1021/acs.biomac.8b01659.
- T. Debsharma, V. Amfilochiou, A. A. Wróblewska, I. De Baere, W. Van Paepegem and F. E. Du Prez, Fast Dynamic Siloxane Exchange Mechanism for Reshapable Vitrimer Composites, J. Am. Chem. Soc., 2022, 144, 12280–12289, DOI:10.1021/jacs.2c03518.
- N. J. Van Zee and R. Nicolaÿ, Vitrimers: Permanently crosslinked polymers with dynamic network topology, Prog. Polym. Sci., 2020, 104, 101233, DOI:10.1016/j.progpolymsci.2020.101233.
- H. Memon, Y. Wei and C. Zhu, Recyclable and reformable epoxy resins based on dynamic covalent bonds – Present, past, and future, Polym. Test., 2022, 105, 1–20, DOI:10.1016/j.polymertesting.2021.107420.
- M. Capelot, M. M. Unterlass, F. Tournilhac and L. Leibler, Catalytic control of the vitrimer glass transition, ACS Macro Lett., 2012, 1, 789–792, DOI:10.1021/mz300239f.
- M. Capelot, D. Montarnal, F. Tournilhac and L. Leibler, Metal-catalyzed transesterification for healing and assembling of thermosets, J. Am. Chem. Soc., 2012, 134, 7664–7667, DOI:10.1021/ja302894k.
- M. Fei, W. Liu, L. Shao, Y. Cao, B. J. Bliss, B. Zhao and J. Zhang, Hemp fiber reinforced dual dynamic network vitrimer biocomposites with direct incorporation of amino silane, Chem. Eng. J., 2024, 480, 148091, DOI:10.1016/j.cej.2023.148091.
- P. Verdugo, D. Santiago, S. De la Flor and À. Serra, A Biobased Epoxy Vitrimer with Dual Relaxation Mechanism: A Promising Material for Renewable, Reusable, and Recyclable Adhesives and Composites, ACS Sustainable Chem. Eng., 2024, 12, 5965–5978, DOI:10.1021/acssuschemeng.4c00205.
- E. Albertini, S. Dalle Vacche, R. Bongiovanni, I. Bianco, G. A. Blengini and A. Vitale, Biobased and Reprocessable Vitrimers Based on Cardanol-Derived Epoxy for More Sustainable Thermosets, ACS Sustainable Chem. Eng., 2025, 13, 2120–2131, DOI:10.1021/acssuschemeng.4c09035.
- S. Guggari, F. Magliozzi, S. Malburet, A. Graillot, M. Destarac and M. Guerre, Vanillin-Based Epoxy Vitrimers: Looking at the Cystamine Hardener from a Different Perspective, ACS Sustainable Chem. Eng., 2023, 11, 6021–6031, DOI:10.1021/acssuschemeng.3c00379.
- S. Tripathi, H. Supriya and S. Bose, Covalent adaptable network offers “sustainable” closed-loop circularity in epoxy vitrimers, SPE Polym., 2023, 5, 95–111, DOI:10.1002/pls2.10113.
- C. Li, B. Ju and S. Zhang, Twin-screw extrusion molding of a cellulose-based vitrimer containing a crosslinkable macromolecular plasticizer, Int. J. Biol. Macromol., 2023, 225, 1487–1493, DOI:10.1016/j.ijbiomac.2022.11.205.
- C. Taplan, M. Guerre, J. M. Winne and F. E. Du Prez, Fast processing of highly crosslinked, low-viscosity vitrimers, Mater. Horiz., 2020, 7, 104–110, 10.1039/c9mh01062a.
- A. Khan, N. Ahmed and M. Rabnawaz, Covalent adaptable network and self-healingmaterials: Current trends and future prospectsin sustainability, Polymers, 2020, 12, 2027, DOI:10.3390/POLYM12092027.
- K. Cerdan, M. Thys, A. Costa Cornellà, F. Demir, S. Norvez, R. Vendamme, N. Van den Brande, P. Van Puyvelde and J. Brancart, Sustainability of self-healing polymers: A holistic perspective towards circularity in polymer networks, Prog, Polym. Sci., 2024, 152, 101816, DOI:10.1016/j.progpolymsci.2024.101816.
- E. Chabert, J. Vial, J. P. Cauchois, M. Mihaluta and F. Tournilhac, Multiple welding of long fiber epoxy vitrimer composites, Soft Matter, 2016, 12, 4838–4845, 10.1039/c6sm00257a.
- M. Röttger, T. Domenech, R. Van Der Weegen, A. Breuillac, R. Nicolaÿ and L. Leibler, High-performance vitrimers from commodity thermoplastics through dioxaborolane metathesis, Science, 1979, 356(2017), 62–65, DOI:10.1126/science.aah5281.
- M. L. Lepage, C. Simhadri, C. Liu, M. Takaffoli, L. Bi, B. Crawford, A. S. Milani and J. E. Wulff, A broadly applicable cross-linker for aliphatic polymers containing C-H bonds, Science, 2019, 366, 875–878, DOI:10.1126/science.aay6230.
- R. Dinu, U. Lafont, O. Damiano and A. Mija, Sustainable and recyclable thermosets with performances for high technology sectors. An environmental friendly alternative to toxic derivatives, Front. Mater., 2023, 10(1242507), 1–11, DOI:10.3389/fmats.2023.1242507.
- H. Okada, T. Tokunaga, X. Liu, S. Takayanagi, A. Matsushima and Y. Shimohigashi, Direct evidence revealing structural elements essential for the high binding ability of bisphenol a to human estrogen-related receptor-γ, Environ. Health Perspect., 2008, 116, 32–38, DOI:10.1289/ehp.10587.
- R. Auvergne, S. Caillol, G. David, B. Boutevin and J. P. Pascault, Biobased thermosetting epoxy: Present and future, Chem. Rev., 2014, 114, 1082–1115, DOI:10.1021/cr3001274.
- P. Ares-Elejoste, R. Seoane-Rivero, I. Gandarias, A. Iturmendi and K. Gondra, Sustainable Alternatives for the Development of Thermoset Composites with Low Environmental Impact, Polymers, 2023, 15(2939), 1–16, DOI:10.3390/polym15132939.
- T. L. Chen, H. Kim, S. Y. Pan, P. C. Tseng, Y. P. Lin and P. C. Chiang, Implementation of green chemistry principles in circular economy system towards sustainable development goals: Challenges and perspectives, Sci. Total Environ., 2020, 716, 1–16, DOI:10.1016/j.scitotenv.2020.136998.
- P. C. Chang, H. Y. Hsu and G. W. Jang, Biological routes to itaconic and succinic acids, Phys. Sci. Rev., 2016, 1, 1–17, DOI:10.1515/psr-2016-0052.
- A. Aden, J. Bozell, J. Holladay and J. White, Amy Manheim, Top Value Added Chemicals from Biomass Volume I-Results of Screening for Potential Candidates from Sugars and Synthesis Gas, 2004. https://www.osti.gov/biblio/15008859 (accessed January 9, 2025).
- X. Pan, Z. Luo, F. Liu, Q. Yi and Y. Zeng, Construction of robust and fully bio-based epoxy vitrimers with facile malleability, self-healing ability, and recyclability from peach gum polysaccharide and itaconic acid, Ind. Crops Prod., 2024, 208, 117779, DOI:10.1016/j.indcrop.2023.117779.
- W. Yang, Q. Yi, F. Liu, X. Pan and Y. Zeng, Fabrication of malleable, repairable, weldable, recyclable and robust epoxy vitrimers from itaconic acid for recycled adhesion, Eur. Polym. J., 2023, 196, 112278, DOI:10.1016/j.eurpolymj.2023.112278.
- T. S. Coope, D. H. Turkenburg, H. R. Fischer, R. Luterbacher, H. Van Bracht and I. P. Bond, Novel Diels-Alder based self-healing epoxies for aerospace composites, Smart Mater. Struct., 2016, 25 DOI:10.1088/0964-1726/25/8/084010.
- P. Wu, H. Cheng, X. Wang, R. Shi, C. Zhang, M. Arai and F. Zhao, A self-healing and recyclable polyurethane-urea Diels-Alder adduct synthesized from carbon dioxide and furfuryl amine, Green Chem., 2021, 23, 552–560, 10.1039/d0gc03695a.
- M. Chrysanthos, J. Galy and J. P. Pascault, Preparation and properties of bio-based epoxy networks derived from isosorbide diglycidyl ether, Polymer, 2011, 52, 3611–3620, DOI:10.1016/j.polymer.2011.06.001.
- A. Adjaoud, L. Puchot and P. Verge, High-Tg and Degradable Isosorbide-Based Polybenzoxazine Vitrimer, ACS Sustainable Chem. Eng., 2022, 10, 594–602, DOI:10.1021/acssuschemeng.1c07093.
- R. Yang, W. Li, R. Mo and X. Zhang, Recyclable and Degradable Vinylogous Urethane Epoxy Thermosets with Tunable Mechanical Properties from Isosorbide and Vanillic Acid, ACS Appl. Polym. Mater., 2023, 5, 2553–2561, DOI:10.1021/acsapm.2c02186.
- D. Stewart, Lignin as a base material for materials applications: Chemistry, application and economics, Ind. Crops Prod., 2008, 27, 202–207, DOI:10.1016/j.indcrop.2007.07.008.
- K. M. Hambleton, Synthesis And Characterization Of Thermosetting Epoxy Resins From Lignin-Inspired Phenolics, Theses and dissertations, Rowan University, 2020. https://rdw.rowan.edu/etd/2841 (accessed September 26, 2025) Search PubMed.
- M. Kleinert and T. Barth, Phenols from lignin, Chem. Eng. Technol., 2008, 31, 736–745, DOI:10.1002/ceat.200800073.
- A. Adjaoud, N. Brosse and P. Verge, Lignin-derived thermosets and vitrimers: Bridging the gap between renewable and recyclable materials, Mater. Today Chem., 2024, 42, 102342, DOI:10.1016/j.mtchem.2024.102342.
- P. K. Karoki, S. Zhang, Y. Pu and A. J. Ragauskas, Lignin-based vitrimers: valorization and utilization of lignin in high-value applications, Mater. Adv., 2024, 5, 7075–7096, 10.1039/d4ma00281d.
- F. G. Sales, C. A. M. Abreu and J. A. F. R. Pereira, Catalytic wet-air oxidation of lignin in a three-phase reactor with aromatic aldehyde production, Braz. J. Chem. Eng., 2004, 21, 211–218, DOI:10.1590/S0104-66322004000200010.
- J. C. Villar, A. Caperos and F. García-Ochoa, , Oxidation of hardwood kraft-lignin to phenolic derivatives with oxygen as oxidant, Wood Sci. Technol., 2001, 35, 245–255, DOI:10.1007/s002260100089.
- S. Nikafshar, O. Zabihi, S. Hamidi, Y. Moradi, S. Barzegar, M. Ahmadi and M. Naebe, A renewable bio-based epoxy resin with improved mechanical performance that can compete with DGEBA, RSC Adv., 2017, 7, 8694–8701, 10.1039/c6ra27283e.
- J. F. Stanzione, J. M. Sadler, J. J. La Scala, K. H. Reno and R. P. Wool, Vanillin-based resin for use in composite applications, Green Chem., 2012, 14, 2346–2352, 10.1039/c2gc35672d.
- H. Memon, H. Liu, M. A. Rashid, L. Chen, Q. Jiang, L. Zhang, Y. Wei, W. Liu and Y. Qiu, Vanillin-Based Epoxy Vitrimer with High Performance and Closed-Loop Recyclability, Macromolecules, 2020, 53, 621–630, DOI:10.1021/acs.macromol.9b02006.
- M. A. Rashid, M. M. Mian, Y. Wei and W. Liu, A vanillin-derived hardener for recyclable, degradable and self-healable high-performance epoxy vitrimers based on transimination, Mater. Today Commun., 2023, 35, 106178, DOI:10.1016/j.mtcomm.2023.106178.
- L. Y. Li, W. Chen, C. H. Hu, Y. D. Li and J. B. Zeng, One-Pot Solvent-Free Synthesis of Imine-Based Epoxidized Soybean Oil Vitrimers for Sustainable Adhesives, ACS Sustainable Chem. Eng., 2025, 13, 547–558, DOI:10.1021/acssuschemeng.4c08530.
- C. Monteserin, M. Blanco, N. Uranga, J. Sanchez, J. M. Laza, J. L. Vilas and E. Aranzabe, Sustainable biobased epoxy thermosets with covalent dynamic imine bonds for green composite development, Polymer, 2023, 285, 126339, DOI:10.1016/j.polymer.2023.126339.
- Q. Yu, X. Peng, Y. Wang, H. Geng, A. Xu, X. Zhang, W. Xu and D. Ye, Vanillin-based degradable epoxy vitrimers: Reprocessability and mechanical properties study, Eur. Polym. J., 2019, 117, 55–63, DOI:10.1016/j.eurpolymj.2019.04.053.
- R. Morales-Cerrada, S. Molina-Gutierrez, P. Lacroix-Desmazes and S. Caillol, Eugenol, a Promising Building Block for Biobased Polymers with Cutting-Edge Properties, Biomacromolecules, 2021, 22, 3625–3648, DOI:10.1021/acs.biomac.1c00837.
- D. Matykiewicz and K. Skórczewska, Characteristics and Application of Eugenol in the Production of Epoxy and Thermosetting Resin Composites: A Review, Materials, 2022, 15, 4824, DOI:10.3390/ma15144824.
- B. G. Harvey, C. M. Sahagun, A. J. Guenthner, T. J. Groshens, L. R. Cambrea, J. T. Reams and J. M. Mabry, A high-performance renewable thermosetting resin derived from eugenol, ChemSusChem, 2014, 7, 1964–1969, DOI:10.1002/cssc.201400019.
- I. Faye, M. Decostanzi, Y. Ecochard and S. Caillol, Eugenol bio-based epoxy thermosets: From cloves to applied materials, Green Chem., 2017, 19, 5236–5242, 10.1039/c7gc02322g.
- J. Qin, H. Liu, P. Zhang, M. Wolcott and J. Zhang, Use of eugenol and rosin as feedstocks for biobased epoxy resins and study of curing and performance properties, Polym. Int., 2014, 63, 760–765, DOI:10.1002/pi.4588.
- C. François, S. Pourchet, G. Boni, S. Fontaine, Y. Gaillard, V. Placet, M. V. Galkin, A. Orebom, J. Samec and L. Plasseraud, Diglycidylether of iso-eugenol: A suitable lignin-derived synthon for epoxy thermoset applications, RSC Adv., 2016, 6, 68732–68738, 10.1039/c6ra15200g.
- J. T. Miao, L. Yuan, Q. Guan, G. Liang and A. Gu, Biobased Heat Resistant Epoxy Resin with Extremely High Biomass Content from 2,5-Furandicarboxylic Acid and Eugenol, ACS Sustainable Chem. Eng., 2017, 5, 7003–7011, DOI:10.1021/acssuschemeng.7b01222.
- S. Günther, P. Lamprecht and G. A. Luinstra, ADMET-polymerization of dienes based on sustainable chemicals, in: Macromol. Symp, 2010, pp. 15–19. DOI:10.1002/masy.200900033.
- H. Niu, Y. Li, P. Zhang, W. Guo, X. Wang and Y. Hu, Self-healable, reprocessable and anti-flammable eugenol-derived covalent adaptable thermosets based on dynamic covalent boronic esters and thiol-ene click chemistry, Polym. Chem., 2024, 4952, 10.1039/d4py00684d.
- C. Ocando, Y. Ecochard, M. Decostanzi, S. Caillol and L. Avérous, Dynamic network based on eugenol-derived epoxy as promising sustainable thermoset materials, Eur. Polym. J., 2020, 135, 109860, DOI:10.1016/j.eurpolymj.2020.109860.
- A. Roig, M. Agizza, À. Serra and S. De la Flor, Disulfide vitrimeric materials based on cystamine and diepoxy eugenol as bio-based monomers, Eur. Polym. J., 2023, 194, 112185, DOI:10.1016/j.eurpolymj.2023.112185.
- R. Tannert, A. Taubert and J. Weber, Biobased Epoxy Eugenol Vitrimers: Influence of Impurities of Technical Grade Monomers on the Network Characteristics, Macromol. Mater. Eng., 2024, 309, 2300236, DOI:10.1002/mame.202300236.
- E. D. Hernandez, A. W. Bassett, J. M. Sadler, J. J. La Scala and J. F. Stanzione, Synthesis and Characterization of Bio-based Epoxy Resins Derived from Vanillyl Alcohol, ACS Sustainable Chem. Eng., 2016, 4, 4328–4339, DOI:10.1021/acssuschemeng.6b00835.
- Y.-T. Wong, J. S. Mahajan, S. Synnott and L. T. J. Korley, Lignin-derivable, thermally healable thiol-acrylate vitrimers with improved mechanical performance and reprocessability via transesterification, RSC Sustainability, 2025, 3, 4067–4078, 10.1039/d5su00182j.
- T. Liu, C. Hao, S. Zhang, X. Yang, L. Wang, J. Han, Y. Li, J. Xin and J. Zhang, A Self-Healable High Glass Transition Temperature Bioepoxy Material Based on Vitrimer Chemistry, Macromolecules, 2018, 51, 5577–5585, DOI:10.1021/acs.macromol.8b01010.
- Y. Y. Liu, J. He, Y. D. Li, X. L. Zhao and J. B. Zeng, Biobased, reprocessable and weldable epoxy vitrimers from epoxidized soybean oil, Ind. Crops Prod., 2020, 153, 112576, DOI:10.1016/j.indcrop.2020.112576.
- C. Di Mauro, A. Genua and A. Mija, Fully bio-based reprocessable thermosetting resins based on epoxidized vegetable oils cured with itaconic acid, Ind. Crops Prod., 2022, 185, 115116, DOI:10.1016/j.indcrop.2022.115116.
- N. C. Nepomuceno, C. Bakkali-Hassani, R. Wellen, S. Caillol and C. Negrell, Fully bio-sourced catalyst-free covalent adaptable networks from epoxidized soybean oil and L-tartaric acid, Eur. Polym. J., 2024, 212, 113074, DOI:10.1016/j.eurpolymj.2024.113074.
- C. Li, B. Ju and S. Zhang, Fully bio-based hydroxy ester vitrimer synthesized by crosslinking epoxidized soybean oil with doubly esterified starch, Carbohydr. Polym., 2023, 302, 120442, DOI:10.1016/j.carbpol.2022.120442.
- J. Jeon and C. Choi, High biomass content in epoxy vitrimers: a study on bio-based and reprocessable thermosets, Macromol. Res., 2024, 33, 345–354, DOI:10.1007/s13233-024-00333-y.
- F. I. Altuna, V. Pettarin and R. J. J. Williams, Self-healable polymer networks based on the cross-linking of epoxidised soybean oil by an aqueous citric acid solution, Green Chem., 2013, 15, 3360–3366, 10.1039/c3gc41384e.
- A. Veloso-Fernández, L. Ruiz-Rubio, I. Yugueros, M. I. Moreno-Benítez, J. M. Laza and J. L. Vilas-Vilela, Improving the Recyclability of an Epoxy Resin through the Addition of New Biobased Vitrimer, Polymers, 2023, 15, 3737, DOI:10.3390/polym15183737.
- X. L. Zhao, Y. Y. Liu, Y. Weng, Y. D. Li and J. B. Zeng, Sustainable Epoxy Vitrimers from Epoxidized Soybean Oil and Vanillin, ACS Sustainable Chem. Eng., 2020, 8, 15020–15029, DOI:10.1021/acssuschemeng.0c05727.
- J. Thomas, J. Nwosu and M. D. Soucek, Acid-Cured Norbornylized Seed Oil Epoxides for Sustainable, Recyclable, and Reprocessable Thermosets and Composite Application, ACS Appl. Polym. Mater., 2023, 5, 2230–2242, DOI:10.1021/acsapm.2c02225.
- S. K. Sahoo, V. Khandelwal and G. Manik, Development of completely bio-based epoxy networks derived from epoxidized linseed and castor oil cured with citric acid, Polym. Adv. Technol., 2018, 29, 2080–2090, DOI:10.1002/pat.4316.
- C. Di Mauro, T. N. Tran, A. Graillot and A. Mija, Enhancing the Recyclability of a Vegetable Oil-Based Epoxy Thermoset through Initiator Influence, ACS Sustainable Chem. Eng., 2020, 8, 7690–7700, DOI:10.1021/acssuschemeng.0c01419.
- C. Di Mauro, S. Malburet, A. Genua, A. Graillot and A. Mija, Sustainable Series of New Epoxidized Vegetable Oil-Based Thermosets with Chemical Recycling Properties, Biomacromolecules, 2020, 21, 3923–3935, DOI:10.1021/acs.biomac.0c01059.
- M. Nardi, L. Ceseracciu, V. Scribano, M. Contardi, A. Athanassiou and A. Zych, Sustainable adhesives: Exploring boronic ester vitrimers containing lignin microparticles, Chem. Eng. J., 2024, 495, 153400, DOI:10.1016/j.cej.2024.153400.
- T. A. McKeon, Chapter 4 - Castor (Ricinus communis L.), in Industrial Oil Crops, ed. T. A. McKeon, D. G. Hayes, D. F. Hildebrand, R. J. Weselake, AOCS Press, 2016, pp. 75–112. DOI:10.1016/B978-1-893997-98-1.00004-X.
- Y. Zhang, M. Zhai, L. Shi, Q. Lei, S. Zhang, L. Zhang, B. Lyu, S. Zhao, J. Ma and V. K. Thakur, Sustainable castor oil-based vitrimers: Towards new materials with reprocessability, self-healing, degradable and UV-blocking characteristics, Ind. Crops Prod., 2023, 193, 116210, DOI:10.1016/j.indcrop.2022.116210.
- J. Zhang, C. Zhang, F. Song, Q. Shang, Y. Hu, P. Jia, C. Liu, L. Hu, G. Zhu, J. Huang and Y. Zhou, Castor-oil-based, robust, self-healing, shape memory, and reprocessable polymers enabled by dynamic hindered urea bonds and hydrogen bonds, Chem. Eng. J., 2022, 429, 131848, DOI:10.1016/j.cej.2021.131848.
- Y. Zhang, S. Zhang, M. Zhai, B. Wei, B. Lyu and L. Liu, Self-Healing and Recyclable Castor Oil-Based Epoxy Vitrimer Based on Dual Dynamic Bonds of Disulfide and Ester Bonds, ACS Appl. Polym. Mater., 2024, 6, 8399–8408, DOI:10.1021/acsapm.4c01208.
- Y. Zhu, F. Gao, J. Zhong, L. Shen and Y. Lin, Renewable castor oil and DL-limonene derived fully bio-based vinylogous urethane vitrimers, Eur. Polym. J., 2020, 135, 109865, DOI:10.1016/j.eurpolymj.2020.109865.
- Y. Shao, H. Zhu, K. Chen, T. Jin, Z. Wang, Z. Luo, J. Wang, H. Sun, S. Wei and Z. Gao, Castor Oil-Based Epoxy Vitrimer Based on Dual Dynamic Network with Intrinsic Photothermal Self-Healing Capability, Polymers, 2025, 17, 897, DOI:10.3390/polym17070897.
- L. Chen, Z. Li, Y. Ma, Q. Shang, Y. Hu, Q. Huang, M. Zhang, P. Jia and Y. Zhou, Tung Oil-Based Degradable Vitrimer for Reprocessable and Recyclable Vitrimer-MWCNT Composites with Self-Healing Ability Triggered by Multiple Stimuli, ACS Appl. Polym. Mater., 2023, 5, 9203–9212, DOI:10.1021/acsapm.3c01681.
- L. Xiao, W. Li, Z. Liu, K. Zhang, S. Li, Y. Wang, J. Chen, J. Huang and X. Nie, Tung Oil-Derived Epoxy Vitrimers with High Mechanical Strength, Toughness, and Excellent Recyclability, ACS Sustainable Chem. Eng., 2022, 10, 9829–9840, DOI:10.1021/acssuschemeng.2c01653.
- Q. Chang, W. Li, L. Xiao, K. Zhang, J. Huang, Y. Wang, X. Nie and J. Chen, Catalyst-Free, Self-Healing, and Recyclable Tung Oil-Based Epoxy Vitrimers with Dynamic Ester Bonds, Macromol. Chem. Phys., 2023, 224, 2300272, DOI:10.1002/macp.202300272.
- F. Ferretti, G. Damonte, F. Cantamessa, R. Arrigo, A. Athanassiou, A. Zych, A. Fina and O. Monticelli, On a Biobased Epoxy Vitrimer from a Cardanol Derivative Prepared by a Simple Thiol-Epoxy “Click” Reaction, ACS Omega, 2024, 9, 1242–1250, DOI:10.1021/acsomega.3c07459.
- H. H. Liu, Y. Ma, Y. H. Zhou and G. D. Feng, A Biobased Vitrimer: Self-Healing, Shape Memory, and Recyclability Induced by Dynamic Covalent Bond Exchange, ACS Omega, 2024, 9, 36497–36508, DOI:10.1021/acsomega.4c04264.
- A. Trejo-Machin, L. Puchot and P. Verge, A cardanol-based polybenzoxazine vitrimer: Recycling, reshaping and reversible adhesion, Polym. Chem., 2020, 11, 7026–7034, 10.1039/d0py01239d.
- F. Chen, Q. Cheng, F. Gao, J. Zhong, L. Shen, C. Lin and Y. Lin, The effect of latent plasticity on the shape recovery of a shape memory vitrimer, Eur. Polym. J., 2021, 147, 110304, DOI:10.1016/j.eurpolymj.2021.110304.
- C. Bo, Y. Sha, F. Song, M. Zhang, L. Hu, P. Jia and Y. Zhou, Renewable benzoxazine-based thermosets from cashew nut: Investigating the self-healing, shape memory, recyclability and antibacterial activity, J. Cleaner Prod., 2022, 341, 130898, DOI:10.1016/j.jclepro.2022.130898.
- T. Liu, M. E. Fei, B. M. Zhao and J. W. Zhang, Progress in biobased vitrimers, Acta Polym. Sin., 2020, 51, 817–832, DOI:10.11777/j.issn1000-3304.2020.20047.
- K. L. Chong, J. C. Lai, R. A. Rahman, N. Adrus, Z. H. Al-Saffar, A. Hassan, T. H. Lim and M. U. Wahit, A review on recent approaches to sustainable bio-based epoxy vitrimer from epoxidized vegetable oils, Ind. Crops Prod., 2022, 189, 115857, DOI:10.1016/j.indcrop.2022.115857.
- H. T. T. Tran, S. S. Nisha, R. Radjef, M. Nikzad, R. Bjekovic and B. Fox, Recyclable and Biobased Vitrimers for Carbon Fibre-Reinforced Composites—A Review, Polymers, 2024, 16, 1025, DOI:10.3390/polym16081025.
- V. Kumar, W. Kuang and L. S. Fifield, Carbon Fiber-Based Vitrimer Composites: A Path toward Current Research That Is High-Performing, Useful, and Sustainable, Materials, 2024, 17, 3265, DOI:10.3390/ma17133265.
- Y. Wu, Y. Wei and Y. Ji, Carbon material/vitrimer composites: Towards sustainable, functional, and high-performance crosslinked polymeric materials, Giant, 2023, 13, 100136, DOI:10.1016/j.giant.2022.100136.
- F. M. Arano, U. Casado, I. Z. Ferrero, J. Rivera, M. J. Churruca, F. I. Altuna, E. S. Rodríguez, C. E. Hoppe and R. J. J. Williams, Self-Healing of Microcracks and Scratches in a Carbon-Fiber Reinforced Epoxy Vitrimer by Conventional or Remote Heating, ACS Appl. Mater. Interfaces, 2025, 17, 13170–13178, DOI:10.1021/acsami.4c18025.
- Y. Xu, H. Ma, H. Zhang, S. Xu, X. Cheng, L. Bi, J. Jiang and Y. Chen, A dual dynamic network self-healing bio-based vitrimer and its application in multiply recyclable carbon fiber reinforced polymers, Ind. Crops Prod., 2023, 198, 116755, DOI:10.1016/j.indcrop.2023.116755.
- W. Zhang, J. Wu, L. Gao, B. Zhang, J. Jiang and J. Hu, Recyclable, reprocessable, self-adhered and repairable carbon fiber reinforced polymers using full biobased matrices from camphoric acid and epoxidized soybean oil, Green Chem., 2021, 23, 2763–2772, 10.1039/d1gc00648g.
- Y. Y. Liu, G. L. Liu, Y. D. Li, Y. Weng and J. B. Zeng, Biobased High-Performance Epoxy Vitrimer with UV Shielding for Recyclable Carbon Fiber Reinforced Composites, ACS Sustainable Chem. Eng., 2021, 9, 4638–4647, DOI:10.1021/acssuschemeng.1c00231.
- J. Hong, Y. Hong, J. Jeong, D. Oh and M. Goh, Robust Biobased Vitrimers and Its Application to Closed-Loop Recyclable Carbon Fiber-Reinforced Composites, ACS Sustainable Chem. Eng., 2023, 11, 14112–14123, DOI:10.1021/acssuschemeng.3c03468.
- D. Sangaletti, L. Ceseracciu, L. Marini, A. Athanassiou and A. Zych, Biobased boronic ester vitrimer resin from epoxidized linseed oil for recyclable carbon fiber composites, Resour., Conserv. Recycl., 2023, 198, 107205, DOI:10.1016/j.resconrec.2023.107205.
- K. Dong, D. Zhao, Y. Pang, B. Liu, Q. Liu, T. Mu and C. Zhao, Multiple-reprocessable guaiacol-derived epoxy vitrimer with disulfide crosslinks and closed-loop recycling of carbon fiber-reinforced composites, Chem. Eng. J., 2025, 508, 160754, DOI:10.1016/j.cej.2025.160754.
- K. Bourdon, V. Boulic, D. Abdallah Boina, P. Verge, D. F. Schmidt and T. Huber, Hybrid effects in biobased benzoxazine vitrimer composites reinforced with flax and carbon fibres, Mater. Des., 2025, 258, 114532, DOI:10.1016/j.matdes.2025.114532.
- V. A. Yiga, S. Subramaniyan, N. K. Kalita, M. Lubwama and M. Hakkarainen, Modified rice husk as component in recyclable and biodegradable epoxy thermosets, Discover Appl. Sci., 2024, 6, 175, DOI:10.1007/s42452-024-05834-0.
- R. Tang, B. Xue, J. Tan, Y. Guan, J. Wen, X. Li and W. Zhao, Regulating Lignin-Based Epoxy Vitrimer Performance by Fine-Tuning the Lignin Structure, ACS Appl. Polym. Mater., 2022, 4, 1117–1125, DOI:10.1021/acsapm.1c01541.
- J. Zheng, Z. M. Png, S. H. Ng, G. X. Tham, E. Ye, S. S. Goh, X. J. Loh and Z. Li, Vitrimers: Current research trends and their emerging applications, Mater. Today, 2021, 51, 586–625, DOI:10.1016/j.mattod.2021.07.003.
- A. Sharma, A. Chand, I. Singh and B. Gaur, Vitrimers for 3D Printing Technology: Current Status and Future Perspectives, Ind. Eng. Chem. Res., 2025, 64, 2491–2515, DOI:10.1021/acs.iecr.4c03705.
- M. Fei, T. Liu, B. Zhao, A. Otero, Y. C. Chang and J. Zhang, From Glassy Plastic to Ductile Elastomer: Vegetable Oil-Based UV-Curable Vitrimers and Their Potential Use in 3D Printing, ACS Appl. Polym. Mater., 2021, 3, 2470–2479, DOI:10.1021/acsapm.1c00063.
- J. M. Capannelli, S. Dalle Vacche, A. Vitale, K. Bouzidi, D. Beneventi and R. Bongiovanni, A biobased epoxy vitrimer/cellulose composite for 3D printing by Liquid Deposition Modelling, Polym. Test., 2023, 127, 108172, DOI:10.1016/j.polymertesting.2023.108172.
- Q. Shi, K. Yu, X. Kuang, X. Mu, C. K. Dunn, M. L. Dunn, T. Wang and H. Jerry Qi, Recyclable 3D printing of vitrimer epoxy, Mater. Horiz., 2017, 4, 598–607, 10.1039/c7mh00043j.
- M. Surós, P. Verdugo, D. Santiago and S. De la Flor, Improving the reversible adhesion performance of an epoxy vitrimer adhesive through surface treatment technology, J. Adhes., 2026, 102(4), 1–21, DOI:10.1080/00218464.2025.2532693.
- C. Jiang, C. Wang, S. Zhang, H. Bi, Y. Wu, J. Wang, Y. Zhu, J. Qin, Y. Zhao, X. Shi and G. Zhang, Reprocessable and self-healable boronic-ester based poly(styrene-b-isoprene-b-styrene, vitrimeric elastomer with improved thermo-mechanical property and adhesive performance, React. Funct. Polym., 2024, 198, 105893, DOI:10.1016/j.reactfunctpolym.2024.105893.
- D. Santiago, D. Guzmán, J. Padilla, P. Verdugo, S. De la Flor and À. Serra, Recyclable and Reprocessable Epoxy Vitrimer Adhesives, ACS Appl. Polym. Mater., 2023, 5, 2006–2015, DOI:10.1021/acsapm.2c02063.
- S. Zhang, T. Liu, C. Hao, L. Wang, J. Han, H. Liu and J. Zhang, Preparation of a lignin-based vitrimer material and its potential use for recoverable adhesives, Green Chem., 2018, 20, 2995–3000, 10.1039/c8gc01299g.
- Y. Yang, H. Zhou, X. Chen, T. Liu, Y. Zheng, L. Dai and C. Si, Green and ultrastrong polyphenol lignin-based vitrimer adhesive with photothermal conversion property, wide temperature adaptability, and solvent resistance, Chem. Eng. J., 2023, 477, 147216, DOI:10.1016/j.cej.2023.147216.
- L. Du, X. Jin, G. Qian, W. Yang, L. Su, Y. Ma, S. Ren and S. Li, Lignin-based vitrimer for circulation in plastics, coatings, and adhesives with tough mechanical properties, catalyst-free and good chemical solvent resistance, Ind. Crops Prod., 2022, 187, 115439, DOI:10.1016/j.indcrop.2022.115439.
- Y. Ma, Y. Hu, Z. Kou, M. Zhang, L. Hu, S. Li, Q. Huang, P. Jia, Y. Zhou and F. Chu, Soybean oil-based random copolymers based on hydrogen bond crosslinked networks as reprocessable, recyclable and multifunctional adhesive materials, Ind. Crops Prod., 2024, 209, 118048, DOI:10.1016/j.indcrop.2024.118048.
- N. Tratnik, N. R. Tanguy, D. J. Davidson and N. Yan, Starch-based reversible adhesive: Effect of off-stoichiometric ratios on transesterification vitrimer properties, Carbohydr. Polym., 2025, 356, 138610, DOI:10.1016/j.carbpol.2025.123384.
- A. K. Biswal, P. Hong, Z. Zhang, Y. Zheng, S. Gupta, D. Nepal, V. Iyer and A. Vashisth, Flexible and Stretchable Vitrimers for Sustainable Electronics, ACS Appl. Mater. Interfaces, 2025, 17, 9736–9747, DOI:10.1021/acsami.4c16995.
- P. Li and B. Tian, Printed circuit boards made greener, Nat. Sustain., 2024, 7, 521–522, DOI:10.1038/s41893-024-01328-4.
- D. H. Ho, M. Jiang, R. Tutika, J. C. Worch and M. D. Bartlett, Liquid Metal-Vitrimer Conductive Composite for Recyclable and Resilient Electronics, Adv. Mater., 2025, 2501341, DOI:10.1002/adma.202501341.
- H. Wang, Y. Yang, M. Zhang, Q. Wang, K. Xia, Z. Yin, Y. Wei, Y. Ji and Y. Zhang, Electricity-Triggered Self-Healing of Conductive and Thermostable Vitrimer Enabled by Paving Aligned Carbon Nanotubes, ACS Appl. Mater. Interfaces, 2020, 12, 14315–14322, DOI:10.1021/acsami.9b21949.
- S. Yang, T. Li, Y. Wang, X. Zhang, S. Wang, J. Huang and W. Dong, Sustainable epoxy composites with high thermal conductivity using poly(lipoic acid, modified liquid metal as crosslinkers, Compos. Commun., 2023, 44, 101747, DOI:10.1016/j.coco.2023.101747.
- H. Xu, Y. Luo, Z. Sun, Z. Mao, J. Chen, B. Tian, P. Zhu and R. Sun, Epoxy adhesive of high Tg for electronic packaging with stress relaxation and self-healing, in: 2023 24th International Conference on Electronic Packaging Technology, ICEPT 2023, Institute of Electrical and Electronics Engineers Inc., 2023. DOI:10.1109/ICEPT59018.2023.10492103.
- Y. Tian, X. Feng, C. Wang, S. Shang, H. Liu, X. Huang, J. Jiang, Z. Song and H. Zhang, Fully Biobased Degradable Vitrimer Foams: Mechanical Robust, Catalyst-Free Self-Healing, and Shape Memory Properties, ACS Appl. Mater. Interfaces, 2024, 16, 6523–6532, DOI:10.1021/acsami.4c00267.
- Y. Tian, S. Dai, J. Wang, X. Huang, H. Zhang and Y. Chen, Degradable and programmable shape-color dual-response tung oil-based foams with dynamic covalent bonds, Ind. Crops Prod., 2024, 222, 120121, DOI:10.1016/j.indcrop.2024.120121.
- W. Liu, Y. Zhang, P. Chen, L. Shao, Y. Cao, B. Zhao, E. C. Lee, X. Wang and J. Zhang, Scalable manufacturing and reprocessing of vitrimerized flexible polyurethane foam (PUF, based on commercial soy polyols, Ind. Chem. Mater., 2025, 3, 231–245, 10.1039/d4im00117f.
- W. Han and X. Liao, Construction of biobased PEF/PTMC composite vitrimer with advanced performance in mechanical properties, reprocessability and foamability via catalyst-free method, Appl. Mater. Today, 2025, 43, 102668, DOI:10.1016/j.apmt.2025.102668.
| This journal is © The Royal Society of Chemistry 2026 |