Open Access Article
Open Access ArticleA thiophene-based and sulfur-enriched highly conjugated hyper-crosslinked polymer for efficient sequestration of iodine from various media
Sk Abdul Wahed ,
Mustafa Maqbool
,
Mustafa Maqbool ,
Atikur Hassan
,
Atikur Hassan * and
Neeladri Das
* and
Neeladri Das *
*
Department of Chemistry, Indian Institute of Technology Patna, Patna, Bihar-800106, India. E-mail: atikurhassan@gmail.com; neeladri@iitp.ac.in; neeladri2002@yahoo.co.in
First published on 3rd March 2026
Abstract
A thiophene-based hyper-crosslinked polymer (Bi-Thio HCP) was synthesized using commercially available 2,2′-bithiophene as a monomer via Friedel–Crafts alkylation reaction. The obtained Bi-Thio HCP exhibits a high surface area, hierarchical porosity, and an abundance of electron-rich binding sites, facilitating efficient and rapid iodine uptake. The Bi-Thio HCP demonstrated a modest iodine capture capabilities, with an adsorption capacity of 1.96 g g−1 in the vapor phase at 350 K and a maximum uptake capacity of 1.66 g g−1 for triiodide (I3−) in aqueous phase at 298 K. The distribution coefficient (Kd) values greater than 105 mL g−1 associated with I3− capture indicate the high affinity of an adsorbent material towards the adsorbate. The trapped iodine can be readily released by simple heating at an elevated temperature or by soaking in an alcoholic solvent (such as methanol and ethanol) at room temperature. The Bi-Thio HCP can be recycled for at least five cycles without any significant decrease in removal efficiency, enabling its use in sustainable applications. In this study, the adsorption capacities and mechanisms of iodine adsorption by Bi-Thio HCP material were examined using Density Functional Theory (DFT) computations. Overall, the Bi-Thio HCP is a highly competitive adsorbent for iodine sequestration, thanks to its ease of synthesis from readily available monomers and scalable reaction conditions.
1. Introduction
The use of nuclear energy to generate electricity is gaining popularity worldwide.1 According to the “World Nuclear Association”, more than four hundred nuclear power reactors are operational worldwide, and these contribute to approximately 13% of the global electricity generation.2,3 Since electricity generated from nuclear energy has a significantly lower carbon footprint than the thermal power plants,4,5 the contribution of nuclear energy to global electricity production is likely to experience substantial growth in the next few decades.6 However, on the flipside, electricity production from nuclear power plants is associated with the generation of radioactive waste.7 Disposal of nuclear waste (having a high level of radioactivity) safely and effectively remains a significant challenge. During the processing of the spent nuclear fuel (SNF) with concentrated HNO3, substantial quantities of the radioisotopes of iodine undergo oxidation to produce volatile molecular iodine, which is released into off-gas streams (OGS).8,9 In addition, nuclear reactor cooling systems are also known to release radioactive iodine into water bodies, harming aquatic ecosystems and posing significant health hazards.10 Of the many radionuclides found in nuclear power plant waste, iodine-129 (129I) and iodine-131 (131I) are particularly dangerous. A unique characteristic of iodine-129 is its exceptionally long half-life of approximately 15.7 million years.11 On the other hand, iodine-131 (131I) has a relatively much shorter half-life of 8.02 days.12 131I is used as a nuclear medicine for the treatment of thyroid cancer or other thyroid problems because it emits radiations of significantly higher energies.13,14 However, upon bioaccumulation of 131I in healthy cells (of the thyroid), it can also cause cancer.The release of radioactive iodine isotopes into water bodies has far-reaching effects not only on aquatic ecosystems but also on human health. As an example, the human metabolic system has been exposed to radioactive iodine that emerged from the chernobyl nuclear accident,15,16 leading to a rise in diseases such as thyroid cancer among the human population in those regions.17 Therefore, it is essential to ensure the selective capture and sequestration of radioactive iodine-containing species from the environment.18 In the near future, it will also support the safer and sustainable usage of nuclear energy over alternative power generation sources.
The current method for capturing radioactive gases from OGS is wet scrubbing.19 However, there are some practical drawbacks, including high operating costs, ineffective separation, and the creation of secondary contaminants in the form of hazardous, corrosive, and carcinogenic liquid wastes. The challenges associated with “wet scrubbing techniques” can be resolved by the use of solid-state porous adsorbents.20 In this context, several porous materials have been proposed as potential adsorbents,21 including metal–organic frameworks (MOFs),22,23 Ag-exchanged zeolites,24 graphene-based aerogels,25 activated carbons,26 and porous organic polymers (POPs).26,27 But every material has its own set of advantages and disadvantages. For example, robust and simple-to-make Ag-exchanged zeolites are costly, have a low potential for adsorption, and are not highly recyclable.28–30 The yield of radioactive AgI precipitate, a secondary pollutant, is another concern. Since the dissolver solution in the SNF facility is highly acidic, MOFs that are unstable at acidic pH are ineffective as adsorbent.31,32 Compared to these materials, amorphous POPs such as Hyper-cross-linked Polymers (HCPs), Conjugated Microporous Polymers (CMPs), Polymers of Intrinsic Microporosity (PIMs), and Porous Aromatic Frameworks (PAFs) are more appropriate for real-world applications like reversible iodine capture because of their favourable properties, such as good thermal and physicochemical stabilities.18,33–35As a result, POPs have recently been developed and synthesized especially for energy storage, gas capture, catalysis, drug delivery, and other various uses.36–39 In the context of iodine capture, the affinity and the maximum adsorption capacity of the POPs for the gaseous iodine molecules may be significantly raised by incorporation of electron-rich heteroatom groups [such as nitrogen (N), oxygen (O), sulfur (S)] within the polymeric framework.40–43 Hyper-crosslinked polymers (HCPs) are a unique class of POPs that were first identified by Davankov and Tsyurupa in 1969.44 To date, a wide variety of HCPs have been synthesized that are decorated with various structural motifs and heteroatoms in their polymeric network. While there are many reports of HCPs with N or O as the heteroatoms, there are only a handful of HCPs that contain softer Lewis basic S atoms incorporated in the polymeric backbone.45–47
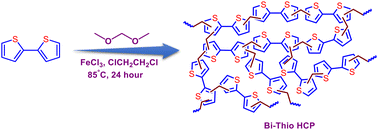
In this work, we design a thiophene-based hyper-crosslinked polymer (HCP) using commercially available yet inexpensive 2,2′-bithiophene as a monomer via the facile Friedel–Crafts alkylation reaction. The obtained thiophene-based adsorbent (Bi-Thio HCP) has an ample number of electron-rich sulphur heteroatoms. After complete characterization using various techniques, we were curious to explore the performance of this S-rich HCP as an improved adsorbent for iodine species, including gaseous I2. The iodine adsorption capability of Bi-Thio HCP was measured to be 1.96 wt%. The mechanism of interaction between the trapped iodine and the thiophene motifs is also examined in this work. Experimental data obtained using various techniques were used to investigate the chemical and physical interactions between iodine molecules and the thiophene motif of Bi-Thio HCP. Additionally, using DFT calculations, valuable insights related to the corresponding structure–property interactions are presented. The ensuing sections provide a detailed description of the experimental results.
2. Experimental section
2.1 Materials
2,2′-Bithiophene, formaldehyde dimethyl acetal (FDA), and anhydrous ferric chloride (FeCl3) was purchased from Sigma-Aldrich. 1,2-Dichloroethane, methanol, acetone, cyclohexane, THF, and molecular iodine were purchased from commercial sources such as Spectrochem, Finar, or CDH. All reagents and solvents used in this work were of analytical grade and used without further purification.2.2 Synthesis of Bi-Thio HCP
Bi-Thio HCP was synthesized via the well-known Friedel–Crafts alkylation reaction using 2,2′-bithiophene as the monomer (Scheme 1). Concisely, 2,2′-bithiophene (166.26 mg, 1.0 mmol) was added to a 50 mL round-bottom flask under a nitrogen (N2) atmosphere and dissolved in 25 mL of 1,2-dichloroethane (DCE). 5 mL of formaldehyde dimethylacetal (FDA) and anhydrous ferric chloride (811 mg, 5 mmol) were added in sequence. The reaction mixture was refluxed at 85 °C for 24 h under an N2 atmosphere. Following the completion of the reaction, the obtained precipitate was filtered and thoroughly washed with methanol, tetrahydrofuran (THF), and acetone to remove FeCl3 from the polymer matrix. Furthermore, the washed product was suspended in methanol for 24 hours to remove any soluble impurities using the Soxhlet extraction technique. The desired product, Bi-Thio HCP, was obtained as a dark brown powder by drying the purified material in a vacuum oven at 100 °C for 12 hours.2.3 Iodine capture experiment
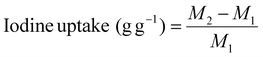 | (1) |
Herein, M1 is the weight of the Bi-Thio HCP before iodine uptake, and M2 is the weight of the Bi-Thio HCP after iodine uptake.
To investigate the kinetics of the iodine capture by Bi-Thio HCP and obtain mechanistic insights, the iodine adsorption data were fitted with the following linear equations corresponding to the pseudo-first-order (eqn (2)) and pseudo-second-order (eqn (3)) rates.
ln(Qe − Qt) = ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Qe − k1t Qe − k1t
| (2) |
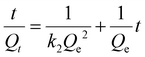 | (3) |
Here, Qe and Qt are the iodine uptake capacity of Bi-Thio HCP at equilibrium (e), and at a particular time (t) interval, respectively, while k1 (h−1) and k2 (g mg−1 h−1) are rate constants of the pseudofirst-order and second-order reaction, respectively.
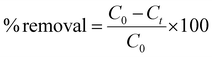 | (4) |
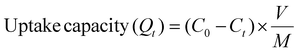 | (5) |
Here, C0 (ppm) and Ct (ppm) represent the initial concentration and concentrations of I3− at a particular time (t), respectively. Qt represents the capture capacity (g g−1) at time (t). V represents the volume of the I3− solution, and M is the weight of the Bi-Thio HCP polymer used in the adsorption experiment.
In adsorption isotherm studies, I3− solution concentrations ranged from 50 to 2000 ppm. In these studies, 2.5 mg of adsorbent (Bi-Thio HCP) was soaked in 5 ml of the respective I3− solution for 12 hours. Later, 2 mL of the supernatant was removed for UV-vis measurement. To gain insight into the sorption mechanism, the I3− sorption isotherm data were fitted with the equations corresponding to the Freundlich isotherm model (eqn (6)) and the Langmuir isotherm model (eqn (7)).
 | (6) |
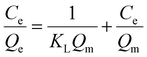 | (7) |
Here, Qe (mg g−1) is the quantity of I3− adsorbed at equilibrium, and Ce (ppm) is the corresponding equilibrium concentration; Qm (mg g−1) is the maximum quantity of I3− adsorbed to form a monolayer on the surface of the adsorbent. KL (L mg−1) is the Langmuir constant, and KF is the Freundlich constant.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 was maintained between I3− and another anion to assess the performance of the adsorbent (Bi-Thio HCP) in the presence of competing ions. The adsorbent (2.5 mg) was suspended in 5 mL of the prepared 50 ppm solution, and the mixture was stirred for 3 hours. Subsequently, 2 mL of the supernatant was collected, and UV-vis spectra were recorded to measure the residual concentration of I3− anions in the solution.
1 was maintained between I3− and another anion to assess the performance of the adsorbent (Bi-Thio HCP) in the presence of competing ions. The adsorbent (2.5 mg) was suspended in 5 mL of the prepared 50 ppm solution, and the mixture was stirred for 3 hours. Subsequently, 2 mL of the supernatant was collected, and UV-vis spectra were recorded to measure the residual concentration of I3− anions in the solution.| ΔE = E(Bi-Thio HCP+iodine species) − E(Bi-Thio HCP) − E(iodine species) |
Here, E(Bi-Thio HCP+iodine species) represents the optimized energy after adsorption of iodine species, E(Bi-Thio HCP) denotes optimized energy of Bi-Thio HCP alone, and E(iodine species) corresponds to the optimized energy of various iodine species such as I2, I3−.
3. Results and discussion
3.1 Characterization of Bi-Thio HCP
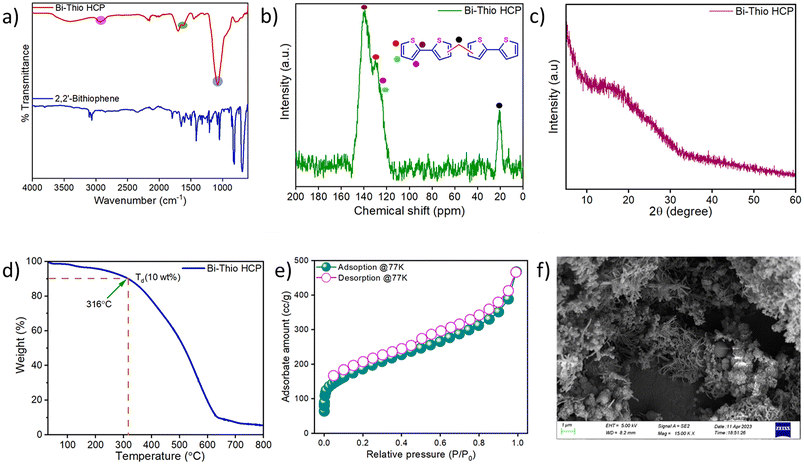
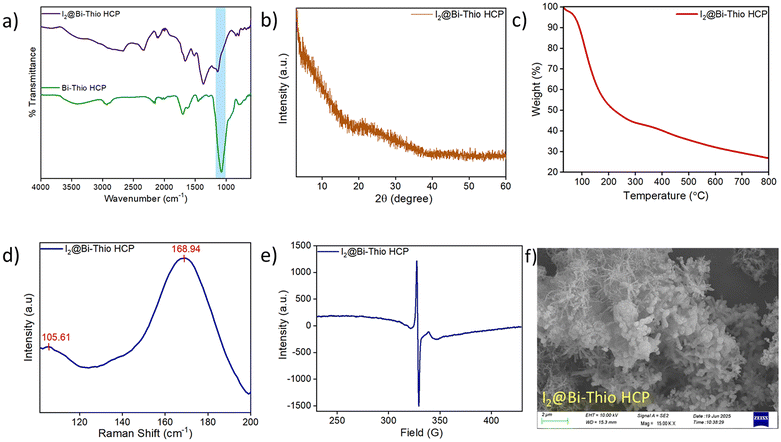
POPs have emerged as a popular class of functional materials due to their unique characteristics, such as facile synthesis with high yields, adjustable porous structures, hierarchical porosity with high surface areas, and ease of incorporating various building blocks and functional groups. Using appropriately functionalized monomers, it might be feasible to develop porous materials with numerous Lewis basic sites that can interact with the electron-deficient iodine species. With this objective, the Bi-Thio HCP was synthesized (Scheme 1) via the well-known Friedel−Crafts alkylation reaction using 2,2′-bithiophene as the monomer (detailed synthesis procedures are provided in the Experimental section). To obtain a pristine polymer free from by-products and unreacted monomers, the resulting crude product was washed with several solvents, including methanol, THF, and acetone. Fourier transform infrared (FTIR) spectrum of Bi-Thio HCP was recorded to identify the distinct functional groups present. In the FT-IR spectrum of the polymer, the appearance of new bands at ∼2871 and ∼2965 cm−1 corresponds to the symmetric and antisymmetric stretching vibrations of the methylene group, which are absent in the monomer spectrum and thus confirm the successful formation of the polymer network (Fig. 1a).48 Furthermore, several bands appeared between 1620 cm−1 and 1060 cm−1, which correspond to the stretching and bending of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C and C
C and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S bonds present in the thiophene units.49 In the solid-state 13C CP-MAS NMR spectrum of Bi-Thio HCP, peaks appearing in between 120 ppm and 140 ppm were attributed to substituted and non-substituted aromatic carbon atoms. Additionally, the resonance peak at 22 ppm corresponds to the methylene carbons in the Bi-Thio HCP network that crosslink one monomer unit with another (Fig. 1b).50 The powder X-ray diffraction (PXRD) pattern of Bi-Thio HCP revealed broad peak suggesting its amorphous nature (Fig. 1c). To investigate the thermal stability of Bi-Thio HCP, a sample was subjected to thermogravimetric analysis (TGA) by heating it between 30 and 800 °C under nitrogen environment (Fig. 1d). The TGA thermogram of Bi-Thio HCP shows the polymeric material has good thermal stability with the thermal degradation temperature (Td = 10% weight loss) at around 316 °C. The nitrogen sorption isotherm was recorded at 77 K to gather information related to the pore structure and surface area. Before surface area analysis, the Bi-Thio HCP sample was activated by heating under vacuum at 120 °C for 12 hours. In the N2 sorption isotherm curves (Fig. 1e), the Bi-Thio HCP demonstrated rapid N2 gas uptake at low relative pressures (P/P0 < 0.01), suggesting the presence of a significant number of micropores in the polymeric network. The BET surface area of Bi-Thio HCP was calculated from the low-temperature nitrogen sorption isotherm and was found to be 664.10 m2 g−1 (Fig. S1). Using nonlocal density functional theory (NL-DFT), the pore size distribution of Bi-Thio HCP was obtained, and it was concluded that the material was microporous with an average pore size of around 1.61 nm (Fig. S2). Due to the inherent porosity in Bi-Thio HCP, the CO2 sorption capacity of Bi-Thio HCP was measured at two temperatures and atmospheric pressure. The CO2 uptake capacities of Bi-Thio HCP were found to be 83 mg g−1 at 273 K (Fig. S3) and 42 mg g−1 at 298 K (Fig. S4). These data suggest the effective CO2 adsorption capabilities of the Bi-Thio HCP polymer, and its performance is comparable to that of several other previously reported polymers under identical conditions. The Field Emission Scanning Electron Microscopy (FESEM) data were used to gain insights into the morphology of Bi-Thio HCP. The micrograph showed a highly porous and rough surface texture with an interconnected network of aggregated particles (Fig. 1f). The presence of carbon (C) and sulfur (S) elements in the network of Bi-Thio HCP was confirmed by energy dispersive X-ray spectroscopy (EDS) mapping analysis (Fig. S5). CHNS elemental analysis was performed to confirm the composition of the synthesized Bi-Thio HCP. CHNS elemental analysis showed that Bi-Thio HCP consists of 66.06% carbon, 3.26% hydrogen and 22.86% sulfur, respectively (Fig. S6). The presence of ample heteroatoms and high surface area (>650 m2 g−1) due to the microporous nature of Bi-Thio HCP motivated us to explore the polymeric material's suitability as an adsorbent for iodine sequestration applications.
S bonds present in the thiophene units.49 In the solid-state 13C CP-MAS NMR spectrum of Bi-Thio HCP, peaks appearing in between 120 ppm and 140 ppm were attributed to substituted and non-substituted aromatic carbon atoms. Additionally, the resonance peak at 22 ppm corresponds to the methylene carbons in the Bi-Thio HCP network that crosslink one monomer unit with another (Fig. 1b).50 The powder X-ray diffraction (PXRD) pattern of Bi-Thio HCP revealed broad peak suggesting its amorphous nature (Fig. 1c). To investigate the thermal stability of Bi-Thio HCP, a sample was subjected to thermogravimetric analysis (TGA) by heating it between 30 and 800 °C under nitrogen environment (Fig. 1d). The TGA thermogram of Bi-Thio HCP shows the polymeric material has good thermal stability with the thermal degradation temperature (Td = 10% weight loss) at around 316 °C. The nitrogen sorption isotherm was recorded at 77 K to gather information related to the pore structure and surface area. Before surface area analysis, the Bi-Thio HCP sample was activated by heating under vacuum at 120 °C for 12 hours. In the N2 sorption isotherm curves (Fig. 1e), the Bi-Thio HCP demonstrated rapid N2 gas uptake at low relative pressures (P/P0 < 0.01), suggesting the presence of a significant number of micropores in the polymeric network. The BET surface area of Bi-Thio HCP was calculated from the low-temperature nitrogen sorption isotherm and was found to be 664.10 m2 g−1 (Fig. S1). Using nonlocal density functional theory (NL-DFT), the pore size distribution of Bi-Thio HCP was obtained, and it was concluded that the material was microporous with an average pore size of around 1.61 nm (Fig. S2). Due to the inherent porosity in Bi-Thio HCP, the CO2 sorption capacity of Bi-Thio HCP was measured at two temperatures and atmospheric pressure. The CO2 uptake capacities of Bi-Thio HCP were found to be 83 mg g−1 at 273 K (Fig. S3) and 42 mg g−1 at 298 K (Fig. S4). These data suggest the effective CO2 adsorption capabilities of the Bi-Thio HCP polymer, and its performance is comparable to that of several other previously reported polymers under identical conditions. The Field Emission Scanning Electron Microscopy (FESEM) data were used to gain insights into the morphology of Bi-Thio HCP. The micrograph showed a highly porous and rough surface texture with an interconnected network of aggregated particles (Fig. 1f). The presence of carbon (C) and sulfur (S) elements in the network of Bi-Thio HCP was confirmed by energy dispersive X-ray spectroscopy (EDS) mapping analysis (Fig. S5). CHNS elemental analysis was performed to confirm the composition of the synthesized Bi-Thio HCP. CHNS elemental analysis showed that Bi-Thio HCP consists of 66.06% carbon, 3.26% hydrogen and 22.86% sulfur, respectively (Fig. S6). The presence of ample heteroatoms and high surface area (>650 m2 g−1) due to the microporous nature of Bi-Thio HCP motivated us to explore the polymeric material's suitability as an adsorbent for iodine sequestration applications.
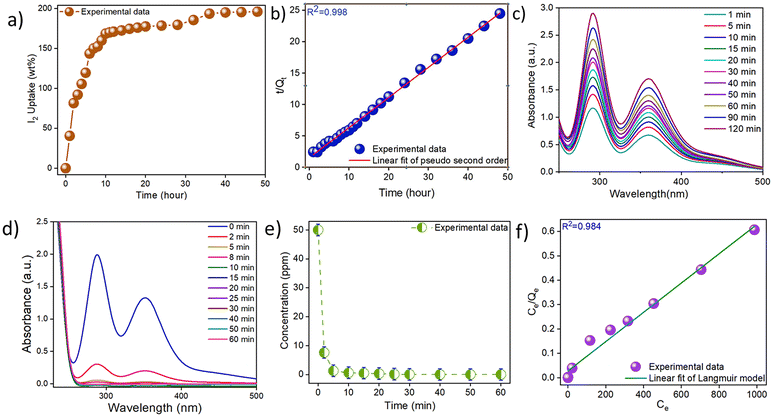
3.2 Iodine capture study
To perform iodine vapor adsorption studies, a pre-weighed sample of Bi-Thio HCP was placed inside a small glass vial, which was subsequently kept in a larger container (containing a crystal of molecular iodine) that was capped securely and put in an oven preheated to ∼77 °C.51 Periodically, the inner glass vial was brought out and weighed to measure the quantity of iodine trapped by Bi-Thio HCP. For nearly complete saturation of the adsorbent (Bi-Thio HCP) with the adsorbate (I2) and to reach the equilibrium conditions, the material has to be exposed to iodine vapors for nearly 48 hours. Upon iodine uptake, a prominent color change was observed in Bi-Thio HCP from dark brown to black (Fig. S7). Upon saturation of Bi-Thio HCP with iodine (I2@Bi-Thio HCP), as evident from negligible weight increment, the weight of the sample was noted to calculate the maximum iodine uptake capacity using eqn (1). Thus, the iodine uptake capacity of Bi-Thio HCP was found to be 196 wt% (Fig. 2a) using the gravimetric analysis method. The iodine uptake of Bi-Thio HCP is comparable to that of several previously reported adsorbents, which are considered high-performing materials (Table S1). The efficiency of Bi-Thio HCP as an adsorbent for iodine in the vapor phase might be due to its aromatic backbone and heteroatom-rich polymer network. Subsequently, the kinetics associated with iodine capture were explored. Experimental data (Fig. S8) fitted well with the pseudo-second-order kinetics rate equation, yielding a correlation coefficient value of 0.998 (Fig. 2b). Iodine recovery from I2@Bi-Thio HCP was thoroughly investigated by heating the iodine-loaded sample or immersing it in methanol solvent. In this context, a known quantity of I2@Bi-Thio HCP sample was heated at 125 °C in an open glass vial, and the weight loss was recorded periodically due to the gradual release of the trapped iodine. Approximately 60% of the adsorbed iodine was released in the first hour, and 86% of iodine was released over the following six hours (Fig. S9). Because of the irregular/non-uniform distribution of pore sizes, some iodine molecules remained trapped in the polymeric network. To conduct an iodine desorption experiment under wet conditions and without the application of heat, a sample of I2@Bi-Thio HCP was immersed in 15 mL of neat methanol placed inside a 20 mL glass vial. Periodically, the UV-Vis spectra of the methanol solution were recorded. The iodine release from I2@Bi-Thio HCP was monitored for 120 minutes using UV-vis spectroscopy (Fig. 2c). As more iodine was released over time, the solvent's color slowly changed from colorless to light yellow and finally deep brown (Fig. S10). Accordingly, the absorbance of the peaks with maxima at 290 and 358 nm continued to increase, indicating the presence of iodine species in methanol.52 This confirmed the effective release of iodine species present on the surface or trapped within the pores of Bi-Thio HCP. This further confirmed the presence of iodine on the surface and within the pores of Bi-Thio HCP that was gradually released into the methanol solution. Reusability of the material is a desirable and essential characteristic in any good adsorbent from the perspective of its sustainable use. Experimental data showed that Bi-Thio HCP could efficiently capture iodine (163 wt%) in the fifth cycle, indicating a minor decrease in its performance as an adsorbent (Fig. S11). Thus, Bi-Thio HCP performs effectively as an iodine adsorbent since it can be easily regenerated while rapidly releasing most of the trapped iodine under ambient conditions.The performance of Bi-Thio HCP as an adsorbent of I3− ions was examined by adding 50 mg of Bi-Thio HCP to a 50 mL solution of I3− (50 ppm) and using UV-vis spectroscopy to monitor the adsorption kinetics. With the gradual passage of time, monitoring the changes in the absorbance of the peaks centered at 352 and 287 nm provided valuable insights into the gradual adsorption of triiodide from the aqueous medium.51 The absorbance of these peaks gradually decreased, indicating that Bi-Thio HCP was effective as an adsorbent in removing triiodide from the aqueous solution (Fig. 2d). Almost 90% adsorption of I3− ions was trapped within five minutes. More than 99% of triiodide were adsorbed within twenty minutes (Fig. 2e and Fig. S12). The results were fitted using pseudo-first-order (Fig. S13) and pseudo-second-order (Fig. S14) rate equations to gain a better understanding of the kinetics associated with triiodide capture. A high correlation coefficient (0.999) was obtained upon fitting the data to the pseudo-second-order rate equation. Compared to the Freundlich model (Fig. S15), a better fit of the adsorption isotherm data was obtained using the Langmuir model (Fig. 2f), suggesting monolayer adsorption of adsorbate species (I3−) on the surface of the adsorbent (Bi-Thio HCP). Using the Langmuir isotherm model, the maximum iodide adsorption capacity of Bi-Thio HCP from water was calculated to be 1663.34 mg g−1. A comparison with previously reported adsorbents of iodide species from aqueous solution suggested that Bi-Thio HCP has superior performance (Table S2). A variety of anions, such as Br−, NO3−, OAc−, CO32−, SO42−, and PO43−, are commonly found in water samples in nature. Therefore, iodine capture experiments were performed in the presence of other ions to test whether Bi-Thio HCP selectivity traps iodide. Experimental data (Fig. S16) clearly indicated that the adsorption of the iodide remained unaffected in the presence of other competing anions that were present at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio in simulated samples of iodine-contaminated wastewater. Thus, capture studies using a binary mixture of anions ratified Bi-Thio HCP's effectiveness in selective uptake of iodide. The distribution coefficient (Kd) is commonly used to assess the affinity of a sorbent toward different sorbates. Adsorbent materials with a Kd value above 104 mL g−1 are considered promising adsorbents. The Kd value of Bi-Thio HCP for I3− removal was determined to be 4.4 × 105 mL g−1.
1 molar ratio in simulated samples of iodine-contaminated wastewater. Thus, capture studies using a binary mixture of anions ratified Bi-Thio HCP's effectiveness in selective uptake of iodide. The distribution coefficient (Kd) is commonly used to assess the affinity of a sorbent toward different sorbates. Adsorbent materials with a Kd value above 104 mL g−1 are considered promising adsorbents. The Kd value of Bi-Thio HCP for I3− removal was determined to be 4.4 × 105 mL g−1.
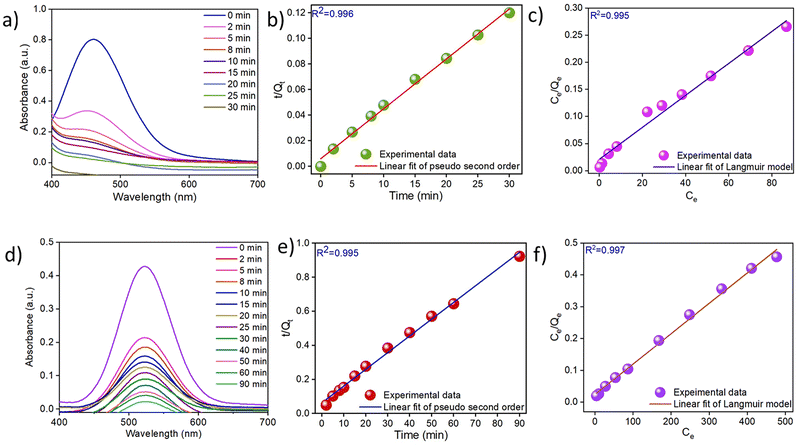
To measure the efficiency of Bi-Thio HCP as an adsorbent of molecular iodine present in aqueous media, we performed adsorption tests using saturated aqueous iodine solutions (1 mM). In these experiments, 50 mg of Bi-Thio HCP was added to iodine solution (50 mL, 1 mM). The color of the solution changed from yellow to colorless, which the unaided eye could easily perceive. This change was quantified using UV-vis spectral data, which showed a decrease in peak intensity at 461 nm (Fig. 3a).53 The Bi-Thio HCP was able to remove almost 80% of the iodine from the aqueous solution in 8 minutes and more than 95% of the iodine in 20 minutes, according to the time-dependent UV-vis data (Fig. S17). Overall, the Bi-Thio HCP was able to remove 99% of iodine from an aqueous solution. Both pseudo-first-order (Fig. S18) and pseudo-second-order (Fig. 3b) kinetics models were used to investigate the mechanism of iodine uptake from aqueous solutions. The kinetics data of the iodine removal process fitted well with the pseudo-second-order model (R2 > 0.99). On the other hand, the pseudo-first-order kinetics model showed a lower correlation coefficient (R2 = 0.954). Subsequently, the maximum iodine uptake capacity from water was estimated using iodine solutions with concentrations ranging from 50 to 250 ppm. The adsorption isotherm data showed an excellent fit to the Langmuir isotherm model (Fig. 3c), with a high correlation coefficient (R2 = 0.995), compared to the Freundlich isotherm model (Fig. S19) indicating that the adsorption process follows Langmuir isotherm model. Using the Langmuir isotherm model (Fig. 3c), the maximum adsorption capacity of iodine from aqueous solutions was calculated to be 348.98 mg g−1. Next, we were interested in assessing the performance of Bi-Thio HCP as an adsorbent to trap iodine dissolved in organic solvents, such as hexanes. A pristine sample of Bi-Thio HCP was added to a purple-colored hexane solution of iodine. The UV-vis spectra of the iodine n-hexane solution were recorded at various time intervals to monitor the rate of iodine uptake by Bi-Thio HCP (Fig. 3d). The decrease in the intensity of the absorption maxima of the UV-vis spectra with the gradual progress of time indicated that the Bi-Thio HCP was successful in removing dissolved iodine from its n-hexane solution. The UV-vis data showed that over 90% of the dissolved iodine was captured by Bi-Thio HCP within 60 minutes (Fig. S20). The kinetics of iodine capture by Bi-Thio HCP from n-hexane solution (Fig. 3e and Fig. S21) were calculated from the spectral data. It was found that the data fitted more precisely with the pseudo-second-order model (correlation coefficient of 0.995) than the pseudo-first-order equation. Additionally, iodine capture experiments were performed using solutions of varying concentration (100 to 1000 ppm) to understand the mechanism of iodine adsorption by Bi-Thio HCP from n-hexane solutions (Fig. 3f and Fig. S22). Using the Langmuir model for data fitting (Fig. 3f), the maximal absorption capacity was determined to be 1049.31 mg g−1. The appreciably high iodine absorption capacity of Bi-Thio HCP suggests that several variables, including surface area, pore size, and the presence of an electron-rich heteroatom in the framework, contribute to its performance. Iodine adsorption in the Bi-Thio HCP is primarily driven by donor–acceptor and charge-transfer interactions between sulfur lone pairs in the bithiophene units and iodine species. Molecular iodine (I2) is destabilized via electron donation to the σ* antibonding orbital of the I–I bond, while triiodide (I3−) is non-covalently stabilized through electron acceptance at terminal iodine atoms. These interactions, together with chalcogen bonding and π-electron delocalization within the conjugated network, enhance iodine adsorption.
3.3 Post capture characterization
Iodine-loaded Bi-Thio HCP (I2@Bi-Thio HCP) samples were examined using several analytical techniques, including FT-IR, PXRD, Raman spectroscopy, EPR, and FESEM), to gain a better understanding of the iodine adsorption mechanism. These characterizations provide valuable information regarding the chemical and morphological changes in the Bi-Thio HCP after iodine adsorption, which in turn help understand the adsorption process. The FT-IR spectra of the Bi-Thio HCP before and after iodine capture are compared in Fig. 4a, wherein noticeable changes in the peak positions of interacting functional groups are observed. For example, the bands corresponding to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S and C
S and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibrations in the pristine polymer (Bi-Thio HCP) shifted after iodine adsorption (I2@Bi-Thio HCP). The observed spectral shift suggested that the sulfur atoms and the arene rings serve as interaction or binding sites for the molecular iodine. The PXRD pattern of I2@Bi-Thio HCP showed that the polymer remained amorphous after iodine capture (Fig. 4b) and there were no diffraction peaks corresponding to crystalline iodine. The thermogravimetric analysis (TGA) of I2@Bi-Thio HCP was performed by heating the sample under a nitrogen atmosphere. The corresponding TGA thermogram revealed a significant weight loss between 80–400 °C due to the progressive release of adsorbed iodine from the I2@Bi-Thio HCP (Fig. 4c). Raman spectral data were further used to find out the nature of iodine species present in I2@Bi-Thio HCP. The I2@Bi-Thio HCP showed two distinct peaks at 105.6 and 168.9 cm−1, which are characteristic of polyiodide anions (Fig. 4d).54 These peaks represent the “V or L” shaped configurations of I5−, suggesting that these polyiodide species were formed on the surface of the Bi-Thio HCP after iodine adsorption.20,55 The presence of charge transfer interactions in I2@Bi-Thio HCP was confirmed by EPR analysis.56 The existence of radical species was evidently revealed by a strong signal in a sample of I2@Bi-Thio HCP (Fig. 4e). Radicals are generated because of facile electron transfer between iodine and the electron-rich groups in the HCP network of Bi-Thio HCP.57 While a comparison of the FESEM images of pristine Bi-Thio HCP (Fig. 1f) and I2@Bi-Thio HCP (Fig. 4f) revealed no noticeable morphological changes, a significant iodine content in the I2@Bi-Thio HCP sample was further confirmed by the elemental mapping data and EDX analysis, with the latter suggesting a uniform distribution of iodine inside the polymer network (Fig. S23).
C stretching vibrations in the pristine polymer (Bi-Thio HCP) shifted after iodine adsorption (I2@Bi-Thio HCP). The observed spectral shift suggested that the sulfur atoms and the arene rings serve as interaction or binding sites for the molecular iodine. The PXRD pattern of I2@Bi-Thio HCP showed that the polymer remained amorphous after iodine capture (Fig. 4b) and there were no diffraction peaks corresponding to crystalline iodine. The thermogravimetric analysis (TGA) of I2@Bi-Thio HCP was performed by heating the sample under a nitrogen atmosphere. The corresponding TGA thermogram revealed a significant weight loss between 80–400 °C due to the progressive release of adsorbed iodine from the I2@Bi-Thio HCP (Fig. 4c). Raman spectral data were further used to find out the nature of iodine species present in I2@Bi-Thio HCP. The I2@Bi-Thio HCP showed two distinct peaks at 105.6 and 168.9 cm−1, which are characteristic of polyiodide anions (Fig. 4d).54 These peaks represent the “V or L” shaped configurations of I5−, suggesting that these polyiodide species were formed on the surface of the Bi-Thio HCP after iodine adsorption.20,55 The presence of charge transfer interactions in I2@Bi-Thio HCP was confirmed by EPR analysis.56 The existence of radical species was evidently revealed by a strong signal in a sample of I2@Bi-Thio HCP (Fig. 4e). Radicals are generated because of facile electron transfer between iodine and the electron-rich groups in the HCP network of Bi-Thio HCP.57 While a comparison of the FESEM images of pristine Bi-Thio HCP (Fig. 1f) and I2@Bi-Thio HCP (Fig. 4f) revealed no noticeable morphological changes, a significant iodine content in the I2@Bi-Thio HCP sample was further confirmed by the elemental mapping data and EDX analysis, with the latter suggesting a uniform distribution of iodine inside the polymer network (Fig. S23).
3.4 DFT study
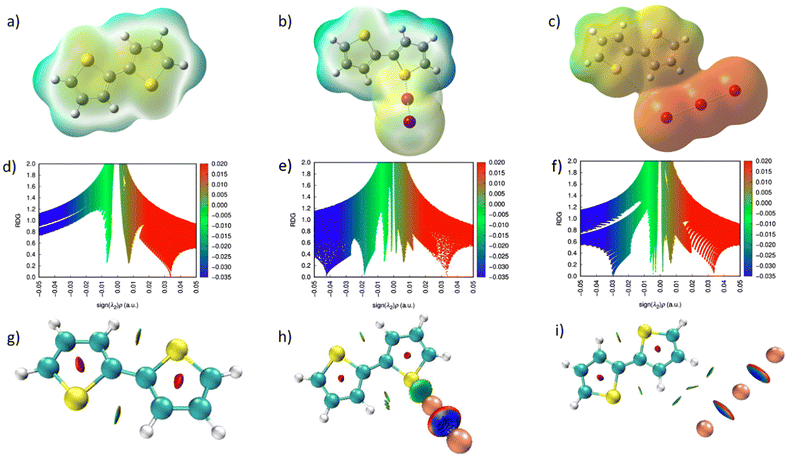
To better comprehend the experimental findings, theoretical investigations using DFT were performed utilizing B3LYP hybrid density functional with a 6-311G or SDD basis set (details in section 4 of SI).58 Initially, DFT computations were performed on the single repeating unit of Bi-Thio HCP to understand the extent of iodine adsorption by aromatic heterocyclic rings containing an S atom. Additionally, RDG (Reduced Density Gradient) and QTAIM (Quantum Theory of Atoms in Molecules) were employed for analysis, as these techniques are frequently used to gain a deeper understanding of molecular structure, chemical bonding and nonbonding interactions, and reactivity. The model of the DFT-optimized structures of Bi-Thio HCP, I2@Bi-Thio HCP, and I3−@Bi-Thio HCP is presented in Fig. S24. The calculated binding energies for I2@Bi-Thio HCP and I3−@Bi-Thio HCP were found to be −15.7 and −13.1 kJ mol−1, respectively. These negative binding energy values led us to conclude that a strong and favorable interaction exists between the iodine and the polymeric network. The interactions between various iodine species are evident from the HOMO and LUMO of the energy-minimized structure of Bi-Thio HCP (Fig. S25). The electrostatic potential (ESP) distribution of Bi-Thio HCP and its iodine-loaded samples (Fig. 5a–c) indicated that the thiophene pendant units produced high local polarization, which promotes electrostatic interactions with the incoming iodine species. The Quantum Theory of Atoms in Molecules (QTAIM) analysis was employed further to investigate the nature and strength of these interactions. The presence of non-covalent stabilizing interactions is confirmed by the QTAIM plots (Fig. 5g–i), which display unique bond critical points (BCPs) between iodine atoms and the S-functional sites of Bi-Thio HCP. Areas of weak-to-moderate overlap in electron density are highlighted by the blue and green isosurfaces seen in the iodine-loaded Bi-Thio HCP. Additionally, RDG analysis provided a more prominent description of the halogen-bonding interaction. In the comparative RDG analysis of the Bi-Thio HCP and the iodine-loaded Bi-Thio HCP (Fig. 5d–f), spikes appear in the blue and green regions, indicating the presence of iodine nonbonding interactions within the optimized polymeric network. These theoretical results provide strong evidence that the thiophene-rich environment of Bi-Thio HCP is essential for stabilizing iodine species (I3−, I2) via orbital polarizations, multi-point noncovalent interactions, and electrostatic attractions.4. Conclusions
Herein, we describe a facile one-step synthesis of a highly conjugated hyper-cross-linked polymer (HCP) using commercially available yet inexpensive 2,2′-bithiophene as a monomer via the well-known Friedel−Crafts alkylation reaction. After a thorough characterization, the hyper-cross-linked polymer (HCP) was investigated as a potential material for iodine adsorption, which occurs in various phases. The iodine uptake capacities of Bi-Thio HCP are superior compared to those of several previously reported porous polymers for similar applications. The adsorbent (Bi-Thio HCP) could be easily regenerated by solvent washing or mild heat treatment. In this process, the Bi-Thio HCP can be recycled without any significant decrease in removal efficiency in further sorption cycles. Using additional experiments and theoretical calculations, the mechanism of interaction between the polymeric network and iodine was studied. Our results suggest that thiophene-functionalized Bi-Thio HCP can be an excellent alternative material for the efficient and selective capture of radioactive iodine vapor during the reprocessing of nuclear fuel. In summary, the porous polymer (Bi-Thio HCP) described here has the potential to be used as an effective adsorbent for environmental remediation applications. We believe our work will enhance the literature on HCP materials, offering improved performance for iodine sequestration.Conflicts of interest
The authors declare that they have no conflict of interest.Data availability
The data that support the findings of this study are available in the supplementary information (SI). Supplementary information: general characterization and physical measurements, characterization of Bi-Thio HCP, iodine capture data, post capture characterization of Bi-Thio HCP, recyclability experiment data, selectivity experiment data, DFT study, comparison tables. See DOI: https://doi.org/10.1039/d5lp00400d.Acknowledgements
N. D. acknowledges the Indian Institute of Technology Patna for providing the research facilities. Sk A. W., M. M., and A. H. are thankful to IIT Patna for their respective institute research fellowships. The authors also acknowledge SAIF-IIT Patna for providing access to the TGA and solid-state 13C NMR facilities.References
- D. F. Sava, M. A. Rodriguez, K. W. Chapman, P. J. Chupas, J. A. Greathouse, P. S. Crozier and T. M. Nenoff, J. Am. Chem. Soc., 2011, 133, 12398–12401 CrossRef CAS PubMed.
- A. Davidson, The role of nuclear energy in the global energy transition, Oxford Institute for Energy Studies, 2022 Search PubMed.
- N. Tang, J. Liang, C. Niu, H. Wang, Y. Luo, W. Xing, S. Ye, C. Liang, H. Guo and J. Guo, J. Mater. Chem. A, 2020, 8, 7588–7625 RSC.
- R. E. Sims, H.-H. Rogner and K. Gregory, Energy Policy, 2003, 31, 1315–1326 CrossRef.
- P. Bagla, Science, 2015, 350, 726–727 CrossRef CAS PubMed.
- F. Gralla, D. J. Abson, A. P. Møller, D. J. Lang and H. von Wehrden, Renewable Sustainable Energy Rev., 2017, 70, 1251–1265 CrossRef.
- J. Fu, J.-Y. Kang, W. Gao, Z.-W. Huang, L.-Q. Kong, K. Xie, Q.-H. Zhu, G.-H. Zhang, G.-H. Tao and L. He, Chem. Commun., 2025, 61, 2235–2256 RSC.
- T. J. Robshaw, J. Turner, S. Kearney, B. Walkley, C. A. Sharrad and M. D. Ogden, SN Appl. Sci., 2021, 3, 843 CrossRef CAS.
- L. He, L. Chen, X. Dong, S. Zhang, M. Zhang, X. Dai, X. Liu, P. Lin, K. Li and C. Chen, Chem, 2021, 7, 699–714 CAS.
- R. M. Asmussen, J. Turner, S. Chong and B. J. Riley, Front. Chem., 2022, 10, 1043653 CrossRef CAS PubMed.
- C. Muhire, A. T. Reda, D. Zhang, X. Xu and C. Cui, Chem. Eng. J., 2022, 431, 133816 CrossRef CAS.
- F. C. Küpper, M. C. Feiters, B. Olofsson, T. Kaiho, S. Yanagida, M. B. Zimmermann, L. J. Carpenter, G. W. Luther III, Z. Lu and M. Jonsson, Angew. Chem., Int. Ed., 2011, 50, 11598–11620 CrossRef PubMed.
- S. Abubakar, T. Skorjanc, D. Shetty and A. Trabolsi, ACS Appl. Mater. Interfaces, 2021, 13, 14802–14815 CrossRef CAS PubMed.
- A. Hassan and N. Das, ACS Appl. Polym. Mater., 2023, 5, 5349–5359 CrossRef CAS.
- J. E. Ten Hoeve and M. Z. Jacobson, Energy Environ. Sci., 2012, 5, 8743–8757 RSC.
- J. Li, X. Zhang, M. Fan, Y. Chen, Y. Ma, G. L. Smith, I. J. Vitorica-Yrezabal, D. Lee, S. Xu and M. Schroder, J. Am. Chem. Soc., 2024, 146, 14048–14057 CrossRef CAS PubMed.
- R. M. Grossi, Science, 2021, 372, 1131 CrossRef CAS PubMed.
- Z. Zheng, Q. Lin, Y. Gao, X. Ji, J. L. Sessler and H. Wang, Coord. Chem. Rev., 2024, 511, 215860 CrossRef CAS.
- S. U. Nandanwar, K. Coldsnow, V. Utgikar, P. Sabharwall and D. E. Aston, Chem. Eng. J., 2016, 306, 369–381 CrossRef CAS.
- T. Pan, K. Yang, X. Dong and Y. Han, J. Mater. Chem. A, 2023, 11, 5460–5475 RSC.
- Y. Xie, L. Yu, L. Chen, C. Chen, L. Wang, F. Liu, Y. Liao, P. Zhang, T. Chen and Y. Yuan, Sci. China: Chem., 2024, 67, 3515–3577 CrossRef CAS.
- W. Xie, D. Cui, S.-R. Zhang, Y.-H. Xu and D.-L. Jiang, Mater. Horiz., 2019, 6, 1571–1595 RSC.
- Y. Zhang, L. He, T. Pan, J. Xie, F. Wu, X. Dong, X. Wang, L. Chen, S. Gong and W. Liu, CCS Chem., 2023, 5, 1540–1548 CrossRef CAS.
- Z. Chen, Q. Lei, Y. Ma, J. Wang, Y. Yan, J. Yin, J. Li, J. Shen, G. Li and T. Pan, Nat. Commun., 2025, 16, 1169 CrossRef CAS PubMed.
- B. Liu, X. Ren, L. Chen, X. Ma, Q. Chen, Q. Sun, L. Zhang, P. Si and L. Ci, J. Hazard. Mater., 2019, 373, 705–715 CrossRef CAS PubMed.
- J. Huve, A. Ryzhikov, H. Nouali, V. Lalia, G. Augé and T. J. Daou, RSC Adv., 2018, 8, 29248–29273 RSC.
- M. Hao, Y. Xie, M. Lei, X. Liu, Z. Chen, H. Yang, G. I. Waterhouse, S. Ma and X. Wang, J. Am. Chem. Soc., 2023, 146, 1904–1913 CrossRef PubMed.
- D. Shetty, J. Raya, D. S. Han, Z. Asfari, J.-C. Olsen and A. Trabolsi, Chem. Mater., 2017, 29, 8968–8972 CrossRef CAS.
- X. Zhang, J. Maddock, T. M. Nenoff, M. A. Denecke, S. Yang and M. Schröder, Chem. Soc. Rev., 2022, 51, 3243–3262 RSC.
- L. Wang, Z. Li, Q. Wu, Z. Huang, L. Yuan, Z. Chai and W. Shi, Environ. Sci.:Nano, 2020, 7, 724–752 RSC.
- I. Ahmed, Z. Hasan, N. A. Khan and S. H. Jhung, Appl. Catal., B, 2013, 129, 123–129 CrossRef CAS.
- M. Sharma, P. Sharma, V. C. Janu and R. Gupta, J. Mater. Chem. A, 2024, 12, 26833–26847 RSC.
- J. Wu, F. Xu, S. Li, P. Ma, X. Zhang, Q. Liu, R. Fu and D. Wu, Adv. Mater., 2019, 31, 1802922 CrossRef PubMed.
- S. Kumar, S. A. Wahed and N. Das, ACS Appl. Polym. Mater., 2025, 7, 11216–11226 CrossRef CAS.
- G. Das, T. Skorjanc, S. K. Sharma, F. Gándara, M. Lusi, D. S. Shankar Rao, S. Vimala, S. Krishna Prasad, J. Raya and D. S. Han, J. Am. Chem. Soc., 2017, 139, 9558–9565 CrossRef CAS PubMed.
- M. S. Lohse and T. Bein, Adv. Funct. Mater., 2018, 28, 1705553 CrossRef.
- N. Baig, S. Shetty, S. A. Wahed, A. Hassan, N. Das and B. Alameddine, ACS Appl. Mater. Interfaces, 2024, 17, 17783–17793 CrossRef PubMed.
- P. Bhanja, A. Modak and A. Bhaumik, ChemCatChem, 2018, 11 Search PubMed.
- L. Tan and B. Tan, Chem. Soc. Rev., 2017, 46, 3322–3356 RSC.
- A. Hassan, S. Goswami, A. Alam, R. Bera and N. Das, Sep. Purif. Technol., 2021, 257, 117923 CrossRef CAS.
- K. Junthod, B. Todee, K. Khamphaijun, T. Chutimasakul, T. Sangtawesin, T. Ratvijitvech, J. Tantirungrotechai, U. Suriya and T. Bunchuay, ACS Appl. Polym. Mater., 2024, 6, 7124–7136 CrossRef CAS.
- Y. Wang, D. Chen, J. Zhang, C. Wang and X. Li, Chem. Eng. J., 2025, 525, 170103 CrossRef CAS.
- C.-J. Sun, P.-F. Wang, H. Wang and B.-H. Han, Polym. Chem., 2016, 7, 5031–5038 RSC.
- J. Huang and S. R. Turner, Polym. Rev., 2018, 58, 1–41 CrossRef CAS.
- H. Ma, J.-J. Chen, L. Tan, J.-H. Bu, Y. Zhu, B. Tan and C. Zhang, ACS Macro Lett., 2016, 5, 1039–1043 CrossRef CAS PubMed.
- B. Todee, T. Chutimasakul, K. Junthod, A. Docker, P. Saetear, M. Kongkaew, T. Ratvijitvech, J. Tantirungrotechai and T. Bunchuay, Mater. Chem. Front., 2022, 6, 3023–3032 RSC.
- L. Monnereau, C. Grandclaudon, T. Muller and S. Bräse, RSC Adv., 2015, 5, 23152–23159 RSC.
- X. Yang and H. Liu, ACS Appl. Mater. Interfaces, 2019, 11, 26474–26482 CrossRef CAS PubMed.
- C. Zhang, Y. He, P. Mu, X. Wang, Q. He, Y. Chen, J. Zeng, F. Wang, Y. Xu and J. X. Jiang, Adv. Funct. Mater., 2018, 28, 1705432 CrossRef.
- A. Hassan, A. Kumar, S. A. Wahed, N. Mishra, A. Kumar and N. Das, Chem. – Eur. J., 2025, e01909 CrossRef CAS PubMed.
- A. Hassan, A. Alam, M. Ansari and N. Das, Chem. Eng. J., 2022, 427, 130950 CrossRef CAS.
- A. Hassan, A. Alam, S. Chandra and N. Das, Environ. Sci.:Adv., 2022, 1, 320–330 CAS.
- Z.-Y. Zeng, Z.-C. Lou, L. Hu, W. Dou, X. Zhao, X. Li, J. Fang, X. Qian, H.-B. Yang and L. Xu, Chem. Eng. J., 2024, 496, 154091 CrossRef CAS.
- T. Liu, Y. Zhao, M. Song, X. Pang, X. Shi, J. Jia, L. Chi and G. Lu, J. Am. Chem. Soc., 2023, 145, 2544–2552 CrossRef CAS PubMed.
- S. A. Wahed, A. Hassan, A. Alam, R. Bera and N. Das, ACS Appl. Mater. Interfaces, 2025, 7, 5127–5137 CAS.
- Z. Zheng, Q. Lin, L. Xie, X. Chen, H. Zhou, K. Lin, D. Zhang, X. Chi, J. L. Sessler and H. Wang, J. Mater. Chem. A, 2023, 11, 13399–13408 RSC.
- J. Chang, H. Li, J. Zhao, X. Guan, C. Li, G. Yu, V. Valtchev, Y. Yan, S. Qiu and Q. Fang, Chem. Sci., 2021, 12, 8452–8457 RSC.
- A. Hassan, R. K. Pandey, A. Chakraborty, S. A. Wahed, T. R. Rao and N. Das, Soft Matter, 2024, 20, 7832–7842 RSC.
| This journal is © The Royal Society of Chemistry 2026 |