Open Access Article
Open Access ArticleSustainable synthesis methods of lignin-based copolymers: recyclable non-carbodiimide catalytic systems in aqueous solvent
Christian
Gonzalez†
,
Arijit
Ghorai†
 and
Hoyong
Chung
and
Hoyong
Chung
 *
*
Department of Chemical and Biomedical Engineering, FAMU-FSU College of Engineering, Tallahassee, Florida 32310, USA. E-mail: hchung@eng.famu.fsu.edu; cag20f@fsu.edu; ag22db@fsu.edu
First published on 9th February 2026
Abstract
Lignin is a valuable biopolymer that serves as a raw material for producing functional polymers due to its abundance, low price, sustainability, and high aromatic content. However, conventional lignin modification methods often rely on environmentally harmful reagents, solvents, and catalysts, primarily because these are common in traditional organic and polymer chemistry. Therefore, utilizing more sustainable solvents, reagents, and catalysts for synthesizing biomass-based, biodegradable polymers is crucial for developing more sustainable production methods. In this work, a lignin-containing copolymer, lignin-graft-polycaprolactone (lignin-graft-PCL), was synthesized using more environmentally friendly solvents, reagents, and catalysts. The first innovation focuses on natural lignin modification. While conventional methods use N,N′-dicyclohexylcarbodiimide (DCC) and 4-dimethylaminopyridine (DMAP) to introduce carboxylic acid functionality into lignin, this study employs 2-chloro-1-methylpyridinium iodide (Mukaiyama reagent), which eliminates the formation of harmful byproducts such as N,N′-dicyclohexylurea (DCU) while maintaining high efficiency. Additionally, a tetrahydrofuran (THF)/water cosolvent system was used for lignin modification, replacing the traditionally used dimethylformamide (DMF). The second key innovation is the use of a recyclable catalyst system for lignin-based graft copolymer synthesis, which requires separate synthesis of PCL prior to graft onto lignin through covalent linkage. Unlike conventional approaches, this study presents a new recyclable polymer bound catalyst, 1,3,4,6,7,8-Hexahydro-2H-pyrimido[1,2-a] pyrimidine, polymer-bound (PS-TBD), for a ε-caprolactone polymerization. The PS-TBD catalyst was successfully recycled and demonstrated sustained performance over four consecutive cycles, showing only a gradual reduction in efficiency. Comprehensive spectroscopic analyses confirmed the chemical structures of all synthesized products obtained from the esterification in lignin modification, ring-opening polymerization, and copolymerization, demonstrating high efficiency.
1. Introduction
The growing effort to diversify raw material sources beyond petroleum-based feedstocks has driven significant research into bio-based alternatives, particularly renewable biomass feedstocks, as viable and sustainable options.1 One renewable biopolymer that has gained significant attention in recent years is lignin. Lignin, the second most abundant macromolecule globally alongside cellulose, is a cost-effective and widely available resource from various industries, making it a promising candidate for biodegradable/non-biodegradable polymer production.2,3 Lignin is found mostly in the cell walls of vascular plants and accounts for 15–30 weight percent of lignocellulosic biomass.4,5 The majority of lignin is currently combusted for energy generation, primarily in the paper and pulp industry, with only a small fraction utilized for real world applications.6 Recent research aims have been the utilization of lignin to produce novel and effective materials that are, most importantly, biobased, and sustainable.7 Due to its aromatic structure, biodegradability, and ease of chemical modification, lignin has potential applications in bioplastics, adhesives, coatings, and composites, offering a sustainable alternative to petroleum-based materials.On a chemical level, lignin is a randomly crosslinked phenolic biopolymer network with a variety of functional groups, including hydroxyl, carboxyl, methoxy, and ether groups. The main repeating units found in lignin consist of coumaryl, coniferyl, and sinapyl alcohols. This variety of functional groups leads to difficulties in characterization and modification of lignin, as well as poor and undefined solubility in commonly used solvents in chemical science.8,9 However, the presence of large contents of aliphatic and phenolic hydroxyl groups allows for great modification potential within the structure of lignin.10–12 The extraction method and plant source of lignin strongly influence both chemical structure and solubility. Dealkaline Kraft lignin is obtained as a by-product of alkaline sulfate pulping. Kraft lignins exhibit lower solubility than organosolv or lignosulfonate lignins, owing to their higher degree of condensation and lower content of hydrophilic substituents.13,14
The above-mentioned poor solubility issue presents significant challenges in working with and processing dealkaline Kraft lignin for various applications, including the development of lignin-based polymers. Dimethylformamide (DMF) is one of the most common organic solvents used to solubilize lignin.15,16 This solvent is highly polar and possesses high boiling point, making it possible to work with and solubilize dealkaline Kraft lignin.17,18 However, DMF is classified as less environmentally friendly due to its toxicity and potential for environmental contamination.19
One promising, green, solvent alternative for lignin modification is dihydrolevoclucosenone (Cyrene). Cyrene is a cellulose biomass-derived solvent, which can be synthesized in a two-step process.20,21 In addition, this solvent is biodegradable, non-mutagenic, and non-toxic. Therefore, cyrene can replace commonly employed dipolar organic solvents such as DMF and NMR. However, cyrene does not dissolve all varieties of lignin efficiently.22 Because of this, it requires intensive additional studies and employing Cyrene on a large scale right now involving lignin would not be advantageous.
Another promising and sustainable solvent is γ-valerolactone (GVL), which is a naturally occurring and non-toxic compound. One of the most attractive properties is its remarkably low vapor pressure, which enhances its safety and usability as a sustainable solvent.23 Additionally, GVL does not form hazardous peroxides when exposed to air, making it a stable option for various industrial processes.24 These characteristics position GVL as a promising candidate for replacing harmful organic solvents. However, GVL alone cannot dissolve lignin, and thus a cosolvent system is required that typically includes DMF or DMSO.25,26 In addition, there is also still a need to develop economical production technologies that can efficiently convert carbohydrate-based biomass into GVL.27 These advancements are crucial for making GVL viable for broader goals of sustainable solvents.
The next sustainable solvent is 2-methyl-tetrahydrofuran (2-MeTHF), which can be derived from renewable resources, such as furfural and levulinic acid.28–30 The solvent possesses practically useful physical and chemical properties, including low water miscibility, a moderate boiling point, and remarkable stability compared to other cyclic chemical structure solvents.31
The examples provided above represent viable greener solvent alternatives, yet several factors hinder their effective implementation in practice. Economic drawbacks, the current challenges of scaling up for industrial use, and difficulties in solubilizing lignin are among the key obstacles facing these solvents. Therefore, improved solvent systems to facilitate lignin modification and polymerization are required.
Unlike the commonly used lignin solvent, DMF, which has a high boiling point of 153 °C, tetrahydrofuran (THF)'s boiling point is 66 °C.32 The low boiling point of THF provides multiple advantages including easy removal from a reaction mixture, which minimizes energy costs and carbon footprints that produce high heating for purification processes.
Another important synthesis step of the lignin-based polymer production is graft copolymerization to covalently link biomass lignin and other polymers that customize functionalities for advanced applications. For this graft copolymerization, carbodiimide catalysts are widely used due to their ability to facilitate the formation of ester bonds.33,34 These catalysts, such as dicyclohexylcarbodiimide (DCC), activate the carboxylic acids, enabling them to react with alcohols. While carbodiimide catalysts are effective and well-known for ester bond linking, they present significant challenges such as the generation of a hazardous byproduct, N,N′-dicyclohexylurea (DCU), which is difficult to remove from the reaction product.35,36 The potential toxicity and environmental impact of these byproducts make the use of carbodiimide catalysts less desirable in sustainable practices. Hence, finding an alternative route to introduce greener and more efficient esterification catalysts is paramount for advancing sustainable polymer synthesis.
2-Chloro-1-methylpyridinium iodide (Mukaiyama reagent) is a viable replacement for traditional DCC and DMAP reagents used for esterification reactions. This catalyst has been used as a condensation reagent in the creation of esters from carboxylic acids and alcohols, as first reported by Mukaiyama et al. in 1975.37 The benefits of using this novel catalyst in place of DCC/DMAP are mainly environmental, as they provide a greener approach to carrying out esterification reactions without the production of the hazardous byproduct, DCU.38
Ring-opening polymerizations (ROP) commonly employ 1,8-diazabicyclo[5.4.0]-undec-7-ene (DBU), 2-tert-butylimino-2-diethylamino-1,3-dimethylperhydro-1,3,2-diazaphosphorine (BEMP), and 1,5,7-Triazabicyclo [4.4.0] dec-5-ene (TBD) catalysts particularly for lactones and cyclic esters.39–43 However, the TBD catalysts are not recoverable and reusable after the polymerization reaction. This not only raises economic concerns but also poses challenges from an environmental standpoint, as the loss of catalyst contributes to waste and increases purification efforts that lead to additional solvent use. Polymer-bound catalysts can solve these outstanding issues. Because these catalysts are immobilized by insoluble or soluble polymer networks or chains, the polymer bound catalyst can be recovered and repeatedly used at the end of the desired chemical reaction, including polymerization. Particularly, the separation of polymer bound catalysts from solution can be easily done by simple filtration, as the polymer support is typically designed to be insoluble and can be easily filtered out, leaving the reaction product in the solution. Depending on the catalyst reactivity, those removed (or recovered) polymer bound catalysts can be reused in other reactions multiple times. Thus, the use of polymer bound catalysts may significantly reduce solvent waste for purification and amount of catalyst required.44–46 Their tunable properties, such as changes in pore size or the hydrophobicity of polymers, also allow for advanced modifications with controllable solubility of the polymer bound catalysts. Therefore, a polymer-bound catalyst could be a good replacement for traditional ROP catalysts and is investigated in this work.
The sustainable PS-TBD presents a promising alternative to traditional TBD catalysts. PS-TBD is a polymer-supported organocatalyst, consisting of a guanidine-based TBD moiety covalently linked to polystyrene resin. It has been successfully applied in a range of reactions, including epoxide ring-opening, aldol-type condensation, Knoevenagel condensation, and Michael additions.47,48 While TBD is widely recognized as the most common catalyst for the ROP of cyclic esters, PS-TBD involved ROP is rare with only a few examples.49–51
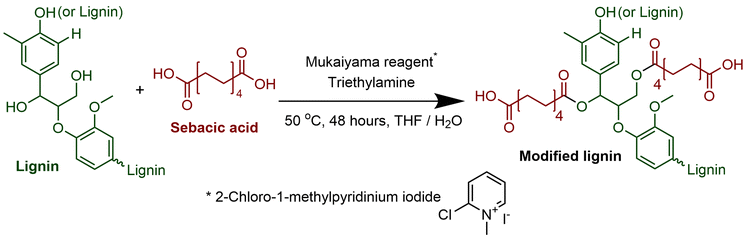
This report focuses on the synthesis of lignin-based polymers using more sustainable solvent, reagent, and catalyst systems. The model polymer synthesized was lignin-graft-polycaprolactone (lignin-graft-PCL). Although similar lignin-based polyesters were reported previously, this work employed higher sustainable synthesis methods compared to classic methods.52,53 The esterification of lignin and the covalent linking of polycaprolactone (PCL) to the modified lignin was achieved using 2-chloro-1-methylpyridinium iodide (Mukaiyama reagent) and a THF/water cosolvent system (Scheme 1, Step 1 and Scheme 3, Step 3). The ROP of PCL was successfully carried out using 1,3,4,6,7,8-Hexahydro-2H-pyrimidine, polymer bound catalyst system (PS-TBD) (Scheme 2, Step 2). The formation of each synthesis step's product is thoroughly established by multiple spectrometry methods, including 1H NMR, 13C NMR, and FT-IR spectroscopy. Thermal properties of the final copolymer were also investigated in comparison to previously reported lignin-based copolymers.
2. Experimental
2.1. Materials
1,3,4,6,7,8-Hexahydro-2H-pyrimido[1,2-a] pyrimidine polymer-bound (polystyrene-bound TBD, PS-TBD), 2-chloro-1-methylpyridinium iodide (Mukaiyama reagent), ε-Caprolactone, triethylamine, and sebacic acid were purchased from MilliporeSigma. Dealkaline Kraft lignin (product number L0045) and 4-methylbenzyl alcohol were purchased from TCI America. According to the product details in TCI America, the sodium ligninsulfonate was initially obtained from needle-leaved trees and broad-leaved trees through the treatment of sodium sulfite. Then, the dealkaline lignin is attained from sodium ligninsulfonate through the chemical modifications such as partial desulfonation, oxidation, hydrolysis and demethylation. All chemicals were used without further purification unless otherwise stated.2.2. Synthesis of sebacic acid modified lignin (modified lignin)
Lignin (2.00 g, OH content: 4.5 mmol g−1, 1 equivalent), sebacic acid (1.82 g, 9 mmol, 1 equivalent), and triethylamine (3 mL, 21.4 mmol, 2.4 equivalent) were taken in a 100 mL two-necked round-bottom flask. Then, 20 mL water/tetrahydrofuran (THF) cosolvent system (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 volume ratio) was added into the flask and stirred to obtain the homogeneous mixture solution. Subsequently, a separate solution of Mukaiyama reagent (2.53 g, 9.9 mmol, 1.1 equivalent) in 20 mL water/THF cosolvent was prepared by stirring at room temperature. Both reaction solutions were degassed under continuous argon atmosphere for 15 minutes. Afterwards, the solution of Mukaiyama reagent was added slowly to the initial solution of lignin and sebacic acid. The final reaction mixture was continuously stirred at 50 °C for 48 hours. The resulting dark solution was precipitated in 1 M HCl solution at 0 °C. The precipitation was filtered under vacuum and rinsed multiple times with de-ionized water. Finally, a brown solid product was obtained after drying under vacuum. Yield: 82 wt% (3.14 g).
1 volume ratio) was added into the flask and stirred to obtain the homogeneous mixture solution. Subsequently, a separate solution of Mukaiyama reagent (2.53 g, 9.9 mmol, 1.1 equivalent) in 20 mL water/THF cosolvent was prepared by stirring at room temperature. Both reaction solutions were degassed under continuous argon atmosphere for 15 minutes. Afterwards, the solution of Mukaiyama reagent was added slowly to the initial solution of lignin and sebacic acid. The final reaction mixture was continuously stirred at 50 °C for 48 hours. The resulting dark solution was precipitated in 1 M HCl solution at 0 °C. The precipitation was filtered under vacuum and rinsed multiple times with de-ionized water. Finally, a brown solid product was obtained after drying under vacuum. Yield: 82 wt% (3.14 g).
2.3. Synthesis of polycaprolactone (PCL)
ε-Caprolactone (4.0 mL, 36.10 mmol, 1 equivalent), PS-TBD (139 mg, 0.361 mmol, 0.01 equivalent), and 4-methylbenzyl alcohol (44.1 mg, 0.361 mmol, 0.01 equivalent) were added to a glass vial and degassed under an argon atmosphere. The reaction mixture was stirred at 110 °C for 48 hours. Then, the mixture was precipitated with cold methanol and precipitation allowed to settle down for 10 minutes. Afterwards, a vacuum filtration was used to separate the solid components (polymer precipitation and polymer bound catalyst) from the methanol. Then, dichloromethane (DCM) was added to the solid products, where the synthesized PCL polymer dissolved in DCM, while the PS-TBD remained insoluble in DCM. The polymer dissolved in DCM solution and the solid yellow PS bound catalyst were collected separately. Lastly, solid PCL polymer was obtained by evaporating DCM under vacuum. Notably, the recovered solid polymer-bound catalyst (PS-TBD) was reused in new polymerization reactions. Yield: 80 wt% (3.3 g).2.4. Synthesis of lignin-graft-polycaprolactone
Modified lignin (0.33 g, OH content: 4.5 mmol g−1, 1.492 mmol, 1 equivalent), PCL (1.0 g), and triethylamine (0.36 g, 3.58 mmol, 2.4 equivalent) were taken in a two-necked round bottom flask. 14 mL of THF/water cosolvent (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 volume ratio) was added to the flask and stirred until dissolved. Simultaneously, a second reaction solution of Mukaiyama reagent (0.46 g, 1.79 mmol, 1.2 equivalent) in 14 mL of THF/water cosolvent was prepared by stirring at room temperature under continuous argon flow for 15 minutes. At this time, the second solution of Mukaiyama reagent was added slowly to the initial mixture of modified lignin, PCL, and triethylamine. The combined mixture solution was continuously stirred at 50 °C for 48 hours. The final reaction mixture was poured into the 1 M HCl solution at 0 °C. The brown solid product was ultimately obtained by vacuum filtering the mixture and rinsing it several times with deionized water. Yield: 80 wt% (1.06 g).
1 volume ratio) was added to the flask and stirred until dissolved. Simultaneously, a second reaction solution of Mukaiyama reagent (0.46 g, 1.79 mmol, 1.2 equivalent) in 14 mL of THF/water cosolvent was prepared by stirring at room temperature under continuous argon flow for 15 minutes. At this time, the second solution of Mukaiyama reagent was added slowly to the initial mixture of modified lignin, PCL, and triethylamine. The combined mixture solution was continuously stirred at 50 °C for 48 hours. The final reaction mixture was poured into the 1 M HCl solution at 0 °C. The brown solid product was ultimately obtained by vacuum filtering the mixture and rinsing it several times with deionized water. Yield: 80 wt% (1.06 g).
2.5. Instrumentation
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 022
022![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 to 580 Da. The calibration curve is utilized to determine the molecular weight of the samples. GPC measurements of the PCL samples were carried out using tetrahydrofuran as the eluent at a flow rate of 1 mL min−1 at room temperature. An injection volume of 50 μL and a sample concentration of 10 mg mL−1 were used.
000 to 580 Da. The calibration curve is utilized to determine the molecular weight of the samples. GPC measurements of the PCL samples were carried out using tetrahydrofuran as the eluent at a flow rate of 1 mL min−1 at room temperature. An injection volume of 50 μL and a sample concentration of 10 mg mL−1 were used.
3. Results and discussion
In this current study, the key features are (1) the utilization of a more sustainable water based cosolvent THF/water, (2) the use of the environmentally friendly Mukaiyama reagent for lignin modification, and (3) exploring the multiple recyclable nature of the PS-TBD catalyst during the ROP of ε-caprolactone (Table 1). This lignin modification reaction has previously been conducted using DCC and DMAP along with high boiling point DMF (153 °C) as the choice solvent.54 The primary focus of this research was the replacement of all three of these chemicals (DCC, DMAP, and DMF) for a more effective and less environmentally impactful alternative without changing the overall chemistry and mechanism of the lignin modification. Overall, lignin-graft-PCL copolymer, is synthesized by three steps: modification of natural lignin, synthesis of PCL, and the covalent integration of modified lignin and PCL. The spectroscopic evidence (1H NMR and 13C NMR) of synthesized products (modified lignin, ε-caprolactone, PCL, lignin-graft-PCL, and “Model Product”), photos of various modified lignins, solubility visuals of raw and modified lignin, and degree of modified lignin acid modification calculations are displayed in Fig. S1–18 in the SI. The schematic representations of these reaction steps can be found in Schemes 1–3, Steps 1–3 and Fig. S12, 15, and 18.| Aspect | This work (improved sustainable methods) | Conventional methods (non-sustainable methods) | Sustainability benefits of this work |
|---|---|---|---|
| Solvent system | Water/tetrahydrofuran (THF) cosolvent system (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 ratio) 50 ratio) |
Dimethylformamide (DMF)55 | Reduces toxicity: Reduces the need for organic solvents and toxicity associated with DMF.19Lower boiling point: Easier removal of solvent and less energy consumption.54 |
| Esterification reagent | Mukaiyama reagent (2-chloro-1-methyl pyridinium iodide) | 4-Dimethylamino pyridine (DMAP) and N,N′-dicyclohexyl carbodiimide (DCC), generates toxic N,N′-dicyclohexylurea (DCU)56 | Reduces toxicity: The Mukaiyama reagent eliminates the formation of the toxic byproduct DCU, which is harmful to both the environment and human health.35,38 |
| ROP catalyst recycling | Polymer-supported TBD (PS-TBD) catalyst | Single-use TBD catalyst57 | Waste reduction: The ability to recycle the PS-TBD catalyst significantly reduces material waste.48Cost-effective: Recycling the catalyst eliminates the need for purchasing new catalyst with each cycle, making the process more cost-effective. |
3.1. Cosolvent system for lignin modification and graft copolymerization
In previous reports, lignin modification commonly used DMF, a solvent with low sustainability. In this study, we established a more sustainable lignin modification method using a THF/water cosolvent system in place of traditional DMF solvent. The synthesis scheme is shown in (Scheme 1, Step 1). While the THF/water mixture represents an improvement over DMF, it should be noted that 50% THF is not a fully green solvent since THF is petroleum-derived. A promising future direction would be to explore 2-methyltetrahydrofuran (2-MeTHF), which can be produced from bio-based raw materials and offers a more sustainable alternative to THF.28–30 The efficiency of Scheme 1, Step 1 reaction was compared with the product obtained using the traditional solvent, DMF, versus the product obtained using the new and more sustainable THF/water cosolvent. It is important to note that neither THF nor water can dissolve the lignin on their own as seen in Fig. S5a and S5b found in the SI. To solve this issue, a cosolvent system was employed using THF and water in a 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 volume ratio. In Fig. S3, found in the SI, a noticeably darker color was observed in the modified lignin reaction using DMF as the solvent. The product was stickier and more viscous than the product found using the novel THF/water cosolvent system, which was a much lighter brown color and was more of a powder with a much lower viscosity. Another metric used to analyze the effectiveness of each reaction is a weight yield. The conventional lignin modification using DMF resulted in a 62 wt% yield (Table S1). In contrast, performing the same reaction in a THF/water cosolvent increased the yield to 71 wt% (Table S1). This reaction scheme and 1H and 13C NMR can be found in the SI in Fig. S12–14. Note that these solvent comparison tests were conducted using DCC/DMAP catalysts, not the Mukaiyama reagent. These results additionally highlight not only the increased sustainability of our novel scheme, but also the surprisingly superior effectiveness of the method employed, and the quality of the novel product acquired.
50 volume ratio. In Fig. S3, found in the SI, a noticeably darker color was observed in the modified lignin reaction using DMF as the solvent. The product was stickier and more viscous than the product found using the novel THF/water cosolvent system, which was a much lighter brown color and was more of a powder with a much lower viscosity. Another metric used to analyze the effectiveness of each reaction is a weight yield. The conventional lignin modification using DMF resulted in a 62 wt% yield (Table S1). In contrast, performing the same reaction in a THF/water cosolvent increased the yield to 71 wt% (Table S1). This reaction scheme and 1H and 13C NMR can be found in the SI in Fig. S12–14. Note that these solvent comparison tests were conducted using DCC/DMAP catalysts, not the Mukaiyama reagent. These results additionally highlight not only the increased sustainability of our novel scheme, but also the surprisingly superior effectiveness of the method employed, and the quality of the novel product acquired.
Additionally, this new cosolvent system was tested on a basic esterification reaction using DCC/DMAP (Fig. S18). While this reaction did not proceed, the focus of this research remains on lignin, leaving this reaction open for future work.
For Scheme 3, Step 3, the graft copolymerization step, the cosolvent system using THF and water in a 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 ratio was also employed.
50 ratio was also employed.
3.2. Sustainable catalyst for lignin modification
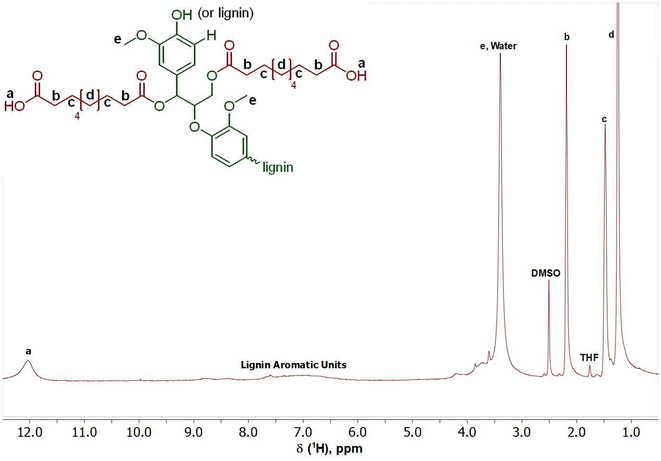
The first step to prepare the lignin-graft-copolymer is the modification of natural lignin. A new and sustainable catalytic system, (Mukaiyama reagent), was utilized to modify lignin.37 The Mukaiyama reagent facilitated the esterification reaction between the hydroxyl groups in natural lignin and the carboxylic acid groups of sebacic acid, as demonstrated in Scheme 1, Step 1. The terminal carboxylic acid groups of the modified lignin were later used for the graft copolymerization with PCL. The successful completion of the reaction in Scheme 1, Step 1 was confirmed using both 1H and 13C NMR (Fig. 1 and 2). The degree of acid modification was found to be 5.1532 mmol g−1 for the modified lignin. Details of the calculations are available in SI 6, and the SI 1H NMR spectrum is also provided in Fig. S6.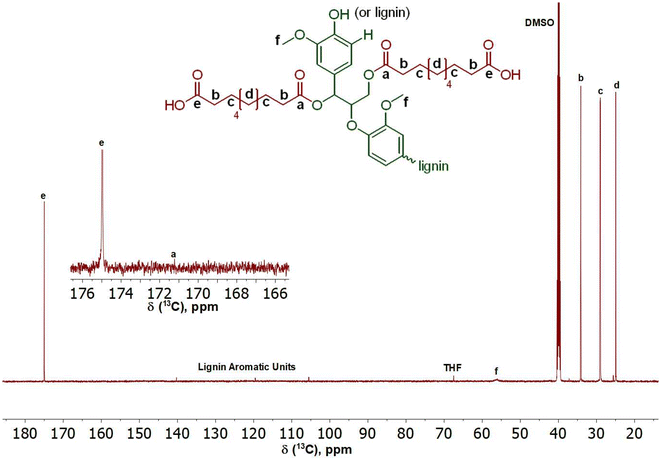 | ||
| Fig. 2 13C NMR of Modified Lignin; hydroxyl groups on lignin were consumed through esterification. New ester linkages of the modified lignin can be seen in peak “a”. | ||
The appearance of a small and broad peak in Fig. 1 around 12.00 ppm, labeled “a”, appeared from the end carboxylic acid groups’ hydrogen in the modified lignin after the reaction between lignin and sebacic acid.52 The presence of methoxy groups can be confirmed by the peak labeled “e” at 3.40 ppm. The peaks labeled “b, c, and d” correspond to the aliphatic carbon chains found within the structure of the attached sebacic acid that subsequently forms the modified lignin.52 The range of peaks found between 6.00 ppm and 9.00 ppm correspond to the various protons found within the unmodified structure of lignin.58,59 According to the 1H NMR data, the modification of natural lignin to introduce carboxylic acid functionalities was successfully occurred, as illustrated in Scheme 1, Step 1, using a more effective and sustainable esterification route.
In 13C NMR (Fig. 2), the presence of a small peak at 172.82 ppm in Fig. 2, labeled “a”, confirms the successful grafting of sebacic acid onto the natural lignin and the presence of an ester within the final structure.52 The larger peak at 175.03 ppm, labeled “e”, represents the carboxylic end groups of the introduced sebacic acid present in the final product.52 Additionally, peaks “b, c, and d” of the chemical structure of the modified lignin in Fig. 2 can be seen between 25.00 ppm and 35.00 ppm.52 These peaks represent the aliphatic carbon chains found within the structure of the attached sebacic acid that subsequently forms the modified lignin. The central and unmodified lignin structure appears between 105.00–140.00 ppm.
The 2D HSQC (1H–13C) NMR spectra shown in Fig. 3(a–c) offer detailed insights into the structural differences between the unmodified lignin and modified lignin. In Fig. 3(a), the characteristic signals corresponding to hydroxyl groups at the α, β, and γ positions of unmodified lignin are observed at δH/δC = 3.2–4.9 ppm/59–78 ppm.52,60–62 The methoxy group (–OCH3) signal displayed at δH/δC: 3.80 ppm/56 ppm.52,60,62 Altogether, the HSQC NMR spectrum of unmodified lignin showed the typical chemical structure of lignin with abundant hydroxyl groups (Fig. 3(a)). After modification, the signals corresponding to the α and γ hydroxyl groups disappeared in the HSQC NMR spectrum of the modified lignin (Fig. 3(b)). Moreover, the signals of the methylene unit (–CH2–) from the sebacic acid segment appeared at δH/δC: 2.18–2.20 ppm/28–40 ppm in Fig. 3(b).52 Thus, the consumption of α and γ hydroxyl groups after the reaction, along with the appearance of methylene signals in the modified lignin (Fig. 3(b)), confirms the successful incorporation of the sebacic acid unit into lignin.
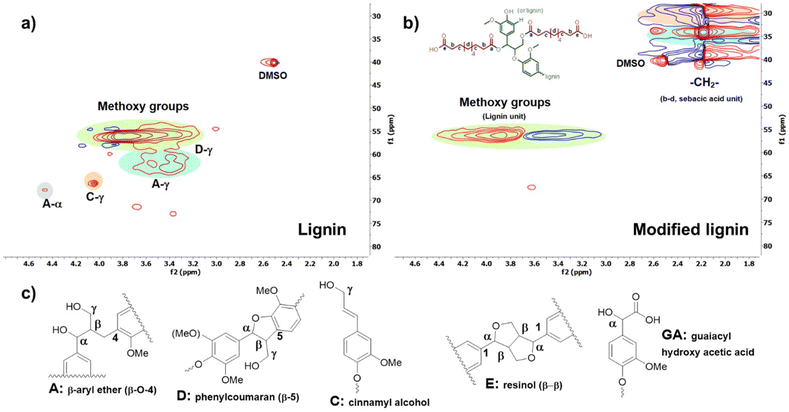 | ||
| Fig. 3 (a) 2D HSQC (1H–13C) NMR spectrum of raw lignin. (b) 2D HSQC (1H–13C) NMR spectrum of modified lignin. (c) Major chemical structural units of lignin. | ||
To test the effectiveness of the new Mukaiyama reagent system as a replacement for DCC/DMAP, both DMF and the THF/Water cosolvent system were applied in the catalyst reactions. The reaction scheme and 1H and 13C NMR for the reaction using the Mukaiyama reagent and DMF can be found in the SI in Fig. S15–17. The Mukaiyama reagent in DMF resulted in a yield of 69 wt% of the initial reactant mass (Table S1) and when the reaction was performed in the co-solvent system, the yield increased to 82 wt% of the initial reactant mass (Table S1). These findings, paired with detailed 1H and 13C NMR spectroscopy, demonstrate that the combined use of the new cosolvent system with the Mukaiyama reagent enhances the yield when compared to either system by itself.
Both natural and synthesized modified lignin demonstrated excellent solubility in the THF/water cosolvent, dimethyl formamide (DMF), dimethyl sulfoxide (DMSO), and N-methylpyrrolidone (NMP) as displayed in Fig. S5c and S5d (SI). The modified lignin produced using the THF/water cosolvent system retained a solubility profile similar to that of natural lignin because of the preserved aromatic lignin core. This comparable solubility of modified lignin is utilized to perform the esterification reaction in THF/water cosolvent using the Mukaiyama reagent in the graft polymerization (Scheme 3, Step 3).
3.3. Sustainable and recyclable polymer-bound catalyst for ring-opening polymerization
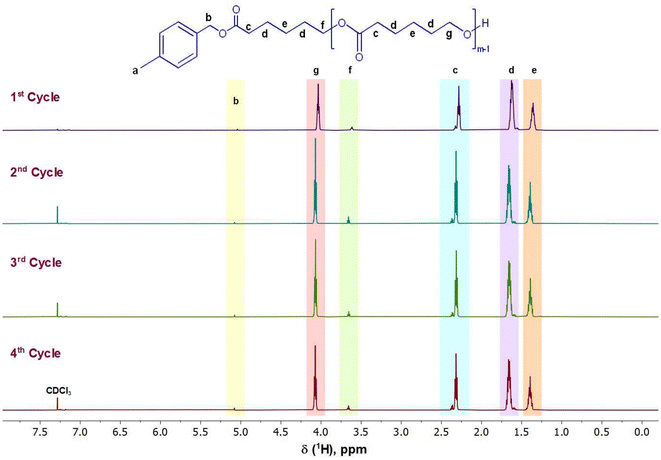
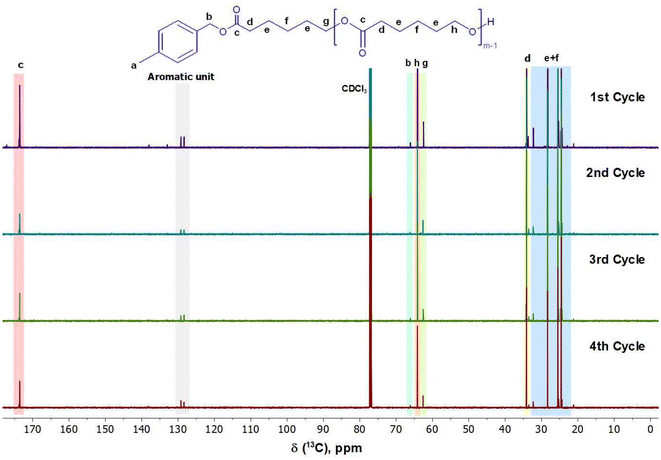
The second step in preparing the lignin-graft-copolymer was to synthesize the aliphatic polymer, PCL. In this work, we established a new sustainable catalytic system, using the PS-TBD catalyst to carry out a ROP of ε-caprolactone according to Scheme 2, Step 2. ε-Caprolactone was chosen for its simple lactone structure, ease of availability, degradability, and cost-effectiveness.63 The initiator, 4-methylbenzyl alcohol, was also used in conjunction with the catalyst. Although the ROP of ε-caprolactone is a well-established polymerization, the use of polymer-bound catalyst, PS-TBD is rare. The ROP was conducted under bulk conditions without a solvent. PS-TBD is easily separated from the polymer product solution by filtration during purification, as PS-TBD is not soluble in the solvent. The 1H NMR spectra in Fig. 4 confirms the successful ROP, validating the precise chemical structure of the synthesized PCL through specific proton assignments. For comparison, the ROP of ε-caprolactone was also conducted without the PS-TBD catalyst, while keeping all other conditions identical, to assess the catalytic role of PS-TBD. Although the PCL synthesis procedure still employs non-green solvents such as methanol, dichloromethane (DCM), and initiator (4-methylbenzyl alcohol), greener alternatives have been explored in the literature.64,65 Including ethanol to replace methanol; ethyl acetate could substitute for DCM; and bio-based alcohol could replace 4-methylbenzyl alcohol. Incorporating such alternatives would further enhance the sustainability of the synthesis and represent a promising direction for future work. It is also worth noting that while PS-bound TBD is effective for laboratory-scale demonstrations, industrial separation/recycling processes should be designed and evaluated for a large-scale application, while still preserving the key principles of the synthesis. The corresponding 1H and 13C NMR spectra of the resulting polymer products are presented in Fig. S9 and S10. Although polymer formation was observed, the yield was extremely low (<14%) compared to the ROP conducted with PS-TBD, which consistently achieved yields above 90% in repeated experiments. These outcomes show that PS-TBD is key to efficient polymerization.The recycled catalyst was used in four separate and repeated polymerizations, as shown in the four spectra in Fig. 4. More specifically, a single batch of PS-TBD catalyst is repeatedly used for conducting four separate ROP reactions in order to test the recyclability of PS-TBD. The reactions followed the same procedure and scheme, with the only variation being the use of the same catalyst in four consecutive runs to assess its durability and effectiveness after repeated use. The amount of catalyst recovered after each reaction was greater than 90% by weight, and the main reason for this minor weight loss can be attributed to physical loss (fractionation of PS resin solid and its bound catalysts) through handling and moving the catalyst. These 1H and 13C NMR spectra demonstrated that the polymerization of ε-caprolactone proceeded according to Scheme 2, Step 2.
In Fig. 4, one of the key peaks to focus on in this 1H NMR spectrum is the small peak labeled “b” at 5.04 ppm. This peak corresponds to the benzyl carbon found between the ester and the benzyl ring of the initiator.66,67 The inclusion of this peak demonstrates the integration of both the initiator and the ε-caprolactone within the final PCL structure. The other significant peak is labeled “g” found at 3.61 ppm.66,67 This proton is found at the hydroxyl-terminating end of PCL and confirms the successful ROP of ε-caprolactone into PCL. Other peaks present include “c, d, e, and f”. These four peaks correspond to the aliphatic carbon chains found within the repeating structure of the ring-opened ε-caprolactone.66,67
One of the key peaks to focus on in the 13C NMR spectrum, in Fig. 5, is the presence of peak “c” at 173.66 ppm. This peak indicates ester groups found within the PCL structure.66,67 Peak “b” at 66.11 ppm represents the benzyl carbon found between the ester and the benzyl ring of the initiator.66,67 Other peaks within the 13C NMR spectra include “d, e, f, g, and h” which are the aliphatic carbon chains found within the repeating unit of PCL.66,67 The aromatic unit of the initiator is observed between 125.00–130.00 ppm.
Another notable transformation is the slight shift between the cyclic structure of the ε-caprolactone and the aliphatic region of the PCL after ring-opening, indicating a change in the proton environment. The spectra of monomer, ε-caprolactone, can be found in Fig. S7 and S8 within the SI. A final point to consider is the height difference in Fig. 5, where the peaks of the repeating aliphatic unit are significantly larger than those of the terminating units at the ends of the PCL chain.66,67 This indicates the successful formation of multiple successive repeating units that make up the PCL chain.
Table 2 shows the molecular weights and molecular weight distribution (PDI: Polydispersity index) of the synthesized polymers The molecular weight and PDI data (Table 2) demonstrate that the polymer–bound PS–TBD catalyst remains highly recyclable and retains substantial catalytic activity over multiple reuse cycles. A gradual decrease in molecular weight from 3.78 to 2.37 kDa is observed with each successive cycle, corresponding to a 37% reduction in PCL molecular weight over the four cycles. This trend indicates a modest decline in catalytic efficiency with reuse. However, even in cycle 4, the reaction continues to produce polymer with a well-defined molecular weight (2.37 kDa) and a narrow molecular weight distribution (PDI = 1.11). This conclusion is further reflected by the consistently high product yields of 80 wt%, 78 wt%, 87 wt%, and 82 wt% for cycles 1–4, respectively. Further analysis of its recyclability beyond four cycles is currently underway, and the results will be reported in future work.
| Cyclea (#) | M w, GPC (kDa) | PDIc |
|---|---|---|
| a Each cycle represents a trial. b The Mw, GPC, measured in kDa, is the number-average molecular weight of each PCL product. c PDI represents the polydispersity index (molecular weight distribution) of each PCL as measured by GPC in THF solvent. | ||
| Cycle 1 | 3.78 | 1.13 |
| Cycle 2 | 3.05 | 1.08 |
| Cycle 3 | 2.44 | 1.18 |
| Cycle 4 | 2.37 | 1.11 |
3.4. Sustainable catalyst for graft copolymerization
After lignin modification and PCL preparation via ROP, the final copolymer, lignin-graft-PCL, was synthesized. The final graft copolymerization step, an esterification reaction, was successfully accomplished using the THF/Water cosolvent system and 2-chloro-1-methylpyridinium iodide (Mukaiyama reagent).37 This sustainable esterification route forms ester linkages between the carboxylic acid groups of the modified lignin and the terminal hydroxyl groups of PCL. The yield of the final copolymer (lignin-graft-PCL) was 80 wt% of the initial reactant mass. This represents the highest yield obtained in the study, attributed to the complementary effects of the cosolvent system and the Mukaiyama reagent found in the lignin modification step.
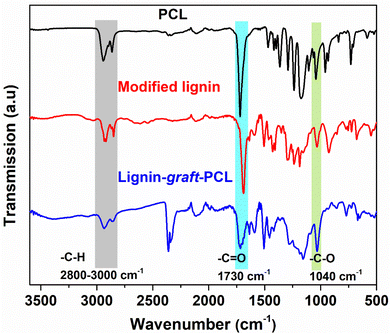
Fig. 6 displays the FT-IR spectra of PCL, modified lignin, and the final copolymer, lignin-graft-PCL. The strong bands at 1730 cm−1 attributed to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration of the ester groups, which were present in PCL (black), modified lignin (red), and lignin-graft-PCL (blue, Fig. 6).52,58,68 The ester linkages in the PCL correspond to the repeating ester units that originated after the ring-opening of ε-caprolactone. The ester bonds in the modified lignin correspond to the incorporated sebacic acid unit within the structure. The lignin-graft-PCL exhibited two peaks for the C
O stretching vibration of the ester groups, which were present in PCL (black), modified lignin (red), and lignin-graft-PCL (blue, Fig. 6).52,58,68 The ester linkages in the PCL correspond to the repeating ester units that originated after the ring-opening of ε-caprolactone. The ester bonds in the modified lignin correspond to the incorporated sebacic acid unit within the structure. The lignin-graft-PCL exhibited two peaks for the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching band within the region of 1710–1730 cm−1, signifying the presence of two distinct types of ester linkages from PCL and modified lignin units.52,58,68 The characteristic C–H stretching peaks from the aliphatic linkages were found between 2800–3000 cm−1 (highlighted in gray, Fig. 6). Moreover, the characteristic peak of C–O stretching from the ester group was observed at 1040 cm−1 (highlighted in green, Fig. 6).52,58,68 Thus, the results confirm the successful integration of PCL and modified lignin into the graft copolymer, lignin-graft-PCL.
O stretching band within the region of 1710–1730 cm−1, signifying the presence of two distinct types of ester linkages from PCL and modified lignin units.52,58,68 The characteristic C–H stretching peaks from the aliphatic linkages were found between 2800–3000 cm−1 (highlighted in gray, Fig. 6). Moreover, the characteristic peak of C–O stretching from the ester group was observed at 1040 cm−1 (highlighted in green, Fig. 6).52,58,68 Thus, the results confirm the successful integration of PCL and modified lignin into the graft copolymer, lignin-graft-PCL.
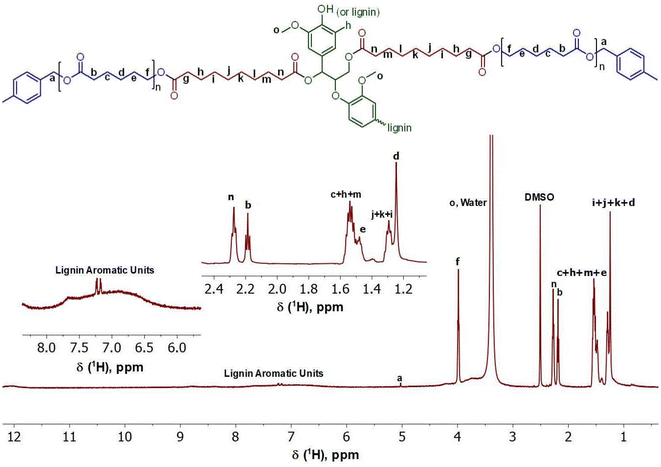
As shown in Fig. 7 and 8, 1H NMR and 13C NMR were performed to analyze the chemical structure of the lignin-graft-PCL. For the 1H NMR (Fig. 7), the peak labeled “f” located at 3.98 ppm corresponds to the linkage of PCL with the modified lignin to create the copolymer structure.52,59,66 The inclusion of the protons found on the benzyl carbon between the ester and the benzyl ring of the initiator can be seen in peak “a” located at 5.04 ppm.66,67 The range of peaks found between 6.00 ppm and 9.00 ppm correspond to the various protons found within the unmodified lignin structure of lignin along with the methoxy groups seen in peak “o” at 3.39 ppm.52,59 A large number of protons on aliphatic carbon chains are also found in the final copolymer structure, labeled “b–n”.52,66
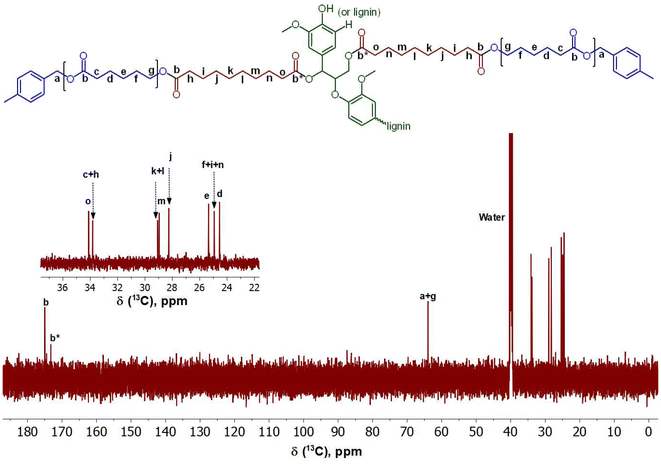 | ||
| Fig. 8 13C NMR of lignin-graft-PCL; The newly formed ester bonds between lignin and PCL, as well as the PCL terminal's ester, are observed at peak “a + g”. | ||
In the 13C NMR (Fig. 8), the linkage between the PCL and modified lignin can also be seen at 63.96 ppm in peak “g”.52,59,66 The peak at 63.96 ppm contains the signal for the benzyl carbon “a”, located between the ester and the benzyl ring of the initiator in the PCL, further confirming the successful synthesis of lignin-graft-PCL.66,67 Peaks “b” and “b*” represent the ester linkages found within the copolymer. Peak “b*” represents the ester linkage between sebacic acid and natural lignin present in the modified lignin.52,59,66 The unmodified lignin appears in the range of 70.00–140.00 ppm, but high baseline noise prevents clear resolution of distinct peaks. The inset in Fig. 8 shows the carbons of the aliphatic chain present in the copolymer. These include peaks “c–f” and “h–o” between 24.00 and 34.00 ppm.52,59,66
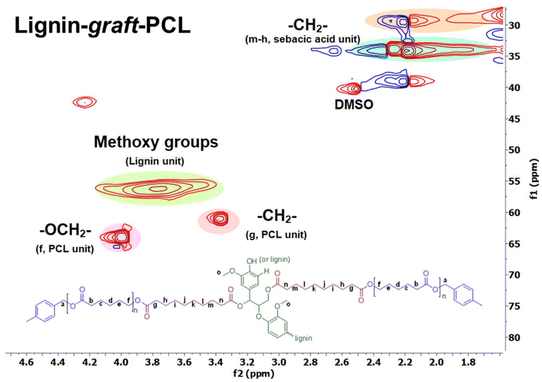
Fig. 9 presents the 2D HSQC (1H–13C) NMR spectrum of lignin-graft-PCL. The signals of the PCL segment are observed at δH/δC = 4.00 ppm/64 ppm (–OCH2–) and 3.37 ppm/61 ppm (–CH2–). The methoxy group signal from the lignin unit appears at δH/δC = 3.78 ppm/56 ppm (Fig. 9). In addition, signals corresponding to the sebacic acid segment are detected at δH/δC = 2.18–2.22 ppm/28–40 ppm, consistent with those of the modified lignin. Hence, the overall results confirm the inclusion of all structural components within the lignin-graft-PCL copolymer.
3.5. Thermal properties of lignin-graft-PCL
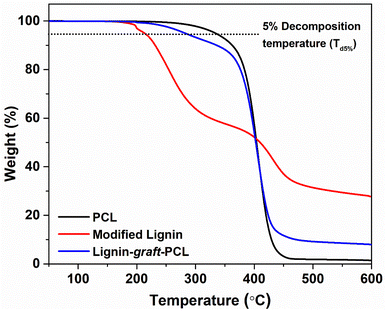
Thermal property analysis was performed on the final copolymer to confirm its structure and assess thermal stability. The results showed a significant improvement in the thermal stability of modified lignin after incorporation of PCL into the copolymer. As shown in Fig. 10, the temperature (Td5%) at which 5% of the total mass degraded was calculated for all three samples, PCL, modified lignin, and lignin-graft-PCL. The Td5% was found to be 213 °C for modified lignin, 334 °C for PCL, and 282 °C for lignin-graft-PCL.69 It is worthwhile to compare the copolymer's thermal behavior with that of similar lignin-based structures to better assess the potential of the proposed green alternatives. According to previous reports, the Td5% for lignin-graft-poly(lactide) is near 300 °C and that of lignin-graft-polyethylene brassylate is found to be around 350 °C.52,70 These similar polymers exhibit slightly higher thermal stability than PCL-graft-lignin, which can be attributed to the use of ε-caprolactone as the monomer building block for the copolymer. The observed thermal stability of lignin-graft-PCL suggests it can be processed under conventional melt-processing conditions and could be suited for applications such as biodegradable films or thermally stable composite materials, owing to its ability to efficiently absorb and release thermal energy.69 This research enables the selection of monomers to be tailored to the specific requirements of the intended application. Incorporating different monomers into the lignin copolymer structure can result in a range of thermal properties.4. Conclusions
In this work, we report a similar lignin-based polyester to that of previous research, using the Mukaiyama reagent and a THF/water cosolvent system as an effective replacement for a DMF solvent, and DCC and DMAP reactant system. The modified lignin component of the reaction was found in higher yields using the Mukaiyama reagent and a THF/water cosolvent in place of conventional DMF. As a grafting polymeric unit, PCL was created by the ring-opening polymerization of ε-caprolactone using a novel PS-TBD catalyst in the presence of an initiator. The new catalyst shows evidence of reasonably high effectiveness through multiple recycles during the ROP reaction. Finally, both the modified lignin and PCL are utilized in the synthesis of lignin-graft-PCL resulting in a yield 80 wt% of the initial reactant mass. Subsequently, chemical structures were confirmed using 1H NMR, 13C NMR, and FT-IR spectroscopy. The thermal property studies of lignin-graft-PCL showed comparable thermal stability to copolymers of similar size and composition, with a Td5% of 283 °C. Therefore, this research is a step forward in the field of green and sustainable biomass-based polymer chemistry and leaves the door open to potential improvements and further research, specifically by investigating the recyclability of polymer-bound catalysts.Conflicts of interest
There are no conflicts to declare.Data availability
All characterization data of the synthesized compounds are included in the manuscript and supplementary information. Additional information, such as extremely detailed experimental procedures, optimized reaction conditions, and data not discussed in the article, are available from the corresponding author upon reasonable request.Supplementary information (SI): characterization data for all synthesized compounds, are provided in the Figures, Tables, SI, and Experimental section. See DOI: https://doi.org/10.1039/d5lp00387c.
Acknowledgements
The authors would like to thank Dr Banghao Chen for the support of NMR facilities and Dr J. S. Raaj Vellore Winfred for the support of FT-IR and SDT Thermal instrumentation facilities. This project was conducted with the support of the Industrial Technology Innovation Program ([RS-2024-00434688, Development of polyurethane coatings prepared from more than 40% CO2-captured monomers: a non-isocyanate synthetic process and processing technologies for textile products]) funded by the Ministry of Trade, Industry & Energy of the Republic of Korea. Research was sponsored by the Army Research Office and was accomplished under Grant Number W911NF2510129. The views and conclusions contained in this document are those of the authors and should not be interpreted as representing the official policies, either expressed or implied, of the Army Research Office or the U.S. Government. The U.S. Government is authorized to reproduce and distribute reprints for Government purposes notwithstanding any copyright notation herein.References
- T. H. Epps, L. T. J. Korley, T. Yan, K. L. Beers and T. M. Burt, Sustainability of Synthetic Plastics: Considerations in Materials Life-Cycle Management, JACS Au, 2022, 2(1), 3–11 CrossRef PubMed.
- D. E. Fagnani, J. L. Tami, G. Copley, M. N. Clemons, Y. D. Y. L. Getzler and A. J. McNeil, 100th Anniversary of Macromolecular Science Viewpoint: Redefining Sustainable Polymers, ACS Macro Lett., 2021, 10(1), 41–53 CrossRef CAS PubMed.
- B. M. Upton and A. M. Kasko, Strategies for the Conversion of Lignin to High-Value Polymeric Materials: Review and Perspective, Chem. Rev., 2016, 116(4), 2275–2306 CrossRef CAS PubMed.
- W. Schutyser, T. Renders, S. Van den Bosch, S. Koelewijn, G. T. Beckham and B. F. Sels, Chemicals from Lignin: An Interplay of Lignocellulose Fractionation, Depolymerisation, and Upgrading A Critical Review of Past and Recent Developments in the Production of Chemicals from Lignin, Focusing on Lignocellulose Fractionation, Lignin Depolymerisation, and Upgrading. Chemicals from Lignin: An Interplay of Lignocellulose Fractionation, Depolymerisation, and Upgrading†, Chem. Soc. Rev., 2018, 47, 852 RSC.
- R. M. O′dea, J. A. Willie and T. H. Epps, 100th Anniversary of Macromolecular Science Viewpoint: Polymers from Lignocellulosic Biomass. Current Challenges and Future Opportunities, 2020 Search PubMed.
- Q. Li, D. Cao, M. T. Naik, Y. Pu, X. Sun, P. Luan, A. J. Ragauskas, T. Ji, Y. Zhao, F. Chen, Y. Zheng and H. Zhu, Molecular Engineering of Biorefining Lignin Waste for Solid-State Electrolyte, ACS Sustainable Chem. Eng., 2022, 10(27), 8704–8714 CrossRef CAS.
- B. M. Upton and A. M. Kasko, Strategies for the Conversion of Lignin to High-Value Polymeric Materials: Review and Perspective, Chem. Rev., 2016, 116(4), 2275–2306 CrossRef CAS PubMed.
- F. H. B. Sosa, D. O. Abranches, A. M. Da Costa Lopes, J. A. P. Coutinho and M. C. Da Costa, Kraft Lignin Solubility and Its Chemical Modification in Deep Eutectic Solvents, ACS Sustainable Chem. Eng., 2020, 8(50), 18577–18589 CrossRef CAS.
- T. Akiba, A. Tsurumaki and H. Ohno, Induction of Lignin Solubility for a Series of Polar Ionic Liquids by the Addition of a Small Amount of Water, Green Chem., 2017, 19, 2260 RSC.
- A. Tolbert, H. Akinosho, R. Khunsupat, A. K. Naskar and A. J. Ragauskas, Characterization and Analysis of the Molecular Weight of Lignin for Biorefining Studies, Biofuels, Bioprod. Biorefin., 2014, 8(6), 836–856 CrossRef CAS.
- J. D. Wade, I. Bakunina, A. J. Ragauskas, M. Li and Y. Pu, Current Understanding of the Correlation of Lignin Structure with Biomass Recalcitrance, Front. Chem., 2016, 4, 45 Search PubMed.
- H. Wang, H. Ben, H. Ruan, L. Zhang, Y. Pu, M. Feng, A. J. Ragauskas and B. Yang, Effects of Lignin Structure on Hydrodeoxygenation Reactivity of Pine Wood Lignin to Valuable Chemicals, ACS Sustainable Chem. Eng., 2017, 5(2), 1824–1830 CrossRef CAS.
- J. V. Vermaas, M. F. Crowley and G. T. Beckham, Molecular Lignin Solubility and Structure in Organic Solvents, ACS Sustainable Chem. Eng., 2020, 8(48), 17839–17850 CrossRef CAS.
- E. I. Evstigneyev and S. M. Shevchenko, Structure, Chemical Reactivity and Solubility of Lignin: A Fresh Look, Wood Sci. Technol., 2019, 53(1), 7–47 CrossRef CAS.
- H. Liu and H. Chung, Lignin-Based Polymers via Graft Copolymerization, J. Polym. Sci., Part A: Polym. Chem., 2017, 55(21), 3515–3528 CrossRef CAS.
- A. Duval, F. Vilaplana, C. Crestini and M. Lawoko, Solvent Screening for the Fractionation of Industrial Kraft Lignin, Holzforschung, 2016, 70(1), 11–20 CrossRef CAS.
- B. S. Furniss, A. J. Hannaford, V. Rogers, P. W. G. Smith and A. R. Tatchell, Vogel's Textbook of Practical Organic Cgemistry, 1989 Search PubMed.
- C. D. Mino, A. J. Clancy, A. Sella, C. A. Howard, T. F. Headen, A. G. Seel and N. T. Skipper, Weak Interactions in Dimethyl Sulfoxide (DMSO)−Tertiary Amide Solutions: The Versatility of DMSO as a Solvent, J. Phys. Chem. B, 2023, 127, 10 Search PubMed.
- View of An Overview on Common Organic Solvents and Their Toxicity.
- J. Zhang, G. B. White, M. D. Ryan, A. J. Hunt and M. J. Katz, Dihydrolevoglucosenone (Cyrene) As a Green Alternative to N,N-Dimethylformamide (DMF) in MOF Synthesis, ACS Sustainable Chem. Eng., 2016, 4(12), 7186–7192 CrossRef CAS.
- T. Brouwer and B. Schuur, Dihydrolevoglucosenone (Cyrene), a Biobased Solvent for Liquid–Liquid Extraction Applications, ACS Sustainable Chem. Eng., 2020, 8, 14807–14817 CrossRef CAS.
- N. A. Stini, P. L. Gkizis and C. G. Kokotos, Cyrene: A Bio-Based Novel and Sustainable Solvent for Organic Synthesis, Green Chem., 2022, 24(17), 6435–6449 RSC.
- I. T. Horváth, H. Mehdi, V. Fábos, L. Boda and L. T. Mika, γ-Valerolactone—a Sustainable Liquid for Energy and Carbon-Based Chemicals, Green Chem., 2008, 10(2), 238–242 RSC.
- V. Fábos, G. Koczó, H. Mehdi, L. Boda and I. T. Horváth, Bio-Oxygenates and the Peroxide Number: A Safety Issue Alert, Energy Environ. Sci., 2009, 2(7), 767–769 RSC.
- Z. Xue, X. Zhao, R. C. Sun and T. Mu, Biomass-Derived γ-Valerolactone-Based Solvent Systems for Highly Efficient Dissolution of Various Lignins: Dissolution Behavior and Mechanism Study, ACS Sustainable Chem. Eng., 2016, 4(7), 3864–3870 CrossRef CAS.
- G. Wang, X. Liu, B. Yang, C. Si, A. M. Parvez, J. Jang and Y. Ni, Using Green γ-Valerolactone/Water Solvent to Decrease Lignin Heterogeneity by Gradient Precipitation, ACS Sustainable Chem. Eng., 2019, 7(11), 10112–10120 CrossRef CAS.
- N. I. Haykir and J. Viell, Challenges and Opportunities for Gamma-Valerolactone in Biomass Pretreatment, Ind. Eng. Chem. Res., 2024, 63(28), 12251–12264 CrossRef CAS.
- V. Pace, P. Hoyos, L. Castoldi, P. Domínguez De María and A. R. Alcántara, 2-Methyltetrahydrofuran (2-MeTHF): A Biomass-Derived Solvent with Broad Application in Organic Chemistry, ChemSusChem, 2012, 5(8), 1369–1379 CrossRef CAS PubMed.
- M. E. Farahat, G. C. Welch, M. E. Farahat and G. C. Welch, Slot-Die Coated Organic UV Indicators and Filters Processed from Green Solvents, Adv. Sustainable Syst., 2022, 6(2), 2100055 CrossRef CAS.
- G. W. Huber, S. Iborra and A. Corma, Synthesis of Transportation Fuels from Biomass: Chemistry, Catalysts, and Engineering, Chem. Rev., 2006, 106(9), 4044–4098, DOI:10.1021/CR068360D/ASSET/IMAGES/MEDIUM/CR068360DF00034.GIF.
- 2-Methyltetrahydrofuran BioRenewable, anhydrous, = 99, Inhibitor-free 96-47-9.
- D. M. Kialengila, K. Wolfs, J. Bugalama, A. Van Schepdael and E. Adams, Full Evaporation Headspace Gas Chromatography for Sensitive Determination of High Boiling Point Volatile Organic Compounds in Low Boiling Matrices, J. Chromatogr. A, 2013, 1315, 167–175 CrossRef PubMed.
- B. Neises and W. Steglich, Simple Method for the Esterification of Carboxylic Acids, Angew. Chem., Int. Ed. Engl., 1978, 17(7), 522–524 CrossRef.
- O. Coulembier and P. Dubois, 4-Dimethylaminopyridine-Based Organoactivation: From Simple Esterification to Lactide Ring-Opening “Living” Polymerization, J. Polym. Sci., Part A: Polym. Chem., 2012, 50(9), 1672–1680 CrossRef CAS.
- A. Jordan, K. D. Whymark, J. Sydenham and H. F. Sneddon, A Solvent-Reagent Selection Guide for Steglich-Type Esterification of Carboxylic Acids, Green Chem., 2021, 23(17), 6405–6413 RSC.
- J. Demarteau, N. Vora, J. D. Keasling, B. A. Helms and C. D. Scown, Lower-Cost, Lower-Carbon Production of Circular Polydiketoenamine Plastics, ACS Sustainable Chem. Eng., 2022, 10(8), 2740–2749 CrossRef CAS.
- T. Mukaiyama, M. Usui, E. Shimada and K. Saigo, A convenient method for the synthesis of carboxylic esters, Chem. Lett., 1975, 4(10), 1045–1048 CrossRef.
- W. J. Nahm, A. Schreck, J. Prasad, E. Rapoport, J. Fragoso and C. Vega, Dicyclohexylcarbodiimide Commonly Causes Contact Dermatitis but Can Also Be Acutely Tissue Cytotoxic, Contact Dermatitis, 2022, 86(1), 48–50 CrossRef CAS PubMed.
- L. Simón and J. M. Goodman, The Mechanism of TBD-Catalyzed Ring-Opening Polymerization of Cyclic Esters, J. Org. Chem., 2007, 72(25), 9656–9662 CrossRef PubMed.
- M. Bouyahyi, M. P. F. Pepels, A. Heise and R. Duchateau, ω-Pentandecalactone Polymerization and ω-Pentadecalactone/Îμ -Caprolactone Copolymerization Reactions Using Organic Catalysts, Macromolecules, 2012, 45(8), 3356–3366 CrossRef CAS.
- E. Fritz-Langhals, Unique Superbase TBD (1,5,7-Triazabicyclo[4.4.0]Dec-5-Ene): From Catalytic Activity and One-Pot Synthesis to Broader Application in Industrial Chemistry, Org. Process Res. Dev., 2022, 26(11), 3015–3023 CrossRef CAS.
- R. M. Shakaroun, P. Jéhan, A. Alaaeddine, J. F. Carpentier and S. M. Guillaume, Organocatalyzed Ring-Opening Polymerization (ROP) of Functional β-Lactones: New Insights into the ROP Mechanism and Poly(Hydroxyalkanoate)s (PHAs) Macromolecular Structure, Polym. Chem., 2020, 11(15), 2640–2652 RSC.
- C. G. Jaffredo, J. F. Carpentier and S. M. Guillaume, Organocatalyzed Controlled ROP of β-Lactones towards Poly(Hydroxyalkanoate)s: From β-Butyrolactone to Benzyl β-Malolactone Polymers, Polym. Chem., 2013, 4(13), 3837–3850 Search PubMed.
- R. Papagna, D. Kutzinski and S. M. Huber, Polymer-Bound Halogen Bonding Organocatalysis**, Eur. J. Org. Chem., 2022, 2022(31), e202200852 Search PubMed.
- R. K. Meena, P. Fageria, A. Sharma, A. Gaur, S. Gulati and R. Kumari, Achiral Polymer-Supported Organic Reagents, Polymer Supported Organic Catalysts, 2024, pp. 31–63 Search PubMed.
- C. A. McNamara, M. J. Dixon and M. Bradley, Recoverable Catalysts and Reagents Using Recyclable Polystyrene-Based Supports, Chem. Rev., 2002, 102(10), 3275–3300 Search PubMed.
- M. Alonzi, M. P. Bracciale, A. Broggi, D. Lanari, A. Marrocchi, M. L. Santarelli and L. Vaccaro, Synthesis and Characterization of Novel Polystyrene-Supported TBD Catalysts and Their Use in the Michael Addition for the Synthesis of Warfarin and Its Analogues, J. Catal., 2014, 309, 260–267 Search PubMed.
- S. Matsukawa and S. Fujikawa, Polystyrene-Supported TBD as an Efficient and Reusable Organocatalyst for Cyanosilylation of Aldehydes, Ketones, and Imines, Tetrahedron Lett., 2012, 53(9), 1075–1077 Search PubMed.
- N. D. Vala, H. H. Jardosh and M. P. Patel, PS-TBD, Triggered General Protocol for the Synthesis of 4H-Chromene, Pyrano[4,3-b]Pyran and Pyrano[3,2-c]Chromene Derivatives of 1H-Pyrazole and Their Biological Activities, Chin. Chem. Lett., 2016, 27(1), 168–172 CrossRef CAS.
- S. Matsukawa and Y. Mouri, A Mild and Regioselective Ring-Opening of Aziridines with Acid Anhydride Using TBD or PS-TBD as a Catalyst, Molecules, 2015, 20(10), 18482–18495 CrossRef CAS PubMed.
- S. Matsukawa, J. Kimura and M. Yoshioka, TBD- or PS-TBD-Catalyzed One-Pot Synthesis of Cyanohydrin Carbonates and Cyanohydrin Acetates from Carbonyl Compounds, Molecules, 2016, 21(8), 1030 CrossRef PubMed.
- S. Kim and H. Chung, Synthesis and Characterization of Lignin- Graft-Poly(Ethylene Brassylate): A Biomass-Based Polyester with High Mechanical Properties, ACS Sustainable Chem. Eng., 2021, 9(44), 14766–14776 CrossRef CAS.
- S. Kim and H. Chung, Fully Biomass-Based Biodegradable Polymers from Lignin and Raw Castor Oil: Lignin- Graft -Castor Oil, Polym. Chem., 2023, 14(35), 4126–4137 Search PubMed.
- Bentley's Textbook of Pharmaceutics – E-Book – Sanjay Kumar Jain – Google Books.
- O. Gordobil, E. Robles, I. Egüés and J. Labidi, Lignin-Ester Derivatives as Novel Thermoplastic Materials, RSC Adv., 2016, 6(90), 86909–86917 Search PubMed.
- R. Buhaibeh, T. Richard, R. M. Gauvin, M. Sauthier and C. Dumont, Catalytic Hydroesterification of Lignin: A Versatile and Efficient Entry into Fully Biobased Tunable Materials, Green Chem., 2023, 25(5), 1842–1851 RSC.
- A. Dzienia, P. Maksym, B. Hachuła, M. Tarnacka, T. Biela, S. Golba, A. ZiÈ©ba, M. Chorażewski, K. Kaminski and M. Paluch, Studying the Catalytic Activity of DBU and TBD upon Water-Initiated ROP of ε-Caprolactone under Different Thermodynamic Conditions, Polym. Chem., 2019, 10(44), 6047–6061 Search PubMed.
- Z. Shi, G. Xu, J. Deng, M. Dong, V. Murugadoss, C. Liu, Q. Shao, S. Wu and Z. Guo, Structural Characterization of Lignin from D. Sinicus by FTIR and NMR Techniques, Green Chem. Lett. Rev., 2019, 12(3), 235–243 Search PubMed.
- E. A. Capanema, M. Y. Balakshin and J. F. Kadla, A Comprehensive Approach for Quantitative Lignin Characterization by NMR Spectroscopy, J. Agric. Food Chem., 2004, 52(7), 1850–1860 CrossRef CAS PubMed.
- L. Shuai, M. T. Amiri, Y. M. Questell-Santiago, F. Héroguel, Y. Li, H. Kim, R. Meilan, C. Chapple, J. Ralph and J. S. Luterbacher, Formaldehyde Stabilization Facilitates Lignin Monomer Production during Biomass Depolymerization, Science, 2016, 354(6310), 329–333 CrossRef CAS PubMed.
- A. Ghorai and H. Chung, CO2 and Lignin-Based Sustainable Polymers with Closed-Loop Chemical Recycling, Adv. Funct. Mater., 2024, 34(39), 2403035 CrossRef CAS.
- A. Ghorai and H. Chung, Ionic Lignin Polymers for Controlled CO2 Capture, Release, and Conversion into High-Value Chemicals, Adv. Mater., 2024, 36(38), 2406610 CrossRef CAS PubMed.
- D. Goldberg, A Review of the Biodegradability and Utility of Poly(Caprolactone), J. Environ. Polym. Degrad., 1995, 3(2), 61–67 CrossRef CAS.
- R. K. Henderson, C. Jimenez-Gonzalez, D. J. C. Constable, S. A. Alston, G. G. A. Inglis, G. Fisher, J. Sherwood, S. P. Binks and A. D. Curzons, Expanding GSK's solvent selection guide-Embedding sustainability into solvent selection starting at medicinal chemistry, Green Chem., 2011, 13, 854–862 RSC.
- F. P. Byrne, S. Jin, G. Paggiola, T. H. M. Petchey, J. H. Clark, T. J. Farmer, A. J. Hunt, C. Robert McElroy and J. Sherwood, Tools and Techniques for Solvent Selection: Green Solvent Selection Guides, Sustainable Chem. Processes, 2016, 4(1), 1–24 Search PubMed.
- K. Takizawa, C. Tang and C. J. Hawker, Molecularly Defined Caprolactone Oligomers and Polymers: Synthesis and Characterization, J. Am. Chem. Soc., 2008, 130(5), 1718–1726 CrossRef CAS PubMed.
- K. Schäler, A. Achilles, R. Bärenwald, C. Hackel and K. Saalwächter, Dynamics in Crystallites of Poly(ε-Caprolactone) as Investigated by Solid-State NMR, Macromolecules, 2013, 46(19), 7818–7825 CrossRef.
- O. Derkacheva and D. Sukhov, Investigation of Lignins by FTIR Spectroscopy, Macromol. Symp., 2008, 265(1), 61–68 Search PubMed.
- S. Kim and H. Chung, Biodegradable Polymers: From Synthesis Methods to Applications of Lignin- Graft –Polyester, Green Chem., 2024, 26(21), 10774–10803 RSC.
- L. E. Chile, S. J. Kaser, S. G. Hatzikiriakos and P. Mehrkhodavandi, Synthesis and Thermorheological Analysis of Biobased Lignin-Graft-Poly(Lactide) Copolymers and Their Blends, ACS Sustainable Chem. Eng., 2018, 6(2), 1650–1661 Search PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |