Open Access Article
Open Access ArticlePrecision surface engineering of metallic biomaterials: translating cell-instructive nanoscale topographies from bench to bone-interfacing implants
Alp Ozgun a and
Fabio Variola
a and
Fabio Variola *bcde
*bcde
aIndependent Researcher, Ottawa, K1G 1L6, Canada
bFaculty of Engineering, Department of Mechanical Engineering, University of Ottawa, Ottawa K1N 6N5, Canada. E-mail: fabio.variola@uottawa.ca
cOttawa-Carleton Institute for Biomedical Engineering (OCIBME), Ottawa K1N 6N5, Canada
dDepartment of Cellular and Molecular Medicine, University of Ottawa, Ottawa K1N 6N5, Canada
eChildren's Hospital of Eastern Ontario (CHEO), Ottawa K1H 8L1, Canada
First published on 27th April 2026
Abstract
Metallic biomaterials remain foundational to orthopedic, spinal and dental implants owing to their mechanical properties, corrosion resistance and biocompatibility. Yet, unsatisfactory osseointegration and implant failure persist, often driven by limited stability, poor bone quality and dysregulated host immune responses. Over the past two decades, nanoscale surface engineering has consolidated its role as a powerful strategy to tune early cell–material interactions and downstream tissue remodeling, with compelling evidence that nanotopographical features regulate cell- and tissue-level functions. Despite a large mechanistic and preclinical literature, clinical translation of cell-instructive nanotopographies remains constrained by manufacturing scalability on complex 3D implant geometries, metrological and process reproducibility, and an enduring in vitro–in vivo disconnect driven by factors such as simplified test systems, dynamic protein adsorption phenomena and interspecies variability, among others. In this perspective, we examine precision nanoscale topographical control as a design variable for bone-interfacing metallic implants and synthesize advances in top-down and bottom-up nanofabrication routes, from deterministic lithographies to scalable anodization and laser texturing. We critically evaluate preclinical model systems spanning 2D assays, 3D and microphysiological platforms, ex vivo tissues and animal studies, emphasizing how model selection shapes mechanistic inference and translational predictability. Finally, we discuss potential pathways toward clinical adoption to enable next-generation implant surfaces that deliver effective osseointegration and long-term clinical performance.
1. Introduction
Metallic biomaterials (e.g., titanium, stainless steels, cobalt–chromium alloys, tantalum) have revolutionized orthopedic, spinal and dental implants by combining a native biocompatibility with mechanical strength, tribological performance, corrosion resistance and scalable manufacturing processes.1–3 Despite these benefits and their widespread use, challenges in achieving optimal osseointegration (i.e., the creation of a stable interface between the host bone tissue and the implant surface for structural and functional support) and long-term success are frequently associated with factors such as poor implant stability, insufficient bone quality at the interface and adverse host immune response, all of which can compromise the biological fixation process and may ultimately result in implant loosening and/or premature failure.1,4–6 To address these limitations and enhance the biological integration of metallic biomaterials, considerable research has focused on modifying the physicochemical properties of implant surfaces to improve their interactions with the surrounding tissues.7–9 Among the strategies adopted to promote a more effective and durable osseointegration, designing the nanoscale surface topography has rapidly emerged as a particularly effective approach to control key cell- and tissue-level events at the bone–implant interface.1,10–12 In fact, it is now well know that the nanotopographical features of surfaces influence cell adhesion, alignment, migration, proliferation and differentiation in vitro,13–16 while also modulating the early immune response17,18 and bone remodeling processes in vivo,19,20 which together are cell- and tissue-level phenomena that contribute to determine peri-implant bone formation and mechanical fixation over time. However, despite a large body of literature reporting extremely promising nanostructured biomaterials for prospective use in patients, the translation of scientific discoveries into medically approved devices requires clinical validation and regulatory approval,21 as well as scalable and cost-effective manufacturing processes. Several persistent roadblocks continue to hinder the successful implementation of cell-instructive surfaces to the biomedical implant manufacturing sector. For example, aside from scalability issues preventing promising surface modification technologies developed in the laboratory to be applicable to the large surface areas and complex geometries of biomedical implants (e.g., dental screw, spinal fixation devices) without compromising accuracy and reproducibility, there is also a large knowledge gap between in vitro research and the performance of biomaterials in humans. The exciting potential of candidate biomaterials demonstrated in vitro under the simplified and physiologically unrealistic conditions of conventional methods (i.e., 2-dimensional cell cultures) does not directly translate to the more complex architecture and dynamic nature of biological tissues.22–27 In addition, interspecies differences and large experimental variability create a supplementary bottleneck in biomaterials development during data extrapolation from animal testing to humans.22–27This perspective article examines precision surface engineering and discusses challenges and opportunities for the clinical adoption of cell-instructive nanotopographies for bone-interfacing implants. Through a critical analysis of current limitations related, for example, to scalability, throughput, reproducibility and the in vitro–in vivo disconnect, this work identifies the foundational requirements for advancing nanoscale surface modification strategies toward next-generation orthopedic, spinal, and dental implants designed to promote robust osseointegration and long-term clinical success. Given the breadth and continued expansion of the field, this work does not aim at providing an exhaustive survey of the literature, but instead focuses on representative and illustrative studies to distill key principles, unresolved challenges and emerging opportunities relevant to clinical translation.
2. The biological rationale for nanoscale topographical precision
2.1. Cell–surface interactions: from native ECM to synthetic metallic biomaterials
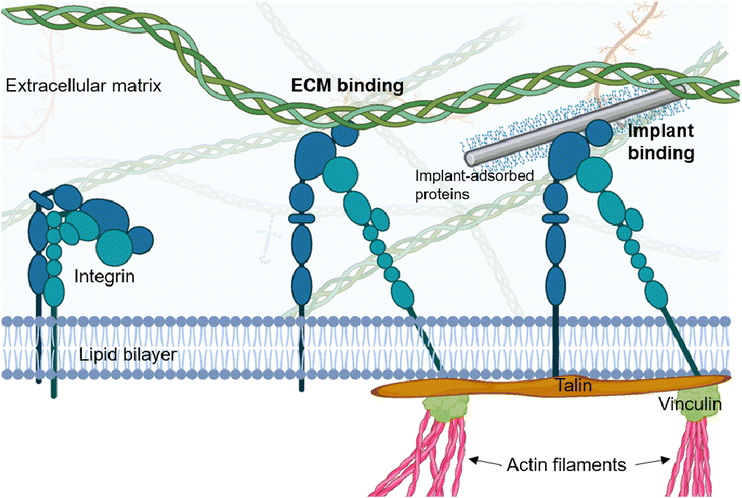
Cells engage with biomaterial surfaces through integrins, transmembrane receptors capable of recognizing specific motifs within the extracellular matrix (ECM),28 including the RGD (arginine–glycine–aspartic acid) sequence found in proteins such as fibronectin and vitronectin.29 Upon ligand binding, as shown in Fig. 1, integrins undergo conformational changes that promote proteins clustering, initiating the formation of focal adhesions (FAs). These adhesion complexes serve as sites for signal transduction and mechanical linkage to the actin cytoskeleton, enabling cells to sense and respond to the mechanical properties of their surroundings.30–32 The assembly of FAs involves the recruitment of proteins such as paxillin, talin and focal adhesion kinase (FAK). Vinculin, another critical FA protein, is recruited to sites of adhesion, linking the integrin–talin complex to the actin cytoskeleton. This connection enables the transmission of mechanical signals from the ECM to the cell interior, a process known as mechanotransduction.30–32When compared to the native ECM, cells interact differently with the rigid surfaces of metallic biomaterials, leading to distinct mechanotransductive processes that influence cell behavior and functions. In the natural ECM, which exhibits a range of stiffness from soft (0.1–10 kPa for brain and adipose tissues) to stiff (>1 MPa for bone and cartilage),33 cells engage with a three-dimensional, fibrillar network composed of proteins like collagen, fibronectin and laminin.28 Conversely, metallic surfaces present a two-dimensional, non-fibrillar and much stiffer environment (e.g., Young's modulus of ∼110 GPa for titanium implants).13 In addition, cellular sensing during the first stage of cell–material interactions is mediated by a surface-bound adlayer of soluble matrix proteins whose structural conformation, distribution and availability of cell-binding domains are determined by the physicochemical properties of the underlying nanostructured substrate (section 2.2).16 When cells adhere to biomaterial surfaces, they form larger and more stable FAs compared to those on softer, more compliant substrates.34 This phenomenon is attributed to increased traction forces generated by the cell, which are balanced by a high cytoskeletal tension that is necessary for spreading. In turn, such strain results in higher forces on the FAs, which are subsequently redistributed across a larger area via enhanced recruitment and activation of FA-associated proteins to reduce the load for each ECM–cytoskeletal linkage, ultimately preventing the adhesive cluster from detaching.35The elevated tension across these adhesions promotes the assembly of robust stress fibers and activates downstream signaling pathways, such as those involving FAK and Rho GTPases,36 which regulate cytoskeletal dynamics. The mechanical signals propagated through FA-associated pathways also critically influence nuclear mechanotransduction via transcriptional regulators. One of the principal downstream effectors of cytoskeletal tension is the Yes-associated protein (YAP), a mechanosensitive transcriptional co-activator that translocates to the nucleus in response to elevated intracellular tension.16,37,38 The activation of FAK and Rho GTPases enhances actomyosin contractility, and the resulting increase in cytoskeletal tension promotes the nuclear localization of YAP,36,39 where it regulates gene expression programs involved in cell proliferation, survival and differentiation. This mechanosensitive response enables cells to convert the mechanical properties of their substrate into transcriptional outputs and, in turn, into long-term changes in cell behavior.
2.2. Nanoscale topography as a regulator of cell functions
Common nanotopographical features employed in the design of metallic biomaterials can be broadly classified into two main categories: nanoscale depressions/cavities (e.g., nanopits) and raised features (e.g., nanopillars).16,40 Additionally, more complex architectures such as nanowires, nanogratings and hierarchical multiscale structures have been developed to direct cell- and tissue-level functions.16,40Nanotopographies have been shown to modulate the spatial organization of integrins and influence the clustering dynamics necessary for FA assembly.13,15 These physical features can either promote or inhibit focal adhesion maturation by altering local adhesion strength and force transmission at the cell–material interface. The resulting changes in FA size, density and spatial arrangement in turn influence cytoskeletal tension, direct the alignment of actin stress fibers and modulate cell polarity, migration and differentiation via the activation of mechanosensitive signaling pathways such as those mediated by FAK and Rho GTPases.36,39,41 Notably, experimental evidence indicates that nanoscale topography modulates cellular mechanosensing to such an extent that it mimics, and even overrides, the effects of substrate stiffness. Specifically, it was shown that neurons and stem cells cultured on rigid glass substrates engineered with nanopillars behaved as if they were cultured on soft hydrogels, in response to the fact that both cues regulate integrin receptor availability and focal adhesion dynamics.13 The reduction in available integrins caused by the nanotopography-induced membrane curvature disrupted focal adhesion assembly, decreased cytoskeletal tension and promoted the disassembly of stress fibers,13 thereby providing a mechanistic framework for the rational design of nanoscale surface features on rigid materials that can effectively recapitulate the mechanobiological cues of native tissues.
From a mechanistic perspective, it was postulated that arrays of nanoscale protrusions with spacings below 70 nm and diameters exceeding 70 nm generally do not affect integrin engagement and the subsequent focal-adhesion reinforcement.42 As the distance between protrusions increases within the submicron range, particularly when the protrusion height remains below 70 nm, cells regain access to the underlying substrate, thereby enhancing interactions with the nanostructured surface. In contrast, taller protrusions restrict integrin binding to the planar surface and confine focal-adhesion formation primarily to the feature apexes.42 Notably, when feature diameters fall below 70 nm and spacing exceeds 70 nm, integrin clustering becomes markedly disrupted, leading to weakened cell adhesion and altered cytoskeletal organization. Similarly, the formation and reinforcement of focal adhesions are profoundly influenced by the geometry of nanoscale pits. When pit diameters are below 70 nm, integrin clustering and focal-adhesion assembly remain largely unaffected, irrespective of pit depth.42 As the lateral dimensions increase beyond this threshold, particularly when the pit depth exceeds 100 nm, integrin organization becomes perturbed, leading to diminished adhesion strength. Conversely, broader but shallower pits promote integrin clustering, thereby reinforcing focal-adhesion formation. Notably, pits in the 70–300 nm range arranged with minimal separation (<70 nm) disrupt integrin clustering and cellular attachment, highlighting the delicate interplay between pit size, depth and spatial distribution in orchestrating cell behaviour on nanostructured surfaces.42 It is important to note, however, that these threshold values described above are drawn from a single mechanistic framework. As discussed in section 5, other studies employing different feature types, fabrication methods and biological models report partially divergent dimensional ranges associated with optimal integrin engagement and focal adhesion assembly.
In this context, while much attention has been given to the shape, size and density of nanoscale features, an often overlooked yet critical parameter in the design of nanostructured surfaces is the spatial distribution of these features, in particular the geometry, symmetry and order of their planar arrangement.43–45 In fact, subtle variations in feature spacing are believed to determine whether integrins can form stable clusters capable of supporting FA assembly and, in turn, initiating the downstream mechanotransductive signaling.43–45 Seminal work by Dalby et al. demonstrated experimentally that disordered nanopit arrays enhance focal-adhesion maturation and promote osteogenic differentiation of mesenchymal stem cells,46,47 indicating that cells do not only respond to size/depth/spacing but also to the degree of spatial variation. Therefore, incorporating spatial statistics into surface design offers a new complementary strategy alongside topographical and mechanical cues for directing cell behavior on biomaterials.
Importantly, it should be noted that regardless of the geometrical characteristics of nanotopographies, cells do not interact directly with the nanostructures but rather with the layer of proteins adsorbed from the surrounding extracellular matrix (ECM) – or from the biochemical microenvironment of in vitro systems (e.g., culture medium) – onto the material surface.16 The resulting surface-bound protein adlayer influences focal adhesion assembly and guides cytoskeletal organization as they probe the substrate.48 Notably, not only the quantity but also the conformation of these adsorbed proteins critically influences the subsequent cellular colonization of surfaces. Specifically, surface topography at the nanoscale can modulate protein folding, orientation and packing density, thereby altering the exposure of functional domains essential for integrin recognition and downstream signaling.16,49,50 In particular, these nanotopography-induced conformational variations determine how effectively adhesion ligands are presented to cells, influencing adhesion strength, focal adhesion assembly and the activation of mechanotransductive pathways that ultimately guide cell fate decisions.49
Taken together, the interplay between nanoscale geometry, spatial disorder and protein-adsorption dynamics underscores some of the complex and multiscale mechanisms by which nanotopography modulates cellular function and signaling, highlighting the need to translate these mechanistic insights into predictive design principles for next-generation metallic implants. Capturing this complexity through more realistic in vitro systems and computational modeling will be essential to bridge mechanistic understanding with predictive in vivo performance in the next generation of metallic implants.
3. Advances in precision nanofabrication of metallic surfaces
The capacity to engineer metallic surfaces with nanoscale precision has transformed how biomaterials are envisioned, and with the extensive body of in vitro and in vivo evidence now available (section 5), the field is increasingly closer to integrate these insights into next-generation metallic implants with precisely tailored biological performance. As part of this broader evolution in surface engineering, coating-based strategies (e.g., plasma spray, sol–gel deposition, PVD/CVD processes, ion implantation) produce micro- and nano-structured thin films used to tailor functional properties such as corrosion resistance, osseointegration and antimicrobial resistance, among others.51–53 However, the resulting topographical features belong to the deposited layer rather than the metallic substrate itself, and their structure and functional role differ markedly from features engineered within the metal. Because this perspective article is mainly centered on nanoscale architectures formed intrinsically within metallic substrates, we do not provide an exhaustive overview of coating technologies, except where such approaches are directly relevant to clinically deployed implant systems and/or offer realistic pathways toward next-generation implant designs (section 6).Several fabrication strategies possess the prerequisite for designing nanoscale surface topographies of metals. Top-down approaches such as electron-beam (e-beam) lithography, nanoimprint lithography (NIL), focused-ion-beam (FIB) milling, and laser ablation provide deterministic control over feature dimensions and layout. A closer examination of their respective benefits and constraints highlights the trade-offs inherent in each technique. Specifically, e-beam lithography, although costly and low-throughput, allows a spatial resolution down to a few nanometers, and has been instrumental in fabricating ordered nanopatterns on titanium substrates.54,55 In parallel, NIL offers a scalable, high-throughput route to replicate sub-nanometer patterns over large areas and transfer them into metallic substrates.56 However, it requires a master mold fabricated using another high-resolution patterning method, which adds an additional processing step. Moreover, because NIL is fundamentally a planar replication technique, it cannot readily produce complex 3D or multi-level nanostructures.57 In addition, while FIB milling enables direct modification to create custom-designed patterns,58–60 its practical deployment is constrained by the very high capital and maintenance costs of the instrumentation, as well as the need for exceptionally stable ion sources and highly trained operators.57 Finally, ultrafast laser ablation offers rapid, mask-free patterning over comparatively large areas and is compatible with a wide range of metallic substrates, including medically relevant materials such as titanium, steel and NiTi alloy.61–63 However, its achievable resolution (typically ∼100 nm) is typically lower than that of beam-based lithographies, and thermal effects may compromise feature fidelity depending on pulse energy and material properties.64
In parallel, bottom-up methods have leveraged self-organization processes to generate nanoscale architectures directly on metallic substrates. Among these, electrochemical anodization has become a widely adopted strategy for producing arrays of nanotubes and additional nanostructures (e.g., nanowires, honey-comb architectures) on passivating metals, mostly titanium and its alloys.65,66 Its simplicity, tunability and scalability make it particularly attractive for biomedical applications (section 6), as pore diameter and wall thickness can be adjusted by voltage, electrolyte composition and time to control specific cellular functions.65–69 Similarly, chemical oxidation and hydrothermal treatments can induce TiO2 nanowires or Mg(OH)2 nanosheets formation,70–73 although these methods produce less uniform or less periodic features compared with nanotubular surfaces. Electrodeposition and templated growth provide versatile bottom-up routes to form nanostructured metallic films with tailored morphology.74–77 Their crystallinity, aspect ratio and uniformity, however, remain partly constrained by template quality and mass-transport conditions. For instance, optimized pulsed electrodeposition can achieve highly crystalline, nearly fully filled nanowire arrays when pore accessibility and nucleation are controlled.75 Similarly, titania nanotube templates can direct the spatially controlled electrodeposition of gold nanoparticles, enabling tunable nanoscale features with biological relevance.77 Despite their scalability and low cost, such bottom-up methods still offer less deterministic long-range spatial ordering than lithographic approaches and often exhibit intrinsic stochasticity in feature size and arrangement, particularly when using low-purity templates or single-step anodization processes designed for high-throughput production.76 Conversely, their compatibility with complex geometries, curved or micro-porous surfaces, and large-area processing makes electrodeposition uniquely suited for translating nanoscale cues onto fully three-dimensional implant platforms and biosensing architectures.74
Notably, the strategies described above have been developed and characterized predominantly on titanium and its alloys, reflecting a broader imbalance in the biomaterials literature that itself stems from the more predominant clinical employment of Ti-based implants in orthopedic, spinal and dental applications. However, many of these approaches are also applicable to other clinically important metallic biomaterials. For example, CoCr alloys are used in total knee and hip replacements and dental prosthetics, and present a fundamentally different surface chemistry governed by a Cr2O3 passive film. Nonetheless, nanostructuring of CoCr alloys has been explored through laser-directed energy deposition combined with biocorrosion to generate surface structures of controlled aspect ratio.78 Similarly, 316L stainless steel (SS) has been nanostructured via femtosecond laser texturing, which produces hierarchical micro/nano patterns.79,80 In this context, nanosecond laser texturing was applied comparatively across 316L SS, CoCr and Ti alloys under the same conditions, revealing that the resulting surface topography and roughness are highly material-dependent,81 thereby underscoring that processing parameters optimized for Ti cannot be uncritically transferred to other alloys. In parallel, anodization with an atypical electrolyte successfully created a mesoporous surface on 304 and 316L stainless steels that selectively promotes mammalian cell activity and limits bacterial adhesion.82 Taken together, while Ti and its alloys remain the most thoroughly characterized systems for precision nanotopographical engineering, the literature on CoCr and stainless steel demonstrates both the feasibility and the material-specificity of nanostructuring approaches for non-Ti metallic biomaterials, and calls for a more systematic extension of mechanistic studies to these clinically prevalent substrates.
A critical challenge that cuts across all nanostrucuring strategies and metals is the metrological framework used to characterize the nanotopographies. In the majority of published studies, surface features are quantified via high-resolution imaging techniques (e.g. Scanning Electron Microscopy – SEM, Atomic Force Microscopy – AFM) over areas of a few square micrometers. While this scale captures local feature geometry, it cannot reflect the statistical distribution of feature size, spacing and height across an implant surface. This scale mismatch is a fundamental reproducibility problem: two surfaces described by nominally identical feature dimensions may exhibit markedly different large-area uniformity and, consequently, divergent biological performance. Addressing this requires broader adoption of standardized surface texture parameters (e.g. as defined in ISO 25178 – Geometrical Product Specifications (GPS) – surface texture: areal) measured over areas that are representative of the implant surface at a clinically relevant scale. Consistent reporting of these parameters across studies would substantially improve comparability, providing quantitative surface descriptors needed to evaluate process reproducibility and batch-to-batch consistency.
4. Preclinical models for the evaluation of cell and tissue response
Understanding how nanoscale features influence biological behavior requires an experimental framework that spans multiple levels of complexity. Over the past decades, researchers have relied on a wide array of in vitro technologies (Fig. 2) and in vivo models, each offering different types of insight while also imposing distinct limitations. These preclinical approaches form a continuum, from simplified two-dimensional (2D) assays to fully integrated animal systems, that together have shaped our current understanding of how biomaterials interact with cells and tissues.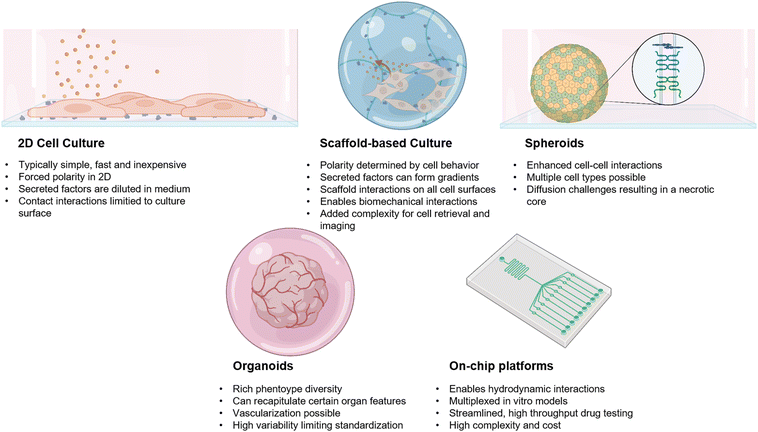 | ||
| Fig. 2 Comparison between different in vitro modeling systems, outlining their important advantages and drawbacks. Created with https://Biorender.com. | ||
Much of the foundational work in the field has relied on conventional 2D cell culture assays. Their simplicity, accessibility and compatibility with high-resolution imaging have made them indispensable for probing the direct relationship between nanoscale design parameters and cellular behaviors such as adhesion, spreading, cytoskeletal organization and lineage specification, among others. However, it is now established that these insights arise in a 2D environment that differs fundamentally from native tissues. Specifically, cells experience an artificial polarity, stiff planar substrates and minimal extracellular matrix constraints.22,83 As a result, the in vitro response of biomaterials may not fully capture their behavior in vivo.
To address these limitations, the field has steadily moved toward more sophisticated in vitro systems that reintroduce essential aspects of 3D tissue organization. For example, scaffold-based cultures,84–87spheroids,85,88,89 organoids90 and bone-on-a-chip devices91 allow to integrate matrix mechanics, soluble gradients and multicellular interactions. These models offer a remarkable degree of self-organization and functional complexity closer to those of the native bone tissues, bringing investigators closer to assessing how metallic nanostructures interact with tissue-level architectures. However, these systems introduce practical challenges: their 3D architecture, dynamic culture conditions and inherent heterogeneity complicate the placement, assessment and imaging of cells at the interface with metallic substrates.
Between in vitro and in vivo systems lies a useful but narrower class of ex vivo models, such as bone explants.87 These offer the advantage of native extracellular matrix composition and mechanical structure, providing a testbed for how biomaterials integrate with intact tissues. They can capture early events in bone remodeling, including osteocyte viability, osteogenic response and mechanosensitive signaling, within a native extracellular matrix environment that is biologically richer than conventional in vitro cultures.92 However, ex vivo tissues lack systemic vascularization, immune recruitment and healing processes, limiting their ability to capture long-term or multifactorial outcomes.
Ultimately, it is in vivo models that provide the most comprehensive picture of how nanostructured metallic surfaces behave under realistic physiological conditions. Implantation in rodents, rabbits and large animals exposes materials to the full complexity of host biology.93,94 These studies remain essential for assessing crucial aspects such as osseointegration, immune modulation and mechanical stability (section 5.2). Yet, the in vivo environment also introduces variability that can obscure the direct contributions of nanoscale features, and inter-species differences can hinder translation to humans. For these reasons, mechanistic insight obtained from in vitro systems must be interpreted alongside in vivo outcomes to build a coherent understanding of how design features translate into functional performance. Taken together, this spectrum of models illustrates both the power and the challenges of evaluating nanostructured metallic implants prior to clinical trials. No single platform fully captures the interplay of tissue architecture, mechanics, biochemistry and cellular complexity. Progress will require experimental strategies that intentionally bridge these domains, linking controlled mechanistic experiments in 2D and 3D systems with validation in ex vivo tissues and in vivo environments (section 6). Only by integrating these multiscale perspectives can we reliably predict how precisely engineered nanotopographies will perform in clinical settings.
5. From model to function: in vitro and in vivo evidence
5.1. In vitro studies
Over the past decade, in vitro studies have provided compelling evidence that nanoscale surface topographies on metallic biomaterials function as active, cell-instructive cues rather than passive structural modifications. Using titanium, titanium alloys and additional clinically relevant metals as model substrates, these investigations demonstrate that nanoscale geometry, independently of surface chemistry, can regulate cell adhesion, proliferation, differentiation, and intercellular signaling. Specifically, initial cell attachment and proliferation are strongly influenced by nanoscale surface features, with numerous studies reporting enhanced cell adhesion on nanostructured metallic substrates relative to smooth or microscale controls.95 Titanium dioxide nanotubes produced via anodization represent one of the most extensively studied topographies in this context. Across multiple reports, nanotubular Ti surfaces consistently promote adhesion and proliferation of osteoblast-like cells and mesenchymal stem cells (MSCs), although the magnitude of the response is highly dependent on nanotube diameter.96–98 In particular, studies employing human Saos-2, murine MC3T3-E1, and primary human osteoblasts have shown that nanotube diameters in the approximate range of 20–70 nm enhance focal adhesion formation, cytoskeletal organization, and early proliferation, whereas excessively large or small features attenuate these effects.96,98 Similar enhancement of adhesion and proliferation has been observed on nanowire-structured titanium alloys, including TNZT and Ti–Nb–Zr systems, indicating that nanoscale geometry remains a dominant regulator even when alloy composition varies.97,99 Beyond nanotubes, alternative nanotopographies such as nanodots, nanospikes and nanopits also support robust initial cell attachment. Specifically, carefully defined nanopit arrays promoted osteogenic activity by colocalizing integrins and BMP-2 receptors100 as demonstrated in Fig. 3.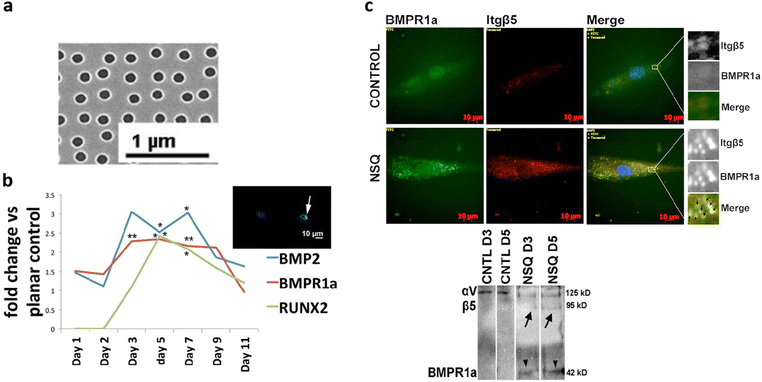 | ||
| Fig. 3 a) Partially disordered nanopit pattern (NSQ) developed by Dalby et al.46 and b) enhancement of the expression of RUNX, BMP2 and BMP receptor.100 c) Co-staining and immunoprecipitation studies of the BMP receptor and integrin beta 5 show the NSQ pattern causes colocalization of these proteins, while it is absent on unpatterned controls. Figure adapted from Yang et al.100 under CC-BY 4.0. | ||
In addition to regulating early adhesion, nanoscale metallic topographies have been repeatedly shown to promote osteogenic differentiation in vitro, even in the absence of osteogenic supplements. Titanium nanotopographies generated via anodization, chemical etching and oxidative treatments enhance alkaline phosphatase activity, extracellular matrix mineralization, and expression of key osteogenic markers, including RUNX2, OPN, OCN, and COL1A1.101–103
Diameter and geometry-dependent effects are particularly pronounced during differentiation. For example, TiO2 nanotubes of intermediate diameters (30–70 nm) consistently induce stronger osteogenic responses than either smooth titanium or larger-scale structures, as demonstrated in both Saos-2 and MC3T3-E1 models.96,102 Nanostructured Ti–Nb–Zr and TNZT alloys similarly promote osteogenic maturation, confirming that nanoscale topography-driven differentiation is conserved across multiple clinically relevant metallic systems.97,99 In parallel, mixed-oxide nanotubular systems incorporating Nb or Zr into TiO2 architectures enhance biocompatibility and osteoblast attachment in vitro when compared to bare Ti alloys, highlighting the synergistic effects of nanoscale geometry and alloying strategies.104
These diameter- and geometry-dependent effects, however, are not uniform across studies, and the specific dimensional ranges associated with optimal responses vary considerably depending on the feature type, fabrication method, and biological model employed. Table 1 presents a selection of comparative studies examining dimensional variations within specific nanofeature types on titanium, chosen not as a comprehensive survey but to illustrate (i) how reported optimal dimensions and biological outcomes vary across studies and (ii) how these values frequently diverge from, or cannot be directly reconciled with, the mechanistic threshold framework described in section 2.2. As biological outcomes are simultaneously governed by multiple nanofeature parameters and by the specific biological models employed, direct quantitative comparison across studies or nanostructure designs remains inherently limited, and threshold values should therefore be interpreted as a mechanistic reference point rather than universal design criteria.
| Source | Feature type | Biological outcome |
|---|---|---|
| Park et al.105 | TiO2 nanotubes (in vitro) | 15 nm diameter optimizes primary human osteoblast adhesion and osteogenic differentiation; wider tubes progressively impair response |
| Voltrova et al.96 | TiO2 nanotubes (in vitro) | 66 nm diameter improves vinculin, talin, osteocalcin and collagen I expression in human Saos-2 osteoblast-like cells. 24 nm diameter less effective, indicating 70 nm threshold42 is not a binary predictor for nanotubular features |
| Luo et al.98 | Nano-flat, -convex, -concave TiO2 nanotopographies (in vitro) | Convex geometry of ∼80 nm in diameter promotes larger FAs and longer actin stress fibers on nanoconvex surfaces in primary human osteoblasts. Feature shape modulates response beyond size alone |
| Ballo et al.106 | Semi-spherical protrusions on Ti (in vivo, rat tibia – section 5.2) | Higher BIC and peri-implant bone formation at 60 nm in vivo; reduced bone apposition at ≥120 nm in vivo |
| Wilmowsky et al.107 | TiO2 nanotubes (in vivo, minipig – section 5.2) | Diameter-dependent osseointegration; highest BIC and osteogenic gene expression at variable nanotube diameter |
Taken together, Table 1 reveals both areas of agreement and notable discrepancies across studies. The 70 nm threshold for protrusion-type features proposed in ref. 42 receives partial in vivo corroboration from Ballo et al. (60 nm optimal). However, Voltrova et al.96 demonstrate that among nanotubular features below 70 nm, the 66 nm diameter produces the strongest osteogenic response, while Park et al.105 identify 15 nm as the optimal nanotube length scale. Luo et al.98 further show that feature geometry (convexity vs. concavity) modulates the cell response independently of diameter. While the studies included in Table 1 represent only a narrow cross-section of the available literature, the discrepancies they reveal are sufficient to illustrate that no single dimensional threshold reliably predicts biological outcomes across conditions. This observation further reinforces the importance of multi-model validation strategies discussed in section 6.
Beyond osteogenic cells, nanoscale metallic topographies influence endothelial behavior, a critical consideration for implant vascularization. Anodized TiO2 nanotube surfaces improved endothelial cell viability, proliferation, and angiogenic behavior relative to untreated titanium, suggesting that nanoscale geometry may support coordinated bone–vascular integration at implant interfaces.108
More complex in vitro models reveal that nanotopography also modulates intercellular signaling. In osteoblast–osteoclast co-culture systems, nanostructured titanium surfaces promoted osteoblast activity while suppressing osteoclast differentiation, indicating that nanoscale geometry can influence bone remodeling dynamics rather than isolated cell phenotypes.109 Similarly, studies using gingiva-derived MSCs on commercially relevant nano- and microstructured implant surfaces showed enhanced protein adsorption and stem cell adhesion, underscoring the relevance of nanotopographical effects across multiple tissue-specific progenitor populations.110
From a mechanistic point of view, a large body of literature has consistently identified integrin-mediated mechanotransduction as a central pathway linking nanoscale geometry to cell functions. For example, nanotopographical titanium surfaces upregulate integrin subunits, particularly αV and β1, leading to enhanced focal adhesion kinase (FAK) activation and downstream signaling cascades associated with osteogenic commitment.100,102 These effects are accompanied by increased actin organization and cytoskeletal tension, reinforcing the role of physical force transmission in nanoscale sensing. More recent studies extend these mechanisms to include intracellular stress-response pathways. Titanium nanotopographies have been shown to activate β-catenin signaling through autophagy-mediated mechanisms, linking nanoscale geometry to intracellular metabolic regulation and differentiation pathways.103 Gradient nanostructured titanium surfaces further demonstrate that spatial variations in nanoscale geometry can locally regulate adhesion strength, cytoskeletal organization, and osteogenic marker expression, offering a powerful platform for dissecting cell–material interactions with high spatial resolution.111
In vitro evidence on non-Ti metallic substrates remains comparatively limited. On nanostructured CoCrMo alloys generated by laser-directed energy deposition and biocorrosion, MC3T3 preosteoblasts and human bone marrow mesenchymal stem cells showed enhanced adhesion, spreading and osteogenic differentiation relative to flat controls.78 On femtosecond and nanosecond laser-textured 316L stainless steel, hierarchical micro/nano surface patterns supported osteoblast adhesion, alignment and contact guidance,79 while co-culture studies demonstrated a reduction in myofibroblast differentiation without impairing endothelial cell proliferation; a response profile of particular relevance for stent applications.80
Taken together, in vitro studies demonstrate that precisely engineered nanoscale topographies on metallic biomaterials can deterministically regulate cell adhesion, proliferation, differentiation, angiogenic behavior, and multicellular signaling. These effects are reproducible across fabrication methods, metallic compositions, and cell types, reinforcing nanoscale geometry as a dominant and tunable design parameter.
5.2. In vivo studies
While in vitro studies establish nanoscale topography as a powerful regulator of cell behavior, in vivo models are essential to validate whether these effects persist within the complex biological environment of bone healing. Over the past decade, a growing number of animal studies have demonstrated that nanoscale-engineered metallic implants can enhance early bone formation, improve mechanical fixation, and modulate immune and remodeling responses. Collectively, these studies provide strong evidence that nanoscale surface precision translates into measurable functional benefits at the bone–implant interface.One of the most consistent findings across in vivo studies is the ability of nanoscale surface topographies to accelerate early bone formation and increase bone–implant contact (BIC). Using well-defined nanotopographical model implants, de novo bone formation has been shown to be highly sensitive to feature size. Titanium implants decorated with hemispherical nanoprotrusions demonstrated significantly higher BIC and peri-implant bone formation in rat tibiae when feature sizes were approximately 60 nm, whereas larger nanoscale features (≥120 nm) resulted in reduced bone apposition, highlighting a narrow optimal nanoscale regime for osteogenic activity.106
Similarly, titanium implants with anodized TiO2 nanotubes of variable diameter consistently enhanced cellular functions of osteoblasts and osteoclasts in vivo, including differentiation and protein expression.107 In rodent long-bone implantation studies, nanotubular surfaces promoted greater bone ongrowth and more mature bone tissue formation at early healing time points compared to smooth titanium controls.112,113 Hydrogenated, superhydrophilic TiO2 nanotubes further amplified these effects, leading to increased new bone regeneration and more intimate bone–implant contact during the first four weeks post-implantation.114
Large-animal models reinforce these findings and underscore their translational relevance. In minipig cranial defect models, titanium implants with nanotube diameters ranging from 30 to 100 nm all improved osseointegration relative to flat controls, with intermediate diameters (∼70 nm) producing the highest BIC and strongest osteogenic gene expression profiles in peri-implant tissue.115 Comparable enhancements in bone volume fraction and bone density were reported in Beagle dog models using nanotextured and Sr-loaded titanium implants placed immediately after tooth extraction, demonstrating that nanotopography can support rapid bone formation even in clinically challenging scenarios.116
Beyond histological outcomes, several in vivo studies demonstrate that nanoscale surface engineering improves the mechanical performance of metallic implants. Removal torque and pull-out tests consistently show stronger implant fixation for nanostructured surfaces compared to smooth or purely microstructured controls. Hierarchical micro/nano-structured TiO2 surfaces generated via hydrothermal or anodic treatments significantly increased removal torque values in rabbit and rodent models, indicating superior mechanical anchorage at the bone–implant interface.117
Nanoporous and nanotubular titanium implants also improve biomechanical stability by promoting faster healing and more homogeneous bone ingrowth.113,118,119 In rodent implantation studies, nanoporous titanium surfaces exhibited higher push-out strength and improved load transfer characteristics, correlating with increased bone maturity and reduced fibrous tissue formation around the implant.113,117 These findings suggest that nanoscale topographies not only enhance bone quantity but also improve bone quality and functional integration.
Dental implant models further support these conclusions. Nanospike-textured titanium dental implants demonstrated significantly higher BIC and improved mechanical engagement compared to conventional surfaces, while maintaining structural integrity under functional loading conditions.120 Importantly, these improvements were observed without compromising surrounding tissue health, reinforcing the safety and efficacy of nanoscale geometries when precisely controlled.
More recent in vivo investigations have moved beyond static measures of osseointegration to explore how nanoscale topographies influence immune responses and bone remodeling. Nanoporous titanium implants suppress osteoclastogenesis while promoting osteoblast activity, leading to a net increase in bone formation during early healing phases.113 Histological and molecular analyses revealed altered macrophage polarization profiles around nanotextured implants, suggesting that nanoscale geometry can shift the local immune environment toward a pro-healing, osteogenic phenotype.
This immune-modulatory role of nanotopography is further supported by studies examining local and systemic responses to titanium nanotube implants. Intramedullary implantation studies demonstrated that nanotubular surfaces were well tolerated, elicited no adverse systemic inflammatory responses, and promoted stable bone ongrowth over extended implantation periods.112 These findings indicate that nanoscale features can actively regulate the foreign body response rather than merely avoiding adverse reactions.
Coordinated regulation of bone formation and resorption was also observed in studies evaluating molecular signaling at the bone–implant interface. Controlled nanoscale topographies enhanced early osteogenic signaling pathways while reducing markers associated with excessive bone resorption, contributing to more balanced and stable bone remodeling in vivo.118
Animal studies also demonstrate that nanoscale surface engineering can deliver multifunctional benefits beyond enhanced osseointegration. For example, nanotextured titanium designed to be bactericidal maintained excellent bone integration while simultaneously reducing bacterial colonization, illustrating that antimicrobial and osteogenic functions are not mutually exclusive when nanoscale geometry is carefully optimized.120–122 In conclusion, in vivo studies provide strong and convergent evidence that precisely engineered nanoscale topographies on metallic implants enhance early bone formation, improve mechanical anchorage, and actively modulate immune and remodeling responses.
6. Outlook and future directions
The rapid advances in precision nanoscale engineering of metallic biomaterials have brought the field to an inflection point: mechanistic understanding is now sufficiently mature to inspire rational design principles, yet significant scientific, technological and translational barriers may still prevent these discoveries from transforming clinical practice. A critical evaluation of these challenges, together with emerging opportunities, contributes to highlight the path toward next-generation bone-interfacing implants capable of providing reproducible and patient-relevant biological outcomes.A central limitation of the current landscape is that most breakthroughs in nanotopographical design have relied on techniques and substrates specifically selected to allow the precise control over surface architecture. To achieve this level of accuracy, researchers predominantly employed planar metallic substrates, as their flat geometry facilitates well-defined nanostructuring, high-resolution imaging and standardized cell culture assays. However, translating these nanoscale modifications to implants with complex 3D geometries introduces a host of additional challenges. For example, curved or threaded geometries disrupt the planar alignment required for lithography or templating, making consistent feature replication during fabrication technically complex. Such geometric constraints increase the likelihood of incomplete or poorly controlled pattern transfer, which may, in turn, propagate variability into downstream biological responses, thereby reinforcing the need for standardized and more stringent surface-processing workflows.123–128 These fabrication-related challenges are further exacerbated by the pronounced mismatch in scale between the relatively small test coupons typically used in laboratory studies and the substantially larger, multi-surface dental, spinal and hip implants. In parallel, and often overlooked, the oxide layer on passive metals, whether native or induced by oxidative processes such as chemical etching, adds an additional level of complexity, as its thickness, composition and crystalline structure are highly sensitive to both processing parameters and environmental exposure. Variations in oxide chemistry can subsequently alter key surface properties, including wettability and functional-group availability, thereby modulating cell- and tissue-level signaling pathways, while the oxide itself may crack and/or delaminate locally under mechanical stresses,129 thereby creating a heterogeneous bone–implant interface. These obstacles ultimately underscore the need for manufacturing technologies that offer nanoscale precision on medically relevant metals and large, non-planar, clinically realistic geometries. To this end, (electro)chemical treatments (e.g. anodization, acid etching) and laser texturing exhibit translational potential by enabling uniform nanoscale surface modification over implant-relevant dimensions and geometries, as demonstrated on titanium dental and orthopedic implants evaluated in vivo (Fig. 4), where these approaches have been shown to enhance osseointegration and early bone–implant contact.107,119,130–133
 | ||
| Fig. 4 Nanotopographical features with translational potential. a) Electron micrographs of a machined implant surface following etching (top) and anodization (bottom). b) Electron micrographs of the resulting nanotube arrays following anodization. c) Low to high magnification electron micrographs (top to bottom) of machined (left tiles) and laser modified (right tiles) titanium implants that result in improved mechanical anchorage and osseointegration. Figures adapted from Shah et al.132 under CC-BY 4.0 and Lee et al.,130 reprinted by permission of Informa UK Limited, trading Taylor & Francis Group https://www.tandfonline.com. This is an Open Access article distributed under the terms of the Creative Commons Attribution – Non Commercial (unported, v3.0) license (http://creativecommons.org/licenses/by-nc/3.0/) which permits non-commercial re-use, distribution, and reproduction in any medium, provided the original work is properly cited. | ||
Indeed, clinically successful commercial solutions already rely on some of these approaches, including anodized and chemically treated titanium dental implants developed by manufacturers such as Nobel Biocare (TiUnite™ and TiUltra™) and Straumann (SLActive™). These systems exemplify the scalability and regulatory viability of chemical treatments on complex implant geometries, demonstrating that robust osseointegration can be achieved even in the absence of precisely defined nanotopographical features. However, this clinical success is likely associated with multiscale surface roughness and enhanced hydrophilicity, rather than with the deliberate encoding of mechanistically instructive nanoscale cues. Consequently, translation in this space has largely converged on surface designs that are clinically effective, but not mechanistically optimized, with limited incentive to move beyond empirically beneficial yet weakly defined nanotopographies. It follows that the effectiveness of these implants should not be interpreted as validation of stochastic nanotopographies as optimal design solutions, but rather as evidence that current translational pathways have stabilized around surfaces that meet clinical and regulatory benchmarks without fully exploiting the biological potential of precision nanoscale engineering. Achieving truly instructive nanotopographies will hinge on stringent control over process parameters and long-range feature uniformity across complex implant geometries. Without such control, even scalable techniques risk introducing variability, ultimately eroding the benefits established in preclinical model systems and further complicating cross-study comparisons as well as regulatory approval pathways. Compounding this manufacturing challenge is a largely overlooked metrological one: the vast majority of studies characterize nanotopographies using SEM/AFM measurements taken over scan areas of only a few square micrometers, which captures local feature geometry but cannot represent the statistical uniformity of the surface across the complex three-dimensional geometry of a clinical implant. For example, a nanopillar array that appears well-ordered over a few μm2 scan field may exhibit significant spatial heterogeneity at the scale of a dental screw thread or an acetabular cup, yet this variability goes undetected and unreported. Moving toward standardized areal characterization measured over areas representative of the functional implant surface, is a prerequisite for the kind of process validation that clinical translation and regulatory approval will ultimately demand.
Beyond these fabrication constraints, even the most promising nanopatterning techniques must contend with additional challenges that emerge upon implantation, including surface contamination and potential alteration of nanoscale features during insertion. This complexity is further compounded by the presence of whole blood, which delivers a highly concentrated mixture of proteins, platelets and coagulation factors that rapidly mask engineered nanotopographies. Because of this multifactorial environment, many idealized findings from in vitro assays may not fully predict interfacial events in vivo, reinforcing the importance of incorporating hemocompatibility and blood–material interactions into implant evaluation frameworks. In this context, commonly used in vitro protein-adsorption assays, while indispensable for probing early mechanistic trends, capture only a limited and highly simplified subset of the interfacial processes that ultimately govern biological outcomes in vivo. Specifically, many in vitro investigations of nanotopography-mediated protein adsorption still rely on simplified, single-protein systems (e.g., albumin, fibronectin, vitronectin), which may not capture the competitive, dynamic protein exchange and multilayer corona formation occurring in vivo. Indeed, studies on nanotextured metals have largely focused on individual proteins or serum-free conditions, leaving the behavior of complex, multi-component protein mixtures comparatively unexplored. This stands in contrast to physiological fluids, where adsorption reflects competitive, time-dependent exchange rather than simple binding: most abundant, rapidly diffusing proteins initially adsorb to the surface but, unable to spread and strengthen their surface contacts, are displaced by lower-abundance species that arrive later yet undergo conformational rearrangements that increase their binding affinity (Vroman effect).134 Consequently, the protein layers forming in vitro under static, compositionally simplified conditions are poor surrogates for the complex and evolving interfaces encountered in vivo, particularly within the circulatory and/or interstitial microenvironments surrounding implants. Taken together, these limitations underscore the need for surface-engineering strategies that remain functional and biologically instructive under the complex and dynamic conditions encountered during implantation.
To address these challenges for clinical translation, functional coatings can be envisioned as effective strategies that help reconcile the precision of in vitro discoveries with the complexity of in vivo deployment. Indeed, this strategy has already been adopted clinically through bioactive coatings that enhances bone–implant interactions, such as nano-hydroxyapatite layers deposited via wet-chemical processes, as exemplified by commercial technologies developed by Promimic (i.e., HAnano Surface™). When appropriately designed, coatings can in fact provide a functional biochemical interface during the critical early phases of implantation. Importantly, a key requirement for such approaches is conformality over complex three-dimensional implant geometries, including threads, porous regions and internal surfaces. In this respect, additional techniques such as plasma-based treatments and sol–gel processes,51,67 are particularly attractive, as they can uniformly modify large, non-planar metallic surfaces while offering control over the physicochemical makeup of surfaces. Notably, coatings may also act as protective or sacrificial layers during implantation, mitigating mechanical abrasion, contamination and the immediate masking of underlying engineered nanotopographies by blood-derived components. By transiently shielding the underlying surface from direct blood contact, such layers can help preserve the intended biological readout of the nanotopography during the early and subsequent stages of healing. Viewed in this way, coatings provide a practical strategy for decoupling nanoscale design intent from the inevitable perturbations associated with surgical handling and the complex in vivo environment.
Alongside advances in structural design, the functional integration of metallic implants is expected to shift toward dynamic, responsive and patient-tailored systems. Future devices may be envisaged to incorporate precision and personalization, with surfaces tuned to patient-specific factors such as bone quality, anatomical site, age or pathological/genetic background,135 thereby acknowledging the heterogeneity of biological environments into which implants are placed. In fact, even within a single patient, the local biochemical milieu is not static: changes in ionic composition, pH, oxygen concentration, metabolic states and soluble mediators across the different phases of healing can continuously modulate how cells perceive and interpret surface cues over time. Recognizing that structural optimization alone cannot accommodate this biochemical and temporal variability, next-generation surfaces may be required to couple topographical design with mechanisms capable of actively responding to, or even shaping, the evolving microenvironment. To this end, functional coatings could also provide smart, self-healing, stimuli-responsive abilities in response to local signals such as pH, enzymatic activity or oxidative stress.136–139 In parallel, surface architectures will likely be engineered to remain functional across diverse and dynamically changing biochemical microenvironments, such as variations in inflammatory cytokines, hypoxia and pH, factors known to influence the cascade of osteogenic or inflammatory events. Finally, implants may also integrate embedded sensing elements that enable real-time monitoring of healing progression, inflammation or mechanical loading, providing clinicians with continuous feedback on implant performance.140–142
In this context, antibacterial performance, whether achieved through inherently nanotopographies or functional coatings, represents an equally critical design goal, given the substantial clinical burden of peri-implant infections.143 Multifunctional strategies that simultaneously promote osseointegration and resist microbial colonization are therefore expected to define the next generation of implant surfaces. Collectively, these developments position future metallic implants not merely as structural supports but as intelligent, adaptive interfaces capable of responding to and communicating with their biological surroundings.
Complementing these strategies, future efforts should leverage high-throughput platforms (e.g., nanotopographical gradients and automated imaging pipelines) to screen several surface designs in parallel, thereby accelerating the discovery of clinically relevant architectures. To this end, equally important will be the evolution of biological testing systems that reflect the complexity of the human tissue microenvironment. While animal models remain indispensable, their translational accuracy is limited by interspecies differences and difficulties in isolating nanoscale effects from systemic processes. Emerging human-relevant in vitro systems, including 3D spheroids, organoids, perfused bone-on-chip devices and ex vivo bone explants, offer a more physiologically relevant platform for interrogating protein adsorption dynamics, osteoimmune interactions, vascular penetration and early bone remodeling. Incorporating nanotopographical gradients with biomimetic systems will be essential to better simulate in vitro how the host microenvironment jointly regulates the interpretation of nanotopographical and biochemical cues. These models should thus become standard tools for evaluating next-generation nanotopographies before advancing to in vivo studies.
Regulatory considerations impose additional constraints on the translation of advanced surface-engineering strategies. Regulatory agencies such as the U.S. Food and Drug Administration (FDA) and standards bodies including the International Organization for Standardization (ISO) place strong emphasis on safety and reproducibility. While surface treatments that preserve bulk material properties may be generally viewed favorably, the introduction of new or highly specific nanoscale features could raise additional questions related to long-term stability, corrosion behavior, wear debris generation and biological safety. A further translational bottleneck lies in the generation of regulator-relevant preclinical evidence: although in vitro studies are indispensable for establishing mechanistic insight, regulatory approval ultimately depends on in vivo and clinical data demonstrating robust and reproducible performance. Notably, the enactment of the FDA Modernization Act 3.0, which formally recognizes human-relevant in vitro methodologies for therapeutic development, creates a timely opportunity to strengthen the regulatory value of advanced nonclinical platforms. The integration of physiologically relevant in vitro systems into surface-engineering research may therefore not only accelerate mechanistic validation but also align emerging technologies with evolving regulatory frameworks.
Importantly, the clinical success rates of existing implant surfaces have established a demanding benchmark for adoption, thereby reducing the incentive to introduce more complex or less familiar surface technologies unless they offer clear, reproducible and clinically meaningful advantages. Moving beyond this plateau will require a concerted effort to align precision nanoscale surface design with scalable manufacturing, standardized nanoscale metrology and performance metrics that are simultaneously meaningful to regulators, clinicians and patients, thereby enabling the next generation of truly instructive, clinically transformative metallic implants.
Conflicts of interest
Authors declare no conflict of interest.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.References
- D. Losic, Advancing of titanium medical implants by surface engineering: recent progress and challenges, Expert Opin. Drug Delivery, 2021, 18, 1355–1378 CrossRef CAS PubMed.
- L. Fan, S. Chen, M. Yang, Y. Liu and J. Liu, Metallic Materials for Bone Repair, Adv. Healthcare Mater., 2024, 13, 2302132 CrossRef CAS PubMed.
- D. S. Ashish, P. A. P. Suya, J. Naveen, T. Khan and S. H. Khahro, Advancement in biomedical implant materials—a mini review, Front. Bioeng. Biotechnol., 2024, 12, 1400918 Search PubMed.
- N. A. Hodges, E. M. Sussman and J. P. Stegemann, Aseptic and septic prosthetic joint loosening: Impact of biomaterial wear on immune cell function, inflammation, and infection, Biomaterials, 2021, 278, 121127 CrossRef CAS PubMed.
- M. M. Soliman, M. T. Islam, M. E. H. Chowdhury, A. Alqahtani, F. Musharavati, T. Alam, A. S. Alshammari, N. Misran, M. S. Soliman, S. Mahmud and A. Khandakar, Advancement in total hip implant: a comprehensive review of mechanics and performance parameters across diverse novelties, J. Mater. Chem. B, 2023, 11, 10507–10537 RSC.
- L. Coelho, M.-C. Manzanares-Céspedes, J. Mendes, V. Tallón-Walton, W. Astudillo-Rozas, C. Aroso and J. M. Mendes, Coating Materials to Prevent Screw Loosening in Single Dental Implant Crowns: A Systematic Review, Materials, 2024, 17, 5053 CrossRef CAS PubMed.
- F. Melo-Fonseca, G. Miranda, H. S. Domingues, I. M. Pinto, M. Gasik and F. S. Silva, Reengineering Bone-Implant Interfaces for Improved Mechanotransduction and Clinical Outcomes, Stem Cell Rev. Rep., 2020, 16, 1121–1138 CrossRef CAS PubMed.
- S. Kligman, Z. Ren, C.-H. Chung, M. A. Perillo, Y.-C. Chang, H. Koo, Z. Zheng and C. Li, The Impact of Dental Implant Surface Modifications on Osseointegration and Biofilm Formation, J. Clin. Med., 2021, 10, 1641 CrossRef CAS PubMed.
- T. Stich, F. Alagboso, T. Křenek, T. Kovářík, V. Alt and D. Docheva, Implant-bone-interface: Reviewing the impact of titanium surface modifications on osteogenic processes in vitro and in vivo, Bioeng. Transl. Med., 2022, 7, e10239 CrossRef CAS PubMed.
- M. B. Berger, P. Slosar, Z. Schwartz, D. J. Cohen, S. B. Goodman, P. A. Anderson and B. D. Boyan, A Review of Biomimetic Topographies and Their Role in Promoting Bone Formation and Osseointegration: Implications for Clinical Use, Biomimetics, 2022, 7, 46 CrossRef CAS PubMed.
- Y. He, Y. Gao, Q. Ma, X. Zhang, Y. Zhang and W. Song, Nanotopographical cues for regulation of macrophages and osteoclasts: emerging opportunities for osseointegration, J. Nanobiotechnol., 2022, 20, 510 CrossRef PubMed.
- K. Komatsu, T. Matsuura, J. Cheng, D. Kido, W. Park and T. Ogawa, Nanofeatured surfaces in dental implants: contemporary insights and impending challenges, Int. J. Implant Dent., 2024, 10, 34 CrossRef PubMed.
- X. Li, L. H. Klausen, W. Zhang, Z. Jahed, C.-T. Tsai, T. L. Li and B. Cui, Nanoscale Surface Topography Reduces Focal Adhesions and Cell Stiffness by Enhancing Integrin Endocytosis, Nano Lett., 2021, 21, 8518–8526 Search PubMed.
- V. D. Tran and S. Kumar, Transduction of cell and matrix geometric cues by the actin cytoskeleton, Curr. Opin. Cell Biol., 2021, 68, 64–71 CrossRef CAS PubMed.
- I. Casanellas, J. Samitier and A. Lagunas, Recent advances in engineering nanotopographic substrates for cell studies, Front. Bioeng. Biotechnol., 2022, 10, 1002967 CrossRef PubMed.
- J. Luo, M. Walker, Y. Xiao, H. Donnelly, M. J. Dalby and M. Salmeron-Sanchez, The influence of nanotopography on cell behaviour through interactions with the extracellular matrix – A review, Bioact. Mater., 2022, 15, 145–159 CAS.
- F. Batool, H. Özçelik, C. Stutz, P.-Y. Gegout, N. Benkirane-Jessel, C. Petit and O. Huck, Modulation of immune-inflammatory responses through surface modifications of biomaterials to promote bone healing and regeneration, J. Tissue Eng., 2021, 12, 20417314211041428 CrossRef PubMed.
- Y. Liu, M. Gao, Y. Yan, X. Wang, Z. Dong, L. Cheng and Y. Xu, Immunology in Osseointegration After Implantation, J. Biomed. Mater. Res., 2025, 113, e35566 CrossRef CAS PubMed.
- Z. Liu, X. Liu and S. Ramakrishna, Surface engineering of biomaterials in orthopedic and dental implants: Strategies to improve osteointegration, bacteriostatic and bactericidal activities, Biotechnol. J., 2021, 16, 2000116 CrossRef CAS PubMed.
- D. Wähnert, J. Greiner, S. Brianza, C. Kaltschmidt, T. Vordemvenne and B. Kaltschmidt, Strategies to Improve Bone Healing: Innovative Surgical Implants Meet Nano-/Micro-Topography of Bone Scaffolds, Biomedicines, 2021, 9, 746 CrossRef PubMed.
- M. F. Kunrath, F. M. Diz, R. Magini and M. E. Galárraga-Vinueza, Nanointeraction: The profound influence of nanostructured and nano-drug delivery biomedical implant surfaces on cell behavior, Adv. Colloid Interface Sci., 2020, 284, 102265 CrossRef CAS PubMed.
- E. Martinez, J. P. St-Pierre and F. Variola, Advanced bioengineering technologies for preclinical research, Adv. Phys.: X, 2019, 4, 1622451, DOI:10.1080/23746149.2019.1622451.
- A. Wang, L. A. Madden and V. N. Paunov, Advanced biomedical applications based on emerging 3D cell culturing platforms, J. Mater. Chem. B, 2020, 8, 10487–10501 Search PubMed.
- I. Yuste, F. C. Luciano, E. González-Burgos, A. Lalatsa and D. R. Serrano, Mimicking bone microenvironment: 2D and 3D in vitro models of human osteoblasts, Pharmacol. Res., 2021, 169, 105626 CrossRef CAS PubMed.
- C. Ma, X. Duan and X. Lei, 3D cell culture model: From ground experiment to microgravity study, Front. Bioeng. Biotechnol., 2023, 11, 1136583 CrossRef PubMed.
- O. Urzì, R. Gasparro, E. Costanzo, A. De Luca, G. Giavaresi, S. Fontana and R. Alessandro, Three-Dimensional Cell Cultures: The Bridge between In Vitro and In Vivo Models, Int. J. Mol. Sci., 2023, 24, 12046 CrossRef PubMed.
- R. De Pace, M. R. Iaquinta, A. Benkhalqui, A. D'Agostino, L. Trevisiol, R. Nocini, C. Mazziotta, J. C. Rotondo, I. Bononi, M. Tognon, F. Martini and E. Mazzoni, Revolutionizing bone healing: the role of 3D models, Cell Regener., 2025, 14, 7 CrossRef CAS PubMed.
- C. Frantz, K. M. Stewart and V. M. Weaver, The extracellular matrix at a glance, J. Cell Sci., 2010, 123, 4195–4200 CrossRef CAS PubMed.
- M. Barczyk, S. Carracedo and D. Gullberg, Integrins, Cell Tissue Res., 2010, 339, 269–280 CrossRef CAS PubMed.
- K. Legerstee and A. Houtsmuller, A Layered View on Focal Adhesions, Biology, 2021, 10, 1189 CrossRef CAS PubMed.
- Y. G. Mishra and B. Manavathi, Focal adhesion dynamics in cellular function and disease, Cell. Signalling, 2021, 85, 110046 CrossRef CAS PubMed.
- P. Kanchanawong and D. A. Calderwood, Organization, dynamics and mechanoregulation of integrin-mediated cell–ECM adhesions, Nat. Rev. Mol. Cell Biol., 2023, 24, 142–161 CrossRef CAS PubMed.
- J. R. W. Conway, A. Isomursu, G. Follain, V. Härmä, E. Jou-Ollé, N. Pasquier, E. P. O. Välimäki, J. K. Rantala and J. Ivaska, Defined extracellular matrix compositions support stiffness-insensitive cell spreading and adhesion signaling, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, e2304288120 CrossRef CAS PubMed.
- J. T. Parsons, A. R. Horwitz and M. A. Schwartz, Cell adhesion: integrating cytoskeletal dynamics and cellular tension, Nat. Rev. Mol. Cell Biol., 2010, 11, 633–643 CrossRef CAS PubMed.
- D. W. Zhou, T. T. Lee, S. Weng, J. Fu and A. J. García, Effects of substrate stiffness and actomyosin contractility on coupling between force transmission and vinculin–paxillin recruitment at single focal adhesions, Mol. Biol. Cell, 2017, 28, 1901–1911 CrossRef CAS PubMed.
- J. Hoon, M. Tan and C.-G. Koh, The Regulation of Cellular Responses to Mechanical Cues by Rho GTPases, Cells, 2016, 5, 17 CrossRef PubMed.
- M. Fischer, P. Rikeit, P. Knaus and C. Coirault, YAP-Mediated Mechanotransduction in Skeletal Muscle, Front. Physiol., 2016, 7 DOI:10.3389/fphys.2016.00041.
- A. Elosegui-Artola, I. Andreu, A. E. M. Beedle, A. Lezamiz, M. Uroz, A. J. Kosmalska, R. Oria, J. Z. Kechagia, P. Rico-Lastres, A.-L. Le Roux, C. M. Shanahan, X. Trepat, D. Navajas, S. Garcia-Manyes and P. Roca-Cusachs, Force Triggers YAP Nuclear Entry by Regulating Transport across Nuclear Pores, Cell, 2017, 171, 1397–1410.e14 CrossRef CAS PubMed.
- I. Dasgupta and D. McCollum, Control of cellular responses to mechanical cues through YAP/TAZ regulation, J. Biol. Chem., 2019, 294, 17693–17706 CrossRef CAS PubMed.
- C. Mas-Moruno, B. Su and M. J. Dalby, Multifunctional Coatings and Nanotopographies: Toward Cell Instructive and Antibacterial Implants, Adv. Healthcare Mater., 2019, 8, 1801103 CrossRef PubMed.
- H. Miyoshi and T. Adachi, Topography Design Concept of a Tissue Engineering Scaffold for Controlling Cell Function and Fate Through Actin Cytoskeletal Modulation, Tissue Eng., Part B, 2014, 20, 609–627 CrossRef CAS PubMed.
- M. J. P. Biggs, R. G. Richards and M. J. Dalby, Nanotopographical modification: a regulator of cellular function through focal adhesions, Nanomedicine, 2010, 6, 619–633 CrossRef CAS PubMed.
- L. Iturriaga, K. D. Van Gordon, G. Larrañaga-Jaurrieta and S. Camarero-Espinosa, Strategies to Introduce Topographical and Structural Cues in 3D-Printed Scaffolds and Implications in Tissue Regeneration, Adv. NanoBiomed Res., 2021, 1, 2100068 Search PubMed.
- P. Rougerie, R. Silva Dos Santos, M. Farina and K. Anselme, Molecular Mechanisms of Topography Sensing by Osteoblasts: An Update, Appl. Sci., 2021, 11, 1791 CrossRef CAS.
- R. Berthelot and F. Variola, Investigating the interplay between environmental conditioning and nanotopographical cueing on the response of human MG63 osteoblastic cells to titanium nanotubes, Biomater. Sci., 2025, 13, 946–968 RSC.
- M. J. Dalby, N. Gadegaard, R. Tare, A. Andar, M. O. Riehle, P. Herzyk, C. D. W. Wilkinson and R. O. C. Oreffo, The control of human mesenchymal cell differentiation using nanoscale symmetry and disorder, Nat. Mater., 2007, 6, 997–1003 CrossRef CAS PubMed.
- M. J. Dalby, N. Gadegaard and R. O. C. Oreffo, Harnessing nanotopography and integrin–matrix interactions to influence stem cell fate, Nat. Mater., 2014, 13, 558–569 CrossRef CAS PubMed.
- E. Ngandu Mpoyi, M. Cantini, P. M. Reynolds, N. Gadegaard, M. J. Dalby and M. Salmerón-Sánchez, Protein Adsorption as a Key Mediator in the Nanotopographical Control of Cell Behavior, ACS Nano, 2016, 10, 6638–6647 CrossRef CAS PubMed.
- P. R. L. Dabare, A. Bachhuka, E. Parkinson-Lawrence and K. Vasilev, Surface nanotopography mediated albumin adsorption, unfolding and modulation of early innate immune responses, Mater. Today Adv., 2021, 12, 100187 CrossRef CAS.
- Y. Yang, S. Knust, S. Schwiderek, Q. Qin, Q. Yun, G. Grundmeier and A. Keller, Protein Adsorption at Nanorough Titanium Oxide Surfaces: The Importance of Surface Statistical Parameters beyond Surface Roughness, Nanomaterials, 2021, 11, 357 CrossRef CAS PubMed.
- A. Dehghanghadikolaei and B. Fotovvati, Coating Techniques for Functional Enhancement of Metal Implants for Bone Replacement: A Review, Materials, 2019, 12, 1795 CrossRef CAS PubMed.
- X. Guo, Y. Hu, K. Yuan and Y. Qiao, Review of the Effect of Surface Coating Modification on Magnesium Alloy Biocompatibility, Materials, 2022, 15, 3291 CrossRef CAS PubMed.
- R. Zhang, B. Han and X. Liu, Functional Surface Coatings on Orthodontic Appliances: Reviews of Friction Reduction, Antibacterial Properties, and Corrosion Resistance, Int. J. Mol. Sci., 2023, 24, 6919 CrossRef CAS PubMed.
- H. Shahali, J. Hasan, H.-H. Cheng, S. Ramarishna and P. K. Yarlagadda, A systematic approach towards biomimicry of nanopatterned cicada wings on titanium using electron beam lithography, Nanotechnology, 2021, 32, 065301 CrossRef CAS PubMed.
- L. Liu, A. Holzer, N. Raney and V. E. Babicheva, Nanoantennas and metasurfaces tailored by electron beam lithography and substrate conductivity, Sci. Rep., 2025, 15, 29339 CrossRef CAS PubMed.
- Y. Cao, D. Ma, H. Li, G. Cui, J. Zhang and Z. Yang, Review of Industrialization Development of Nanoimprint Lithography Technology, Chips, 2025, 4, 10 CrossRef.
- L. Vincenti, P. Pellegrino, M. Cascione, V. D. Matteis, I. Farella, F. Quaranta and R. Rinaldi, Crafting at the nanoscale: A comprehensive review of mechanical Atomic force microscopy-based lithography methods and their evolution, Mater. Des., 2024, 243 DOI:10.1016/j.matdes.2024.113036.
- D. Petit, C. C. Faulkner, S. Johnstone, D. Wood and R. P. Cowburn, Nanometer scale patterning using focused ion beam milling, Rev. Sci. Instrum., 2005, 76 DOI:10.1063/1.1844431.
- M. G. Stanford, B. B. Lewis, K. Mahady, J. D. Fowlkes and P. D. Rack, Review Article: Advanced nanoscale patterning and material synthesis with gas field helium and neon ion beams, J. Vac. Sci. Technol., B: Nanotechnol. Microelectron.: Mater., Process., Meas., Phenom., 2017, 35 DOI:10.1116/1.4981016.
- R. Córdoba, P. Orús, S. Strohauer, T. E. Torres and J. M. D. Teresa, Ultra-fast direct growth of metallic micro- and nano-structures by focused ion beam irradiation, Sci. Rep., 2019, 9 DOI:10.1038/s41598-019-50411-w.
- K. O. Böker, F. Kleinwort, J. H. Klein-Wiele, P. Simon, K. Jäckle, S. Taheri, W. Lehmann and A. F. Schilling, Laser ablated periodic nanostructures on titanium and steel implants influence adhesion and osteogenic differentiation of mesenchymal stem cells, Materials, 2020, 13 DOI:10.3390/MA13163526.
- Y. Ma, L. Jiang, J. Hu, H. Liu, S. Wang, P. Zuo, P. Ji, L. Qu and T. Cui, Multifunctional 3D Micro-Nanostructures Fabricated through Temporally Shaped Femtosecond Laser Processing for Preventing Thrombosis and Bacterial Infection, ACS Appl. Mater. Interfaces, 2020, 12, 17155–17166 CrossRef CAS PubMed.
- H. Yao, X. Zou, S. Zheng, Y. Hu, S. Zhang, C. Liang, H. Zhou, D. Wang, H. Wang, L. Yang and Q. Li, Femtosecond laser-induced nanoporous layer for enhanced osteogenesis of titanium implants, Mater. Sci. Eng., C, 2021, 127 DOI:10.1016/j.msec.2021.112247.
- L. Gao, Q. Zhang and M. Gu, Femtosecond laser micro/nano processing: from fundamental to applications, Int. J. Extreme Manuf., 2024, 7 DOI:10.1088/2631-7990/ad943e.
- J. Alipal, T. C. Lee, P. Koshy, H. Z. Abdullah and M. I. Idris, Evolution of anodised titanium for implant applications, Heliyon, 2021, 7 DOI:10.1016/j.heliyon.2021.e07408.
- K. Gulati, Y. Zhang, P. Di, Y. Liu and S. Ivanovski, Research to Clinics: Clinical Translation Considerations for Anodized Nano-Engineered Titanium Implants, ACS Biomater. Sci. Eng., 2022, 8, 4077–4091 CrossRef CAS PubMed.
- Q. Wang, P. Zhou, S. Liu, S. Attarilar, R. L. W. Ma, Y. Zhong and L. Wang, Multi-scale surface treatments of titanium implants for rapid osseointegration: A review, Nanomaterials, 2020, 10, 1–27 Search PubMed.
- K. Gulati, R. D. O. Martinez, M. Czerwiński and M. Michalska-Domańska, Understanding the influence of electrolyte aging in electrochemical anodization of titanium, Adv. Colloid Interface Sci., 2022, 302 DOI:10.1016/j.cis.2022.102615.
- T. Guo, J. C. Scimeca, S. Ivanovski, E. Verron and K. Gulati, Enhanced Corrosion Resistance and Local Therapy from Nano-Engineered Titanium Dental Implants, Pharmaceutics, 2023, 15 DOI:10.3390/pharmaceutics15020315.
- Y. He, M. Yao, J. Zhou, J. Xie, C. Liang, D. Yin, S. Huang, Y. Zhang, F. Peng and S. Cheng, Mg(OH)2 nanosheets on Ti with immunomodulatory function for orthopedic applications, Regener. Biomater., 2022, 9 DOI:10.1093/rb/rbac027.
- İ. Erol, G. Khamidov, E. Efe, S. Yurdakal and L. Palmisano, Hydrothermal synthesis of bare TiO2 nanowires and polystyrene (PS)-TiO2 nanowires used for selective photocatalytic oxidation of 3-pyridinemethanol in water and PS photodegradation in solid state, Res. Chem. Intermed., 2024, 50, 4641–4671 CrossRef.
- A. M. Tarasov, L. I. Sorokina, D. A. Dronova, O. Volovlikova, A. Y. Trifonov, S. S. Itskov, A. V. Tregubov, E. N. Shabaeva, E. S. Zhurina, S. V. Dubkov, D. V. Kozlov and D. Gromov, Influence of the Structure of Hydrothermal-Synthesized TiO2 Nanowires Formed by Annealing on the Photocatalytic Reduction of CO2 in H2O Vapor, Nanomaterials, 2024, 14 DOI:10.3390/nano14161370.
- D. V. Wellia, R. M. Rahma, S. Arief, R. Subagyo and Y. Kusumawati, Controlling the formation of 1D TiO2 nanowires and their performance in photoreduction of chromium Cr(VI), Case Stud. Chem. Environ. Eng., 2024, 9 DOI:10.1016/j.cscee.2024.100719.
- B. Lam, R. D. Holmes, J. Das, M. Poudineh, A. Sage, E. H. Sargent and S. O. Kelley, Optimized templates for bottom-up growth of high-performance integrated biomolecular detectors, Lab Chip, 2013, 13, 2569–2575 Search PubMed.
- M. Arefpour, M. A. Kashi, A. Ramazani and A. H. Montazer, Electrochemical pore filling strategy for controlled growth of magnetic and metallic nanowire arrays with large area uniformity, Nanotechnology, 2016, 27 DOI:10.1088/0957-4484/27/27/275605.
- C. Fernández-González, J. C. Guzmán-Mínguez, A. Guedeja-Marrón, E. García-Martín, M. Foerster, M. Á. Niño, L. Aballe, A. Quesada, L. Pérez and S. Ruiz-Gómez, Scaling up the production of electrodeposited nanowires: A roadmap towards applications, Nanomaterials, 2021, 11 DOI:10.3390/nano11071657.
- B. Xu, Y. He, Y. Zhang, Z. Ma, Y. Zhang and W. Song, In Situ Growth of Tunable Gold Nanoparticles by Titania Nanotubes Templated Electrodeposition for Improving Osteogenesis through Modulating Macrophages Polarization, ACS Appl. Mater. Interfaces, 2022, 14, 50520–50533 CrossRef CAS PubMed.
- K. Man, S. Mazumder, N. B. Dahotre and Y. Yang, Surface Nanostructures Enhanced Biocompatibility and Osteoinductivity of Laser-Additively Manufactured CoCrMo Alloys, ACS Omega, 2023, 8, 47658–47666 Search PubMed.
- S. Kedia, S. K. Bonagani, A. G. Majumdar, V. Kain, M. Subramanian, N. Maiti and J. P. Nilaya, Nanosecond laser surface texturing of type 316L stainless steel for contact guidance of bone cells and superior corrosion resistance, Colloid Interface Sci. Commun., 2021, 42, 100419 Search PubMed.
- M. Oberringer, E. Akman, J. Lee, W. Metzger, C. K. Akkan, E. Kacar, A. Demir, H. Abdul-Khaliq, N. Pütz, G. Wennemuth, T. Pohlemann, M. Veith and C. Aktas, Reduced myofibroblast differentiation on femtosecond laser treated 316LS stainless steel, Mater. Sci. Eng., C, 2013, 33, 901–908 CrossRef CAS PubMed.
- M. A. Bashir, T. Khan and R. Nair, Evaluation of hydrophilicity and surface morphology of nanosecond-pulsed laser-engineered surface textures on stainless steel, cobalt-chromium, and titanium alloys, J. Mater. Sci.: Mater. Med., 2025, 36, 106 CrossRef CAS PubMed.
- A. Rodriguez-Contreras, D. Guadarrama Bello, S. Flynn, F. Variola, J. D. Wuest and A. Nanci, Chemical nanocavitation of surfaces to enhance the utility of stainless steel as a medical material, Colloids Surf., B, 2018, 161, 677–687 CrossRef CAS PubMed.
- A. Ozgun, D. Lomboni, H. Arnott, W. A. Staines, J. Woulfe and F. Variola, Biomaterials-based strategies for in vitro neural models, Biomater. Sci., 2022, 10, 1134–1165 RSC.
- I. Chiesa, C. D. Maria, A. Lapomarda, G. M. Fortunato, F. Montemurro, R. D. Gesù, R. S. Tuan, G. Vozzi and R. Gottardi, Endothelial cells support osteogenesis in an in vitro vascularized bone model developed by 3D bioprinting, Biofabrication, 2020, 12 DOI:10.1088/1758-5090/ab6a1d.
- V. Prabhakaran, F. P. W. Melchels, L. M. Murray and J. Z. Paxton, Engineering three-dimensional bone macro-tissues by guided fusion of cell spheroids, Front. Endocrinol., 2023, 14 DOI:10.3389/fendo.2023.1308604.
- B. W. M. de Wildt, E. E. A. Cramer, L. S. de Silva, K. Ito, D. Gawlitta and S. Hofmann, Evaluating material-driven regeneration in a tissue engineered human in vitro bone defect model, Bone, 2023, 166 DOI:10.1016/j.bone.2022.116597.
- M. Maglio, M. Fini, M. Sartori, G. Codispoti, V. Borsari, D. Dallari, S. Ambretti, M. Rocchi and M. Tschon, An Advanced Human Bone Tissue Culture Model for the Assessment of Implant Osteointegration In Vitro, Int. J. Mol. Sci., 2024, 25 DOI:10.3390/ijms25105322.
- S. Vermeulen, K. Knoops, H. Duimel, M. Parvizifard, D. van Beurden, C. López-Iglesias, S. Giselbrecht, R. Truckenmüller, P. Habibović and Z. T. Birgani, An in vitro model system based on calcium- and phosphate ion-induced hMSC spheroid mineralization, Mater. Today Bio, 2023, 23 DOI:10.1016/j.mtbio.2023.100844.
- D. Boscaro and P. Sikorski, Spheroids as a 3D in vitro model to study bone and bone mineralization, Biomater. Adv., 2024, 157 DOI:10.1016/j.bioadv.2023.213727.
- S. Frenz-Wiessner, S. D. Fairley, M. Buser, I. Goek, K. Salewskij, G. Jonsson, D. Illig, B. zu Putlitz, D. Petersheim, Y. Li, P. H. Chen, M. Kalauz, R. Conca, M. Sterr, J. Geuder, Y. Mizoguchi, R. T. A. Megens, M. I. Linder, D. Kotlarz, M. Rudelius, J. M. Penninger, C. Marr and C. Klein, Generation of complex bone marrow organoids from human induced pluripotent stem cells, Nat. Methods, 2024, 21, 868–881 CrossRef CAS PubMed.
- M. K. Kim, K. Paek, S. M. Woo and J. A. Kim, Bone-on-a-Chip: Biomimetic Models Based on Microfluidic Technologies for Biomedical Applications, ACS Biomater. Sci. Eng., 2023, 9, 3058–3073 CrossRef CAS PubMed.
- E. E. A. Cramer, K. Ito and S. Hofmann, Ex vivo Bone Models and Their Potential in Preclinical Evaluation, Curr. Osteoporos. Rep., 2021, 19, 75–87 CrossRef CAS PubMed.
- A. Scarano, A. G. A. Khater, S. A. Gehrke, F. Inchingolo and S. R. Tari, Animal Models for Investigating Osseointegration: An Overview of Implant Research over the Last Three Decades, J. Funct. Biomater., 2024, 15 DOI:10.3390/jfb15040083.
- R. M. Varut, D. M. Trasca, G. A. Stoica, C. Sirbulet, C. C. Arsenie and C. Popescu, Animal Models as Foundational Tools in Preclinical Orthopedic Implant Research, Biomedicines, 2025, 13 DOI:10.3390/biomedicines13102468.
- S. E. A. Camargo, X. Xia, C. Fares, F. Ren, S.-M. Hsu, D. Budei, C. Aravindraja, L. Kesavalu and J. F. Esquivel-Upshaw, Nanostructured Surfaces to Promote Osteoblast Proliferation and Minimize Bacterial Adhesion on Titanium, Materials, 2021, 14, 4357 Search PubMed.
- B. Voltrova, V. Hybasek, V. Blahnova, J. Sepitka, V. Lukasova, K. Vocetkova, V. Sovkova, R. Matejka, J. Fojt, L. Joska, M. Daniel and E. Filova, Different diameters of titanium dioxide nanotubes modulate Saos-2 osteoblast-like cell adhesion and osteogenic differentiation and nanomechanical properties of the surface, RSC Adv., 2019, 9, 11341–11355 RSC.
- L. Fanton, F. Loria, M. Amores, M. R. Pazos, C. Adán, R. A. García-Muñoz and J. Marugán, Proliferation of osteoblast precursor cells on the surface of TiO2 nanowires anodically grown on a β-type biomedical titanium alloy, Sci. Rep., 2022, 12, 7895 CrossRef CAS PubMed.
- J. Luo, S. Zhao, X. Gao, S. N. Varma, W. Xu, M. Tamaddon, R. Thorogate, H. Yu, X. Lu, M. Salmeron-Sanchez and C. Liu, TiO2 Nanotopography-Driven Osteoblast Adhesion through Coulomb's Force Evolution, ACS Appl. Mater. Interfaces, 2022, 14, 34400–34414 CrossRef CAS PubMed.
- M.-K. Lee, H. Lee, H.-E. Kim, E.-J. Lee, T.-S. Jang, H.-D. Jung, M.-K. Lee, H. Lee, H.-E. Kim, E.-J. Lee, T.-S. Jang and H.-D. Jung, Nano-Topographical Control of Ti-Nb-Zr Alloy Surfaces for Enhanced Osteoblastic Response, Nanomaterials, 2021, 11 DOI:10.3390/nano11061507.
- J. Yang, L. E. McNamara, N. Gadegaard, E. V. Alakpa, K. V. Burgess, R. M. D. Meek and M. J. Dalby, Nanotopographical Induction of Osteogenesis through Adhesion, Bone Morphogenic Protein Cosignaling, and Regulation of MicroRNAs, ACS Nano, 2014, 8, 9941–9953 CrossRef CAS PubMed.
- D. G. Costa, E. P. Ferraz, R. P. F. Abuna, P. T. de Oliveira, M. Morra, M. M. Beloti and A. L. Rosa, The effect of collagen coating on titanium with nanotopography on in vitro osteogenesis, J. Biomed. Mater. Res., Part A, 2017, 105, 2783–2788 CrossRef CAS PubMed.
- H. B. Lopes, G. P. Freitas, D. M. C. Fantacini, V. Picanço-Castro, D. T. Covas, A. L. Rosa and M. M. Beloti, Titanium with nanotopography induces osteoblast differentiation through regulation of integrin αV, J. Cell. Biochem., 2019, 120, 16723–16732 CrossRef CAS PubMed.
- L. Li, S. Yang, L. Xu, Y. Li, Y. Fu, H. Zhang and J. Song, Nanotopography on titanium promotes osteogenesis via autophagy-mediated signaling between YAP and β-catenin, Acta Biomater., 2019, 96, 674–685 CrossRef CAS PubMed.
- M. Qadir, J. Lin, A. Biesiekierski, Y. Li and C. Wen, Effect of Anodized TiO2-Nb2O5-ZrO2 Nanotubes with Different Nanoscale Dimensions on the Biocompatibility of a Ti35Zr28Nb Alloy, ACS Appl. Mater. Interfaces, 2020, 12, 6776–6787 CrossRef CAS PubMed.
- J. Park, S. Bauer, K. A. Schlegel, F. W. Neukam, K. Von Der Mark and P. Schmuki, TiO2 Nanotube Surfaces: 15 nm—An Optimal Length Scale of Surface Topography for Cell Adhesion and Differentiation, Small, 2009, 5, 666–671 CrossRef CAS PubMed.
- A. Ballo, H. Agheli, J. Lausmaa, P. Thomsen and S. Petronis, Nanostructured model implants for in vivo studies: influence of well-defined nanotopography on de novo bone formation on titanium implants, Int. J. Nanomed., 2011, 6, 3415–3428 CrossRef CAS PubMed.
- C. von Wilmowsky, S. Bauer, S. Roedl, F. W. Neukam, P. Schmuki and K. A. Schlegel, The diameter of anodic TiO2 nanotubes affects bone formation and correlates with the bone morphogenetic protein-2 expression in vivo, Clin. Oral Implants Res., 2012, 23, 359–366, DOI:10.1111/j.1600-0501.2010.02139.x.
- E. Beltrán-Partida, B. Valdéz-Salas, A. Moreno-Ulloa, A. Escamilla, M. A. Curiel, R. Rosales-Ibáñez, F. Villarreal, D. M. Bastidas and J. M. Bastidas, Improved in vitro angiogenic behavior on anodized titanium dioxide nanotubes, J. Nanobiotechnol., 2017, 15, 10 CrossRef PubMed.
- R. L. Bighetti-Trevisan, E. P. Ferraz, M. B. F. Silva, G. C. Zatta, M. B. de Almeida, A. L. Rosa and M. M. Beloti, Effect of osteoblasts on osteoclast differentiation and activity induced by titanium with nanotopography, Colloids Surf., B, 2023, 229, 113448 CrossRef CAS PubMed.
- V. Campos-Bijit, N. C. Inostroza, R. Orellana, A. Rivera, A. V. Marttens, C. Cortez, C. Covarrubias, V. Campos-Bijit, N. C. Inostroza, R. Orellana, A. Rivera, A. V. Marttens, C. Cortez and C. Covarrubias, Influence of Topography and Composition of Commercial Titanium Dental Implants on Cell Adhesion of Human Gingiva-Derived Mesenchymal Stem Cells: An In Vitro Study, Int. J. Mol. Sci., 2023, 24 DOI:10.3390/ijms242316686.
- N.-J. Cao, Y.-H. Zhu, F. Gao, C. Liang, Z.-B. Wang, Y. Zhang, C.-P. Hao and W. Wang, Gradient nanostructured titanium stimulates cell responses in vitro and enhances osseointegration in vivo, Ann. Transl. Med., 2021, 9, 531–531 CrossRef CAS PubMed.
- E. A. Baker, M. M. Fleischer, A. D. Vara, M. R. Salisbury, K. C. Baker, P. T. Fortin, C. R. Friedrich, E. A. Baker, M. M. Fleischer, A. D. Vara, M. R. Salisbury, K. C. Baker, P. T. Fortin and C. R. Friedrich, Local and Systemic In Vivo Responses to Osseointegrative Titanium Nanotube Surfaces, Nanomaterials, 2021, 11 DOI:10.3390/nano11030583.
- Y. He, Z. Li, X. Ding, B. Xu, J. Wang, Y. Li, F. Chen, F. Meng, W. Song and Y. Zhang, Nanoporous titanium implant surface promotes osteogenesis by suppressing osteoclastogenesis via integrin β1/FAKpY397/MAPK pathway, Bioact. Mater., 2022, 8, 109–123 CAS.
- C. Wang, S. Gao, R. Lu, X. Wang and S. Chen, In Vitro and In Vivo Studies of Hydrogenated Titanium Dioxide Nanotubes with Superhydrophilic Surfaces during Early Osseointegration, Cells, 2022, 11, 3417 CrossRef CAS PubMed.
- A. Klymov, L. Prodanov, E. Lamers, J. A. Jansen and X. Frank Walboomers, Understanding the role of nano-topography on the surface of a bone-implant, Biomater. Sci., 2013, 1, 135–151 RSC.
- Y. Li, L. Tang, M. Shen, Z. Wang and X. Huang, A comparative study of Sr-loaded nano-textured Ti and TiO2 nanotube implants on osseointegration immediately after tooth extraction in Beagle dogs, Front. Mater., 2023, 10 DOI:10.3389/fmats.2023.1213163.
- L. Lin, H. Wang, M. Ni, Y. Rui, T.-Y. Cheng, C.-K. Cheng, X. Pan, G. Li and C. Lin, Enhanced osteointegration of medical titanium implant with surface modifications in micro/nanoscale structures, J. Orthop. Translat., 2014, 2, 35–42 CrossRef.
- D. Karazisis, L. Rasmusson, S. Petronis, A. Palmquist, F. A. Shah, H. Agheli, L. Emanuelsson, A. Johansson, O. Omar and P. Thomsen, The effects of controlled nanotopography, machined topography and their combination on molecular activities, bone formation and biomechanical stability during osseointegration, Acta Biomater., 2021, 136, 279–290 CrossRef CAS PubMed.
- R. M. Wazen, S. Kuroda, C. Nishio, K. Sellin, J. B. Brunski and A. Nanci, Gene Expression Profiling and Histomorphometric Analyses of The Early Bone Healing Response Around Nanotextured Implants, Nanomedicine, 2013, 8, 1385–1395 CrossRef CAS PubMed.
- J. Gil and M. Sanz, Bactericidal nanotopography of titanium dental implants: in vitro and in vivo studies, Clin. Oral Investig., 2025, 29, 351 CrossRef PubMed.
- M. B. Ariganello, D. Guadarrama Bello, A. Rodriguez-Contreras, S. Sadeghi, G. Isola, F. Variola and A. Nanci, Surface nanocavitation of titanium modulates macrophage activity, Int. J. Nanomed., 2018, 13, 8297–8308 CrossRef CAS PubMed.
- D. Guadarrama Bello, A. Fouillen, A. Badia and A. Nanci, A nanoporous titanium surface promotes the maturation of focal adhesions and formation of filopodia with distinctive nanoscale protrusions by osteogenic cells, Acta Biomater., 2017, 60, 339–349 CrossRef CAS PubMed.
- G. Zhu, G. Wang and J. J. Li, Advances in implant surface modifications to improve osseointegration, Mater. Adv., 2021, 2, 6901–6927 RSC.
- C. Hou, J. An, D. Zhao, X. Ma, W. Zhang, W. Zhao, M. Wu, Z. Zhang and F. Yuan, Surface Modification Techniques to Produce Micro/Nano-scale Topographies on Ti-Based Implant Surfaces for Improved Osseointegration, Front. Bioeng. Biotechnol., 2022, 10 DOI:10.3389/fbioe.2022.835008.
- S. Yang, W. Jiang, X. Ma, Z. Wang, R. L. Sah, J. Wang and Y. Sun, Nanoscale Morphologies on the Surface of 3D-Printed Titanium Implants for Improved Osseointegration: A Systematic Review of the Literature, Int. J. Nanomed., 2023, 18, 4171–4191 CrossRef CAS PubMed.
- P. Yuan, M. Chen, X. Lu, H. Yang, L. Wang, T. Bai, W. Zhou, T. Liu and S. Yu, Application of advanced surface modification techniques in titanium-based implants: latest strategies for enhanced antibacterial properties and osseointegration, J. Mater. Chem. B, 2024, 12, 10516–10549 RSC.
- H. Ghodrati, A. Goodarzi, M. Golrokhian, F. Fattahi, R. M. Anzabi, M. Mohammadikhah, S. Sadeghi and S. Mirhadi, A narrative review of recent developments in osseointegration and anti-corrosion of titanium dental implants with nano surface, Bone Rep., 2025, 25 DOI:10.1016/j.bonr.2025.101846.
- J. Li, H. Fan, H. Li, L. Hua, J. Du, Y. He and Y. Jin, Recent Advancements in the Surface Modification of Additively Manufactured Metallic Bone Implants, Addit. Manuf. Front., 2025, 4, 200195, DOI:10.1016/j.amf.2025.200195.
- A. Ketabchi, A. Weck and F. Variola, Influence of oxidative nanopatterning and anodization on the fatigue resistance of commercially pure titanium and Ti–6Al–4V, J. Biomed. Mater. Res., 2015, 103, 563–571 CrossRef PubMed.
- J. K. Lee, D. S. Choi, I. Jang and W. Y. Choi, Improved osseointegration of dental titanium implants by tio2 nanotube arrays with recombinant human bone morphogenetic protein-2: A pilot in vivo study, Int. J. Nanomed., 2015, 10, 1145–1154 CAS.
- S. Ozan, Ş. Kasman, I. C. Uçar and C. Wen, Laser surface texturing for enhanced implant biocompatibility and osseointegration: A critical review of its effects on surface properties, Clin. Biomech., 2025, 126 DOI:10.1016/j.clinbiomech.2025.106540.
- F. A. Shah, M. L. Johansson, O. Omar, H. Simonsson, A. Palmquist and P. Thomsen, Laser-Modified Surface Enhances Osseointegration and Biomechanical Anchorage of Commercially Pure Titanium Implants for Bone-Anchored Hearing Systems, PLoS One, 2016, 11, e0157504 CrossRef PubMed.
- M. J. Coathup, G. W. Blunn, N. Mirhosseini, K. Erskine, Z. Liu, D. R. Garrod and L. Li, Controlled laser texturing of titanium results in reliable osteointegration, J. Orthop. Res., 2017, 35, 820–828 CrossRef CAS PubMed.
- J. Kim, Mathematical modeling approaches to describe the dynamics of protein adsorption at solid interfaces, Colloids Surf., B, 2018, 162, 370–379 CrossRef CAS PubMed.
- D. Guadarrama Bello, F. Variola and A. Nanci, Bone Healing Around Implants in Normal and Medically Compromised Conditions: Osteoporosis and Diabetes, Adv. Healthcare Mater., 2026, e04701 CrossRef CAS PubMed.
- C. Hu, D. Ashok, D. R. Nisbet and V. Gautam, Bioinspired surface modification of orthopedic implants for bone tissue engineering, Biomaterials, 2019, 219 DOI:10.1016/j.biomaterials.2019.119366.
- V. Hernández-Montes, R. Buitrago-Sierra, M. Echeverry-Rendón and J. F. Santa-Marín, Ceria-based coatings on magnesium alloys for biomedical applications: a literature review, RSC Adv., 2023, 13, 1422–1433 RSC.
- S. Tripathi, A. Raheem, M. Dash, P. Kumar, A. Elsebahy, H. Singh, G. Manivasagam and H. S. Nanda, Surface engineering of orthopedic implants for better clinical adoption, J. Mater. Chem. B, 2024, 12, 11302–11335 RSC.
- C. Verma, S. Y. Lee, J. Gautam, S. Dubey, P. Singh, K. Y. Rhee, E. E. Ebenso, A. Alfantazi and S. J. Park, Hydroxyapatite-based coatings for corrosion resistance and self-healing in biomedical and industrial applications, Acta Biomater., 2025, 206, 1–42 CrossRef CAS PubMed.
- E. Abyzova, E. Dogadina, R. D. Rodriguez, I. Petrov, Y. Kolesnikova, M. Zhou, C. Liu and E. Sheremet, Beyond Tissue replacement: The Emerging role of smart implants in healthcare, Mater. Today Bio, 2023, 22 DOI:10.1016/j.mtbio.2023.100784.
- J. Huang, H. Dong, W. Zhang, J. Cui, Q. Li, J. Wang, Z. Zhao and X. Zang, Sensor-enabled Orthopedic Implants for Musculoskeletal Monitoring, Adv. Sens. Res., 2025, 4, 2400138, DOI:10.1002/adsr.202400138.
- P. H. Noordhuis, P. C. Jutte, A. G. P. Kottapalli, C. J. C. Lamoth and C. C. Roossien, Advancements in Biomedical Sensors for Early Detection of Failure in Hip and Knee Implants: Scoping Review on Potential Sensors for Implant Integration, Ann. Biomed. Eng., 2025, 53, 2392–2407 CrossRef PubMed.
- Y. Li, X. Li, D. Guo, L. Meng, X. Feng, Y. Zhang and S. Pan, Immune dysregulation and macrophage polarization in peri-implantitis, Front. Bioeng. Biotechnol., 2024, 12, 1291880 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |