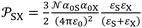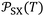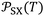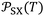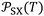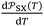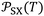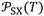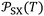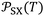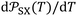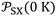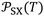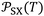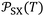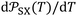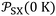Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Effect of temperature dependence of deformation polarizability and ionization energy of solvents on surface properties of solid materials
Tayssir
Hamieh
 *abc
*abc
aFaculty of Science and Engineering, Maastricht University, 6200 MD Maastricht, The Netherlands. E-mail: t.hamieh@maastrichtuniversity.nl
bInstitut de Science des Matériaux de Mulhouse, Université de Haute-Alsace, CNRS, IS2M UMR 7361, F-68100 Mulhouse, France
cLaboratory of Materials, Catalysis, Environment and Analytical Methods (MCEMA), Faculty of Sciences, Lebanese University, P.O. Box 6573/14, Beirut, Lebanon
First published on 13th April 2026
Abstract
In recent studies, an original approach based on the London interaction equation has been proposed for the determination of the dispersive and polar surface properties of solid materials. The reported results revealed significant deviations in surface parameters compared with those obtained using classical methods. However, in these earlier works, the ionization energy and the deformation polarizability of solids and probe molecules were assumed to be temperature-independent. In the present work, the effect of temperature on these two fundamental molecular parameters is investigated, and the influence of their temperature dependence on the surface properties associated with the adsorption of organic solvents on oxide materials such as alumina, titania, and magnesium oxide is examined. Inverse gas chromatography (IGC) at infinite dilution is employed to determine the net retention volumes of probe molecules, enabling the calculation of the free energy of adsorption, as well as its dispersive and polar components, the Lewis acid–base parameters, and the corresponding surface energies. The results demonstrate that thermal variations in ionization energy and deformation polarizability—although relatively small—have a pronounced impact on surface thermodynamic parameters. In particular, changes of up to 100% are observed in the dispersive and polar components of the adsorption free energy for several probe molecules, while variations in the Lewis acid–base constants of solid surfaces reach up to 200% in the case of magnesium oxide. These findings clearly highlight the high sensitivity of surface properties to temperature-dependent molecular parameters and emphasize the necessity of explicitly accounting for thermal effects in the analysis of surface–molecule interactions.
1. Introduction
In a previous study,1 an original method was introduced to determine the surface properties of solid materials by exploiting the London dispersion interaction energy between model organic probe molecules and solid surfaces. This approach enabled a more accurate separation of dispersive and polar contributions to the adsorption free energy, leading to a significant correction of the Lewis acid–base parameters of solid surfaces compared with classical chromatographic models. Such an improved separation is of considerable importance for the reliable prediction of physicochemical surface properties of materials and nanomaterials.1Inverse gas chromatography (IGC) at infinite dilution has been widely employed to quantify dispersive and polar free energies of adsorption through the measurement of retention volumes of probe molecules on solid materials.2–46 In this framework, the standard free energy of adsorption, ΔG0a(T), is determined as a function of temperature. The London-equation-based approach has demonstrated clear superiority over traditional methods relying on empirical or semi-empirical solvent parameters, such as the boiling point TBP,2 vapor pressure P0,3,4 dispersive surface tension γdl,8 topological indices,6,7 standard enthalpy of vaporization ΔH0vap, or deformation polarizability α0,L.5
Several subsequent studies1,8,13–17,26,27,34 have further refined the determination of surface properties—including dispersive surface energy, polar free energy, and Lewis acid–base constants—by incorporating thermal models that account for the temperature dependence of the surface area of organic probe molecules. These developments have confirmed the essential role of temperature in surface thermodynamics.
More recently, the London-equation-based formalism introduced a new thermodynamic parameter, 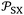 , to describe solid–molecule interactions, defined as:
, to describe solid–molecule interactions, defined as:
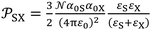 | (1) |
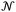 is Avogadro's number, ε0 is the vacuum permittivity, and εS and εX are their corresponding ionization energies. In previous applications of this model, both deformation polarizability and ionization energy were assumed to be independent of temperature.
is Avogadro's number, ε0 is the vacuum permittivity, and εS and εX are their corresponding ionization energies. In previous applications of this model, both deformation polarizability and ionization energy were assumed to be independent of temperature.
In the present work, this framework is extended by explicitly considering the temperature dependence of deformation polarizability and ionization energy for both solids and probe molecules. This refined approach is applied to the determination of surface properties of alumina, titania, and magnesium oxide, examining its impact on the dispersive and polar components of the adsorption free energy, the Lewis acid–base parameters, and the corresponding acid and base surface energies. This study provides a more physically consistent description of surface–molecule interactions and highlights the critical role of thermal effects in surface thermodynamic analyses.
2. Materials and methods
The solid materials and organic solvents were used in previous studies1,16,17 using chromatographic methods and models. The non-polar organic solvents were n-hexane, n-heptane, n-octane, and n-nonane, whereas the polar molecules were dichloromethane, chloroform, carbon tetrachloride, benzene, ethyl acetate, diethyl ether, acetone, tetrahydrofuran (THF), acetone, toluene, and acetonitrile.In the context of inverse gas chromatography, molecular polarity is not solely defined by the presence of a permanent dipole moment. According to the Gutmann donor–acceptor concept, molecules such as benzene, toluene, and carbon tetrachloride—although nonpolar in the classical electrostatic sense—exhibit weak but measurable electron-donor or electron-acceptor character arising from π-electron delocalization or highly polarizable bonds. These molecules are therefore treated as weakly polar probes in IGC analyses, as they are capable of engaging in specific Lewis acid–base interactions with solid surfaces. Table S1 summarizes the corrected Gutmann donor (DN) and acceptor (AN) electron numbers together with the permanent dipole moments of the probe molecules, highlighting that several solvents classified as weakly polar or amphoteric in inverse gas chromatography exhibit specific Lewis acid–base character despite possessing negligible or very small permanent dipole moments.
The solid materials were alumina (Al2O3), magnesium oxide (MgO), and titania (TiO2) and previously characterized.1Table 1 lists for all chemicals the CAS registry number, source of chemicals, and reported purity. The net retention time of organic solvents adsorbed on the different solid surfaces was determined at different temperatures using inverse gas chromatography (IGC) at infinite dilution with the help of a Focus GC gas chromatograph equipped with a flame ionization detector of high sensitivity (Sigma-Aldrich, Paris, France). A mass of 1 g of solid particles was packed into a stainless-steel column of a length of 30 cm and 2 mm internal diameter. Helium was used as carrier gas with a flow rate equal to 25 mL min−1. The retention times of the different injected organic solvents were measured at infinite dilution, supposing that there is no interaction between the probe molecules themselves. The column temperatures varied from 30 to 200 °C. Average retention times and volumes were determined by repeating each solvent injection three times with a standard deviation less than 1% in all chromatographic measurements. Uncertainty propagation was evaluated by considering experimental uncertainties in retention times (1%), temperature control (±0.1 K), and molecular parameters such as deformation polarizability and ionization energy. The resulting uncertainties in derived adsorption energies and acid–base parameters remain within ±5%, indicating that the observed temperature trends are robust and not dominated by experimental error. All reported values represent the mean of repeated measurements, and the associated uncertainties are expressed as standard deviations. The number of significant figures has been adjusted to reflect the experimental precision.
| Chemical | CAS no. | Supplier and location | Reported purity |
|---|---|---|---|
| n-Hexane | 110-54-3 | Aldrich, Paris, France | ≥99% |
| n-Heptane | 142-82-5 | Aldrich, Paris, France | ≥99% |
| n-Octane | 111-65-9 | Aldrich, Paris, France | ≥99% |
| n-Nonane | 111-84-2 | Aldrich, Paris, France | ≥99% |
| Dichloromethane | 75-09-2 | Aldrich, Paris, France | ≥99% |
| Chloroform | 67-66-3 | Aldrich, Paris, France | ≥99% |
| Carbon tetrachloride | 56-23-5 | Aldrich, Paris, France | ≥99% |
| Benzene | 71-43-2 | Aldrich, Paris, France | ≥99% |
| Ethyl acetate | 141-78-6 | Aldrich, Paris, France | ≥99% |
| Diethyl ether | 60-29-7 | Aldrich, Paris, France | ≥99% |
| Acetone | 67-64-1 | Aldrich, Paris, France | ≥99% |
| Tetrahydrofuran (THF) | 109-99-9 | Aldrich, Paris, France | ≥99% |
| Toluene | 108-88-3 | Aldrich, Paris, France | ≥99% |
| Acetonitrile | 75-05-8 | Aldrich, Paris, France | ≥99% |
| α-Alumina (Al2O3) | 1344-28-1 | Aldrich, Paris, France | ≥99% |
| Magnesium oxide (MgO) | 1309-48-4 | Aldrich, Paris, France | ≥99% |
| Titanium dioxide (TiO2) | 13463-67-7 | Aldrich, Paris, France | ≥99% |
The IGC technique2–46 allows the characterization of the surface properties of solid materials through the determination of the net retention volumes of probe molecules adsorbed on the solid surfaces. This approach enables the determination of the free energy of adsorption ΔG0a of the adsorbed molecules by using the following fundamental equation of IGC:
ΔG0a(T) = −RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Vn + C(T) Vn + C(T) | (2) |
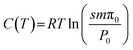 | (3) |
- Kemball and Rideal reference state47 given for T0 = 0 °C by P0 = 1.013 × 105 Pa and π0 = 6.08 × 10−5 N m−1.
- De Boer and Kruyer reference state48 given for T0 = 0 °C by P0 = 1.013 × 105 Pa and π0 = 3.38 × 10−5 N m−1.
The total free energy of adsorption ΔG0a(T) is composed of the respective London dispersive energy ΔGda(T) and polar energy ΔGpa(T):
| ΔG0a(T) = ΔGda(T) + ΔGpa(T) | (4) |
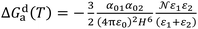 | (5) |
In the case of adsorption of organic solvents on solid materials, the solid molecule (molecule 1) was denoted S and the probe molecule (molecule 2) denoted by X and combining the previous equations. The free energy of adsorption ΔG0a(T) can be written as:
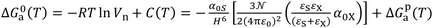 | (6) |
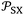 was proposed as new chromatographic indicator variable given by:
was proposed as new chromatographic indicator variable given by:eqn (6) becomes as follows:
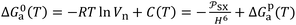 | (7) |
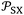 led to quantify both the dispersive and polar contributions of the total free energy of adsorption using n-alkanes and polar solvents. The free energy of n-alkanes gave the dispersive component ΔGda(T) because they only exhibit dispersive interactions, while the distance between the representative point of the polar solvent and the n-alkanes straight-line led to ΔGpa as it was shown in Fig. 1.
led to quantify both the dispersive and polar contributions of the total free energy of adsorption using n-alkanes and polar solvents. The free energy of n-alkanes gave the dispersive component ΔGda(T) because they only exhibit dispersive interactions, while the distance between the representative point of the polar solvent and the n-alkanes straight-line led to ΔGpa as it was shown in Fig. 1.
 | ||
Fig. 1 Variations of the free energy ΔG0a (in kJ mol−1) of adsorption of organic solvents on alumina surfaces as a function of the parameter 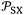 (in 10−54 J Å6) at 323.15 K. (in 10−54 J Å6) at 323.15 K. | ||
Knowing the polar free energy of polar solvents and their total free energy, it was possible to obtain the dispersive free energy of these solvents using eqn (4). On the other hand, the application of eqn (5) to n-alkanes and polar organic molecules allowed to determine the separation distance H between the solvents and solid material.
In previous studies,1,26,27 the ionization energy and deformation polarizability of solid and solvents were supposed constants independent from the temperature. Even if the variations of these variables slightly vary versus the temperature, the temperature effect of these parameters on the surface properties of solid materials was highlighted in this paper.
For clarity and consistency, a list of abbreviations, definitions, and units used in the theoretical and thermodynamic analysis is provided in the SI.
3. Results
The variations of the ionization energy and deformation polarizability of the different compounds used in this work were determined as a function of temperature using several references from the literature.49–64 Recent studies65–70 further illustrate the growing use of electronic-structure descriptors (e.g., adsorption energies, reaction barriers, charge redistribution, and free-energy relationships) to rationalize chemical reactivity and interfacial phenomena. For example, kinetic and mechanistic analyses of gas-phase radical reactions highlight how electronic features and the underlying potential-energy surface govern reaction pathways and reactivity trends.65,66 In parallel, density functional theory (DFT) investigations have been widely employed to resolve mechanistic questions by locating transition states and quantifying activation energies, thereby linking molecular electronic structure to measurable thermodynamic and kinetic observables.65 DFT has also been used to analyze adsorption on carbon-based substrates (e.g., defective graphene), where adsorption-induced changes in electronic properties are central to sensing and interfacial response. Likewise, recent computational studies correlate molecular electronic properties with functional performance, including organic redox/electrode behavior and acid–base descriptors derived from energetic and vibrational signatures.70 Collectively, these contributions emphasize that surface/interfacial properties are governed by electronic quantities and their coupling to thermodynamic functions. In this context, explicitly incorporating the temperature dependence of ionization energy and deformation polarizability provides a physically consistent route to improve adsorption-energy partitioning and Lewis acid–base surface parameters obtained from inverse gas chromatography.The values of the deformation polarizability α0 of solvents and solid materials as a function of temperature were given in Table S2 showing a slight linear increase and satisfying the following equation:
| α0(T) = (dα0/dT)T + α0(0 K) | (8) |
| Compounds | Equation of α0(T) | dα0/dT (×10−3) | α 0 (0 K) |
|---|---|---|---|
| n-Hexane | α 0(T) = 2.9 × 10−3T + 12.378 | 2.9 ± 0.03 | 12.378 ± 0.124 |
| n-Heptane | α 0(T) = 3.5 × 10−3T + 14.112 | 3.5 ± 0.04 | 14.112 ± 0.141 |
| n-Octane | α 0(T) = 4.3 × 10−3T + 16.420 | 4.3 ± 0.04 | 16.420 ± 0.164 |
| n-Nonane | α 0(T) = 4.8 × 10−3T + 17.876 | 4.8 ± 0.05 | 17.876 ± 0.179 |
| CCl4 | α 0(T) = 2.2 × 10−3T + 11.410 | 2.2 ± 0.02 | 11.410 ± 0.114 |
| Nitromethane | α 0(T) = 1.3 × 10−3T + 7.822 | 1.3 ± 0.01 | 7.822 ± 0.078 |
| CH2Cl2 | α 0(T) = 1.2 × 10−3T + 7.660 | 1.2 ± 0.01 | 7.660 ± 0.077 |
| Chloroform | α 0(T) = 1.7 × 10−3T + 9.352 | 1.7 ± 0.02 | 9.352 ± 0.094 |
| Diethyl ether | α 0(T) = 2.1 × 10−3T + 9.900 | 2.1 ± 0.02 | 9.900 ± 0.099 |
| THF | α 0(T) = 1.8 × 10−3T + 8.602 | 1.8 ± 0.02 | 8.602 ± 0.086 |
| Ethyl acetate | α 0(T) = 2.5 × 10−3T + 9.45 | 2.5 ± 0.03 | 9.450 ± 0.095 |
| Acetone | α 0(T) = 1.1 × 10−3T + 6.762 | 1.1 ± 0.01 | 6.762 ± 0.068 |
| Acetonitrile | α 0(T) = 0.7 × 10−3T + 4.72 | 0.7 ± 0.01 | 4.720 ± 0.047 |
| Toluene | α 0(T) = 2.4 × 10−3T + 12.410 | 2.4 ± 0.02 | 12.410 ± 0.124 |
| Benzene | α 0(T) = 2.1 × 10−3T + 10.898 | 2.1 ± 0.02 | 10.898 ± 0.109 |
| Alumina | α 0(T) = 0.3 × 10−3T + 5.86 | 0.3 ± 0.00 | 5.860 ± 0.059 |
| Titania | α 0(T) = 0.7 × 10−3T + 7.71 | 0.7 ± 0.01 | 7.710 ± 0.077 |
| MgO | α 0(T) = 0.3 × 10−3T + 6.23 | 0.3 ± 0.00 | 6.230 ± 0.062 |
The uncertainties associated with the thermal coefficients dα0/dT and deformation polarizability α0(T) were determined by propagation of the standard deviations obtained from inverse gas chromatography (IGC) measurements. Each probe injection was repeated three times, resulting in an overall relative error of less than 1%.
The variations of the ionization energy ε(T) of solvents and solid materials were given in Table S3 as a function of temperature. The obtained results showed very slight variations of ε(T) against the temperature. However, the variations of both α0(T) and ε(T) affect the values of the interaction parameter 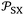 versus the temperature.
versus the temperature.
The new method was based on the temperature effect on the chromatographic parameter 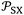 of different solvents respectively adsorbed on alumina, titania, and magnesium oxide.
of different solvents respectively adsorbed on alumina, titania, and magnesium oxide.
Although the International System of Units is used as the reference framework, intermolecular distances are expressed in angstroms for convenience. Accordingly, the parameter 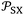 is reported in J Å6 mol−1, ensuring that the ratio
is reported in J Å6 mol−1, ensuring that the ratio 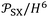 retains the correct unit of adsorption free energy (J mol−1).
retains the correct unit of adsorption free energy (J mol−1).
Table 3 gave the variations of 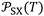 as a function of temperature of the adsorbed organic molecules on solid materials using the values given in previous paper.34 It was observed a slight variation of
as a function of temperature of the adsorbed organic molecules on solid materials using the values given in previous paper.34 It was observed a slight variation of 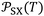 versus the temperature. However, there is an important variation of
versus the temperature. However, there is an important variation of 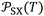 depending on both solvents and solid surfaces. This was more elucidated in Table 4 giving the equations
depending on both solvents and solid surfaces. This was more elucidated in Table 4 giving the equations 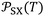 of the different solvents with the extrapolated values of
of the different solvents with the extrapolated values of 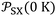 at 0 K.
at 0 K. 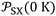 largely varied from solvent to another and from solid to solid. The slope
largely varied from solvent to another and from solid to solid. The slope 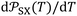 which is equal to the derivative of
which is equal to the derivative of 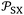 with respect of temperature represents a thermal expansion coefficient. The general equation was given as follows:
with respect of temperature represents a thermal expansion coefficient. The general equation was given as follows:
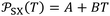 | (9) |
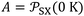 the extrapolated value of
the extrapolated value of 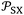 at 0 K and
at 0 K and 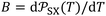 .
.
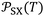 of solvents adsorbed on of the solid materials as a function of temperature. The uncertainty associated with
of solvents adsorbed on of the solid materials as a function of temperature. The uncertainty associated with 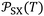 is in the range 0.001–0.012 (×10−54 J Å6 mol−1)
is in the range 0.001–0.012 (×10−54 J Å6 mol−1)
| Parameter (×10−54 J Å6 mol−1) of alumina | ||||
|---|---|---|---|---|
| Temperature (K) | 323.15 | 343.15 | 363.15 | 383.15 |
| n-Hexane | 34.885 | 35.073 | 35.261 | 35.450 |
| n-Heptane | 39.639 | 39.862 | 40.086 | 40.309 |
| n-Octane | 46.084 | 46.354 | 46.624 | 46.896 |
| n-Nonane | 50.093 | 50.393 | 50.693 | 50.993 |
| CCl4 | 33.196 | 33.351 | 33.505 | 33.660 |
| CH2Cl2 | 21.941 | 22.029 | 22.118 | 22.206 |
| Chloroform | 27.036 | 27.157 | 27.278 | 27.399 |
| Ether | 27.060 | 27.196 | 27.332 | 27.468 |
| THF | 23.367 | 23.483 | 23.599 | 23.715 |
| Ethyl acetate | 26.756 | 26.914 | 27.073 | 27.231 |
| Toluene | 32.751 | 32.904 | 33.058 | 33.212 |
| Parameter (×10−54 J Å6 mol−1) of titania | ||||
|---|---|---|---|---|
| Temperature (K) | 323.15 | 343.15 | 363.15 | 383.15 |
| n-Hexane | 60.648 | 61.029 | 61.411 | 61.793 |
| n-Heptane | 68.759 | 69.207 | 69.656 | 70.107 |
| n-Octane | 79.819 | 80.358 | 80.899 | 81.441 |
| n-Nonane | 86.674 | 87.269 | 87.866 | 88.464 |
| CH2Cl2 | 38.621 | 38.810 | 39.000 | 39.190 |
| Chloroform | 47.612 | 47.867 | 48.122 | 48.379 |
| THF | 40.272 | 40.508 | 40.744 | 40.981 |
| Ethyl acetate | 46.454 | 46.770 | 47.087 | 47.405 |
| Acetone | 31.743 | 31.902 | 32.062 | 32.222 |
| Benzene | 50.392 | 50.672 | 50.953 | 51.234 |
| Nitromethane | 39.167 | 39.365 | 39.565 | 39.765 |
| Acetonitrile | 24.545 | 24.661 | 24.778 | 24.895 |
| Parameter (×10−54 J Å6 mol−1) of MgO | ||||
|---|---|---|---|---|
| Temperature (K) | 323.15 | 343.15 | 363.15 | 383.15 |
| n-Hexane | 41.560 | 41.701 | 41.842 | 41.982 |
| n-Heptane | 47.169 | 47.340 | 47.511 | 47.681 |
| n-Octane | 54.795 | 55.007 | 55.219 | 55.429 |
| n-Nonane | 59.530 | 59.767 | 60.004 | 60.240 |
| CH2Cl2 | 26.309 | 26.362 | 26.415 | 26.468 |
| Chloroform | 32.426 | 32.506 | 32.586 | 32.666 |
| Diethyl ether | 32.118 | 32.215 | 32.312 | 32.408 |
| THF | 27.712 | 27.794 | 27.877 | 27.958 |
| Ethyl acetate | 31.854 | 31.979 | 32.103 | 32.227 |
| Acetone | 21.803 | 21.850 | 21.896 | 21.942 |
| Acetonitrile | 16.654 | 16.685 | 16.716 | 16.747 |
| Toluene | 38.700 | 38.804 | 38.908 | 39.012 |
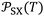 of the different solvents adsorbed on solid materials with the extrapolated values of
of the different solvents adsorbed on solid materials with the extrapolated values of 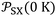 at 0 K and the corresponding slopes
at 0 K and the corresponding slopes 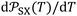 . Values of
. Values of 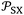 are expressed as ×J Å6 K−1 mol−1 and T in K. The estimated standard deviations are bounded by:
are expressed as ×J Å6 K−1 mol−1 and T in K. The estimated standard deviations are bounded by: 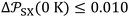 (×10−54 J Å6 mol−1) and
(×10−54 J Å6 mol−1) and 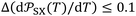 (×J Å6 K−1 mol−1)
(×J Å6 K−1 mol−1)
The uncertainty in 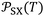 was determined from the propagated uncertainties of the different parameters derived from the experimental data.
was determined from the propagated uncertainties of the different parameters derived from the experimental data.
The determination of polar free energy components of solvents adsorbed on solid surfaces was obtained using eqn (7) and the values of 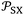 given in Table 3 and S4–S6. The representation of total free energy ΔG0a(T) (n-alkane) (Tables S4-S6) of n-alkanes adsorbed on solid materials as a function of parameter of
given in Table 3 and S4–S6. The representation of total free energy ΔG0a(T) (n-alkane) (Tables S4-S6) of n-alkanes adsorbed on solid materials as a function of parameter of 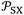 n-alkanes denoted
n-alkanes denoted 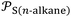 give the n-alkanes-straight-line (Fig. 1) represented by the following equation:
give the n-alkanes-straight-line (Fig. 1) represented by the following equation:
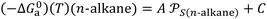 | (10) |
When a polar solvent (X) is adsorbed, it is then characterized by its representative geometric point with two coordinates 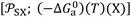 . The difference between the free energy (−ΔG0a)(T)(X) of the polar solvent and that of the fictive point located on the n-alkanes-straight-line having the same abscissa
. The difference between the free energy (−ΔG0a)(T)(X) of the polar solvent and that of the fictive point located on the n-alkanes-straight-line having the same abscissa 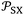 (Fig. 1), gives the corresponding polar free energy (−ΔGpa)(T)(X) of solvent X:
(Fig. 1), gives the corresponding polar free energy (−ΔGpa)(T)(X) of solvent X:
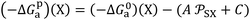 | (11) |
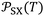 given in Table 3 and those of free energy (−ΔG0a) of adsorption reported in Tables S4–S6, the polar free energy ΔGpa(T) of the adsorbed organic solvents on solid materials were then obtained. The new values of ΔGpa(T) of the various solvents adsorbed on alumina, titania, and MgO were given in Table 5 as a function of temperature.
given in Table 3 and those of free energy (−ΔG0a) of adsorption reported in Tables S4–S6, the polar free energy ΔGpa(T) of the adsorbed organic solvents on solid materials were then obtained. The new values of ΔGpa(T) of the various solvents adsorbed on alumina, titania, and MgO were given in Table 5 as a function of temperature.
| Alumina | ||||
|---|---|---|---|---|
| Temperature (K) | 323.15 | 343.15 | 363.15 | 383.15 |
| CCl4 | 6.848 | 6.591 | 6.442 | 6.291 |
| CH2Cl2 | 38.946 | 36.464 | 34.334 | 31.831 |
| Chloroform | 18.676 | 16.093 | 13.779 | 11.726 |
| Ether | 41.199 | 39.000 | 37.001 | 35.171 |
| THF | 40.653 | 38.187 | 36.030 | 34.111 |
| Ethyl acetate | 43.013 | 40.705 | 38.397 | 36.089 |
| Toluene | 18.913 | 17.415 | 16.269 | 15.598 |
| Titania | ||||
|---|---|---|---|---|
| Temperature (K) | 323.15 | 343.15 | 363.15 | 383.15 |
| CH2Cl2 | 5.965 | 5.575 | 5.287 | 4.801 |
| Chloroform | 2.622 | 1.497 | 0.374 | 0.000 |
| THF | 4.122 | 2.800 | 1.480 | 0.167 |
| Ethyl acetate | 3.193 | 1.611 | 0.032 | 0.000 |
| Acetone | 4.943 | 3.228 | 1.515 | 0.000 |
| Benzene | 0.580 | 0.529 | 0.481 | 0.440 |
| Nitromethane | 9.723 | 8.353 | 6.985 | 5.619 |
| Acetonitrile | 3.610 | 1.506 | 0.000 | 0.000 |
| MgO | ||||
|---|---|---|---|---|
| Temperature (K) | 323.15 | 343.15 | 363.15 | 383.15 |
| CH2Cl2 | 39.945 | 38.903 | 37.861 | 36.819 |
| Chloroform | 10.589 | 10.026 | 9.635 | 9.248 |
| Diethyl ether | 35.873 | 33.635 | 31.689 | 29.375 |
| THF | 21.071 | 18.283 | 15.806 | 13.596 |
| Ethyl acetate | 29.652 | 27.310 | 24.968 | 22.626 |
| Acetone | 46.707 | 44.062 | 41.716 | 39.541 |
| Acetonitrile | 46.573 | 43.625 | 41.094 | 38.803 |
| Toluene | 19.088 | 17.577 | 16.417 | 15.737 |
Table 5 showed that the lowest values of free polar interaction ΔGpa(T) were obtained with the titanium dioxide, whereas MgO gave the highest ΔGpa(T). However, the values of ΔGpa(T) relative alumina are not so far from the those of magnesium oxide.
The determined free energy of adsorption of the polar solvents in Table 5 showed closest values for MgO and alumina very larger than those of titania then proving higher polar interaction for alumina and MgO.
The results showed in Table 5 were compared to those previously obtained without considering the thermal effect on the ionization energy and deformation polarizability.1 It was observed in Table 6 an important deviation between the results of the two methods varying from 7% to 2665% (in the case of THF adsorbed on titania).
| Alumina | ||||
|---|---|---|---|---|
| Temperature (K) | 323.15 | 343.15 | 363.15 | 383.15 |
| CCl4 | 95.1 | 97.5 | 98.7 | — |
| CH2Cl2 | 82.7 | 81.8 | 80.8 | 79.1 |
| Chloroform | 107.8 | 127.7 | 151.6 | 178.1 |
| Ether | 55.0 | 58.4 | 62.1 | 65.0 |
| THF | 1.1 | 2.5 | 3.4 | 4.9 |
| Ethyl acetate | 73.0 | 76.8 | 79.5 | 83.0 |
| Toluene | 114.3 | 120.4 | 123.6 | 123.6 |
| Titania | ||||
|---|---|---|---|---|
| Temperature (K) | 323.15 | 343.15 | 363.15 | 383.15 |
| CH2Cl2 | 57.3 | 65.5 | 76.3 | 84.9 |
| Chloroform | 20.0 | 34.9 | 138.5 | — |
| THF | 84.9 | 136.5 | 279.7 | 2665.2 |
| Ethyl acetate | 24.6 | 50.0 | 2544.8 | — |
| Acetone | 16.9 | 26.0 | 55.9 | — |
| Benzene | 859.4 | 693.7 | 489.8 | 232.6 |
| Nitromethane | 6.9 | 8.0 | 9.6 | 11.8 |
| Acetonitrile | 27.8 | 67.6 | — | — |
| MgO | ||||
|---|---|---|---|---|
| Temperature (K) | 323.15 | 343.15 | 363.15 | 383.15 |
| CH2Cl2 | 91.7 | 90.3 | 88.0 | 85.8 |
| TCM | 44.9 | 73.1 | 83.8 | 76.5 |
| Diethyl ether | 59.8 | 50.8 | 41.1 | 29.5 |
| THF | 9.4 | 36.8 | 70.4 | 111.8 |
| Ethyl acetate | 79.0 | 72.1 | 63.5 | 53.5 |
| Acetone | 66.3 | 53.4 | 39.2 | 23.5 |
Serious consequences resulted from the above results leading to a higher disparity in the values of other surface thermodynamic parameters, particularly on the polar enthalpy and entropy of adsorption, and Lewis acid–base parameters of the solid substrates.
The polar enthalpy (−ΔHpa) and entropy (−ΔSpa) of solvents adsorbed on solid surfaces were obtained from the variations of the free energy of adsorption against the temperature using the following relation:
| ΔGpa(T) = ΔHpa − TΔSpa | (12) |
| Alumina | ||||||
|---|---|---|---|---|---|---|
| Previous results | New results | Error (%) on | Error (%) on | |||
| Solvents | −ΔSpa(J K−1mol−1) | −ΔHpa(kJ mol−1) | −ΔSpa(J K−1mol−1) | −ΔHpa(kJ mol−1) | (−ΔSpa) | (−ΔHpa) |
| CCl4 | 6.2 | 2.314 | 9.1 | 9.7553 | 31.9 | 76.3 |
| CH2Cl2 | 1.9 | 7.3421 | 117.4 | 76.843 | 98.4 | 90.4 |
| CHCl3 | 102.8 | 71.989 | 115.8 | 55.971 | 11.2 | 28.6 |
| Diethyl ether | 104.6 | 52.207 | 100.4 | 73.55 | 4.2 | 29.0 |
| THF | 88.8 | 69.683 | 108.9 | 75.711 | 18.5 | 8.0 |
| Ethyl acetate | 90.4 | 40.683 | 115.4 | 80.305 | 21.7 | 49.3 |
| Toluene | 94.9 | 71.036 | 55.4 | 36.628 | 71.3 | 93.9 |
| Titanium dioxide | ||||||
|---|---|---|---|---|---|---|
| Previous results | New results | Error (%) on | Error (%) on | |||
| Solvents | −ΔSpa(J K−1mol−1) | −ΔHpa(kJ mol−1) | −ΔSpa(J K−1mol−1) | −ΔHpa(kJ mol−1) | (−ΔSpa) | (−ΔHpa) |
| CH2Cl2 | 30.7 | 12.146 | 18.9 | 12.084 | 62.4 | 0.5 |
| CHCl3 | 56.4 | 20.818 | 56.2 | 20.780 | 0.4 | 0.2 |
| THF | 10 | 23.277 | 65.9 | 25.423 | 84.8 | 8.4 |
| Ethyl acetate | 78.1 | 28.448 | 79 | 28.729 | 1.1 | 1.0 |
| Acetone | 85.4 | 32.518 | 85.7 | 32.635 | 0.4 | 0.4 |
| Benzene | 68.3 | 26.965 | 2.3 | 1.335 | 2869.6 | 1920.0 |
| Nitromethane | 68.5 | 31.846 | 68.4 | 31.829 | 0.1 | 0.1 |
| Acetonitrile | 104.6 | 37.37 | 105.1 | 37.586 | 0.5 | 0.6 |
| MgO | ||||||
|---|---|---|---|---|---|---|
| Previous results | New results | Error (%) on | Error (%) on | |||
| Solvents | −ΔSpa(J K−1mol−1) | −ΔHpa(kJ mol−1) | −ΔSpa(J K−1mol−1) | −ΔHpa(kJ mol−1) | (−ΔSpa) | (−ΔHpa) |
| CH2Cl2 | 32.2 | 7.1665 | 52.100 | 56.781 | 38.2 | 87.4 |
| CHCl3 | −60.5 | −24.435 | 22.1 | 17.665 | 373.8 | 238.3 |
| Diethyl ether | 105.1 | 19.543 | 107.2 | 70.503 | 2.0 | 72.3 |
| Ethyl acetate | 71.9 | 17.038 | 124.5 | 61.159 | 42.2 | 72.1 |
| THF | 95.8 | 7.8791 | 117.100 | 67.493 | 18.2 | 88.3 |
| Acetone | 242 | 62.489 | 119.2 | 85.107 | 103.0 | 26.6 |
| Acetonitrile | 81.6 | 2.0138 | 129.2 | 88.148 | 36.8 | 97.7 |
| Toluene | −13.8 | 15.211 | 56.1 | 37.003 | 124.6 | 58.9 |
The results in Table 7 led to the Lewis enthalpic acid–base constants KA and KD using the empirical relation (13):
| −ΔHp = KA × DN + KD × AN | (13) |
The values of KA and KD of solids were deduced by drawing the variations of 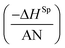 versus
versus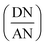 of polar solvents using eqn (14):
of polar solvents using eqn (14):
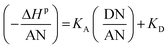 | (14) |
| (−ΔSpa) = ωADN′ + ωDAN′ | (15) |
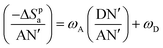 | (16) |
| Previous results | This work | |||||
|---|---|---|---|---|---|---|
| Lewis parameter | Alumina | Titania | MgO | Alumina | Titania | MgO |
| K A | 0.71 | 0.25 | 0.08 | 0.79 | 0.27 | 0.65 |
| K D | 2.21 | 0.87 | 1.13 | 2.69 | 0.89 | 2.37 |
| K D/KA | 3.1 | 3.5 | 14 | 3.41 | 3.26 | 3.65 |
| R 2 | 0.7301 | 0.9874 | 0.1722 | 0.9827 | 0.9895 | 0.9585 |
| 103 × ωA | 0.92 | 0.86 | 1.16 | 1.13 | 0.73 | 1.39 |
| 103 × ωD | 4.21 | 1.8 | 0.57 | 3.92 | 2.03 | 2.00 |
| ω D/ωA | 4.58 | 2.09 | 0.49 | 3.48 | 2.79 | 1.44 |
| R 2 | 0.7739 | 0.9804 | 0.8126 | 0.973 | 0.9885 | 0.9754 |
The results indicate that all three solid materials exhibit an amphoteric character with a predominance of basic behavior. Alumina shows the highest enthalpic and entropic Lewis acid and base constants, followed by MgO, whereas titania presents the lowest Lewis acid and base constants. The Lewis acid–base parameters of MgO and alumina are found to be very close, while titania displays KA and KD values approximately three times lower than those of alumina and MgO. Comparison with previous results1 reveals comparable KA and KD values for alumina and titania, but significantly different values for MgO surfaces. Moreover, the discrepancy between the two methods becomes more pronounced for the entropic acid–base constants ωA and ωD. Overall, the present approach provides a more accurate quantification of the surface properties of solid materials.
4. Discussion
The temperature dependence of the ionization energy and deformation polarizability of solvents adsorbed on alumina, titania, and MgO induces significant variations in the surface thermodynamic properties, particularly in the polar component of the adsorption energy and the Lewis acid–base constants of the solid surfaces. Accordingly, a correction of the surface properties relative to the previous method was performed, clearly highlighting the strong influence of temperature on the thermodynamic parameters governing the Lewis acid–base behavior of these materials.The values of total free energy −ΔG0a(T) of different solvents adsorbed on solid surfaces given in Tables S4–S6 and those of the corresponding polar energy −ΔGpa(T) given in Table 5 led to determine the London dispersive energy of adsorbed solvents as a function of temperature using the following equation:
| ΔGda(T) = ΔG0a(T) − ΔGpa(T) | (17) |
| Alumina | ||||
|---|---|---|---|---|
| Temperature (K) | 323.15 | 343.15 | 363.15 | 383.15 |
| n-Hexane | 28.716 | 28.776 | 28.827 | 28.878 |
| n-Heptane | 31.857 | 31.774 | 31.692 | 31.609 |
| n-Octane | 35.117 | 34.813 | 34.510 | 34.207 |
| n-Nonane | 38.467 | 37.716 | 37.163 | 36.611 |
| CCl4 | 27.666 | 27.858 | 27.993 | 28.129 |
| CH2Cl2 | 20.691 | 21.455 | 22.033 | 22.610 |
| Chloroform | 23.848 | 24.355 | 24.734 | 25.112 |
| Ether | 23.863 | 24.377 | 24.762 | 25.145 |
| THF | 21.575 | 22.277 | 22.808 | 23.338 |
| Ethyl acetate | 23.675 | 24.218 | 24.626 | 25.031 |
| Toluene | 47.776 | 47.508 | 46.754 | 45.523 |
| Titania | ||||
|---|---|---|---|---|
| Temperature (K) | 323.15 | 343.15 | 363.15 | 383.15 |
| n-Hexane | 12.233 | 11.145 | 10.061 | 8.981 |
| n-Heptane | 16.137 | 15.048 | 13.963 | 12.882 |
| n-Octane | 18.889 | 17.739 | 16.593 | 15.451 |
| n-Nonane | 21.792 | 20.513 | 19.239 | 17.968 |
| CH2Cl2 | 4.902 | 3.994 | 3.089 | 2.186 |
| Chloroform | 8.045 | 7.073 | 6.102 | 5.134 |
| THF | 5.479 | 4.571 | 3.665 | 2.761 |
| Ethyl acetate | 7.640 | 6.700 | 5.760 | 4.821 |
| Acetone | 2.497 | 1.646 | 0.797 | |
| Benzene | 9.017 | 8.026 | 7.037 | 6.050 |
| Nitromethane | 5.093 | 4.183 | 3.276 | 2.370 |
| MgO | ||||
|---|---|---|---|---|
| Temperature (K) | 323.15 | 343.15 | 363.15 | 383.15 |
| n-Hexane | 28.716 | 28.776 | 28.827 | 28.878 |
| n-Heptane | 31.857 | 31.774 | 31.692 | 31.609 |
| n-Octane | 35.117 | 34.813 | 34.510 | 34.207 |
| n-Nonane | 38.467 | 37.716 | 37.163 | 36.611 |
| CH2Cl2 | 20.718 | 21.478 | 22.056 | 22.631 |
| Chloroform | 23.925 | 24.424 | 24.800 | 25.172 |
| Diethyl ether | 23.764 | 24.284 | 24.678 | 25.066 |
| THF | 21.453 | 22.165 | 22.706 | 23.242 |
| Ethyl acetate | 23.625 | 24.171 | 24.585 | 24.992 |
| Acetone | 18.355 | 19.315 | 20.047 | 20.775 |
| Acetonitrile | 15.656 | 16.839 | 17.744 | 18.645 |
| Toluene | 27.215 | 27.443 | 27.611 | 27.774 |
The original consequence of this new approach was the determination of the intermolecular distance H(T) between the organic solvents and the solid materials as a function of temperature. Indeed, using eqn (4) and the values of London dispersive free energy −ΔGda(T) of adsorption of solvents on the different solid surfaces given in Table 9 against the temperature, the values of the intermolecular distance H(T) were obtained from the following Equations:
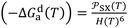 | (18) |
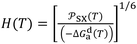 | (19) |
| Alumina | ||||
|---|---|---|---|---|
| Temperature T (K) | 323.15 | 343.15 | 363.15 | 383.15 |
| n-Hexane | 3.267 | 3.268 | 3.270 | 3.272 |
| n-Heptane | 3.280 | 3.284 | 3.289 | 3.293 |
| n-Octane | 3.309 | 3.317 | 3.325 | 3.333 |
| n-Nonane | 3.305 | 3.319 | 3.330 | 3.342 |
| CCl4 | 3.260 | 3.259 | 3.258 | 3.258 |
| CH2Cl2 | 3.193 | 3.176 | 3.164 | 3.153 |
| Chloroform | 3.229 | 3.220 | 3.214 | 3.209 |
| Ether | 3.229 | 3.220 | 3.215 | 3.209 |
| THF | 3.205 | 3.190 | 3.180 | 3.171 |
| Ethyl acetate | 3.227 | 3.218 | 3.213 | 3.207 |
| Toluene | 2.871 | 2.877 | 2.887 | 2.903 |
| Titania | ||||
|---|---|---|---|---|
| Temperature T (K) | 323.15 | 343.15 | 363.15 | 383.15 |
| n-Hexane | 4.129 | 4.198 | 4.275 | 4.361 |
| n-Heptane | 4.026 | 4.078 | 4.134 | 4.194 |
| n-Octane | 4.021 | 4.068 | 4.118 | 4.172 |
| n-Nonane | 3.980 | 4.025 | 4.073 | 4.125 |
| CH2Cl2 | 4.461 | 4.619 | 4.825 | 5.116 |
| Chloroform | 4.253 | 4.349 | 4.461 | 4.596 |
| THF | 4.409 | 4.549 | 4.724 | 4.957 |
| Ethyl acetate | 4.272 | 4.372 | 4.488 | 4.629 |
| Acetone | 4.831 | 5.183 | 5.853 | — |
| Benzene | 4.213 | 4.299 | 4.398 | 4.515 |
| Nitromethane | 4.443 | 4.595 | 4.790 | 5.060 |
| MgO | ||||
|---|---|---|---|---|
| Temperature T (K) | 323.15 | 343.15 | 363.15 | 383.15 |
| n-Hexane | 3.363 | 3.364 | 3.365 | 3.366 |
| n-Heptane | 3.376 | 3.380 | 3.383 | 3.387 |
| n-Octane | 3.406 | 3.413 | 3.420 | 3.427 |
| n-Nonane | 3.401 | 3.414 | 3.425 | 3.436 |
| CH2Cl2 | 3.291 | 3.246 | 3.068 | 3.066 |
| Chloroform | 3.327 | 3.308 | 3.133 | 3.135 |
| Diethyl ether | 3.325 | 3.272 | 2.882 | 2.900 |
| THF | 3.300 | 3.226 | 2.996 | 3.020 |
| Ethyl acetate | 3.324 | 3.263 | 2.802 | 2.817 |
| Acetone | 3.254 | 3.167 | 2.660 | 2.672 |
| Acetonitrile | 3.195 | 3.085 | 2.564 | 2.575 |
| Toluene | 3.353 | 3.327 | 3.098 | 3.105 |
The variations of H(T) between the solvents and the solid substrates reported in Table 10 highlight a clear temperature effect on the intermolecular distance. A linear increase of H(T) with increasing temperature is observed for n-alkanes, whereas a decrease of H(T) is found for polar solvents. The results in Table 10 also reveal significant differences in intermolecular distances that strongly depend on the polarity and surface characteristics of the solid materials. In particular, the lowest H(T) values are obtained for alumina, followed by MgO, while the highest values are observed for titania. This trend is consistent with the Lewis acid–base properties of the solid surfaces, as alumina exhibits the highest acid–base constants, leading to shorter intermolecular distances due to stronger van der Waals and specific interactions.
The temperature dependence of deformation polarizability reflects the progressive softening of the electronic cloud under thermal excitation, leading to enhanced electronic deformation at the solid–molecule interface. Simultaneously, the decrease in ionization energy with temperature indicates a reduction of the electronic potential barrier, facilitating charge displacement and polarization. Together, these effects amplify dispersive and polarization-induced interactions, thereby modifying surface energy, adsorption strength, and Lewis acid–base characteristics of oxide materials.
5. Conclusions
The surface properties of oxide materials such as alumina, titania, and magnesium oxide were determined using a refined approach that explicitly accounts for the temperature dependence of the ionization energy and deformation polarizability of probe molecules, and consequently of the surface thermodynamic parameters of solid materials. Although only slight variations in ionization energy and deformation polarizability of organic solvents were observed with temperature, these changes resulted in significant differences in the calculated surface properties of the oxides. In particular, marked discrepancies were found in the Lewis acid–base constants obtained using the present thermal method compared with those derived from the previous approach, which neglected temperature effects on the dispersive and polar components of the adsorption energy.The substantial differences observed in the intermolecular distances between solvents and the various solid substrates further confirm the superiority of the proposed method, as they reflect a more physically consistent description of solid–molecule interactions.
Overall, these findings highlight the critical importance of incorporating temperature-dependent electronic and polarizability effects when evaluating surface reactivity, adhesion, and interfacial interactions. From a broader materials science perspective, this work establishes a fundamental link between molecular-scale properties of probe molecules and macroscopic surface behavior, providing new insights for the rational design of functional materials, surface coatings, and nanostructured interfaces with tailored thermodynamic and interfacial properties.
Conflicts of interest
The author declares no conflicts of interest.Data availability
The data presented in this study are available in the article.Supplementary information (SI): the data supporting the findings of this study are provided within the article and its SI. The SI contains detailed physicochemical datasets and parameters used throughout the analysis. Specifically, Table S1 reports the corrected donor (DN′) and acceptor (AN′) numbers, along with dipole moments (μ), of the investigated organic solvents, characterizing their Lewis acid–base properties. Table S2 presents the temperature-dependent deformation polarizability of both solvents and solid materials, while Table S3 provides the corresponding temperature-dependent ionization energies. Tables S4–S6 compile the temperature-dependent standard Gibbs free energies of adsorption for solvents on alumina, titania, and MgO surfaces, respectively, as determined by inverse gas chromatography. These datasets form the basis for the thermodynamic and molecular interaction analyses presented in the manuscript. In addition, a comprehensive list of abbreviations and symbols is provided to ensure clarity and reproducibility of the methodology, including definitions of all thermodynamic, molecular, and surface parameters employed in the study. See DOI: https://doi.org/10.1039/d6lf00006a.
References
- T. Hamieh, New Progress on London Dispersive Energy, Polar Surface Interactions, and Lewis's Acid–Base Properties of Solid Surfaces, Molecules, 2024, 29, 949, DOI:10.3390/molecules29050949.
- D. T. Sawyer and D. J. Brookman, Thermodynamically based gas chromatographic retention index for organic molecules using salt-modified aluminas and porous silica beads, Anal. Chem., 1968, 40, 1847–1850, DOI:10.1021/ac60268a015.
- C. Saint-Flour and E. Papirer, Gas-solid chromatography. A method of measuring surface free energy characteristics of short carbon fibers. 1. Through adsorption isotherms, Ind. Eng. Chem. Prod. Res. Dev., 1982, 21, 337–341, DOI:10.1021/i300006a029.
- C. Saint-Flour and E. Papirer, Gas-solid chromatography: Method of measuring surface free energy characteristics of short fibers. 2. Through retention volumes measured near zero surface coverage, Ind. Eng. Chem. Prod. Res. Dev., 1982, 21, 666–669, DOI:10.1021/i300008a031.
- J. B. Donnet, S. J. Park and H. Balard, Evaluation of specific interactions of solid surfaces by inverse gas chromatography, Chromatographia, 1991, 31, 434–440 CrossRef CAS.
- E. Brendlé and E. Papirer, A new topological index for molecular probes used in inverse gas chromatography for the surface nanorugosity evaluation, 2. Application for the Evaluation of the Solid Surface Specific Interaction Potential, J. Colloid Interface Sci., 1997, 194, 217–224 CrossRef PubMed.
- E. Brendlé and E. Papirer, A new topological index for molecular probes used in inverse gas chromatography for the surface nanorugosity evaluation, 1. Method of Evaluation, J. Colloid Interface Sci., 1997, 194, 207–216 CrossRef.
- T. Hamieh and J. Schultz, New approach to characterise physicochemical properties of solid substrates by inverse gas chromatography at infinite dilution. I. II. And III, J. Chromatogr. A, 2002, 969, 17–47, DOI:10.1016/S0021-9673(02)00368-0.
- J. B. Donnet, E. Custodéro, T. K. Wang and G. Hennebert, Energy site distribution of carbon black surfaces by inverse gas chromatography at finite concentration conditions, Carbon, 2002, 40, 163–167, DOI:10.1016/S0008-6223(01)00168-3.
- E. Papirer, E. Brendlé, F. Ozil and H. Balard, Comparison of the surface properties of graphite, carbon black and fullerene samples, measured by inverse gas chromatography, Carbon, 1999, 37, 1265–1274, DOI:10.1016/S0008-6223(98)00323-6.
- J. F. Gamble, R. N. Davé, S. Kiang, M. M. Leane, M. Tobyn and S. S. Y. Wang, Investigating the applicability of inverse gas chromatography to binary powdered systems: An application of surface heterogeneity profiles to understanding preferential probe-surface interactions, Int. J. Pharm., 2013, 445, 39–46 CrossRef CAS PubMed.
- H. Balard, D. Maafa, A. Santini and J. B. Donnet, Study by inverse gas chromatography of the surface properties of milled graphites, J. Chromatogr. A, 2008, 1198–1199, 173–180 CrossRef CAS PubMed.
- T. Hamieh, Study of the temperature effect on the surface area of model organic molecules, the dispersive surface energy and the surface properties of solids by inverse gas chromatography, J. Chromatogr. A, 2020, 1627, 461372 CrossRef CAS PubMed.
- T. Hamieh, A. A. Ahmad, T. Roques-Carmes and J. Toufaily, New approach to determine the surface and interface thermodynamic properties of H-β-zeolite/rhodium catalysts by inverse gas chromatography at infinite dilution, Sci. Rep., 2020, 10, 20894 CrossRef CAS.
- T. Hamieh, New methodology to study the dispersive component of the surface energy and acid–base properties of silica particles by inverse gas chromatography at infinite dilution, J. Chromatogr. Sci., 2022, 60, 126–142, DOI:10.1093/chromsci/bmab066.
- T. Hamieh, Some Irregularities in the Evaluation of Surface Parameters of Solid Materials by Inverse Gas Chromatography, Langmuir, 2023, 39, 17059–17070, DOI:10.1021/acs.langmuir.3c01649.
- T. Hamieh, Inverse Gas Chromatography to Characterize the Surface Properties of Solid Materials, Chem. Mater., 2024, 36(5), 2231–2244, DOI:10.1021/acs.chemmater.3c03091.
- A. Voelkel, B. Strzemiecka, K. Adamska and K. Milczewska, Inverse gas chromatography as a source of physiochemical data, J. Chromatogr. A, 2009, 1216, 1551 CrossRef CAS PubMed.
- Z. Y. Al-Saigh and P. Munk, Study of polymer-polymer interaction coefficients in polymer blends using inverse gas chromatography, Macromolecules, 1984, 17, 803 CrossRef CAS.
- S. K. Papadopoulou and C. Panayiotou, Thermodynamic characterization of poly(1,1,1,3,3,3-hexafluoroisopropyl methacrylate) by inverse gas chromatography, J. Chromatogr. A, 2012, 1229, 230 CrossRef CAS.
- P. Coimbra, M. S. N. Coelho and J. A. F. Gamelas, Surface characterization of polysaccharide scaffolds by inverse gas chromatography regarding application in tissue engineering, Surf. Interface Anal., 2019, 51(11), 1070–1077 CrossRef CAS.
- J. Kołodziejek, A. Voelkel and K. Heberger, Characterization of hybrid materials by means of inverse gas chromatography and chemometrics, J. Pharm. Sci., 2013, 102, 1524 CrossRef.
- H. M. Ryan, J. G. Douglas and W. Rupert, Inverse Gas Chromatography for Determining the Dispersive Surface Free Energy and Acid–Base Interactions of Sheet Molding Compound-Part II 14 Ligno-Cellulosic Fiber Types for Possible Composite Reinforcement, J. Appl. Polym. Sci., 2008, 110, 3880–3888 CrossRef.
- P. N. Jacob and J. C. Berg, Acid-base surface energy characterization of microcrystalline cellulose and two wood pulp fiber types using inverse gas chromatography, Langmuir, 1994, 10, 3086–3093 CrossRef CAS.
- M. G. Carvalho, J. M. R. C. A. Santos, A. A. Martins and M. M. Figueiredo, The Effects of Beating, Web Forming and Sizing on the Surface Energy of Eucalyptus globulus Kraft Fibres Evaluated by Inverse Gas Chromatography, Cellulose, 2005, 12, 371–383 CrossRef CAS.
- T. Hamieh, The Effect of Temperature on the Surface Energetic Properties of Carbon Fibers Using Inverse Gas Chromatography, Crystals, 2024, 14, 28, DOI:10.3390/cryst14010028.
- T. Hamieh, London Dispersive and Lewis Acid-Base Surface Energy of 2D Single-Crystalline and Polycrystalline Covalent Organic Frameworks, Crystals, 2024, 14, 148, DOI:10.3390/cryst14020148.
- H. Chtourou, B. Riedl and B. V. Kokta, Surface characterizations of modified polyethylene pulp and wood pulps fibers using XPS and inverse gas chromatography, J. Adhes. Sci. Technol., 1995, 9, 551–574 CrossRef CAS.
- V. I. Bogillo, V. P. Shkilev and A. Voelkel, Determination of surface free energy components for heterogeneous solids by means of inverse gas chromatography at finite concentrations, J. Mater. Chem., 1998, 8, 1953–1961 RSC.
- S. C. Das, Q. Zhou, D. A. V. Morton, I. Larson and P. J. Stewart, Use of surface energy distributions by inverse gas chromatography to understand mechanofusion processing and functionality of lactose coated with magnesium stearate, Eur. J. Pharm. Sci., 2011, 43, 325–333 CrossRef CAS PubMed.
- S. C. Das and P. J. Stewart, Characterising surface energy of pharmaceutical powders by inverse gas chromatography at finite dilution, J. Pharm. Pharmacol., 2012, 64, 1337–1348 CrossRef CAS.
- W. Bai, E. Pakdel, Q. Li, J. Wang, W. Tang, B. Tang and X. Wang, Inverse gas chromatography (IGC) for studying the cellulosic materials surface characteristics: A mini review, Cellulose, 2023, 30, 3379–3396, DOI:10.1007/s10570-023-05116-9.
- S. Dong, M. Brendlé and J. B. Donnet, Study of solid surface polarity by inverse gas chromatography at infinite dilution, Chromatographia, 1989, 28, 469–472 CrossRef CAS.
- T. Hamieh, Temperature Dependence of the Polar and Lewis Acid–Base Properties of Poly Methyl Methacrylate Adsorbed on Silica via Inverse Gas Chromatography, Molecules, 2024, 29, 1688, DOI:10.3390/molecules29081688.
- J. F. Gamble, M. Leane, D. Olusanmi, M. Tobyn, E. Supuk, J. Khoo and M. Naderi, Surface energy analysis as a tool to probe the surface energy characteristics of micronized materials—A comparison with inverse gas chromatography, Int. J. Pharm., 2012, 422, 238–244 CrossRef CAS PubMed.
- H. E. Newell, G. Buckton, D. A. Butler, F. Thielmann and D. R. Williams, The use of inverse gas chromatography to measure the surface energy of crystalline, amorphous, and recently milled lactose, Pharm. Res., 2001, 18, 662–666 CrossRef CAS.
- H. E. Newell and G. Buckton, Inverse gas chromatography: Investigating whether the technique preferentially probes high energy sites for mixtures of crystalline and amorphous lactose, Pharm. Res., 2004, 21, 1440–1444 CrossRef CAS.
- R. Ho, S. J. Hinder, J. F. Watts, S. E. Dilworth, D. R. Williams and J. Y. Y. Heng, Determination of surface heterogeneity of D-mannitol by sessile drop contact angle and finite concentration inverse gas chromatography, Int. J. Pharm., 2010, 387, 79–86 CrossRef CAS PubMed.
- R. Calvet, S. Del Confetto, H. Balard, E. Brendlé and J. B. Donnet, Study of the interaction polybutadiene/fillers using inverse gas chromatography, J. Chromatogr. A, 2012, 1253, 164–170 CrossRef CAS PubMed.
- S. K. Papadopoulou, G. Dritsas, I. Karapanagiotis, I. Zuburtikudis and C. Panayiotou, Surface characterization of poly(2,2,3,3,3-pentafluoropropyl methacrylate) by inverse gas chromatography and contact angle measurements Eur, Polym. J., 2010, 46, 202–208 CAS.
- G. S. Dritsas, K. Karatasos and C. Panayiotou, Investigation of thermodynamic properties of hyperbranched poly(ester amide) by inverse gas chromatography, J. Polym. Sci. Polym. Phys., 2008, 46, 2166–2172 CrossRef CAS.
- D. L. Chung, Carbon Fiber Composites, Butterworth-Heinemann, Boston, MA, USA, 1994, pp. 3–65, ISBN: 978-0-08-050073-7, DOI:10.1016/C2009-0-26078-8.
- J. B. Donnet and R. C. Bansal, Carbon Fibers, Marcel Dekker, New York, NY, USA, 2nd edn, 1990, p. 584, DOI:10.1201/9781482285390.
- Y. Liu, Y. Gu, S. Wang and M. Li, Optimization for testing conditions of inverse gas chromatography and surface energies of various carbon fiber bundles, Carbon Lett., 2023, 33, 909–920, DOI:10.1007/s42823-023-00472-9.
- A. Pal, A. Kondor, S. Mitra, K. Thua, S. Harish and B. B. Saha, On surface energy and acid–base properties of highly porous parent and surface treated activated carbons using inverse gas chromatography, J. Ind. Eng. Chem., 2019, 69, 432–443, DOI:10.1016/j.jiec.2018.09.046.
- P. K. Basivi, T. Hamieh, V. Kakani, V. R. Pasupuleti, G. Sasikala, S. M. Heo, K. S. Pasupuleti, M.-D. Kim, V. S. Munagapati, N. S. Kumar, J.-H. Wen and C. W. Kim, Exploring advanced materials: Harnessing the synergy of inverse gas chromatography and artificial vision intelligence, TrAC, Trends Anal. Chem., 2024, 173, 117655, DOI:10.1016/j.trac.2024.117655.
- C. Kemball and E. K. Rideal, The adsorption of vapours on mercury I) Non –polar substances, Proc. R. Soc. London, Ser. A, 1946, 187, 53–73 CAS.
- J. H. De Boer and S. Kruyer, Entropy and mobility of adsorbed molecules I) Procedure; atomic gases on charcoal, Proc. K. Ned. Akad. Wet., Ser. B, 1952, 55, 451–463 Search PubMed.
- M. Laib and D. M. Mittleman, Terahertz spectroscopy of liquid n-alkanes: Temperature dependence, J. Infrared, Millimeter, Terahertz Waves, 2010, 31(9), 1015–1024 CrossRef.
- Semiconductors: Data Handbook, ed. O. Madelung, Springer, 3rd edn, 2004 Search PubMed.
- J. Barthel and R. Buchner, Dielectric spectroscopy of ethers over temperature, J. Solution Chem., 1995, 24(12), 1311–1327 Search PubMed.
- R. Buchner, G. Hefter and J. Barthel, Dielectric relaxation of polar aprotic solvents: Temperature and frequency dependence, J. Phys. Chem. A, 1999, 103(1), 1–9 CrossRef CAS.
- J. Barthel, R. Buchner and M. Münsterer, Static and dynamic dielectric permittivity of acetonitrile, Chem. Phys. Lett., 1995, 240(1–3), 193–200 Search PubMed.
- J. Barthel and R. Buchner, Dielectric properties of aromatic hydrocarbons versus temperature, J. Mol. Liq., 1995, 67(1), 1–12 Search PubMed.
- A. S. Barker and A. J. Sievers, Optical studies of Al2O3 dielectric properties vs temperature, Rev. Mod. Phys., 1975, 47(2), S1–S179 CrossRef.
- J. H. Parker, Dielectric response of TiO2 as a function of temperature, Phys. Rev., 1967, 155(3), 712–714 CrossRef CAS.
- R. A. Marcus, Electrostatic free energy and other properties of states having nonequilibrium polarization, J. Chem. Phys., 1956, 24(5), 979–989, DOI:10.1063/1.1742723.
- J. K. Parker and C. E. Brion, Absolute photoabsorption cross-sections and oscillator strengths for valence-shell transitions in acetone and ethanol, Chem. Phys., 1978, 31(2), 317–333, DOI:10.1016/0301-0104(78)85118-1.
- T. W. Bentley and P. v. R. Schleyer, The Grunwald–Winstein equation, Adv. Phys. Org. Chem., 1977, 14, 1–67, DOI:10.1016/S0065-3160(08)60294-2.
- B. K. P. Scaife and A. W. Lyons, Dielectric permittivity and pVT data of some n-alkanes, Proc. R. Soc. London, Ser. A, 1980, 370, 193–212 Search PubMed.
- B. K. P. Scaife and A. W. Lyons, Density and dielectric polarizability of n-alkane liquids, Ber. Bunsenges. Phys. Chem., 1990, 94, 758–765 CrossRef.
- U. Kaatze, Complex permittivity of nitromethane as function of temperature and frequency, J. Chem. Phys., 1989, 90, 3071–3077 CrossRef.
- R. Buchner and J. Barthel, Dielectric relaxation of halogenated methanes as function of temperature, J. Mol. Liq., 1995, 63, 21–39 CrossRef.
- J. Barthel and R. Buchner, Dielectric spectroscopy of ethers over temperature, J. Solution Chem., 1995, 24, 1311–1327 Search PubMed.
- P. Issofa, E. Njabon, S. Njankwa, T. Tchamba and A. Emadak, Kinetic and mechanistic insights into the atmospheric hydrogen abstraction of 3-Hydroxybutanal by chlorine, Chem. Rev. Lett., 2026, 11–16, DOI:10.22034/crl.2025.532470.1648.
- R. Behjatmanesh-Ardakani and H. Imanov, DFT study on the mechanism of benzimidazole synthesis from phenylenediamine and formic acid: Activation energies and transition states' locations, Chem. Rev. Lett., 2025, 8(6), 1188–1199, DOI:10.22034/crl.2025.537649.1665.
- M. Haqgu, Z. Rostami and A. Mengboev, DFT investigation of aflatoxin B1 adsorption on vacancy-defective graphene: electronic properties and sensing potential, Chem. Rev. Lett., 2025, 8(6), 1258–1268, DOI:10.22034/crl.2025.544347.1683.
- R. Omer, A. Anwar, Y. Hussein, H. K. I. Sultan, H. K. Ismail, A. F. Hamasdiq and R. Kareem, Obaid Synthesis and DFT-guided evaluation of PPy-ZnFe2O4@Fe3O4 nanocomposite for pharmaceutical adsorption, J. Chem. Lett., 2026, 240–255, DOI:10.22034/jchemlett.2025.554966.1361.
- M. A. Rafiee and M. Javaheri, Theoretical Study of Benzoquinone Derivatives as Organic Cathode Materials in Lithium-Ion Batteries, J. Chem. Lett., 2025, 6(3), 166–174, DOI:10.22034/jchemlett.2025.516569.1295.
- A.-K. Kabi, DFT-Based Insights into Carboxylic Acid Acidity: Correlating pKa with Free Energy and Vibrational Signatures, J. Chem. Lett., 2025, 6(3), 212–222, DOI:10.22034/jchemlett.2025.543299.1340.
| This journal is © The Royal Society of Chemistry 2026 |