Open Access Article
Open Access ArticleSolar light driven type-II heterojunction TiO2@ZIF-8 nanocomposite for sustainable chlorpyrifos detoxification: physicochemical insights, mineralization pathways and antibacterial performance
Subhashree Mohanty ,
Bibeka Nanda Marai and
Sushanta Kumar Badamali
,
Bibeka Nanda Marai and
Sushanta Kumar Badamali *
*
Department of Chemistry, Utkal University, Vani Vihar, Bhubaneswar-751004, Odisha, India. E-mail: skbuche@utkaluniversity.ac.in
First published on 8th April 2026
Abstract
Chlorpyrifos (CP), a persistent organophosphate pesticide, poses significant environmental and health risks, which necessitate sustainable detoxification strategies. TiO2@ZIF-8 nanocomposite was prepared by a combined hydrothermal–ultrasonication approach and characterized using XRD, FTIR spectroscopy, Raman, XPS, FE-SEM, HR-TEM, BET, DRS, and DLS analyses and TGA to gain insights into its structural characteristics. Moreover, the interfacial charge separation was further confirmed through Mott–Schottky and EIS measurements. Photocatalytic degradation studies showed that ∼90% of CP (100 mg L−1) was removed within 90 min under solar light using 0.6 g L−1 of the catalyst. RSM–CCD optimisation resulted in 86% CP degradation at 90 mg L−1 of CP, 20 mg of adsorbent, and 86 min of solar light. A band energy diagram was constructed based on Tauc plot and Mott–Schottky analyses to determine the VB and CB positions of ZIF-8, TiO2, and TiO2@ZIF-8, indicating the probable formation of a type-II heterojunction, which facilitated efficient charge-carrier separation, broadened the light-absorption range, and suppressed electron–hole recombination. The kinetic analysis revealed that the degradation process followed pseudo-first-order kinetics. Radical scavenging study confirmed that holes, superoxide radicals, and hydroxyl radicals were the preferentially active reactive species in the system. A plausible degradation mechanism was proposed accordingly. The composite exhibited excellent reusability by maintaining ∼85% activity over four cycles. The nanocomposite demonstrated significant antibacterial efficacy against Staphylococcus aureus and Escherichia coli, suggesting multifunctional applicability in water purification. These results demonstrate that the TiO2@ZIF-8 nanocomposite possesses both photocatalytic and antibacterial capabilities independently, enabling its potential for multifunctional applications in the future.
1. Introduction
The rapid expansion of global industrialization and agricultural sectors has raised significant concerns regarding environmental pollution. It has not only intensified the volume of pollutants entering ecosystems but also diversified their chemical complexity by deteriorating the water quality and public health. Among such pollutants, pesticides pose a critical threat due to their pervasive use, environmental persistence, and neurotoxic effects on ecosystems. Agricultural runoff, industrial effluents, and improper waste disposal have further dispersed these hazardous chemicals into water systems. Consequently, ensuring access to safe drinking water has become increasingly challenging, as the economic and technical complexities of purifying such contaminated water remain unresolved.1Among the myriads of pesticides, organophosphorus pesticides (OPPs) particularly pose severe ecological and public health risks arising from their persistence, bioaccumulation potential, and carcinogenic properties. Among them, chlorpyrifos (CP), a widely used insecticide, exemplifies this crisis. Initially patented in 1966, CP has been extensively applied in agriculture, urban pest control, and household settings.2 The detectable residues of CP are found in 89% of global surface water samples, with contaminations found up to 24 km from the application sites.2 Its half-life in aquatic environments ranges from 30 to 120 days, allowing bioaccumulation in aquatic organisms and biomagnification through food chains. It is linked to oxidative DNA damage, chronic poisoning, acute developmental disorders, and endocrine disruption in humans, even at trace concentrations. Despite its efficacy against pests, CP is classified by the WHO as a class II hazardous chemical.3 Its residues are frequently detected in surface waters worldwide, and the metabolites, such as chlorpyrifos-oxon and 3,5,6-trichloro-2-pyridinol (TCP), are more toxic and tenacious. In addition to these, several risks associated with CP and its contaminated aquatic sources have become an urgent priority to develop efficient and sustainable technologies to protect the environment.4,5
Various methods, including oxidative degradation, electrochemical treatment, adsorption, and photodegradation, have been explored for removing pollutants from aqueous solutions. However, conventional treatment methods, such as adsorption and phase-transfer processes, often fail to achieve complete degradation as they merely relocate contaminants rather than eliminating them.6 Among these, photocatalytic degradation stands out as a mostly favourable approach owing to its simplicity, rapid degradation rates, and ability to mineralize pollutants. This process utilizes UV-visible light or sunlight to activate a semiconductor photocatalyst, generating highly reactive hydroxyl radicals (OH·) and superoxide radicals that mineralize organic contaminants into harmless by-products, eventually producing CO2 and H2O.7 Among semiconductor photocatalysts, TiO2 remains the benchmark material because of its unique combination of physicochemical and electronic properties. Compared with other semiconductors, such as ZnO, WO3, and CdS, TiO2 offers enhanced light absorption, superior stability, non-toxicity, and resistance to photo corrosion, making it a relatively safe alternative for environmental applications. The anatase phase is particularly favorable for photocatalytic oxidation, as its conduction band potential (∼−0.5 V vs. NHE) is more negative than the O2/·O2− redox potential (−0.33 V), which facilitates efficient oxygen reduction. In addition, its highly positive valence band (+2.7 V) possesses strong oxidizing ability, facilitating the formation of hydroxyl radicals.8 Pristine TiO2 has a wide gap of about 3.2 eV in the anatase phase, and the absorption edge falls within the UV range, which limits visible absorption. However, it favors the rapid recombination of electron and hole pairs, which reduces the photocatalytic activity. Therefore, rational modifications, such as nanocomposite engineering, surface functionalization, and coupling with porous supports, including MOFs, are being actively explored to enhance visible-light activity, suppress recombination, and maximize their photocatalytic performance in real environmental systems.
Metal–organic frameworks (MOFs) have been developed as a group of porous materials with exceptional versatility in environmental and energy applications, particularly in adsorption and photodegradation processes. Among them, ZIF-8 (zeolitic imidazole framework-8) exhibits unique features, including a high surface area, strong chemical stability, and a sodalite-type nanoporous architecture, which enables the selective adsorption of contaminants, such as pesticides, dyes, pharmaceuticals and heavy metals.9 More importantly, when combined with semiconductors, such as TiO2, ZIF-8 acts as an ideal support, preventing nanoparticle agglomeration, enhancing reactant diffusion, and facilitating charge separation. Its semiconductor-like behavior and ability to form nanocomposites also enhance visible-light absorption and inhibit electron–hole recombination, thereby significantly boosting photocatalytic activity. These attributes make TiO2/ZIF-8 heterostructures highly promising for the sustainable degradation of persistent pollutants and water purification.10–13
Most of the reported studies on the removal of chlorpyrifos (CP) have explored a variety of degradation processes. For instance, F. Zisti et al. demonstrated the use of a Fe3O4@SiO2/PAEDTC@MIL-101 catalyst under UV light, achieving nearly 99% degradation of 25 mg L−1 of CP.14 Hakimeh Teymourinia et al. reported g-C3N5/CdS dendrite/AgNPs nanocomposite, which degrades 95.3% of CP under visible light in 60 min.15 Similarly, Y. Tian et al. reported a CuFe2O4/MIL-101(Fe) catalyst capable of degrading CP with 95% efficiency under a 300 W xenon lamp.16 Xin Liu et al. have degraded ∼94% of CP using bifunctional composite MOF525@BDMTp under UV-vis light for 4 h.17 Although these results are promising, they typically require artificial light sources or complex catalyst formulation, which limit their ecological sustainability, scalability, and cost-effectiveness. The key objectives of sustainable chemistry emphasis the minimization of atom usage, cost-effective material selection, and the design of reusable heterogeneous catalysts. To address these, we design a TiO2@ZIF-8 heterostructure by uniformly dispersing anatase TiO2 nanoparticles onto the surface of ZIF-8 through a hydrothermal–ultrasonication method. The integration of TiO2 with ZIF-8 fulfils the required criteria, providing cost effectiveness, ease of operation, high efficiency, recyclability, heterogeneous nature and functionality under sustainable sunlight. The surface architecture and morphology of the nanocomposite are systematically characterized and discussed in detail. The photocatalytic performance is further optimized using response surface methodology (RSM) and central composite design (CCD), which identify the most efficient conditions for CP degradation. The stability of the catalyst and the heterogeneous nature of the reaction are studied, and a plausible degradation mechanism of CP is elucidated based on GC-MS analysis. Furthermore, electrochemical analyses are employed to determine the type of heterojunction and to assess the enhanced charge-transfer dynamics, along with the migration behaviour of charge carriers. Alongside pollutant removal, the nanocomposite demonstrates significant antibacterial effects against E. coli and S. aureus, highlighting its multifunctional applicability in water detoxification and microbial control.
2. Experimental
2.1 Chemicals and reagents
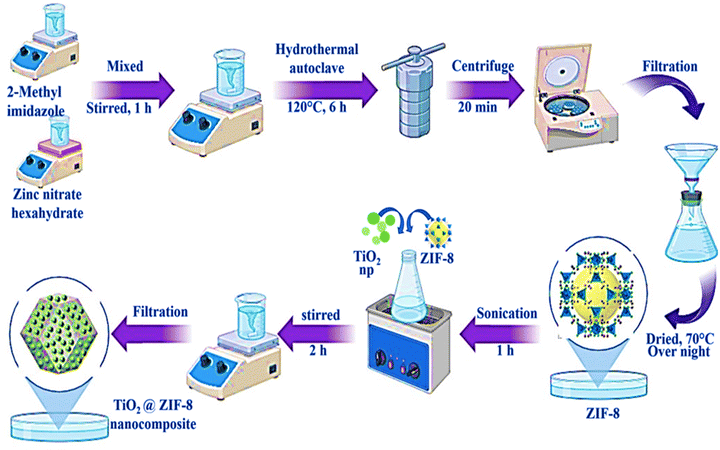
Zinc nitrate hexahydrate (98%), 2-methyl imidazole (98%), titanium dioxide (anatase) nanopowder (98%), and anhydrous methanol (99%) of analytical grades were supplied by Sigma Aldrich, USA. Commercial-grade chlorpyrifos (Chlorpyrifos 20% EC) was bought from the local market (Bhubaneswar, India) and used without any further treatment. All experimental procedures were carried out using double-distilled water to ensure purity.2.2 Preparation of catalysts
2.3 Characterization
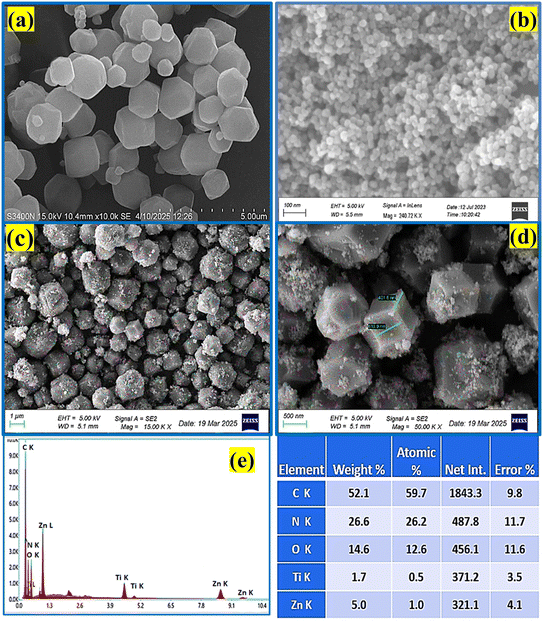
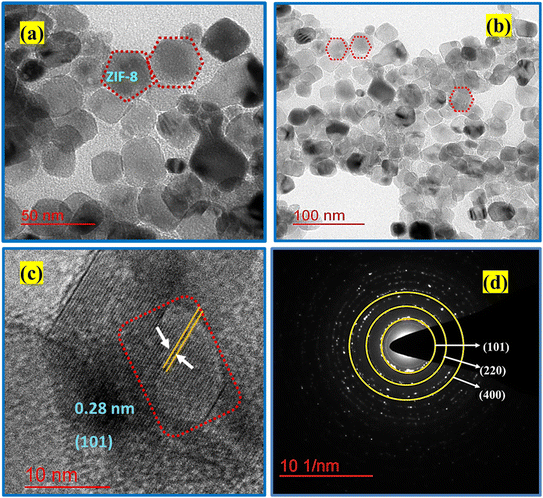
The structural properties of the as-synthesized ZIF-8 and TiO2@ZIF-8 nanocomposites were studied using powder X-ray diffraction (Bruker D8 Advanced) with Cu Kα radiation (λ = 0.1548 nm), scanning 2θ angles from 0.5° to 60° at 0.01° increments with 1 s counting time per step. Surface functional groups were observed by Fourier-transform infrared (FTIR) spectroscopy (Shimadzu). Optical properties and bandgap were measured by UV-vis diffuse reflectance spectroscopy (Agilent Cary 5000) and photoluminescence spectroscopy (Hitachi F-4600), respectively. The specific surface areas were evaluated through nitrogen adsorption–desorption isotherms measured at 77 K using a surface area analyser (Micrometrics ASAP 2020, USA) system, for which samples were degassed at 373 K for 12 h before analysis, and the Brunauer–Emmett–Teller (BET) method. Thermal stability was assessed by thermogravimetry-differential thermal analysis (STA 6000, PerkinElmer) under nitrogen atmosphere with a heating rate of 10 K min−1 from 300 to 1273 K. Morphological characterization was performed using field-emission scanning electron microscopy (Zeiss Auriga) coupled with energy-dispersive X-ray spectroscopy for elemental mapping. HRTEM (JEOL 2100) was used for crystallographic analysis. Surface chemical states were probed by X-ray photoelectron spectroscopy (Kratos AXIS Supra+) using monochromatic Al Kα radiation (1.48 keV). Reaction monitoring was conducted via UV-vis spectroscopy (Perkin Elmer, Germany) with a 1.5 nm s−1 scan rate. The GC-MS spectra were recorded using a Shimadzu GCMS-QP201 (Japan) system.2.4 Experimental design
The photocatalytic degradation of chlorpyrifos (CP) using the TiO2@ZIF-8 nanocomposite was optimized via response surface methodology (RSM), employing a central composite design (CCD). Three critical parameters and their ranges, as presented in Table 1, concentration (A), catalyst dosage (B), and time (C), were investigated for their individual and synergistic effects on degradation efficiency. The experimental design, comprising 18 randomized runs, was generated using Stat-Ease 360, with photodegradation efficiency as the response variable. A quadratic polynomial model was employed to describe the independent variables and the degradation performance. The significance of model terms and their interactions was statistically validated through analysis of variance (ANOVA), with particular emphasis on evaluating linear, quadratic, and two-factor interaction effects. Model adequacy was confirmed by comparing the coefficient of R2 value with the adjusted R2 value, while residual analysis and lack-of-fit tests ensured the robustness of the predictive model. This multivariate approach enabled the simultaneous optimization of multiple parameters while elucidating their complex interdependencies in the photocatalytic system.| Independent variables | Range and levels (−1 to +1) | ||
|---|---|---|---|
| Factors | Code | Units | 50–200 |
| Concentration of CP | A | mg L−1 | |
| Catalyst dose | B | mg | 10–50 |
| Time | C | min | 15–120 |
2.5 Photocatalytic measurement
The evaluation of the photocatalytic efficiency of TiO2@ZIF-8 was evaluated through the sunlight-driven degradation of the CP solutions. Experiments were conducted in 100 mL beakers containing 50 mL of CP solution under natural sunlight (June 2025, 20.300622° N and 85.842409°, 11:00–13:00 local time, 4.8–5.0 kWh m−2 day−1, equivalent to an instantaneous solar intensity of ∼95–100 mW cm−2 under clear-sky conditions) to maintain consistent light intensity. Further, 30 mg of photocatalyst was added to 100 mg L−1 of 50 mL CP solution and stirred in the dark for 15 min to establish adsorption–desorption equilibrium. The reaction was then employed under sunlight exposure for 90 min. The aliquots were collected at 30 min intervals for analysis. The CP degradation progress was monitored by measuring characteristic absorbance changes using UV-vis spectroscopy (λmax = 300 nm), enabling the quantitative determination of the decomposition efficiency. Each experiment was carried out in triplicate using the optimized parameters determined from RSM analysis to ensure reproducibility.The degradation efficiency (%) was calculated using the following equation:
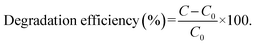 | (1) |
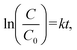 | (2) |
Following the completion of the reaction, the product was isolated by the repeated extraction of the aqueous layer with diethyl ether. The combined organic extracts were dried over anhydrous magnesium sulphate, concentrated, and subsequently analyzed by GC-MS.
2.6 Photoelectrochemical measurement
Electrochemical impedance spectroscopy (EIS) and Mott–Schottky were conducted on a CHI660E electrochemical workstation (CH Instruments) using a standard three-electrode system. The TiO2@ZIF-8 nanocomposite served as the working electrode, a saturated calomel electrode (SCE) as the reference, and a platinum sheet as the counter electrode.17 The Mott–Schottky measurements were performed in impedance–potential mode at a fixed frequency of 1 kHz with an AC perturbation of 10 mV. EIS Nyquist plots were recorded at open-circuit potential under dark conditions in the frequency range of 105–102 Hz, using 0.5 M Na2SO4 aqueous solution as the electrolyte. All measurements were carried out at ambient temperature (25 ± 1 °C) after allowing the system to stabilize for 20 min.2.7 Heterogeneity and recyclability test
The heterogeneous nature of the catalyst was evaluated using the following procedure. The catalyst was dispersed in water in the absence of the substrate and exposed to sunlight for 120 min. After this period, the suspension was filtered to separate the catalyst. The photocatalytic reactions were then carried out independently using the filtrate and the recovered catalyst.For the recycling study, the catalyst was recovered after each photocatalytic run by centrifugation, followed by filtration. After separation, the catalyst was thoroughly washed with water to eliminate any impurities, dried, and thermally regenerated. The regenerated catalyst was subsequently reused in successive degradation cycles under identical run conditions to assess its stability and reusability.
2.8 Antibacterial assessment
The antibacterial efficacy of the TiO2@ZIF-8 nanocomposite was assessed against both Gram-negative (E. coli) and Gram-positive (S. aureus) strains. Mueller–Hinton agar (MHA) medium was prepared and aseptically poured into sterile petri plates. Overnight bacterial cultures were spread evenly across the agar surface, and wells were loaded with the ZIF-8 and TiO2@ZIF-8 nanocomposites at concentrations of 5, 10 and 20 μg mL−1. After incubation at 37 °C for 24 h, the diameters of the inhibition zones were recorded.3. Results and discussion
3.1 Physicochemical characterizations
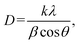 | (3) |
 | (4) |
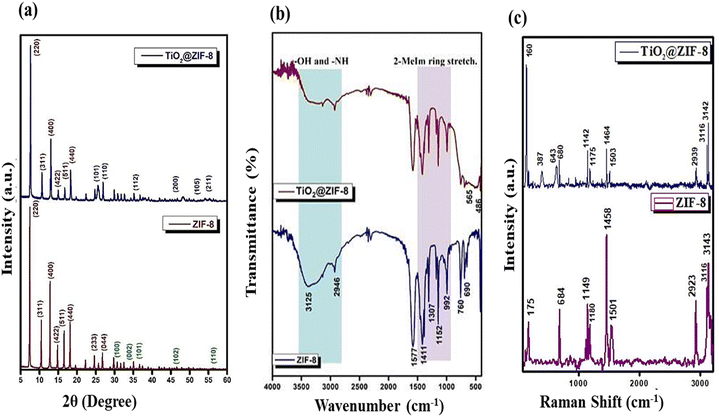
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretching, C–N stretching, imidazole ring bending, and a broad O–H/N–H stretching band at 1580, 1152, 760, and ∼3200–3600 cm−1, respectively.18 For the TiO2@ZIF-8 composite, these ZIF-8 signatures remained intact by preserving the ZIF-8 framework. The peaks around 800 cm−1 corresponded to Ti–O–Ti bridging, while new Ti–O–Ti stretching vibrations appeared around 486 cm−1, confirming the TiO2 incorporation into ZIF-8.20 The observed slight broadening and minor shifts (∼5–10 cm−1) in the C
N stretching, C–N stretching, imidazole ring bending, and a broad O–H/N–H stretching band at 1580, 1152, 760, and ∼3200–3600 cm−1, respectively.18 For the TiO2@ZIF-8 composite, these ZIF-8 signatures remained intact by preserving the ZIF-8 framework. The peaks around 800 cm−1 corresponded to Ti–O–Ti bridging, while new Ti–O–Ti stretching vibrations appeared around 486 cm−1, confirming the TiO2 incorporation into ZIF-8.20 The observed slight broadening and minor shifts (∼5–10 cm−1) in the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N and C–N stretching regions suggested interfacial interactions between TiO2 nanoparticles and the ZIF-8 framework, indicating successful hybrid formation.
N and C–N stretching regions suggested interfacial interactions between TiO2 nanoparticles and the ZIF-8 framework, indicating successful hybrid formation.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretching at 168, 686, 1147 and 1456 cm−1, respectively. The high-frequency peaks at 2923 cm−1 were due to the aliphatic C–H stretching of the methyl group, whereas those at 3116 and 3143 cm−1 were attributed to the aromatic C–H stretching of the imidazole ring.21 The TiO2@ZIF-8 nanocomposite maintained these ZIF-8 signatures while introducing new peaks at 160 cm−1 (Eg), 387 cm−1 (B1g), and 643 cm−1 (Eg), confirming the successful incorporation of anatase TiO2.19 Notably, the shifting of peaks suggested interfacial N–Ti–O bonding between ZIF-8 and TiO2. The spectral changes demonstrated the preservation of ZIF-8, along with the interactions between TiO2 and ZIF-8 at the interface.
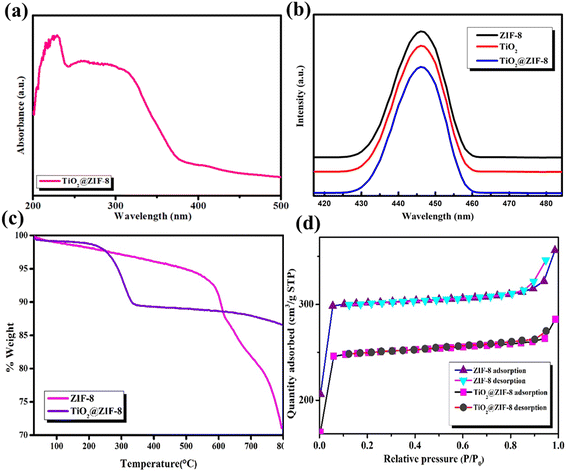
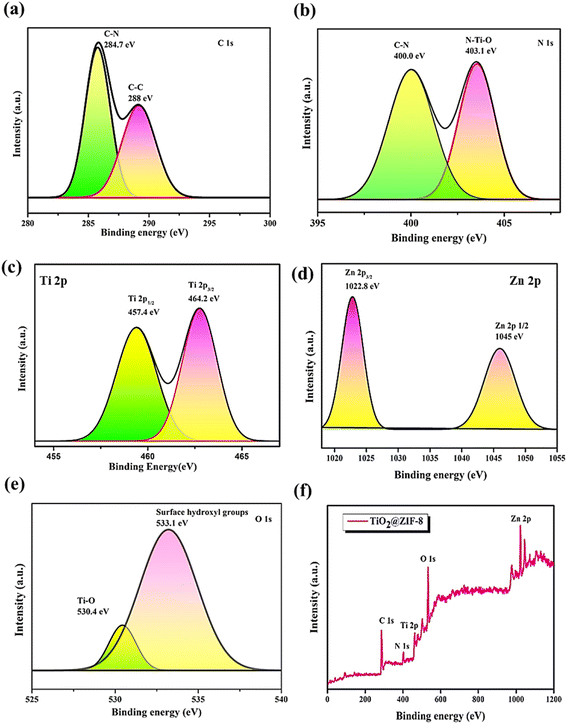
N stretching at 168, 686, 1147 and 1456 cm−1, respectively. The high-frequency peaks at 2923 cm−1 were due to the aliphatic C–H stretching of the methyl group, whereas those at 3116 and 3143 cm−1 were attributed to the aromatic C–H stretching of the imidazole ring.21 The TiO2@ZIF-8 nanocomposite maintained these ZIF-8 signatures while introducing new peaks at 160 cm−1 (Eg), 387 cm−1 (B1g), and 643 cm−1 (Eg), confirming the successful incorporation of anatase TiO2.19 Notably, the shifting of peaks suggested interfacial N–Ti–O bonding between ZIF-8 and TiO2. The spectral changes demonstrated the preservation of ZIF-8, along with the interactions between TiO2 and ZIF-8 at the interface.The zeta potential of TiO2@ZIF-8 was found to be −30 mV, suggesting moderate colloidal stability. Although this value was not significantly high enough, the negative surface charge rendered appreciable electrostatic repulsion, which helped to maintain dispersion.
3.2 Photocatalytic activity study of CP by TiO2@ZIF-8
| Degradation efficiency (%) = 78.61952 − 0.035426A + 0.291972B + 0.139493C + 0.000183AB − 0.000275AC − 0.000279BC + 0.000133A2 − 0.004772B2 − 0.000402C2. | (5) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 115.67, a P-value < 0.0001, and a large sum of squares (105.95), with significant terms including A, B, D, A2, B2, C2, and D2. Model reliability was further confirmed by a low coefficient of variation (CV = 0.0117%) and strong correlation coefficients (R2 = 0.995, adjusted R2 = 0.990, and predicted R2 = 0.981). The adequate precision value (1060.54) indicated a robust signal-to-noise ratio, well above the threshold of 4, which supported the model's validity (Tables S3 and S4).32
115.67, a P-value < 0.0001, and a large sum of squares (105.95), with significant terms including A, B, D, A2, B2, C2, and D2. Model reliability was further confirmed by a low coefficient of variation (CV = 0.0117%) and strong correlation coefficients (R2 = 0.995, adjusted R2 = 0.990, and predicted R2 = 0.981). The adequate precision value (1060.54) indicated a robust signal-to-noise ratio, well above the threshold of 4, which supported the model's validity (Tables S3 and S4).32Diagnostic plots were employed to evaluate the reliability of the predicted model. Fig. S5 illustrates the plot of predicted degradation efficiency against externally studentized residuals, confirming model validity as the predicted values were independent of the actual values. The random distribution of residuals within a ±4 range further supported the model's predictive accuracy. Fig. S5b displays the linear fit of externally studentized residuals against normal probability %, revealing a normally distributed linear trend that validated the model's results. The close alignment of the data points with the regression line indicated a strong correlation between actual and predicted values, validating the model's accuracy in predicting responses to variations in the independent variables. As shown in Fig. S5c, the Box–Cox plot (Fig. S5e) suggested a lambda value of 1, implying no need for response transformation in CP degradation analysis. Additionally, Cook's distance plot (Fig. S5d) showed that out of 18 experimental runs, most exhibited values near zero, indicating minimal influential outliers and robust model predictability. As shown in Fig. S5e, the actual vs. predicted degradation efficiency plot exhibits a strong correlation, reinforcing the model's applicability to experimental data. As shown in Fig. S5f, the externally studentized residual plot against the run number shows no discernible pattern, with residuals scattered randomly around the baseline within a ±4 range, indicating model precision and the absence of data inconsistencies.32–34 These results collectively affirmed the model's suitability for optimizing CP degradation under the studied conditions.
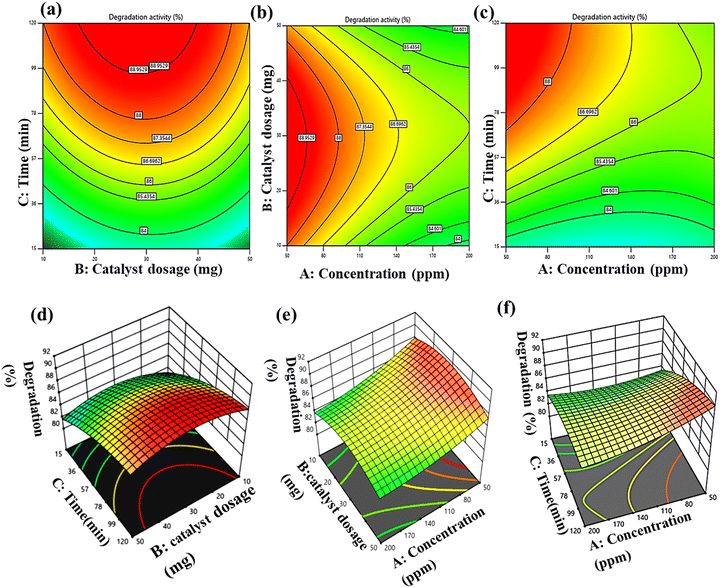
3.3 Influence of operational parameters on CP degradation
The surface model 2D and 3D plots were described for the responses of different parameters as variables and one constant, through which the optimized value of CP degradation efficiency could be obtained. The plots indicated the integrated effect on the degradation of CP.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
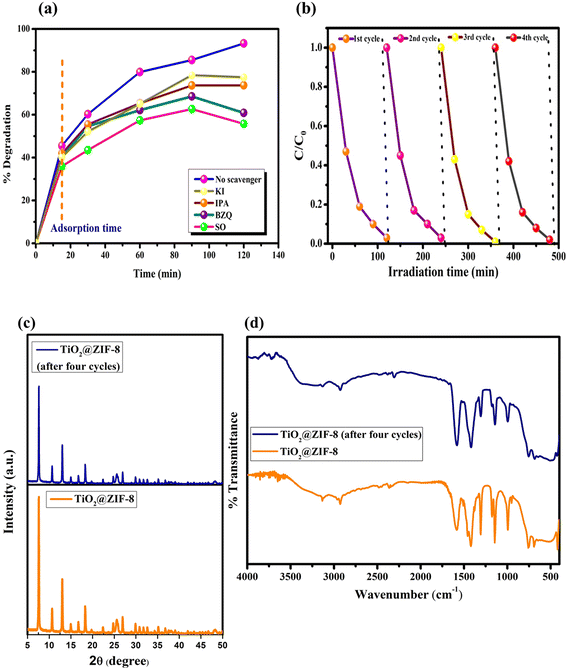
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ZIF-8 ratios (5%, 10%, and 20% of TiO2) were evaluated under identical conditions, revealing efficiencies of 70%, 82% and 90%, respectively, which indicated that ZIF-8 containing 10% of TiO2 was the best among the studied catalysts, as presented in Fig. S6a. Possibly, 5% w/w of TiO2 may not possess sufficient active sites; on the other hand, 20% w/w of TiO2 leads to the aggregation/blockage of active sites, as compared to the optimised 10% w/w TiO2@ZIF-8. Hence, the optimised catalyst selected for further study was 10% w/w of TiO2@ZIF-8. As illustrated in Fig. 7a and d, the efficiency increased with catalyst loading up to 30 mg, due to the relatively high availability of exposed active sites for CP interaction. However, increasing the dosage to 50 mg may result in light scattering, which decreases the degradation efficiency.
ZIF-8 ratios (5%, 10%, and 20% of TiO2) were evaluated under identical conditions, revealing efficiencies of 70%, 82% and 90%, respectively, which indicated that ZIF-8 containing 10% of TiO2 was the best among the studied catalysts, as presented in Fig. S6a. Possibly, 5% w/w of TiO2 may not possess sufficient active sites; on the other hand, 20% w/w of TiO2 leads to the aggregation/blockage of active sites, as compared to the optimised 10% w/w TiO2@ZIF-8. Hence, the optimised catalyst selected for further study was 10% w/w of TiO2@ZIF-8. As illustrated in Fig. 7a and d, the efficiency increased with catalyst loading up to 30 mg, due to the relatively high availability of exposed active sites for CP interaction. However, increasing the dosage to 50 mg may result in light scattering, which decreases the degradation efficiency.
The decrease in the degradation activity was mainly attributed to competitive adsorption and the light-shielding effect, which limited the photon penetration and reduced the availability of reactive oxygen species. Deviations from the optimized conditions led to reduced performance, arising from incomplete degradation at short times, ROS depletion at high pollutant concentrations, or light scattering at excessive catalyst loadings, highlighting the critical interdependence of these parameters in solar-driven photocatalytic systems.
| Pesticides | Variable | Before | After | Degradation efficiency (%) | |
|---|---|---|---|---|---|
| Predicted | Experimental | ||||
| Chlorpyrifos | Concentration (ppm) | 100 | 90 | 86 | 90 |
| Weight of the adsorbent (mg) | 30 | 20 | |||
| Time (min) | 90 | 86 | |||
3.4 Kinetics of the photocatalytic degradation of CP
To further investigate the reaction kinetics, ln(C/C0) was plotted as a function of time under varying CP concentrations (50–200 mg L−1). The linear fit of ln(C/C0) = kt + b confirmed that the degradation followed pseudo-first-order kinetics. The rate constant (k) was 0.025 s−1, and the R2 value was 0.991 for 100 mg L−1 of CP by TiO2@ZIF-8, which was the highest among all the concentrations (Fig. 8b and Table S7). This indicated that at this concentration, the balance of active site availability on the reaction surface and accessibility of CP molecules was optimal, thereby facilitating the efficient generation and utilization of reactive oxygen species (ROS).36 At relatively low CP concentrations, the reduced pollutant availability may limit the interaction with photo-induced charge carriers, while at high concentrations, the excessive adsorption of CP molecules could lead to surface saturation and light-shielding effects, ultimately suppressing photocatalytic efficiency.35 The observed rate constant trend was in excellent agreement with the photocatalytic performance results presented in Table S6, validating the superior efficiency of TiO2@ZIF-8 in driving the solar-light-assisted degradation of CP. | ||
| Fig. 8 Photocatalytic degradation of CP by (a) different photocatalysts and (b) kinetic study (catalyst amount: 30 mg; CP concentration: 100 mg L−1, time: 120 min). | ||
3.5 Electrochemical study
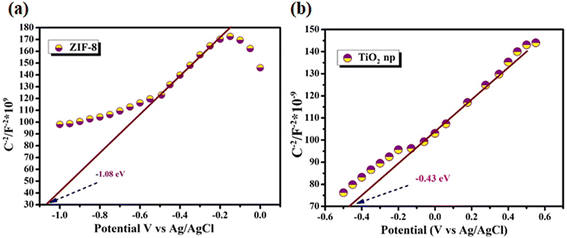
As shown in Fig. 9a and b, the positive slopes of the Mott–Schottky plots for ZIF-8 and TiO2 indicate their classification as n-type semiconductors. The flat-band potentials (Ex6) were estimated to be −1.31 and −0.43 eV for ZIF-8 and TiO2, respectively. The negative flat-band potential of ZIF-8 suggests a higher Fermi level compared with that of TiO2, facilitating efficient electron transfer at the TiO2@ZIF-8 nanocomposite.37 This alignment promoted the separation of photo-induced charge carriers under sunlight, improving photocatalysis. Similarly, EIS Nyquist plots (S8) revealed distinct charge-transfer resistance (Ret) for each material. ZIF-8 and TiO2 displayed relatively large semicircular arcs, indicative of high interfacial resistance and slow charge transfer kinetics.37 In contrast, the TiO2@ZIF-8 composite exhibited a significantly small arc radius, demonstrating reduced charge-transfer resistance due to the synergistic interaction within the heterostructure. This improvement confirmed enhanced electron–hole separation and interfacial charge migration, which were critical for the photocatalytic degradation of CP.3.6 Scavenger study
To identify the major reactive species involved in the photocatalytic removal of CP, various scavengers, such as potassium iodide (KI), benzoquinone (BQ), sodium oxalate (Na2C2O4), and isopropanol (IPA), were employed for radical trapping experiments for surface ·OH, h+, ·O2−, and ·OH, respectively.38 The addition of these scavengers significantly reduced the degradation efficacy of the TiO2@ZIF-8 composite from 90.2% to 58.5%, 50.9% and 50.3% after 90 min in the presence of Na2C2O4, BQ, and IPA, respectively (Fig. 10a). These results indicated that h+, ·O2−, and ·OH were crucial contributors to the degradation process. The significant decrease was obtained using IPA and BQ, emphasizing ·OH, h+ and ·O2− as the major active species, while KI confirmed that the surface ·OH radicals played a minimal role. The quantitative data presented in supplementary information Fig. S6b provide a hierarchical understanding of the reactive species, confirming their synergistic contribution to the high photocatalytic efficiency of the TiO2@ZIF-8 composite. The obtained results supported the proposed type-II heterojunction mechanism, where effective charge separation promoted the formation of multiple reactive oxygen species (ROS), thereby enhancing the overall photocatalytic activity of the TiO2@ZIF-8 heterostructure.3.7 Heterogeneity and recyclability study
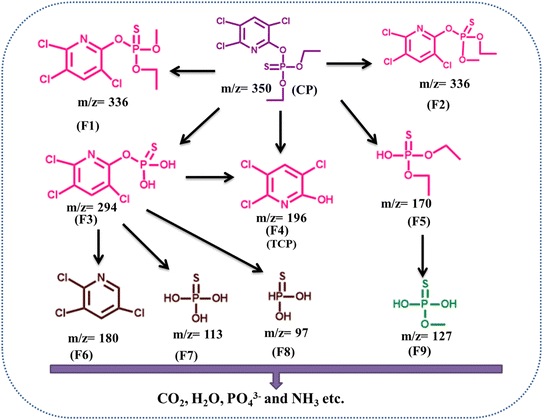
The heterogeneous nature of the catalyst was further examined by conducting photocatalytic degradation experiments using the filtrate obtained after the thorough washing and removing the solid catalyst. The filtrate was separated carefully to ensure the absence of residual catalyst particles, thereby eliminating any contribution from heterogeneous catalytic sites. The degradation efficiency observed in the filtrate system was approximately 30%, which closely corresponded to the extent of degradation achieved under direct photolysis conditions. This similarity strongly indicated that the contribution of dissolved or leached active species from the catalyst to the overall degradation process was negligible. Furthermore, the stability and recyclability of TiO2@ZIF-8 were assessed over four consecutive photocatalytic cycles, as shown in Fig. 10(b), which maintained an efficiency of ∼85%. The regenerated catalyst, after four cycles of characterization by XRD and FT IR (Fig. 10(c) and (d)), revealed no significant changes in crystalline phases and vibrational signatures, indicating structural stability. These findings demonstrate that TiO2@ZIF-8 is a highly stable and efficient photocatalyst for CP degradation.3.8 Enhanced photocatalytic mechanism
The TiO2@ZIF-8 nanocomposite achieved ∼90% degradation of CP within 90 min under solar light. To elucidate the charge-transfer pathways, a band energy diagram was developed using Tauc plots. The VB/CB positions of ZIF-8, TiO2, and TiO2@ZIF-8 suggested the probable formation of a type-II heterojunction. According to the previous analysis, this could be explained based on the electronic structure and interfacial charge transfer characteristics. Under solar irradiation, TiO2 absorbs photons and produces electron–hole pairs, while ZIF-8 facilitates interfacial electron transfer due to its more negative CB position. Mott–Schottky analysis suggests the presence of positive slopes for TiO2 and ZIF-8, indicative of n-type semiconductors. The flat-band potentials were determined to be −0.43 and −1.31 V for TiO2 and ZIF-8, respectively.For n-type semiconductors, the conduction band (CB) potentials were estimated to be approximately 0.2 V more negative than the flat-band potentials, which yielded CB positions of −0.63 and −1.28 V for TiO2 and ZIF-8, respectively. Based on their respective bandgaps (3.2 eV for TiO2 and 4.0 eV for ZIF-8, determined from UV-vis DRS), the valence band (VB) positions were estimated as +2.57 V for TiO2 and +2.49 V vs. NHE for ZIF-8. This band alignment indicated the formation of a type-II heterojunction, where photogenerated electrons transferred from the CB of ZIF-8 to that of TiO2, while holes migrated from the VB of TiO2 to that of ZIF-8, thereby facilitating efficient charge separation and suppressing electron–hole recombination (Fig. 11).
 | ||
| Fig. 11 Schematic presentation of photocatalytic degradation mechanism of chlorpyrifos (CP) by TiO2@ZIF-8. | ||
The VB potential of TiO2 was positive enough to oxidize H2O/OH− into ·OH radicals, while the CB potential of ZIF-8 was more negative than the O2/·O2− redox potential, enabling the reduction of dissolved oxygen into ·O2− radicals. Some of the ·O2− species further underwent proton-assisted reactions to form H2O2, which could decompose into additional ·OH radicals. The resulting spatial separation of charge carriers significantly reduced recombination losses and enhanced the photocatalytic efficiency, as evidenced by the improved degradation performance under solar light. The formation of this heterojunction with favorable band alignment provided a rational explanation for the observed enhancement in the photocatalytic activity of TiO2@ZIF-8. The interfacial electric field formed at the heterojunction further accelerated this charge separation while suppressing recombination. This migration of e− and h+ was driven purely by the energetic gradient and the interfacial electric field at the TiO2@ZIF-8 junction. Additionally, interfacial ligand-to-metal charge transfer (LMCT) and linker defect states in ZIF-8 help slightly extend the absorption of the composite.39 The porous ZIF-8 framework additionally enhanced photocatalytic efficiency by (1) providing a high surface area for reactant adsorption, (2) facilitating mass transport of reactants/products, (3) suppressing electron–hole recombination due to interfacial electric fields, and (4) potentially offering molecular sieving effects.
Under solar light, the photocatalytic degradation of CP proceeded through multiple reactive pathways. Initially, photoexcitation generated electron–hole pairs in the TiO2@ZIF-8 heterostructure, with electrons (e−) promoted to the conduction band (CB) while leaving holes (h+) in the valence band (VB). The thermodynamic potentials drove subsequent redox reactions: (1) the VB potential of TiO2 (+2.57 V vs. NHE) exceeded the oxidation potential of H2O/·OH (+2.4 V vs. NHE), enabling h+ to oxidize adsorbed H2O/OH− to generate hydroxyl radicals (·OH). (2) Simultaneously, the more negative CB potential of ZIF-8 (−1.31 V vs. NHE) compared to O2/·O2− (−0.33 V vs. NHE) facilitated oxygen reduction by photoelectrons to produce superoxide radicals (·O2−).39 (3) These primary reactive species further interacted through secondary reactions, where some ·O2− radicals combined with protons to form additional ·OH.
| TiO2@ZIF-8 + hν → TiO2@ZIF-8 + (h+VB, e−CB) pairs, |
| H2O/(OH−) + TiO2 (h+) → ·OH + H+, |
| O2 + ZIF-8 (e−) → ·O2−, |
| ·O2− + TiO2 (h+) + ·OH + CP → PO43− + CO2 + H2O + small molecules. |
3.9 Probable degradation pathway
GC-MS analysis confirmed the stepwise degradation of chlorpyrifos (CP, m/z = 350) into several intermediates, followed by mineralization, as presented in Fig. 12 and 13. The first step involved oxidative dealkylation, yielding demethylated derivatives F1 and F2 (m/z = 336), followed by the hydrolytic cleavage of the P–O–aryl bond to form F3 (m/z = 294). Again, the hydrolysis of the phosphorothioate group of CP into F4 (m/z = 196), identified as 3,5,6-trichloro-2-pyridinol (TCP), was observed. TCP underwent dechlorination and ring-cleavage, resulting in F6 (m/z = 180), while the parallel degradation of the thiophosphoryl group produced phosphorothioate derivatives F5 (m/z = 170), F7 (m/z = 113), F8, and F9 (m/z = 127) through sequential dealkylation, desulfuration, and oxidation. The disappearance of CP and its intermediates, along with the formation of CO2, H2O, PO43−, and NH3 at the m/z values of 44, 18, 97 and 16, respectively, indicated complete mineralization. Overall, the results suggested that the degradation process involved hydrolysis, oxidative dealkylation, dechlorination, and ring cleavage, demonstrating an efficient pathway for the detoxification and mineralization of chlorpyrifos.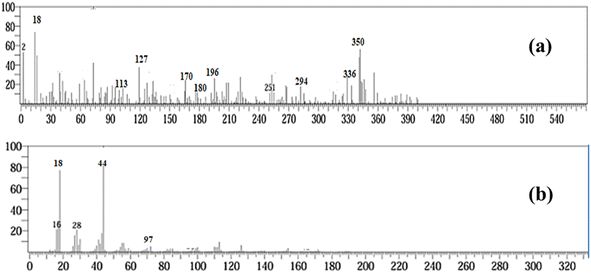 | ||
| Fig. 12 Fragments of CP (a) after the commencement of the reaction and (b) after the completion of the reaction. | ||
3.10 Anti-microbial analysis
The antibacterial activity of the TiO2@ZIF-8 nanocomposite was evaluated by measuring the inhibition zones on agar plates against both E. coli and S. aureus (Fig. S9). The TiO2@ZIF-8 (10 μg mL−1) nanocomposite exhibited significant antibacterial activities compared to other dosages. The positive control exhibited a prominent zone of inhibition, confirming the reliability of the assay, whereas the negative control showed no visible inhibition, validating the absence of any background antibacterial effect. In the case of E. coli, the TiO2@ZIF-8 (10 μg mL−1) nanocomposite demonstrated a distinct inhibition zone of approximately 14–15 mm, indicating significant antibacterial activities. While for S. aureus, the inhibition zone diameters were found to be 4–6 mm, which was less than the positive control but clearly active compared to the negative control. This is because the partial degradation of the ZIF-8 framework in biological media releases Zn2+ ions, which readily penetrate the thin Gram-negative outer membrane of E. coli, disrupt essential enzymes, and induce metabolic toxicity. Simultaneously, direct contact between bacterial cells and the high-surface-area composite promoted membrane disruption through exposure to surface metal nodes, and chelation enhanced penetration. Under light irradiation, TiO2 generated reactive oxygen species (·OH, ·O2−, h+) that oxidatively damaged lipids, proteins, and nucleic acids, and those ROS diffuse more easily through the less-rigid Gram-negative cell envelope, while the thick peptidoglycan layer of S. aureus offered partial protection. Additionally, ligand-exchange interactions (HSAB effect) could accelerate Zn2+ release and generate more reactive surface sites.40 Further, the incorporation of TiO2 into the porous ZIF-8 framework provided a high surface area, improved dispersion, and enhanced light-harvesting capability, which together promoted high ROS generation.41,42 Together, Zn2+ ion toxicity, contact-mediated membrane disruption, ROS-induced oxidative stress, and linker-exchange degradation synergistically accounted for the high susceptibility of E. coli.42 These findings highlight TiO2@ZIF-8 as a potential candidate for antimicrobial applications, with potential utility in water disinfection, biomedical coatings, and environmental remediation.4. Conclusion
TiO2@ZIF-8 heterostructures were successfully fabricated via hydrothermal and ultrasonication methods, and they showed significant photocatalytic and antimicrobial performance. TiO2@ZIF-8 heterostructures achieved the efficient photocatalytic degradation of CP at 90.9% with 100 mg L−1 of CP, using 30 mg of a catalyst within 90 min under solar light, and they exhibited better homogeneity and stability, maintaining activity even after four successive cycles. The superior photocatalytic efficiency of TiO2@ZIF-8 was attributed to its type-II heterojunction structure, which promoted charge carrier dynamics and broadened visible-light utilization. Radical quenching studies confirmed O2·−, h+, and ·OH as the major species in CP degradation. Additionally, the composite demonstrated notable antibacterial efficacy against E. coli and S. aureus, establishing its promise as a multifunctional material for environmental remediation.Author contributions
Sushanta Kumar Badamali is responsible for supervision, formal analysis, and resources. Subhashree Mohanty is responsible for writing – review and editing, writing – original draft, methodology, investigation, formal analysis, data curation, and software, and Bibeka Nanda Marai is responsible for methodology.Conflicts of interest
Authors declare no conflicts of interest.Data availability
Data will be made available by the authors upon request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5lf00405e.
Acknowledgements
The authors acknowledge the Government of Odisha, India, for the award of the MRIP fellowship. We acknowledge the support of JNCASR, Bangalore, and the Material Analysis and Research Center, Karnataka, for XRD measurement and BET analysis. We are also thankful to CRF, KIIT, Bhubaneswar and, IIT Hyderabad for34 FE SEM and, XPS analysis, respectively.References
- F. H. Tang, M. Lenzen, A. McBratney and F. Maggi, Nat. Geosci., 2021, 14, 206–210 CrossRef CAS.
- E. M. John and J. M. Shaike, Environ. Chem. Lett., 2015, 13, 269–291 CrossRef CAS.
- F. H. Tang, M. Lenzen, A. McBratney and F. Maggi, Nat. Geosci., 2021, 14, 206–210 CrossRef CAS.
- E. Wołejko, B. Łozowicka, A. Jabłońska-Trypuć, M. Pietruszyńska and U. Wydro, Int. J. Environ. Res. Public Health, 2022, 19, 12209 CrossRef PubMed.
- K. Jamil, A. P. Shaik, M. Mahboob and D. Krishna, Drug Chem. Toxicol., 2005, 28, 447–459 CrossRef PubMed.
- K. Piaskowski, R. Świderska-Dąbrowska and P. K. Zarzycki, J. AOAC Int., 2018, 101, 1371–1384 CrossRef CAS PubMed.
- D. S. Bhatkhande, V. G. Pangarkar and A. A. C. M. Beenackers, J. Chem. Technol. Biotechnol., 2002, 77, 102–116 CrossRef CAS.
- X. Wei, G. Zhu, J. Fang and J. Chen, Int. J. Photoenergy, 2013, 2013, 726872 Search PubMed.
- L. Ren, Y. Li, M. Mao, L. Lan, X. Lao and X. Zhao, Appl. Surf. Sci., 2019, 490, 283–292 CrossRef CAS.
- H. Dai, X. Yuan, L. Jiang, H. Wang, J. Zhang and T. Xiong, Coord. Chem. Rev., 2021, 441 Search PubMed.
- Y. H. Zou, H. N. Wang, X. Meng, H. X. Sun and Z. Y. Zhou, Nanoscale Adv., 2021, 3, 1455–1463 RSC.
- A. N. Ökte and D. Tuncel, Photochem. Photobiol. Sci., 2025, 1–20 Search PubMed.
- W. Wang, C. Shen, R. Shao, Y. Zhu, W. Yu, X. Wu and Z. Xu, et al., Colloids Surf., A, 2025, 714, 136568 CrossRef CAS.
- F. Zisti, F. J. M. Al-Behadili, M. Nadimpour, R. Rahimpoor, N. Mengelizadeh, A. Alsalamy and D. Balarak, Environ. Res., 2024, 245, 118019 CrossRef CAS PubMed.
- H. Teymourinia, H. A. Alshamsi, A. Al-Nayili and M. Gholami, Chemosphere, 2023, 344, 140325 CrossRef CAS PubMed.
- Y. Tian, Q. Liu, S. Lin, X. Liang, S. Khan, X. Yang and X. Wang, J. Environ. Chem. Eng., 2023, 11, 110054 CrossRef CAS.
- X. Liu, Z. H. Peng, L. Lei, R. X. Bi, C. R. Zhang, Q. X. Luo and J. D. Qiu, Appl. Catal., B, 2024, 342, 123460 CrossRef CAS.
- Y. Pan, Y. Liu, G. Zeng, L. Zhao and Z. Lai, Chem. Commun., 2011, 47, 2071–2073 RSC.
- Z. Ma, B. Guan, J. Guo, Y. Chen, J. Chen, H. Dang and Z. Huang, Ind. Eng. Chem. Res., 2023, 62, 17658–17670 CrossRef.
- R. Li, W. Li, C. Jin, Q. He and Y. Wang, J. Alloys Compd., 2020, 825, 154008 CrossRef CAS.
- K. Zhou, B. Mousavi, Z. Luo, S. Phatanasri, S. Chaemchuen and F. Verpoort, J. Mater. Chem. A, 2017, 5, 952–957 RSC.
- M. R. Barik and S. K. Badamali, ChemistrySelect, 2025, 10, e01792 CrossRef CAS.
- A. Semwal, D. Sajwan, J. Rawat, L. Gambhir, H. Sharma and C. Dwivedi, Environ. Sci. Pollut. Res., 2023, 30, 45827–45839 CrossRef CAS PubMed.
- G. Kumari, K. Jayaramulu, T. K. Maji and C. Narayana, J. Phys. Chem. A, 2013, 117, 11006–11012 CrossRef CAS PubMed.
- L. Cen, T. Tang, F. Yu, H. Wu, C. Li, H. Zhu and Y. Guo, J. Ind. Eng. Chem., 2023, 126, 537–545 CrossRef CAS.
- L. Bogdan, A. Palcic, M. Duplancic, M. Leskovac and V. Tomašic, Processes, 2023, 11, 1234 CrossRef.
- S. Angela, V. B. Lunardi, K. Kusuma, F. E. Soetaredjo, J. N. Putro, S. P. Santoso and S. Ismadji, Environ. Nanotechnol., Monit. Manage., 2021, 16, 100598 CAS.
- Z. Ma, B. Guan, J. Guo, Y. Chen, J. Chen, H. Dang and Z. Huang, Ind. Eng. Chem. Res., 2023, 62, 17658–17670 CrossRef.
- J. A. Thompson, K. W. Chapman, W. J. Koros, C. W. Jones and S. Nair, Microporous Mesoporous Mater., 2012, 158, 292–299 CrossRef CAS.
- C. Muthukumar, E. Iype, K. Raju, S. Pulletikurthi and B. P. Kumar, J. Mol. Struct., 2022, 1263, 133194 CrossRef CAS.
- E. M. Hotze, T. Phenrat and G. V. Lowry, J. Environ. Qual., 2010, 39, 1909–1924 CrossRef CAS PubMed.
- S. H. Khan, B. Pathak and M. H. Fulekar, Nanotechnol. Environ. Eng., 2018, 3, 13 CrossRef.
- M. Mohammadi, S. Sabbaghi, M. Binazadeh, S. Ghaedi and H. Rajabi, Chemosphere, 2023, 336, 139311 CrossRef CAS PubMed.
- M. S. Mohtaram, S. Sabbaghi, J. Rasouli and K. Rasouli, Environ. Pollut., 2024, 347, 123746 CrossRef CAS PubMed.
- A. M. Dugandžić, A. V. Tomašević, M. M. Radišić, N. Ž. Šekuljica, D. Ž. Mijin and S. D. Petrović, J. Photochem. Photobiol., A, 2017, 336, 146–155 CrossRef.
- J. Zhang, X. Gao, W. Guo, Z. Wu, Y. Yin and Z. Li, RSC Adv., 2022, 12, 6676–6682 RSC.
- C. Mohanty, A. Samal, A. K. Behera and N. Das, J. Water Process Eng., 2024, 65, 105821 CrossRef.
- Y. Wang, Q. Wang, X. Zhan, F. Wang, M. Safdar and J. He, Nanoscale, 2013, 5, 8326–8339 RSC.
- S. Navalon, A. Dhakshinamoorthy, M. Alvaro, B. Ferrer and H. Garcia, Chem. Rev., 2022, 123, 445–490 CrossRef PubMed.
- R. K. Alavijeh, S. Beheshti, K. Akhbari and A. Morsali, Polyhedron, 2018, 156, 257–278 CrossRef.
- J. Cui, D. Wu, Z. Li, G. Zhao, J. Wang, L. Wang and B. Niu, Ceram. Int., 2021, 47, 15759–15770 CrossRef CAS.
- S. Sadiq, I. Khan, M. Humayun, P. Wu, A. Khan and M. Bououdina, ACS Omega, 2023, 8(51), 49244–49258 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |