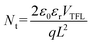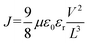Open Access Article
Open Access ArticleSelf-assembled monolayer modified nickel oxide surface for air-processed blue perovskite light-emitting diodes
Jiandong Wua,
Yujie Wanga,
Fankai Lina,
Chaoran Nia,
Shuhao Xionga,
Miao Zhangb,
Hongyue Wang *a and
Hongqiang Wang
*a and
Hongqiang Wang *a
*a
aState Key Laboratory of Solidification Processing, Center for Nano Energy Materials, School of Materials Science and Engineering, Northwestern Polytechnical University and Shaanxi Joint Laboratory of Graphene, Xi'an, 710072, China. E-mail: hongyue.wang@nwpu.edu.cn; hongqiang.wang@nwpu.edu.cn
bMaterials Institute of Atomic and Molecular Science, School of Physics & Information Science, Shaanxi University of Science and Technology, Xi'an 710021, China
First published on 16th March 2026
Abstract
Ambient-air fabrication of blue perovskite light-emitting diodes (PeLEDs) presents a significant pathway toward accelerating the commercialization of perovskite displays. However, a primary challenge still remains in deteriorating perovskite crystallization caused by absorption of moisture on the substrate surface. Herein, we engineered a novel composite interface comprising an inorganic transport layer and an ultra-thin self-assembled monolayer (SAM) to significantly enhance the crystallization quality of air-processed blue perovskites. The molecule of (2-(3,6-dibromo-9H-carbazol-9-yl)ethyl) phosphonic acid (Br-2PACz) features a hydrophobic carbazole headgroup and a strongly anchoring phosphonic acid (P–OH) tail group. The tail group establishes robust covalent P–O–Ni bonding with NiOx nanoparticles, which effectively passivates the surface defects on the NiOx surface and concurrently creates an enhanced interfacial dipole moment, thereby facilitating superior hole injection. Simultaneously, the peripheral carbazole headgroup enhances interfacial hydrophobicity of the NiOx layer, thereby optimizing the crystallization of perovskites in ambient air. Ultimately, the air-processed blue perovskite films on the Br-2PACz-modified layer exhibit suppressed defect densities and enhanced photoluminescence quantum yield (PLQY), leading to a substantial improvement in the external quantum efficiency (EQE) for blue PeLEDs from 0.73% to 3.32% in ambient air (T ≈ 21 °C, RH ≈ 20%).
Introduction
Solution-processed metal halide perovskites (MHPs) have attracted significant attention in the field of next-generation optoelectronics, owing to their high color purity, tunable bandgaps and high photoluminescence quantum yield (PLQY).1–3 The state-of-the-art perovskite light-emitting diodes (PeLEDs) have achieved landmark efficiencies, with record external quantum efficiencies (EQEs) now surpassing 30% for green and red PeLEDs,4,5 as well as reaching above 26% for blue PeLEDs.6 However, the fabrication of these high-performance devices is currently confined to inert-atmosphere gloveboxes with nitrogen environments, which significantly hinders their commercialization.7 The recent development of ambient air-processing techniques for perovskite film fabrication has demonstrated significant potential for low-cost and scalable manufacturing of PeLEDs.8,9 However, there is still a major challenge that moisture and oxygen can disrupt the crystallization process of perovskites, resulting in poor film optical properties.10–12 Particularly for high moisture-sensitivity blue perovskites, ambient humidity substantially degrades film quality and decreases photoluminescence quantum yield (PLQY), constraining the record EQE of air-processed blue PeLEDs to only 5.89%.13,14 Therefore, developing novel strategies that enable efficient and reproducible blue device fabrication under ambient air conditions is crucial for the future commercialization of PeLEDs.Many efforts have been devoted to enhancing the crystallization quality of perovskites under ambient air conditions. For example, typical weakly polar antisolvents such as diethyl ether13 and n-hexane15 effectively narrow the crystallization window of perovskites in ambient air, thereby enhancing the crystallinity and luminescence properties of the blue perovskite films. Also, a series of additive strategies have been developed to mitigate the detrimental effects of moisture by chemical elimination of residual water from perovskite precursors9,16 and strengthening the ionic interaction among perovskite crystals to improve their environmental stability.17 However, these strategies improved the humidity stability of the perovskite active layer, simultaneously deteriorating interfacial properties under moisture attack, which remain the key constraint for high-performance air-processed PeLEDs.18 Particularly at buried interfaces, studies have confirmed that even trace amounts of moisture adsorbed on the substrate hole transport layers (HTLs) can severely disrupt the nucleation and crystallization processes of perovskites, ultimately leading to spectral instability and compromised luminescence performance of the perovskite films.19,20 Inorganic nickel oxide (NiOx) has recently emerged as a preferred hole-transport material for air-processed PeLEDs due to its weak water-absorption nature.21 However, pristine NiOx nanoparticles often suffer from a high density of defect states (e.g., Ni3+ species and oxygen vacancies) and an unideal band structure, which can cause severe non-radiative recombination and create an energy barrier for hole injection.22 Moreover, these defects also function as primary sites for moisture attack under ambient conditions, which markedly accelerates the hydrolytic degradation of prepared perovskite films.23 Therefore, an ideal interfacial modifier is thus urgently needed to resolve the dual challenges in NiOx transport layers, requiring both enhanced hydrophobicity to enable moisture-resistant surfaces for perovskite crystallization in ambient air and optimized energy level alignment to promote hole injection.
Here, we propose and validated a strategy that an ultra-thin modification layer of a self-assembled monolayer (SAM) addresses the dual-challenges of moisture adsorption and energy level mismatch for the air-served NiOx layer. The molecule of (2-(3,6-dibromo-9H-carbazol-9-yl)ethyl) phosphonic acid (Br-2PACz) integrates a hydrophobic carbazole headgroup and a strong phosphonic acid (P–OH) anchoring group.24 The P–OH groups establish a strong covalent P–O–Ni bond with the NiOx substrate, efficiently passivating surface defects of the NiOx layer and increasing the surface potential for enhanced hole injection. Simultaneously, the peripheral carbazole groups enhance interfacial hydrophobicity, thereby optimizing the crystallization process of perovskites in ambient air. Capitalizing on this dual advancement, Br-2PACz-modified HTLs yield air-processed blue perovskite films with suppressed trap states and enhanced PLQY (from 16.8% to 31.0%), ultimately boosting the EQE of air-processed blue PeLEDs from 0.73% to 3.32% (T ≈ 21 °C, RH ≈ 20%). Meanwhile, when the emission area of PeLEDs is scaled up to 1.0 cm × 1.0 cm, they still maintained an EQE of 1.83%.
Results and discussion
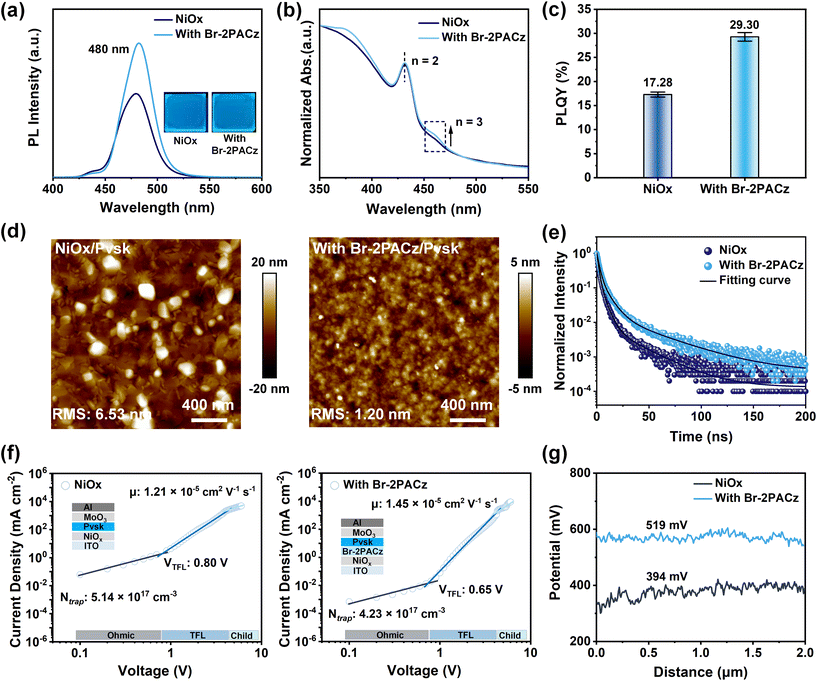
The hole transport layers (HTLs) of Br-2PACz-modified NiOx were fabricated via spin-coating (see details in the SI). The presence of Br-2PACz simultaneously passivates the defects and improves the hydrophobicity of the NiOx surface (Fig. 1a and b). We conducted X-ray photoelectron spectroscopy (XPS) characterization to confirm the interaction between Br-2PACz molecules and NiOx. As shown in Fig. 1c, the characteristic peaks observed at 134.2 eV and 133.2 eV correspond to P 2p1/2 and P 2p3/2 respectively, which are associated with –P–OH and –PO32− functional groups. The electronegative –PO32− group in Br-PACz provides a potential site for chemical interaction with NiOx. Meanwhile, the Ni 2p signal in the Br-2PACz modified film shifts by approximately 0.8 eV to a lower binding energy (Fig. 1d) and exhibits a reduced Ni3+ content compared with that of NiOx film (Table S1), which indicates the formation of a chemical bond between Br-2PACz and NiOx via P–O–Ni. This strong anchoring between Br-2PACz and NiOx is further confirmed by the presence of the P–O–Ni signal at 532.2 eV in the O 1s spectrum (Fig. S1). To evaluate the water resistance of these substrates, we measured the contact angles of water droplets on NiOx and Br-2PACz-modified film, respectively. As shown in Fig. 1e and. S2, the Br-2PACz-modified film exhibits an increased contact angle, rising from 56.1° to 67.4° compared to the NiOx film. The enhanced contact angle effectively prevents the adsorption of atmospheric moisture on the surface (Fig. S3), thereby promoting more uniform nucleation and optimizing the crystal growth process of the perovskite. Additionally, the enhanced interfacial hydrophobicity effectively accelerates the perovskite crystallization rate, thereby shortening the window of exposure to ambient moisture during film formation, which contributes to the formation of high-quality, uniformly blue perovskite films under ambient air conditions (Note S1). Atomic force microscopy (AFM) and Kelvin probe force microscopy (KPFM) were employed to characterize the surface roughness and surface potential of these films. As shown in Fig. S4, the Br-2PACz-modified film exhibits a lower surface roughness of 3.46 nm compared to the NiOx film (4.17 nm), indicating that Br-2PACz makes the NiOx surface flatter and helps reduce surface defects. By leveraging the pronounced conjugation group of carbazole, the Br-2PACz-modified film exhibits a significantly elevated average surface contact potential difference (CPD = 822 mV) compared with that of NiOx film (CPD = 432 mV), as shown in Fig. 1f and g. The increased surface potential effectively reduces the carrier transport barrier at the interface, thereby enhancing hole injection efficiency from the HTLs to the perovskite film.The blue quasi-2D perovskite films were spin-coated onto the substrates and annealed under ambient conditions (T ≈ 21 °C, RH ≈ 20%). As shown in Fig. 2a, these perovskite films display a prominent primary PL emission peak at 480 nm, along with inapparent shoulder peaks at 435 nm and 465 nm, which are attributed to the PL from low-dimensional phases.25 Notably, a significant enhancement in the PL intensity is observed in the film deposited on the Br-2PACz-modified NiOx substrate, indicating a considerable improvement in the radiative recombination efficiency of the perovskite. We employed UV-vis absorption spectroscopy to characterize the phase distribution of these quasi-2D perovskite films. As shown in Fig. 2b, the absorption features at 432 nm and 462 nm can be assigned to the n = 2 and n = 3 phases, respectively. On the Br-2PACz-modified NiOx substrate, the perovskite film exhibits an increased proportion of the n = 3 phase. This effectively alleviates the undesirable electron–phonon coupling induced by the quantum confinement of low-dimensional phases and promotes efficient charge carrier transport within the quasi-2D perovskite film.26 Consequently, the perovskite film on the Br-2PACz-modified NiOx shows a significant increase in PL quantum yield (PLQY) to 29.30%, up from 17.28% on the NiOx substrate (Fig. 2c and S5). Atomic force microscopy (AFM) was used to characterize the surface morphology of these perovskite films. As shown in Fig. 2d, the film on the Br-2PACz-modified NiOx substrate exhibits a much smoother surface with a reduced roughness of 1.20 nm, compared to the 6.53 nm roughness of the film on the NiOx substrate. This enhanced film uniformity is attributed to the substantial reduction of perovskite component segregation on Br-2PACz-modified substrates (Fig. S6), thus effectively passivating crystalline defects in the perovskite film.
We performed time-resolved photoluminescence (TRPL) measurement to characterize the carrier recombination dynamics of these perovskite films (Fig. 2e), and the statistical result is shown in Table S2. The film on the Br-2PACz-modified substrate shows an enhanced average decay time (τavg) of 10.85 ns, which represents a two-fold increase compared to that of the film on NiOx (τavg = 5.40 ns). Notably, on the Br-2PACz-modified NiOx substrate, the increased fraction of τ2 (7.75 ns) in the perovskite film further confirms the reduction of trap-assisted recombination, thereby enhancing the luminescence performance of the perovskite film.27 To further investigates the carrier dynamics and trap density of the perovskite films, space-charge limited current (SCLC) measurements were conducted by fabricating hole-only devices. The dark current density–voltage (J–V) curves are presented in Fig. 2f. The J–V curve in the SCLC model is typically divided into three distinct regions: the ohmic region at low bias, the trap-filling limit (TFL) region, and the trap-free Child's region at high bias.28 The trap-filling limited voltage (VTFL) can be extracted from the turning point between the ohmic and TFL regions, which is used to calculate the trap density (Nt) using the following equation:
where J is the current density, μ is the carrier mobility, V is the applied voltage, ε0 is the vacuum permittivity, εr is the relative dielectric constant (εr ≈ 14.52), VTFL is the trap-filled limit voltage, q is the elemental charge (q = 1.60217 × 10−19) and L is the thickness of perovskite films (L ≈ 50 nm). As shown in Fig. 2f, the VTFL for the device incorporated Br-2PACz is significantly reduced to 0.66 V, compared to 0.80 V for the control device. The calculated trap density (Nt) is also reduced from 5.14 × 1017 cm−3 (NiOx) to 4.23 × 1017 cm−3 (with Br-2PACz). Meanwhile, the hole mobility (μ) increased from 1.21 × 10−5 cm2 V−1 s−1 (NiOx) to 1.45 × 10−5 cm2 V−1 s−1 (with Br-2PACz). This SCLC analysis provides strong support that the Br-2PACz modification not only effectively passivates the trap states within the perovskite film but also promotes the efficiency of hole transport. Kelvin probe force microscopy (KPFM) was employed to characterize the surface potential distribution of these perovskite films. As shown in Fig. 2g and S7, the Br-2PACz-modified NiOx substrate yields a perovskite film with a contact potential difference (CPD) of 519 mV, which is higher than that of the perovskite film on NiOx (394 mV). This enhanced surface potential further confirms improved charge carrier transport capability within the perovskite layer,30 which is consistent with the results of enhanced PLQY and a prolonged carrier lifetime.
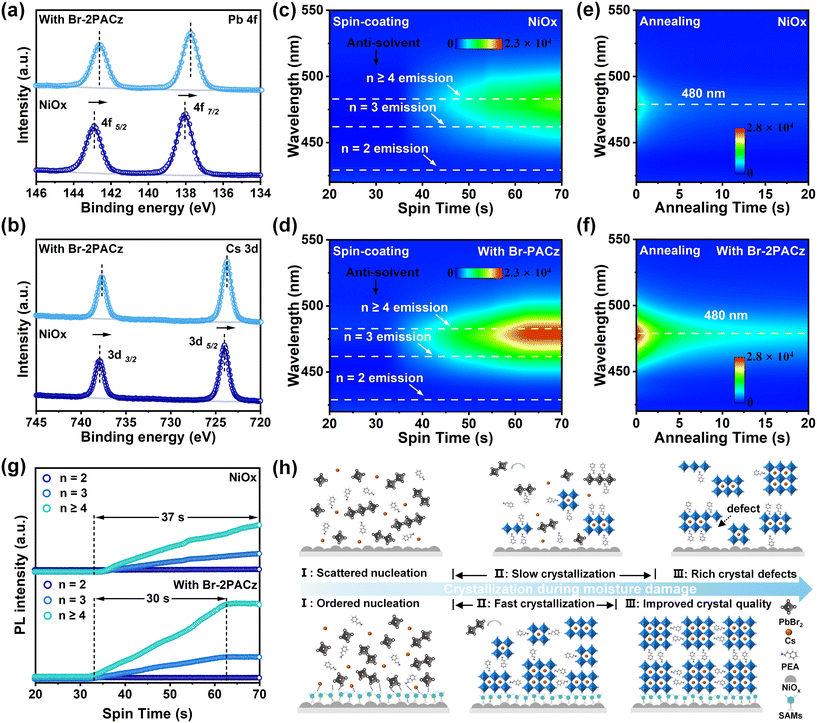
The nucleation and crystallization processes of blue quasi-2D perovskites on substrates with and without Br-2PACz modification were further systematically investigated. To verify potential pre-nucleation interactions, perovskite components (PbBr2 and CsBr) were thoroughly blended with respective hole transport materials (NiOx and Br-2PACz). As shown in Fig. 3a and b, when mixed with NiOx, the characteristic XPS signal of Pb 4f and Cs 3d remained nearly identical to those of the pure PbBr2 and CsBr references, indicating the absence of chemical interaction. In contrast, introduction of Br-2PACz into the mixture induced clear shifts for both peaks toward lower binding energies. Meanwhile, FT-IR analysis reveals that the C–Br vibration peak of the Br-2PACz exhibits a significant redshift from 733.31 cm−1 to 732.34 cm−1 upon mixing with the perovskite components (Fig. S8).31 These results confirm the pre-interaction between Br-2PACz and the perovskite components, which helps establish abundant nucleation sites on the buried interface that direct and facilitate perovskite crystallization on the substrate.32
We continuously monitored the PL intensity evolution during the spin-coating and annealing stages of the perovskite film by using a home-made in situ PL spectroscopy system, aiming to elucidate the perovskite growth dynamics on these substrates. All PL spectra were acquired under identical measurement conditions. As shown in Fig. 3c and d, perovskite films processed on Br-2PACz-modified NiOx substrates exhibited significantly enhanced PL intensity during spin-coating, and an extended PL thermal quenching duration was observed throughout the annealing process (Fig. 3e and f), collectively indicating improved crystallinity and reduced defect density in the perovskite films. The formation of luminescent crystals determines the PL intensity, and the time required to reach the maximum intensity serves as an indicator of the crystallization rate.33 We further investigated the growth kinetics of distinct n-phases by analyzing the temporal evolution of their corresponding PL intensities extracted from Fig. 3c and d during the spin-coating process, as summarized in Fig. 3g. The characteristic emission signals for n = 2, n = 3, and n = 4 phases in the quasi-2D perovskite were monitored at 430 nm, 465 nm, and 479 nm, respectively. On the NiOx substrate, all n-phase PL intensities exhibited continuous enhancement throughout the entire spin-coating duration, indicating ongoing perovskite crystallization. In contrast, on the Br-2PACz-modified NiOx substrate, the PL intensities of all n-phases reached their maximum at 62.7 s and subsequently stabilized during the remaining spin-coating time. This shortened time to reach the PL maximum demonstrates that Br-2PACz significantly accelerates the crystallization rate of all perovskite n-phases. X-ray diffraction (XRD) was employed to characterize these prepared films. As shown in Fig. S9, the perovskite film deposited on the Br-2PACz modified NiOx substrate exhibits a notably higher intensity of the characteristic (200) peak, indicating the enhanced crystallinity of perovskite film.34 Therefore, we conclude that the abundant nucleation sites provided by Br-2PACz at the interface effectively accelerate the crystallization rate of air-processed quasi-2D perovskites, which significantly shortens the timeframe during which moisture hampers perovskite crystallization, thereby enabling the formation of uniform perovskite films with substantially improved luminescence performance (Fig. 3h).
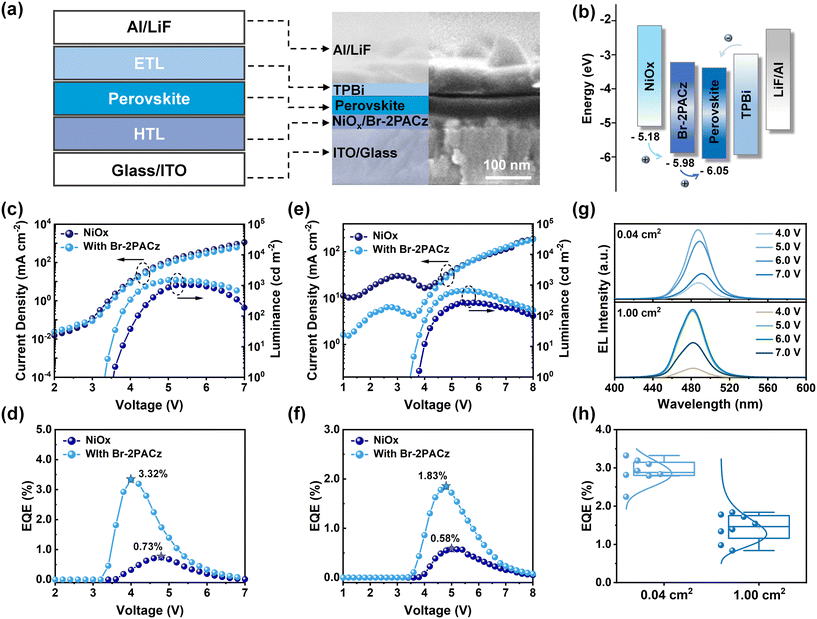
We used the NiOx layer, with and without Br-2PACz modification, as a hole transport substrate layer for spin-coating blue perovskite films under ambient air conditions (T ≈ 21 °C, RH ≈ 20%), and then electron transport layers (ETLs) and aluminum (Al) were evaporated to fabricate blue PeLEDs. The device structure and SEM image in cross-sectional view of corresponding PeLEDs are shown in Fig. 4a. Ultraviolet photoelectron spectrometry (UPS) was used to assess the carrier injection barriers between these HTLs and the perovskite layer. As shown in Fig. 4b and S10, the Br-2PACz modified NiOx layer demonstrated a deeper HOMO energy level of −5.98 eV compared to −5.18 eV for the pure NiOx. Since the blue perovskite has a deeper valence band maximum (VBM), the deepened HOMO level of Br-2PACz modified NiOx effectively mitigates the hole injection barrier, contributing to improve PeLED performance. Fig. 4c presents the current density–voltage–luminance (J–V–L) curves of these blue PeLEDs, where the lower current density and turn-on voltage of Br-2PACz contained PeLEDs indicates reduced interfacial defects, thereby resulting in a maximum brightness of 1512 cd m−2 and a peak EQE of 3.32% for the champion device; these values are higher than the control devices with a maximum brightness of 1036 cd m−2 and a peak EQE of 0.73% (Fig. 4d). To further evaluate the operational stability of these air-processed PeLEDs, we measured their operational half-lifetime (T50) under the initial luminance of 100 cd m2, with the constant voltage of 4 V. As shown in Fig. S11, the T50 of PeLEDs incorporating Br-2PACz reach to 268.2 s, which is approximately 3.17 times higher than that of the NiOx-based device (T50 = 84.5 s).
Benefiting from the enhanced interfacial hydrophobicity and uniform crystallization control of the perovskite, we successfully achieved the scalable fabrication of large-area PeLEDs with a 1.0 cm × 1.0 cm emitting area under ambient air conditions. As shown in Fig. 4e, the large-area devices containing Br-2PACz exhibited an enhanced brightness of 653 cd m−2 and a peak EQE of 1.83% (Fig. 4f), outperforming the control devices (maximum brightness: 271 cd m−2, EQE: 0.57%). Furthermore, all air-processed blue PeLEDs incorporating Br-2PACz maintained exceptional color stability, exhibiting negligible spectral shift as the operating voltage was increased from 4.0 V to 7.0 V (Fig. 4g and S12). Statistical analysis of 8 devices reveals that all air-processed PeLEDs incorporating Br-2PACz exhibited enhanced EQE compared to the control devices, alongside demonstrating excellent performance reproducibility of the Br-2PACz involved blue PeLEDs (Fig. 4h and S13).
Conclusion
In summary, the Br-2PACz modification strategy effectively mitigates the detrimental impact of interfacial moisture adsorption on the crystallization of perovskites in ambient air, owing to the enhanced defect passivation and improved hydrophobicity. Meanwhile, the Br-2PACz molecules on the NiOx surface provide pre-nucleation coordination sites for perovskite components, promoting uniform perovskite film crystallization on the substrate. This optimized crystallization process results in air-processed blue perovskite films with suppressed defect densities and a higher PLQY. Consequently, the EQE of air-processed blue PeLEDs is significantly enhanced from 0.73% to 3.32% compared with control devices. Notably, the emission area of the PeLEDs can be scaled up to 1.0 cm × 1.0 cm while maintaining an EQE of 1.83%. This study offers a novel interfacial design strategy for the ambient-air fabrication of perovskite optoelectronic devices, thereby promotes the commercialization of perovskite-based displays.Conflicts of interest
The authors declare no conflict of interest.Data availability
The authors confirm that all data supporting the findings of this study are contained within the article and the supplementary information (SI), and others available from the corresponding author upon reasonable request.Supplementary information: materials and methods were detailed as supporting information. See DOI: https://doi.org/10.1039/d5lf00384a.
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (22372132), thw National Key R&D Program of China (2023YFE0206300) and the Fundamental Research Funds for the Central Universities (G2025KY06205). We would like to thank the Analytical & Testing Center of Northwestern Polytechnical University for structure and performance characterization.References
- Z.-K. Tan, R. S. Moghaddam, M. L. Lai, P. Docampo, R. Higler, F. Deschler, M. Price, A. Sadhanala, L. M. Pazos, D. Credgington, F. Hanusch, T. Bein, H. J. Snaith and R. H. Friend, Bright light-emitting diodes based on organometal halide perovskite, Nat. Nanotechnol., 2014, 9(9), 687–692 CrossRef CAS PubMed.
- J. Dong, B. Zhao, H. Ji, Z. Zang, L. Kong, C. Chu, D. Han, J. Wang, Y. Fu, Z.-H. Zhang, Y. Yang, L. Zhang, X. Yang and N. Wang, Multivalent-effect immobilization of reduced-dimensional perovskites for efficient and spectrally stable deep-blue light-emitting diodes, Nat. Nanotechnol., 2025, 20(4), 507–514 CrossRef CAS PubMed.
- Y. Yu, B. F. Wang, Y. Shen, Y. T. Wang, Y. H. Zhang, Y. Y. Li, Z. H. Su, L. X. Cao, S. C. Feng, Y. H. Wu, X. Y. Gao, S. Kera, N. Ueno, J. X. Tang and Y. Q. Li, Efficient Blue Perovskite LEDs via Bottom-Up Charge Manipulation for Solution-Processed Active-Matrix Displays, Adv. Mater., 2025, 37(37), 2503234 CrossRef CAS PubMed.
- M. Li, Y. Yang, Z. Kuang, C. Hao, S. Wang, F. Lu, Z. Liu, J. Liu, L. Zeng, Y. Cai, Y. Mao, J. Guo, H. Tian, G. Xing, Y. Cao, C. Ma, N. Wang, Q. Peng, L. Zhu, W. Huang and J. Wang, Acceleration of radiative recombination for efficient perovskite LEDs, Nature, 2024, 630(8017), 631–635 CrossRef CAS PubMed.
- S. Q. Sun, J. W. Tai, W. He, Y. J. Yu, Z. Q. Feng, Q. Sun, K. N. Tong, K. Shi, B. C. Liu, M. Zhu, G. Wei, J. Fan, Y. M. Xie, L. S. Liao and M. K. Fung, Enhancing Light Outcoupling Efficiency via Anisotropic Low Refractive Index Electron Transporting Materials for Efficient Perovskite Light-Emitting Diodes, Adv. Mater., 2024, 36(24), 2400421 CrossRef CAS PubMed.
- Y. Gao, Q. Cai, Y. He, D. Zhang, Q. Cao, M. Zhu, Z. Ma, B. Zhao, H. He, D. Di, Z. Ye and X. Dai, Highly efficient blue light-emitting diodes based on mixed-halide perovskites with reduced chlorine defects, Sci. Adv., 2024, 10, eado5645 CrossRef CAS PubMed.
- M. Zhang, X. Ma, J. L. Esguerra, H. Yu, O. Hjelm, J. Li and F. Gao, Towards sustainable perovskite light-emitting diodes, Nat. Sustain., 2025, 8(3), 315–324 CrossRef.
- J. Cao, X. Zhang, Y. Miao, W. Li, X. Zeng, S. Yang, C. Yan, J. Lu and W. Yang, Interactions between H2O and lead halide perovskites: Recent progress and applications, Matter, 2024, 7(11), 3728–3755 CrossRef CAS.
- Y. Guo, P. Yang, F. Dong, X. Zhang, Q. Zheng, Y. Jiang, H. Wang and H. Wang, Quantum-Well Encapsulation Enabling Air-Processed Quasi-2D Perovskites with Long-Term Stability for Large-Area Deep-Blue Light-Emitting Diodes, Adv. Funct. Mater., 2025, e09277 Search PubMed.
- K. Liu, Y. Luo, Y. Jin, T. Liu, Y. Liang, L. Yang, P. Song, Z. Liu, C. Tian, L. Xie and Z. Wei, Moisture-triggered fast crystallization enables efficient and stable perovskite solar cells, Nat. Commun., 2022, 13(1), 4891 CrossRef CAS PubMed.
- J. Tang, W. Tian, C. Zhao, Q. Sun, C. Zhang, H. Cheng, Y. Shi and S. Jin, Imaging the Moisture-Induced Degradation Process of 2D Organolead Halide Perovskites, ACS Omega, 2022, 7(12), 10365–10371 CrossRef CAS PubMed.
- S. He, F. Yuan, P. Zhu, X. Wang, F. Ali, S. Zhang, P. Wu, W. Deng and Z. Wu, Multifunctional synergy of polydentate ligands enables high-performance all-bromine sky-blue perovskite light-emitting diodes, Chem. Eng. J., 2026, 529, 172867 CrossRef CAS.
- Y. Liu, T. Bu, L. K. Ono, G. Tong, H. Zhang and Y. Qi, Phase Aggregation Suppression of Homogeneous Perovskites Processed in Ambient Condition toward Efficient Light-Emitting Diodes, Adv. Funct. Mater., 2021, 31(32), 2103399 CrossRef CAS.
- Y. Duan, F. E. Oropeza, X. Jin, O. Amargós-Reyes, Y. Atoini, L. M. Cavinato, G. N. Nagy, M. U. Kahaly, V. A. de la Peña O'Shea, D. Y. Wang and R. D. Costa, Holy Water: Photo-Brightening in Quasi-2D Perovskite Films under Ambient Enables Highly Performing Light-Emitting Diodes, Adv. Funct. Mater., 2022, 33(7), 2209249 CrossRef.
- S. Yang, W. Bo, J. Zhou, Y. Ji, Y. Zhang, Y. Hu, W. Zhang, J. Tang, S. Li, H. Wang, Q. Wang, X. He and H. Xu, Regulating the interaction between perovskite precursor solution and antisolvent for efficient air-processed blue perovskite light-emitting diodes, Chem. Eng. J., 2025, 522, 168194 CrossRef CAS.
- H. Wang, Y. Tong, H. Qi, K. Wang, M. Fu and H. Wang, Water assisted chlorination enables ambient air fabrication of mixed halide perovskite films for blue light emitting diodes, J. Lumin., 2023, 263, 120114 CrossRef CAS.
- Y. Guo, P. Yang, F. Dong, H. Li, J. Gao, Z. Cheng, J. Wu, Y. Xu, H. Wang and H. Wang, Lattice Stabilized and Emission Tunable Pure-Bromide Quasi-2D Perovskite for Air-Processed Blue Light-Emitting Diodes, Adv. Sci., 2024, 12(5), 2414499 CrossRef PubMed.
- H. Li, J. Wu, S. Hu, W. Zhang, J. Gao, Y. Guo, P. Guo, C. Liu, G. Zhao, M. Fu, H. Bian, T. Liu, H. Wang and H. Wang, Air-Processed Blue Perovskite Light-Emitting Diodes Enabled by Manipulation of Adsorbed-Moisture-Dominated Crystallization Kinetics, Adv. Funct. Mater., 2023, 33(44), 2303787 CrossRef CAS.
- W. Li, T. Li, Y. Tong, Y. Li, H. Wang, H. Qi, K. Wang and H. Wang, Reducing Nonradiative Losses of Air-Processed Perovskite Films via Interface Modification for Bright and Efficient Light Emitting Diodes, Adv. Funct. Mater., 2023, 34(7), 2311133 CrossRef.
- Y. Guo, H. Li, P. Yang, J. Gao, Z. Cheng, F. Dong, J. Wu, H. Wang and H. Wang, Perfluorooctanoic acid interface modification enabling efficient true-blue perovskite light-emitting diodes and air-processing compatibility, Chem. Eng. J., 2024, 490, 151764 CrossRef CAS.
- J. Wu, H. Li, Y. Yang, Y. Chen, Z. Wang, F. Dong, X. Liu, Y. Guo, T. Yao, Y. Xu, Q. Ye, H. Wang, H. Wang and Y. Fang, Bilateral Embedded Anchoring via Tailored Polymer Brush for Large-Area Air-Processed Blue Light-Emitting Diodes, Angew. Chem., Int. Ed., 2024, e202411361 CAS.
- F. H. Isikgor, S. Zhumagali, L. V. T. Merino, M. De Bastiani, I. McCulloch and S. De Wolf, Molecular engineering of contact interfaces for high-performance perovskite solar cells, Nat. Rev. Mater., 2022, 8(2), 89–108 CrossRef.
- A. Dučinskas, G. Y. Kim, D. Moia, A. Senocrate, Y.-R. Wang, M. A. Hope, A. Mishra, D. J. Kubicki, M. Siczek, W. Bury, T. Schneeberger, L. Emsley, J. V. Milić, J. Maier and M. Grätzel, Unravelling the Behavior of Dion-Jacobson Layered Hybrid Perovskites in Humid Environments, ACS Energy Lett., 2020, 6(2), 337–344 CrossRef.
- S. Liu, J. Li, W. Xiao, R. Chen, Z. Sun, Y. Zhang, X. Lei, S. Hu, M. Kober-Czerny, J. Wang, F. Ren, Q. Zhou, H. Raza, Y. Gao, Y. Ji, S. Li, H. Li, L. Qiu, W. Huang, Y. Zhao, B. Xu, Z. Liu, H. J. Snaith, N.-G. Park and W. Chen, Buried interface molecular hybrid for inverted perovskite solar cells, Nature, 2024, 632(8025), 536–542 CrossRef PubMed.
- Z. Chu and J. You, Blue Light-Emitting Diodes Based on Pure Bromide Perovskites, Adv. Mater., 2025, 37(25), 2409718 CrossRef CAS PubMed.
- H. Li, S. Hu, H. Wang, X. Zhang, Y. Tong, H. Qi, P. Guo, G. Zhao, J. Gao, P. Liu, J. Zang, H. Hao, T. Liu, H. Bian, Y. Zhang, Y. Wei, Y. Guo, L. Zhang, Y. Fang and H. Wang, Control of n-Phase Distribution in Quasi Two-Dimensional Perovskite for Efficient Blue Light-Emitting Diodes, ACS Appl. Mater. Interfaces, 2023, 15(7), 9574–9583 CrossRef CAS PubMed.
- C. Liu, Y. Liu, S. Wang, J. Liang, C. Wang, F. Yao, W. Ke, Q. Lin, T. Wang, C. Tao and G. Fang, Highly Efficient Quasi-2D Green Perovskite Light-Emitting Diodes with Bifunctional Amino Acid, Adv. Opt. Mater., 2022, 10(13), 2200276 CrossRef CAS.
- Q. Cheng, B. Wang, G. Huang, Y. Li, X. Li, J. Chen, S. Yue, K. Li, H. Zhang, Y. Zhang and H. Zhou, Impact of Strain Relaxation on 2D Ruddlesden–Popper Perovskite Solar Cells, Angew. Chem., Int. Ed., 2022, 61(36), e202208264 CrossRef CAS PubMed.
- S. Yuan, B. Han, T. Fang, Q. Shan and J. Song, Flat, Luminescent, and Defect-Less Perovskite Films on PVK for Light-Emitting Diodes with Enhanced Efficiency and Stability, ACS Appl. Electron. Mater., 2020, 2(11), 3530–3537 CrossRef CAS.
- L. Liu, Y. Yang, M. Du, Y. Cao, X. Ren, L. Zhang, H. Wang, S. Zhao, K. Wang and S. Liu, Self-Assembled Amphiphilic Monolayer for Efficient and Stable Wide-Bandgap Perovskite Solar Cells, Adv. Energy Mater., 2022, 13(4), 2202802 CrossRef.
- P. A. Christy, A. M. F. Benial, A. John Peter and C. W. Lee, Structural and spectroscopic studies of bromofullerene, J. Alloys Compd., 2019, 780, 202–211 CrossRef CAS.
- Y. An, N. Zhang, Z. Zeng, Y. Cai, W. Jiang, F. Qi, L. Ke, F. R. Lin, S. W. Tsang, T. Shi, A. K. Y. Jen and H. L. Yip, Optimizing Crystallization in Wide Bandgap Mixed Halide Perovskites for High-Efficiency Solar Cells, Adv. Mater., 2023, 36(17), 2306568 CrossRef PubMed.
- A. Ummadisingu and M. Grätzel, Revealing the detailed path of sequential deposition for metal halide perovskite formation, Sci. Adv., 2018, 4, e1701402 CrossRef PubMed.
- S. He, F. Yuan, P. Zhu, F. Ali, H. Tang, S. Zhang, P. Wu, W. Deng, H. Dong and Z. Wu, Divergent Optoelectronic Tuning in Sky-Blue Quasi-2D Perovskites via Spacers Conjugation and Fluorination, Adv. Funct. Mater., 2025, e23282 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |