Open Access Article
Open Access ArticleLignin-based biopharmaceuticals from nature to medicine: current trends, future prospects and emerging challenges
Kshitij Rawat
a,
Ravneet Kaura,
Anil Kumar Pujaria,
Kunal Gogdea,
Devesh Mohnea,
Seema Kirara,
Yeddula Nikhileshwar Reddy†
 a,
Pooja Ramji Yadava,
Prakash Y. Khandave
a,
Pooja Ramji Yadava,
Prakash Y. Khandave b,
Abhay H. Pande
b,
Abhay H. Pande b and
Jayeeta Bhaumik
b and
Jayeeta Bhaumik *a
*a
aBRIC National Agri-Food and Biomanufacturing Institute (BRIC NABI), Department of Biotechnology (DBT), Government of India, Sector 81 (Knowledge City), S.A.S. Nagar 140306, Punjab, India. E-mail: jayeeta@ciab.res.in; jbhaumi@gmail.com
bDepartment of Biotechnology, National Institute of Pharmaceutical Education and Research (NIPER), Sector 67, S.A.S. Nagar, Punjab 160062, India
First published on 17th February 2026
Abstract
Lignin, a renewable and widespread aromatic biopolymer constituting 15–40% of the dry weight of lignocellulosic biomass, is a principal component of plant cell walls. Its inherent properties, including biocompatibility, antioxidant potential, antimicrobial activity, UV absorption, and higher mechanical strength, make it a highly promising sustainable material. This review comprehensively describes the potential of lignin for biopharmaceutical products by corroborating its fundamental properties, sources, and biomedical applications. A major focus is on the critical translational challenges, including detailed discussions of physicochemical properties, toxicological implications, safety assessment, and regulatory hurdles. Lignin-based biopharmaceuticals hold great promise for sustainable healthcare, but their safe and successful commercialization requires more comprehensive research and the establishment of clear regulatory frameworks to ensure the safe commercialization and sustainable development of lignin-based healthcare solutions urgently.
1. Introduction
1.1 Overview of lignin
Lignin is a highly complex polyphenolic biopolymer that has gained interest as a sustainable material for diverse industrial and biopharmaceutical applications due to its diversity in structure and potential utilization. It is the second most abundant natural polymer and a principal component of lignocellulosic biomass.1–3 More than 50 million tons of lignin-based materials and chemicals are produced every year globally, showing the abundance of lignin and its potential for large-scale utilization.41.2 Extraction and types of lignin
The extraction and solubilization of lignin are challenging due to its structural complexity and poor solubility in most conventional solvents.5 Lignin's solubility depends on the method used in the delignification of biomass.5 In the Kraft process, Kraft lignin (KL) is extracted from biomass using an aqueous solution containing sodium hydroxide and sodium sulfide, yielding a lignin product with a relatively low ash content. Additionally, KL is soluble in alkali solutions and various polar organic solvents due to its sulfur content. In KL, G-type units are dominantly present, as well as its high phenolic content, which may indicate that it would contain many reactive potential active sites for further functionalization, cross-linking, or polymerization.6 On the other hand, for the extraction of alkali lignin, the soda process utilizes alkaline agents such as sodium hydroxide, potassium hydroxide, or calcium hydroxide to hydrolytically break the linkage between lignin and carbohydrates. The surface functionality of alkali lignin is dominated by phenolic hydroxyl (–OH), aliphatic hydroxyl, and methoxyl (–OCH3) groups, which are crucial for its surface-active and antioxidative properties. The organosolv process, which employs organic solvents under milder conditions, yields lignin of higher purity with lower sulfur content, and makes more suitable for biomedical uses. Organosolv lignin with a higher S-type unit content exhibits better reducing power, resulting in greater antioxidant properties.7 Modern techniques such as solvent extraction, membrane ultrafiltration, and enzymatic fractionation are increasingly employed to obtain native-like lignin with improved structural homogeneity and biocompatibility.8 These approaches are particularly valuable because industrial or technical lignin, although abundant, often contains sulfur and metal impurities that limit reproducibility and make it unsuitable for direct biomedical use without further purification.9,101.3 Structural characteristics and monomeric units
Structurally, lignin is an amorphous, highly branched polymer composed of phenolic monomers, predominantly p-coumaryl alcohol, coniferyl alcohol, and sinapyl alcohol. These monomers polymerize into p-hydroxyphenyl, guaiacol, and syringyl units (Fig. 1). Lignin's molecular complexity arises from different chemical bonds that connect its monomers, such as β-O-4, 5-5, β-5, β-1, 4-O-5, and β-β linkages.11 The relative abundance of H, G, and S monomers varies depending on the plant source. The variation in the lignin monomer ratio leads to different functional group densities.12 Different types of lignin and their monomer unit ratios are accountable for the variations in their activity, such as antioxidant and antimicrobial (Table 1). Softwoods are typically rich in guaiacyl units, while hardwoods contain both guaiacyl and syringyl units.13 For example, Sun et al. observed that very high total phenolic –OH (3.28 mmol g−1) and –COOH (3.03 mmol g−1) contents lead to superior reducing ability, nucleation density, and metal coordination, resulting in well-dispersed, uniform nanoparticles. Low or fewer accessible phenolic/carboxyl sites and greater steric hindrance produce less uniform nanoparticles.13 These observations suggest that variation in monomer composition plays a significant role in determining functional group density and application.| Types of lignin | Solubility | M. wt (Da) | Zeta potential (mV) | H/G/S ratio | Dominant unit | Antioxidant activity | Antimicrobial activity |
|---|---|---|---|---|---|---|---|
| Kraft lignin | Alkali, organic solvents | 3700–19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
−40 to −45 | 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 (softwood KL) 5 (softwood KL) |
Guaiacyl | Moderate | Moderate to high |
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45 (hardwood KL) 45 (hardwood KL) |
|||||||
| Lignosulfonate | Water | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000–60 000–60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
−25 to −30 | 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 (softwood) 5 (softwood) |
— | High | Moderate |
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45 (hardwood) 45 (hardwood) |
|||||||
20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 (grass-based) 30 (grass-based) |
|||||||
| Soda lignin | Alkali | 1300–1400 | −20 to −40 | 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 (highly variable depending on the source) 30 (highly variable depending on the source) |
Mixed (H/G) | Very high | High |
| Organosolv | Organic solvents | 4100–10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
−8 to −12 | 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 (softwood) 5 (softwood) |
Syringyl | High | Moderate |
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 55 (hardwood organosolv) 55 (hardwood organosolv) |
Analytical characterization plays a key role in elucidating this complexity. Techniques such as gel permeation chromatography (GPC), 31P NMR, and FT-IR spectroscopy have been utilized to characterize the variety of lignin. They provide insight into the complex and heterogeneous structure of lignin, including its molecular weight, functional groups, and chemical structure. Other techniques, such as UV-vis spectroscopy, SEM/TEM, and XPS, can be used in assessing conjugation and chromophore content, morphological evaluation, and surface chemical analysis. Together, these techniques provide critical insight into lignin's heterogeneity and enable correlations between the molecular structure and biomedical performance.13
1.4 Physicochemical and biological properties relevant to biopharmaceutical use
Lignin's structural intricacy imparts a range of physicochemical and biological functionalities critical for biomedical applications. Its aromatic rings and phenolic hydroxyl and methoxy groups contribute to strong antioxidant and antimicrobial properties. Additionally, the phenolic groups found in lignin may interfere with the bacterial cell membrane, resulting in cell lysis and enhancing the antimicrobial activity.14Lignin has practical application as a substitute for traditional and chemical ingredients in numerous pharmaceuticals based on UV-blocking properties.15–17 Besides biomedical uses, the antioxidant properties of lignin make it a promising additive in food and cosmetic products, where it can prevent oxidative reactions, slow down ageing processes, and enhance product stability.15,16,18
The antioxidant activity of lignin arises from the presence of phenolic hydroxyl (–OH) groups in its structure. These phenolic moieties act as hydrogen atom donors, neutralizing reactive oxygen species (ROS) and free radicals by converting them into more stable, non-reactive molecules. In this process, a phenoxy radical is formed, which is stabilized through resonance across the aromatic lignin backbone, thus preventing further radical propagation. Additionally, the conjugated π-electron system of lignin facilitates electron delocalization, enhancing its radical scavenging efficiency.19,20
The antibacterial activity of lignin is due to the interaction of lignin with microbial cellular structure. The hydrophobic aromatic groups of lignin associate with phospholipid bilayers in bacterial membranes, increasing membrane permeability and causing leakage of intracellular components. Additionally, low-molecular-weight phenolic groups released during lignin degradation can interfere with microbial metabolic enzymes, impair ATP synthesis, and induce oxidative stress within the cell. Consequently, these effects compromise membrane integrity, inhibit cellular respiration, and ultimately lead to pathogen growth inhibition.21,22 All these features make lignin a potential candidate for numerous biomedical applications, such as anti-aging treatments, wound healing, and tissue engineering.
1.5 Suitability for biomedical use and purification needs
Lignin's polyphenolic structure and abundance of functional groups, including hydroxyl, methoxy, and carboxyl moieties, make it an attractive biopolymer for biomedical materials and applications, particularly due to its antioxidant activity and chemical versatility. However, not all types of lignin are suitable for biomedical or biopharmaceutical use in their native form. Technical lignin such as alkali lignin, KL, soda lignin, and LS often contains sulfur residues, metal ions, and other process-related impurities, which contribute to chemical heterogeneity and limited reproducibility.10Recent studies focused on different types of lignin, such as those extracted using organosolv, enzymatic hydrolysis, and mild extraction or fractionation processes, which demonstrated structural homogeneity, low impurity, and high biocompatibility. In addition to their properties, high antioxidant potential makes them suitable for biomedical applications.23,24 Organosolv lignin is more suitable for biomedical applications due to its relatively low molecular weight and high content of phenolic hydroxyl groups, which enhance antioxidant activity through efficient radical scavenging. Its low sulfur content and high chemical purity improve hydrophobicity, interfacial packing, and colloidal stability during nanoparticle or emulsion formulation. In contrast, other types of technical lignin typically exhibit higher molecular weight, greater polydispersity, and higher impurity levels, leading to reduced antioxidant efficiency and poorer formulation stability.25 Techniques like solvent extraction, membrane ultrafiltration, or enzymatic treatment can be utilized to attain extra purity, maintaining the quality and biocompatibility of lignin.8
Despite encouraging laboratory findings, most lignin-based biomedical applications continue at the laboratory or proof-of-concept stage. The industrial scalability and regulatory amenability of lignin-derived biomaterials still need to be authenticated. Thus, recognizing and optimizing lignin extraction and purification approaches remain a critical upstream research challenge for understanding the biomedical potential of lignin and improving the overall economic viability of lignin valorization.
This review comprises recent studies on emergent interest in lignin-based biopharmaceutical as sustainable alternatives to conventional pharmaceuticals. By linking lignin's chemical versatility and biological functionality with formulation design, this review aims to provide a framework for the rational development of lignin-derived materials in pharmaceutical sciences. This review offers insights to provide a new path for potential applications of different lignin-based formulations (solid, semi-solid, and liquid) in various biotherapeutic applications, such as drug delivery and tissue regeneration. As lignin has unique properties such as UV absorption capacity, minimal cytotoxicity, biodegradability, biocompatibility, antioxidant activity, and antibacterial activity, there is great market potential for lignin-based formulations. Despite challenges in applying lignin in pharmaceutical industries, the implications of necessary regulatory approvals can serve as a game-changer in lignin-based biopharmaceuticals.
2. Biopharmaceutical relevance of lignin
Lignin's biological activity extends far beyond its structural role in plants. Its antioxidant, antimicrobial, antiviral, and anticancer properties, together with its mechanical robustness and biocompatibility, make it a promising natural polymer for biomedical innovation.26 The following subsections outline key areas in which lignin and its derivatives contribute to therapeutic and diagnostic applications.2.1 Antimicrobial potential
Lignin exhibits broad-spectrum antimicrobial properties against bacteria and fungi owing to its phenolic hydroxyl groups and aromatic moieties that disrupt microbial membranes and metabolic processes.27 For instance, lignin extracted from Caesalpinia pulcherrima leaves exhibited antifungal activity against various Candida species.28 Lignin from eucalyptus and spruce has demonstrated antifungal activity against Aspergillus niger, with KL exhibiting more potent activity than organosolv lignin.2.2 Antiviral potential
The antiviral potential of lignin has also been widely recognized. Low-molecular-weight lignin fractions suppress HIV-1 replication by interfering with viral transcription.29 Lignin from the paper industry was used as a coating material to inactivate herpes simplex virus type 2 (HSV-2) by generating reactive oxygen species.30 Additionally, lignin has shown effectiveness against herpes simplex virus (HSV-1), with lignosulfonic acid, a lignin derivative, exhibiting antiviral properties against a wide range of viruses.29 In this context, lignin extracted from Lentinus edodes mycelium has demonstrated inhibition against hepatitis C virus infection, and it was also found to inhibit influenza virus.31 Besides its antiviral properties, lignin has also been investigated for its potential to purify water from viral contaminants. Colloidal lignin particles provide a sustainable solution to viral contamination, as they have the potential to aggregate viruses, facilitating their removal from water.322.3 Anticancer activity
Besides antimicrobial and antiviral effects, lignin and its derivatives show notable anticancer potential. Lignin and its derivatives have the potential to inhibit cancer cell proliferation and induce apoptosis.33 Combined with chemotherapeutic agents, lignin-based formulations can enhance the therapeutic efficacy of standard cancer therapies.34,35 Lignin also exhibits immunomodulatory properties, meaning that it can influence immune system activity to stimulate the body's natural defence mechanisms against tumors. Researchers are investigating the process by which lignin-linked adjuvants can augment the efficiency and effectiveness of many vaccinations or immunotherapy treatments.36 Overall, the phenolic structure of lignin underlies its diverse biological actions. It donates hydrogen atoms to neutralize free radicals, chelates metal ions to reduce oxidative stress, and interacts with biological membranes and proteins, providing a multifaceted defence mechanism relevant to infection control, oncology, and immunotherapy.2.4 Tissue engineering
The biocompatibility and mechanical strength of lignin make it an ideal material for use in tissue engineering and regenerative medicine scaffolds or hydrogels that support cell growth and drug delivery. It enhances the scope for various therapeutic applications, including wound healing, bone repair, and soft tissue regeneration.37,38 As the pharmaceutical industry moves toward more sustainable production practices, lignin is a promising candidate for replacing conventional, petroleum-based materials in various applications.392.5 Biosensing
Lignin is beginning to gain attention not just due to its structural and bioactive properties, but also due to its functional applications, e.g., in biosensor development. In biosensing, lignin exhibits redox activity because it contains phenolic, quinone, and hydroquinone functional groups. These redox-active moieties enable lignin to participate directly in electron transfer processes at the electrode–electrolyte interface. Through reversible electron–proton transfer reactions, lignin can act as an electron mediator between the electrode surface and the biorecognition element. When an analyte binds or an enzymatic reaction takes place, the local redox environment is altered, which in turn modulates the redox behavior of lignin. This modulation produces measurable changes in current, potential, or impedance, forming the basis for electrochemical signal transduction in lignin-based biosensing systems.40 These studies highlight that the source and processing of lignin, whether from softwood, hardwood, technical, or nano-particulate sources, are important in defining its reactivity, homogeneity, and biocompatibility.Together, these findings show that lignin has a wide range of useful biological activities that support its role in many therapeutic and diagnostic areas. Its antioxidant, antimicrobial, antiviral, anticancer, and tissue-supporting properties make it a valuable natural material for medical use. These features provide a strong base for developing lignin-based biopharmaceutical products, which are discussed in the next section.
3. Lignin-based biopharmaceutical products
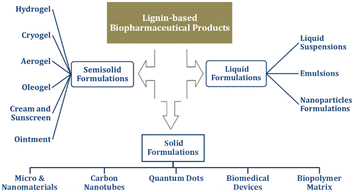
Lignin is now being actively used as a structural and functional component in the development of several biomedical formulations. Its inherent chemical complexity and tunability allow it to interact synergistically with other biomaterials, forming stable, responsive, and bioactive systems. Whether incorporated into gel-based matrices, dispersed in colloidal systems, or structured into solid-state platforms, lignin supports diverse therapeutic objectives such as drug stabilization, controlled release, wound healing, and tissue repair.Recent studies have demonstrated the feasibility of lignin-based hydrogels, emulsions, and nanoparticles in both therapeutic and diagnostic contexts.41,42 These formulations benefit from lignin's antioxidant, antimicrobial, and stabilizing capabilities, which enhance product functionality and therapeutic potential. Additionally, the emergence of technologies such as 3D printing and nanofabrication has opened new avenues for customizing lignin-based biomedical materials.43 These systems based on their physical form are classified into 3 types: semi-solid formulations, liquid formulations, and solid formulations, which provides an overview of different types of lignin-based biopharmaceuticals (Fig. 2).
3.1 Lignin in semisolid formulations
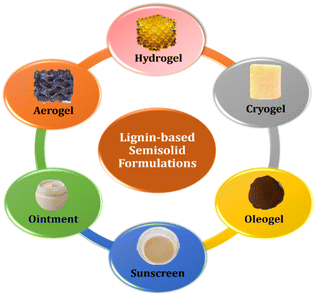
Semisolid formulations, including creams, gels, ointments, and pastes, play a crucial role in biopharmaceuticals, particularly for topical, transdermal, and mucosal drug delivery (Fig. 3). These formulations comprise a polymer matrix with the drug and excipients dispersed or dissolved in aqueous or non-aqueous bases. Typical formulations consist of a polymer matrix containing the drug and necessary excipients, either dissolved or suspended in aqueous or non-aqueous bases.44 Lignin has many advantages, such as antioxidant, UV protective, and antimicrobial, which make it a suitable choice in the development of semisolid formulations such as creams, ointments, and gels.45,46In semisolid formulations, lignin is a versatile ingredient due to its ability to act as a gelling agent, contributing to the desirable consistency and texture of the product.47 Lignin's inherent biocompatibility makes it suitable for skin care and pharmaceutical applications, minimizing the risk of adverse reactions.48 Lignin's compatibility with other formulation components allows for the production of stable and homogeneous products.49 When incorporated into semisolid formulations, lignin can enhance the rheological properties, improving spreadability and skin feel.50 Additionally, it can enhance the stability of proteins, such as enzymes or antibodies, extending the shelf life and efficacy of certain biopharmaceutical products.48,51 Lignin-based semisolid formulations in skincare may provide antioxidant benefits, helping to protect the skin against oxidative stress and providing anti-ageing effects.52 Additionally, the antimicrobial properties of lignin could be advantageous in formulations designed to combat skin infections.53
Currently, the development of lignin-based semisolid formulations is a promising approach to optimizing extraction processes, ensuring consistency in quality, and addressing challenges related to color and odor.54 As the industry continues to explore sustainable and innovative ingredients, lignin-based semisolid formulations present a convincing option for formulators seeking effective, eco-friendly, and versatile solutions in skincare and pharmaceutical applications.55
Lignin-based hydrogels can be synthesized through various methods, including interpenetrating polymer networks, crosslinking grafted lignin with monomers, atom transfer radical polymerization (ATRP), and reversible addition–fragmentation transfer (RAFT) polymerization (Table 2).58 These hydroxyl groups present on the lignin surface act as interfacial crosslinkers and promote hydrogen bonding with other biomolecules. The ionization state of lignin is highly pH-dependent, influencing its physicochemical and biological behavior. At acidic pH, the phenolic groups become protonated, resulting in a decrease in surface charge and an increase in hydrophobicity, which may enhance aggregation and cellular uptake in certain biological environments. Conversely, at basic pH, the phenolic groups in lignin become deprotonated, giving the surface a negative charge. This increases its hydrophilicity and promotes electrostatic interactions with positively charged biomolecules such as proteins and cell surfaces. These changes collectively modify the hydrophilic–hydrophobic balance of the system. This mechanism influences the hydrogel swelling behavior and the release kinetics.59 The aromatic ring structure of lignin exhibits strong π–π stacking interactions with hydrophobic drug molecules. These weak interactions regulate drug release kinetics.60
| Types of cross-linking | Role of lignin | Matrix | Crosslinker | Ref. |
|---|---|---|---|---|
| Lignin-based hydrogels by chemical methods | Lignin as a crosslinked unit | Lignin–amine | PEGDGE | 61 |
| Lignin–agarose | ECH | 62 | ||
| Lignin–xanthan | ECH | 63 | ||
| Lignin–PVA | ECH | 64 | ||
| Modified lignin–PVA | ECH | 65 | ||
| Lignin–Mt–acrylic acid | NMBA | 66 | ||
| Acrylic acid–OMt–grafted lignin | NEBA | 67 | ||
| Lignin as a crosslinking agent | PVA | Aminated lignin | 68 | |
| PMVE/MA | Lignin and lignin-PEG | 69, 70 | ||
| Polymerized acrylic acid–PVA | Sodium lignosulphonate | 71 | ||
| Lignin-based hydrogels by physical methods | Lignin as a crosslinked unit | Lignin and hydrophilic PU | N/A | 72 |
| Lignin as a crosslinking agent | Chitosan | Lignin | 73 | |
| Chitosan–PVA | Lignin | 74 |
In hydrogel systems based on lignin or natural polymers, and especially for biological use, lignin seldom plays the role of the primary structural matrix. Its role changes based on the nature of the formulation and polymer composition used. In the majority of the formulations, lignin functions as a bioactive additive or secondary crosslinking agent incorporated into polymeric matrices such as poly(vinyl alcohol), chitosan, gelatin, or polyethylene glycol. Through these interactions, lignin enhances the mechanical integrity, moisture-retention capacity, and antioxidant properties of the resulting gel, thereby contributing to its overall stability and functional performance.75,76 In some systems, lignin acts primarily as an antioxidant and antimicrobial, protecting the encapsulated drug or bioactive principles from oxidation, while supporting wound healing. In specifically designed lignin-rich formulations, lignin may also influence gelation kinetics and viscoelastic behavior when present in substantial amounts.23,24
Chandna et al. developed a sustainable lignin-based hydrogel incorporating RB@AgLNCs. The lignin-based hydrogel was prepared by crosslinking lignin, PVA, and polyacrylic acid (Fig. 4). This hydrogel coating showed remarkable antifungal activity and maintained effectiveness through photodynamic inactivation, where light-activated photosensitizers (RB) generate reactive oxygen species that damage fungal cells. Its water resistance, transparency, and strong adhesion suggest high potential for biomedical device disinfection.77
 | ||
| Fig. 4 The schematic diagram for the preparation of the lignin-based hydrogel. Adapted from ref. 57 with permission from the American Chemical Society, ACS Applied Polymer Materials, 2022, 4, 12, 8962–8976. Copyright 2022. | ||
Notably, lignin-based hydrogels also serve as essential bio-ink components for 3D printing and as structural matrices in wound dressing and scaffold fabrication. While their multifunctional applications are presented in subsequent sections (e.g., section 3.1.5), here we primarily emphasize their semi-solid dosage form characteristics, rheological properties, and fundamental hydrogel-forming behavior. However, hydrogels have poor mechanical strength and rapid drug release capacity, and are very difficult to handle. Lignin-derived hydrogels promote wound healing capacity in a moist environment and act as a scaffold for the regeneration of infected tissue. They are potential candidates for a targeted drug delivery system, biocompatible, and easy to customize for biotherapeutic applications.
Abudula et al. developed lignin-based injectable cryogels (lignin-co-gelatin cryogels) for biomedical purposes. The developed cryogels were evaluated against surgical site infection-causing pathogens S. aureus and E. coli.78 The synthesis of cryogels involves the alkali-assisted ultrasonication and homogeneous mixing of lignin–gelatin solution (Fig. 5). Further, cryogelation was done in the presence of glutaraldehyde as a cross-linker.78 Lin et al. also developed highly porous lignin-formaldehyde cryogels, which have shown enhanced thermal stability and unique thermal insulation.81 Lignin-derived cryogels are highly sustainable and biobased as compared to petroleum-based conventional cryogels. They are highly porous and lightweight, and have interconnected networks over conventional cryogels which require crosslinkers for building block construction. They also have wide application over biotherapeutics such as water adsorbents, supercapacitors, packaging, and insulation.
 | ||
| Fig. 5 Schematic illustration of lignin–gelatin cryogel preparation. Lignin was dispersed in aqueous gelatin via alkali-assisted ultrasonication, followed by cryogel formation using glutaraldehyde (GA) as a cross-linker during freezing; residual GA was removed during the thawing step. Reproduced from ref. 78 with permission from the American Chemical Society, Biomacromolecules, 2021, 22, 4110–4121. Copyright 2021. | ||
 | ||
| Fig. 6 3D printing (a) meshes and (b) oral capsules. Reproduced from ref. 91, J. Domínguez-Robles et al., Pharmaceutics, 2019, 11, 165, under the terms of the Creative Commons CC-BY 4.0 licence. | ||
Generally, lignin-based creams are prepared as oil-in-water (O/W) emulsions, where lignin is dispersed in the aqueous phase prior to emulsification. Normally, lignin is dissolved in an alkaline aqueous solution (pH 7.5–8.5) and then subjected to ultrasonication. This aqueous lignin phase is heated to 70–75 °C to maintain the same temperature as the oil phase (e.g., fatty alcohols, esters, and emollients), which is heated to 70–75 °C. The two phases are then mixed under homogenization (4000–10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm) to form the emulsion. To stabilize the cream structure, slow cooling under continuous stirring is needed. The aromatic and phenolic groups of lignin interact with emulsifiers and polymeric thickeners through hydrogen bonding and π–π interactions, contributing to viscosity, SPF enhancement, and antioxidant stability. Critical process parameters that influence the texture, spreadability, and uniformity of lignin dispersion include pH (typically maintained between 5.0 and 6.0 for skin compatibility), homogenization speed, and cooling rate.
000 rpm) to form the emulsion. To stabilize the cream structure, slow cooling under continuous stirring is needed. The aromatic and phenolic groups of lignin interact with emulsifiers and polymeric thickeners through hydrogen bonding and π–π interactions, contributing to viscosity, SPF enhancement, and antioxidant stability. Critical process parameters that influence the texture, spreadability, and uniformity of lignin dispersion include pH (typically maintained between 5.0 and 6.0 for skin compatibility), homogenization speed, and cooling rate.
In one of the studies conducted by Antunes et al., lignin was extracted from sugarcane bagasse. The extracted lignin was utilized to prepare a multifunctional blemish balm (BB) cream. The experimental in vitro and in vivo results predicted its broad-spectrum UV protection, cytotoxicity, skin allergenicity, genotoxicity, and acute skin irritation, which was safe for topical applications.50 Additionally, lignin-based sunscreens have gained enormous interest in developing cosmeceutical formulations.14 One of the studies exhibited that lignin has enhanced synergistic properties with the marketed sunscreen. The resulting formulation has boosted the SPF with good sunscreen performance.93 Conclusively, lignin has a large number of phenolic groups in its structure, which is responsible for its anti-inflammatory and skin-calming effects. Consequently, it showed potential benefits for acne, dermatitis, sunburn, redness, and irritation in biotherapeutic applications.
One such study reported by Mahata et al. describes the development of a lignin-graft-polyoxazoline conjugated triazole-based efficient ointment having anti-inflammatory solid activities due to lignin's inherent antioxidant and antimicrobial properties. The study was carried out on biofilm and animal models infected with Pseudomonas aeruginosa HW01-induced burn-wound healing.95 The aqueous-based gel has been incorporated with lignin and exhibited better rheology and drug loading. Further, the in vivo study suggested its safe use in skin burns to avoid secondary infections.97 Lignin-based ointments can be improved further for wound care and controlled drug delivery. Small changes in lignin size, surface treatment, and base composition can enhance spreadability and release behavior. Adding bioactive agents or nanoparticles may strengthen antimicrobial and healing effects. With simpler and greener processing, these ointments can become reliable options for future topical therapies.
3.2 Lignin-based liquid formulations
Nanoformulations in biopharmaceuticals are particularly used for the delivery of poorly water-soluble drugs, biological macromolecules, and nanoparticle-based therapeutics. These formulations consist of finely dispersed drug particles in a liquid medium to prevent sedimentation and aggregation. They offer several advantages, such as ease of administration, rapid action, and avoiding difficulty in swallowing.98,99 The engineering of lignin properties has gained significant attention due to its potential for wide-range applications. For a material to be suitable for industrial use, it must be widely available and cost-effective, exhibit good technological performance, and possess low toxicity. Lignin fulfils many of these criteria and has been utilized in various liquid formulations, such as emulsions, suspensions, and formulations, highlighting its versatility. These properties allow lignin-based liquid systems to serve as carriers for therapeutic molecules, excipients in dermatological and nutraceutical preparations, and stabilizers in injectable formulations. | ||
| Fig. 7 The schematic representation of the synthesis of the lignin nanospray using lignin. Reproduced from ref. 103 with permission from The Royal Society of Chemistry, Journal of Materials Chemistry B, 2021, 9, 1592–1603, Copyright 2021. | ||
Lignin-based emulsions can be used as a template for the synthesis of polymers with porous architectures, such as fuel emulsions, organic carriers, and others.107,108 Emulsion investigations were conducted using LS and KL with varying molecular weights. However, their use in emulsions is restricted due to LS's high sulfur content and KL's poor solubility.109,110 KL shows low surface activity and exhibits a hydrophilic character at high pH. To overcome the hydrophobicity and low charge density of KL, it was reacted with tannic acid under alkaline conditions. KL–tannic acid acts as a green emulsifier/stabilizer for oil-in-water emulsions (Fig. 8).111 The bioavailability of lipophilic therapeutic molecules such as curcumin, essential oils, and hydrophobic anticancer agents can be enhanced through encapsulation within lignin-based systems, where lignin-stabilized emulsions improve their dispersion, stability, and sustained-release behavior. The research was carried out to determine the behaviour of emulsions stabilized by polymer-grafted lignin particles, in which lignin particles were grafted by RAFT using two distinct polymers of polyacrylamide and polyacrylic acid.112 Technological characteristics of lignin as an emulsifier were determined to add value to this biopolymer for water purification, water treatment, food, cosmetic, paint, biomedical, chemical, and agricultural industries.107,113 In another study, the reactor formed stable oil-in-water emulsions.114 Recently, researchers looked at how well water-soluble carboxymethylated lignin produced stable kerosene in a water emulsion. They observed that lignin particles with a high degree of substitution could keep emulsions reasonably stable throughout various conditions.115 Lignin-based emulsifiers can lead to safer and more sustainable emulsion systems for medical, cosmetic, and industrial use. Chemical modification of lignin can further improve its surface activity and compatibility with different oils and solvents. Better control of particle size and charge may enhance emulsion stability and drug-loading ability. These advances can support future applications in therapeutic delivery, functional foods, and eco-friendly formulations.
 | ||
| Fig. 8 Tannic acid and KL as a stabilizer for oil-in-water emulsions. Reproduced from ref. 113 with permission from the American Chemical Society, ACS Sustainable Chem. Eng., 2019, 7, 2370–2379, Copyright 2018. | ||
 | ||
| Fig. 9 Synthesis of lignin-based nanocarriers. Reproduced from ref. 121, Gigli et al., Frontiers in Plant Science, 2022, 13, 976410, under the terms of the Creative Commons CC-BY 4.0 licence. | ||
3.3 Lignin-based solid formulations
Solid formulations have gained significant attention in biopharmaceutical applications due to their ability to enhance therapeutic agents' stability, efficacy, and patient compliance. They offer better shelf life and reduced susceptibility to environmental degradation.124 Advances in formulation science have also enabled the integration of novel biomaterials, including biopolymers such as lignin, into solid dosage forms, enhancing biocompatibility and enabling eco-friendly drug delivery platforms. These discoveries are opening new opportunities for using lignin in both materials science and biomaterials development. Different types of lignin-based solid formulations have been developed for pharmaceutical applications such as drug delivery, bioimaging agents, etc.Many drugs face limitations in clinical use due to poor water solubility and adverse effects on normal tissues. To address this, improving targeted delivery and tissue penetration is crucial. Liu et al. developed folic acid–polyethylene glycol–alkaline lignin conjugates (FA-PEG-AL) to create targeted nanoparticle vehicles for delivering hydroxyl camptothecin (HCPT).128 Chen et al. demonstrated that various lignin types effectively dissolve poorly water-soluble drugs like doxorubicin and gatifloxacin in a hydrotropic system. This method achieves 90% drug encapsulation efficiency, making it suitable for scalable, high-yield production of drug-loaded lignin nanoparticles.129
Figueiredo et al. prepared lignin nanoparticles complexed with iron and magnetic Fe3O4-infused lignin nanoparticles (Fe3O4-LNPs) to assess their potential in nanomedicine. Fe3O4-LNPs showed the most promising results for cancer therapy and diagnosis. While high concentrations of these nanoparticles can cause cytotoxicity and haemolysis, no significant toxicity was observed at concentrations up to 100 mg mL−1, suggesting their suitability for nanomedicine with controllable concentration and time-dependent effects.130 Dai et al. loaded trans-RSV (insoluble anticancer drug) with Fe3O4 into lignin nanoparticles for passive and magnetic targeting. In mice, AL/RSV/Fe3O4 NPs retained more trans-RSV in tumours than free trans-RSV. It was found that the developed NPs effectively inhibited tumor growth and improved survival rates, with the latter benefiting from sustained drug release, optimal particle size, magnetic properties, and superior biocompatibility and dispersibility.131 Figueiredo et al. conjugated lignin nanoparticles with Asp-Ser-Ser (DSS) polypeptide, known for its cell-penetrating properties, to improve interactions between cells and lignin nanoparticles. The DSS polypeptides were chemically conjugated to carboxyl groups on lignin nanoparticles using EDC/NHS coupling chemistry in a mild buffer, preserving loading efficiency. The developed system improved the dissolution rate and stability under tumor-like conditions, enhancing cellular uptake and antiproliferative activity in both 2D and 3D tumor models.130
Nanoparticle formulations can enhance drug bioavailability and promote quick oral absorption of poorly soluble drugs. Lignin, which is insoluble in acidic solutions but soluble under alkaline conditions, holds potential for targeted drug release in the intestine. Alqahtani et al. developed a formulation for curcumin by cross-linking lignin in a citrate buffer. This achieved excellent stability under gastric conditions and sustained release in simulated intestinal fluid. This formulation improved curcumin permeability and bioavailability compared to the curcumin suspension.132 Lignin nanoparticles demonstrate enhanced thermal performance and stability across a broad pH range and exhibit antiproliferative effects against cancer cells. They are also incorporated into cosmetics, particularly sunscreens, for UV protection. Lignin nanomaterials can be further refined for precise targeting, improved penetration, and safer delivery of difficult-to-solubilize drugs. Combining lignin with peptides, metals, or polymers may expand their use in cancer therapy, imaging, and oral drug systems. These advances position lignin nanomaterials as strong candidates for future biomedical and pharmaceutical technologies.
These materials benefit from lignin's renewable origin and inherent surface functionality, which differentiates them from conventionally synthesized CNTs derived from fossil-based precursors. Compared to conventional CNTs, lignin-based CNTs often exhibit higher surface functionality and defect density, thereby enhancing dispersibility and interfacial interactions. These features are particularly advantageous for biomedical applications such as drug delivery, imaging, and tissue engineering. With their high surface area and potential for targeted drug release, lignin-based CNTs offer new possibilities for improving therapeutic efficacy and precision. Additionally, their unique properties, including antioxidant and antimicrobial effects, are significantly effective in wound healing and infection control. This combination of sustainability and performance positions lignin-based CNTs as a ground-breaking solution for developing next-generation biopharmaceutical products.134,135 However, lignin-based CNTs have promising applications in drug delivery systems, tissue engineering, and biosensor development. They also have a lower environmental impact, generating fewer greenhouse gases, promoting a circular bioeconomy, and reducing waste compared to conventional CNT production.
Chen et al. produced lignin-based carbon dots through hydrothermal treatment with an H2O2–Fe2+/Fe3+ system for bioimaging. The optimal carbon dots exhibited strong blue photoluminescence at 430 nm when excited at 320 nm and maintained stable fluorescence intensity for over 100 minutes, outperforming CdTe quantum dots in stability.140 Niu et al. developed lignin-based carbon dots for cellular imaging. These carbon dots formed through molecular aggregation during dialysis, averaging 2.4 nm in diameter. After assessing potential cytotoxicity, carbon dots were tested for their cellular imaging capabilities in HeLa cells. The carbon dots were in cells within 1 hour, and they could be observed in the cell nuclei after three hours via fluorescence.141
Rai et al. developed carbon dots from LS lignin for cellular imaging and drug delivery with a high quantum yield of up to 47.3%. The synthesis process involved microwave irradiation followed by reduction with NaBH4. The presence of sulfur in LS enhanced fluorescence stability and the quantum yield.138 Myint et al. synthesized fluorescent carbon dots (CDs) from KL using the compressed liquid CO2 antisolvent method, which involves carbonization and oxidation. These dots exhibited bright yellow fluorescence under 458 nm excitation. CDs showed no cytotoxicity to HeLa cells at a 500 μg mL−1 concentration after 48 hours and maintained strong fluorescence.139 Lignin-based quantum dots can be advanced for safer bioimaging, real-time tracking, and combined imaging–therapy systems. Fine control over their size, surface groups, and emission range may enhance brightness and targeting accuracy. Their low toxicity also supports future use in in vivo diagnostics, biosensing, and image-guided drug delivery. These developments position lignin-derived quantum dots as promising tools for next-generation biomedical technologies.
Domínguez-Robles et al. utilized lignin as a vehicle for manufacturing drug-containing solid oral dosage forms. Their findings indicated that incorporating lignin could potentially alter the pattern of drug release. Additionally, the antioxidant properties of lignin could effectively protect the active ingredients from oxidative degradation within the formulation.144 Pishnamazi et al. found that Alcell lignin enhances the compressibility of microcrystalline cellulose/lactose monohydrate composites, improving flowability during roll compaction. It was observed that incorporating lignin as an excipient reduces disintegration time and accelerates drug release rates in aspirin pills. This is attributed to lignin's amorphous structure and unique interaction with aspirin, enhancing bioavailability.145 On the other hand, carboxylated lignin decreases pill hardness and accelerates the disintegration and release rates of paracetamol due to reduced interaction with the drug. Lignin-containing solid oral dosage forms also exhibit controlled release behaviour under simulated intestinal conditions, showcasing potential in pharmaceutical formulations.146 However, the biological performance of lignin in pill formulations requires further investigation across different therapeutic applications. In biomedical imaging, essential for detecting and treating diseases like cancer, lignin-based biomaterials play a role in photodynamic and photothermal therapies. They are synthesized with metal ions, carbon dots, or porphyrins in several steps to improve their functionality.137,147 In photodynamic therapy, lignin acts as a carrier or matrix for photosensitizers, enabling light-triggered reactive oxygen species generation. In photothermal therapy, lignin-based composites convert absorbed light into localized heat for therapeutic action when combined with suitable photothermal agents. Native lignin exhibits intrinsic autofluorescence mainly in the UV-visible region due to its aromatic structure. The near-infrared fluorescence used for bioimaging generally arises from incorporated photosensitizers or fluorescent dyes within the composite system, while lignin functions as a biocompatible carrier.148 In addition, the redox activity and surface functionality of lignin support its integration into biosensing platforms, where changes in optical or electrochemical signals can be used for analyte detection. Lignin-based materials can be further developed for safer and more efficient drug-delivery devices, microneedles, and wound-care systems. Their tunable chemistry may allow better control of release rates, mechanical strength, and imaging responses. Combining lignin with metals, polymers, or fluorescent agents could expand its use in biosensing and image-guided therapies. These improvements can help establish lignin as a reliable and sustainable component in future biomedical devices.
Electrospun fibres, known for their high surface area-to-volume ratios, are used in tissue engineering scaffolds for cell cultivation.149 Lignin, used as a polymeric additive in electrospinning, faces challenges in forming homogeneous and elastic nanofibers due to its incompatibility with nonpolar polymers like polylactic acid (PLA), polyhydroxybutyrate (PHB), and polycaprolactone (PCL).102 To address this, researchers like Kai et al. have developed methods to chemically graft polymer chains onto lignin, improving its compatibility with polymer matrices. Their lignin-graft-PLA copolymer, for example, creates well-dispersed, homogeneous nanofibers with diameters of 350–500 nm. These fibres show good compatibility with PLLA, antioxidant activity, and biocompatibility with various cell types. Other lignin-containing polymer nanofibers have also been developed, including combinations with PHB, PCL, and PMMA. Liang et al. have reported a PCL–lignin nanofiber membrane with intrinsic antioxidant activity, excellent mechanical properties, low cytotoxicity, and anti-inflammatory effects, showing promise for osteoarthritis treatment as an alternative to traditional antioxidants.150
Bone regeneration scaffolds, crucial in tissue engineering, greatly rely on mechanical stability. Rekola et al. developed a metal-free, fibre-reinforced composite bone material by heat-treating wood inspired by the porous structure of wood and bone. This treatment improved the osteoconductivity of the artificial implants, demonstrating lignin's good biocompatibility.151 Additionally, recent research found that unmodified PCL and lignin can form a uniform solution in chloroform/DMF, creating mineralized, hydroxyapatite-coated fibres through electrospinning. Lignin's reactive phenolic and aliphatic hydroxyl groups help in the chelation of Ca2+, crucial for hydroxyapatite formation, enhancing osteoblast adhesion and proliferation, making this platform a promising bone regeneration material.152 Reesi et al. synthesized a lignin-based nanofibrous dressing with arginine for wound healing, which mimics the extracellular matrix and accelerates wound closure in vivo. The combined antibacterial and antioxidant properties of lignin and controlled arginine release boost nitric oxide levels and collagen deposition, further enhancing wound healing properties.153 Lignin-based biopolymer matrices can be further optimized for stronger, more cell-responsive scaffolds in tissue repair. Simple chemical modifications may improve fibre formation, mineralization, and compatibility with other biomedical polymers. Their natural antioxidant and antimicrobial traits also support future use in smart wound dressings and regenerative implants. With better control of the structure and processing, lignin-derived matrices could become reliable, sustainable alternatives to conventional scaffold materials.
Lignin appears in multiple dosage form categories across this review; each section highlights a distinct functional state (semi-solid hydrogel, printable formulation, or scaffold matrix), supporting the rationale of classifying biomaterials based on their physical form and administration characteristics (Table 3). The lignin-derived biopolymer matrix inhibits bacterial growth, fungal colonization, and the formation of biofilms. Thus, there is a wide range of applications in food packaging films, biomedical scaffolds, and wound dressings, replacing chemical-based biopolymer matrices.
| Types | Matrix and processing method | Application | Main findings | Ref. |
|---|---|---|---|---|
| Hydrogel | Doxorubicin was conjugated on lignin–folic acid derived CDs via EDC-NHS reaction and loaded with PEGMA, PAA, and ZcPC-based hydrogels | Chemo-phototherapy of breast cancer | Lignin-derived photochemo-hydrogel showing synergistic effect for treatment of breast cancer | 41 |
| Cryogel | The cryogel was prepared by mixing lignin and gelatin using the orbital shaking method | Antibacterial, antioxidant, and intravenous delivery | The developed lignin-derived gelatin cryogel showed 7 times higher activity than the pure gelatin cryogel | 78 |
| Aerogel | CNF/lignin-based aerogel was developed by a crosslinking method | Antioxidant, antimicrobial, and sustained release properties | The CNF/lignin-based aerogel showed potent antimicrobial activity of tetracycline as well as sustained release properties | 83 |
| Oleogel | Lignin castor oil-based oleogel was prepared using a facile synthetic method that employed two saline coupling reagents | Antioxidation | The synthesized oleogel showed good oxidation properties when modified with 20 wt% of lignin | 87 |
| Sunscreen | Flavonoids containing lignin sunscreen using the coextraction method | Antioxidant and UV protectant | Musa basjoo and Moso bamboo leaves were used to extract flavonoid-containing lignin using L-cysteine (L-cys)-mediated extraction. Utilizing non-dewaxed sources might boost the flavonoid content of lignin | 154 |
| Ointment | Lignin-graft-polyoxazoline conjugated triazole: a novel anti-infective ointment | Antimicrobial and antibiofilm | In vivo tests of this innovative lignin-based hydrogel have demonstrated its potential as an anti-inflammatory dressing material, to promote healing, and to prevent infection of burn wounds | 95 |
| Emulsion | Lignin was dissolved in acetone/water, and then the cosolvent was added to MilliQ water | Drug vehicle and microencapsulation | LNPs prepared were shown to be a highly efficient stabilizer for oil-in-water pickering emulsions | 155 |
| Suspensions | The synthesis of lignin aqueous hydrotropic solution by a greener and facile method | Drug delivery | The synthesized LNP solution has potential for the delivery of drugs and bioactive compounds in biomedical applications | 129 |
| Nanoformulations | Synthesized nano and microparticles using a green synthetic method | Antioxidant | LPs showed good antioxidant activity against E. coli and Salmonella enterica, reducing the intracellular ROS level | 156 |
| CNTs | Developed nitrogen-doped lignin-derived carbon nanotubes by the pyrolysis method | Drug delivery | High photothermal conversion efficiency of 58.8% was obtained | 157 |
| Carbon dots | Lignin-based CQDs were prepared using a hydrothermal method | Bioimaging | L-CQDs exhibited low cytotoxicity and good cellular biocompatibility, demonstrating significant potential for bioimaging applications | 143 |
| Biomedical devices | GOx–SiO2/Lig system was integrated with single-walled carbon nanotubes and platinum nanoparticles | Biosensors for medical and bacterial sensing | The glucose biosensor presented a high sensitivity of 0.78 μA mM−1. The linear response range of 0.5–9 mM has a detection limit (LOD) of 145 μM | 158 |
| Biopolymer matrix | Lignin blended with different biopolymeric blends like starch, polylactic acids, polyhydroxybutyrate, and polyamides | Tissue engineering & regenerative medicine, and therapeutic delivery systems | Biodegradability, strong mechanical reinforcement ability, unique structural properties, renewability, and diverse possible modifications of its unique chemical structure | 159 |
3.4 Mechanistic insights of lignin formulations
Numerous researchers have investigated lignin-based biomedical formulations, including hydrogels, polyurethanes, nanoparticles, and carbon nanotube (CNT) composites; nevertheless, most studies have primarily focused on synthesis routes and formulation approaches. However, there is limited discussion about the underlying mechanisms involving lignin's performance in these systems. A mechanistic understanding of lignin's role in such formulations is critical, as its functional groups vary widely depending on the chemical structure and source.Phenolic hydroxyl groups in lignin contribute to hydrogen-bonding or covalent crosslinking.75 In hydrogels, phenolic hydroxyl groups of lignin act as interfacial crosslinkers by establishing hydrogen-bonding networks with hydroxyl and amino groups in polymers such as PVA and chitosan. This is accompanied by π–π stacking and hydrophobic interactions of lignin's aromatic domains, thereby enhancing interfacial adhesion.160,161 These studies show that the functional contribution of lignin goes beyond mere coloration or stabilization, but actively affects the nature of the bioactivity and mechanical integrity of such hydrogels.
In general, lignin-based composite biogels exhibit enhanced fatigue resistance, toughness, and adhesion to the substrate due to the interaction of the aromatic and aliphatic components of lignin with the polymeric framework of the composite. This observation highlights the importance of the lignin type, source, and functionalization for the mechanical and biological properties of the resulting products.162,163 In addition to systems based on gels, lignin has also been incorporated into chitosan composites and lignin–cellulose complexes for other biomedical-relevant applications where its bioactive groups and interfacial interactions contribute to beneficial functional effects.162,163
In CNT composites, lignin acts as a dispersant and facilitates interfacial charge transfer. Due to its aromatic rings, it attaches to the nanotubes through π–π interactions, which helps keep the formulation dispersed and stable.164 In polyurethane systems, lignin plays a different role; it can act as a chain extender or a reactive filler. Its aliphatic hydroxyls and aromatic rings help in making the material tougher, more resistant to UV light, and even more eco-friendly since they improve biodegradability.23
Lignin-based nanoparticles present another mechanistically interesting system due to the amphiphilic nature of lignin, which can self-assemble into these well-ordered tiny core–shell structures. This allows it to act as both the carrier and the protective shell for bioactive molecules.24 These reflect the reactivity and capacity for crosslinking and interfacial properties important in inducing biocompatibility and surface interactions. The source of the lignin and its purity are other significant factors affecting performance. Native lignin and organosolv lignin are usually more uniform and reactive than technical Kraft or soda lignin, which contain some sulfur and heterogeneous linkages, which limit their potential biomedical utility.
The mechanistic aspects of lignin-based formulations are summarized in terms of their physical state, functional role, and key chemical groups (Table 4). These features determine their main biomedical applications, along with the advantages and limitations influencing their overall performance. In summary, this comparative analysis gives further evidence of the adaptability of lignin as a multifunctional component biomaterial and defines some of the structural features that lead to the variations in performance in relation to the diverse biomedical applications.23,24,165
| Formulation type | Physical state | Role of lignin | Functional groups | Applications | Advantages | Limitations/challenges |
|---|---|---|---|---|---|---|
| Hydrogels | Semi-solid | Crosslinker, antioxidant, antimicrobial agent | Phenolic –OH, COOH | Wound dressings, tissue scaffolds | Biocompatibility, antioxidant protection | Mechanical weakness, limited tunability |
| Polyurethanes | Solid/elastic | Chain extender, UV stabilizer, filler | Aliphatic –OH, aromatic rings | Soft biomaterials, coatings | Improved strength, UV resistance | Variability in reactivity across lignin types |
| CNT/lignin composites | Solid/conductive | Dispersant, electron mediator, mechanical enhancer | Phenolic –OH, aromatic rings | Biosensors, conductive scaffolds | Enhanced dispersion, redox activity | Processing complexity |
| Nanoparticles | Colloidal/liquid | Carrier, stabilizer, active agent | Phenolic –OH, COOH, methoxy | Drug delivery, antioxidant nanocarriers | High surface reactivity, biodegradability | Scale-up, stability in physiological media |
| Films/coatings | Solid | UV barrier, antimicrobial layer | Aromatic rings, phenolic –OH | Biomedical packaging, wound coatings | Antimicrobial and antioxidant protection | Brittleness, uneven morphology |
Despite these advances, the majority of reports are from the laboratory scale, and further work is required to help translate lignin materials to clinical (or commercial) applications in biomedicine. This requires robust purification, reproducibility studies, and safety evaluations. Continued mechanistic insight is thus necessary, alongside controlled lignin extraction and functionalization, to fully unlock the biomedical avenues for this abundant natural polymer.
4. Frontiers of lignin-based biopharmaceutical development
Lignin, a naturally abundant and renewable biopolymer, holds immense potential in the field of biopharmaceuticals due to its unique structural properties. Recent advancements in lignin functionalization and nanotechnology have opened new avenues for its application in biopharmaceuticals. Despite its shortcomings, due to the wide range of applications, such as in pharmaceutics, cosmetics, and agricultural industries, it is anticipated that lignin will be crucial to future innovations. In biopharmaceutical systems, lignin is predominantly employed as a functional, non-active component, where it serves as a carrier, stabilizing matrix, or performance-enhancing excipient rather than as a standalone therapeutic agent.Current research demonstrates that lignin contributes to therapeutic outcomes by improving drug stability, encapsulation efficiency, controlled release, and targeting performance, particularly in advanced drug delivery systems. The increasing interest in lignin-based biomedical applications is due to high stability, pH-responsive behaviour, and high encapsulation efficiency.100 Accordingly, this section discusses established applications of lignin in drug delivery, biomedical platforms, combination strategies, and bioactive lignin-derived compounds, while also highlighting emerging directions and future opportunities in lignin-based biopharmaceutical development.
4.1 Drug delivery system
In pharmaceutical systems, lignin biomaterials have been utilized extensively to develop stimuli-responsive drug delivery systems, and substantial research will be required to completely understand lignin's potential. Moreno et al. reported thermo-responsive lignin-based materials incorporated with a low critical solution temperature (LCST). Below the LCST, the lignin-based polymer chain starts existing as random coils due to hydrogen bond interactions. The phase separation occurs due to the difference between temperatures above and below the LCST, resulting in the controlled drug release in the specific area.166 Similarly, Dinari et al. assessed the dual response of a lignin-based nano gel for curcumin loading and release. The nano gel named lignin-g-P(NIPAM-co-DMAEMA) was studied for its biodegradability and stimuli-response, overcoming its limitations and enhancing its other properties.167 Due to lignin's amphiphilic nature, both hydrophilic and hydrophobic drugs can be encapsulated, which can help to protect the encapsulated drugs and also help in their controlled release.168 Amphiphilic lignin acts as a micelle due to the availability of both hydrophobic and hydrophilic parts. The hydrophobic part generally consists of a phenyl propane skeleton, and the hydrophilic groups consist of phenolic hydroxyl and carboxyl groups.169 A Π–Π conjugated effect was observed between benzene rings which allowed lignin-based micelles to transport hydrophobic drugs and nutrients through the formation of lignin nano-micelles, which have proven to enhance molecular drug delivery because they exhibit a low critical micelle concentration.170Another area in which lignin is expected to play an important role is targeted drug delivery, particularly for anticancer therapeutics, where lignin-based systems have been widely explored as carriers to enhance drug stability, targeting efficiency, and therapeutic performance. Liu et al. developed lignin-based targeted polymeric nanoparticles for efficient and targeted drug delivery of anticancer drug hydroxyl camptothecin (HCPT) via folic acid–polyethylene glycol–alkaline lignin conjugates (FAPEG-AL).128 The high drug loading efficiency, robust stability, moderate particle size, and efficient delivery capacity of FA-PEG-AL/HCPT NPs enhanced the properties and effectiveness of HCPT. In this context, it also enhances blood circulation time and cellular uptake. Another application that can be further explored in the field of drug delivery is incorporating lignin with some already available pharmaceutical drugs to enhance their therapeutic properties. For example, Pishnamazi et al. modified lignin to form carboxylated lignin and then studied the pH-dependent release behaviours of carboxylated lignin and its effects on releasing a drug.146 The drug that was incorporated with carboxylated lignin was paracetamol, a standard antipyretic and analgesic prescribed by doctors worldwide. The results were quite optimistic and illustrated an increase in the rate of release of paracetamol when incorporated with carboxylated lignin. This can be explained by the lower degree of interaction between lignin and the API due to the deprotonation of the –COOH group. Recently, lignin has also been incorporated into the formulation for the SNEDDS (solid self-nanoemulsifying drug delivery system) as a surfactant, as reported by Dai et al., in which a lignin-based surfactant in the SNEDDS was prepared to improve the stability, absorption, and effectiveness of trans-RSV.171
These findings collectively highlight the versatility of lignin as a functional component in advanced drug delivery systems, capable of influencing both targeting efficiency and release behaviour. In drug delivery applications, the specific location of lignin and the binding mode within the gel matrix critically impact drug loading, diffusion, and release performance. Structures where lignin is covalently attached are inclined to display slower, diffusion-controlled release, while physically incorporated lignin frequently aids rapid and responsive release profiles. Thus, lignin should be regarded not only as a universal matrix polymer, but also as a multifunctional additive whose contribution to mechanical, chemical, or biological properties depends on its integration strategy and chemical structure.
4.2 Biomedical applications
Biomedical application is the application of technology and engineering in life sciences, especially related to materials with therapeutic or medicinal properties.172 Biomedical applications have a wide range of applications in medicine, including diagnostic imaging technologies such as MRI, CT scans, and other novel research areas of genetic engineering, gene therapy, and biosensors. Jedrzak et al. developed an enzyme-based biosensor with the help of a functional biohybrid silica–lignin, which was used to immobilize glucose oxidase on its surface for glucose sensing.158 The synthesized biosensor has high sensitivity and a linear response range of 0.5–9 mM and has a detection limit (LOD) of 145 μM.Lignin will have substantial applications in tissue engineering soon due to its emerging nature and diverse applications. Lignin-based nanomaterials are developed into nanofibers and hydrogels, which are fabricated to mimic the extracellular matrix and promote tissue growth and regeneration.173 Subsequently, the porous structure helps us to influence the geometry, density, surface areas, and permeability, which makes it suitable for different medical uses as its pores can be used to lighten weight in bone tissue engineering applications, and also apply its absorption ability that can be used to control the drug loading and release activity.43
Lignin's application in wound healing has only reached a surface level so far, and there is still much to be done; however, the results reported to date have been fruitful. For instance, Ravishankar et al. reported a hydrogel of chitosan–alkali lignin, which was synthesized using cross-linkages, and no toxicity was observed in either in vitro or in vivo studies.73 Lignin can also help regulate bone metabolism due to its ability to scavenge ROS, which will help restore the uncoupling bone remodelling and prevent and cure many orthopaedic diseases.174
4.3 Combination therapy
Combination therapy refers to the use of two or more therapeutic agents or treatment modalities together to achieve enhanced or synergistic effects. It has become a cornerstone in many biomedical applications, and numerous compounds are being evaluated and explored for their synergistic potential and enhanced therapeutic efficacy. Combinational therapy is of great value due to certain reasons, like fewer side effects, environmentally friendly drug synthesis, and enhanced drug efficacy with a material possessing the same properties or another therapeutic property. Siddiqui et al. reported a novel 4-in-1 strategy on how to combat colon cancer, in which the first strategy involves the action of a chemotherapeutic drug on cancer cells. Secondly, antioxidants present in lignin and quercetin act on cells under high oxidative stress to prevent cancer relapse. Subsequently, quercetin modulates to overcome drug efflux. Lastly, hyaluronic acid acts as a ligand to target lignin nanoparticles to cancer cells through CD44 receptors.175 Combination strategies in cancer research often aim to enhance therapeutic efficacy through synergistic mechanisms. In this context, Pylypchuk et al. synthesized lignin nanoparticles from eucalyptus and spruce wood lignin.176 These nanoparticles show anticancer properties against hepatocellular carcinoma (HCC). There was also high dose dependency in both cases, although E-LNPs more efficiently inhibited the growth of aggressive HCC cells than less polar S-LNPs. Importantly, the study revealed a direct correlation between lignin surface chemistry and anticancer activity, where a higher number of carbohydrate subunits on the lignin nanoparticle surface was associated with enhanced biological activity.Synergistic combination is another area where lignin's potential could be unleashed. Liu et al. reported using lignin and quercetin as a natural, effective, and cheap antioxidant.177 The free radical scavenging ability was even better than that of pure quercetin, which could be explained by the encapsulation of quercetin into lignin that preserved its antioxidant properties under UV radiation for a longer time. Another research conducted by Figueiredo et al. explained the effectiveness of biodegradable lignin-based nanoparticles in pharmaceutical sciences.130 Three different kinds of nanoparticles were made, including Fe3O4-LNPs, LNPs, and pLNPs. The efficacy of loading poorly water-soluble drugs was evaluated, leading to improved release profiles, and the antiproliferative effect was enhanced. With the increase in their surface structure, LNPs were modified to target moieties to increase the cellular interaction with cancer cells, which can be useful for cancer therapy.
4.4 Bioactive compounds from lignin
Bioactive compounds are the naturally occurring compounds found in plants, animals, and microorganisms that have a positive impact on human health. There is a long list of compounds that fall under the category of bioactive compounds, which includes flavonoids, antioxidants, terpenes, alkaloids, probiotics, prebiotics, and many more. Bioactive compounds found in vegetables and fruits possess the potential to prevent certain chronic conditions, such as cancer, cardiac diseases, and diabetes.178 Lignin, being rich in polyphenols, is an immense source of bioactive compounds, which are responsible for its therapeutic behaviour.179 If such bioactive compounds are efficiently extracted from lignin, they can be useful in multiple therapeutic applications.The presence of polyphenolic bioactive compounds imparts high antioxidant properties in lignin, which makes it a suitable material for use in cosmetics and pharmaceuticals. Vinardell et al. examined the antioxidant properties of lignin extracted from different sources, such as LS, bagasse, steam explosion, and curan.180 The results confirmed that lignin obtained from all four of these possesses antioxidant properties, with bagasse being the most potent. The extracted lignin from different sources was also tested to check its toxicity to human skin and eyes, which also confirmed that lignin is safe when it is exposed to skin and eyes. Water-soluble lignin generally has better antioxidant properties than traditional lignin due to its low molecular weight.181 Apart from antioxidant and anticancer properties, lignin also possesses anti-inflammatory properties, but this field has not been explored as much as lignin's application in antioxidant and anticancer drugs. Wang et al. worked on two types of water-soluble lignin; one was derived from bamboo (WSL BM) and the other from the wheat stalk (WSL-WS).181 Anti-inflammatory properties were tested when some mice were given the treatment for colitis, and anti-inflammatory effects were observed by regulating key signalling pathways involved in inflammation by activating the Nrf2 pathway and inhibiting NFκB to treat bowel diseases. The neuroprotective effect is the ability of some molecules or substances to protect the neurons present in the brain and nervous system, and it has become a beacon of hope in the treatment of diseases like Alzheimer's disease, Parkinson's disease, and amyotrophic lateral sclerosis (ALS). Lignin applications have started to creep into the area of neuroprotective effects; however, this area is new, and further studies need to be done to evaluate its full potential. For instance, Ito et al. converted lignin into its bioactive lignophenol derivative.182 It was found that a modified lignocresol derivative from bamboo protected SH-SY5Y cells from H2O2-induced apoptosis and had neuroprotective effects against oxidative stress, OGD stress, tunicamycin stress, PSI stress, and retinal damage, which eventually suggests that it could have application in treating several neurodegenerative diseases.
5. Challenges and future prospects
Lignin, despite its wide range of applications and promising developments, still has several challenges that hinder its clinical and industrial translation. Here, key issues including structural heterogeneity, lack of standardization in extraction and characterization methods, and limited understanding of its biological interactions were discussed. Additionally, regulatory uncertainties and scalability concerns pose further obstacles. Addressing these limitations through interdisciplinary research, advanced processing techniques, and policy frameworks will be crucial for unlocking lignin's full potential in biomedical applications.5.1 Structural heterogeneity and standardization
Standardization of lignin may be the most significant problem related to lignin because the chemical structure and molecular properties of lignin are still uncertain, as they change every time with the change in the method of extraction.183,184 The structure and chemical composition of lignin also depend upon the source or species of the plant that is being used to extract lignin.185 The primary goal of scaling up lignin processing is its valorization into value-added materials or products. However, achieving cost competitiveness in industrial-scale biorefinery plants remains challenging, particularly in the lignin isolation step, as lignin is a by-product of the extraction process rather than the primary target.186 Some of the methods to extract lignin are detrimental to the environment due to the use of high temperatures and pressures, along with the use of harsh chemicals. To combat these existential issues, the extraction of lignin via biorefineries is a good alternative that revolves around minimizing the extent of alternative energy and maximizing the efficacy of the lignin that is extracted.187 Similarly, deep eutectic solvents (DESs) are another way to extract lignin in a green and sustainable way by increasing the reactivity of lignin, which makes its functional group more accessible.187Isolation of lignin, its transformation, and minimization of structural changes, as well as keeping the extraction procedure low-cost, are big challenges.188 Lignin degradation is a multistep process that involves various reactions and also produces large amounts of side products.189 There's a need to use potent oxidation agents to help the enzymes attack any lignin site that was found to be accessible, but this could also lead to dehydrogenation, which could lead to oxidation coupling via phenolic moieties.190 The heterogeneity of lignin results in the heterogeneity of aromatic compounds, which is another problem. However, a new approach called biological funnelling is being used in which some microbes are used to convert aromatic molecules into intermediates, which are funnelled into the central carbon atom.191 Lignin degradation poses another problem that we need to tackle before we can start utilizing lignin at its full potential.
5.2 Translational hurdles in lignin biopharmaceutical development
Despite growing academic interest, lignin remains a preclinical biomaterial. The absence of validated PK/PD data, standardized biocompatibility testing, reliable sterilization and endotoxin control, and reproducible CMC documentation continues to impede translation. Bridging this gap will require coordinated studies that integrate pharmacological testing, regulatory-grade material characterization, and long-term toxicology in line with ICH, ISO, and FDA/EMA expectations before lignin can progress toward clinical evaluation.
5.3 Toxicity mechanisms and long-term biodegradation of CNTs and quantum dots
Direct evidence linking the long-term toxicity, persistence, or metabolic fate of carbon nanotubes (CNTs) and carbon quantum dots (CQDs) remains limited; however, multiple studies indicate characteristic biological responses that warrant attention. CNTs can exert cytotoxic or inflammatory effects in cells and animals through mechanical injury, oxidative stress, and interference with intracellular pathways. Their surfaces promote the generation of reactive oxygen species (ROS), which in turn deplete glutathione (GSH) and disrupt the redox balance, ultimately leading to mitochondrial damage and cell death.193,200 The extent of these effects depends on the tube length, surface functionalization, and the degree of aggregation.The breakdown of CNTs proceeds either by chemical oxidation or biological transformation. Enzymatic routes, particularly those involving peroxidases such as myeloperoxidase (MPO), horseradish peroxidase (HRP), and lactoperoxidase (LPO), have been extensively examined. HRP was first reported to oxidize single-walled CNTs, but later work showed that much of this activity originates from a Fenton-type reaction driven by iron released when the enzyme's heme group decomposes in the presence of hydrogen peroxide.199,201–203 The resulting hydroxyl radicals (·OH) attack the graphitic lattice, producing fragmented and oxygenated structures. Reported degradation rates vary from a few days to several decades, illustrating the strong dependence of the process on reaction conditions.204
Microbial activity can also modify CNTs. Bacteria such as Labrys sp., Shewanella, and Burkholderia species generate H2O2 and Fe(II) simultaneously, enabling biogenic Fenton-like oxidation of CNTs.205,206 This pathway converts resistant carbon frameworks into smaller, oxidized fragments, although complete mineralization is a slow process and may require months under favorable conditions. The persistence of CNTs in soils and aquatic systems, consequently, remains a realistic concern.
Morphologically, some needle-like multiwalled CNTs resemble asbestos fibers, raising the possibility of similar pathogenic outcomes. Experimental inhalation studies have linked such materials to pleural fibrosis, mesothelioma, and chronic pulmonary inflammation.207,208 Besides human health, CNTs also affect plants and microorganisms: growth inhibition, reduced germination, and membrane disruption have all been documented.209,210 As production and use increase, environmental accumulation and ecotoxicological exposure will require systematic risk evaluation.
In contrast, lignin-functionalized CNTs and CQDs appear less harmful. The lignin coating supplies antioxidant phenolic groups that can quench ROS and limit oxidative stress during photothermal activation. Experimental work demonstrates lower cytotoxicity and improved cellular tolerance for lignin-coated nanostructures compared to unmodified carbon analogues.200 Even so, detailed in vivo studies addressing biodistribution, chronic exposure, and degradation kinetics are still missing. Collectively, lignin modification offers a promising route to enhance the biocompatibility and environmental stability of carbon nanomaterials; however, a clear understanding of their long-term fate remains essential for safe biomedical and industrial applications.
5.4 Regulatory hurdles
A specific framework for lignin-based materials does not currently exist, especially for biopharmaceuticals. Rules and regulations are stringent when it comes to medicines, and no traces of toxic materials are generally accepted.211 Lignin has already shown various biological activities in clinical studies. However, lignin, being a compound or an excipient in formulation, has also been evaluated for metabolism studies. Metabolism studies point out that lignin is primarily undigested in the gastrointestinal tract; the lack of biodegradation permits careful attention. The nano-scale non-digestible lignin-based materials may persist at the intestinal interface and interact with the mucus layer and epithelial surface. The abundant phenolic hydroxyl groups and aromatic groups present in lignin can help adhesion to mucins and epithelial membranes through hydrogen bonding, hydrophobic interactions, and π–π stacking, possibly leading to interfacial accumulation. Such a buildup of undigested lignin raises alarms regarding long-term exposure, increased epithelial permeability, local inflammation, or disruption of gut microbiota equilibrium. Thus, the absence of digestive metabolism highlights the need for targeted studies, such as evaluations of mucus penetration, epithelial retention, and long-term exposure. These drawbacks emphasize the necessity of a lignin-specific regulation framework that considers interfacial persistence and nano-bio interactions rather than relying solely on conventional bulk-material toxicity models.212The use of lignin in the pharmaceutical field has not yet been approved by major regulatory agencies worldwide, including the Food and Drug Administration (FDA) and the European Medicines Agency (EMA).24 Apart from that, there are also no certified criteria for engineered nanomaterials, but the FDA and the European Commission are working to design strict regulations regarding the optimum use of nanoparticles.213,214 For pharmaceutical applications, lignin-derived systems would be expected to comply with the International Council for Harmonisation (ICH) guidelines, particularly Q8 (R2) on Pharmaceutical Development and Q12 on Pharmaceutical Product Lifecycle Management. These frameworks emphasize the definition of Critical Quality Attributes (CQAs), implementation of Quality by Design (QbD) principles, and establishment of a robust Chemistry, Manufacturing, and Controls (CMC) framework to ensure batch reproducibility and product consistency throughout the development lifecycle.215,216 For biomedical device applications—such as lignin-based wound dressings, hydrogels, or 3D-printed scaffolds—evaluation is guided by the ISO 10993 standards for biological safety and the European Union Medical Device Regulation.217 Depending on the degree of invasiveness and intended use, such products are generally categorized as class IIb or class III, requiring a comprehensive biological evaluation that encompasses cytotoxicity, sensitization, systemic toxicity, implantation, and degradation analyses.218
Thus, most pharmaceutical companies will be in a dilemma to give lignin a chance, as it will be time-consuming and also require a lot of capital without knowing whether it will ever be approved or not. Safety and efficacy standards should be met to come on par with the pharmaceutical regulations that are set for other biomaterials. General protocols of testing should be followed, having a global vision in mind to meet the demands of the whole of mankind. We still have a lot to explore about lignin's properties and its toxicity, molecular behaviour at different sizes, and how it will affect its medicinal and therapeutic properties. Addressing these aspects will be essential for transforming lignin from a promising experimental biomaterial into a clinically viable and regulatory-compliant therapeutic platform.
5.5 Integration with existing pharmaceutical processes
Integration of biomaterials with existing pharmaceutical processes has also been on the rise to increase the therapeutic properties of existing drugs and minimize the side effects. The biomaterial, which is generally incorporated with the drug, is known as an active pharmaceutical ingredient (API). Lignin can be used as a modifier to enhance the bioavailability of APIs because of its amorphous nature, which often results in higher free energy.219 The problem with APIs, in general, is that many pharmaceutical companies are always looking for faster and more economical methods to produce APIs without any thought of the safety or rules and regulations.220 This is detrimental to both human health and the environment if a company uses hazardous chemicals just for the sake of making a profit. For instance, a big pharmaceutical corporation can make lignin-based APIs without following safety and regulation guidelines, as there are no specific guidelines for lignin biomaterials. Pishnamazi et al. reported one example where lignin was integrated into existing pharmaceutics (acetylsalicylic acid tablets), where organosolv lignin, along with microcrystalline cellulose (MCC) and lactose, was added to aspirin, resulting in increased hardness and release rate of aspirin.145Lignin-based biopharmaceuticals have a great deal of potential to advance biomedical applications, combination treatments, drug delivery methods, and the extraction of bioactive components. It is essential to standardize lignin sources and quality, taking into account extraction techniques, quality control, and source variability. One must overcome regulatory barriers, such as the absence of certain laws, safety requirements, and international harmonization. Also, the incorporation of lignin into current pharmaceutical processes demands that compatibility, processing difficulties, scale-up issues, interactions with other components, and cost implications should be carefully considered. Regardless of these challenges, addressing them all at once will pave the way for the successful integration of lignin into the biopharmaceutical sectors, offering creative and long-lasting remedies for medical demands in the future.
5.6 Comparative context with existing biomaterials
The widely used biopolymers, such as hyaluronic acid and collagen, which are often expensive and sourced from animal tissues, are compared to lignin, an agri-residue-based biopolymer, which offers high cost and supply advantages. In contrast to alginate and collagen hydrogels, which primarily provide structural support, lignin inherently exhibits antioxidant and antimicrobial activities due to its phenolic and aromatic domains.221–223 Additionally, the molecular structure of lignin allows extensive chemical modification, enabling the tuning of stiffness, degradation rate, and surface interaction features that are comparatively more limited in natural polymers with fixed backbone chemistries.224 However, the enzymatic biodegradation of lignin is slower than that of polysaccharide-based hydrogels; this property can be advantageous for applications requiring prolonged material stability or sustained release. Collectively, these factors position lignin as a functionally competitive and economically scalable biomaterial candidate.225,2266. Conclusion
Lignin, a sustainable biopolymer, has shown significant potential in the development of various biopharmaceutical formulations due to its structural diversity and inherent biological activities. Its antioxidant, antimicrobial, and anti-inflammatory properties, along with biocompatibility and availability, make it a strong candidate for use in pharmaceutical and biomedical applications. In this review, we have explored a wide range of lignin-based formulations, including semisolid systems such as hydrogels and cryogels, liquid forms like suspensions and emulsions, and solid platforms like nanoparticles, nanocomposites, and biomedical devices. These materials offer innovative solutions for drug delivery, wound healing, and other therapeutic strategies, often enhancing performance while also contributing to sustainability. However, its use is still limited by variability in composition, extraction methods, and the lack of standardized purification and testing protocols. Most studies remain at the laboratory scale, and systematic data on long-term safety, pharmacokinetics, and biodegradation are still missing. For lignin to move closer to real pharmaceutical use, greater attention is needed to ensure batch uniformity, reproducibility, and regulatory compliance. Overall, lignin represents a sustainable and promising platform for future healthcare materials. Continued collaboration between materials science, pharmacology, and regulatory research will be key to turning this potential into safe, effective, and commercially viable biopharmaceutical products.Abbreviations
| AIDS | Acquired immune deficiency syndrome |
| ALS | Amyotrophic lateral sclerosis |
| API | Active pharmaceutical ingredient |
| ATRP | Atom transfer radical polymerization |
| BB | Blemish balm |
| BM | Bamboo |
| CDs | Carbon dots |
| CNTs | Lignin-based carbon nanotubes |
| DMI | Dimethyl isosorbide |
| DESs | Deep eutectic solvents |
| E. coli | Escherichia coli |
| ECH | Epichlorohydrin |
| EMA | European Medicines Agency |
| FA-PEG-AL | Folic acid–polyethylene glycol–alkaline lignin conjugates |
| FAPEG-AL | Folic acid–polyethylene glycol–alkaline lignin conjugates |
| FDA | Food and Drug Administration |
| Fe3O4-LNPs | Fe3O4-infused lignin nanoparticles |
| HCC | Hepatocellular carcinoma |
| HCPT | Hydroxyl camptothecin |
| HIV-1 | Herpes simplex virus type 1 |
| HSEs | Human skin equivalents |
| HSV-2 | Herpes simplex virus type 2 |
| KL | Kraft lignin |
| KL-PEG | Kraft lignin–polyethylene glycol conjugate |
| K-cLNPs | Kraft-colloidal lignin nanoparticles |
| LCST | Low critical solution temperature |
| LNCs | Lignin nanocapsules |
| LNPs | Lignin nanoparticles |
| LS | Lignosulfonates |
| MCC | Microcrystalline cellulose |
| Mt | Montmorillonite |
| NEBA | N,N′-Ethylenebisacrylamide |
| NIPAM-co-DMAEMA | Nanogel named lignin-g-P |
| NMBA | N,N′-Methylenebisacrylamide |
| OMt | Organo-montmorillonite |
| PCL | Polycaprolactone |
| PEG | Poly(ethylene glycol) |
| PEGDGE | Poly(ethylene glycol) diglycidyl ether |
| PHB | Polyhydroxybutyrate |
| PLA | Polylactic acid |
| PMVE/MA | Poly(methyl vinyl ether-co-maleic acid) |
| PU | Polyurethane |
| PVA | Polyvinyl alcohol |
| RAFT | Reversible addition–fragmentation transfer |
| ROS | Reactive oxygen species |
| S. aureus | Staphylococcus aureus |
| SNEDDS | Solid self-nanoemulsifying drug delivery system |
| SPF | Sun protection factor |
| WS | Wheat stalk |
| WSL | Water-soluble lignin |
Author contributions
Kshitij Rawat: conceptualization, visualization, investigation, validation, writing original draft – review & editing; Ravneet Kaur: investigation, validation, writing original draft – review & editing; Anil Kumar Pujari: investigation, validation, writing original draft – review & editing; Kunal Gogde: investigation, validation, writing original draft – review & editing; Devesh Mohne: investigation, validation, writing original draft – review & editing; Seema Kirar: validation, review & editing; Yeddula Nikhileshwar Reddy: validation, review & editing; Pooja Ramji Yadav: validation, review & editing; Prakash Y. Khandave: validation, review & editing; Abhay H. Pande: fund acquisition, validation, review & editing; Jayeeta Bhaumik: conceptualization, visualization, fund acquisition, supervision, validation, review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software, or code have been included, and no new data were generated or analysed as part of this review.Acknowledgements
All the authors are thankful to the BRIC NABI, the Department of Biotechnology (DBT), Government of India (GOI), for providing funding, infrastructural support, and instrumental facilities. The authors thank the Department of Biotechnology for funding the lignin valorization project (Grant No. BT/PR42965/PBD/26/821/2021). K. R. appreciates the University Grants Commission (UGC) for the senior research fellowship. P. R. Y. is grateful to the Indian Council of Medical Research (ICMR) for the junior research fellowship. S. K. thanks the Department of Science and Technology (DST) for the fellowship under the WOSA scheme. The authors thank Prof. Deepa Ghosh, Scientist G, INST Mohali, for her insightful comments.References
- Y. Mukheja, J. Kaur, K. Pathania, S. P. Sah, D. B. Salunke, A. T. Sangamwar and S. V. Pawar, Int. J. Biol. Macromol., 2023, 241, 124601 CrossRef CAS PubMed.
- G. Kaur, P. Kumar and C. Sonne, RSC Appl. Interfaces, 2024, 1, 846–867 RSC.
- L. Lorenzo, W. Pitacco, N. Mattar, I. Faye, B. Maestro and P. Ortiz, RSC Appl. Polym., 2025, 3, 420–427 RSC.
- V. K. Garlapati, A. K. Chandel, S. P. J. Kumar, S. Sharma, S. Sevda, A. P. Ingle and D. Pant, Renewable Sustainable Energy Rev., 2020, 130, 109977 CrossRef CAS.
- J. A. Diaz-Baca and P. Fatehi, Biotechnol. Adv., 2024, 70, 108281 CrossRef CAS PubMed.
- R. Eloy de Souza, F. J. B. Gomes, E. O. Brito, R. C. C. Lelis, L. A. R. Batalha, F. A. Santos and D. L. Junior, J. Appl. Biotechnol. Bioeng., 2020, 7, 100–105 Search PubMed.
- S. Kirar, D. Mohne, M. Singh, V. Sagar, A. Bhise, S. Goswami and J. Bhaumik, Sustainable Mater. Technol., 2024, 40, e00864 CrossRef CAS.
- M. M. Alam, A. Greco, Z. Rajabimashhadi and C. Esposito Corcione, Cleaner Mater., 2024, 13, 100253 CrossRef CAS.
- E. Sheridan, S. Filonenko, A. Volikov, J. A. Sirviö and M. Antonietti, Green Chem., 2024, 26, 2967–2984 Search PubMed.
- P. Figueiredo, K. Lintinen, J. T. Hirvonen, M. A. Kostiainen and H. A. Santos, Prog. Mater. Sci., 2018, 93, 233–269 Search PubMed.
- Y. Zhang and M. Naebe, ACS Sustainable Chem. Eng., 2021, 9, 1427–1442 CrossRef CAS.
- A. V. Faleva, A. V. Belesov, A. Y. Kozhevnikov, D. I. Falev, D. G. Chukhchin and E. V. Novozhilov, Int. J. Biol. Macromol., 2021, 166, 913–922 CrossRef CAS PubMed.
- H. Sun, G. Wang, J. Ge, N. Wei, W. Sui, Z. Chen, H. Jia, A. M. Parvez and C. Si, Ind. Crops Prod., 2022, 180, 114685 CrossRef CAS.
- D. Piccinino, E. Capecchi, E. Tomaino, S. Gabellone, V. Gigli, D. Avitabile and R. Saladino, Antioxidants, 2021, 10, 274 CrossRef CAS PubMed.
- J. F. Nash and P. R. Tanner, Photodermatol. Photoimmunol. Photomed., 2014, 30, 88–95 CrossRef CAS PubMed.
- Y. Huang, J. C. F. Law, T. K. Lam and K. S. Y. Leung, Sci. Total Environ., 2020, 755, 142486 CrossRef PubMed.
- Anushikha and K. K. Gaikwad, Biomass Convers. Biorefin., 2024, 14, 16755–16767 CrossRef CAS.
- L. Korkina, V. Kostyuk, A. Potapovich, W. Mayer, N. Talib and C. De Luca, Cosmetics, 2018, 5, 32 Search PubMed.
- D. Kai, M. J. Tan, P. L. Chee, Y. K. Chua, Y. L. Yap and X. J. Loh, Green Chem., 2016, 18, 1175–1200 Search PubMed.
- M. P. Vinardell and M. Mitjans, Int. J. Mol. Sci., 2017, 18, 1219 Search PubMed.
- A. G. Morena and T. Tzanov, Nanoscale Adv., 2022, 4, 4447–4469 RSC.
- S. Li, Y. Zhang, X. Ma, S. Qiu, J. Chen, G. Lu, Z. Jia, J. Zhu, Q. Yang, J. Chen and Y. Wei, Biomacromolecules, 2022, 23, 1622–1632 Search PubMed.
- D. Kai, K. Zhang, L. Jiang, H. Z. Wong, Z. Li, Z. Zhang and X. J. Loh, ACS Sustainable Chem. Eng., 2017, 5, 6016–6025 Search PubMed.
- J. Domínguez-Robles, Á. Cárcamo-Martínez, S. A. Stewart, R. F. Donnelly, E. Larrañeta and M. Borrega, Sustainable Chem. Pharm., 2020, 18, 100320 Search PubMed.
- X. Pan, J. F. Kadla, K. Ehara, N. Gilkes and J. N. Saddler, J. Agric. Food Chem., 2006, 54, 5806–5813 Search PubMed.
- J. Zhao, M. Zhu, W. Jin, J. Zhang, G. Fan, Y. Feng, Z. Li, S. Wang, J. S. Lee, G. Luan, Z. Dong and Y. Li, J. Nanobiotechnol., 2025, 23(1), 538 Search PubMed.
- K. Bhavna, S. Kirar, Y. N. Reddy, K. Rawat and J. Bhaumik, ACS Appl. Nano Mater., 2024, 7, 17101–17110 Search PubMed.
- C. M. L. de Melo, I. J. da Cruz Filho, G. F. de Sousa, G. A. de Souza Silva, D. K. D. do Nascimento Santos, R. S. da Silva, B. R. de Sousa, R. G. de Lima Neto, M. do Carmo Alves de Lima and G. J. de Moraes Rocha, Int. J. Biol. Macromol., 2020, 162, 1725–1733 Search PubMed.
- M. Oeyen, S. Noppen, E. Vanhulle, S. Claes, B. O. Myrvold, K. Vermeire and D. Schols, Virus Res., 2019, 274, 197760 CrossRef CAS PubMed.
- A. Boarino, H. Wang, F. Olgiati, F. Artusio, M. Özkan, S. Bertella, N. Razza, V. Cagno, J. S. Luterbacher, H. A. Klok and F. Stellacci, ACS Sustainable Chem. Eng., 2022, 10, 14001–14010 CrossRef CAS PubMed.
- K. Matsuhisa, S. Yamane, T. Okamoto, A. Watari, M. Kondoh, Y. Matsuura and K. Yagi, Biochem. Biophys. Res. Commun., 2015, 462, 52–57 Search PubMed.
- G. N. Rivière, A. Korpi, M. H. Sipponen, T. Zou, M. A. Kostiainen and M. Österberg, ACS Sustainable Chem. Eng., 2020, 8, 4167–4177 CrossRef PubMed.
- S. Liu, S. Xiao, B. Wang, Y. Cai, R. Xie, X. Wang and J. Wang, Int. J. Biol. Macromol., 2024, 263, 130509 CrossRef CAS PubMed.
- G. J. Gil-Chávez, S. S. P. Padhi, C. V. Pereira, J. N. Guerreiro, A. A. Matias and I. Smirnova, Int. J. Biol. Macromol., 2019, 136, 697–703 Search PubMed.
- S. Paul, A. De, S. Kirar, N. S. Thakur, A. K. Pujari, K. Gogde and J. Bhaumik, ACS Appl. Nano Mater., 2024, 7, 10984–10997 Search PubMed.
- M. S. Alqahtani, M. Kazi, M. Z. Ahmad, R. Syed, M. A. Alsenaidy and S. A. Albraiki, Int. J. Biol. Macromol., 2020, 163, 1314–1322 CrossRef CAS PubMed.
- T. Luo, C. Wang, X. Ji, G. Yang, J. Chen, S. Janaswamy and G. Lyu, Molecules, 2021, 26, 218 Search PubMed.
- T. O. MacHado, S. J. Beckers, J. Fischer, B. Müller, C. Sayer, P. H. De Araújo, K. Landfester and F. R. Wurm, Biomacromolecules, 2020, 21, 2755–2763 Search PubMed.
- J. Y. Zhu, U. P. Agarwal, P. N. Ciesielski, M. E. Himmel, R. Gao, Y. Deng, M. Morits and M. Österberg, Biotechnol. Biofuels, 2021, 14, 1–31 Search PubMed.
- Y. Mukheja, A. T. Sangamwar and S. V. Pawar, iotechnol. Sustainable Mater., 2025, 2, 1–14 Search PubMed.
- S. Paul, B. Yadav, M. D. Patil, A. K. Pujari, U. Singh, V. Rishi and J. Bhaumik, Mater. Adv., 2024, 5, 1903–1916 Search PubMed.
- K. Rawat, R. Kaur, A. K. Pujari, S. Kirar and J. Bhaumik, ACS Appl. Bio Mater., 2024, 7(11), 7666–7674 Search PubMed.
- N. Nan, W. Hu and J. Wang, Biomedicines, 2022, 10, 747 Search PubMed.
- D. Ziental, B. Czarczynska-Goslinska, M. Wysocki, M. Ptaszek and Ł. Sobotta, Eur. J. Pharm. Biopharm., 2024, 204, 114485 Search PubMed.
- M. A. Delgado, E. Cortés-Triviño, C. Valencia and J. M. Franco, Tribol. Int., 2021, 14(2), 356–379 Search PubMed.
- R. Kaur, N. S. Thakur, S. Chandna and J. Bhaumik, J. Mater. Chem. B, 2020, 8, 260–269 Search PubMed.
- J. Domínguez-Robles, E. Cuartas-Gómez, S. Dynes, E. Utomo, Q. K. Anjani, U. Detamornrat, R. F. Donnelly, N. Moreno-Castellanos and E. Larrañeta, Sustainable Mater. Technol., 2023, 35, e00581 CrossRef.
- N. Sajinčič, O. Gordobil, A. Simmons and A. Sandak, Cosmetics, 2021, 8(3), 78 Search PubMed.
- S. Chandna, N. S. Thakur, R. Kaur and J. Bhaumik, Biomacromolecules, 2020, 21, 3216–3230 Search PubMed.
- F. Antunes, I. F. Mota, J. F. Fangueiro, G. Lopes, M. Pintado and P. S. Costa, Int. J. Biol. Macromol., 2023, 15(234), 12359 Search PubMed.
- Y. Mukheja, S. N. Kethavath, L. Banoth and S. V. Pawar, Int. J. Biol. Macromol., 2024, 280, 135940 Search PubMed.
- H. A. Ariyanta, E. B. Santoso, L. Suryanegara, E. T. Arung, I. W. Kusuma, M. N. A. M. Taib, M. H. Hussin, Y. Yanuar, I. Batubara and W. Fatriasari, Sustainable Chem. Pharm., 2023, 32, 100966 CrossRef CAS.
- A. K. Pujari, R. Kaur, Y. N. Reddy, S. Paul, K. Gogde and J. Bhaumik, J. Med. Chem., 2023, 67, 2004–2018 CrossRef PubMed.
- M. Siahkamari, S. Emmanuel, D. B. Hodge and M. Nejad, ACS Sustainable Chem. Eng., 2022, 10, 3430–3441 Search PubMed.
- P. S. Chauhan, Bioresour. Technol. Rep., 2020, 9, 100374 Search PubMed.
- L. Passauer, T. Hallas, E. Bäucker, G. Ciesielski, S. Lebioda and U. Hamer, ACS Sustainable Chem. Eng., 2015, 3, 1955–1964 CrossRef CAS.
- S. Chandna, S. Paul, R. Kaur, K. Gogde and J. Bhaumik, ACS Appl. Polym. Mater., 2022, 4, 8962–8976 CrossRef CAS.
- C. Hachimi Alaoui, G. Réthoré, P. Weiss and A. Fatimi, Int. J. Mol. Sci., 2023, 24(17), 13493 CrossRef CAS PubMed.
- Z. Zhang, F. Li, J. W. Heo, J. W. Kim, M. S. Kim, Q. Xia and Y. S. Kim, Ind. Crops Prod., 2023, 205, 117403 CrossRef CAS.
- A. K. Pujari, S. Kirar, K. Gogde, K. Rawat and J. Bhaumik, J. Med. Chem., 2025, 68, 10314–10328 CrossRef CAS PubMed.
- X. Teng, H. Xu, W. Song, J. Shi, J. Xin, W. C. Hiscox and J. Zhang, ACS Omega, 2017, 2, 251–259 Search PubMed.
- S. Sathawong, W. Sridach and K. A. Techato, J. Environ. Chem. Eng., 2018, 6, 5879–5888 CrossRef CAS.
- I. E. Raschip, G. E. Hitruc, C. Vasile and M. C. Popescu, Int. J. Biol. Macromol., 2013, 54, 230–237 CrossRef CAS PubMed.
- L. Wu, S. Huang, J. Zheng, Z. Qiu, X. Lin and Y. Qin, Int. J. Biol. Macromol., 2019, 140, 538–545 CrossRef CAS PubMed.
- S. Akhramez, A. Fatimi, O. V. Okoro, M. Hajiabbas, A. Boussetta, A. Moubarik, A. Hafid, M. Khouili, J. Simińska-Stanny, C. Brigode and A. Shavandi, Sustainability, 2022, 14(14), 8791 CrossRef CAS.
- X. F. Sun, Y. Hao, Y. Cao and Q. Zeng, Int. J. Biol. Macromol., 2019, 127, 511–519 CrossRef CAS PubMed.
- R. Kaur, R. Sharma and G. K. Chahal, Chem. Pap., 2021, 75, 4465–4478 Search PubMed.
- M. Li, X. Jiang, D. Wang, Z. Xu and M. Yang, Colloids Surf., B, 2019, 177, 370–376 Search PubMed.
- J. Domínguez-Robles, M. S. Peresin, T. Tamminen, A. Rodríguez, E. Larrañeta and A. S. Jääskeläinen, Int. J. Biol. Macromol., 2018, 115, 1249–1259 Search PubMed.
- E. Larrañeta, M. Imízcoz, J. X. Toh, N. J. Irwin, A. Ripolin, A. Perminova, J. Domínguez-Robles, A. Rodríguez and R. F. Donnelly, ACS Sustainable Chem. Eng., 2018, 6, 9037–9046 CrossRef PubMed.
- X. Wang, Z. Zhou, X. Guo, Q. He, C. Hao and C. Ge, RSC Adv., 2016, 6, 35550–35558 Search PubMed.
- F. Oveissi, S. Naficy, T. Y. L. Le, D. F. Fletcher and F. Dehghani, ACS Appl. Bio Mater., 2018, 1, 2073–2081 CrossRef CAS PubMed.
- K. Ravishankar, M. Venkatesan, R. P. P. Desingh, A. Mahalingam, B. Sadhasivam, R. Subramaniyam and R. Dhamodharan, Mater. Sci. Eng., C, 2019, 102, 447–457 CrossRef CAS PubMed.
- Y. Zhang, M. Jiang, Y. Zhang, Q. Cao, X. Wang, Y. Han, G. Sun, Y. Li and J. Zhou, Mater. Sci. Eng., C, 2019, 104, 110002 CrossRef CAS PubMed.
- A. K. Mondal, D. Xu, S. Wu, Q. Zou, W. Lin, F. Huang and Y. Ni, Int. J. Biol. Macromol., 2022, 207, 48–61 Search PubMed.
- Z. Shan, B. Jiang, P. Wang, W. Wu and Y. Jin, Int. J. Biol. Macromol., 2025, 285, 138327 CrossRef CAS PubMed.
- S. Chandna, K. Gogde, S. Paul and J. Bhaumik, RSC Sustainability, 2024, 2, 2348–2356 Search PubMed.
- T. Abdullah, T. Colombani, T. Alade, S. A. Bencherif and A. Memić, Biomacromolecules, 2021, 22, 4110–4121 CrossRef CAS PubMed.
- A. Barros, S. Quraishi, M. Martins, P. Gurikov, R. Subrahmanyam, I. Smirnova, A. R. C. Duarte and R. L. Reis, Chem. Ing. Tech., 2016, 88, 1770–1778 Search PubMed.
- A. Memic, T. Abudula, H. S. Mohammed, K. Joshi Navare, T. Colombani and S. A. Bencherif, ACS Appl. Bio Mater., 2019, 2, 952–969 CrossRef CAS PubMed.
- K. T. Lin, R. Ma, P. Wang, J. Xin, J. Zhang, M. P. Wolcott and X. Zhang, Macromolecules, 2019, 52, 227–235 Search PubMed.
- L. Manzocco, K. S. Mikkonen and C. A. García-González, Food Struct., 2021, 28, 100188 CrossRef CAS.
- L. M. Sanchez, A. K. Hopkins, E. Espinosa, E. Larrañeta, D. Malinova, A. N. McShane, J. Domínguez-Robles and A. Rodríguez, Chem. Biol. Technol. Agric., 2023, 10, 1–11 Search PubMed.
- S. Chandna, C. A. Olivares M., E. Baranovskii, G. Engelmann, A. Böker, C. C. Tzschucke and R. Haag, Angew. Chem., Int. Ed., 2024, 63, e202313945 CrossRef CAS PubMed.
- C. W. Wu, P. H. Li, Y. M. Wei, C. Yang and W. J. Wu, RSC Adv., 2022, 12, 10755–10765 RSC.
- S. Quraishi, M. Martins, A. A. Barros, P. Gurikov, S. P. Raman, I. Smirnova, A. R. C. Duarte and R. L. Reis, J. Supercrit. Fluids, 2015, 105, 1–8 Search PubMed.
- Z. Wu, P. P. Thoresen, L. Matsakas, U. Rova, P. Christakopoulos and Y. Shi, ACS Sustainable Chem. Eng., 2023, 11, 12552–12561 CrossRef CAS.
- A. M. Borrero-López, L. Wang, H. Li, T. V. Lourençon, C. Valencia, J. M. Franco and O. J. Rojas, Int. J. Biol. Macromol., 2023, 242, 124941 Search PubMed.
- Z. Wan, H. Zhang, M. Niu, Y. Guo and H. Li, Int. J. Biol. Macromol., 2023, 253, 126660 Search PubMed.
- B. Jiang, H. Jiao, X. Guo, G. Chen, J. Guo, W. Wu, Y. Jin, G. Cao and Z. Liang, Adv. Sci., 2023, 10, 2206055 Search PubMed.
- J. Domínguez-Robles, N. K. Martin, M. L. Fong, S. A. Stewart, N. J. Irwin, M. I. Rial-Hermida, R. F. Donnelly and E. Larrañeta, Pharmaceutics, 2019, 11, 165 CrossRef PubMed.
- L. Jaiswal, S. Shankar, J. W. Rhim and D. H. Hahm, Int. J. Biol. Macromol., 2020, 159, 859–869 CrossRef CAS PubMed.
- S. C. Lee, T. M. T. Tran, J. W. Choi and K. Won, Int. J. Biol. Macromol., 2019, 122, 549–554 Search PubMed.
- J. S. Stine, B. J. Harper, C. G. Conner, O. D. Velev and S. L. Harper, Nanomaterials, 2021, 11, 111 CrossRef CAS PubMed.
- D. Mahata, M. Jana, A. Jana, A. Mukherjee, N. Mondal, T. Saha, S. Sen, G. B. Nando, C. K. Mukhopadhyay, R. Chakraborty and S. M. Mandal, Sci. Rep., 2017, 7, 1–16 Search PubMed.
- N. Tarannum, K. Pooja, S. Jakhar and A. Mavi, Discover Nano, 2024, 19, 1–29 CrossRef PubMed.
- Scientists develop new wound-healing cream from wood, https://www.downtoearth.org.in/health/scientists-develop-new-wound-healing-cream-from-wood-57744, (accessed 22 January 2025).
- B. Ozturk and D. J. McClements, Curr. Opin. Food Sci., 2016, 7, 1–6 CrossRef.
- N. Riquelme, R. N. Zúñiga and C. Arancibia, LWT--Food Sci. Technol., 2019, 111, 760–766 CrossRef CAS.
- M. Stanisz, Ł. Klapiszewski, M. N. Collins and T. Jesionowski, Mater. Today Chem., 2022, 26, 101198 CrossRef CAS.
- A. Naseem, S. Tabasum, K. M. Zia, M. Zuber, M. Ali and A. Noreen, Int. J. Biol. Macromol., 2016, 93, 296–313 CrossRef CAS PubMed.
- S. Sen, S. Patil and D. S. Argyropoulos, Green Chem., 2015, 17, 4862–4887 RSC.
- S. Paul, N. S. Thakur, S. Chandna, Y. N. Reddy and J. Bhaumik, J. Mater. Chem. B, 2021, 9, 1592–1603 RSC.
- L. Bai, L. G. Greca, W. Xiang, J. Lehtonen, S. Huan, R. W. N. Nugroho, B. L. Tardy and O. J. Rojas, Langmuir, 2019, 35, 571–588 Search PubMed.
- Y. Yang, Z. Wei, C. Wang and Z. Tonga, Chem. Commun., 2013, 49, 7144–7146 Search PubMed.
- E. Park, M. S. Park, J. Lee, K. J. Kim, G. Jeong, J. H. Kim, Y. J. Kim and H. Kim, ChemSusChem, 2015, 8, 688–694 CrossRef CAS PubMed.
- S. Li, D. Ogunkoya, T. Fang, J. Willoughby and O. J. Rojas, J. Colloid Interface Sci., 2016, 482, 27–38 CrossRef CAS PubMed.
- T. E. Nypelö, C. A. Carrillo and O. J. Rojas, Soft Matter, 2015, 11, 2046–2054 RSC.
- C. Gupta and N. R. Washburn, Langmuir, 2014, 30, 9303–9312 CrossRef CAS PubMed.
- M. Ago, S. Huan, M. Borghei, J. Raula, E. I. Kauppinen and O. J. Rojas, ACS Appl. Mater. Interfaces, 2016, 8, 23302–23310 CrossRef CAS PubMed.
- A. Duval, D. Vidal, A. Sarbu, W. René and L. Avérous, Mater. Today Chem., 2022, 24, 100793 CrossRef CAS.
- S. Beisl, A. Friedl and A. Miltner, Int. J. Mol. Sci., 2017, 18, 2367 CrossRef PubMed.
- S. Gharehkhani, N. Ghavidel and P. Fatehi, ACS Sustainable Chem. Eng., 2019, 7, 2370–2379 CrossRef CAS.
- J. C. de Haro, J. F. Rodríguez, Á. Pérez and M. Carmona, J. Cleaner Prod., 2016, 138, 77–82 CrossRef CAS.
- S. H. F. da Silva, I. Egüés and J. Labidi, Ind. Crops Prod., 2019, 137, 687–693 CrossRef CAS.
- X. Pan and J. N. Saddler, Biotechnol. Biofuels, 2013, 6, 1–10 Search PubMed.
- A. N. Hayati, D. A. C. Evans, B. Laycock, D. J. Martin and P. K. Annamalai, Ind. Crops Prod., 2018, 117, 149–158 CrossRef CAS.
- S. Gómez-Fernández, M. Günther, B. Schartel, M. A. Corcuera and A. Eceiza, Ind. Crops Prod., 2018, 125, 346–359 CrossRef.
- E. Delebecq, J. P. Pascault, B. Boutevin and F. Ganachaud, Chem. Rev., 2013, 113, 80–118 CrossRef CAS PubMed.
- Y. Chevalier and M. A. Bolzinger, Colloids Surf., A, 2013, 439, 23–34 CrossRef CAS.
- M. Gigli, G. Fellet, L. Pilotto, M. Sgarzi, L. Marchiol and C. Crestini, Front. Plant Sci., 2022, 13, 976410 CrossRef CAS PubMed.
- T. D. H. Bugg, M. Ahmad, E. M. Hardiman and R. Rahmanpour, Nat. Prod. Rep., 2011, 28, 1883–1896 RSC.
- D. A. Ali and M. M. Mehanna, Int. J. Biol. Macromol., 2022, 221, 934–953 CrossRef CAS PubMed.
- T. C. Ezike, U. S. Okpala, U. L. Onoja, C. P. Nwike, E. C. Ezeako, O. J. Okpara, C. C. Okoroafor, S. C. Eze, O. L. Kalu, E. C. Odoh, U. G. Nwadike, J. O. Ogbodo, B. U. Umeh, E. C. Ossai and B. C. Nwanguma, Heliyon, 2023, 9, e17488 CrossRef CAS PubMed.
- W. D. H. Schneider, A. J. P. Dillon and M. Camassola, Biotechnol. Adv., 2021, 47, 107685 CrossRef CAS PubMed.
- S. Chandna, N. S. Thakur, Y. N. Reddy, R. Kaur and J. Bhaumik, ACS Biomater. Sci. Eng., 2019, 5, 3212–3227 CrossRef CAS PubMed.
- K. Gogde, S. Paul, A. K. Pujari, A. K. Yadav and J. Bhaumik, J. Med. Chem., 2023, 66, 13058–13071 CrossRef CAS PubMed.
- K. Liu, D. Zheng, H. Lei, J. Liu, J. Lei, L. Wang and X. Ma, ACS Biomater. Sci. Eng., 2018, 4, 1730–1737 CAS.
- L. Chen, X. Zhou, Y. Shi, B. Gao, J. Wu, T. B. Kirk, J. Xu and W. Xue, Chem. Eng. J., 2018, 346, 217–225 CrossRef CAS.
- P. Figueiredo, K. Lintinen, A. Kiriazis, V. Hynninen, Z. Liu, T. Bauleth-Ramos, A. Rahikkala, A. Correia, T. Kohout, B. Sarmento, J. Yli-Kauhaluoma, J. Hirvonen, O. Ikkala, M. A. Kostiainen and H. A. Santos, Biomaterials, 2017, 121, 97–108 Search PubMed.
- L. Dai, R. Liu, L. Q. Hu, Z. F. Zou and C. L. Si, ACS Sustainable Chem. Eng., 2017, 5, 8241–8249 CrossRef CAS.
- M. S. Alqahtani, A. Alqahtani, A. Al-Thabit, M. Roni and R. Syed, J. Mater. Chem. B, 2019, 7, 4461–4473 Search PubMed.
- F. Liu, Q. Wang, G. Zhai, H. Xiang, J. Zhou, C. Jia, L. Zhu, Q. Wu and M. Zhu, Nat. Commun., 2022, 13, 1–10 Search PubMed.
- L. Sonowal and S. Gautam, Nano-Struct. Nano-Objects, 2024, 38, 101117 CrossRef CAS.
- B. Feng, W. Zhao, M. Zhang, X. Fan, T. He, Q. Luo, J. Yan and J. Sun, Chem. – Asian J., 2024, 19(19), e202400611 CrossRef CAS PubMed.
- S. Paul, N. S. Thakur, S. Chandna, V. Sagar and J. Bhaumik, ACS Appl. Nano Mater., 2022, 5, 2748–2761 Search PubMed.
- B. Zhang, Y. Liu, M. Ren, W. Li, X. Zhang, R. Vajtai, P. M. Ajayan, J. M. Tour and L. Wang, ChemSusChem, 2019, 12, 4202–4210 Search PubMed.
- S. Rai, B. K. Singh, P. Bhartiya, A. Singh, H. Kumar, P. K. Dutta and G. K. Mehrotra, J. Lumin., 2017, 190, 492–503 Search PubMed.
- A. A. Myint, W. K. Rhim, J. M. Nam, J. Kim and Y. W. Lee, J. Ind. Eng. Chem., 2018, 66, 387–395 CrossRef CAS.
- W. Chen, C. Hu, Y. Yang, J. Cui and Y. Liu, Materials, 2016, 9(3), 184 Search PubMed.
- N. Niu, Z. Ma, F. He, S. Li, J. Li, S. Liu and P. Yang, Langmuir, 2017, 33, 5786–5795 CrossRef CAS PubMed.
- P. Trucillo, Materials, 2024, 17, 456 Search PubMed.
- B. Xue, Y. Yang, Y. Sun, J. Fan, X. Li and Z. Zhang, Int. J. Biol. Macromol., 2019, 122, 954–961 Search PubMed.
- J. Domínguez-Robles, S. A. Stewart, A. Rendl, Z. González, R. F. Donnelly and E. Larrañeta, Biomolecules, 2019, 9(9), 423 Search PubMed.
- M. Pishnamazi, J. Iqbal, S. Shirazian, G. M. Walker and M. N. Collins, Int. J. Biol. Macromol., 2019, 124, 354–359 CrossRef CAS PubMed.
- M. Pishnamazi, H. Hafizi, S. Shirazian, M. Culebras, G. M. Walker and M. N. Collins, Polymer, 2019, 11, 1–10 Search PubMed.
- K. Gogde, S. Kirar, A. K. Pujari, D. Mohne, A. K. Yadav and J. Bhaumik, J. Mater. Chem. B, 2024, 13, 288–304 RSC.
- J. S. Jayan, S. S. Jayan, B. D. S. Deeraj and A. Saritha, Ind. Crops Prod., 2024, 213, 118402 CrossRef CAS.
- H. S. Yoo, T. G. Kim and T. G. Park, Adv. Drug Delivery Rev., 2009, 61, 1033–1042 Search PubMed.
- D. Kai, H. M. Chong, L. P. Chow, L. Jiang, Q. Lin, K. Zhang, H. Zhang, Z. Zhang and X. J. Loh, Compos. Sci. Technol., 2018, 158, 26–33 CrossRef CAS.
- J. Rekola, A. J. Aho, J. Gunn, J. Matinlinna, J. Hirvonen, P. Viitaniemi and P. K. Vallittu, Acta Biomater., 2009, 5, 1596–1604 CrossRef CAS PubMed.
- R. M. Kalinoski and J. Shi, Ind. Crops Prod., 2019, 128, 323–330 Search PubMed.
- F. Reesi, M. Minaiyan and A. Taheri, Drug Delivery Transl. Res., 2018, 8, 111–122 CrossRef CAS PubMed.
- S. Zhang, S. Hu, Y. Zhou, M. Xie, Y. Liao and F. Yue, ACS Sustainable Chem. Eng., 2025, 13(11), 4560–4569 CrossRef CAS.
- X. Li, J. Shen, B. Wang, X. Feng, Z. Mao and X. Sui, ACS Sustainable Chem. Eng., 2021, 9, 5470–5480 CrossRef CAS.
- F. M. C. Freitas, M. A. Cerqueira, C. Gonçalves, S. Azinheiro, A. Garrido-Maestu, A. A. Vicente, L. M. Pastrana, J. A. Teixeira and M. Michelin, Int. J. Biol. Macromol., 2020, 163, 1798–1809 CrossRef CAS PubMed.
- Y. Peng, B. Guo, W. Wang, P. Yu, Z. Wu, L. Shao and W. Luo, Int. J. Biol. Macromol., 2023, 238, 124127 CrossRef CAS PubMed.
- A. Jędrzak, T. Rębiś, Ł. Klapiszewski, J. Zdarta, G. Milczarek and T. Jesionowski, Sens. Actuators, B, 2018, 256, 176–185 CrossRef.
- M. Mariana, T. Alfatah, H. P. S. Abdul Khalil, E. B. Yahya, N. G. Olaiya, A. Nuryawan, E. M. Mistar, C. K. Abdullah, S. N. Abdulmadjid and H. Ismail, J. Mater. Res. Technol., 2021, 15, 2287–2316 CrossRef CAS.
- J. Preet, K. Pathania, J. Kaur, R. Singh, D. B. Salunke and S. V. Pawar, Mater. Adv., 2024, 5, 9445–9457 RSC.
- M. A. L. Huët, I. C. Phul, N. Goonoo, Z. Li, X. Li and A. Bhaw-Luximon, J. Mater. Chem. B, 2024, 12, 5496–5512 RSC.
- X. Liu, H. J. Zhang, S. Xi, Y. Zhang, P. Rao, X. You and S. Qu, Adv. Funct. Mater., 2025, 35, 2413464 CrossRef CAS.
- Y. Gu, C. Chen, Y. Yuan, X. Guo, C. Zhang, W. Wu, M. M. Rahman, B. Jiang and Y. Jin, Adv. Compos. Hybrid Mater., 2025, 8, 1–13 Search PubMed.
- H. Li, G. Wang, Y. Wu, N. Jiang and K. Niu, Polymer, 2024, 16, 770 Search PubMed.
- H. Li, Y. Liang, P. Li and C. He, J. Bioresour. Bioprod., 2020, 5, 163–179 CAS.
- A. Moreno and M. H. Sipponen, Mater. Horiz., 2020, 7, 2237–2257 Search PubMed.
- A. Dinari, M. Abdollahi and M. Sadeghizadeh, Sci. Rep., 2021, 11, 1962 CrossRef CAS PubMed.
- C. J. Wijaya, S. Ismadji and S. Gunawan, Molecules, 2021, 26, 676 CrossRef CAS PubMed.
- Y. Li, X. Qiu, Y. Qian, W. Xiong and D. Yang, Chem. Eng. J., 2017, 327, 1176–1183 CrossRef CAS.
- L. Cheng, B. Deng, W. Luo, S. Nie, X. Liu, Y. Yin, S. Liu, Z. Wu, P. Zhan, L. Zhang and J. Chen, J. Agric. Food Chem., 2020, 68, 5249–5258 CrossRef CAS PubMed.
- L. Dai, W. Zhu, R. Liu and C. Si, Part. Part. Syst. Charact., 2018, 35(4), 1700447 CrossRef.
- M. Seyedi, B. Kibret, S. Salibindla and D. T. H. Lai, in Encyclopedia of Information Science and Technology, ed. D. B. A. Mehdi Khosrow-Pour, IGI Global Scientific Publishing, Hershey, PA, 3rd edn, 2015, pp. 469–478, DOI:10.4018/978-1-4666-5888-2.ch045.
- R. Kumar, A. Butreddy, N. Kommineni, P. Guruprasad Reddy, N. Bunekar, C. Sarkar, S. Dutt, V. K. Mishra, K. R. Aadil, Y. K. Mishra, D. Oupicky and A. Kaushik, Int. J. Nanomed., 2021, 16, 2419–2441 CrossRef CAS PubMed.
- W. Pei, J. Deng, P. Wang, X. Wang, L. Zheng, Y. Zhang and C. Huang, Int. J. Biol. Macromol., 2022, 212, 547–560 CrossRef CAS PubMed.
- L. Siddiqui, H. Mishra, P. K. Mishra, Z. Iqbal and S. Talegaonkar, Med. Hypotheses, 2018, 121, 10–14 CrossRef CAS PubMed.
- I. V. Pylypchuk, H. Suo, C. Chucheepchuenkamol, N. Jedicke, P. A. Lindén, M. E. Lindström, M. P. Manns, O. Sevastyanova and T. Yevsa, Front. Bioeng. Biotechnol., 2022, 9, 817768 CrossRef PubMed.
- D. Liu, Y. Li, Y. Qian, Y. Xiao, S. Du and X. Qiu, ACS Sustainable Chem. Eng., 2017, 5, 8424–8428 CrossRef CAS.
- B. S. Patil, G. K. Jayaprakasha, K. N. Chidambara Murthy and A. Vikram, J. Agric. Food Chem., 2009, 57, 8142–8160 CrossRef CAS PubMed.
- F. Verdini, E. C. Gaudino, E. Canova, S. Tabasso, P. J. Behbahani and G. Cravotto, Molecules, 2022, 27, 3598 CrossRef CAS PubMed.
- M. P. Vinardell, V. Ugartondo and M. Mitjans, Ind. Crops Prod., 2008, 27, 220–223 CrossRef CAS.
- R. Wang, L. Zheng, Q. Xu, L. Xu, D. Wang, J. Li, G. Lu, C. Huang and Y. Wang, Int. J. Biol. Macromol., 2021, 191, 1087–1095 CrossRef CAS PubMed.
- Y. Ito, Y. Akao, M. Shimazawa, N. Seki, Y. Nozawa and H. Hara, CNS Drug Rev., 2007, 13, 296–307 CrossRef CAS PubMed.
- C. Huang, J. He, R. Narron, Y. Wang and Q. Yong, ACS Sustainable Chem. Eng., 2017, 5, 11770–11779 CrossRef CAS.
- Z. Chen, L. Chen, K. S. Khoo, V. K. Gupta, M. Sharma, P. L. Show and P. S. Yap, Biotechnol. Adv., 2023, 69, 108265 CrossRef CAS PubMed.
- Ł. Klapiszewski, T. J. Szalaty and T. Jesionowski, Lignin - Trends and Applications, 2017, DOI:10.5772/INTECHOPEN.70376.
- B. Kumar and P. Verma, Fuel, 2021, 288, 119622 CrossRef CAS.
- Z. Chen, A. Ragauskas and C. Wan, Ind. Crops Prod., 2020, 147, 112241 CrossRef CAS.
- S. Sethupathy, G. Murillo Morales, L. Gao, H. Wang, B. Yang, J. Jiang, J. Sun and D. Zhu, Bioresour. Technol., 2022, 347, 126696 Search PubMed.
- F. N. U. Asina, I. Brzonova, E. Kozliak, A. Kubátová and Y. Ji, Renewable Sustainable Energy Rev., 2017, 77, 1179–1205 CrossRef CAS.
- J. Ralph, K. Lundquist, G. Brunow, F. Lu, H. Kim, P. F. Schatz, J. M. Marita, R. D. Hatfield, S. A. Ralph, J. H. Christensen and W. Boerjan, Phytochem. Rev., 2004, 3, 29–60 CrossRef CAS.
- G. T. Beckham, C. W. Johnson, E. M. Karp, D. Salvachúa and D. R. Vardon, Curr. Opin. Biotechnol., 2016, 42, 40–53 Search PubMed.
- P. P. Fu, Q. Xia, H. M. Hwang, P. C. Ray and H. Yu, J. Food Drug Anal., 2014, 22, 64–75 Search PubMed.
- L. Siddiqui, J. Bag, D. Seetha, A. Mittal, H. Leekha, M. Mishra, A. K. Mishra, P. K. Verma, A. Mishra, Z. Iqbal Ekielski and S. Talegaonkar, Int. J. Biol. Macromol., 2020, 152, 786–802 Search PubMed.
- C. Bragato, A. Persico, G. Ferreres, T. Tzanov and P. Mantecca, Int. J. Nanomed., 2024, 19, 7731–7750 CrossRef CAS PubMed.
- R. Jagels, Am. J. Ind. Med., 1985, 8, 241–251 CrossRef CAS PubMed.
- S. Sugiarto, Y. Leow, C. L. Tan, G. Wang and D. Kai, Bioact. Mater., 2022, 8, 71–94 Search PubMed.
- R. Tsuji, H. Koizumi, D. Aoki, Y. Watanabe, Y. Sugihara, Y. Matsushita, K. Fukushima and D. Fujiwara, J. Biol. Chem., 2015, 290, 4410–4421 CrossRef CAS PubMed.
- L. Greco, S. Ullo, L. Rigano, M. Fontana and E. Berardesca, Cosmetics, 2019, 6, 38 CrossRef CAS.
- S. Takahashi and K. Hori, Front. Microbiol., 2023, 14, 1298323 CrossRef PubMed.
- W. Lee and S. Ko, Molecules, 2025, 30, 667 CrossRef CAS PubMed.
- V. E. Kagan, N. V. Konduru, W. Feng, B. L. Allen, J. Conroy, Y. Volkov, I. I. Vlasova, N. A. Belikova, N. Yanamala, A. Kapralov, Y. Y. Tyurina, J. Shi, E. R. Kisin, A. R. Murray, J. Franks, D. Stolz, P. Gou, J. Klein-Seetharaman, B. Fadeel, A. Star and A. A. Shvedova, Nat. Nanotechnol., 2010, 5, 354–359 CrossRef CAS PubMed.
- B. L. Allen, P. D. Kichambare, P. Gou, I. I. Vlasova, A. A. Kapralov, N. Konduru, V. E. Kagan and A. Star, Nano Lett., 2008, 8, 3899–3903 CrossRef CAS PubMed.
- K. Bhattacharya, R. El-Sayed, F. T. Andón, S. P. Mukherjee, J. Gregory, H. Li, Y. Zhao, W. Seo, A. Fornara, B. Brandner, M. S. Toprak, K. Leifer, A. Star and B. Fadeel, Carbon, 2015, 91, 506–517 CrossRef CAS.
- D. X. Flores-Cervantes, H. M. Maes, A. Schäffer, J. Hollender and H. P. E. Kohler, Environ. Sci. Technol., 2014, 48, 4826–4834 CrossRef CAS PubMed.
- R. Sekar and T. J. Dichristina, Environ. Sci. Technol., 2014, 48, 12858–12867 CrossRef CAS PubMed.
- J. Wang, Q. Ma, Z. Zhang, S. Li, C. S. Diko, C. Dai, H. Zhang and Y. Qu, Sci. Total Environ., 2020, 746, 141020 CrossRef CAS PubMed.
- K. Donaldson, C. A. Poland, F. A. Murphy, M. MacFarlane, T. Chernova and A. Schinwald, Adv. Drug Delivery Rev., 2013, 65, 2078–2086 CrossRef CAS PubMed.
- C. A. Poland, R. Duffin, I. Kinloch, A. Maynard, W. A. H. Wallace, A. Seaton, V. Stone, S. Brown, W. MacNee and K. Donaldson, Nat. Nanotechnol., 2008, 3, 423–428 CrossRef CAS PubMed.
- L. Kong, G. Yan, X. Huang, Y. Wu, X. Che, J. Liu, J. Jia, H. Zhou and B. Yan, Sci. Total Environ., 2023, 864, 161059 CrossRef CAS PubMed.
- M. Hatami, Ecotoxicol. Environ. Saf., 2017, 142, 274–283 CrossRef CAS PubMed.
- M. H. Sipponen, H. Lange, C. Crestini, A. Henn and M. Österberg, ChemSusChem, 2019, 12, 2039–2054 CrossRef CAS PubMed.
- M. H. Lahtinen, E. Kynkäänniemi, C. Jian, A. Salonen, A. M. Pajari and K. S. Mikkonen, Mol. Nutr. Food Res., 2023, 67, 2300201 CrossRef CAS PubMed.
- J. Allan, S. Belz, A. Hoeveler, M. Hugas, H. Okuda, A. Patri, H. Rauscher, P. Silva, W. Slikker, B. Sokull-Kluettgen, W. Tong and E. Anklam, Regul. Toxicol. Pharmacol., 2021, 122, 104885 CrossRef CAS PubMed.
- J. A. Chávez-Hernández, A. J. Velarde-Salcedo, G. Navarro-Tovar and C. Gonzalez, Nanoscale Adv., 2024, 6, 1583–1610 RSC.
- International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use ICH Harmonised Guideline Technical and Regulatory Considerations for Pharmaceutical Product Lifecycle Management Q12 Final Version, 2019.
- International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use Pharmaceutical Development Q8(R2), 2009.
- https://eur-lex.europa.eu/eli/reg/2017/745/oj/eng, (accessed 9 November 2025).
- ISO 10993-1:2018(en), Biological evaluation of medical devices — Part 1: Evaluation and testing within a risk management process, https://www.iso.org/obp/ui/#iso:std:iso:10993:-1:ed-5:v2:en, (accessed 9 November 2025).
- M. Pishnamazi, H. Y. Ismail, S. Shirazian, J. Iqbal, G. M. Walker and M. N. Collins, Cellulose, 2019, 26, 6165–6178 CrossRef CAS.
- V. Kumar, V. Bansal, A. Madhavan, M. Kumar, R. Sindhu, M. K. Awasthi, P. Binod and S. Saran, Bioengineered, 2022, 13, 4309–4327 Search PubMed.
- J. A. Burdick and G. D. Prestwich, Adv. Mater., 2011, 23, H41 Search PubMed.
- E. Rezvani Ghomi, N. Nourbakhsh, M. Akbari Kenari, M. Zare and S. Ramakrishna, J. Biomed. Mater. Res., Part B, 2021, 109, 1986–1999 Search PubMed.
- K. Y. Lee and D. J. Mooney, Prog. Polym. Sci., 2012, 37, 106–126 CrossRef CAS PubMed.
- R. Liu, L. Dai, C. Xu, K. Wang, C. Zheng and C. Si, ChemSusChem, 2020, 13, 4266–4283 CrossRef CAS PubMed.
- E. Barbera, A. Guarise, A. Bertucco, R. L. Maglinao and S. Kumar, J. Supercrit. Fluids, 2025, 223, 106631 CrossRef CAS.
- Z. Ebrahimpourboura, M. Mosalpuri, C. Yang, A. Ponukumati, C. Stephenson, M. Foston and M. M. Wright, Green Chem., 2024, 26, 11303–11315 RSC.
Footnote |
| † Present address: Department of Biotechnology, School of Bioengineering, SRM Institute of Science and Technology, SRM Nagar, Chengalpattu Dist., Kattankulathur, 603203, Tamil Nadu, India. |
| This journal is © The Royal Society of Chemistry 2026 |